Abstract
This overview of reviews (i.e., an umbrella review) is designed to reappraise the validity of systematic reviews (SRs) and meta-analyses related to the performance of Aspergillus PCR tests for the diagnosis of invasive aspergillosis in immunocompromised patients. The methodological quality of the SRs was assessed using the AMSTAR-2 checklist; the quality of the evidence (QOE) within each SR was appraised following the GRADE approach. Eight out of 12 SRs were evaluated for qualitative and quantitative assessment. Five SRs evaluated Aspergillus PCR on bronchoalveolar lavage fluid (BAL) and three on blood specimens. The eight SRs included 167 overlapping reports (59 evaluating PCR in blood specimens, and 108 in BAL), based on 107 individual primary studies (98 trials with a cohort design, and 19 with a case−control design). In BAL specimens, the mean sensitivity and specificity ranged from 0.57 to 0.91, and from 0.92 to 0.97, respectively (QOE: very low to low). In blood specimens (whole blood or serum), the mean sensitivity ranged from 0.57 to 0.84, and the mean specificity from 0.58 to 0.95 (QOE: low to moderate). Across studies, only a low proportion of AMSTAR-2 critical domains were unmet (1.8%), demonstrating a high quality of methodological assessment. Conclusions. Based on the overall methodological assessment of the reviews included, on average we can have high confidence in the quality of results generated by the SRs.
1. Introduction
Invasive aspergillosis (IA) is a life-threatening opportunistic invasive mold disease of the immunocompromised host [1] and, as such, requires early diagnosis and prompt systemic antifungal treatment to enhance survival [2]. Consequently, there is an urgent need for new diagnostic tools and optimization of the use of existing tests individually, or in combination, to better complement antifungal treatment [3,4]. Aspergillus polymerase chain reaction (PCR) testing of blood and respiratory samples has recently been included in the second revision of the EORTC/MSGERC definitions for classifying invasive fungal disease [5,6].
Due to the large number of published papers on Aspergillus PCR (>2500 papers to date (PubMed search for Aspergillus PCR)), there is a significant amount of clinical data available, and a number of systematic reviews (SRs) and meta-analyses on the performance of PCR for the diagnosis of IA have been published [7,8]. Their conclusions show extensive heterogeneity among studies in terms of design, conduct, and reporting. The current study is an overview of SRs, termed an umbrella review, aimed at reappraising the validity of the conclusions of SRs and the diagnostic accuracy of PCR-based tests on blood and respiratory specimens for the diagnosis of IA in immunocompromised patients published in meta-analyses. The decision to perform this overview was based on the continuing importance of the review question, and on the availability of new data from SRs/meta-analyses. Increasing the number of studies can improve precision of effect estimates, allowing additional comparisons or subgroup analyses to be performed [9]. An umbrella review is a review of reviews, and only considers other systematic reviews as eligible for inclusion (in other words, the unit of analysis for overview of reviews is the systematic review/meta-analysis and not individual patient data). An umbrella review collects evidence from multiple existing reviews and provides perhaps the highest levels of evidence. In this umbrella review, we have also applied new review methods such as the AMSTAR-2 tool, and a GRADE assessment, with the aim of enhancing the existing results in terms of the certainty of the review’s findings [10].
2. Material and Methods
The protocol of this overview of reviews is available on the International Prospective Register of Systematic Reviews (PROSPERO) with the registration number CRD42021259625. There were no amendments from the pre-specified criteria reported in the protocol throughout the review process. The results are reported according to Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies (PRISMA-DTA) [11].
2.1. Inclusion and Exclusion Criteria
This overview includes SRs evaluating clinical trials (i) aimed at comparing the performance of PCR tests with reference to the consensus definitions of IA published by the European Organization for Research and Treatment of Cancer/Mycoses Study Group (EORTC/MSGERC [6,12,13]); (ii) reporting data on false-positive, true-positive, false-negative, and true-negative results for the diagnostic tests under investigation separately; and (iii) evaluating the test(s) in cohorts of patients from a relevant clinical population, defined as a group of individuals at high risk for IA.
2.2. Search Strategy
Relevant studies in three bibliographic databases (Embase, PubMed, and Cochrane library) were searched up to March 2023. The searches were performed without language restriction using Medical Subjects Heading: (“Aspergillosis/Aspergillus” or “Invasive fungal Infection”) AND “diagnosis” AND (“systematic review” OR “meta-analysis). Furthermore, reference lists of the reviews were checked to identify potentially eligible studies not captured by the electronic literature search.
2.3. Study Selection and Data Extraction
All titles were screened by two independent assessors (MC and LW). Eligibility assessment was initially based on the title or abstract, and on the full text, when required. Full texts of potentially eligible articles were obtained and assessed independently by two reviewers (MC and LW) against the stated inclusion criterion. The study selection decision of each reviewer was compared for concordance. The two assessors also independently extracted quantitative and qualitative data from each selected study, with disagreements resolved through discussion and through the opinion of a third reviewer (RB). Findings are presented in tabular format with Table S1. Tabulation of results includes the following: first author name and year of publication, clinical setting, number and design of studies included in the SR, index test and reference standard, subgroup analyses, and the main conclusion of the review as reported by authors.
2.4. Assessment of Methodological Quality of Systematic Reviews
We used the AMSTAR-2 critical appraisal checklist, a tool for SRs that includes randomized or non-randomized studies, or both [14]. The tool is suitable for reviews of intervention, but can also be adapted to explore SRs of diagnostic tests. It includes 16 domains, of which 7 are considered critical, relating to the research question, review design, search strategy, study selection, data extraction, justification for excluded studies, description of included studies, risk of bias, sources of funding, meta-analysis, heterogeneity, publication bias, and conflicts of interest. Two review authors (MC, LW) independently assessed the quality of evidence and the methodological quality of the SRs. We resolved discrepancies through discussion or, if needed, through a third review author (RB). Reviews were not excluded based on AMSTAR 2 ratings, but the ratings were considered in interpretation of the results.
2.5. Summary of the Evidence, Subgroups Analisis, and Appraisal of the Quality of Evidence
For the quantitative synthesis, the sensitivity and specificity were reported (when available) with the 95% confidence intervals (CIs), as stated in the individual reviews. Where available, other measures of diagnostic accuracy such as predictive values, likelihood ratios, and diagnostic odds ratio were reported. Moreover, the impacts of several variables on the diagnostic performance of PCR were evaluated, as reported in the SRs by subgroups analysis or meta-regression. To this end, we focused on differences in study design (e.g., cohort vs. case−control studies), patient selection (e.g., hematology vs. other at-risk patients), variations in the index test and reference standard, and the use of antifungal agents.
The quality of evidence was appraised following the GRADE approach (Grades of Recommendation, Assessment, Development, and Evaluation) [15,16]. Whenever available, the grading of the quality of evidence reported in the SRs was considered to define the quality of evidence. When grading of evidence was not reported by the authors of the study, the GRADE approach was applied based on the information available in the individual review. Studies can be downgraded because of concerns over the risk of bias, indirectness (applicability of the results to the question), inconsistency (heterogeneity between study results), imprecision (low number of studies and/or participants), and publication bias [16]. The GRADE approach has four levels of certainty: very low (the true effect is probably markedly different from the estimated effect), low (the true effect might be markedly different from the estimated effect), moderate (the true effect is probably close to the estimated effect), and high (the true effect is similar to the estimated effect) [16].
3. Results
The electronic and manual search retrieved 118 references. The Preferred Reporting Items for SRs and Meta-Analyses (PRISMA) flow diagram is reported in Figure 1.
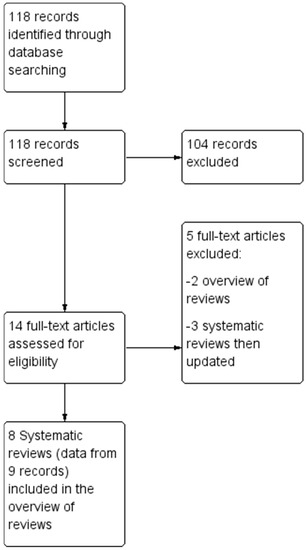
Figure 1.
Flow chart of study selection process.
At the first stage of screening titles and abstracts, 14 references were selected [7,8,17,18,19,20,21,22,23,24,25,26,27,28]. After the full texts were examined with regards to eligibility (i.e., inclusion and exclusion criteria), twelve records were considered for this umbrella review [17,18,19,20,21,22,23,24,25,26,27,28] but, to avoid the inclusion of duplicate records, data were extracted from nine records (eight SRs), with the exclusion of three reviews [17,18,19], due to the availability of updated versions which were included [23,26]. Two previous overviews of reviews were also excluded [7,8].
3.1. Description of the Studies
Of the eight SRs included in the overview, five evaluated PCR on bronchoalveolar lavage fluid (BAL) specimens, with three on blood specimens. Four reviews focused exclusively on PCR, while four compared the diagnostic performance of PCR test to galactomannan and/or beta-d glucan [22,23,24,25]. The eight SRs included 167 overlapping reports (59 evaluating PCR in blood specimens, and 108 in BAL), based on 107 individual primary studies. The 107 primary studies included 98 trials with a cohort design (47 with blood specimens, 41 with BAL specimens), and 19 with a case−control design (3 with blood specimens, 16 with BAL specimens). The main characteristics of the SRs included are summarized in Table 1.

Table 1.
Main characteristics of the systematic reviews (SRs) on Aspergillus PCR.
3.2. Methodological Quality of the SRs with the AMSTAR-2
Three of the SRs met all the AMSTAR-2 methodological requirements, and three SRs partially met one or two of the methodologic requirements, while fully meeting the rest (Table 2). Two SRs had one unmet methodologic requirement, plus between two and six partially met requirements. Of the 120 methodological requirements assessed (Domain 10 excluded) across all eight studies, a total of 12 (10%) methodological requirements were only partially met, with only 1.7% unmet and 88.3% (106/120) of domains fully met. Of the seven critical domains (see footnote of Table 2), only one (1.8% of critical domains across all the SRs) item was judged to have been unmet, and six (10.7%) partially met, leaving 87.5% of critical domains fully met across all SRs. Based on the overall methodological assessment and considering an unmet requirement as an indicator of lower confidence, there is high confidence in the results generated from the majority (75%) of SRs included in the overview [14]. For this overview, one item (sources of funding for the studies included in the review) was not included, given that the large majority of primary studies evaluated were in-house PCR tests and the process of SR and meta-analysis was independent of financial support.

Table 2.
Methodological quality of Systematic reviews assessed with the AMSTAR-2 tool.
Amstar-2 domains:
- Did the research questions and inclusion criteria for the review include the components of PICO (patients, index test, comparator, accuracy as outcome)?
- Did the report of the review contain an explicit statement that the review methods were established prior to the conduct of the review and did the report justify any significant deviations from the protocol?
- Did the review authors explain their selection of the study designs for inclusion in the review?
- Did the review authors use a comprehensive literature search strategy?
- Did the review authors perform study selection in duplicate?
- Did the review authors perform data extraction in duplicate?
- Did the review authors provide a list of excluded studies and justify the exclusions?
- Did the review authors describe the included studies in adequate detail?
- Did the review authors use a satisfactory technique for assessing the risk of bias (RoB) in individual studies that were included in the review?
- Did the review authors report on the sources of funding for the studies included in the review?
- If meta-analysis was performed did the review authors use appropriate methods for statistical combination of results?
- If meta-analysis was performed, did the review authors assess the potential impact of RoB in individual studies on the results of the meta-analysis or other evidence synthesis?
- Did the review authors account for RoB in individual studies when interpreting/discussing the results of the review?
- Did the review authors provide a satisfactory explanation for, and discussion of, any heterogeneity
- If they performed quantitative synthesis did the review authors carry out an adequate investigation of publication bias (small study bias) and discuss its likely impact on the results of the review?
- Did the review authors report any potential sources of conflict of interest, including any funding they received for conducting the review?
Although AMSTAR 2 consists of 16 items, critical domains include items 2, 4, 7, 9, 11, 13, and 15. Item 10 was not assessed (na).
3.3. Summary of the Performance of PCR for the Diagnosis of Invasive Aspergillosis
Mean sensitivity and specificity and 95% CIs (if available) of PCR as calculated for each meta-analysis are summarized in Table 3. In BAL specimens, the mean sensitivity ranged from 0.57 to 0.91, and the mean specificity from 0.92 to 0.97. The level of certainty of these findings for studies within individual SRs was considered low to very low, mostly due to the risk of bias (selection bias in case−control studies) and inconsistency (due to heterogeneity) in primary studies included in the reviews.

Table 3.
Summary of findings table (SOT), including the diagnostic accuracy of PCR for Aspergillus in BAL and blood specimens. Studies meeting/partially meeting all AMSTAR-2 requirements in bold text.
In blood specimens (whole blood or serum), the mean sensitivity ranged from 0.57 to 0.84, and the mean specificity from 0.58 to 0.95. The results of two studies that performed subgroup analysis according to the number of specimens required to define the test positive (a single positive specimen or ≥2 consecutive positive specimens) were consistent [23,26]. In these two SRs, mean sensitivity values were 0.79 and 0.84 for 1 positive test, and 0.57 and 0.59 for ≥2 positive tests, and mean specificity values were 0.79 for 1 positive test, and 0.93 and 0.95 for ≥2 positive tests. Lower specificity (0.58) was found in the review of PCR performance in pediatric patients [25]. We graded the level of certainty of these findings as being low to moderate. The overall quality of the evidence according to the GRADE assessment was very low for three SRs, low for four SRs, and moderate for one SR (Table 3).
3.4. Other Measures of Diagnostic Performance and Subgroup Analyses
In BAL specimens, positive and negative likelihood ratios from three reviews [20,21,28] ranged from 10.4 to 11.9 and 0.10 to 0.27, respectively. In one review, positive and negative predictive values were 81.6 and 97.7 in the overall analysis [24]. DOR from three reviews ranged from 44 to 243, reflecting the heterogeneity of sensitivity and specificity values reported in the primary studies included in the reviews [21,22,28]. In blood specimens, data from two reviews [23,26] with similar prevalence of IA showed consistent results for predictive values: 0.38–0.42 for positive predictive values and 0.96–0.95 for negative predictive values with a single positive test, and 0.67–0.70 for positive predictive values and 0.93–0.94 for negative predictive values for ≥2 positive tests. Higher positive predictive values (0.88 and 0.96) and DOR (135) were found when both PCR and GM were positive [23]. Similar DORs were seen in two reviews (17 and 15 for a single positive specimen, and 30 and 34 for ≥2 positive specimens) [18,23] (Table 4).

Table 4.
Summary of the measure of diagnostic accuracy other than sensitivity and specificity, and subgroup analyses. Studies meeting/partially meeting all AMSTAR-2 requirements in bold text.
The results of the main subgroup analyses to control for sources of heterogeneity are summarized in Table 4. In two reviews of Aspergillus PCR performance on BAL specimens [22,28], antifungal prophylaxis significantly reduced sensitivity of PCR; conversely, results of a review in blood specimens did not find substantial differences in sensitivity values, but a significant decrease in specificity values related to anti-mold prophylaxis [27]. As expected, sensitivity was lower in cohort studies compared to case−control studies, and with the degree of adherence to EORTC/MSGERC criteria [21,28].
4. Discussion
This umbrella review of SRs was aimed at providing an overall summary of the diagnostic accuracy of PCR-based tests on blood and BAL to diagnose IA in immunocompromised patients. Umbrella reviews collate several SRs on the same topic and consider the inclusion of the highest level of evidence available, such as SRs and meta-analyses [28,29,30]. Umbrella reviews of diagnostic tests provide an opportunity to gain greater insights into test accuracy, as data are summarized across different populations, settings, type of specimen, or other variables, while also considering the overall strength of each study included in the review.
In this umbrella review, the results of eight SRs (twelve records) evaluating the performance of Aspergillus PCR tests for the diagnosis of IA in immunocompromised patients, published between 2007 and 2023, were reappraised. The SRs included present data from 167 overlapping reports based on 107 primary studies (98 with a cohort design, 19 with a case−control design) making this, to our knowledge, the largest review of the subject area to date.
When testing BAL specimens, results from five SRs showed a mean sensitivity ranging from 0.57 to 0.91, and mean specificity from 0.92 to 0.97. We graded the level of certainty of these findings as being very low to low due to the risk of bias and due to the heterogeneity (in clinical setting, index test, reference standard adherence, use of antifungal agents) in the primary studies included in the reviews. In blood specimens, results from three SRs showed marked heterogeneity in both sensitivity (ranging from 0.57 to 0.84), and specificity (from 0.58 to 0.95). We graded the level of certainty of these findings as being low (due to the risk of bias and inconsistency) or moderate (due to inconsistency). Lower specificity (0.58) was found in the review of pediatric patients [25], while in two systematic reviews including mostly adult patients, the mean specificity values were higher (0.79 for a single positive test, and 0.93–0.95 for ≥2 positive tests) [23,26]. When pediatric and adult studies are compared, the sensitivity of PCR when testing blood is similar (0.76 in the pediatric review, and from 0.79 and 0.84 for a single positive test, and 0.57 and 0.59 for ≥2 positive tests in the two reviews in adult patients). As with other biomarker tests [31], the use of antifungal therapy was shown to affect performance. In two reviews of BAL specimens [22,28], mold-active antifungals reduced PCR sensitivity, but this effect is not consistent across studies and SRs. Results of a large review in blood specimens [27] did not find substantial differences in PCR sensitivity values, but a significant decrease in specificity values related to mold active agents. It is possible that anti-mold prophylaxis reduces the clinical progression of IA, limiting the manifestations typically associated with IA that are essential when classifying probable IA using the EORTC/MSGERC definitions. Furthermore, given that anti-mold prophylaxis has been associated with reduced GM-EIA sensitivity, the use of mold active agents could result in false-negative GM-EIA results, preventing cases of possible IA being upgraded to probable IA and so compromising the specificity of PCR [27].
There has been considerable progress in standardizing Aspergillus PCR protocols and blood-based assays have been shown to be analytically valid. Now it is necessary to consider how the tests can be best used in practice to maximize clinical utility [31]. The use of a standardized PCR may improve performance, and recent evidence suggests that PCR testing in combination with GM-EIA may provide the optimal management strategy [8].
The sensitivity of Aspergillus PCR using plasma is superior to that using serum [31,32,33]. PCR positivity occurs earlier when testing plasma and provides sufficient sensitivity for the screening of invasive aspergillosis while maintaining methodological simplicity [32]. However, this level of technical detail has not be assessed in the current SRs. Understanding the influence of Aspergillus spp. on PCR performance is also important given analytical sensitivity appears to be reduced when testing for non-fumigatus species [34]. Unfortunately, this level of technical detail is generally not provided in the SRs, further compounded by the fact that many cases of IA are diagnosed in the absence of a positive culture or using genus specific tests. While a wide range of both in-house and commercial Aspergillus PCR assays are available, performance appears to be comparable [26]. The nucleic acid extraction protocol is critical to optimal performance and is important to understand how variations in this process may be influencing SR data, although currently, the wide variation in combined extraction and amplification protocols may limit/prevent this statistical analysis. Nevertheless, compliance with methodological recommendations when testing blood specimens are associated with improved performance, with significant improvements in specificity [19]. The specificity of Aspergillus PCR when testing BAL fluid and when requiring two consecutive positives in blood specimens remains high (>0.92) and subsequent positive likelihood ratios are sufficient to support a diagnosis of IA and the inclusion of Aspergillus PCR as a mycological criterion in the 2020 revision of the EORTC/MSGERC definitions for invasive fungal disease [6]. However, clarification on the interpretation of the Aspergillus PCR criterion when testing BAL fluid is still needed [35]. While the negative predictive values for Aspergillus PCR remain high, the influence of a low pre-test probability (incidence) needs to be observed and the use of negative likelihood ratios better employed (rather than negative predictive values) when test sensitivity is <90%.
As time advances, there will be changes in antifungal prophylactic strategies, treatment of underlying hematological conditions (e.g., CAR-T, monoclonal antibodies), and definitions of invasive fungal disease, which may impact the performance of biomarker assays. These will need to be accounted for when performing future SRs that include both historic and novel datasets. When performing umbrella reviews, the influence of data duplication between individual SR also needs to be considered.
To conclude, this overview summarizes the existing evidence about the diagnostic accuracy of Aspergillus PCR assays in immunocompromised patients and allows us to further investigate the evidence available in the existing systematic reviews, assessing variations in study populations, procedures used to conduct the tests, and other variables, with potential to reduce the impact of data heterogeneity by drawing on a broader evidence base. As determined by GRADE assessment, the level of certainty (evidence) for the individual studies included in each SR is variable (very low to low in SRs of PCR on BAL specimens, and from low to moderate in SRs of PCR on blood specimens). However, based on the overall methodological assessment of the SRs included in this umbrella review, in general, we can have high confidence in the methodological quality provided by the SRs, with 75% of SRs meeting or partially meeting all requirements on the AMSTAR-2 checklist and 98.2% of all critical domains being met/partially met across studies.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jof9100967/s1, Table S1: the PRISMA-DTA checklist” here.
Author Contributions
Conceptualization: M.C. and P.L.W. Methodology: M.C. Data extraction: M.C., P.L.W. Writing—preparation original draft: M.C. and P.L.W. Writing—review and editing: M.C., P.L.W., R.A.B., J.L., J.P.D., T.R.R., W.J.H., A.W., C.O.M., M.L., L.K., D.E.A.L. and B.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Marr, K.A.; Carter, R.A.; Boeckh, M.; Martin, P.; Corey, L. Invasive aspergillosis in allogeneic stem cell transplant recipients: Changes in epidemiology and risk factors. Blood 2002, 100, 4358–4366. [Google Scholar] [CrossRef] [PubMed]
- Barnes, R.A. Early diagnosis of fungal infection in immunocompromised patients. J. Antimicrob. Chemother. 2008, 61 (Suppl. 1), i3–i6. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.J.; Anaissie, E.J.; Denning, D.W.; Herbrecht, R.; Kontoyiannis, D.P.; Marr, K.A.; Morrison, V.A.; Segal, B.H.; Steinbach, W.J.; Stevens, D.A.; et al. Treatment of aspergillosis: Clinical practice guidelines of the Infectious Diseases Society of America. Clin. Infect. Dis. 2008, 46, 327–360. [Google Scholar] [CrossRef]
- Donnelly, J.P. Polymerase chain reaction for diagnosing invasive aspergillosis: Getting closer but still a ways to go. Clin. Infect. Dis. 2006, 42, 487–489. [Google Scholar] [CrossRef] [PubMed]
- White, P.L.; Bretagne, S.; Caliendo, A.M.; Loeffler, J.; Patterson, T.F.; Slavin, M.; Wingard, J.R. Aspergillus Polymerase Chain Reaction-An Update on Technical Recommendations, Clinical Applications, and Justification for Inclusion in the Second Revision of the EORTC/MSGERC Definitions of Invasive Fungal Disease. Clin. Infect. Dis. 2021, 72 (Suppl. 2), S95–S101. [Google Scholar] [CrossRef]
- Donnelly, J.P.; Chen, S.C.; Kauffman, C.A.; Steinbach, W.J.; Baddley, J.W.; Verweij, P.E.; Clancy, C.J.; Wingard, J.R.; Lockhart, S.R.; Groll, A.H.; et al. Revision and Update of the Consensus Definitions of Invasive Fungal Disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin. Infect. Dis. 2020, 71, 1367–1376. [Google Scholar] [CrossRef]
- Haydour, Q.; Hage, C.A.; Carmona, E.M.; Epelbaum, O.; Evans, S.E.; Gabe, L.M.; Knox, K.S.; Kolls, J.K.; Wengenack, N.L.; Prokop, L.J.; et al. Diagnosis of Fungal Infections. A Systematic Review and Meta-Analysis Supporting American Thoracic Society Practice Guideline. Ann. Am. Thorac. Soc. 2019, 16, 1179–1188. [Google Scholar] [CrossRef]
- White, P.L.; Wingard, J.R.; Bretagne, S.; Löffler, J.; Patterson, T.F.; Slavin, M.A.; Barnes, R.A.; Pappas, P.G.; Donnelly, J.P. Aspergillus Polymerase Chain Reaction: Systematic Review of Evidence for Clinical Use in Comparison with Antigen Testing. Clin. Infect. Dis. 2015, 61, 1293–1303. [Google Scholar] [CrossRef]
- Cumpston, M.; Chandler, J. Chapter IV: Updating a review. In Cochrane Handbook for Systematic Reviews of Interventions Version 6.3; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M.L.T., Page, M.J., Welch, V.A., Eds.; Cochrane: London, UK, 2022; Available online: www.training.cochrane.org/handbook (accessed on 1 September 2023).
- Pollock, M.; Fernandes, R.M.; Becker, L.A.; Pieper, D.; Hartling, L. Chapter V: Overviews of Reviews. In Cochrane Handbook for Systematic Reviews of Interventions Version 6.3; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M.L.T., Page, M.J., Welch, V.A., Eds.; Cochrane: London, UK, 2022; Available online: www.training.cochrane.org/handbook (accessed on 1 September 2023).
- McInnes, M.D.F.; Moher, D.; Thombs, B.D.; McGrath, T.A.; Bossuyt, P.M.; the PRISMA-DTA Group; Clifford, T.; Cohen, J.F.; Deeks, J.J.; Gatsonis, C.; et al. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies: The PRISMA-DTA Statement. JAMA 2018, 319, 388–396. [Google Scholar] [CrossRef]
- Ascioglu, S.; Rex, J.H.; de Pauw, B.; Bennett, J.E.; Bille, J.; Crokaert, F.; Denning, D.W.; Donnelly, J.P.; Edwards, J.E.; Erjavec, Z.; et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: An international consensus. Clin. Infect. Dis. 2002, 34, 7–14. [Google Scholar] [CrossRef]
- De Pauw, B.; Walsh, T.J.; Donnelly, J.P.; Stevens, D.A.; Edwards, J.E.; Calandra, T.; Pappas, P.G.; Maertens, J.; Lortholary, O.; Kauffman, C.A.; et al. Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin. Infect. Dis. 2008, 46, 1813–1821. [Google Scholar] [PubMed]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef] [PubMed]
- Brozek, J.L.; Akl, E.A.; Jaeschke, R.; Lang, D.M.; Bossuyt, P.; Glasziou, P.; Helfand, M.; Ueffing, E.; Alonso-Coello, P.; Meerpohl, J.; et al. GRADE Working Group. Grading quality of evidence and strength of recommendations in clinical practice guidelines: Part 2 of 3. The GRADE approach to grading quality of evidence about diagnostic tests and strategies. Allergy 2009, 64, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.; Brożek, J.; Guyatt, G.; Oxman, A. (Eds.) Handbook for Grading the Quality of Evidence and the Strength of Recommendations Using the GRADE Approach (Updated October 2013); GRADE Working Group: Melbourne, Australia, 2013; Available online: gdt.guidelinedevelopment.org/app/handbook/handbook.html (accessed on 1 September 2023).
- Mengoli, C.; Cruciani, M.; Barnes, R.A.; Loeffler, J.; Donnelly, J.P. Use of PCR for diagnosis of invasive aspergillosis: Systematic review and meta-analysis. Lancet Infect. Dis. 2009, 9, 89–96. [Google Scholar] [CrossRef]
- Cruciani, M.; Mengoli, C.; Loeffler, J.; Donnelly, P.; Barnes, R.; Jones, B.L.; Klingspor, L.; Morton, O.; Maertens, J. Polymerase chain reaction blood tests for the diagnosis of invasive aspergillosis in immunocompromised people. Cochrane Database Syst. Rev. 2015, 9, CD009551. [Google Scholar] [CrossRef]
- Arvanitis, M.; Ziakas, P.D.; Zacharioudakis, I.M.; Zervou, F.N.; Caliendo, A.M.; Mylonakis, E. PCR in diagnosis of invasive aspergillosis: A meta-analysis of diagnostic performance. J. Clin. Microbiol. 2014, 52, 3731–3742. [Google Scholar] [CrossRef]
- Tuon, F.F. A systematic literature review on the diagnosis of invasive aspergillosis using polymerase chain reaction (PCR) from bronchoalveolar lavage clinical samples. Rev. Iberoam. Micol. 2007, 24, 89–94. [Google Scholar]
- Sun, W.; Wang, K.; Gao, W.; Su, X.; Qian, Q.; Lu, X.; Song, Y.; Guo, Y.; Shi, Y. Evaluation of PCR on bronchoalveolar lavage fluid for diagnosis of invasive aspergillosis: A bivariate metaanalysis and systematic review. PLoS ONE 2011, 6, e28467. [Google Scholar] [CrossRef]
- Avni, T.; Levy, I.; Sprecher, H.; Yahav, D.; Leibovici, L.; Paul, M. Diagnostic accuracy of PCR alone compared to galactomannan in bronchoalveolar lavage fluid for diagnosis of invasive pulmonary aspergillosis: A systematic review. J. Clin. Microbiol. 2012, 50, 3652–3658. [Google Scholar] [CrossRef]
- Arvanitis, M.; Anagnostou, T.; Mylonakis, E. Galactomannan and Polymerase Chain Reaction-Based Screening for Invasive Aspergillosis Among High-Risk Hematology Patients: A Diagnostic Meta-analysis. Clin. Infect. Dis. 2015, 61, 1263–1272. [Google Scholar] [CrossRef]
- Heng, S.C.; Morrissey, O.; Chen, S.C.; Thursky, K.; Manser, R.L.; Nation, R.L.; Kong, D.C.; Slavin, M. Utility of bronchoalveolar lavage fluid galactomannan alone or in combination with PCR for the diagnosis of invasive aspergillosis in adult hematology patients: A systematic review and meta-analysis. Crit. Rev. Microbiol. 2015, 41, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Lehrnbecher, T.; Robinson, P.D.; Fisher, B.T.; Castagnola, E.; Groll, A.H.; Steinbach, W.J.; Zaoutis, T.E.; Negeri, Z.F.; Beyene, J.; Phillips, B.; et al. Galactomannan, β-D-Glucan, and Polymerase Chain Reaction-Based Assays for the Diagnosis of Invasive Fungal Disease in Pediatric Cancer and Hematopoietic Stem Cell Transplantation: A Systematic Review and Meta-Analysis. Clin. Infect. Dis. 2016, 63, 1340–1348. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, M.; Mengoli, C.; Barnes, R.; Donnelly, J.P.; Loeffler, J.; Jones, B.L.; Klingspor, L.; Maertens, J.; Morton, C.O.; White, L.P. Polymerase chain reaction blood tests for the diagnosis of invasive aspergillosis in immunocompromised people. Cochrane Database Syst. Rev. 2019, 9, CD009551. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, M.; White, P.L.; Mengoli, C.; Löffler, J.; Morton, C.O.; Klingspor, L.; Buchheidt, D.; Maertens, J.; Heinz, W.J.; Rogers, T.R.; et al. Fungal PCR Initiative. The impact of anti-mould prophylaxis on Aspergillus PCR blood testing for the diagnosis of invasive aspergillosis. J. Antimicrob. Chemother. 2021, 76, 635–638. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Wu, X.; Jiang, G.; Guo, A.; Jin, Z.; Ying, Y.; Lai, J.; Li, W.; Yan, F. Bronchoalveolar lavage fluid polymerase chain reaction for invasive pulmonary aspergillosis among high-risk patients: A diagnostic meta-analysis. BMC Pulm. Med. 2023, 23, 58. [Google Scholar] [CrossRef]
- Ioannidis, J.P. Integration of evidence from multiple meta-analyses: A primer on umbrella reviews, treatment networks and multiple treatments meta-analyses. CMAJ 2009, 181, 488–493. [Google Scholar] [CrossRef]
- Aromataris, E.; Fernandez, R.; Godfrey, C.M.; Holly, C.; Khalil, H.; Tungpunkom, P. Summarizing systematic reviews: Methodological development, conduct and reporting of an umbrella review approach. Int. J. Evid. Based Health 2015, 13, 132–140. [Google Scholar] [CrossRef]
- Barnes, R.A.; White, P.L.; Morton, C.O.; Rogers, T.R.; Cruciani, M.; Loeffler, J.; Donnelly, J.P. Diagnosis of aspergillosis by PCR: Clinical considerations and technical tips. Med. Mycol. 2018, 56 (Suppl. 1), 60–72. [Google Scholar] [CrossRef]
- White, P.L.; Barnes, R.A.; Springer, J.; Klingspor, L.; Cuenca-Estrella, M.; Morton, C.O.; Lagrou, K.; Bretagne, S.; Melchers, W.J.; Mengoli, C.; et al. EAPCRI. Clinical Performance of Aspergillus PCR for Testing Serum and Plasma: A Study by the European Aspergillus PCR Initiative. J. Clin. Microbiol. 2015, 53, 2832–2837. [Google Scholar] [CrossRef]
- Loeffler, J.; Mengoli, C.; Springer, J.; Bretagne, S.; Cuenca-Estrella, M.; Klingspor, L.; Lagrou, K.; Melchers, W.J.; Morton, C.O.; Barnes, R.A.; et al. European Aspergillus PCR Initiative. Analytical Comparison of In Vitro-Spiked Human Serum and Plasma for PCR-Based Detection of Aspergillus fumigatus DNA: A Study by the European Aspergillus PCR Initiative. J. Clin. Microbiol. 2015, 53, 2838–2845. [Google Scholar] [CrossRef]
- Morton, C.O.; White, P.L.; Barnes, R.A.; Klingspor, L.; Cuenca-Estrella, M.; Lagrou, K.; Bretagne, S.; Melchers, W.; Mengoli, C.; Caliendo, A.M.; et al. EAPCRI. Determining the analytical specificity of PCR-based assays for the diagnosis of IA: What is Aspergillus? Med. Mycol. 2017, 55, 402–413. [Google Scholar] [CrossRef] [PubMed]
- Huygens, S.; Dunbar, A.; Buil, J.B.; Klaassen, C.H.W.; Verweij, P.E.; van Dijk, K.; de Jonge, N.; Janssen, J.J.W.M.; van der Velden, W.J.F.M.; Biemond, B.J.; et al. Clinical impact of PCR-based Aspergillus and azole resistance detection in invasive aspergillosis. A prospective multicenter study. Clin. Infect. Dis. 2023, 77, ciad141. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).