A Novel Distachionate from Breynia distachia Treats Inflammations by Modulating COX-2 and Inflammatory Cytokines in Rat Liver Tissue
Abstract
:1. Introduction
2. Results
2.1. Phytochemical Analysis
2.1.1. Solubility Analysis
2.1.2. Metal Analysis
2.2. Phytochemical Characterization
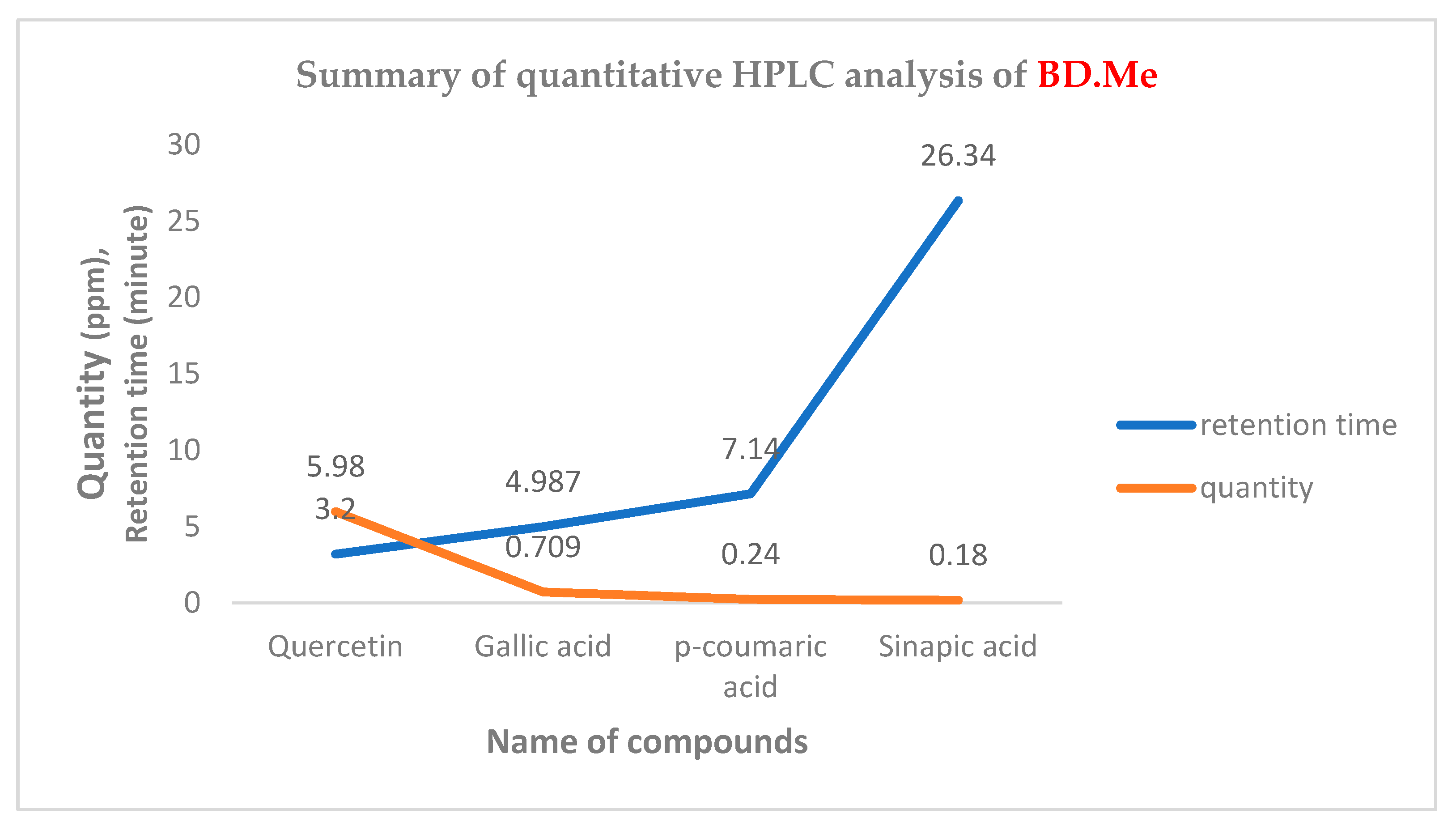
Quantitative Analysis by HPLC
2.3. Qualitative Analysis
Total Phenolic Content
2.4. Purification of Novel Compound
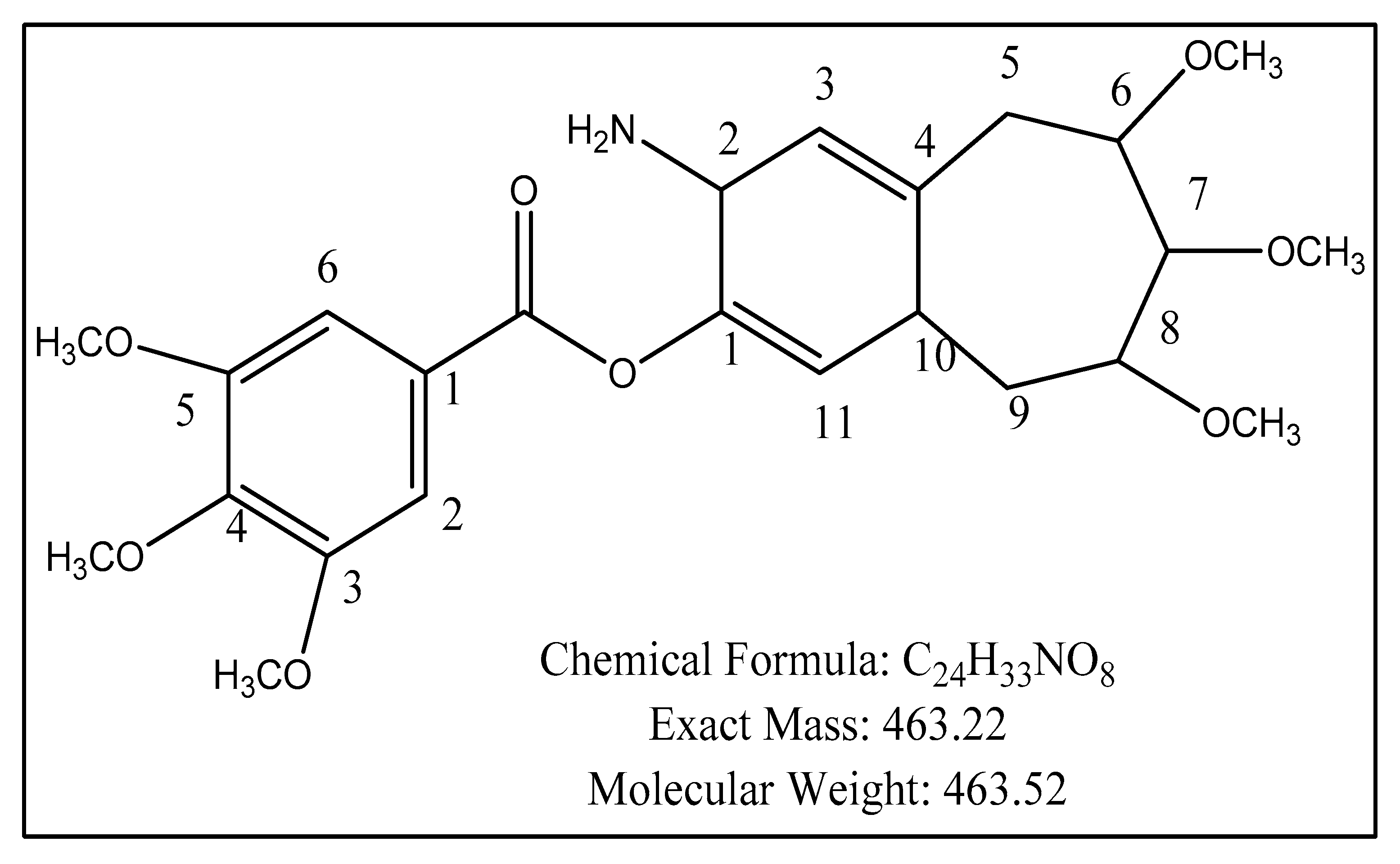
2.5. Structure Elucidation
2.6. Antioxidant Assays
In Vitro
2.7. In Vivo
Antioxidant Enzymes
2.8. Histopathological Results
2.9. Wound Healing Potential of BD.Me
2.10. Carrageenan Paw Edema Model
2.11. Cotton-Pellet Edema Model
2.12. Statistical Analysis of Anti-Inflammatory Study
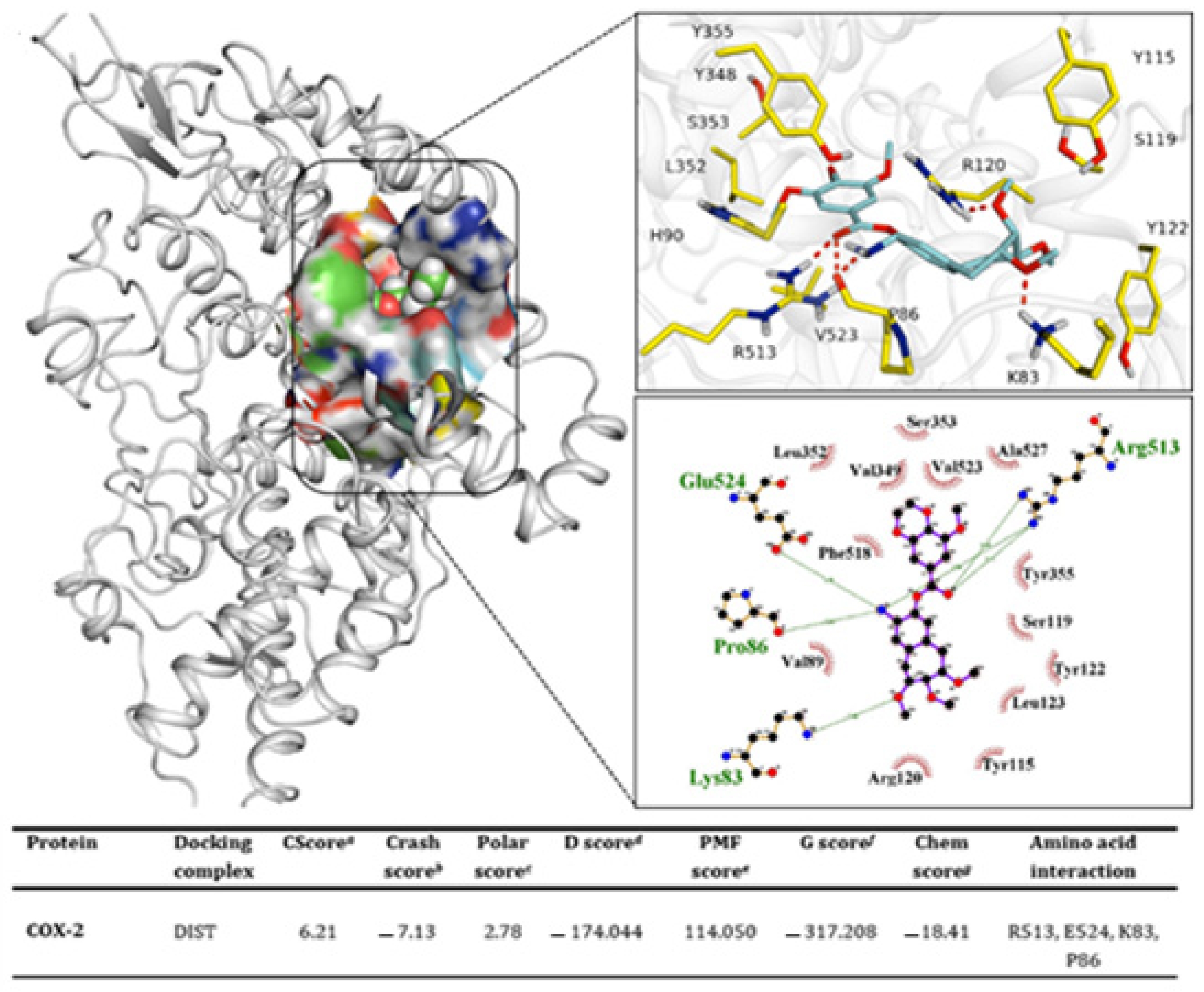
2.13. Docking
3. Discussion
4. Materials and Methods
4.1. Plant Collection
4.2. Plant Extraction
4.3. Phytochemical Analysis
4.4. Solubility Test
4.5. Trace Metal Analysis
4.6. Total Phenolic Content Assay
4.7. Phytochemical Characterization
4.7.1. High Performance Liquid Chromatography (HPLC)
4.7.2. Thin Layer Chromatography
4.7.3. Column Chromatography
4.8. Purification
4.9. Structure Elucidation
4.10. Pharmacological Assays
4.10.1. Antioxidant Assays
4.10.2. Antioxidant Enzymes
4.11. Pharmacological Activities
4.11.1. Acute Toxicity Study
Animals
Experimental Design
4.12. Wound Healing Study
4.12.1. Study Design
4.12.2. Excision Wound Healing Model
4.12.3. Burn Wound Healing Model
4.13. Anti-Inflammatory Study
4.13.1. Carrageenan Paw Edema Model
4.13.2. Cotton Pellet Edema Model
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Simon, L.S. Role and regulation of cyclooxygenase-2 during inflammation. Am. J. Med. 1999, 106, 37S–42S. [Google Scholar] [CrossRef]
- Zhang, J.M.; An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, R.; Rana, C. Plant secondary metabolites: A review. Int. J. Eng. Res. Generic Sci. 2015, 3, 661–670. [Google Scholar]
- Fu, G.M.; Yu, B.Y.; Zhu, D.N. Studies on the chemical constituents from the aerial parts of Breynia fruticosa. Chin. Med. J. 2004, 29, 1052–1054. [Google Scholar]
- Nguyen, T.M.; Le, X.T.; Nguyen, M.T.K. Chemical constituents of Breynia vitis-idaea (Burm. f.) C.E.C. Fischer. AIP Conf. Proc. 2017, 1878, 020041. [Google Scholar] [CrossRef]
- Meng, D.-H.; Wu, J.; Wang, L.-Y.; Zhao, W.-M. Two new glycosides from Breynia vitis-idaea. J. Asian Nat. Prod. Res. 2010, 12, 535–541. [Google Scholar] [CrossRef]
- Zhu, H.; Pollock, N.K.; Kotak, I.; Gutin, B.; Wang, X.; Bhagatwala, J.; Parikh, S.; Harshfield, G.A.; Dong, Y. Dietary sodium, adiposity, and inflammation in healthy adolescents. Pediatrics 2014, 133, 635–642. [Google Scholar] [CrossRef]
- Nielsen, F.H. Magnesium deficiency and increased inflammation: Current perspectives. J. Inflamm. Res. 2018, 11, 25. [Google Scholar] [CrossRef]
- Visweswari, G.; Christopher, R.; Rajendra, W. Phytochemical screening of active secondary metabolites present in Withania somnifera root: Role in traditional medicine. Int. J. Pharm. Sci. 2013, 4, 2770. [Google Scholar]
- Ambriz-Pérez, D.L.; Leyva-López, N.; Gutierrez-Grijalva, E.P.; Heredia, B.J. Phenolic compounds: Natural alternative in inflammation treatment. A Review. Cogent Food Agric. 2016, 2, 1131412. [Google Scholar]
- Shi, Y.-S.; Zhang, Y.; Hu, W.Z.; Chen, X.; Fu, X.; Lv, X.; Zhang, L.-H.; Zhang, N.; Li, G. Anti-Inflammatory Triterpene Glycosides from the Roots of Ilex dunniana Levl. Molecules 2017, 22, 1206. [Google Scholar] [CrossRef]
- Jang, K.-J.; Kim, H.K.; Han, M.H.; Na Oh, Y.; Yoon, H.-M.; Chung, Y.H.; Kim, G.Y.; Hwang, H.J.; Kim, B.W.; Choi, Y.H. Anti-inflammatory effects of saponins derived from the roots of Platycodon grandiflorus in lipopolysaccharide-stimulated BV2 microglial cells. Int. J. Mol. Med. 2013, 31, 1357–1366. [Google Scholar] [CrossRef]
- Gopalakrishnan, A.; Ram, M.; Kumawat, S.; Tandan, S.K.; Kumar, D. Quercetin accelerated cutaneous wound healing in rats by increasing levels of VEGF and TGF-β1. Natl. Inst. Sci. Commun. Inf. Resour. 2016, 54, 187–195. [Google Scholar]
- Arulselvan, P.; Fard, M.T.; Tan, W.S.; Gothai, S.; Fakurazi, S.; Norhaizan, M.E.; Kumar, S.S. Role of antioxidants and natural products in inflammation. Oxid. Med. Cell. Longev. 2016, 2016, 5276130. [Google Scholar] [CrossRef]
- Younus, H. Therapeutic potentials of superoxide dismutase. Int. J. Health Sci. 2018, 12, 88. [Google Scholar]
- Topçu, K.M.Ç.; Kırıcı, D.Ö.; Evcil, M.S. Catalase activity in healthy and inflamed pulp tissues of permanent teeth in young people. Niger. J. Clin. Pract. 2016, 19, 600–602. [Google Scholar]
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. 2013, 58, 187–205. [Google Scholar] [CrossRef]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 2014, 6, 016295. [Google Scholar] [CrossRef]
- Zelová, H.; Hošek, J. TNF-α signalling and inflammation: Interactions between old acquaintances. J. Inflamm. Res. 2013, 62, 641–651. [Google Scholar] [CrossRef]
- Meshram, G.G.; Kumar, A.; Rizvi, W.; Tripathi, C.D.; Khan, R.A. Evaluation of the anti-inflammatory activity of the aqueous and ethanolic extracts of the leaves of Albizzia lebbeck in rats. J. Tradit. Complement. Med. 2016, 6, 172–175. [Google Scholar] [CrossRef]
- Osama, A.; Awdelkarim, S.; Fadul, E.; Mohamed, G.; Siddig, M.; Sheikh, A.; Abdelmoneim, A.; Khalid, A. In vitro Evaluation of Acacia nilotica Pods for its Antioxidant, Acetylcholinesterase Inhibitory Activities and Phytochemical Screening. Eur. Acad. Res. 2015, 3, 3059–3069. [Google Scholar]
- Ramamurthy, V.; Sathiyadevi, M. Preliminary phytochemical screening of methanol extract of Indigotera trita Linn. J. Plant. Biochem. Physiol. 2017, 5, 2. [Google Scholar]
- Güven, D.E.; Akinci, G. Comparison of acid digestion techniques to determine heavy metals in sediment and soil samples. Gazi Univ. J. Sci. 2011, 24, 29–34. [Google Scholar]
- Samad, N.A.; Asif, M.; Tabana, Y.M.; Ahmed, M.B.K.; Majid, A.S.A.; Majid, A.M.S.A. Inhibitory effect of Labisia pumila leaf extract on angiogenesis via down-regulation of vascular endothelial growth factor. Trop. J. Pharm. Res. 2018, 17, 1087–1094. [Google Scholar] [CrossRef]
- Seo, J.-H.; Kim, J.E.; Shim, J.H.; Yoon, G.; Bang, M.A.; Bae, C.S.; Lee, K.J.; Park, D.H.; Cho, S.S. HPLC analysis, optimization of extraction conditions and biological evaluation of Corylopsis coreana Uyeki Flos. Molecules 2016, 21, 94. [Google Scholar] [CrossRef]
- Kumar, S.; Jyotirmayee, K.; Sarangi, M. Thin layer chromatography: A tool of biotechnology for isolation of bioactive compounds from medicinal plants. Int. J. Pharm. Sci. Rev. Res. 2013, 18, 126–132. [Google Scholar]
- Houghton, P. Current trends in the isolation and structural elucidation of medicinal plant constituents in Standardization and utilization of herbal medicines: Challenges of the 21st century. In Proceedings of the 1st International Workshop on Herbal Medicinal Products, Ibadan, Nigeria, 22–24 November 1998. [Google Scholar]
- Hameed, S.; Imran, A.; Nisha, M.; Arshad, M.S.; Saeed, F.; Arshad, M.U.; Khan, M.A. Characterization of extracted phenolics from black cumin (Nigella sativa linn), coriander seed (Coriandrum sativum L.), and fenugreek seed (Trigonella foenum-graecum). Int. J. Food Prop. 2019, 22, 714–726. [Google Scholar] [CrossRef]
- Wheeler, C.R.; Salzman, J.A.; Elsayed, N.M.; Omaye, S.T.; Korte, D.W. Automated assays for superoxide dismutase, catalase, glutathione peroxidase, and glutathione reductase activity. Anal. Biochem. 1990, 184, 193–199. [Google Scholar] [CrossRef]
- John-Africa, L. Studies on the acute toxicity of the aqueous extract of Alysicarpus ovalifolius in mice. Sci. World J. 2019, 14, 43–46. [Google Scholar]
- Jahandideh, M.; Hajimehdipoor, H.; Mortazavi, S.A.; Dehpour, A.; Hassanzadeh, G. Evaluation of the wound healing activity of a traditional compound herbal product using rat excision wound model. Iran. J. Pharm. Sci. 2017, 16 (Suppl.), 153. [Google Scholar]
- Cai, E.Z.; Ang, C.H.; Raju, A.; Tan, K.B.; Hing, E.C.H.; Loo, Y.; Wong, Y.C.; Lee, H.; Lim, J.; Moochhala, S.M.; et al. Creation of consistent burn wounds: A rat model. Arch. Plast. Surg. 2014, 41, 317. [Google Scholar] [CrossRef]
- Zaynab, M.; Fatima, M.; Abbas, S.; Sharif, Y.; Jamil, K.; Ashraf, A.; Aslam, M.M.; Shabbir, A.; Batool, W. Proteomics approach reveals importance of herbal plants in curing diseases. Mol. Microbiol. 2018, 1, 23–28. [Google Scholar]
- Owoyele, B.V.; Adebukola, O.M.; Funmilayo, A.A.; Soladoye, A.O. Anti-inflammatory activities of ethanolic extract of Carica papaya leaves. Inflammopharmacology 2008, 16, 168–173. [Google Scholar] [CrossRef]




| Sr. No. | Name of Element | Dry Powder/Breynia distachia’s Plant Material (50 mg) |
|---|---|---|
| 1 | Iron (Fe) | 0.004 mg |
| 2 | Manganese (Mn) | 0.005 mg |
| 3 | Potassium (K) | 0.830 mg |
| 4 | Zinc (Zn) | 0.004 mg |
| 5 | Sodium (Na) | 4.056 mg |
| 6 | Magnesium (Mg) | 2.357 mg |
| Phytochemicals | Presence or Absence |
|---|---|
| Alkaloids | +ve |
| Phenols | +ve |
| Glycosides | +ve |
| Saponins | +ve |
| Tannins | +ve |
| Carbohydrates | −ve |
| Proteins | −ve |
| Carbon No. | Multiplicity DEPT | 13C-NMR (δ) | 1H-NMR | J-Value |
|---|---|---|---|---|
| C-1 | C | 143.8 | - | - |
| C-2 | CH | 53.4 | 3.99 dd | 1.6 Hz |
| C-3 | C | 114.3 | - | - |
| C-4 | C | 137.2 | - | - |
| C-5 | CH2 | 34.3 | 2.21, 1.96 d | 3.2 Hz |
| C-6 | CH | 82.2 | 3.12 | 3.0 Hz |
| C-7 | CH | 94.43 | 3.20 d | 2.4 Hz |
| C-8 | CH | 84.25 | 2.99 dd | 2.6 Hz |
| C-9 | CH2 | 30.4 | 1.50, 1.23 d | 3.50 Hz |
| C-10 | CH | 31.55 | 2.50 dd | 2.1 Hz |
| C-11 | CH | 109.65 | 4.74 d | 3.25 Hz |
| C-12 | C | 121.2 | - | - |
| C-13 | CH | 104.45 | 7.1 | 2.20 Hz |
| C-14 | C | 149.26 | 6.85 d | - |
| C-15 | C | 139.15 | 6.90 s | - |
| C-16 | C | 148.5 | 7.54 s | - |
| C-17 | CH | 104.33 | 6.97 s | 1.60 Hz |
| C-18 | C | 159.25 | - | - |
| C-19 | OCH3 | 54.33 | 3.80 s | - |
| C-20 | OCH3 | 57.2 | 3.82 s | - |
| C-21 | OCH3 | 54.62 | 3.90 s | - |
| C-22 | OCH3 | 55.35 | 3.18 d | 5.2 Hz |
| C-23 | OCH3 | 55.62 | 3.21 d | 4.3 Hz |
| C-24 | OCH3 | 56.1 | 3.22 d | 4.0 Hz |
| Antioxidant Power | Sample | ||
|---|---|---|---|
| Ascorbic Acid (Standard) | BD.Me | Distachionate | |
| ABTS IC50 (μg/mL) | 3.33 ± 1.07 | 60.85 ± 5.1 | 30.75 ± 3.2 |
| DPPH IC50 (μg/mL) | 2.93 ± 0.32 | 79.50 ± 2.64 | 23.10 ± 2.40 |
| FRAP (nmol Fe + 2 per mg extract) | - | 390.13 ± 14.25 | 28.25 ± 3.50 |
| Antioxidant Enzymes | Effect of Groups | ||||
|---|---|---|---|---|---|
| Control (Distilled Water) | BD.Me 10% Cream | BD.Me 20% Cream | Distachionate | Standard | |
| TSP (mg g−1 FW) | 3.29 ± 0.01 | 3.26 ± 0.01 | 2.62 ± 0.02 | 2.15 ± 0.14 | 1.92 ± 0.12 |
| Catalase enzyme (U/mg protein) | 23.30 ± 0.20 | 23.79 ± 0.20 | 28.81 ± 0.16 | 35.45 ± 0.25 | 44.32 ± 0.08 |
| Suboxide dismutase enzyme (U/mg protein) | 15.11 ± 0.01 | 15.24 ± 0.02 | 16.70 ± 0.06 | 28.10 ± 0.05 | 19.49 ± 0.08 |
| Excision Wound Healing Model | ||||||
|---|---|---|---|---|---|---|
| Groups | Days | |||||
| 0 | 3rd | 7th | 10th | 14th | 21st | |
| Control | 0.0 ± 0.0 | 1.72 ± 0.61 | 19.0 ± 0.26 | 66.8 ± 1.21 | 81.4 ± 0.45 | - |
| BD.Me 10% Cream | 0.0 ± 0.0 ns | 3.85 ± 0.85 ns | 17 ± 0.36 ns | 72.2 ± 1.17 * | 88.7 ± 0.35 *** | - |
| BD.Me 20% Cream | 0.0 ± 0.0 ns | 6.46 ± 0.98 **/ns | 29 ± 2.52 */** | 79.2 ± 0.88 ***/** | 92.4 ± 0.58 ***/** | - |
| Novel Distachionate | 0.0 ± 0.0 ns | 8.2 ± 0.04 ***/ns | 32.6 ± 0.40 **/ns | 78.5 ± 0.75 ***/ns | 93.2 ± 0.55 ***/** | - |
| Standard Contractobex® | 0.0 ± 0.0 ns | 9.5 ± 1.02 ***/ns | 30.8 ± 3.55 **/ns | 81.3 ± 1.93 ***/ns | 96.1 ± 0.92 ***/** | - |
| Burn Wound Healing Model | ||||||
|---|---|---|---|---|---|---|
| Groups | Days | |||||
| 0 | 3rd | 7th | 10th | 14th | 21st | |
| Control (Base cream) | 0.0 ± 0.0 | −19.4 ± 0.74 | 24.3 ± 1.30 | 33.1 ± 2.97 | 46.2 ± 3.29 | 72.4 ± 0.85 |
| BD.Me 10% Cream | 0.0 ± 0.0 ns | −19.9 ± 1.16 ns | 32.9 ± 2.66 * | 42.3 ± 4.11 ns | 58.8 ± 2.50 * | 82.6 ± 1.12 *** |
| BD.Me 20% Cream | 0.0 ± 0.0 ns | −17.5 ± 0.67 ns/ns | 36.5 ± 1.45 ***/ns | 45.9 ± 2.11 */ns | 71.0 ± 2.11 ***/* | 87.2 ± 0.99 ***/** |
| Novel Distachionate | 0.0 ± 0.0 ns | −17.2 ± 0.81 ns/ns | 28.5 ± 1.65 ***/ns | 49.1 ± 3.10 ***/ns | 67.2 ± 1.45 | 90.1 ± 0.55 ***/ns |
| Standard Contractobex® | 0.0 ± 0.0 ns | −16 ± 1.21 ns/ns | 38.2 ± 1.39 ***/ns | 56.7 ± 2.46 ***/ns | 70.5 ± 2.66 | 88.6 ± 0.84 ***/ns |
| Group | Dose mg Kg−1 | Increase in Paw Volume (mL) | ||||
|---|---|---|---|---|---|---|
| 1 h | 2 h | 3 h | 4 h | 5 h | ||
| Saline | - | 0.680 ± 0.040 | 0.690 ± 0.040 | 0.700 ± 0.030 | 0.710 ± 0.030 | 0.720 ± 0.040 |
| BD.ME | 50 | 0.590 ± 0.030 (16.14%) | 0.540 ± 0.040 (21.15%) | 0.490 ± 0.030 (30.11%) | 0.500 ± 0.040 (31.40%) | 0.520 ± 0.040 (31.35%) |
| Distachionate | 5 | 0.470 ± 0.054 (32.82%) | 0.490 ± 0.051 (33.34%) | 0.370 ± 0.040 ** (49.86%) | 0.320 ± 0.060 ** (54.12%) | 0.310 ± 0.050 ** (58.25%) |
| Indomethacin | 5 | 0.230 ± 0.030 ** (68.92%) | 0.200 ± 0.021 ** (72.61%) | 0.180 ± 0.023 ** (75.34%) | 0.190 ± 0.027 ** (72.47%) | 0.190 ± 0.033 ** (74.33%) |
| Treatment | Scab Formation | Complete Reepithelization | Subepidermal Tissue Fibroblast | Granulated Tissues | Blood Vessel | Inflammatory Cell Type | Inflammation Intensity | Epidermal Changes |
|---|---|---|---|---|---|---|---|---|
| Control | − | − | − | + | − | lymphocytes | 2 | − |
| BD.Me 10% cream | − | − | − | − | + | Lymphocytes | 1 | − |
| BD.Me 20% cream | − | − | − | − | − | Neutrophils | 1 | − |
| Standard | − | − | − | − | − | lymphocytes | 0 | − |
| Treatment and Groups | Scab Formation | Complete Reepithelization | Sub-Epidermal Tissue Fibroblast | Granulated Tissues | Blood Vessel | Inflammatory Cell Type | Intensity of Inflammation | Epidermal Changes | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 14 | 21 | 14 | 21 | 14 | 21 | 14 | 21 | 14 | 21 | 14 | 21 | 14 | 21 | 14 | 21 | |
| Control | + | + | − | + | − | − | + | + | − | − | − | Lymphocyte | 2 | 1 | + | − |
| BD.Me 10% cream | + | − | + | + | − | − | + | − | − | − | − | − | 1 | − | + | − |
| BD.Me 20% cream | + | − | + | + | − | − | + | − | − | − | Neutrophil | − | 1 | − | − | − |
| Standard | − | − | + | + | − | − | − | − | − | − | − | − | − | − | − | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saadullah, M.; Asif, M.; Farid, A.; Naseem, F.; Rashid, S.A.; Ghazanfar, S.; Muzammal, M.; Ahmad, S.; Bin Jardan, Y.A.; Alshaya, H.; et al. A Novel Distachionate from Breynia distachia Treats Inflammations by Modulating COX-2 and Inflammatory Cytokines in Rat Liver Tissue. Molecules 2022, 27, 2596. https://doi.org/10.3390/molecules27082596
Saadullah M, Asif M, Farid A, Naseem F, Rashid SA, Ghazanfar S, Muzammal M, Ahmad S, Bin Jardan YA, Alshaya H, et al. A Novel Distachionate from Breynia distachia Treats Inflammations by Modulating COX-2 and Inflammatory Cytokines in Rat Liver Tissue. Molecules. 2022; 27(8):2596. https://doi.org/10.3390/molecules27082596
Chicago/Turabian StyleSaadullah, Malik, Muhammad Asif, Arshad Farid, Faiza Naseem, Sheikh Abdur Rashid, Shakira Ghazanfar, Muhammad Muzammal, Sohail Ahmad, Yousef A. Bin Jardan, Huda Alshaya, and et al. 2022. "A Novel Distachionate from Breynia distachia Treats Inflammations by Modulating COX-2 and Inflammatory Cytokines in Rat Liver Tissue" Molecules 27, no. 8: 2596. https://doi.org/10.3390/molecules27082596
APA StyleSaadullah, M., Asif, M., Farid, A., Naseem, F., Rashid, S. A., Ghazanfar, S., Muzammal, M., Ahmad, S., Bin Jardan, Y. A., Alshaya, H., Saleem, M. H., Ali, S., Adetunji, C. O., & Arif, S. (2022). A Novel Distachionate from Breynia distachia Treats Inflammations by Modulating COX-2 and Inflammatory Cytokines in Rat Liver Tissue. Molecules, 27(8), 2596. https://doi.org/10.3390/molecules27082596









