Abstract
The analysis of the IR carbonyl bands of some 3-(4′-substituted phenylsulfanyl)-1-methyl-2-piperidones 1–6 bearing substituents: NO2 (compound 1), Br (compound 2), Cl (compound 3), H (compound 4) Me (compound 5) and OMe (compound 6) supported by B3LYP/6-31+G(d,p) and PCM calculations along with NBO analysis (for compound 4) and X-ray diffraction (for 2) indicated the existence of two stable conformations, i.e., axial (ax) and equatorial (eq), the former corresponding to the most stable and the least polar one in the gas phase calculations. The sum of the energy contributions of the orbital interactions (NBO analysis) and the electrostatic interactions correlate well with the populations and the νCO frequencies of the ax and eq conformers found in the gas phase. Unusually, in solution of the non-polar solvents n-C6H14 and CCl4, the more intense higher IR carbonyl frequency can be ascribed to the ax conformer, while the less intense lower IR doublet component to the eq one. The same νCO frequency trend also holds in polar solvents, that is νCO (eq) < νCO (ax). However, a reversal of the ax/eq intensity ratio occurs going from non-polar to polar solvents, with the ax conformer component that progressively decreases with respect to the eq one in CHCl3 and CH2Cl2, and is no longer detectable in the most polar solvent CH3CN. The PCM method applied to compound 4 supports these findings. In fact, it predicts the progressive increase of the eq/ax population ratio as the relative permittivity of the solvent increases. Moreover, it indicates that the computed νCO frequencies of the ax and eq conformers do not change in the non–polar solvents n-C6H14 and CCl4, while the νCO frequencies of the eq conformer become progressively lower than that of the ax one going from CHCl3 to CH2Cl2 and to CH3CN, in agreement with the experimental IR values. The analysis of the geometries of the ax and eq conformers shows that the carbonyl oxygen atom of the eq conformer is free for solvation, while the O[CO]…H[o-Ph] hydrogen bond that takes place in the ax conformer partially hinders the approach of the solvent molecules to the carbonyl oxygen atom. Therefore, the larger solvation that occurs in the carbonyl oxygen atom of the eq conformer is responsible for the observed and calculated decrease of the corresponding frequency. The X-ray single crystal analysis of 2 indicates that this compound adopts the most polar eq geometry in the solid. In fact, in order to obtain the largest energy gain, the molecules are arranged in the crystal in a helical fashion due to dipole moment coupling along with C-H…O and C-H…πPh hydrogen bonds.
1. Introduction
The analysis of the infrared (νCO) band and theoretical B3LYP/6-311++G(d,p) calculations of some N,N-diethyl-2-[(4′-substituted) phenylsulfanyl acetamides Et2NC(O)CH2SC6H4-Y (Y=OMe, Me, H, Cl, Br, NO2) [1] indicated the existence of two pairs (anti and syn) of cis (c) and gauche (g) conformers in the gas phase, with the gauche conformers being significantly more stable (ca. 90%) than the cis ones. The anti-geometry is more stable than the syn one for each pair of cis and gauche conformers. The summing up of the σC-S→π*C=O, πC=O→σ*C-S, π*C=O→σ*C-S (NBO) orbital interactions is the main factor that stabilises the gauche conformers to a larger extent than the cis ones for which these interactions are absent. The similarity of the carbonyl frequencies in the gas phase for the anti and syn pairs of the cis and gauche conformers originates a carbonyl doublet in solution for which the less intense, higher frequency component is ascribed to the pair of the cis conformers and the lower frequency component corresponds to the more stable pair of the gauche conformers.
In solution of the non-polar solvents (n-C6H14 and CCl4), the least polar gauche conformers (ca. 85%) are predominant with respect to the most polar cis ones. In the polar solvent, CHCl3, a decrease of the gauche/cis population ratio is observed, i.e., 60% / 40%, and in the highly polar solvent, CH3CN, the most polar cis conformer is the only one present. This trend was supported by the PCM solvation model performed by the B3LYP/6-311++G(d,p) method.
Theoretical and spectroscopic (IR, NMR and microwave) studies [2,3,4,5] of 2-piperidones showed that the piperidone ring lies in a half-chair or slightly twisted half-chair conformation. X-ray single crystal structure determination of the 3-chloro-2-piperidone [6] has shown that the piperidone ring is in a slightly twisted half-chair conformation with the 3-substituent assuming the quasi-equatorial [Cl-C-C=O (dihedral angle) = −30°] geometry. Moreover, our theoretical and X-ray single crystal studies of some 3-(4′-substituted phenylsulfonyl)-1-methyl-2-piperidones [7,8] have shown that, in these compounds, the 2-piperidone ring assumes a slightly distorted half-chair conformation. In the gas phase the 3-substituent adopts the quasi-equatorial, sin-clinal and quasi-axial geometries with respect to S-C-C=O dihedral angles of ca. 31°, 46° and 71°, respectively, while in the solid state the preferred geometry is quasi-axial with respect to the S-C-C=O dihedral angle of ca. 72°.
Aiming to throw more light on the nature of the orbital and electrostatic interactions that influence the stability of the cis-gauche conformers of 2-(4′-substituted)-phenylsulfanylamides [1], this paper reports the IR study of some six-member ring lactams, i.e., 3-(4′-substituted phenylsulfanyl)-1-methyl-2-piperidones bearing in the 4′ position the substituents, i.e., Y = NO2 (compound 1), Br (compound 2), Cl (compound 3), H (compound 4) Me (compound 5) and OMe (compound 6) (Scheme 1) along with density functional theory (DFT), polarizable continuum model (PCM) and natural bond orbital (NBO) calculations for 4 taken as the representative compound for the whole series, along with the X-ray diffraction analysis of 2. These compounds were chosen taking into account some conformational rigidity of the piperidone ring, which allows the 3-substituent to assume almost exclusively the quasi-equatorial and axial conformations.
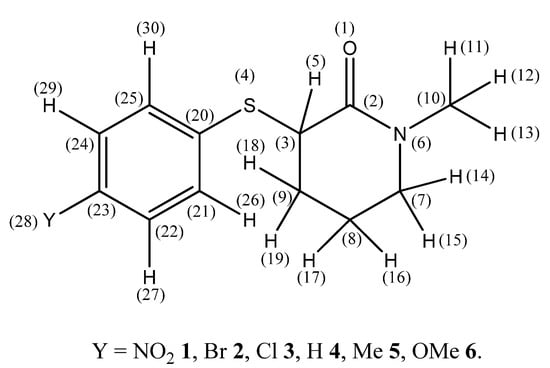
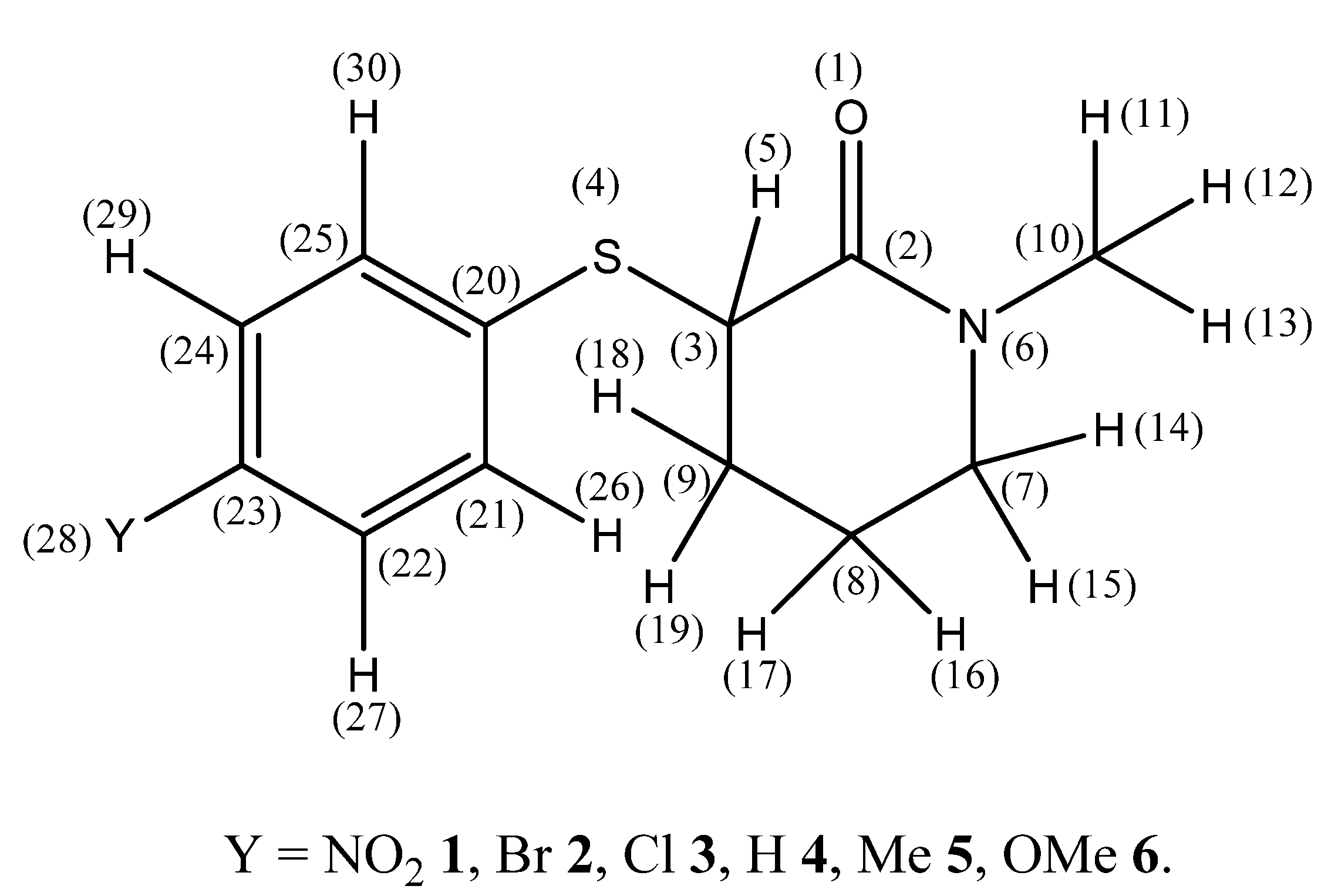
Scheme 1.
Atom labeling of 3-(4′-substituted phenylsulfanyl)-1-methyl-2-piperidones and definition of relevant torsion angles.
Scheme 1.
Atom labeling of 3-(4′-substituted phenylsulfanyl)-1-methyl-2-piperidones and definition of relevant torsion angles.
α = O(1)-C(2)-C(3)-S(4)
β = C(2)-C(3)-S(4)-C(20)
γ = C(3)-S(4)-C(20)-C(25)
δ = O(1)-C(2)-N(6)-C(10)
θ = O(1)-C(2)-N(6)-C(7)
θ’ = O(1)-C(2)-C(3)-C(9)
ω = C(2)-N(6)-C(7)-C(8)
ω’ = C(2)-C(3)-C(9)-C(8)
2. Results and Discussion
Table 1 displays the stretching frequencies and the absorbance percentage of the analytically resolved carbonyl band components for compounds 1–6 in solvents of increasing relative permittivity [9], i.e., n-C6H14 (ε = 1.9), CCl4 (ε = 2.2), CHCl3 (ε = 4.8), CH2Cl2 (ε = 9.1) and CH3CN (ε = 38). A doublet is shown in n-hexane for derivatives 2–6 and in CCl4 (fundamental and first overtone regions) for derivatives 1–6, with the higher frequency component being the more intense, i.e., ca. 80% and ca. 58%, respectively. However, as the relative permittivity increases, a reversal of the carbonyl doublet intensity is observed, with the lower frequency component progressively increasing up to ca. 70% in CHCl3, ca. 88% in CH2Cl2 and emerges as a singlet in the highest relative permittivity solvent, CH3CN. The solvent effect on the carbonyl band components for 4, chosen as a prototype for the series 1–6 is illustrated in Figure 1.
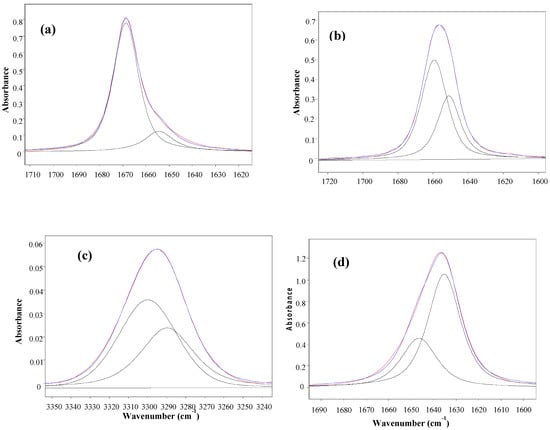
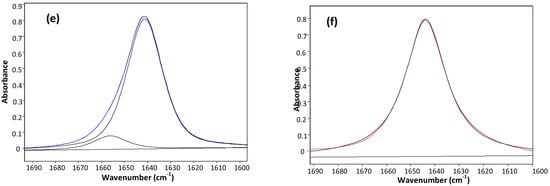
Figure 1.
IR spectra of 3-(phenylsulfanyl)-1-methyl-2-piperidone(4) showing the analytically resolved carbonyl stretching band in: n-hexane (a), carbon tetrachloride [fundamental (b) and first overtone (c)], chloroform (d) dichloromethane (e) and acetonitrile (f).
Figure 1.
IR spectra of 3-(phenylsulfanyl)-1-methyl-2-piperidone(4) showing the analytically resolved carbonyl stretching band in: n-hexane (a), carbon tetrachloride [fundamental (b) and first overtone (c)], chloroform (d) dichloromethane (e) and acetonitrile (f).
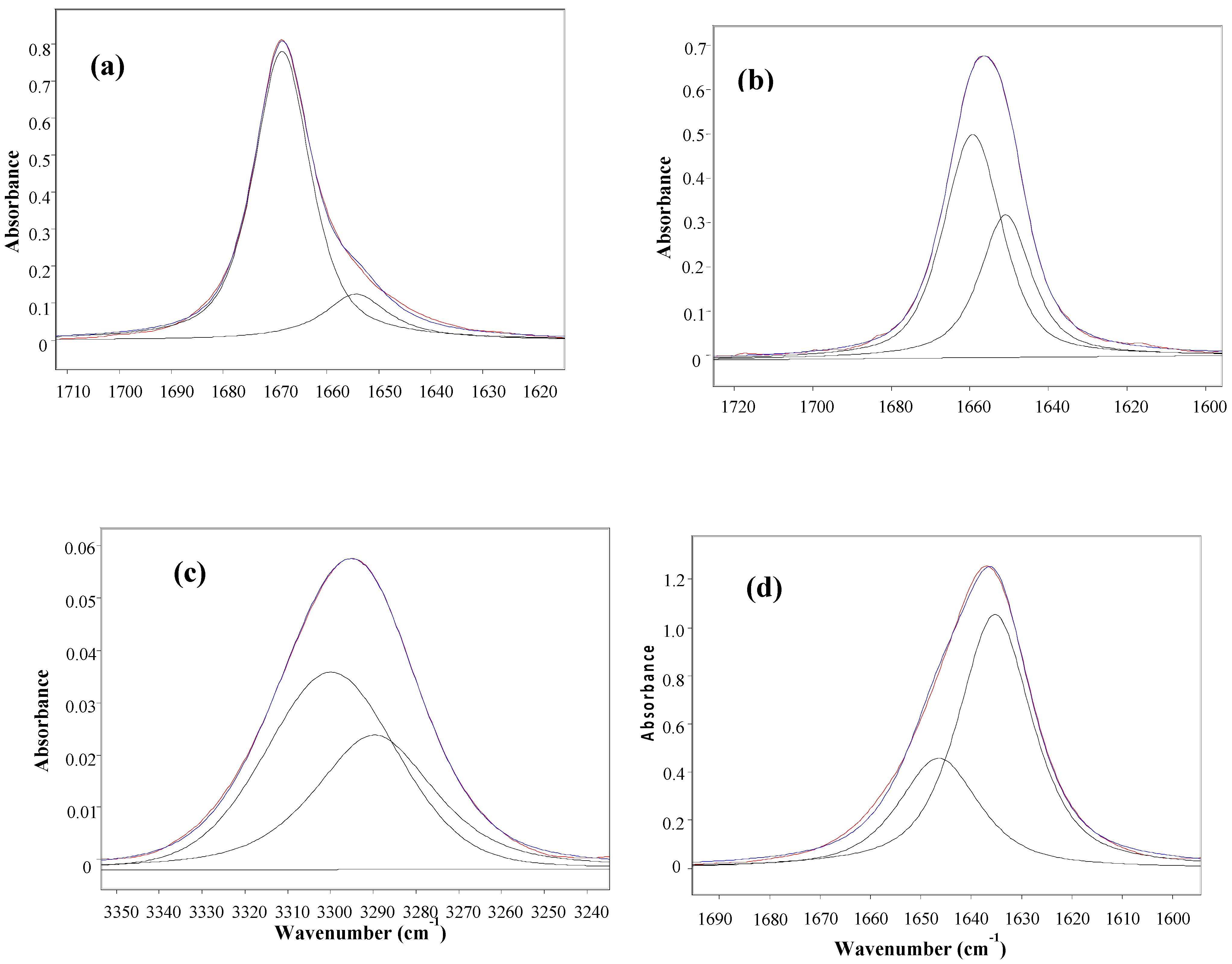
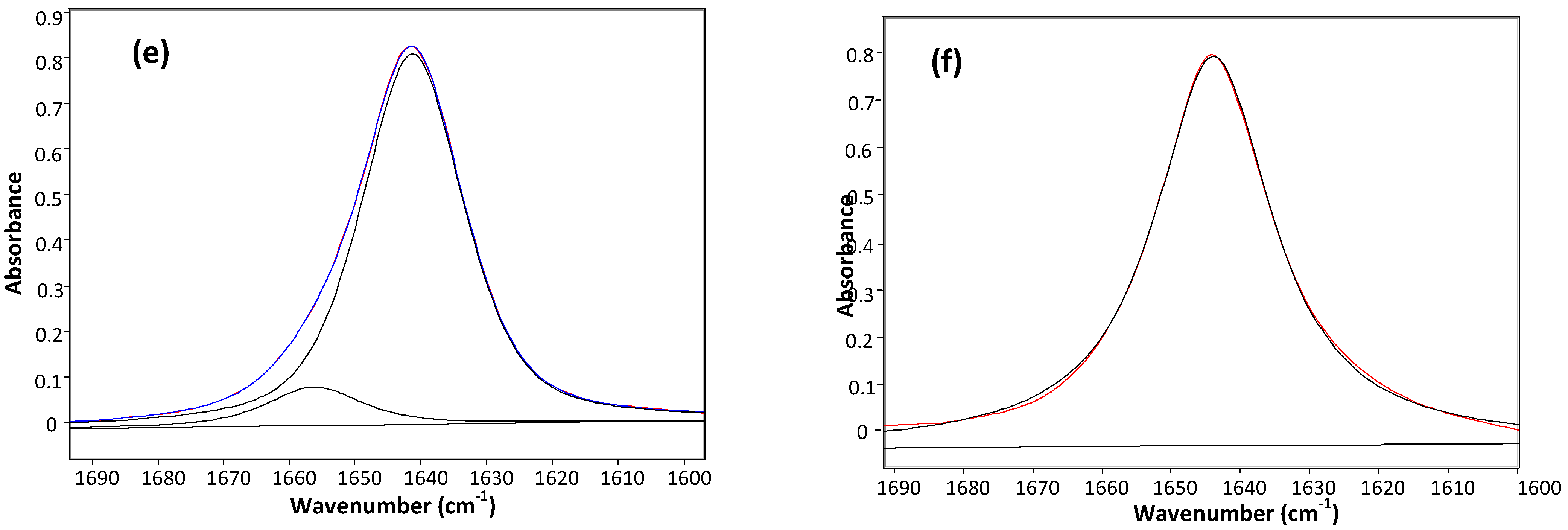
The unusual observed solvent effect (Table 1) might not be a clear evidence of the existence of conformational isomerism. However, the occurrence of two carbonyl band components in the first overtone region for 1–6 in carbon tetrachloride, with comparable intensity ratios and at frequencies twice that of the fundamental minus ca. 15 cm−1 (that is two times the mechanical anharmonicity [10]), is indicative of the occurrence of two stable conformations for all compounds in this solvent, ruling out the existence of any vibrational effect in the fundamental transition of the νCO mode [11,12]. Therefore, the IR data strongly suggest the occurrence of two conformers for compounds 1–6 in all solvents, with the exception of CH3CN, for which only one conformer is present.

Table 1.
Frequencies (ν, cm−1) and intensities of the carbonyl stretching bands in the IR spectra of 3-(4′-substituted-phenyl)sulfanyl-1-methyl-2-piperidones 1–6.
| Compound | Y | n-C6H14 | CCl4 | CHCl3 | CH2Cl2 | CH3CN | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ν | Pa | ν | Pa | νb | P | ν | P | ν | P | ν | P | |||
| 1 | NO2 | -c | - | 1660 | 61 | 3300 | 60 | 1648 | 43 | 1656 | 9 | - | - | |
| - | - | 1652 | 39 | 3289 | 40 | 1637 | 57 | 1641 | 91 | 1645 | 100 | |||
| 2 | Br | 1668 | 84 | 1658 | 68 | 3299 | 63 | 1645 | 33 | 1652 | 12 | - | - | |
| 1656 | 16 | 1650 | 32 | 3291 | 37 | 1635 | 67 | 1641 | 88 | 1644 | 100 | |||
| 3 | Cl | 1668 | 79 | 1660 | 45 | 3306 | 49 | 1647 | 30 | 1656 | 13 | - | - | |
| 1656 | 21 | 1652 | 55 | 3292 | 51 | 1635 | 70 | 1641 | 87 | 1644 | 100 | |||
| 4 | H | 1669 | 83 | 1659 | 62 | 3300 | 59 | 1647 | 32 | 1656 | 9 | - | - | |
| 1654 | 17 | 1651 | 38 | 3290 | 41 | 1635 | 68 | 1641 | 91 | 1644 | 100 | |||
| 5 | Me | 1668 | 72 | 1657 | 57 | 3300 | 57 | 1645 | 16 | 1652 | 12 | - | - | |
| 1659 | 28 | 1651 | 43 | 3286 | 43 | 1634 | 84 | 1640 | 88 | 1643 | 100 | |||
| 6 | OMe | 1668 | 78 | 1660 | 44 | 3301 | 41 | 1647 | 25 | 1652 | 15 | - | - | |
| 1657 | 22 | 1651 | 56 | 3288 | 59 | 1635 | 75 | 1639 | 85 | 1643 | 100 | |||
a Relative intensity of each component of the carbonyl doublet, expressed as a percentage of absorbance; b First overtone; c Compound slightly soluble in this solvent.
Aiming to estimate the geometry of the minimum energy conformations in the gas phase, B3LYP/6-31+G(d,p) calculations were performed for compound 4, taken as representative of the whole series 1–6. Moreover, in order to estimate the solvent effect on the minimum energy conformations of 6, the PCM method was applied to fully optimise the referred conformations in the n-C7H16, CCl4, CHCl3, CH2Cl2 and CH3CN solvents, and to compute the corresponding vibrational frequencies. The relevant data obtained for 4 are presented in Table 2.
Theoretical calculations in the gas phase show the existence of two stable conformations for compound 4. Due to the significant contribution of the [-O‒C=N+] resonance structure for the hybrid [13,14], the [C(O)NC(10)C(7)] carboxamide moiety (Scheme 1) is almost planar (δ and θ dihedral angles are ca. 3° and ca. 170°, respectively). This induces the piperidone ring to adopt a slightly distorted half-chair geometry, with the ω and ω′ dihedral angles being ca. 27° and ca. 51°, respectively (Table 2 and Figure 2) and the 3-phenylsulfanyl substituent assuming the almost axial (ax) and equatorial (eq) geometries with respect to the carbonyl group, at α angles of ca. 83° and ca. 29°, respectively. Moreover, as shown by the θ, θ′, ω and ω′ dihedral angles for the equatorial conformer (Table 2), in the gas phase of 4 and in the solid state of 2, the substituents at the 4′ position have no influence on the geometry of the lactam moiety.

Table 2.
Relative energies (E, kJ mol−1), dipole moments (μ, D), carbonyl frequencies (ν, cm−1), and selected torsion angles (deg) calculated for the minimum energy conformations of 3-phenylthio-1-methyl-2-piperidone 4 in gas phase and in solvents with the PCM method at the B3LYP/6-31+G(d,p) level, and the X-ray geometric data of 2.
| Conf. a | E b | P c | μ | νCO | Torsion angles/º d | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| α | β | γ | δ | θ | θ’ | ω | ω' | ||||||
| gas | ax | 0 | 96.8 | 3.62 | 1713.4 | 82.0 | −101.3 | 48.2 | 2.13 | 167.9 | −154.3 | 23.6 | −50.8 |
| eq | 8.45 | 3.2 | 5.39 | 1720.1 | 30.4 | −176.5 | 73.2 | −3.90 | −170.8 | 157.1 | −23.7 | 50.8 | |
| C7H16 | ax | 0 | 84.4 | 4.08 | 1696.7 | 82.0 | −101.9 | 48.4 | 2.06 | 168.2 | −154.5 | 23.3 | −50.7 |
| eq | 4.20 | 15.6 | 6.21 | 1699.0 | 31.6 | −177.4 | 72.9 | −3.74 | −172.2 | 158.2 | −22.8 | 50.3 | |
| CCl4 | ax | 0 | 80.0 | 4.16 | 1694.2 | 81.9 | −102.1 | 49.1 | 2.06 | 168.1 | −154.5 | 23.4 | −50.7 |
| eq | 3.43 | 20.0 | 6.37 | 1694.3 | 31.1 | −176.2 | 73.3 | −3.75 | −171.7 | 157.7 | −23.1 | 50.6 | |
| CHCl3 | ax | 0.17 | 48.3 | 4.61 | 1676.8 | 81.9 | −102.4 | 47.7 | 2.11 | 168.3 | −154.5 | 23.2 | −50.7 |
| eq | 0 | 51.7 | 7.20 | 1673.0 | 33.4 | −178.1 | 72.1 | −3.28 | −174.9 | 159.9 | −20.6 | 49.7 | |
| CH2Cl2 | ax | 2.01 | 30.8 | 4.89 | 1668.9 | 80.6 | −104.3 | 48.5 | 2.57 | 169.3 | −155.7 | 23.1 | −50.1 |
| eq | 0 | 69.2 | 7.62 | 1663.8 | 32.7 | −178.4 | 71.4 | −3.20 | −174.3 | 159.1 | −20.8 | 50.9 | |
| CH3CN | ax | 3.93 | 16.9 | 5.18 | 1659.5 | 80.0 | −105.6 | 48.9 | 2.60 | 169.8 | −156.2 | 22.6 | −49.8 |
| eq | 0 | 83.1 | 8.10 | 1652.3 | 32.1 | −178.4 | 70.7 | −3.11 | −173.2 | 158.5 | −21.7 | 50.2 | |
| X-ray e | - | - | - | - | - | 31.0 (3) | −176.2 (2) | −155.5(2) | −4.1 (4) | −177.6(3) | 157.2(3) | −14.3(4) | 53.1 (3) |
a Conformer designations ax and eq refers to the axial and equatorial conformers, respectively; b Relative energy; c Molar fraction in percentage; d See Scheme 1; e Refers to compound 2.
The ax conformer is the most stable (ca. 97%) and the least polar (ca. 3.6 D) and the eq conformer is the least stable (ca. 3%) and the most polar (ca. 5.4 D). Moreover, in the gas phase of 4, the carbonyl frequencies for the ax and eq conformers are ca. 1713 cm−1 and ca. 1720 cm−1, respectively. As expected, this trend is in agreement with the computations of the N,N-diethyl-2-[(4′-substituted) phenylsulfanyl acetamides [1] for which, in the gas phase, the less polar gauche conformers are the most stable (lower carbonyl frequency) than the more polar cis conformers (higher carbonyl frequency) (see above). However, the theoretical (gas phase) carbonyl frequencies and the relative population values for the axial and equatorial conformers for 4 (Table 2) do not match with the IR frequencies and the relative intensities of each carbonyl component in solution (Table 1). Unusually, it seems reasonable to assign the more intense (ca. 80%) higher carbonyl frequency component in solution of non-polar solvent (n-C6H14) to the ax conformer and the less intense lower carbonyl frequency component to the eq one.
Table 2 shows that the torsional angles α-ω’ for the ax and eq conformers for 4 remain almost the same going from the gas phase to solvents of increasing relative permittivity, i.e., from n-C7H16 to CH3CN. Furthermore, in this direction, there is a progressive decrease of the calculated ax/eq conformer population ratio, i.e., 97%/3% (gas), 84%/16% (n-C7H16), 80%/20% (CCl4), 48%/52% (CHCl3), 31%/69% (CH2Cl2) and 17%/83% (CH3CN) followed by a simultaneous decrease of carbonyl frequency shifts (Δν = νeq – νax) between the eq and ax conformer frequencies that is: ca. 7 cm−1 (gas), ca. 2 cm−1 (n-C7H16), ca. 0 cm−1 (CCl4), ca. −4 cm−1 (CHCl3), ca. −5 cm−1 (CH2Cl2) and ca. −7 cm−1 (CH3CN). These trends cannot be justified on the grounds of the dipole moment (μ) analysis as the dipole moment for the ax and eq conformers increases almost to the same extent going from gas to acetonitrile solution, i.e., ca. 2.7 D.
A close match was not found between the computed ax/eq population ratios (PCM) and the experimental IR ones. However, the progressive decrease of the prevalence of the population of the ax conformer over the eq one going from n-C7H16 to CCl4, along with the progressive increase of the prevalence of the population of the eq conformer over the ax one going from CHCl3 to CH2Cl2 and to CH3CN, seems to be in line with the experimental IR trend of the ax/eq population ratio. The fact that the calculated (PCM) carbonyl frequencies for the ax and eq conformers becomes practically coincident in the non-polar solvents n-C7H16 and CCl4, and that the νCO frequency of the eq conformer becomes progressively smaller than the νCO frequency of the ax conformer going from CHCl3 to CH2Cl2 and to CH3CN, gives support to the experimental (IR) ax-eq carbonyl frequencies assignments (see above).
It should be pointed out that MP2/6-31+G(d,p) calculations for 4 [15] have shown that the population ratio for the ax/eq conformers (ca. 78%/22%) as well as the torsional angles α-ω’ values for both conformers, are close to those obtained at the B3LYP/6-31+G(d,p) level (Table 2). The computed MP2 carbonyl frequencies indicate that the ax and eq conformers have almost the same carbonyl frequency of ca. 1716 cm−1. Moreover the single-point PCM calculations at the MP2 level [16] for the ax and eq conformers of 4 have shown that the solvent effect on the ax/eq population ratio is quite similar to that obtained with the B3LYP method.
Table 3 presents the ChELPG atomic charges [17] for selected atoms computed at the B3LYP/6-31+G(d,p) level for compound 4, while Table 4 displays the interatomic distances between some selected atoms and the difference between these contacts and the sum of the van der Waals (ΣvdW) radii.

Table 3.
ChElPG charge (e) at selected atoms obtained at the B3LYP/6-31+G(d,p) level for 3-phenylsulfanyl-1-methyl-piperidone 4.
| Conf. | O(1) | C(2) | S(4) | N(6) | H(26) | H(30) | H(5) | H(11) | H(16) | H(17) |
|---|---|---|---|---|---|---|---|---|---|---|
| ax | −0.557 | 0.525 | −0.321 | −0.187 | 0.131 | 0.102 | 0.048 | 0.088 | 0.028 | 0.065 |
| eq | −0.537 | 0.605 | −0.320 | −0.311 | 0.103 | 0.061 | 0.027 | 0.058 | 0.030 | 0.045 |

Table 4.
Selected interatomic distances (Å) for the ax and eq conformers of 3-phenylsulfanyl-1-methyl-2-piperidone 4 at the B3LYP/6-31+G(d,p) level.
| Conf. a | O[1]...S[4] b | Δl c | O[1]...H[11] d | Δl | O[1]...H[26] d | Δl | O[1]...H[5] d | Δl |
|---|---|---|---|---|---|---|---|---|
| ax | 3.37 | +0.05 | 2.28 | −0.44 | 2.35 | −0.37 | 2.52 | −0.20 |
| eq | 2.89 | −0.43 | 2.27 | −0.45 | 4.75 | +2.03 | 2.83 | +0.11 |
a Conformer designation; b Sum of the van der Waals radii = 3.32 Å; c Difference between non bonded atoms distance and the sum of their van der Waals radii; d Sum of the van der Waals radii = 2.72 Å.
Due to the decrease of the α dihedral angle going from the ax (ca. 80°) to the eq (ca. 31°) conformer there is in the eq conformer a short contact between the negatively charged O[1] (≅ −0.55e)…S[4] (≅ −0.32e) atoms whose interatomic distance is significantly shorter than the sum of the van der Waals radii (ΣvdW) (Δl ≅ −0.43Å) while for the ax conformer the same contact is close to the ΣvdW radii (Δl ≅ +0.05Å). The referred shorter O…S contact of the eq conformer should be responsible for the stronger Repulsive Field Effect [11,18] between the Cδ+=Oδ- and Cδ+─Sδ− dipoles in the eq conformer, being responsible for both the destabilisation of this conformer relative to the ax one and for the higher carbonyl frequency observed in the gas phase of the eq conformer with respect to that of the ax one whose carbonyl frequency shift (νeq – νax) is ca. +7 cm−1 (Table 2).
As expected from the geometry of the carboxamide moiety of the 1-methyl-2-piperidones, there is a short contact between the oppositely charged O[1] (≅ −0.55e)…H[11] (≅ 0.09e) atoms whose interatomic distance is significantly shorter than the ΣvdW radii (Δl ≅ −0.44Å; C-H…O angle ca. 103°) for bothax and eq conformers (Figure 2). The O[1] (≅ −0.55e) and H[5] (≅ 0.05e) contact is shorter than ΣvdW radii by (Δl ≅ −0.20Å; C-H…O angle ca. 84°) for theax conformer only. Moreover, as a consequence of the smaller γ dihedral angle in the ax conformer (ca. 48°) as compared with that of the eq one (ca. 73°) for 4 (Table 2) the positively charged o-phenylsulfanyl hydrogen atom H[26] (≅ 0.13e) gets closer to the negatively charged carbonyl oxygen atom O[1] (≅ −0.55e) Therefore, the interatomic distance becomes shorter than the ΣvdW radii by (Δl ≅ −0.37 Å; C-H…O angle ca. 148°) for the ax conformer and larger than the ΣvdW radii (Δl ≅ +2.0 Å) for the eq one.
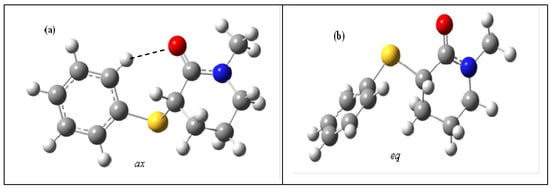
Figure 2.
Axial (ax) (a) and equatorial (eq) (b) conformers for 4 obtained at the B3LYP/6-31+G(d,p) level.
Figure 2.
Axial (ax) (a) and equatorial (eq) (b) conformers for 4 obtained at the B3LYP/6-31+G(d,p) level.
From these Hδ+…Oδ- contacts analysis it may be concluded that the O[1]…H[11] short contact stabilises electrostatically both the eq and ax conformers to the same extent. However, the O[1]…H[5] contact and mainly the O[1]...H[26] one stabilise electrostatically the ax conformer only.
In order to rationalise the orbital interactions that stabilise the ax and eq conformers, NBO analysis [19] was performed and the results obtained for selected NBO energy interactions between donor and acceptor orbitals [20] are reported in Table 5.

Table 5.
Comparison of significant NBO energies (kcal mol−1) of the corresponding interacting orbitals for the conformers ax and eq of 3-phenylsulfanyl-1-methyl-2-piperidone 4 at the B3LYP/6-31+G(d,p) level.
| Orbitals | ax | eq |
|---|---|---|
| LPN6→π*C2=O1 | 65.3 | 50.6 |
| LPO1→σ*C2-N6 | 25.4 | 25.5 |
| LPO1→σ*C2-C3 | 18.9 | 20.3 |
| LPO1→σ*C21-H26 | 3.0 | - |
| LPO1→σ*C10-H11 | 1.0 | 1.2 |
| LPO1→ σ*S4-C20 | - a | 0.9 |
| LPS4→ π*C2=O1 | 2.0 | - |
| LPS4→ π *C20-C21 | 8.3 | 2.7 |
| σC3-S4→π*C2=O1 | 4.2 | 0.7 |
| σC3-S4→σ*C2-N6 | - | 3.4 |
| πC2=O1→σ*C3-S4 | 1.7 | 0.6 |
| π*C2=O1→σ*C3-S4 | 5.5 | 0.8 |
| ∑ Eb | 135.3 | 106.7 |
a Interaction energy smaller than 0.5 kcal mol−1; b Summing up of the energies of the interacting orbitals.
The most important orbital interaction, the LPN6→π*C2=O1 corresponding to the [O=C–N ↔ −O–C=N+] conjugation, has a value of ca. 65 kcal moL−1 for the ax conformer, significantly higher than that of ca. 51 kcal mol−1 for theeq one. The smaller orbital interaction value for the latter conformer is in agreement with the smaller contribution of the [−O–C=N+] canonical form to the resonance hybrid due to the strong Repulsive Field Effect between the Cδ+=Oδ- and Cδ+-Sδ− dipoles, as outlined above. On the other hand, the very short Oδ−[1]…H δ+[13] contact with a suitable C─H…O angle of ca. 148° in the ax conformer determines the LPO1→σ*C21-H26 orbital interaction (ca. 3 kcal mol−1) (hydrogen bond) in the ax conformer. This interaction increases the weight of the polar [-O‒C=N+] canonical form for the resonance hybrid and is partially responsible for the higher value of the LPN6→π*C2=O1 orbital interaction cited above. In addition, the higher occupancy value [20] of the σ*C21-H26 orbital in the ax conformer (0.134 vs. 0.014) gives further support for the occurrence of the significant LPO1→σ*C21-H26 hydrogen bond interaction.
In the carboxamide moiety, there are two additional higher energy interactions (through bond coupling [21]), LPO1→σ*C2-N6 and LPO1→ σ*C2-C3, at mean values of ca.26 kcal mol−1 and ca.20 kcal mol−1, respectively, for the eq and ax conformers.
The more favourable γ dihedral angle of the phenylsulfanyl group (ca. 48°) for the ax conformer allows a reasonable 3pS-πPh* orbital overlap that contributes to a larger stabilisation of the ax conformer with respect to the eq one [LPS4→π*C20-C21 orbital interaction of ca. 8.3 kcal mol−1 (ax) and ca.2.7 kcal mol−1 (eq)].
The O[1]…H[11] short contacts are responsible for the weak LPO1→σ*C10-H11 orbital interactions of ca. 1.1 kcal mol−1 that stabilise to the same extent both conformers.
It should be pointed out that the eq conformer is stabilised through two weak LPO1→σ*S4-C20 and σC3-S4→σ*C2-N6 orbital interactions whose delocalisation energy values are ca. 0.9 kcal mol−1 and 3.4 kcal mol−1, respectively.
The suitable geometry of the ax conformer allows the occurrence of superjacent LPS4→ π*C2-O1 orbital interaction [22] whose value is 2.0 kcal mol−1, which is absent for the eq conformer. Moreover, in the [O=C-C-S] moiety there are three π(π*)/σ(σ*) orbital interactions: πC2=O1→σ*C3S4, σC3-S4→π*C2=O1 and π*C2=O1→σ*C3-S4 whose energy values progressively decrease as the π/σ overlap decreases, i.e., they present the maximum value for the ax conformer and the minimum one for the eq one. In fact, the sum of the energies of these interactions displays the value ca.11.4 kcal mol−1 and ca. 2.1 kcal mol−1 for the ax and eq conformers, respectively.
In conclusion, since the total sum of the delocalisation energies of the selected interactions has the maximum value of ca. 135 kcal mol−1 for the ax conformer and significantly decreases to ca. 107 kcal mol−1 for the eq one, the NBO analysis and the trend of the electrostatic interactions are in agreement with the higher relative abundance of the ax (ca. 97%) conformer with respect to the eq one, found in the gas phase, whose computed carbonyl frequencies are ca. 1713 cm−1 and 1720 cm−1, respectively.
However, there is no matching between the IR experimental and the theoretical gas phase data. As pointed out above, since a reversal of the IR carbonyl frequencies has been observed experimentally in solution, the higher frequency component has been ascribed to the ax conformer, while the lower frequency component to the eq one. This behaviour may be rationalised as follows.
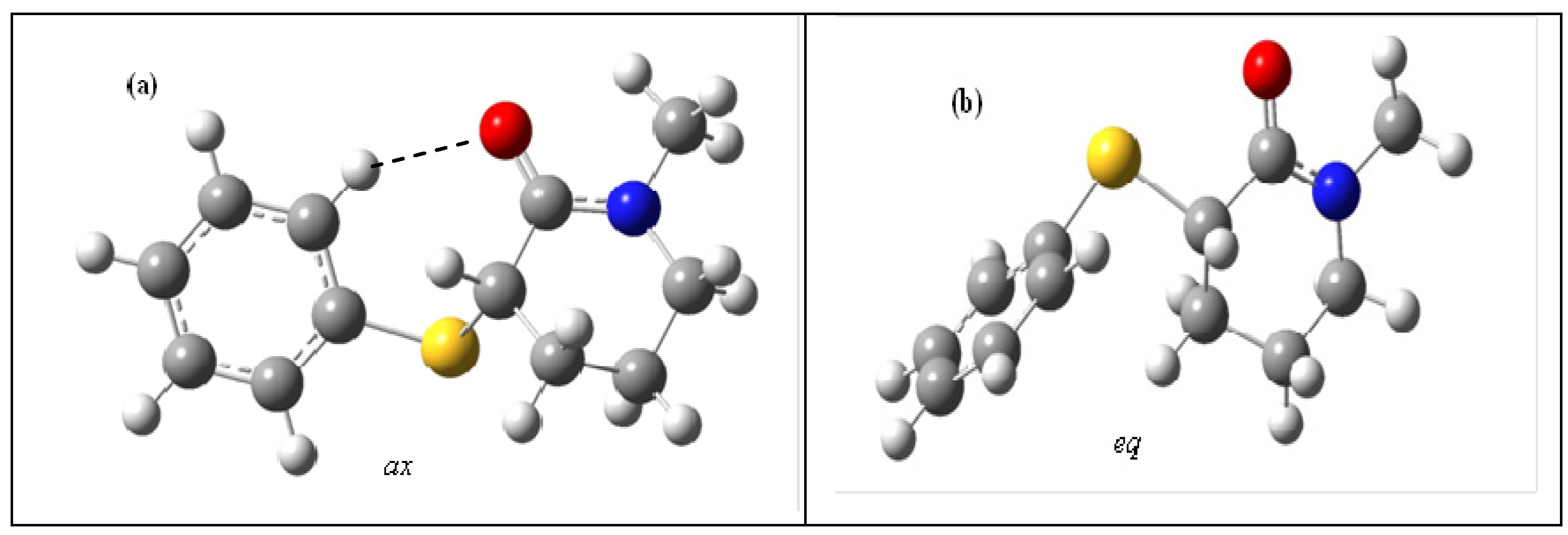
A close inspection of the ax (a) and eq (b) conformers in the gas phase (Figure 2 and Scheme 2) shows that the eq conformer carbonyl oxygen atom is free for solvation, while the hydrogen bond that takes place between the ortho-phenylsulfanyl hydrogen atom and the carbonyl oxygen atom lone pair in the ax conformer partially prevents the solvent molecules' approach to the carbonyl oxygen atom.
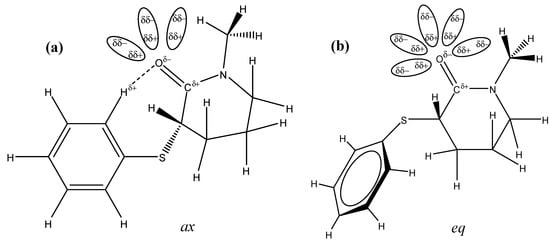
Scheme 2.
Differential solvent effect for 4 showing that the carbonyl oxygen atom in the axial conformer (a) is partially hindered for the approach of the solvent molecules due to [H(o-Ph)…O(CO)] hydrogen bond in comparison to the equatorial conformer (b) for which the carbonyl oxygen atom is free for salvation.
Scheme 2.
Differential solvent effect for 4 showing that the carbonyl oxygen atom in the axial conformer (a) is partially hindered for the approach of the solvent molecules due to [H(o-Ph)…O(CO)] hydrogen bond in comparison to the equatorial conformer (b) for which the carbonyl oxygen atom is free for salvation.
Therefore, the larger solvation at the carbonyl oxygen atom in the more polar eq conformer contributes to the decrease of the carbonyl bond order and, as a consequence, lowers its frequency to a larger extent than that of the ax conformer.
X-ray single crystal analysis of 2 showed that in the solid state this compound assumes the equatorial geometry with respect to the torsion angle α (Scheme 1) and the 2-piperidone ring, similar to the gas phase of 4, displays a slightly distorted half-chair conformation (Table 2 and Figure 3).
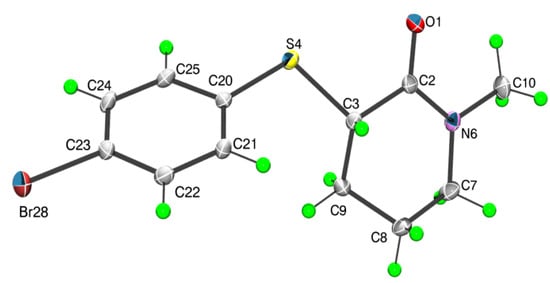
Figure 3.
The molecular structure of 2 showing atom labelling scheme and displacement ellipsoids at the 50% probability level (arbitrary spheres for the H-atoms).
Figure 3.
The molecular structure of 2 showing atom labelling scheme and displacement ellipsoids at the 50% probability level (arbitrary spheres for the H-atoms).
The absolute values of the relevant torsional angles (α-ω’) in the solid state of 2 (Table 2) are similar to the corresponding torsional angles in the gas phase of the equatorial conformer of 4, except for the absolute value of the γ angle of ca. 155° for 2, which differs from the γ angle of ca. 73° for 4. The larger γ torsional angle for 2 allows the hydrogen bonding between the π phenyl orbital and the methylene [H14] atom (Figure 4).
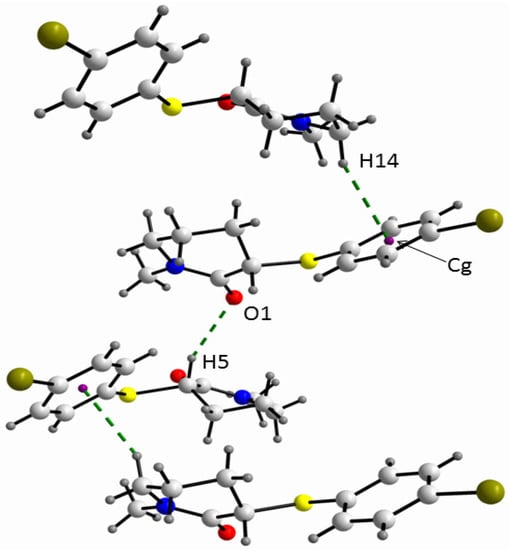
Figure 4.
A view of the supramolecular array of 2 where the C-H…O and C-H….πPh interactions are shown.
Figure 4.
A view of the supramolecular array of 2 where the C-H…O and C-H….πPh interactions are shown.
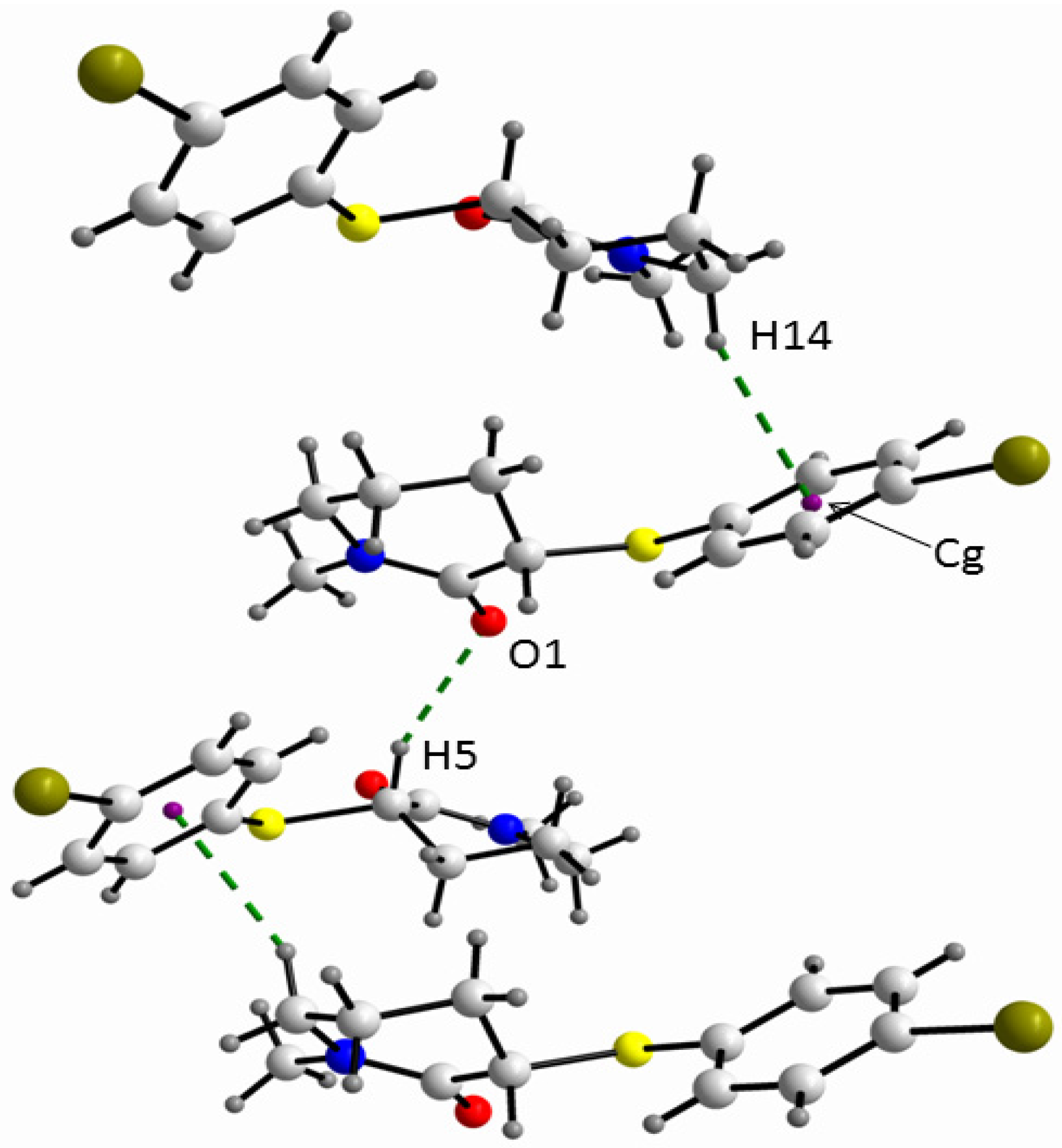
In fact, in order to obtain the largest energy gain from the crystal packing, the molecules of the equatorial conformer of 2 are stabilised in the crystal through dipole moment coupling along with two hydrogen bond interactions in a helical fashion, i.e., C-H…O (C3i-H5i…O1: C3i…O1 = 3.346(3), H5i…O1 = 2.43 Å; C3i-H5i…O1 = 155°; symmetry operation i = 1/2 + x, 1/2-y, 1-z) and C-H…π (C7…Cgii = 3.618(4), H14…Cgii = 2.72 Å; C7-H14…Cgii = 155°; symmetry operation ii = x − 1/2, −1/2−y, 1 − z). Additionally, similar to the gas phase of 4, in the crystal of 2 an intramolecular short contact [C10-H11...O1: C...O = 2.676(4); H…O = 2.23 Å; C-H...O = 107°] was found.
3. Experimental
3.1. Materials
All solvents for IR measurements were spectrograde and used without further purification. The 3-[4′-substituted phenylsulfanyl)]-1-methyl-2-piperidones 1–3, 5 and 6 are new compounds. Derivatives 2–6 were obtained from the reaction of 1-methyl-2-piperidone (a) with lithium diisopropylamide (b), in tetrahydrofuran (THF), followed by the addition of the THF solution of 4-substituted diphenyl disulphide (c), in the presence of hexamethylphosphoramide (d), in the molar ratio of 1(a):2(b):1(c):1(d) as described in the literature for 4 [23]. The appropriate diphenyl disulphide was prepared by the reaction of 4-substituted-thiophenol with bromine in dichloromethane on hydrated silica gel support [24]. The pure phenylsulfanyl piperidones 2–6 were obtained in 20–45% yield. The 4′-[nitrophenylsulfanyl]-1-methyl-2-piperidone 1 was obtained as follows: initially, the THF solution of the lithium enolate of 1-methyl-2-piperidone obtained as described above [23] reacted with the tosylate chloride [25] in a 1:1 ratio to obtain the 3-chloro-1-methyl-2-piperidone. This crude product was allowed to react with an ethanolic solution of 4-nitrothiophenol in the presence of equimolar proportion of potassium hydroxide. After usual workup and recrystallization from ethanol-water, the pure compound 1 was obtained in 24% yield. Suitable crystals for X-ray analysis for 2 were obtained by vapour diffusion from chloroform/n-hexane at 283K. The 1H- and 13C-NMR data for compounds 1–6 are collated in Table 6 and elemental analysis results are shown in Table 7.
3.2. IR Measurements
The IR spectra were obtained on a FTIR Michelson-Bomem-MB100 spectrophotometer, with 1.0 cm-1 resolution. The carbonyl region (1800–1600 cm−1) was recorded for n-hexane, carbon tetrachloride, chloroform, dichloromethane and acetonitrile solutions, at a concentration of 2.0 × 10−2 mol dm−3, using a 0.519 mm sodium chloride cell. The carbonyl first overtone region (3500–3100 cm−1) was recorded for carbon tetrachloride and chloroform solutions (2.0 × 10−2 mol dm−3) using a 1.00 cm quartz cell. The overlapped carbonyl bands (fundamental and first overtone) were deconvoluted by means of the Grams/32 curve-fitting program, version 4.04 Level II [26]. The populations of the axial and equatorial conformers were estimated from the maximum of each component of the resolved carbonyl doublet expressed in percentage of absorbance, assuming equal molar absorptivity coefficients for all the studied compounds 1–6, in each solvent.

Table 6.
Physical, 1H- and 13C-NMR data for 3-[(4′-substituted-phenyl)sulfanyl]-1-methyl-2-piperidones 1–6.
| Compd. | Y | Mp (°C) | 1H- and 13C-NMR a |
|---|---|---|---|
| 1 | NO2 | 77–78 | 1H-NMR: 8.14–8.12 (m, 2H), 7.64–7.63 (m, 2H), 4.06 (t, 1H, J = 5.5 Hz), 3.41–3.32 (m, 2H), 2.99 (s, 3H), 2.30–2.27 (m, 1H), 2.09–2.03 (m, 2H), 1.92–1.88 (m, 1H). 13C-NMR: 167.83, 146.07, 145.77, 128.67, 123.86, 49.82, 46.90, 35.36, 28.68, 20.76. |
| 2 | Br | 64–64 | 1H-NMR: 7.44–7.40 (m, 4H), 3.82 (t, 1H, J = 5.7Hz), 3.33–3.26 (m. 2H), 2.96 (s, 3H), 2.11–2.04 (m, 2H), 1.99–1.94 (m, 1H), 1.82–1.77 (m, 1H). 13C-NMR: 168.36, 134.27, 134.07, 132.19, 121.82, 50.06, 49.02, 35.47, 28.83, 20.59. |
| 3 | Cl | b | 1H-NMR: 7.53–7.51 (m, 2H), 7.30–7.29 (m, 2H), 3.84 (t, 1H, J = 5.7Hz), 3.36–3.28 (m, 2H), 2.99 (s, 3H), 2.17–2.08 (m, 2H), 2.02–1.96 (m, 1H), 1.83–1.80 (m, 1H) 13C-NMR: 168.19, 133.77, 133.64, 133.33, 129.06, 49.86, 48.96, 35.27, 28.62, 20.38. |
| 4 | H | 38–39 | 1H-NMR: 7.56–7.54 (m, 2H), 7.32–7.28 (m, 2H), 7.26–7.24 (m, 1H), 3.88 (t, 1H, J = 5.0 Hz), 3.30–3.24 (m, 2H), 2.97 (s, 3H), 2.12–2.07 (m, 2H), 1.98–1.94 (m, 1H), 1.77–1.74 (m, 1H). 13C-NMR: 168.47, 134.96, 132.61, 129.12, 127.61, 50.08, 49.03, 35.44, 28.75, 20.46. |
| 5 | CH3 | 44–45 | 1H-NMR: 7.47–7.46 (m, 2H), 7.15–7.13 (m, 2H), 3.83 (t, 1H, J = 5,5Hz), 3.34–3.25 (m, 2H), 2.98 (s, 3H), 2.35 (s, 3H), 2.13–2.05 (m, 2H), 1.96-1.92 (m, 1H), 1.80–1.75 (m, 1H) 13C-NMR: 168.37, 137.77, 133.25, 130.79, 129.71, 49.89, 49.21, 35.23, 28.35, 21.12, 20.12. |
| 6 | OCH3 | b | 1H-NMR: δ (ppm): 7.51–7.49 (m, 2H), 6.85–6.83 (m, 2H), 3.79 (s, 3H), 3,71 (t, 1H, J = 6,0Hz), 3.25–3,23 (m, 2H), 2.94 (s, 3H), 2.10–2,01 (m, 2H), 1.94–1,89 (m, 1H), 1.75–1,71 (m, 1H). 13C-NMR: 168.36, 159.85, 135.93, 124.61, 114.50, 55.31, 49.89, 49.80, 35.20, 28.28, 20.12 |
a1H- and 13C- chemical shifts in ppm relative to TMS and coupling constants in Hz at 500 MHz. in CDCl3; b liquid at room temperature; c From [4]: b.p 130–133° / 0.05 Torr.

Table 7.
Elemental analysis data for 3-[(4′-substituted-phenyl)sulfanyl]-1-methyl-2-piperidones 1–6.
| Compd | Y | Molecular formula | Analysis (%) | |||
|---|---|---|---|---|---|---|
| C | H | N | ||||
| 1 | NO2 | C12H14N2O3S | Calc.Found | 54.1254.34 | 5.305.23 | 10.5210.46 |
| 2 | Br | C12H14BrNOS | Calc. Found | 48.0147.91 | 4.704.77 | 4.674.79 |
| 3 | Cl | C12H14ClNOS | Calc. Found | 56.3556.26 | 5.525.22 | 5.485.27 |
| 4 | H | C12H15NOS | Calc. Found | 65.1265.12 | 6.836.56 | 6.336.46 |
| 5 | CH3 | C13H17NOS | Calc. Found | 66.3466.23 | 7.287.20 | 5.956.01 |
| 6 | OCH3 | C13H17NO2S | Calc. Found | 62.1262.13 | 6.826.81 | 5.575.55 |
3.3. X-ray Measurements
Crystal data for C12H14BrNOS (2): M = 300.21, T = 290(2) K, orthorhombic, P212121, a = 7.3328(5), b = 7.7903(6), c = 21.502(2)Å, V = 1228.30(17) Å3, Z = 4, Dx = 1.623 g cm−3, F(000) = 608, λ (Mo Kα) = 0.71073, μ = 3.495 mm−1, R = 0.0275.
3.3.1. Data Collection and Processing
Data were collected on a Bruker APEX-II CCD diffractometer using Mo Kα radiation so that θmax = 25.4°, no. of unique data = 2229, no. of parameters = 146, R (1964 data with I ≥ 2σ(I)) = 0.0275, wR (all data) = 0.0663. The structure was solved by direct methods [27] and refined with anisotropic displacement parameters for non- hydrogen atoms. The H atoms were geometrically placed (C—H = 0.93–0.98 Å) and refined as riding with Uiso(H) = 1.2–1.5Ueq(C). The weighting scheme used was w = 1/[σ2(Fo2) + 0.0193P2 + 0.000P] where P = (Fo2 + 2Fc2)/3) with SHELXL-97 [28] on F2. The programs WinGX [29], ORTEP3 for Windows [30], PLATON [31], MarvinSketch 5.1.10 [32] and DIAMOND [33] were used for geometric calculations and to prepare crystallographic material for publication and depositing. Crystallographic data for the structural analysis have been deposited with the Cambridge Crystallographic Data Centre as CCDC 934425. Copies of this information may be obtained free of charge on application to CCDC, 12 Union Road, Cambridge CB2 1EZ, UK (fax: 44 1223 336 033; e-mail: deposit@ccdc.cam.ac.uk or www: http://www.ccdc.cam.ac.uk).
3.4. Theoretical Calculations
All calculations were carried out (at 298 K) using methods and basis sets implemented in the GAUSSIAN package of programs (G03.E01) [34]. Full geometry optimisations (GJF files for the eq and ax conformers of 4 are available in the Supplementary material as eq.gjf and ax.gjf) and analytical vibrational frequency calculations were performed on all the orientations with respect to the carbonyl group resultant from a systematic conformational search using the ab initio Hartree–Fock method with the 3–21G basis set [35,36,37,38,39], allowing complete relaxation of all internal parameters. The obtained conformations were refined using B3LYP and MP2 methods with the 6-31+G(d,p) basis set [35,36,37,38,39]. Frequency analyses were carried out to verify the nature of the minimum state of all the stationary points obtained. To estimate the solvation effects on the relative stability of the most relevant conformers, a full optimisation and frequency calculations were done starting from the gas-phase optimised structures using the PCM model [40] at the B3LYP/6-31+G(d,p) level as implemented in the GAUSSIAN 03. Additionally, the solvent effect was estimated by PCM single-point calculations MP2/6-31+G(d,p) [41] using the corresponding gas-phase optimised structures. The NBO 3.1 program [19] was used as implemented in the GAUSSIAN 03 package, and the reported NBO delocalisation energies (E2) are those given by second-order perturbation theory. The partial atomic charges were calculated using a grid based method (ChElPG) [42].
4. Conclusions
The preferred conformations of some 3-(4′-substituted phenylsulfanyl)-1-methyl-2-piperidones 1–6 bearing as substituents NO2 (compound 1), Br (compound 2), Cl (compound 3), H (compound 4) Me (compound 5) and OMe (compound 6) were determined by νCO IR analysis, B3LYP/6-31+G(d,p) and PCM calculations along with NBO analysis (for 4) and X-ray diffraction (for 2). Theoretical data indicated the existence of two stable conformations. The 2-piperidone ring of the title compounds assumes a slightly distorted half-chair geometry, while the phenylsulfanyl substituent adopts the almost axial (ax) and equatorial (eq) geometries with respect to the carbonyl group. The ax conformer is the most stable (ca. 97%), the least polar, and exhibits a lower νCO frequency.
Unusually, the higher νCO frequency component can be ascribed to the ax conformer in solution of non-polar (C6H14 and CCl4) and polar solvents (CHCl3 and CH2Cl2). As far as the relative abundance is concerned, the ax conformer component progressively decreases with respect to the eq one as the relative permittivity increases, and is no longer detectable in the most polar solvent CH3CN.
The PCM method was applied to derivative 4 to fully optimise the ax and eq conformations in all solvents. The torsional angles α-ω’ of the ax and eq conformers remain practically the same going from the gas phase to solvents of increasing relative permittivity.
The progressive decrease of the ax/eq population ratio as the solvent polarity increases is in line with the experimental IR trend. Moreover, the computed PCM νCO frequencies of the two conformers do not significantly differ in the non-polar solvents. In contrast, the νCO frequency of the eq conformer becomes progressively smaller than that of the ax one as the solvent polarity increases. These results further support the experimental IR frequency assignments. The lower νCO IR frequency of the eq conformer in solution may be rationalised by the analysis of the computed geometries, which indicate that in the eq conformer the carbonyl oxygen atom is free for solvation, while in the ax conformer the presence of the [OCO…Ho-Ph] hydrogen bond partially prevents the solvent molecules' approach to the carbonyl oxygen atom. Thus, the larger solvation at the carbonyl oxygen atom of the most polar eq conformer decreases the carbonyl bond order and consequently its frequency to a larger extent than in the ax conformer.
The sum of the energy contributions of the orbital interactions (NBO analysis) and the electrostatic interactions correlates well with the populations and the νCO frequencies of both conformers in the gas phase. The smaller contribution of the LPN→π*CO orbital interaction for the eq conformer with respect to the ax conformer is in line with the smaller contribution of the [−O‒C=N+] canonical form to the resonance hybrid due to the strong Repulsive Field Effect between the Cδ+=Oδ− and Cδ+-Sδ− dipoles, which destabilises the eq conformer and increases its νCO frequency relative to that of the ax conformer.
X-ray single crystal analysis of 2 indicates that this compound assumes, in the solid, the most polar eq geometry with respect to the [O=C-C-SPh] moiety and that the 2-piperidone ring displays a slightly distorted half-chair conformation. In order to obtain the largest energy gain from the crystal packing, the molecules of the eq conformer of 2 are arranged in the solid through a helical fashion due to hydrogen bond interactions, i.e., C(3)-H(5)…O(1) and C(7)-H(14)…π(Ph).
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/18/7/7492/s1.
Acknowledgments
The Brazilian authors thank the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for financial support of this research and for a fellowship to E.V., the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for scholarships to C.R.C.Jr., J.M.M.S., D.N.S.R. and B.C. and fellowships to P.R.O and J.Z-S, and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior CAPES (808/2009) to J.Z-S and for a scholarship to D.N.S.R.. The authors thank Prof. R.H.A. Santos (IQSC-USP) for the X-ray data collection. The Italian author thanks MIUR (PRIN 2009) for financial support.
Conflicts of Interest
The authors declare no conflict of interest.
References and Notes
- Vinhato, E.; Olivato, P.R.; Zukerman-Schpector, J.; Dal Colle, M. Conformational Analysis of some N,N-diethyl-2-[(4′-substituted) phenylthio] acetamides. Spectrochim. Acta Part A 2013, submitted. [Google Scholar]
- Warshel, A.; Levitt, M.; Lifson, S. Consistent Force Field for Calculation of Vibrational Spectra and Conformations of Some Amides and Lactam Rings. J. Mol. Spectrosc. 1970, 33, 84–99. [Google Scholar] [CrossRef]
- Treschanke, L.; Rademacher, P. Electronic structure and conformational properties of the amide linkage: Part 1. Geometric and electronic structure of lactams as determined by MNDO calculations. J. Mol. Struct. (Theochem) 1985, 122, 35–45. [Google Scholar] [CrossRef]
- Kuze, N.; Funahashi, H.; Ogawa, M.; Tariji, H.; Ohta, Y.; Usami, T.; Sakaizumi, T.; Ohashi, O. Microwave Spectrum and Molecular Conformation of delta-Valerolactam. J. Mol. Spectrosc. 1999, 198, 381–386. [Google Scholar] [CrossRef]
- Boudreault, N.; Ball, R.G.; Bayly, C.; Bernstein, M.A.; Leblanc, Y. Conformational analysis of δ-lactams. Tetrahedron 1994, 50, 7947–7968. [Google Scholar] [CrossRef]
- Romers, C.; Rutten, E.W.M.; van Driel, C.A.A.; Sanders, W.W. a-chloro-δ-valerolactam. Acta Cryst. 1967, 22, 893–899. [Google Scholar] [CrossRef]
- Zukerman-Schpector, J.; Olivato, P.R.; Cerqueira, C.R., Jr.; Vinhato, E.; Tiekink, E.R.T. 1-Methyl-3-phenylsulfonyl-2-piperidone. Acta Cryst. 2008, E64, o835–o836. [Google Scholar]
- Olivato, P.R.; Santos, J.M.M.; Cerqueira, C.R., Jr.; Vinhato, E.; Zukerman-Schpector, J.; Ng, S.W.; Tiekink, E.R.T.; Dal Colle, M. Conformational preferences for some 3-(4´-substituted phenylsulfonyl)-1-methyl-2-piperidones through spectroscopic and theoretical studies. J. Mol. Struct. 2012, 1028, 97–106. [Google Scholar]
- Lide, D.R. (Ed.) CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 2005.
- Colthup, N.B.; Daly, L.H.; Wiberley, S.E. Introduction to Infrared and Raman Spectroscopy, 3rd ed.; Academic Press: New York, NY, USA, 1990; pp. 27–33. [Google Scholar]
- Bellamy, L.J. Advances in Infrared Group Frequencies; Chapman and Hall: London, UK, 1975; pp. 141–142. [Google Scholar]
- Gaset, A.; Lafaille, A.; Verdier, A.; Lattes, A. Infrared spectra of α-aminoketones: conformational study and evidence of an enol form. Bull. Soc. Chim. Fr. 1968, 4108–4112. [Google Scholar]
- Jones, D.; Modelli, A.; Olivato, P.R.; Dal Colle, M.; De Palo, M.; Distefano, G. Ab initio and electron spectroscopy study of carbonyl derivatives. J. Chem. Soc. Perkin Trans. 2 1994, 7, 1651–1656. [Google Scholar]
- Mujika, J.I.; Matxain, J.M.; Eriksson, L.A.; Lopez, X. Resonance structures of the amide bond: The advantages of planarity. Chem. Eur. J. 2006, 12, 7215–7224. [Google Scholar] [CrossRef]
- MP2/6-31G+(d,p) calculations for 4 in the gas phase are presented in Table S1 (Supplementary information).
- Single point PCM calculations at the MP2/6-31G+(d,p) level for 4 in solvents of increasing relative permissivity are presented in Table S2.
- Mulliken charges are presented in Table S3.
- Katritzky, A.R.; Topsom, R. Infrared intensities: A guide to intramolecular interactions in conjugated systems. Chem. Rev. 1977, 77, 639–658. [Google Scholar] [CrossRef]
- Glendening, E.D.; Reed, A.E.; Carpenter, J.E.; Weinhlod, F. NBO Version 3.1 (Implemented in Gaussian 03 package of Programs). Available online: http://archive.osc.edu/supercomputing/software/apps/gaussian03.shtml (accessed on 25 June 2013).
- Occupancies for donor and acceptor orbital for the ax and eq conformers of 4 are presented in TableS4.
- Levin, C.C.; Hoffmann, R.; Hehre, W.J.; Hudec, J. Orbital Interaction in Amino-ketones. J. Chem. Soc. Perkin 2 1973, 210–220. [Google Scholar]
- Eisenstein, O.; Ahn, N.T.; Devaquet, J.A.; Cantacuzene, J.; Salem, L. Lone pairs in organic molecules: Energetic and orientational non-equivalence: Stereochemical consequences. Tetrahedron 1974, 30, 1717–1723. [Google Scholar] [CrossRef]
- Zoretic, P.A.; Soja, P. Sulfenylation and selenenylation of lactams. J. Org. Chem. 1976, 41, 3587–3589. [Google Scholar] [CrossRef]
- Hashmat, A.M.; McDermott, M. Oxidation of thiols to disulfides with molecular bromine on hydrated silica gel support. Tetrahedron Lett. 2002, 43, 6271–6273. [Google Scholar] [CrossRef]
- Brummond, K.M.; Gesenberg, K.D. Alpha-chlorination of ketones using p-toluenesulfonyl chloride. Tetrahedron Lett. 1999, 40, 2231–2234. [Google Scholar] [CrossRef]
- Grams/32 curve-fitting program, version 4.04 Level II; Galactic Industries Corporation: Salem, NH, USA, 1991–1998.
- Altomare, A.; Cascarano, G.; Giacovazzo, C.; Guagliardi, A.J. Completion and Refinement of Crystal Structures with SIR92. J. Appl. Crystallogr. 1993, 26, 343–350. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar]
- Farrugia, L.J. WinGX suite for small-molecule single-crystal crystallography J. Appl. Crystallogr. 1999, 32, 837–838. [Google Scholar] [CrossRef]
- Farrugia, L.J. ORTEP-3 for Windows - A version of ORTEP-III with a Graphical User Interface (GUI) J. Appl. Crystallogr. 1997, 30, 565. [Google Scholar] [CrossRef]
- Spek, A.L. PLATON, A Multipurpose Crystallographic Tool. J. Appl. Crystallogr. 2003, 36, 7–13. [Google Scholar] [CrossRef]
- ChemAxon, Marvin 5.1.10. Available online: http://www.chemaxon.com (accessed on 25 June 2013).
- Brandenburg, K.; Putz, H. Diamond—Crystal and Molecular Structure Visualization; Crystal Impact: Bonn, Germany, 2012. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A., Jr.; Vreven, T.; Kudin, K.N.; Burant, J.C.; et al. Gaussian 03; Revision E.01; Gaussian Inc.: Wallingford, CT., USA, 2004. [Google Scholar]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. 1988, A 38, 3098–3100. [Google Scholar]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti Correlation-Energy Formula into a Functional of the Electron Density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Becke, A.D. Density-Functional Thermochemistry. III. The Role of Exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Stephens, P.J.; Devlin, F.J.; Chabalowski, C.F.; Frisch, M.J. Ab Initio Calculation of Vibrational Absorption and Circular Dichroism Spectra Using Density Functional Force Fields. J. Phys. Chem. 1994, 98, 11623–11627. [Google Scholar]
- Hehre, W.J.; Radom, L.; Schleyer, P.V.R.; Pople, J.A. Ab Initio Molecular Orbital Theory; Wiley: New York, NY, USA, 1986. [Google Scholar]
- Cossi, M.; Rega, N.; Scalmani, G.; Barone, V. New developments in the polarizable continuum model for quantum mechanical and classical calculations on molecules in solution. J. Chem. Phys. 2002, 117, 43–54. [Google Scholar] [CrossRef]
- Head-Gordon, M.; Pople, J.A.; Frisch, M.J. MP2 energy evaluation by direct methods. Chem. Phys. Lett. 1988, 153, 503–506. [Google Scholar] [CrossRef]
- Breneman, C.M.; Wiberg, K.B. Determining atom-centered monopoles from molecular electrostatic potentials. The need for high sampling density in formamide conformational analysis. J. Comp. Chem. 1990, 11, 361–373. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–6 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
