Crosstalk between DnaA Protein, the Initiator of Escherichia coli Chromosomal Replication, and Acidic Phospholipids Present in Bacterial Membranes
Abstract
:1. Introduction
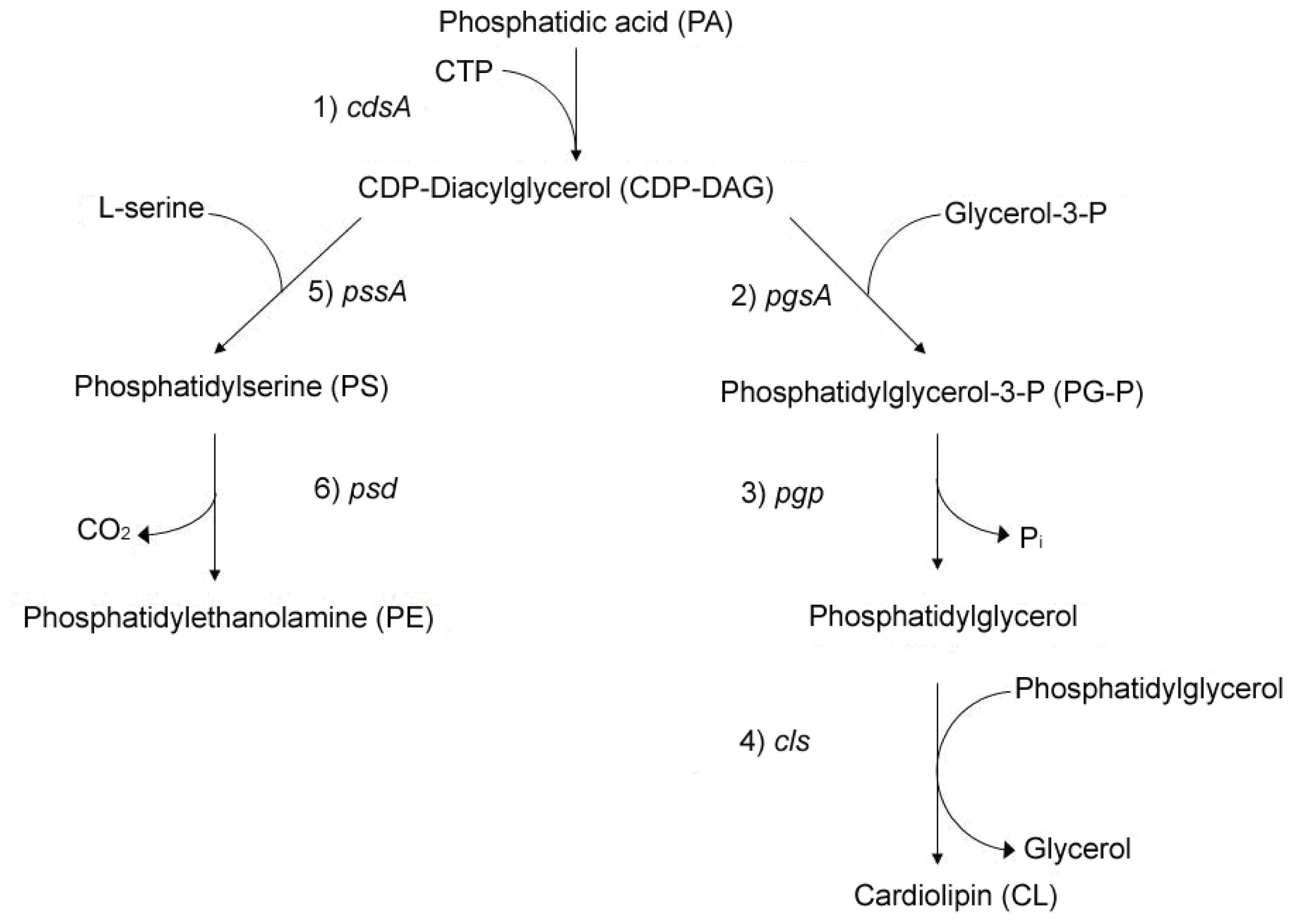
2. Linkage between Bacterial Growth and Membrane Acidic Phospholipids
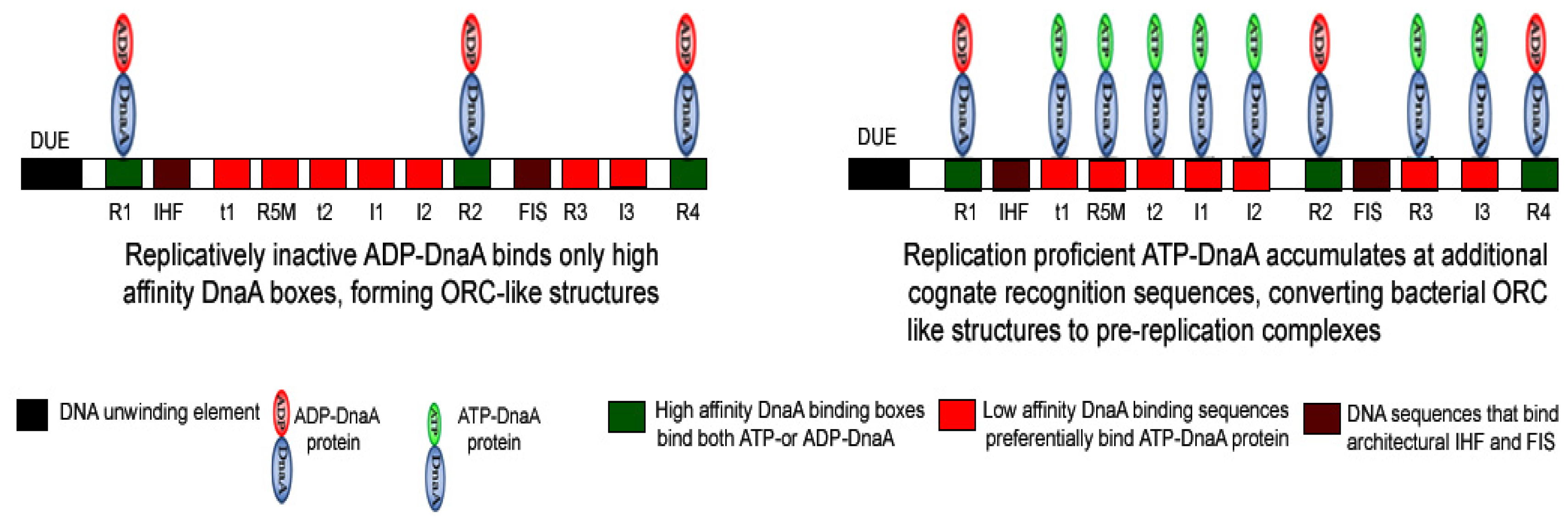
3. Importance of the Nucleotide State of DnaA Protein in Determining the Functional Status of Nucleoprotein Complex Generated at oriC
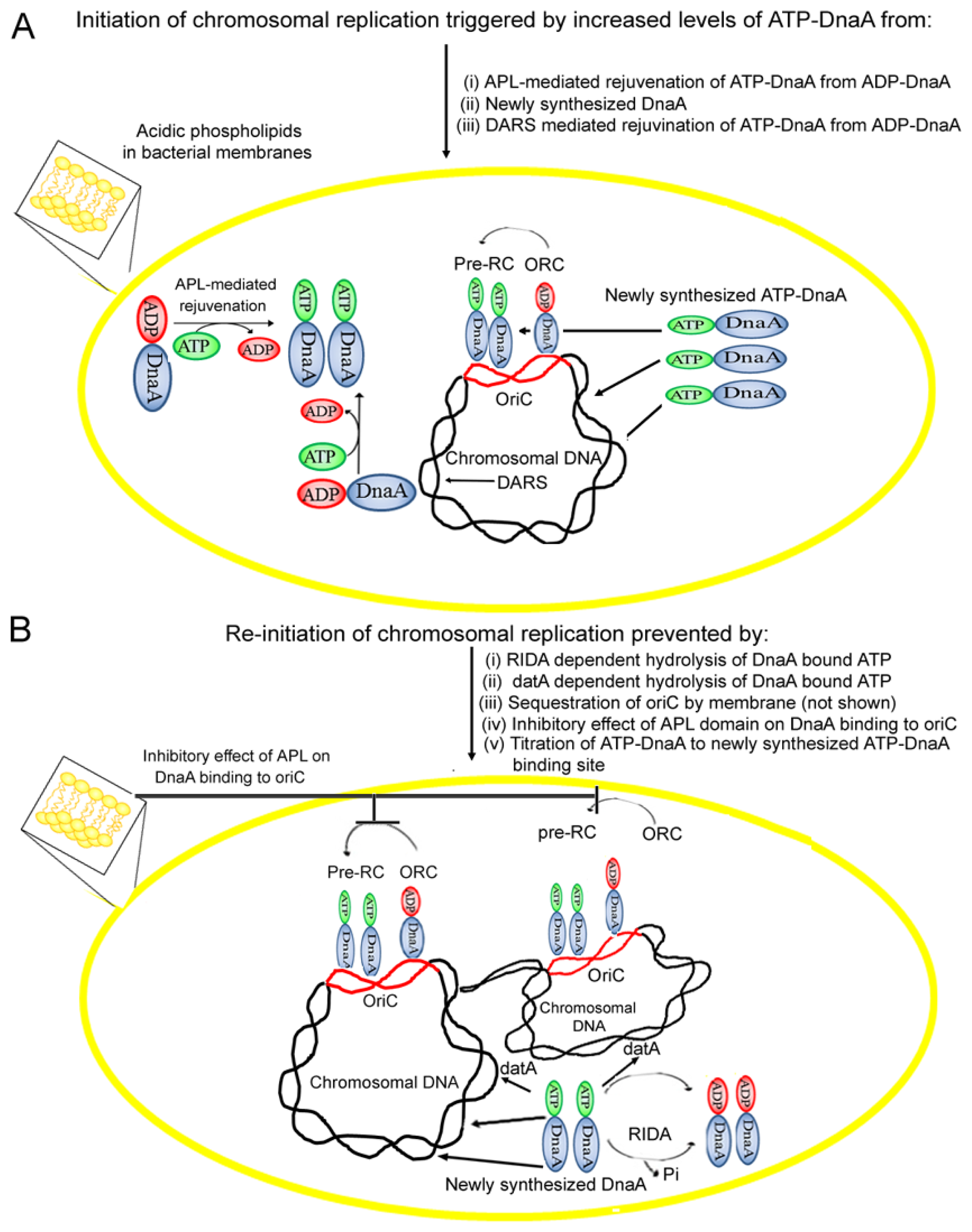
4. Acidic Phospholipids Promote Conversion of Replicatively Feeble ADP-DnaA to the Replicatively Active ATP-Form
5. Membrane Fluidity Determines the Rate of ADP to ATP-DnaA Exchange
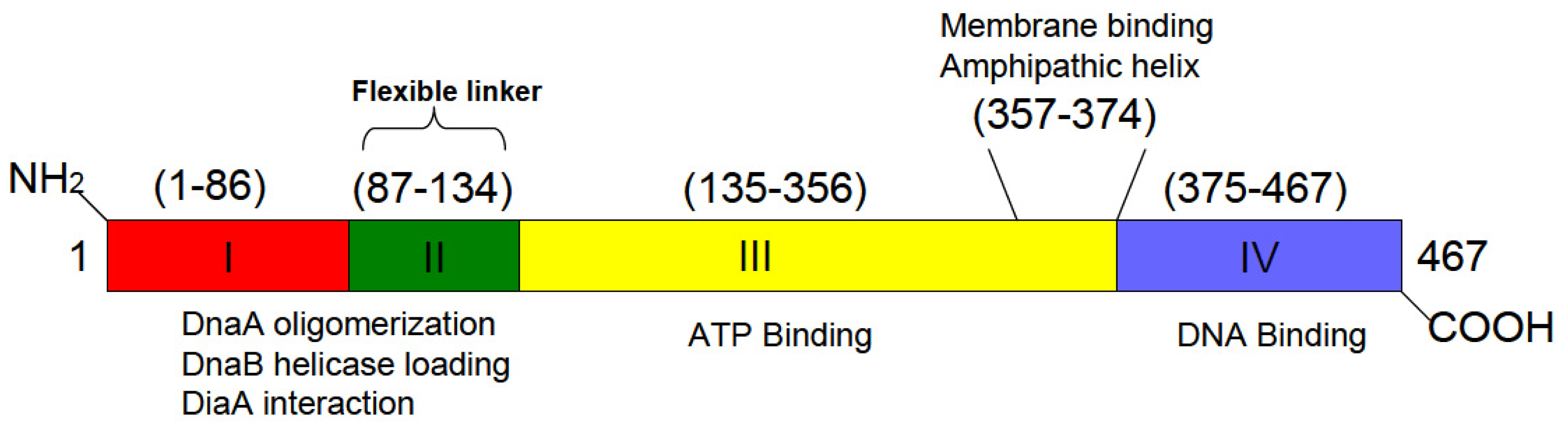
6. A Discrete Region of DnaA Associates with Fluid Bilayers Possessing Acidic Phospholipids
7. DnaA, Acidic Phospholipids, and Electrostatic Interactions
8. Acidic Phospholipids Inhibit DnaA Binding to E. coli oriC DNA
9. Cardiolipin Helps Sub-Cellular Localization of Certain Bacterial Proteins
10. Concluding Remarks
Acknowlegements
Abbreviations
| PG | Phosphatidylglycerol |
| CL | cardiolipin |
| E. coli | Escherichia coli |
| PE | phosphatidylethanolamine |
| CDP-DAG | cytosine diphosphate diacylglycerol |
| PssA | phosphatidylserine synthase |
| PS | phosphatidylserine |
| PgsA | phosphatidylglycerophosphate synthase |
| Pgp | phosphatidylglycerophosphate phosphatase |
| ClsA | cardiolipin synthase |
| B. Subtilis | Bacillus subtilis |
| PLD | phospholipase D |
| oriC | origin of chromosomal replication |
| NAO | 10-N-nonyl acridine orange |
| cSDR | constitutively stable DNA replication |
| MANT-ATP | 2′-(or-3′)-O-(N-methylanthraniloyl) adenosine 5′ tri-phosphate |
| ORC | origin recognition complex |
| pre-RC | pre-replication complex |
| RIDA | regulatory inactivation of DnaA |
| PC | phosphatidylcholine |
| DARS | DnaA reactivating sequences |
| DNAC | 3-decynoyl-N-acetylcysteamine |
| PI | phosphatidylinositol |
| GM1 | monosialotetrahexosylganglioside |
| PA | Phosphatadic acid |
| S. aureus. | Staphylococcus aureus |
Conflict of Interest
References
- Raetz, C.R. Molecular genetics of membrane phospholipid synthesis. Annu. Rev. Genet 1986, 20, 253–295. [Google Scholar]
- Kanfer, J.N.; Kennedy, E.P. Synthesis of phosphatidylserine by Escherichia coli. J. Biol. Chem 1962, 237, 270–271. [Google Scholar]
- Kanfer, J.N.; Kennedy, E.P. Metabolism and function of bacterial lipids. ii. Biosynthesis of phospholipids in Escherichia coli. J. Biol. Chem 1964, 239, 1720–1726. [Google Scholar]
- Raetz, C.R.; Dowhan, W. Biosynthesis and function of phospholipids in Escherichia coli. J. Biol. Chem 1990, 265, 1235–1238. [Google Scholar]
- Cronan, J.E. Bacterial membrane lipids: Where do we stand? Annu. Rev. Microbiol 2003, 57, 203–224. [Google Scholar]
- Ohta, A.; Obara, T.; Asami, Y.; Shibuya, I. Molecular cloning of the cls gene responsible for cardiolipin synthesis in Escherichia coli and phenotypic consequences of its amplification. J. Bacteriol 1985, 163, 506–514. [Google Scholar]
- Nishijima, S.; Asami, Y.; Uetake, N.; Yamagoe, S.; Ohta, A.; Shibuya, I. Disruption of the Escherichia coli cls gene responsible for cardiolipin synthesis. J. Bacteriol 1988, 170, 775–780. [Google Scholar]
- Tropp, B.E. Cardiolipin synthase from Escherichia coli. Biochim. Biophys. Acta 1997, 1348, 192–200. [Google Scholar]
- Depalo, N.; Catucci, L.; Mallardi, A.; Corcelli, A.; Agostiano, A. Enrichment of cardiolipin content throughout the purification procedure of photosystem II. Bioelectrochemistry 2004, 63, 103–106. [Google Scholar]
- Ventrella, A.; Catucci, L.; Mascolo, G.; Corcelli, A.; Agostiano, A. Isolation and characterization of lipids strictly associated to PSII complexes: Focus on cardiolipin structural and functional role. Biochim. Biophys. Acta 2007, 1768, 1620–1627. [Google Scholar]
- Laczkó-Dobos, H.; Ughy, B.; Tóth, S.Z.; Komenda, J.; Zsiros, O.; Domonkos, I.; Párducz, A.; Bogos, B.; Komura, M.; Itoh, S.; et al. Role of phosphatidylglycerol in the function and assembly of Photosystem II reaction center, studied in a cdsA-inactivated PAL mutant strain of Synechocystis sp. PCC6803 that lacks phycobilisomes. Biochim. Biophys. Acta 2008, 1777, 1184–1194. [Google Scholar]
- Mileykovskaya, E.; Dowhan, W. Visualization of phospholipid domains in Escherichia coli by using the cardiolipin-specific fluorescent dye 10-N-nonyl acridine orange. J. Bacteriol 2000, 182, 1172–1175. [Google Scholar]
- Matsumoto, K.; Kusaka, J.; Nishibori, A.; Hara, H. Lipid domains in bacterial membranes. Mol. Microbiol 2006, 61, 1110–1117. [Google Scholar]
- Schlame, M. Cardiolipin synthesis for the assembly of bacterial and mitochondrial membranes. J. Lipid. Res 2008, 49, 1607–1620. [Google Scholar]
- Mileykovskaya, E.; Dowhan, W. Cardiolipin membrane domains in prokaryotes and eukaryotes. Biochim. Biophys. Acta 2009, 1788, 2084–2091. [Google Scholar]
- Daum, G. Lipids of mitochondria. Biochim. Biophys. Acta 1985, 822, 1–42. [Google Scholar]
- Schlame, M.; Haldar, D. Cardiolipin is synthesized on the matrix side of the inner membrane in rat liver mitochondria. J. Biol. Chem 1993, 268, 74–79. [Google Scholar]
- De Andrade Rosa, I.; Einicker-Lamas, M.; Bernardo, R.R.; Previatto, L.M.; Mohana-Borges, R.; Morgado-Díaz, J.A.; Benchimol, M. Cardiolipin in hydrogenosomes: Evidence of symbiotic origin. Eukaryot. Cell 2006, 5, 784–787. [Google Scholar]
- Lensink, M.F.; Govaerts, C.; Ruysschaert, J.M. Identification of specific lipid-binding sites in integral membrane proteins. J. Biol. Chem 2010, 285, 10519–10526. [Google Scholar]
- Kitchen, J.L.; Li, Z.; Crooke, E. Electrostatic interactions during acidic phospholipid reactivation of DnaA protein, the Escherichia coli initiator of chromosomal replication. Biochemistry 1999, 38, 6213–6221. [Google Scholar]
- Murray, D.; Arbuzova, A.; Honig, B.; McLaughlin, S. The role of electrostatic and nonpolar interactions in the association of peripheral proteins with membranes Pept. Lipid Interact 2002, 52, 277–307. [Google Scholar]
- Mulgrew-Nesbitt, A.; Diraviyam, K.; Wang, J.Y.; Singh, S.; Murray, P.; Li, Z.H.; Rogers, L.; Mirkovic, N.; Murray, D. The role of electrostatics in protein-membrane interactions. Biochim. Biophys. Acta 2006, 1761, 812–826. [Google Scholar]
- Vorobyov, I.; Allen, T.W. On the role of anionic lipids in charged protein interactions with membranes. Biochim. Biophys. Acta 2011, 1808, 1673–1683. [Google Scholar]
- McMillin, J.B.; Dowhan, W. Cardiolipin and apoptosis. Biochim. Biophys. Acta 2002, 1585, 97–107. [Google Scholar]
- Gonzalvez, F.; Gottlieb, E. Cardiolipin: Setting the beat of apoptosis. Apoptosis 2007, 12, 877–885. [Google Scholar]
- Schug, Z.T.; Gottlieb, E. Cardiolipin acts as a mitochondrial signalling platform to launch apoptosis. Biochim. Biophys. Acta 2009, 1788, 2022–2031. [Google Scholar]
- Haines, T.H.; Dencher, N.A. Cardiolipin: A proton trap for oxidative phosphorylation. FEBS Lett 2002, 528, 35–39. [Google Scholar]
- Gohil, V.M.; Hayes, P.; Matsuyama, S.; Schägger, H.; Schlame, M.; Greenberg, M.L. Cardiolipin biosynthesis and mitochondrial respiratory chain function are interdependent. J. Biol. Chem 2004, 279, 42612–42618. [Google Scholar]
- Arias-Cartin, R.; Grimaldi, S.; Pommier, J.; Lanciano, P.; Schaefer, C.; Arnoux, P.; Giordano, G.; Guigliarelli, B.; Magalon, A. Cardiolipin-based respiratory complex activation in bacteria. Proc. Natl. Acad. Sci. USA 2011, 108, 7781–7786. [Google Scholar]
- Arias-Cartin, R.; Grimaldi, S.; Arnoux, P.; Guigliarelli, B.; Magalon, A. Cardiolipin binding in bacterial respiratory complexes: Structural and functional implications. Biochim. Biophys. Acta 2012, 1817, 1937–1949. [Google Scholar]
- Wenz, T.; Hielscher, R.; Hellwig, P.; Schägger, H.; Richers, S.; Hunte, C. Role of phospholipids in respiratory cytochrome bc(1) complex catalysis and supercomplex formation. Biochim. Biophys. Acta 2009, 1787, 609–616. [Google Scholar]
- Laskowska, E.; Kuczyńska-Wiśnik, D.; Skórko-Glonek, J.; Taylor, A. Degradation by proteases Lon, Clp and HtrA, of Escherichia coli proteins aggregated in vivo by heat shock; HtrA protease action in vivo and in vitro. Mol. Microbiol 1996, 22, 555–571. [Google Scholar]
- Rosen, R.; Biran, D.; Gur, E.; Becher, D.; Hecker, M.; Ron, E.Z. Protein aggregation in Escherichia coli: Role of proteases. FEMS Microbiol. Lett 2002, 207, 9–12. [Google Scholar]
- Minami, N.; Yasuda, T.; Ishii, Y.; Fujimori, K.; Amano, F. Regulatory role of cardiolipin in the activity of an ATP-dependent protease, Lon, from Escherichia coli. J. Biochem 2011, 149, 519–527. [Google Scholar]
- Mileykovskaya, E.; Dowhan, W. Role of membrane lipids in bacterial division-site selection. Curr. Opin. Microbiol 2005, 8, 135–142. [Google Scholar]
- Xia, W.; Dowhan, W. In vivo evidence for the involvement of anionic phospholipids in initiation of DNA replication in Escherichia coli. Proc. Natl. Acad. Sci. USA 1995, 92, 783–787. [Google Scholar]
- Zheng, W.; Li, Z.; Skarstad, K.; Crooke, E. Mutations in DnaA protein suppress the growth arrest of acidic phospholipid-deficient Escherichia coli cells. EMBO J 2001, 20, 1164–1172. [Google Scholar]
- Boeneman, K.; Crooke, E. Chromosomal replication and the cell membrane. Curr. Opin. Microbiol 2005, 8, 143–148. [Google Scholar]
- Fingland, N.; Flåtten, I.; Downey, C.D.; Fossum-Raunehaug, S.; Skarstad, K.; Crooke, E. Depletion of acidic phospholipids influences chromosomal replication in Escherichia coli. Microbiologyopen 2012, 1, 450–466. [Google Scholar]
- Heacock, P.N.; Dowhan, W. Construction of a lethal mutation in the synthesis of the major acidic phospholipids of Escherichia coli. J. Biol. Chem 1987, 262, 13044–13049. [Google Scholar]
- Heacock, P.N.; Dowhan, W. Alteration of the phospholipid composition of Escherichia coli through genetic manipulation. J. Biol. Chem 1989, 264, 14972–14977. [Google Scholar]
- Pluschke, G.; Hirota, Y.; Overath, P. Function of phospholipids in Escherichia coli. Characterization of a mutant deficient in cardiolipin synthesis. J. Biol. Chem 1978, 253, 5048–5055. [Google Scholar]
- Tan, B.K.; Bogdanov, M.; Zhao, J.; Dowhan, W.; Raetz, C.R.; Guan, Z. Discovery of a cardiolipin synthase utilizing phosphatidylethanolamine and phosphatidylglycerol as substrates. Proc. Natl. Acad. Sci. USA 2012, 109, 16504–16509. [Google Scholar]
- Kawai, F.; Shoda, M.; Harashima, R.; Sadaie, Y.; Hara, H.; Matsumoto, K. Cardiolipin domains in Bacillus subtilis marburg membranes. J. Bacteriol 2004, 186, 1475–1483. [Google Scholar]
- Guo, D.; Tropp, B.E. A second Escherichia coli protein with CL synthase activity. Biochim. Biophys. Acta 2000, 1483, 263–274. [Google Scholar]
- Sekimizu, K.; Kornberg, A. Cardiolipin activation of dnaA protein, the initiation protein of replication in Escherichia coli. J. Biol. Chem 1988, 263, 7131–7135. [Google Scholar]
- Yung, B.Y.; Kornberg, A. Membrane attachment activates dnaA protein, the initiation protein of chromosome replication in Escherichia coli. Proc. Natl. Acad. Sci. USA 1988, 85, 7202–7205. [Google Scholar]
- Castuma, C.E.; Crooke, E.; Kornberg, A. Fluid membranes with acidic domains activate DnaA, the initiator protein of replication inEscherichia coli. J. Biol. Chem. 1993, 268, 24665–24668. [Google Scholar]
- Crooke, E.; Castuma, C.E.; Kornberg, A. The chromosome origin of Escherichia coli stabilizes DnaA protein during rejuvenation by phospholipids. J. Biol. Chem 1992, 267, 16779–16782. [Google Scholar]
- Makise, M.; Mima, S.; Katsu, T.; Tsuchiya, T.; Mizushima, T. Acidic phospholipids inhibit the DNA-binding activity of DnaA protein, the initiator of chromosomal DNA replication in Escherichia coli. Mol. Microbiol 2002, 46, 245–256. [Google Scholar]
- Mozharov, A.D.; Shchipakin, V.N.; Fishov, I.L.; Evtodienko, YuV. Changes in the composition of membrane phospholipids during the cell cycle of Escherichia coli. FEBS Lett 1985, 186, 103–106. [Google Scholar]
- Hiraoka, S.; Matsuzaki, H.; Shibuya, I. Active increase in cardiolipin synthesis in the stationary growth phase and its physiological significance in Escherichia coli. FEBS Lett 1993, 336, 221–224. [Google Scholar]
- Renner, L.D.; Weibel, D.B. Cardiolipin microdomains localize to negatively curved regions of Escherichia coli membranes. Proc. Natl. Acad. Sci. USA 2011, 108, 6264–6269. [Google Scholar]
- Szeto, T.H.; Rowland, S.L.; Rothfield, L.I.; King, G.F. Membrane localization of MinD is mediated by a C-terminal motif that is conserved across eubacteria, archaea, and chloroplasts. Proc. Natl. Acad. Sci. USA 2002, 99, 15693–15698. [Google Scholar]
- Mileykovskaya, E.; Fishov, I.; Fu, X.; Corbin, B.D.; Margolin, W.; Dowhan, W. Effects of phospholipid composition on MinD-membrane interactions in vitro and in vivo. J. Biol. Chem 2003, 278, 22193–22198. [Google Scholar]
- Romantsov, T.; Helbig, S.; Culham, D.E.; Gill, C.; Stalker, L.; Wood, J.M. Cardiolipin promotes polar localization of osmosensory transporter ProP in Escherichia coli. Mol. Microbiol 2007, 64, 1455–1465. [Google Scholar]
- Mileykovskaya, E. Subcellular localization of Escherichia coli osmosensory transporter ProP: Focus on cardiolipin membrane domains. Mol. Microbiol 2007, 64, 1419–1422. [Google Scholar]
- Romantsov, T.; Battle, A.R.; Hendel, J.L.; Martinac, B.; Wood, J.M. Protein localization in Escherichia coli cells: Comparison of the cytoplasmic membrane proteins ProP, LacY, ProW, AqpZ, MscS, and MscL. J. Bacteriol 2010, 192, 912–924. [Google Scholar]
- Breyton, C.; Haase, W.; Rapoport, T.A.; Kühlbrandt, W. Collinson I. Three-dimensional structure of the bacterial protein-translocation complex SecYEG. Nature 2002, 418, 662–665. [Google Scholar]
- Gold, V.A.; Robson, A.; Bao, H.; Romantsov, T.; Duong, F.; Collinson, I. The action of cardiolipin on the bacterial translocon. Proc. Natl. Acad. Sci. USA 2010, 107, 10044–10049. [Google Scholar]
- Newman, G.; Crooke, E. DnaA, the initiator of Escherichia coli chromosomal replication, is located at the cell membrane. J. Bacteriol 2000, 182, 2604–2610. [Google Scholar]
- Boeneman, K.; Fossum, S.; Yang, Y.; Fingland, N.; Skarstad, K.; Crooke, E. Escherichia coli DnaA forms helical structures along the longitudinal cell axis distinct from MreB filaments. Mol. Microbiol 2009, 72, 645–657. [Google Scholar]
- Torrey, T.; Atlung, T.; Kogoma, T. dnaA suppressor (dasF) mutants of Escherichia coli are stable DNA replication (sdrA/rnh) mutants. Mol. Gen. Genet 1984, 196, 350–355. [Google Scholar]
- Von Meyenburg, K.; Boye, E.; Skarstad, K.; Koppes, L.; Kogoma, T. Mode of initiation of constitutive stable DNA replication in RNase H-defective mutants of Escherichia coli K-12. J. Bacteriol 1987, 169, 2650–2658. [Google Scholar]
- Kikuchi, S.; Shibuya, I.; Matsumoto, K. Viability of an Escherichia coli pgsA null mutant lacking detectable phosphatidylglycerol and cardiolipin. J. Bacteriol 2000, 182, 371–376. [Google Scholar]
- Inouye, S.; Franceschini, T.; Sato, M.; Itakura, K.; Inouye, M. Prolipoprotein signal peptidase of Escherichia coli requires a cysteine residue at the cleavage site. EMBO J 1983, 2, 87–91. [Google Scholar]
- Sankaran, K.; Wu, H.C. Lipid modification of bacterial prolipoprotein. Transfer of diacylglyceryl moiety from phosphatidylglycerol. J. Biol. Chem 1994, 269, 19701–19706. [Google Scholar]
- Suzuki, M.; Hara, H.; Matsumoto, K. Envelope disorder of Escherichia coli cells lacking phosphatidylglycerol. J. Bacteriol 2002, 184, 5418–5425. [Google Scholar]
- Li, Z.; Kitchen, J.L.; Boeneman, K.; Anand, P.; Crooke, E. Restoration of growth to acidic phospholipid-deficient cells by DnaA(L366K) is independent of its capacity for nucleotide binding and exchange and requires DnaA. J. Biol. Chem 2005, 280, 9796–9801. [Google Scholar]
- Saxena, R.; Rozgaja, T.; Grimwade, J.; Crooke, E. Remodeling of nucleoprotein complexes is independent of the nucleotide state of a mutant AAA+ protein. J. Biol. Chem 2011, 286, 33770–33777. [Google Scholar]
- Weigel, C.; Schmidt, A.; Seitz, H.; Tüngler, D.; Welzeck, M.; Messer, W. The N-terminus promotes oligomerization of the Escherichia coli initiator protein DnaA. Mol. Microbiol 1999, 34, 53–66. [Google Scholar]
- Simmons, L.A.; Felczak, M.; Kaguni, J.M. DnaA Protein of Escherichia coli: Oligomerization at the E. coli chromosomal origin is required for initiation and involves specific N-terminal amino acids. Mol. Microbiol 2003, 49, 849–858. [Google Scholar]
- Felczak, M.M.; Simmons, L.A.; Kaguni, J.M. An essential tryptophan of Escherichia coli DnaA protein functions in oligomerization at the E. coli replication origin. J. Biol. Chem 2005, 280, 24627–24633. [Google Scholar]
- Aranovich, A.; Parola, A.H.; Fishov, I. The reactivation of DnaA(L366K) requires less acidic phospholipids supporting their role in the initiation of chromosome replication in Escherichia coli. FEBS Lett 2007, 581, 4439–4442. [Google Scholar]
- Kimura, M.; Miki, T.; Hiraga, S.; Nagata, T.; Yura, T. Conditionally lethal amber mutations in the dnaA region of the Escherichia coli chromosome that affect chromosome replication. J. Bacteriol 1979, 140, 825–834. [Google Scholar]
- Fuller, R.S.; Funnell, B.E.; Kornberg, A. The dnaA protein complex with the Escherichia coli chromosomal replication origin (oriC) and other DNA sites. Cell 1984, 38, 889–900. [Google Scholar]
- Sekimizu, K.; Bramhill, D.; Kornberg, A. ATP activates dnaA protein in initiating replication of plasmids bearing the origin of the Escherichia coli chromosome. Cell 1987, 50, 259–265. [Google Scholar]
- Bramhill, D.; Kornberg, A. Duplex opening by dnaA protein at novel sequences in initiation of replication at the origin of the Escherichia coli chromosome. Cell 1988, 52, 743–755. [Google Scholar]
- Ozaki, S.; Kawakami, H.; Nakamura, K.; Fujikawa, N.; Kagawa, W.; Park, S.Y.; Yokoyama, S.; Kurumizaka, H.; Katayama, T. A common mechanism for the ATP-DnaA-dependent formation of open complexes at the replication origin. J. Biol. Chem. 2008, 283, 8351–8362. [Google Scholar]
- Marszalek, J.; Kaguni, J.M. DnaA protein directs the binding of DnaB protein in initiation of DNA replication in Escherichia coli. J. Biol. Chem 1994, 269, 4883–4890. [Google Scholar]
- Sutton, M.D.; Carr, K.M.; Vicente, M.; Kaguni, J.M. Escherichia coli DnaA protein. The N-terminal domain and loading of DnaB helicase at the Escherichia coli chromosomal origin. J. Biol. Chem 1998, 273, 34255–34262. [Google Scholar]
- Kaguni, J.M. Replication initiation at the Escherichia coli chromosomal origin. Curr. Opin. Chem. Biol 2011, 15, 606–613. [Google Scholar]
- Matsui, M.; Oka, A.; Takanami, M.; Yasuda, S.; Hirota, Y. Sites of dnaA protein-binding in the replication origin of the Escherichia coli K-12 chromosome. J. Mol. Biol 1985, 184, 529–533. [Google Scholar]
- McGarry, K.C.; Ryan, V.T.; Grimwade, J.E.; Leonard, A.C. Two discriminatory binding sites in the Escherichia coli replication origin are required for DNA strand opening by initiator DnaA-ATP. Proc. Natl. Acad. Sci. USA 2004, 101, 2811–2816. [Google Scholar]
- Kawakami, H.; Keyamura, K.; Katayama, T. Formation of an ATP-DnaA-specific initiation complex requires DnaA Arginine 285, a conserved motif in the AAA+ protein family. J. Biol. Chem 2005, 280, 27420–27430. [Google Scholar]
- Nievera, C.; Torgue, J.J.; Grimwade, J.E.; Leonard, A.C. SeqA blocking of DnaA-oriC interactions ensures staged assembly of the Escherichia coli pre-RC. Mol Cell 2006, 24, 581–592. [Google Scholar]
- Miller, D.T.; Grimwade, J.E.; Betteridge, T.; Rozgaja, T.; Torgue, J.J.; Leonard, A.C. Bacterial origin recognition complexes direct assembly of higher-order DnaA oligomeric structures. Proc. Natl. Acad. Sci. USA 2009, 106, 18479–18484. [Google Scholar]
- Cassler, M.R.; Grimwade, J.E.; Leonard, A.C. Cell cycle-specific changes in nucleoprotein complexes at a chromosomal replication origin. EMBO J 1995, 14, 5833–5841. [Google Scholar]
- Katayama, T.; Kubota, T.; Kurokawa, K.; Crooke, E.; Sekimizu, K. The initiator function of DnaA protein is negatively regulated by the sliding clamp of the Escherichia coli chromosomal replicase. Cell 1998, 94, 61–71. [Google Scholar]
- Kato, J.; Katayama, T. Hda, a novel DnaA-related protein, regulates the replication cycle in Escherichia coli. EMBO J 2001, 20, 4253–4262. [Google Scholar]
- Nakamura, K.; Katayama, T. Novel essential residues of Hda for interaction with DnaA in the regulatory inactivation of DnaA: Unique roles for Hda AAA Box VI and VII motifs. Mol. Microbiol 2010, 76, 302–317. [Google Scholar]
- Keyamura, K.; Katayama, T. DnaA protein DNA-binding domain binds to Hda protein to promote inter-AAA+ domain interaction involved in regulatory inactivation of DnaA. J. Biol. Chem 2011, 286, 29336–29346. [Google Scholar]
- Camara, J.E.; Breier, A.M.; Brendler, T.; Austin, S.; Cozzarelli, N.R.; Crooke, E. Hda inactivation of DnaA is the predominant mechanism preventing hyperinitiation of Escherichia coli DNA replication. EMBO Rep 2005, 6, 736–741. [Google Scholar]
- Riber, L.; Olsson, J.A.; Jensen, R.B.; Skovgaard, O.; Dasgupta, S.; Marinus, M.G.; Løbner-Olesen, A. Hda-mediated inactivation of the DnaA protein and dnaA gene autoregulation act in concert to ensure homeostatic maintenance of the Escherichia coli chromosome. Genes Dev 2006, 20, 2121–2134. [Google Scholar]
- Katayama, T.; Fujimitsu, K.; Ogawa, T. Multiple pathways regulating DnaA function in Escherichia coli: Distinct roles for DnaA titration by the datA locus and the regulatory inactivation of DnaA. Biochimie 2001, 83, 13–17. [Google Scholar]
- Kasho, K.; Katayama, T. DnaA binding locus datA promotes DnaA-ATP hydrolysis to enable cell cycle-coordinated replication initiation. Proc. Natl. Acad. Sci. USA 2013, 110, 936–941. [Google Scholar]
- Kurokawa, K.; Nishida, S.; Emoto, A.; Sekimizu, K.; Katayama, T. Replication cycle-coordinated change of the adenine nucleotide-bound forms of DnaA protein in Escherichia coli. EMBO J 1999, 18, 6642–6652. [Google Scholar]
- Aranovich, A.; Gdalevsky, G.Y.; Cohen-Luria, R.; Fishov, I.; Parola, A.H. Membrane-catalyzed nucleotide exchange on DnaA. Effect of surface molecular crowding. J. Biol Chem 2006, 281, 12526–12534. [Google Scholar]
- Fujimitsu, K.; Katayama, T. Reactivation of DnaA by DNA sequence-specific nucleotide exchange in vitro. Biochem. Biophys. Res. Commun 2004, 322, 411–419. [Google Scholar]
- Fujimitsu, K.; Senriuchi, T.; Katayama, T. Specific genomic sequences of Escherichia coli promote replicational initiation by directly reactivating ADP-DnaA. Genes Dev 2009, 23, 1221–1233. [Google Scholar]
- Fralick, J.A.; Lark, K.G. Evidence for the involvement of unsaturated fatty acids in initiating chromosome replication in Escherichia coli. J. Mol. Biol 1973, 80, 459–475. [Google Scholar]
- Marr, A.G.; Ingraham, J.I. Effect of temperature on the composition of fatty acids in Escherichia coli. J. Bacteriol 1962, 84, 1260–1267. [Google Scholar]
- De Mendoza, D.; Cronan, J.E. Thermal regulation of membrane lipid fluidity in bacteria. Trends. Biochem. Sci 1983, 8, 49–52. [Google Scholar]
- Suzuki, E.; Kondo, T.; Makise, M.; Mima, S.; Sakamoto, K.; Tsuchiya, T.; Mizushima, T. Alteration in the contents of unsaturated fatty acids in dnaA mutants of Escherichia coli. Mol. Microbiol 1998, 28, 95–102. [Google Scholar]
- Katayama, T.; Kornberg, A. Hyperactive initiation of chromosomal replication in vivo and in vitro by a mutant initiator protein, DnaAcos, of Escherichia coli. J. Biol. Chem 1994, 269, 12698–12703. [Google Scholar]
- Garner, J.; Crooke, E. Membrane regulation of the chromosomal replication activity of Escherichia coli DnaA requires a discrete site on the protein. EMBO J 1996, 15, 3477–3485. [Google Scholar]
- Garner, J.; Durrer, P.; Kitchen, J.; Brunner, J.; Crooke, E. Membrane-mediated release of nucleotide from an initiator of chromosomal replication, Escherichia coli DnaA, occurs with insertion of a distinct region of the protein into the lipid bilayer. J. Biol. Chem 1998, 273, 5167–5173. [Google Scholar]
- Hase, M.; Yoshimi, T.; Ishikawa, Y.; Ohba, A.; Guo, L.; Mima, S.; Makise, M.; Yamaguchi, Y.; Tsuchiya, T.; Mizushima, T. Site-directed mutational analysis for the membrane binding of DnaA protein. Identification of amino acids involved in the functional interaction between DnaA protein and acidic phospholipids. J. Biol. Chem 1998, 273, 28651–28656. [Google Scholar]
- Makise, M.; Mima, S.; Tsuchiya, T.; Mizushima, T. Identification of amino acids involved in the functional interaction between DnaA protein and acidic phospholipids. J. Biol. Chem 2000, 275, 4513–4518. [Google Scholar]
- Erzberger, J.P.; Pirruccello, M.M.; Berger, J.M. The structure of bacterial DnaA: Implications for general mechanisms underlying DNA replication initiation. EMBO J 2002, 21, 4763–4773. [Google Scholar]
- Yamaguchi, Y.; Hase, M.; Makise, M.; Mima, S.; Yoshimi, T.; Ishikawa, Y.; Tsuchiya, T.; Mizushima, T. Involvement of Arg-328, Arg-334 and Arg-342 of DnaA protein in the functional interaction with acidic phospholipids. Biochem J 1999, 340, 433–438. [Google Scholar]
- Makise, M.; Mima, S.; Tsuchiya, T.; Mizushima, T. Molecular mechanism for functional interaction between DnaA protein and acidic phospholipids: Identification of important amino acids. J. Biol. Chem 2001, 276, 7450–7456. [Google Scholar]
- Makise, M.; Mima, S.; Koterasawa, M.; Tsuchiya, T.; Mizushima, T. Biochemical analysis of DnaA protein with mutations in both Arg328 and Lys372. Biochem J. 2002, 362, 453–458. [Google Scholar]
- Rauch, J.; Tannenbaum, H.; Stollar, B.D.; Schwartz, R.S. Monoclonal anti-cardiolipin antibodies bind to DNA. Eur. J. Immunol 1984, 14, 529–534. [Google Scholar]
- Kumar, S.; Kalsi, J.; Bunting, K.; Ravirajan, C.T.; Latchman, D.S.; Pearl, L.H.; Isenberg, D.A. Fine binding characteristics of human autoantibodies-partial molecular characterization. Mol. Immunol 2004, 41, 495–510. [Google Scholar]
- Ichihashi, N.; Kurokawa, K.; Matsuo, M.; Kaito, C.; Sekimizu, K. Inhibitory effects of basic or neutral phospholipid on acidic phospholipid-mediated dissociation of adenine nucleotide bound to DnaA protein, the initiator of chromosomal DNA replication. J. Biol. Chem 2003, 278, 28778–28786. [Google Scholar]
- Adler, H.I.; Fisher, W.D.; Cohen, A.; Hardigree, A.A. Miniature Escherichia coli cells deficient in DNA. Proc. Natl. Acad. Sci. USA 1967, 57, 321–326. [Google Scholar]
- Koppelman, C.M.; Tanneke, D.B.; Duursma, M.C.; Heeren, R.M.; Nanninga, N. Escherichia coli minicell membranes are enriched in cardiolipin. J. Bacteriol 2001, 183, 6144–6147. [Google Scholar]
- Huang, K.C.; Mukhopadhyay, R.; Wingreen, N.S. A curvature-mediated mechanism for localization of lipids to bacterial poles. PLoS Comput. Biol 2006, 2, 1357–1364. [Google Scholar]
- Shiomi, D.; Margolin, W. The C-terminal domain of MinC inhibits assembly of the Z ring in Escherichia coli. J. Bacteriol 2007, 189, 236–243. [Google Scholar]
- Lenarcic, R.; Halbedel, S.; Visser, L.; Shaw, M.; Wu, L.J.; Errington, J.; Marenduzzo, D.; Hamoen, L.W. Localisation of DivIVA by targeting to negatively curved membranes. EMBO J 2009, 28, 2272–2282. [Google Scholar]
- Jones, L.J.; Carballido-López, R.; Errington, J. Control of cell shape in bacteria: Helical, actin-like filaments in Bacillus subtilis. Cell 2001, 104, 913–922. [Google Scholar]
- Shih, Y.L.; Le, T.; Rothfield, L. Division site selection in Escherichia coli involves dynamic redistribution of Min proteins within coiled structures that extend between the two cell poles. Proc. Natl. Acad. Sci. USA 2003, 100, 7865–7870. [Google Scholar]
- Nozaki, S.; Niki, H.; Ogawa, T. Replication initiator DnaA of Escherichia coli changes its assembly form on the replication origin during the cell cycle. J. Bacteriol 2009, 191, 4807–4814. [Google Scholar]
- Regev, T.; Myers, N.; Zarivach, R.; Fishov, I. Association of the chromosome replication initiator DnaA with the Escherichia coli inner membrane in vivo: Quantity and mode of binding. PLoS One 2012, 7, e36441. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Saxena, R.; Fingland, N.; Patil, D.; Sharma, A.K.; Crooke, E. Crosstalk between DnaA Protein, the Initiator of Escherichia coli Chromosomal Replication, and Acidic Phospholipids Present in Bacterial Membranes. Int. J. Mol. Sci. 2013, 14, 8517-8537. https://doi.org/10.3390/ijms14048517
Saxena R, Fingland N, Patil D, Sharma AK, Crooke E. Crosstalk between DnaA Protein, the Initiator of Escherichia coli Chromosomal Replication, and Acidic Phospholipids Present in Bacterial Membranes. International Journal of Molecular Sciences. 2013; 14(4):8517-8537. https://doi.org/10.3390/ijms14048517
Chicago/Turabian StyleSaxena, Rahul, Nicholas Fingland, Digvijay Patil, Anjali K. Sharma, and Elliott Crooke. 2013. "Crosstalk between DnaA Protein, the Initiator of Escherichia coli Chromosomal Replication, and Acidic Phospholipids Present in Bacterial Membranes" International Journal of Molecular Sciences 14, no. 4: 8517-8537. https://doi.org/10.3390/ijms14048517



