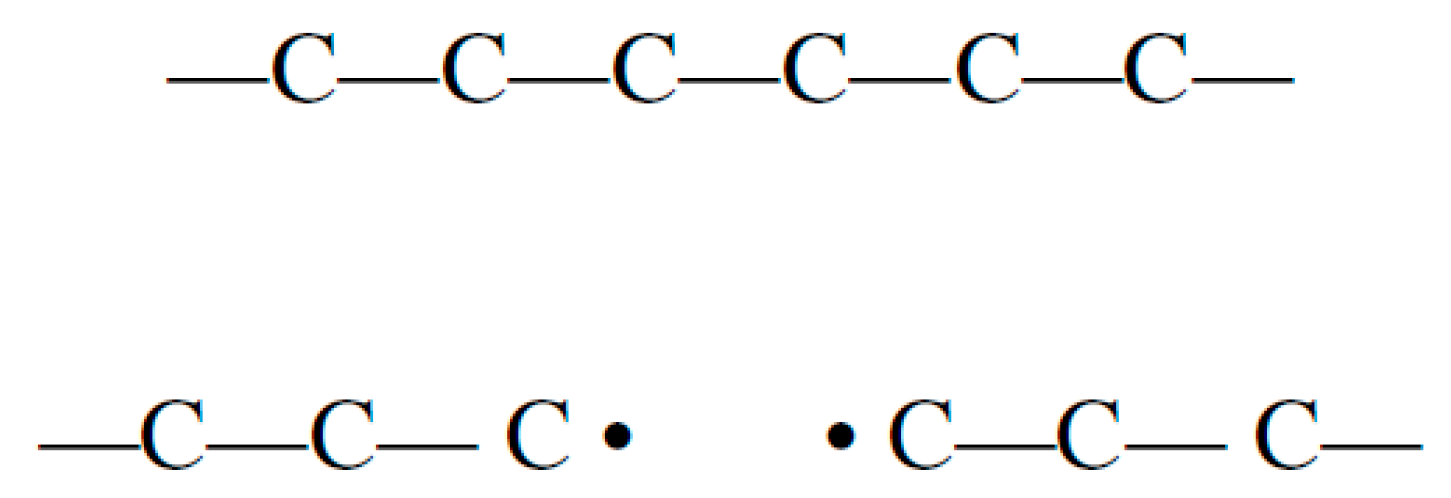1. Introduction
Nowadays plastic consumption is increasing exponentially and the annual consumption of plastics in Europe is about 60 million tons, of which 40% are used in packing and packaging [
1]. The excellent properties of plastics have been used to replace the use of wood and metals thus increasing the annual plastic consumption about 20 times from around 5 million tons in the 1950s to nearly 100 million tons in 2011 [
2]. It was also reported that 33 million tons of plastic waste are generated in the U.S. based on 2013 statistics [
3]. Most recycled plastics are mechanically recycled, while less than 2% is chemically recycled [
4]. Growing worldwide concerns regarding the environmental consequences of heavy dependence on fossil fuels, particularly climate change, are likely to constrain excessive use of fossil fuels in the near future. Thus, plastic to oil pyrolysis could have a positive environmental impact by providing alternative fuels and reducing the carbon footprint of incineration [
5]. Also, the steady increase in energy consumption coupled with environmental pollution promotes the evolution of alternative fuels such as plastic pyrolysis oil. Because of the increasing cost and decreasing space of landfills, traditional methods of plastic waste recycling such as landfilling and incineration cause irreversible environmental problems, including high NO
2 and CO
2 emissions [
6]. In this research work diesel range oil (DRO) in range of C
10–C
28 is produced by pyrolysis of plastics and identified using a GC-FID method applied to methanol solutions.
Thermoplastic pyrolysis is a tertiary recycling method that produces liquid hydrocarbons in an inert thermal cracking chemical reaction [
7]. The pyrolysis chemical reactions involve scission of long chain polymers to smaller chains forming hydrocarbon products in the C
4–C
40 range [
8]. Polyethylene is a major plastic component and was used as a feedstock for our experimental work. Oils produced from plastic pyrolysis have non-acidic, sulphur-free and non-corrosive characteristics, which is a major advantage of pyrolysis oils over fossil fuels and they can be used as feedstock in fluid catalytic cracking (FCC) refinery units [
9,
10]. Pyrolysis final products are affected by reaction parameters such as operating temperature, residence time, operating pressure, heating rate and catalysts [
11].
In pyrolysis, the polymeric materials are heated to high temperatures causing the macromolecular structures to break down into smaller molecules forming a wide range of hydrocarbons as [
12,
13]. Polyethylene is chosen since it is a major component of plastic waste from domestic disposal. Many studies have been conducted on the reaction kinetics of plastic wastes to determine rates of polymer conversion against heating rate and operating temperature [
14]. As seen in
Table 1, the operating temperature is inversely proportional to residence time. Below are the first order reactions used [
15]:
Pyrolysis reaction conversion
The LDPE reaction order can be assumed as
n = 1 while the E
a varies with molecular weight of sample [
16]. LDPE pyrolysis activation energy is estimated at 179–188 KJ/mol [
14]. LDPE pyrolysis has less environmental impact, unlike the incineration of plastics which releases toxic and harmful emissions such as 5.04 CO
2 kg/LDPE kg [
17,
18]. Combustion of pyrolysis oils releases less CO
2 in comparison with LDPE incineration, with an average of 6 to 11.5% wt. % (i.e., 0.06–0.115 kg CO
2/kg of combusted pyrolysis oil) depending on the engine load [
19].
As seen below, as the operating temperature increases, the residence time decreases due to the higher rate of thermal cracking [
10]. The chosen residence time in the experimental work is 30 min since thermal plasma has the ability to achieve a temperature higher than 500 °C in that period. As seen in
Table 1, the operating temperature is inversely proportional to the residence time.
In this research work, slow pyrolysis is used with a residence time of 30 min without using a catalyst [
5]. The minimum cracking temperature of LDPE is 450 °C The main pyrolysis products derived from polyethylene plastics are 1-alkenes,
n-alkanes, and α,ω-dialkenes ranging from C
1 to C
45+ [
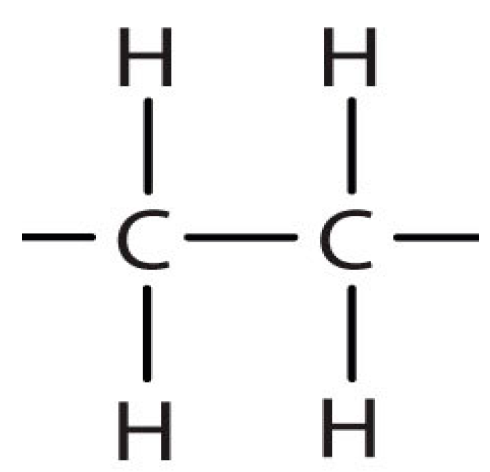
20]. The repetitive molecular structure of a low density polyethylene (LDPE) chain is shown in
Figure 1, below:
The hydrocarbon liquids produced were analyzed by a FID-GC method using methanol as a solvent [
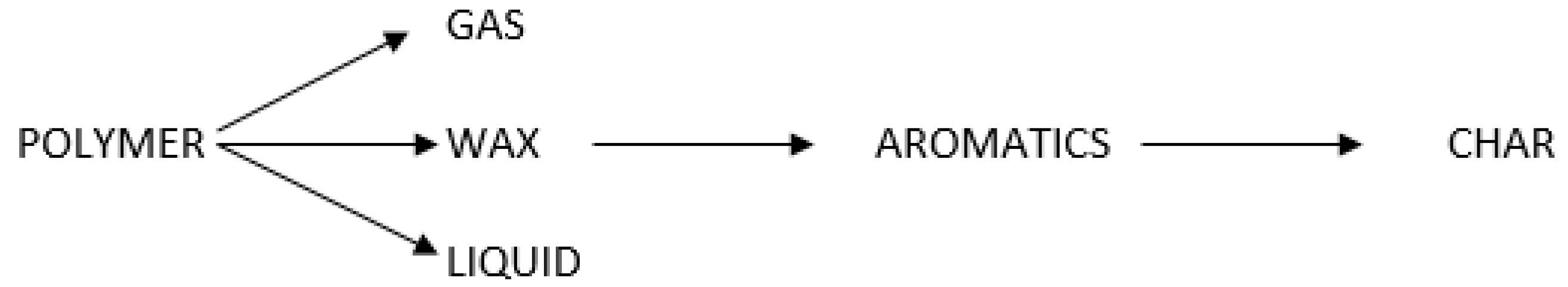
13]. The expected pyrolysis products are shown in
Figure 2, including the following hydrocarbon fractions:
Fraction 1: C6 to C10 hydrocarbon liquid
Fraction 2: C10 to C16 hydrocarbon liquid
Fraction 3: C16 to C34 hydrocarbon liquid
Fraction 4: C34 to C50 hydrocarbon liquid
1.1. DC Thermal Plasma
Thermal plasma technology has been under active development and is well-established in metallurgical processing and material synthesis. Thermal plasmas can achieve high temperatures through plasma torches and have the ability to decrease the reactant volume to non-leachable residue [
23]. The classification of the thermal plasma used in our project is low temperature plasma (quasi-equilibrium plasma) in the temperature range of 2 × 10
3–3 × 10
4 K, as shown in
Table 2.
Thermal plasma technology can help develop pyrolysis reactors due to its ability to generate free radicals which accelerate chemical reactions and thermal cracking rates, causing better reaction kinetics [
23,
24]. Plasma-assisted chemical reactions are more energy efficient with less environmental impact and in addition, they increase the process stability and enhance the pyrolysis chemistry [
25]. A constant heating rate in plastic pyrolysis achieves higher oil yield, and lower gas and tar yield [
26]. The thermal plasma can achieve better process chemistry control due to its high energy density, free radical reactivity and rapid startup and shutdown capability [
27]. Thermal plasmas can also achieve high thermal efficiencies of up to 70%, as well as long electrode lifetimes (up to 1000 h) which in the long term can justify the high initial capital cost [
27].
DC plasma provides a high energy density and high temperature generation between two electrodes and in the presence of a sufficiently high gas flow, the plasma extends beyond one of the electrodes in the form of a plasma jet. In a non-transferred arc torch, the two electrodes do not participate in the process and have the sole function of plasma generation, unlike the transferred arc torch which is only suitable for conductive reactants [
24]. Using thermal plasma in plastic waste reactions achieves better environmental performance as well as better life cycle assessment and higher process efficiency [
28]. The thermal plasma also accelerates reaction kinetics and improve thermal cracking and the emission enthalpy can be adjusted easily by electric power tuning [
29]. Therefore, DC thermal plasma torches can act as a catalyst due to their excellent properties such as high energy density, high temperature range, and high chemical reactivity [
30].
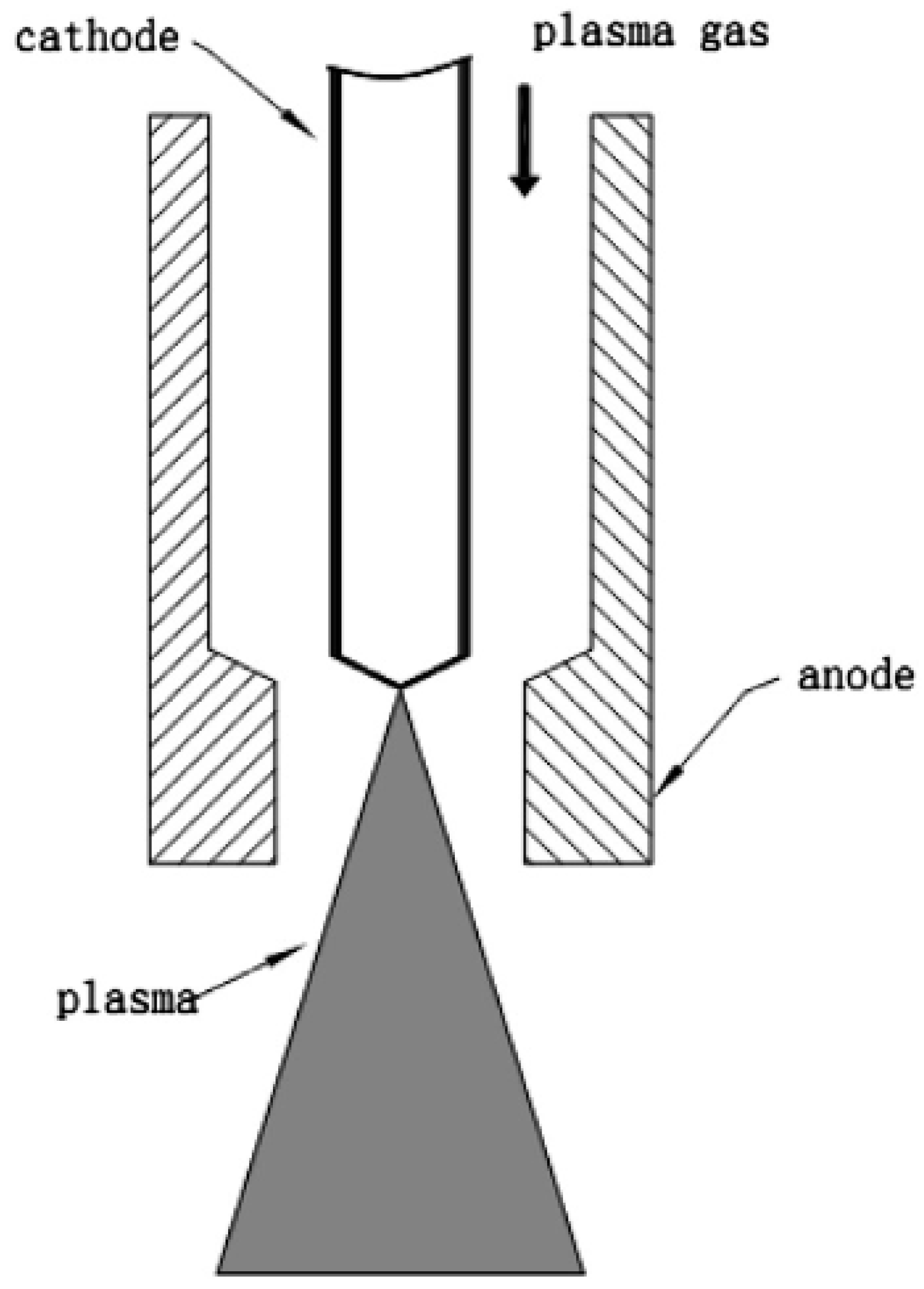
Figure 3 shows a non-transferred thermal DC plasma torch schematic diagram.
The advantage of DC thermal plasma is the ease of operation, expected thermal efficiency of 90% and a possible disadvantage of small jet volume [
31]. The thermal plasma used in this research work releases thermal energy as arc discharge [
24]. The DC thermal plasma used in the experiment is a low temperature thermal plasma with an initial steep ramp in the first minute from 25 to 625 °C, followed by a constant heating rate of 7.8 °C/min.
1.2. Goal and Scope
The goal of this study was to demonstrate a reliable DC thermal plasma system that provides enough thermal energy (approximately 1047 KJ/kg) for pyrolysis reactions. The pyrolysis reaction is allowed to proceed for τ = 30 min and the hydrocarbon products obtained are condensed using a cooling water condensation system at 25 °C. The oil sample is analyzed using FID-gas chromatography and its chemical components are identified. The objective of the experiment is to assess the DC thermal plasma’s ability to work in a LDPE pyrolysis reaction system.
1.3. LDPE Degradation Mechanism
The dissociation during thermoplastic pyrolysis involves free radical transfer through three steps: random scission, free radical saturation and group transfer and the rate of these reactions increases under higher operating temperatures. The first step involves long chain polymer cracking into smaller chains, including shorter chains like methane and ethane that includes free radicals. The second step involves side group saturation leaving free radicals in the middle carbons of the polymer. The final step is chain termination by adding an R group as seen below (
Figure 4) [
32]:
The pyrolysis reaction as seen above is initiated by breaking weaker bonds producing shorter chains with free radicals. The second step aims to have more stable intramolecular arrangements by hydrogen transfer to free radicals, while the third step involves R group addition and formation of smaller hydrocarbon molecules.
2. Methodology
In order to test the thermal performance of DC thermal plasma in pyrolysis reactors, Step 1 involves gathering reaction kinetics and thermal energy required by thermal plasma jet which includes understanding chemical and physical properties of LDPE, activation energies and reaction constant, k. In Step 2, the DC thermal plasma circuit is designed to achieve the operating temperature required by LDPE pyrolysis. Step 3 includes experimental work in an inert reactor, followed by yield calculations and GC-FID analysis of the pyrolysis oil collected from LDPE. In this step, the LDPE operating conditions are gathered, including optimum cracking temperature, reaction residence time, heating rate, expected product yield, chemical composition of oil produced, and dissociation or thermal energy needed per kg of LDPE. The first order reaction is derived from Equation (2) to anticipate mass loss per unit time for the batch reactor. Step 3 involves experimental setup of main equipment, operating procedure, oil products collection and FID-GC chemical analysis.
3. Experimental Setup
The experimental setup converts LDPE into oil using a DC thermal plasma as a heating source in a pyrolysis reaction at pressure = −0.95 bar and temperature = 550 °C which is maintained throughout the reaction. The equipment used in the pyrolysis reactor setup is described below in
Figure 5.
Vacuum Pump: All other valves should be closed and V-1 opened before starting the experiment. The vacuum pump is used to remove air before nitrogen supply to reactor.
Pure Nitrogen Gas Cylinder: All other valves should be closed and V-2 opened before allowing nitrogen gas to flow to the reactor at 2 bar. The vessel is filled with nitrogen till the pressure increases from −0.95 bar to 1 bar.
Condensation System: The condensation system cools gaseous hydrocarbon products at a constant rate of 35 °C/min. The thermal plasma system is switched off before starting the condensation process.
K-Type Thermocouple: A k-type thermocouple is inserted inside the closed vessel to get an accurate temperature profile. The thermocouple has an initial temperature of 23.5 °C before starting the experiment. The temperature profile is measured per min for 30 min.
DC Thermal Plasma: A 270 W thermal plasma probe at the top of the reactor is used as the heating source on a 15 g LDPE sample and the temperature profile is recorded using the k-type thermocouple. Both electrodes are 316 L stainless steel in a non-transferred thermal plasma setup (i.e., both electrodes are inside the reactor and have a sole plasma generation function) in a nitrogen atmoshphere. The direct current thermal plasma setup is not cooled by any cooling fluid but rather operates under pure nitrogen gas.
3.1. Operating Conditions
In non-catalytic LDPE pyrolysis, light oil starts to form at a low reaction temperature of 430 °C The reaction temperature and reaction residence time τ have an impact on the product yield and the composition of the oil produced. At 550 °C, under a nitrogen atmosphere, polyethylene can form 93 wt. % oil and 7 wt. % gas under optimum conditions [
33]. In order to minimize undesired products, nitrogen gas is used and any leak or rupture is avoided. When LDPE pyrolysis is performed at an operating temperature of 500 °C, the liquid yield obtained is 75.6 wt. %. The main components expected of LDPE are benzene, toluene, xylene, dimethyl-benzene, trimethyl-benzene, indane, indene, methyl-indenes, naphthalene, methyl-naphthalenes, ethyl-naphthalene, and other naphthalene derivatives [
34]. A slow heating rate and longer reaction residence time maximizes char production, while char content also increases with higher operating temperatures [
35]. Oxidation and combustion is avoided and its occurrence can increase char by up to 40–55 wt. % [
35]. Since the reaction residence time is longer than few seconds, a batch reactor or pressurized batch reactor is used. In the case of catalyst usage, a fluidized bed reactor is used [
35]. The heating rate has a direct impact on tar production and the stability of the pyrolysis oil produced. Thus, recommended heating rates are between 6 to 10 °C/min. Advantages of a batch reactor are a high conversion rate and stable hydrocarbon products [
36]. Disadvantages of a batch reactor include batch to batch product variability, high labor cost per batch and the difficulty of large scale production due to the extended residence time [
3].
3.2. DC Thermal Plasma Circuit Design
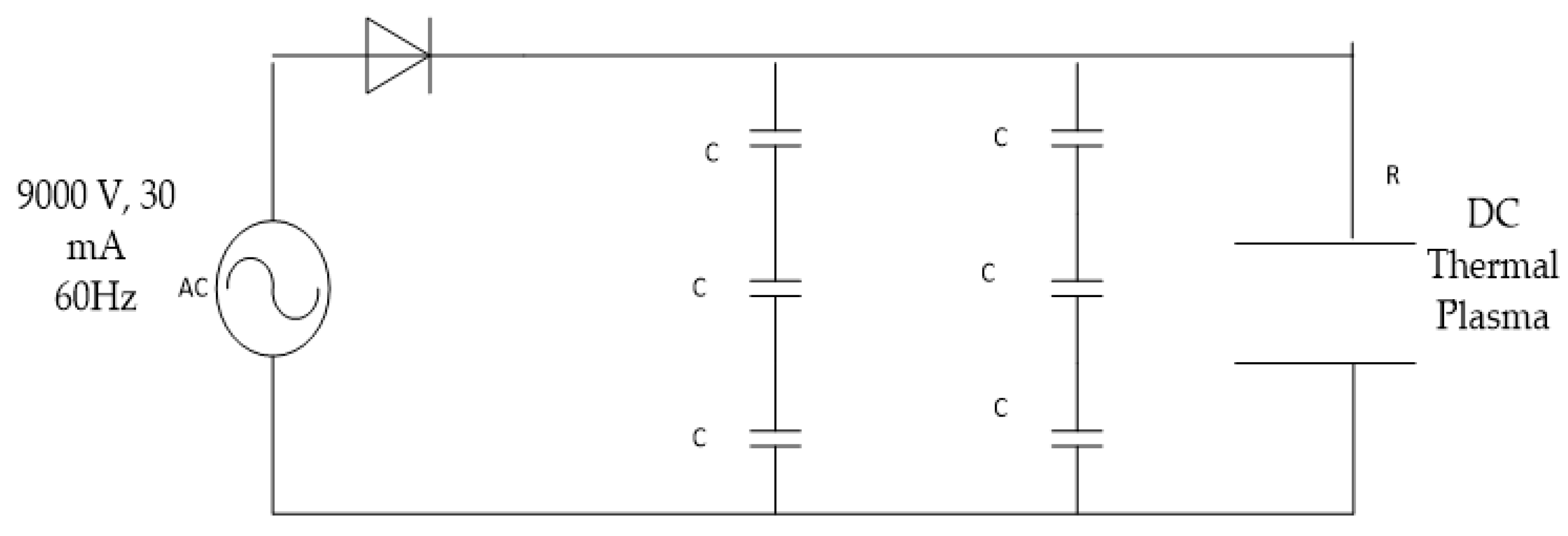
A non-transferred DC circuit of 9000 V, 30 mA at 60 Hz frequency is shown in
Figure 6. The circuit consists of capacitors, ceramic plates, diode and resistors. The input power source of the thermal plasma circuit is alternating current (AC), which is converted to direct current and the output impulse power is 270 W.
As shown above, C stands for capacitors, each of which has 1500 pF in two parallel loops connected to a diode that restricts the current to pass to the capacitors which store the electric energy. The thermal plasma circuit has three capacitors with 1500 pF each in series. The total circuit capacitance is shown below:
Using the above equation to calculate the total capacitance of three 1500 pF capacitors in series:
Thus the total capacitance in the two parallel loops is 1000 pF (i.e., 1 nF). Energy density emitted by DC thermal plasma over emission volume of 3.14 × 10
−7 m
3 is calculated below [
37]:
The density of LDPE is 0.92 g/cm
3 [
38] so the volume of a 15 g sample is 16.3 cm
3. The thermal energy needed for the reaction sample is 15.705 KJ. Thus, the energy released by the 270 W DC thermal plasma system satisfies the energy required.
The function of the diode is to convert the alternating current to direct current for thermal plasma generation creating a half wave rectifier as shown below in
Figure 7. A half cycle is used to charge the capacitors, and in the response time of absence of current, the capacitors release the charge load at the electrodes generating a thermal plasma torch at the vacuum operating pressure of −0.95 bar.
The thermal plasma impulse power is calculated: P
Impulse: 270 W. A K-type thermocouple installed few millimeters away from the LDPE sample shows 625.6 °C as an initial temperature and a maximum temperature of 890 °C is achieved. The temperature profile is presented in the Results section. Thermal plasma can achieve very high temperatures and special precautions need to be taken for safety and health standards [
39]. The temperature limit for the experiment is set to 1000 °C. Several precautions should be taken is a higher temperature is detected using the K-type thermocouple:
Switching off the main power supply.
Pressure test before switching on the thermal plasma system to prevent leaks during operations.
Ensure the pressure is below atmospheric for optimum plasma operation.
4. Experimental Results
As seen below, a 270 W thermal plasma operating at a pressure = −0.95 bar using non-transferred DC thermal plasma in an inert vacuum chamber over a ceramic nozzle as shown in
Figure 8 and
Figure 9.
The DC thermal plasma is integrated inside a 1 L pyrolysis reactor and tested on a 15 g LDPE sample. After few min the LDPE sample shrinks and changes into molten plastic, followed by formation of gaseous hydrocarbon products. The gaseous products are allowed to condense into oil products. In
Figure 10, DC thermal plasma emissions on a 15 g LDPE setup is shown.
At the end of the reaction, the gaseous hydrocarbon products are allowed to escape at atmospheric pressure through a condensation system to form liquid hydrocarbons and waxes are allowed to condense as shown in
Figure 11 and identified using GC-FID.
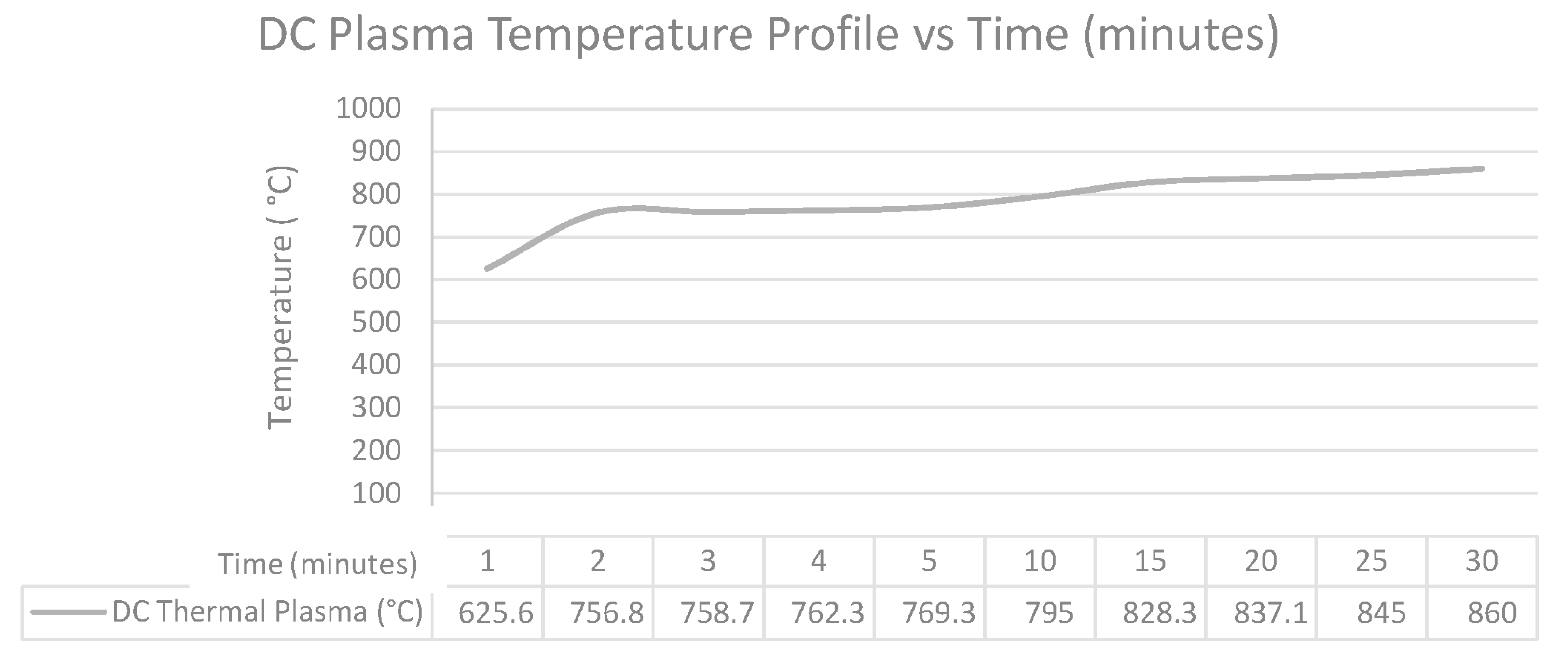
4.1. Temperature Profile
The temperature profile is recorded using the K-type thermocouple for the DC thermal plasma. As seen in the temperature profile, the initial temperature reading is 625.6 °C and a maximum temperature of 860 °C is reached. The thermal plasma showed an acceptable gradual temperature increase which is favored in LDPE pyrolysis.
As seen in
Figure 12, the DC thermal plasma produces a high temperature profile on the 15 g thermoplastic sample and can be easily controlled by the current input to the plasma circuit. It can also be concluded that the DC thermal plasma with 270 W can achieve higher temperatures than required for pyrolysis (up to 860 °C).
4.2. Collection of Oil Samples
The hydrocarbon gaseous products from the pyrolysis experiment pass through a condensation system and the volatile oil sample is analyzed using FID gas chromatography. The oil sample collected from a 15 g sample of LDPE and condensed oil is shown in
Figure 13.
The hydrocarbon gaseous products are condensed and the liquid products are collected using potable cooling water at 25 °C at a cooling rate of 35 °C/min for 15 min as shown in Figure 15.
4.3. Headspace GC-FID Analysis
The oil sample is analyzed using a head space FID-GC with methanol solution. The oil sample shows the existence of the following hydrocarbon compounds:
The first chromatogram in
Figure 14 was identified using headspace GC with an FID to determine the C
6 to C
10 hydrocarbon fraction.
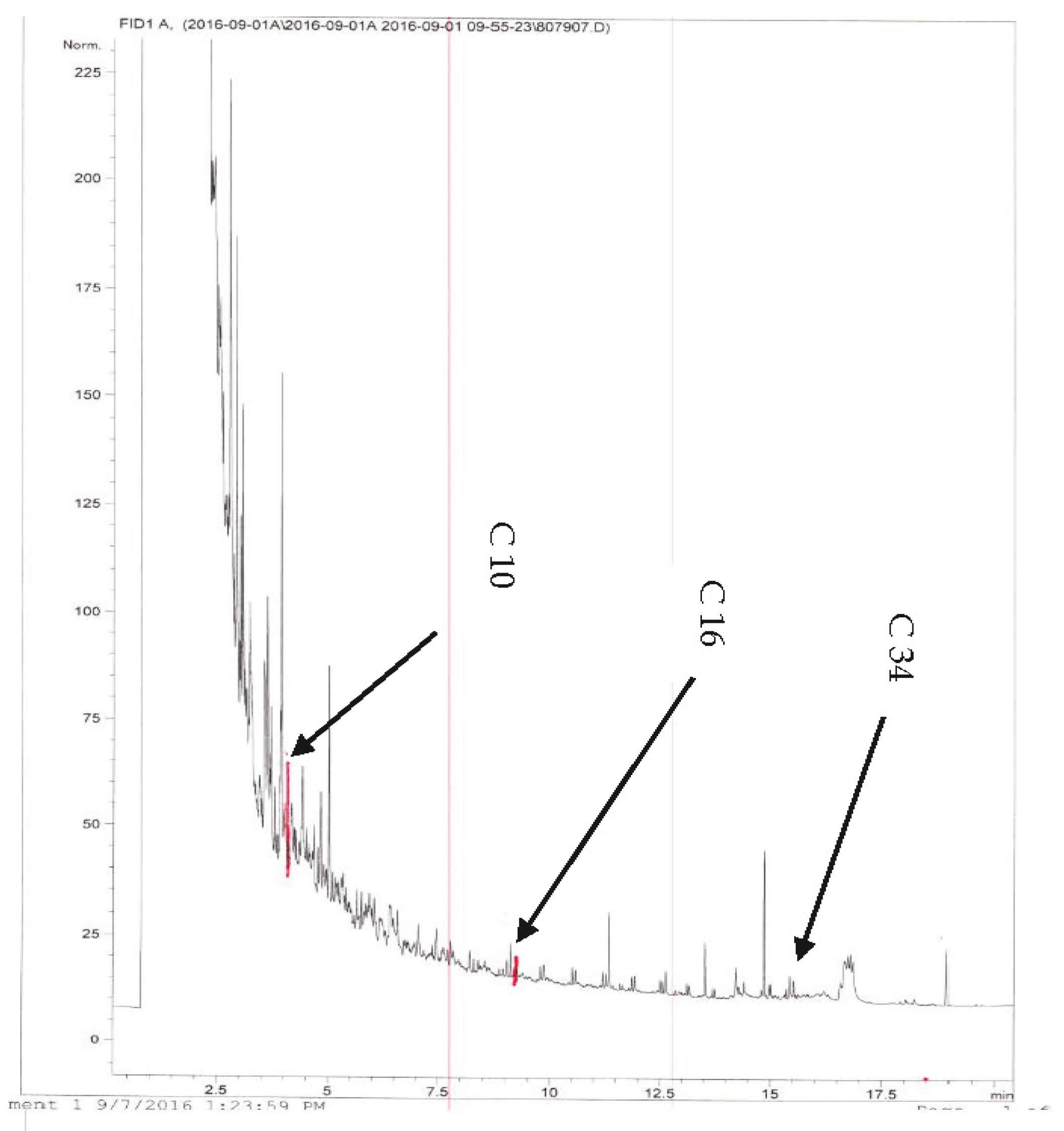
The second chromatogram was analyzed using GC-FID with methanol solvent. The labeled retention times are for decane (C
10), hexadecane (C
16) and tetratriacontane (C
34) as shown in
Figure 15.
The quantitative analysis of the pyrolysis oil is shown in
Table 3. The data displayed in
shows 87.9 wt. % 1,4-dichlorobenzene, minor percentages of benzene, and ethylbenzene. In terms of hydrocarbon analysis it shows the highest concentration of 2340
for (C
10–C
16), followed by C
16–C
34 and small traces of heavier C
34–C
50 hydrocarbon content.
4.4. Product Yield Results
As mentioned in the Introduction, the product yield from LDPE pyrolysis at 550 °C is 93 wt. %. The lower hydrocarbon liquid yields can be due to higher operating temperatures which increases the formation of light hydrocarbon products such as methane and ethane. The final tar and wax sample are weighted and considered undesired product. Below (
Table 4) are the results from a 15 g LDPE sample.