Abstract
SrCl2·6H2O has been shown to act as an efficient catalyst for the conversion of aldehydes or ketones into the corresponding gem-dihydroperoxides (DHPs) by treatment with aqueous H2O2 (30%) in acetonitrile. The reactions proceed under mild and neutral conditions at room temperature to afford good to excellent yields of product.
1. Introduction
In recent years, much research has been directed towards gem-dihydroperoxides (DHPs) [1], due to their importance as useful intermediates in the synthesis of various peroxides, including tetraoxanes [2,3,4,5,6,7,8,9], and their analogues such as silatetroxanes [10], spirobisperoxyketals [11], and tetroxycycloalkanes [12], and epoxidation of α,β-unsaturated ketones [13]. These compounds have also recently been utilized as effective reagents in: (i) oxidation of various compounds [14] such as sulfides [15], (ii) enantioselective oxidation of 2-substituted 1,4-naphthoquinones [16], and (iii) as initiators in polymerization reactions [17,18]. It is also remarkable that gem-dihydroperoxides are relevant to peroxidic antimalarial drugs [2,19,20,21,22,23] possessing the gem-peroxy linkage as a salient structural feature [23,24,25,26] in common with many well-known antimalarial cyclic organic peroxides [1,2,27,28,29,30,31,32,33,34,35]. Most of the documented protocols for the synthesis of gem-dihydroperoxides in the literature suffer from significant drawbacks such as the use of strong acidic media, concentrated H2O2 and low yields [1]. These methods mainly utilize a Brönsted or Lewis acid e.g., HCO2H [12,20,36], NaHSO4-SiO2 [37], H2SO4 [38], F3CCO2H [39], H2WO4 [29,38], and BF3·OEt2 [30,39] to promote the conversion of ketones, ketals or enol ethers into the corresponding DHPs on treatment with aqueous H2O2. Other catalysts such as methyltrioxorhenium (prepared from Re2O7) [2], ceric ammonium nitrate (CAN) [32], and iodine [33] have also been reported to promote such transformations. However, these methods are not mild enough to offer general applicability and have limitations such as low yields, long reaction times, use of high concentration of H2O2 and incompatibility with sensitive functional groups. Recently, Dussault has reported a remarkably mild and highly efficient protocol for Re2O7-catalyzed conversion of ketones, aldehydes or acetals into 1,1-dihydroperoxides by H2O2 which represents a major improvement [34].
2. Results and Discussion
As part of our ongoing efforts to develop new methods for the synthesis of DHPs, we report here another new and highly efficient and inexpensive catalyst SrCl2·6H2O to promote the synthesis of gem-DHPs from ketones and aldehydes employing aqueous H2O2 (30%) at room temperature. To achieve suitable reaction conditions, i.e., lower reaction times and higher yields, for the conversion of the ketones and aldehydes into their corresponding DHPs, various Lewis acid catalysts and solvents were investigated using 3-pentanone, cyclohexanone, acetophenone, and benzaldehyde as test compounds at room temperature (Table 1).

Table 1.
Effects of catalyst and solvent in the synthesis of gem-DHPs. a
| Entry | Ketone 1/Aldehyde 3 | Catalyst | Solvent | Time (h) | Yield (%)b |
|---|---|---|---|---|---|
| 1 | 3-pentanone | SrCl3·6H2O | CH3CN | 3 | 95 |
| 2 | 3-pentanone | SrCl3·6H2O | CH2Cl2 | 6 | 78 |
| 3 | 3-pentanone | SrCl3·6H2O | Et2O | 8 | 56 |
| 4 | 3-pentanone | SrCl3·6H2O | AcOEt | 6 | 82 |
| 5 | 3-pentanone | SbCl3 | CH3CN | 8 | 48 |
| 6 | 3-pentanone | CeO2 | CH3CN | 10 | 45 |
| 7 | 3-pentanone | CrCl3·6H2O | CH3CN | 8 | 75 |
| 8 | 3-pentanone | KF-Al2O3 | CH3CN | 10 | Trace |
| 9 | Cyclohexanone | SrCl3·6H2O | CH3CN | 3 | 94 |
| 10 | Cyclohexanone | SbCl3 | CH3CN | 7 | 55 |
| 11 | Cyclohexanone | CeO2 | CH3CN | 8 | 50 |
| 12 | Cyclohexanone | CrCl3·6H2O | CH3CN | 6 | 70 |
| 13 | Cyclohexanone | KF-Al2O3 | CH3CN | 10 | Trace |
| 14 | Acetophenone | SrCl3·6H2O | CH3CN | 10 | 45 |
| 15 | Acetophenone | SbCl3 | CH3CN | 12 | 23 |
| 16 | Acetophenone | CeO2 | CH3CN | 12 | 15 |
| 17 | Acetophenone | CrCl3·6H2O | CH3CN | 10 | 28 |
| 18 | Acetophenone | KF-Al2O3 | CH3CN | 20 | 0 |
| 19 | Benzaldehyde | SrCl3·6H2O | CH3CN | 10 | 54 |
| 20 | Benzaldehyde | SbCl3 | CH3CN | 15 | 32 |
| 21 | Benzaldehyde | CeO2 | CH3CN | 15 | 15 |
| 22 | Benzaldehyde | CrCl3·6H2O | CH3CN | 12 | 22 |
| 23 | Benzaldehyde | KF-Al2O3 | CH3CN | 20 | 0 |
a Conditions: Ketone and aldehyde (1 mmol), solvent (4 mL), catalyst (0.1 mmol), 30% aq. H2O2 (3 mL), reactions are carried out at rt. b Isolated yields.
As can be seen in Table 1, the reaction worked best in terms of yield and reaction time with aqueous H2O2 (30%) when SrCl2·6H2O (10 mol %) was used as a catalyst. The other catalysts such as SbCl3, CeO2 and CrCl3·6H2O gave moderate to low yields while KF-Al2O3 was found to be completely unsuitable for the synthesis of these DHPs. Effects of the solvents such as CH2Cl2, Et2O, MeCN and AcOEt on the yields of the products were tested and the results are summarized in Table 1. Acetonitrile appeared as a much better solvent compared with other ones. This suggests that solvent polarity plays an important role in the synthesis of DHPs.
This success encouraged us to extend these reaction conditions to a variety of cyclic and acyclic aliphatic ketones 1a-g using aqueous H2O2 (30%) in the presence of 10 mol% amount of SrCl2·6H2O as a chosen catalyst in acetonitrile at room temperature. The corresponding gem-dihydroperoxides 2a-g were produced in high to excellent yields (90–98%) within 3–12 h (Table 2, Scheme 1). Similarly, aromatic ketones 1h-j and aromatic aldehydes 1l-p were converted to their corresponding gem-DHPs 2h-j and 2l-p in (45–68%) and (52–75%) yields respectively (Table 1). However, under the same reaction condition no conversion to gem-DHP was observed for benzophenone 1k and it was recovered almost intact after 12 hours. This can possibly be accounted for by the strong resonance stabilization and steric effects exerted by two phenyl groups.
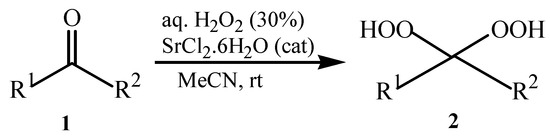
Scheme 1.
Synthesis of gem-dihydroperoxides 2a-g.
Scheme 1.
Synthesis of gem-dihydroperoxides 2a-g.
As previously reported by Rieche [40] and Žmitek et al. [41], we observed in the present protocol that simple, nonaromatic aldehydes such as octanal 3q and dihydrocinnamaldehyde 3r, which easily undergo hydration [42], reacted differently from the ketones and aromatic aldehydes. Under the same reaction conditions which converted ketones and aromatic aldehydes into their corresponding DHPs, both alkyl aldehydes-octanal 3q and dihydrocinnamaldehyde 3r-were not converted into their corresponding DHPs but instead into hydroxyl-hydroperoxides 4q and 4r in high yields (Table 1, Scheme 2), that is the addition of only one molecule of hydrogen peroxide to the carbonyl group has occurred. This implies that our protocol can furnish another hitherto unreported approach to 1,1-hydroxyhydroperoxides from aliphatic aldehydes.
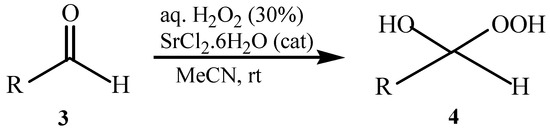
Scheme 2.
Syntheiss of hydroxyl-hydroperoxides 4q and 4r.
Scheme 2.
Syntheiss of hydroxyl-hydroperoxides 4q and 4r.

Table 2.
Synthesis of gem-dihydroperoxides with SrCl2·6H2O (cat.)/30% aq. H2O2. a
| Entry | Ketone 1/ Aldehyde 3 | Product2 or 4 b | Time (h) | Yield (%)c |
|---|---|---|---|---|
| a | 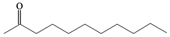 | 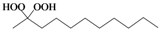 | 4 | 96 |
| b |  |  | 4 | 98 |
| c | 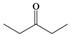 | 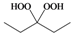 | 3 | 95 |
| d | 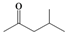 | 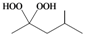 | 3 | 92 |
| e | 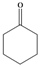 | 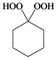 | 3 | 94 |
| f |  | 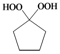 | 4 | 97 |
| g |  | 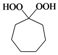 | 3 | 90 |
| h |  | 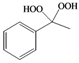 | 10 | 45 |
| i | 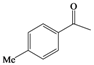 | 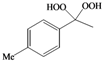 | 9 | 68 |
| j | 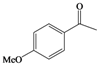 | 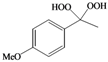 | 8 | 62 |
| k |  |  | 12 |  |
| l | 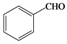 | 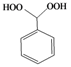 | 10 | 54 |
| m | 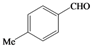 |  | 11 | 52 |
| n | 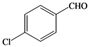 | 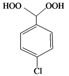 | 9 | 75 |
| o | 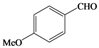 | 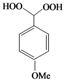 | 9 | 72 |
| Pd | 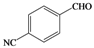 | 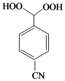 | 10 | 73 |
| q | 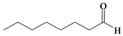 | 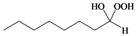 | 5 | 90 |
| r |  |  | 5 | 92 |
a Conditions: Ketone and aldehyde (1 mmol), acetonitrile (4 mL), SrCl2·6H2O (0.1 mmol), 30% aq. H2O2 (3 mL), reactions are carried out at rt. b The structures of the products were established from their physical properties and spectral (1H-, 13C-NMR and MS) analysis and compared with the literature. c Isolated Yield. d A new derivative.
3. Experimental
3.1. General
Chemicals were obtained from Merck. FT-IR spectra were recorded on a Shimadzu 435-U-04 spectrophotometer (KBr pellets). 1H- and 13C-NMR spectra were recorded on a 200 (50) MHz Varian or JEOL FX 90 MHz spectrometers in CDCl3 and DMSO-d6 solution, and are reported in δ units with TMS as internal standard. Melting points were determined in open capillary tubes in a Stuart SMP3 apparatus and uncorrected.
3.2. General procedure for synthesis of gem-dihydroperoxides
Caution: Peroxidic compounds are potentially explosive and require precautions in handling (shields, fume hoods, absence of transition metal salts and heating).
A mixture of carbonyl substrates 1 or 3 (1 mmol), 30% aqueous H2O2 (3 mL) and SrCl2·6H2O (0.1 mmol) in MeCN (4 mL) was stirred at room temperature for 3-10 h (Table 1). After the completion of the reaction, the mixture was diluted with water (5 mL), extracted with EtOAc (3 × 5 mL). The combined organic layer was washed with saturated aqueous sodium bicarbonate solution (3 mL), dried over anhydrous Na2SO4 and concentrated in vacuo. The residue was purified by column chromatography (silica gel, hexane-EtOAc) to afford pure gem-dihydroperoxides 2 or hydroxyl-hydroperoxides 4 (Table 1). The products were characterized on the basis of their physical properties and spectral (1H-, 13C-NMR and MS) analyses and compared with literature data [32,33,37,40,41]. The spectral (1H-, 13C-NMR and MS) data of some representative products are given below.
Undecane-2,2-dihydroperoxide (2a) [32]. 1H-NMR (200 MHz, CDCl3): δ 9.51 (br s, 2H), 1.76–1.60 (m, 2H), 1.38 (s, 3H), 1.32–1.19 (br s,14H), 0.82 (t, J = 7 Hz, 3H); 13C-NMR (50 MHz, CDCl3): 112.3, 33.4, 32.0, 29.4, 29.1, 28.4, 23.6, 22.5, 17.6, 13.8, 13.5; FABMS: m/z 243 [M+Na]+.
4-Methylpentane-2,2-dihydroperoxide (2d) [32]. 1H-NMR (200 MHz, CDCl3): δ 9.54 (br s, 2H), 1.80 (m, 1H), 1.62 (d, J = 7 Hz, 2H), 1.42 (s, 3H), 0.98 (d, J = 7 Hz, 6H); FABMS: m/z 173 [M+Na]+.
Cyclohexane-1,1-dihydroperoxide (2e) [37]. 1H-NMR (200 MHz, CDCl3): δ 9.70 (br s, 2H), 1.93–1.70 (m, 4H), 1.67–1.39 (m, 6H); FABMS: m/z 171 [M+Na]+.
Methy-phenyl-1,1-dihydroperoxide (2h) [33]. 1H-NMR (200 MHz, CDCl3): δ 9.16 (br s, 2H), 7.50–7.43 (m, 2H), 7.38–7.26 (m, 3H), 1.69 (s, 3H); FABMS: m/z 193 [M+Na]+.
Phenylmethylene-1,1-dihydroperoxide (2l) [33]. 1H-NMR (200 MHz, CDCl3): δ 9.57 (br s, 2H), 7.42–7.28 (m, 5H), 6.24 (s, 1H); FABMS: m/z 179 [M+Na]+.
(4-Methylphenyl)methylene-1,1-dihydroperoxide (2m) [41]. 1H-NMR (200 MHz, CDCl3): δ 9.71 (br s, 2H), 7.30 (d, J = 8 Hz, 2H), 7.15 (d, J = 8 Hz, 2H), 6.28 (s,1H), 2.32 (s, 3H); 13C-NMR (50 MHz, CDCl3): 139.5, 129.4, 129.0, 126.7, 109.8, 21.1; FABMS: m/z 193 [M+Na]+.
(4-Chlorophenyl)methylene-1,1-dihydroperoxide (2n) [32]. 1H-NMR (200 MHz, CDCl3): δ 9.94 (br s, 2H), 7.85–7.34 (m, 4H), 6.26 (s, 1H); 13C-NMR (50 MHz, CDCl3): 139.6, 129.4, 129.0, 126.8, 10.02; FABMS: m/z 213 [M+Na]+.
(4-Cyanophenyl)methylene-1,1-dihydroperoxide (2p). White solid; m.p. 107–110 ºC; IR (KBr): 3,414, 2,916, 2,235, 1,611, 1,405, 1,333, 1,243, 1,199, 1,122, 1,083, 977, 824 cm-1; 1H-NMR (200 MHz, CDCl3): δ 10.08 (s, 2H), 8.04–7.78 (m, 4H), 7.24 (s, 1H); 13C-NMR (50 MHz, CDCl3): δ 139.3, 129.4, 128.0, 126.1, 117.0, 112.1; FABMS: m/z 204 [M+Na]+; Anal. Calcd for C8H7NO4: C, 53.04; H, 3.86; N, 7.73. Found: C, 53.15; H, 3.98; N, 7.78.
Octane-1,1-hydroxyhydroperoxide (4q) [42]. Colorless oil; IR (KBr): 3,374, 3,028, 2,931, 2,863, 1,496, 1,454, 1,357, 1,242, 1,078, 1,030, 924, 748, 699 cm-1; 1H-NMR (200 MHz, CDCl3): δ 8.20 (br s, 1H), 7.00 (br s, 1H), 4.90 (t, J = 7 Hz, 1H), 2.10–0.70 (m, 15H); 13C-NMR (50 MHz, CDCl3): δ 101.2, 32.6, 30.0, 28.5, 24.0, 20.1, 14.0; FABMS: m/z 185 [M+Na]+.
3-Phenylpropane-1,1-hydroxyhydroperoxide (4r) [42]. Colorless oil; IR (KBr): 3384, 3062, 3027, 2902, 2861, 1496, 1457, 1376, 1242, 1079, 1031, 923, 747, 700 cm-1; 1H NMR (200 MHz, CDCl3): δ 9.78 (br s, 1H), 8.65 (br s, 1H), 7.60–7.00 (m, 5H), 5.10 (t, J = 7 Hz, 1H), 2.60 (t, J = 8 Hz, 2H), 2.15–1.60 (m, 2H); 13C NMR (50 MHz, CDCl3): δ 141.5, 127.5, 125.0, 100.0, 32.2, 28.5; FABMS: m/z 191 [M+Na]+.
4. Conclusions
In summary, a new efficient homogeneous catalyst SrCl2·6H2O has been shown to promote the synthesis of gem-dihydroperoxides from aliphatic and aromatic ketones and aldehydes using aqueous H2O2 (30%) in acetonitrile at room temperature. The attractive features of this new approach are the readily available and non-expensive catalyst, high yields of the products, mild reaction conditions and the operational simplicity of the procedure.
Acknowledgements
The authors are thankful to Bu-Ali Sina University research council for the financial support.
- Sample Availability: Samples of the compounds are available from the authors.
References
- Zmitek, K.; Zupan, M.; Iskra, J. Synthetic strategies for a biologically important class of gem-dihydroperoxide and perketal derivatives. Org. Biomol. Chem. 2007, 5, 3895–3908. [Google Scholar]
- Iskra, J.; Bonnet-Delpon, D.; Begue, J.P. One-pot synthesis of non-symmetric tetraoxanes with the H2O2/MTO/fluorous alcohol system. Tetrahedron Lett. 2003, 44, 6309–6312. [Google Scholar]
- Terent’ev, A.O.; Kutkin, A.V.; Starikova, Z.A.; Antipia, M.Y.; Ogibin, Y.N.; Nikishina, G.I. New preparation of 1,2,4,5-tetraoxanes. Synthesis 2004, 2356–2366. [Google Scholar]
- Ito, T.; Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K.J. Synthesis of novel macrocyclic peroxides by bis(sym-collidine)iodine (I) hexafluorophosphate-mediated cyclization of unsaturated hydroperoxides and unsaturated alcohol. Tetrahedron 2003, 59, 525–536. [Google Scholar] [CrossRef]
- Zmitek, K.; Stavber, S.; Zupan, M.; Bonnet-Delpon, D.; Iskra, J. Fluorinated alcohol directed formation of dispiro-1,2,4,5-tetraoxanes by hydrogen peroxide under acid conditions. Tetrahedron 2006, 62, 1479–1484. [Google Scholar] [CrossRef]
- Dong, Y.X.; Vennerstrom, J.L. Dispiro-1,2,4,5-tetraoxanes via ozonolysis of cycloalkanone O-methyl oximes: a comparison with the peroxidation of cycloalkanones in acetonitrile sulfuric acid media. J. Org. Chem. 1998, 63, 8582–8585. [Google Scholar] [CrossRef]
- Zmitek, K.; Stavber, S.; Zupan, M.; Bonnet-Delpon, D.; Charneau, S.; Grellier, P.; Iskra, J. Synthesis and antimalarial activities of novel 3,3,6,6-tetraalkyl-1,2,4,5-tetraoxanes. J. Bioorg. Med. Chem. 2006, 14, 7790–7795. [Google Scholar]
- Opsenica, D.; Pocsfalvi, G.; Juranic, Z.; Tinant, B.; Declercq, J.P.; Kyle, D.E.; Milhous, W.K.; Solaja, B.A. Cholic acid derivatives as 1,2,4,5-tetraoxane carriers: structure, antimalarial and antiproliferative activity. J. Med. Chem. 2000, 43, 3274–3282. [Google Scholar]
- Dong, Y. Synthesis and antimalarial activity of 1,2,4,5-tetraoxanes. Mini-Rev. Med. Chem. 2002, 2, 113–123. [Google Scholar]
- Terent’ev, A.O.; Platonov, M.M.; Tursina, A.I.; Chemyshev, V.V.; Nikishin, G.I. Synthesis of cyclic peroxides containing the Si-gem-bisperoxide fragment 1,2,4,5,7,8-hexaoxa-3-silonanes as a new class of peroxides. J. Org. Chem. 2008, 73, 3169–3174. [Google Scholar] [CrossRef]
- Ghorai, P.; Dussault, P.H.; Hu, C. Synthesis of spiro-bisperoxyketals. Org. Lett. 2008, 10, 2401–2404. [Google Scholar] [CrossRef]
- Kim, H.S.; Nagai, Y.; Ono, K.; Begum, K.; Wataya, Y.; Hamada, Y.; Tsuchiya, K.; Masuyama, A.; Nojima, M.; McCullough, K.J. Synthesis and antimalarial activity of novel medium-sized 1,2,4,5-tetraoxacycloalkanes. J. Med. Chem. 2001, 44, 2357–2361. [Google Scholar]
- Jakka, K.; Liu, J.; Zhao, C.G. Facile epoxidation of α,β-unsaturated ketones with cyclohexylidenebishydroperoxide. Tetrahedron Lett. 2007, 48, 1395–1398. [Google Scholar]
- Saneyyoshi, H.; Miyata, K.; Seio, K.; Sekine, M. 1,1-Dihydroperoxycyclododecane as a new, crystalline non-hygroscopic oxidizer for the chemical synthesis of oligodeoxyribonucleotides. Tetrahedron Lett. 2006, 47, 8945–8947. [Google Scholar]
- Jon Paul Selvam, J.; Suresh, V.; Rajesh, K.; Chanti Babu, D.; Suryakiran, N.; Venkateswarlu, Y. A novel rapid sulfoxidation of sulfides with cyclohexylidenebishydroperoxide. Tetrahedron Lett. 2008, 49, 3463–3465. [Google Scholar] [CrossRef]
- Bunge, A.; Hamann, H.J.; McCalmont, E.; Leibscher, J. Enantioselective epoxidation of 2-substituted 1,4-naphthoquinones using gem-dihydroperoxides. Tetrahedron Lett. 2009, 50, 4629–4632. [Google Scholar] [CrossRef]
- Adam, W. Peroxide Chemistry: Mechanistic And Preparative Aspects Of Oxygen Transfer; Wiley-VCH: Weinheim, Germany, 2000. [Google Scholar]
- Ando, W. Organic Peroxides; John Wiley & Sons: Chichester, UK, 1992. [Google Scholar]
- Tang, Y.Q.; Dong, Y.X.; Vennerstrom, J.L. Synthetic peroxides as antimalarials. Med. Res. Rev. 2004, 24, 425–448. [Google Scholar] [CrossRef]
- Masuyama, A.; Wu, J.M.; Nojima, M.; Kim, H.S.; Wataya, Y. 1,2,4,5-Tetraoxacycloalkanes: synthesis and antimalarial activity. Mini-Rev. Med. Chem. 2005, 5, 1035–1043. [Google Scholar]
- Borstnik, K.; Mpaik, I.H.; Shapiro, T.A.; Posner, G.H. Antimalarial chemotherapeutic peroxides: artemisinin, yingzhaosu A and related compounds. Int. J. Parasitol. 2002, 32, 1661–1667. [Google Scholar] [CrossRef]
- Wiesner, J.; Ortmann, R.; Jomaa, H.; Schlitzer, M. New antimalarial drugs. Angew. Chem. Int. Ed. 2003, 42, 5274–5293. [Google Scholar] [CrossRef]
- Hamada, Y.; Tokuhara, H.; Masuyama, A.; Nojima, M.; Kim, H.S.; Ono, K.; Ogura, N.; Wataya, Y. Synthesis and notable antimalarial activity of acyclic peroxides, L-(alkyldioxy)-L-(methyldioxy)cyclododecanes. J. Med. Chem. 2002, 45, 1374–1378. [Google Scholar] [CrossRef]
- Kim, H.S.; Tsuchiya, K.; Shibata, Y.; Wataya, Y.; Ushigoe, Y.; Masuyama, A.; Nojima, M.; McCullough, K.J. Synthetic methods for unsymmetrically-substituted 1,2,4,5-tetroxanes and of 1,2,4,5,7-pentoxocanes. J. Chem. Soc., Perkin Trans. 1999, 1, 1867–1870. [Google Scholar]
- Tsuchiya, K.; Hamada, Y.; Masuyama, A.; Nojima, M. Synthesis, crystal structure and anti-malarialactivity of novel spiro-1,2,4,5-tetraoxacycloalkanes. Tetrahedron Lett. 1999, 40, 4077–4080. [Google Scholar]
- Dong, Y.; Matile, H.; Chollet, J.; Kaminsky, R.; Wood, J.K.; Vennerstrom, J.L. Synthesis and antimalarial activity of eleven dispiro-1,2,4,5-tetraoxane analogs of WR 148999. 7,8,15,16-tetraoxadispiro[5.2.5.2]hexadecanes substituted at the 1 and 10 positions with unsaturated and polar functional groups. J. Med. Chem. 1999, 42, 1477–1480. [Google Scholar]
- Ledaal, T.; Solbjor, T. 1,1-Dihydroperoxycyclododecane. Acta Chem. Scand. 1967, 21, 1658–1659. [Google Scholar] [CrossRef]
- Terent’ev, A.O.; Platonov, M.M.; Ogibin, Y.N.; Nikishin, G.I. Convenient synthesis of geminal bishydroperoxides by the reaction of ketones with hydrogen peroxide. Synth. Cmmun. 2007, 37, 1281–1287. [Google Scholar] [CrossRef]
- Jefford, C.W.; Li, W.; Jaber, A.; Boukouvalas, J. A new method for the synthesis of gem-dihydroperoxides. Synth. Cmmun. 1990, 20, 2589–2596. [Google Scholar] [CrossRef]
- Terent’ev, A.O.; Kutkin, A.V.; Platonov, M.M.; Ogibin, Y.N.; Nikishin, G.I. A new method for the synthesis of bishydroperoxides based on a reaction of ketals with hydrogen peroxide catalyzed by boron trifluoride complexe. Tetrahedron Lett. 2003, 44, 7359–7363. [Google Scholar] [CrossRef]
- Bunge, A.; Hamann, H.J.; Liebscher, J. A simple, efficient and versatile synthesis of primary gem-dihydroperoxides from aldehydes and hydrogen peroxide. Tetrahedron Lett. 2009, 50, 524–526. [Google Scholar] [CrossRef]
- Das, B.; Krishnaiah, M.; Veeranjaneyulu, B.; Ravikanth, B. A simple and efficient synthesis of gem-dihydroperoxides from ketones using aqueous hydrogen peroxide and catalytic ceric ammonium nitrate. Tetrahedron Lett. 2007, 48, 6286–6289. [Google Scholar] [CrossRef]
- Zmitek, K.; Zupan, M.; Stavber, S.; Iskra, J. Iodine as a catalyst for efficient conversion of ketones to gem-dihydroperoxides by aqueous hydrogen peroxide. Org. Lett. 2006, 8, 2491–2494. [Google Scholar] [CrossRef]
- Ghorai, P.; Dussault, P.H. Mild and efficient Re(VII)-catalyzed synthesis of 1,1-dihydroperoxides. Org. Lett. 2008, 10, 4577–4579. [Google Scholar] [CrossRef]
- Ghorai, P.; Dussault, P.H. Broadly applicable synthesis of 1,2,4,5-tetraoxanes. Org. Lett. 2009, 11, 213–216. [Google Scholar] [CrossRef]
- Ledaal, T.; Solbjor, T. 1,1-Dihydroperoxycyclododecane. Acta Chem. Scand. 1967, 21, 1658–1659. [Google Scholar] [CrossRef]
- Das, B.; Veeranjaneyulu, B.; Krishnaiah, M.; Balasubramanyam, P. Synthesis of gem-dihydroperoxides from ketones using silica-supported sodium hydrogen sulfate as a heterogeneous catalyst. J. Mol. Catal. A 2008, 284, 116–119. [Google Scholar] [CrossRef]
- Ramirez, A.; Woerpel, K.A. Synthesis of 1,2-dioxolanes by annulation reactions of peroxzcarbenium ions with alkenes. Org. Lett. 2005, 7, 4617–4620. [Google Scholar] [CrossRef]
- Terent’ev, A.O.; Kutkin, A.V.; Troizky, N.A.; Ogibin, Y.N.; Nikishin, G.I. Synthesis of geminal bisperoxides by acid-catalyzed reaction of acetals and enol ethers with tert-butyl hydroperoxide. Synthesis 2005, 2215–2219. [Google Scholar]
- Rieche, A. Über oxyalkyl-hydroperoxyde. (VII. Mitteil. über alkylperoxyde). Chem. Ber. 1931, 64, 2328–2335. [Google Scholar]
- Zmitek, K.; Zupan, M.; Stavber, S.; Iskra, J. The effect of iodine on the peroxidation of carbonyl compounds. J. Org. Chem. 2007, 72, 6534–6540. [Google Scholar] [CrossRef]
- McClelland, R.A.; Coe, M. Structure-reactivity effects in the hydration of benzaldehydes. J. Am. Chem. Soc. 1983, 105, 2718–2725. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
