Locoregional Treatment of Metastatic Pancreatic Cancer Utilizing Resection, Ablation and Embolization: A Systematic Review
Abstract
Simple Summary
Abstract
1. Introduction
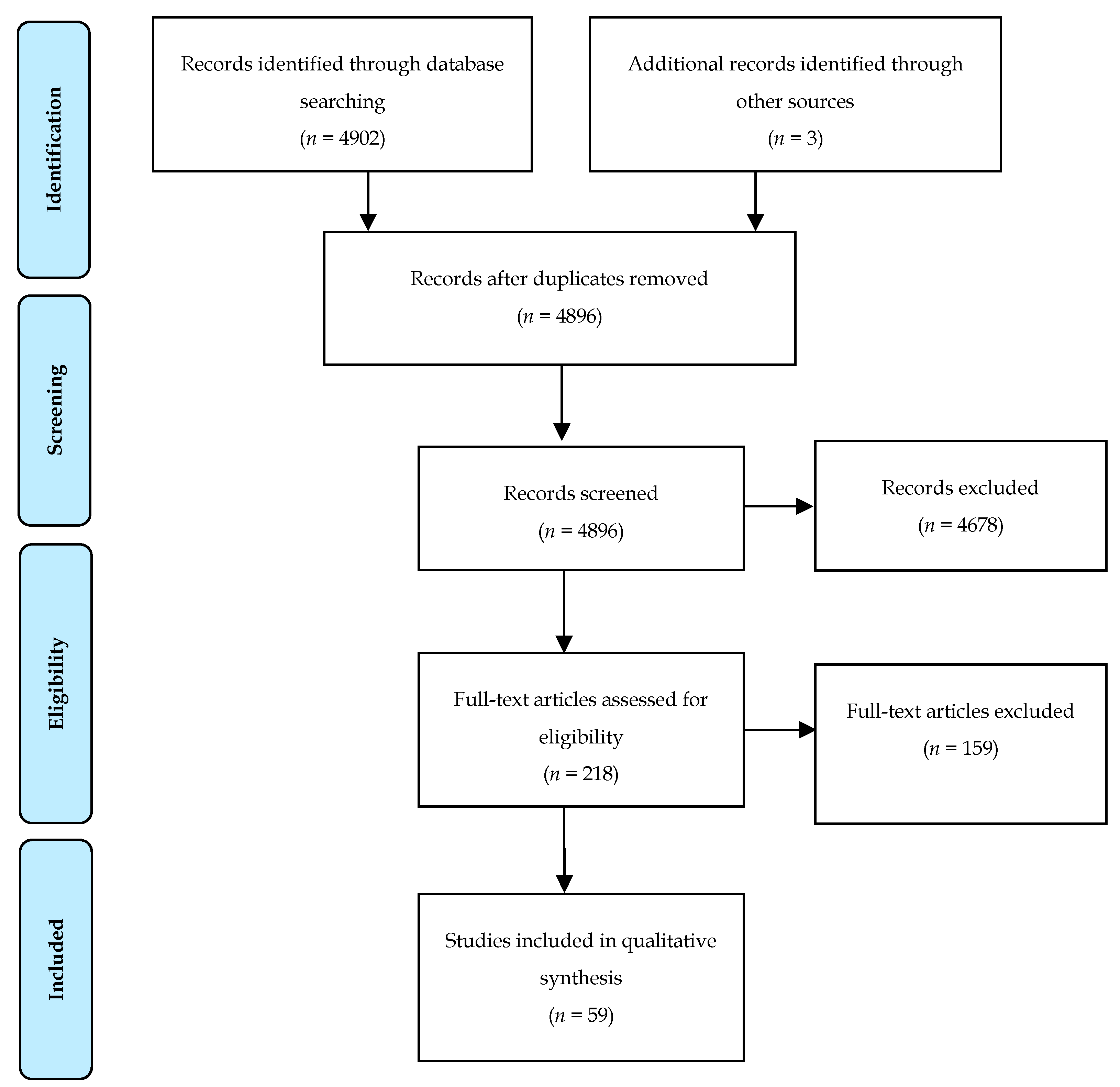
2. Materials and Methods
Definitions
3. Results
3.1. Resection
3.1.1. Primary Pancreatic Tumor Resection
3.1.2. Hepatic Metastasectomy
3.1.3. Pulmonary Metastasectomy
3.2. Ablation
3.2.1. Radiofrequency Ablation (RFA)
3.2.2. Irreversible Electroporation (IRE)
3.2.3. Stereotactic Body Radiotherapy (SBRT)
3.2.4. High-Intensity Focused Ultrasound (HIFU)
3.3. Embolization
3.3.1. Selective Internal Radiation Therapy (SIRT)/Transarterial Radioembolization (TARE)
3.3.2. Transarterial Chemoembolization (TACE)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McGuigan, A.; Kelly, P.; Turkington, R.C.; Jones, C.; Coleman, H.G.; McCain, R.S. Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes. World J. Gastroenterol. 2018, 24, 4846–4861. [Google Scholar] [CrossRef]
- Feig, C.; Gopinathan, A.; Neesse, A.; Chan, D.S.; Cook, N.; Tuveson, D.A. The pancreas cancer microenvironment. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 4266–4276. [Google Scholar] [CrossRef]
- Parisi, L.; Gini, E.; Baci, D.; Tremolati, M.; Fanuli, M.; Bassani, B.; Farronato, G.; Bruno, A.; Mortara, L. Macrophage Polarization in Chronic Inflammatory Diseases: Killers or Builders? J. Immunol. Res. 2018, 2018, 8917804. [Google Scholar] [CrossRef]
- Ryan, D.P.; Hong, T.S.; Bardeesy, N. Pancreatic adenocarcinoma. N. Engl. J. Med. 2014, 371, 2140–2141. [Google Scholar] [CrossRef]
- Le Large, T.Y.S.; Bijlsma, M.F.; Kazemier, G.; van Laarhoven, H.W.M.; Giovannetti, E.; Jimenez, C.R. Key biological processes driving metastatic spread of pancreatic cancer as identified by multi-omics studies. Semin. Cancer Biol. 2017, 44, 153–169. [Google Scholar] [CrossRef]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Hellman, S.; Weichselbaum, R.R. Oligometastases. J. Clin. Oncol. 1995, 13, 8–10. [Google Scholar] [CrossRef]
- Chow, F.C.; Chok, K.S. Colorectal liver metastases: An update on multidisciplinary approach. World J. Hepatol. 2019, 11, 150–172. [Google Scholar] [CrossRef] [PubMed]
- Tsitskari, M.; Filippiadis, D.; Kostantos, C.; Palialexis, K.; Zavridis, P.; Kelekis, N.; Brountzos, E. The role of interventional oncology in the treatment of colorectal cancer liver metastases. Ann. Gastroenterol. 2019, 32, 147–155. [Google Scholar] [CrossRef]
- Grandhi, M.S.; Lafaro, K.J.; Pawlik, T.M. Role of Locoregional and Systemic Approaches for the Treatment of Patients with Metastatic Neuroendocrine Tumors. J. Gastrointest. Surg. 2015, 19, 2273–2282. [Google Scholar] [CrossRef]
- van Veldhuisen, E.; van den Oord, C.; Brada, L.J.; Walma, M.S.; Vogel, J.A.; Wilmink, J.W.; Del Chiaro, M.; van Lienden, K.P.; Meijerink, M.R.; van Tienhoven, G.; et al. Locally Advanced Pancreatic Cancer: Work-Up, Staging, and Local Intervention Strategies. Cancers 2019, 11, 976. [Google Scholar] [CrossRef]
- Hofland, J.; de Herder, W.W.; Kann, P.H. Turning Up the Heat: Endoscopic Ablation of Pancreatic Neuroendocrine Neoplasms. J. Clin. Endocrinol. Metab. 2019, 104, 5053–5055. [Google Scholar] [CrossRef] [PubMed]
- Venkat, S.R.; Mohan, P.P.; Gandhi, R.T. Colorectal Liver Metastasis: Overview of Treatment Paradigm Highlighting the Role of Ablation. AJR Am. J. Roentgenol. 2018, 210, 883–890. [Google Scholar] [CrossRef]
- Shiina, S.; Sato, K.; Tateishi, R.; Shimizu, M.; Ohama, H.; Hatanaka, T.; Takawa, M.; Nagamatsu, H.; Imai, Y. Percutaneous Ablation for Hepatocellular Carcinoma: Comparison of Various Ablation Techniques and Surgery. Can. J. Gastroenterol. Hepatol. 2018, 2018, 4756147. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Watanabe, H.; Okada, M.; Kaji, Y.; Satouchi, M.; Sato, Y.; Yamabe, Y.; Onaya, H.; Endo, M.; Sone, M.; Arai, Y. New response evaluation criteria in solid tumours-revised RECIST guideline (version 1.1). Gan To Kagaku Ryoho 2009, 36, 2495–2501. [Google Scholar]
- Trotti, A.; Colevas, A.D.; Setser, A.; Rusch, V.; Jaques, D.; Budach, V.; Langer, C.; Murphy, B.; Cumberlin, R.; Coleman, C.N.; et al. CTCAE v3.0: Development of a comprehensive grading system for the adverse effects of cancer treatment. Semin. Radiat. Oncol. 2003, 13, 176–181. [Google Scholar] [CrossRef]
- Oweira, H.; Petrausch, U.; Helbling, D.; Schmidt, J.; Mannhart, M.; Mehrabi, A.; Schöb, O.; Giryes, A.; Decker, M.; Abdel-Rahman, O. Prognostic value of site-specific metastases in pancreatic adenocarcinoma: A Surveillance Epidemiology and End Results database analysis. World J. Gastroenterol. 2017, 23, 1872–1880. [Google Scholar] [CrossRef]
- Wang, L.; Yang, L.; Chen, L.; Chen, Z. Do Patients Diagnosed with Metastatic Pancreatic Cancer Benefit from Primary Tumor Surgery? A Propensity-Adjusted, Population-Based Surveillance, Epidemiology and End Results (SEER) Analysis. Med. Sci. Monit. 2019, 25, 8230–8241. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, R.; Michalski, C.W.; Liu, B.; Liao, Q.; Kleeff, J. Surgery for synchronous and metachronous single-organ metastasis of pancreatic cancer: A SEER database analysis and systematic literature review. Sci. Rep. 2020, 10, 4444. [Google Scholar] [CrossRef]
- Tao, L.; Yuan, C.; Ma, Z.; Jiang, B.; Xiu, D. Surgical resection of a primary tumor improves survival of metastatic pancreatic cancer: A population-based study. Cancer Manag. Res. 2017, 9, 471–479. [Google Scholar] [CrossRef]
- McKenzie, S.; Mailey, B.; Artinyan, A.; Kim, J.; Ellenhorn, J.D. The incidence and outcomes of pancreatectomy in patients with metastatic pancreatic adenocarcinoma. Jop 2010, 11, 341–347. [Google Scholar]
- Yamada, H.; Hirano, S.; Tanaka, E.; Shichinohe, T.; Kondo, S. Surgical treatment of liver metastases from pancreatic cancer. HPB 2006, 8, 85–88. [Google Scholar] [CrossRef][Green Version]
- Gleisner, A.L.; Assumpcao, L.; Cameron, J.L.; Wolfgang, C.L.; Choti, M.A.; Herman, J.M.; Schulick, R.D.; Pawlik, T.M. Is resection of periampullary or pancreatic adenocarcinoma with synchronous hepatic metastasis justified? Cancer 2007, 110, 2484–2492. [Google Scholar] [CrossRef]
- Shrikhande, S.V.; Kleeff, J.; Reiser, C.; Weitz, J.; Hinz, U.; Esposito, I.; Schmidt, J.; Friess, H.; Büchler, M.W. Pancreatic resection for M1 pancreatic ductal adenocarcinoma. Ann. Surg. Oncol. 2007, 14, 118–127. [Google Scholar] [CrossRef]
- De Jong, M.C.; Farnell, M.B.; Sclabas, G.; Cunningham, S.C.; Cameron, J.L.; Geschwind, J.F.; Wolfgang, C.L.; Herman, J.M.; Edil, B.H.; Choti, M.A.; et al. Liver-directed therapy for hepatic metastases in patients undergoing pancreaticoduodenectomy: A dual-center analysis. Ann. Surg. 2010, 252, 142–148. [Google Scholar] [CrossRef]
- Dünschede, F.; Will, L.; von Langsdorf, C.; Möhler, M.; Galle, P.R.; Otto, G.; Vahl, C.F.; Junginger, T. Treatment of metachronous and simultaneous liver metastases of pancreatic cancer. Eur. Surg. Res. 2010, 44, 209–213. [Google Scholar] [CrossRef]
- Klein, F.; Puhl, G.; Guckelberger, O.; Pelzer, U.; Pullankavumkal, J.R.; Guel, S.; Neuhaus, P.; Bahra, M. The impact of simultaneous liver resection for occult liver metastases of pancreatic adenocarcinoma. Gastroenterol. Res. Pract. 2012, 2012, 939350. [Google Scholar] [CrossRef]
- Zanini, N.; Lombardi, R.; Masetti, M.; Giordano, M.; Landolfo, G.; Jovine, E. Surgery for isolated liver metastases from pancreatic cancer. Updates Surg. 2015, 67, 19–25. [Google Scholar] [CrossRef]
- Bahra, M.; Pratschke, J.; Klein, F.; Neuhaus, P.; Boas-Knoop, S.; Puhl, G.; Denecke, T.; Pullankavumkal, J.R.; Sinn, M.; Riess, H.; et al. Cytoreductive Surgery for Pancreatic Cancer Improves Overall Outcome of Gemcitabine-Based Chemotherapy. Pancreas 2015, 44, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Tachezy, M.; Gebauer, F.; Janot, M.; Uhl, W.; Zerbi, A.; Montorsi, M.; Perinel, J.; Adham, M.; Dervenis, C.; Agalianos, C.; et al. Synchronous resections of hepatic oligometastatic pancreatic cancer: Disputing a principle in a time of safe pancreatic operations in a retrospective multicenter analysis. Surgery 2016, 160, 136–144. [Google Scholar] [CrossRef]
- Hackert, T.; Niesen, W.; Hinz, U.; Tjaden, C.; Strobel, O.; Ulrich, A.; Michalski, C.W.; Büchler, M.W. Radical surgery of oligometastatic pancreatic cancer. Eur. J. Surg. Oncol. 2017, 43, 358–363. [Google Scholar] [CrossRef]
- Crippa, S.; Bittoni, A.; Sebastiani, E.; Partelli, S.; Zanon, S.; Lanese, A.; Andrikou, K.; Muffatti, F.; Balzano, G.; Reni, M.; et al. Is there a role for surgical resection in patients with pancreatic cancer with liver metastases responding to chemotherapy? Eur. J. Surg. Oncol. 2016, 42, 1533–1539. [Google Scholar] [CrossRef]
- Wright, G.P.; Poruk, K.E.; Zenati, M.S.; Steve, J.; Bahary, N.; Hogg, M.E.; Zuriekat, A.H.; Wolfgang, C.L.; Zeh, H.J., 3rd; Weiss, M.J. Primary Tumor Resection Following Favorable Response to Systemic Chemotherapy in Stage IV Pancreatic Adenocarcinoma with Synchronous Metastases: A Bi-institutional Analysis. J. Gastrointest. Surg. 2016, 20, 1830–1835. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, S.C.; Song, K.B.; Kim, J.; Kang, D.R.; Lee, J.H.; Park, K.M.; Lee, Y.J. Improved survival after palliative resection of unsuspected stage IV pancreatic ductal adenocarcinoma. HPB 2016, 18, 325–331. [Google Scholar] [CrossRef]
- Andreou, A.; Knitter, S.; Klein, F.; Malinka, T.; Schmelzle, M.; Struecker, B.; Schmuck, R.B.; Noltsch, A.R.; Lee, D.; Pelzer, U.; et al. The role of hepatectomy for synchronous liver metastases from pancreatic adenocarcinoma. Surg. Oncol. 2018, 27, 688–694. [Google Scholar] [CrossRef]
- Kandel, P.; Wallace, M.B.; Stauffer, J.; Bolan, C.; Raimondo, M.; Woodward, T.A.; Gomez, V.; Ritter, A.W.; Asbun, H.; Mody, K. Survival of Patients with Oligometastatic Pancreatic Ductal Adenocarcinoma Treated with Combined Modality Treatment Including Surgical Resection: A Pilot Study. J. Pancreat. Cancer 2018, 4, 88–94. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, J.; Lui, W.; Huo, Y.; Fu, X.; Yang, M.; Hua, R.; Wang, L.; Sun, Y. Patients with hepatic oligometastatic pancreatic body/tail ductal adenocarcinoma may benefit from synchronous resection. HPB 2020, 22, 91–101. [Google Scholar] [CrossRef]
- Gu, J.; Xu, Z.; Ma, Y.; Chen, H.; Wang, D.; Deng, X.; Cheng, D.; Xie, J.; Jin, J.; Zhan, X.; et al. Surgical resection of metastatic pancreatic cancer: Is it worth it?-a 15-year experience at a single Chinese center. J. Gastrointest. Oncol. 2020, 11, 319–328. [Google Scholar] [CrossRef]
- Schwarz, C.; Fitschek, F.; Primavesi, F.; Stättner, S.; Margonis, G.A.; Weiss, M.A.; Stavrou, G.A.; Oldhafer, K.J.; Kornprat, P.; Wundsam, H.; et al. Metachronous hepatic resection for liver only pancreatic metastases. Surg. Oncol. 2020, 35, 169–173. [Google Scholar] [CrossRef]
- Arnaoutakis, G.J.; Rangachari, D.; Laheru, D.A.; Iacobuzio-Donahue, C.A.; Hruban, R.H.; Herman, J.M.; Edil, B.H.; Pawlik, T.M.; Schulick, R.D.; Cameron, J.L.; et al. Pulmonary Resection for Isolated Pancreatic Adenocarcinoma Metastasis: An Analysis of Outcomes and Survival. J. Gastrointest. Surg. 2011, 15, 1611–1617. [Google Scholar] [CrossRef]
- Thomas, R.M.; Truty, M.J.; Nogueras-Gonzalez, G.M.; Fleming, J.B.; Vauthey, J.N.; Pisters, P.W.; Lee, J.E.; Rice, D.C.; Hofstetter, W.L.; Wolff, R.A.; et al. Selective reoperation for locally recurrent or metastatic pancreatic ductal adenocarcinoma following primary pancreatic resection. J. Gastrointest. Surg. 2012, 16, 1696–1704. [Google Scholar] [CrossRef]
- Downs-Canner, S.; Zenati, M.; Boone, B.A.; Varley, P.R.; Steve, J.; Hogg, M.E.; Zureikat, A.; Zeh, H.J.; Lee, K.K. The indolent nature of pulmonary metastases from ductal adenocarcinoma of the pancreas. J. Surg. Oncol. 2015, 112, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Robinson, L.A.; Tanvetyanon, T.; Springett, G.; Fontaine, J.; Toloza, E.; Hodul, P.; Pimiento, J.M.; Malafa, M. Pulmonary metastasectomy for suspected pancreaticobiliary cancer. J. Thorac. Cardiovasc. Surg. 2016, 152, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Kruger, S.; Haas, M.; Burger, P.J.; Ormanns, S.; Modest, D.P.; Westphalen, C.B.; Michl, M.; Kleespies, A.; Angele, M.K.; Hartwig, W.; et al. Isolated pulmonary metastases define a favorable subgroup in metastatic pancreatic cancer. Pancreatology 2016, 16, 593–598. [Google Scholar] [CrossRef]
- Nakajima, M.; Ueno, T.; Suzuki, N.; Matsui, H.; Shindo, Y.; Sakamoto, K.; Tokuhisa, Y.; Tokumitsu, Y.; Takeda, S.; Yoshino, S.; et al. Novel Indications for Surgical Resection of Metachronous Lung Metastases From Pancreatic Cancer After Curative Resection. J. Clin. Gastroenterol. 2017, 51, e34–e38. [Google Scholar] [CrossRef] [PubMed]
- Okui, M.; Yamamichi, T.; Asakawa, A.; Harada, M.; Horio, H. Resection for Pancreatic Cancer Lung Metastases. Korean J. Thorac. Cardiovasc. Surg. 2017, 50, 326–328. [Google Scholar] [CrossRef]
- Yasukawa, M.; Kawaguchi, T.; Kawai, N.; Tojo, T.; Taniguchi, S. Surgical Treatment for Pulmonary Metastasis of Pancreatic Ductal Adenocarcinoma: Study of 12 Cases. Anticancer Res. 2017, 37, 5573–5576. [Google Scholar] [CrossRef]
- Ilmer, M.; Schiergens, T.S.; Renz, B.W.; Schneider, C.; Sargut, M.; Waligora, R.; Weniger, M.; Hartwig, W.; Ceyhan, G.O.; Friess, H.; et al. Oligometastatic pulmonary metastasis in pancreatic cancer patients: Safety and outcome of resection. Surg. Oncol. 2019, 31, 16–21. [Google Scholar] [CrossRef]
- Groot, V.P.; Blair, A.B.; Gemenetzis, G.; Ding, D.; Burkhart, R.A.; van Oosten, A.F.; Molenaar, I.Q.; Cameron, J.L.; Weiss, M.J.; Yang, S.C.; et al. Isolated pulmonary recurrence after resection of pancreatic cancer: The effect of patient factors and treatment modalities on survival. HPB 2019, 21, 998–1008. [Google Scholar] [CrossRef]
- Kaiho, T.; Suzuki, H.; Yamamoto, T.; Morimoto, J.; Sakairi, Y.; Wada, H.; Nakajima, T.; Yoshino, I. Surgical outcomes of pulmonary metastasis from hepatopancreatobiliary carcinomas: A comparison with pulmonary metastasis from colorectal carcinomas. Surg. Today 2019, 49, 762–768. [Google Scholar] [CrossRef]
- Shimizu, T.; Taniguchi, K.; Asakuma, M.; Komeda, K.; Inoue, Y.; Lee, S.W.; Hirokawa, F.; Uchiyama, K. Initial pulmonary metastasis after pancreatectomy for pancreatic ductal adenocarcinoma. Surg. Today 2020, 50, 413–418. [Google Scholar] [CrossRef]
- Liu, K.H.; Hung, C.Y.; Hsueh, S.W.; Chang, P.H.; Chen, Y.Y.; Lu, C.H.; Chen, P.T.; Yeh, K.Y.; Huang, P.W.; Tsang, N.M.; et al. Lung Metastases in Patients with Stage IV Pancreatic Cancer: Prevalence, Risk Factors, and Survival Impact. J. Clin. Med. 2019, 8, 1402. [Google Scholar] [CrossRef]
- Park, J.B.; Kim, Y.H.; Kim, J.; Chang, H.M.; Kim, T.W.; Kim, S.C.; Kim, P.N.; Han, D.J. Radiofrequency ablation of liver metastasis in patients with locally controlled pancreatic ductal adenocarcinoma. J. Vasc. Interv. Radiol. 2012, 23, 635–641. [Google Scholar] [CrossRef]
- Hua, Y.Q.; Wang, P.; Zhu, X.Y.; Shen, Y.H.; Wang, K.; Shi, W.D.; Lin, J.H.; Meng, Z.Q.; Chen, Z.; Chen, H. Radiofrequency ablation for hepatic oligometastatic pancreatic cancer: An analysis of safety and efficacy. Pancreatology 2017, 17, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Kim, J.H.; Kim, S.Y.; Won, H.J.; Shin, Y.M.; Kim, P.N. Percutaneous Radiofrequency Ablation for Metachronous Hepatic Metastases after Curative Resection of Pancreatic Adenocarcinoma. Korean J. Radiol. 2020, 21, 316–324. [Google Scholar] [CrossRef]
- Hong, Y.; Rice, J.; Sharma, D.; Martin, R.C.G., 2nd. The use of IRE in multi-modality treatment for oligometastatic pancreatic cancer. Am. J. Surg. 2018, 216, 106–110. [Google Scholar] [CrossRef]
- Gkika, E.; Adebahr, S.; Kirste, S.; Schimek-Jasch, T.; Wiehle, R.; Claus, R.; Wittel, U.; Nestle, U.; Baltas, D.; Grosu, A.L.; et al. Stereotactic body radiotherapy (SBRT) in recurrent or oligometastatic pancreatic cancer: A toxicity review of simultaneous integrated protection (SIP) versus conventional SBRT. Strahlenther. Onkol. 2017, 193, 433–443. [Google Scholar] [CrossRef]
- Su, T.S.; Liang, P.; Lu, H.Z.; Liang, J.N.; Liu, J.M.; Zhou, Y.; Gao, Y.C.; Tang, M.Y. Stereotactic body radiotherapy using CyberKnife for locally advanced unresectable and metastatic pancreatic cancer. World J. Gastroenterol. 2015, 21, 8156–8162. [Google Scholar] [CrossRef]
- Lischalk, J.W.; Burke, A.; Chew, J.; Elledge, C.; Gurka, M.; Marshall, J.; Pishvaian, M.; Collins, S.; Unger, K. Five-Fraction Stereotactic Body Radiation Therapy (SBRT) and Chemotherapy for the Local Management of Metastatic Pancreatic Cancer. J. Gastrointest. Cancer 2018, 49, 116–123. [Google Scholar] [CrossRef]
- Chang, D.T.; Schellenberg, D.; Shen, J.; Kim, J.; Goodman, K.A.; Fisher, G.A.; Ford, J.M.; Desser, T.; Quon, A.; Koong, A.C. Stereotactic radiotherapy for unresectable adenocarcinoma of the pancreas. Cancer 2009, 115, 665–672. [Google Scholar] [CrossRef]
- Scorsetti, M.; Comito, T.; Franceschini, D.; Franzese, C.; Prete, M.G.; D’Alessio, A.; Bozzarelli, S.; Rimassa, L.; Santoro, A. Is there an oligometastatic state in pancreatic cancer? Practical clinical considerations raise the question. Br. J. Radiol. 2020, 93, 20190627. [Google Scholar] [CrossRef]
- Li, X.; Wang, K.; Zheng, L.; Meng, Z. Retrospective analysis of high intensity focused ultrasound combined with S-1 in the treatment of metastatic pancreatic cancer after failure of gemcitabine. Am. J. Cancer Res. 2016, 6, 84–90. [Google Scholar]
- Ouyang, H.; Ma, W.; Zhang, T.; Liu, F.; Zhao, L.; Fang, M.; Quan, M.; Pan, Z. Systemic chemotherapy in combination with liver-directed therapy improves survival in patients with pancreatic adenocarcinoma and synchronous liver metastases. Pancreatology 2018, 18, 983–989. [Google Scholar] [CrossRef]
- Cao, C.; Yan, T.D.; Morris, D.L.; Bester, L. Radioembolization with yttrium-90 microspheres for pancreatic cancer liver metastases: Results from a pilot study. Tumori 2010, 96, 955–958. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Michl, M.; Haug, A.R.; Jakobs, T.F.; Paprottka, P.; Hoffmann, R.T.; Bartenstein, P.; Boeck, S.; Haas, M.; Laubender, R.P.; Heinemann, V. Radioembolization with Yttrium-90 microspheres (SIRT) in pancreatic cancer patients with liver metastases: Efficacy, safety and prognostic factors. Oncology 2014, 86, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, P.; Do, C.; Lipton, L.; Cade, D.N.; Tapner, M.J.; Price, D.; Bower, G.D.; Dowling, R.; Lichtenstein, M.; van Hazel, G.A. Phase II trial of selective internal radiation therapy and systemic chemotherapy for liver-predominant metastases from pancreatic adenocarcinoma. BMC Cancer 2015, 15, 802. [Google Scholar] [CrossRef]
- Kim, A.Y.; Unger, K.; Wang, H.; Pishvaian, M.J. Incorporating Yttrium-90 trans-arterial radioembolization (TARE) in the treatment of metastatic pancreatic adenocarcioma: A single center experience. BMC Cancer 2016, 16, 492. [Google Scholar] [CrossRef]
- Kim, A.Y.; Frantz, S.; Brower, J.; Akhter, N. Radioembolization with Yttrium-90 Microspheres for the Treatment of Liver Metastases of Pancreatic Adenocarcinoma: A Multicenter Analysis. J. Vasc. Interv. Radiol. 2019, 30, 298–304.e292. [Google Scholar] [CrossRef]
- Nezami, N.; Camacho, J.C.; Kokabi, N.; El-Rayes, B.F.; Kim, H.S. Phase Ib trial of gemcitabine with yttrium-90 in patients with hepatic metastasis of pancreatobiliary origin. J. Gastrointest. Oncol. 2019, 10, 944–956. [Google Scholar] [CrossRef]
- Kayaleh, R.; Krzyston, H.; Rishi, A.; Naziri, J.; Frakes, J.; Choi, J.; El-Haddad, G.; Parikh, N.; Sweeney, J.; Kis, B. Transarterial Radioembolization Treatment of Pancreatic Cancer Patients with Liver-Dominant Metastatic Disease Using Yttrium-90 Glass Microspheres: A Single-Institution Retrospective Study. J. Vasc. Interv. Radiol. 2020, 31, 1060–1068. [Google Scholar] [CrossRef]
- Kim, J.H.; Choi, E.K.; Yoon, H.K.; Ko, G.Y.; Sung, K.B.; Gwon, D.I. Transcatheter arterial chemoembolization for hepatic recurrence after curative resection of pancreatic adenocarcinoma. Gut Liver 2010, 4, 384–388. [Google Scholar] [CrossRef]
- Azizi, A.; Naguib, N.N.; Mbalisike, E.; Farshid, P.; Emami, A.H.; Vogl, T.J. Liver metastases of pancreatic cancer: Role of repetitive transarterial chemoembolization (TACE) on tumor response and survival. Pancreas 2011, 40, 1271–1275. [Google Scholar] [CrossRef]
- Kotoyan, R.; Metzger, T.; Tatum, C.; Robbins, K.; Martin, R.C., 2nd. Hepatic arterial therapy with drug-eluting beads in the management of metastatic pancreatic carcinoma to the liver: A multi-institutional registry. J. Oncol. 2012, 2012, 168303. [Google Scholar] [CrossRef]
- Sun, J.H.; Zhou, T.Y.; Zhang, Y.L.; Zhou, G.H.; Nie, C.H.; Zhu, T.Y.; Chen, S.Q.; Wang, B.Q.; Ye, S.; Shen, Y.; et al. Efficacy of transcatheter arterial chemoembolization for liver metastases arising from pancreatic cancer. Oncotarget 2017, 8, 39746–39755. [Google Scholar] [CrossRef]
- Vogl, T.J.; Mohamed, S.A.; Albrecht, M.H.; Gruber-Roh, T.; Lin, H.; Nour Eldin, N.E.A.; Bednarova, I.; Naguib, N.N.; Panahi, B. Transarterial chemoembolization in pancreatic adenocarcinoma with liver metastases: MR-based tumor response evaluation, apparent diffusion coefficient (ADC) patterns, and survival rates. Pancreatology 2018, 18, 94–99. [Google Scholar] [CrossRef]
- Das, S.K.; Wang, J.L.; Li, B.; Zhang, C.; Yang, H.F. Clinical effectiveness of combined interventional therapy as a salvage modality for unresectable pancreatic carcinoma. Oncol. Lett. 2019, 18, 375–385. [Google Scholar] [CrossRef]
- Tanaka, M.; Mihaljevic, A.L.; Probst, P.; Heckler, M.; Klaiber, U.; Heger, U.; Büchler, M.W.; Hackert, T. Meta-analysis of recurrence pattern after resection for pancreatic cancer. Br. J. Surg. 2019, 106, 1590–1601. [Google Scholar] [CrossRef]
- Suenaga, M.; Fujii, T.; Kanda, M.; Takami, H.; Okumura, N.; Inokawa, Y.; Kobayashi, D.; Tanaka, C.; Yamada, S.; Sugimoto, H.; et al. Pattern of first recurrent lesions in pancreatic cancer: Hepatic relapse is associated with dismal prognosis and portal vein invasion. Hepatogastroenterology 2014, 61, 1756–1761. [Google Scholar]
- Yachida, S.; Jones, S.; Bozic, I.; Antal, T.; Leary, R.; Fu, B.; Kamiyama, M.; Hruban, R.H.; Eshleman, J.R.; Nowak, M.A.; et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 2010, 467, 1114–1117. [Google Scholar] [CrossRef]
- Kamisawa, T.; Isawa, T.; Koike, M.; Tsuruta, K.; Okamoto, A. Hematogenous metastases of pancreatic ductal carcinoma. Pancreas 1995, 11, 345–349. [Google Scholar] [CrossRef]
- Sakaguchi, T.; Valente, R.; Tanaka, K.; Satoi, S.; Del Chiaro, M. Surgical treatment of metastatic pancreatic ductal adenocarcinoma: A review of current literature. Pancreatology 2019, 19, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Satoi, S.; Yamamoto, T.; Yamaki, S.; Sakaguchi, T.; Sekimoto, M. Surgical indication for and desirable outcomes of conversion surgery in patients with initially unresectable pancreatic ductal adenocarcinoma. Ann. Gastroenterol. Surg. 2020, 4, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Frigerio, I.; Regi, P.; Giardino, A.; Scopelliti, F.; Girelli, R.; Bassi, C.; Gobbo, S.; Martini, P.T.; Capelli, P.; D’Onofrio, M.; et al. Downstaging in Stage IV Pancreatic Cancer: A New Population Eligible for Surgery? Ann. Surg. Oncol. 2017, 24, 2397–2403. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Singh, T.; Chaudhary, A. Synchronous resection of solitary liver metastases with pancreaticoduodenectomy. Jop 2010, 11, 434–438. [Google Scholar] [PubMed]
- Ballehaninna, U.K.; Chamberlain, R.S. Serum CA 19-9 as a Biomarker for Pancreatic Cancer-A Comprehensive Review. Indian J. Surg. Oncol. 2011, 2, 88–100. [Google Scholar] [CrossRef]
- Collisson, E.A.; Bailey, P.; Chang, D.K.; Biankin, A.V. Molecular subtypes of pancreatic cancer. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Geboers, B.; Ruarus, A.H.; Nieuwenhuizen, S.; Puijk, R.S.; Scheffer, H.J.; de Gruijl, T.D.; Meijerink, M.R. Needle-guided ablation of locally advanced pancreatic cancer: Cytoreduction or immunomodulation by in vivo vaccination? Chin. Clin. Oncol. 2019, 8, 61. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.; Chen, J.; He, L.; Liao, M.; Yuan, Y.; Zeng, J.; Li, J.; Zuo, J.; Xu, K. Combination treatment with comprehensive cryoablation and immunotherapy in metastatic pancreatic cancer. Pancreas 2013, 42, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Wen, X.; Tian, L.; Li, T.; Xu, C.; Wen, X.; Melancon, M.P.; Gupta, S.; Shen, B.; Peng, W.; et al. Irreversible electroporation reverses resistance to immune checkpoint blockade in pancreatic cancer. Nat. Commun. 2019, 10, 899. [Google Scholar] [CrossRef]
- Narayanan, J.S.S.; Ray, P.; Hayashi, T.; Whisenant, T.C.; Vicente, D.; Carson, D.A.; Miller, A.M.; Schoenberger, S.P.; White, R.R. Irreversible Electroporation Combined with Checkpoint Blockade and TLR7 Stimulation Induces Antitumor Immunity in a Murine Pancreatic Cancer Model. Cancer Immunol. Res. 2019, 7, 1714–1726. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Verma, V.; Lazenby, A.; Ly, Q.P.; Berim, L.D.; Schwarz, J.K.; Madiyalakan, M.; Nicodemus, C.F.; Hollingsworth, M.A.; Meza, J.L.; et al. Phase I/II Trial of Neoadjuvant Oregovomab-based Chemoimmunotherapy Followed by Stereotactic Body Radiotherapy and Nelfinavir For Locally Advanced Pancreatic Adenocarcinoma. Am. J. Clin. Oncol. 2019, 42, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Yasmin-Karim, S.; Bruck, P.T.; Moreau, M.; Kunjachan, S.; Chen, G.Z.; Kumar, R.; Grabow, S.; Dougan, S.K.; Ngwa, W. Radiation and Local Anti-CD40 Generate an Effective in situ Vaccine in Preclinical Models of Pancreatic Cancer. Front. Immunol. 2018, 9, 2030. [Google Scholar] [CrossRef]
- Lin, M.; Liang, S.; Wang, X.; Liang, Y.; Zhang, M.; Chen, J.; Niu, L.; Xu, K. Short-term clinical efficacy of percutaneous irreversible electroporation combined with allogeneic natural killer cell for treating metastatic pancreatic cancer. Immunol. Lett. 2017, 186, 20–27. [Google Scholar] [CrossRef] [PubMed]

| Authors | Year | Design | Nr. of Patients (mPDAC) | Study Details | Synchronous/Metachronous | Morbidity, Grade 3+ | Mortality | Chemotherapy | Median Overall Survival (Months) |
|---|---|---|---|---|---|---|---|---|---|
| McKenzie [19] | 2010 | R | 4649 | 92 P 4557 NR | 92/0 | NS | P: 0–17.5 NR: 5.8–34.1% | P: 36% NR: 38.4% | From primary res: P: 6.3 NR: 4.9 |
| Tao [20] | 2017 | R | 28,918 | 467 P 28,451 NR | NS | NS | NS | P: 53% NR: 45% | From primary diagnosis: P: 7 NR: 2 |
| Oweira [21] | 2017 | R | 13,233 | 160 P 504 M 14,812 NR | NS | NS | NS | NS | NS |
| Wang [22] | 2019 | R | 2694 | 365 P 2329 NR | NS | NS | NS | PSM 1: P: 69% NR: 66% | From primary res (PSM 1): P: 11.6 NR: 9 |
| Liu [23] | 2020 | R | 11,541 | 133 P 24 PM 6999 NR | NS | NS | NS | NS | From primary diagnosis: P: 10–14 NR: 4–6 |
| Treatment | Positive Prognostic Factors— Patient Characteristics | Positive Prognostic Factors— Disease Characteristics (Primary Tumor) | Positive Prognostic Factors— Disease Characteristics (Metastases) | Positive Prognostic Factors—Treatment(s) |
|---|---|---|---|---|
| Primary resection | Younger age [20,21] Caucasian ethnicity [20,21] Married [20,21] Female [20,21] Later era of diagnosis [22] | Tumor in pancreatic head [20] Well/moderate differentiation of tumor (histological grade) [20,22,23] | Longer DFI [23] Single-organ metastatic disease [23] | Chemotherapy (pre- and/or post local treatment) [20,22] Primary tumor resection [20,21,22,23] Local metastatic treatment [21] |
| Hepatic resection | Lower tumor stage [24,25] Tumor in pancreatic body/tail [26] Smaller primary tumor size [27] Well/moderate differentiation of tumor (histological grade) [25] Lower tumor marker CA19-9 levels pre- or post-treatment(s) [25,28,29] | Fewer and/or smaller liver lesions [28] Metachronous (instead of synchronous) [30] Absence of lymph node status [25,31] | Chemotherapy (pre- and/or post local treatment) [25,28,29] Primary tumor resection [28,29] Better primary resection status [24,31] Better liver resection status [25] | |
| Pulmonary resection | Well/moderate differentiation of tumor (histological grade) [32] Lower tumor marker CA19-9 levels pre- and/or post-treatment(s) [33,34] | Fewer and/or smaller lung lesions [34,35] Longer DFI [32,34,36] | ||
| Ablation | Younger age [37] Performance status 0–1 [37] | Lower tumor stage [38] Tumor in pancreatic body/tail [39] Well/moderate differentiation of tumor (histological grade) [37,38,40] Neutrophil-to-lymphocyte ratio (NLR) < 2.5 [38] | Fewer and/or smaller liver/lung lesions [37,38,39,40,41] Longer DFI [38,42] Single-organ metastatic disease [38] | |
| Embolization | Male [43] | Smaller primary tumor size [44] Lower tumor marker CA19-9 levels pre- or post-treatment(s) [45] Absence of ascites [44] Decreased albumin and bilirubin prior to local treatment [46] | Fewer and/or smaller liver/lung lesions [47] Single-organ metastatic disease [48] | Primary tumor resection [46] Local metastatic treatment [44] |
| Authors | Year | Design | Nr. of Patients (Hepatic mPDAC) | Study Details | Resection Details | Synchronous/Metachronous | Morbidity, Grade 3+ | Peri-Procedural Mortality | Chemotherapy, Neoadjuvant/Adjuvant | Median Overall Survival (Months), Hepatic mPDAC Only |
|---|---|---|---|---|---|---|---|---|---|---|
| Yamada [49] | 2006 | R | 6 | 6 Primary + liver res (PM) | NS | 5/1 | NS | NS | NS | NS |
| Gleisner [50] | 2007 | R | 68 | 17 Primary + liver res (PM) 66 Palliative bypass (control) | NS | 88/0 | NS | 9% | N: NS A: 32% | From primary/meta res: -PM: 5.9 -Control (PB): 5.6 |
| Shrikhande [31] | 2007 | R | 10 | 29 Primary res ± ILN res or liver res or multi-organ res (PM) | R0: 86% R1: 14% | 29/0 | NS | 0% | N: 3% A: 69% | From primary/meta res: -PM: 11.4 -Control (SE/PB): 5.9 |
| De Jong [51] | 2010 | Pro | 42 | 126 Primary res + LDT (incl. liver res, ablation, embolization) (PM) | NS | 15/28 | Sc: 12.4% Mc: 20.3% | Sc: 2% Mc: 3% | NS | From primary/meta diagnosis or resection (unknown): -PM: 13.6 |
| Dünschede [52] | 2010 | Pro | 23 | 13 Primary + liver res (PM) 10 Chemotherapy (control) | R0 pancreas: 100% R0 liver: Sc: 89% Mc: 100% | 14/9 | NS | Sc: 0% Mc: 0% | N: NS A: -PM: 50% -Control: 100% | Sc, from primary/meta diagnosis: -PM: 8 -Control (chemo): 11 Mc, from meta diagnosis: -PM: 31 -Control (chemo): 11 |
| Klein [53] | 2012 | R | 22 | 22 Primary + liver res (PM) 22 Primary res only (M0) | Liver: R0: 32% R1: 46% R2: 23% | 22/0 | 18% | 0% | N: NS A: 100% | From primary/meta diagnosis or resection (unknown): -PM: 7.5 |
| Zanini [30] | 2015 | R | 15 | 15 Primary + liver res (PM) | R0: 47% R1: 53% | 11/4 | Sc: 9% Mc: 25% | 0% | N: NS A: 100% | From meta res: -Sc PM: 8.3 -Mc PM: 11.4 |
| Bahra [54] | 2015 | R | 29 | 21 Primary + liver res (PM) 24 Primary res only (control) 45 Chemotherapy (control) | R0M1: 27% R1M1: 27% R2M1: 13% R2M0: 33% | PM: 29/0 Control: NS | 20% | 2% | N: NS A: -PM: 98% -Control: 100% | From primary/meta res: -PM: 10.4 -Control (chemo): 7.2 |
| Tachezy [24] | 2016 | R | 138 | 69 Primary + liver res (PM) 69 Liver res only (control, M) | R0: 58% R1: 32% R2: 10% | 138/0 | PM: 7% SE: 5% | PM: 1% SE: 1% | N: -PM: 14% -Control: 1% A: -PM: 80% -Control: 82% | From primary/meta treatment: -PM: 14.5 -Control (M): 7.5 |
| Hackert [55] | 2016 | Pro | 85 | 85 Primary + liver res (PM) 43 Primary + ILN res | Liver: R0: 19% R1: 60% Rx: 21% | Liver: 62/23 | NS | Sc: 3% Mc: 4% | N: 16% A: 75% | From meta res: -Sc + Mc PM: 12.3 |
| Crippa [28] | 2016 | R | 127 | 11 Primary ± liver res (PM) 116 Chemotherapy (control) | R0: 82% R1: 18% | 3/8 | NS | PM: 0% Control: NS | N: 100% A: -PM: 82% | From primary diagnosis: -Sc + Mc PM: 39 -Control (chemo): 11 |
| Wright [56] | 2016 | R | 16 | 23 Primary ± liver/lung res (PM) | R0: 91% R1: 9% | 23/0 | 13% | 0% | N: 100% A: NS | NS |
| Kim [29] | 2016 | R | 45 | 35 Primary res ± metastasectomy (PM) 35 No res (matched controls) | NS | 70/0 | PM: 20% Control: 6% | 0% | N: NS A: -PM: 83% -Control: 57% | NS |
| Andreou [25] | 2018 | R | 76 | 76 Primary + liver res (PM) | R0: 82% R1: 18% | 76/0 | 16% | 5% | N: 5% A: 72% | NS |
| Kandel [57] | 2018 | R | 18 | 6 M1PDAC: Primary res + meta res/RFA/embolization (PM) 18 M1PDAC: No res, chemo | M1R0: 83% M1R1: 17% | NS | NS | NS | N: -PM: 100% -Control: 44% A: 100% all groups | NS |
| Yang [26] | 2020 | R | 89 | 48 Primary + liver res (PM) 10 Surgical exploration, chemo (control) 31 No res, chemo (control) | R0: 100% | 89/0 | NS | PM: 4% | N: 27% A: -PM: 79% -Control (SE): 100% -Control: 100% | From primary/meta res: -PM: 7.8 -Control (SE, chemo): 4.3 -Control (chemo): 7.6 |
| Gu [27] | 2020 | R | 73 | 36 Primary + meta res (PM) 60 Surgical exploration (control) 54 Palliative bypass (control) | R0: 94% R1: 6% | 150/0 | PM: 3% SE: 0% PB: 2% | PM: 0% SE: 3% PB: 4% | N: 0% A: -PM: 19% -Control (SE): NS -Control (PB): NS | NS |
| Schwarz [58] | 2020 | R | 33 | 25 Primary + liver res (PM) 8 Primary res + chemo (matched controls) | Liver: R0: 96% R1: 4% | 0/33 | 12% | 0% | N: NS A: -PM: 88% -Control: 100% | From meta diagnosis: -PM: 36.8 -Controls (P + chemo): 9.2 |
| Authors | Year | Design | Nr. of Patients (Pulmonary mPDAC) | Study Details | Synchronous/Metachronous | Morbidity, Grade 3+ | Peri-Procedural Mortality | Chemotherapy *, Neoadjuvant/Adjuvant | Median Overall Survival (Months), Pulmonary mPDAC Only |
|---|---|---|---|---|---|---|---|---|---|
| Arnaoutakis [59] | 2011 | R | 31 | 9 Primary res + CRT + lung res (PM) 22 Primary res + CRT (control) | 0/31 | NS | Lung res: 0% Control: NS | N: 100% A: NS | From primary res: -PM: 51 -Control (P + chemo): 23 |
| Thomas [36] | 2012 | R | 7 | 14 Primary + meta res/RFA (7 lung, PM) 405 Primary res only (incl. lung, other sites) | 0/7 | NS | 0% | N: 76% A: 29% | From primary res: -PM: 92.3 |
| Downs-Canner [60] | 2015 | R | 58 | Data available on 41 patients: 8 Primary res/SBRT + lung res (PM) 23 Primary res + chemo (control) 10 Primary res + BSC (control) | 0/58 | NS | NS | N: 88% A: 50% | From primary diagnosis: -PM: 67.5 -Control (P + chemo): 33.8 -Control (P + BSC): 29.9 From meta diagnosis: -PM: 27 -Control (P + chemo): 18.9 -Control (P + BSC): 11.5 |
| Robinson [33] | 2016 | R | 16 | 15 Primary res + lung res (PM) 1 Primary SBRT + lung res (PM) | 1/15 | NS | 0% | N: 88% A: 56% | From primary res: -PM: 52 From meta res: -PM: 28 |
| Kruger [35] | 2016 | R | 40 | 13 S Primary res + lung res (PM) 22 M Primary res + lung res (PM) 5 M CRT primary + lung res (LAPC, M) | 13/27 | NS | NS | N: 71% A: NS | From meta diagnosis: -Sc PM: 22.8 -Mc PM: 31.3 -Mc M: 10.7 |
| Nakajima [61] | 2017 | R | 16 | 16 Primary res + lung res (PM) | 0/16 | NS | 0% | N: 59% A: 71% | From primary res: -PM: 92 From meta res: -PM: 37 |
| Okui [62] | 2017 | R | 6 | 6 Primary + lung res (PM) | 0/6 | NS | NS | N: 100% A: NS | Median follow-up, since all patients were alive From primary res: -PM: 81.7 From meta res: -PM: 37.3 |
| Yasukawa [63] | 2017 | R | 12 | 11 Primary res + lung res (PM) 1 CRT + lung res | 0/12 | NS | 0% | N: 100% A: 100% | From primary res: -PM: 121 From meta res: -PM: 47 |
| Ilmer [32] | 2019 | R | 11 | 11 Primary + lung res (PM) | 0/11 | 0% | 0% | N: 91% A: 100% | From primary res: -PM: 37.7 From meta res: -PM: 26 |
| Groot [34] | 2019 | R | 96 | 19 Primary + lung res (PM) 77 Primary res only (controls): -45 CRT -32 BSC | 0/96 | Lung res: 0% | 0% | N: -PM: 5% A: -PM: 53% | From primary res: -PM: 68.9 -Control (P + CRT): 34.2 -Control (P + BSC): 24.5 From meta res: -PM: 35 -Control (P + CRT): 20.2 -Control (P + BSC): 8.1 |
| Kaiho [64] | 2019 | R | 12 | Primary + lung res (PM) | NS | NS | 0% | N: -mPDAC: 75% A: -mPDAC: 75% | NS |
| Shimizu [65] | 2020 | R | 13 | 6 Primary + lung res (PM) 7 Primary res only (control) | 0/13 | NS | 0% | N: -PM: 100% -Control: 71% A: -PM: 50% -Control: 0% | From primary res: -PM: 39 -Control (P + BSC): 33 |
| Authors | Year | Design | Nr. of Patients (mPDAC) | Study Details | Synchronous/Metachronous | Morbidity, Grade 3+ | Peri-Procedural Mortality | Chemotherapy, Neoadjuvant/Adjuvant | Median Overall Survival (Months), mPDAC Only |
|---|---|---|---|---|---|---|---|---|---|
| RFA | |||||||||
| Park [40] | 2012 | R | 34 liver | 34 Primary res + liver RFA (PM) | 6/28 | NS | 0% | N: 68% A: 62% | From primary res: -PM: 18 From meta RFA: -PM: 14 |
| Hua [39] | 2017 | R | 102 liver | 102 no primary res (unresectable) + liver RFA (M) | 102/0 | 0% | 0% | N/A: 100% Unclear whether prior to or after liver RFA | From primary/meta diagnosis: -M: 11.4 |
| Lee [38] | 2020 | R | 126 liver | 60 Primary res + liver RFA (PM) 66 Primary res + chemo for meta (control, P) | 0/126 | 13% | 0% | N: 80% A: NS | From meta RFA: -PM: 12 -Control (P + chemo): 9.1 |
| IRE | |||||||||
| Hong [67] | 2018 | R | 7 liver, peritoneum, omentum | Primary res + meta IRE (PM) or Primary IRE + metastasectomy (PM) | 7/0 | NS | 0% | N: 100% A: 57% | From initial local treatment: -PM: 16 |
| Authors | Year | Design | Nr. of Patients (mPDAC) | Study Details | Synchronous/Metachronous | Morbidity, acute/late, Grade 3+ | Peri-Procedural Mortality | Chemotherapy, Neoadjuvant/Adjuvant | Median Overall Survival (Months), mPDAC Only |
| SBRT | |||||||||
| Chang [68] | 2009 | R | 15 Metastatic sites not specified | 15 primary SBRT only (P) | 15/0 | Ac: 1% | 0% | Prior: 19% Concurrent: 77% | From primary SBRT: -P: 10.5 |
| Su [69] | 2015 | R | 16 Metastatic sites not specified | 16 Primary SBRT only (P) | 16/0 | Ac: 0% L: NS | 0% | N: 8% A: 8% | From primary SBRT: -P: 8.5 |
| Gkika [70] | 2017 | R | 14 liver, LN | -5 Primary res + primary SBRT (P) -9 Primary res + meta SBRT (PM) -2 Primary SBRT + meta SBRT (PM) -2 Primary SBRT (P) | Sc + Mc Numbers not specified | Ac: 6% L: 6% | 0% | N: NS A: 78% | NS |
| Lischalk [41] | 2018 | R | 20 Metastatic sites not specified | 20 Primary SBRT only (P) | 20/0 | Ac: NS L: 0% | 0% | N: 60% A: 100% | From primary SBRT: -P: 13.6 |
| Scorsetti [42] | 2020 | R | 41 liver, lung, LN | 33 Primary res (± CRT) + meta SBRT (PM) 8 Meta SBRT only (M) | 2/39 | NS | NS | N: 83% A: 22% | From SBRT: -M ± P: 23 |
| HIFU | |||||||||
| Li [37] | 2016 | R | 120 liver, lung, LN | 61 HIFU meta + chemo ± primary res (M ± P) 59 Chemo ± primary res (control, ± P) | NS | 0% | 0% | Concomitant: 100% | From meta HIFU/chemo: -M ± P: 10.3 -Control (chemo ± P): 6.6 |
| Authors | Year | Design | Nr. of Patients (mPDAC) | Study Details | Synchronous/Metachronous | Morbidity, Grade 3+ | Peri-Procedural Mortality | Chemotherapy, Neoadjuvant/Adjuvant | Median Overall Survival (Months), mPDAC Only |
|---|---|---|---|---|---|---|---|---|---|
| SIRT/TARE | |||||||||
| Cao [71] | 2010 | Pro | 7 liver (±EHD) | 3 Primary res + liver SIRT (PM) 4 liver SIRT only (M) | 6/1 | Ac: 0% L: 0% | 0% | N: 100% A: NS | NS |
| Michl [45] | 2014 | R | 19 liver (±EHD) | 15 Primary res + liver SIRT (PM) 4 liver SIRT only (M) | 9/10 | Ac: 9% L: 43–64% | 16% (likely TARE related) | N: 84% A: 47% | From meta SIRT: -M ± P: 9 |
| Gibbs [72] | 2015 | Pro | 14 liver (±EHD) | 4 Primary res + liver SIRT (PM) 10 liver SIRT only (M) | Sc + Mc Numbers not specified | Ac: 36% L: 50% | 14% | Concomitant: 100% | From enrolment/SIRT: -PM: 13.6 -M: 4.2 |
| Kim [73] | 2016 | R | 16 liver (±EHD) | 6 Primary res/SBRT + liver SIRT (PM) 10 liver SIRT only (M) | NS | 6% | 0% | Concomitant: 94% | From meta diagnosis: -M ± P: 22 From meta SIRT: -M ± P: 12.5 |
| Kim [46] | 2019 | R | 33 liver (±EHD) | 23 Primary res/SBRT + liver SIRT (PM) 10 liver SIRT only (M) | NS | Clinical: 15% Lab: 9% | 3% (likely TARE related) | N: 82% A: 30% | From primary diagnosis: -M ± P: 20.8 From meta SIRT: -M ± P: 8.1 |
| Nezami [74] | 2019 | Pro | 3 liver | 3 Primary treatment NS + liver SIRT (M ± P) | NS | Clinical: 38.5% Lab: 38.5% | 0% | Concomitant: 100% | NS |
| Kayaleh [47] | 2020 | R | 26 liver (±EHD) | 8 Primary res + liver SIRT (PM) 18 no primary res + liver SIRT (M) | 13/13 | Clinical: 3 in 77 pts Lab: 9 in 77 pts | 0% | N: 100% A: 73% | From primary diagnosis: -M ± P: 33 From meta diagnosis: -M ± P: 21.8 From meta SIRT: -M ± P: 7 |
| TACE | |||||||||
| Kim [75] | 2010 | R | 15 liver | 15 Primary res + liver TACE (PM) | 0/15 | 13% | 0% | N: 13% A: NS | From meta diagnosis: -PM: 9.6 From meta TACE: -PM: 7.5 |
| Azizi [43] | 2011 | R | 32 liver | 32 Primary res + liver TACE (PM) | NS | 0% | 0% | N: 100% A: NS | From meta TACE: -PM: 16 |
| Kotoyan [76] | 2012 | Pro | 6 liver (± EHD) | 6 Primary NS + liver TACE (M ± P) | NS | 30% | 0% | N: 100% A: NS Concomitant: 100% | From unknown: -M ± P: 9.3 |
| Sun [48] | 2017 | R | 18 liver (± EHD) | 18 liver TACE ± primary res (M ± P) | NS | 0% | 0% | N: 44% A: NS | NS |
| Vogl [77] | 2018 | R | 112 liver | 112 Primary res + liver TACE (PM) | NS | 0% | 0% | N: 100% A: NS | From TACE: -PM: 19 |
| Das [78] | 2019 | R | 182 liver | -84 RFA/ISI + TACE -59 TACE -123 matched control: syst chemo All groups include M1 and M0 disease | NS | RFA/ISI + TACE: 13 in 75 pts TACE only: 16 in 143 pts Controls: 28 in 123 pts | 0% | N: NS A: NS Control: 100% | NS |
| TARE/TACE | |||||||||
| Ouyang [44] | 2018 | R | 184 liver (±EHD) | No primary resection, some pts may have received primary SIRT -64 LDT + systemic chemo (M):
| 184/0 | M: 30% Control (chemo): 18% | TACE: 1 pt | N: 100% A: NS | From primary/meta diagnosis: -M: 8.7 -Control (chemo): 6.3 |
| (Locoregional) Treatments | Hepatic mPDAC mOS from Metastatic Diagnosis/Treatment | Pulmonary mPDAC mOS from Metastatic Diagnosis/Treatment |
|---|---|---|
| P + M | 7.8–19 (Sc + Mc) | 22.8–47 (Sc + Mc) |
| P only | 9.1–9.2 (Mc) | 8.1–20.2 (Mc) |
| M only | 7.5 (Sc) | 10.7 (Mc) |
| CRT/BSC | 4.3–7.6 (Sc + Mc) | 11.8 * (Sc + Mc) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timmer, F.E.F.; Geboers, B.; Nieuwenhuizen, S.; Schouten, E.A.C.; Dijkstra, M.; de Vries, J.J.J.; van den Tol, M.P.; Meijerink, M.R.; Scheffer, H.J. Locoregional Treatment of Metastatic Pancreatic Cancer Utilizing Resection, Ablation and Embolization: A Systematic Review. Cancers 2021, 13, 1608. https://doi.org/10.3390/cancers13071608
Timmer FEF, Geboers B, Nieuwenhuizen S, Schouten EAC, Dijkstra M, de Vries JJJ, van den Tol MP, Meijerink MR, Scheffer HJ. Locoregional Treatment of Metastatic Pancreatic Cancer Utilizing Resection, Ablation and Embolization: A Systematic Review. Cancers. 2021; 13(7):1608. https://doi.org/10.3390/cancers13071608
Chicago/Turabian StyleTimmer, Florentine E. F., Bart Geboers, Sanne Nieuwenhuizen, Evelien A. C. Schouten, Madelon Dijkstra, Jan J. J. de Vries, M. Petrousjka van den Tol, Martijn R. Meijerink, and Hester J. Scheffer. 2021. "Locoregional Treatment of Metastatic Pancreatic Cancer Utilizing Resection, Ablation and Embolization: A Systematic Review" Cancers 13, no. 7: 1608. https://doi.org/10.3390/cancers13071608
APA StyleTimmer, F. E. F., Geboers, B., Nieuwenhuizen, S., Schouten, E. A. C., Dijkstra, M., de Vries, J. J. J., van den Tol, M. P., Meijerink, M. R., & Scheffer, H. J. (2021). Locoregional Treatment of Metastatic Pancreatic Cancer Utilizing Resection, Ablation and Embolization: A Systematic Review. Cancers, 13(7), 1608. https://doi.org/10.3390/cancers13071608







