Abstract
Prunus tenella is a rare and precious relict plant in China. It is an important genetic resource for almond improvement and an indispensable material in ecological protection and landscaping. However, the research into molecular breeding and genetic evolution has been severely restricted due to the lack of genome information. In this investigation, we created a chromosome-level genomic pattern of P. tenella, 231 Mb in length with a contig N50 of 18.1 Mb by Hi-C techniques and high-accuracy PacBio HiFi sequencing. The present assembly predicted 32,088 protein-coding genes, and an examination of the genome assembly indicated that 94.7% among all assembled transcripts were alignable to the genome assembly; most (97.24%) were functionally annotated. By phylogenomic genome comparison, we found that P. tenella is an ancient group that diverged approximately 13.4 million years ago (mya) from 13 additional closely related species and about 6.5 Mya from the cultivated almond. Collinearity analysis revealed that P. tenella is highly syntenic and has high sequence conservation with almond and peach. However, this species also exhibits many presence/absence variants. Moreover, a large inversion at the 7588 kb position of chromosome 5 was observed, which may have a significant association with phenotypic traits. Lastly, population genetic structure analysis in eight different populations indicated a high genetic differentiation among the natural distribution of P. tenella. This high-quality genome assembly provides critical clues and comprehensive information for the systematic evolution, genetic characteristics, and functional gene research of P. tenella. Moreover, it provides a valuable genomic resource for in-depth study in protection, developing, and utilizing P. tenella germplasm resources.
1. Introduction
Prunus tenella is one of the oldest relict plants left from the Tertiary Miocene epoch and is mainly distributed in China and Kazakhstan [1]. In China, P. tenella is an endangered and rare species with economic, scientific, and cultural importance and is scattered only in the northern mountainous areas of Xinjiang province [2]. It is considered to be in danger of extinction due to natural and human-caused disturbances, and as a result, is on the list of nationally significant protected wild plants (Figure 1).

Figure 1.
Morphological characteristics of the P. tenella: (A) flower; (B) fruit; and (C) seed.
As a relatively wild species of cultivated almond, P. tenella has adapted to extremely harsh environments, displaying impressive cold and drought tolerance [3]. For example, it can grow normally in the hills and mountains in Tacheng and Altay at extremely low temperatures reaching −35 °C. Additionally, P. tenella possesses exceptional agronomic traits and unique genomic characteristics, making it highly valuable for diverse applications in various fields, such as food processing and medicine [2]. Additionally, these characteristics provide precious genetic materials for studying the adaptive evolution of the P. tenella genome and allow for the genetic improvement of the cultivated almond, thus improving the resistance of cultivated almond to biotic and abiotic stresses and improving the yield.
With the increased attention to wild resources, many desirable traits have been mined and applied to agricultural varieties [4,5,6,7,8]. Moreover, many breakthrough varieties have also benefited from the discovery of wild-breeding genetic resources [9,10,11,12], especially for wheat, rice, soybean, and other economically important food crops [13,14,15,16]. Together, these lines of evidence have shown that introducing wild gene resources has greatly improved disease resistance, insect resistance, and the growth of cultivated varieties [17,18,19,20,21].
In P. tenella, some genes such as self-incompatibility-related genes SBPI, Petcullin1, SFB, SSK1, S-RNase, and the cold resistance-related gene AlsCBF1-A were identified by homologous cloning technology [22,23,24]. Population diversity and structure were studied using molecular markers, including chloroplast DNA (cp DNA), ISSR, and SSR markers. There was a significant genetic structure among the wild almond populations, and the genetic variation mainly came from among the populations. Some regions with high genetic diversity were also found. These studies provide a basis for the distribution, evolution and conservation of wild almond populations [25,26]. However, thorough knowledge of the economically significant genetic features has been severely hampered by a paucity of genomic resources. More research into the evolutionary adaptations and the development process of distinct features is required before the genetic characteristics of P. tenella can be properly analyzed.
Obtaining high-quality reference genome sequences is the key to revealing allelic variation, genetic relationship, and evolutionary history [27,28,29,30,31]. The present paper describes a highly accurate chromosome-scale standard genome of P. tenella assembled from scratch using the Hi-C technique and lengthy PacBio SMRT reads. Additionally, the genetic variation and geographical differentiation of 130 individual plants from eight wild natural populations were assessed using genome-wide high-resolution molecular markers, which provided an important basis for advancing our understanding of origin, formation, and geographical distribution profile of P. tenella.
2. Results
2.1. De Novo Assembly of the P. tenella Genome
After quality assessment (GC distribution statistics, quality value Q20, Q30 assessment) and filtering, 29.55 Gb clean data were obtained for genome size assessment and assembly. The sequencing depth totaled approximately 137.00×, with a GC content of approximately 38.15%. Additionally, the proportion of Q20 was greater than 97.19%, and the proportion of Q30 was greater than 91.81%. Based on k-mer distribution analysis, the sample genome size was estimated at around 215.65 Mb, consisting of 35.76% repeat sequences and 0.47% heterozygosity. A k-mer distribution of k = 21 was observed (Supplementary Figure S1).
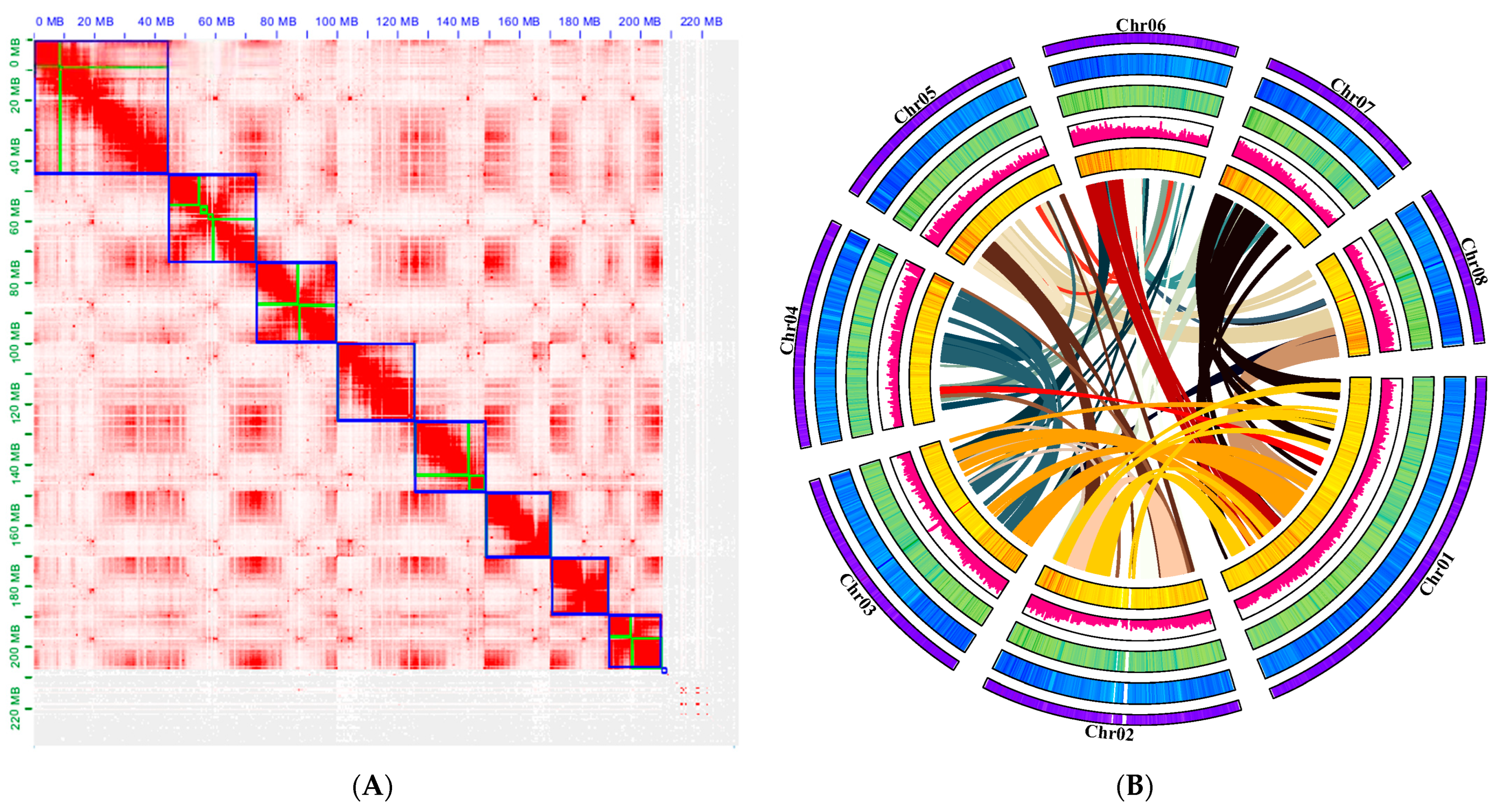
Using PacBio SMRT single-molecule sequencing, 34.6 GB high-accuracy HiFi reads were obtained for de novo genome assembly. The average length of ccs produced was more than 1765 bp, and the longest was 34,337 bp. After assembly and deduplication, a contig level assembly of 231 M sequences was obtained, a contig-level assembly of 231 M sequences was obtained, comprising 3073 contigs (longest = 35.9 Mb) with a 18.1 Mb contig N50 (Table 1). After sequencing quality control, 36.05 Gb Hi-C Fastq clean data were obtained by 3D de novo assembly with the proportion of Q30 was more than 93.10% and the effect rate of 32.5%. Finally, 231 Mb of sequences were anchored onto eight pseudochromosomes (Figure 2), covering 479 scaffolds (longest = 44.8 Mb, scaffold N50 = 25.6 Mb) (Table 1). A 89.7% mounting ration was estimated (Table 2). The BUSCO results showed that more than 2009 (94.7%) genes could be compared to the lineal homologous database, among which 62.5% were single-copy and 32.7% were duplicates (Table 3).

Table 1.
Statistical findings of the assembled P. tenella genome.
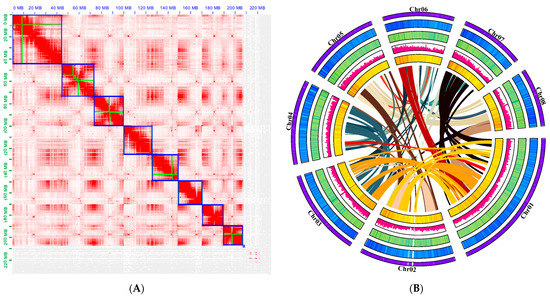
Figure 2.
Hi–C assisted assembly of P. tenella genome pseudomolecules: (A) High-resolution (100 kb) Hi-C interaction heat map among eight chromosomes. The regions in the blue box represent chromosomes and the green within the box represents contig; (B) Chromosome characteristics of the P. tenella genome. The following items follow the outermost ring: The pseudochromosomal visualization of gene density, GC content, repeat content, SNP density, and gene collinearity from a genome assembly.

Table 2.
Statistical findings of chromosomal level assembly of P. tenella.

Table 3.
Findings of P. tenella genome integrality assessment by BUSCO.
2.2. Functional Annonations, Gene Prediction, and Repetitive Sequences
To further describe the P. tenella genome, we categorized all sequences that recur using a combination of de novo and homology-based methods. We estimated that transposable elements made up 28.97% of the genome, with TIRs making up to 7.56% and non-TIRs making up to 3.04%. Only LTR retrotransposons were found, accounting for 18.37%, while non-LTR retrotransposons were not detected (Table 4). The present assembly projected a total of 32,088 protein-coding genes, among which 97.24% had at least one public database functional annotation (Table 5).

Table 4.
Statistical findings of repeated sequences TE annotations in P. tenella genome.

Table 5.
Protein-coding genes-related functional annotations in P. tenella genome.
2.3. Synteny Analysis, and Genome Evolution and Phylogeny
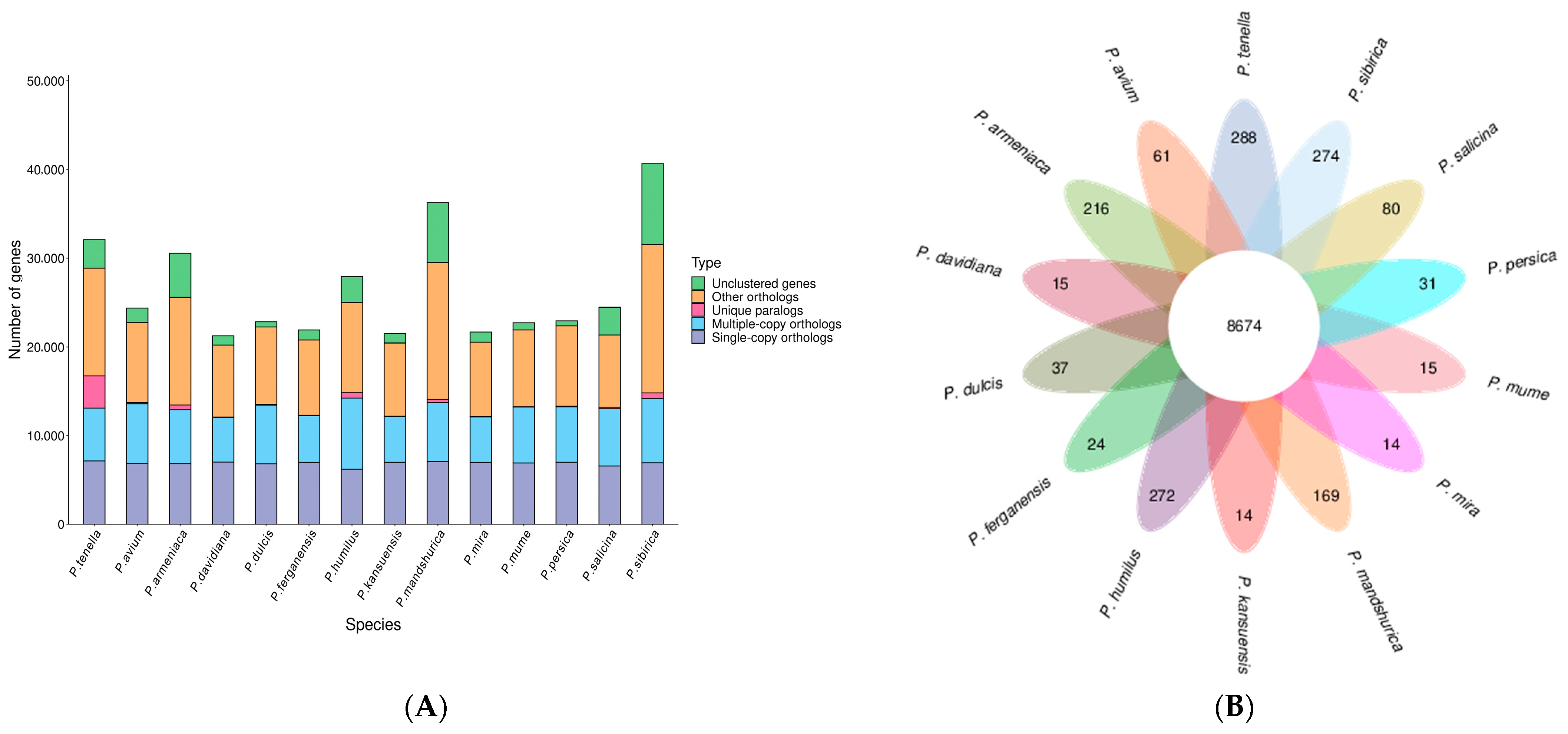
We utilized 10,184 gene families from related species, including, P. avium, P. armeniaca, P. kansuensis, P. ferganensis, P. dulcis, P. davidiana, P. humilus, P. mandshurica, P. mira, P. mume, P. persica, P. salicina, and P. sibirica. Among them, 8674 were common to all species, while 288 were unique to P. tenella (Figure 3).
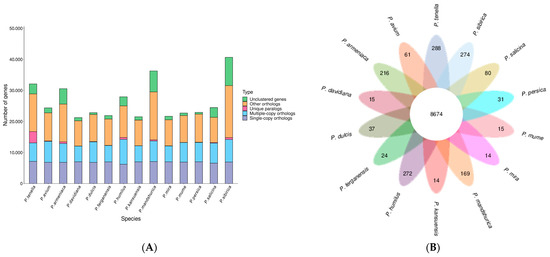
Figure 3.
Number of homologous genes in the species and gene family clustering: (A) Homologous gene statistics; (B) Gene family clustering petal map.
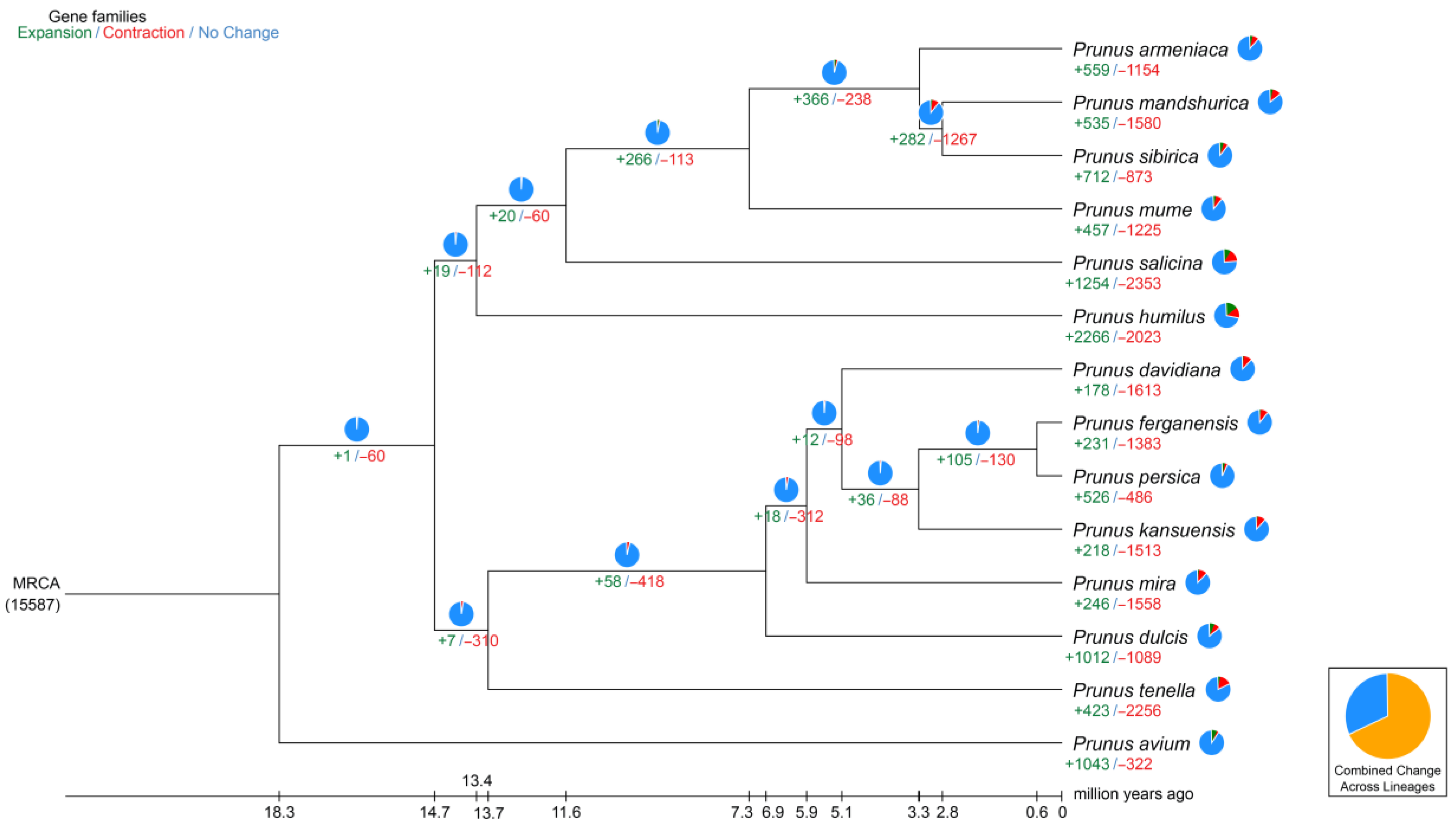
The period of divergence was then approximated by comparing the protein sequences from every single-copy gene families common to those species, and the resulting phylogenetic trees were created. Bootstrap values greater than 90% provided strong evidence for the association between the species (Figure 4). Indeed, all seven species of the peach genus are on the same branch; among them, P. tenella is an ancient group that diverged approximately 13.4 million years ago (Mya). Furthermore, P. tenella almost diverged about 6.5 Mya from the cultivated almond. A total of 2679 genes involved in expansion and contraction were identified by gene family analysis, among which 1581 genes in 105 gene families were significantly expanded, and 26 genes in 23 gene families were significantly contracted. Together, these data indicate that the P. tenella gene family has undergone a significant contraction (2256 genes) relative to other species (486–1613 genes) in the peach genus, which may be related to natural selection.
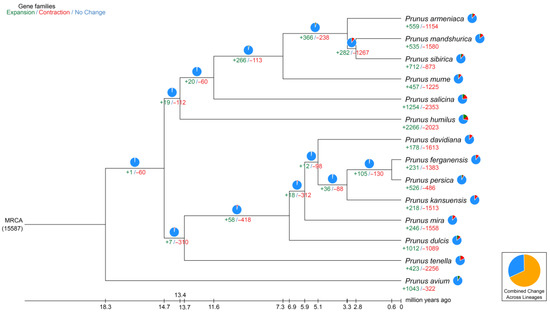
Figure 4.
Divergence time and phylogenetic correlation among species. The percentages of conserved (blue), contracted (red), and expanded (green) gene families among all gene families in the 14 species is shown as a pie chart.
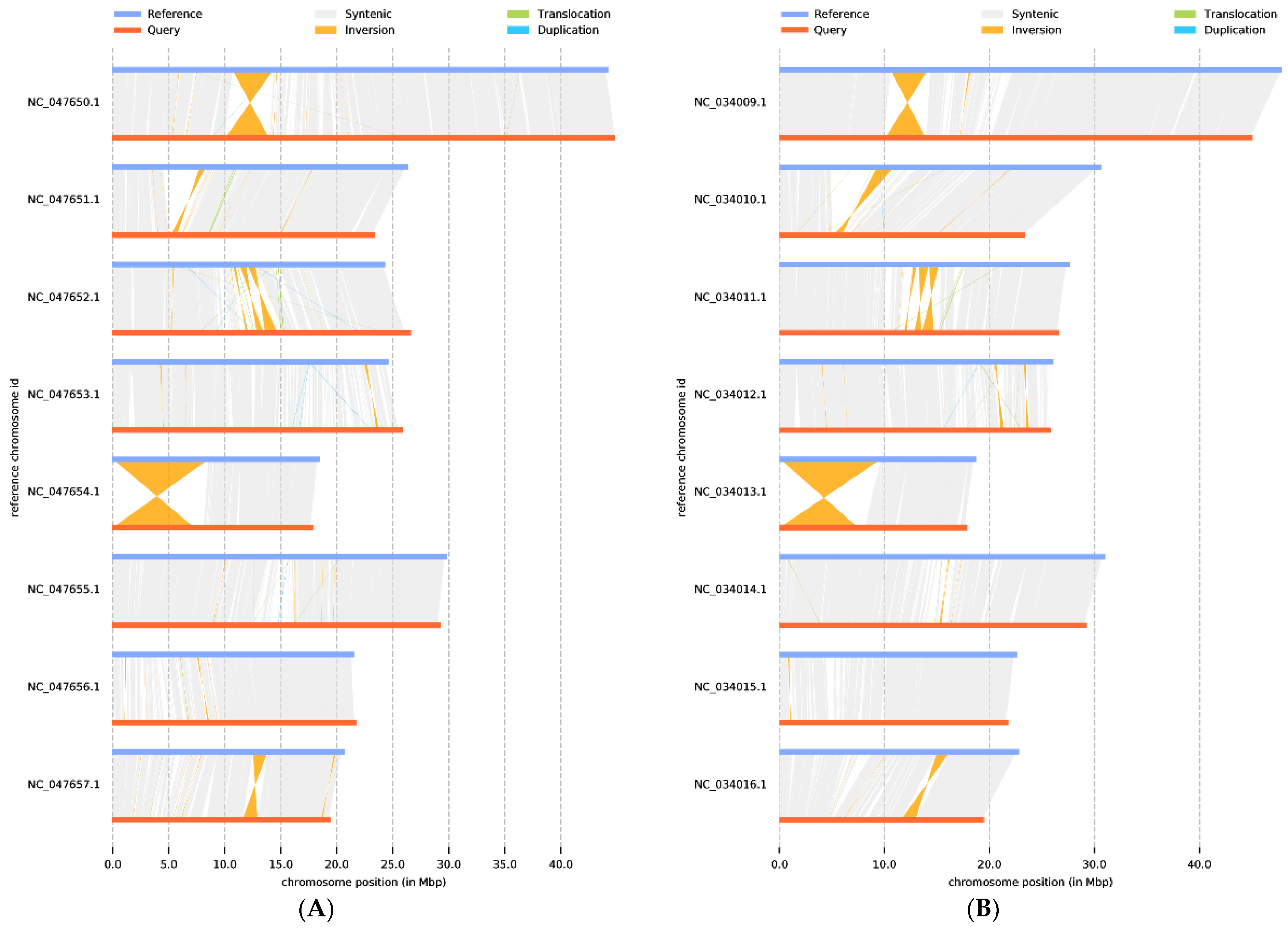
Next, synteny analysis was performed to further understand genes’ position relationship on homologous chromosomes and the variation of genome structure. Coding gene and genome-wide collinearity analysis showed a highly linear relationship between P. tenella, cultivated almond, and peach. Meanwhile, 1,540,264 SNPs, 105,280 deletions, and 155,863 insertions (including absence/presence variations) sequences were identified when compared with almond (Supplementary Table S1). Compared with peach, the 1,574,620 SNP, 106,957 deletions, and 161,747 insertions (including absence/presence variations) sequences were identified (Supplementary Table S2). These structural variations are the main source of genomic variation and may have a significant association with phenotypic traits. In addition, a large inversion (7588 kb) was identified on chromosome 5, which is valuable for further understanding gene regulation and epigenetic inheritance in P. tenella (Figure 5).
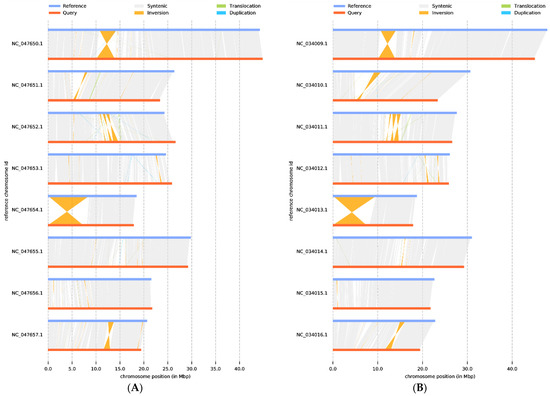
Figure 5.
Genome structure map: (A) P. dulcis vs. P. tenella, (B) P. persica vs. P. tenella. The grey area is the collinear relationship area, yellow is the inversion area, green is the translocation area, and blue is the area where reduplication occurs.
2.4. Population Genetic Structure and Genetic Diversity Analysis
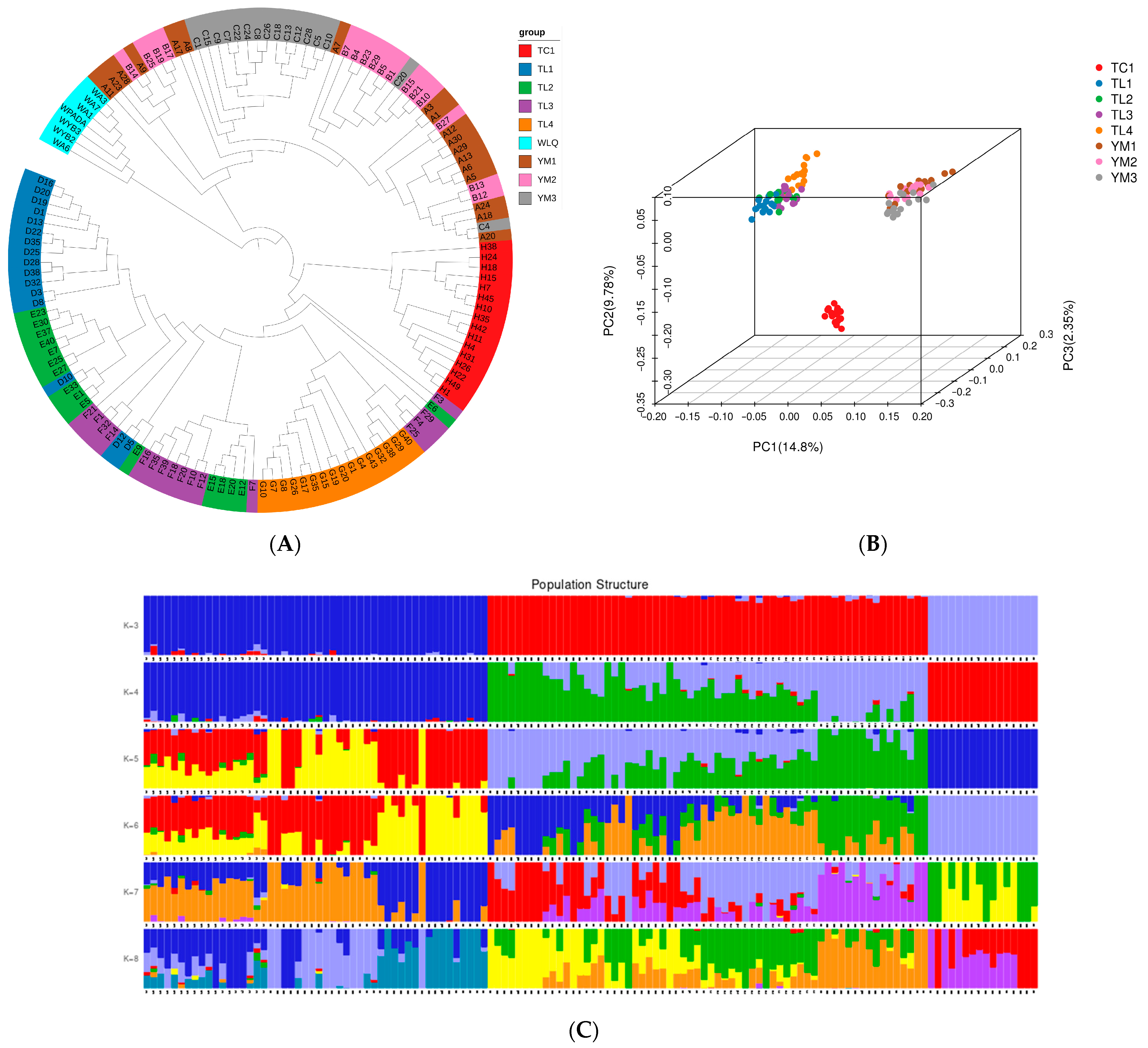
Using the Illumina HiSeq X Ten platform, 512.5 Gb of clean PE150 paired-end data (approximately 15 average sequencing depth) were generated from 130 separate specimens gathered from the native area in Xinjiang Province, revealing the genetic variants in P. tenella across different geographic populations. Based on the genetic distances derived from the genotypes at all the SNP sites of the eight subpopulations in the three areas, a maximum-likelihood and neighbor-joining (Figure 6A) phylogenetic tree was created using the SNPs/genotypes. Some individuals within the subgroups were grouped in other subgroups, but overall, the groupings from the three locations exhibited strong genetic isolation and constituted distinct groups within the phylogenetic tree. The phylogenetic tree’s conclusion was confirmed by principal component analysis. Tacheng, Yuoli, and Yumin samples clustered together in a distinct subgroup of the PCA (Figure 6B).
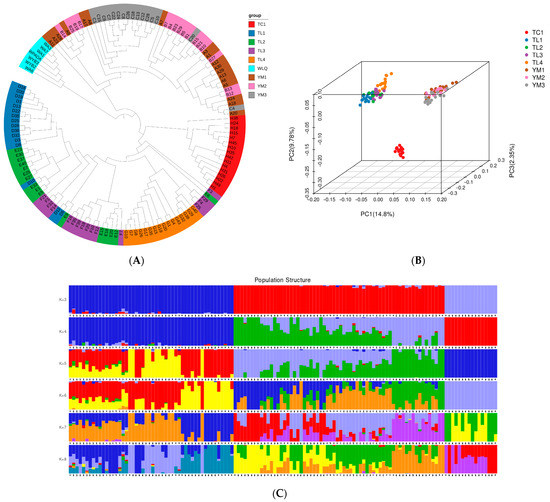
Figure 6.
Phylogeny and population structure of different populations. (A) Phylogenetic tree; (B) PCA three-dimensional cluster diagram of samples; and (C) Admixture sample clustering results corresponding to K values (3–8). The length of the different color segments indicates the proportion of a particular ancestor in the individual’s genome.
Structure analysis results also showed that the 130 samples were mainly from three ancestral populations, consistent with their distribution areas. The samples from the Tacheng are a pure population, while the TuoLi and Yumin samples are hybridized populations with slight levels of admixture (K = 3; Figure 6C). Genetic diversity analysis showed that Expected_heterozygous_number, Observed_allele_number, Nei_diversity_index, and Shnnon_Wiener_index were 0.17–0.29, 1.31–1.49, 0.18–0.30, and 0.25–0.43, respectively (Supplementary Table S3). Compared with other populations, the genetic diversity of the three populations in Yumin was relatively high, while the Tacheng population was relatively low.
2.5. Genome-Wide Selection Signatures’ Analysis of Differentiation
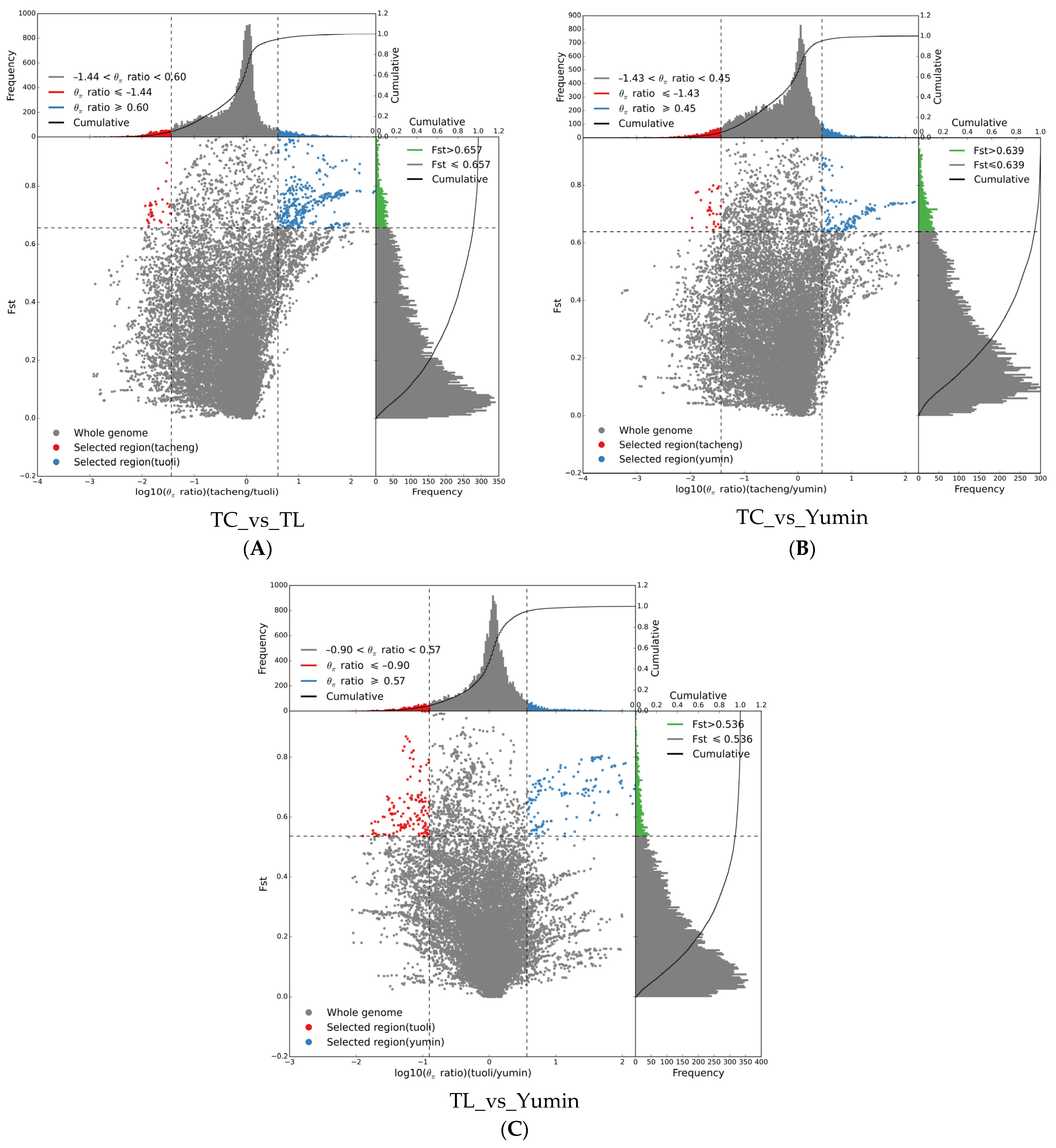
To understand the genetic differentiation among populations, we conducted selective sweep analyses and calculated all pairs of Fst values between eight sampling populations in three different regions. The results showed that the genetic differentiation between the Tacheng and Yumin populations was 0.29–0.32. The genetic differentiation between Tacheng and Tuoli was 0.28–0.3, and the genetic differentiation between Tuoli and Yumin was 0.21–0.27. The inter-population genetic differentiation within the region was low, with the differentiation coefficient falling between 0.05 and 0.1 in the four sample populations of Tuoli and between 0.05 and 0.09 in the three sample populations of Yumin. These results indicate that there had been strong genetic differentiation and geographical variation among the different geographical regions of P. tenella, which may be due to the obstruction of gene flow caused by geographical isolation.
Based on population differentiation and genomic heterozygosity detection, Tacheng, Tuoli and Yumin regions all experienced strong selection(Figure 7). When Fst ≥ 0.657, the differentiation level between Tacheng and Tuoli populations reached a significant level, among which Tacheng had relatively few selection sites (36) and Tuoli had more selection sites (379), indicating that the Tuoli population had a relatively high selection degree. When Fst ≥ 0.639, the differentiation level between the Tacheng and the Yumin population reached a significant level, and compared with Tacheng population (33), the Yumin population was also more selected in loci 43. When Fst ≥ 0.536, the level of differentiation between the Yumin and Tuoli populations reached a significant level, and the selected sites of the two populations were relatively balanced, ranging from 147 to 153. The analysis of selection sites showed that the selection degree of the Tacheng population was relatively small, and the selection mainly occurred in the Tuoli and Yumin populations.
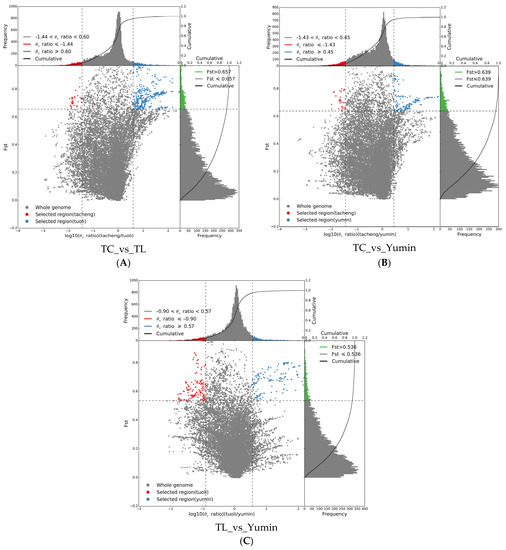
Figure 7.
Schematic of selection signatures between populations: (A) Selective sweep analysis of Tacheng vs. Tuoli; (B) Selective sweep analysis of Tacheng vs. Yumin; and (C) Selective sweep analysis of Tuoli vs. Yumin. The ratio of π in the abscissa and Fst in the ordinate corresponding to the frequency distribution diagram above and the frequency distribution diagram on the right, respectively. The dot plot in the middle represents the corresponding Fst and π ratios in different windows. The blue and red areas at the top are the top 5% of the region selected by π, the green area at the right is the top 5% of the region selected by Fst, and the blue and red areas in the middle are the intersection of Fst and π, which are the candidate sites.
3. Discussion
Through the use of various sequencing methods, we assembled the first complete reference genome for P. tenella in this work. The Prunus genus has several commercially and ecologically significant species in forestry and agriculture, and these data are essential for learning more about P. tenella and the genus as a whole. These findings will also aid in the development of genome-enabled P. tenella breeding initiatives. Last but not least, P. tenella’s status as a relict species makes it a useful model for studying the genetic basis of population formation, evolution, and adaptation to environmental effects under conditions of geographic isolation.
Given the high quality of our P. tenella genome assembly, high-depth PacBio long-read, and whole genome re-sequencing data, we now have a comprehensive understanding of the genome of the P. tenella. Single-copy, multi-copy, and species-specific gene families were obtained, the evolutionary status was inferred, and the genome’s evolutionary history was traced, laying the foundation for further exploration and research. Additionally, we found many variation sites, including SNPs, insertions, deletions, and inversion. Many of these variants may be associated with phenotypic traits, which will help understand the phylogenetic evolution of P. tenella. Moreover, these various sites can be used as important molecular markers for germplasm identification, genetic analysis, functional gene extraction, and assisted breeding. Compared with the genome of P. persica and P. dulcis, we identified a large inversion on chromosome 5, which may be related to the unique characteristics of P. tenella such as dwarfing and freezing resistance. In order to further determine the authenticity of this inversion, we re-checked the assembly process and the contig connection. Since the chromosome we assembled was composed of two contig and there was a break point at about 7588 kb on one side, we divided 500 kb sequences at both ends of the break point and calculated the degree of linkage disequilibrium between the two connection modes. By calculation, the degree of linkage disequilibrium of the current connection mode is 0.09415, which is significantly greater than that of the other connection mode (0.05116), indicating that our present assembly results are reasonable.
Through the analysis, we also found some different characteristics of the P. tenella genome relative to other species of the same genus. Compared with other tree species, P. tenella had relatively more endemic gene families (288), while cultivated almond and peach had relatively few, only 31–37, which may reflect the unique evolutionary characteristics of P. tenella. More gene families were contracted in P. tenella, which might be related to natural selection caused by the extreme natural environment. The evolutionary status inferred by the phylogenetic tree shows that the P. tenella were clustered into a large clade with amygdala subgenus, but compared with other species of amygdala subgenus, P. tenella differentiated earlier, at 13.4 million years ago, while cultivated almond differentiated only 6.9 million years ago, indicating that P. tenella is a relatively old species. But the P. tenella is still the closest relative to cultivated almond. Importantly, this observation indicates that P. tenella has the potential utilization value of providing genetic resources for cultivated almond, and this lays the foundation for further exploration and research.
Population genetic structure can be used to analyze the evolutionary dynamics of a population by describing gene transmission, gene frequency change, and genotype distribution [32,33,34,35]. Based on the SNP information derived from the whole-genome re-sequencing data, thousands of single SNP markers can be used for the fine-scale description of genetic structure [36,37,38]. The results showed that, compared with the Tacheng (Nei’s = 0.18; Ho = 0.16) and Tuoli (Nei’s = 0.23–0.26; Ho = 0.16–0.22) populations, the Yumin variety (Nei’s = 0.26–0.3; Ho = 0.17–0.22) has relatively high genetic diversity. This observation is consistent with the study on genetic diversity using chloroplast sequences [4]. Additionally, the results indicated a high genetic differentiation among the natural distribution of P. tenella, as the pairwise genetic differentiation (Fst) in a different region is 0.23–0.32, especially within the Tacheng and Yumin group where Fst reached 0.29–0.32, values much higher than wright’s high differentiation coefficient [39,40,41]. However, there is little differentiation between subgroups within the Tuoli and Yumin group (0.05–0.1). These results suggest that geographical isolation is an important factor affecting the genetic evolution of P. tenella. This higher differentiation may result from the long-term natural selection without gene flow.
Selective Sweep analysis showed that the Tacheng population received fewer selection sites, while the Tuoli and Yumin populations received stronger selection sites. This indicates that there is a tendency to decrease genetic polymorphism and increasing purity and degree in Yumin and Tuoli populations. Due to the severe geographical isolation of P. tenella, this phenomenon may be related to the small population and inbreeding of the population. Since Tuoli and Yumin are in the same mountain range, and Tacheng is in another mountain range, this conclusion also shows that, under the influence of climatic and geographical conditions, the evolution and selection among the groups are relatively independent, resulting in different evolutionary directions. In summary, we assembled the first chromosome-level genome of P. tenella and assessed the genetic variation and geographical differentiation of eight natural populations, which laid a solid foundation for further research on genetic improvement and formation mechanism of important characters in the future.
4. Materials and Methods
4.1. Utilized Materials
P. tenella sample materials used for genome assembly were obtained from the germplasm conservation nursery of Xinjiang Academy of Forestry Sciences, Xinjiang, China. Fresh leaves were utilized for Hi-C library development, PacBio HiFi sequencing, and Illumina sequencing. To aid in genome assembly and annotation, fruit, leaf, root, and stem tissues were taken for RNA-seq study.
The fresh leaves used for whole genome re-sequencing were collected from Yumin County, Tuoli County, and Tacheng City, Xinjiang, China. A total of 8 P. tenella populations were collected, including 3 from Yumin County, 4 from Tuoli County, and 1 from Tacheng City (Table 6). Approximately 15–18 samples were collected from each population. In addition, 7 cultivated almond samples were collected for population evolution analysis.

Table 6.
Latitude and longitude information of the sample population.
4.2. Genome Sequencing and Transcriptome Sequencing
The experiments were carried out in accordance with Illumina’s recommended methodology. The ultrasonic shock was used to physically fragment the qualifying genomic DNA into fragments (350 bp), and then end restoration, adding A, an adapter, and target fragment picking and PCR were used to generate the tiny fragment sequencing library. By using a bridge PCR, the library was transferred to the sequencing chip. An Illumina sequencer performed double-ended 150 bp (PE 150) library sequencing.
DNA capture and purification, cyclization, end repair, endonuclease digestion, cell cross-linking, and on-machine sequencing were all necessary steps for HI-C sequencing to be completed. The mRNA was utilized to synthesize full-length cDNA with the help of the SMARTerTM PCR cDNA Synthesis Kit, which was then used to generate sequencing libraries. Using the PacBio system, we sequenced the whole transcriptome.
Library sequencing, library quality testing, library creation, and sample quality testing were all carried out as per Illumina’s recommended approach for re-sequencing a variety of population samples. In order to prepare the DNA for sequencing, it was first physically fragmented (using ultrasonic waves), then purified, the ends were mended, the 3’ end was augmented with A, and the sequencing joint was linked. Finally, agarose gel electrophoresis was used to determine the optimal fragment size, and PCR amplification was carried out to form the sequencing library.
Transcriptome sequencing of the stem, root, leaf, and fruit tissues was performed on the NovaSeq 6000 platform.
4.3. Assurance of Sequencing Data Quality
Low-quality sequences and duplicated readings in the sequencing data were removed using stringent filtering algorithms that were optimized for the particular platform utilized to ensure data integrity and accuracy. Filtering criteria included the following actions for Illumina Hi-Seq data: Firstly, polyG tails were removed. Secondly, paired reads of less than 100 bp in length were discarded. Thirdly, read pairs containing more than 10% of bases that are the same as the next base were removed. Fourthly, read pairs with over 50% low-quality bases (quality score less than 10) were discarded. The last step was to clean the data of read pairings with a typical quality rating below 20. The Hi-C sequencing results went through a comparable filtering procedure as Illumina Hi-Seq Data before being processed in 3D. With the default settings of the pbccs pipeline, subreads from the PacBio HiFi long readings were filtered and corrected immediately. Approximately 2000 PacBio HiFi (CCS) reads were randomly selected from the sequencing data and compared with the NT library to evaluate whether the sequencing data contained contamination.
4.4. Heterozygosity and Genome Size Estimation
Heterozygosity and genome size were analyzed before HiFi library construction and sequencing. From the Illumina data, Jellyfish v.2.2.10 [42] examined frequency distributions of quality-filtered short fragments (21-mers). Then, based on Jellyfish’s results, genome escope22 was used for genome analysis. This strategy obtained the genomic information of P. tenella (Supplementary Figure S1), such as heterozygosity, genome size, and proportion of repeat sequences.
4.5. Genome Assembly
Following correction and filtering, HiFi circular consensus sequencing (CCS) reading could be used in the de novo assembly using hifiasm (v0.14-r312) with default parameters. Purge haplotigs was used to remove redundant haploids [43]. In 2017, Dudchenko et al. [44] used the 3D de novo assembly (3D-DNA) software for scaffolding the haploid contigs. The Hi-C readings could be aligned within the draft genome 3D-DNA and Juicebox v1.9.8 was used for the candidate assembly. Assembly Tools (JBAT) [45] was utilized for reviewing the candidate assembly and corrected artificially. The eudicotyledons_odb10 database was employed in conjunction with the BUSCO v3.0.2 (Benchmarking Universal Single-Copy Orthologs) [46] algorithm for assessing genome integrality and gene annotation. A combination of the BWA-MEM method and HISAT2 (v2.1.0) [47] was utilized for mapping the small reads’ filtration obtained by Illumina and the assembled transcripts to the assembly.
4.6. Repetitive Element Annotations
To annotate the TEs or transposable elements [48], the EDTA genome annotation pipeline was utilized. TEs include retrotransposons and DNA transposons. RepeatModeler was used to identify DNA transposons, including long interspersed nuclear elements (LINEs) of the terminal inverted repeats (TIRs) and retrotransposons, and long tandem repeats (LTRs), as well as helitrons found in DNA transposons. To do this, we used Repbase and RepeatMasker (v4.0.7) and Repbase with the optimal settings to generate a de novo repeat library for repeat sequence identification [49,50].
4.7. Functional Annonations and Gene Prediction
The StringTie (v1.3.5) and HISAT2 (v2.1.0) pipeline was used for mapping the RNA-seq data within the fruits, leavers, stems, and roots to the genome. Gene prediction together with de novo transcripts assembly were conducted through Trinity [51]. PASA (v2.4.1) pipeline transdecoder4 was also applied to annotate the transcripts-relevant coding regions [52]. Exonerate v2.2.0 carried out homolog predictions. GlimmerHMM (v3.0.4) and the protein sequences of P. dulcis, P. mira, P. persica, P. armeniaca, P. mume, and P. salicina could also be mapped to the genome [53]. For de novo gene speculation, genes from the PASA results were trained by AUGUSTUS (v3.3.3) and SNAP [54,55]. In order to combine the gene models, EVidenceModeler (v1.1.1) was used [56]. The predicted protein sequences were compared to the EuKaryotic Orthologous Groups (KOG), Nr databases, Pfam, SwissProt, Kyoto Encyclopedia of Genes and Genomes (KEGG), and Gene Ontology (GO) to infer possible functions for the protein-coding genes.
4.8. Phylogenetic and Gene Family Analysis
Thirteen closely related species were selected for phylogenetic and gene family analysis along with P.tenella. Additionally, P.avium was selected as the outgroup. The genome database for Rosaceae “www.rosaceae.org (accessed on 11 April 2023)”, and the NCBI database “www.ncbi.nlm.nih.gov (accessed on 5 January 2023)” was used for obtaining the protein sequences of these species. Alignment quality was ensured by excluding sequences with lengths < 100 bp. OrthoFinder (v2.5.2) were deployed for identifying single-copy homologous genes and classifying families, using the settings “-M msa -S diamond -T raxml-ng” [57]. RaxML [58] have been approached for estimating and evaluating the phylogenetic connection tree of 14 species using 100 bootstrap repetitions. Time to diverge was computed using PAML’s MCMC tree [59]. CAFE (v3.1) was used for examining relevant growth patterns and gene families-related declines, as described by Han et al. [60]. By counting the number of ancestral gene families on each branch of the phylogenetic tree, we were able to determine the rate at which gene family sizes shrank or grew. Cafetutorial_clade_and_size_filter.py was used to filter gene families characterized by very high variations in gene copy numbers in an effort to decrease prediction mistakes. Exact data on the contraction and expansion gene families of 14 species were utilized using the script cafetutorial_report_analysis.py, and these data were then analyzed. For selected gene families, we used Fisher’s exact test to analyze GO functional enrichment.
4.9. Whole-Genome Synteny Analysis
Almond and peach were selected for whole genome replication (WGD) analysis. Four-fold synonymous (degenerative) third-codon transversion (4DTv) values and synonymous mutation distributions for each synonymous site (Ks) were calculated to analyze the genome replication events. The YN substitution model was used to calculate the 4DTv rates based on four-fold degenerate sites. KaKs_Calculator (v2.0) [61] with default parameters was used to calculate Ks values. The minimap2 software “https://lh3.github.io/minimap2/minimap2.html (accessed on 15 November 2022)” was used for genome-wide comparison, and syri software “https://github.com/schneebergerlab/syri (accessed on 12 November 2022)” was used to identify collinear regions between the two genomes, structural rearrangements (inversion, translocation, and duplication), local variations (SNP, indel, and CNV), and unaligned regions. The nucmer (4.0.0beta2) program in MUMmer4 [62] was used to determine whether similar gene pairs on chromosomal were adjacent in different species.
4.10. Single-Nucleotide Polymorphism (SNP) Calling
Trimmomatic v0.36 was used to eliminate adaptors and low-quality sequences during the preprocessing phase. Every sample’s clean reads have been planned using Burrows-Wheeler Aligner to the P. tenella standard genome. Next, Picard “http://broadinstitute.github.io/picard/ (accessed on 12 November 2022)” was employed to identify and align the PCR duplicated sample findings. SNP sites in re-sequencing people from diverse geographical regions were identified using GATK v4 (Genome Analysis Toolkit) for SNP recalling. Each genome’s VCF files were generated using variant calling with GATK Hap-lotypeCaller, and then the VCF files for all 137 genomes were combined to create a single VCF file. Only SNPs that had a Hardy–Weinberg equilibrium < 0.001, minor allele frequency > 0.05, and genotype missing rate of 10% for each were kept for further study, narrowing the analysis down to just biallelic variation sites.
4.11. Phylogenetic Analysis
A phylogenetic tree was generated using the distance matrix produced by MEGA-CC3.5 software (MEGAX) [63] and 1000 bootstrap repetitions to assess the phylogenetic connection of various individuals in order to study the evolutionary links between different populations. In addition, the SMARTPCA application included in the EIGENSOFT software “https://github.com/chrchang/eigensoft (accessed on 16 November 2022)” was utilized to carry out principal component analysis (PCA) and ascertain the subpopulations’ clustering status [64].
4.12. Population Genetic Structure and Genetic Diversity Analysis
In order to learn about the genetic makeup of populations, including their variety, structure, and differentiation, nucleotide diversity was assessed by dividing each population into 10 kb chunks and analyzing a 100 kb window [65]. Using a Bayesian-based strategy, the K-values (the hypothesized number of populations) ranged between 1 and 10 in ADMIXTURE [66]. The optimal K-value was determined using cross-validation statistics across five separate studies. Bar graphs of the Q matrix for each K-value were made with the aid of the R package Pophelper “http://royfrancis.github.io/pophelper (accessed on 25 November 2022)”. The fixation index (FST) and nucleotide diversity ratios (π) were computed using VCF methods to identify genomic areas possibly experiencing natural selection sweeps throughout the adaptation process.
4.13. Selective Sweep Analysis
Genome-wide detection on selective sweep region was processed by calculating the population genetic index of all SNPs within a sliding window of 100 kb and a certain step (10 kb). The indicators include population differentiation fixation index (Fst) and nucleotide polymorphism (π). The index was calculated by the PopGenome package based on consensus SNPs with a pre-defined bin and step.
Supplementary Materials
The supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms241411735/s1.
Author Contributions
G.Z. and H.Z. conceived and coordinated the research. H.Z. and H.H. investigated and collected the samples. Y.Q. and Z.W. assembled and analyzed the data. Y.Q. wrote the manuscript, which has been critically revised by F.L. and G.Z. All authors contributed to the article and approved the submitted version. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the “National Key R&D Program of China (2022YFD2200400)” and the “Key R&D Program of Xinjiang Uygur Autonomous Region (2022292937)”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The whole genome sequence data reported in this paper were deposited in the Genome Warehouse in National Genomics Data Center, Beijing Institute of Genomics, Chinese Academy of Sciences/China National Center for Bioinformation, under accession number GWHCBGA00000000 that is publicly accessible at https://ngdc.cncb.ac.cn/gwh. The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
The authors would like to thank all the reviewers who participated in the review.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Li, J.; Zeng, B.; Luo, S.P.; Li, H.L.; Madaniyati, W. Protection and propagation of Amygdalus ledebouriana Schleche in China. Xinjiang Agric. Sci. 2006, 43, 61–62. [Google Scholar]
- Yin, L.K.; Tan, L.X.; Wang, B. Rare Endangered Endemic Higher Plants in Xinjiang of China; Urumqi Xinjiang Science & Technology Publishing House: Urumqi, China, 2006. [Google Scholar]
- Zhong, H.X.; Lu, C.S.; Luo, S.P.; Li, J. The study of cold resistance test of dormancy branches and buds of Amygdalus ledebouriana Schleche in Xinjiang. Xinjiang Agric. Sci. 2016, 53, 120–125. [Google Scholar]
- Perazzolli, M.; Malacarne, G.; Baldo, A.; Righetti, L.; Bailey, A.; Fontana, P.; Velasco, R.; Malnoy, M. Characterization of resistance gene analogues (RGAs) in apple (Malus × domestica Borkh.) and their evolutionary history of the Rosaceae family. PLoS ONE 2014, 9, e83844. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, B.; Elias, M.; Azimov, R.; Turdieva, M.; Aaliev, S.; Bobokalonov, F.; Butkov, E.; Kaparova, E.; Mukhsimov, N.; Shamuradova, S.; et al. Home gardens of Central Asia: Reservoirs of diversity of fruit and nut tree species. PLoS ONE 2022, 17, e0271398. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Singh, C.; Ambika; Chandana, B.S.; Mahto, R.K.; Patial, R.; Gupta, A.; Gahlaut, V.; Gayacharan; Hamwieh, A.; et al. Exploring Chickpea Germplasm Diversity for Broadening the Genetic Base Utilizing Genomic Resourses. Front. Genet. 2022, 13, 905771. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Jacob, S.R.; Mir, R.R.; Vikas, V.K.; Kulwal, P.; Chandra, T.; Kaur, S.; Kumar, U.; Kumar, S.; Sharma, S.; et al. Indian Wheat Genomics Initiative for Harnessing the Potential of Wheat Germplasm Resources for Breeding Disease-Resistant, Nutrient-Dense, and Climate-Resilient Cultivars. Front. Genet. 2022, 13, 834366. [Google Scholar] [CrossRef]
- Kefale, H.; Wang, L. Discovering favorable genes, QTLs, and genotypes as a genetic resource for sesame (Sesamum indicum L.) improvement. Front. Genet. 2022, 13, 1002182. [Google Scholar] [CrossRef]
- García-Gómez, B.E.; Salazar, J.A.; Nicolás-Almansa, M.; Razi, M.; Rubio, M.; Ruiz, D.; Martínez-Gómez, P. Molecular Bases of Fruit Quality in Prunus Species: An Integrated Genomic, Transcriptomic, and Metabolic Review with a Breeding Perspective. Int. J. Mol. Sci. 2020, 22, 333. [Google Scholar] [CrossRef]
- Filip, E.; Woronko, K.; Stępień, E.; Czarniecka, N. An Overview of Factors Affecting the Functional Quality of Common Wheat (Triticum aestivum L.). Int. J. Mol. Sci. 2023, 24, 7524. [Google Scholar] [CrossRef]
- Li, Z.; Xue, Y.; Zhou, H.; Li, Y.; Usman, B.; Jiao, X.; Wang, X.; Liu, F.; Qin, B.; Li, R.; et al. High-resolution mapping and breeding application of a novel brown planthopper resistance gene derived from wild rice (Oryza. rufipogon Griff). Rice 2019, 12, 41. [Google Scholar] [CrossRef]
- Mamidi, S.; Healey, A.; Huang, P.; Grimwood, J.; Jenkins, J.; Barry, K.; Sreedasyam, A.; Lovell, J.T.; Feldman, M.; Wu, J.; et al. A genome resource for green millet Setaria viridis enables discovery of agronomically valuable loci. Nat. Biotechnol. 2020, 38, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Prahalada, G.D.; Shivakumar, N.; Lohithaswa, H.C.; Sidde Gowda, D.K.; Ramkumar, G.; Kim, S.R.; Ramachandra, C.; Hittalmani, S.; Mohapatra, T.; Jena, K.K. Identification and fine mapping of a new gene, BPH31 conferring resistance to brown planthopper biotype 4 of India to improve rice, Oryza sativa L. Rice 2017, 10, 41. [Google Scholar] [CrossRef] [PubMed]
- Laugerotte, J.; Baumann, U.; Sourdille, P. Genetic control of compatibility in crosses between wheat and its wild or cultivated relatives. Plant Biotechnol. J. 2022, 20, 812–832. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Schulthess, A.W.; Bassi, F.M.; Badaeva, E.D.; Neumann, K.; Graner, A.; Özkan, H.; Werner, P.; Knüpffer, H.; Kilian, B. Introducing Beneficial Alleles from Plant Genetic Resources into the Wheat Germplasm. Biology 2021, 10, 982. [Google Scholar] [CrossRef]
- Aleem, M.; Aleem, S.; Sharif, I.; Aleem, M.; Shahzad, R.; Khan, M.I.; Batool, A.; Sarwar, G.; Farooq, J.; Iqbal, A.; et al. Whole-Genome Identification of APX and CAT Gene Families in Cultivated and Wild Soybeans and Their Regulatory Function in Plant Development and Stress Response. Antioxidants 2022, 11, 1626. [Google Scholar] [CrossRef]
- Mk, A.; Shw, B.; Ss, A. Wheat wild germplasm: A hidden treasure. Wild Germplasm Genet. Improv. Crop Plants 2011, 2021, 55–63. [Google Scholar]
- Yumurtaci, A. Utilization of wild relatives of wheat, barley, maize and oat in developing abiotic and biotic stress tolerant new varieties. Emir. J. Food Agric. 2015, 27, 1. [Google Scholar] [CrossRef]
- Haus, M.J.; Pierz, L.D.; Jacobs, J.L.; Wiersma, A.T.; Cichy, K.E. Preliminary evaluation of wild bean (Phaseolus spp.) germplasm for resistance to Fusarium cuneirostrum and Fusarium oxysporum. Crop Sci. 2021, 61, 3264–3274. [Google Scholar] [CrossRef]
- Rostad, H.E.; Reen, R.A.; Mumford, M.H.; Zwart, R.; Thompson, J.P. Resistance to root-lesion nematode Pratylenchus neglectus identified in a new collection of two wild chickpea species (Cicer reticulatum and C. echinospermum) from turkey. Plant Pathol. 2022, 5, 71. [Google Scholar] [CrossRef]
- Jeff, E.; Olumide, S.T.; Bruce, D.; Andre, H.; Julianne, A.; Olufemi, A. Resistance in wild macadamia germplasm to Phytophthora cinnamomi and Phytophthora multivora. Ann. Appl. Biol. 2021, 178, 519–526. [Google Scholar] [CrossRef]
- Wang, B.; Yu, Z.F.; Zeng, B.; Xia, J.H.; Ma, X.X. Self-incompatibility Gene Cullin1 Cloning and Bioinformatics Analysis of Wild Almond in Xinjiang. Chin. Agric. Sci. Bull. 2017, 33, 63–68. [Google Scholar]
- Zeng, B.; Liu, N.N.; Xia, J.H.; Liu, M.W.; Wang, J.Y.; Wang, B. Molecular Cloning and Bioinformatics Analysis of SFB Genes Controlling Self-incompatibility in Xinjiang Wild Almond (Prunus tenella Batsch.). Chin. Agric. Sci. Bull. 2017, 33, 22–30. [Google Scholar]
- Yu, Z.F.; Wang, B.; Zeng, B.; Wang, J.Y. Cloning and sequence analysis of self—Incompatibility gene SBPI of wild almond in Xinjiang. Mol. Plant Breed. 2018, 16, 6955–6960. [Google Scholar]
- Zeng, B.; Li, J.; Luo, S.P.; Cheng, Y.J. Identification of Genetic Relationship of Amygdalus Plants by SSR. Xinjiang Agric. Sci. 2009, 46, 18–22. [Google Scholar]
- Lu, Z.J.; Li, J.; Omir, S.T.; Zeng, B.; Luo, S.P. ISSR analysis for genetic diversity of Amygdalus ledebouriana germplasm from Xinjiang, China. J. Fruit Sci. 2010, 27, 918–923. [Google Scholar]
- Chen, D.X.; Pan, Y.; Wang, Y.; Cui, Y.Z.; Li, L.Y. The chromosome-level reference genome of coptis chinensis provides insights into genomic evolution and berberine biosynthesis. Hortic. Res. 2021, 8, 11. [Google Scholar] [CrossRef]
- Rush, D.W.; Epstein, E. Breeding and selection for salt tolerance by the incorporation of wild germplasm into a domestic tomato. J. Am. Soc. Hortic. Sci. 1981, 106, 699–704. [Google Scholar] [CrossRef]
- D’Amico-Willman, K.M.; Ouma, W.Z.; Meulia, T.; Sideli, G.M.; Gradziel, T.M.; Fresnedo-Ramírez, J. Whole-genome sequence and methylome profiling of the almond (Prunus dulcis [mill.] d.a.webb) cultivar ‘nonpareil’. G3 Genes Genomes Genet. 2022, 12, jkac065. [Google Scholar] [CrossRef]
- Liu, J.F.; Wei, H.; Zhang, X.; Wang, D. Chromosome-level genome assembly and hazelomics database construction provides insights into unsaturated fatty acid synthesis and cold resistance in hazelnut (Corylus heterophylla). Front. Plant Sci. 2021, 12, 766548. [Google Scholar] [CrossRef]
- Verde, I.; Abbott, A.G.; Scalabrin, S.; Jung, S.; Rokhsar, D.S. The high-quality draft genome of peach (Prunus persica) identifies unique patterns of genetic diversity, domestication and genome evolution-Supplementary Information. Nat. Genet. 2014, 45, 486–495. [Google Scholar]
- Suguiyama, V.F.; Vasconcelos, L.A.B.; Rossi, M.M.; Biondo, C.; de Setta, N. The population genetic structure approach adds new insights into the evolution of plant LTR retrotransposon lineages. PLoS ONE 2019, 14, e0214542. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H. Decades-long phylogeographic issues: Complex historical processes and ecological factors on genetic structure of alpine plants in the Japanese Archipelago. J. Plant Res. 2022, 135, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Mori, G.M.; Zucchi, M.I.; Souza, A.P. Multiple-geographic-scale genetic structure of two mangrove tree species: The roles of mating system, hybridization, limited dispersal and extrinsic factors. PLoS ONE 2015, 10, e0118710. [Google Scholar] [CrossRef]
- Nishio, S.; Takada, N.; Terakami, S.; Takeuchi, Y.; Kimura, M.K.; Isoda, K.; Saito, T.; Iketani, H. Genetic structure analysis of cultivated and wild chestnut populations reveals gene flow from cultivars to natural stands. Sci. Rep. 2021, 11, 240. [Google Scholar] [CrossRef]
- Deb, S.; Della Lucia, M.C.; Ravi, S.; Bertoldo, G.; Stevanato, P. Transcriptome-Assisted SNP Marker Discovery for Phytophthora infestans Resistance in Solanum lycopersicum L. Int. J. Mol. Sci. 2023, 24, 6798. [Google Scholar] [CrossRef]
- Bali, S.; Robinson, B.R.; Sathuvalli, V.; Bamberg, J.; Goyer, A. Single Nucleotide Polymorphism (SNP) markers associated with high folate content in wild potato species. PLoS ONE 2018, 13, e0193415. [Google Scholar] [CrossRef]
- Roncallo, P.F.; Beaufort, V.; Larsen, A.O.; Dreisigacker, S.; Echenique, V. Genetic diversity and linkage disequilibrium using SNP (KASP) and AFLP markers in a worldwide durum wheat (Triticum turgidum L. var durum) collection. PLoS ONE 2019, 14, e0218562. [Google Scholar] [CrossRef]
- Castilla, A.R.; Méndez-Vigo, B.; Marcer, A.; Martínez-Minaya, J.; Conesa, D.; Picó, F.X.; Alonso-Blanco, C. Ecological, genetic and evolutionary drivers of regional genetic differentiation in Arabidopsis thaliana. BMC Evol. Biol. 2020, 20, 71. [Google Scholar] [CrossRef] [PubMed]
- Oh, A.; Oh, B.U. Genetic differentiation that is exceptionally high and unexpectedly sensitive to geographic distance in the absence of gene flow: Insights from the genus Eranthis in East Asian regions. Ecol. Evol. 2022, 12, e9007. [Google Scholar] [CrossRef]
- Santangelo, J.S.; Johnson, M.T.J.; Ness, R.W. Modern spandrels: The roles of genetic drift, gene flow and natural selection in the evolution of parallel clines. Proceedings. Biol. Sci. 2018, 285, 20180230. [Google Scholar] [CrossRef]
- Marcais, G.; Kingsford, C. A fast, lock-free approach for efficient parallel counting of occurrences of k-mers. Bioinformatics 2011, 27, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Roach, M.J.; Schmidt, S.A.; Borneman, A.R. Purge Haplotigs: Allelic contig reassignment for third-gen diploid genome assemblies. BMC Bioinform. 2018, 19, 460. [Google Scholar] [CrossRef] [PubMed]
- Dudchenko, O.; Batra, S.S.; Omer, A.D.; Nyquist, S.K.; Hoeger, M.; Durand, N.C. De novo assembly of the Aedes aegypti genome using Hi-C yields chromosome-length scaffolds. Science 2017, 356, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Durand, N.; Robinson, J.; Shamim, S.; Aiden, E.L. Juicebox provides a visualization system for Hi-C contact maps with unlimited zoom. Cell Syst. 2016, 3, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Simão, F.A.; Waterhouse, R.M.; Loannidis, P.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO: Assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 2015, 31, 3210–3212. [Google Scholar] [CrossRef]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef]
- Ou, S.; Su, W.; Liao, Y.; Chougule, K.; Doreen, W.; Thomas, P.; Ning, J.; Candice, N.H.; Hufford, M.B. Benchmarking transposable element annotation methods for creation of a streamlined, comprehensive pipeline. Cold Spring Harb. Lab. 2019, 1, 275–281. [Google Scholar] [CrossRef]
- Bao, W.; Kojima, K.K.; Kohany, O. Repbase Update, a database of repetitive elements in eukaryotic genomes. Mob. DNA 2015, 6, 11. [Google Scholar] [CrossRef]
- Tempel, S. Using and Understanding Repeat Masker. Methods Mol. Biol. 2012, 859, 29–51. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef]
- Haas, B.J.; Delcher, A.L.; Mount, S.M.; Wortman, J.R.; Smith, J.; Hannick, L.I.; Rama, M.; Ronning, C.M.; Rusch, D.B.; Town, C.D. Improving the Arabidopsis genome annotation using maximal transcript alignment assemblies. Nucleic Acids Res. 2003, 31, 5654–5666. [Google Scholar] [CrossRef] [PubMed]
- Majoros, W.; Pertea, M.; Salzberg, S. TigrScan and Glimmer HMM: Two open source ab initio eukaryotic gene-finders. Bioinformatics 2004, 20, 2878–2879. [Google Scholar] [CrossRef]
- Stanke, M.; Keller, O.; Gunduz, I.; Hayes, A.; Waack, S.; Morgenstern, B. AUGUSTUS: Ab initio prediction of alternative transcripts. Nucleic Acids Res. 2006, 34, W435–W439. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.D.; Handsaker, R.E.; Pulit, S.L.; Nizzari, M.M.; O’donnell, C.J.; De Bakker, P.I. SNAP: A web-based tool for identification and annotation of proxy SNPs using HapMap. Bioinformatics 2008, 24, 2938–2939. [Google Scholar] [CrossRef] [PubMed]
- Haas, B.J.; Salzberg, S.L.; Zhu, W.; Pertea, M.; Allen, J.E.; Orvis, J.; White, O.; Buell, C.R.; Wortman, J.R. Automated eukaryotic gene structure annotation using evidence modeler and the program to assemble spliced alignments. Genome Biol. 2008, 9, R7. [Google Scholar] [CrossRef] [PubMed]
- Emms, D.M.; Kelly, S. Ortho Finder: Phylogenetic orthology inference for comparative genomics. Genome Biol. 2019, 20, 238. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Yang, Z. PAML 4: Phylogenetic analysis by maximum likelihood. Mol. Bio. Evol. 2007, 24, 1586–1591. [Google Scholar] [CrossRef]
- Han, M.V.; Thomas, G.W.C.; Lugo-Martinez, J.; Hahn, M.W. Estimating gene gain and loss rates in the presence of error in genome assembly and annotation using CAFE 3. Mol Biol. Evol. 2013, 30, 1987–1997. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Y.; Zhang, Z.; Zhu, J.; Yu, J. KaKs_Calculator 2.0:a toolkit incorporating gamma-series methods and sliding window strategies. Genom. Proteom. Bioinform. 2010, 8, 77–80. [Google Scholar] [CrossRef]
- Marais, G.; Delcher, A.L.; Phillippy, A.M.; Coston, R.; Zimin, A. MUMmer4: A fast and versatile genome alignment system. PLOS Comput. Biol. 2018, 14, e1005944. [Google Scholar] [CrossRef] [PubMed]
- Sudhir, K.; Glen, S.; Michael, L.; Christina, K.; Koichiro, T. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar]
- Price, A.L.; Patterson, N.J.; Plenge, R.M.; Weinblatt, M.E.; Shadick, N.A.; Reich, D. Principal components analysis corrects for stratification in genome-wide association studies. Nat. Genet. 2006, 38, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Vilella, A.J.; Blanco-Garcia, A.; Hutter, S.; Rozas, J. VariScan: Analysis of evolutionary patterns from large-scale DNA sequence polymorphism data. Bioinformatics 2005, 21, 2791–2793. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.H.; Novembre, J.; Lange, K. Fast model-based estimation of ancestry in unrelated individuals. Genome Res. 2009, 19, 1655–1664. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).