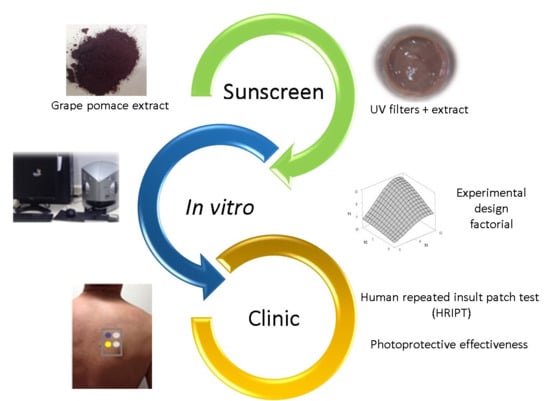
Safety and Photoprotective Efficacy of a Sunscreen System Based on Grape Pomace (Vitis vinifera L.) Phenolics from Winemaking
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Crude Extract
2.3. Design of Experiment (DoE)
2.3.1. Formulations
2.3.2. Antioxidant Activity
2.3.3. Sun Protection Factor (In Vitro SPF) and Photostability
2.4. Clinical Trials
2.4.1. Subjects
2.4.2. Primary and Cumulative Cutaneous Irritability and Sensitization Tests
- Induction (1st to 3rd week): hypoallergenic adhesive tape (Finn Chambers, Epitest, Rannankoukku, Tuusula, Finland) containing the test samples (E and F) and distilled water as negative control was applied at random sites on the back of each volunteer. If any volunteer showed signs at this negative control site, they would have been excluded from the research. Clinical signs and discomfort were reported to the dermatologist and the samples were reapplied in the same area;
- Rest (4th to 5th week): no product application;
2.4.3. Phototoxicity and Photosensitization Test
- Induction (1st to 2nd week): hypoallergenic adhesive tape (Finn Chambers, Epitest) containing the samples (E and F) and distilled water as negative control was randomly applied on the back skin of the subject and maintained for 48 h, after which the adhesive tape was removed from the skin and the areas were exposed to UVA and UVB radiation for 6 min 4 s. Clinical signs were reported to the dermatologist and products were reapplied in the same area;
- Rest (3rd to 4th week): no sample was applied;
- Challenge (5th week): samples were in contact with the skin for 48 h, after which the adhesive tape was removed from the skin and the areas were re-irradiated.
2.4.4. Clinical Photoprotective Effectiveness
2.4.5. UVA Protection Factor (UVA-PF)
2.5. Statistical Analysis
3. Results
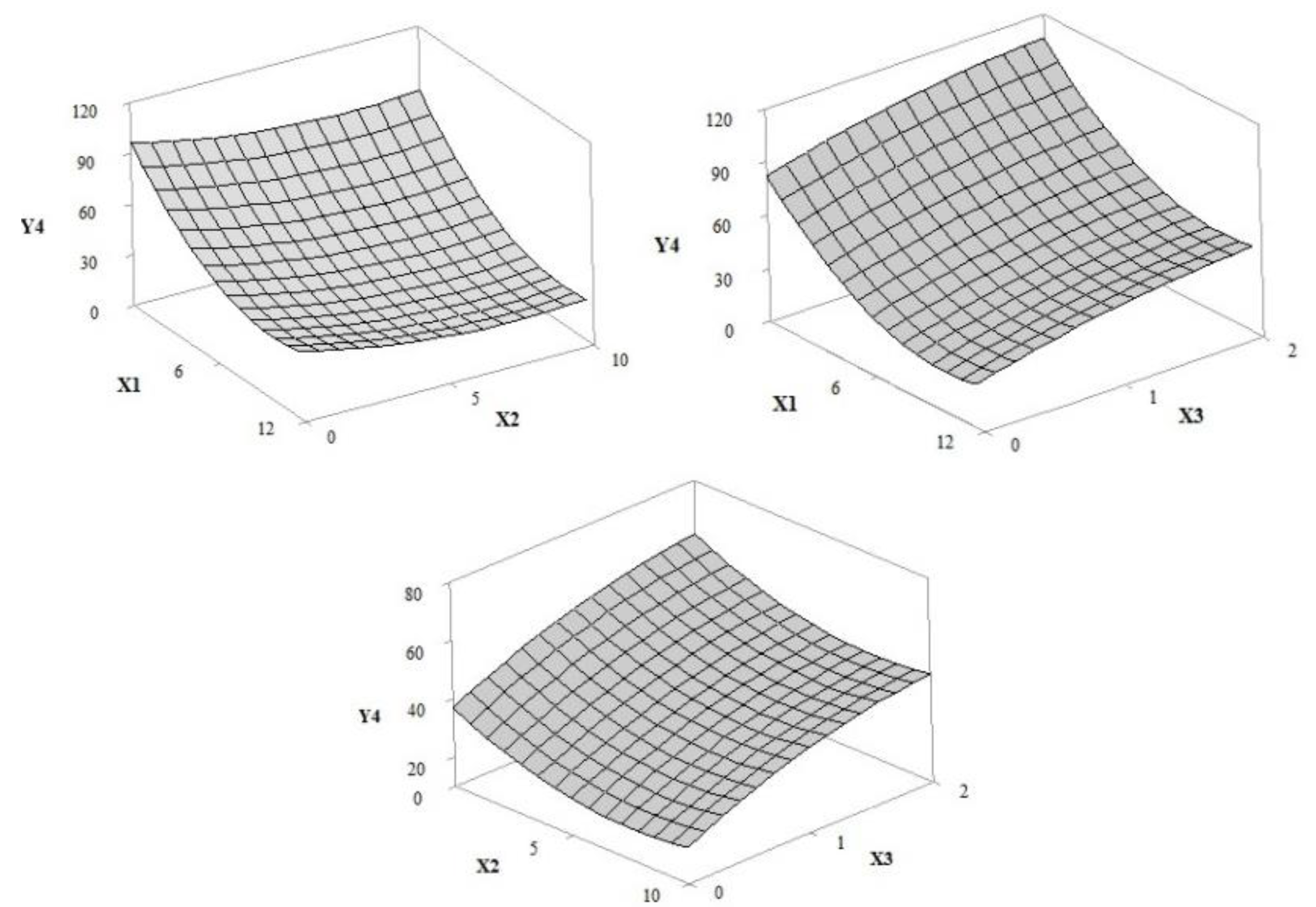
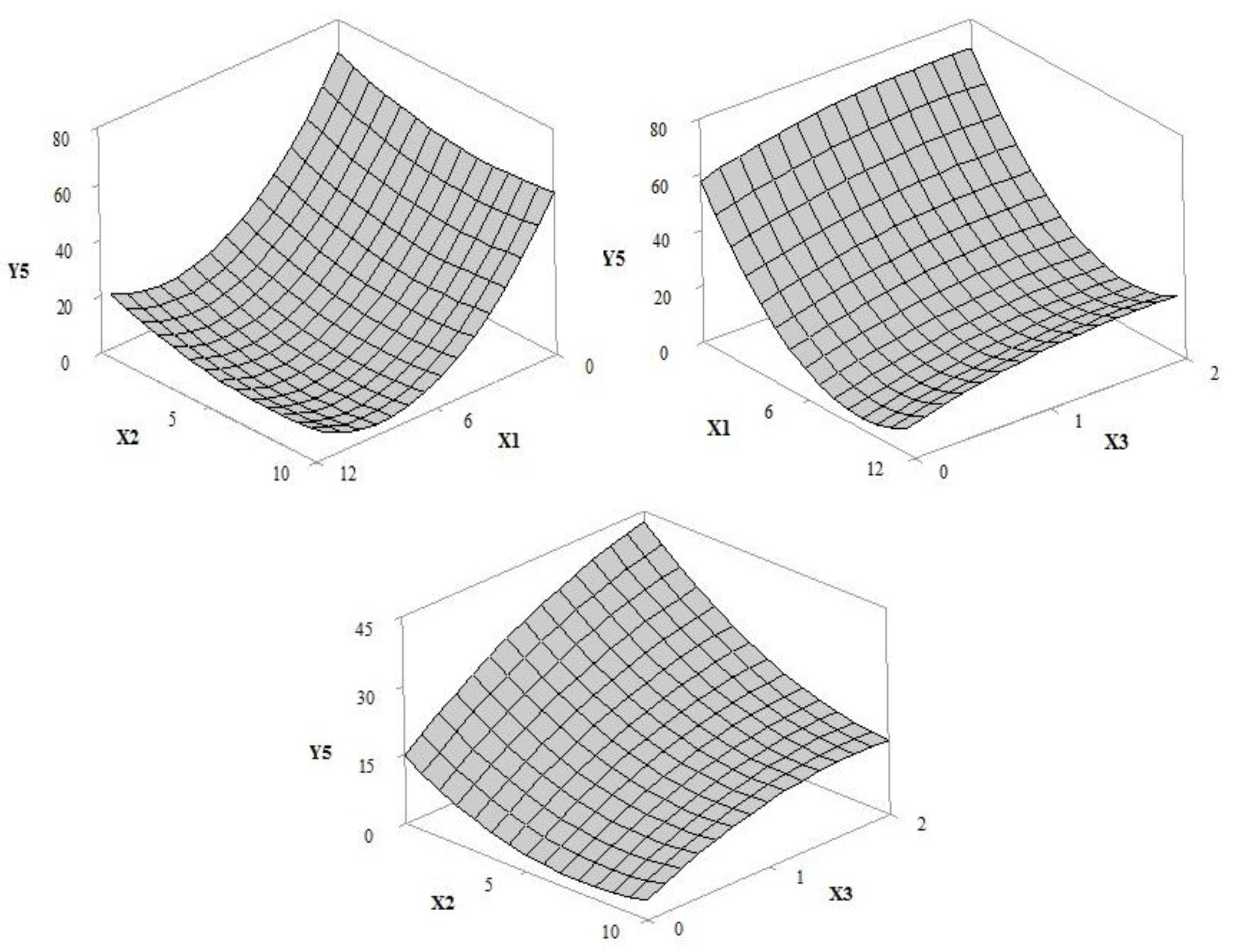
3.1. Design of Experiment (DoE)
3.1.1. Formulations
3.1.2. Antioxidant Activity
3.1.3. Sun Protection Factor (In Vitro SPF) and Photostability
3.1.4. Critical Wavelength
3.1.5. UVA Transmittance
3.1.6. UVB Transmittance
3.1.7. UVA/UVB Ratio
3.2. Clinical Trials
3.2.1. Primary, Accumulated Dermal Irritability Test, and Sensitization
3.2.2. Phototoxicity and Photosensitization Test
3.2.3. Photoprotective Effectiveness
3.2.4. UVA Protection Factor (UVA-PF)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brenner, M.; Hearing, V.J. The Protective Role of Melanin Against UV Damage in Human Skin. Photochem. Photobiol. 2008, 84, 539–549. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Q.; Feng, C.; Ruan, Z. Inhibitory effect of a genistein derivative on pigmentation of guinea pig skin. RSC Adv. 2017, 7, 7914–7919. [Google Scholar] [CrossRef] [Green Version]
- Gragnani, A.; Mac Cornick, S.; Chominski, V.; De Noronha, S.M.R.; De Noronha, S.A.A.C.; Ferreira, L.M. Review of Major Theories of Skin Aging. Adv. Aging Res. 2014, 3, 265–284. [Google Scholar] [CrossRef] [Green Version]
- Lucas, R.M.; Norval, M.; Neale, R.E.; Young, A.; De Gruijl, F.R.; Takizawa, Y.; Van Der Leun, J.C. The consequences for human health of stratospheric ozone depletion in association with other environmental factors. Photochem. Photobiol. Sci. 2015, 14, 53–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rai, R.; Shanmuga, S.C.; Srinivas, C.R. Update on photoprotection. Indian J. Dermatol. 2012, 57, 335–342. [Google Scholar] [CrossRef]
- Peres, D.D.; Ariede, M.B.; Candido, T.M.; De Almeida, T.S.; Lourenço, F.R.; Consiglieri, V.O.; Kaneko, T.M.; Velasco, M.V.R.; Baby, A.R. Quality by design (QbD), Process Analytical Technology (PAT), and design of experiment applied to the development of multifunctional sunscreens. Drug Dev. Ind. Pharm. 2016, 43, 246–256. [Google Scholar] [CrossRef]
- Deuschle, V.C.K.N.; Deuschle, R.A.N.; Bortoluzzi, M.R.; Athayde, M.L. Physical chemistry evaluation of stability, spreadability, in vitro antioxidant, and photo-protective capacities of topical formulations containing Calendula officinalis L. leaf extract. Braz. J. Pharm. Sci. 2015, 51, 63–75. [Google Scholar] [CrossRef] [Green Version]
- Ngoc, L.T.N.; Van Tran, V.; Moon, J.-Y.; Chae, M.; Park, D.; Lee, Y.-C. Recent Trends of Sunscreen Cosmetic: An Update Review. Cosmetics 2019, 6, 64. [Google Scholar] [CrossRef] [Green Version]
- Radice, M.; Manfredini, S.; Ziosi, P.; Dissette, V.; Buso, P.; Fallacara, A.; Vertuani, S. Herbal extracts, lichens and biomolecules as natural photo-protection alternatives to synthetic UV filters. A systematic review. Fitoterapia 2016, 114, 144–162. [Google Scholar] [CrossRef]
- Williams, S.; Tamburic, S.; Lally, C. Eating chocolate can significantly protect the skin from UV light. J. Cosmet. Dermatol. 2009, 8, 169–173. [Google Scholar] [CrossRef]
- Fiume, M.M.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.J.G.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; et al. Safety Assessment of Vitis vinifera (Grape)-Derived Ingredients as Used in Cosmetics. Int. J. Toxicol. 2014, 33, 48S–83S. [Google Scholar] [CrossRef] [PubMed]
- Komes, D.; Horžić, D.; Belščak, A.; Ganić, K.K.; Vulić, I. Green tea preparation and its influence on the content of bioactive compounds. Food Res. Int. 2010, 43, 167–176. [Google Scholar] [CrossRef]
- The Plant List. Botanical Name. Available online: http://www.theplantlist.org/tpl1.1/record/ild-10365 (accessed on 30 June 2018).
- Saewan, N.; Jimtaisong, A. Photoprotection of natural flavonoids. J. Appl. Pharm. Sci. 2013, 3, 129–141. [Google Scholar]
- Mansur, M.C.P.R.; Leitão, S.G.; Cerqueira-Coutinho, C.; Vermelho, A.B.; Silva, R.S.; Presgrave, O.A.; Leitão, Á.A.; Leitão, G.G.; Ricci-Júnior, E.; Santos, E.P. In vitro and in vivo evaluation of efficacy and safety of photoprotective formulations containing antioxidant extracts. Rev. Bras. de Farm. 2016, 26, 251–258. [Google Scholar] [CrossRef] [Green Version]
- Hubner, A.; Sobreira, F.; Neto, A.V.; Pinto, C.A.S.D.O.; Dario, M.F.; Díaz, I.E.C.; Lourenço, F.R.; Rosado, C.; Baby, A.R.; Bacchi, E.M. The Synergistic Behavior of Antioxidant Phenolic Compounds Obtained from Winemaking Waste’s Valorization, Increased the Efficacy of a Sunscreen System. Antioxidants 2019, 8, 530. [Google Scholar] [CrossRef] [Green Version]
- Yilmaz, Y.; Toledo, R.T. Major Flavonoids in Grape Seeds and Skins: Antioxidant Capacity of Catechin, Epicatechin, and Gallic Acid. J. Agric. Food Chem. 2004, 52, 255–260. [Google Scholar] [CrossRef]
- FAO, Food and Agriculture Organization of the United Nations. Agribusiness Handbook: Grapes Wine; FAO: Rome, Italy, 2009; pp. 1–39.
- Baxter, R.A. Anti-aging properties of resveratrol: Review and report of a potent new antioxidant skin care formulation. J. Cosmet. Dermatol. 2008, 7, 2–7. [Google Scholar] [CrossRef]
- Ky, I.; Teissedre, P.-L. Characterisation of Mediterranean Grape Pomace Seed and Skin Extracts: Polyphenolic Content and Antioxidant Activity. Molecules 2015, 20, 2190–2207. [Google Scholar] [CrossRef] [Green Version]
- Mello, L.M.R. Vitivinicultura Brasileira: Panorama 2018, Embrapa: Bento Gonçalves, RS. 2019. Available online: https://ainfo.cnptia.embrapa.br/digital/bitstream/item/203100/1/Comunicado-Tecnico-210.pdf (accessed on 14 July 2019).
- SEBRAE. Serviço Brasileiro de Apoio às Micro e Pequenas Empresas. Produção de Vinhos e Espumantes no Brasil. 2014. Available online: https://respostas.sebrae.com.br/producao-de-vinhos-e-espumantes-no-brasil/ (accessed on 27 January 2016).
- Gunst, R.F.; Myers, R.H.; Montgomery, D.C. Response Surface Methodology: Process and Product Optimization Using Designed Experiments. Technometrics 1996, 38, 285. [Google Scholar] [CrossRef]
- Department of Health and Human Services, Food and Drug Administration, United States. Labeling and effectiveness testing; sunscreen drug products for over-the-counter human use. Final Rule. Fed. Regist. 2011, 76, 35620–35665. [Google Scholar]
- COLIPA. Cosmetics Europe. In In Vitro Method for the Determination of the UVA Protection Factor and “Critical Wavelength” Values of Sunscreen Products; Colipa: Brussels, Belgium, 2011; pp. 1–29. [Google Scholar]
- Rietschel, R.L.; Fowler, J.F., Jr. Fisher’s Contact Dermatitis, 6th ed.; PMPH: Shelton, CT, USA, 2008; p. 862. [Google Scholar]
- COLIPA. Cosmetics Europe. Guidelines for the Assessment of Skin Toleranceof Potentially Irritant Cosmetic Ingredients; Colipa: Brussels, Belgium, 2004; pp. 1–25. [Google Scholar]
- BRASIL Agência Nacional de Vigilância Sanitária. Farmacopeia dos Estados Unidos do Brasil, 5th ed.; Anvisa: Brasília, Brazil, 2017; pp. 311–330.
- ANVISA; Agência Nacional De Vigilância Sanitária. Resolução—RDC n.69, de 23 de Março de 2016. Regulamento Técnico Mercosul Sobre Lista de Filtros Ultravioletas Permitidos para Produtos de Higiene Pessoal, Cosméticos e Perfumes. 2016. Available online: http://portal.anvisa.gov.br/documents/10181/2863150/RDC_69_2016_COMP.pdf/5689ac91-e621-45b7-a122-b3163e4b3cc3 (accessed on 3 September 2016).
- Baby, A.R.; Haroutiounian-Filho, C.A.; Sarruf, F.D.; Tavante-Júnior, C.R.; Pinto, C.A.S.O.; Zague, V.; Arêas, E.P.G.; Kaneko, T.M.; VElasco, M.V.R. Stability and in vitro penetration study of rutin incorporated in a cosmetic emulsion through an alternative model biomembrane. Rev. Bras. Cien. Farm. 2008, 44, 233–248. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Springsteen, A.; Yurek, R.; Frazier, M.; Carr, K.F. In vitro measurement of sun protection factor of sunscreens by diffuse transmittance1This paper is dedicated to the memory of our colleague and friend, Mona Yurek (1959–1998), who was involved in the quantitation and methodology of the sample preparation along with most of our in vitro sunscreen measurements.1. Anal. Chim. Acta 1999, 380, 155–164. [Google Scholar] [CrossRef]
- Diffey, B.L.; Tanner, P.R.; Matts, P.J.; Nash, J. In vitro assessment of the broad-spectrum ultraviolet protection of sunscreen products. J. Am. Acad. Dermatol. 2000, 43, 1024–1035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ANVISA; Agência Nacional de Vigilânia Sanitária. Guia para Avaliação de Segurança de Produtos Cosméticos, 2nd ed.; Anvisa: Brasília, Brazil, 2012; pp. 34–37.
- United States. Food and Drug Administration. 21 CRF Parts 347 and 352. Sunscreen drug products for over-the-counter human use; Proposed amendment of final monograph. Proposed Rule Fed. Regist. 2007, 72, 49070–49122. [Google Scholar]
- COLIPA. Cosmetics Europe. In International Sun Protection Factor (SPF) Test Method; Colipa: Brussels, Belgium, 2006; pp. 1–78. [Google Scholar]
- Fitzpatrick, T.B. Peau et soleil. J. Med. Esthet. 1975, 2, 33–34. [Google Scholar]
- Herzog, B.; Giesinger, J.; Schnyder, M. Solubility of UV Absorbers for sunscreens is essential for the creation of light feel formulations. SOFW J. 2013, 139, 7–14. [Google Scholar]
- Balogh, T.S.; Velasco, M.V.R.; Moraes, C.A.P.; Kaneko, T.M.; Baby, A.R. Ultraviolet radiation protection: Current available resources in photoprotection. An. Bras. Dermatol. 2011, 86, 732–742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, R.R.; Machado, P.F.L.; Rocha, R.J.; Silva, S.C.F. A luz e os filtros solares: Uma temática sociocientífica. Rev. Virtual Quim. 2015, 7, 218–241. [Google Scholar]
- Brooke, D.N.; Burs, J.S.; Crookes, M.J.E. Using science to create a better place: UV-filters in cosmetics-prioritisation for environmental assessment. Environ. Agency 2008, 15–48. [Google Scholar]
- Kligman, A.M.; Wooding, W.M. A method for the measurement and evaluation of irritants of human skin. J. Investig. Dermatol. 1967, 49, 78–94. [Google Scholar] [CrossRef] [Green Version]
- ANVISA; Agência Nacional de Vigilância Sanitária. Guia de estabilidade de produtos cosméticos; Anvisa: Brasília, Brazil, 2004; pp. 1–45.
- Lavelli, V.; Kerr, W. Moisture properties and stability of novel bioactive ingredients. In Food Quality and Shelf Life; Elsevier BV: Amsterdam, The Netherlands, 2019; pp. 33–54. [Google Scholar]
- Bakhshabadi, H.; Mirzaei, H.; Ghodsvali, A.; Jafari, S.M.; Ziaiifar, A.M.; Farzaneh, V. The effect of microwave pretreatment on some physico-chemical properties and bioactivity of Black cumin seeds’ oil. Ind. Crop. Prod. 2017, 97, 1–9. [Google Scholar] [CrossRef]
- Gil, E.S.; Couto, R.O. Flavonoid electrochemistry: A review on the electroanalytical applications. Rev. Bras. Farm. 2013, 23, 542–558. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Burton, S.; Kim, C.; Sismour, E.N. Phenolic compounds, antioxidant, and antibacterial properties of pomace extracts from four Virginia-grown grape varieties. Food Sci. Nutr. 2016, 4, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Şahin, S.; Bilgin, M. Olive tree (Olea europaea L.) leaf as a waste by-product of table olive and olive oil industry: A review. J. Sci. Food Agric. 2018, 98, 1271–1279. [Google Scholar] [CrossRef]
- Scotti, M.T.; Cardoso, C.; Castro-Gamboa, I.; Velasco, M.V.R.; Menezes, C.M.D.S.; Scotti, L.; Pauletti, P.; Bolzani, V.D.S.; Ferreira, E.I. Modelagem molecular aplicada ao desenvolvimento de moléculas com atividade antioxidante visando ao uso cosmético. Revista Brasileira de Ciências Farmacêuticas 2007, 43, 153–166. [Google Scholar] [CrossRef] [Green Version]
- Qin, X.; Lu, Y.; Peng, Z.; Fan, S.; Yao, Y. Systematic Chemical Analysis Approach Reveals Superior Antioxidant Capacity via the Synergistic Effect of Flavonoid Compounds in Red Vegetative Tissues. Front. Chem. 2018, 6, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Korać, R.R.; Khambholja, K.M. Potential of herbs in skin protection from ultraviolet radiation. Pharmacogn. Rev. 2011, 5, 164–173. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, A.C.; Paiva, J.P.; Diniz, R.R.; Dos Anjos, V.M.; Silva, A.B.S.; Pinto, A.V.; Dos Santos, E.P.; Leitão, A.C.; Cabral, L.M.; Rodrigues, C.R.; et al. Photoprotection assessment of olive (Olea europaea L.) leaves extract standardized to oleuropein: In vitro and in silico approach for improved sunscreens. J. Photochem. Photobiol. B Biol. 2019, 193, 162–171. [Google Scholar] [CrossRef]
- Gimenis, J.M.; Gomes, A.C.; Dos Santos, V.H.M.; Ferreira, P.C.; Oliveira, C.A.; Baby, A.R.; Da Silva, R.M.G. Antioxidant and photoprotective potential of Moringa oleifera Lam (Moringaceae). Biosci. J. 2018, 34, 1365–1378. [Google Scholar] [CrossRef]
- Napagoda, M.; Malkanthi, B.M.A.S.; Abayawardana, S.A.K.; Qader, M.; Jayasinghe, L. Photoprotective potential in some medicinal plants used to treat skin diseases in Sri Lanka. BMC Complement. Altern. Med. 2016, 16, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Hübner, A.A. Caracterização Fitoquímica e Eficácia Fotoprotetora Clínica de Formulações Cosméticas Contendo Extrato do Bagaço de uva Cabernet Sauvignon. Master’s Thesis, Universidade def São Paulo, São Paulo, Brasil, 2017. [Google Scholar]
- Aleixandre-Tudo, J.L.; Du Toit, W. The Role of UV-Visible Spectroscopy for Phenolic Compounds Quantification in Winemaking. Front. New Trends Sci. Fermented Food Beverages 2019, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Serpone, N.; Dondi, D.; Albini, A. Inorganic and organic UV filters: Their role and efficacy in sunscreens and suncare products. Inorganica Chim. Acta 2007, 360, 794–802. [Google Scholar] [CrossRef]
- Paris, C.; Lhiaubet-Vallet, V.; Jiménez, O.; Trullas, C.; Miranda, M.A. A Blocked Diketo Form of Avobenzone: Photostability, Photosensitizing Properties and Triplet Quenching by a Triazine-derived UVB-filter. Photochem. Photobiol. 2009, 85, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Afonso, S.; Horita, K.; E Silva, J.S.; Almeida, I.; Amaral, M.; Lobão, P.; Costa, P.; Miranda, M.S.; Da Silva, J.C.E.; Lobo, J.S. Photodegradation of avobenzone: Stabilization effect of antioxidants. J. Photochem. Photobiol. B Biol. 2014, 140, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Shaath, N.A. Ultraviolet filters. Photochem. Photobiol. Sci. 2010, 9, 464–469. [Google Scholar] [CrossRef] [PubMed]
- United States. Food and Drug Administration. Appraisal of the Safety of Chemicals in Foods, Drugs, and Cosmetics; Association of Food and Drug Officials of the United States: Austin, TX, USA, 1959.
- Pinnagoda, J.; Tupkek, R.A.; Agner, T.; Serup, J. Guidelines for transepidermal water loss (TEWL) measurement. Contact Dermat. 1990, 22, 164–178. [Google Scholar] [CrossRef] [PubMed]
- Viciolle, E.; Castilho, P.C.; Rosado, C. In vitroandin vivoassessment of the effect of Laurus novocanariensisoil and essential oil in human skin. Int. J. Cosmet. Sci. 2012, 34, 546–550. [Google Scholar] [CrossRef]
- Bircher, A.J.; De Boer, E.M.; Agner, T.; Wahlberg, J.E. Guidelines for measurement of cutaneous blood flow by laser Doppler flowme-try: A report from the standardization group of the european Ssciety of contact dermatitis. Contact. Derm. 1994, 30, 65–72. [Google Scholar] [CrossRef]
- United States. Food and Drug Administration. Division of Biotechnology and GRAS Notice Review (HFS-255). Determination of the GRAS Status of the Addition of Grape Pomace Extract to Conventional Foods. GRAS Notice (GRN) 2012, 446. [Google Scholar]
- Sharif, A.; Akhtar, N.; Khan, M.S.; Menaa, A.; Menaa, B.; Khan, B.A.; Menaa, F. Formulation and evaluation on human skin of a water-in-oil emulsion containing M uscat hamburg black grape seed extract. Int. J. Cosmet. Sci. 2015, 37, 253–258. [Google Scholar] [CrossRef]
- Soto, M.L.; Falqué, E.; Domínguez, H. Relevance of Natural Phenolics from Grape and Derivative Products in the Formulation of Cosmetics. Cosmetics 2015, 2, 259–276. [Google Scholar] [CrossRef] [Green Version]
- Yuan, X.-Y.; Liu, W.; Hao, J.-C.; Gu, W.-J.; Zhao, Y.-S. Topical Grape Seed Proanthocyandin Extract Reduces Sunburn Cells and Mutant p53 Positive Epidermal Cell Formation, and Prevents Depletion of Langerhans Cells in an Acute Sunburn Model. Photomed. Laser Surg. 2012, 30, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Montagner, S.; Costa, A. Bases biomoleculares do fotoenvelhecimento. An. Bras. Dermatol. 2009, 84, 263–269. [Google Scholar] [CrossRef] [Green Version]
- Yamasaki, R.; Jidoi, J.; Dekio, S. Contact dermatitis from grape bud. Contact Dermat. 1985, 12, 226–227. [Google Scholar] [CrossRef] [PubMed]
- Wróblewska, K.B.; Baby, A.R.; Guaratini, M.T.G.; Moreno, P.R.H. In vitro antioxidant and photoprotective activity of five native Brazilian bamboo species. Ind. Crop. Prod. 2019, 130, 208–215. [Google Scholar] [CrossRef]
- Fontana, A.; Antoniolli, A.; Fernández, M.A.D.; Bottini, R. Phenolics profiling of pomace extracts from different grape varieties cultivated in Argentina. RSC Adv. 2017, 7, 29446–29457. [Google Scholar] [CrossRef] [Green Version]
- Choquenet, B.; Couteau, C.; Paparis, E.; Coiffard, L. Quercetin and Rutin as Potential Sunscreen Agents: Determination of Efficacy by an in Vitro Method. J. Nat. Prod. 2008, 71, 1117–1118. [Google Scholar] [CrossRef]
- Hruza, L.L.; Pentland, A.P. Mechanisms of UV-induced inflammation. J. Investig. Dermatol. 1993, 100, 35. [Google Scholar] [CrossRef] [Green Version]
- Kolbe, L.; Pissavini, M.; Tricaud, C.; Cabanas, C.T.; Dietrich, E.; Matts, P.J. Anti-inflammatory/anti-oxidant activity of ingredients of sunscreen products? Implications for SPF. Int. J. Cosmet. Sci. 2019, 41, 320–324. [Google Scholar] [CrossRef] [Green Version]
- Couteau, C.; Chauvet, C.; Paparis, E.; Coiffard, L. Influence of certain ingredients on the SPF determined in vivo. Arch. Dermatol. Res. 2012, 304, 817–821. [Google Scholar] [CrossRef]
- Fang, F.; Li, J.-M.; Pan, Q.-H.; Huang, W. Determination of red wine flavonoids by HPLC and effect of aging. Food Chem. 2007, 101, 428–433. [Google Scholar] [CrossRef]
- Sisa, M.; Bonnet, S.L.; Ferreira, D.; Van Der Westhuizen, J.H. Photochemistry of Flavonoids. Molecules 2010, 15, 5196–5245. [Google Scholar] [CrossRef] [PubMed]
- Yeh, S.L.; Wang, W.Y.; Huang, C.H.; Hu, L.M. Pro-oxidative effect of β-carotene and the interaction with flavonoids on UVA-induced DNA strand breaks in mouse fibroblast C3H10T1/2 cells. J. Nutr. Biochem. 2005, 16, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Kale, S.; Kavade, E.; Yadav, A.V. Formulation and In-vitro Evaluation for Sun Protection Factor of Crinum asiaticum Linn flower (Family-Amaryllidaceae) Extract Sunscreen Creams. Indian J. Pharm. Educ. 2012, 46, 112–119. [Google Scholar]
- De Oliveira, C.A.; Peres, D.D.; Rugno, C.M.; Kojima, M.; Pinto, C.A.S.D.O.; Consiglieri, V.O.; Kaneko, T.M.; Rosado, C.; Mota, J.P.; Velasco, M.V.R.; et al. Functional photostability and cutaneous compatibility of bioactive UVA sun care products. J. Photochem. Photobiol. B Biol. 2015, 148, 154–159. [Google Scholar] [CrossRef]
- Stevanato, R.; Bertelle, M.; Fabris, S. Photoprotective characteristics of natural antioxidant polyphenols. Regul. Toxicol. Pharmacol. 2014, 69, 71–77. [Google Scholar] [CrossRef] [PubMed]






| INCI 1. | Composition% (w/w) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | G | H | I | J | K | |
| Aqueous phase | |||||||||||
| Ammonium acryloyldimethyltaurate/VP copolymer, trilaureth-4 phosphate, rapeseed oil sorbitol esters, mineral oil and isopropyl palmitate | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 |
| Ammonium acryloyldimethyltaurate vinylpyrrolidone | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 |
| Propylene glycol | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 |
| Ethyl alcohol | 2.50 | 2.50 | 2.50 | 2.50 | 2.50 | 2.50 | 2.50 | 2.50 | 2.50 | 2.50 | 2.50 |
| Disodium EDTA | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Grape pomace extract | 1.46 | 8.54 | 1.46 | 8.54 | 0.00 | 10.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 |
| Water purified q.s. | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
| Oil phase | |||||||||||
| Butylmethoxydibenzoyl methane 2 | 0.73 | 0.73 | 4.27 | 4.27 | 2.50 | 2.50 | 0.00 | 5.00 | 2.50 | 2.50 | 2.50 |
| Ethylhexyl methoxycinnamate 3 | 1.46 | 1.46 | 8.54 | 8.54 | 5.00 | 5.00 | 0.00 | 10.00 | 5.00 | 5.00 | 5.00 |
| Ethylhexyl dimethyl PABA 3 | 1.17 | 1.17 | 6.83 | 6.83 | 4.00 | 4.00 | 0.00 | 8.00 | 4.00 | 4.00 | 4.00 |
| Butyl hydroxy toluene | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Mixture of phenoxyethanol and parabens others 4 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 |
| Source | Y1 | Y2 | Y3 | Y4 | Y5 | Y6 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Df | SS | P | Df | SS | P | Df | SS | P | Df | SS | P | Df | SS | P | Df | SS | P | |
| Regression | 6 | 10.26 | 0.000 | 4 | 599.9 | 0.002 | 6 | 6150 | 0.000 | 6 | 11710 | 0.000 | 7 | 8358 | 0.000 | 3 | 0.756 | 0.000 |
| Linear | ||||||||||||||||||
| X1 | 1 | 0.89 | 0.000 | 1 | 36.7 | 0.021 | 1 | 3016 | 0.000 | 1 | 1645 | 0.000 | 1 | 1665 | 0.000 | 1 | 0.171 | 0.000 |
| X2 | 1 | 0.32 | 0.005 | 1 | 65.2 | 0.010 | 1 | 15 | 0.174 | 1 | 380 | 0.009 | 1 | 209 | 0.014 | 1 | 0.050 | 0.000 |
| X3 | 1 | 0.90 | 0.000 | 1 | 105 | 0.002 | 1 | 541 | 0.003 | 1 | 484 | 0.001 | ||||||
| Quadratic | ||||||||||||||||||
| X1 × X1 | 1 | 0.39 | 0.002 | 1 | 2146 | 0.000 | 1 | 753 | 0.001 | 1 | 942 | 0.000 | 1 | 0.085 | 0.000 | |||
| X2 × X2 | 1 | 0.07 | 0.133 | 1 | 28.2 | 0.030 | 1 | 183 | 0.057 | 1 | 127 | 0.048 | ||||||
| X3 × X3 | 1 | 0.17 | 0.029 | 1 | 103 | 0.144 | 1 | 82 | 0.106 | |||||||||
| Interaction | ||||||||||||||||||
| X1 × X2 | 1 | 8.2 | 0.128 | 1 | 39 | 0.035 | ||||||||||||
| X1 × X3 | ||||||||||||||||||
| X2 × X3 | 1 | 34 | 0.048 | 1 | 98 | 0.079 | ||||||||||||
| Error | 17 | 0.51 | 3 | 5.6 | 17 | 128 | 17 | 44 | 16 | 444 | 20 | 0.043 | ||||||
| Total | 23 | 10.77 | 7 | 605.5 | 23 | 6278 | 23 | 12456 | 23 | 8802 | 23 | 0.799 | ||||||
| Regression Equation | 1 R2 | 2 R2adj | 3 R2pred |
|---|---|---|---|
| ln(Y1) = 0.341 + 0.2817 × X1 + 0.1258 × X2 − 0.857 × X3 − 0.01409 × X1 × X1 − 0.00579 × X2 × X2 + 0.1795 × X3 × X3 | 0.9522 | 0.9354 | 0.8849 |
| Y2 = 24.35 − 1.017 × X1 + 3.631 × X2 − 0.1831 × X2 × X2 + 0.0861 × X1 × X2 | 0.9907 | 0.9783 | 0.8295 |
| Y3 = 328.35 + 16.112 × X1 + 0.722 × X2 − 4.91 × X3 − 0.9844 × X1 × X1 − 0.1092 × X1 × X2 + 0.477 × X2 × X3 | 0.9796 | 0.9723 | 0.9361 |
| Y4 = 96.51 − 12.11 × X1 − 4.31 × X2 + 20.96 × X3 + 0.617 × X1 × X1 + 0.285 × X2 × X2 − 4.39 × X3 × X3 | 0.9401 | 0.9190 | 0.8581 |
| Y5 = 68.08 − 12.19 × X1 − 3.41 × X2 + 21.78 × X3 + 0.691 × X1 × X1 + 0.238 × X2 × X2 − 3.91 × X3 × X3 − 0.808 × X2 × X3 | 0.9495 | 0.9275 | 0.8681 |
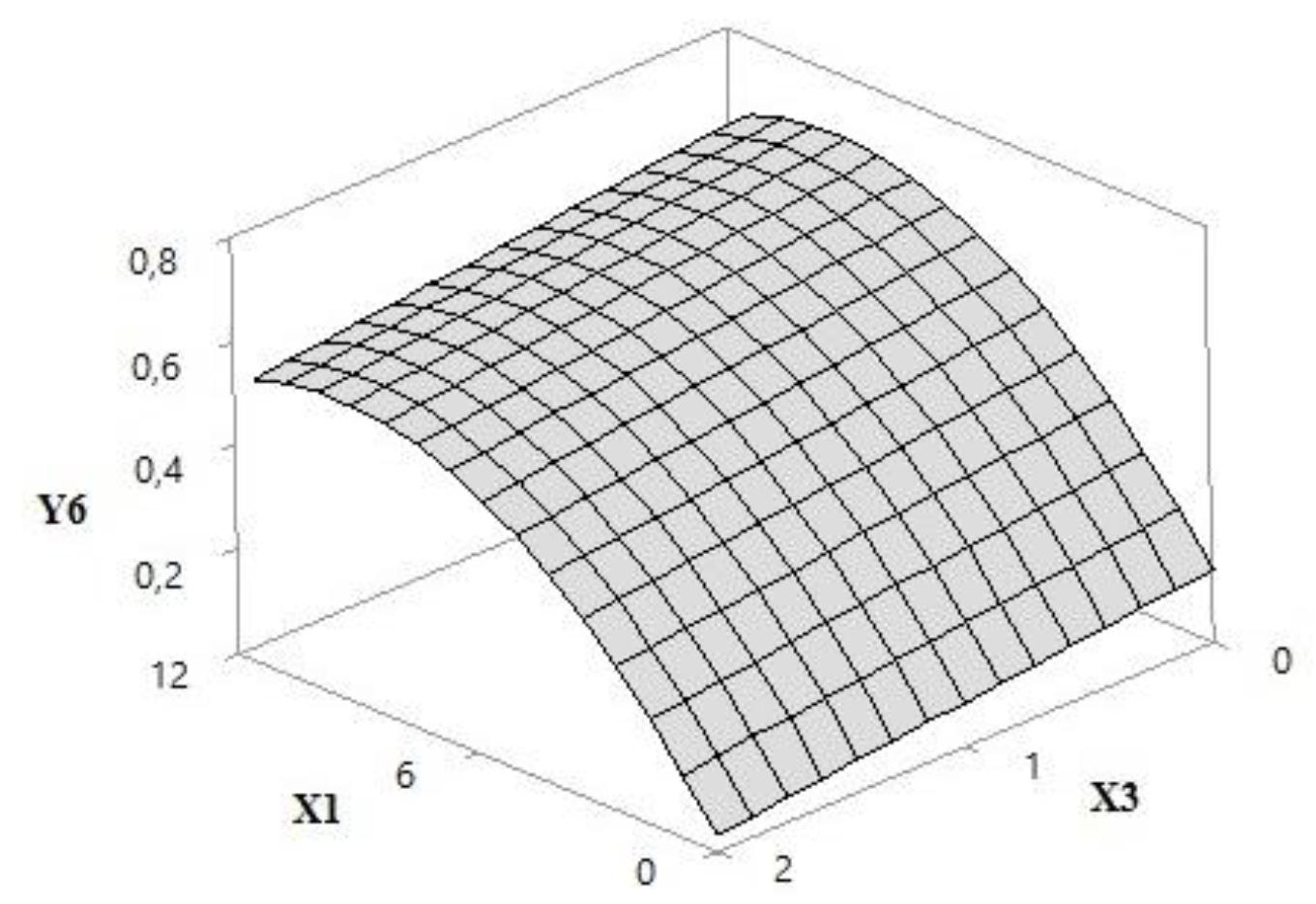
| Y6 = 0.1409 + 0.1159 × X1 − 0.0559 × X3 − 0.006207 × X1 × X1 | 0.9459 | 0.9378 | 0.9082 |
| Formulation | Concentration (% w/w) | Organoleptic Characteristics | ||||
|---|---|---|---|---|---|---|
| UV Filters 1 | GP 2 | Aspect 3 | Color | Odor 4 | pH | |
| A | 3.37 | 1.46 | N | Light purple | C | 5.40 |
| B | 3.37 | 8.54 | N | Wine | C | 5.46 |
| E | 11.50 | 0.00 | N | Yellow | C | 5.40 |
| F | 11.50 | 10.00 | SI | Dark purple | C | 5.49 |
| G | 0.00 | 5.00 | N | Wine | C | 5.46 |
| I | 11.50 | 5.00 | N | Purple | C | 5.51 |
| J | 11.50 | 5.00 | N | Purple | C | 5.45 |
| K | 11.50 | 5.00 | N | Purple | C | 3.38 |
| Formulation | % AAO 1 | Trolox (µmol g−1) 2 |
|---|---|---|
| A | 26.88 ± 0.02 | 306.76 ± 0.02 |
| B | 41.68 ± 0.01 | 545.53 ± 0.01 |
| E | 12.20 ± 0.00 | 64.92 ± 0.00 |
| F | 40.10 ± 0.00 | 519.92 ± 0.00 |
| G | 36.99 ± 0.00 | 469.30 ± 0.00 |
| I | 32.64 ± 0.00 | 398.22 ± 0.00 |
| J | 30.79 ± 0.01 | 368.05 ± 0.01 |
| K | 30.02 ± 0.01 | 355.59 ± 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hübner, A.A.; Sarruf, F.D.; Oliveira, C.A.; Neto, A.V.; Fischer, D.C.H.; Kato, E.T.M.; Lourenço, F.R.; Baby, A.R.; Bacchi, E.M. Safety and Photoprotective Efficacy of a Sunscreen System Based on Grape Pomace (Vitis vinifera L.) Phenolics from Winemaking. Pharmaceutics 2020, 12, 1148. https://doi.org/10.3390/pharmaceutics12121148
Hübner AA, Sarruf FD, Oliveira CA, Neto AV, Fischer DCH, Kato ETM, Lourenço FR, Baby AR, Bacchi EM. Safety and Photoprotective Efficacy of a Sunscreen System Based on Grape Pomace (Vitis vinifera L.) Phenolics from Winemaking. Pharmaceutics. 2020; 12(12):1148. https://doi.org/10.3390/pharmaceutics12121148
Chicago/Turabian StyleHübner, Alexandra A., Fernanda D. Sarruf, Camila A. Oliveira, Alberto V. Neto, Dominique C. H. Fischer, Edna T. M. Kato, Felipe R. Lourenço, André Rolim Baby, and Elfriede M. Bacchi. 2020. "Safety and Photoprotective Efficacy of a Sunscreen System Based on Grape Pomace (Vitis vinifera L.) Phenolics from Winemaking" Pharmaceutics 12, no. 12: 1148. https://doi.org/10.3390/pharmaceutics12121148