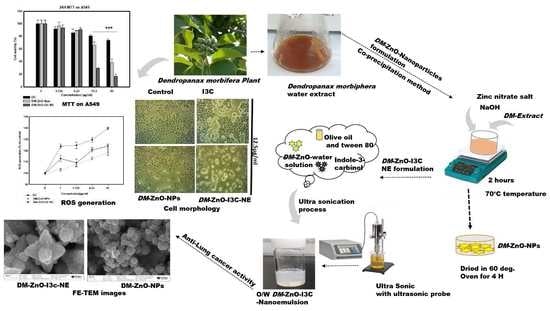
Dendropanax Morbifera Extract-Mediated ZnO Nanoparticles Loaded with Indole-3-Carbinol for Enhancement of Anticancer Efficacy in the A549 Human Lung Carcinoma Cell Line
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Chemicals
2.3. Preparation of DM Extract
2.4. Synthesis of DM-ZnO NPs from the Extract
2.5. Synthesis of DM-ZnO-I3C NE
2.6. Characterization
Cell culture
3. Results and Discussion
3.1. Physicochemical Properties of the Synthesized Nano Formulation
3.2. UV-Vis Analysis
3.3. FTIR Spectroscopic Analysis
3.4. XRD Analysis
3.5. FE-TEM, Elemental Mapping, and FE-SEM Analysis
3.6. Size Measurement Analysis
3.7. In Vitro Cell Cytotoxicity Analysis
3.8. ROS Generation Induced by DM-ZnO-I3C-NE
3.9. Effect of DM-ZnO-I3C-NE on Cell Morphology
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kim, C.G.; Castro-Aceituno, V.; Abbai, R.; Lee, H.A.; Simu, S.Y.; Han, Y.; Hurh, J.; Kim, Y.-J.; Yang, D.C. Caspase-3/MAPK pathways as main regulators of the apoptotic effect of the phyto-mediated synthesized silver nanoparticle from dried stem of Eleutherococcus senticosus in human cancer cells. Biomed. Pharmacother. 2018, 99, 128–133. [Google Scholar] [CrossRef]
- Wang, C.; Mathiyalagan, R.; Kim, Y.J.; Castro-Aceituno, V.; Singh, P.; Ahn, S.; Wang, D.; Yang, D.C. Rapid green synthesis of silver and gold nanoparticles using Dendropanax morbifera leaf extract and their anticancer activities. Int. J. Nanomed. 2016, 11, 3691. [Google Scholar]
- Mahaling, B.; Verma, M.; Mishra, G.; Chaudhuri, S.; Dutta, D.; Sivakumar, S. Fate of GdF3 nanoparticles-loaded PEGylated carbon capsules inside mice model: A step toward clinical application. Nanotoxicology 2020, 14, 577–594. [Google Scholar] [CrossRef]
- Chen, F.; Ehlerding, E.B.; Cai, W. Theranostic nanoparticles. J. Nucl. Med. 2014, 55, 1919–1922. [Google Scholar] [CrossRef] [Green Version]
- Rupa, E.J.; Anandapadmanaban, G.; Chokkalingam, M.; Li, J.F.; Markus, J.; Soshnikova, V.; Perez, Z.; Yang, D.-C. Cationic and anionic dye degradation activity of Zinc oxide nanoparticles from Hippophae rhamnoides leaves as potential water treatment resource. Optik 2019, 181, 1091–1098. [Google Scholar]
- Yusof, H.M.; Mohamad, R.; Zaidan, U.H. Microbial synthesis of zinc oxide nanoparticles and their potential application as an antimicrobial agent and a feed supplement in animal industry: A review. J. Anim. Sci. Biotechnol. 2019, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Aljabali, A.A.; Akkam, Y.; Al Zoubi, M.S.; Al-Batayneh, K.M.; Al-Trad, B.; Abo Alrob, O.; Alkilany, A.M.; Benamara, M.; Evans, D.J. Synthesis of gold nanoparticles using leaf extract of Ziziphus zizyphus and their antimicrobial activity. Nanomaterials 2018, 8, 174. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.Y.; Kim, K.-S.; Lee, Y.-C.; Moon, H.-I.; Lee, J.-H. Oleifolioside A, a new active compound, attenuates LPS-stimulated iNOS and COX-2 expression through the downregulation of NF-κB and MAPK activities in RAW 264.7 macrophages. Evid. Based Complement. Altern. Med. 2012, 2012, 637512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.-E.; Sapkota, K.; Choi, J.-H.; Kim, M.-K.; Kim, Y.H.; Kim, K.M.; Kim, K.J.; Oh, H.-N.; Kim, S.-J.; Kim, S. Rutin from Dendropanax morbifera Leveille protects human dopaminergic cells against rotenone induced cell injury through inhibiting JNK and p38 MAPK signaling. Neurochem. Res. 2014, 39, 707–718. [Google Scholar] [CrossRef]
- Jin, C.-Y.; Yu, H.Y.; Park, C.; Han, M.H.; Hong, S.H.; Kim, K.-S.; Lee, Y.-C.; Chang, Y.-C.; Cheong, J.; Moon, S.-K. Oleifolioside B-mediated autophagy promotes apoptosis in A549 human non-small cell lung cancer cells. Int. J. Oncol. 2013, 43, 1943–1950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duvall, M.N. FDA Regulation of Nanotechnology; Beveridge and Diamond, PG: Washington, DC, USA, 2012. [Google Scholar]
- Zhang, Z.-Y.; Xiong, H.-M. Photoluminescent ZnO nanoparticles and their biological applications. Materials 2015, 8, 3101–3127. [Google Scholar] [CrossRef]
- Akhtar, M.J.; Ahamed, M.; Kumar, S.; Khan, M.M.; Ahmad, J.; Alrokayan, S.A. Zinc oxide nanoparticles selectively induce apoptosis in human cancer cells through reactive oxygen species. Int. J. Nanomed. 2012, 7, 845. [Google Scholar]
- Bailey, G.S.; Tilton, S.; Williams, D.E. Mechanisms of Tumor Inhibition, and Tumor Promotion, by Dietary Indole-3-Carbinol; AACR: Philadelphia, PA, USA, 2006. [Google Scholar]
- Rupa, E.J.; Anandapadmanaban, G.; Mathiyalagan, R.; Yang, D.-C. Synthesis of zinc oxide nanoparticles from immature fruits of Rubus coreanus and its catalytic activity for degradation of industrial dye. Optik 2018, 172, 1179–1186. [Google Scholar] [CrossRef]
- Teo, B.S.X.; Basri, M.; Zakaria, M.R.S.; Salleh, A.B.; Rahman, R.N.Z.R.A.; Rahman, M.B.A. A potential tocopherol acetate loaded palm oil esters-in-water nanoemulsions for nanocosmeceuticals. J. Nanobiotechnol. 2010, 8, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira, E.; Boock, K.; Maruno, M.; Rocha-Filho, P.A.d. Accelerated stability and moisturizing capacity of emulsions presenting lamellar gel phase obtained from brazilian natural raw material. J. Dispers. Sci. Technol. 2011, 32, 1135–1139. [Google Scholar] [CrossRef]
- Junyaprasert, V.B.; Teeranachaideekul, V.; Souto, E.B.; Boonme, P.; Müller, R.H. Q10-loaded NLC versus nanoemulsions: Stability, rheology and in vitro skin permeation. Int. J. Pharm. 2009, 377, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, D.S.; Pereira, T.A.; Maciel, N.R.; Bortoloto, J.; Viera, G.S.; Oliveira, G.C.; Rocha-Filho, P.A. Formation and stability of oil-in-water nanoemulsions containing rice bran oil: In vitro and in vivo assessments. J. Nanobiotechnol. 2011, 9, 44. [Google Scholar] [CrossRef] [PubMed]
- Aleixandre-Tudo, J.L.; du Toit, W. The role of UV-visible spectroscopy for phenolic compounds quantification in winemaking. In Frontiers and New Trends in the Science of Fermented Food and Beverages; IntechOpen: London, UK, 2018. [Google Scholar]
- Mabry, T.; Markham, K.R.; Thomas, M.B. The Systematic Identification of Flavonoids; Springer Science & Business Media: Berlin, Germany, 2012. [Google Scholar]
- Chikkanna, M.M.; Neelagund, S.E.; Rajashekarappa, K.K. Green synthesis of zinc oxide nanoparticles (ZnO NPs) and their biological activity. SN Appl. Sci. 2019, 1, 117. [Google Scholar] [CrossRef] [Green Version]
- Talam, S.; Karumuri, S.R.; Gunnam, N. Synthesis, characterization, and spectroscopic properties of ZnO nanoparticles. ISRN Nanotechnol. 2012, 2012, 6. [Google Scholar] [CrossRef] [Green Version]
- Namvar, F.; Azizi, S.; Rahman, H.S.; Mohamad, R.; Rasedee, A.; Soltani, M.; Rahim, R.A. Green synthesis, characterization, and anticancer activity of hyaluronan/zinc oxide nanocomposite. OncoTargets Ther. 2016, 9, 4549. [Google Scholar]
- Markus, J.; Mathiyalagan, R.; Kim, Y.-J.; Han, Y.; Jiménez-Pérez, Z.E.; Veronika, S.; Yang, D.-C. Synthesis of hyaluronic acid or O-carboxymethyl chitosan-stabilized ZnO–ginsenoside Rh2 nanocomposites incorporated with aqueous leaf extract of Dendropanax morbifera Léveille: In vitro studies as potential sunscreen agents. N. J. Chem. 2019, 43, 9188–9200. [Google Scholar] [CrossRef]
- Handore, K.; Bhavsar, S.; Horne, A.; Chhattise, P.; Mohite, K.; Ambekar, J.; Pande, N.; Chabukswar, V. Novel green route of synthesis of ZnO nanoparticles by using natural biodegradable polymer and its application as a catalyst for oxidation of aldehydes. J. Macromol. Sci. Part A 2014, 51, 941–947. [Google Scholar] [CrossRef]
- Upadhyaya, L.; Singh, J.; Agarwal, V.; Pandey, A.; Verma, S.P.; Das, P.; Tewari, R. In situ grafted nanostructured ZnO/carboxymethyl cellulose nanocomposites for efficient delivery of curcumin to cancer. J. Polym. Res. 2014, 21, 550. [Google Scholar] [CrossRef]
- Ramesh, M.; Anbuvannan, M.; Viruthagiri, G. Green synthesis of ZnO nanoparticles using Solanum nigrum leaf extract and their antibacterial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 136, 864–870. [Google Scholar] [CrossRef]
- Ali, K.; Dwivedi, S.; Azam, A.; Saquib, Q.; Al-Said, M.S.; Alkhedhairy, A.A.; Musarrat, J. Aloe vera extract functionalized zinc oxide nanoparticles as nanoantibiotics against multi-drug resistant clinical bacterial isolates. J. Colloid Interface Sci. 2016, 472, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, S.-B.; Sarrafzadeh, M.-H.; Fakhroueian, Z.; Khorramizadeh, M.R. Flower-like curcumin-loaded folic acid-conjugated ZnO-MPA-βcyclodextrin nanostructures enhanced anticancer activity and cellular uptake of curcumin in breast cancer cells. Mater. Sci. Eng. C 2019, 103, 109827. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Vo, C.; Taylor, M.; Smith, B.R. Non-spherical micro-and nanoparticles in nanomedicine. Mater. Horiz. 2019, 6, 1094–1121. [Google Scholar] [CrossRef]
- Sultana, S.; Djaker, N.; Boca-Farcau, S.; Salerno, M.; Charnaux, N.; Astilean, S.; Hlawaty, H.; de La Chapelle, M.L. Comparative toxicity evaluation of flower-shaped and spherical gold nanoparticles on human endothelial cells. Nanotechnology 2015, 26, 055101. [Google Scholar] [CrossRef] [Green Version]
- Singh, P.; Singh, H.; Castro-Aceituno, V.; Ahn, S.; Kim, Y.J.; Farh, M.E.-A.; Yang, D.C. Engineering of mesoporous silica nanoparticles for release of ginsenoside CK and Rh2 to enhance their anticancer and anti-inflammatory efficacy: In vitro studies. J. Nanopart. Res. 2017, 19, 257. [Google Scholar] [CrossRef]
- Ma, D.; Chen, C.; Jhanji, V.; Xu, C.; Yuan, X.; Liang, J.; Huang, Y.; Cen, L.; Ng, T.K. Expression of SARS-CoV-2 receptor ACE2 and TMPRSS2 in human primary conjunctival and pterygium cell lines and in mouse cornea. Eye 2020, 34, 1–8. [Google Scholar] [CrossRef]
- Ostrovsky, S.; Kazimirsky, G.; Gedanken, A.; Brodie, C. Selective cytotoxic effect of ZnO nanoparticles on glioma cells. Nano Res. 2009, 2, 882–890. [Google Scholar] [CrossRef] [Green Version]
- Chung, I.M.; Kim, M.Y.; Park, S.D.; Park, W.H.; Moon, H.I. In vitro evaluation of the antiplasmodial activity of Dendropanax morbifera against chloroquine-sensitive strains of Plasmodium falciparum. Phytother. Res. 2009, 23, 1634–1637. [Google Scholar] [CrossRef] [PubMed]
- Aceituno, V.C.; Ahn, S.; Simu, S.Y.; Wang, C.; Mathiyalagan, R.; Yang, D.C. Silver nanoparticles from Dendropanax morbifera Léveille inhibit cell migration, induce apoptosis, and increase generation of reactive oxygen species in A549 lung cancer cells. Vitr. Cell. Dev. Biol. Anim. 2016, 52, 1012–1019. [Google Scholar] [CrossRef]
- Hanley, C.; Layne, J.; Punnoose, A.; Reddy, K.á.; Coombs, I.; Coombs, A.; Feris, K.; Wingett, D. Preferential killing of cancer cells and activated human T cells using ZnO nanoparticles. Nanotechnology 2008, 19, 295103. [Google Scholar] [CrossRef] [Green Version]
- Hsin, Y.-H.; Chen, C.-F.; Huang, S.; Shih, T.-S.; Lai, P.-S.; Chueh, P.J. The apoptotic effect of nanosilver is mediated by a ROS-and JNK-dependent mechanism involving the mitochondrial pathway in NIH3T3 cells. Toxicol. Lett. 2008, 179, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Babizhayev, M.A.; Savel’yeva, E.L.; Moskvina, S.N.; Yegorov, Y.E. Telomere length is a biomarker of cumulative oxidative stress, biologic age, and an independent predictor of survival and therapeutic treatment requirement associated with smoking behavior. Am. J. Ther. 2011, 18, e209–e226. [Google Scholar] [CrossRef]
- Das, T.K.; Wati, M.R.; Fatima-Shad, K. Oxidative stress gated by Fenton and Haber Weiss reactions and its association with Alzheimer’s disease. Arch. Neurosci. 2015, 2. [Google Scholar] [CrossRef] [Green Version]
- Jaiswal, M.; Dudhe, R.; Sharma, P. Nanoemulsion: An advanced mode of drug delivery system. 3 Biotech. 2015, 5, 123–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kassie, F.; Melkamu, T.; Endalew, A.; Upadhyaya, P.; Luo, X.; Hecht, S.S. Inhibition of lung carcinogenesis and critical cancer-related signaling pathways by N-acetyl-S-(N-2-phenethylthiocarbamoyl)-l-cysteine, indole-3-carbinol and myo-inositol, alone and in combination. Carcinogenesis 2010, 31, 1634–1641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camerlingo, R.; Miceli, R.; Marra, L.; Rea, G.; D’Agnano, I.; Nardella, M.; Montella, R.; Morabito, A.; Normanno, N.; Tirino, V. Conditioned medium of primary lung cancer cells induces EMT in A549 lung cancer cell line by TGF-ß1 and miRNA21 cooperation. PLoS ONE 2019, 14, e0219597. [Google Scholar] [CrossRef] [PubMed] [Green Version]










| Sample (Name) | DM-ZnO NPs (Aqueous Solution) | Olive Oil | I3C Drug | Surfactant (Tween 80) |
|---|---|---|---|---|
| S1 | 82% (37.5 mg) | 10% | 5% (12.5 mg) | 3% |
| S2 | 80% (37.5 mg) | 8% | 5% (12.5 mg) | 7% |
| S3 | 80% (37.5 mg) | 5% | 5% (12.5 mg) | 10% |
| Lattice Plane | Position of Peak (2θ) | d-Spacing Value (A0) | FWHM Value (2θ) | Size (nm) | Average (nm) |
|---|---|---|---|---|---|
| 100 | 31.81 | d = 2.81 | 0.8242 | 10.03 | |
| 002 | 34.45 | d = 2.60 | 0.5691 | 14.63 | 11.52 |
| 101 | 36.29 | d = 2.47 | 0.8438 | 9.92 | |
| 102 | 47.77 | d = 1.90 | 2.3549 | 3.69 | |
| 110 | 56.67 | d = 1.62 | 1.3541 | 6.67 | |
| 103 | 62.89 | d = 1.48 | 1.9036 | 4.90 | |
| 112 | 67.99 | d = 1.37 | 2.7474 | 3.49 |
| Lattice Plane | Position of Peak (2θ) | d-Spacing Value (A0) | FWHM Value (2θ) | Size (nm) | Average (nm) |
|---|---|---|---|---|---|
| 100 | 31.78 | d = 2.80 | 0.7261 | 11.39 | |
| 002 | 34.47 | d = 2.59 | 0.3336 | 24.95 | 16.0708 |
| 101 | 36.29 | d = 2.47 | 0.7065 | 11.84 | |
| 102 | 47.62 | d = 1.90 | 2.5904 | 3.35 | |
| 110 | 56.69 | d = 1.62 | 1.1382 | 7.94 | |
| 103 | 62.89 | d = 1.47 | 2.0606 | 4.52 | |
| 112 | 68.05 | d = 1.37 | 2.1587 | 4.44 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rupa, E.J.; Arunkumar, L.; Han, Y.; Kang, J.P.; Ahn, J.C.; Jung, S.-K.; Kim, M.; Kim, J.Y.; Yang, D.-C.; Lee, G.J. Dendropanax Morbifera Extract-Mediated ZnO Nanoparticles Loaded with Indole-3-Carbinol for Enhancement of Anticancer Efficacy in the A549 Human Lung Carcinoma Cell Line. Materials 2020, 13, 3197. https://doi.org/10.3390/ma13143197
Rupa EJ, Arunkumar L, Han Y, Kang JP, Ahn JC, Jung S-K, Kim M, Kim JY, Yang D-C, Lee GJ. Dendropanax Morbifera Extract-Mediated ZnO Nanoparticles Loaded with Indole-3-Carbinol for Enhancement of Anticancer Efficacy in the A549 Human Lung Carcinoma Cell Line. Materials. 2020; 13(14):3197. https://doi.org/10.3390/ma13143197
Chicago/Turabian StyleRupa, Esrat Jahan, Lakshminarayanan Arunkumar, Yaxi Han, Jong Pyo Kang, Jong Chan Ahn, Seok-Kyu Jung, Mia Kim, Jong Yun Kim, Deok-Chun Yang, and Gyong Jai Lee. 2020. "Dendropanax Morbifera Extract-Mediated ZnO Nanoparticles Loaded with Indole-3-Carbinol for Enhancement of Anticancer Efficacy in the A549 Human Lung Carcinoma Cell Line" Materials 13, no. 14: 3197. https://doi.org/10.3390/ma13143197