Usage of Fermental Traps for the Study of the Species Diversity of Coleoptera
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Placement of Traps
2.2. Usage of Attractive Liquids
- (1)
- beer with sugar (BS),
- (2)
- beer without sugar (B),
- (3)
- red wine with sugar (RvS),
- (4)
- red wine without sugar (Rv),
- (5)
- white wine with sugar (WvS),
- (6)
- white wine without sugar (Wv).
2.3. Calculations and Used Terms
2.4. Format
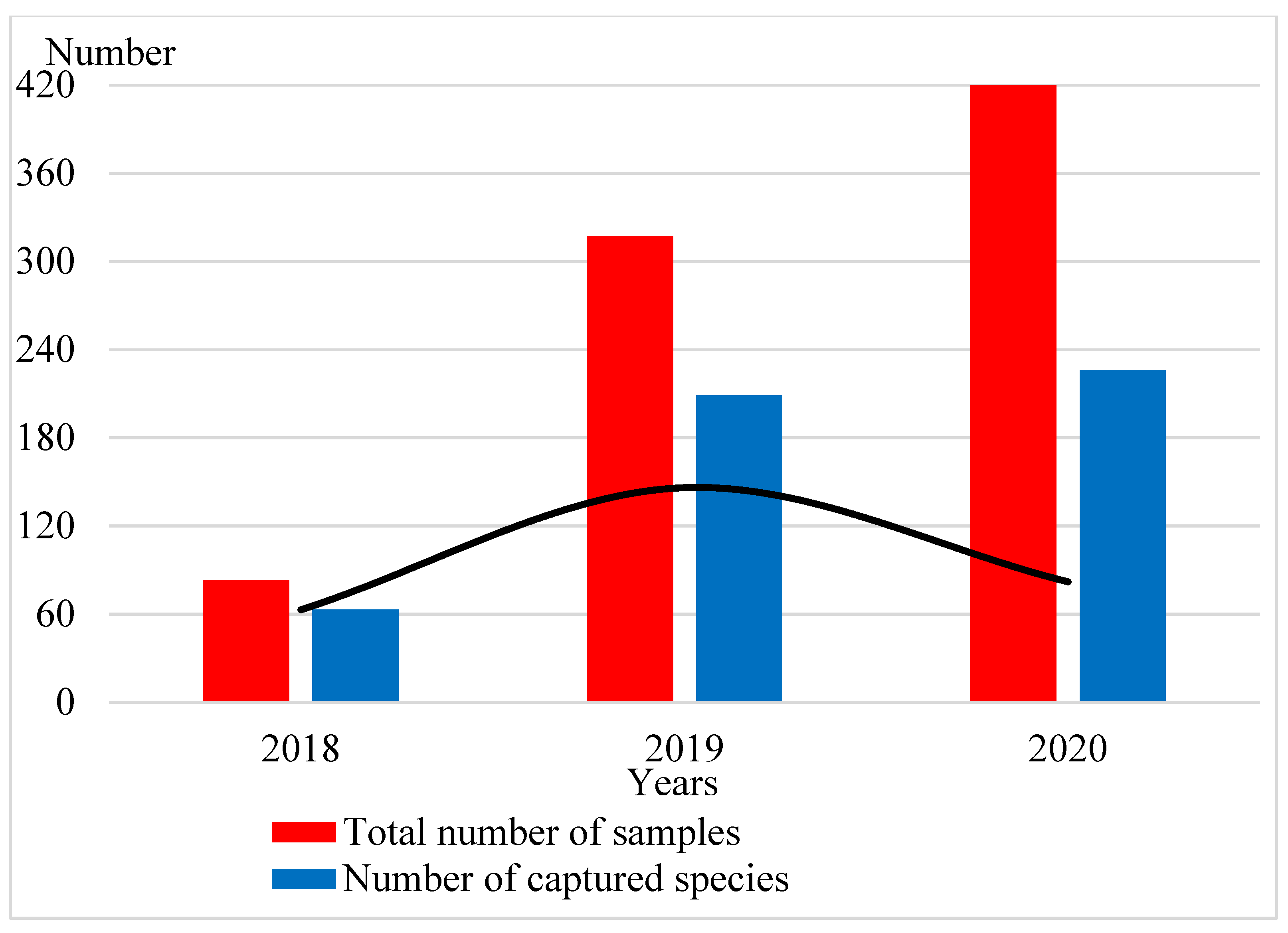
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Family, Species | 2018 | 2019 | 2020 | |||
|---|---|---|---|---|---|---|
| Number of Specimens | Occurrence, % | Number of Specimens | Occurrence, % | Number of Specimens | Occurrence, % | |
| Carabidae | ||||||
| Dromius agilis (Fabricius, 1787) | 1 | 0.24 | ||||
| Dromius quadraticollis A. Morawitz, 1862 | 3 | 0.95 | ||||
| Harpalus distinguendus (Duftschmid, 1812) | 1 | 0.24 | ||||
| Harpalus signaticornis (Duftschmid, 1812) | 1 | 0.32 | ||||
| Harpalus xanthopus winkleri Schauberger, 1923 | 2 | 0.48 | ||||
| Lebia marginata (Geoffroy, 1785) | 1 | 0.32 | ||||
| Limodromus assimilis (Paykull, 1790) | 2 | 0.48 | ||||
| Limodromus krynickii (Sperk, 1835) | 1 | 0.32 | ||||
| Tachyta nana (Gyllenhal, 1810) | 1 | 0.32 | ||||
| Dytiscidae | ||||||
| Ilybius erichsoni (Gemminger & Harold, 1868) | 1 | 0.24 | ||||
| Ilybius fuliginosus (Fabricius, 1792) | 1 | 0.32 | ||||
| Hydrochidae | ||||||
| Hydrochus brevis (Herbst, 1793) | 1 | 0.32 | ||||
| Histeridae | ||||||
| Atholus duodecimstriatus (Schrank, 1781) | 1 | 0.32 | ||||
| Gnathoncus buyssoni Auzat, 1917 | 7 | 1.89 | 6 | 1.19 | ||
| Platysoma elongatum (Thunberg, 1787) | 8 | 1.58 | 4 | 0.71 | ||
| Platysoma lineare Erichson, 1834 | 7 | 1.89 | 1 | 0.24 | ||
| Silphidae | ||||||
| Dendroxena quadrimaculata (Scopoli, 1771) | 3 | 0.95 | 41 | 5 | ||
| Necrodes littoralis (Linnaeus, 1758) | 2 | 2.4 | 21 | 1.58 | 45 | 4.76 |
| Nicrophorus humator (Gleditsch, 1767) | 1 | 1.2 | 4 | 0.71 | ||
| Nicrophorus interruptus Stephens, 1830 | 14 | 1.67 | ||||
| Nicrophorus sepultor Charpentier, 1825 | 1 | 0.24 | ||||
| Nicrophorus vespillo (Linnaeus, 1758) | 1 | 0.24 | ||||
| Nicrophorus vespilloides Herbst, 1783 | 20 | 2.4 | 3 | 0.32 | 4 | 0.71 |
| Oiceoptoma thoracicum (Linnaeus, 1758) | 11 | 9.5 | 24 | 4.1 | 13 | 2.62 |
| Silpha tristis Illiger, 1798 | 2 | 0.24 | ||||
| Staphylinidae | ||||||
| Staphylinidae sp. | 13 | 13.1 | 423 | 22.08 | 252 | 28.57 |
| Philonthus sp. | 1 | 1.2 | ||||
| Quedius dilatatus (Fabricius, 1787) | 6 | 6 | 329 | 23.66 | 221 | 16.67 |
| Lucanidae | ||||||
| Lucanus cervus (Linnaeus, 1758) | 4 | 0.71 | ||||
| Platycerus caprea (De Geer, 1774) | 1 | 0.24 | ||||
| Platycerus caraboides (Linnaeus, 1758) | 1 | 0.32 | 1 | 0.24 | ||
| Sinodendron cylindricum (Linnaeus, 1758) | 1 | 0.24 | ||||
| Scarabaeidae | ||||||
| Cetonia aurata (Linnaeus, 1758) | 60 | 21.4 | 122 | 17.03 | 635 | 20.48 |
| Esymus pusillus (Herbst, 1789) | 1 | 0.24 | ||||
| Gnorimus variabilis (Linnaeus, 1758) | 16 | 7.1 | 33 | 5.99 | 12 | 2.14 |
| Osmoderma barnabita Motschulsky, 1845 | 2 | 0.32 | ||||
| Protaetia affinis (Andersch, 1797) | 1 | 1.2 | 2 | 0.24 | ||
| Protaetia fieberi (Kraatz, 1880) | 56 | 33.3 | 250 | 30.28 | 617 | 29.29 |
| Protaetia marmorata (Fabricus, 1792) | 750 | 82.1 | 2443 | 67.51 | 2550 | 67.14 |
| Protaetia speciosissima (Scopoli, 1786) | 16 | 10.7 | 64 | 8.52 | 34 | 5 |
| Protaetia cuprea volhyniensis (Gory & Percheron, 1833) | 13 | 11.9 | 100 | 17.03 | 327 | 15.71 |
| Serica brunnea (Linnaeus, 1758) | 1 | 0.32 | 1 | 0.24 | ||
| Trichius fasciatus (Linnaeus, 1758) | 4 | 1.26 | ||||
| Scirtidae | ||||||
| Contacyphon padi (Linnaeus, 1758) | 4 | 1.26 | ||||
| Contacyphon pubescens (Fabricius, 1792) | 3 | 0.63 | ||||
| Contacyphon sp. | 1 | 0.32 | 2 | 0.48 | ||
| Microcara testacea (Linnaeus,1767) | 2 | 1.2 | 1 | 0.32 | 3 | 0.71 |
| Buprestidae | ||||||
| Agrilus sulcicollis Lacordaire, 1835 | 2 | 0.63 | ||||
| Agrilus angustulus (Illiger, 1803) | 1 | 0.24 | ||||
| Anthaxia quadripunctata (Linnaeus, 1758) | 1 | 0.24 | ||||
| Buprestis haemorrhoidalis Herbst, 1780 | 1 | 0.32 | ||||
| Dicerca alni (Fischer von Waldheim, 1824) | 1 | 0.32 | ||||
| Phaenops cyanea (Fabricius, 1775) | 2 | 0.63 | ||||
| Trachys minutus (Linnaeus, 1758) | 1 | 0.32 | ||||
| Eucnemidae | ||||||
| Melasis buprestoides (Linnaeus, 1760) | 1 | 0.24 | ||||
| Otho sphondyloides (Germar, 1818) | 1 | 0.32 | ||||
| Throscidae | ||||||
| Trixagus sp. | 4 | 0.95 | 9 | 1.13 | ||
| Aulonothroscus sp. | 1 | 0.32 | ||||
| Elateridae | ||||||
| Agriotes lineatus (Linnaeus, 1767) | 1 | 0.24 | ||||
| Agriotes obscurus (Linnaeus, 1758) | 1 | 0.24 | ||||
| Agrypnus murinus (Linnaeus, 1758) | 7 | 2.21 | 29 | 5.48 | ||
| Ampedus balteatus (Linnaeus, 1758) | 2 | 0.63 | 5 | 1.19 | ||
| Ampedus cinnabarinus (Eschscholtz, 1829) | 9 | 2.21 | 97 | 5 | ||
| Ampedus elongatulus (Fabricius, 1787) | 3 | 0.95 | 1 | 0.24 | ||
| Ampedus nigerrimus (Lacordaire in Boisduval & Lacordaire, 1835) | 1 | 0.24 | ||||
| Ampedus nigrinus (Herbst, 1784) | 1 | 0.32 | ||||
| Ampedus nigroflavus (Goeze, 1777) | 13 | 1.9 | ||||
| Ampedus pomonae (Stephens, 1830) | 1 | 0.32 | 13 | 1.67 | ||
| Ampedus pomorum (Herbst, 1784) | 4 | 1.26 | 97 | 9.05 | ||
| Ampedus praeustus (Fabricius, 1792) | 1 | 0.32 | 13 | 1.67 | ||
| Ampedus sanguinolentus (Schrank, 1776) | 3 | 0.95 | 45 | 4.05 | ||
| Ampedus sanguineus (Linnaeus, 1758) | 3 | 0.48 | ||||
| Ampedus tristis (Linnaeus, 1758) | 1 | 0.24 | ||||
| Aplotarsus incanus (Gyllenhal, 1827) | 1 | 0.32 | ||||
| Athous haemorrhoidalis (Fabricius, 1801) | 1 | 0.24 | ||||
| Athous subfuscus (O.F. Müller, 1764) | 2 | 0.32 | 2 | 0.48 | ||
| Athous vittatus (Fabricius, 1792) | 1 | 0.32 | 4 | 0.95 | ||
| Cardiophorus ruficollis (Linnaeus, 1758) | 1 | 0.32 | 1 | 0.24 | ||
| Dalopius marginatus (Linnaeus, 1758) | 1 | 1.2 | 24 | 3.79 | 15 | 3.33 |
| Danosoma fasciatum (Linnaeus, 1758) | 1 | 0.32 | ||||
| Denticollis borealis (Paykull, 1800) | 1 | 0.32 | 2 | 0.48 | ||
| Ectinus aterrimus (Linnaeus, 1760) | 1 | 0.32 | ||||
| Elater ferrugineus Linnaeus, 1758 | 2 | 2.4 | 6 | 1.89 | ||
| Hemicrepidius niger (Linnaeus, 1758) | 1 | 0.24 | ||||
| Lacon lepidopterus (Panzer, 1800) | 1 | 0.24 | ||||
| Limonius minutus (Linnaeus, 1758) | 3 | 0.95 | 4 | 0.95 | ||
| Melanotus castanipes (Paykull, 1800) | 8 | 2.52 | 9 | 1.19 | ||
| Melanotus villosus (Geoffroy, 1785) | 5 | 0.95 | ||||
| Mosotalesus nigricornis (Panzer, 1799) | 3 | 0.63 | 1 | 0.24 | ||
| Prosternon tesselatum (Linnaeus, 1758) | 1 | 1.2 | 14 | 3.47 | 124 | 9.76 |
| Selatosomus aeneus (Linnaeus, 1758) | 1 | 0.32 | 17 | 1.43 | ||
| Sericus brunneus (Linnaeus, 1758) | 1 | 0.24 | ||||
| Lycidae | ||||||
| Lygistopterus sanguineus (Linnaeus, 1758) | 8 | 1.26 | ||||
| Cantharidae | ||||||
| Cantharis flavilabris Fallén, 1807 | 1 | 0.32 | ||||
| Cantharis livida Linnaeus, 1758 | 12 | 2.4 | 6 | 1.26 | 196 | 4.29 |
| Cantharis nigricans O.F. Müller, 1776 | 2 | 1.2 | 9 | 2.84 | 90 | 6.9 |
| Cantharis pallida Goeze, 1777 | 7 | 0.95 | ||||
| Cantharis pellucida Fabricius, 1792 | 10 | 1.26 | 83 | 5.24 | ||
| Cantharis rufa Linnaeus, 1758 | 4 | 0.95 | 1 | 0.24 | ||
| Cantharis rustica Fallén, 1807 | 1 | 1.2 | 12 | 0.95 | ||
| Malthodes guttifer Kiesenwetter, 1852 | 2 | 0.48 | ||||
| Malthodes sp. | 1 | 0.24 | ||||
| Podabrus alpinus (Paykull, 1798) | 1 | 0.24 | ||||
| Rhagonycha fulva (Scopoli, 1763) | 1 | 1.2 | 1 | 0.24 | ||
| Rhagonycha fugax Mannerheim, 1843 | 4 | 0.71 | ||||
| Rhagonycha lignosa (O.F. Müller, 1764) | 4 | 0.95 | ||||
| Rhagonycha nigriventris Motschulsky, 1860 | 2 | 0.48 | ||||
| Dermestidae | ||||||
| Attagenus schaefferi (Herbst, 1792) | 107 | 4.42 | 16 | 2.38 | ||
| Anthrenus museorum (Linnaeus, 1760) | 1 | 0.32 | ||||
| Ctesias serra (Fabricius, 1792) | 39 | 4.42 | ||||
| Dermestes laniarius Illiger, 1801 | 1 | 0.32 | ||||
| Dermestes lardarius Linnaeus, 1758 | 1 | 0.32 | 1 | 0.24 | ||
| Dermestinus murinus Linnaeus, 1758 | 4 | 0.71 | ||||
| Globicornis emarginata (Gyllenhal, 1808) | 27 | 2.52 | 17 | 2.14 | ||
| Megatoma undata (Linnaeus, 1758) | 1 | 0.32 | 2 | 0.48 | ||
| Trogoderma glabrum (Herbst, 1783) | 2 | 2.4 | 88 | 5.68 | 18 | 3.33 |
| Ptinidae | ||||||
| Dorcatoma dresdensis Herbst, 1792 | 1 | 0.32 | ||||
| Dorcatoma flavicornis (Fabricius, 1792) | 1 | 0.32 | ||||
| Dorcatoma robusta A. Strand, 1938 | 6 | 1.58 | 1 | 0.24 | ||
| Lymexylidae | ||||||
| Elateroides dermestoides (Linnaeus, 1760) | 1 | 0.24 | ||||
| Cleridae | ||||||
| Allonyx quadrimaculatus (Schaller, 1783) | 2 | 0.63 | ||||
| Thanasimus femoralis (Zetterstedt, 1828) | 3 | 0.95 | 2 | 0.48 | ||
| Thanasimus formicarius (Linnaeus, 1758) | 4 | 1.26 | 3 | 0.71 | ||
| Tillus elongatus (Linnaeus, 1758) | 1 | 0.32 | ||||
| Trichodes apiarius (Linnaeus, 1758) | 1 | 1.2 | 2 | 0.63 | 3 | 0.71 |
| Melyridae | ||||||
| Cordylepherus viridis (Fabricius, 1787) | 1 | 0.32 | 3 | 0.71 | ||
| Dasytes niger (Linnaeus, 1760) | 1 | 1.2 | 10 | 3.15 | 12 | 1.9 |
| Dasytes fusculus (Illiger, 1801) | 1 | 0.32 | 7 | 0.95 | ||
| Malachius bipustulatus (Linnaeus, 1758) | 5 | 0.95 | ||||
| Erotylidae | ||||||
| Triplax russica (Linnaeus, 1758) | 2 | 0.63 | 1 | 0.24 | ||
| Tritoma subbasalis (Reitter, 1896) | 2 | 0.48 | ||||
| Monotomidae | ||||||
| Rhizophagus fenestralis (Linnaeus, 1758) | 15 | 2.84 | 39 | 4.29 | ||
| Nitidulidae | ||||||
| Carpophilus hemipterus (Linnaeus, 1758) | 10 | 2.21 | 2 | 0.48 | ||
| Carpophilus marginellus Motschulsky, 1858 | 2 | 0.24 | ||||
| Carpophilus sp. | 1 | 0.32 | ||||
| Cryptarcha strigata (Fabricius, 1787) | 249 | 60.7 | 1227 | 44.79 | 1406 | 48.09 |
| Cryptarcha undata (G.-A. Olivier, 1790) | 8 | 3.6 | 14 | 3.78 | 105 | 8.57 |
| Cychramus luteus (Fabricius, 1787) | 9 | 6 | 834 | 12.3 | 101 | 6.9 |
| Cychramus variegatus (Herbst, 1792) | 4 | 1.2 | 56 | 5.05 | 15 | 1.43 |
| Cyllodes ater (Herbst, 1792) | 1 | 1.2 | 1 | 0.24 | ||
| Epuraea sp. | 8 | 8.3 | 268 | 15.77 | 437 | 19.52 |
| Glischrochilus grandis (Tournier, 1872) | 47 | 25 | 652 | 25.24 | 4876 | 50.48 |
| Glischrochilus hortensis (Geoffroy, 1785) | 71 | 25 | 885 | 28.08 | 783 | 32.38 |
| Glischrochilus quadriguttatus (Fabricius, 1777) | 1 | 1.2 | 33 | 5.68 | 3 | 0.71 |
| Glischrochilus quadripunctatus (Linnaeus, 1758) | 2 | 2.4 | 96 | 11.04 | 105 | 11.19 |
| Glischrochilus quadrisignatus (Say, 1835) | 2 | 2.4 | 13 | 2.52 | 75 | 6.19 |
| Meligethes sp. | 3 | 0.63 | 6 | 0.24 | ||
| Omosita discoidea (Fabricius, 1775) | 1 | 0.32 | ||||
| Pocadius ferrugineus (Fabricius, 1775) | 1 | 0.24 | ||||
| Soronia grisea (Linnaeus, 1758) | 47 | 21.4 | 363 | 25.24 | 654 | 39.05 |
| Soronia punctatissima (Illiger, 1794) | 3 | 0.95 | 5 | 0.48 | ||
| Silvanidae | ||||||
| Uleiota planatus (Linnaeus, 1760) | 1 | 0.32 | ||||
| Cucujidae | ||||||
| Pediacus depressus (Herbst, 1797) | 15 | 3.47 | 29 | 2.86 | ||
| Laemophloeidae | ||||||
| Cryptolestes sp. | 1 | 0.32 | ||||
| Cerylonidae | ||||||
| Cerylon ferrugineum Stephens, 1830 | 2 | 0.32 | ||||
| Cerylon histeroides (Fabricius, 1792) | 1 | 0.32 | ||||
| Latridiidae | ||||||
| Corticaria sp. | 2 | 0.48 | ||||
| Cortinicara gibbosa (Herbst, 1793) | 1 | 0.32 | 1 | 0.24 | ||
| Enicmus histrio Joy & Tomlin, 1910 | 1 | 0.32 | ||||
| Stephostethus pandellei (C.N.F. Brisout de Barneville, 1863) | 2 | 0.63 | ||||
| Coccinellidae | ||||||
| Adalia bipunctata (Linnaeus, 1758) | 1 | 0.24 | ||||
| Adalia decempunctata (Linnaeus, 1758) | 1 | 0.24 | ||||
| Anatis ocellata (Linnaeus, 1758) | 1 | 0.32 | 4 | 0.95 | ||
| Calvia decemguttata (Linnaeus, 1767) | 1 | 1.2 | 6 | 1.26 | 10 | 1.9 |
| Calvia quatuordecimguttata (Linnaeus, 1758) | 7 | 2.21 | 14 | 2.38 | ||
| Chilocorus renipustulatus (L.G. Scriba, 1791) | 2 | 0.63 | ||||
| Coccinella magnifica L. Redtenbacher, 1843 | 2 | 0.48 | ||||
| Coccinella septempunctata Linnaeus, 1758 | 1 | 0.24 | ||||
| Exochomus quadripustulatus (Linnaeus, 1758) | 1 | 0.24 | ||||
| Halyzia sedecimguttata (Linnaeus, 1758) | 1 | 1.2 | 5 | 1.58 | 9 | 2.14 |
| Harmonia axyridis (Pallas, 1773) | 1 | 0.24 | ||||
| Harmonia quadripunctata (Pontoppidan, 1763) | 2 | 0.63 | 6 | 1.19 | ||
| Hippodamia variegata (Goeze, 1777) | 1 | 0.32 | 1 | 0.24 | ||
| Mysia oblongoguttata (Linnaeus, 1758) | 1 | 0.32 | 5 | 1.19 | ||
| Oenopia conglobata (Linnaeus, 1758) | 1 | 0.32 | 2 | 0.48 | ||
| Propylea quatuordecimpunctata (Linnaeus, 1758) | 2 | 0.48 | ||||
| Sospita vigintiguttata (Linnaeus, 1758) | 1 | 0.32 | 1 | 0.24 | ||
| Vibidia duodecimguttata (Poda von Neuhaus, 1761) | 1 | 0.24 | ||||
| Mycetophagidae | ||||||
| Litargus connexus (Geoffroy, 1785) | 17 | 3.47 | 11 | 1.19 | ||
| Mycetophagus quadripustulatus (Linnaeus, 1760) | 1 | 0.32 | 2 | 0.48 | ||
| Melandryidae | ||||||
| Osphya bipunctata (Fabricius, 1775) | 1 | 0.24 | ||||
| Phloiotrya subtilis (Reitter, 1897) | 1 | 0.24 | ||||
| Mordellidae | ||||||
| Tomoxia bucephala A. Costa, 1854 | 7 | 1.89 | 1 | 0.24 | ||
| Mordella sp. | 3 | 0.63 | 1 | 0.24 | ||
| Tenebrionidae | ||||||
| Bolitophagus reticulatus (Linnaeus, 1767) | 3 | 0.71 | ||||
| Corticeus unicolor Piller & Mitterpacher, 1783 | 2 | 0.48 | ||||
| Lagria hirta (Linnaeus, 1758) | 19 | 4.73 | 3 | 0.71 | ||
| Mycetochara axillaris (Paykull, 1799) | 1 | 0.32 | ||||
| Mycetochara flavipes (Fabricius, 1792) | 1 | 0.32 | 1 | 0.24 | ||
| Upis ceramboides (Linnaeus, 1758) | 4 | 0.95 | 1 | 0.24 | ||
| Oedemeridae | ||||||
| Chrysanthia geniculata W.L.E. Schmidt, 1846 | 6 | 1.19 | ||||
| Chrysanthia viridissima (Linnaeus, 1758) | 2 | 2.4 | 4 | 0.71 | ||
| Nacerdes carniolica (Gistel, 1834) | 38 | 1.67 | ||||
| Oedemera femorata (Scopoli, 1763) | 1 | 0.24 | ||||
| Oedemera virescens (Linnaeus, 1767) | 1 | 0.24 | ||||
| Boridae | ||||||
| Boros schneideri (Panzer, 1796) | 1 | 0.24 | ||||
| Pyrochroidae | ||||||
| Pyrochroa coccinea (Linnaeus, 1760) | 1 | 0.32 | 9 | 0.71 | ||
| Schizotus pectinicornis (Linnaeus, 1758) | 1 | 0.32 | 14 | |||
| Salpingidae | ||||||
| Salpingidae sp. | 1 | 0.32 | ||||
| Salpingus ruficollis (Linnaeus, 1760) | 1 | 0.24 | ||||
| Aderidae | ||||||
| Phytobaenus amabilis R.F. Sahlberg, 1834 | 1 | 0.24 | ||||
| Scraptiidae | ||||||
| Anaspis frontalis (Linnaeus, 1758) | 1 | 1.2 | 1 | 0.32 | 5 | 0.95 |
| Anaspis thoracica (Linnaeus, 1758) | 1 | 0.24 | ||||
| Cerambycidae | ||||||
| Aegomorphus clavipes (Schrank, 1781) | 1 | 1.2 | 1 | 0.32 | ||
| Alosterna ingrica (Baeckmann, 1902) | 1 | 0.32 | ||||
| Alosterna tabacicolor (De Geer, 1775) | 1 | 1.2 | 17 | 1.58 | 1 | 0.24 |
| Anaesthetis testacea (Fabricius, 1781) | 1 | 0.32 | ||||
| Anastrangalia reyi (L. Heyden, 1889) | 1 | 0.32 | 2 | 0.48 | ||
| Anoplodera rufipes ventralis Heyden, 1886 | 1 | 0.24 | ||||
| Anoplodera sexguttata (Fabricius, 1775) | 1 | 1.2 | 23 | 3.15 | 1 | 0.24 |
| Arhopalus rusticus (Linnaeus, 1758) | 5 | 0.95 | ||||
| Aromia moschata (Linnaeus, 1758) | 23 | 11.9 | 58 | 10.09 | 23 | 3.33 |
| Chlorophorus herbstii (Brahm, 1790) | 1 | 0.32 | ||||
| Cortodera femorata (Fabricius, 1787) | 4 | 0.71 | ||||
| Dinoptera collaris (Linnaeus, 1758) | 4 | 1.26 | 5 | 0.95 | ||
| Etorofus pubescens (Fabricius, 1787) | 1 | 0.32 | ||||
| Euracmaeops marginatus (Fabricius, 1781) | 1 | 0.32 | ||||
| Euracmaeops septentrionis (C.G. Thomson, 1866) | 1 | 0.32 | ||||
| Judolia sexmaculata (Linnaeus, 1758) | 1 | 0.32 | ||||
| Leiopus linnei Wallin, Nylander & Kvamme, 2009 | 1 | 0.32 | 2 | 0.48 | ||
| Leptura aurulenta Fabricius, 1793 | 2 | 2.4 | 1 | 0.32 | ||
| Leptura thoracica Creutzer, 1799 | 68 | 22.6 | 751 | 31.55 | 1113 | 21.43 |
| Leptura quadrifasciata Linnaeus, 1758 | 104 | 35.7 | 489 | 37.22 | 433 | 19.29 |
| Lepturalia nigripes (De Geer, 1775) | 2 | 1.2 | 14 | 1.89 | 57 | 4.76 |
| Lepturobosca virens (Linnaeus, 1758) | 5 | 0.48 | ||||
| Mesosa myops (Dalman, 1817) | 5 | 3.6 | 13 | 2.84 | 1 | 0.24 |
| Molorchus minor (Linnaeus, 1758) | 3 | 0.95 | 12 | 2.38 | ||
| Monochamus sutor (Linnaeus, 1758) | 1 | 0.32 | ||||
| Necydalis major Linnaeus, 1758 | 9 | 7.1 | 47 | 10.73 | 32 | 5.71 |
| Nivellia sanguinosa (Gyllenhal, 1827) | 1 | 0.32 | ||||
| Obrium cantharinum (Linnaeus, 1767) | 1 | 1.2 | 45 | 5.68 | 104 | 7.14 |
| Oedecnema gebleri (Ganglbauer, 1889) | 2 | 0.48 | ||||
| Pachyta quadrimaculata (Linnaeus, 1758) | 4 | 3.6 | 5 | 1.58 | 3 | 0.71 |
| Phymatodes testaceus (Linnaeus, 1758) | 2 | 0.32 | 24 | 2.86 | ||
| Plagionotus arcuatus (Linnaeus, 1758) | 2 | 0.48 | ||||
| Plagionotus detritus (Linnaeus, 1758) | 1 | 1.2 | 8 | 2.21 | 119 | 2.38 |
| Prionus coriarius (Linnaeus, 1758) | 1 | 0.32 | ||||
| Purpuricenus globulicollis Dejean, 1839 | 5 | 0.95 | 1 | 0.24 | ||
| Purpuricenus kaehleri (Linnaeus, 1758) | 7 | 7.1 | 124 | 11.04 | 167 | 7.62 |
| Rhagium inquisitor (Linnaeus, 1758) | 24 | 4.73 | 10 | 1.9 | ||
| Rhagium mordax (De Geer, 1775) | 49 | 20.2 | 422 | 24.29 | 778 | 34.05 |
| Rhagium sycophanta (Schrank, 1781) | 2 | 2.4 | ||||
| Rhamnusium bicolor (Schrank, 1781) | 1 | 1.2 | ||||
| Ropalopus clavipes (Fabricius, 1775) | 1 | 0.24 | ||||
| Ropalopus macropus (Germar, 1823) | 2 | 0.24 | ||||
| Rutpela maculata (Poda von Neuhaus, 1761) | 9 | 7.1 | 20 | 3.15 | 16 | 2.38 |
| Saperda scalaris (Linnaeus, 1758) | 1 | 0.24 | ||||
| Spondylis buprestoides (Linnaeus, 1758) | 2 | 0.63 | 1 | 0.24 | ||
| Stenocorus meridianus (Linnaeus, 1758) | 7 | 8.3 | 93 | 11.67 | 38 | 1.9 |
| Stenurella melanura (Linnaeus, 1758) | 1 | 0.24 | ||||
| Stictoleptura maculicornis (De Geer, 1775) | 2 | 0.63 | 2 | 0.48 | ||
| Stictoleptura rubra (Linnaeus, 1758) | 2 | 0.63 | 1 | 0.24 | ||
| Stictoleptura variicornis (Dalman, 1817) | 1 | 0.32 | ||||
| Strangalia attenuata (Linnaeus, 1758) | 8 | 1.89 | 2 | 0.48 | ||
| Trichoferus campestris (Faldermann, 1835) | 2 | 2.4 | 7 | 0.95 | 1 | 0.24 |
| Xylotrechus antilope (Schoenherr, 1817) | 17 | 3.47 | 34 | 4.29 | ||
| Xylotrechus arvicola (Olivier, 1795) | 1 | 1.2 | 2 | 0.48 | ||
| Xylotrechus capricornus (Gebler, 1830) | 1 | 0.24 | ||||
| Xylotrechus pantherinus (Savenius, 1825) | 2 | 0.63 | 1 | 0.24 | ||
| Xylotrechus rusticus (Linnaeus, 1758) | 3 | 0.95 | 1 | 0.24 | ||
| Chrysomelidae | ||||||
| Altica sp. | 5 | 1.58 | 8 | 1.43 | ||
| Aphthona sp. | 1 | 0.24 | ||||
| Chrysomela vigintipunctata (Scopoli, 1763) | 1 | 0.24 | ||||
| Crepidodera aurata (Marsham, 1802) | 1 | 0.32 | ||||
| Crepidodera nitidula (Linnaeus, 1758) | 1 | 0.32 | ||||
| Galerucella lineola (Fabricius, 1781) | 2 | 0.63 | 2 | 0.48 | ||
| Gonioctena viminalis (Linnaeus, 1758) | 1 | 0.24 | ||||
| Hypocassida subferruginea (Schrank, 1776) | 1 | 0.24 | ||||
| Lochmaea caprea (Linnaeus, 1758) | 2 | 0.32 | ||||
| Orsodacne cerasi (Linnaeus, 1758) | 2 | 0.48 | ||||
| Phyllotreta undulata Kutschera, 1860 | 1 | 0.24 | ||||
| Plagiosterna aenea (Linnaeus, 1758) | 1 | 0.32 | 1 | 0.24 | ||
| Anthribidae | ||||||
| Dissoleucas niveirostris (Fabricius, 1798) | 1 | 0.24 | ||||
| Tropideres albirostris (Schaller, 1783) | 2 | 0.63 | 5 | 1.19 | ||
| Attelabidae | ||||||
| Byctiscus betulae (Linnaeus, 1758) | 1 | 0.24 | ||||
| Brentidae | ||||||
| Betulapion simile (Kirby, 1811) | 1 | 0.32 | 1 | 0.24 | ||
| Curculionidae | ||||||
| Anisandrus dispar (Fabricius, 1792) | 1 | 1.2 | 386 | 10.73 | 2012 | 17.86 |
| Anthonomus incurvus (Panzer, 1795) | 1 | 0.32 | ||||
| Bagous puncticollis Boheman, 1845 | 1 | 0.32 | ||||
| Brachyderes incanus (Linnaeus, 1758) | 2 | 0.63 | ||||
| Coeliodinus rubicundus (Herbst, 1795) | 2 | 0.63 | ||||
| Curculio glandium Marsham, 1802 | 1 | 0.32 | 1 | 0.24 | ||
| Curculio nucum Linnaeus, 1758 | 2 | 2.4 | 1 | 0.32 | 16 | 0.48 |
| Curculio venosus (Gravenhorst, 1807) | 1 | 0.32 | ||||
| Curculio villosus Fabricius, 1781 | 4 | 0.95 | ||||
| Ellescus bipunctatus (Linnaeus, 1758) | 1 | 0.32 | ||||
| Ellescus scanicus (Paykull, 1792) | 1 | 0.32 | ||||
| Hylastes opacus Erichson, 1836 | 2 | 0.63 | ||||
| Ips acuminatus (Gyllenhal, 1827) | 1 | 0.32 | ||||
| Ips typographus (Linnaeus, 1758) | 1 | 0.24 | ||||
| Orchestes rusci (Herbst, 1795) | 1 | 0.32 | 1 | 0.24 | ||
| Phyllobius arborator (Herbst, 1797) | 1 | 0.32 | ||||
| Phyllobius argentatus (Linnaeus, 1758) | 6 | 1.26 | 5 | 1.19 | ||
| Phyllobius maculicornis Germar, 1823 | 2 | 0.63 | 1 | 0.24 | ||
| Phyllobius pomaceus Gyllenhal, 1834 | 1 | 0.32 | ||||
| Phyllobius pyri (Linnaeus, 1758) | 4 | 0.63 | 6 | 1.43 | ||
| Pissodes piniphilus (Herbst, 1797) | 1 | 0.24 | ||||
| Polydrusus cervinus (Linnaeus, 1758) | 1 | 0.32 | ||||
| Polydrusus flavipes (De Geer, 1775) | 1 | 1.2 | 1 | 0.32 | ||
| Polydrusus sp. | 1 | 0.24 | ||||
| Polydrusus tereticollis (De Geer, 1775) | 1 | 0.24 | ||||
| Polygraphus subopacus C.G. Thomson, 1871 | 1 | 0.32 | ||||
| Scolytus intricatus (Ratzeburg, 1837) | 1 | 0.24 | ||||
| Sitona ambiguus Gyllenhal, 1834 | 1 | 0.24 | ||||
| Sitona macularius (Marsham, 1802) | 1 | 0.24 | ||||
| Strophosoma capitatum (De Geer, 1775) | 5 | 1.26 | 7 | 1.43 | ||
| Trypodendron signatum (Fabricius, 1792) | 1 | 0.32 | ||||
| Xyleborus saxesenii (Ratzeburg, 1837) | 92 | 1.26 | 123 | 3.09 | ||
| TOTAL | 1750 | 11,655 | 19,864 | |||
References
- Lindenmayer, D.B.; Cunningham, R.B.; Donelly, C.F.; Lesslie, R. On the use of landscape surrogates as ecological indicators in fragmented forests. For. Ecol. Manag. 2002, 159, 203–216. [Google Scholar] [CrossRef]
- Mason, F.; Zapponi, L. The forest biodiversity artery: Towards forest management for saproxylic conservation. iForest Biogeosci. For. 2016, 9, 205–216. [Google Scholar] [CrossRef]
- Bazhina, E.V. Siberian fir (Abies sibirica) state and chemical element allocation in tree crown in forest ecosystems of Protected Areas in south of Krasnoyarsk Region (Russia). Nat. Conserv. Res. 2018, 3 (Suppl. 2), 40–53. [Google Scholar] [CrossRef]
- Uwalaka, N.O.; Muoghalu, J.I.; Osewole, A.O. Species diversity and successional dynamics in the secondary forest of Obafemi Awolowo University Biological Gardens Ile-Ife, Nigeria. Nat. Conserv. Res. 2018, 3, 21–34. [Google Scholar] [CrossRef]
- Rozhkov, Y.F.; Kondakova, M.Y. Assessment of the post-fire forest restoration dynamics in the Olekminsky State Nature Reserve (Russia) according to data of Landsat satellite images. Nat. Conserv. Res. 2019, 4 (Suppl. 1), 1–10. [Google Scholar] [CrossRef]
- Ruchin, A.B.; Khapugin, A.A. Red data book invertebrates in a protected area of European Russia. Acta Zool. Acad. Sci. Hung. 2019, 65, 349–370. [Google Scholar] [CrossRef]
- Arkhipova, M.V. Forest cover changes in the center of East European plain over the last 150 years. Russ. J. For. Sci. 2020, 1, 35–45. [Google Scholar] [CrossRef]
- Polevoi, A.V. Fungus gnats (Diptera: Bolitophilidae, Diadocidiidae, Keroplatidae, Mycetophilidae) in the Kostomuksha State Nature Reserve, Russia. Nat. Conserv. Res. 2021, 6 (Suppl. 1). [Google Scholar] [CrossRef]
- Carnus, J.M.; Parrotta, J.; Brockerhoff, E.; Arbez, M.; Jactel, H.; Kremer, A.; Walters, B. Planted forests and biodiversity. J. For. 2006, 104, 65–77. [Google Scholar]
- Cicort-Lucaciu, A.Ș. Road-killed ground beetles prove the presence of Carabus hungaricus (Coleoptera: Carabidae) in North-Western Romania. Nat. Conserv. Res. 2020, 5, 134–138. [Google Scholar] [CrossRef]
- Thuiller, W. Biodiversity–climate change and the ecologist. Nature 2007, 448, 550–552. [Google Scholar] [CrossRef] [PubMed]
- Ruchin, A.B.; Egorov, L.V. Overview of insect species included in the Red Data Book of Russian Federation in the Mordovia State Nature Reserve. Nat. Conserv. Res. 2017, 2 (Suppl. 1), 2–9. (In Russian) [Google Scholar] [CrossRef]
- Kovac, M.; Hladnik, D.; Kutnar, L. Biodiversity in (the Natura 2000) forest habitats is not static: Its conservation calls for an active management approach. J. Nat. Conserv. 2018, 43, 250–260. [Google Scholar] [CrossRef]
- Aleinikov, A.A. The fire history in pine forests of the plain area in the Pechora-Ilych Nature Biosphere Reserve (Russia) before 1942: Possible anthropogenic causes and long-term effects. Nat. Conserv. Res. 2019, 4 (Suppl. 1), 21–34. [Google Scholar] [CrossRef]
- Ruchin, A.B.; Alekseev, S.K.; Khapugin, A.A. Post-fire fauna of carabid beetles (Coleoptera, Carabidae) in forests of the Mordovia State Nature Reserve (Russia). Nat. Conserv. Res. 2019, 4 (Suppl. 1), 11–20. [Google Scholar] [CrossRef]
- Dedyukhin, S.V. Phytophagous beetles (Coleoptera: Chrysomelidae and Curculionoidea), protected and recommended for protection in the regions of the Middle Volga and the Urals. Nat. Conserv. Res. 2020, 5, 1–27. (In Russian) [Google Scholar] [CrossRef]
- Stork, N.E.; Grimbacher, P.S. Beetle assemblages from an Australian tropical rainforest show that the canopy and the ground strata contribute equally to biodiversity. Proc. Biol. Sci. 2006, 273, 1969–1975. [Google Scholar] [CrossRef] [PubMed]
- Dodds, K.J. Effects of trap height on captures of arboreal insects in pine stands of northeastern United States of America. Can. Entomol. 2014, 146, 80–89. [Google Scholar] [CrossRef]
- Skvarla, M.J.; Dowling, A.P.G. A comparison of trapping techniques (Coleoptera: Carabidae, Buprestidae, Cerambycidae, and Curculionoidea excluding Scolytinae). J. Insect Sci. 2017, 17, 1–28. [Google Scholar] [CrossRef]
- Tomaszewska, W.; Egorov, L.V.; Ruchin, A.B.; Vlasov, D.V. First record of Clemmus troglodytes (Coleoptera: Coccinelloidea, Anamorphidae) for the fauna of Russia. Nat. Conserv. Res. 2018, 3, 103–105. [Google Scholar] [CrossRef]
- Volf, M.; Klimeš, P.; Lamarre, G.P.A.; Redmond, C.M.; Seifert, C.L.; Abe, T.; Auga, J.; Anderson-Teixeira, K.; Basset, Y.; Beckett, S.; et al. Quantitative assessment of plant-arthropod interactions in forest canopies: A plot-based approach. PLoS ONE 2019, 14, e0222119. [Google Scholar] [CrossRef]
- Bondarenko, A.S.; Zamotajlov, A.S.; Belyi, A.I.; Khomitskiy, E.E. Fauna and ecological characteristics of ground beetles (Coleoptera, Carabidae) of the Nature Sanctuaries ”Prichernomorskiy“ and “Tuapsinskiy” (Russia). Nat. Conserv. Res. 2020, 5, 66–85. [Google Scholar] [CrossRef]
- Jackman, J.A.; Nelson, C.R. Diversity and phenology of tumbling flower beetles (Coleoptera: Mordellidae) captured in a Malaise trap. Entomol. News 1995, 106, 97–107. [Google Scholar]
- Leksono, A.S.; Takada, K.; Koji, S.; Nakagoshi, N.; Anggaeni, T.; Nakamura, K. Vertical and seasonal distribution of flying beetles in a suburban temperate deciduous forest collected by water pan trap. Insect Sci. 2005, 12, 199–206. [Google Scholar] [CrossRef]
- Marques, M.I.; Adis, J.; Brizzola dos Santos, G.; Battirola, L.D. Terrestrial arthropods from tree canopies in the Pantanal of Mato Grosso, Brazil. Rev. Bras. Entomol. 2006, 50, 257–267. [Google Scholar] [CrossRef][Green Version]
- Campbell, J.W.; Hanula, J.L. Efficiency of Malaise traps and colored pan traps for collecting flower visiting insects from three forested ecosystems. J. Insect Conserv. 2007, 11, 399–408. [Google Scholar] [CrossRef]
- Hodge, S.; Marshall, S.A.; Oliver, H.; Berry, J.; Marris, J.; Andrew, I. A preliminary survey of the insects collected using mushroom baits in native and exotic New Zealand woodlands. N. Z. Entomol. 2010, 33, 43–54. [Google Scholar] [CrossRef]
- Fagundes, C.K.; Di Mare, R.A.; Wink, C.; Manfio, D. Diversity of the families of Coleoptera captured with pitfall traps in five different environments in Santa Maria, RS, Brazil. Braz. J. Biol. 2011, 71, 381–390. [Google Scholar] [CrossRef]
- Schmeelk, T.C.; Millar, J.G.; Hanks, L.M. Influence of trap height and bait type on abundance and species diversity of cerambycid beetles captured in forests of East-Central Illinois. J. Econ. Entomol. 2016, 109, 1750–1757. [Google Scholar] [CrossRef] [PubMed]
- Alexeev, S.C.; Aleksanov, V.V. Pitfall trap construction affects the efficacy of ground beetle counts. Zool. Zhurnal 2017, 96, 295–304. [Google Scholar] [CrossRef]
- Byk, A.; Węgrzynowicz, P. The Structure and Seasonal Dynamics of Coprophagous Scarabaeoidea (Coleoptera) Communities in Later Developmental Stages of Pine Stands in NW Poland. J. Entomol. Res. Soc. 2015, 17, 39–57. [Google Scholar]
- Kazantsev, S.V.; Egorov, L.V.; Ruchin, A.B. Discovery of Lopheros lineatus (Gorham, 1883) (Coleoptera, Lycidae) in Mordovia, Central Russia. Entomol. Rev. 2019, 99, 656–659. [Google Scholar] [CrossRef]
- MacRae, T.C. Beetle Collecting 101: Fermenting Bait Traps for Collecting Longhorned Beetles. 2015. Available online: https://beetlesinthebush.wordpress.com/2015/12/28/beetle-collecting-101-fermenting-bait-traps-for-collecting-longhorned-beetles/ (accessed on 3 October 2020).
- Hodge, S.; Williams, A. Beetles collected using rotting vegetable baits in a Kent Garden. Entomol. Mon. Mag. 2011, 146, 179–188. [Google Scholar]
- Allemand, R.; Aberlenc, H.-P. Une méthode efficace d’echantillonage de l’entomofaune des frondaisons: Le piège attractif aérien. Bull. Société Entomol. Suisse 1991, 64, 293–305. [Google Scholar]
- Williams, R.N.; Ellis, M.S.; Keeney, G. A bait attractant study of the Nitidulidae (Coleoptera) at Shawnee State Forest in Southern Ohio. Great Lakes Entomol. 1995, 27, 229–234. [Google Scholar]
- MacRae, T.C.; Rice, M.E. Distributional and biological observations on North American Cerambycidae (Coleoptera). Coleopt. Bull. 2007, 61, 227–263. [Google Scholar] [CrossRef]
- Guarnieri, F.G. A survey of longhorned beetles (Coleoptera: Cerambycidae) from Paw Paw, Morgan County, West Virginia. Md. Entomol. 2009, 5, 11–22. [Google Scholar]
- Bardiani, M.; Tini, M.; Carpaneto, G.M.; Audisio, P.; Bussola, E.; Campanaro, A.; Cini, A.; Maurizi, E.; Mason, F.; Peverieri, G.S.; et al. Effects of trap baits and height on stag beetle and lower chafer monitoring: Ecological and conservation implications. J. Insect Conserv. 2017, 21, 157–168. [Google Scholar] [CrossRef]
- Redolfi De Zan, L.; Bardiani, M.; Antonini, G.; Campanaro, A.; Chiari, S.; Mancini, E.; Maura, M.; Sabatelli, S.; Solano, E.; Zauli, A.; et al. Guidelines for the monitoring of Cerambyx cerdo. Nat. Conserv. 2017, 20, 129–164. [Google Scholar] [CrossRef]
- Rukavina, I.; Kostanjšek, F.; Jelaska, S.D.; Pirnat, A.; Šerić, J.L. Distribution and habitat suitability of two rare saproxylic beetles in Croatia—A piece of puzzle missing for South-Eastern Europe. iForest 2018, 11, 765–774. [Google Scholar] [CrossRef]
- Barros, R.C.; Fonseca, M.G.; Jardim, M.T.; Vendramini, V.E.; Damiani, B.C.B.; Julio, C.E.A. Species of Cerambycinae (Insecta, Coleoptera, Cerambycidae) from east Paraná State (Brazil), with new geographic records. Zootaxa 2020, 4845, 001–025. [Google Scholar] [CrossRef]
- Ruchin, A.B.; Egorov, L.V.; Khapugin, A.A. Seasonal activity of Coleoptera attracted by fermental crown traps in forest ecosystems of Central Russia. Ecol. Quest. 2021, 32, 37–53. [Google Scholar] [CrossRef]
- Dvořák, L.; Dvořáková, K.; Oboňa, J.; Ruchin, A.B. Selected Diptera families caught with beer traps in the Republic of Mordovia (Russia). Nat. Conserv. Res. 2020, 5, 65–77. [Google Scholar] [CrossRef]
- Ruchin, A.B.; Egorov, L.V. The beetles (Insecta: Coleoptera) of Smolny National Park (based on insect collecting by fermental crown traps in 2020). Sci. Proc. State Nat. Reserve Prisursky 2020, 35, 221–225. (In Russian) [Google Scholar]
- Ruchin, A.B.; Egorov, L.V.; Khapugin, A.A.; Vikhrev, N.E.; Esin, M.N. The use of simple crown traps for the insects collection. Nat. Conserv. Res. 2020, 5, 87–108. [Google Scholar] [CrossRef]
- MacRae, T.C. Review of the genus Purpuricenus Dejean (Coleoptera: Cerambycidae) in North America. Pan-Pac. Entomol. 2000, 76, 137–169. [Google Scholar]
- Philips, T.K.; Whorrall, K.A.; Gearner, O.M.; Huchet, J.B. A new genus of spider beetle (Coleoptera, Ptinidae) from western Peru. ZooKeys 2020, 934, 81–91. [Google Scholar] [CrossRef]
- Ruchin, A.B.; Egorov, L.V. Beetles (Insecta, Coleoptera), collected using fermental crown trap in the Republic of Mordovia. Report. 1. Mordovia State Nature Reserve. Sci. Proc. State Nat. Reserve Prisursky 2018, 33, 209–215. (In Russian) [Google Scholar]
- Bouchard, P.; Bousquet, Y.; Davies, A.E.; Alonso-Zarazaga, M.A.; Lawrence, J.F.; Lyal, C.H.C.; Newton, A.F.; Ried, C.A.M.; Schmitt, M.; Ślipiński, S.A.; et al. Family-group names in Coleoptera (Insecta). ZooKeys 2011, 88, 1–972. [Google Scholar] [CrossRef]
- Bouchard, P.; Bousquet, Y. Additions and corrections to “Family-group names in Coleoptera (Insecta)”. ZooKeys 2020, 922, 65–139. [Google Scholar] [CrossRef] [PubMed]
- Löbl, I.; Smetana, A. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 7: Curculionoidea I; Apollo Books: Stenstrup, Denmark, 2011; p. 373. [Google Scholar]
- Löbl, I.; Smetana, A. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 8: Curculionoidea II; Apollo Books: Stenstrup, Denmark, 2013; p. 707. [Google Scholar]
- Löbl, I.; Löbl, D. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 2/1. Revised and Updated Version. Hy-Drophiloidea–Staphylinoidea; Brill: Leiden, The Netherlands; Boston, MA, USA, 2015; p. 1702. [Google Scholar]
- Löbl, I.; Löbl, D. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 3. Revised and Updated Version. Scara-Baeoidea–Scirtoidea–Dascilloidea–Buprestoidea–Byrrhoidea; Brill: Leiden, The Netherlands; Boston, MA, USA, 2016; p. 983. [Google Scholar]
- Löbl, I.; Löbl, D. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 1. Revised and Updated Version. Archostemata–Adephaga–Myxophaga; Brill: Leiden, The Netherlands; Boston, MA, USA, 2017; p. 1443. [Google Scholar]
- Iwan, D.; Löbl, I. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 5. Revised and Updated Second Edition. Tenebrionoidea; Brill: Leiden, The Netherlands; Boston, MA, USA, 2020; p. 945. [Google Scholar]
- Danilevsky, M. (Ed.) Catalogue of Palaearctic Coleoptera. Vol. 6/1. Updated and Revised Second Edition. Chrysomeloidea I (Vesperidae, Disteniidae, Cerambycidae); Brill: Leiden, The Netherlands; Boston, MA, USA, 2020; p. 712. [Google Scholar]
- Robertson, J.; Ślipiński, A.; Moulton, M.; Shockley, F.W.; Giorgi, A.; Lord, N.P.; McKenna, D.D.; Tomaszewska, W.; Forrester, J.; Miller, K.B.; et al. Phylogeny and classification of Cucujoidea and the recognition of a new superfamily Coccinelloidea (Coleoptera: Cucujiformia). Syst. Entomol. 2015, 40, 745–778. [Google Scholar] [CrossRef]
- Alonso-Zarazaga, M.A.; Barrios, H.; Borovec, R.; Bouchard, P.; Caldara, R.; Colonnelli, E.; Gültekin, L.; Hlaváč, P.; Korotyaev, B.; Lyal, C.H.C.; et al. Cooperative Catalogue of Palaearctic Coleoptera Curculionoidea. Monogr. Electrón. SEA 2017, 8, 1–729. [Google Scholar]
- Löbl, I.; Smetana, A. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 4. Elateroidea–Derodontoidea–Bos-trichoidea–Lymexyloidea–Cleroidea–Cucujoidea; Apollo Books: Stenstrup, Denmark, 2007; p. 935. [Google Scholar]
- Löbl, I.; Smetana, A. (Eds.) Catalogue of Palaearctic Coleoptera. Vol. 6: Chrysomeloidae; Apollo Books: Stenstrup, Denmark, 2010; p. 924. [Google Scholar]
- Bousquet, Y. Litteratura Coleopterologica (1758–1900): A guide to selected books related to the taxonomy of Coleoptera with publication dates and notes. ZooKeys 2016, 583, 1–776. [Google Scholar] [CrossRef]
- Red Data Book of Vladimir Region; TPS Publ.: Tambov, Russia, 2018; p. 432. (In Russian)
- Red Data Book of the Nizhny Novgorod Region. Vol. 1: Animals; DEKOM Publ.: Nizhny Novgorod, Russia, 2014; p. 448. (In Russian)
- Red Data Book of Penza Region. Vol. 2. Animals; E. A. Bolkhovitinov Publ.: Voronezh, Russia, 2019; p. 264. (In Russian)
- Red Data Book of the Republic of Mordovia. Vol. 2: Animals; Mordovia Book Publ.: Saransk, Russia, 2015; p. 336. (In Russian)
- Red Data Book of Ryazan Region; Golos Gubernii Publ.: Ryazan, Russia, 2011; p. 626. (In Russian)
- Red Data Book of Saratov Region; Papirus Ltd.: Saratov, Russia, 2021; p. 496. Available online: http://redbook.ch56058.tmweb.ru/#page=300 (accessed on 3 October 2020). (In Russian)
- Red Data Book of the Tambov Region; Yulis Publ.: Tambov, Russia, 2012; p. 352. (In Russian)
- Red Data Book of Ulyanovsk Region; Buki Vedi Publ.: Ulyanovsk, Russia, 2015; p. 550. (In Russian)
- List of Objects of the Animal World Listed in the Red Book of the Russian Federation. 2020. Available online: https://minjust.consultant.ru/documents/45937 (accessed on 11 May 2020). (In Russian).
- Basset, Y. Invertebrates in the canopy of tropical rain forests. How much do we really know? Plant. Ecol. 2001, 153, 87–107. [Google Scholar] [CrossRef]
- McCaig, T.; Sam, L.; Nakamura, A.; Stork, N.E. Is insect vertical distribution in rainforests better explained by distance from the canopy top or distance from the ground? Biodivers. Conserv. 2020, 29, 1081–1103. [Google Scholar] [CrossRef]
- Lowman, M.D. Canopy research in the twenty-first century: A review of Arboreal Ecology. Trop. Ecol. 2009, 50, 125–136. [Google Scholar]
- Neves, F.S.; Araújo, L.S.; Espírito-Santo, M.M.; Fagundes, M.; Fernandes, G.W.; Sanchez-Azofeifa, G.A.; Quesada, M. Canopy herbivory and insect herbivore diversity in a dry forest–savanna transition in Brazil. Biotropica 2010, 42, 112–118. [Google Scholar] [CrossRef]
- Nadkarni, N.M.; Parker, G.G.; Lowman, M.D. Forest canopy studies as an emerging field of science. Ann. For. Sci. 2011, 68, 217. [Google Scholar] [CrossRef]
- Viana-Junior, A.B.; Quijano-Cuervo, L.G.; Ferreira, J.C.; do Nascimento Reis, R.R.; dos Santos, I.A.; Martins, M.B. Collecting arboreal arthropods: A technique for sampling plant-inhabiting arthropod communities in a tropical forest understory. Entomol. Exp. Et Appl. 2021, 169, 312–321. [Google Scholar] [CrossRef]
- Sanchez-Gracia, A.; Vieira, F.G.; Almeida, F.C.; Rozas, J. Comparative genomics of the major chemosensory gene families in Arthropods. In Encyclopedia of Life Sciences; John Wiley & Sons, Ltd.: Chichester, UK, 2011. [Google Scholar]
- Engsontia, P.; Sangket, U.; Chotigeat, W.; Satasook, C. Molecular Evolution of the Odorant and Gustatory Receptor Genes in Lepidopteran Insects: Implications for Their Adaptation and Speciation. J. Mol. Evol. 2014, 79, 21–39. [Google Scholar] [CrossRef] [PubMed]
- Xu, W. How do moth and butterfly taste?—Molecular basis of gustatory receptors in Lepidoptera. Insect Sci. 2020, 27, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- Kent, L.; Robertson, H. Evolution of the sugar receptors in insects. BMC Evolutionary Biology 2009, 9, 41. [Google Scholar] [CrossRef]
- Tooming, E.; Merivee, E.; Must, A.; Luik, A.; Williams, I.H. Antennal sugar sensitivity in the click beetle Agriotes obscurus. Physiol. Entomol. 2012, 37, 345–353. [Google Scholar] [CrossRef]
- Stensmyr, M.C.; Larsson, M.C.; Bice, S.; Hansson, B.S. Detection of fruit- and flower-emitted volatiles by olfactory receptor neurons in the polyphagous fruit chafer Pachnoda marginata (Coleoptera: Cetoniinae). J. Comp. Physiol. A 2001, 187, 509–519. [Google Scholar] [CrossRef]
- Leroy, P.D.; Heuskin, S.; Sabri, A.; Verheggen, F.J.; Farmakidis, J.; Lognay, G.; Thonart, P.; Wathelet, J.-P.; Brostaux, Y.; Haubruge, E. Honeydew volatile emission acts as a kairomonal message for the Asian lady beetle Harmonia axyridis (Coleoptera: Coccinellidae). Insect Sci. 2012, 19, 498–506. [Google Scholar] [CrossRef]
- Kirmse, S.; Chaboo, C.S. Flowers are essential to maintain high beetle diversity (Coleoptera) in a Neotropical rainforest canopy. J. Nat. Hist. 2020, 54, 1661–1696. [Google Scholar] [CrossRef]
- Tauzin, P. Ethologie et chorologie de Protaetia (Liocola) lugubris Herbst, 1786 sur le territoire français (Coleoptera, Cetoniidae, Cetoniinae, Cetoniini). Cetoniimania 2006, 3, 4–38. [Google Scholar]
- Oleksa, A.; Chybicki, I.J.; Gawronski, R.; Svensson, G.P.; Burczyk, J. Isolation by distance in saproxylic beetles may increase with niche specialization. J. Insects Conserv. 2013, 17, 219–233. [Google Scholar] [CrossRef]
- Urban, P.; Schulze, W. Ein aktueller Nachweis des Marmorierten Rosenkäfers Protaetia marmorata (Fabricius, 1792) in der Senne (Nordrhein-Westfalen) (Mitteilungen zur Insektenfauna Westfalens XXII). Mitt. Der Arb. Westfälischer Entomol. 2017, 33, 15–19. [Google Scholar]
- Kurochkin, A.S. Fauna and bionomy of sap beetles (Coleoptera, Nitidulidae) and kateretid beetles (Coleoptera, Kateretidae) of Krasnosamarskoe forestry farm (Samara Region, Russia). Vestn. Samara Univ. Nat. Sci. Ser. 2007, 8, 120–128. (In Russian) [Google Scholar]
- Alekseev, V.I.; Nikitsky, N.B. Rare and new for the fauna of the Baltic States Beetles (Coleoptera) from the Kaliningrad region. Acta Zool. Litu. 2008, 18, 254–259. [Google Scholar] [CrossRef]
- Lasoń, A.; Holly, M. Glischrochilus grandis Tournier, 1872—New species of beetle for the Polish fauna and new data on the occurrence of genus Glischrochilus Reitter, 1873 (Coleoptera: Nitidulidae: Cryptarchinae). Acta Entomol. Sil. 2015, 23, 1–4. [Google Scholar]
- Nikitsky, N.B.; Osipov, I.N.; Chemeris, M.V.; Semenov, V.B.; Gusakov, A.A. The beetles of the Prioksko-Terrasny Biosphere Reserve—Xylobiontes, mycetobiontes and Scarabaeidae. Arch. Zool. Mus. Mosc. State Univ. 1996, XXXVI, 1–197. (In Russian) [Google Scholar]
- Nikitsky, N.B.; Mamontov, S.N.; Vlasenko, A.S. New data of beetles from Tula abatis forests (Coleoptera: Nitidulidae–Scolytidae) collected in window traps. Bull. Mosc. Soc. Nat. Biol. Ser. 2016, 121, 25–37. (In Russian) [Google Scholar]
- Clayhills, T. Coleoptera species new to Finland (1) (Coleoptera). Koleopterol. Rundsch. 2011, 81, 311–319. [Google Scholar]
- Tauzin, P. Chorologie et éco-éthologie de Protaetia (Potosia) fieberi Kraatz 1880 en France (Coleoptera, Cetoniinae, Cetoniini). Cetoniimania 2007, 3, 115–146. [Google Scholar]
- Ruchin, A.B.; Egorov, L.V.; Sazhnev, A.S.; Polumordvinov, O.A.; Ishin, R.N. Present distribution of Protaetia fieberi (Kraatz, 1880) (Insecta, Coleoptera, Scarabaeidae) in the European part of Russia. Biharean Biol. 2019, 13, 12–16. [Google Scholar]
- Bílý, S.; Mehl, O. Longhorn Beetles (Coleoptera, Cerambycidae) of Fennoscandia and Denmark; Brill: Leiden, The Netherlands, 1989; p. 200. [Google Scholar]
- Avgin, S.S.; Antonini, G.; Lasoń, A.; Jansson, N.; Abacigil, T.Ö.; Varli, S.V.; De Biase, A.; Audisio, P. New data on distribution, ecology, and taxonomy of Turkish Nitidulidae (Coleoptera). Turk. J. Zool. 2015, 29, 314–322. [Google Scholar] [CrossRef]
- Oude, J.E. Naamlijst van de glanskevers van Nederland en het omliggende gebied (Coleoptera: Nitidulidae and Brachypteridae). Ned. Faun. Meded. 1999, 8, 11–32. [Google Scholar]
- Ruchin, A.B.; Egorov, L.V. Fauna of longicorn beetles (Coleoptera: Cerambycidae) of Mordovia. Russ. Entomol. J. 2018, 27, 161–177. [Google Scholar] [CrossRef]
- Cherepanov, A.I. The Longhorn Beetles of Northern Asia (Prioninae, Disteniinae, Lepturinae, Aseminae); Nauka Publ.: Novosibirsk, Russia, 1979; p. 472. [Google Scholar]
- Gutowski, J.M.; Ługowoj, J.; Maciejewski, K.H. Leptura thoracica Creutzer, 1799 (Coleoptera: Cerambycidae) in Poland. Wiad. Entomol. 1994, 13, 157–165. (In Polish) [Google Scholar]
- Sama, G. Atlas of the Cerambycidae of Europe and the Mediterranean Area. Vol. 1. Northern, Western, Central and Eastern Europe British Isles and Continental Europe from France (Excl. Corsica) to Scandinavia and Urals; Kabourek: Zlín, Czechia, 2002; p. 173. [Google Scholar]
- Karpiński, L.; Szczepański, W.T.; Boldgiv, B.; Walczak, M. New data on the longhorn beetles of Mongolia with particular emphasis on the genus Eodorcadion Breuning, 1947 (Coleoptera, Cerambycidae). ZooKeys 2018, 739, 107–150. [Google Scholar] [CrossRef]
- Danilevsky, M.L.; Ruchin, A.B.; Egorov, L.V. Mass collection of two rare longicorn-species (Coleoptera, Cerambycidae) in Central Russia. Humanit. Space 2019, 8, 1179–1183. [Google Scholar]
- Karolyi, F.; Gorb, S.N.; Krenn, H.W. Trapping pollen by the moist mouth: Structure and function of the mouthparts in the flower visiting Cetonia aurata (Scarabeidae, Coleoptera). Arthropod Plant. Interact. 2009, 3, 1–8. [Google Scholar] [CrossRef]
- Landvik, M.; Niemelä, P.; Roslin, T. Mother knows the best mould: An essential role for non-wood dietary components in the life cycle of a saproxylic scarab beetle. Oecologia 2016, 182, 163–176. [Google Scholar] [CrossRef]
- Strassen, R. Zur Oekologie des Velleius dilatatus Fabricius, eines als Raumgast bei Vespa crabro Linnaeus lebenden Staphyliniden (Insecta, Coleoptera). Z. Für Morphol. Und Ökologie Der Tiere 1957, 46, 243–292. (In German) [Google Scholar] [CrossRef]
- Konwerski, S.; Melke, A.; Miłkowski, M.; Ruta, R.; Sienkiewicz, P. Nowe stanowiska Velleius dilatatus (Fabricius, 1787) w Polsce (Coleoptera: Staphylinidae) oraz uwagi o jego ochronie. Chrońmy Przyr. Ojczystą 2010, 66, 111–115. (In Polish) [Google Scholar]
- Burakowski, B.; Mroczkowski, M.; Stefańska, J. Chrząszcze Coleoptera, Scarabaeoidea, Dascilloidea, Byrrhoidea i Parnoidea. Kat. Fauny Pol. 1983, 23, 9. [Google Scholar]
- Parmentier, T.; Dekoninck, W.; Wenseleers, T. A highly diverse microcosm in a hostile world: A review on the associates of red wood ants (Formica rufa group). Insectes Sociaux 2014, 61, 229–237. [Google Scholar] [CrossRef]
- Valdés, E.M.E.; Aldana, L.L.; Figueroa, B.; Gutiérrez, M.O.; Hernández, R.M.C.; Chavelas, M.T. Trapping of Scyphophorus acupunctatus (Coleoptera: Curculionidae) with two natural baits in a field of Polianthes tuberosa (Liliales: Agavaceae) in the state of Morelos, México. Fla. Entomol. 2005, 88, 338–340. [Google Scholar] [CrossRef]
- Yang, K.-L.; Wen, X.-J.; Zhang, G.-Y.; Wen, J.-B. Evaluation of trap designs and food attractants for trapping Eucryptorrhynchus scrobiculatus (Coleoptera: Curculionidae). Biocontrol Sci. Technol. 2019, 29, 28–43. [Google Scholar] [CrossRef]
- Bardiani, M.; Chiari, S.; Maurizi, E.; Tini, M.; Toni, I.; Zauli, A.; Campanaro, A.; Carpaneto, G.M.; Audisio, P. Guidelines for the monitoring of Lucanus cervus. Nat. Conserv. 2017, 20, 37–78. [Google Scholar] [CrossRef]
- Mudge, A.D.; Orozco, J.; Philips, T.K.; Antoine, P. The cetoniine fauna of the Upper Guinean forests and savannas of Ghana (Coleoptera: Scarabaeidae: Cetoniinae). Terr. Arthropod Rev. 2012, 5, 113–174. [Google Scholar] [CrossRef]
- Correa, C.M.A.; Puker, A.; Lara, M.A.; Rosa, C.S.; Korasaki, V. Evaluation of baits for trapping of Neotropical flower chafer beetles (Coleoptera: Scarabaeoidea: Cetoniinae). Entomol. Sci. 2019, 22, 365–372. [Google Scholar] [CrossRef]
- Wong, J.C.H.; Hanks, L.M. Influence of fermenting bait and vertical position of traps on attraction of cerambycid beetles to pheromone lures. J. Econ. Entomol. 2016, 109, 2145–2150. [Google Scholar] [CrossRef]
- Ausden, M.; Drake, M. Invertebrates. In Ecological Census Techniques, a Handbook, 2nd ed.; Sutherland, W.J., Ed.; Cambridge University Press: Cambridge, UK, 2006; pp. 214–249. [Google Scholar]
- Laaksonen, J.; Laaksonen, T.; Itämies, J.; Rytkönen, S.; Välimäki, P. A new efficient bait-trap model for Lepidoptera surveys—The «Oulu» model. Entomol. Fenn. 2006, 17, 153–160. [Google Scholar] [CrossRef]
- Makarkin, V.N.; Ruchin, A.B. New data on Neuroptera and Raphidioptera of Mordovia (Russia). Kavk. Entomol. Bull. 2019, 15, 147–157. (In Russian) [Google Scholar] [CrossRef]




| Region | 2018 | 2019 | 2020 | Total |
|---|---|---|---|---|
| Republic of Mordovia | 83 1750 | 266 10,617 | 226 10,901 | 575 23,268 |
| Penza region | 0 0 | 18 495 | 86 4968 | 104 5463 |
| Ulyanovsk region | 0 0 | 20 278 | 32 1767 | 52 2045 |
| Nizhny Novgorod region | 0 0 | 13 265 | 29 880 | 42 1145 |
| Vladimir region | 0 0 | 0 0 | 21 143 | 21 143 |
| Ryazan region | 0 0 | 0 0 | 19 850 | 19 850 |
| Saratov region | 0 0 | 0 0 | 4 60 | 4 60 |
| Tambov region | 0 0 | 0 0 | 3 295 | 3 295 |
| Total | 83 1750 | 317 11,655 | 420 19,864 | 820 33,269 |
| Species | Red Data Book of Russia | Red Data Book | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Vladimir Region | Ryazan Region | Republic of Mordovia | Penza Region | Nizhny Novgorod Region | Ulyanovsk Region | Saratov Region | Tambov Region | ||
| Carabidae | |||||||||
| Lebia marginata (Geoffroy, 1785) | – | – | – | 1 (2) * | – | – | – | – | – |
| Staphylinidae | |||||||||
| Quedius dilatatus (Fabricius, 1787) | – | – | – | 57 (10) | – | – | 10 (5) | – | – |
| Silphidae | |||||||||
| Dendroxena quadrimaculata (Scopoli, 1771) | – | 0 (5) | – | 10 (3) | – | – | – | – | – |
| Lucanidae | |||||||||
| Lucanus cervus (Linnaeus, 1758) | + | 0 (1) | – | 0 (3) | 0 (25) | 0 (2) | 3 (43) | 0 (30) | 0 (7) |
| Scarabaeidae | |||||||||
| Gnorimus variabilis (Linnaeus, 1758) | – | 0 (6) | 0 (2) | 29 (6) | 2 (6) | – | 0 (9) | 0 (10) | 0 (2) |
| Osmoderma barnabita Motschulsky, 1845 | + | 0 (3) | 0 (9) | 3 (4) | 0 (7) | 0 (11) | 0 (7) | 0 (5) | 0 (1) |
| Protaetia fieberi (Kraatz, 1880) | + | 7 (4) | 5 (–) | 125 (12) | 48 (6) | 11 (–) | 40 (–) | 2 (9) | 1 (–) |
| Protaetia marmorata (Fabricus, 1792) | – | – | 15 (7) | – | – | – | – | – | – |
| Protaetia speciosissima (Scopoli, 1786) | + | 2 (–) | 1 (2) | 25 (8) | 20 (11) | 4 (1) | 9 (25) | 1 (14) | 1 (4) |
| Elateridae | |||||||||
| Elater ferrugineus Linnaeus, 1758 | + | – | – | 6 (2) | – | – | 1 (–) | 0 (1) | – |
| Coccinellidae | |||||||||
| Adalia bipunctata (Linnaeus, 1758) | – | – | – | 1 (4) | – | – | – | – | |
| Cerambycidae | |||||||||
| Leptura thoracica (Creutzer, 1799) | – | – | 6 (2) | – | – | – | – | – | – |
| Purpuricenus globulicollis Dejean, 1839 | – | – | – | – | 1 (1) | – | – | – | 0 (1) |
| Purpuricenus kaehleri (Linnaeus, 1758) | – | – | 1 (1) | 31 (2) | – | – | – | – | 1 (4) |
| Necydalis major Linnaeus, 1758 | – | – | 0 (7) | 32 (8) | – | – | – | 0 (8) | 0 (6) |
| Leptura aurulenta Fabricius, 1793 | – | – | – | 3 (1) | – | – | – | – | – |
| Aromia moschata (Linnaeus, 1758) | – | – | – | 30 (10) | – | – | – | – | 0 (8) |
| Cleridae | |||||||||
| Allonyx quadrimaculatus (Schaller, 1783) | – | – | – | 2 (1) | – | 0 (2) | – | – | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruchin, A.B.; Egorov, L.V.; Khapugin, A.A. Usage of Fermental Traps for the Study of the Species Diversity of Coleoptera. Insects 2021, 12, 407. https://doi.org/10.3390/insects12050407
Ruchin AB, Egorov LV, Khapugin AA. Usage of Fermental Traps for the Study of the Species Diversity of Coleoptera. Insects. 2021; 12(5):407. https://doi.org/10.3390/insects12050407
Chicago/Turabian StyleRuchin, Alexander B., Leonid V. Egorov, and Anatoliy A. Khapugin. 2021. "Usage of Fermental Traps for the Study of the Species Diversity of Coleoptera" Insects 12, no. 5: 407. https://doi.org/10.3390/insects12050407
APA StyleRuchin, A. B., Egorov, L. V., & Khapugin, A. A. (2021). Usage of Fermental Traps for the Study of the Species Diversity of Coleoptera. Insects, 12(5), 407. https://doi.org/10.3390/insects12050407








