Abstract
In this work, the hydrogen bonds and halogen bonds in the complexes between Y2CTe (Y = H, F, CH3) and XF (X = F, Cl, Br, I) have been studied by quantum chemical calculations. We found three interesting abnormalities regarding the interactions. Firstly, the strength of halogen bonds increases in the order of IF < BrF < ClF < F2. Secondly, the halogen bonds formed by F2 are very strong, with an interaction energy in the range between −199.8 and −233.1 kJ/mol. Thirdly, all the halogen bonds are stronger than the hydrogen bonds in the systems we examined. All these results are against the general understanding of halogen bonds. These apparent abnormal properties are reconciled with the high polarizability of the Te atom and the strong inducing effect of F on the Te atom of Y2CTe. These findings provide a new perspective on halogen bonds. Additionally, we also proposed bonding distance-based methods to compare the strength of halogen/hydrogen bonds formed between different donor atoms and the same acceptor atom.
1. Introduction
A halogen bond is formed between an electrophilic region of a halogen atom X (X = F, Cl, Br, and I) in a molecule R-X (R is an electron-withdrawing atom/group) and a nucleophilic region of a molecule Y-R’ [1], denoted as R-X∙∙∙Y-R’. The electrophilic region, or the electron-deficient region of the X atom, is located along the R-X σ-bond, denoted as σ-hole, which is surrounded by a belt of negative electrostatic potential [2]. Nowadays, halogen bonds have received extensive attention due to their important roles in many fields such as supramolecular chemistry, organocatalysis, synthetic coordination chemistry, polymer chemistry, and drug discovery [3,4,5,6,7,8,9,10,11,12,13]. For example, halogen bonding has been a popular and much exploited supramolecular synthon in the crystal field [5,9]. The application of halogen atoms as pharmaceutically active ligand substituents has been widespread in recent medicinal chemistry [10,11].
The properties of halogen bonds are related to their strength, which is not only dependent on the halogen donor atom and the acceptor atom, but is also affected by substituents. Normally, the halogen bond becomes stronger with the halogen donor varying from F to I [1,12,13,14,15,16]. An electron-donating group in the halogen bond acceptor strengthens the halogen bond, while an electron-withdrawing group in the acceptor has a weakening effect [16]. The type of the halogen bond acceptor varies from anions and neutral molecules with lone pairs to π-electron molecules, radicals, metal hydrides, and carbenes [17,18]. Specially, the molecules containing N and O atoms are often taken as the halogen bond acceptor.
It is interesting to study the differences between hydrogen bonds and halogen bonds, since both types of interactions have comparable strength and may coexist in the same systems [19,20,21,22,23,24,25,26,27]. Usually, hydrogen bonds are stronger than halogen bonds, except for when an iodine atom acts as the halogen donor [24,28]. Thus, some studies have tried to make halogen bonds stronger than hydrogen bonds [28,29,30]. When the halogen bond acceptor H2CO binds with the hydrogen/halogen donor HOBr, the interaction energy of the hydrogen bond is larger by 7 kJ/mol than that of the halogen bond [19]. Inversely, the interaction energy of the halogen bond is larger by 1 kJ/mol than that of the hydrogen bond if H2CO is changed to H2CS [28]. This difference is enlarged to 2 kJ/mol when one H atom of H2CS is replaced by a Li atom [28]. These results indicate that the differences between hydrogen bonds and halogen bonds can be regulated by changing the halogen bond acceptor atoms and/or adding substituents. Nevertheless, these comparisons are not very convincing, because, for HOBr to participate in hydrogen bonding and halogen bonding interactions, the remaining moieties are -OBr and -OH, respectively, meaning that they are not identical. To overcome this difficulty, Li and coauthors designed a molecule called 6-OX-fulvene (X = H, Cl, Br, I), where the moiety of fulvene increases the acidity of the X atom. Then, they examined the interactions between this molecule and ZH3/H2Y (Z = N, P, As, and Sb; Y = O, S, Se, and Te) [31]. It was found that the hydrogen bond is weakened with the Lewis base atom growing in size; however, the effect of the same on halogen bonds is very limited [31]. If SbH3 and H2Te are selected as the acceptors, the halogen bonds are much stronger than the hydrogen bonds, and the largest difference in their interaction energies is 40 kJ/mol in the SbH3∙∙∙6-OCl-fulvene complex [31].
H2CTe is a homologue of H2CO and H2CS; thus, it can also work as an acceptor to form hydrogen bonds or halogen bonds. Considering that Te is a semimetal located on the dividing line between metals and non-metals, we expect that the halogen bond formed by it may have different patterns. In this study, we investigated the complexes between Y2CTe (Y = H, F, and CH3) and XF (X = H, F, Cl, Br, and I), wherein XF is a hydrogen/halogen bond donor and Y2CTe is an acceptor. With the strong electronegativity of F, the designated molecules XF are expected to be prominent halogen bond donors. The following questions are addressed by the method of quantum chemical calculations: (1) Whether the halogen bond is stronger than the hydrogen bond. (2) Whether the strength of halogen bond follows the order of F2 < ClF < BrF < IF. (3) What is the nature of the hydrogen bond and halogen bond in these complexes?
2. Results
2.1. Molecular Electrostatic Potential (MEP) Analyses
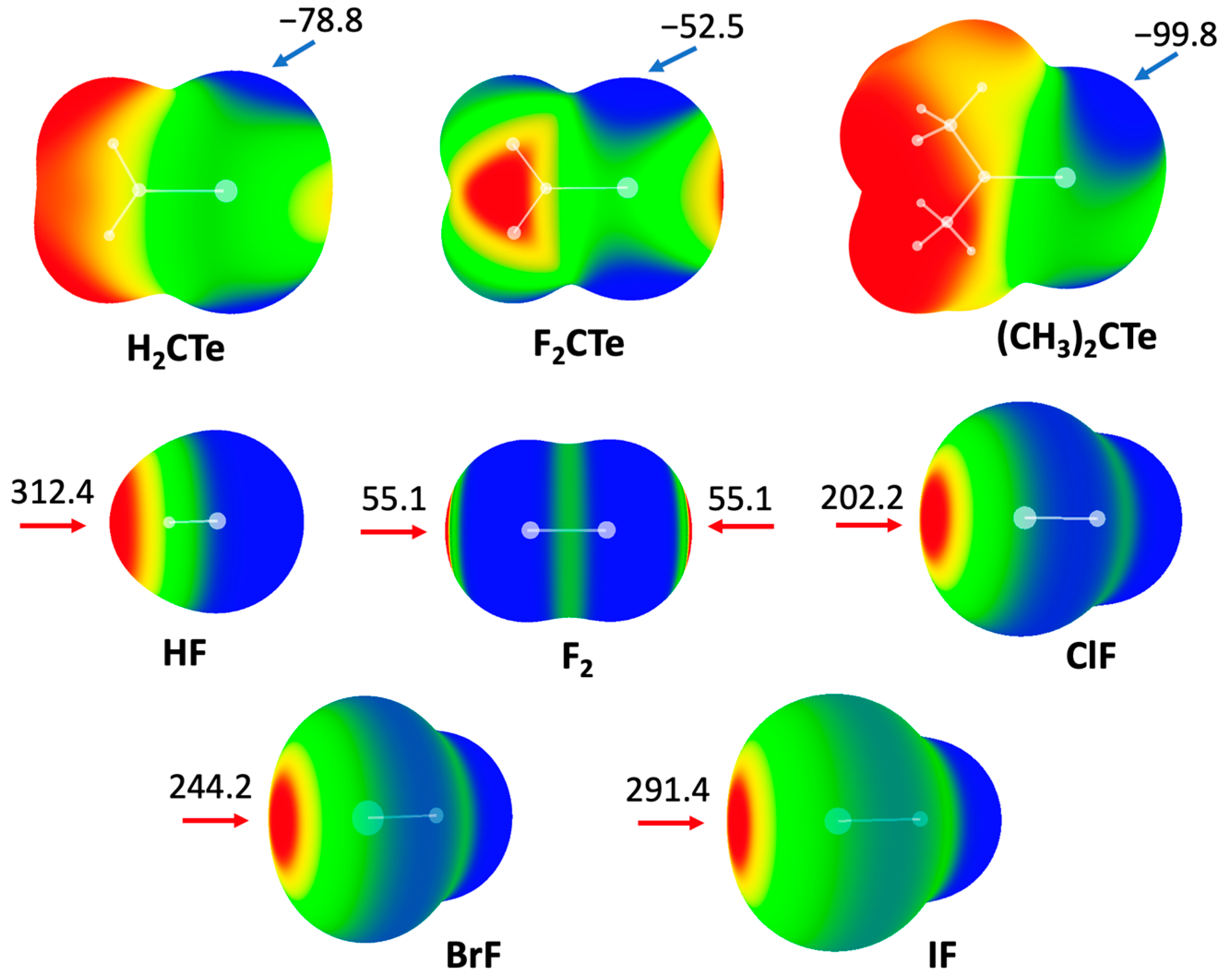
It is well known that the MEP diagram of a molecule is helpful to effectively predict noncovalent interactions involving that molecule [32]. Figure 1 shows the MEP maps of two families of molecules: Y2CTe (Y = F, H, and CH3) and XF (X = H, F, Cl, Br, and I). The MEP distributions in both families are anisotropic. For Y2CTe, we focus on the negative areas of the MEPs (blue colored areas). As expected, there are mainly two negative areas in each molecule, which correspond to the lone pairs of the Te atom. Compared with H2CTe (−78.8 kJ/mol), the minimal MEP value of the Te atom decreases in F2CTe (−52.5 kJ/mol) but increases in (CH3)2CTe (−99.8 kJ/mol), which can be attributed to the electron-withdrawing nature of F atoms and the electron-donating ability of the methyl groups, respectively.
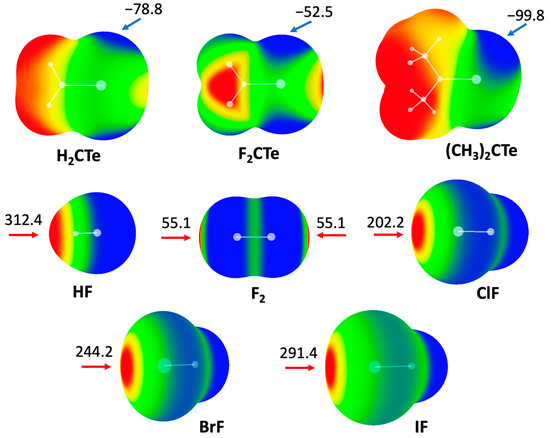
Figure 1.
MEP diagrams of molecules studied in this work. Color ranges, in kJ/mol: red, greater than 52.5; yellow, between 52.5 and 0; green, between 0 and −52.5; blue, less than −52.5. Arrows refer to values of maxima and minima.
For XF, we focus on the positive areas of the MEPs (red areas). In the case of HF, the atom H exhibits positive electrostatic potential, while F is negative. For dihalogen molecules XF, there is a positive MEP region (σ-hole) at the X atom along the X-F bond. The magnitude of the σ-hole on the halogen atom increases with an increasing atomic mass of X. It is also found that the maximal MEP on the H atom is larger than that on the halogen atoms, including iodine.
2.2. Geometries
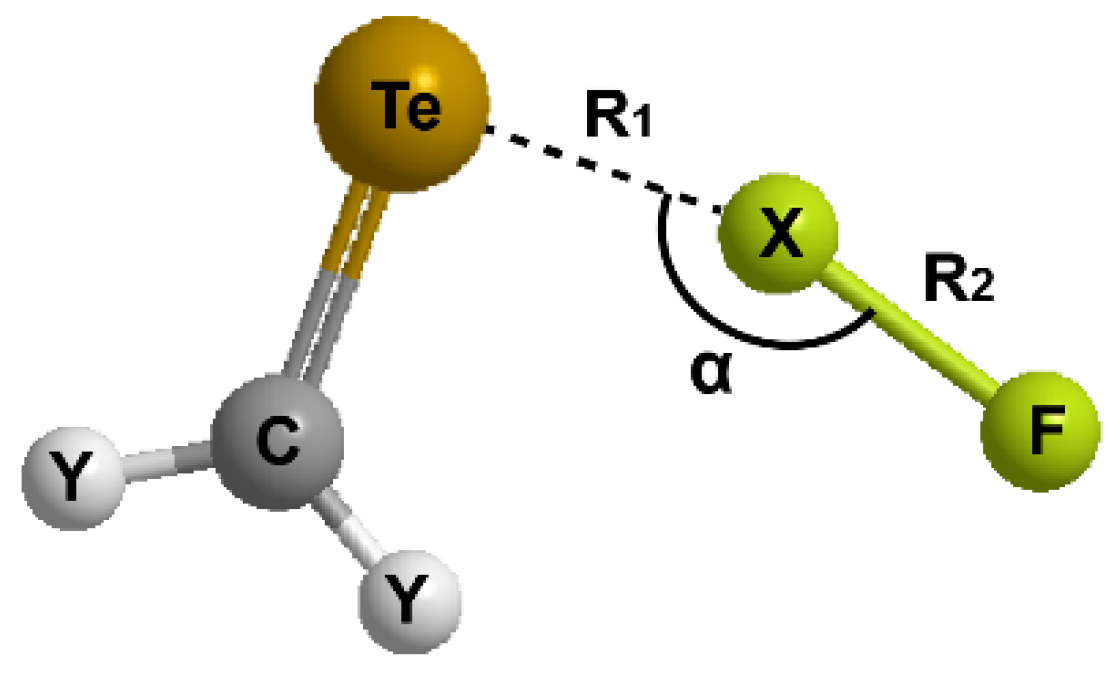
For the hydrogen bonding or halogen bonding interactions (Y2CTe∙∙∙XF), the general geometry of the complexes and the involved parameters are shown in Figure 2. We focus on the Te∙∙∙X distance (R1), the change in the X-F bond length (∆R2), and the Te∙∙∙X-F angle (α). The data of the optimized structures are listed in Table 1.
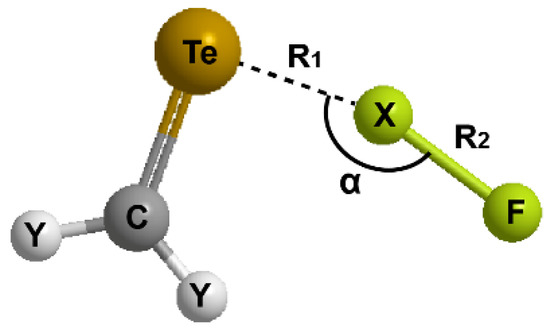
Figure 2.
Illustration of the general structure of Y2CTe∙∙∙XF complex.

Table 1.
Binding distance (R1, Å), ΔR1%; elongation of the X-F bond length (ΔR2, Å), ΔR2%; bond angle (α, deg) of the complexes.
As can be seen in the table, all the values of R1 are much shorter than the sum of the van der Waals radii of the respective atoms (3.3 Å for Te and H, 3.6 Å for Te and F, 4.0 Å for Te and Cl, 4.2 Å for Te and Br, and 4.4 Å for Te and I) [33,34]. This justifies the formation of hydrogen/halogen bonds. Further, the interactions between the electron-donor and acceptor molecules seem to be quite strong because stronger interaction is known to result in shorter bond length (R1). To compare the relative strength between the halogen bonds between different interaction partners and the hydrogen bond, we define a quantity ΔR1% in the following equation:
where Rc is the sum of the van der Waals radii of the two atoms representing the critical distance to judge the presence of a hydrogen/halogen bond. After normalization with Rc, the shortening of the Te∙∙∙X distance could be used to evaluate the strength of hydrogen/halogen bonds. Thus, for each of the three molecules (H2CTe, F2CTe, and (CH3)2CTe), the ΔR1% are all in the sequential order F2 > ClF > BrF > IF > HF when they form interaction pairs. This implies that all the halogen bonds are stronger than the hydrogen bonds. Most interestingly, the ΔR1% values suggest that the halogen bond strength decreases with an increasing size of the halogen atom in the donor molecule XF. This is different from the general understanding of halogen bonds.
The change in the X-F bond length R2 of a donor could also reflect the interaction strength of the hydrogen/halogen bond. Here, we calculated the change of R2 relative to the R2 in the monomer, denoted as ΔR2%, using the following formula:
The ΔR2% represents the elongation percentage of the X-F bond, and the larger ΔR2% implies more significant weakening of the bond and, thus, stronger interaction. As indicated by ΔR2%, the value of X-F bond length is larger in the halogen-bonded complex than that in the hydrogen-bonded analogue. This relative elongation in the halogen-bonded complex decreases in the order of F2 > ClF > BrF > IF. These data are supportive of the conclusions from ΔR1%.
The Te∙∙∙X-F angle (α) is in the range of 168–180°, confirming a good direction of the hydrogen/halogen bonds. The angles are less than 180° in the majority of the complexs due to the attraction between the Y atom/group in Y2CTe and XF.
2.3. Energies
Here, we consider the interaction energy to be the most credible criteria to judge the strength of interactions. Therefore, we calculated the interaction energies (Eint) of the various complexes for comparing the hydrogen and the halogen bonds. We used the counterpoise correction method to eliminate the basis set superposition error (BSSE), and the corrected energy is denoted as Eint,BSSE. In addition, the more accurate energy Eint,CBS,BSSE with complete basis set (CBS) was also calculated. The results with and without BSSE correction, as well as with CBS, are all listed in Table 2. The main concern of our study is that the changing trends of the interaction energy with the variation of X in XF are the same based on all the three methods. It is worth clarifying that the following discussions about energies in the full text are all according to their absolute values. As shown in Table 2, the interaction energies of hydrogen bonds in all of the three series of complexes are smaller than those of the halogen bonds, indicating that the hydrogen bonds are weaker than all of the halogen bonds. For the strength order of the halogen bonds, both Eint,BSSE and Eint,CBS,BSSE increased in the order of IF < BrF < ClF < F2 for the series of H2CTe∙∙∙XF and (CH3)2CTe∙∙∙XF complexes. This result is abnormal compared to the common perception that the halogen bond becomes stronger with the halogen donor varying from F to I. For F2CTe∙∙∙XF complexes, the Eint,BSSE/Eint,CBS,BSSE of ClF, BrF, and IF was close. For all of the three series of the complexes, F2 molecules formed the strongest halogen bonds. The absolute values of the interaction energy were very large, up to 228.8 kJ/mol for Eint,BSSE and 233.1 kJ/mol for Eint,CBS,BSSE. To compare the interaction energies of the halogen bonds formed by different acceptors (Y2CTe), when Y is F, an electron-withdrawing atom, the Y2CTe∙∙∙XF interaction, was weakened and compared to that of H2CTe. On the contrary, when Y was the electron-donating methyl group, the interaction was strengthened.

Table 2.
Interaction energies (Eint) corrected with and without BSSE in the complexes at the MP2/aug-cc-pVTZ(PP) level, all in kJ/mol.
To understand the attribution of the interaction energy, we partitioned it into five terms: electrostatic energy (Ees), exchange energy (Eex), repulsion energy (Erep), polarization energy (Epol), and dispersion energy (Edisp), and the data are listed in Table 3. Obviously, Eex is the largest attractive term in each complex; thus, it plays the most important role in the stabilization of hydrogen/halogen bonds [35,36]. This term increases in the order of HF < IF < BrF < ClF < F2, which is consistent with the results of orbital interaction discussed in the following section. The large Eex of each complex suggests a strong orbital interaction between the two respective monomers. For the repulsive term, Erep was very large and even exceeds 1000 kJ/mol in each F2-related complex. This can be attributed to the much shorter Te∙∙∙X distances. It is seen that Erep was almost twice as much as Eex and both terms have a good linear relationship (Figure S1), confirming their dependency each other.

Table 3.
Electrostatic (Ees), exchange energy (Eex), repulsion energy (Erep), polarization (Epol), and dispersion (Edisp) energies in the complexes, all in kJ/mol.
Now, we examine the three attractive terms (Ees, Epol, and Edisp) in Table 3 in some detail (intuitively in Figure S2). For the hydrogen bond complex Y2CTe∙∙∙HF, Ees was the largest attractive interaction among the three terms, followed by Epol. For the interaction energies of halogen bonds formed by ClF, BrF, or IF with all the three acceptors Y2CTe, the contributions of electrostatic and polarization interactions are comparable. However, for the interaction energies of the halogen bonds formed by F2 with the Y2CTe, the polarization interaction is the dominating contribution. This may be attributed to the special property of F, namely the largest electronegativity in the periodic table, thus possessing a very strong inducing ability.
2.4. Atoms in Molecules (AIM) Analyses
The hydrogen/halogen bonds can be characterized by the Te∙∙∙X bond critical points (BCPs, Figure S3). The most important properties of each bond critical point are summarized in Table 4, where ρ refers to the electron density, ∇2ρ its Laplacian, and H the energy density [37,38,39]. Generally, the larger electron density ρ reflects the stronger interaction. For all the investigated systems, ρ increases in the sequential order of HF < IF < BrF < ClF < F2, in agreement with the order of interaction energies (Table 2). For the Laplacian, it was seen that ∇2ρ > 0 for all the complexes, demonstrating that the interactions studied were closed shell interaction. The energy density H is a more sensitive parameter than ∇2ρ. The negative values of H further demonstrate that the interactions are partially covalent in nature. In the complexes involving (CH3)2CTe, there are also BCPs between the methyl H and the halogen X in HX or XFs (Figure S3), indicating the presence of C-H∙∙∙F/X hydrogen bonds. The interaction energies of the C-H∙∙∙F/X hydrogen bonds were estimated by E = 0.5V(r) [40,41], where V(r) is the potential energy density at a BCP in each case. The corresponding data are −5.6, −17.8, −10.3, −9.9, and −8.1 kJ/mol for (CH3)2CTe∙∙∙HF, (CH3)2CTe∙∙∙F2, (CH3)2CTe∙∙∙ClF, (CH3)2CTe∙∙∙BrF, and (CH3)2CTe∙∙∙IF, respectively. Clearly, these hydrogen bonds contributed to the stability of the complexes; however, their shares in the total interaction energies (Table 2) are small. Subtracting them from the total interaction energies, the residual results still have the same change trend with the total interaction energy. Thus, the presence of C-H∙∙∙F/X hydrogen bonds does not affect the abnormality of halogen bonds.

Table 4.
Electron density (ρ), Laplacian (∇2ρ), and total energy density (H) at the intermolecular BCP in the complexes (all values in a.u.).
2.5. Natural Bond Orbital (NBO) Analyses
The charge transfers (CTs) from Y2CTe to XF are listed in Table 5, which are calculated as the sum of the charge on all atoms in Y2CTe in the complexes. The charge transfer reflects the electrons bias from electron donor (Y2CTe) to electron acceptor (XF), providing information about the interaction strength in one aspect. As can be seen, the CTs are all larger than 0.2 e in the halogen bonds and even exceed 0.8 e in the F2 complexes. On the contrary, the hydrogen-bonded complexes have much smaller CTs than the halogen-bonded analogues. Additionally, for all three of the series of complexes, the CT value decreases in the order F2 > ClF > BrF > IF > HF, which is the same as the interaction strength order. Besides, when the Y is the electron-withdrawing atom F in Y2CTe, the CT becomes smaller than that in H2CTe. When the Y is the electron-donating methyl group, the CT increases.

Table 5.
Charge transfer (CT, e), second-order perturbation energy (E2, kJ/mol), and dipole moment (μ, D) in the complexes.
There is an orbital interaction between the lone pair orbital on the Te atom of Y2CTe and the anti-bonding orbital of the X-F bond (LpTe→σ*x-F), and this orbital interaction can be measured with the second-order perturbation energy (E2), which is also listed in Table 5. This orbital interaction is not detected in the F2-containing complexes since the F-F bond is taken as two subunits in the NBO analysis. The E2 has a consistent change order with the charge transfer. This orbital interaction is strong; therefore, it makes an important contribution to the formation of hydrogen/halogen bond. We also calculated the dipole moments of the complexes (Table 5). It was found that the order of the dipole moment is consistent with the interaction energy. Further, the relationship between the CTs and the population of the σ*x-F orbital was analyzed, and positive correlation was found (Figure S4). This suggests that the main destination/receiver of the CT is the σ*x-F orbital in each complex.
3. Discussion
Hydrogen bonds and halogen bonds are two important noncovalent interactions, and they often coexist; thus, it is interesting to compare their strength. Generally speaking, hydrogen bonds are considered to be stronger than halogen bonds [28]. Interestingly, in the present study, using Y2CTe (Y = H, F, and CH3) as electron donors, we found that their halogen bonding interactions with dihalogen molecules XF (X = F, Cl, Br, and I) are stronger than their hydrogen bonding interactions with HF. This apparent abnormality was also seen in a previous study on the interactions between PH3/AsH3/H2Te and 6-OX-fulvene (X = H, Cl, Br, I) [31]. The second abnormality found in this work was that, when the X changes from F to I, the halogen bond becomes weaker, in contrast to the normal understanding that stronger halogen bonds accompany heavier halogen donors [16]. The abnormalities can be explained by the high polarizability of the Te atom, which is easily polarized when the electronegative XF approaches it. The greater the electronegativity of the approaching atom, the greater the polarization of the Te atom. Therefore, when the X atom of XF varies from I to F, the dipole moment of the complex increases, as seen in Table 5, and the polarization energy (the major contribution to the interaction energy) also increases. Based on the data in Table 3 and Table 5, a near positive correlation between the polarization energy and the dipole moment of the complex is found (Figure S5).
Another surprising result was that F2 participates in the strongest halogen bond with the interaction energy up to −233.1 kJ/mol in the (CH3)2CTe∙∙∙F2 complex. Such large interaction energy is abnormal because it shows the least MEP at the end of the X atom among the four XF molecules. The apparent contradiction can be reconciled as follows. The F atom of F2, due to it having the highest electronegativity among the halogens, would cause the largest polarization on the Te atom and, thus, the largest dipole moment of the Y2CTe∙∙∙XF complex. This is evidenced by the largest polarization energies being seen in the three Y2CTe∙∙∙F2 complexes. The polarization mechanism is also consistent with the charge transfer data, which are the biggest in the Y2CTe∙∙∙F2 complexes, even as big as 0.943e in the complex of (CH3)2CTe∙∙∙F2. Such big charge transfers (>0.8e) mean that the molecule F2 holds nearly an extra electron in each of the three complexes, similar to the process of becoming an anion.
In order to test if the above abnormal results are found only for Y2CTe, the Te atom of H2CTe was replaced by O, S, and Se. The corresponding interaction energies are listed in Table 6. For the lighter chalcogen atoms O and S, the halogen bond strength increases in the order of F2 < ClF < BrF < IF, which is the “normal” understanding of halogen bonds. For the heavier Se as the electron donor, the halogen bonds formed by ClF, BrF, and IF turned out to be comparable, while that formed by F2 increased tremendously. This complicated situation is explained as follows. On one hand, a Se atom with a larger radius is more easily polarized than O and S. On the other hand, it is not as easily polarized as Te. Thus, only the most electronegative F2 is able to assert marked influence on the electron distribution of H2CSe, making it the strongest interaction in the H2CSe∙∙∙XF complexes.

Table 6.
Interaction energies (Eint,BSSE, kJ/mol) of complexes between H2CZ (Z = O, S, Se, and Te) and XF (X = H, F, Cl, Br, and I) at the MP2/aug-cc-pVTZ(PP) level.
As discussed above, the size/polarizability of the chalcogen atoms in H2CZ (Z = O, S, Se, and Te) plays a very important role in the strength of halogen bonds. The data in Table 6 demonstrate that, for a given XF (X = F, Cl, Br, and I), the strength of its halogen bond with H2CZ increases monotonously when Z varies from O to Te. On the contrary, the strength of the hydrogen bond between HF and H2CZ decreases monotonously. As a result, for H2CO, only IF forms a stronger halogen bond than the hydrogen bond. For H2CS, each dihalogen molecule, excluding F2, participates in a stronger halogen bond than the hydrogen bond formed by HF. For H2CSe and H2CTe, all the dihalogen molecules form stronger halogen bonds than hydrogen bonds formed by HF.
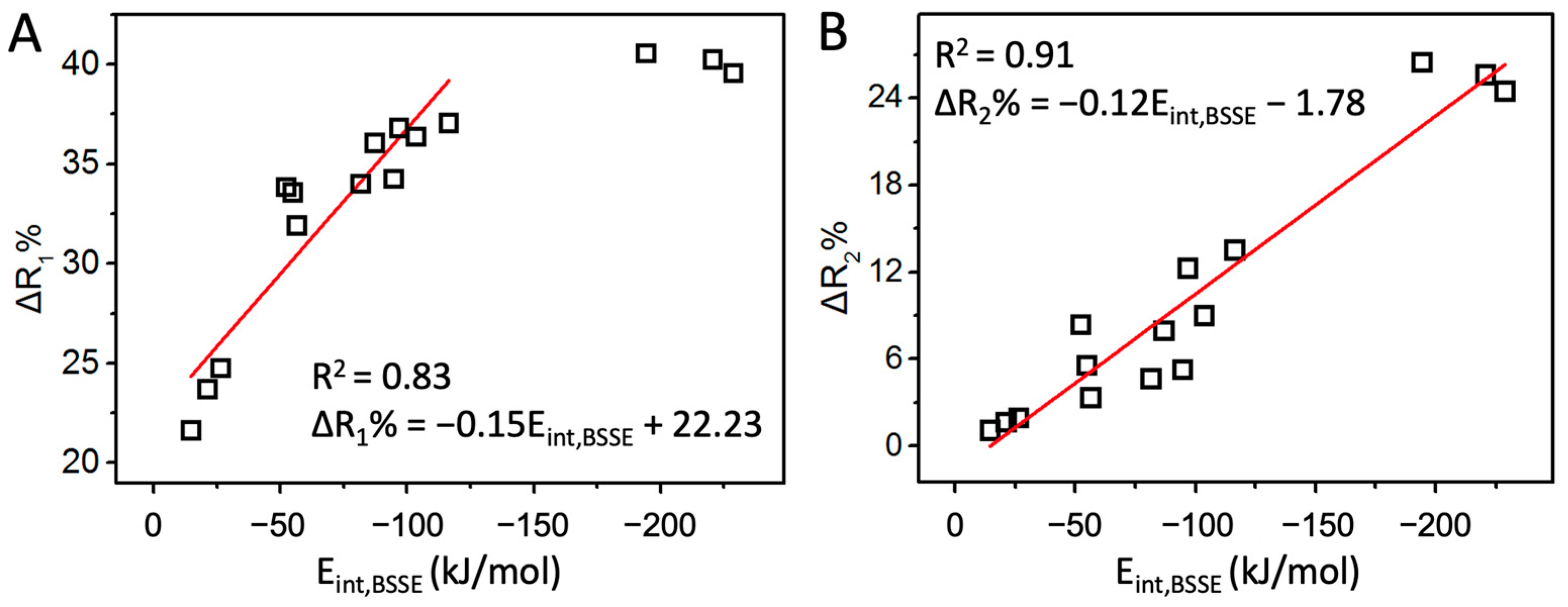
The above discussions about the interaction strength of halogen/hydrogen bonds are based on their interaction energies, often regarded as golden criteria. Practically, other parameters such as electrostatic potential (MEP), halogen/hydrogen bond length (R1), and donor bond length (R2) are often used to compare the interaction strength. MEP sometimes provides correct indications. For example, the MEP order is Cl < Br < I at the end of X atom in XF, and the halogen bond strength order is ClF < BrF < IF in the F2CTe∙∙∙XF complexes. However, the F has the least MEP at the end of the X atom among the four XF molecules, but it forms the strongest interaction with F2CTe. Clearly, MEP cannot always provide correct results because it only contains the information of isolated molecules. For R1 and R2, due to the different radii of the halogen atoms, we defined two quantities, ΔR1% and ΔR2%, to compare the halogen/hydrogen bond strength. The correlations between the two quantities and the respective interaction energies are plotted in Figure 3 (the trend comparisons are shown in Figure S6). Undoubtedly, they are all positively correlated, suggesting that both ΔR1% and ΔR2% can be used to compare the halogen/hydrogen bond strength qualitatively. Quantitatively, ΔR2% is a better choice.
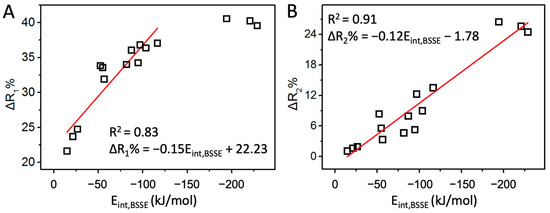
Figure 3.
The correlation of Eint,BSSE with ΔR1% (A) and ΔR2% (B).
4. Computational Methods
All calculations were carried out with the Gaussian 09 program [42]. The geometries of all the monomers and complexes were optimized at the MP2 level with the aug-cc-pVTZ basis set for all atoms except I and Te, where the aug-cc-pVTZ-PP basis set was adopted to account for relativistic corrections [43]. For all atoms in all the complexes, collectively, aug-cc-pVTZ(PP) represents the basis set used in this work. The extrema of molecular electrostatic potentials (MEPs) were calculated on the 0.001 a.u. isodensity surface at the MP2/aug-cc-pVTZ(PP) level using the WFA-SAS program [32]. The interaction energy (Eint) of a complex was computed as the difference between the energy of the complex and the sum of the energies of the monomers with their geometries frozen in the optimized complex. For this supermolecular method of calculating the MP2 interaction energy, the dispersion correction was taken into account, since MP2 contains certain correlation terms such as the uncoupled Hartree-Fock (UCHF) dispersion energy, the corresponding Hartree-Fock exchange-dispersion energy, and a deformation-correlation term [44]. Interaction energies were corrected for basis set superposition error (BSSE) using the counterpoise procedure (CP) proposed by Boys and Bernardi [45]. The two-point extrapolated energies with a complete basis set (CBS) proposed by Halkier et al. [46,47] were obtained with two basis sets of aug-cc-pVDZ(PP) and aug-cc-pVTZ(PP). The localized molecular orbital-energy decomposition analysis was used to decompose the interaction energy into five terms of electrostatic, exchange, repulsion, polarization, and dispersion at the MP2/aug-cc-pVTZ(PP) level with the GAMESS program [48]. The dispersion energy was obtained as a difference between the MP2 and CCSD(T) energy in the GAMESS program. The AIM2000 package [49] was used to assess the topological parameters at each bond critical point (BCP), including electron density, as well as its Laplacian, and energy density. Using the natural bond orbital (NBO) method [50] within the Gaussian 09 program, the charge transfer and second-order perturbation energy were obtained.
5. Conclusions
Quantum chemical calculations have been performed to study the interactions between Y2CTe (Y = H, F, and CH3) and XF (X = H, F, Cl, Br, and I). The results show that the electron-withdrawing groups F in F2CTe weaken the interactions, while the electron-donating methyl groups in (CH3)2CTe strengthen them. More importantly, we found three abnormalities regarding halogen bonds in this work. The first one is that the strength of halogen bond increases in the sequential order IF < BrF < ClF < F2 in H2CTe∙∙∙XF and (CH3)2CTe∙∙∙XF complexes. This is contrary to the normal understanding that the stronger halogen bonds accompany heavier halogen donors. The second one is that the halogen bonds formed by F2 are very strong, even up to −233.1 kJ/mol with (CH3)2CTe. Contrary to this, the halogen bonds formed by F-R are normally considered to be very weak or even negligible. The last one is that all halogen bonds are stronger than the hydrogen bonds in the complexes, which is abnormal compared with the majority of previous studies. These abnormalities are discussed in the context of the high polarizability of the Te atom in the halogen acceptors. Because the Te atom is easily polarized when the electronegative XF approaches it, the greater the electronegativity of the X atom, the greater the polarization of the Te atom. Particularly, the F atom has the largest electronegativity in the periodic table and possesses a very strong inducing ability. Consequently, F-F forms tremendously strong interactions with Y2CTe.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules27238523/s1, Figure S1: The relationship between repulsion energy Erep and exchange energy Eex in the complexes between Y2CTe (Y = H, F, and CH3) and XF (X = H, F, Cl, Br, and I); Figure S2: Electrostatic (Ees), polarization (Epol), and dispersion (Edisp) energies in complexes between Y2CTe (Y = H, F, and CH3) and XF (X = H, F, Cl, Br, and I); Figure S3: Molecular graphs of the complexes between Y2CTe (Y = H, F, CH3) and XF (X = H, F, Cl, Br, I). Small red balls indicate the Te∙∙∙X bond critical point; Figure S4: The relationship between the population of the σ*x-F orbitals and the charge transfer in the complexes formed by Y2CTe (Y = H, F, and CH3) and XF (X = H, F, Cl, Br, and I); Figure S5: The relationship between the polarization energy Epol and the dipole moment of the complexes formed by Y2CTe (Y = H, F, and CH3) and XF (X = F, Cl, Br, and I); Figure S6: Trend comparison of Eint,BSSE with ΔR1% (A) and ΔR2% (B) in H2CTe∙∙∙XF systems; The coordinates of optimized monomer Y2CTe (Y = H, F, CH3), XF (X = H, F, Cl, Br, I), and their complexes.
Author Contributions
Data curation, Y.-Q.W. and R.-J.W.; Formal analysis, Y.-Q.W. and Q.-Z.L.; Supervision, Q.-Z.L. and Z.-W.Y.; Writing—original draft, Y.-Q.W. and Q.-Z.L.; Writing—review & editing, Z.-W.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 21733011 and 22233006).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The author declares no conflict of interest.
Sample Availability
Samples of the compounds are not available from the authors.
References
- Politzer, P.; Murray, J.S. Halogen Bonding: An Interim Discussion. ChemPhysChem 2013, 14, 278–294. [Google Scholar] [CrossRef] [PubMed]
- Clark, T.; Hennemann, M.; Murray, J.S.; Politzer, P. Halogen Bonding: The Sigma-Hole. J. Mol. Model. 2007, 13, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Gilday, L.C.; Robinson, S.W.; Barendt, T.A.; Langton, M.J.; Mullaney, B.R.; Beer, P.D. Halogen Bonding in Supramolecular Chemistry. Chem. Rev. 2015, 115, 7118–7195. [Google Scholar] [CrossRef]
- Bertani, R.; Metrangolo, P.; Moiana, A.; Perez, E.; Pilati, T.; Resnati, G.; Rico-Lattes, I.; Sassi, A. Supramolecular Route to Fluorinated Coatings: Self-Assembly Between Poly(4-vinylpyridines) and Haloperfluorocarbons. Adv. Mater. 2002, 14, 1197–1201. [Google Scholar] [CrossRef]
- Fourmigue, M.; Batail, P. Activation of Hydrogen- and Halogen-Bonding Interactions in Tetrathiafulvalene-Based Crystalline Molecular Conductors. Chem. Rev. 2004, 104, 5379–5418. [Google Scholar] [CrossRef]
- Jungbauer, S.H.; Walter, S.M.; Schindler, S.; Rout, L.; Kniep, F.; Huber, S.M. Activation of a Carbonyl Compound by Halogen Bonding. Chem. Commun. 2014, 50, 6281–6284. [Google Scholar] [CrossRef] [PubMed]
- Libri, S.; Jasim, N.A.; Perutz, R.N.; Brammer, L. Metal Fluorides Form Strong Hydrogen Bonds and Halogen Bonds: Measuring Interaction Enthalpies and Entropies in Solution. J. Am. Chem. Soc. 2008, 130, 7842–7844. [Google Scholar] [CrossRef]
- Mele, A.; Metrangolo, P.; Neukirch, H.; Pilati, T.; Resnati, G. A Halogen-Bonding-Based Heteroditopic Receptor for Alkali Metal Halides. J. Am. Chem. Soc. 2005, 127, 14972–14973. [Google Scholar] [CrossRef]
- Adler, M.; Kochanny, M.J.; Ye, B.; Rumennik, G.; Light, D.R.; Biancalana, S.; Whitlow, M. Crystal Structures of Two Potent Nonamidine Inhibitors Bound to Factor Xa. Biochemistry 2002, 41, 15514–15523. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, Y.; Zhu, W. Nonbonding Interactions of Organic Halogens in Biological Systems: Implications for Drug Discovery and Biomolecular Design. Phys. Chem. Chem. Phys. 2010, 12, 4543–4551. [Google Scholar] [CrossRef]
- Matter, H.; Nazaré, M.; Güssregen, S.; Will, D.W.; Schreuder, H.; Bauer, A.; Urmann, M.; Ritter, K.; Wagner, M.; Wehner, V. Evidence for C−Cl/C−Br∙∙∙π Interactions as an Important Contribution to Protein–Ligand Binding Affinity. Angew. Chem. Int. Ed. 2009, 48, 2911–2916. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. The Halogen Bond. Chem. Rev. 2016, 116, 2478–2601. [Google Scholar] [PubMed]
- Erdelyi, M. Halogen Bonding in Solution. Chem. Soc. Rev. 2012, 41, 3547–3557. [Google Scholar] [CrossRef] [PubMed]
- Dipaolo, T.; Sandorfy, C. On the Biological Importance of the Hydrogen Bond Breaking Potency of Fluorocarbons. Chem. Phys. Lett. 1974, 26, 466–473. [Google Scholar] [CrossRef]
- Lu, Y.; Li, H.; Zhu, X.; Zhu, W.; Liu, H. How Does Halogen Bonding Behave in Solution? A Theoretical Study Using Implicit Solvation Model. J. Phys. Chem. A 2011, 115, 4467–4475. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Clark, T. Halogen bonding: An Electrostatically-Driven Highly Directional Noncovalent Interaction. Phys. Chem. Chem. Phys. 2010, 12, 7748–7757. [Google Scholar] [CrossRef]
- Fourmigue, M. Halogen Bonding: Recent Advances. Curr. Opin. Solid. St. M. 2009, 13, 36–45. [Google Scholar] [CrossRef]
- Tepper, R.; Schubert, U.S. Halogen Bonding in Solution: Anion Recognition, Templated Self-Assembly, and Organocatalysis. Angew. Chem. Int. Ed. 2018, 57, 6004–6016. [Google Scholar] [CrossRef]
- Li, Q.Z.; Xu, X.S.; Liu, T.; Jing, B.; Li, W.Z.; Cheng, J.B.; Gong, B.A.; Sun, J.Z. Competition Between Hydrogen Bond and Halogen bond in Complexes of Formaldehyde with Hypohalous Acids. Phys. Chem. Chem. Phys. 2010, 12, 6837–6843. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Lane, P. σ-Hole Bonding and Hydrogen Bonding: Competitive Interactions. Int. J. Quantum Chem. 2007, 107, 3046–3052. [Google Scholar] [CrossRef]
- Aakeroy, C.B.; Fasulo, M.; Schultheiss, N.; Desper, J.; Moore, C. Structural Competition between Hydrogen Bonds and Halogen Bonds. J. Am. Chem. Soc. 2007, 129, 13772–13773. [Google Scholar] [CrossRef] [PubMed]
- Nagels, N.; Geboes, Y.; Pinter, B.; De Proft, F.; Herrebout, W.A. Tuning the Halogen/Hydrogen Bond Competition: A Spectroscopic and Conceptual DFT Study of Some Model Complexes Involving CHF2I. Chem. Eur. J. 2014, 20, 8433–8443. [Google Scholar] [CrossRef] [PubMed]
- An, X.L.; Yang, X.; Xiao, B.; Cheng, J.B.; Li, Q.Z. Comparison of Hydrogen and Halogen Bonds Between Dimethyl Sulfoxide and Hypohalous Acid: Competition and Cooperativity. Mol. Phys. 2017, 115, 1614–1623. [Google Scholar] [CrossRef]
- An, X.L.; Zhuo, H.Y.; Wang, Y.Y.; Li, Q.Z. Competition Between Hydrogen Bonds and Halogen Bonds in Complexes of Formamidine and Hypohalous Acids. J. Mol. Model. 2013, 19, 4529–4535. [Google Scholar] [CrossRef] [PubMed]
- Geboes, Y.; De Proft, F.; Herrebout, W.A. Towards a Better Understanding of the Parameters Determining the Competition Between Bromine Halogen Bonding and Hydrogen Bonding: An FTIR Spectroscopic Study of the Complexes Between Bromodifluoromethane and Trimethylamine. J. Mol. Struct. 2018, 1165, 349–355. [Google Scholar] [CrossRef]
- Zheng, Y.Z.; Deng, G.; Zhou, Y.; Sun, H.Y.; Yu, Z.W. Comparative Study of Halogen- and Hydrogen-Bond Interactions between Benzene Derivatives and Dimethyl Sulfoxide. ChemPhysChem 2015, 16, 2594–2601. [Google Scholar] [CrossRef]
- Zheng, Y.Z.; Wang, N.N.; Zhou, Y.; Yu, Z.W. Halogen-Bond and Hydrogen-Bond Interactions between Three Benzene Derivatives and Dimethyl Sulphoxide. Phys. Chem. Chem. Phys. 2014, 16, 6946–6956. [Google Scholar] [CrossRef]
- Li, Q.Z.; Jing, B.; Li, R.; Liu, Z.B.; Li, W.Z.; Luan, F.; Cheng, J.B.; Gong, B.A.; Sun, J.Z. Some Measures for Making Halogen Bonds Stronger than Hydrogen Bonds in H2CS-HOX (X = F, Cl, and Br) Complexes. Phys. Chem. Chem. Phys. 2011, 13, 2266–2271. [Google Scholar] [CrossRef]
- Lv, H.; Zhuo, H.Y.; Li, Q.Z.; Yang, X.; Li, W.Z.; Cheng, J.B. Halogen Bonds with N-heterocyclic Carbenes as Halogen Acceptors: A Partially Covalent Character. Mol. Phys. 2015, 112, 3024–3032. [Google Scholar] [CrossRef]
- Zhuo, H.Y.; Yu, H.; Li, Q.Z.; Li, W.Z.; Cheng, J.B. Some Measures for Mediating the Strengths of Halogen Bonds with the B-B Bond in Diborane(4) as an Unconventional Halogen Acceptor. Int. J. Quantum Chem. 2014, 114, 128–137. [Google Scholar] [CrossRef]
- Hou, M.C.; Li, Q.Z.; Scheiner, S. Comparison between Hydrogen and Halogen Bonds in Complexes of 6-OX-Fulvene with Pnicogen and Chalcogen Electron Donors. ChemPhysChem 2019, 20, 1978–1984. [Google Scholar] [CrossRef] [PubMed]
- Bulat, F.A.; Toro-Labbe, A.; Brinck, T.; Murray, J.S.; Politzer, P. Quantitative Analysis of Molecular Surfaces: Areas, Volumes, Electrostatic Potentials and Average Local Ionization Energies. J. Mol. Model. 2010, 16, 1679–1691. [Google Scholar] [CrossRef]
- Pauling, L. The Nature of the Chemical Bond; Cornell University Press: Ithaca, NY, USA, 1960. [Google Scholar]
- Pauling, L.; Pauling, P. Chemistry; W. H. Freeman Company: San Francisco, CA, USA, 1975. [Google Scholar]
- Miranda, M.O.; Duarte, D.J.R. Halogen Bonds Stabilised by an Electronic Exchange Channel. ChemistrySelect 2021, 6, 680–684. [Google Scholar] [CrossRef]
- Duarte, D.J.R.; Buralli, G.J.; Peruchena, N.M. Is σ-Hole an Electronic Exchange Channel in YX⋯CO Interactions? Chem. Phys. Lett. 2018, 710, 113–117. [Google Scholar] [CrossRef]
- Koch, U.; Popelier, P.L.A. Characterization of C-H-O Hydrogen Bonds on the Basis of the Charge Density. J. Phys. Chem. 1995, 99, 9747–9754. [Google Scholar] [CrossRef]
- Popelier, P.L.A. Characterization of a Dihydrogen Bond on the Basis of the Electron Density. J. Phys. Chem. A 1998, 102, 1873–1878. [Google Scholar] [CrossRef]
- Arnold, W.D.; Oldfield, E. The Chemical Nature of Hydrogen Bonding in Proteins via NMR: J-Couplings, Chemical Shifts, and AIM Theory. J. Am. Chem. Soc. 2000, 122, 12835–12841. [Google Scholar] [CrossRef]
- Espinosa, E.; Molins, E.; Lecomte, C. Hydrogen Bond Strengths Revealed by Topological Analyses of Experimentally Observed Electron Densities. Chem. Phys. Lett. 1998, 285, 170–173. [Google Scholar] [CrossRef]
- Liu, N.; Li, Q.Z.; Scheiner, S.; Xie, X.Y. Resonance-Assisted Intramolecular Triel Bonds. Phys. Chem. Chem. Phys. 2022, 24, 15015–15024. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision A.02; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Dunning, T.H. Gaussian Basis Sets for Use in Correlated Molecular Calculations. I. The Atoms Boron Through Neon and Hydrogen. J. Chem. Phys. 1989, 90, 1007–1023. [Google Scholar] [CrossRef]
- Pitonak, M.; Hesselmann, A. Accurate Intermolecular Interaction Energies from a Combination of MP2 and TDDFT Response Theory. J. Chem. Theory Comput. 2010, 6, 168–178. [Google Scholar] [CrossRef]
- Boys, S.F.; Bernardi, F. The Calculation of Small Molecular Interactions by the Differences of Separate Total Energies. Some Procedures with Reduced Errors. Mol. Phys. 1970, 19, 553–556. [Google Scholar] [CrossRef]
- Halkier, A.; Helgaker, T.; Jorgensen, P.; Klopper, W.; Olsen, J. Basis-Set Convergence of the Energy in Molecular Hartree-Fock Calculations. Chem. Phys. Lett. 1999, 302, 437–446. [Google Scholar] [CrossRef]
- Halkier, A.; Klopper, W.; Helgaker, T.; Jorgensen, P.; Taylor, P.R. Basis Set Convergence of the Interaction Energy of Hydrogen-Bonded Complexes. J. Chem. Phys. 1999, 111, 9157–9167. [Google Scholar] [CrossRef]
- Su, P.F.; Li, H. Energy Decomposition Analysis of Covalent Bonds and Intermolecular Interactions. J. Chem. Phys. 2009, 131, 014102. [Google Scholar] [CrossRef] [PubMed]
- Bader, R.F.W. AIM2000 Program, v. 2.0; McMaster University: Hamilton, ON, Canada, 2000. [Google Scholar]
- Reed, A.E.; Curtiss, L.A.; Weinhold, F. Intermolecular Interactions from a Natural Bond Orbital, Donor-Acceptor Viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).