Clusters of Hydroxyl-Functionalized Cations Stabilized by Cooperative Hydrogen Bonds: The Role of Polarizability and Alkyl Chain Length
Abstract
1. Introduction
2. Density Functional Theory (DFT) Calculations of Cationic and Neutral Clusters
3. NMR Proton Chemical Shifts and Deuteron Quadrupole Coupling Constants as H-Bond Sensors
4. Influence of Cation Polarizability on Energies, Geometries, and Spectroscopic Properties
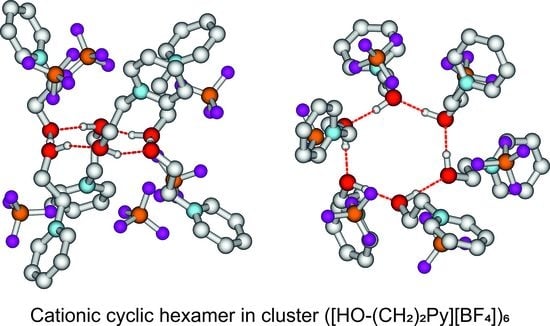
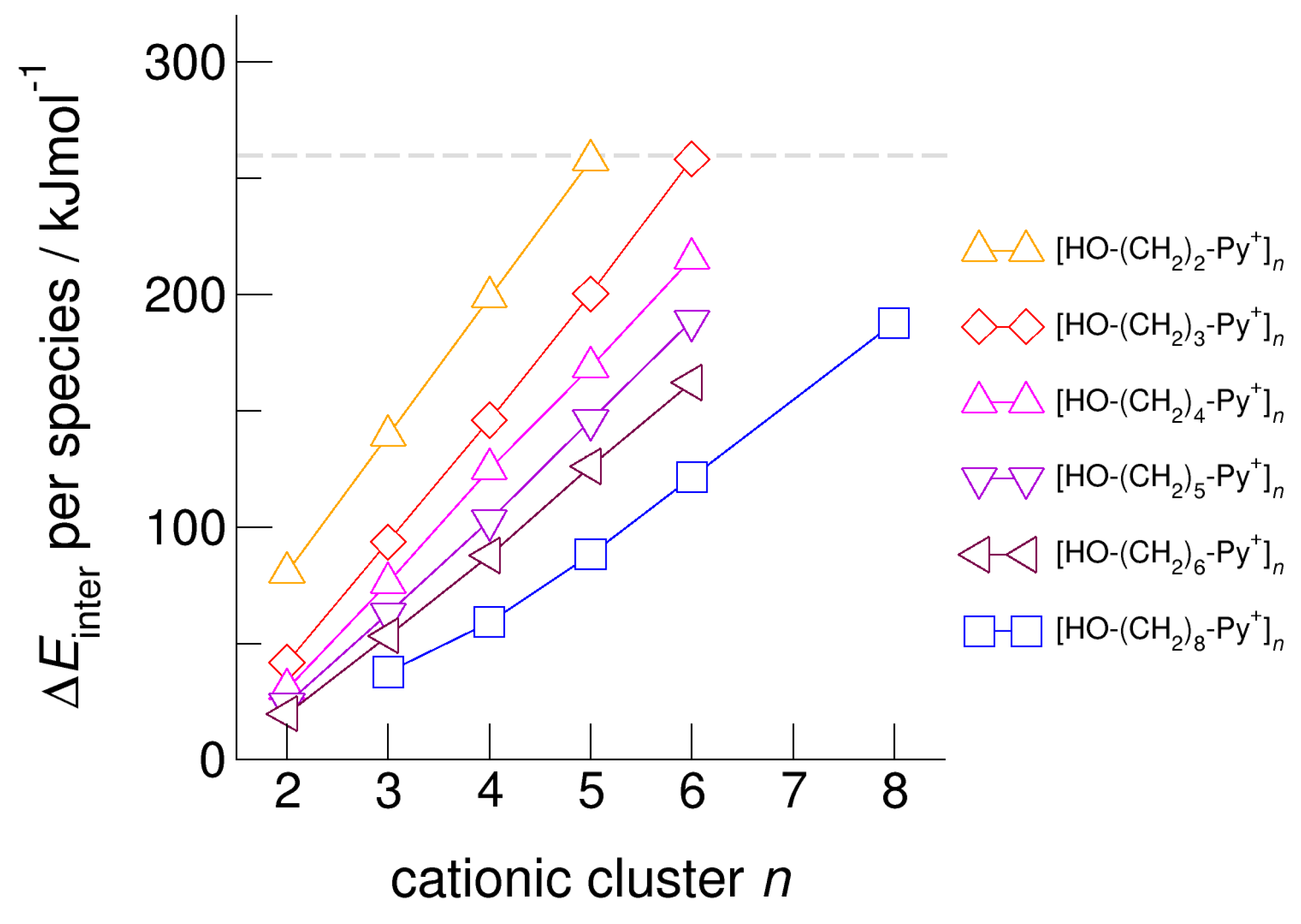
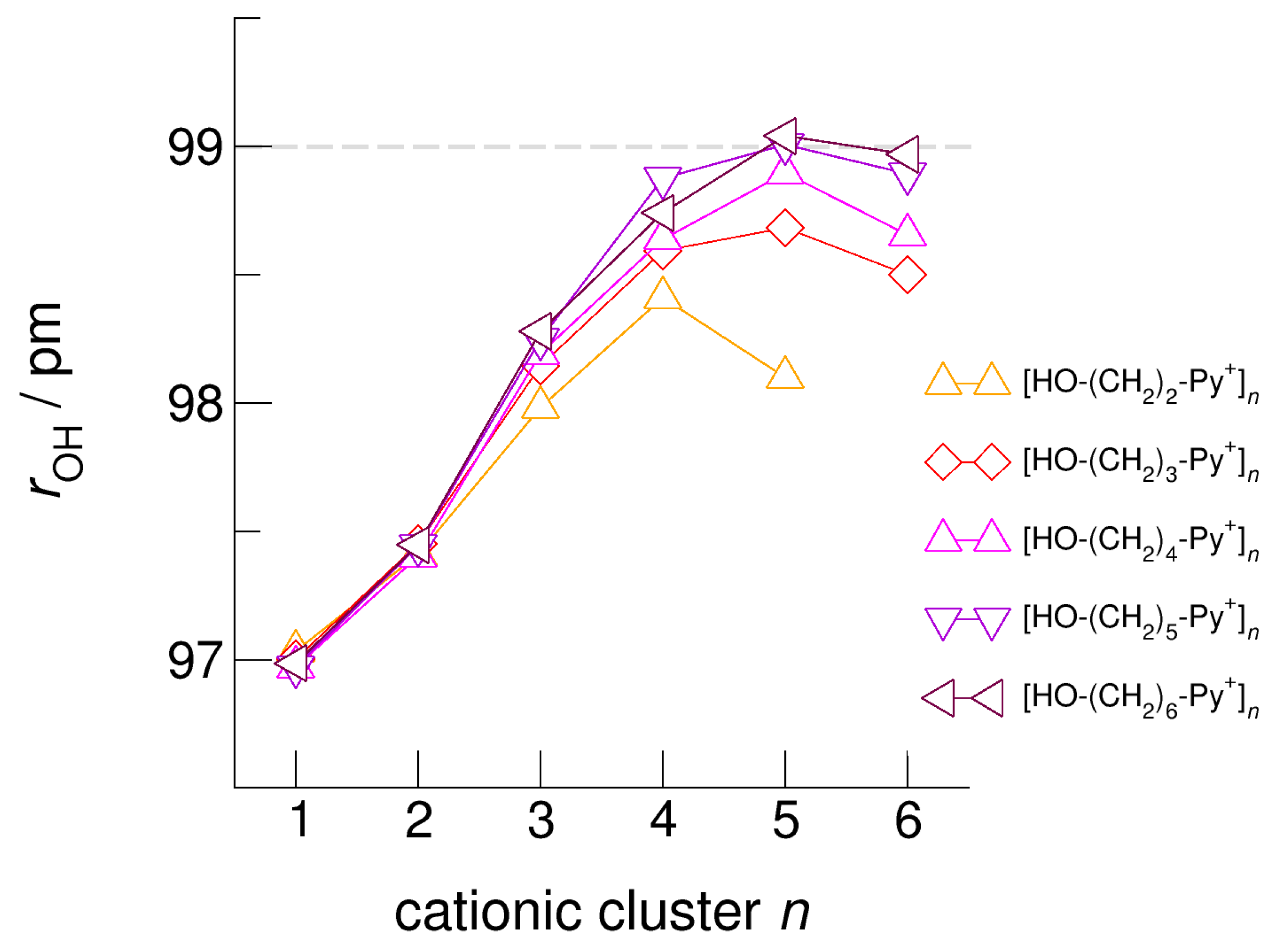
5. Role of Hydroxyl Alkyl Chain Length on Energies, Geometries, and Spectroscopic Properties
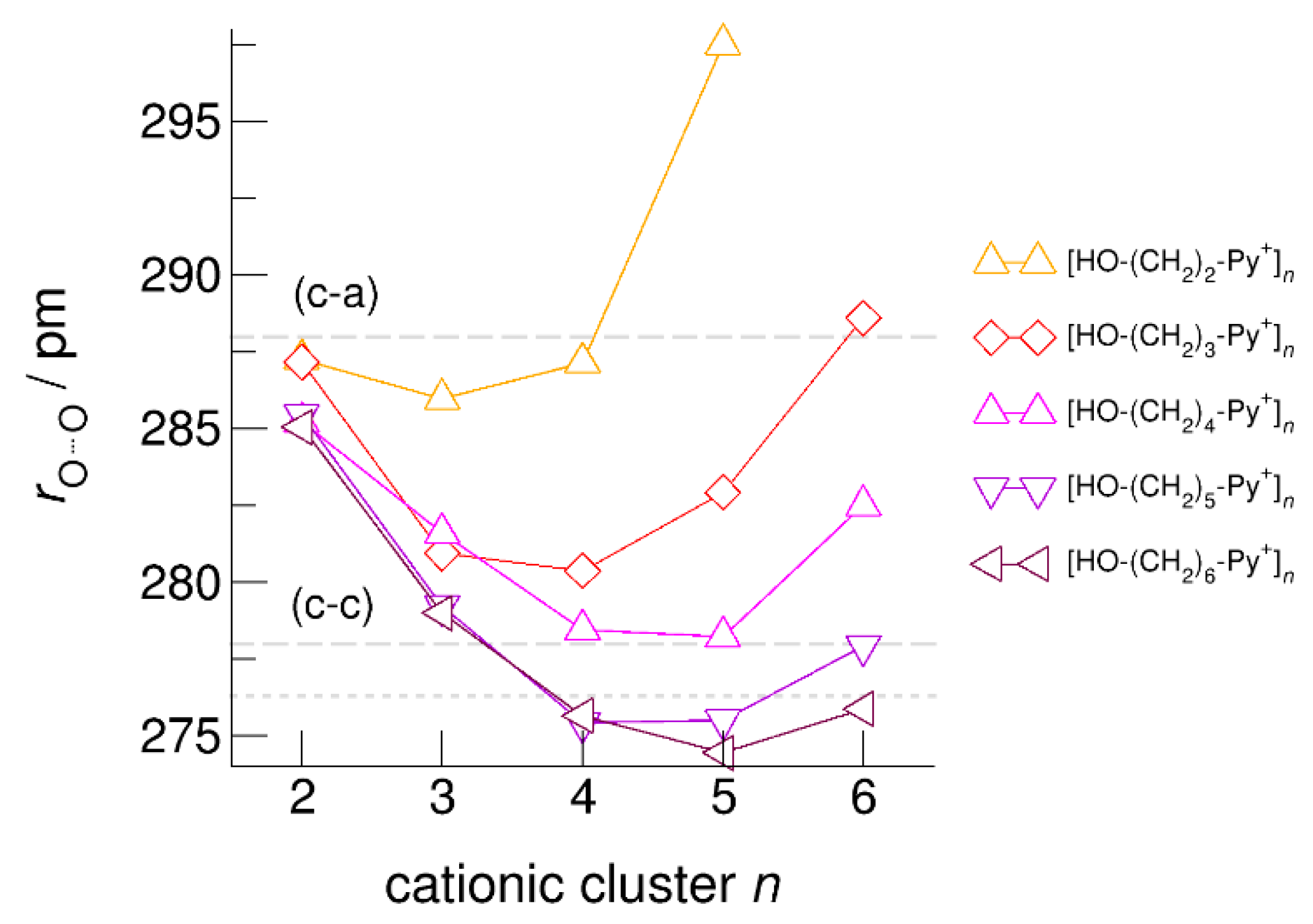
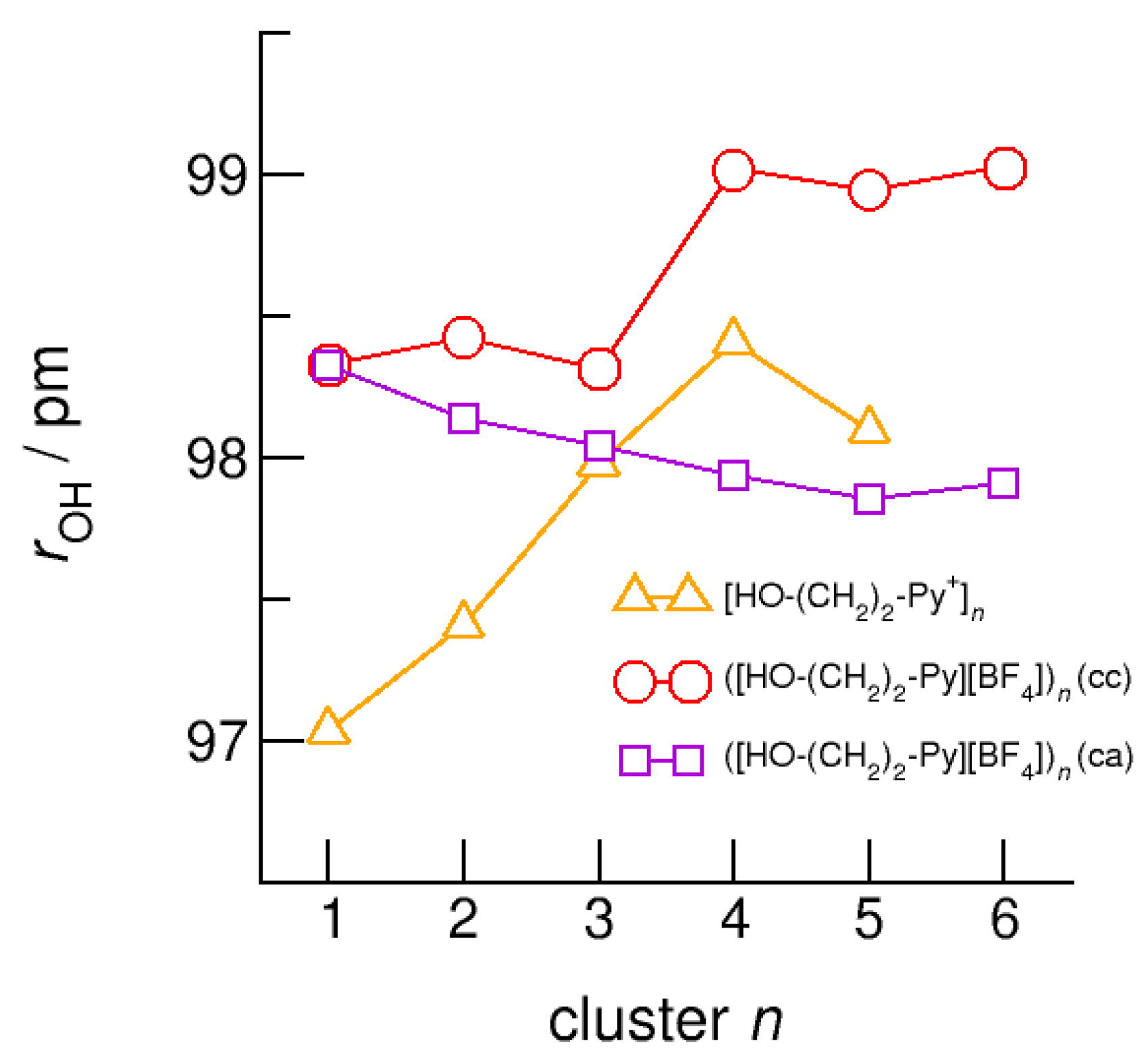
6. Neural Clusters: Competition between (c–a) and (c–c) Hydrogen Bonds
7. Universal Linear Relation between Δ(χD) and δ1H Allows Prediction of Properties
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smid, J. Structure of Ion Pair Solvation Complexes. Angew. Chem. Int. Ed. 1972, 11, 112–127. [Google Scholar] [CrossRef]
- Reichhardt, C. Solvents and Solvent Effects in Organic Chemistry; Wiley-VCH: Weinheim, Germany, 1990. [Google Scholar]
- Marcus, Y.; Hefter, G. Ion Pairing. Chem. Rev. 2006, 106, 4585–4621. [Google Scholar] [CrossRef] [PubMed]
- Holz, M.; Patil, K.J. Cation-Cation Association of Tetramethylammonium Ions in Aqueous Mixed Electrolyte Solutions. Ber. Bunsen-Ges. 1991, 95, 107–113. [Google Scholar] [CrossRef]
- Shih, O.; England, A.H.; Dallinger, G.C.; Smith, J.W.; Duffey, K.C.; Cohen, R.C.; Prendergast, D.; Saykally, R.J. Cation-cation contact pairing in water: Guanidinium. J. Chem. Phys. 2013, 139, 035104. [Google Scholar] [CrossRef] [PubMed]
- Benrraou, M.; Bales, B.L.; Zana, R. Effect of the Nature of the Counterion on the Properties of Anionic Surfactants. 1. Cmc, Ionization Degree at the Cmc and Aggregation Number of Micelles of Sodium, Cesium, Tetramethylammonium, Tetraethylammonium, Tetrapropylammonium, and Tetrabutylammonium Dodecyl Sulfates. J. Phys. Chem. B 2003, 107, 13432–13440. [Google Scholar]
- Caplan, M.R.; Moore, P.N.; Zhang, S.; Kamm, R.D.; Lauffenburger, D.A. Self-assembly of a β-sheet protein governed by relief of electrostatic repulsion relative to van der Waals attraction. Biomacromolecules 2000, 1, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Kuron, M.; Arnold, A. Role of geometrical shape in like-charge attraction of DNA. Eur. Phys. J. E 2015, 38, 20. [Google Scholar] [CrossRef] [PubMed]
- Fumino, K.; Wulf, A.; Ludwig, R. The Cation–Anion Interaction in Ionic Liquids Probed by Far-Infrared Spectroscopy. Angew. Chem. Int. Ed. Engl. 2008, 47, 3830–3834. [Google Scholar] [CrossRef]
- Fumino, K.; Wulf, A.; Ludwig, R. Strong, localized, and directional hydrogen bonds fluidize ionic liquids. Angew. Chem. Int. Ed. Engl. 2008, 47, 8731–8734. [Google Scholar] [CrossRef]
- Lehmann, S.B.C.; Roatsch, M.; Schöppke, M.; Kirchner, B. On the physical origin of the cation-anion intermediate bond in ionic liquids Part I Placing a (weak) hydrogen bond between two charges. Phys. Chem. Chem. Phys. 2010, 12, 7473–7486. [Google Scholar] [CrossRef]
- Pensado, A.S.; Costa Gomes, M.F.; Canongia Lopes, J.N.; Malfreyt, P.; Pádua, A.A.H. Effect of alkyl chain length and hydroxyl group functionalization on the surface properties of imidazolium ionic liquids. Phys. Chem. Chem. Phys. 2011, 13, 13518–13526. [Google Scholar] [CrossRef] [PubMed]
- Fumino, K.; Ludwig, R. Analyzing the interaction energies between cation and anion in ionic liquids: The subtle balance between Coulomb forces and hydrogen bonding. J. Mol. Liq. 2014, 192, 94–102. [Google Scholar] [CrossRef]
- Fumino, K.; Reimann, S.; Ludwig, R. Probing molecular interaction in ionic liquids by low frequency spectroscopy: Coulomb energy, hydrogen bonding and dispersion forces. Phys. Chem. Chem. Phys. 2014, 40, 21903–21929. [Google Scholar] [CrossRef] [PubMed]
- Hunt, P.A.; Ashworth, C.R.; Matthews, R.P. Hydrogen bonding in ionic liquids. Chem. Soc. Rev. 2015, 44, 1257–1288. [Google Scholar] [CrossRef] [PubMed]
- Ashwoth, C.R.; Matthews, R.P.; Welton, T.; Hunt, P.A. Doubly ionic hydrogen bond interactions within the choline chloride–urea deep eutectic solvent. Phys. Chem. Chem. Phys. 2016, 18, 18145–18160. [Google Scholar] [CrossRef] [PubMed]
- Fumino, K.; Peppel, T.; Geppert-Rybczynska, M.; Zaitsau, D.H.; Lehmann, J.K.; Verevkin, S.P.; Köckerling, M.; Ludwig, R. The influence of hydrogen bonding on the physical properties of ionic liquids. Phys. Chem. Chem. Phys. 2011, 13, 14064–14075. [Google Scholar] [CrossRef]
- Katsyuba, S.A.; Vener, M.V.; Zvereva, E.E.; Fei, Z.; Scopelliti, R.; Laurenczy, G.; Yan, N.; Paunescu, E.; Dyson, P.J. How Strong Is Hydrogen Bonding in Ionic Liquids? Combined X-ray Crystallographic, Infrared/Raman Spectroscopic, and Density Functional Theory Study. J. Phys. Chem. B 2013, 117, 9094–9105. [Google Scholar] [CrossRef] [PubMed]
- Fakhraee, M.; Zandkarimi, B.; Salari, H.; Gholami, M.R. Hydroxyl-Functionalized 1-(2-Hydroxyethyl)-3-methyl Imidazolium Ionic Liquids: Thermodynamic and Structural Properties using Molecular Dynamics Simulations and ab Initio Calculations. J. Phys. Chem. B 2014, 118, 14410–14428. [Google Scholar] [CrossRef] [PubMed]
- Knorr, A.; Fumino, K.; Bonsa, A.-M.; Ludwig, R. Spectroscopic evidence of ‘jumping and pecking’ of cholinium and H-bond enhanced cation–cation interaction in ionic liquids. Phys. Chem. Chem. Phys. 2015, 17, 30978–30982. [Google Scholar] [CrossRef] [PubMed]
- Knorr, A.; Ludwig, R. Cation-cation clusters in ionic liquids: Cooperative hydrogen bonding overcomes like-charge repulsion. Sci. Rep. 2015, 5, 17505. [Google Scholar] [CrossRef] [PubMed]
- Knorr, A.; Stange, P.; Fumino, K.; Weinhold, F.; Ludwig, R. Spectroscopic Evidence for Clusters of Like-Charged Ions in Ionic Liquids Stabilized by Cooperative Hydrogen Bonding. Chem. Phys. Chem. 2016, 17, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Strate, A.; Niemann, T.; Stange, P.; Michalik, D.; Ludwig, R. When Like Charged Ions Attract in Ionic Liquids: Controlling the Formation of Cationic Clusters by the Interaction Strength of the Counterions. Angew. Chem. Int. Ed. Engl. 2017, 56, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Niemann, T.; Neumann, J.; Stange, P.; Gärtner, S.; Youngs, T.G.A.; Paschek, D.; Atkin, R.; Ludwig, R. The Double-Faced Nature of Hydrogen Bonding in Hydroxy-Functionalized Ionic Liquids Shown by Neutron Diffraction and Molecular Dynamics Simulations. Angew. Chem. Int. Ed. Engl. 2019, 58, 12887–12892. [Google Scholar] [CrossRef] [PubMed]
- Khudozhitkov, A.E.; Overbeck, V.; Stange, P.; Paschek, D.; Stepanov, A.G.; Kolokolov, D.I.; Ludwig, R. Hydrogen Bonding Between Ions of Like Charge in Ionic Liquids Characterized by NMR Deuteron Quadrupole Coupling Constants—Comparison with Salt Bridges and Molecular Systems. Angew. Chem. Int. Ed. 2019, 58, 17863–17871. [Google Scholar] [CrossRef]
- Niemann, T.; Zaitsau, D.H.; Strate, A.; Stange, P.; Ludwig, R. Controlling “like–likes–like” charge attraction in hydroxy-functionalized ionic liquids by polarizability of the cations, interaction strength of the anions and varying alkyl chain length. Phys. Chem. Chem. Phys. 2020, 22, 2763–2774. [Google Scholar] [CrossRef] [PubMed]
- Khudozhitkov, A.E.; Niemann, T.; Stange, P.; Donoshita, M.; Stepanov, A.G.; Kitagawa, H.; Kolokolov, D.I.; Ludwig, R. Freezing the Motion in Hydroxy-Functionalized Ionic Liquids–Temperature Dependent NMR Deuteron Quadrupole Coupling Constants for Two Types of Hydrogen Bonds Far below the Glass Transition. J. Phys. Chem. Lett. 2020, 11, 6000–6006. [Google Scholar] [CrossRef] [PubMed]
- Menges, F.S.; Zeng, H.J.; Kelleher, P.J.; Gorlova, O.; Johnson, M.A.; Niemann, T.; Strate, A.; Ludwig, R. Structural Motifs in Cold Ternary Ion Complexes of Hydroxyl-Functionalized Ionic Liquids: Isolating the Role of Cation–Cation Interactions. J. Phys. Chem. Lett. 2018, 9, 2979–2984. [Google Scholar] [CrossRef]
- Niemann, T.; Strate, A.; Ludwig, R.; Zeng, H.J.; Menges, F.S.; Johnson, M.A. Spectroscopic Evidence for an Attractive Cation–Cation Interaction in Hydroxy-Functionalized Ionic Liquids: A Hydrogen-Bonded Chain-like Trimer. Angew. Chem. Int. Ed. Engl. 2018, 57, 15364–15368. [Google Scholar] [CrossRef]
- Niemann, T.; Strate, A.; Ludwig, R.; Zeng, H.J.; Menges, F.; Johnson, M.A. Cooperatively enhanced hydrogen bonds in ionic liquids: Closing the loop with molecular mimics of hydroxy-functionalized cations. Phys. Chem. Chem. Phys. 2019, 21, 18092–18098. [Google Scholar] [CrossRef]
- Zeng, H.; Menges, F.; Johnson, M.; Niemann, T.; Strate, A.; Ludwig, R. Chain Length Dependence of Hydrogen Bond Linkages between Cationic Constituents in Hydroxy-Functionalized Ionic Liquids: Tracking Bulk Behavior to the Molecular Level with Cold Cluster Ion Spectroscopy. J. Phys. Chem. Lett. 2020, 14, 683–688. [Google Scholar] [CrossRef]
- Niemann, T.; Stange, P.; Strate, A.; Ludwig, R. When hydrogen bonding overcomes Coulomb repulsion: From kinetic to thermodynamic stability of cationic dimers. Phys. Chem. Chem. Phys. 2019, 21, 8215–8220. [Google Scholar] [CrossRef] [PubMed]
- Niemann, T.; Stange, P.; Strate, A.; Ludwig, R. Like-likes-Like: Cooperative Hydrogen Bonding Overcomes Coulomb Repulsion in Cationic Clusters with Net Charges up to Q=+6e. Chem. Phys. Chem. 2018, 19, 1691–1695. [Google Scholar] [CrossRef] [PubMed]
- Nauta, K.; Miller, R.E. Formation of Cyclic Water Hexamer in Liquid Helium: The Smallest Piece of Ice. Science 2000, 287, 293–295. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Cruzan, J.D.; Saykally, R.J. Water Clusters. Science 1996, 271, 929–933. [Google Scholar] [CrossRef]
- Buck, U.; Siebers, J.-G.; Wheatley, R.J. Structure and vibrational spectra of methanol clusters from a new potential model. J. Chem. Phys. 1998, 108, 20–32. [Google Scholar] [CrossRef]
- Sulaiman, M.I.; Yang, S.; Ellis, A. Infrared Spectroscopy of Methanol and Methanol/Water Clusters in Helium Nanodroplets: The OH Stretching Region. J. Phys. Chem. A 2017, 121, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Malloum, A.; Fifen, J.J. Conradie, J. Solvation energies of the proton in methanol revisited and temperature effects. J. Phys. Chem. Chem. Phys. 2018, 20, 29184–29206. [Google Scholar] [CrossRef] [PubMed]
- Weinhold, F.; Klein, R.A. Anti-Electrostatic Hydrogen Bonds. Angew. Chem. Int. Ed. 2014, 53, 11214–11217, Angew. Chem.2014, 126, 11396–11399. [Google Scholar] [CrossRef]
- Weinhold, F. Polyion Covalency: Exotic Species from the Unexplored World of Electrostatically Shielded Molecular Ion Chemistry. Angew. Chem. Int. Ed. 2017, 56, 14577–14581. [Google Scholar] [CrossRef] [PubMed]
- Weinhold, F. Theoretical Prediction of Robust Second-Row Oxyanion Clusters in the Metastable Domain of Antielectrostatic Hydrogen Bonding. Inorg. Chem. 2018, 57, 2035–2044. [Google Scholar] [CrossRef] [PubMed]
- Reed, A.E.; Weinhold, F. Natural bond orbital analysis of near-Hartree–Fock water dimer. J. Chem. Phys. 1983, 78, 4066–4073. [Google Scholar] [CrossRef]
- Weinhold, F.; Klein, R.A. What is a hydrogen bond? Mutually consistent theoretical and experimental criteria for characterizing H-bonding interactions. Mol. Phys. 2012, 110, 565–579. [Google Scholar] [CrossRef]
- Grabowski, S.J. What is the Covalency of the Hydrogen bonding. Chem. Rev. 2011, 111, 2597–2625. [Google Scholar] [CrossRef] [PubMed]
- Gaussian 09, Revision A.02 ed; Gaussian, Inc.: Wallingford, CT, USA, 2016.
- Ludwig, R. The effect of hydrogen bonding on the thermodynamic and spectroscopic properties of molecular clusters and liquids. Phys. Chem. Chem. Phys. 2002, 4, 5481–5487. [Google Scholar] [CrossRef]
- Murdoch, K.M.; Ferris, T.D.; Wright, J.C.; Farrar, T.C. Infrared spectroscopy of ethanol clusters in ethanol–hexane binary solutions. J. Chem. Phys. 2002, 116, 5717. [Google Scholar] [CrossRef]
- Ludwig, R. The Structure of Liquid Methanol. ChemPhysChem 2005, 6, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, S.; Moellmann, J.; Reckien, W.; Bredow, T.; Grimme, S. System-Dependent Dispersion Coefficients for the DFT-D3 Treatment of Adsorption Processes on Ionic Surfaces. Chem. Phys. Chem. 2011, 12, 3414–3420. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Jansen, A. Dispersion-Corrected Mean-Field Electronic Structure Methods. Chem. Rev. 2016, 116, 5105–5154. [Google Scholar] [CrossRef] [PubMed]
- Graton, J.; Besseau, F.; Brossard, A.-M.; Charpentier, E.; Deroche, A.; Le Questel, J.-Y. Hydrogen-Bond Acidity of OH Groups in Various Molecular Environments (Phenols, Alcohols, Steroid Derivatives, and Amino Acids Structures): Experimental Measurements and Density Functional Theory Calculations. J. Phys. Chem. A 2013, 117, 13184–13193. [Google Scholar] [CrossRef] [PubMed]
- Lomas, J.S. 1H NMR spectra of alcohols in hydrogen bonding solvents: DFT/GIAO calculations of chemical shifts. Magn. Reson. Chem. 2016, 54, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Kontogianni, V.G.; Charisiadis, P.; Primikyri, A.; Pappas, C.G.; Exarchou, V.; Tzakosa, A.G.; Gerothanassis, I.P. Hydrogen bonding probes of phenol –OH groups. Org. Biomol. Chem. 2013, 11, 1013–1025. [Google Scholar] [CrossRef] [PubMed]
- Huber, H. Deuterium quadrupole coupling constants. A theoretical investigation. J. Chem. Phys. 1985, 83, 4591–4598. [Google Scholar] [CrossRef]
- Eggenberger, R.; Gerber, S.; Huber, H.; Searles, D.; Welker, M. Ab initio calculation of the deuterium quadrupole coupling in liquid water. J. Chem. Phys. 1992, 97, 5898–5904. [Google Scholar] [CrossRef]
- Searles, D.J.; Huber, H. Accurate Determination of Nuclear Quadrupole Coupling Constants and other NMR Parameters in Liquids from the Combination of Molecular Dynamics Simulations and ab initio Calculations. In Encyclopedia of Nuclear Magnetic Resonance; Grant, D.M., Harris, R.K., Eds.; Wiley: New York, NY, USA, 2002; Volume 9. [Google Scholar]
- Cummins, P.L.; Bacskay, G.B.; Hush, N.S.; Halle, B.; Engström, S. The effect of intermolecular interactions on the 2H and 17O quadrupole coupling constants in ice and liquid water. J. Chem. Phys. 1985, 82, 2002–2013. [Google Scholar] [CrossRef]
- Philipp, J.K.; Fritsch, S.; Ludwig, R. Cyclic Octamer of Hydroxyl-functionalized Cations with Net Charge Q=+8e Kinetically Stabilized by a ‘Molecular Island’ of Cooperative Hydrogen Bonds. Chem. Phys. Chem. 2020. [Google Scholar] [CrossRef]
- Weinhold, F.; Glendening, E.D. Comment on “Natural Bond Orbitals and the Nature of the Hydrogen Bond”. J. Phys. Chem. A 2018, 122, 724–732. [Google Scholar] [CrossRef]
- Xantheas, S.S.; Dunning, T.H. Ab initio studies of cyclic water clusters (H2O)n, n=1–6. I. Optimal structures and vibrational spectra. J. Chem. Phys. 1993, 99, 8774–8792. [Google Scholar] [CrossRef]
- Tanaka, Y.; Ohtomo, N.; Arakawa, K. The Structure of Liquid Alcohols by Neutron Diffraction. I. Molecular Structure of Methyl Alcohol. Bull. Chem. Soc. Jpn. 1984, 57, 644–647. [Google Scholar] [CrossRef]
- Sarkar, S.; Joarder, R.N. Molecular clusters in liquid ethanol at room temperature. J. Chem. Phys. 1994, 100, 5118–5122. [Google Scholar] [CrossRef]
- Wendt, M.A.; Zeidler, M.D.; Farrar, T.C. The temperature dependence of the deuterium quadrupole coupling constant and the molecular rotational correlation time in liquid methanol. Mol. Phys. 1999, 97, 753–756. [Google Scholar] [CrossRef]
- O’Reilly, D.E.; Peterson, E.M. Self-Diffusion Coefficients and Rotational Correlation Times in Polar Liquids. II. J. Chem. Phys. 1971, 55, 2155. [Google Scholar] [CrossRef]
- Ludwig, R.; Gill, D.S.; Zeidler, M.D. Molecular Reorientation in Liquid Methanol. Z. Naturforsch. 1991, 46a, 89–94. [Google Scholar] [CrossRef]
- Ludwig, R.; Gill, D.S.; Zeidler, M.D. Molecular Reorientation of Methanol in Mixtures with Carbon Tetrachloride. Z. Naturforsch. Sect. A 1992, 47, 857–864. [Google Scholar]
- Strauch, M.; Bonsa, A.-M.; Golub, B.; Overbeck, V.; Michalik, D.; Paschek, D.; Ludwig, R. Deuteron quadrupole coupling constants and reorientational correlation times in protic ionic liquids. Phys. Chem. Chem. Phys. 2016, 18, 17788–17794. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |












Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Philipp, J.K.; Ludwig, R. Clusters of Hydroxyl-Functionalized Cations Stabilized by Cooperative Hydrogen Bonds: The Role of Polarizability and Alkyl Chain Length. Molecules 2020, 25, 4972. https://doi.org/10.3390/molecules25214972
Philipp JK, Ludwig R. Clusters of Hydroxyl-Functionalized Cations Stabilized by Cooperative Hydrogen Bonds: The Role of Polarizability and Alkyl Chain Length. Molecules. 2020; 25(21):4972. https://doi.org/10.3390/molecules25214972
Chicago/Turabian StylePhilipp, Jule K., and Ralf Ludwig. 2020. "Clusters of Hydroxyl-Functionalized Cations Stabilized by Cooperative Hydrogen Bonds: The Role of Polarizability and Alkyl Chain Length" Molecules 25, no. 21: 4972. https://doi.org/10.3390/molecules25214972
APA StylePhilipp, J. K., & Ludwig, R. (2020). Clusters of Hydroxyl-Functionalized Cations Stabilized by Cooperative Hydrogen Bonds: The Role of Polarizability and Alkyl Chain Length. Molecules, 25(21), 4972. https://doi.org/10.3390/molecules25214972