1. Introduction
Organic perovskite CH
3NH
3PbI
3 (MAPbI
3) is considered to be the most potential light-absorbing material for perovskite solar cells (PSCs) due to its high optical absorption characteristics and long diffusion length [
1]. Compared with silicon solar cells, and although they dominate the solar industry with efficiencies of over 20%, silicon solar cells remain relatively expensive to manufacture [
2]. In the industry, in order to ensure large-scale production and meet future energy consumption needs, there is an urgent need to significantly reduce manufacturing costs. In recent years, perovskite solar cells (PSCs) have received widespread attention based on very low material costs. According to previous reports, the conversion efficiency of organic perovskite solar cells has rapidly increased from 9.2% to 20.5% [
3,
4], and the mobility of perovskite samples is calculated to be 60–75 cm
2 V
−1 s
−1 [
5]. However, organic perovskite MAPbI
3 still has many problems that need to be overcome. For example, it is easily degraded for organic perovskite in air and the hygroscopicity of methylammonium (MA) cations will trap moisture in the air, which will increase the crystal size and cause pollution [
6,
7]. Therefore, improving the organic perovskite MAPbI
3 has become a concern in recent years. Amalie Dualeh et al. used control of the film formation temperature to improve the photoelectric conversion efficiency (PCE) of MAPbI
3 [
8]; Xiao Bing et al. used inorganic PbCl
2 to increase the carrier mobility of perovskite solar cells [
9]; LC Chen et al. used doped FAPbI
3 quantum dots (QDs) to enhance the photoelectric conversion efficiency of MAPbI
3 [
10]. It can be found that passivation treatment and doping with inorganic materials have become an important basis for improving organic perovskite MAPbI
3. Based on the above, doping inorganic quantum dots (CsPbI
3) into MAPbI
3 is still poorly studied. Therefore, in this article, a detailed investigation of improvements in the light-absorption capacity and carrier mobility of MAPbI
3 by doping with inorganic quantum dots CsPbI
3 and changing the filming temperature is presented.
2. Results
As shown in
Figure 1a, when the filming temperature is 80–100 °C, pristine MAPbI
3 can still show a typical perovskite absorption spectrum; however, when the filming temperature is further heated to 120–140 °C, the pristine MAPbI
3 shows a significant decrease in the absorption spectrum, and the decomposed to PbI
2 phase dominated. The decomposition of MAPbI
3 can change from dark brown to yellow, similar to previous reports [
11,
12].
Figure 1b shows the absorption spectrum for CsPbI
3-QD doped perovskite thin films. It can be found that when the filming temperature exceeds 120 °C, the typical perovskite absorption peak can still be observed at 750 nm. This is due to the addition of CsPbI
3-QDs, which stabilize the structure of the perovskite film surface and make MAPbI
3 difficult to degrade. In addition, after increasing the filming temperature, the absorption area increases significantly in the entire spectral range (350–850 nm), and the long-wavelength absorption (750 nm) is significantly improved. This is because the energy gap of CsPbI
3 QD is wider and a small strain occurs at the QDs–MAPbI
3 interface [
12,
13,
14]. Therefore, adding CsPbI
3 QDs can not only stabilize the MAPbI
3 film at a higher filming temperature, but also improve the absorption of the film at long wavelengths, and further enhance the absorption capacity of CsPbI
3-QD doped perovskite thin films in the active layer of perovskite solar cells.
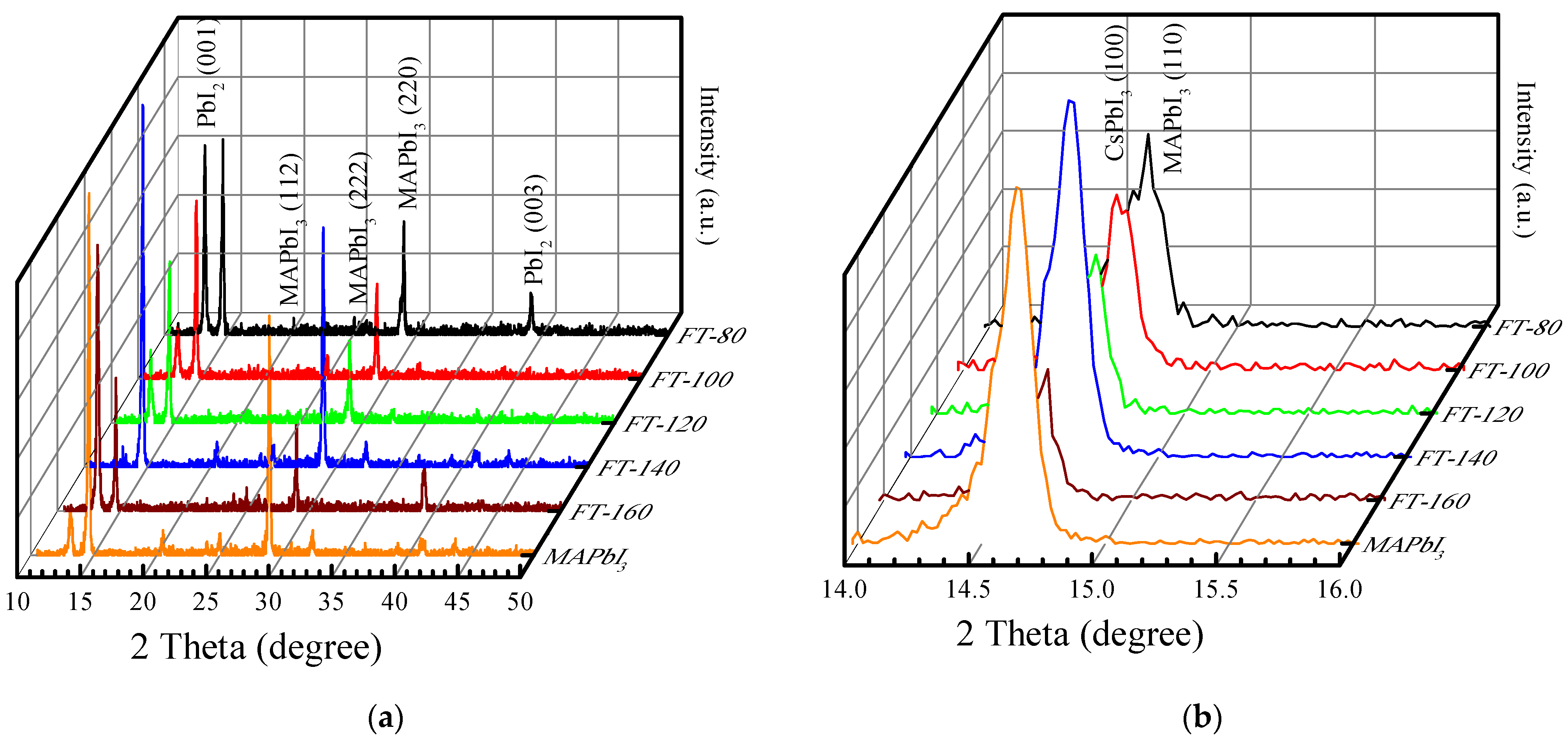
Figure 2a demonstrates the X-ray diffraction (XRD) pattern of CsPbI
3-QD doped perovskite thin films when the filming temperature is 80–160 °C. Based on the spectra of conventional MAPbI
3 films [
14], the peak position for MAPbI
3 in CsPbI
3-QD doped perovskite thin films under different filming temperatures appeared at 14° and 28° and all the films demonstrated strongest intensity along (110). There is an additional new peak at 12.7°, which is attributed to PbI
2. The intensity of the PbI
2 peak of the control sample (pristine MAPbI
3) is much greater than that of BT-140, and there is almost no PbI
2 peak in FT-140. This is due to the better thermal stability that effectively inhibits the formation of PbI
2 and the doping of CsPbI
3 QDs avoids the degradation of MAPbI
3 which is due to the decrease in hydrogen bonds in MAPbI
3 and the increase in the octahedral tilt due to the Cs-ion exchange process [
15]. When the filming temperature is increased to 160 °C, the peak intensity of PbI
2 (001) is much stronger than the perovskite peak. Generally, the change in the filming temperature can be used to remove impurities or organic substances from the surface of the film to optimize it. When the filming temperature is lower than 140 °C, excess ligands (oleylamine, oleic acid) or PbI
2 is removed, but when the filming temperature is 160 °C, MAPbI
3 degrades, resulting in a large amount of PbI
2 that will damage the structure of the doped thin film.
Figure 2b shows the details of the preferred peaks of the QD doped film. According to previous research, it is found that when the filming temperature is up to 140 °C, the ratio of the peak area CsPbI
3/MAPbI
3 is close to 1 and the perovskite crystallinity is optimal [
12].
In order to explain the charge recombination effect introduced by CsPbI
3-QDs, the XPS spectrum of the film was measured and it was understood that changing the film formation temperatures may affect the surface stability of the MAPbI
3 film.
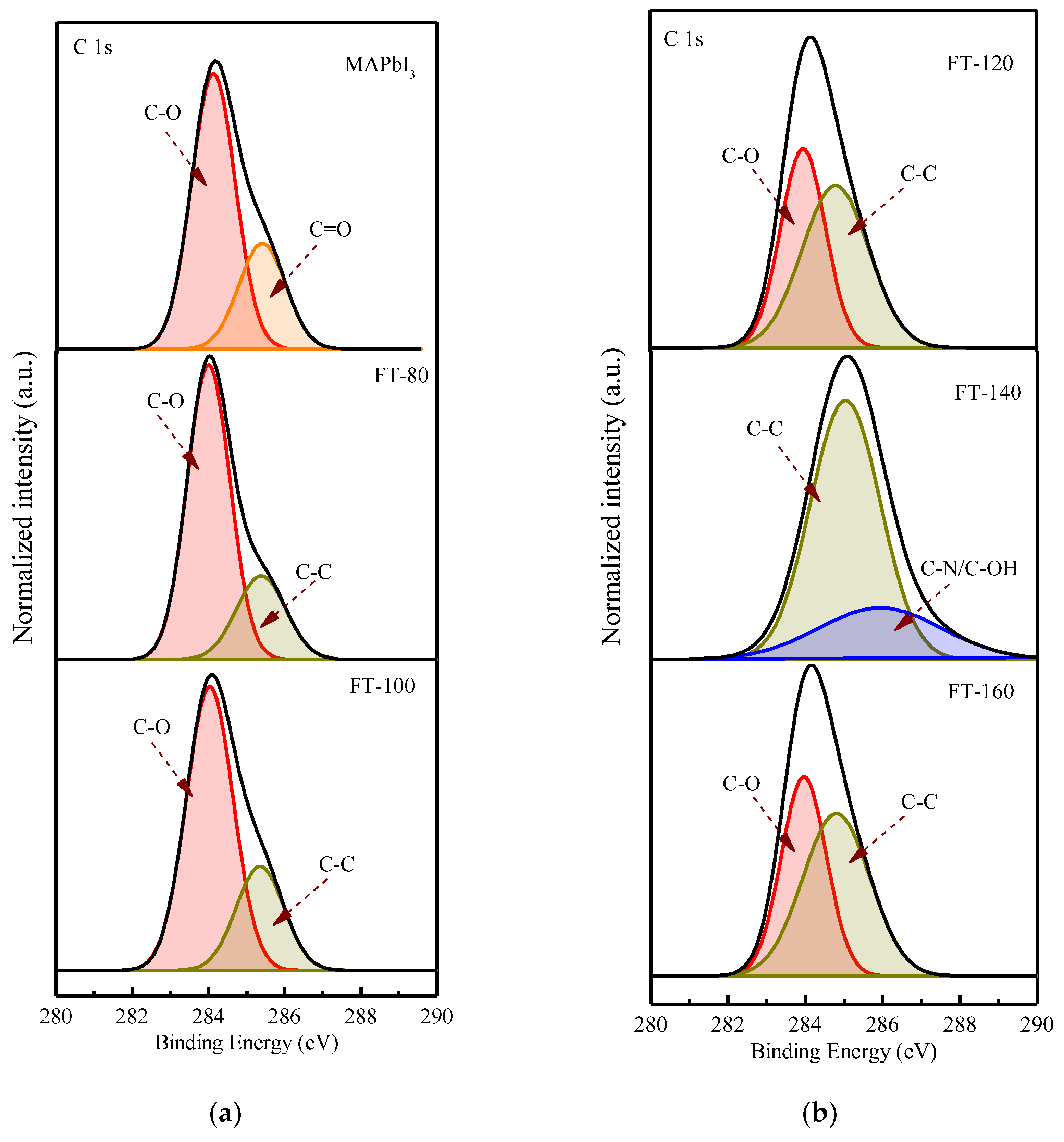
Figure 3 shows the core-level spectra of CsPbI
3-QD doped perovskite thin films at different filming temperatures. The deconvolution characteristic of the carbon peak shows the binding state of carbon material and atmospheric oxygen. The peak at 283.97 eV corresponds to C-O and the peak at 285.4 eV corresponds to C=O [
16]; the carbon configuration combined with oxygen can be found in the spectra of the control group (pristine MAPbI
3), which is due to the moisture absorption of the MAPbI
3 film when it is exposed to air and the perovskite thin films surface will be oxidized; therefore, it will lead to the appearance of a C-O peak and C=O peak. After adding CsPbI
3-QDs, the C=O peak disappeared and was converted to a C-C peak; even after the filming temperature was increased to 140 °C, the C-O peak disappeared. This could be due to the higher temperature which will eliminate the weakly bound organic components.
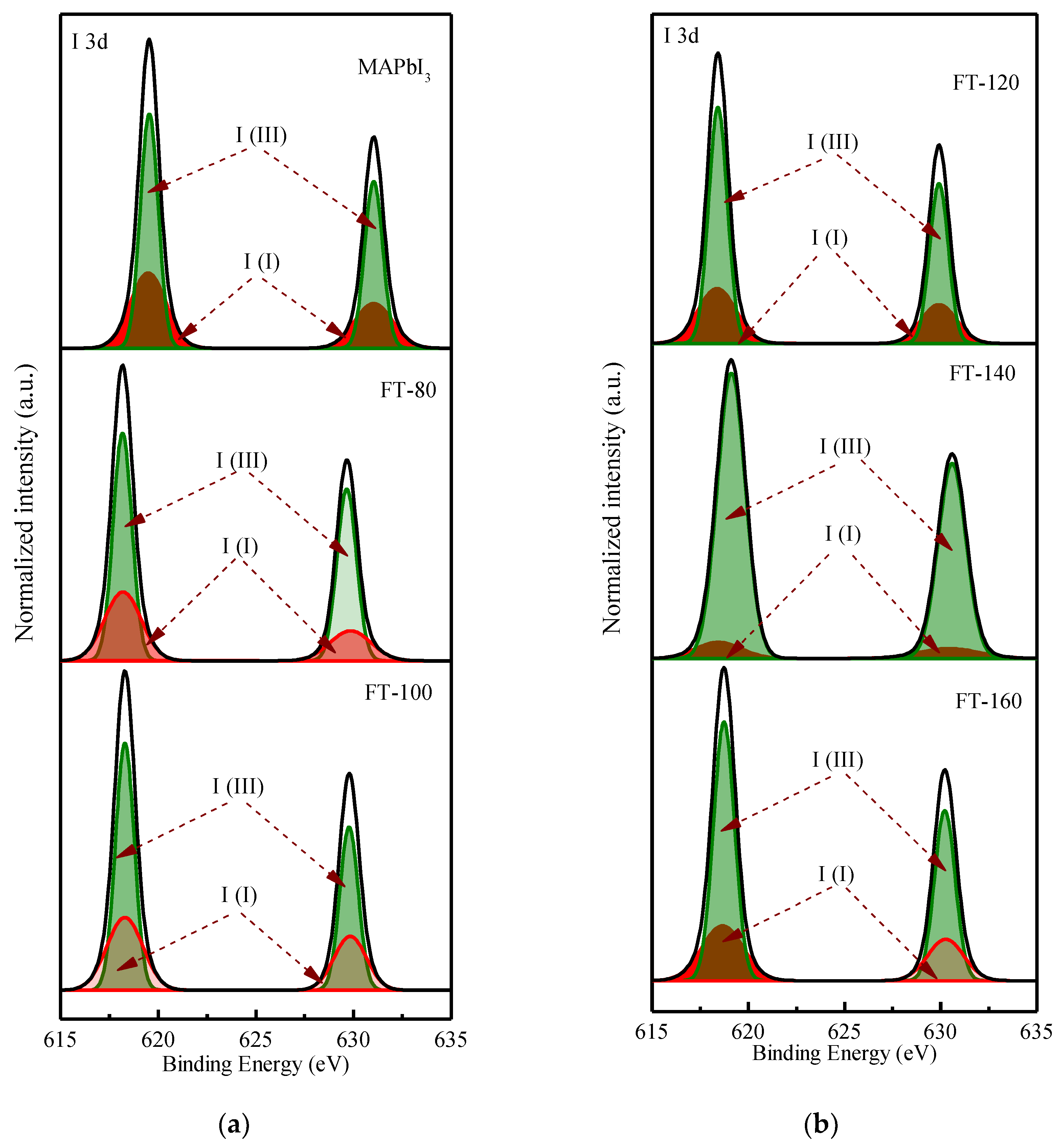
Figure 4 shows the deconvoluted XPS spectrum of the I 3d doublet. The values of 619.5 and 631 eV correspond to the I
3− charge state, while 619.37 and 630.87 eV correspond to the I
2+ charge state.
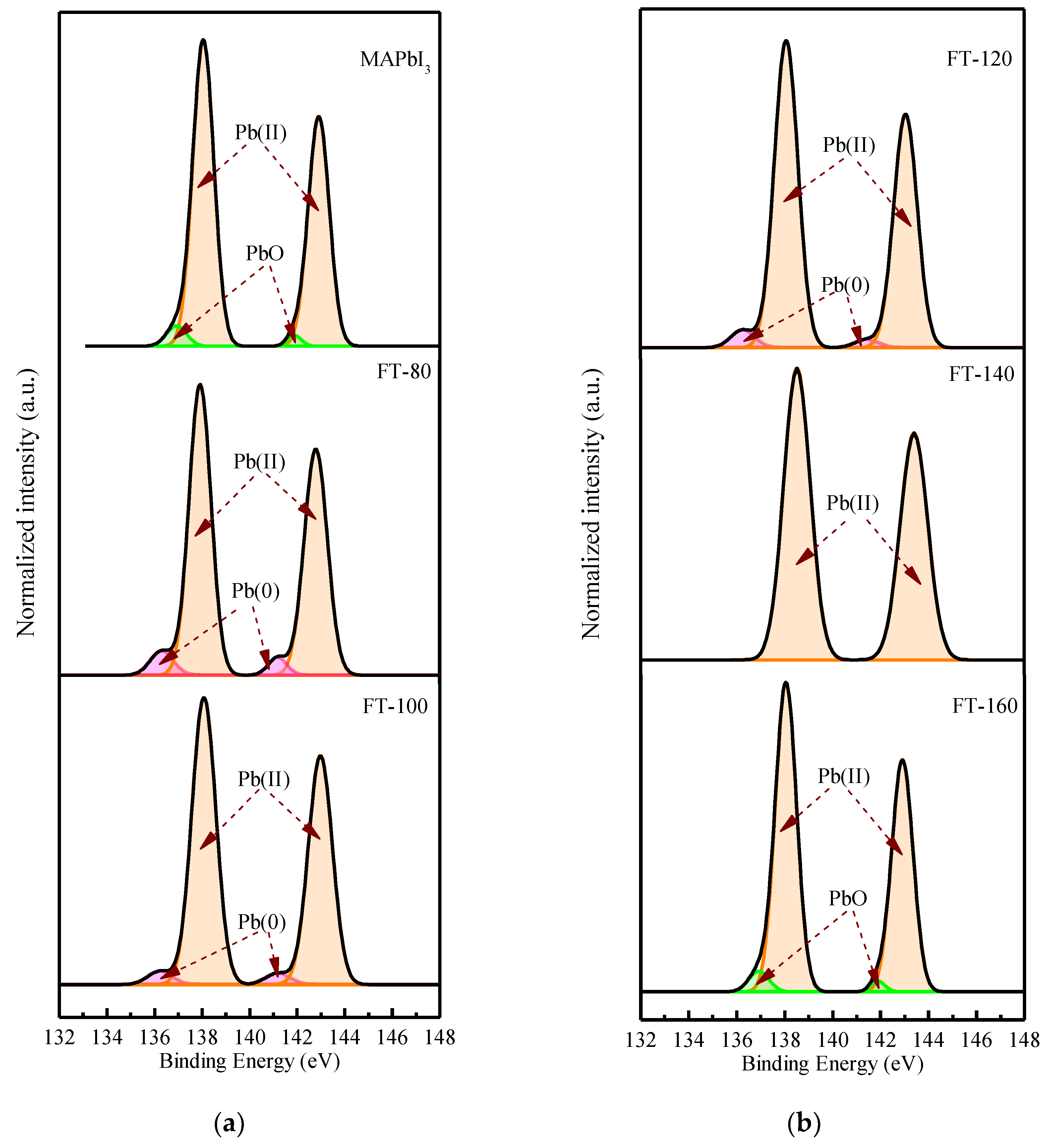
Figure 5 shows the deconvoluted XPS spectrum of the Pb 4f doublet. The values of 136.23 and 141.18 eV correspond to metallic lead (Pb), while 138.07 and 142.97 eV correspond to Pb (II) in perovskite. From the Pb XPS spectra, it can be found that after adding quantum dots and increasing the film forming temperature, the percentage of Pb (II) species is relatively higher than that of metal Pb, even if the temperature is increased to 160 °C. This shows that the iodine atom interacts with the lead atom and forms a donor–acceptor complex. This is because the low electronegativity Pb atom provides the excess unpaired electrons to the high electronegativity I(I), and in the process of electron transfer, the Pb atom is oxidized to Pb
2+ and provides two electrons to reduce the iodine atom to 2I
−, and is further reduced to triiodide(I
3−). It can be clearly understood by the following equation:
However, when the filming temperature is increased to 140 °C, the peak of metallic lead disappears. Recent studies have shown that the peak of metallic lead is derived from unsaturated lead, and the presence of unsaturated lead atoms is related to the lack of iodide [
17], and the metal lead is compounded as recombination point, leading to poor performance. Due to its thermal stability, Cs atoms replace some MA, resulting in the loss of molecular groups and fewer iodine atoms at the A site of the perovskite, and unsaturated Pb is effectively suppressed.
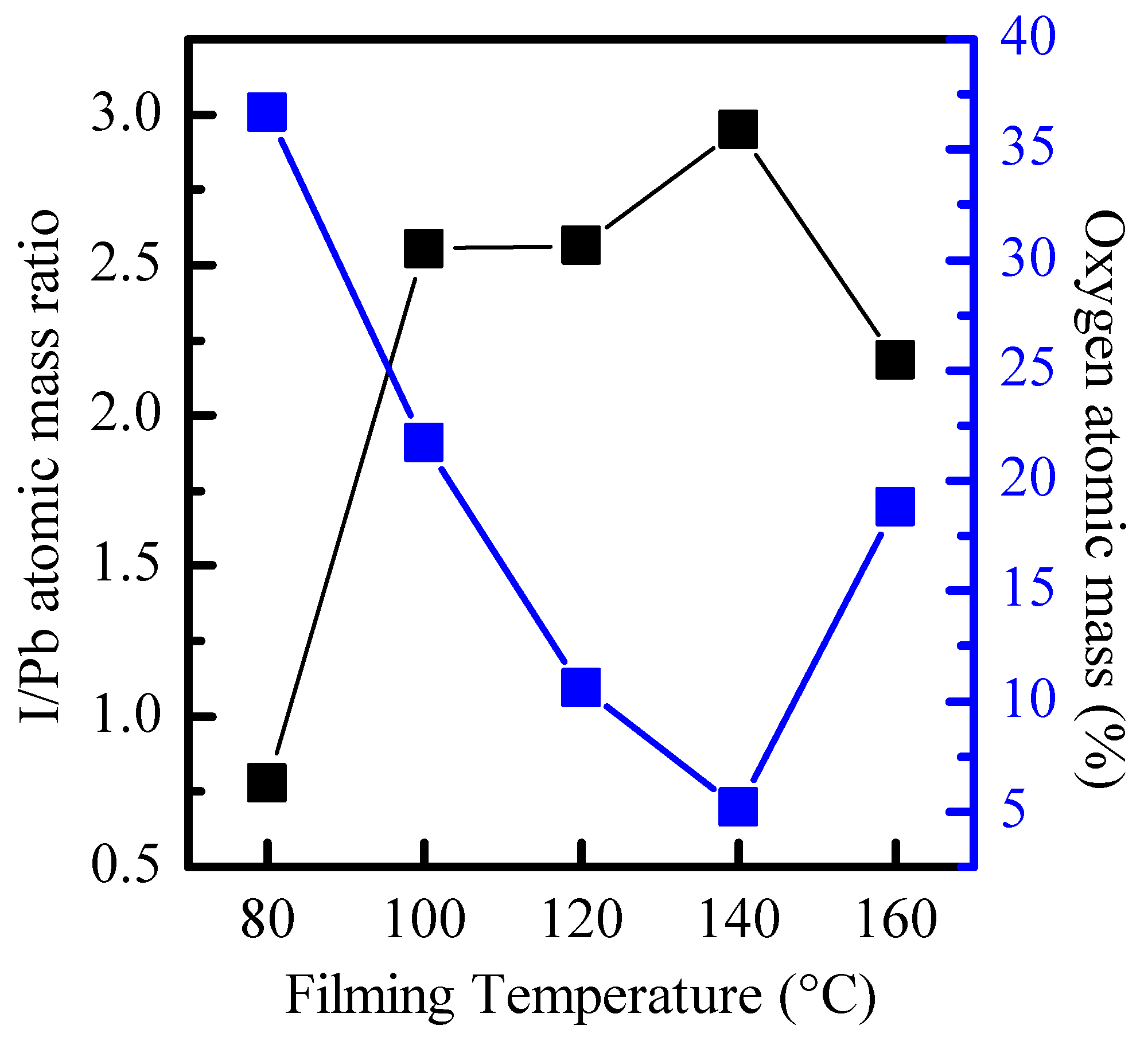
Figure 6 shows the relationship between the I/Pb atomic mass ratio calculated from the integral area of Pb 4f and I 3d and the total atomic mass percentage of O 1s and the filming temperature. Research has pointed out that the thickness of the film is related to the combination of surface oxygen [
18]; however, the thickness of the film is 295 nm at different filming temperatures. Therefore, it can be further inferred that the total atomic concentration of the I 3d peak gradually increases relative to the total concentration of the Pb 4f peak, which is related to the reduction in surface oxides. Therefore, it can be seen that when the filming temperature is 140 °C (I/Pb ratio is closest to 3), the CsPbI
3-QD doped perovskite thin films surface can be effectively stabilized and prevented from oxidation.
It can be found from
Table 1 that after the filming temperature is increased, the mobility is significantly increased. This is attributed to the addition of CsPbI
3 QDs, which effectively prevents the formation of metallic lead and reduces the chance of electron-hole recombination.
3. Materials and Methods
3.1. Materials
All materials contain cesium carbonate (Cs
2CO
3, 99.9%), lead(II) iodide (PbI
2, 99.9985%), oleic acid (C
18H
34O
2, analytical reagent 90%), oleyl amine (C
18H
35NH
2, 90%), 1-octadecene (ODE, technical grade 90%), toluene (anhydrous, 99.8%), hexane (analytical reagent, 97%), methyl acetate (MeOAc, anhydrous 99.5%), methylammonium iodide (CH
3NH
3I, 99%), dimethyl sulfoxide ((CH
3)
2SO, 99%) and gamma-butyrolactone (C
4H
6O
2, 99.9%), as shown in
Table 1. All the chemicals in this work were used without further treatment.
3.2. Solution Preparation and Synthesis for Cs-Oleate Precursor, CsPbI3 QDs and CH3NH3PbI3
The experimental method is the modified hot-injection method previously reported [
12]. All experiments were performed in a glove box filled with nitrogen, H
2O < 1 ppm and O
2 < 1 ppm.
3.3. Synthesis of Cs-Oleate
Cs2CO3 (0.1 g), OA (0.5 mL) and ODE (10 mL) were loaded into a 50 mL sample bottle and stirred for 1 h at 120 °C. We used heating and air extraction to remove moisture and internal air. Then, the solution was heated at 150 °C until the solution was clear. Finally, the Cs-oleate was stored at 100 °C to avoid precipitation.
3.4. Synthesis of CsPbI3 QDs
Both ODE (10 mL) and PbI2 (0.173 g) were added into a 50 mL sample bottle and were dried at 120 °C for 1 h. Then, 1 mL of OA and 1 mL of OAM (preheated at 70 °C) were poured. The solution was degassed until the PbI2 completely dissolved and the solution became clear. The solution was then heated to 185 °C. The Cs-oleate (0.0625 M, 1.6 mL) precursor was swiftly injected into the solution. After 5 s, the reaction solution was cooled by immediately immersing the sample bottle into an ice bath.
3.5. Purification of CsPbI3 QD
The prepared CsPbI3 QDs were separated by adding MeOAc (volume ratio of crude solution/MeOAc is 1:3), and then they were centrifuged at 8000 rpm for 5 min. After that, the supernatant was discarded, and the precipitation that contained the QDs was dissolved in 3 mL of hexane. Then, the CsPbI3 QDs were precipitated again by adding MeOAc (volume ratio of crude solution/MeOAc is 1:1) and centrifuging at 8000 rpm for 2 min. Finally, the QDs were dispersed in 3 mL of hexane and centrifuged at 4000 rpm for 5 min to remove excess PbI2 and precursor.
3.6. Synthesis of CH3NH3I
We added CH3NH3I (198.75 mg) and PbI2 (576.25 mg) into the 50 mL sample bottle, and then added C2H6OS (0.5 mL) and C6H6O2 (0.5 mL) into the sample bottle in the glove box and stirred at 300 rpm for 24 h.
3.7. Fabrication of Thin Films
CH3NH3I (50 μL) and CsPbI3 (1 mg) were mixed and spin-coated on the glass substrate in the glove box and then filmed by different filming temperatures from 80–160 °C.
3.8. Characteristic Measurements
The absorption spectra of the thin film were measured by ultraviolet/visible (UV/vis) absorption spectroscopy (HITACHI, U-3900, Hitachi High-Technologies Corporation Tokyo, Japan). X-ray diffraction (XRD) data of films were recorded by the Bruker D8 Discover (Bruker AXS Gmbh, Karlsruhe, Germany) X-ray diffractometer with Grazing Incidence X-ray Diffraction (GIXRD) and X-ray photoelectron spectroscopy (XPS) data of films were recorded by a PHI 5000 (ULVAC-PHI, Kanagawa Prefecture, Japan) VersaProbe/Scanning ESCA Microprobe.