Assessing Nasal Epithelial Dynamics: Impact of the Natural Nasal Cycle on Intranasal Spray Deposition
Abstract
:1. Introduction
- (1)
- Prepare a set of multi-sectioned transparent 3D-printed nasal cast models based on a patient-specific nasal geometry, incorporating varying degrees of turbinate thickness to reflect different levels of congestion/decongestion resulting from the nasal cycle.
- (2)
- Visualize the distribution of deposited intranasally administered sprays within the various sections of each nasal model under the influence of inhalation flow and various head positions.
- (3)
- Quantify the deposition in the various model sections: front nose, turbinate regions (anterior, middle, posterior), and nasopharynx, and evaluate the outcomes of the experimental scenarios.
2. Results
2.1. Nasal Cycle Models and 3D-Printed Casts
2.2. Spray Deposition vs. Nasal Cycle at a 22.5° Backward Head Tilt
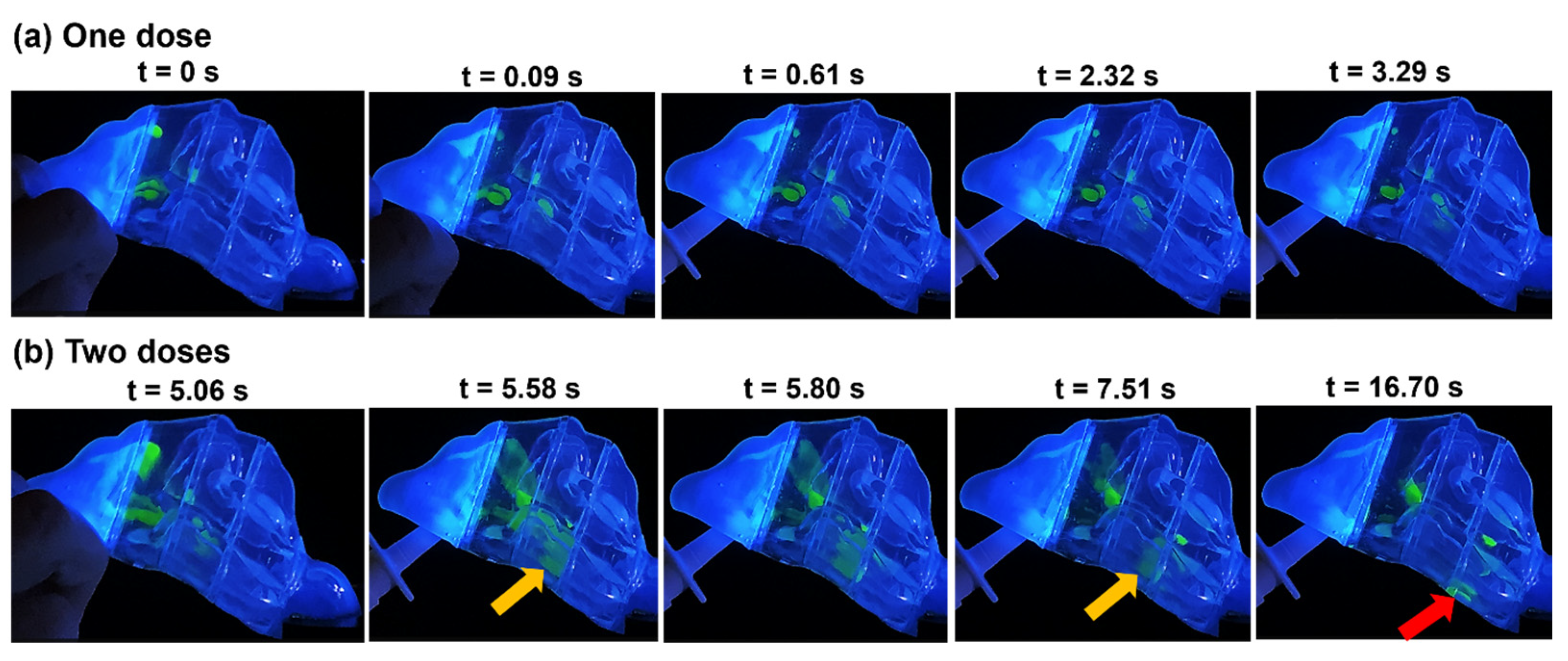
2.2.1. Time-Sequence Visualization of Deposition in Model N1
2.2.2. Comparison of Spray Deposition Patterns among N1, N0, P1, and P2
2.3. Spray Deposition vs. Nasal Cycle at Upright and 45° Backward Head Tilt
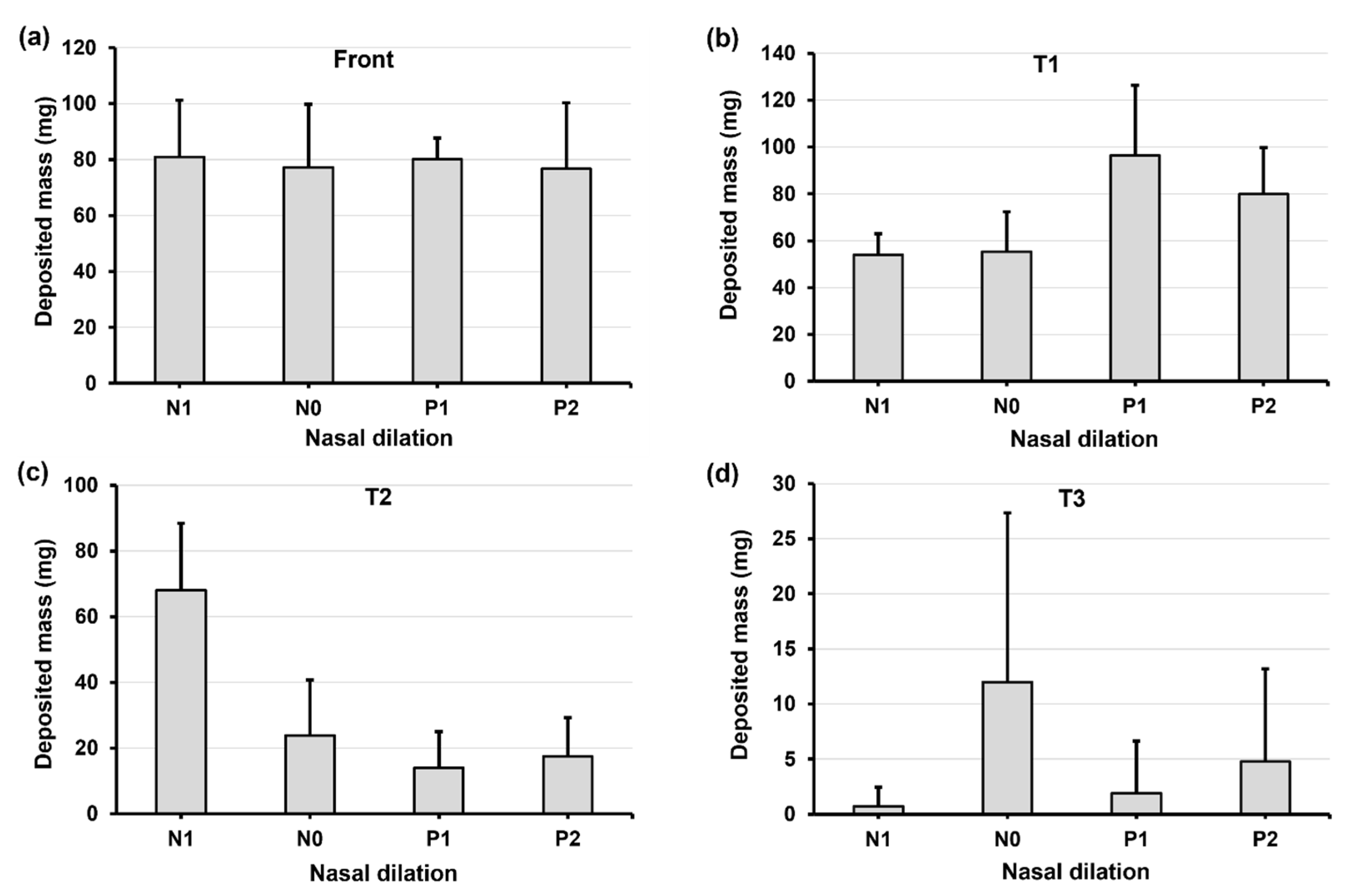
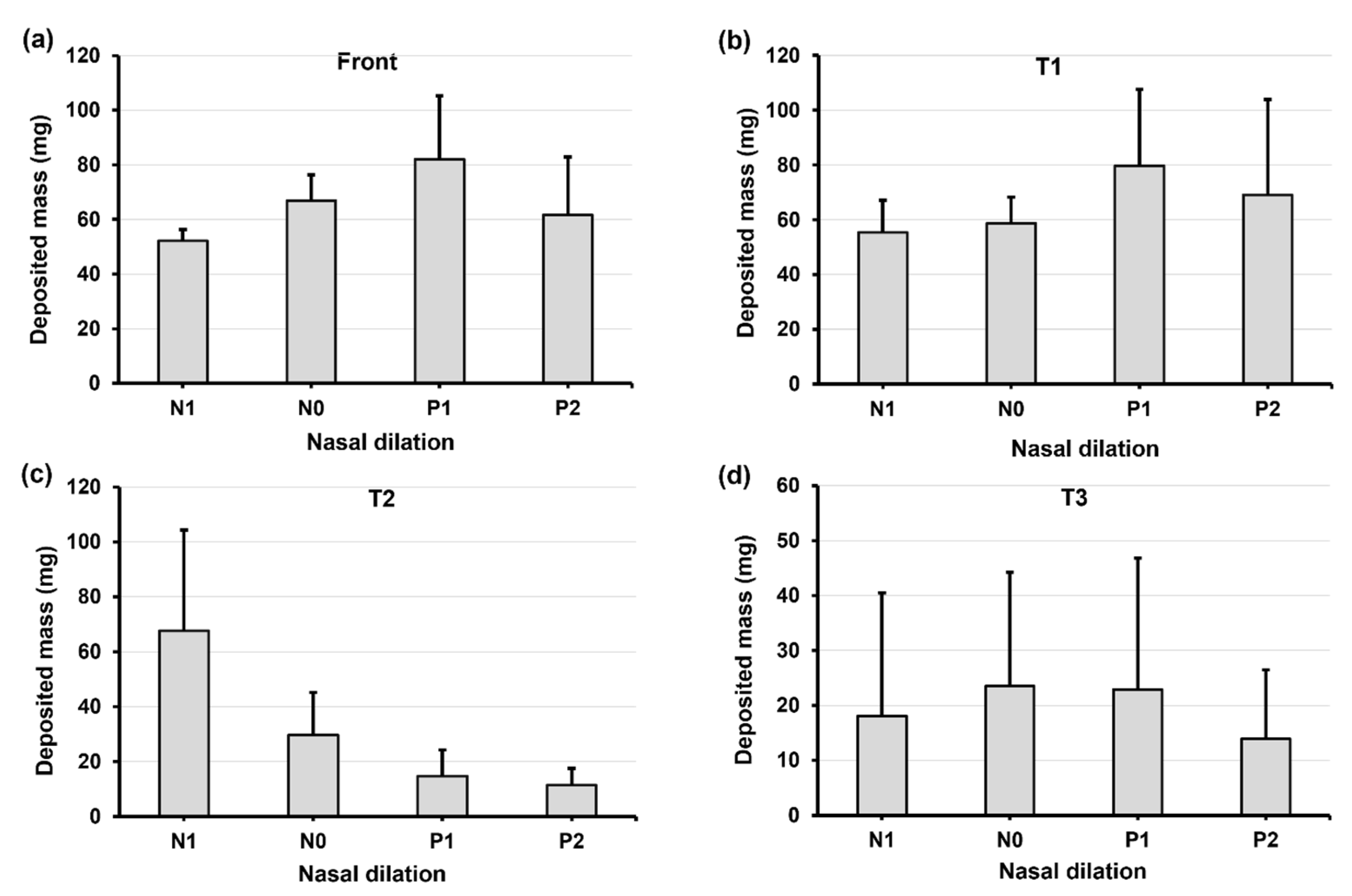
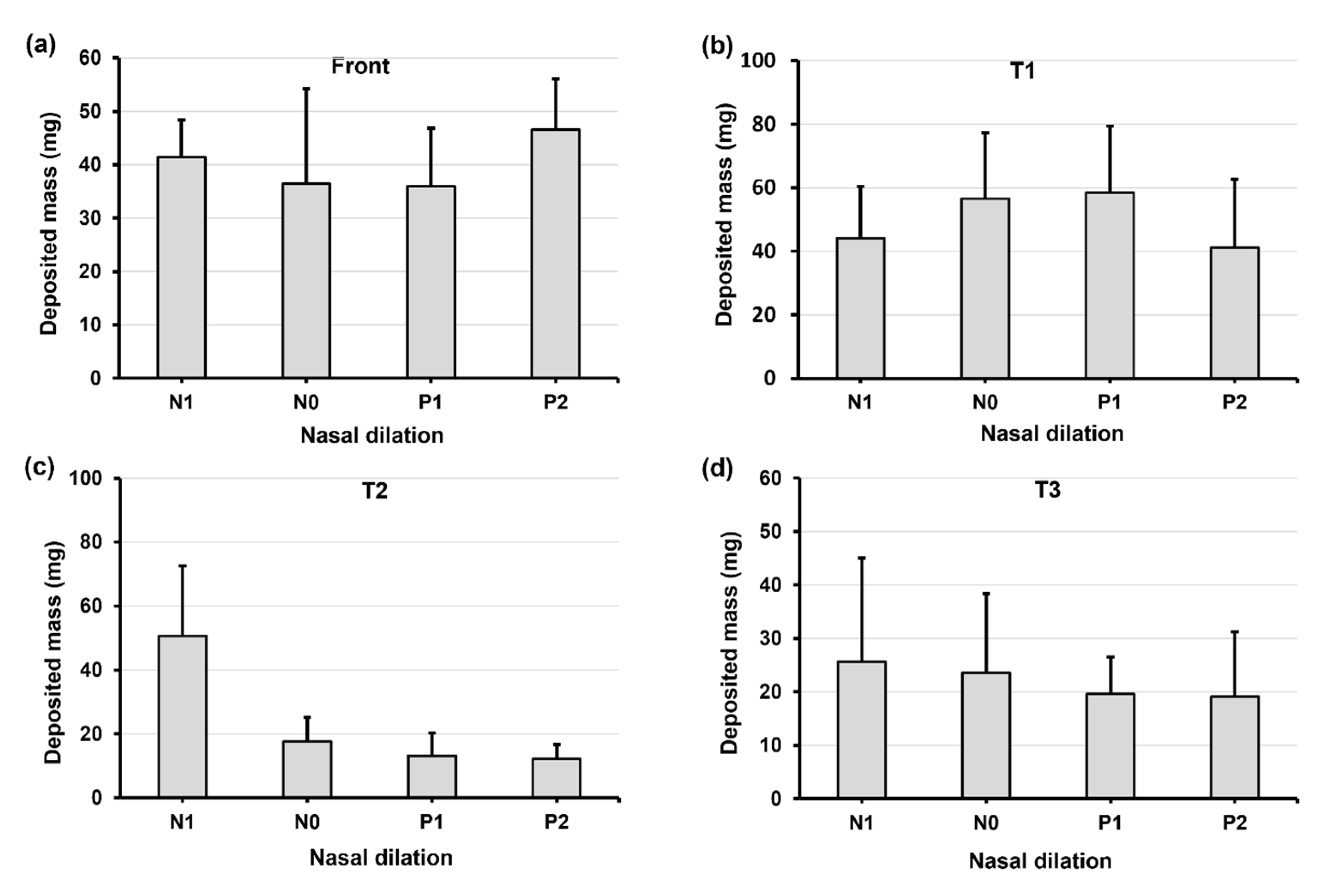
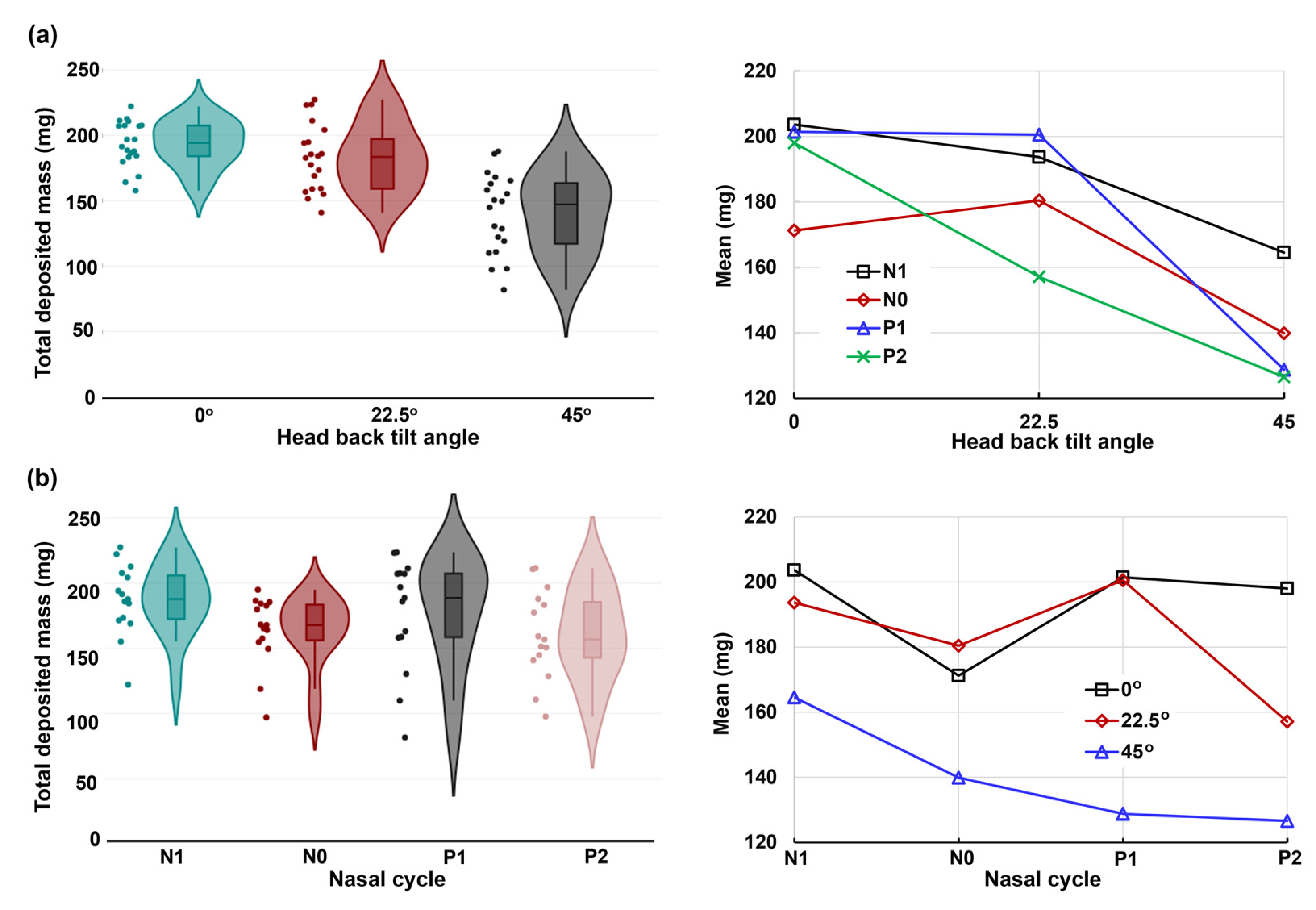
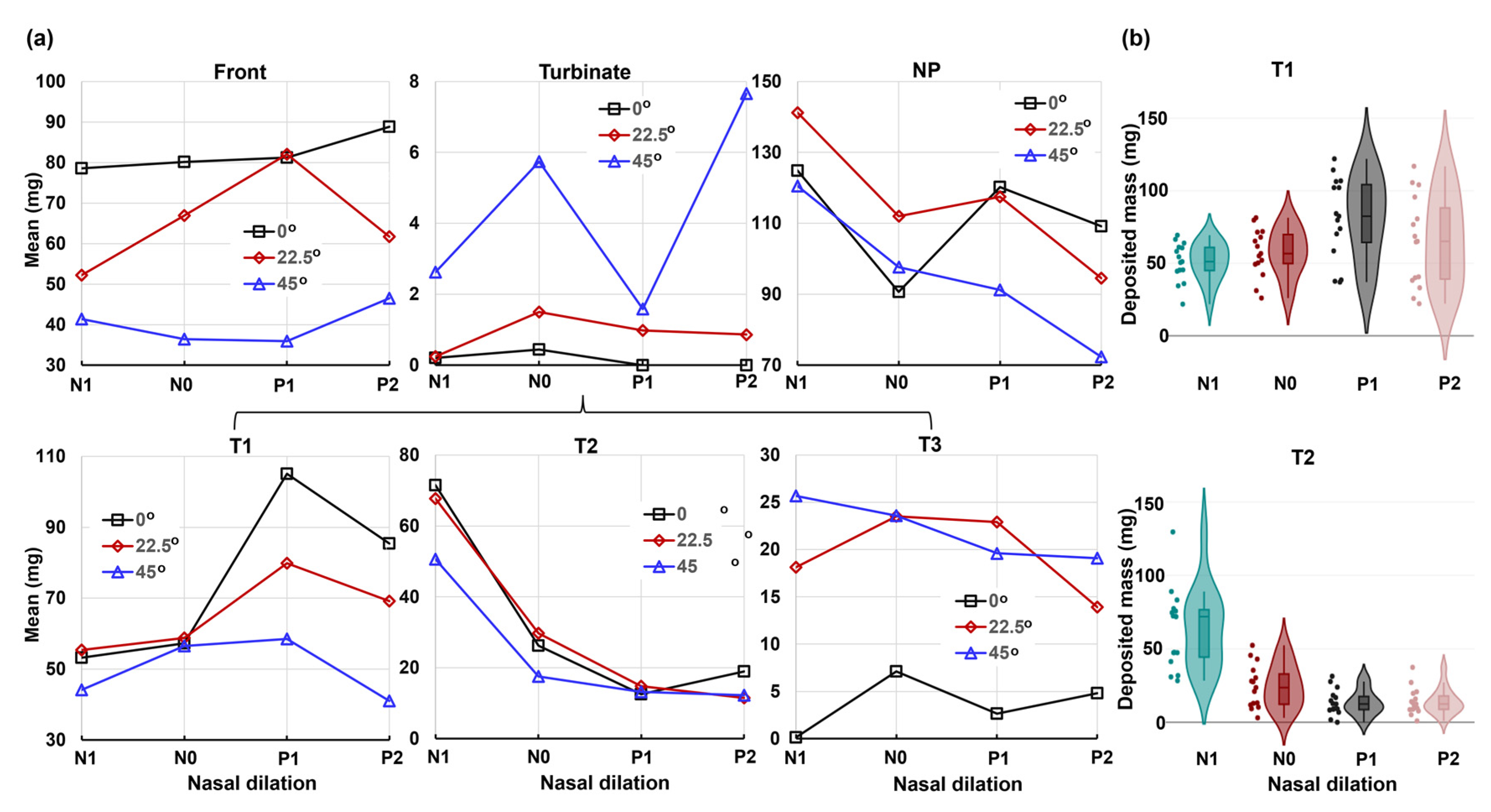
2.4. Quantification of Regional Deposition vs. Nasal Cycle and Head Angle
2.5. Factorial Analyses of Effects of Nasal Cycle and Head Angle
2.5.1. Total Deposition
2.5.2. Regional Deposition
3. Discussion
3.1. Effects of Nasal Cycle on Spray Deposition Distribution
3.2. Effects of Head Angle and Interactions with Nasal Cycle
3.3. Deposition Variance Related to Nasal Cycle and Head Angle
3.4. Implications for Intranasal Spray Administration
3.5. Limitations
4. Materials and Methods
4.1. Study Design
4.2. Nasal Cast Models
4.3. Experimental Protocol
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ulusoy, S.; Bayar Muluk, N.; Scadding, G.K.; Passali, G.S.; Dilber, M.; Gevaert, P.; Passali, D.; Resuli, A.S.; Van Gerven, L.; Kalogjera, L.; et al. The intranasal trigeminal system: Roles in rhinitis (allergic and non-allergic). Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 25–37. [Google Scholar] [PubMed]
- Kahana-Zweig, R.; Geva-Sagiv, M.; Weissbrod, A.; Secundo, L.; Soroker, N.; Sobel, N. Measuring and characterizing the human nasal cycle. PLoS ONE 2016, 11, e0162918. [Google Scholar] [CrossRef] [PubMed]
- Soane, R.J.; Carney, A.S.; Jones, N.S.; Frier, M.; Perkins, A.C.; Davis, S.S.; Illum, L. The effect of the nasal cycle on mucociliary clearance. Clin. Otolaryngol. Allied Sci. 2001, 26, 9–15. [Google Scholar] [CrossRef] [PubMed]
- White, D.E.; Bartley, J.; Nates, R.J. Model demonstrates functional purpose of the nasal cycle. Biomed. Eng. Online 2015, 14, 38. [Google Scholar] [CrossRef] [PubMed]
- Eccles, R. A role for the nasal cycle in respiratory defence. Eur. Respir. J. 1996, 9, 371–376. [Google Scholar] [CrossRef]
- Eccles, R.; Reilly, M.; Eccles, K.S. Changes in the amplitude of the nasal cycle associated with symptoms of acute upper respiratory tract infection. Acta Otolaryngol. 1996, 116, 77–81. [Google Scholar] [CrossRef]
- Hanif, J.; Jawad, S.S.; Eccles, R. The nasal cycle in health and disease. Clin. Otolaryngol. Allied Sci. 2000, 25, 461–467. [Google Scholar] [CrossRef]
- Braun, T.; Rich, M.; Berghaus, A.; Kramer, M.F. Effects of oxymetazoline nasal spray on the nasal cycle assessed by long-term rhinoflowmetry. Rhinology 2012, 50, 370–375. [Google Scholar] [CrossRef]
- Van Cauwenberge, P.B.; Deleye, L. Nasal cycle in children. Arch. Otolaryngol. 1984, 110, 108–110. [Google Scholar] [CrossRef]
- Williams, M.R.; Eccles, R. The nasal cycle and age. Acta Otolaryngol. 2015, 135, 831–834. [Google Scholar] [CrossRef]
- Lindemann, J.; Weindel, C.; Hoffmann, T.K.; Sommer, F.; Scheithauer, M.O.; Stupp, F.; Reins, E.F. Re-evaluating the nasal cycle by long-term rhinoflowmetry: Most individuals demonstrate a ‘mixed’ nasal cycle. Rhinology 2021, 59, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Baraniuk, J.N.; Kim, D. Nasonasal reflexes, the nasal cycle, and sneeze. Curr. Allergy Asthma Rep. 2007, 7, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Letzel, J.; Darbinjan, A.; Hummel, T. The nasal cycle before and after nasal septoplasty. Eur. Arch. Otorhinolaryngol. 2022, 279, 4961–4968. [Google Scholar] [CrossRef] [PubMed]
- Pendolino, A.L.; Scarpa, B.; Ottaviano, G. Relationship between nasal cycle, nasal symptoms and nasal cytology. Am. J. Rhinol. Allergy 2019, 33, 644–649. [Google Scholar] [CrossRef] [PubMed]
- Cingi, C.; Bayar Muluk, N.; Mitsias, D.I.; Papadopoulos, N.G.; Klimek, L.; Laulajainen-Hongisto, A.; Hytönen, M.; Toppila-Salmi, S.K.; Scadding, G.K. The nose as a route for therapy: Part 1. pharmacotherapy. Front. Allergy 2021, 2, 638136. [Google Scholar] [CrossRef] [PubMed]
- Blaiss, M.S.; Benninger, M.S.; Fromer, L.; Gross, G.; Mabry, R.; Mahr, T.; Marple, B.; Stoloff, S. Expanding choices in intranasal steroid therapy: Summary of a roundtable meeting. Allergy Asthma Proc. 2006, 27, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Merkus, P.; Ebbens, F.A.; Muller, B.; Fokkens, W.J. Influence of anatomy and head position on intranasal drug deposition. Eur. Arch. Otorhinolaryngol. 2006, 263, 827–832. [Google Scholar] [CrossRef]
- Naclerio, R.M.; Bachert, C.; Baraniuk, J.N. Pathophysiology of nasal congestion. Int. J. Gen. Med. 2010, 3, 47–57. [Google Scholar] [CrossRef]
- Salati, H.; Bartley, J.; White, D.E. Nasal saline irrigation—A review of current anatomical, clinical and computational modelling approaches. Respir. Physiol. Neurobiol. 2020, 273, 103320. [Google Scholar] [CrossRef]
- Xiao, Q.; Bates, A.J.; Cetto, R.; Doorly, D.J. The effect of decongestion on nasal airway patency and airflow. Sci. Rep. 2021, 11, 14410. [Google Scholar] [CrossRef]
- Basu, S.; Holbrook, L.T.; Kudlaty, K.; Fasanmade, O.; Wu, J.; Burke, A.; Langworthy, B.W.; Farzal, Z.; Mamdani, M.; Bennett, W.D.; et al. Numerical evaluation of spray position for improved nasal drug delivery. Sci. Rep. 2020, 10, 10568. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Dong, J.; Zhang, Y.; Tian, L.; Tu, J. Numerical study of the effect of nasopharynx airway obstruction on the transport and deposition of nanoparticles in nasal airways. Exp. Comput. Multiph. Flow 2022, 4, 399–408. [Google Scholar] [CrossRef]
- Shang, Y.; Dong, J.; He, F.; Inthavong, K.; Tian, L.; Tu, J. Detailed comparative analysis of environmental microparticle deposition characteristics between human and monkey nasal cavities using a surface mapping technique. Sci. Total Environ. 2022, 853, 158770. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Maza, G.; Farag, A.A.; Krebs, J.P.; Deshpande, B.; Otto, B.A.; Zhao, K. Asymptomatic vs. symptomatic septal perforations: A computational fluid dynamics examination. Int. Forum Allergy Rhinol. 2019, 9, 883–890. [Google Scholar] [CrossRef] [PubMed]
- Frank-Ito, D.O.; Kimbell, J.S.; Borojeni, A.A.T.; Garcia, G.J.M.; Rhee, J.S. A hierarchical stepwise approach to evaluate nasal patency after virtual surgery for nasal airway obstruction. Clin. Biomech. 2019, 61, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, M.G.; Garcia, G.J.M.; Frank-Ito, D.O.; Kimbell, J.S.; Rhee, J.S. Virtual septoplasty: A method to predict surgical outcomes for patients with nasal airway obstruction. Int. J. Comput. Assist. Radiol. Surg. 2020, 15, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Rhee, J.S.; Cannon, D.E.; Frank, D.O.; Kimbell, J.S. Role of virtual surgery in preoperative planning: Assessing the individual components of functional nasal airway surgery. Arch. Facial Plast. Surg. 2012, 14, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Vanhille, D.L.; Garcia, G.J.M.; Asan, O.; Borojeni, A.A.T.; Frank-Ito, D.O.; Kimbell, J.S.; Pawar, S.S.; Rhee, J.S. Virtual surgery for the nasal airway: A preliminary report on decision support and technology acceptance. JAMA Facial Plast. Surg. 2018, 20, 63–69. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, X.; Lou, M.; Gong, M.; Zhang, J.; Ma, R.; Zhang, L.; Huang, F.; Sun, B.; Zhu, K.; et al. Computational fluid dynamics (CFD) investigation of aerodynamic characters inside nasal cavity towards surgical treatments for secondary atrophic rhinitis. Math. Probl. Eng. 2019, 2019, 6240320. [Google Scholar] [CrossRef]
- Bickford, L.; Shakib, S.; Taverner, D. The nasal airways response in normal subjects to oxymetazoline spray: Randomized double-blind placebo-controlled trial. Br. J. Clin. Pharmacol. 1999, 48, 53–56. [Google Scholar] [CrossRef]
- Xi, J.; Wang, Z.; Si, X.A.; Zhou, Y. Nasal dilation effects on olfactory deposition in unilateral and bi-directional deliveries: In Vitro tests and numerical modeling. Eur. J. Pharm. Sci. 2018, 118, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, P.; Eccles, R. Spontaneous changes of unilateral nasal airflow in man. A re-examination of the ‘nasal cycle’. Acta Otolaryngol. 1997, 117, 590–595. [Google Scholar] [CrossRef]
- Cherobin, G.B.; Voegels, R.L.; Gebrim, E.; Garcia, G.J.M. Sensitivity of nasal airflow variables computed via computational fluid dynamics to the computed tomography segmentation threshold. PLoS ONE 2018, 13, e0207178. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Chung, S.K. Investigation on the respiratory airflow in human airway by PIV. J. Vis. 2009, 12, 259–266. [Google Scholar] [CrossRef]
- Wang, Y.D.; Lee, H.P.; Gordon, B.R. Impacts of fluid dynamics simulation in study of nasal airflow physiology and pathophysiology in realistic human three-dimensional nose models. Clin. Exp. Otorhinolaryngol. 2012, 5, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Xi, J. Development and challenges of nasal spray vaccines for short-term COVID-19 protection. Curr. Pharm. Biotechnol. 2022, 23, 1671–1677. [Google Scholar] [CrossRef] [PubMed]
- Alu, A.; Chen, L.; Lei, H.; Wei, Y.; Tian, X.; Wei, X. Intranasal COVID-19 vaccines: From bench to bed. EBioMedicine 2022, 76, 103841. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Lei, L.R.; Zouzas, W.; April Si, X. Nasally inhaled therapeutics and vaccination for COVID-19: Developments and challenges. MedComm 2021, 2, 569–586. [Google Scholar] [CrossRef]
- Wu, S.; Huang, J.; Zhang, Z.; Wu, J.; Zhang, J.; Hu, H.; Zhu, T.; Zhang, J.; Luo, L.; Fan, P.; et al. Safety, tolerability, and immunogenicity of an aerosolised adenovirus type-5 vector-based COVID-19 vaccine (Ad5-nCoV) in adults: Preliminary report of an open-label and randomised phase 1 clinical trial. Lancet Infect. Dis. 2021, 21, 1654–1664. [Google Scholar] [CrossRef]
- Nian, X.; Zhang, J.; Huang, S.; Duan, K.; Li, X.; Yang, X. Development of nasal vaccines and the associated challenges. Pharmaceutics 2022, 14, 1983. [Google Scholar] [CrossRef]
- Xi, J.; Si, X.A. A next-generation vaccine for broader and long-lasting COVID-19 protection. MedComm 2022, 3, e138. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xi, J.; Han, P.; Wongwiset, N.; Pontius, J.; Dong, H. Computational analysis of a flapping uvula on aerodynamics and pharyngeal wall collapsibility in sleep apnea. J. Biomech. 2019, 94, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Seifelnasr, A.; Si, X.A.; Xi, J. Visualization and estimation of nasal spray delivery to olfactory mucosa in an image-based transparent nasal model. Pharmaceutics 2023, 15, 1657. [Google Scholar] [CrossRef]
- Jamalabadi, M.Y.; Xi, J. Olfactory drug aerosol delivery with acoustic radiation. Biomedicines 2022, 10, 1347. [Google Scholar]
- Hintze, J.L.; Nelson, R.D. Violin plots: A box plot-density trace synergism. Am. Stat. 1998, 52, 181–184. [Google Scholar]





| Region | Total | Front | Turbinate | NP | T1 | T2 | T3 |
|---|---|---|---|---|---|---|---|
| Head angle | <0.001 | <0.001 | 0.034 | 0.001 | 0.001 | 0.244 | 0.001 |
| Nasal dilation | 0.003 | 0.223 | 0.001 | 0.061 | 0.003 | <0.001 | 0.78 |
| Interaction | 0.031 | 0.074 | 0.364 | 0.075 | 0.164 | 0.794 | 0.941 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seifelnasr, A.; Si, X.; Xi, J. Assessing Nasal Epithelial Dynamics: Impact of the Natural Nasal Cycle on Intranasal Spray Deposition. Pharmaceuticals 2024, 17, 73. https://doi.org/10.3390/ph17010073
Seifelnasr A, Si X, Xi J. Assessing Nasal Epithelial Dynamics: Impact of the Natural Nasal Cycle on Intranasal Spray Deposition. Pharmaceuticals. 2024; 17(1):73. https://doi.org/10.3390/ph17010073
Chicago/Turabian StyleSeifelnasr, Amr, Xiuhua Si, and Jinxiang Xi. 2024. "Assessing Nasal Epithelial Dynamics: Impact of the Natural Nasal Cycle on Intranasal Spray Deposition" Pharmaceuticals 17, no. 1: 73. https://doi.org/10.3390/ph17010073





