Structure−Activity Relationship (SAR) Study of trans-Cinnamic Acid and Derivatives on the Parasitic Weed Cuscuta campestris
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Plant Material and Chemicals
3.2. Inhibitory Activity In Vitro Test against Cuscuta campestris Growth
3.3. Calculations and Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Riopel, J.L.; Timko, M.P. Haustorial initiation and differentiation. In Parasitic Plants; Press, M.C., Graves, J.D., Eds.; Chapman & Hall: London, UK, 1995; pp. 39–79. [Google Scholar]
- Fernández-Aparicio, M.; Delavault, P.; Timko, M.P. Management of infection by parasitic weeds: A review. Plants 2020, 9, 1184–1210. [Google Scholar] [CrossRef]
- Parker, C. Observations on the current status of Orobanche and Striga problems worldwide. Pest Manag. Sci. 2009, 65, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Nickrent, D.L. Parasitic angiosperms: How often and how many? Taxon 2020, 69, 5–27. [Google Scholar] [CrossRef]
- Dawson, J.H.; Musselman, L.J.; Wolswinkel, P.; Dörr, I. Biology and control of Cuscuta. Rev. Weed Sci. 1994, 6, 265–317. [Google Scholar]
- Lanini, W.T.; Kogan, M. Biology and management of Cuscuta in crops. Int. J. Agric. Nat. Resour. 2005, 32, 165–179. [Google Scholar] [CrossRef]
- Goldwasser, Y.; Miryamchik, H.; Sibony, M.; Rubin, B. Detection of resistant chickpea (Cicer arietinum) genotypes to Cuscuta campestris (field dodder). Weed Res. 2012, 52, 122–130. [Google Scholar] [CrossRef]
- Córdoba, E.M.; Fernández-Aparicio, M.; González-Verdejo, C.I.; López-Grau, C.; Muñoz-Muñoz, M.V.; Nadal, S. Search for resistant genotypes to Cuscuta campestris infection in two legume species, Vicia sativa and Vicia ervilia. Plants 2021, 10, 738–751. [Google Scholar] [CrossRef]
- Fernández-Aparicio, M.; Soriano, G.; Masi, M.; Carretero, P.; Vilariño-Rodríguez, S.; Cimmino, A. (4Z)-Lachnophyllum lactone, an acetylenic furanone from Conyza bonariensis, identified for the first time with allelopathic activity against Cuscuta campestris. Agriculture 2022, 12, 790–801. [Google Scholar] [CrossRef]
- Yang, X.X.; Choi, H.W.; Yang, S.F.; Li, N. A UV-light activated cinnamic acid isomer regulates plant growth and gravitropism via an ethylene receptor-independent pathway. Aust. J. Plant Physiol. 1999, 26, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.Q.; Wong, W.S.; Ye, W.C.; Li, N. Biologically active cis-cinnamic acid occurs naturally in Brassica parachinensis. Chin. Sci. Bull. 2003, 48, 555–558. [Google Scholar] [CrossRef]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin biosynthesis. Annu. Rev. Plant. Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant 2010, 3, 2–20. [Google Scholar] [CrossRef]
- Vanholme, B.; El Houari, I.; Boerjan, W. Bioactivity: Phenylpropanoids’ best kept secret. Curr. Opin. Biotechnol. 2019, 56, 156–162. [Google Scholar] [CrossRef] [PubMed]
- del Río, J.C.; Rencoret, J.; Gutiérrez, A.; Elder, T.; Kim, H.; Ralph, J. Lignin monomers from beyond the canonical monolignol biosynthetic pathway: Another brick in the wall. ACS Sustain. Chem. Eng. 2020, 8, 4997–5012. [Google Scholar] [CrossRef]
- Macías, F.A.; Mejías, F.J.; Molinillo, J.M. Recent advances in allelopathy for weed control: From knowledge to applications. Pest Manag. Sci. 2019, 75, 2413–2436. [Google Scholar] [CrossRef] [PubMed]
- Bonner, J.; Galston, A.W. Toxic substances from the culture media of guayule which may inhibit growth. Bot. Gaz. 1944, 106, 185–198. [Google Scholar] [CrossRef]
- Baziramakenga, R.; Simard, R.R.; Leroux, G.D. Effects of benzoic and cinnamic acids on growth, mineral composition, and chlorophyll content of soybean. J. Chem. Ecol. 1994, 20, 2821–2833. [Google Scholar] [CrossRef] [PubMed]
- Chon, S.U.; Kim, Y.M.; Lee, J.C. Herbicidal potential and quantification of causative allelochemicals from several Compositae weeds. Weed Res. 2003, 43, 444–450. [Google Scholar] [CrossRef]
- Haagen-Smit, S.A.J.; Went, F.W. A physiological analysis of the growth substance. In Proceedings Koninklijke Akademie van Wetenschappen the Amsterdam; University of Illinois at Urbana-Champaign: Champaign, IL, USA, 1935; Volume 38, pp. 852–857. [Google Scholar]
- Koepfli, J.B.; Thimann, K.B.; Went, F.W. Plant hormones: Structure and physiological activity. I. J. Biol. Chem. 1938, 122, 763–780. [Google Scholar] [CrossRef]
- Went, F.W. Analysis and integration of various auxin effects. II. In Proceedings Koninklijke Akademie van Wetenschappen the Amsterdam; University of Illinois at Urbana-Champaign: Champaign, IL, USA, 1939; Volume 42, pp. 731–739. [Google Scholar]
- Steenackers, W.; Klíma, P.; Quareshy, M.; Cesarino, I.; Kumpf, R.P.; Corneillie, S.; Araújo, P.; Viaene, T.; Goeminne, G.; Nowack, M.K.; et al. cis-Cinnamic acid is a novel, natural auxin efflux inhibitor that promotes lateral root formation. Plant Physiol. 2017, 173, 552–565. [Google Scholar] [CrossRef]
- Hiradate, S.; Morita, S.; Furubayashi, A.; Fujii, Y.; Harada, J. Plant growth inhibition by cis-cinnamoyl glucoside and cis-cinnamic acid. J. Chem. Ecol. 2005, 31, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.; Wand Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Robles, A.; Cala Peralta, A.; Zorrilla, J.G.; Soriano, G.; Masi, M.; Vilariño-Rodríguez, S.; Cimmino, A.; Fernández-Aparicio, M. Identification of structural features of hydrocinnamic acid related to its allelopathic activity against the parasitic weed Cuscuta campestris. Plants 2022, 11, 2846. [Google Scholar] [CrossRef] [PubMed]
- Abe, M.; Nishikawa, K.; Fukuda, H.; Nakanishi, K.; Tazawa, Y.; Taniguchi, T.; Park, S.Y.; Hiradate, S.; Fujii, Y.; Okuda, K.; et al. Key structural features of cis-cinnamic acid as an allelochemical. Phytochemistry 2012, 84, 56–67. [Google Scholar] [CrossRef]
- Nishikawa, K.; Fukuda, H.; Abe, M.; Nakanishi, K.; Taniguchi, T.; Nomura, T.; Yamaguchi, C.; Hiradate, S.; Fujii, Y.; Okuda, K.; et al. Substituent effects of cis-cinnamic acid analogues as plant growh inhibitors. Phytochemistry 2013, 96, 132–147. [Google Scholar] [CrossRef]
- Moreno-Robles, A.; Cala Peralta, A.; Soriano, G.; Zorrilla, J.G.; Masi, M.; Vilariño, S.; Cimmino, A.; Fernández-Aparicio, M. Identification of allelochemicals with differential modes of phytotoxicity on Cuscuta campestris. Agriculture 2022, 12, 1746. [Google Scholar] [CrossRef]
- Dewick, P.M. Medicinal Natural Products: A Biosynthetic Approach; John Wiley & Sons: Chichester, UK, 2001. [Google Scholar]
- Lorenzo, P.; Reboredo-Durán, J.; Muñoz, L.; Freitas, H.; González, L. Herbicidal properties of the commercial formulation of methyl cinnamate, a natural compound in the invasive silver wattle (Acacia dealbata). Weed Sci. 2019, 68, 69–78. [Google Scholar] [CrossRef]
- Vasilakoglou, I.; Dhima, K.; Paschalidis, K.; Ritzoulis, C. Herbicidal potential on Lolium rigidum of nineteen major essential oil components and their synergy. J. Essent. Oil Res. 2013, 25, 1–10. [Google Scholar] [CrossRef]
- Khanh, T.D.; Cong, L.C.; Xuan, T.D.; Lee, S.J.; Kong, D.S.; Chung, I.M. Weed-suppressing potential of dodder (Cuscuta hygrophilae) and its phytotoxic constituents. Weed Sci. 2008, 56, 119–127. [Google Scholar] [CrossRef]
- Peralta, A.C.; Soriano, G.; Zorrilla, J.G.; Masi, M.; Cimmino, A.; Fernández-Aparicio, M. Characterization of Conyza bonariensis allelochemicals against broomrape weeds. Molecules 2022, 27, 7421. [Google Scholar] [CrossRef]
- Tice, C.M. Selecting the right compounds for screening: Does Lipinski’s Rule of 5 for pharmaceuticals apply to agrochemicals? Pest Manag. Sci. 2001, 57, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Gaertner, E.E. Studies of seed germination, seed identification, and host relationship in dodders, Cuscuta spp. Mem. Cornell Agric. Exp. Stn. 1950, 294, 1–56. [Google Scholar]
- Cala, A.; Zorrilla, J.G.; Rial, C.; Molinillo, J.M.G.; Varela, R.M.; Macías, F.A. Easy access to alkoxy, amino, carbamoyl, hydroxy, and thiol derivatives of sesquiterpene lactones and evaluation of their bioactivity on parasitic weeds. J. Agric. Food Chem. 2019, 67, 10764–10773. [Google Scholar] [CrossRef] [PubMed]
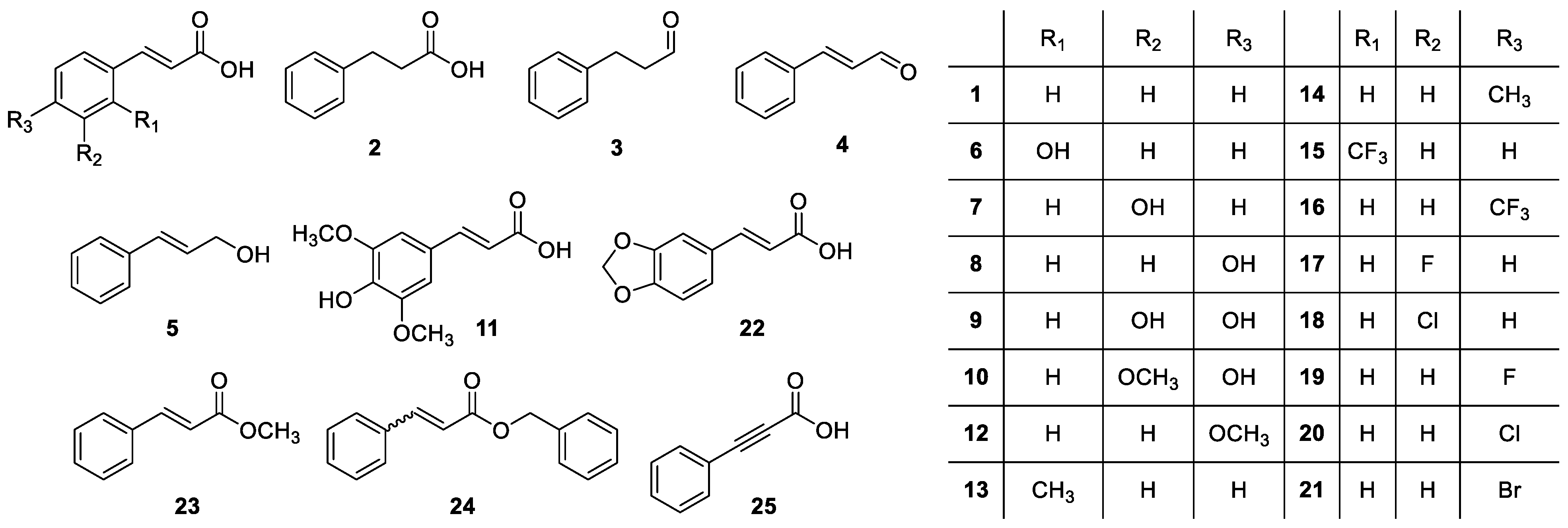



| CLogP | IC50 (μM) | CLogP | IC50 (μM) | CLogP | IC50 (μM) | |||
|---|---|---|---|---|---|---|---|---|
| 1 | 2.239 | >1000 | 11 | 1.204 | - | 21 | 3.102 | 876 |
| 2 | 1.903 | 777 | 12 | 2.158 | >1000 | 22 | 2.204 | >1000 |
| 3 | 1.873 | 497 | 13 | 2.738 | - | 23 | 2.465 | 331 |
| 4 | 2.049 | 408 | 14 | 2.738 | >1000 | 24 | 4.233 | - |
| 5 | 1.608 | >1000 | 15 | 3.122 | - | 25 | 1.785 | - |
| 6 | 1.572 | >1000 | 16 | 3.122 | 399 | |||
| 7 | 1.572 | - | 17 | 2.382 | ~1000 | |||
| 8 | 1.572 | - | 18 | 2.952 | 461 | |||
| 9 | 0.975 | - | 19 | 2.382 | ~1000 | |||
| 10 | 1.421 | - | 20 | 2.952 | 955 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreno-Robles, A.; Cala Peralta, A.; Zorrilla, J.G.; Soriano, G.; Masi, M.; Vilariño-Rodríguez, S.; Cimmino, A.; Fernández-Aparicio, M. Structure−Activity Relationship (SAR) Study of trans-Cinnamic Acid and Derivatives on the Parasitic Weed Cuscuta campestris. Plants 2023, 12, 697. https://doi.org/10.3390/plants12040697
Moreno-Robles A, Cala Peralta A, Zorrilla JG, Soriano G, Masi M, Vilariño-Rodríguez S, Cimmino A, Fernández-Aparicio M. Structure−Activity Relationship (SAR) Study of trans-Cinnamic Acid and Derivatives on the Parasitic Weed Cuscuta campestris. Plants. 2023; 12(4):697. https://doi.org/10.3390/plants12040697
Chicago/Turabian StyleMoreno-Robles, Antonio, Antonio Cala Peralta, Jesús G. Zorrilla, Gabriele Soriano, Marco Masi, Susana Vilariño-Rodríguez, Alessio Cimmino, and Mónica Fernández-Aparicio. 2023. "Structure−Activity Relationship (SAR) Study of trans-Cinnamic Acid and Derivatives on the Parasitic Weed Cuscuta campestris" Plants 12, no. 4: 697. https://doi.org/10.3390/plants12040697










