Evaluation of the Biological Activity of Glucosinolates and Their Enzymolysis Products Obtained from Lepidium meyenii Walp. (Maca)
Abstract
:1. Introduction
2. Results and Discussion
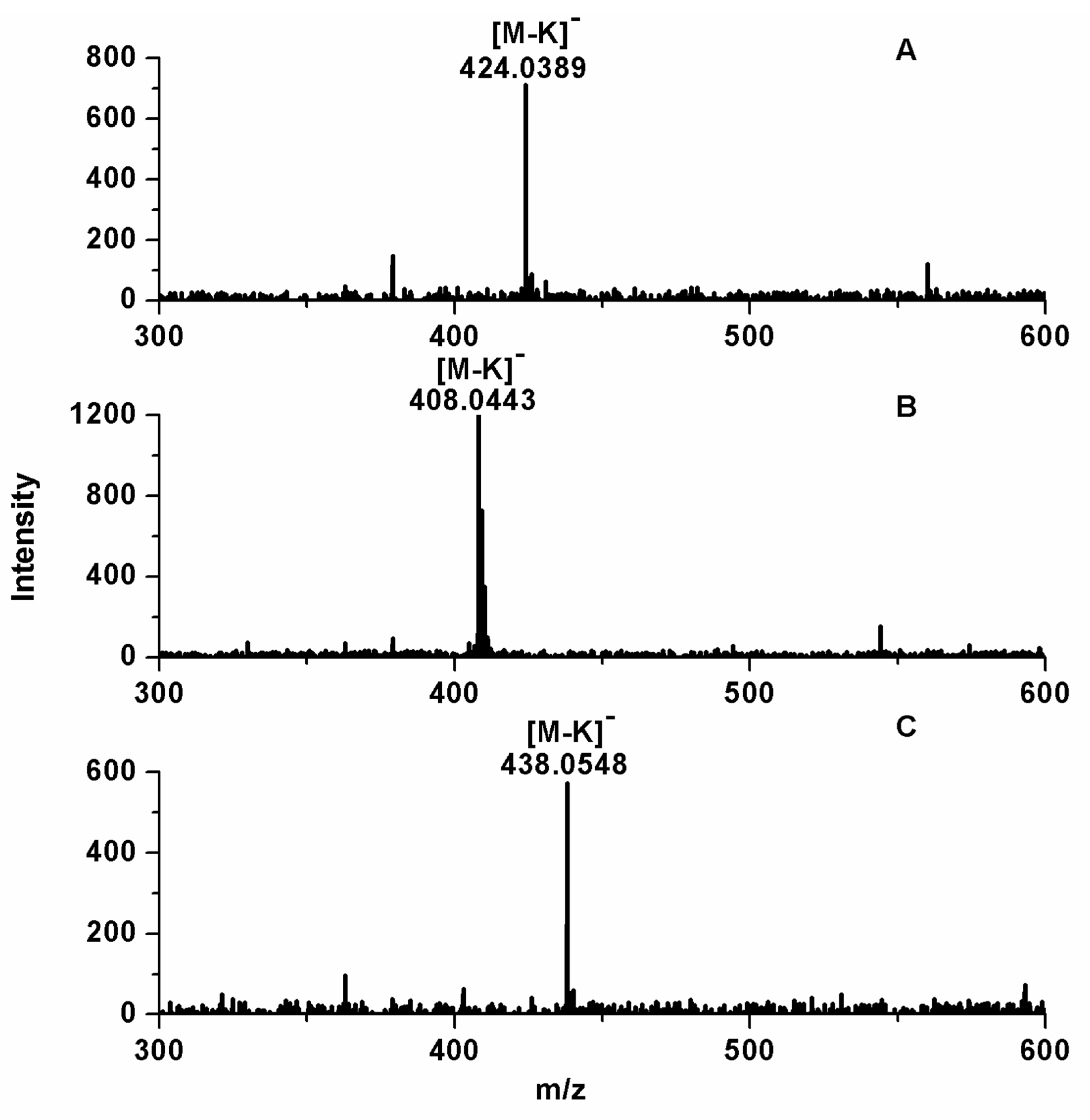
2.1. Analysis of GLS
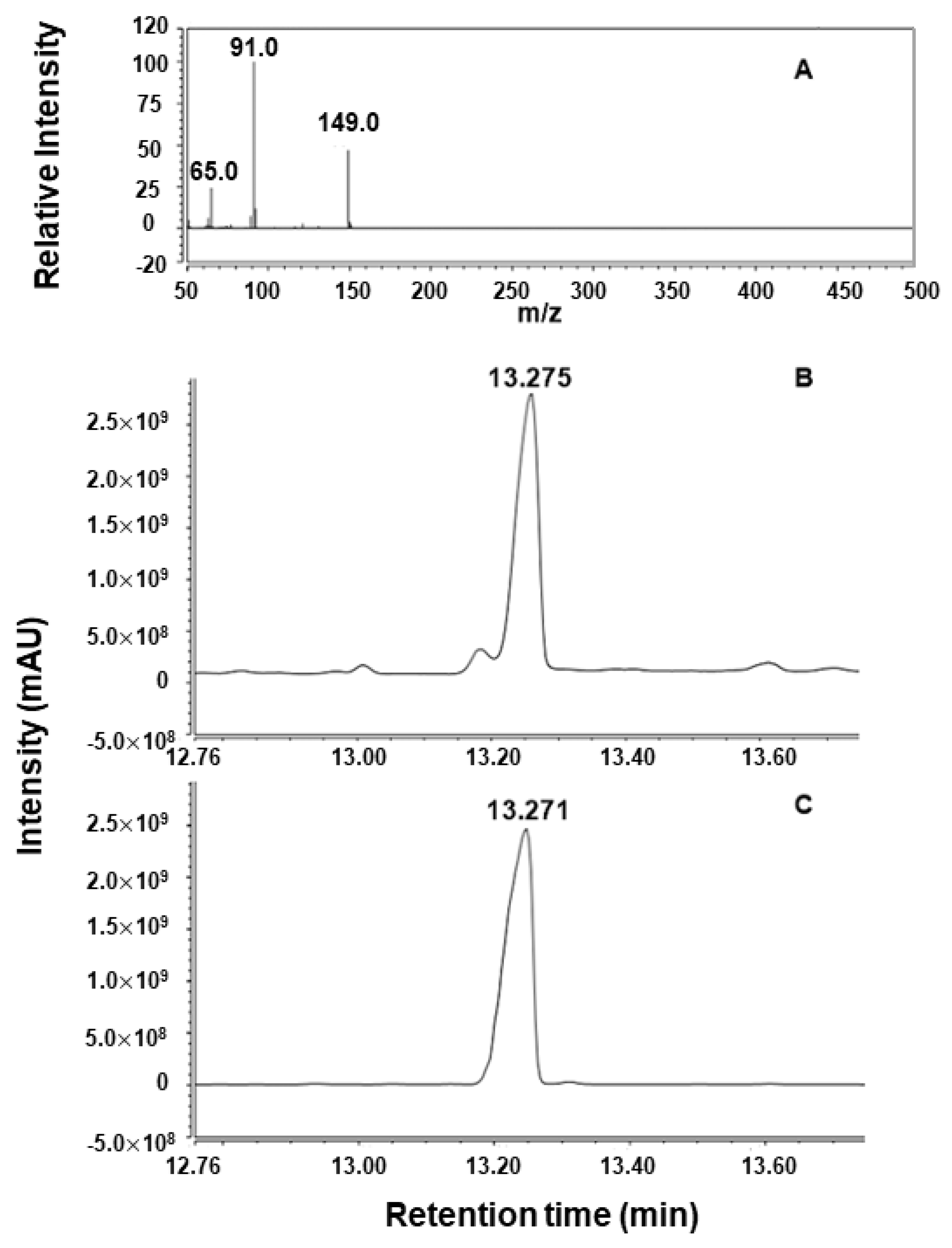
2.2. Analysis of Enzymatic Hydrolysis Products
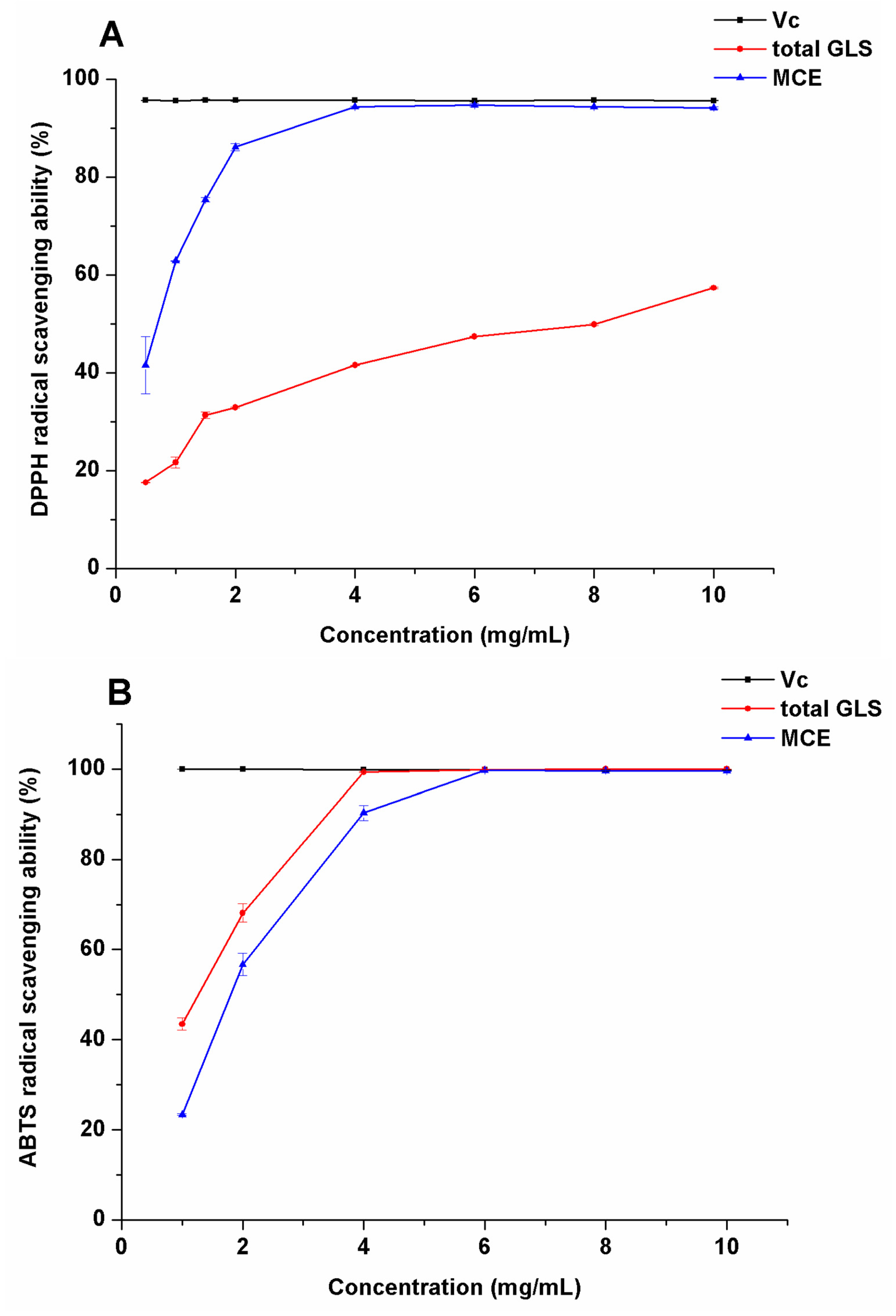
2.3. Evaluation of Antioxidation Activity
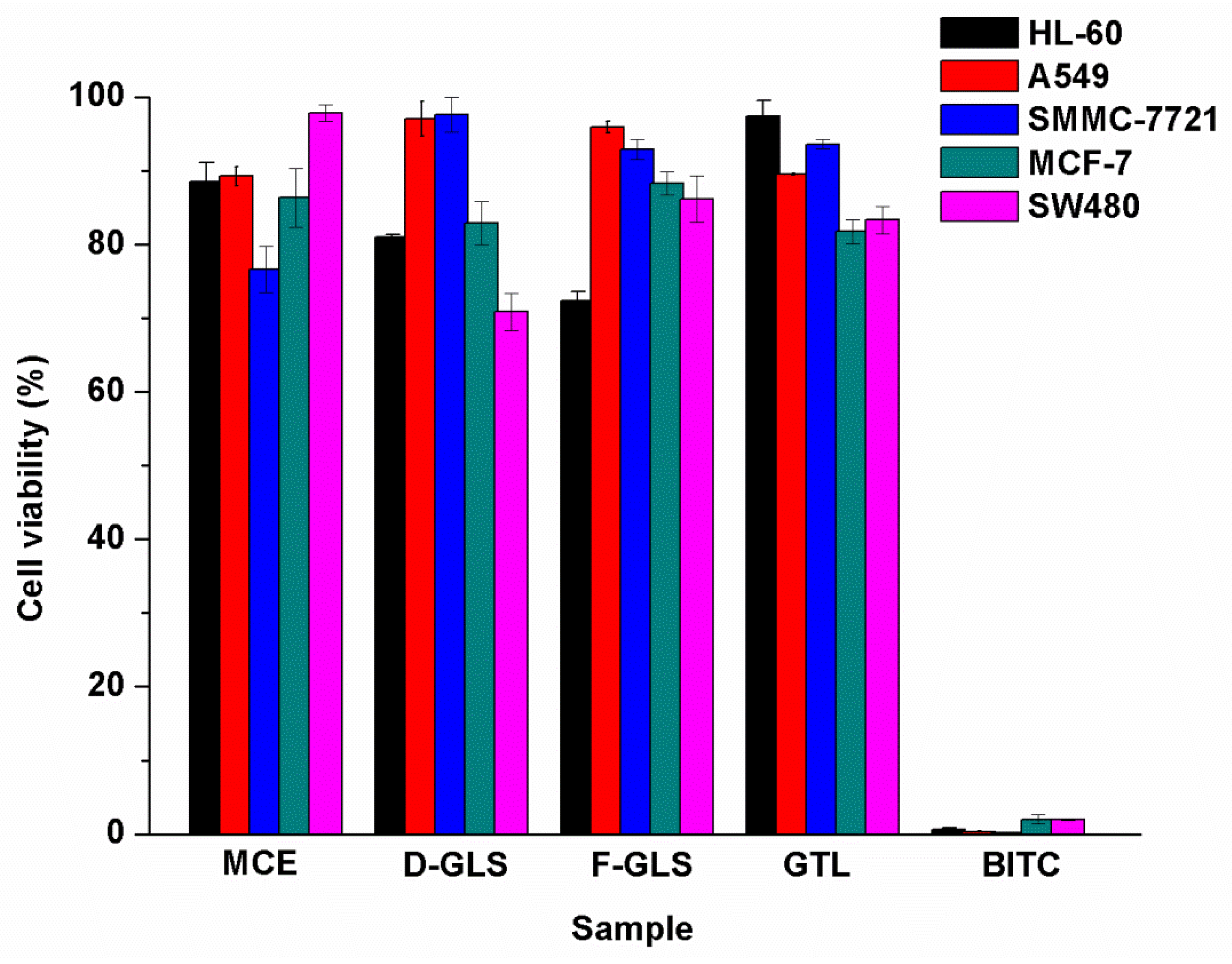
2.4. Anti-Cancer Activity of Extracts, GTL, and BITC
2.5. Anti-Inflammatory Activity
3. Materials and Methods
3.1. Materials
3.2. Extraction and Purification
3.3. Enzymatic Hydrolysis
3.4. Characterization
3.5. Antioxidant Activity In Vitro
3.6. Cytotoxic Assay
3.7. Anti-Inflammatory Assay
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, L.; Li, G.; Wang, S.; Yao, W.; Zhu, F. Physicochemical properties of Maca starch. Food Chem. 2017, 218, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhu, F. Chemical composition and health effects of Maca (Lepidium meyenii). Food Chem. 2019, 288, 422–443. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, F.V.; Ferraz, C.G.; Ribeiro, P.R. Pharmacological activities of the nutraceutical plant Lepidium meyenii: A critical review. J. Food Chem. Nanotechnol. 2020, 6, 107–116. [Google Scholar] [CrossRef]
- Marsh, R.E.; Waser, J. Refinement of the crystal structure of sinigrin. Acta Crystallogr. 1970, 26, 1030–1037. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, V.P.T.; Stewart, J.; Lopez, M.; Ioannou, I.; Allais, F. Glucosinolates: Natural occurrence, biosynthesis, accessibility, isolation, structures, and biological activities. Molecules 2020, 25, 4537. [Google Scholar] [CrossRef]
- Rosa, E.A.S.; Heaney, R.K.; Fenwick, G.R.; Portas, C.A.M. Glucosinolates in crop plants. Hortic. Rev. 1996, 19, 99. [Google Scholar]
- Li, G.; Ammermann UQuirόs, C.F. Glucosinolate contents in Maca (Lepidium peruvianum chacon) seeds, sprouts, mature plants and several derived commercial products. Econ. Bot. 2001, 55, 255–262. [Google Scholar] [CrossRef]
- Huang, Y.J.; Peng, X.R.; Qiu, M.H. Progress on the chemical constituents derived from glucosinolates in Maca (Lepidium meyenii Walp.). Nat. Prod. Bioprospect. 2018, 8, 405–412. [Google Scholar] [CrossRef] [Green Version]
- Cartea, M.E.; Velasco, P. Glucosinolates in Brassica foods: Bioavailability in food and significance for human health. Phytochem. Rev. 2008, 7, 213–229. [Google Scholar] [CrossRef]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Mithen, R. Leaf glucosinolate profiles and their relationship to pest and disease resistance in oilseed rape. Euphytica 1992, 63, 71–83. [Google Scholar] [CrossRef]
- Getahun, S.M.; Chung, F.L. Conversion of glucosinolates to isothiocyanates in humans after ingestion of cooked watercress. Cancer Epidemiol. Biomark. Prev. 1999, 8, 447–451. [Google Scholar]
- Xu, K.; Thornalley, P.J. Studies on the mechanism of the inhibition of human leukaemia cell growth by dietary isothiocyanates and their cysteine adducts in vitro. Biochem. Pharmacol. 2000, 60, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Hudlikar, R.; Wang, L.; Wu, R.; Li, S.; Kong, A.N. Epigenetics/epigenomics and prevention of early stages of cancer by isothiocyanates. Cancer Prev. Res. 2020, 14, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Popović, M.; Maravić, A.; Čulić, V.Č.; Đulovi, A.; Burčul, F.; Blažević, I. Biological effects of glucosinolate degradation products from Horseradish: A horse that wins the race. Biomolecules 2020, 10, 343. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.-H.; Shivendra Singh, V. The role of polycomb group protein bmi-1 and notch4 in breast cancer stem cell inhibition by benzyl isothiocyanate. Breast Cancer Res. Treat. 2015, 149, 681–692. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.H.; Sehrawat, A.; Singh, S.V. Dietary chemopreventative benzyl isothiocyanate inhibits breast cancer stem cells in vitro and in vivo. Cancer Prev. Res. 2013, 6, 782–790. [Google Scholar] [CrossRef] [Green Version]
- Keum, Y.S.; Jeong, W.S.; Kong, A.N. Chemoprevention by isothiocyanates and their underlying molecular signaling mechanisms. Mol. Mech. Mutagen. 2004, 555, 191–202. [Google Scholar] [CrossRef]
- Keum, Y.S.; Jeong, W.S.; Kong, A.N. Chemopreventive functions of isothiocyanates. Drug News Perspect. 2005, 18, 445–451. [Google Scholar] [CrossRef]
- Pereira, C.; Calado, A.M.; Sampaio, A.C. The effect of benzyl isothiocyanate on Candida albicans growth, cell size, morphogenesis, and ultrastructure. World J. Microbiol. Biotechnol. 2020, 36, 153. [Google Scholar] [CrossRef]
- Dinkova-Kostova, A.T. Chemoprotection against cancer by isothiocyanates: A focus on the animal models and the protective mechanisms. Top. Curr. Chem. 2012, 329, 179–201. [Google Scholar]
- Yuan, L.; Guo, W.; Wang, Z. Extraction, separation and structural identification of 4-pentenyl glucosinolate from rapeseed. Chin. J. Anal. Chem. 2004, 32, 1513–1516. [Google Scholar]
- Yábar, E.; Pedreschi, R.; Chirinos, R.; Campos, D. Glucosinolate content and myrosinase activity evolution in three maca (Lepidium meyenii Walp.) ecotypes during preharvest, harvest and postharvest drying. Food Chem. 2011, 127, 1576–1583. [Google Scholar] [CrossRef]
- Okunade, O.A.; Ghawi, S.K.; Methven, L.; Niranjan, K. Thermal and pressure stability of myrosinase enzymes from black mustard (Brassica nigra L. W.D.J. Koch. var. nigra), brown mustard (Brassica juncea L. Czern. var. juncea) and yellow mustard (Sinapsis alba L. subsp. maire) seeds. Food Chem. 2015, 187, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Guo, L.; Wang, Z.; Zhuang, Y.; Gu, Z. Response surface optimization and identification of isothiocyanates produced from broccoli sprouts. Food Chem. 2013, 141, 1580–1586. [Google Scholar] [CrossRef]
- Fu, L.; Wei, J.; Gao, Y.; Chen, R. Antioxidant and antitumoral activities of isolated macamide and macaene fractions from Lepidium meyenii (Maca). Talanta 2021, 221, 121635. [Google Scholar] [CrossRef]
- Wang, C.C.; Chang, S.C.; Inbaraj, B.S.; Chen, B.H. Isolation of carotenoids, flavonoids and polysaccharides from Lycium barbarum L. and evaluation of antioxidant activity. Food Chem. 2010, 120, 184–192. [Google Scholar] [CrossRef]
- Schiestl, F.P. Correlation analyses between volatiles and glucosinolates show no evidence for chemical defense signaling in brassica rapa. Front. Ecol. Evol. 2014, 2, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Arumugam, A.; Razis, A. Apoptosis as a mechanism of the cancer chemopreventive activity of glucosinolates: A review. Asian Pac. J. Cancer Prev. 2018, 19, 1439–1448. [Google Scholar]
- Dias, C.; Aires, A.; Saavedra, M. Antimicrobial activity of isothiocyanates from Cruciferous plants against methicillin-resistant Staphylococcus aureus (MRSA). Int. J. Mol. Sci. 2014, 15, 19552–19561. [Google Scholar] [CrossRef] [Green Version]
- Cory, A.H.; Owen, T.C.; Barltrop, J.A.; Cory, J.G. Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture. Cancer Commun. 1991, 3, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Theoduloz, C.; Delporte, C.; Valenzuela-Barra, G.; Silva, X.; Cádiz, S.; Bustamante, F.; Pertino, M.W.; Schmeda-Hirschmann, G. Topical anti-inflammatory activity of new hybrid molecules of terpenes and synthetic drugs. Molecules 2015, 20, 11219–11235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, G.; Lee, K.; Lee, M.; Ham, I.; Choi, H.Y. Inhibition of lipopolysaccharide-induced nitric oxide and prostaglandin E2 production by chloroform fraction of Cudrania tricuspidata in RAW 264.7 macrophages. BMC Complement. Altern. Med. 2012, 12, 250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hussain, S.P.; Harris, C.C. Inflammation and cancer: An ancient link with novel potentials. Int. J. Cancer 2007, 121, 2373–2380. [Google Scholar] [CrossRef]
- Li, J.; Sun, Q.; Meng, Q.; Lei, W.; Zhang, L. Anti-fatigue activity of polysaccharide fractions from Lepidium meyenii walp. (Maca). Int. J. Biol. Macromol. 2017, 95, 1305–1311. [Google Scholar] [CrossRef]
- Wang, T.; Liang, H.; Yuan, Q. Separation and purification of sinigrin and gluconapin from defatted Indian mustard seed meals by macroporous anion exchange resin and medium pressure liquid chromatography. Sep. Sci. Technol. 2014, 49, 1838–1847. [Google Scholar] [CrossRef]
- Su, L.; Su, G.; Wang, X.; Du, Q.; Wang, K. Endogenous and exogenous enzymolysis of vegetable-sourced glucosinolates and influencing factors. Food Chem. 2010, 119, 987–994. [Google Scholar]
- Blažević, I.; Đulović, A.; Burčul, F.; Popović, M.; Montaut, S.; Bilušić, T.; Vrca, I.; Markić, J.; Ljubenkov, I.; Ruščić, i.; et al. Stability and bioaccessibility during ex vivo digestion of glucoraphenin and glucoraphasatin from Matthiola incana (L.) R. Br. J. Food Compos. Anal. 2020, 90, 103483. [Google Scholar] [CrossRef]
- Gan, J.; Feng, Y.; Zhang, H.; He, Z.; Xu, L.; Chen, X. Analysis on composition and content of glucosinolate in three color types of Maca (Lepidium meyenii). Sci. Agric. Sin. 2012, 81, 244–256. [Google Scholar]
- Sun, T.; Powers, J.R.; Tang, J. Loss of rutin and antioxidant activity of asparagus juice caused by a pectolytic enzyme preparation from Aspergillus niger. Food Chem. 2007, 105, 173–178. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, F.; Li, Q.; Chen, H.; Zhang, W.; Yu, P.; Zhao, T.; Mao, G.; Feng, W.; Yang, L.; et al. Structure characterization of one polysaccharide from Lepidium meyenii Walp., and its antioxidant activity and protective effect against H2O2-induced injury RAW264.7 cells. Int. J. Biol. Macromol. 2018, 118, 816–833. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.E.; Songe, M.M.; Skaar, I. Colorimetric assay for the in vitro evaluation of Saprolegnia biofilm inhibitors. J. Fish Dis. 2019, 42, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Avilés-Gaxiola, S.; León-Félix, J.; Jiménez-Nevárez, Y.B.; Angulo-Escalante, M.A.; Ramos-Payán, R.; Colado-Velázquez, I.I.I.J.; Heredia, J.B. Antioxidant and anti-inflammatory properties of novel peptides from Moringa oleifera Lam. Leaves. South Afr. J. Bot. 2021, 141, 466–473. [Google Scholar] [CrossRef]

| Glucosinolate | Content in Fresh Maca (mg/g) | Content in Dry Maca (mg/g) |
|---|---|---|
| GTL | 24.31 ± 2.5 | 19.37 ± 0.67 |
| GLH | 3.72 ± 0.63 | 2.81 ± 0.31 |
| GSB | 6.09 ± 0.43 | 4.14 ± 0.39 |
| GLS | 34.12 ± 2.4 | 26.32 ± 1.4 |
| Concentration (μg/mL) | Cell Viability (%) | ||||
|---|---|---|---|---|---|
| HL-60 | A549 | SMMC-7721 | MCF-7 | SW480 | |
| 0.16 | 88.31 ± 2.10 | 95.12 ± 0.90 | 86.22 ± 2.70 | 92.79 ± 4.70 | 88.50 ± 1.60 |
| 0.8 | 53.34 ± 2.53 | 55.29 ± 0.38 | 54.75 ± 0.66 | 71.24 ± 0.93 | 58.58 ± 1.30 |
| 4 | 0.89 ± 0.17 | 21.74 ± 2.07 | 7.54 ± 2.08 | 31.15 ± 1.72 | 24.57 ± 0.15 |
| 20 | 0.54 ± 0.14 | 3.75 ± 0.48 | −0.34 ± 0.09 | 8.05 ± 0.74 | 25.26 ± 2.43 |
| Sample | IC50 ± SD (μg/mL) | ||||
|---|---|---|---|---|---|
| HL-60 | A549 | SMMC-7721 | MCF-7 | SW480 | |
| BITC | 0.886 ± 0.065 * | 1.021 ± 0.032 * | 0.941 ± 0.023 * | 1.880 ± 0.088 * | 1.200 ± 0.057 * |
| paclitaxel | <0.006 | <0.006 | 0.033 ± 0.004 | <0.006 | <0.006 |
| DDP | 1.797 ± 0.318 * | 3.669 ± 0.33 * | 1.298 ± 0.306 * | 6.669 ± 1.09 * | 6.309 ± 0.98 * |
| Sample | Concentration (μg/mL) | Inhibition Rate of NO (%) |
|---|---|---|
| NOSI | 12.41 | 58.07 ± 0.89 |
| F-GLS | 50 | −10.73 ± 0.57 |
| D-GLS | 50 | −10.13 ± 2.81 |
| GTL | 50 | 16.19 ± 2.53 |
| BITC | 50 | 99.26 ± 0.62 * |
| 1.56 | 88.44 ± 1.58 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, S.; Wei, J.; Chen, R. Evaluation of the Biological Activity of Glucosinolates and Their Enzymolysis Products Obtained from Lepidium meyenii Walp. (Maca). Int. J. Mol. Sci. 2022, 23, 14756. https://doi.org/10.3390/ijms232314756
Yan S, Wei J, Chen R. Evaluation of the Biological Activity of Glucosinolates and Their Enzymolysis Products Obtained from Lepidium meyenii Walp. (Maca). International Journal of Molecular Sciences. 2022; 23(23):14756. https://doi.org/10.3390/ijms232314756
Chicago/Turabian StyleYan, Suitong, Jinchao Wei, and Rui Chen. 2022. "Evaluation of the Biological Activity of Glucosinolates and Their Enzymolysis Products Obtained from Lepidium meyenii Walp. (Maca)" International Journal of Molecular Sciences 23, no. 23: 14756. https://doi.org/10.3390/ijms232314756






