Lysozyme and Its Application as Antibacterial Agent in Food Industry
Abstract
:1. Introduction
2. Types of Lysozymes
3. Mode of Action of Lysozyme
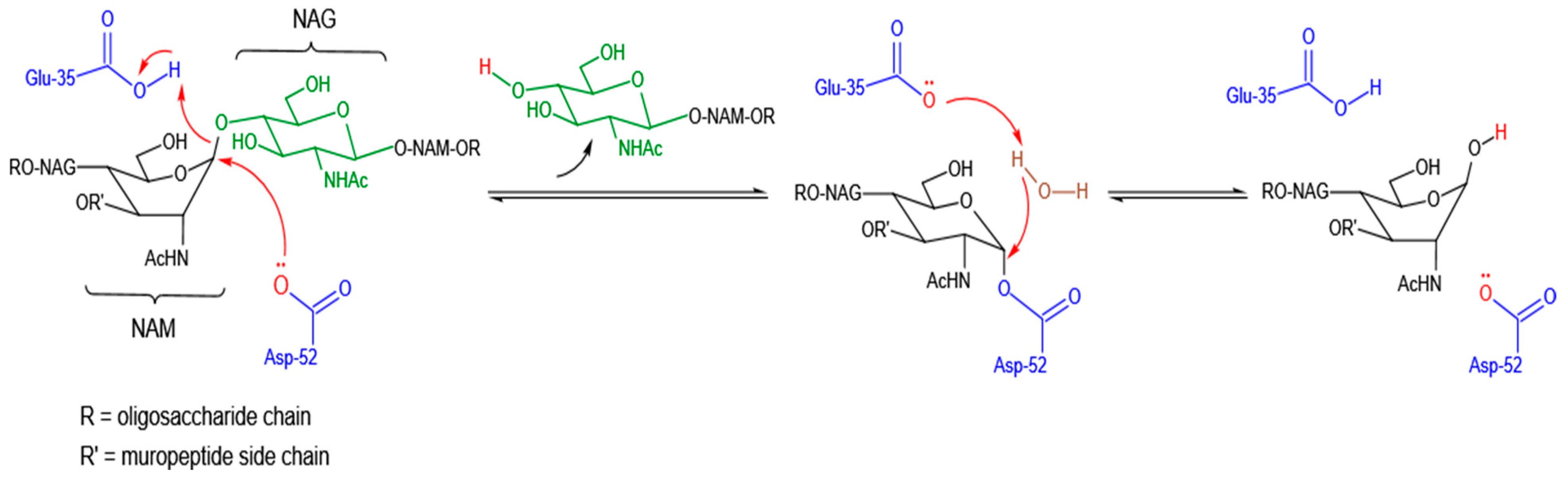
3.1. Catalytic Mode of Antibacterial Action
3.2. Non-Catalytic Mode of Antibacterial Action
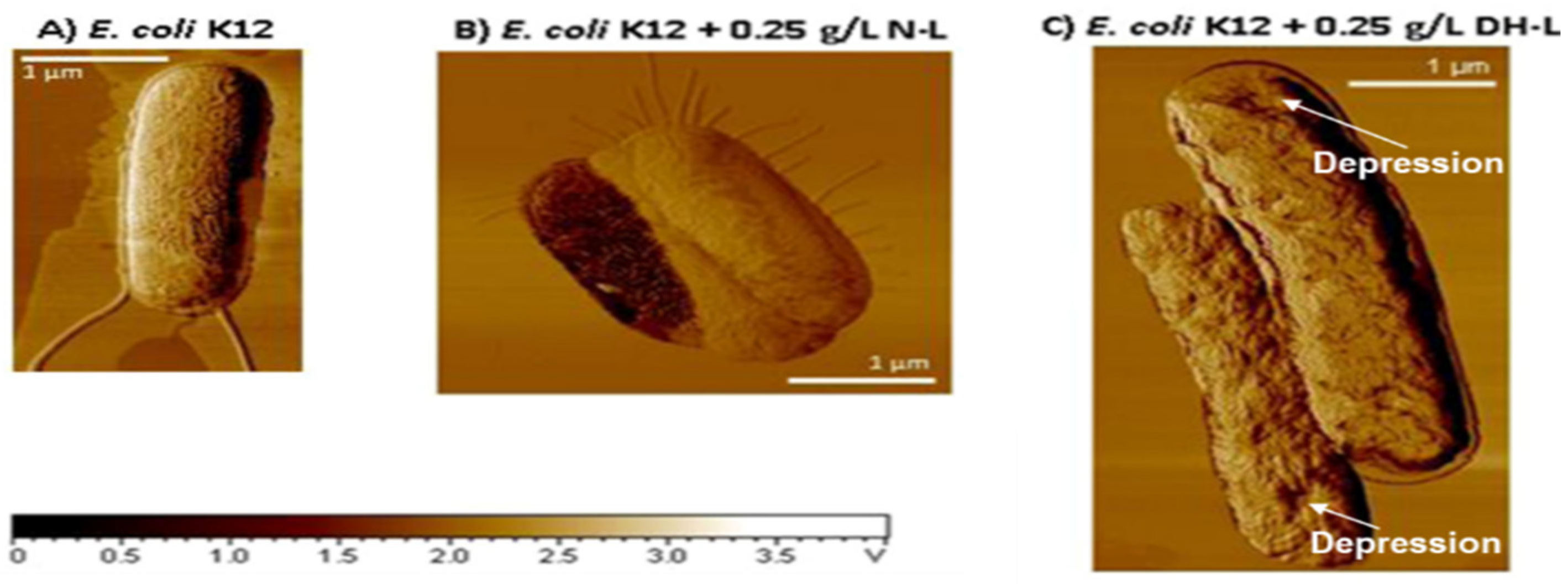
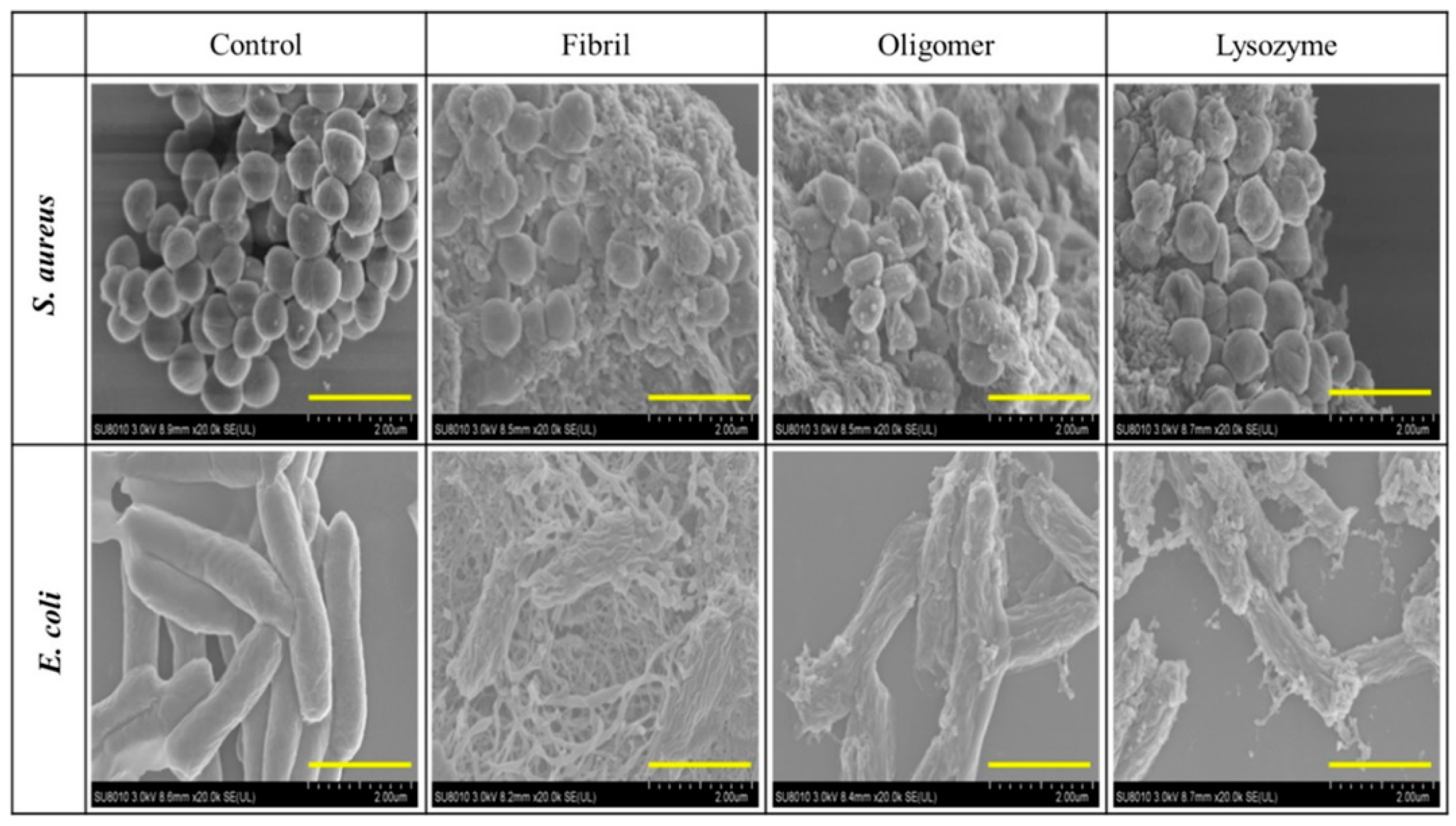
4. Susceptibility and Resistance of Bacteria to Lysozyme
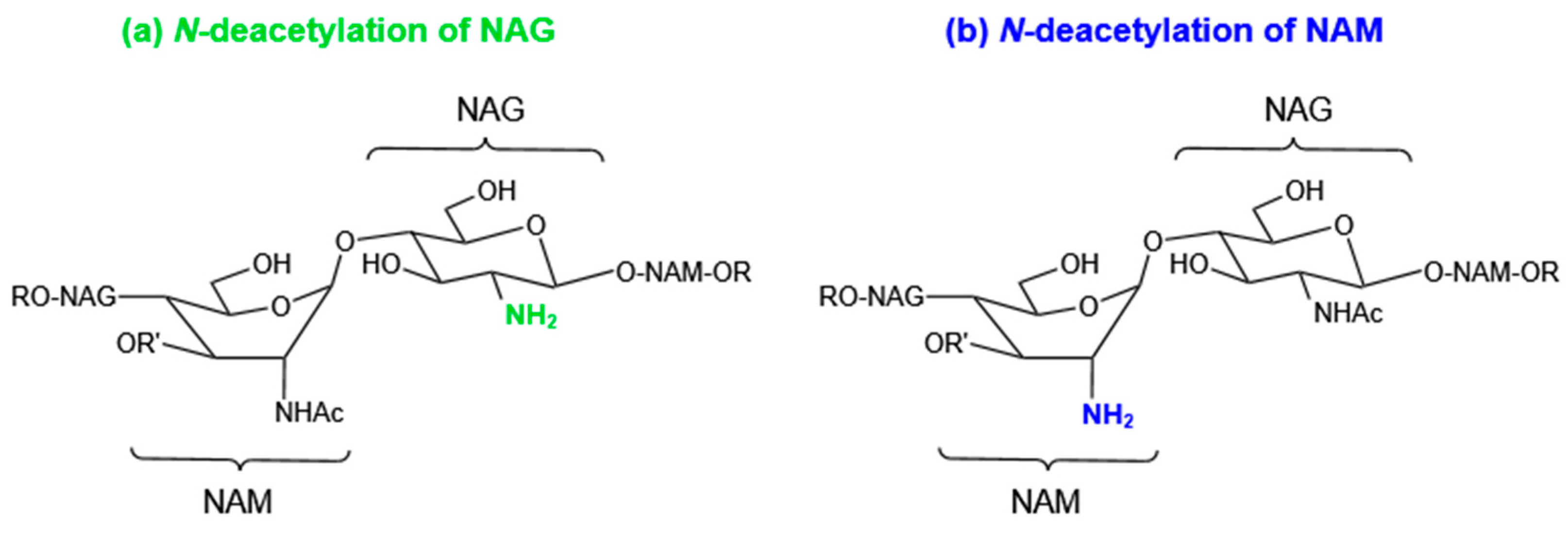
4.1. Modifications of Peptidoglycan
4.1.1. N-Deacetylation of NAG or NAM
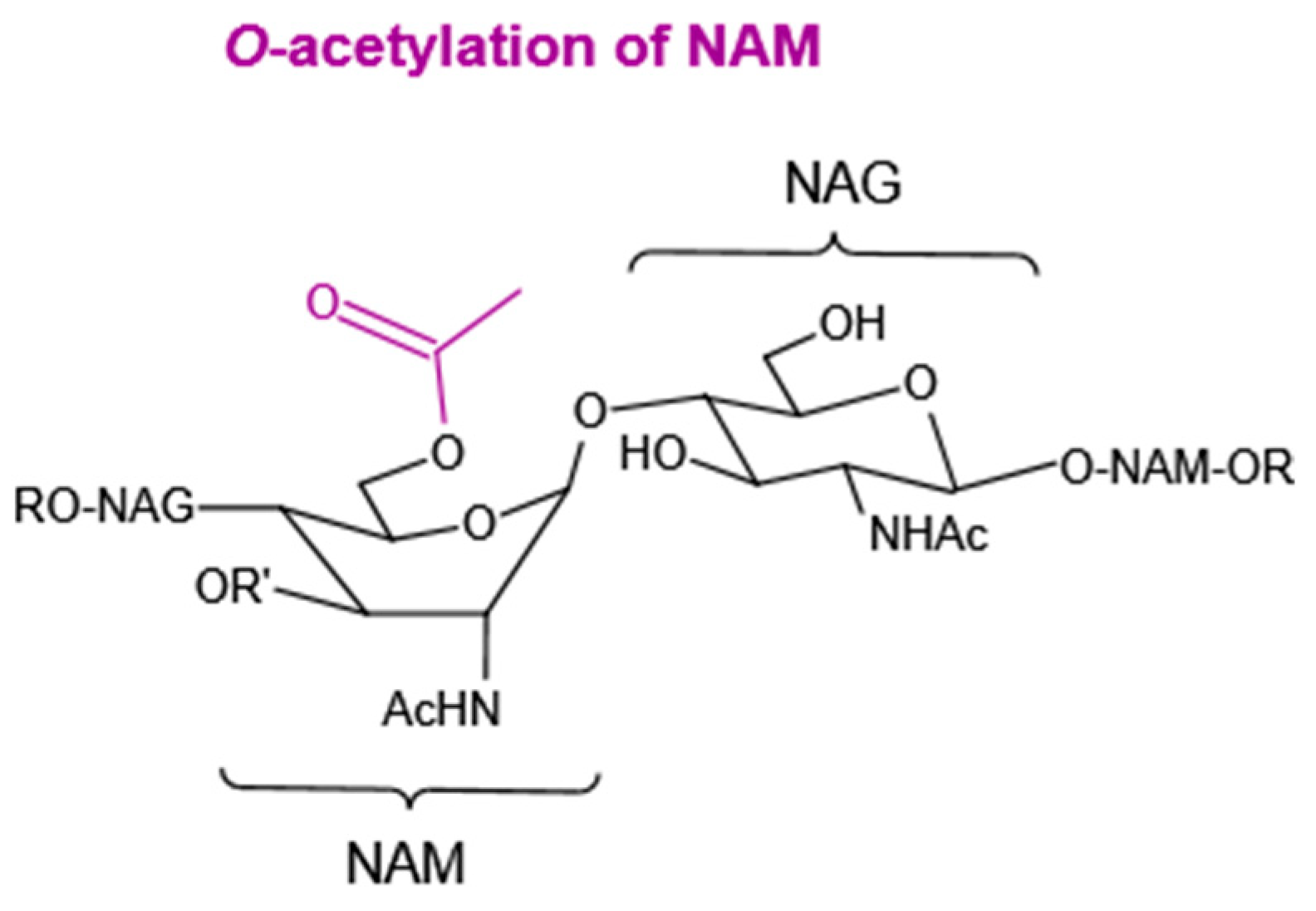
4.1.2. O-Acetylation of NAM
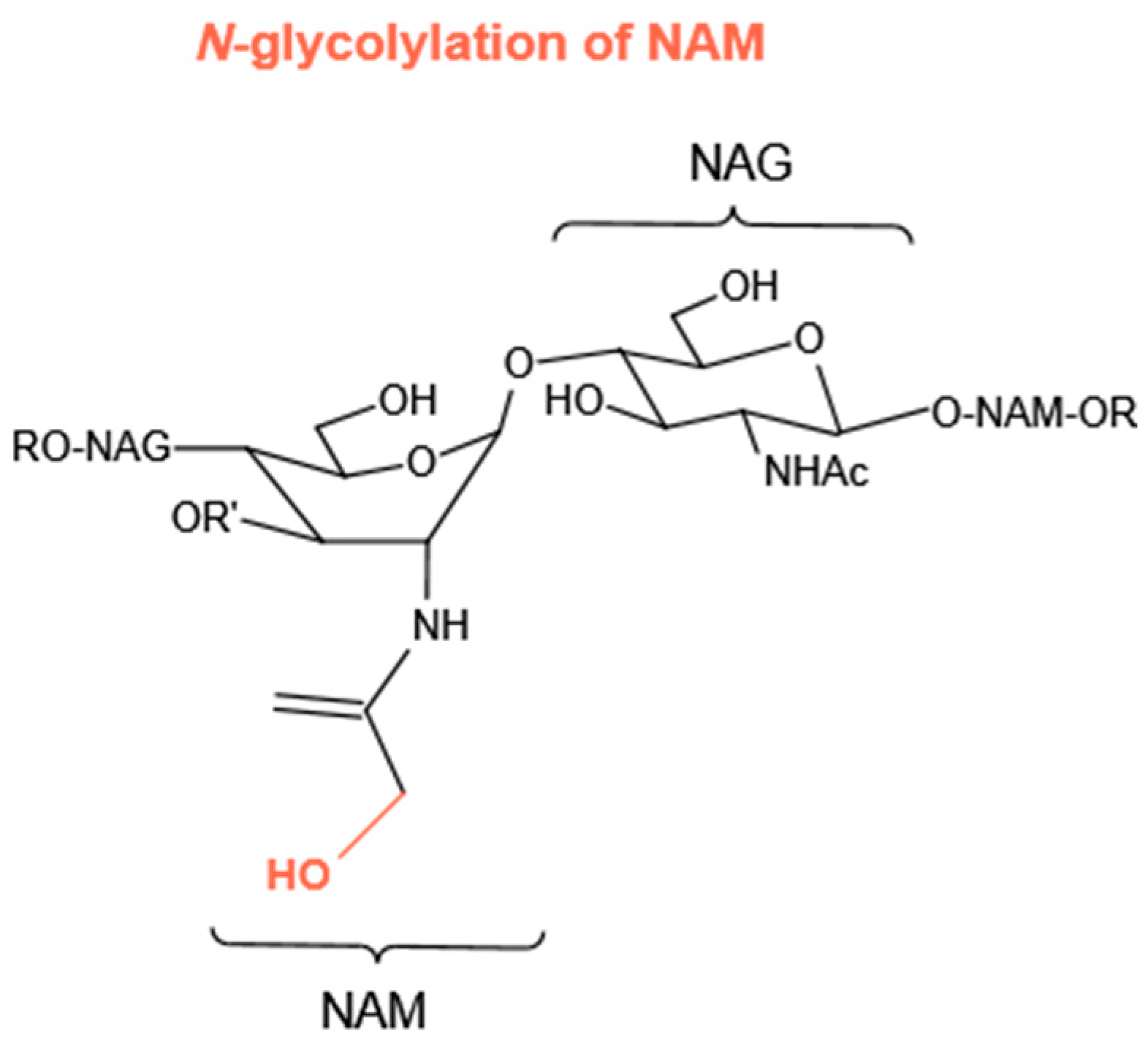
4.1.3. N-Glycolylation of NAM
4.2. Specific Proteinaceous Inhibitors of Lysozyme
4.3. Modifications of Anionic Glycopolymers of Cell Wall
5. Modification of Lysozyme
5.1. Lipophilization of Lysozyme
5.2. Modifications with Polysaccharides
6. Application of Lysozymes in the Food Industry
7. Conclusions
Funding
Conflicts of Interest
Abbreviations
| AH | amphipathic helix | MDP | muramyl dipeptide |
| AMPs | antimicrobial peptides | MliC | membrane associated inhibitor of c-type lysozyme |
| BPS | bisphenol S | NAG | N-acetylglucosamine |
| DL | destabilase-lysozyme | NAM | N-acetylmuramic acid |
| GEWL | goose egg-white lysozyme | NSC | N-succinyl chitosan |
| GFC | gold fluorescent clusters | OatA | O-acetyltransferase A |
| GRAS | generally recognized as safe | PG | peptidoglycan |
| HEWL | hen/chicken egg white lysozyme | PET | polyethylene terephthalate |
| HLH | helix-loop-helix | PliC | periplasmic inhibitor of C-type lysozyme |
| II-2 | interleukin-2 | PliI | periplasmic inhibitors of I-type lysozyme |
| IL-6 | interleukin-6 | PliG | periplasmic inhibitors of G-type lysozymes |
| Ivy | inhibitor of vertebrate lysozyme | PVOH | polyvinyl alcohol |
| LP | lactoperoxidase | SIC | streptococcal inhibitor of complement |
| LPS | lipopolysaccharides | TNFα | tumor necrosis factor |
| LYZ | lysozyme | TWAs | wall teichoic acids |
| LYZ-NSC | lysozyme-N-succinyl chitosan |
References
- Juneja, V.K.; Dwivedi, H.P.; Yan, X. Novel natural food antimicrobials. Annu. Rev. Food Sci. Technol. 2012, 3, 381–403. [Google Scholar] [CrossRef] [PubMed]
- Lesnierowski, G.; Stangierski, J. What’s new in chicken egg research and technology for human health promotion?—A review. Trends Food Sci. Technol. 2018, 71, 46–51. [Google Scholar] [CrossRef]
- Liburdi, K.; Benucci, I.; Esti, M. Lysozyme in wine: An overview of current and future applications. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1062–1073. [Google Scholar] [CrossRef]
- Ragland, S.A.; Criss, A.K. From bacterial killing to immune modulation: Recent insights into the functions of lysozyme. PLoS Pathog. 2017, 13, e1006512. [Google Scholar] [CrossRef] [PubMed]
- Tagashira, A.; Nishi, K.; Matsumoto, S.; Sugahara, T. Anti-inflammatory effect of lysozyme from hen egg white on mouse peritoneal macrophages. Cytotechnology 2018, 70, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Dowarha, D.; Katte, R.; Chou, R.-H.; Filipek, A.; Yu, C. Lysozyme as the anti-proliferative agent to block the interaction between S100A6 and the RAGE V domain. PLoS ONE 2019, 14, e0216427. [Google Scholar] [CrossRef] [PubMed]
- Liao, A.-H.; Hung, C.-R.; Lin, C.-F.; Lin, Y.-C.; Chen, H.-K. Treatment effects of lysozyme-shelled microbubbles and ultrasound in inflammatory skin disease. Sci. Rep. 2017, 7, 41325. [Google Scholar] [CrossRef]
- Shalhoub, S. Interferon beta-1b for COVID-19. Lancet 2020, 395, 1670–1671. [Google Scholar] [CrossRef]
- Acharya, D.; Liu, G.; Gack, M.U. Dysregulation of type I interferon responses in COVID-19. Nat. Rev. Immunol. 2020, 20, 397–398. [Google Scholar] [CrossRef]
- Zhang, W.; Rhim, J.W. Functional edible films/coatings integrated with lactoperoxidase and lysozyme and their application in food preservation. Food Control 2022, 133, 108670. [Google Scholar] [CrossRef]
- Leśnierowski, G.; Yang, T. Lysozyme and its modified forms: A critical appraisal of selected properties and potential. Trends Food Sci. Technol. 2021, 107, 333–342. [Google Scholar] [CrossRef]
- Kim, J.W.; Yoe, S.M. Analysis of a lysozyme gene from sweet potato hornworm, Agrius convolvuli. Entomol. Res. 2003, 33, 237–240. [Google Scholar] [CrossRef]
- Daffre, S.; Kylsten, P.; Samakovlis, C.; Hultmark, D. The lysozyme locus in Drosophila melanogaster: An expanded gene family adapted for expression in the digestive tract. Mol. Genet. Genom. 1994, 242, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Fujita, A.; Minamoto, T.; Shimizu, I.; Abe, T. Molecular cloning of lysozyme-encoding cDNAs expressed in the salivary gland of a wood-feeding termite, Reticulitermes speratus. Insect Biochem. Mol. Biol. 2002, 32, 1615–1624. [Google Scholar] [CrossRef]
- Araújo, C.A.; Waniek, P.J.; Stock, P.; Mayer, C.; Jansen, A.M.; Schaub, G.A. Sequence characterization and expression patterns of defensin and lysozyme encoding genes from the gut of the reduviid bug Triatoma brasiliensis. Insect Biochem. Mol. Biol. 2006, 36, 547–560. [Google Scholar] [CrossRef]
- Takano, K.; Yamagata, Y.; Yutani, K. Role of amino acid residues at turns in the conformational stability and folding of human lysozyme. Biochemistry 2000, 39, 8655–8665. [Google Scholar] [CrossRef]
- Huang, J.; Wu, L.; Yalda, D.; Adkins, Y.; Kelleher, S.L.; Crane, M.; Lonnerdal, B.; Rodriguez, R.L.; Huang, N. Expression of functional recombinant human lysozyme in transgenic rice cell culture. Transgenic Res. 2002, 11, 229–239. [Google Scholar] [CrossRef]
- Yang, B.; Wang, J.; Tang, B.; Liu, Y.; Guo, C.; Yang, P.; Yu, T.; Li, R.; Zhao, J.; Zhang, L.; et al. Characterization of bioactive recombinant human lysozyme expressed in milk of cloned transgenic cattle. PLoS ONE 2011, 6, e17593. [Google Scholar] [CrossRef]
- Dan, L.; Liu, S.; Shang, S.; Zhang, H.; Zhang, R.; Li, N. Expression of recombinant human lysozyme in bacterial artificial chromosome transgenic mice promotes the growth of Bifidobacterium and inhibits the growth of Salmonella in the intestine. J. Biotechnol. 2018, 272, 33–39. [Google Scholar] [CrossRef]
- Gao, Y.; Zhao, H.L.; Feng, X.; Zhai, R.D.; Zhu, S.; DU, C.T.; Sun, C.J.; Lei, L.C. Expression of recombinant human lysozyme-tachyplesin I (hLYZ-TP I) in Pichia pastoris and analysis of antibacterial activity. Biomed. Environ. Sci. 2013, 26, 319–322. [Google Scholar]
- Pooart, J.; Torikata, T.; Araki, T. The primary structure of a novel goose-type lysozyme from rhea egg white. Biosci. Biotechnol. Biochem. 2004, 68, 159–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thammasirirak, S.; Torikata, T.; Takami, K.; Murata, K.; Araki, T. The primary structure of cassowary (Casuarius casuarius) goose type lysozyme. Biosci. Biotechnol. Biochem. 2002, 66, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Song, L.; Li, C.; Zou, H.; Ni, D.; Wang, W.; Xu, W. Molecular cloning of an invertebrate goose-type lysozyme gene from Chlamys farreri, and lytic activity of the recombinant protein. Mol. Immunol. 2007, 44, 1198–1208. [Google Scholar] [CrossRef] [PubMed]
- Nilsen, I.W.; Myrnes, B.; Edvardsen, R.B.; Chourrout, D. Urochordates carry multiple genes for goose-type lysozyme and no genes for chicken-or invertebrate-type lysozymes. Cell. Mol. Life Sci. 2003, 60, 2210–2218. [Google Scholar] [CrossRef] [PubMed]
- Paskewitz, S.; Li, B.; Kajla, M. Cloning and molecular characterization of two invertebrate-type lysozymes from Anopheles gambiae. Insect Mol. Biol. 2008, 17, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.G.; Itoh, N.; Schey, K.L.; Li, Y.L.; Cooper, R.K.; La Peyre, J.F. A new lysozyme from the eastern oyster (Crassostrea virginica) indicates adaptive evolution of i-type lysozymes. Cell. Mol. Life Sci. 2007, 64, 82–95. [Google Scholar] [CrossRef] [PubMed]
- Kurdyumov, A.S.; Manuvera, V.A.; Baskova, I.P.; Lazarev, V.N. A comparison of the enzymatic properties of three recombinant isoforms of thrombolytic and antibacterial protein—Destabilase-Lysozyme from medicinal leech. BMC Biochem. 2015, 16, 27. [Google Scholar] [CrossRef]
- Peters, C.W.; Kruse, U.; Pollwein, R.; Grzeschik, K.H.; Sippel, A.E. The human lysozyme gene: Sequence organization and chromosomal localization. Eur. J. Biochem. 1989, 182, 507–516. [Google Scholar] [CrossRef]
- Simpson, R.J.; Morgan, F.J. Complete amino acid sequence of Embden goose (Anser anser) egg-white lysozyme. BBA Protein Struct. Mol. Enzymol. 1983, 744, 349–351. [Google Scholar] [CrossRef]
- Hikima, J.; Minagawa, S.; Hirono, I.; Aoki, T. Molecular cloning, expression and evolution of the Japanese flounder goose-type lysozyme gene, and the lytic activity of its recombinant protein. BBA-Gene Struct. Expr. 2001, 1520, 35–44. [Google Scholar] [CrossRef]
- Larsen, A.N.; Solstad, T.; Svineng, G.; Seppola, M.; Jørgensen, T.Ø. Molecular characterisation of a goose-type lysozyme gene in Atlantic cod (Gadus morhua L.). Fish Shellfish Immunol. 2009, 26, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Irwin, D.M.; Gong, Z.M. Molecular evolution of vertebrate goose-type lysozyme genes. J. Mol. Evol. 2003, 56, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, K.; Hashimoto, Y.; Thujihata, Y.; So, T.; Ueda, T.; Iomoto, T. Determination of the complete cDNA sequence, construction of expression systems, and elucidation of fibrinolytic activity for Tapes japonica lysozyme. Protein Expr. Purif. 2004, 36, 254–262. [Google Scholar] [CrossRef]
- Cong, L.; Yang, X.; Wang, X.; Tada, M.; Lu, M.; Liu, H.; Zhu, B. Characterization of an i-type lysozyme gene from the sea cucumber Stichopus japonicus, and enzymatic and nonenzymatic antimicrobial activities of its recombinant protein. J. Biosci. Bioeng. 2009, 107, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Vocadlo, D.J.; Davies, G.J.; Laine, R.; Withers, S.G. Catalysis by hen egg-white lysozyme proceeds via a covalent intermediate. Nature 2001, 412, 835–838. [Google Scholar] [CrossRef] [PubMed]
- Wohlkönig, A.; Huet, J.; Looze, Y.; Wintjens, R. Structural relationships in the lysozyme superfamily: Significant evidence for glycoside hydrolase signature motifs. PLoS ONE 2010, 5, e15388. [Google Scholar] [CrossRef]
- Weaver, L.; Grütter, M.; Matthews, B. The refined structures of goose lysozyme and its complex with a bound trisaccharide show that the “Goose-type” ly’sozymes lack a catalytic aspartate residue. J. Mol. Biol. 1995, 245, 54–68. [Google Scholar] [CrossRef]
- Vollmer, W.; Blanot, D.; De Pedro, M.A. Peptidoglycan structure and architecture. FEMS Microbiol. Rev. 2008, 32, 149–167. [Google Scholar] [CrossRef]
- Yang, M.M.; Wang, J.; Dong, L.; Kong, J.; Teng, Y.; Liu, P.; Fan, J.J.; Yu, X.H. Lack of association of C3 gene with uveitis: Additional insights into the genetic profile of uveitis regarding complement pathway genes. Sci. Rep. 2017, 7, 879. [Google Scholar] [CrossRef]
- Shockman, G.D. The autolytic (‘suicidase’) system of Enterococcus hirae: From lysine depletion autolysis to biochemical and molecular studies of the two muramidases of Enterococcus hirae ATCC 9790. FEMS Microbiol. Lett. 1992, 100, 261–267. [Google Scholar] [CrossRef]
- Joris, B.; Englebert, S.; Chu, C.P.; Kariyama, R.; Daneo-Moore, L.; Shockman, G.D.; Ghuysen, J.M. Modular design of the Enterococcus hirae muramidase-2 and Streptococcus faecalis autolysin. FEMS Microbiol. Lett. 1992, 91, 257–264. [Google Scholar] [CrossRef]
- Massidda, O.; Kariyama, R.; Daneo-Moore, L.; Shockman, G.D. Evidence that the PBP 5 synthesis repressor (psr) of Enterococcus hirae is also involved in the regulation of cell wall composition and other cell wall-related properties. J. Bacteriol. 1996, 178, 5272–5278. [Google Scholar] [CrossRef] [PubMed]
- López, R.; García, E. Recent trends on the molecular biology of pneumococcal capsules, lytic enzymes, and bacteriophage. FEMS Microbiol. Rev. 2004, 28, 553–580. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, N.; Campillo, N.E.; García, E.; Gallego, C.; Pera, B.; Diakun, G.P.; Sáiz, J.L.; García, P.; Díaz, J.F.; Menéndez, M. Cpl-7, a lysozyme encoded by a pneumococcal bacteriophage with a novel cell wall-binding motif. J. Biol. Chem. 2010, 285, 33184–33196. [Google Scholar] [CrossRef]
- García, P.; García, J.L.; García, E.; Sánchez-Puelles, J.M.; López, R. Modular organization of the lytic enzymes of Streptococcus pneumoniae and its bacteriophages. Gene 1990, 86, 81–88. [Google Scholar] [CrossRef]
- Oliveira, H.; Thiagarajan, V.; Walmagh, M.; Sillankorva, S.; Lavigne, R.; Neves-Petersen, M.T.; Kluskens, L.D.; Azeredo, J. A thermostable Salmonella phage endolysin, Lys68, with broad bactericidal properties against Gram-negative pathogens in presence of weak acids. PLoS ONE 2014, 9, e108376. [Google Scholar] [CrossRef]
- Perez, S.; Tvaroška, I. Carbohydrate–protein interactions: Molecular modeling insights. Adv. Carbohydr. Chem. Biochem. 2014, 71, 9–136. [Google Scholar]
- Henrissat, B. Classification of chitinases modules. EXS 1999, 87, 137–156. [Google Scholar]
- Moreira, L.R.S.; Filho, E.X.F. Insights into the mechanism of enzymatic hydrolysis of xylan. Appl. Microbiol. Biotechnol. 2016, 100, 5205–5214. [Google Scholar] [CrossRef]
- Li, S.; Yang, X.; Bao, M.; Wu, Y.; Yu, W.; Han, F. Family 13 carbohydrate-binding module of alginate lyase from Agarivorans sp. L11 enhances its catalytic efficiency and thermostability, and alters its substrate preference and product distribution. FEMS Microbiol. Lett. 2015, 362, fnv054. [Google Scholar] [CrossRef]
- Masschalck, B.; Michiels, C.W. Antimicrobial properties of lysozyme in relation to foodborne vegetative bacteria. Crit. Rev. Microbiol. 2003, 29, 191–214. [Google Scholar] [CrossRef] [PubMed]
- Derde, M.; Guérin-Dubiard, C.; Lechevalier, V.; Cochet, M.F.; Jan, S.; Baron, F.; Gautier, M.; Vié, V.; Nau, F. Dry-heating of lysozyme increases its activity against Escherichia coli membranes. J. Agric. Food Chem. 2014, 62, 1692–1700. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Wu, S.; Xia, J.; Shao, P.; Sun, P.; Xiang, N. Enhanced antibacterial activity of hen egg-white lysozyme against Staphylococcus aureus and Escherichia coli due to protein fibrillation. Biomacromolecules 2021, 22, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Kummer, N.; Wu, T.; De France, K.J.; Zuber, F.; Ren, Q.; Fischer, P.; Campioni, S.; Nyström, G. Self-Assembly Pathways and Antimicrobial Properties of Lysozyme in Different Aggregation States. Biomacromolecules 2021, 22, 4327–4336. [Google Scholar] [CrossRef]
- Derde, M.; Lechevalier, V.; Guérin-Dubiard, C.; Cochet, M.F.; Jan, S.; Baron, F.; Gautier, M.; Vié, V.; Nau, F. Hen egg white lysozyme permeabilizes Escherichia coli outer and inner membranes. J. Agric. Food Chem. 2013, 61, 9922–9929. [Google Scholar] [CrossRef]
- Pellegrini, A.; Thomas, U.; Bramaz, N.; Klauser, S.; Hunziker, P.; von Fellenberg, R. Identification and isolation of a bactericidal domain in chicken egg white lysozyme. J. Appl. Microbiol. 1997, 82, 372–378. [Google Scholar] [CrossRef]
- Düring, K.; Porsch, P.; Mahn, A.; Brinkmann, O.; Gieffers, W. The non-enzymatic microbicidal activity of lysozymes. FEBS Lett. 1999, 449, 93–100. [Google Scholar] [CrossRef]
- Thammasirirak, S.; Pukcothanung, Y.; Preecharram, S.; Daduang, S.; Patramanon, R.; Fukamizo, T.; Araki, T. Antimicrobial peptides derived from goose egg white lysozyme. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2010, 151, 84–91. [Google Scholar] [CrossRef]
- Zavalova, L.L.; Yudina, T.G.; Artamonova, I.I.; Baskova, I.P. Antibacterial non-glycosidase activity of invertebrate destabilase-lysozyme and of its helical amphipathic peptides. Chemotherapy 2006, 52, 158–160. [Google Scholar] [CrossRef]
- Ibrahim, H.R.; Thomas, U.; Pellegrini, A. A helix-loop-helix peptide at the upper lip of the active site cleft of lysozyme confers potent antimicrobial activity with membrane permeabilization action. J. Biol. Chem. 2001, 276, 43767–43774. [Google Scholar] [CrossRef]
- Drin, G.; Antonny, B. Amphipathic helices and membrane curvature. FEBS Lett. 2010, 584, 1840–1847. [Google Scholar] [CrossRef] [PubMed]
- Zschornig, O.; Paasche, G.; Thieme, C.; Korb, N.; Fahrwald, A.; Arnold, K. Association of lysozyme with phospholipid vesicles is accompanied by membrane surface dehydration. Gen. Physiol. Biophys. 2000, 19, 85–102. [Google Scholar] [PubMed]
- Giuliani, A.; Pirri, G.; Nicoletto, S. Antimicrobial peptides: An overview of a promising class of therapeutics. Open Life Sci. 2007, 2, 1–33. [Google Scholar] [CrossRef]
- Dimroth, P.; Kaim, G.; Matthey, U. Crucial role of the membrane potential for ATP synthesis by F (1) F (o) ATP synthases. J. Exp. Biol. 2000, 203, 51–59. [Google Scholar] [CrossRef]
- Wen, S.; Yao, D.; Liu, X.; Wang, F. A novel fluorescence resonance energy transfer-based high-throughput screening method for generation of lysozyme with improved antimicrobial activity against Escherichia coli strains. J. Agric. Food Chem. 2019, 67, 12584–12589. [Google Scholar] [CrossRef]
- Ray, B.; Bhunia, A.K. Fundamental Food Microbiology, 5th ed.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Carpenter, T.S.; Parkin, J.; Khalid, S. The free energy of small solute permeation through the Escherichia coli outer membrane has a distinctly asymmetric profile. J. Phys. Chem. Lett. 2016, 7, 3446–3451. [Google Scholar] [CrossRef]
- Nikaido, H. Molecular basis of bacterial outer membrane permeability revisited. Microbiol. Mol. Biol. Rev. 2003, 67, 593–656. [Google Scholar] [CrossRef]
- Papo, N.; Shai, Y. A molecular mechanism for lipopolysaccharide protection of Gram-negative bacteria from antimicrobial peptides. J. Biol. Chem. 2005, 280, 10378–10387. [Google Scholar] [CrossRef]
- Rice, A.; Wereszczynski, J. Atomistic scale effects of lipopolysaccharide modifications on bacterial outer membrane defenses. Biophys. J. 2018, 114, 1389–1399. [Google Scholar] [CrossRef]
- Brandenburg, K.; Koch, M.H.; Seydel, U. Biophysical characterisation of lysozyme binding to LPS Re and lipid A. Eur. J. Biochem. 1998, 258, 686–695. [Google Scholar] [CrossRef]
- Ragland, S.A.; Schaub, R.E.; Hackett, K.T.; Dillard, J.P.; Criss, A.K. Two lytic transglycosylases in Neisseria gonorrhoeae impart resistance to killing by lysozyme and human neutrophils. Cell. Microbiol. 2017, 19, e12662. [Google Scholar] [CrossRef] [PubMed]
- Vollmer, W.; Tomasz, A. The pgdA gene encodes for a peptidoglycanN-acetylglucosamine deacetylase in Streptococcus pneumoniae. J. Biol. Chem. 2000, 275, 20496–20501. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.M.; Weiser, J.N. Modifications to the peptidoglycan backbone help bacteria to establish infection. Infect. Immun. 2011, 79, 562–570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vollmer, W.; Tomasz, A. Peptidoglycan N-acetylglucosamine deacetylase, a putative virulence factor in Streptococcus pneumoniae. Infect. Immun. 2002, 70, 7176–7178. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Maier, S.E.; Lo, L.F.; Maier, G.; Dosi, S.; Maier, R.J. Peptidoglycan deacetylation in Helicobacter pylori contributes to bacterial survival by mitigating host immune responses. Infect. Immun. 2010, 78, 4660–4666. [Google Scholar] [CrossRef]
- Kaoukab-Raji, A.; Biskri, L.; Bernardini, M.L.; Allaoui, A. Characterization of SfPgdA, a Shigella flexneri peptidoglycan deacetylase required for bacterial persistence within polymorphonuclear neutrophils. Microbes Infect. 2012, 14, 619–627. [Google Scholar] [CrossRef]
- Melnyk, J.E.; Mohanan, V.; Schaefer, A.K.; Hou, C.W.; Grimes, C.L. Peptidoglycan modifications tune the stability and function of the innate immune receptor Nod2. J. Am. Chem. Soc. 2015, 137, 6987–6990. [Google Scholar] [CrossRef]
- Moynihan, P.J.; Clarke, A.J. O-Acetylated peptidoglycan: Controlling the activity of bacterial autolysins and lytic enzymes of innate immune systems. Int. J. Biochem. Cell Biol. 2011, 43, 1655–1659. [Google Scholar] [CrossRef] [PubMed]
- Pushkaran, A.C.; Nataraj, N.; Nair, N.; Götz, F.; Biswas, R.; Mohan, C.G. Understanding the structure–function relationship of lysozyme resistance in Staphylococcus aureus by peptidoglycan O-acetylation using molecular docking, dynamics, and lysis assay. J. Chem. Inf. Model. 2015, 55, 760–770. [Google Scholar] [CrossRef]
- Shimada, T.; Park, B.G.; Wolf, A.J.; Brikos, C.; Goodridge, H.S.; Becker, C.A.; Reyes, C.N.; Miao, E.A.; Aderem, A.; Götz, F.; et al. Staphylococcus aureus evades lysozyme-based peptidoglycan digestion that links phagocytosis, inflammasome activation, and IL-1β secretion. Cell Host Microbe 2010, 7, 38–49. [Google Scholar] [CrossRef]
- Laaberki, M.H.; Pfeffer, J.; Clarke, A.J.; Dworkin, J. O-Acetylation of peptidoglycan is required for proper cell separation and S-layer anchoring in Bacillus anthracis. J. Biol. Chem. 2011, 286, 5278–5288. [Google Scholar] [CrossRef] [PubMed]
- Dillard, J.P.; Hackett, K.T. Mutations affecting peptidoglycan acetylation in Neisseria gonorrhoeae and Neisseria meningitidis. Infect. Immun. 2005, 73, 5697–5705. [Google Scholar] [CrossRef] [PubMed]
- Raymond, J.B.; Mahapatra, S.; Crick, D.C.; Pavelka, M.S., Jr. Identification of the namH gene, encoding the hydroxylase responsible for the N-glycolylation of the mycobacterial peptidoglycan. J. Biol. Chem. 2005, 280, 326–333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bera, A.; Biswas, R.; Herbert, S.; Kulauzovic, E.; Weidenmaier, C.; Peschel, A.; Götz, F. Influence of wall teichoic acid on lysozyme resistance in Staphylococcus aureus. J. Bacteriol. 2007, 189, 280–283. [Google Scholar] [CrossRef]
- Fernie-King, B.A.; Seilly, D.J.; Davies, A.; Lachmann, P.J. Streptococcal inhibitor of complement inhibits two additional components of the mucosal innate immune system: Secretory leukocyte proteinase inhibitor and lysozyme. Infect. Immun. 2002, 70, 4908–4916. [Google Scholar] [CrossRef]
- Callewaert, L.; Van Herreweghe, J.M.; Vanderkelen, L.; Leysen, S.; Voet, A.; Michiels, C.W. Guards of the great wall: Bacterial lysozyme inhibitors. Trends Microbiol. 2012, 20, 501–510. [Google Scholar] [CrossRef]
- Dostal, S.M.; Fang, Y.; Guerrette, J.C.; Scanlon, T.C.; Griswold, K.E. Genetically enhanced lysozyme evades a pathogen derived inhibitory protein. ACS Chem. Biol. 2015, 10, 1110–1117. [Google Scholar] [CrossRef]
- Peschel, A.; Vuong, C.; Otto, M.; Götz, F. The D-alanine residues of Staphylococcus aureus teichoic acids alter the susceptibility to vancomycin and the activity of autolytic enzymes. Antimicrob. Agents Chemother. 2000, 44, 2845–2847. [Google Scholar] [CrossRef]
- Masschalck, B.; Deckers, D.; Michiels, C.W. Sensitization of outer-membrane mutants of Salmonella typhimurium and Pseudomonas aeruginosa to antimicrobial peptides under high pressure. J. Food Prot. 2003, 66, 1360–1367. [Google Scholar] [CrossRef]
- Bader, M.W.; Navarre, W.W.; Shiau, W.; Nikaido, H.; Frye, J.G.; McClelland, M.; Fang, F.C.; Miller, S.I. Regulation of Salmonella typhimurium virulence gene expression by cationic antimicrobial peptides. Mol. Microbiol. 2003, 50, 219–230. [Google Scholar] [CrossRef]
- Napier, B.A.; Burd, E.M.; Satola, S.W.; Cagle, S.M.; Ray, S.M.; McGann, P.; Pohl, J.; Lesho, E.P.; Weiss, D.S. Clinical use of colistin induces cross-resistance to host antimicrobials in Acinetobacter baumannii. mBio 2013, 4, e00021-13. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.Y.; Jiang, S.T.; Pan, L.J.; Zhai, Y.S. Characteristics and functional properties of wheat germ protein glycated with saccharides through Maillard reaction. Int. J. Food Sci. Technol. 2011, 46, 2197–2203. [Google Scholar] [CrossRef]
- Aminlari, M.; Ramezani, R.; Jadidi, F. Effect of Maillard-based conjugation with dextran on the functional properties of lysozyme and casein. J. Sci. Food Agric. 2005, 85, 2617–2624. [Google Scholar] [CrossRef]
- Alahdad, Z.; Ramezani, R.; Aminlari, M.; Majzoobi, M. Preparation and Properties of Dextran Sulfate—Lysozyme Conjugate. J. Agric. Food Chem. 2009, 57, 6449–6454. [Google Scholar] [CrossRef] [PubMed]
- Corzo-Martínez, M.; Moreno, J.F.; Villamiel, M.; Harte, F.M. Characterization and improvement of rheological properties of sodium caseinate glycated with galactose, lactose and dextran. Food Hydrocoll. 2010, 24, 88–97. [Google Scholar] [CrossRef]
- Seo, S.; Karboune, S.; L’Hocine, L.; Yaylayan, V. Characterization of glycated lysozyme with galactose, galactooligosaccharides and galactan: Effect of glycation on structural and functional properties of conjugates. LWT-Food Sci. Technol. 2013, 53, 44–53. [Google Scholar] [CrossRef]
- Lesnierowski, G.; Kijowski, J. Lysozyme. In Bioactive Egg Compounds; Springer: Berlin/Heidelberg, Germany, 2007; pp. 33–42. [Google Scholar]
- Liu, S.T.; Sugimoto, T.; Azakami, H.; Kato, A. Lipophilization of lysozyme by short and middle chain fatty acids. J. Agric. Food Chem. 2000, 48, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Azakami, H.; Kato, A. Improvement in the yield of lipophilized lysozyme by the combination with Maillard-type glycosylation. Nahrung 2000, 44, 407–410. [Google Scholar] [CrossRef]
- Syngai, G.G.; Ahmed, G. Lysozyme: A natural antimicrobial enzyme of interest in food applications. In Enzymes in Food Biotechnology; Academic Press: London, UK, 2019; pp. 169–179. [Google Scholar]
- Abdou, A.M.; Kim, M.; Sato, K. Functional proteins and peptides of hen’s egg origin. In Bioactive Food Peptides in Health and Disease; IntechOpen: London, UK, 2013; pp. 115–116. [Google Scholar]
- Dickinson, E. Hydrocolloids at interfaces and the influence on the properties of dispersed systems. Food Hydrocoll. 2003, 17, 25–39. [Google Scholar] [CrossRef]
- Aminlari, L.; Hashemi, M.M.; Aminlari, M. Modified lysozymes as novel broad spectrum natural antimicrobial agents in foods. J. Food Sci. 2014, 79, R1077–R1090. [Google Scholar] [CrossRef]
- Amiri, S.; Ramezani, R.; Aminlari, M. Antibacterial activity of dextran-conjugated lysozyme against Escherichia coli and Staphylococcus aureus in cheese curd. J. Food Prot. 2008, 71, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Jiang, Q.; Wu, D.; Hu, Y.; Chen, S.; Ding, T.; Ye, X.; Liu, D.; Chen, J. What is new in lysozyme research and its application in food industry? A review. Food Chem. 2019, 274, 698–709. [Google Scholar] [CrossRef] [PubMed]
- Ferraboschi, P.; Ciceri, S.; Grisenti, P. Applications of lysozyme, an innate immune defense factor, as an alternative antibiotic. J. Antibiot. 2021, 10, 1534. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.R.; Kato, A.; Kobayashi, K. Antimicrobial effects of lysozyme against Gram-negative bacteria due to covalent binding of palmitic acid. J. Agric. Food Chem. 1991, 39, 2077–2082. [Google Scholar] [CrossRef]
- Nakamura, S.; Kato, A.; Kobayashi, K. New antimicrobial characteristics of lysozyme-dextran conjugate. J. Agric. Food Chem. 1991, 39, 647–650. [Google Scholar] [CrossRef]
- Nakamura, S.; Kato, A. Multi-functional biopolymer prepared by covalent attachment of galactomannan to egg-white proteins through naturally occurring Maillard reaction. Nahrung 2000, 44, 201–206. [Google Scholar] [CrossRef]
- Song, Y.; Babiker, E.E.; Usui, M.; Saito, A.; Kato, A. Emulsifying properties and bactericidal action of chitosan–lysozyme conjugates. Int. Food Res. J. 2002, 35, 459–466. [Google Scholar] [CrossRef]
- Park, S.I.; Daeschel, M.; Zhao, Y. Functional properties of antimicrobial lysozyme-chitosan composite films. J. Food Sci. 2004, 69, M215–M221. [Google Scholar] [CrossRef]
- Gaare, M.; Hussain, S.A.; Mishra, S.K.; Ram, C. Natural antimicrobials for preservation of food. In Dairy and Food Processing Industry Recent Trends-Part I, 1st ed.; Biotech Books: New Delhi, India, 2013; pp. 204–230. [Google Scholar]
- Cegielska-Radziejewska, R.; Lesnierowski, G.; Kijowski, J. Properties and application of egg white lysozyme and its modified preparations—A review. Pol. J. Food Nutr. Sci. 2008, 58, 5–10. [Google Scholar]
- Mangalassary, S.; Han, I.; Rieck, J.; Acton, J.; Dawson, P. Effect of combining nisin and/or lysozyme with in-package pasteurization for control of Listeria monocytogenes in ready-to-eat turkey bologna during refrigerated storage. Food Microbiol. 2008, 25, 866–870. [Google Scholar] [CrossRef]
- Stocco, G.; Cipolat-Gotet, C.; Cecchinato, A.; Calamari, L.; Bittante, G. Milk skimming, heating, acidification, lysozyme, and rennet affect the pattern, repeatability, and predictability of milk coagulation properties and of curd-firming model parameters: A case study of Grana Padano. J. Dairy Sci. 2015, 98, 5052–5067. [Google Scholar] [CrossRef] [PubMed]
- Silvetti, T.; Brasca, M.; Lodi, R.; Vanoni, L.; Chiolerio, F.; de Groot, M.; Bravi, A. Effects of lysozyme on the microbiological stability and organoleptic properties of unpasteurized beer. J. Inst. Brew. 2010, 116, 33–40. [Google Scholar] [CrossRef]
- Leone, S.; Pica, A.; Merlino, A.; Sannino, F.; Temussi, P.A.; Picone, D. Sweeter and stronger: Enhancing sweetness and stability of the single chain monellin MNEI through molecular design. Sci. Rep. 2016, 6, 34045. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wang, Z.; Yang, L.; Tian, S.; Hou, C.; Lu, Y. Lysozyme-stabilized gold fluorescent cluster: Synthesis and application as Hg2+ sensor. Analyst 2010, 135, 1406–1410. [Google Scholar] [CrossRef] [Green Version]
- Becerril, R.; Gómez-Lus, R.; Goñi, P.; López, P.; Nerín, C. Combination of analytical and microbiological techniques to study the antimicrobial activity of a new active food packaging containing cinnamon or oregano against E. coli and S. aureus. Anal. Bioanal. Chem. 2007, 388, 1003–1011. [Google Scholar] [CrossRef]
- Pramanik, U.; Kongasseri, A.A.; Shekhar, S.; Mathew, A.; Yadav, R.; Mukherjee, S. Structural Compactness in Hen Egg White Lysozyme Induced by Bisphenol S: A Spectroscopic and Molecular Dynamics Simulation Approach. ChemPhysChem 2021, 22, 1745–1753. [Google Scholar] [CrossRef]
- Jebali, A.; Hekmatimoghaddam, S.; Behzadi, A.; Rezapor, I.; Mohammadi, B.H.; Jasemizad, T.; Yasini, S.A.; Javadzadeh, M.; Amiri, A.; Soltani, M.; et al. Antimicrobial activity of nanocellulose conjugated with allicin and lysozyme. Cellulose 2013, 20, 2897–2907. [Google Scholar] [CrossRef]
- Malhotra, B.; Keshwani, A.; Kharkwal, H. Antimicrobial food packaging: Potential and pitfalls. Front. Microbiol. 2015, 6, 611. [Google Scholar] [CrossRef]
- Rollini, M.; Nielsen, T.; Musatti, A.; Limbo, S.; Piergiovanni, L.; Munoz, P.H.; Gavara, R. Antimicrobial performance of two different packaging materials on the microbiological quality of fresh salmon. Coatings 2016, 6, 6. [Google Scholar] [CrossRef]
- Lucera, A.; Costa, C.; Conte, A.; Del Nobile, M.A. Food applications of natural antimicrobial compounds. Front. Microbiol. 2012, 3, 287. [Google Scholar] [CrossRef]
- Li, H.; Pan, Y.; Li, C.; Yang, Z.; Rao, J.; Chen, B. Design, synthesis and characterization of lysozyme–gentisic acid dual-functional conjugates with antibacterial/antioxidant activities. Food Chem. 2022, 370, 131032. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Zhu, L.; Xi, L.; Guo, L.; Wang, H. An antimicrobial agent prepared by N-succinyl chitosan immobilized lysozyme and its application in strawberry preservation. Food Control 2020, 108, 106829. [Google Scholar] [CrossRef]
| Source of Lysozyme | Amount of Lysozyme |
|---|---|
| Tears | 3000–5000 µg/mL |
| Chicken egg white | 2500–3500 µg/mL |
| Duck egg white | 1000–1300 µg/mL |
| Goose egg white | 500–700 µg/mL |
| Human milk | 55-75 µg/mL |
| Cow milk | 10–15 µg/mL |
| Cauliflower juice | 25–28 µg/mL |
| Cabbage juice | 7–8 µg/mL |
| Papaya juice | 9 µg/mL |
| Spleen | 50–160 mg/kg |
| Thymus | 60–80 mg/kg |
| Pancreas | 20–35 mg/kg |
| Type of Lysozyme | Class | Organism | Type of Identification | References |
|---|---|---|---|---|
| c-type | Birds | Chicken | AA sequence | [21] |
| Mammals | Human | AA sequence | [28] | |
| Insects | Lepidoptera | cDNA isolation | [12] | |
| Diptera | cDNA isolation | [13] | ||
| Isoptera | cDNA isolation | [14] | ||
| Hemiptera | cDNA isolation | [15] | ||
| g-type | Birds | Goose | AA sequence | [29] |
| Cassowary | AA sequence | [22] | ||
| Rhea | AA sequence | [21] | ||
| Fish | Japanese flounder | cDNA isolation | [30] | |
| Atlantic cod | cDNA isolation | [31] | ||
| Mammals | Human | Similarity search with chicken lysozyme in databases | [32] | |
| Invertebrates | Mollusks | cDNA isolation | [23] | |
| Urochordates | cDNA isolation | [24] | ||
| i-type | Mollusks | Tapes japonica | cDNA isolation and AA sequence | [33] |
| Echinodermata | Sea cucumber | cDNA isolation | [34] |
| Modified Lysozyme | Properties | Reference |
|---|---|---|
| Palmitic acid | Antimicrobial activity against E. coli and Edwardsiella tarda. | [108] |
| Short and middle chain saturated fatty acids | Improve the bactericidal action | [99] |
| Dextran | Excellent in vitro antibacterial effect against S. aureus and E. coli. | [109] |
| Dextran | Preparation of a dextran-lysozyme conjugate for antibacterial effect against S. aureus and E. coli in a natural food system (cheese curd) | [105] |
| Dextran | Increased heat stability, better emulsion and higher solubility | [109] |
| Glactomannan | Antimicrobial activity against the Gram-negative pathogen E. tarda | [110] |
| Chitosan | Exhibits antimicrobial action towards E. coli K-12 | [111] |
| Chitosan | lysozyme-chitosan composite film activated against E. coli and Streptococcus faecalis | [112] |
| Xanthan gum | Used as a thickener, stabilizer, and an emulsifier in the food industry. Inhibited the growth of S. aureus and E. coli. | [104] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nawaz, N.; Wen, S.; Wang, F.; Nawaz, S.; Raza, J.; Iftikhar, M.; Usman, M. Lysozyme and Its Application as Antibacterial Agent in Food Industry. Molecules 2022, 27, 6305. https://doi.org/10.3390/molecules27196305
Nawaz N, Wen S, Wang F, Nawaz S, Raza J, Iftikhar M, Usman M. Lysozyme and Its Application as Antibacterial Agent in Food Industry. Molecules. 2022; 27(19):6305. https://doi.org/10.3390/molecules27196305
Chicago/Turabian StyleNawaz, Nida, Sai Wen, Fenghuan Wang, Shiza Nawaz, Junaid Raza, Maryam Iftikhar, and Muhammad Usman. 2022. "Lysozyme and Its Application as Antibacterial Agent in Food Industry" Molecules 27, no. 19: 6305. https://doi.org/10.3390/molecules27196305
APA StyleNawaz, N., Wen, S., Wang, F., Nawaz, S., Raza, J., Iftikhar, M., & Usman, M. (2022). Lysozyme and Its Application as Antibacterial Agent in Food Industry. Molecules, 27(19), 6305. https://doi.org/10.3390/molecules27196305







