Abstract
Skeletal muscle is capable of changing its structural parameters, metabolic rate and functional characteristics within a wide range when adapting to various loading regimens and states of the organism. Prolonged muscle inactivation leads to serious negative consequences that affect the quality of life and work capacity of people. This review examines various conditions that lead to decreased levels of muscle loading and activity and describes the key molecular mechanisms of muscle responses to these conditions. It also details the theoretical foundations of various methods preventing adverse muscle changes caused by decreased motor activity and describes these methods. A number of recent studies presented in this review make it possible to determine the molecular basis of the countermeasure methods used in rehabilitation and space medicine for many years, as well as to identify promising new approaches to rehabilitation and to form a holistic understanding of the mechanisms of gravity force control over the muscular system.
1. Introduction
Skeletal muscle is an extremely flexible organ, since it undergoes various structural and functional changes in response to external and internal stimuli. The key regulatory signal for muscles comes from muscle activity, i.e., the level and duration of the muscle loading [1]. The adaptation of muscles to increased load has been the subject of interest since ancient times; however, much less attention has been paid to muscle adaptation to a reduced load or to complete inactivity. In recent decades, the development of scientific and technological progress has led to a change in lifestyle and a decrease in the motor activity of a huge number of people. The progress in medicine has made it possible to recover from severe injuries, often resulting in prolonged confinement to bed. The inactivation of skeletal muscles is often a side effect of the treatment of various diseases that require prolonged bed rest, which can complicate the rehabilitation of patients [2]. Nowadays it is obvious that a person’s health and well-being directly depend on the state of their skeletal muscles, which affects the functioning of the endocrine, nervous, cardiovascular and immune systems [3]. Therefore, the development of countermeasures against changes that occur in skeletal muscles as a result of their decreased activity (functional unloading) is important for human health. This review is devoted to the description of the main changes in disused skeletal muscles (cast immobilization, head-down tilt bed rest, “dry” immersion, space flight and various animal models) and countermeasures against changes occurring under these conditions, including both widely used approaches and those just starting to be put into practice.
According to their functions, skeletal muscles can be divided into two types: locomotor muscles, which are primarily responsible for voluntary locomotions, and “tonic” (postural) muscles, which play a key role in maintaining posture and resisting gravity [4]. These two types of muscles differ in the type of work they perform (primarily in the pattern of neuronal impulses and in the duration of work per day), and in the relative proportion of fibers of different types (“fast-twitch” or “slow-twitch”), which determines their differences in the type of metabolism and contractile activity. Moreover, the properties of fibers of the same type differ significantly depending on whether they belong to a predominantly “locomotor” muscle or a predominantly “tonic” muscle [5]. Data on different models of muscle inactivation that have been accumulated since the 1960s show that tonic and locomotor muscles have different sensitivities to various types of decreases in contractile activity. Postural muscles, predominantly consisting of muscle fibers of the “slow-twitch” type, are the most vulnerable to conditions of disuse and support elimination. The most complete elimination of the support stimulus is observed under space flight conditions, in the model of hindlimb unloading in rodents and unilateral lower limb suspension (ULLS) or “dry” immersion in humans; some weakening of the support stimulus for the muscles of the lower limbs is observed in bed rest as well. In contrast, the locomotor muscles undergo atrophy under when there is a decrease in mechanical loading, even if the support stimulus is present (the most typical model of such an effect is a restricted activity model for humans or animals).
In real-life clinical practice, the conditions of muscle inactivation usually combine both the elimination of support and the elimination of mechanical loading, affecting both locomotor and tonic muscles. Therefore, when developing countermeasures against changes caused by some form of decrease in muscle contractile activity, it is necessary to take into account both the ratio of supportlessness and the decrease in mechanical loading in a particular model, and assess the involvement of tonic and locomotor muscles in negative changes separately (Figure 1).
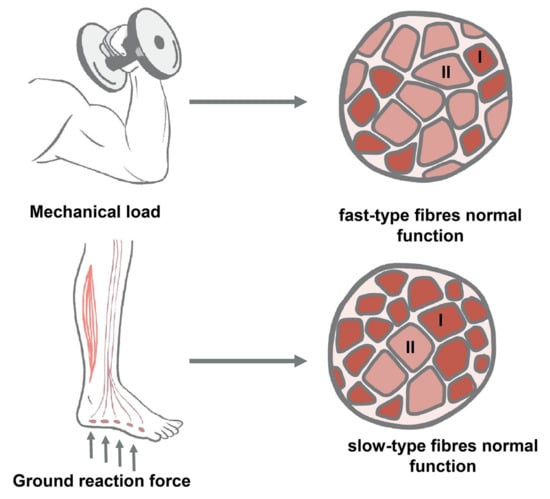
Figure 1.
Involvement of mechanical load and ground reaction force into the regulation of slow-type (type I) or fast-type (type II) skeletal muscle fibers.
2. Models of Decreased Activity of Skeletal Muscles and Their Functional Consequences
Currently, the decreased activity of skeletal muscles is studied in a wide range of models which differ significantly both in effects and in the dynamics and degree of adverse changes manifested. Key research models are head-down tilt bed rest (HDT BR), immobilization (with a cast or leg brace), step reduction, space flight, “dry” immersion and hindlimb unloading [6].
Head-down tilt bed rest is one of the most popular models used for studying the functional unloading of skeletal muscle in humans. HDT BR with a 6-degree tilt is a model for studying the chronic atrophic and adaptive effects of the functional unloading of the skeletal muscles [7]. The effects of HDT BR include both a decrease in the mechanical loading of muscles and a partial elimination of the support stimulus due to the redistribution of the support reaction force from the soles of the feet to the posterior surface of the human body. Atrophy of different muscles with different contents of fast-twitch and slow-twitch fibers was documented for HDT BR. Many studies have shown that HDT BR leads to a significant reduction in muscle strength and mass. A 60-day HDT BR led to a decrease in the cross-sectional area (CSA) of the lower back muscles, whereas a 120-day HDT BR to a decrease in the CSA of the soleus muscle; in both cases, the atrophy correlated with a decrease in the contraction strength of these muscles [8,9]. After 84-day HDT BR, magnetic resonance imaging showed a 17% reduction in mass and a 40% reduction in the strength of the lateral femoris muscle [10]. A different 90-day experiment showed a 26% decrease in the CSA of the lower leg muscles, as well as an 8% decrease in the CSA of the forearm muscles [11]. Moreover, similar results were obtained in a 35-day experiment, in which the CSA of the “slow-twitch” fibers of the lateral femoris muscle decreased to a significantly higher degree than the CSA of the “fast-twitch” fibers of the same muscle [12]. Short-duration HDT BR (17–20 days) also resulted in a 10–12% decrease in the CSA of the lower leg muscles [13]. On the other hand, it was also shown that not all muscles of the lower limbs are affected by prolonged HDT BR; in particular, after a case of 60-day HDT BR, there was no significant decrease in the volume of the rectus femoris, adductor brevis, gracilis or pectineus muscles in comparison with their state before the experiment [14].
The immobilization of limbs makes it possible to study the local effect of a decrease in motor activity on various muscle groups, as well as to identify the consequences of muscle immobilization in traumatology. The immobilized muscles of the lower limbs also exhibit the effect of eliminating the support stimulus on the soles of the feet (when the limb function is disabled). One of these models is limb immobilization using a cast or a special cuff that fixes the limb. A two-week cast immobilization leads to a decrease in the mass, CSA and strength of the quadriceps femoris muscle by 9%, 5–8% and 23%, respectively [15,16]. A 6-week cast immobilization of the lower limb muscles leads to the atrophy of the soleus muscle (19%), as well as the tibialis (10.7%), gastrocnemius medialis (23%) and gastrocnemius lateralis (17%) muscles; the atrophy of the muscles of the contralateral leg is also observed, although to a much lesser extent. Shorter (5-day) periods of lower limb cast immobilization led to a similar decrease in muscle mass [17]. A 5-day study showed a 9% decrease in the strength and a 4% decrease in the CSA of the quadriceps femoris muscle [18]. Thus, in the model of cast immobilization, as well as in bed rest, both fast-twitch and slow-twitch skeletal muscles fibers are subjected to atrophy to a comparable extent.
Space flight exerts a unique influence on skeletal muscles, since under these conditions both the support reaction force and the direct effect of weight are eliminated, which cannot be achieved on Earth. Thus, the conditions of space flight can be called an “ideal” model for studying the role of support, reaction force and weight on the functioning of skeletal muscles. Numerous works devoted to studies of the influence of space flight conditions indicate a significant decrease in skeletal muscle mass, the CSA of muscle fibers and muscle strength [6]. The longer the stay in zero gravity, the more pronounced is the atrophy of the skeletal muscles; 115–197 days of weightlessness lead to a 20% decrease in the mass of the soleus and gastrocnemius muscles and a 10% decrease in the mass of the quadriceps of the thigh [19]. Another study showed that a 180-day space flight significantly reduces the strength and size of the fibers of the soleus and gastrocnemius muscles. In addition, that study showed that the degree of atrophy depends on the type of muscle fibers. For example, the fibers that were the most susceptible to atrophy were the “slow-twitch” postural fibers of the soleus muscle. Furthermore, the fibers most resistant to microgravity conditions were the “fast-twitch” fibers of the gastrocnemius muscle, which mainly perform the locomotor function [20]. Thus, prolonged or brief elimination of the effect of gravity leads to reduced weight bearing by postural muscles and a decrease in their contractile activity. A decrease in the activity of these muscles results in their atrophy, a decrease in their contractile properties, and growing muscle weakness. However, even under terrestrial conditions, there are ways to eliminate the support stimulus.
In the early 1970s, E.B. Shulzhenko and I.F. Will-Williams at the Institute of Biomedical Problems of RAS developed a method for the functional unloading of skeletal muscles which they named “dry immersion” (DI) [21]. This method makes it possible to study the influence of negligible values of the support reaction force on the human motor system and to simulate a number of effects of space flight under terrestrial conditions. The volunteer lies down on a waterproof fabric placed in a bathtub and is thus submerged under the water to the level of the neck, with the fabric area significantly exceeding the surface area of the water. Excess fabric allows the subject’s body to be submerged from all sides [22]. This model makes it possible to simulate the effects of weightlessness, since the force of gravity in this case is balanced by the buoyant force. Thus, DI reproduces three effects of weightlessness: the decrease in muscle activity, the elimination of support signals and the elimination of the vertical distribution of the fluid in the blood vessels [22,23]. Moreover, under the conditions of this model (as well as under the conditions of space flight, especially for animals placed in tight compartments) there is also a limitation of muscle mobility. Staying under conditions of “dry” immersion for 7 days results in a number of changes at the cellular level in the human postural muscles [24,25,26]. Under these conditions, researchers observed a decrease in soleus muscle fiber diameter and the maximum value of the isometric force, calcium sensitivity and transverse stiffness, CSA of predominantly “slow-twitch”-type fibers, and of the relative content of titin, nebulin, desmin and α-actinin. These changes developed along with a decrease in the electrical activity of this muscle [27]. Earlier studies showed that as early as on the 3rd day of “dry” immersion, the slow-twitch motor units did not participate in the work of maintaining a small load [28]. It is interesting to note that the depth of atrophic changes that were found after 7 days of immersion is usually developed after longer periods of bed rest [29,30]. After 7 days of DI, a 24% decrease in the CSA of the slow-twitch fibers of the soleus muscle was detected, whereas there was no decrease in the CSA of the fast-twitch fibers of the same muscle [31]. Nevertheless, after 3 days of DI, a significant decrease in the CSA of the slow-twitch fibers of the vastus lateralis muscle of 7.5% and in the fast-twitch fibers of 8.9% was observed [23]. After 7 days of DI, a decrease in the CSA of slow-twitch and fast-twitch fibers of the vastus lateralis muscle had already reached 17.3% and 15.3%, respectively [23]. Under conditions of dry immersion, the contractile properties of the human soleus muscle were also studied. After 7-day DI, the maximum strength of the fibers of the soleus muscle decreased by 32% [26,31]. Similar data were obtained after 7-day DI and 7-day space flight using isokinetic dynamometry [32,33]. It should be noted that the decrease in the maximum strength of voluntary contraction in the DI experiment was more pronounced.
The most complete limitation of skeletal muscle mobility with the concomitant elimination of support afferentation occurs after various injuries affecting the nervous system, such as denervation of a muscle or muscle group, spinal cord injuries, various paralyses, as well as in intensive care units. In such conditions, both the afferent signal from the muscle and neuronal stimulation are absent. In some disorders, there is also no trophic effect of the nerve on the muscle fibers, which leads to catastrophic changes in both the fast-twitch and slow-twitch fibers of skeletal muscles [34,35,36]. In the case of life-threatening diseases (critical illness), muscle damage is also caused by the diseases themselves or their complications (sepsis, inflammation), as well as by drug therapy (for example, the use of glucocorticoids), and thus the data obtained in the study of such conditions cannot be used when considering the functional unloading of skeletal muscles. Therefore, a detailed description of the models associated with denervation is not included in this review.
There are also models studying a partial decrease in muscle activity with the full preservation of the support stimulus. One such model is step reduction (SR). In the SR experiment, subjects reduce the amount of daily walking (while counting each step with a pedometer) to the minimum values (750–5000 steps) [37,38]. The lower threshold for the number of steps (~750) approximately corresponds to the activity of outpatients [39]. The reduction of physical activity in the SR model to such low values does not lead to a complete functional unloading of skeletal muscles, but has serious physiological consequences [40]. Restriction of activity by 91% (from 13,054 to 1192 steps per day) reduces protein synthesis in skeletal muscle fibers by 22% [41]. The physiological consequences of even short periods of reduced activity in the SR model lead to a decrease in skeletal muscle strength and mass, as well as a decrease in insulin sensitivity. A reduction in the number of steps to less than 1500 per day for two weeks leads to a decrease in the maximum oxygen consumption [38] and a significant decrease in the maximum voluntary strength of the lower limbs [42,43].
Studies in animals (mainly in rodents and primates) also include models with the elimination of the support stimulus as the primary effect and models with the restriction of muscle contractile activity. At present, it is questionable whether animal reduced-activity models can be compared with human models. Nonetheless, the main functional and structural changes (muscle fiber atrophy, transformation of the phenotype, decrease in maximum strength), as well as changes at the molecular level (a decrease in protein synthesis and the level of mitochondrial biogenesis, an increase in the expression of proteolytic markers, changes in the expression pattern of myosin genes) are observed in both rodents and humans. Obviously, the dynamics of changes occurring during functional unloading of the muscles should differ in rodents and in humans. To date, there are insufficient data for most models and it is not possible to clearly compare the timing of the development of certain molecular responses to functional muscle unloading in different mammalian species. Nevertheless, when translating a number of results from animal models to humans, it is necessary to take into account the type of skeletal muscles, whether “tonic” or “locomotor”. A number of species of small animals, in particular mice (in contrast to rats), do not have purely “tonic” muscles at all, which imposes certain limitations on studies of the effects of supportlessness in these animals [44]. Rodents also have a type of myosin heavy-chain IIB isoform which is absent in humans [45].
The model of rodent hindlimb unloading is widely used to study muscle plasticity. One of the reasons this model is widely used is the similarity of many of its effects to those of space flight. It has been approved by many researchers as adequate for simulating microgravity, studying changes in skeletal muscle during space flight and the adaptation of skeletal muscles to decreased activity [46,47,48]. In this model, the animal is suspended by the tail in such a way that its hindlimbs do not touch the floor of the cage, whereas it is able to freely move around the cage. Thus, the support reaction force is eliminated.
In rat models of immobilization and hindlimb unloading, muscle loss is usually greater in the lower leg extensors (soleus, gastrocnemius muscles) than in flexors (extensor digitorum longus) [5,49,50]. A reduction in the CSA is observed first in slow-twitch, then in fast-twitch fibers of the IIa type, and then in IIx and IIb types [49,51], which is usually associated with elimination of the postural function in conditions of a supportless environment [52]. Atrophy of myofibrils under conditions of simulated hypogravity leads to a deterioration of the muscle contractile properties [53,54,55]. After 7-day hindlimb unloading, the relative weight of the soleus muscle decreased by 21% compared to the control, the maximum tetanic tension normalized to muscle weight was significantly reduced, and the contraction time decreased by 25% [56]. After 16-day hindlimb unloading, the absolute weight of the soleus muscle decreased by 50% and 65%, the fiber size decreased by 66% and the fiber length and the amount of sarcomeres decreased by 14% compared to control animals [57]. After 4-week hindlimb unloading, the weight of the unloaded soleus muscle was 42% lower than in control animals, whereas the strengths of maximum isometric contraction and tetanic tension were reduced by 62% and 69%.
The method of restricting the activity of animals while preserving the support stimulus is a partial limitation of mobility. In this model, the rodents are kept in small cages that restrict the movement of the animals. In [58], the weight of the soleus and plantaris muscles decreased by 10% and 27%, respectively, after 21 days in such cages. A decrease in the CSA was observed only for the plantar muscle, with the limitation of activity having a greater effect on the fast-twitch muscle fibers. These results differ from the earlier data obtained under the conditions of weightlessness or hindlimb unloading, which showed a greater atrophy of slow-twitch muscle fibers than fast-twitch ones [5,49]. In a recent experiment with a 21-day activity restriction in rodents, a decrease in the mass of both the soleus muscle and EDL was found; however, a decrease in protein synthesis and mTOR activity (determined by the level of phosphorylation of p70S6k) was observed only in the “fast-twitch” EDL [59]. Moreover, in connection with the problem of activity restriction, it is necessary to consider the data obtained on primates, when comparing the conditions of space flight and staying in the same capsule as those in the flight, but on Earth. Animals in space flight showed a significant decrease in the CSA of the vastus lateralis muscle, which was also observed in animals staying on Earth, whereas the sizes of the slow-twitch fibers of the soleus muscle decreased only under conditions of space flight [60,61]. These data demonstrate the different effects of supportlessness and mobility limitation for the locomotor and tonic components of the muscular system.
Cast immobilization can also be studied in animals. During immobilization, the joint of the hindlimb of the animal is fixed in the desired position using a cast bandage or needles. Experimental immobilization (with a needle) was used by Fischbach and Robbins [62]. They observed a significant reduction in impulse activity to 5–15% [62], <10% of the control [63], as well as a significant reduction in the strength of muscle contraction. Depending on the angle of fixation of the joint, the degree of muscle atrophy can be different, whereas the length of the muscle in a fixed state is very important [64]. A shortened muscle undergoes atrophy more strongly than a stretched muscle, and the decrease in functional activity in a shortened state is stronger [65,66,67]. Presumably, this effect is associated with the role of mechanical tension in the regulation of muscle mass.
Another model of the functional unloading of skeletal muscles of animals is tenotomy. In this model, a transection or partial dissection of the tendon is performed, which leads to a decrease in the mechanical loading and weakens the afferent response of the muscle due to the inactivation of muscle spindles. At the initial stages of tenotomy, in contrast to the hindlimb unloading model, the EMG activity of the tenotomized muscles of the hindlimbs is retained for at least one week [68,69]. After tenotomy, muscle weight and its strength characteristics decrease; atrophy occurs faster than in other models of functional unloading of the muscle. Weight loss can reach 50% or more. The weight of the soleus muscle of the rat after a 12-day tenotomy decreased approximately twofold [70,71]. After tenotomy, predominantly slow-twitch muscle fibers of tenatomized muscles undergo atrophy [72]. The key muscle inactivity models and their effects on slow-type and fast-type fibers are listed in Table 1.

Table 1.
Classification of the types of in vivo inactivation of skeletal muscles.
3. Molecular Signaling Alterations in Skeletal Muscles under Disuse Conditions
The regulatory mechanisms involved in negative changes in the skeletal muscles under various disuse conditions have been studied for decades and researchers are still far from reaching a complete understanding. Since there are a lot of excellent and detailed reviews dedicated to various aspects of muscle signaling under muscle inactivation, this chapter of our review briefly describes the key molecular mechanisms determining muscle atrophy, fiber-type transition, as well as decreases in oxidative capacity and muscle functional characteristics.
The key signaling events of early muscle inactivity (caused by supportlessness or immobilization) are a dramatic decrease in energy consumption (ATP/ADP ratio increase, glycogen accumulation), calcium ion accumulation (accompanied by ROS production), a nitric oxide production decrease and the inactivation of mechanosensors [73,74,75]. These early changes cause an “intermediate” signaling state between actively working muscle (which is characterized by a low ATP/ADP ratio, high calcium and ROS levels, a high level of NO and active mechanosensors) and the resting muscle (high ATP/ADP ratio, low calcium and ROS levels, low NO and inactive mechanosensors). In this state, the negative effects of calcium/ROS accumulation cannot be compensated for by activity-induced defense mechanisms, so this leads to development of muscle atrophy and subsequent alterations. In the next chapters we describe the detailed regulatory mechanisms mediating the response of skeletal muscles to these changes.
It should be noted that the complete disappearance of postural soleus muscle EMG activity, observed during the first 1–3 days of muscle disuse, is followed by the restoration of EMG to control levels between the 6th and 9th days of rodent hindlimb suspension, and the further increase of EMG above the control level [76] A similar effect is observed in humans and animals after spinal cord injuries, although the time before the reappearance of EMG is longer [77]. In the hindlimb suspension model, the reappearance of EMG is accompanied by a decrease in the ATP/ADP ratio and activation of some activity-dependent signaling pathways (AMPK activation, NFAT entrance into muscle nuclei, myosin IIa expression restoration) after 14 days of rat hindlimb unloading [78,79]. It should be underlined that in the earlier stage of muscle unloading (1–3 days) AMPK is inactivated, the ATP/ADP ratio is high and the NFATc1 myonuclear content is twofold lower than that in the control [80,81,82]. However, these changes during later unloading stages do not lead to the restoration of muscle function. Moreover, some of these changes even enhance atrophy development [74]. These facts indicate that the time-course of signaling events during muscle disuse (especially the early stages of muscle disuse, 2 weeks and earlier) is not just a linear developing process, and the key mechanisms that determine atrophy development as well as other alterations are different at different time-points of disuse, even in the same disuse model and the same muscle. Nowadays the causes and effects of disuse-induced slow-tonic muscle activity are almost completely unclear; however, the presence of this activity, as well as its non-linear effect on muscle signaling, cannot be ignored when studying disused muscle functioning.
3.1. Effect of Reduced Motor Activity on Protein Synthesis in Skeletal Muscle
The loss of skeletal muscle mass (muscle atrophy) is caused by alterations of protein homeostasis in skeletal muscle fibers, i.e., by a decrease in the level of protein synthesis and/or by an increase in the rate of protein degradation. The relative contribution of these two processes to the atrophy caused by functional unloading depends on the studied model, as well as on the duration of the unloading. As early as at the 1st day of the hindlimb unloading of rodents, a significant decrease in the level of protein synthesis was observed in the soleus muscle [83]; at the same time, there was not yet any significant decrease in muscle mass observed. A reduced level of protein synthesis, determined using the SuNSET method, was also observed at later stages of hindlimb unloading (3rd, 7th, 14th days) [83,84,85]. A decrease in the level of protein synthesis assessed using radioactive labeling was observed after 3-, 4-, 7- and 28-day or hindlimb unloading of rodents [86,87,88,89].
A decrease in the level of protein synthesis as a result of the functional unloading of skeletal muscles was also found in humans; for example, Gibson et al. showed that people exposed to ULLS under fasting conditions had a ~30% lower protein synthesis rate compared to a contralateral non-immobilized limb [90]. Subsequent studies confirmed the decrease in protein synthesis, for example, a ~50% decrease in the synthesis rate was observed after 2-week HDT BR [91,92] and cast immobilization [15]. Later, Kortebein et al. reported a ~30% reduction in protein synthesis measured over a 24-h period in older adults after 10 days of bed rest [93].
Nowadays, the intensity of protein synthesis in mammalian skeletal muscles is believed to depend both on the efficiency of translation and on the translational capacity [94]. Translation efficiency is the rate of protein synthesis per ribosome, whereas translational capacity is determined by the amount of ribosomes per unit of tissue and, accordingly, depends on the amount of ribosomes in muscle fibers [94,95]. It should also be borne in mind that in order to fully understand the mechanisms that control protein synthesis in the skeletal muscles, it is necessary to take into account the duration of gravitational unloading [95,96].
The mTOR signaling protein (the mechanistic target of rapamycin) is the main center of the signaling network involved in the regulation of mRNA translation and thus translation efficiency [97]. Studies in vitro and on rodents show that this system responds to important stimuli responsible for the regulation of protein synthesis: (1) insulin and insulin-like growth factor-1 (IGF-1) via the IRS (insulin receptor substrate) and PI3K (phosphatidylinositol-3) signaling pathways, (2) amino acid levels via the leucyl-tRNA/Rag-mTORc1 pathways, (3) energy stress via the AMPK/eEF2 (AMP-activated protein kinase/eukaryotic elongation factor 2) pathway, and (4) mechanical stress via various mechanosensory molecules [97,98]. In conditions of normal activity in mammals, the IGF-1/Akt/mTORC1 signaling pathway acts as a regulator of translation initiation in the skeletal muscle [99,100]. This signaling pathway is initially activated via IGF-1 binding to its specific receptor (IGF-1R), which triggers a signaling cascade, resulting in the phosphorylation of the insulin receptor 1 substrate (IRS-1). Next, the phosphorylated IRS-1 binds to phosphatidylinositol-3-kinase (PI3K), which results in its activation [101]. After that, the phosphatidylinositol-3-phosphate interacts with phosphoinositide-dependent protein kinase 1 (PDK1), which phosphorylates protein kinase B (Akt). The phosphorylated Akt then activates mTORC1 via the inhibition of TSC2 [102,103,104]. The activated mTORC1 phosphorylates two molecules: 4E-binding factor 1 (4E-BP1) and ribosomal kinase 70 (p70S6k), which leads to an increase in protein synthesis [105]. It was shown that under disuse conditions IRS-1 is rapidly ubiquitinated by cblb ubiquitin ligase, which lead to its degradation. Cblb expression was prevented by antioxidant administration, such as N-acetylcysteine and TEMPOL, indicating the ROS-induced degradation of IRS [106]. IRS-1 degradation in turn leads to PI3K-dependent signal weakening and Akt dephosphorylation.
In addition to the main pathway, Akt is also capable of phosphorylating and inhibiting glycogen synthase kinase (GSK-3β) at Ser 9, which can lead to an increase in total protein synthesis via translation initiation factor 2B (eIF2B) [107]. GSK-3β phosphorylation at Ser 9 is significantly downregulated as early as after 1 day of rat hindlimb suspension and remains decreased at least during the first two weeks of rat hindlimb unloading [78,83]. Nevertheless, the functioning of the mTOR pathway under functional unloading of the skeletal muscles (in particular, hindlimb unloading) remains unclear. A number of studies have shown an increase in the phosphorylation of p70S6k (which implies an increase in mTOR activity) on days 1–3 of hindlimb unloading [78,83,108]; however, this activation is accompanied by a decrease in the level of protein synthesis. This transitory activation may be caused by inactivity-dependent AMPK downregulation, as AICAR treatment during 1 day of rat HS prevented an increase in p70S6K phosphorylation [80]. At the same time, rapamycin treatment during 1-day HS prevented an increase in the expression nof MuRF-1 and atrogin-1 E3 ubiquitin ligases, so we can conclude that transient mTOR activation contributes to the early development of disuse-induced proteolysis [80].
At the same time, in the later stages of the mechanical unloading of the hindlimbs, as a rule, there is a decrease in the phosphorylation of p70S6k [109,110,111]. Phosphorylation of 4E-BP1, which is also an effector of mTORC1, decreased in the rat soleus muscle in the first days of functional unloading [78,112]. In the soleus muscle of the mouse, it did not change after 3- and 7-day hindlimb unloading [113]. After 14-day hindlimb unloading, according to some authors, the phosphorylation of 4E-BP1 significantly decreased in the rat soleus muscle [109,114], whereas according to other authors, it did not differ from control values [96,115]. Thus, it cannot be ruled out that the contribution of mTOR-dependent signaling pathways to a decrease in protein synthesis in skeletal muscles depends on the stage of functional unloading. The reasons for the nonlinear changes in the activity of mTOR must, apparently, be considered in the context of the fact that mTORC1 receives signals about the energy state of the cell and decreases its activity in case of a deficiency of high-energy phosphates and the activation of AMP-dependent protein kinase (AMPK). A deficiency of ATP and an increase in the level of AMP lead to the activation of AMPK (phosphorylation at Thr172), which is a key energy sensor in the cell. When activated, AMPK can suppress mTORC1 signaling through the phosphorylation and activation of TSC2 [116] or the phosphorylation of Raptor (regulatory-associated protein of mammalian target of rapamycin, a part of the mTORC1 complex), which leads to its subsequent sequestration by regulatory proteins of the 14-3-3 family [117]. The data obtained in our laboratory indicate that AMPK becomes dephosphorylated (at Thr172) in the rat soleus muscle as early as the 1st day after hindlimb unloading [78,118], as well as in the soleus muscle of humans after a 3-day immersion [119], suggesting that the increased activity of mTORC1 at the initial stage of gravitational unloading may be associated with the inactivation of AMPK at this stage. A further increase in AMPK activity (twofold in comparison with the control after 14 days of hindlimb unloading) can lead to the inactivation of mTOR and an increase in atrophy [78].
The molecular mechanisms of mTOR regulation in conditions of the functional unloading of skeletal muscles in humans remain insufficiently clear. In response to unloading, a decrease in the phosphorylation of mTOR and p70S6K was observed after 2 weeks of immobilization [120]. In contrast, other studies did not find a decrease in signal transmission from Akt to mTOR, despite a decrease in protein synthesis [98,121]. It is worth noting that the peak activation of signaling proteins (4E-BP1 and p70S6K) occurs 1–2 h after a meal, whereas muscle biopsies in many studies were taken 3–6 h after a meal, when the response was weakened or possibly absent [122,123,124]. This effect may complicate the analysis of mTOR signaling in humans under disuse conditions.
Another problem related to the unloading-induced changes in protein synthesis is insulin resistance. Insulin resistance (i.e., an impaired metabolic response to endogenous or exogenous insulin) has repeatedly been observed in humans after HDT BR [125,126] and immobilization [127], as well as in rats in a model of hindlimb unloading [128]. Experiments with hindlimb unloading showed that insulin resistance led to a weakening of the Akt/mTORC1 signaling pathway [105] in the soleus and medial gastrocnemius muscles of the rat.
The level of protein synthesis is also influenced by translational capacity, which is characterized by the amount of ribosomes [94]. A decrease in the content of total rRNA, as well as 18S and 28S rRNA, was registered in the soleus muscle after 1, 3, 7 and 14 days of hindlimb unloading in rats [84,110] and after a 21-day “dry” immersion in humans in our laboratory (unpublished data). A decrease in the total concentration of rRNA was also shown in experiments with gravitational muscle unloading in humans [129,130], as well as during periods of inactivity after surgery [131]. Although changes in the content of ribosomes after immobilization were not observed in humans [90,132], the above results indicate that ribosome biogenesis can be disrupted in various variants of functional unloading, contributing to a decrease in protein synthesis [94].
Recent studies in our laboratory showed that nitric oxide plays an important role in the regulation of protein synthesis and GSK-3 activity, as well as the content of ribosomal RNA at later stages of hindlimb unloading (day 7) [85]. Mechanical stimulation of the feet support zones prevented a decrease in the content of 18S and 28S rRNA after 7 days of hindlimb unloading in rats due to nitric oxide production. It also partially prevented a decrease in protein synthesis, which suggests that a decrease in translational capacity under conditions of functional unloading may contribute in a decrease in protein synthesis and is apparently regulated by the level of nitric oxide in the myoplasm of the skeletal muscle [85]. It cannot be ruled out that the NO/GSK-3 (or NO/rRNA) signaling pathway contributes to a decrease in the level of protein synthesis at the earlier stages of hindlimb unloading, including the period of increased mTOR activity. On the 3rd day of the experiment with the mechanical stimulation of the feet support zones, the Akt-independent inactivation of GSK-3 was detected, which is likely to be the result of an increase in NO [81]. NO donor treatment also prevented muscle atrophy after 7 days of mice hindlimb unloading [133]. However, the role of NO in disuse-induced muscle alterations is a subject of a scientific discussion and it will be revealed in detail in the next chapter.
3.2. Effect of Reduced Motor Activity on Protein Degradation in Skeletal Muscle
To date, it is impossible to deny the contribution of increased protein degradation to skeletal muscle atrophy in various models of the functional unloading of skeletal muscles [134]. After only 3 days of ULLS, an increase in the content of 3-methylhistidine, a marker of myofibril destruction, in the interfiber space of the vastus lateralis muscle, was registered in the volunteers [135]. Nonetheless, direct measurement of the protein degradation rate, in contrast to measurements of the protein synthesis rate, is rarely carried out. In most studies, conclusions about the rate of protein degradation are made by analyzing the functioning of molecular proteolytic systems.
A number of authors in recent years have turned their attention to the fact that studies on the effects of bed rest in humans showed a significant decrease in the level of protein synthesis and the content of anabolic signaling markers, whereas no changes in the activity and expression of proteolytic enzymes were found [see review [136]]. These authors even suggest the existence of fundamental biological differences between humans and laboratory rodents. For example, the hindlimb unloading model is always accompanied by a high activity of proteolytic systems. However, all arguments in favor of this concept are based on studies in humans on the vastus lateralis muscle, a mixed locomotor muscle with a high proportion of fast-twitch fibers, whereas in studies on laboratory rodents, samples of predominantly slow-twitch postural soleus muscles are used. Even in studies on the vastus lateralis muscle of humans, an increase in the proteolytic enzymes’ expression is observed after a short-term exposure to unloading. Studies of human soleus muscles under conditions of unloading also show increases in proteolytic enzymes [see review [134]].
The main protein degradation systems in skeletal muscle are Са2+-dependent proteases (calpains), the autophagosomal system and the ubiquitin-proteasome system [137]. Calpains promote the degradation of muscle proteins and may be responsible for the destruction of the cytoskeletal structure of myofibrils [138].
The main cellular event that activates calpain is the accumulation of calcium ions in the myoplasm. Such an increase was predicted by G.A. Nasledov [139] and discovered by C. Ingalls [140]. In our laboratory, it was shown that blocking L-type calcium channels in vivo can partially prevent destructive processes in the soleus muscle during unloading [for an overview, see [141]]. The levels of resting cytosolic calcium in the slow-tonic muscles of the hindlimb-unloaded animals were increased on the second day of unloading and remained elevated at least during the next two weeks [140,142]. The overexpression of calpastatin, an endogenous calpain inhibitor, prevented the atrophy of rodent soleus muscles by 30% after 10 days of hindlimb unloading [143]. The administration of the calpain inhibitor prevented the increased ubiquitination of proteins, phosphorylation of FOXO3, and an increase in the expression of ubiquitin ligase MAFbx [144], which indicates the coordinated activation of various proteolytic systems under functional unloading. Moreover, calpain inhibitor administration protects cytoskeletal proteins, including desmin and titin, from degradation, and prevents the unloading-induced decrease in soleus muscle stiffness after 3-days unloading [145]. In recent years, new data indicating a more complex nature of the interaction of calpain-dependent and ubiquitin-proteasome proteolysis in muscle atrophy were obtained. It was recently shown that the intense degradation of desmin, which is characteristic of the initiation stage of denervation atrophy, begins with intense phosphorylation of the substrate by glycogen synthase kinase (GSK3β) and then the ubiquitination of the molecule by the E3 ubiquitin ligase MuRF-1. Only after this does the cytoskeletal protein molecule undergo calpain-dependent fragmentation and processing in the 26S proteasome [146].
Nitric oxide is one of endogenous inhibitors of both calpains and GSK-3, so the lack of NO under conditions of a high resting cytoplasmic calcium level may be one of the causes of the unloading-induced proteolysis upregulation. Moreover, in a recent article we have shown that calpastatin expression in the rat soleus muscle is regulated in an NO-dependent manner [147]. In this study, we have also showed that plantar mechanical stimulation during 7-days with hindlimb suspension prevented the unloading-induced decrease in muscle stiffness as well as the downregulation of desmin, titin and nebulin protein contents in tbe soleus muscles, whereas treatment by nNOS inhibitor L-NAME completely blocked these effects.
However, nNOS activity was shown to trigger the entrance of the FOXO transcription factor into muscle nuclei after 6 h of rat hindlimb unloading in soleus muscle [148], so the role of NO (or more likely the combination of NO and ROS, causing nitrosative stress) during the first 24 h of unloading could seem rather controversial. We previously showed that hindlimb unloading of the rat leads to a significant and profound decrease in the concentration of nitric oxide in the soleus muscle [149,150]. At the same time, Suzuki et al., using a similar model and the same analytical method (EPR spectroscopy), observed a significant increase in the content of nitric oxide in the soleus muscle of mice [151]. These studies differ in a number of technical details. In particular, Suzuki et al. used unfrozen tissue samples, which made it possible for NO synthases to maintain their activity during measurements. A detailed analysis of methodological issues was carried out in the study [150]. These considerations and a number of indirect data (in particular, a profound decrease in the expression of nitric oxide synthase during unloading [148,149,152]) suggest a significant decrease in the level of nitric oxide in the fibers of unloaded soleus muscle. Lomonosova et al. also obtained data indicating the role of this decrease in the activation of the calpain-dependent proteolysis of cytoskeletal proteins: the intensity of their degradation significantly decreased when the animals were administered with the main substrate of nitric oxide synthase L-arginine [149].
Proteolytic fragments produced via the activity of calpains are ubiquitinated and degraded in proteasomes to amino acids by cathepsin peptidases. Much evidence suggests that calpains [153], caspase-3 [154], the autophagolysosomal system of the cell [155] and the ubiquitin-proteasome system [156] are all involved in muscle atrophy caused by unloading. However, under conditions of unloading, the ubiquitin-proteasome system is often regarded as the most important proteolytic system that leads to muscle atrophy [96,137]. For protein cleavage through this system, three different enzymatic components of the ubiquitin-proteasome pathway are required: E1 (ubiquitin-activating enzyme), E2 (ubiquitin-conjugated enzyme) and E3 (ubiquitin ligase, a key enzyme that regulates proteolysis, since it recognizes numerous target protein substrates) [157]. Two muscle-specific E3 ligases (MuRF1 and MAFbx/atrogin-1) play an important role in skeletal muscle atrophy, including that caused by functional unloading [156,158,159]. In various animal models of muscle atrophy under conditions of mechanical/gravitational unloading (hindlimb unloading, immobilization, space flight and denervation), the mRNA levels of both ubiquitin ligases (MuRF1/MAFbx) rapidly increased and supposedly played a decisive role in the initiation of the atrophy process [96,160,161,162,163,164]. Studies using MAFbx- and MuRF1-knockdown mouse models further confirmed the role of these two genes in the development of muscle atrophy. For example, mice deficient in either MAFbx or MuRF1 were found to be more resistant to denervation-induced atrophy [161]. Another study also showed an attenuated atrophy response of the soleus muscle of MuRF1-knockout mice during 10 days of hindlimb unloading [165]. In the studies by our laboratory, the activation of the expression of MuRF-1 and MAFbx was shown to occur already after the first 24 h of the hindlimb unloading of rats [80]; at this stage, the increase in their expression was triggered by the mTOR activation caused by the AMPK inactivation. Moreover, at the conditions of 3-day hindlimb unloading of rats, the administration of the MAP kinase inhibitor p38 was shown to prevent an increase in MuRF-1 expression, as well as the atrophy of the soleus muscles of experimental animals [166]. Inhibition of xanthine oxidase, an activator of the p38 MAP kinase, also prevents an increase in MuRF-1 expression in animals subjected to hindlimb unloading [167].
Apparently, the activation of ubiquitin ligase expression during the functional unloading of skeletal muscles is caused by both the accumulation of high-energy phosphates and the accumulation of reactive oxygen species, which is likely to be triggered, among other factors, by the accumulation of calcium ions in the cytoplasm of muscle fibers. In a recent study, researchers investigated one more calcium-dependent pathway regulating proteolysis under disuse conditions. The “slow” path of Ca2+ accumulation in the myoplasm in between muscle contractions, dependent on ATP/ADP accumulation, their exit from pannexin 1 channels, P2Y receptor activation and PI3/PLC-dependent opening of IP3 calcium channels, was described by E Jaimovich and colleagues [168]. In our laboratory it was shown that the blocking of pannexin 1 channels during 3 days of rat hindlimb suspension results in downregulation of key proteolytic markers MuRF-1 and MaFbx expression, so the described mechanism of calcium accumulation is likely to contribute to the unloading-induced muscle atrophy [82].
Despite the fact that in many works MuRF-1 and MAFbx are considered together, there is evidence that the degree and timing of activation of these two enzymes are different for different muscles, as well as the regulatory mechanisms that determine their expression. Under conditions of unloading, the expression of E3 ubiquitin ligases MuRF1 and MAFbx is known to be regulated by various upstream factors or signaling pathways [156]. For example, in the plantar flexors of the ankle joint (soleus and medial gastrocnemius muscles), more significant and prolonged increases in the expression of the MuRF1/MAFbx genes occurred compared to the dorsi flexor of the foot (tibialis anterior muscle) [160,161]. In our laboratory, the expression of MaFbx, but not MuRF-1, was shown to be regulated by histone deacetylase 1 after 3 days of hindlimb unloading in rats [169]. Mechanical stimulation of the support zones of the feet during 3 days of hindlimb unloading, on the contrary, prevents an increase in the expression of MuRF-1, but not MaFbx, as well as the inhibition of MAP kinase p38 [83].
It should also be noted that there are many studies on human models that show upregulation of MuRF1 and MAFbx during atrophy, caused by unloading [162]. For example, the MuRF1 and MAFbx mRNA content increased significantly after 2-day [170] and 5-day immobilization [18], or after 3-day ULLS [171]. However, in a 20-day bed rest study, an elevated content of MAFbx mRNA, but not MuRF1 mRNA, in the vastus lateralis muscle was observed [172]; the same results were observed during a 14-day immobilization of the legs [173]. On the other hand, an elevated expression of MuRF1 mRNA, but not of MAFbx mRNA, was observed from days 1 to 10 of leg immobilization [121]. Moreover, the MuRF1 protein expression was found to increase in the soleus muscle but not in the vastus lateralis muscle after 60 days of bed rest [174].
The identification of “atrogenes”—genes that are activated independently by atrophy stimulus (denervation, unloading, thermal damage)—attracted much attention from many laboratories [161,173,175]. This led researchers to identify the FOXO transcription factors (FOXO1 and FOXO3) as the main regulators of the expression of both MuRF1 and MAFbx [176,177]. It is noteworthy that the functioning of the FOXO factors is determined by their cellular location; they are regulated via the IGF-1-PI3K-Akt signaling pathway [178,179]. Under normal physiological conditions, Akt phosphorylates FOXO at specific threonine and serine residues, which leads to the retention of FOXO in the cytoplasm instead of translocation to the nucleus [178]. Under conditions of muscle unloading, dephosphorylated FOXO translocates into the nucleus and activates several different types of atrogenes (E3 ligases) or genes associated with autophagy [177]. Several studies reported that levels of FOXO1 or FOXO3 mRNA and protein expression in “slow-twitch” soleus and “fast-twitch” gastrocnemius and plantar muscles are increased in various models of muscle atrophy [163,180,181,182,183]. Sandri et al. found that constitutively active FOXO3 acts on the MAFbx promoter, inducing MAFbx transcription and the severe atrophy of muscle fibers. In addition, they showed that the stimulation of two main proteolytic ubiquitin ligases (MuRF1/MAFbx) through the overexpression of FOXO1 and FOXO3 leads to an additional reduction in muscle mass and strength during atrophy caused by functional unloading [176]. Moreover, direct or indirect inhibition of the transcriptional activity of FOXO and its interaction with other transcription factors results in a decrease in muscle atrophy [184,185,186]. It is interesting that constitutively active FOXO3 also controls the stimulation of the autophagic/lysosomal proteolysis pathway, thus leading to muscle atrophy in fasting and denervation models [187].
The third mechanism of proteolysis in skeletal muscle is autophagy. Autophagy is responsible for the removal of unfolded, damaged and dysfunctional proteins and organelles through the formation of autophagosomes for degradation by lysosomes [155]. By the 24th day of bed rest, the activation of autophagy markers such as beclin-1 was detected, suggesting an increase in the formation of autophagosomes and therefore a higher activity of macroautophagosomes (autophagosomes for cleaving organelles); however, other markers of autophagy, such as P62, the ratio of LC3II/I and cathepsin-L, were not activated [12].
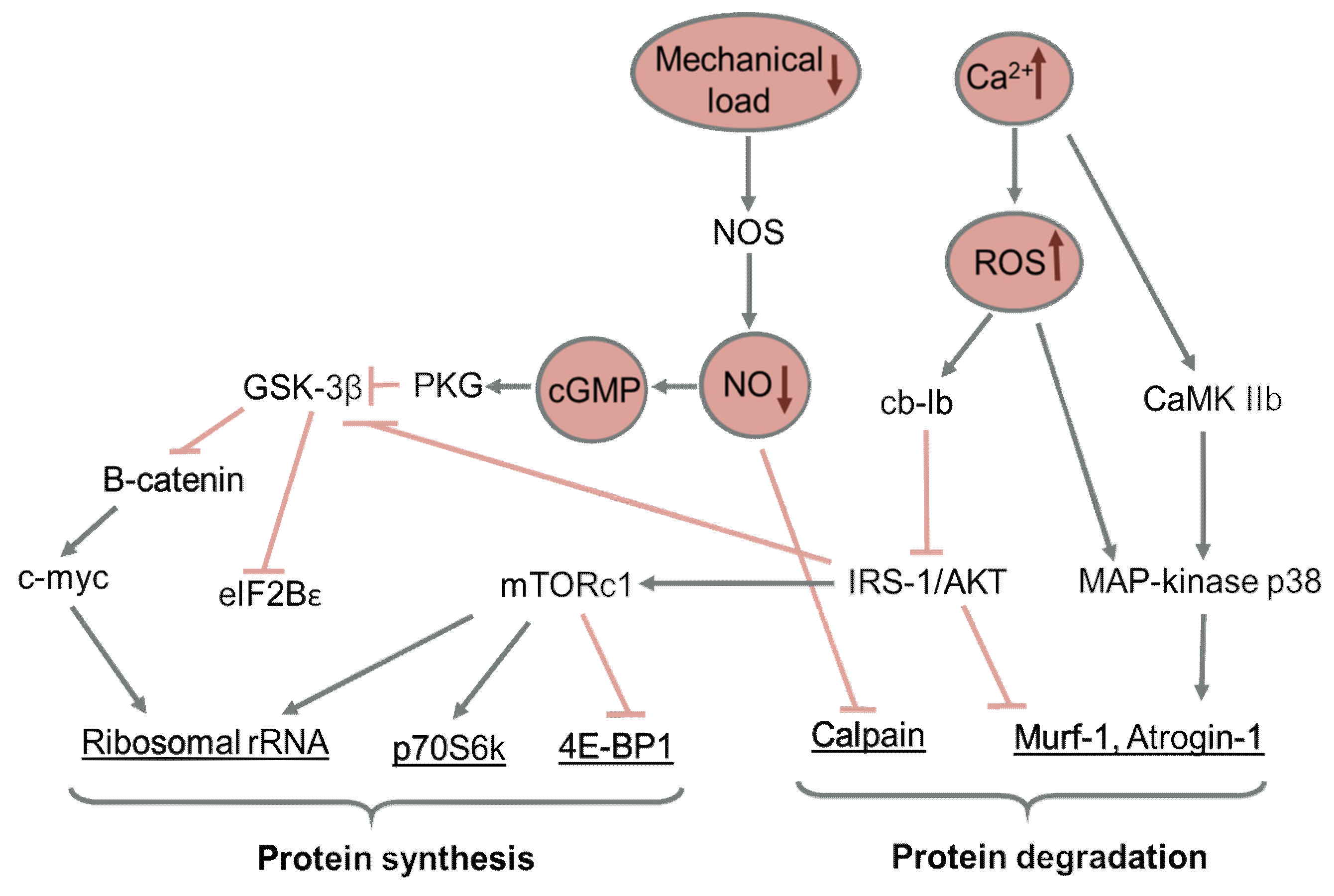
A scheme summarizing the key signaling pathways regulating protein turnover during muscle disuse is presented in Figure 2.
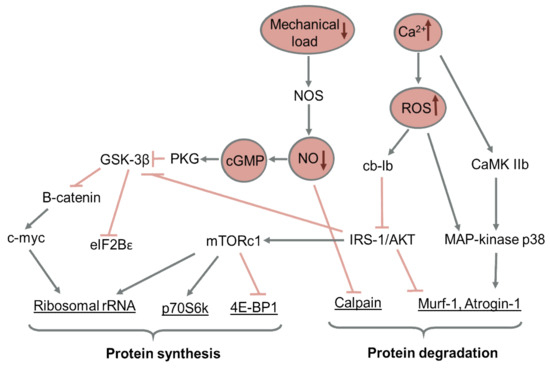
Figure 2.
Key signaling pathways regulating protein turnover during muscle disuse. Pointed arrows show activation; blocked arrows show inhibition.
3.3. Effect of Reduced Activity on Changes in Muscle Fiber Types
As mentioned above, skeletal muscle fibers are divided into two types, “fast-twitch” and “slow-twitch”. “Fast-twitch” muscle fibers demonstrate a high strength and speed of contraction, along with rapid fatigability. “Slow-twitch” muscle fibers show low fatigability and a relatively low maximum strength and speed of contraction. The properties of muscle fibers are determined by the predominance of one or another isoform of myosin heavy chains (MyHCs) in the fibers. In the skeletal muscle of mammals, there are four main isoforms of MyHCs: the “slow-twitch” isoform MyHC I (β) and three “fast-twitch” isoforms IIa, IId/x- and IIb- [45]. In humans, the MyHC isoform IIb is absent [45]. Functional unloading of skeletal muscles caused by the absence of contractile activity leads to a shift in the myosin phenotype, i.e., slow-twitch MyHC isoform expression decreases and fast-twitch isoform expression increases. This slow-to-fast fiber-type shift has been observed as early as after 1 week of “dry” immersion [188]. In astronauts, a decrease in the proportion of slow-twitch skeletal muscle fibers was observed after a 6-month space flight [189] and an 11-day flight [190]. The transformation of the myosin phenotype to the “fast-twitch” type was also observed in volunteers after 84-day bed rest [10] and 35-day bed rest [191]. To the greatest extent, the slow-to-fast transformation of muscle fibers is manifested when the spinal cord is damaged [192].
In an animal model of gravitational unloading, mRNA of the slow-twitch MyHC isoform was observed in the soleus muscle already after the 1st day of hindlimb unloading [118,193]. Moreover, after the 1st day of hindlimb unloading of the rodents, the expression of mRNA of the “fast-twitch” MyHC isoforms IId/x and IIb increased in the soleus muscle and remained elevated for at least 28 days of hindlimb unloading [193,194]. After the 7th day of hindlimb unloading and later, numerous studies showed a significant decrease in the expression of the slow-twitch MyHC isoform both at the mRNA and protein levels [149,150,193,194,195]. From day 3 to the end of the 1st week of hindlimb unloading, the expression of MyHC IIa mRNA was decreased, but after that its expression began to increase and after 14 days of hindlimb unloading it was as high as the control values [194,196].
Studies of the reasons for the change in the expression pattern of myosin genes in the conditions of functional unloading have been carried out mainly on models of the hindlimb unloading of rodents. A key role in the decrease of the slow-twitch MyHC isoform during hindlimb unloading is played by the AMPK/HDAC4 and NO/GSK-3/NFATc1 signaling pathways. Both of these signaling pathways are activated when a muscle is activated The AMP-activated protein kinase (AMPK) is activated upon the consumption of ATP in muscle and the accumulation of ADP and especially AMP, as well as upon a decrease in glycogen content in the muscle; activated AMPK phosphorylates histone deacetylase 4 (HDAC4) is the slow-twitch MyHC transcriptional repressor, and removes HDAC4 from the muscle nuclei. The administration of an AMPK activator AICAR on the 1st day of hindlimb unloading prevents both the accumulation of histone deacetylase 4 in the muscle nuclei of the soleus muscle and a decrease in the expression of mRNA of the slow-twitch MyHC isoform [118]. The application of β-guanidine propionic acid, an activator of AMPK which decreases the ATP/ADP+AMP ratio, prevents the slow-to-fast transformation of the myosin phenotype of soleus muscles after 10 days of hindlimb unloading [197].
The second pathway regulating the expression of slow-twitch MyHC fibers under the conditions of functional unloading is the NO/GSK-3/NFATc1 pathway. NFATc1 (nuclear factor of activated T-cells, cytoplasmic 1) is a transcription factor that, when in the nucleus, binds to the promoter of the gene encoding the slow-twitch MyHC isoform and activates its expression. NFATc1 can be phosphorylated and thereby removed from the nucleus by protein kinases such as GSK-3β, MAP kinase p38 and JNK [198,199]. Calcium-dependent phosphatase calcineurin dephosphorylates NFATc1, which leads to its return to the muscle nuclei and the restoration of its ability to activate the expression of the gene encoding the slow-twitch MyHC isoform.
In experiments with fluorescent reporters of the transcriptional activity of NFATc1 in vivo in skeletal muscles of rats, NFATc1 was shown to be a sensor of neuromuscular activity of skeletal muscles: the level of NFATc1-dependent transcription is higher in slow-twitch muscles than in fast-twitch ones, whereas in denervated slow-twitch muscles its transcriptional activity decreases, but after a low-frequency electrical stimulation of denervated muscles it is restored [200].
The content of NFATc1 in muscle nuclei, as well as its transcriptional activity, decrease already on the 1st day of hindlimb unloading, which is accompanied by GSK-3β kinase activation [81]. By the 7th day of hindlimb unloading, the content of NFATc1 in the muscle nuclei and its transcriptional activity are also reduced; the pharmacological blocking of GSK-3β kinase or an increase in the level of nitric oxide in the muscles both lead to the restoration of the NFATc1 content in the muscle nuclei and to the restoration of slow-twitch MyHC isoform expression [150,201]. It should be noted that mechanical stimulation of the support zones of the feet restores the relative proportion of fast-twitch and slow-twitch fibers and leads to the restoration of NFATc1 myonuclear content and its transcriptional activity, and this effect depends on the activity of the NO synthase [152]. The MAP kinase p38 also contributes to NFAT-dependent transcriptional activity and to the expression of the slow-twitch MyHC isoform during the hindlimb unloading of rats [202].
3.4. Effect of Reduced Muscle Activity on Muscle Oxidative Capacity
Aerobic performance is the ability to perform long-term work (several minutes/tens of minutes), the energy supply of which is mainly provided due to oxidative reactions. Aerobic performance depends on the ability of the cardiovascular system to deliver O2 to skeletal muscles and on the ability of muscles to utilize the O2 [203]. In untrained people and people with reduced muscle functional capacities, aerobic performance is limited first of all by the low oxidative capacity of skeletal muscles and their high fatigability, rather than by the oxygen delivery [204].
Muscle capillary density is one of the key parameters affecting the diffusion of O2 to the mitochondria of muscle fibers [203]. Most studies have shown a decrease in the number of capillaries per fiber [174,205,206] and capillary density, [207,208,209,210] in the vastus lateralis muscle in humans after several weeks of restricted physical activity, whereas other studies did not detect any changes in these parameters [211]. In accordance with these data, three weeks of one-leg immobilization caused multidirectional changes in the expression of genes associated with the regulation of angiogenesis in the vastus lateralis muscle; less than half of them (including the key regulators of angiogenesis VEGFA and VEGFB) decreased their expression, whereas the remaining genes’ expression increased [212]. Numerous studies have convincingly shown that in human muscles, a decrease in motor activity is accompanied by a decrease in the density of mitochondria and the maximum respiration rate, which negatively affects the maximum rate of oxidation of fats and carbohydrates and reduces aerobic performance at the level of the whole organism (see reviews [213,214]). Two-dimensional gel electrophoresis and mass spectrometric analysis made it possible to study changes in the content of several tens of muscle proteins. So, after 8 and 35 days, as well as for longer periods (55 and 65 days), of bed rest in the vastus lateralis muscle, a similar decrease in the content of several enzymes of the Krebs cycle and oxidative phosphorylation was observed [12,215,216]. That is, with a decrease in motor activity, the oxidative capacities of skeletal muscles rapidly decrease and then are kept at the achieved level. It is interesting to note that after two days of leg immobilization in the vastus lateralis muscle, the expression of some genes encoding mitochondrial proteins decreases [170]. With an increase in the duration of hypokinesia, the expression of several tens or even hundreds of genes encoding mitochondrial proteins decreases—these include enzymes of the tricarboxylic acid cycle, electron transport chain, ubiquinone biosynthesis, as well as beta-oxidation enzymes (HADH, ACADL, ACADM, ACADVL) and fatty acid transporters (CD36) [170,217,218,219,220,221,222].
The regulation of fiber metabolism is closely related to the slow-to-fast fiber-type transformation. In general, slow-twitch fibers in humans contain a greater number of mitochondria and the content and activity of respiratory enzymes than fast-twitch fibers. Under various types of unloading conditions mitochondria undergo alterations in function (including a decreased respiratory rate and increase in ROS production and a decline in mitochondrial biogenesis and in activated mytophagy) [223]. At the same time, the markers of mitochondrial fusion and fission are both downregulated, but the changes in fusion markers are usually more profound than in fission markers, thereby activating mitochondrial fission [223]. Alterations of mitochondrial functioning precede the downregulation of mitochondrial biogenesis, occurring as early as after 24 h of muscle immobilization [224]. A number of reviews describe the causes of the increased ROS production by mitochondria in the unloaded muscles, and among the main causes of these alterations are increases in myoplasmic and intramitochondrial calcium levels [223,225]. However, there is a lack of experimental data directly showing the mechanisms connecting unloading-induced cytoplasmic calcium accumulation and the production of ROS by mitochondria. Moreover, the regulation of mitochondrial functioning, dynamics and biogenesis may be controlled by different signaling mechanisms, although all these parameters contribute to muscle oxidative capacity. Some evidence shows that calpain activation, as well as an increase in the ATP/ADP ratio, also contribute to alterations in mitochondrial functions under various disuse conditions [197,226]. Many authors suggest that mitochondrial alterations, especially PGC-1α inactivation, induced by muscle inactivity are the main cause of the unloading-induced muscle atrophy [113,227]; however, we did not detect any countermeasure effect of PGC-1α expression decrease prevention on soleus muscle weight in the hindlimb-unloading experiments conducted in our laboratory [150]. However, the most studied pathway regulating mitochondrial oxidative capacity under disuse conditions is still the PGC-1α-dependent pathway.
In humans, the content of enzymes of the mitochondrial respiratory chain in the vastus lateralis muscle decreases already after 10 days of bed rest [228]; a decrease in the expression of the key regulator of mitochondrial biogenesis, PGC-1α, is also observed [229]. A decrease in PGC-1α expression and the content of the mitochondrial respiratory chain enzymes, along with the disruption of mitochondrial functions, are observed in the muscles of patients in intensive care; however, it is not known whether this decrease is caused by the functional unloading of the muscles or concomitant factors [207,230]. By the 3rd day of hindlimb unloading, the content of PGC-1α in the soleus muscle of rats significantly decreases [83]. Both after 7 and 14 days of hindlimb unloading, the expression and content of PGC-1α in the soleus muscles remains decreased, as does the content of enzymes of the mitochondrial respiratory chain [150,231,232]. Studies on transgenic animals show that PGC-1α is closely related to muscle fatigue resistance [233,234]. Thus, a decrease in the expression of PGC-1α, along with the slow-to-fast transformation of skeletal muscle fibers, can be the cause of increased muscle fatigue during functional unloading.
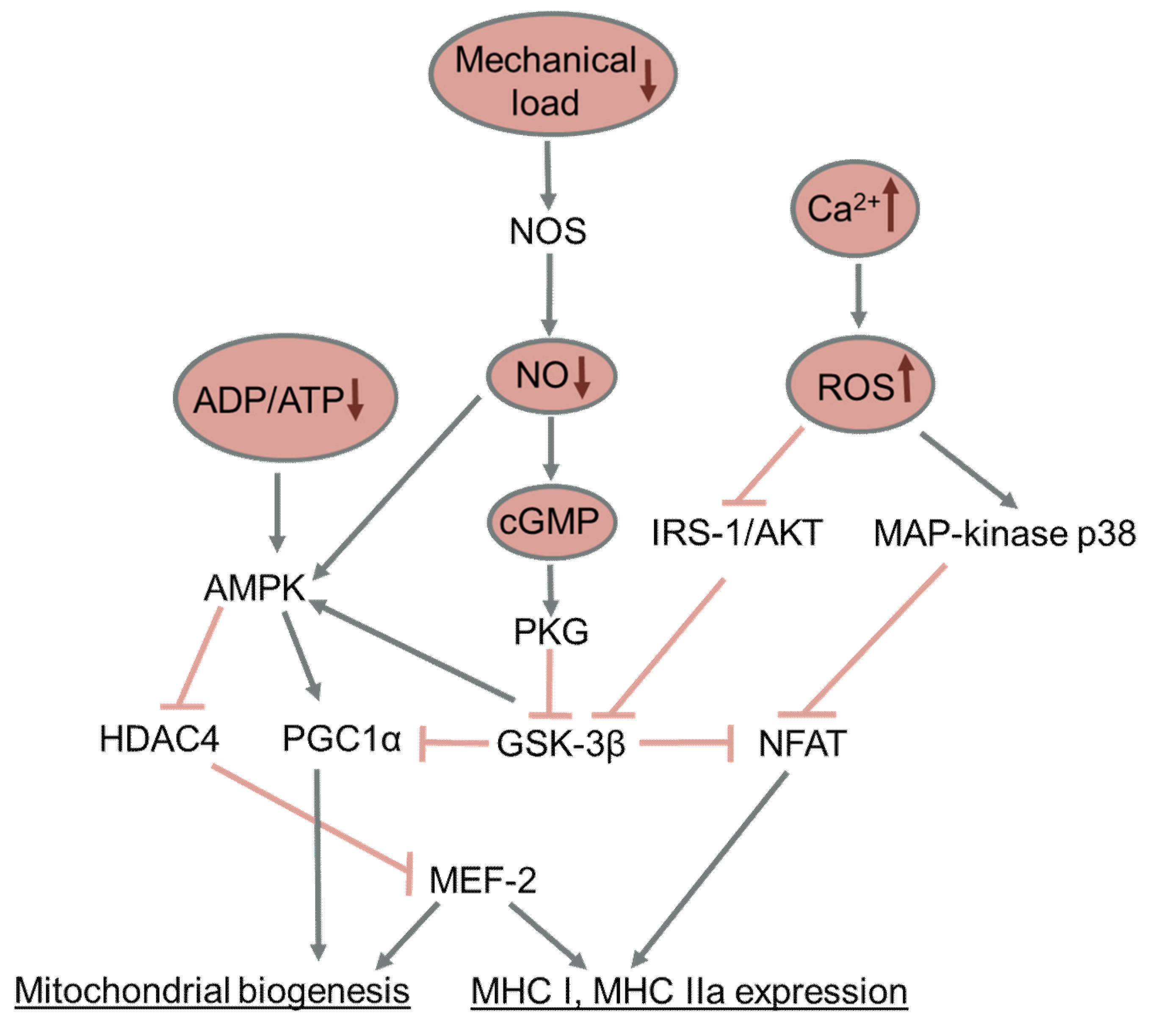
Little is known about the molecular mechanisms of the decreased expression of PGC-1α under the conditions of functional unloading. Inhibition of GSK-3β during 2-week hindlimb unloading was shown to prevent a decrease in the content of PGC-1α and components of the mitochondrial respiratory chain [231]. The administration of nitric oxide donors during hindlimb unloading and mechanical stimulation of the support zones of the feet both prevented the activation of GSK-3β and decreased the expression of PGC-1α, along with the prevention of a decrease in the expression of the slow-twitch MyHC isoform [83,150]. It was shown on myotubes C2C12 that PGC-1α expression can be regulated via the action of GSK-3β on the TFEB transcription factor [235]. It was also shown that the slow-twitch MyHC isoform can regulate the expression of PGC-1α by means of micro-RNA-499 [236]. Thus, GSK-3β kinase is a key regulator linking the activity of skeletal muscle and the slow-twitch, oxidative phenotype of muscle fibers, which determines muscle resistance to fatigue. The key signaling pathways affecting slow-to-fast fiber-type transformation and mitochondrial biogenesis are summarized in Figure 3.
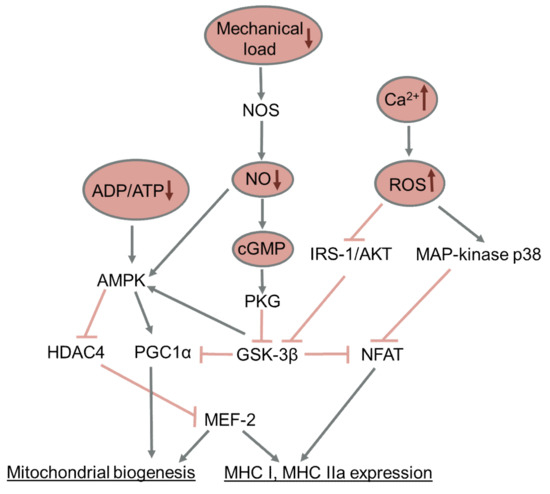
Figure 3.
Key signaling pathways regulating slow-to-fast fiber-type transformation and mitochondrial biogenesis during muscle disuse. Pointed arrows show activation; blocked arrows show inhibition.
4. Countermeasures to Changes Caused by Reduced Motor Activity
As can be seen from the above, the several negative effects of chronic decrease on the contractile activity of the muscles have a dramatic effect on their work capacity and lead to profound systemic changes that threaten human health and quality of life. It is clear that it is necessary to use a complex of measures to prevent the negative consequences of complete or partial muscle inactivation. Great progress in the development of such a complex has been achieved in space medicine.
Based on the results of studies carried out for the medical support of space flights, as well as international studies of the effects of microgravity on the musculoskeletal system of humans and animals, Professor I.B. Kozlovskaya developed a number of theoretical generalizations that made it possible to systematically analyze the effects of microgravity and create the appropriate countermeasures. According to her theory, the key effect of microgravity on the musculoskeletal system is the elimination of support afferentation [23]. The elimination of support afferentation leads to inactivation of the postural muscles motor units, which in turn leads to atrophy, atony, transformation of the myosin phenotype and subsequent functional disorders of skeletal muscles [23,237]. At the same time, the mechanical loading of skeletal muscles also contributes to atrophy of both slow-twitch and fast-twitch muscle fibers [1,238]. Countermeasures against the disorders caused by muscle disuse are based on providing support afferentation and/or mechanical loads for muscles in one way or another. To provide a mechanical load, it is recommended to use various exercise systems and devices that provide an axial load (which are suited to providing a load on the muscles and the force of acceleration in centrifuges) [239]. For the restoration of support afferentation and, accordingly, muscle activity, there are devices that stimulate the mechanoreceptors of the feet, which lead to neuromuscular activation [240,241]. Electrical stimulation makes it possible to directly activate fast-twitch and slow-twitch motor units in accordance with the impulse patterns specific to a particular type of muscle fibers and thus activate the muscle in the absence of load, which provides an effect similar to the restoration of support afferentation [25]. In recent years, methods of pharmacological countermeasures against the effects of microgravity are also developing, which make it possible to reproduce the effects of muscle activity and/or loading on various intramuscular molecular pathways, but these methods are still rarely used in clinical practice [150,231,242]. A classification of countermeasure methods according to the sensory system they affect is listed in Table 2.

Table 2.
Main countermeasure methods used to prevent unloading-induced muscle alterations, and the sensory structures that respond to them.
Countermeasures against functional and structural changes in skeletal muscles caused by gravitational unloading play an important role in the medical support of manned space flights. Numerous studies conducted with the participation of cosmonauts and astronauts on the orbital stations Salyut, Mir and the International Space Station, as well as under the simulated microgravity conditions on Earth, show that the changes caused by microgravity can be partially or completely prevented by means of countermeasures [240,241,243,244].
In the 2010s, the “Profilaktika” experiment was carried out on the ISS with the participation of 15 Russian cosmonauts. Within the framework of this experiment, two protocols of countermeasures were compared—intensive interval exercise combining aerobic and anaerobic loading zones, and prolonged low intensive aerobic exercise. Interval exercise in space flight led to the preservation of muscle function almost at the pre-flight level [243]. In 2020, the effectiveness of high-intensity exercise on orbit was confirmed in an experiment by NASA [244].
The workout system of Russian cosmonauts in orbit includes two 1-h sessions during the day, with three days of exercise, followed by one day of rest. The exercise includes treadmill running and bicycle ergometer cycling, with the energy cost ranging from 380 to 600 kcal per day [245]. Since not all cosmonauts perform the prescribed exercises in full throughout the entire flight, the most informative studies are those of the effectiveness of the countermeasures against microgravity-induced disorders in terrestrial conditions, when the effects of microgravity are simulated using HDT BR, unilateral lower limb suspension and “dry” immersion.
Intense exercise on a bicycle ergometer was not found to be efficient in preventing changes caused by functional unloading in the lower leg muscles [246]. At the same time, high-intensity jumping exercises prevented a decrease in maximum muscle strength and in muscle mass after 60-day HDT BR [208], whereas resistive exercises prevented atrophy of vastus lateralis muscle fibers after a 14-day bed rest [29], which correlates well with data indicating the higher efficiency of high-intensity workouts under weightlessness conditions. Nevertheless, high-intensity jumping exercises during a 60-day HDT BR had a rather weak countermeasure effect on changes in the myosin phenotype of fibers and the decrease in the oxidative capacity of the lower limbs’ skeletal muscles [209].
In addition to exercise, there are other methods of increasing the level of muscles’ mechanical loading. The use of the “Penguin” axial loading suit during a 2-month and 4-month HDT BR prevented the atrophy of the slow-twitch soleus muscle fibers [247]. Vibration stimulation of the plantar fascia during rat hindlimb unloading or cast immobilization leads to a partial prevention of muscle fiber atrophy, but does not prevent the slow-to-fast fiber-type transformation [210,248]. In 2019, vibration stimulation of the plantar fascia was shown to activate the mechano-dependent regulators YAP1 and ERK [248], which suggests that the effects of vibration stimulation are caused not only by the activation of sole proprioceptors and the restoration of muscle activity, but also via the activation of muscle mechanosensors and by triggering signaling cascades that respond to loading. The use of massage as a method of activating muscle mechanoreceptors increased the level of protein metabolism in the gastrocnemius muscle of rats during 7 days of hindlimb unloading, but did not lead to the atrophy prevention or a decrease in the rate of degradation of cytoskeletal proteins [249].
It some studies, it was shown that reductions in skeletal muscle mass and functional properties could be prevented through the application of artificial gravity via centrifugation. The use of artificial gravity by rotation of the test subjects in a centrifuge with an acceleration of 2.5 G in the area of the feet for one hour per day prevented a decrease in protein synthesis after 21-day HDT BR, as well as soleus and vastus lateralis atrophy and the “fast-twitch” transformation of soleus muscle fibers [250,251]. Another work shows that intermittent centrifugation can partially maintain the mass of the rat soleus muscle during unloading [252]. After a 5-day HDBR, there was a positive effect on the maximum strength of leg muscle contractions and the height of jumps in groups with a 30-min centrifugation in a short-radius centrifuge, as well as with six 5-min centrifugation sessions [253]. In the experiments with rodents, it was shown that 30-day 2G centrifugation induces a distinct anabolic response in mouse soleus and tibialis anterior muscles [254]. Unfortunately, there are very few studies on the effect of artificial gravity as a countermeasure against changes caused by functional unloading.
Another approach to countermeasures is the restoration of support afferentation, which leads to the activation of skeletal muscles’ motor units (that is, the muscle fiber and the motor neuron that innervates it). The effects of support afferentation on the state of the human musculoskeletal system were first shown in the 1983 Soviet–Cuban experiment during a 7-day space flight [32]. In a subsequent experimental series using the “dry” immersion model, it was found that the use of mechanical stimulation of the support zones of the feet allows the maintenance of a normal level of electrical activity and reflex tonus of the skeletal muscle [188]. In this experimental series, a pressure of 40 kPa was applied to a foot daily during 6-h sessions, with a regimen of a 20-min stimulation, followed by a 40-min rest period. The stimulation comprised two modes, corresponding to slow walking (75 steps per min) and fast walking (120 steps per min), with each mode implemented for 10 min. Mechanical stimulation of the support zones of the feet prevented the atrophy of soleus muscle slow-twitch fibers, as well as slow-to-fast fiber-type transformation after a 7-day “dry” immersion and partially prevented the degradation of cytoskeletal proteins [26,31,255]. Studies in rats with mechanical stimulation of the feet support zones during hindlimb unloading showed that the restoration of support afferentation prevents a change in the pattern of myosin gene expression, a decrease in the level of PGC-1α expression and protein synthesis, an increase in ubiquitin ligase expression under conditions of 1 and 3 days of functional unloading, as well as preventing the atrophy of the soleus muscle [81,83,256]. Recent studies show that a key role in most of the countermeasure effects of support stimulation is played by the activity of neuronal NO synthase [85,152].
An effect comparable to the restoration of support afferentation for slow-twitch skeletal fibers is exerted by electrical stimulation of skeletal muscle. Electrical stimulation is based on the restoration of the normal electrical activity of muscles using electric currents with characteristics similar to the patterns of fast-twitch (50–60 Hz) or slow-twitch (10 Hz) innervation. In sports medicine, however, higher-frequency stimulation patterns are used (up to 100 Hz) [257]. Electrical stimulation is employed both directly (electrodes are applied to the skin or to the muscle and the current directly impacts the muscle) and indirectly (by stimulating the nerve that innervates the muscle). Electrical stimulation leads to the activation of muscle fibers and thus allows one to maintain muscle activity. The use of electrical stimulation as a countermeasure against changes caused by functional unloading is the only possible measure for patients who for some reason cannot perform physical exercises, for example, for those who are unconscious. A randomized study showed the effectiveness of high-frequency (45 Hz) electrical stimulation in the prevention of quadriceps femoris atrophy of patients in intensive care [258]. High-frequency stimulation (50 Hz) led to hypertrophy of “fast-twitch” muscle fibers in the vastus lateralis muscle in cancer patients undergoing chemotherapy, but did not prevent a reduction in the number of mitochondria [259]. In sports medicine, electrical stimulation is often used to help an athlete to recover after injuries, after surgery on the ligaments or during cast immobilization. The use of stimulation with a frequency of 50 Hz in athletes after surgery for the restoration of the anterior cruciate knee ligament led to the partial prevention of a decrease in quadriceps strength, but did not prevent the atrophy of the studied muscle [260]. Sessions of electrical stimulation with a frequency of 30 Hz throughout 40 days of leg immobilization due to a fracture of the lower leg led to prevention of a decrease in the level of protein synthesis and atrophy of the quadriceps in comparison with the contralateral leg [132]. High-frequency stimulation also prevented an increase in the expression of ubiquitin ligases MuRF-1, MaFbx and myostatin during a 4-day leg immobilization in the knee joint [18].
Low-frequency electrical stimulation is used in the rehabilitation of patients with chronic systemic damage of skeletal muscles, for example, as a result of spinal cord injury. Unlike high-frequency stimulation, low-frequency stimulation primarily affects the mitochondrial enzyme content and oxidative capacity of the muscle, as well as muscle resistance to fatigue. Studies in rodents conducted in the 1980s showed that chronic low-frequency stimulation leads to the fast-to-slow fiber-type transformation, as well as increases in capillarization, the level of mitochondrial enzymes and muscle resistance to fatigue [261]. An increase in resistance to fatigue with low-frequency stimulation was also observed in the tibialis anterior muscle in humans [262].
Low-frequency electrical stimulation (2–10 Hz) leads to an increase in the expression of the key factor of mitochondrial biogenesis, PGC-1α, in the vastus lateralis muscle of paralyzed patients with spinal cord injury 3 h after the end of the stimulation session [263]. In patients with chronic heart failure, low-frequency (15 Hz) electrical stimulation led to an increase in maximum oxygen consumption (VO2), an increase in citrate synthase activity, and transformation of the vastus lateralis muscle fibers to the “slow-twitch” fiber type [264].
In the 1970s, Soviet scientists carried out studies on the effects of electrical stimulation during prolonged bed rest. For instance, in two series of studies the efficacy of electrostimulation training was assessed using a “Tonus-2” apparatus [265]. In the first series, two different modes of electromyostimulation were used on two groups of volunteers (each consisting of four people) subjected to 49-day bed rest, whereas the volunteers of the third (control) group were under conditions of bed rest without additional influences. The volunteers of the control group had a significant decrease in the cross-sectional area of the muscle fibers of all types in soleus muscle. The size of the muscle fibers of the volunteers subjected to electromyostimulation did not change. Currently, Russian cosmonauts in orbit use the Tonus-3 electrostimulator for stimulation of the quadriceps, abdominal muscles and muscles of the neck, back and shoulders at a frequency of 60 Hz and Stimul-01-LF for muscle stimulation at a frequency of 50 to 25 Hz, depending on the operating mode [241].
However, the use of low-frequency stimulation as a countermeasure against changes caused by functional unloading has certain disadvantages; in particular, the use of chronic 15 Hz stimulation in healthy volunteers may decrease the maximum voluntary strength of knee extensors and the size of muscle fibers [266]. Nevertheless, electrical stimulation in combination with muscle stretching in this experiment prevented the decreases in the size of the fibers and muscle strength.
Studies of changes in molecular regulatory mechanisms during the electrical stimulation of skeletal muscles have mainly been performed on the model of rodent hindlimb unloading and on cultures of myoblasts. Among the triggering mechanisms determining the response of muscle fibers to low-frequency electrical stimulation, various authors mention a decrease in the ATP/ADP ratio, which leads to the activation of AMP-activated protein kinase and calcium ion accumulation, activating the calcineurin/NFATc1 signaling pathway in the myoplasm of skeletal muscle fibers [267,268,269]. The effect of low-frequency electrical stimulation on soleus muscle atrophy during rat hindlimb unloading differs depending on the mode of electrical stimulation. A study with 8-h stimulation reported the partial prevention of atrophy and a decrease in the stiffness of the rat soleus muscle [270], whereas studies with continuous electrical stimulation did not find any prevention of atrophy [109,271]. At the same time, the prevention of the slow-to-fast fiber-type transformation was observed in all three of the above studies. However, the poor effect of continuous electrical stimulation is rather predictable, as it is well-known that the processes of protein synthesis in skeletal muscles are activated during the periods of rest when muscle does not contract.
The least-studied type of countermeasures against disorders caused by muscle functional unloading is the use of various pharmaceutical drugs and food additives. Attempts to prevent muscle atrophy under conditions of bed rest with amino acid or protein supplements have showed contradictory results. Some researchers believe that the use of protein supplements in excess of the recommended intake of 1 g of protein per 1 kg of body weight per day does not lead to the prevention of unloading-induced atrophy. Accordingly, in those studies which detected a positive effect of protein supplements on muscle state, it was caused by replenishing the protein deficiency in the diet of test subjects [272]. Nevertheless, it was shown that the use of amino acid supplements during immobilization and bed rest has a positive effect on protein synthesis, but only partially prevents atrophy [15,273]. The supplementation of food with essential amino acids partially prevents atrophy during bed rest [273,274]. The combination of resistive exercise and the supplementation of food with essential amino acids during a 28-day bed rest effectively prevented a decrease in the mass and strength of the thigh muscles of test subjects [275]. Interestingly, testosterone treatment (especially in combination with exercise) is effective to prevent decreases in lean body mass and muscle strength, aerobic performance and the muscle content of some structural and mitochondrial proteins under conditions of 70-day bed rest. The literature on the use of antioxidants as countermeasure food additives against changes in skeletal muscles under functional unloading is also very controversial. Despite the fact that an increase in the level of reactive oxygen species in various forms of functional unloading has been shown in numerous studies, the main source of reactive oxygen species during unloading has not yet been reliably identified [276]. Antioxidants such as vitamin E, curcumin, astaxanthin and green tea extract have shown both the partial prevention of atrophy during immobilization or hindlimb unloading and the absence of any effect, depending on the dose of the antioxidant and the stage and model of functional unloading [277,278,279].
In recent years, research has begun on pharmaceutical drugs that modulate various molecular signaling pathways involved in the muscle response to functional unloading. The use of such drugs in humans is limited, since it is necessary for these substances to be preliminarily tested for safety. However, there are a number of promising pharmaceuticals already in clinical use in other fields that are also capable of preventing some of the effects of functional unloading. These pharmaceuticals include the p38 MAP kinase inhibitor; its use during 3-day hindlimb unloading in rats led both to the prevention of atrophy and to the prevention of a decrease in the expression of PGC-1α and mRNA of the slow-twitch myosin isoform [166,202]. The use of GSK-3β kinase inhibitors during hindlimb unloading in rats led to the prevention of a decrease in the expression of PGC-1α and mitochondrial proteins and the prevention of the slow-to-fast fiber-type transformation [201,231]. In recent years, GSK-3β was shown to play a significant role in the development of changes in postural muscles during variants of muscle unloading; the inhibition of GSK-3 can lead to an increase in the work capacity of skeletal muscles in rodents, which makes GSK-3β inhibitors some of the most promising pharmacological agents as a countermeasure against unloading-induced changes [280,281].
5. Conclusions
The functional unloading of skeletal muscles occurs both in the conditions of space flight and, in one form or another, in terrestrial conditions. To date, all the forms of the functional unloading of skeletal muscles are known to result in muscle atrophy, and the transformation of fibers from the “slow-twitch” type to the “fast-twitch” type, which leads to a decrease in the maximum strength and speed of muscle contraction, as well as to a decrease in their work capacity. The signaling mechanisms of these changes, as well as the triggers that cause them, are far from fully known; there are many questions that still remain open, e.g., about the relationship between the role of muscle electrical activity and its mechanical loading in the development of changes caused by unloading, about signaling mechanisms that initiate a decrease in protein synthesis and an increase in its degradation, as well as a decrease in the oxidative capacity of skeletal muscles. The use of muscle electrical stimulation and mechanical stimulation of the support zones of the feet allows us to prevent a number of negative changes caused by functional unloading, but only in recent years is it beginning to become clear through which molecular mechanisms these countermeasures work. To date, however, enough data have been accumulated that it is now possible to commence the development of pharmacological methods for the correction of changes caused by functional unloading by targeting the signaling pathways that control protein synthesis and degradation, as well as the myosin phenotype and mitochondrial biogenesis in skeletal muscles. Further studies of the molecular mechanisms that trigger skeletal muscle changes occurring during unloading will allow us to expand the range of possible pharmacological targets and physiotherapeutic methods, as well as to improve existing methods and maintain human health in conditions of reduced or completely absent skeletal muscle activity.
Author Contributions
K.S., E.T.; D.V.P., A.A.S., S.A.T. and B.S.S. wrote the manuscript, prepared the tables and figures and approved the final version of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The study was supported by the Ministry of Science and Higher Education of the Russian Federation under agreement No. 075-1502020-919 from 16 November 2020 about the grant in the form of subsidy from the federal budget to provide government support for the creation and development of a world-class research center, the “Pavlov Center for Integrative Physiology to Medicine, High-tech Healthcare and Stress Tolerance Technologies”.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the interpretation of data; in the writing of the manuscript, or in the decision to publish the review.
Abbreviations
| 4E-BP1 | eukaryotic initiation factor 4E binding protein |
| AKT | protein kinase B |
| AMPK | AMP-activated protein kinase |
| cGMP | cyclic guanosine monophosphate |
| c-Myc | c-myelocytomatosis oncogene (transcription factor) |
| CSA | cross-sectional area |
| eIF2B | eukaryotic initiation factor 2B |
| FoxO | forkhead box O protein |
| GS1 | glycogen synthase-1 |
| GSK-3β | glycogen synthase kinase-3β |
| HDT BR | head down tilt bed rest |
| HS | hindlimb suspension |
| HU | hindlimb unloading |
| IGF-1 | insulin-like growth factor 1 |
| L-NAME | N(gamma)-nitro-L-arginine methyl ester |
| MAFbx | muscle atrophy F-box protein/atrogin 1 |
| MAPK | mitogen-activated protein kinase |
| MCIP 1.4 | modulatory calcineurin-interaction protein 1.4 |
| MEF-2 | myocyte enhancer factor 2 |
| mir-208 | micro-RNA 208 |
| mTORC1 | mammalian/mechanistic target of rapamycin complex 1 |
| MuRF1 | muscle RING finger protein |
| MyHC | myosin heavy chain |
| NFAT | nuclear factor of activated T-cells |
| NO | nitric oxide |
| NRF | nuclear respiratory factor |
| p70S6K | ribosomal protein S6 kinase p70 |
| PGC-1α | peroxisome proliferator-activated receptor gamma coactivator 1-alpha |
| PI3K | phosphatidylinositol 3-kinase |
| PKA | protein kinase A |
| PKB | protein kinase B (AKT) |
| PKC | protein kinase C |
| PKD1 | protein kinase D1 |
| PKG | protein kinase G |
| PMS | plantar mechanical stimulation |
| PuRA | purine Rich Element Binding Protein A |
| PuRB | purine Rich Element Binding Protein B |
| rRNA | ribosomal RNA |
| SOX-6 | transcription factor |
| TFAM | mitochondrial transcription factor A |
References
- Falempin, M.; Mounier, Y. Muscle atrophy associated with microgravity in rat: Basic data for countermeasures. Acta Astronaut. 1998, 42, 489–502. [Google Scholar] [CrossRef]
- Powers, S.K.; Lynch, G.S.; Murphy, K.T.; Reid, M.B.; Zijdewind, I. Disease-Induced Skeletal Muscle Atrophy and Fatigue. Med. Sci. Sports Exerc. 2016, 48, 2307–2319. [Google Scholar] [CrossRef]
- Hoffmann, C.; Weigert, C. Skeletal Muscle as an Endocrine Organ: The Role of Myokines in Exercise Adaptations. Cold Spring Harb. Perspect. Med. 2017, 7. [Google Scholar] [CrossRef]
- Shenkman, B.S.; Grigoriev, A.I.; Kozlovskaya, I.B. Gravity mechanisms in tonic motor system. Neurophysiological and muscle aspects. Hum. Physiol. 2017, 43, 578–590. [Google Scholar] [CrossRef]
- Roy, R.R.; Baldwin, K.M.; Edgerton, V.R. The plasticity of skeletal muscle: Effects of neuromuscular activity. Exerc. Sport Sci. Rev. 1991, 19, 269–312. [Google Scholar] [CrossRef]
- Gao, Y.; Arfat, Y.; Wang, H.; Goswami, N. Muscle Atrophy Induced by Mechanical Unloading: Mechanisms and Potential Countermeasures. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef]
- Bodine, S.C. Disuse-induced muscle wasting. Int. J. Biochem. Cell. Biol. 2013, 45, 2200–2208. [Google Scholar] [CrossRef] [PubMed]
- Koryak, Y.A. Influence of simulated microgravity on mechanical properties in the human triceps surae muscle in vivo. I: Effect of 120 days of bed-rest without physical training on human muscle musculo-tendinous stiffness and contractile properties in young women. Eur. J. Appl. Physiol. 2014, 114, 1025–1036. [Google Scholar] [CrossRef]
- Holt, J.A.; Macias, B.R.; Schneider, S.M.; Watenpaugh, D.E.; Lee, S.M.C.; Chang, D.G.; Hargens, A.R. WISE 2005: Aerobic and resistive countermeasures prevent paraspinal muscle deconditioning during 60-day bed rest in women. J. Appl. Physiol. 2016, 120, 1215–1222. [Google Scholar] [CrossRef] [PubMed]
- Trappe, S.; Trappe, T.; Gallagher, P.; Harber, M.; Alkner, B.; Tesch, P. Human single muscle fibre function with 84 day bed-rest and resistance exercise. J. Physiol. 2004, 557, 501–513. [Google Scholar] [CrossRef]
- Rittweger, J.; Frost, H.M.; Schiessl, H.; Ohshima, H.; Alkner, B.; Tesch, P.; Felsenberg, D. Muscle atrophy and bone loss after 90 days’ bed rest and the effects of flywheel resistive exercise and pamidronate: Results from the LTBR study. Bone 2005, 36, 1019–1029. [Google Scholar] [CrossRef]
- Brocca, L.; Cannavino, J.; Coletto, L.; Biolo, G.; Sandri, M.; Bottinelli, R.; Pellegrino, M.A. The time course of the adaptations of human muscle proteome to bed rest and the underlying mechanisms. J. Physiol. 2012, 590, 5211–5230. [Google Scholar] [CrossRef]
- Akima, H.; Kubo, K.; Imai, M.; Kanehisa, H.; Suzuki, Y.; Gunji, A.; Fukunaga, T. Inactivity and muscle: Effect of resistance training during bed rest on muscle size in the lower limb. Acta Physiol. Scand. 2001, 172, 269–278. [Google Scholar] [CrossRef]
- Miokovic, T.; Armbrecht, G.; Gast, U.; Rawer, R.; Roth, H.J.; Runge, M.; Felsenberg, D.; Belavy, D.L. Muscle atrophy, pain, and damage in bed rest reduced by resistive (vibration) exercise. Med. Sci. Sports Exerc. 2014, 46, 1506–1516. [Google Scholar] [CrossRef]
- Glover, E.I.; Phillips, S.M.; Oates, B.R.; Tang, J.E.; Tarnopolsky, M.A.; Selby, A.; Smith, K.; Rennie, M.J. Immobilization induces anabolic resistance in human myofibrillar protein synthesis with low and high dose amino acid infusion. J. Physiol. 2008, 586, 6049–6061. [Google Scholar] [CrossRef] [PubMed]
- Suetta, C.; Hvid, L.G.; Justesen, L.; Christensen, U.; Neergaard, K.; Simonsen, L.; Ortenblad, N.; Magnusson, S.P.; Kjaer, M.; Aagaard, P. Effects of aging on human skeletal muscle after immobilization and retraining. J. Appl. Physiol. 2009, 107, 1172–1180. [Google Scholar] [CrossRef]
- Wall, B.T.; Dirks, M.L.; Snijders, T.; Senden, J.M.; Dolmans, J.; van Loon, L.J. Substantial skeletal muscle loss occurs during only 5 days of disuse. Acta Physiol. 2014, 210, 600–611. [Google Scholar] [CrossRef] [PubMed]
- Dirks, M.L.; Wall, B.T.; Snijders, T.; Ottenbros, C.L.; Verdijk, L.B.; van Loon, L.J. Neuromuscular electrical stimulation prevents muscle disuse atrophy during leg immobilization in humans. Acta Physiol. 2014, 210, 628–641. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, A.; Lin, C.; Shackelford, L.; Sinitsyn, V.; Evans, H.; Belichenko, O.; Schenkman, B.; Kozlovskaya, I.; Oganov, V.; Bakulin, A.; et al. Muscle volume, MRI relaxation times (T2), and body composition after spaceflight. J. Appl. Physiol. 2000, 89, 2158–2164. [Google Scholar] [CrossRef] [PubMed]
- Fitts, R.H.; Trappe, S.W.; Costill, D.L.; Gallagher, P.M.; Creer, A.C.; Colloton, P.A.; Peters, J.R.; Romatowski, J.G.; Bain, J.L.; Riley, D.A. Prolonged space flight-induced alterations in the structure and function of human skeletal muscle fibres. J. Physiol. 2010, 588, 3567–3592. [Google Scholar] [CrossRef] [PubMed]
- Шульженкo, Е.Б.; Виль-Вильямс, И.Ф. Вoзмoжнoсть прoведения длительнoй вoднoй иммерсии метoдoм «сухoгo» пoгружения. Кoсмическая Биoлoгия Авиакoсмическая Медицина 1976, 10, 82–84. [Google Scholar]
- Tomilovskaya, E.; Shigueva, T.; Sayenko, D.; Rukavishnikov, I.; Kozlovskaya, I. Dry Immersion as a Ground-Based Model of Microgravity Physiological Effects. Front. Physiol. 2019, 10, 284. [Google Scholar] [CrossRef]
- Shenkman, B.S.; Kozlovskaya, I.B. Cellular Responses of Human Postural Muscle to Dry Immersion. Front. Physiol. 2019, 10, 187. [Google Scholar] [CrossRef]
- Litvinova, K.S.; Vikhlyantsev, I.M.; Kozlovskaya, I.B.; Podlubnaya, Z.A.; Shenkman, B.S. Effects of artificial support stimulation on fiber and molecular characteristics of soleus muscle in men exposed to 7-day dry immersion. J. Gravit. Physiol. 2004, 11, P131–P132. [Google Scholar] [PubMed]
- Moukhina, A.; Shenkman, B.; Blottner, D.; Nemirovskaya, T.; Lemesheva, Y.; Puttmann, B.; Kozlovskaya, I. Effects of support stimulation on human soleus fiber characteristics during exposure to “dry” immersion. J. Gravit. Physiol. 2004, 11, P137–P138. [Google Scholar] [PubMed]
- Ogneva, I.V.; Shenkman, B.S.; Kozlovskaya, I.B. The contents of desmin and alpha-actinin-1 in the human soleus muscle after seven-day “dry” immersion. Dokl. Biol. Sci. 2011, 436, 20–22. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.F.; Saenko, I.V.; Popov, D.V.; Vinogradova, O.L.; Kozlovskaya, I.B. Effect of mechanical stimulation of the support zones of soles on the muscle stiffness in 7-day dry immersion. J. Gravit. Physiol. 2004, 11, P135–P136. [Google Scholar] [PubMed]
- Kirenskaia, A.V.; Kozlovskaia, I.B.; Sirota, M.G. [Effect of immersion hypokinesia on the characteristics of the rhythmic activity of the motor units of the soleus muscle]. Fiziol. Cheloveka 1986, 12, 627–632. [Google Scholar] [PubMed]
- Bamman, M.M.; Clarke, M.S.; Feeback, D.L.; Talmadge, R.J.; Stevens, B.R.; Lieberman, S.A.; Greenisen, M.C. Impact of resistance exercise during bed rest on skeletal muscle sarcopenia and myosin isoform distribution. J. Appl. Physiol. 1998, 84, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Hikida, R.S.; Gollnick, P.D.; Dudley, G.A.; Convertino, V.A.; Buchanan, P. Structural and metabolic characteristics of human skeletal muscle following 30 days of simulated microgravity. Aviat. Space Env. Med. 1989, 60, 664–670. [Google Scholar]
- Shenkman, B.S.; Podlubnaya, Z.A.; Vikhlyantsev, I.M.; Litvinova, K.S.; Udaltsov, S.N.; Nemirovskaya, T.L.; Lemesheva, Y.S.; Mukhina, A.M.; Kozlovskaya, I.B. Contractile characteristics and sarcomeric cytoskeletal proteins of human soleus fibers in muscle unloading: Role of mechanical stimulation from the support surface. Biophysics 2004, 49, 807–815. [Google Scholar]
- Hernandez Corvo, R.; Kozlovskaia, I.B.; Kreidich Iu, V.; Martinez Fernandez, S.; Rakhamanov, A.S. Effect of a 7-day space flight on the structure and function of the human locomotor apparatus. Kosm. Biol. I Aviakosmicheskaia Meditsina 1983, 17, 37–44. [Google Scholar]
- Kozlovskaia, I.B.; Grigor’eva, L.S.; Gevlich, G.I. [Comparative analysis of the effect of weightlessness and its model on the velocity-strength properties and tonus of human skeletal muscles]. Kosm. Biol. I Aviakosmicheskaia Meditsina 1984, 18, 22–26. [Google Scholar]
- Friedrich, O.; Diermeier, S.; Larsson, L. Weak by the machines: Muscle motor protein dysfunction—A side effect of intensive care unit treatment. Acta Physiol. 2018, 222, e12885. [Google Scholar] [CrossRef]
- Giangregorio, L.; McCartney, N. Bone loss and muscle atrophy in spinal cord injury: Epidemiology, fracture prediction, and rehabilitation strategies. J. Spinal Cord. Med. 2006, 29, 489–500. [Google Scholar] [CrossRef]
- Cisterna, B.A.; Cardozo, C.; Saez, J.C. Neuronal involvement in muscular atrophy. Front. Cell. Neurosci. 2014, 8, 405. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, S.Y.; McGlory, C.; D’Souza, L.K.; Morgan, A.K.; Saddler, N.I.; Baker, S.K.; Parise, G.; Phillips, S.M. A randomized controlled trial of the impact of protein supplementation on leg lean mass and integrated muscle protein synthesis during inactivity and energy restriction in older persons. Am. J. Clin. Nutr. 2018, 108, 1060–1068. [Google Scholar] [CrossRef]
- Krogh-Madsen, R.; Thyfault, J.P.; Broholm, C.; Mortensen, O.H.; Olsen, R.H.; Mounier, R.; Plomgaard, P.; van Hall, G.; Booth, F.W.; Pedersen, B.K. A 2-wk reduction of ambulatory activity attenuates peripheral insulin sensitivity. J. Appl. Physiol. 2010, 108, 1034–1040. [Google Scholar] [CrossRef]
- Fisher, S.R.; Goodwin, J.S.; Protas, E.J.; Kuo, Y.F.; Graham, J.E.; Ottenbacher, K.J.; Ostir, G.V. Ambulatory activity of older adults hospitalized with acute medical illness. J. Am. Geriatr. Soc. 2011, 59, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, S.Y.; Holloway, T.M.; Phillips, S.M. The Impact of Step Reduction on Muscle Health in Aging: Protein and Exercise as Countermeasures. Front. Nutr. 2019, 6, 75. [Google Scholar] [CrossRef]
- Shad, B.J.; Thompson, J.L.; Holwerda, A.M.; Stocks, B.; Elhassan, Y.S.; Philp, A.; LJC, V.A.N.L.; Wallis, G.A. One Week of Step Reduction Lowers Myofibrillar Protein Synthesis Rates in Young Men. Med. Sci. Sports Exerc. 2019, 51, 2125–2134. [Google Scholar] [CrossRef] [PubMed]
- Devries, M.C.; Breen, L.; Von Allmen, M.; MacDonald, M.J.; Moore, D.R.; Offord, E.A.; Horcajada, M.N.; Breuille, D.; Phillips, S.M. Low-load resistance training during step-reduction attenuates declines in muscle mass and strength and enhances anabolic sensitivity in older men. Physiol. Rep. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Reid, K.F.; Pasha, E.; Doros, G.; Clark, D.J.; Patten, C.; Phillips, E.M.; Frontera, W.R.; Fielding, R.A. Longitudinal decline of lower extremity muscle power in healthy and mobility-limited older adults: Influence of muscle mass, strength, composition, neuromuscular activation and single fiber contractile properties. Eur. J. Appl. Physiol. 2014, 114, 29–39. [Google Scholar] [CrossRef]
- Hitomi, Y.; Kizaki, T.; Watanabe, S.; Matsumura, G.; Fujioka, Y.; Haga, S.; Izawa, T.; Taniguchi, N.; Ohno, H. Seven skeletal muscles rich in slow muscle fibers may function to sustain neutral position in the rodent hindlimb. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2005, 140, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Reggiani, C. Fiber types in mammalian skeletal muscles. Physiol. Rev. 2011, 91, 1447–1531. [Google Scholar] [CrossRef]
- Ilin, E.A.; Novikov, V.E. Stand for modelling the physiological effects of weightlessness in laboratory experiments with rats. Kosm. Biol. I Aviakosmicheskaia Meditsina 1980, 14, 79–80. [Google Scholar]
- Morey-Holton, E.R.; Globus, R.K. Hindlimb unloading rodent model: Technical aspects. J. Appl. Physiol. 2002, 92, 1367–1377. [Google Scholar] [CrossRef]
- Lawler, J.M.; Song, W.; Demaree, S.R. Hindlimb unloading increases oxidative stress and disrupts antioxidant capacity in skeletal muscle. Free Radic. Biol. Med. 2003, 35, 9–16. [Google Scholar] [CrossRef]
- Ohira, Y.; Yoshinaga, T.; Nomura, T.; Kawano, F.; Ishihara, A.; Nonaka, I.; Roy, R.R.; Edgerton, V.R. Gravitational unloading effects on muscle fiber size, phenotype and myonuclear number. Adv. Space. Res. Off. J. Comm. Space. Res. 2002, 30, 777–781. [Google Scholar] [CrossRef]
- Zhong, H.; Roy, R.R.; Siengthai, B.; Edgerton, V.R. Effects of inactivity on fiber size and myonuclear number in rat soleus muscle. J. Appl. Physiol. 2005, 99, 1494–1499. [Google Scholar] [CrossRef]
- Thomason, D.B.; Booth, F.W. Atrophy of the soleus muscle by hindlimb unweighting. J. Appl. Physiol. 1990, 68, 1–12. [Google Scholar] [CrossRef]
- Григoрьев, А.И.; Кoзлoвская, И.Б.; Шенкман, Б.С. Рoль oпoрнoй афферентации в oрганизации тoническoй мышечнoй системы. Рoссийский Физиoлoгический Журнал им. И.М. Сеченoва 2004, 90, 508–521. [Google Scholar]
- Gardetto, P.R.; Schluter, J.M.; Fitts, R.H. Contractile function of single muscle fibers after hindlimb suspension. J. Appl. Physiol. 1989, 66, 2739–2749. [Google Scholar] [CrossRef]
- Stevens, L.; Mounier, Y.; Holy, X.; Falempin, M. Contractile properties of rat soleus muscle after 15 days of hindlimb suspension. J. Appl. Physiol. 1990, 68, 334–340. [Google Scholar] [CrossRef]
- Ponomareva, E.V.; Kravtsova, V.V.; Kachaeva, E.V.; Altaeva, E.G.; Vikhliantsev, I.M.; Podlubnaia, Z.A.; Krivoi, I.I.; Shenkman, B.S. [Contractile properties of the isolated rat musculus soleus and single skinned soleus fibers at the early stage of gravitational unloading: Facts and hypotheses]. Biofizika 2008, 53, 1087–1094. [Google Scholar]
- Pierotti, D.J.; Roy, R.R.; Flores, V.; Edgerton, V.R. Influence of 7 days of hindlimb suspension and intermittent weight support on rat muscle mechanical properties. Aviat. Space Environ. Med. 1990, 61, 205–210. [Google Scholar] [PubMed]
- Wang, X.D.; Kawano, F.; Matsuoka, Y.; Fukunaga, K.; Terada, M.; Sudoh, M.; Ishihara, A.; Ohira, Y. Mechanical load-dependent regulation of satellite cell and fiber size in rat soleus muscle. Am. J. Physiol. Cell Physiol. 2006, 290, C981–C989. [Google Scholar] [CrossRef] [PubMed]
- Takemura, A.; Roy, R.R.; Edgerton, V.R.; Ishihara, A. Biochemical Adaptations in a Slow and a Fast Plantarflexor Muscle of Rats Housed in Small Cages. Aerosp. Med. Hum. Perform. 2016, 87, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Belova, S.P.; Tyganov, S.A.; Mochalova, E.P.; Shenkman, B.S. Restricted Activity and Protein Synthesis in Postural and Locomotor Muscles. Ross. Fiziol. Zhurnal. Im. I.M. Sechenova 2021, 107, 842–853. [Google Scholar] [CrossRef]
- Belozerova, I.N.; Nemirovskaya, T.L.; Shenkman, B.S.; Kozlovskaya, I.B. Characteristic of changes in the structure and metabolism of the vastus lateralis muscles in monkeys after space flight. Neurosci. Behav. Physiol. 2003, 33, 735–740. [Google Scholar] [CrossRef]
- Shenkman, B.S.; Belozerova, I.N.; Lee, P.; Nemirovskaya, T.L.; Kozlovskaya, I.B. Effects of weightlessness and movement restriction on the structure and metabolism of the soleus muscle in monkeys after space flight. Neurosci. Behav. Physiol. 2003, 33, 717–722. [Google Scholar] [CrossRef]
- Fischbach, G.D.; Robbins, N. Changes in contractile properties of disused soleus muscles. J. Physiol. 1969, 201, 305–320. [Google Scholar] [CrossRef] [PubMed]
- Hnik, P.; Vejsada, R.; Goldspink, D.F.; Kasicki, S.; Krekule, I. Quantitative evaluation of electromyogram activity in rat extensor and flexor muscles immobilized at different lengths. Exp. Neurol. 1985, 88, 515–528. [Google Scholar] [CrossRef]
- Jokl, P.; Konstadt, S. The effect of limb immobilization on muscle function and protein composition. Clin. Orthop. Relat. Res. 1983, 222–229. [Google Scholar] [CrossRef]
- Simard, C.P.; Spector, S.A.; Edgerton, V.R. Contractile properties of rat hind limb muscles immobilized at different lengths. Exp. Neurol. 1982, 77, 467–482. [Google Scholar] [CrossRef]
- Booth, F.W. Effect of limb immobilization on skeletal muscle. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1982, 52, 1113–1118. [Google Scholar] [CrossRef]
- Edgerton, V.R.; Barnard, R.J.; Peter, J.B.; Maier, P.A.; Simpson, D.R. Properties of immobilized hind-limb muscles of the Galago senegalensis. Exp. Neurol. 1975, 46, 115–131. [Google Scholar] [CrossRef]
- Elder, G.C.; Toner, L.V. Muscle shortening induced by tenotomy does not reduce activity levels in rat soleus. J. Physiol. 1998, 512 (Pt. 1), 251–265. [Google Scholar] [CrossRef]
- Nelson, P.G. Functional consequences of tenotomy in hind limb muscles of the cat. J. Physiol. 1969, 201, 321–333. [Google Scholar] [CrossRef]
- Jakubiec-Puka, A.; Catani, C.; Carraro, U. Myosin heavy-chain composition in striated muscle after tenotomy. Biochem. J. 1992, 282 (Pt 1), 237–242. [Google Scholar] [CrossRef]
- Buller, A.J.; Lewis, D.M. Further Observations on the Differentiation of Skeletal Muscles in the Kitten Hind Limb. J. Physiol. 1965, 176, 355–370. [Google Scholar] [CrossRef]
- Jamali, A.A.; Afshar, P.; Abrams, R.A.; Lieber, R.L. Skeletal muscle response to tenotomy. Muscle Nerve 2000, 23, 851–862. [Google Scholar] [CrossRef]
- Krivoi, I.I.; Kravtsova, V.V.; Altaeva, E.G.; Kubasov, I.V.; Prokof’ev, A.V.; Drabkina, T.M.; Nikol’skii, E.E.; Shenkman, B.S. [Decrease in the electrogenic contribution of Na,K-ATPase and resting membrane potential as a possible mechanism of calcium ion accumulation in filaments of the rat musculus soleus subjected to the short-term gravity unloading]. Biofizika 2008, 53, 1051–1057. [Google Scholar] [PubMed]
- Vilchinskaya, N.A.; Krivoi, I.I.; Shenkman, B.S. AMP-Activated Protein Kinase as a Key Trigger for the Disuse-Induced Skeletal Muscle Remodeling. Int. J. Mol. Sci. 2018, 19, 3558. [Google Scholar] [CrossRef] [PubMed]
- Shenkman, B.S. How Postural Muscle Senses Disuse? Early Signs and Signals. Int. J. Mol. Sci. 2020, 21, 5037. [Google Scholar] [CrossRef]
- De-Doncker, L.; Kasri, M.; Picquet, F.; Falempin, M. Physiologically adaptive changes of the L5 afferent neurogram and of the rat soleus EMG activity during 14 days of hindlimb unloading and recovery. J. Exp. Biol. 2005, 208, 4585–4592. [Google Scholar] [CrossRef]
- McKay, W.B.; Ovechkin, A.V.; Vitaz, T.W.; Terson de Paleville, D.G.; Harkema, S.J. Long-lasting involuntary motor activity after spinal cord injury. Spinal Cord 2011, 49, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Mirzoev, T.; Tyganov, S.; Vilchinskaya, N.; Lomonosova, Y.; Shenkman, B. Key Markers of mTORC1-Dependent and mTORC1-Independent Signaling Pathways Regulating Protein Synthesis in Rat Soleus Muscle During Early Stages of Hindlimb Unloading. Cell Physiol. Biochem. 2016, 39, 1011–1020. [Google Scholar] [CrossRef]
- Paramonova, I.I.; Sharlo, K.A.; Vilchinskaya, N.A.; Shenkman, B.S. The Time Course of Muscle Nuclear Content of Transcription Factors Regulating the MyHC I(β) Expression in the Rat Soleus Muscle under Gravitational Unloading. Biol. Membr. 2020, 37. [Google Scholar] [CrossRef]
- Belova, S.P.; Vilchinskaya, N.A.; Mochalova, E.P.; Mirzoev, T.M.; Nemirovskaya, T.L.; Shenkman, B.S. Elevated p70S6K phosphorylation in rat soleus muscle during the early stage of unloading: Causes and consequences. Arch. Biochem. Biophys. 2019, 674, 108105. [Google Scholar] [CrossRef] [PubMed]
- Sharlo, K.; Paramonova, I.; Turtikova, O.; Tyganov, S.; Shenkman, B. Plantar mechanical stimulation prevents calcineurin-NFATc1 inactivation and slow-to-fast fiber type shift in rat soleus muscle under hindlimb unloading. J. Appl. Physiol. 2019, 126, 1769–1781. [Google Scholar] [CrossRef] [PubMed]
- Zaripova, K.A.; Kalashnikova, E.P.; Belova, S.P.; Kostrominova, T.Y.; Shenkman, B.S.; Nemirovskaya, T.L. Role of Pannexin 1 ATP-Permeable Channels in the Regulation of Signaling Pathways during Skeletal Muscle Unloading. Int. J. Mol. Sci. 2021, 22, 10444. [Google Scholar] [CrossRef]
- Tyganov, S.A.; Mochalova, E.P.; Belova, S.P.; Sharlo, K.A.; Rozhkov, S.V.; Vilchinskaya, N.A.; Paramonova, I.I.; Mirzoev, T.M.; Shenkman, B.S. Effects of Plantar Mechanical Stimulation on Anabolic and Catabolic Signaling in Rat Postural Muscle Under Short-Term Simulated Gravitational Unloading. Front. Physiol. 2019, 10. [Google Scholar] [CrossRef]
- Rozhkov, S.V.; Sharlo, K.A.; Mirzoev, T.M.; Shenkman, B.S. Temporal changes in the markers of ribosome biogenesis in rat soleus muscle under simulated microgravity. Acta Astronaut. 2021, 186, 252–258. [Google Scholar] [CrossRef]
- Tyganov, S.A.; Mochalova, E.; Belova, S.; Sharlo, K.; Rozhkov, S.; Kalashnikov, V.; Turtikova, O.; Mirzoev, T.; Shenkman, B. Plantar mechanical stimulation attenuates protein synthesis decline in disused skeletal muscle via modulation of nitric oxide level. Sci. Rep. 2021, 11, 9806. [Google Scholar] [CrossRef]
- Loughna, P.; Goldspink, G.; Goldspink, D.F. Effect of inactivity and passive stretch on protein turnover in phasic and postural rat muscles. J. Appl. Physiol. 1986, 61, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Fluckey, J.D.; Dupont-Versteegden, E.E.; Montague, D.C.; Knox, M.; Tesch, P.; Peterson, C.A.; Gaddy-Kurten, D. A rat resistance exercise regimen attenuates losses of musculoskeletal mass during hindlimb suspension. Acta. Physiol. Scand. 2002, 176, 293–300. [Google Scholar] [CrossRef]
- Fluckey, J.D.; Dupont-Versteegden, E.E.; Knox, M.; Gaddy, D.; Tesch, P.A.; Peterson, C.A. Insulin facilitation of muscle protein synthesis following resistance exercise in hindlimb-suspended rats is independent of a rapamycin-sensitive pathway. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E1070–E1075. [Google Scholar] [CrossRef]
- You, J.S.; Anderson, G.B.; Dooley, M.S.; Hornberger, T.A. The role of mTOR signaling in the regulation of protein synthesis and muscle mass during immobilization in mice. Dis. Models Mech. 2015, 8, 1059–1069. [Google Scholar] [CrossRef]
- Gibson, J.N.; Halliday, D.; Morrison, W.L.; Stoward, P.J.; Hornsby, G.A.; Watt, P.W.; Murdoch, G.; Rennie, M.J. Decrease in human quadriceps muscle protein turnover consequent upon leg immobilization. Clin. Sci. 1987, 72, 503–509. [Google Scholar] [CrossRef]
- Ferrando, A.A.; Lane, H.W.; Stuart, C.A.; Davis-Street, J.; Wolfe, R.R. Prolonged bed rest decreases skeletal muscle and whole body protein synthesis. Am. J. Physiol. 1996, 270, E627–E633. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Sheffield-Moore, M.; Zhang, X.J.; Volpi, E.; Wolf, S.E.; Aarsland, A.; Ferrando, A.A.; Wolfe, R.R. Amino acid ingestion improves muscle protein synthesis in the young and elderly. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E321–E328. [Google Scholar] [CrossRef] [PubMed]
- Kortebein, P.; Ferrando, A.; Lombeida, J.; Wolfe, R.; Evans, W.J. Effect of 10 days of bed rest on skeletal muscle in healthy older adults. JAMA 2007, 297, 1772–1774. [Google Scholar] [CrossRef] [PubMed]
- Chaillou, T.; Kirby, T.J.; McCarthy, J.J. Ribosome biogenesis: Emerging evidence for a central role in the regulation of skeletal muscle mass. J. Cell. Physiol. 2014, 229, 1584–1594. [Google Scholar] [CrossRef] [PubMed]
- Mirzoev, T.M.; Shenkman, B.S. Regulation of protein synthesis in inactivated skeletal muscle: Signal inputs, protein kinase cascades, and ribosome biogenesis. Biochemistry 2018, 83, 1299–1317. [Google Scholar] [CrossRef]
- Baehr, L.M.; West, D.W.D.; Marshall, A.G.; Marcotte, G.R.; Baar, K.; Bodine, S.C. Muscle-specific and age-related changes in protein synthesis and protein degradation in response to hindlimb unloading in rats. J. Appl. Physiol. 2017, 122, 1336–1350. [Google Scholar] [CrossRef]
- Hornberger, T.A. Mechanotransduction and the regulation of mTORC1 signaling in skeletal muscle. Int. J. Biochem. Cell. Biol. 2011, 43, 1267–1276. [Google Scholar] [CrossRef]
- Wackerhage, H.; Ratkevicius, A. Signal transduction pathways that regulate muscle growth. Essays Biochem. 2008, 44, 99–108. [Google Scholar] [CrossRef]
- Han, B.; Tong, J.; Zhu, M.J.; Ma, C.; Du, M. Insulin-like growth factor-1 (IGF-1) and leucine activate pig myogenic satellite cells through mammalian target of rapamycin (mTOR) pathway. Mol. Reprod. Dev. 2008, 75, 810–817. [Google Scholar] [CrossRef]
- Erbay, E.; Park, I.H.; Nuzzi, P.D.; Schoenherr, C.J.; Chen, J. IGF-II transcription in skeletal myogenesis is controlled by mTOR and nutrients. J. Cell Biol. 2003, 163, 931–936. [Google Scholar] [CrossRef]
- Jheng, H.F.; Tsai, P.J.; Guo, S.M.; Kuo, L.H.; Chang, C.S.; Su, I.J.; Chang, C.R.; Tsai, Y.S. Mitochondrial fission contributes to mitochondrial dysfunction and insulin resistance in skeletal muscle. Mol. Cell. Biol. 2012, 32, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Inoki, K.; Li, Y.; Zhu, T.; Wu, J.; Guan, K.L. TSC2 is phosphorylated and inhibited by Akt and suppresses mTOR signalling. Nat. Cell. Biol. 2002, 4, 648–657. [Google Scholar] [CrossRef]
- Goodman, C.A.; Miu, M.H.; Frey, J.W.; Mabrey, D.M.; Lincoln, H.C.; Ge, Y.; Chen, J.; Hornberger, T.A. A phosphatidylinositol 3-kinase/protein kinase B-independent activation of mammalian target of rapamycin signaling is sufficient to induce skeletal muscle hypertrophy. Mol. Biol. Cell. 2010, 21, 3258–3268. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, M.; McCarthy, J.J.; Fedele, M.J.; Esser, K.A. Early activation of mTORC1 signalling in response to mechanical overload is independent of phosphoinositide 3-kinase/Akt signalling. J. Physiol. 2011, 589, 1831–1846. [Google Scholar] [CrossRef]
- Gordon, B.S.; Kelleher, A.R.; Kimball, S.R. Regulation of muscle protein synthesis and the effects of catabolic states. Int. J. Biochem. Cell Biol. 2013, 45, 2147–2157. [Google Scholar] [CrossRef] [PubMed]
- Uchida, T.; Sakashita, Y.; Kitahata, K.; Yamashita, Y.; Tomida, C.; Kimori, Y.; Komatsu, A.; Hirasaka, K.; Ohno, A.; Nakao, R.; et al. Reactive oxygen species upregulate expression of muscle atrophy-associated ubiquitin ligase Cbl-b in rat L6 skeletal muscle cells. Am. J. Physiol. Cell Physiol. 2018, 314, C721–C731. [Google Scholar] [CrossRef]
- Welsh, G.I.; Miller, C.M.; Loughlin, A.J.; Price, N.T.; Proud, C.G. Regulation of eukaryotic initiation factor eIF2B: Glycogen synthase kinase-3 phosphorylates a conserved serine which undergoes dephosphorylation in response to insulin. FEBS Lett. 1998, 421, 125–130. [Google Scholar] [CrossRef]
- Chibalin, A.V.; Benziane, B.; Zakyrjanova, G.F.; Kravtsova, V.V.; Krivoi, I.I. Early endplate remodeling and skeletal muscle signaling events following rat hindlimb suspension. J. Cell. Physiol. 2018, 233, 6329–6336. [Google Scholar] [CrossRef]
- Dupont, E.; Cieniewski-Bernard, C.; Bastide, B.; Stevens, L. Electrostimulation during hindlimb unloading modulates PI3K-AKT downstream targets without preventing soleus atrophy and restores slow phenotype through ERK. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Bajotto, G.; Sato, Y.; Kitaura, Y.; Shimomura, Y. Effect of branched-chain amino acid supplementation during unloading on regulatory components of protein synthesis in atrophied soleus muscles. Eur. J. Appl. Physiol. 2011, 111, 1815–1828. [Google Scholar] [CrossRef]
- Gwag, T.; Lee, K.; Ju, H.; Shin, H.; Lee, J.W.; Choi, I. Stress and signaling responses of rat skeletal muscle to brief endurance exercise during hindlimb unloading: A catch-up process for atrophied muscle. Cell. Physiol. Biochem. 2009, 24, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, A.R.; Kimball, S.R.; Dennis, M.D.; Schilder, R.J.; Jefferson, L.S. The mTORC1 signaling repressors REDD1/2 are rapidly induced and activation of p70S6K1 by leucine is defective in skeletal muscle of an immobilized rat hindlimb. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E229–E236. [Google Scholar] [CrossRef]
- Cannavino, J.; Brocca, L.; Sandri, M.; Bottinelli, R.; Pellegrino, M.A. PGC1-alpha over-expression prevents metabolic alterations and soleus muscle atrophy in hindlimb unloaded mice. J. Physiol. 2014, 592, 4575–4589. [Google Scholar] [CrossRef]
- Han, B.; Zhu, M.J.; Ma, C.; Du, M. Rat hindlimb unloading down-regulates insulin like growth factor-1 signaling and AMP-activated protein kinase, and leads to severe atrophy of the soleus muscle. Appl. Physiol. Nutr. Metab. 2007, 32, 1115–1123. [Google Scholar] [CrossRef] [PubMed]
- Mirzoev, T.M.; Tyganov, S.A.; Lomonosova, Y.N.; Musienko, P.E.; Shenkman, B.S. Signaling Pathways Regulating Protein Synthesis in Rat Soleus Muscle during Early Stages of Hindlimb Unloading. Ross. Fiziol. Zhurnal. Im. I.M. Sechenova 2015, 101, 1299–1308. [Google Scholar]
- Thomson, D.M. The Role of AMPK in the Regulation of Skeletal Muscle Size, Hypertrophy, and Regeneration. Int. J. Mol. Sci. 2018, 19, 3125. [Google Scholar] [CrossRef] [PubMed]
- Gwinn, D.M.; Shackelford, D.B.; Egan, D.F.; Mihaylova, M.M.; Mery, A.; Vasquez, D.S.; Turk, B.E.; Shaw, R.J. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol. Cell 2008, 30, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Vilchinskaya, N.A.; Mochalova, E.P.; Nemirovskaya, T.L.; Mirzoev, T.M.; Turtikova, O.V.; Shenkman, B.S. Rapid decline in MyHC I(beta) mRNA expression in rat soleus during hindlimb unloading is associated with AMPK dephosphorylation. J. Physiol. 2017, 595, 7123–7134. [Google Scholar] [CrossRef]
- Vilchinskaya, N.A.; Mirzoev, T.M.; Lomonosova, Y.N.; Kozlovskaya, I.B.; Shenkman, B.S. Human muscle signaling responses to 3-day head-out dry immersion. J. Musculoskelet. Neuronal Interact 2015, 15, 286–293. [Google Scholar]
- Wall, B.T.; Snijders, T.; Senden, J.M.; Ottenbros, C.L.; Gijsen, A.P.; Verdijk, L.B.; van Loon, L.J. Disuse impairs the muscle protein synthetic response to protein ingestion in healthy men. J. Clin. Endocrinol. Metab. 2013, 98, 4872–4881. [Google Scholar] [CrossRef]
- de Boer, M.D.; Selby, A.; Atherton, P.; Smith, K.; Seynnes, O.R.; Maganaris, C.N.; Maffulli, N.; Movin, T.; Narici, M.V.; Rennie, M.J. The temporal responses of protein synthesis, gene expression and cell signalling in human quadriceps muscle and patellar tendon to disuse. J. Physiol. 2007, 585, 241–251. [Google Scholar] [CrossRef]
- Drummond, M.J.; Dickinson, J.M.; Fry, C.S.; Walker, D.K.; Gundermann, D.M.; Reidy, P.T.; Timmerman, K.L.; Markofski, M.M.; Paddon-Jones, D.; Rasmussen, B.B.; et al. Bed rest impairs skeletal muscle amino acid transporter expression, mTORC1 signaling, and protein synthesis in response to essential amino acids in older adults. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E1113–E1122. [Google Scholar] [CrossRef]
- Glover, E.I.; Yasuda, N.; Tarnopolsky, M.A.; Abadi, A.; Phillips, S.M. Little change in markers of protein breakdown and oxidative stress in humans in immobilization-induced skeletal muscle atrophy. Appl. Physiol. Nutr. Metab. 2010, 35, 125–133. [Google Scholar] [CrossRef]
- Churchward-Venne, T.A.; Burd, N.A.; Phillips, S.M. Nutritional regulation of muscle protein synthesis with resistance exercise: Strategies to enhance anabolism. Nutr. Metab. 2012, 9, 40. [Google Scholar] [CrossRef]
- Shangraw, R.E.; Stuart, C.A.; Prince, M.J.; Peters, E.J.; Wolfe, R.R. Insulin responsiveness of protein metabolism in vivo following bedrest in humans. Am. J. Physiol. 1988, 255, E548–E558. [Google Scholar] [CrossRef] [PubMed]
- Stuart, C.A.; Shangraw, R.E.; Prince, M.J.; Peters, E.J.; Wolfe, R.R. Bed-rest-induced insulin resistance occurs primarily in muscle. Metab. Clin. Exp. 1988, 37, 802–806. [Google Scholar] [CrossRef]
- Richter, E.A.; Kiens, B.; Mizuno, M.; Strange, S. Insulin action in human thighs after one-legged immobilization. J. Appl. Physiol. 1989, 67, 19–23. [Google Scholar] [CrossRef]
- Allen, D.L.; Linderman, J.K.; Roy, R.R.; Grindeland, R.E.; Mukku, V.; Edgerton, V.R. Growth hormone/IGF-I and/or resistive exercise maintains myonuclear number in hindlimb unweighted muscles. J. Appl. Physiol. 1997, 83, 1857–1861. [Google Scholar] [CrossRef] [PubMed]
- Haddad, F.; Baldwin, K.M.; Tesch, P.A. Pretranslational markers of contractile protein expression in human skeletal muscle: Effect of limb unloading plus resistance exercise. J. Appl. Physiol. 2005, 98, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Gamrin, L.; Berg, H.E.; Essen, P.; Tesch, P.A.; Hultman, E.; Garlick, P.J.; McNurlan, M.A.; Wernerman, J. The effect of unloading on protein synthesis in human skeletal muscle. Acta Physiol. Scand. 1998, 163, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Petersson, B.; Wernerman, J.; Waller, S.O.; von der Decken, A.; Vinnars, E. Elective abdominal surgery depresses muscle protein synthesis and increases subjective fatigue: Effects lasting more than 30 days. Br. J. Surg. 1990, 77, 796–800. [Google Scholar] [CrossRef]
- Gibson, J.N.; Smith, K.; Rennie, M.J. Prevention of disuse muscle atrophy by means of electrical stimulation: Maintenance of protein synthesis. Lancet 1988, 2, 767–770. [Google Scholar] [CrossRef]
- Anderson, J.E.; Zhu, A.; Mizuno, T.M. Nitric oxide treatment attenuates muscle atrophy during hind limb suspension in mice. Free Radic. Biol. Med. 2018, 115, 458–470. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.B.; Judge, A.R.; Bodine, S.C. CrossTalk opposing view: The dominant mechanism causing disuse muscle atrophy is proteolysis. J. Physiol. 2014, 592, 5345–5347. [Google Scholar] [CrossRef] [PubMed]
- Tesch, P.A.; von Walden, F.; Gustafsson, T.; Linnehan, R.M.; Trappe, T.A. Skeletal muscle proteolysis in response to short-term unloading in humans. J. Appl. Physiol. 2008, 105, 902–906. [Google Scholar] [CrossRef]
- Phillips, S.M.; McGlory, C. CrossTalk proposal: The dominant mechanism causing disuse muscle atrophy is decreased protein synthesis. J. Physiol. 2014, 592, 5341–5343. [Google Scholar] [CrossRef] [PubMed]
- Scicchitano, B.M.; Faraldi, M.; Musaro, A. The Proteolytic Systems of Muscle Wasting. Recent. Adv. DNA Gene Seq. 2015, 9, 26–35. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dayton, W.R.; Goll, D.E.; Zeece, M.G.; Robson, R.M.; Reville, W.J. A Ca2+-activated protease possibly involved in myofibrillar protein turnover. Purification from porcine muscle. Biochemistry 1976, 15, 2150–2158. [Google Scholar] [CrossRef]
- Arutyunyan, R.S.; Kozlovskaya, I.B.; Nasledov, G.A.; Nemirovskaya, T.L.; Radzyukevitch, T.L.; Shenkman, B.S. The contraction of unweighted fast and slow rat muscles in calcium-free solution. Basic Appl. Myol. 1995, 5, 169–175. [Google Scholar]
- Ingalls, C.P.; Warren, G.L.; Armstrong, R.B. Intracellular Ca2+ transients in mouse soleus muscle after hindlimb unloading and reloading. J. Appl. Physiol. 1999, 87, 386–390. [Google Scholar] [CrossRef]
- Shenkman, B.S.; Nemirovskaya, T.L. Calcium-dependent signaling mechanisms and soleus fiber remodeling under gravitational unloading. J. Muscle Res. Cell Motil. 2008, 29, 221–230. [Google Scholar] [CrossRef]
- Ingalls, C.P.; Wenke, J.C.; Armstrong, R.B. Time course changes in [Ca2+]i, force, and protein content in hindlimb-suspended mouse soleus muscles. Aviat. Space Env. Med. 2001, 72, 471–476. [Google Scholar]
- Tidball, J.G.; Spencer, M.J. Expression of a calpastatin transgene slows muscle wasting and obviates changes in myosin isoform expression during murine muscle disuse. J. Physiol. 2002, 545, 819–828. [Google Scholar] [CrossRef]
- Shenkman, B.S.; Belova, S.P.; Lomonosova, Y.N.; Kostrominova, T.Y.; Nemirovskaya, T.L. Calpain-dependent regulation of the skeletal muscle atrophy following unloading. Arch. Biochem. Biophys. 2015, 584, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Petrova, I.O.; Tyganov, S.A.; Mirzoev, T.M.; Tsaturyan, A.K.; Kozlovskaya, I.B.; Shenkman, B.S. Early Desmall es, Cyrillicline in Rat Soleus Passive Tension with Hindlimb Unloading: Inactivation of Cross-bridges or Activation of Calpains? Dokl. Biochem. Biophys. 2018, 481, 205–207. [Google Scholar] [CrossRef] [PubMed]
- Aweida, D.; Rudesky, I.; Volodin, A.; Shimko, E.; Cohen, S. GSK3-beta promotes calpain-1-mediated desmin filament depolymerization and myofibril loss in atrophy. J. Cell. Biol. 2018, 217, 3698–3714. [Google Scholar] [CrossRef]
- Tyganov, S.A.; Mochalova, E.P.; Melnikov, I.Y.; Vikhlyantsev, I.M.; Ulanova, A.D.; Sharlo, K.A.; Mirzoev, T.M.; Shenkman, B.S. NOS-dependent effects of plantar mechanical stimulation on mechanical characteristics and cytoskeletal proteins in rat soleus muscle during hindlimb suspension. FASEB J. 2021, 35, e21905. [Google Scholar] [CrossRef]
- Lechado, I.T.A.; Vitadello, M.; Traini, L.; Namuduri, A.V.; Gastaldello, S.; Gorza, L. Sarcolemmal loss of active nNOS (Nos1) is an oxidative stress-dependent, early event driving disuse atrophy. J. Pathol. 2018, 246, 433–446. [Google Scholar] [CrossRef]
- Lomonosova, Y.N.; Kalamkarov, G.R.; Bugrova, A.E.; Shevchenko, T.F.; Kartashkina, N.L.; Lysenko, E.A.; Shvets, V.I.; Nemirovskaya, T.L. Protective effect of L-Arginine administration on proteins of unloaded m. soleus. Biochemistry 2011, 76, 571–580. [Google Scholar] [CrossRef]
- Sharlo, K.A.; Paramonova, I.I.; Lvova, I.D.; Vilchinskaya, N.A.; Bugrova, A.E.; Shevchenko, T.F.; Kalamkarov, G.R.; Shenkman, B.S. NO-Dependent Mechanisms of Myosin Heavy Chain Transcription Regulation in Rat Soleus Muscle After 7-Days Hindlimb Unloading. Front. Physiol. 2020, 11, 814. [Google Scholar] [CrossRef]
- Suzuki, N.; Motohashi, N.; Uezumi, A.; Fukada, S.; Yoshimura, T.; Itoyama, Y.; Aoki, M.; Miyagoe-Suzuki, Y.; Takeda, S. NO production results in suspension-induced muscle atrophy through dislocation of neuronal NOS. J. Clin. Investig. 2007, 117, 2468–2476. [Google Scholar] [CrossRef]
- Sharlo, K.A.; Paramonova, I.I.; Lvova, I.D.; Mochalova, E.P.; Kalashnikov, V.E.; Vilchinskaya, N.A.; Tyganov, S.A.; Konstantinova, T.S.; Shevchenko, T.F.; Kalamkarov, G.R.; et al. Plantar Mechanical Stimulation Maintains Slow Myosin Expression in Disused Rat Soleus Muscle via NO-Dependent Signaling. Int. J. Mol. Sci. 2021, 22, 1372. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhu, X. The molecular mechanisms of calpains action on skeletal muscle atrophy. Physiol. Res. 2016, 65, 547–560. [Google Scholar] [CrossRef]
- Talbert, E.E.; Smuder, A.J.; Min, K.; Kwon, O.S.; Powers, S.K. Calpain and caspase-3 play required roles in immobilization-induced limb muscle atrophy. J. Appl. Physiol. 2013, 114, 1482–1489. [Google Scholar] [CrossRef]
- Sandri, M. Autophagy in skeletal muscle. FEBS Lett. 2010, 584, 1411–1416. [Google Scholar] [CrossRef] [PubMed]
- Bodine, S.C.; Baehr, L.M. Skeletal muscle atrophy and the E3 ubiquitin ligases MuRF1 and MAFbx/atrogin-1. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E469–E484. [Google Scholar] [CrossRef]
- Rudrappa, S.S.; Wilkinson, D.J.; Greenhaff, P.L.; Smith, K.; Idris, I.; Atherton, P.J. Human Skeletal Muscle Disuse Atrophy: Effects on Muscle Protein Synthesis, Breakdown, and Insulin Resistance-A Qualitative Review. Front. Physiol. 2016, 7, 361. [Google Scholar] [CrossRef]
- Mitch, W.E.; Goldberg, A.L. Mechanisms of muscle wasting. The role of the ubiquitin-proteasome pathway. N. Engl. J. Med. 1996, 335, 1897–1905. [Google Scholar] [CrossRef]
- Foletta, V.C.; White, L.J.; Larsen, A.E.; Leger, B.; Russell, A.P. The role and regulation of MAFbx/atrogin-1 and MuRF1 in skeletal muscle atrophy. Pflug. Arch. Eur. J. Physiol. 2011, 461, 325–335. [Google Scholar] [CrossRef]
- Lecker, S.H.; Jagoe, R.T.; Gilbert, A.; Gomes, M.; Baracos, V.; Bailey, J.; Price, S.R.; Mitch, W.E.; Goldberg, A.L. Multiple types of skeletal muscle atrophy involve a common program of changes in gene expression. FASEB J. 2004, 18, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Bodine, S.C.; Latres, E.; Baumhueter, S.; Lai, V.K.; Nunez, L.; Clarke, B.A.; Poueymirou, W.T.; Panaro, F.J.; Na, E.; Dharmarajan, K.; et al. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 2001, 294, 1704–1708. [Google Scholar] [CrossRef] [PubMed]
- Murton, A.J.; Constantin, D.; Greenhaff, P.L. The involvement of the ubiquitin proteasome system in human skeletal muscle remodelling and atrophy. Biochim. Biophys. Acta 2008, 1782, 730–743. [Google Scholar] [CrossRef]
- Allen, D.L.; Bandstra, E.R.; Harrison, B.C.; Thorng, S.; Stodieck, L.S.; Kostenuik, P.J.; Morony, S.; Lacey, D.L.; Hammond, T.G.; Leinwand, L.L.; et al. Effects of spaceflight on murine skeletal muscle gene expression. J. Appl. Physiol. 2009, 106, 582–595. [Google Scholar] [CrossRef]
- Gambara, G.; Salanova, M.; Ciciliot, S.; Furlan, S.; Gutsmann, M.; Schiffl, G.; Ungethuem, U.; Volpe, P.; Gunga, H.C.; Blottner, D. Microgravity-Induced Transcriptome Adaptation in Mouse Paraspinal longissimus dorsi Muscle Highlights Insulin Resistance-Linked Genes. Front. Physiol. 2017, 8, 279. [Google Scholar] [CrossRef]
- Labeit, S.; Kohl, C.H.; Witt, C.C.; Labeit, D.; Jung, J.; Granzier, H. Modulation of muscle atrophy, fatigue and MLC phosphorylation by MuRF1 as indicated by hindlimb suspension studies on MuRF1-KO mice. J. Biomed. Biotechnol. 2010, 2010, 693741. [Google Scholar] [CrossRef]
- Belova, S.P.; Mochalova, E.P.; Kostrominova, T.Y.; Shenkman, B.S.; Nemirovskaya, T.L. P38alpha-MAPK Signaling Inhibition Attenuates Soleus Atrophy during Early Stages of Muscle Unloading. Int. J. Mol. Sci. 2020, 21, 2756. [Google Scholar] [CrossRef] [PubMed]
- Derbre, F.; Ferrando, B.; Gomez-Cabrera, M.C.; Sanchis-Gomar, F.; Martinez-Bello, V.E.; Olaso-Gonzalez, G.; Diaz, A.; Gratas-Delamarche, A.; Cerda, M.; Vina, J. Inhibition of xanthine oxidase by allopurinol prevents skeletal muscle atrophy: Role of p38 MAPKinase and E3 ubiquitin ligases. PLoS ONE 2012, 7, e46668. [Google Scholar] [CrossRef]
- Casas, M.; Buvinic, S.; Jaimovich, E. ATP signaling in skeletal muscle: From fiber plasticity to regulation of metabolism. Exerc. Sport Sci. Rev. 2014, 42, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Mochalova, E.P.; Belova, S.P.; Mirzoev, T.M.; Shenkman, B.S.; Nemirovskaya, T.L. Atrogin-1/MAFbx mRNA expression is regulated by histone deacetylase 1 in rat soleus muscle under hindlimb unloading. Sci. Rep. 2019, 9, 10263. [Google Scholar] [CrossRef]
- Abadi, A.; Glover, E.I.; Isfort, R.J.; Raha, S.; Safdar, A.; Yasuda, N.; Kaczor, J.J.; Melov, S.; Hubbard, A.; Qu, X.; et al. Limb immobilization induces a coordinate down-regulation of mitochondrial and other metabolic pathways in men and women. PLoS ONE 2009, 4, e6518. [Google Scholar] [CrossRef]
- Gustafsson, T.; Osterlund, T.; Flanagan, J.N.; von Walden, F.; Trappe, T.A.; Linnehan, R.M.; Tesch, P.A. Effects of 3 days unloading on molecular regulators of muscle size in humans. J. Appl. Physiol. 2010, 109, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Furochi, H.; Mameoka, M.; Hirasaka, K.; Onishi, Y.; Suzue, N.; Oarada, M.; Akamatsu, M.; Akima, H.; Fukunaga, T.; et al. Ubiquitin ligase gene expression in healthy volunteers with 20-day bedrest. Muscle Nerve 2006, 34, 463–469. [Google Scholar] [CrossRef]
- Jones, S.W.; Hill, R.J.; Krasney, P.A.; O’Conner, B.; Peirce, N.; Greenhaff, P.L. Disuse atrophy and exercise rehabilitation in humans profoundly affects the expression of genes associated with the regulation of skeletal muscle mass. FASEB J. 2004, 18, 1025–1027. [Google Scholar] [CrossRef]
- Salanova, M.; Schiffl, G.; Puttmann, B.; Schoser, B.G.; Blottner, D. Molecular biomarkers monitoring human skeletal muscle fibres and microvasculature following long-term bed rest with and without countermeasures. J. Anat. 2008, 212, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Milan, G.; Romanello, V.; Pescatore, F.; Armani, A.; Paik, J.H.; Frasson, L.; Seydel, A.; Zhao, J.; Abraham, R.; Goldberg, A.L.; et al. Regulation of autophagy and the ubiquitin-proteasome system by the FoxO transcriptional network during muscle atrophy. Nat. Commun. 2015, 6, 6670. [Google Scholar] [CrossRef]
- Sandri, M.; Sandri, C.; Gilbert, A.; Skurk, C.; Calabria, E.; Picard, A.; Walsh, K.; Schiaffino, S.; Lecker, S.H.; Goldberg, A.L. Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 2004, 117, 399–412. [Google Scholar] [CrossRef]
- Stitt, T.N.; Drujan, D.; Clarke, B.A.; Panaro, F.; Timofeyva, Y.; Kline, W.O.; Gonzalez, M.; Yancopoulos, G.D.; Glass, D.J. The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol. Cell 2004, 14, 395–403. [Google Scholar] [CrossRef]
- Brunet, A.; Bonni, A.; Zigmond, M.J.; Lin, M.Z.; Juo, P.; Hu, L.S.; Anderson, M.J.; Arden, K.C.; Blenis, J.; Greenberg, M.E. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999, 96, 857–868. [Google Scholar] [CrossRef]
- Zhao, J.; Brault, J.J.; Schild, A.; Cao, P.; Sandri, M.; Schiaffino, S.; Lecker, S.H.; Goldberg, A.L. FoxO3 coordinately activates protein degradation by the autophagic/lysosomal and proteasomal pathways in atrophying muscle cells. Cell. Metab. 2007, 6, 472–483. [Google Scholar] [CrossRef]
- Okamoto, T.; Machida, S. Changes in FOXO and proinflammatory cytokines in the late stage of immobilized fast and slow muscle atrophy. Biomed. Res. 2017, 38, 331–342. [Google Scholar] [CrossRef]
- Levine, S.; Biswas, C.; Dierov, J.; Barsotti, R.; Shrager, J.B.; Nguyen, T.; Sonnad, S.; Kucharchzuk, J.C.; Kaiser, L.R.; Singhal, S.; et al. Increased proteolysis, myosin depletion, and atrophic AKT-FOXO signaling in human diaphragm disuse. Am. J. Respir. Crit. Care Med. 2011, 183, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Giresi, P.G.; Stevenson, E.J.; Theilhaber, J.; Koncarevic, A.; Parkington, J.; Fielding, R.A.; Kandarian, S.C. Identification of a molecular signature of sarcopenia. Physiol. Genom. 2005, 21, 253–263. [Google Scholar] [CrossRef]
- Sacheck, J.M.; Hyatt, J.P.; Raffaello, A.; Jagoe, R.T.; Roy, R.R.; Edgerton, V.R.; Lecker, S.H.; Goldberg, A.L. Rapid disuse and denervation atrophy involve transcriptional changes similar to those of muscle wasting during systemic diseases. FASEB J. 2007, 21, 140–155. [Google Scholar] [CrossRef]
- Brocca, L.; Toniolo, L.; Reggiani, C.; Bottinelli, R.; Sandri, M.; Pellegrino, M.A. FoxO-dependent atrogenes vary among catabolic conditions and play a key role in muscle atrophy induced by hindlimb suspension. J. Physiol. 2017, 595, 1143–1158. [Google Scholar] [CrossRef] [PubMed]
- Reed, S.A.; Sandesara, P.B.; Senf, S.M.; Judge, A.R. Inhibition of FoxO transcriptional activity prevents muscle fiber atrophy during cachexia and induces hypertrophy. FASEB J. 2012, 26, 987–1000. [Google Scholar] [CrossRef] [PubMed]
- Senf, S.M.; Dodd, S.L.; McClung, J.M.; Judge, A.R. Hsp70 overexpression inhibits NF-kappaB and Foxo3a transcriptional activities and prevents skeletal muscle atrophy. FASEB J. 2008, 22, 3836–3845. [Google Scholar] [CrossRef]
- Mammucari, C.; Milan, G.; Romanello, V.; Masiero, E.; Rudolf, R.; Del Piccolo, P.; Burden, S.J.; Di Lisi, R.; Sandri, C.; Zhao, J.; et al. FoxO3 controls autophagy in skeletal muscle in vivo. Cell Metab. 2007, 6, 458–471. [Google Scholar] [CrossRef]
- Grigor’ev, A.I.; Kozlovskaia, I.B.; Shenkman, B.S. [The role of support afferents in organisation of the tonic muscle system]. Ross. Fiziol. Zh. Im. I. M. Sechenova. 2004, 90, 508–521. [Google Scholar]
- Trappe, S.; Costill, D.; Gallagher, P.; Creer, A.; Peters, J.R.; Evans, H.; Riley, D.A.; Fitts, R.H. Exercise in space: Human skeletal muscle after 6 months aboard the International Space Station. J. Appl. Physiol. 2009, 106, 1159–1168. [Google Scholar] [CrossRef]
- Zhou, M.Y.; Klitgaard, H.; Saltin, B.; Roy, R.R.; Edgerton, V.R.; Gollnick, P.D. Myosin heavy chain isoforms of human muscle after short-term spaceflight. J. Appl. Physiol. 1995, 78, 1740–1744. [Google Scholar] [CrossRef] [PubMed]
- Borina, E.; Pellegrino, M.A.; D’Antona, G.; Bottinelli, R. Myosin and actin content of human skeletal muscle fibers following 35 days bed rest. Scand. J. Med. Sci. Sports. 2010, 20, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Malisoux, L.; Jamart, C.; Delplace, K.; Nielens, H.; Francaux, M.; Theisen, D. Effect of long-term muscle paralysis on human single fiber mechanics. J. Appl. Physiol. 2007, 102, 340–349. [Google Scholar] [CrossRef][Green Version]
- Giger, J.M.; Bodell, P.W.; Zeng, M.; Baldwin, K.M.; Haddad, F. Rapid muscle atrophy response to unloading: Pretranslational processes involving MHC and actin. J. Appl. Physiol. 2009, 107, 1204–1212. [Google Scholar] [CrossRef]
- Stevens, L.; Sultan, K.R.; Peuker, H.; Gohlsch, B.; Mounier, Y.; Pette, D. Time-dependent changes in myosin heavy chain mRNA and protein isoforms in unloaded soleus muscle of rat. Am. J. Physiol. 1999, 277, C1044–C1049. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, K.M.; Haddad, F.; Pandorf, C.E.; Roy, R.R.; Edgerton, V.R. Alterations in muscle mass and contractile phenotype in response to unloading models: Role of transcriptional/pretranslational mechanisms. Front. Physiol. 2013, 4, 284. [Google Scholar] [CrossRef] [PubMed]
- Lomonosova, Y.N.; Turtikova, O.V.; Shenkman, B.S. Reduced expression of MyHC slow isoform in rat soleus during unloading is accompanied by alterations of endogenous inhibitors of calcineurin/NFAT signaling pathway. J. Muscle Res. Cell Motil. 2016, 37, 7–16. [Google Scholar] [CrossRef]
- Matoba, H.; Wakatuti, Y.; Ohira, Y. 879β-Guanidinopropionic acid suppresses suspension-induced changes in myosin expression in rat skeletal muscle. Med. Sci. Sports Exerc. 1993, 25, 157. [Google Scholar] [CrossRef]
- Braz, J.C.; Bueno, O.F.; Liang, Q.; Wilkins, B.J.; Dai, Y.S.; Parsons, S.; Braunwart, J.; Glascock, B.J.; Klevitsky, R.; Kimball, T.F.; et al. Targeted inhibition of p38 MAPK promotes hypertrophic cardiomyopathy through upregulation of calcineurin-NFAT signaling. J. Clin. Investig. 2003, 111, 1475–1486. [Google Scholar] [CrossRef]
- Liang, Q.; Bueno, O.F.; Wilkins, B.J.; Kuan, C.Y.; Xia, Y.; Molkentin, J.D. c-Jun N-terminal kinases (JNK) antagonize cardiac growth through cross-talk with calcineurin-NFAT signaling. EMBO J. 2003, 22, 5079–5089. [Google Scholar] [CrossRef]
- McCullagh, K.J.; Calabria, E.; Pallafacchina, G.; Ciciliot, S.; Serrano, A.L.; Argentini, C.; Kalhovde, J.M.; Lomo, T.; Schiaffino, S. NFAT is a nerve activity sensor in skeletal muscle and controls activity-dependent myosin switching. Proc. Natl. Acad. Sci. USA 2004, 101, 10590–10595. [Google Scholar] [CrossRef]
- Sharlo, C.A.; Turtikova, Y.N.; Turtikova, O.V.; Mitrofanova, O.V.; Kalamkarov, G.R.; Bugrova, A.E.; Shevchenko, T.F.; Shenkman, B.S. The Role of GSK-3β Phosphorylation in the Regulation of Slow Myosin Expression in Soleus Muscle during Functional Unloading. Biochem. Suppl. Ser. A Membr. Cell. Biol. 2018, 12, 85–91. [Google Scholar] [CrossRef]
- Sharlo, K.A.; Mochalova, E.P.; Belova, S.P.; Lvova, I.D.; Nemirovskaya, T.L.; Shenkman, B.S. The role of MAP-kinase p38 in the m. soleus slow myosin mRNA transcription regulation during short-term functional unloading. Arch. Biochem. Biophys. 2020, 695, 108622. [Google Scholar] [CrossRef]
- Popov, D.V.; Vinogradova, O.L.; A.I., G. Human aerobic performance. (book in russian).
- Gifford, J.R.; Weavil, J.C.; Nelson, A.D. Symmorphosis in patients with chronic heart failure? J. Appl. Physiol. 2016, 121, 1039. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Berg, H.E.; Dudley, G.A.; Hather, B.; Tesch, P.A. Work capacity and metabolic and morphologic characteristics of the human quadriceps muscle in response to unloading. Clin. Physiol. 1993, 13, 337–347. [Google Scholar] [CrossRef]
- Vigelso, A.; Gram, M.; Wiuff, C.; Andersen, J.L.; Helge, J.W.; Dela, F. Six weeks’ aerobic retraining after two weeks’ immobilization restores leg lean mass and aerobic capacity but does not fully rehabilitate leg strength in young and older men. J. Rehabil. Med. 2015, 47, 552–560. [Google Scholar] [CrossRef]
- Puthucheary, Z.A.; Astin, R.; McPhail, M.J.W.; Saeed, S.; Pasha, Y.; Bear, D.E.; Constantin, D.; Velloso, C.; Manning, S.; Calvert, L.; et al. Metabolic phenotype of skeletal muscle in early critical illness. Thorax 2018, 73, 926–935. [Google Scholar] [CrossRef]
- Kramer, A.; Kummel, J.; Mulder, E.; Gollhofer, A.; Frings-Meuthen, P.; Gruber, M. High-Intensity Jump Training Is Tolerated during 60 Days of Bed Rest and Is Very Effective in Preserving Leg Power and Lean Body Mass: An Overview of the Cologne RSL Study. PLoS ONE 2017, 12, e0169793. [Google Scholar] [CrossRef]
- Blottner, D.; Hastermann, M.; Weber, R.; Lenz, R.; Gambara, G.; Limper, U.; Rittweger, J.; Bosutti, A.; Degens, H.; Salanova, M. Reactive Jumps Preserve Skeletal Muscle Structure, Phenotype, and Myofiber Oxidative Capacity in Bed Rest. Front. Physiol. 2019, 10, 1527. [Google Scholar] [CrossRef]
- Falempin, M.; In-Albon, S.F. Influence of brief daily tendon vibration on rat soleus muscle in non-weight-bearing situation. J. Appl. Physiol. 1999, 87, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Arentson-Lantz, E.J.; English, K.L.; Paddon-Jones, D.; Fry, C.S. Fourteen days of bed rest induces a decline in satellite cell content and robust atrophy of skeletal muscle fibers in middle-aged adults. J. Appl. Physiol. 2016, 120, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Lammers, G.; van Duijnhoven, N.T.; Hoenderop, J.G.; Horstman, A.M.; de Haan, A.; Janssen, T.W.; de Graaf, M.J.; Pardoel, E.M.; Verwiel, E.T.; Thijssen, D.H.; et al. The identification of genetic pathways involved in vascular adaptations after physical deconditioning versus exercise training in humans. Exp. Physiol. 2013, 98, 710–721. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hackney, K.J.; Ploutz-Snyder, L.L. Unilateral lower limb suspension: Integrative physiological knowledge from the past 20 years (1991–2011). Eur. J. Appl. Physiol. 2012, 112, 9–22. [Google Scholar] [CrossRef]
- Hyatt, H.; Deminice, R.; Yoshihara, T.; Powers, S.K. Mitochondrial dysfunction induces muscle atrophy during prolonged inactivity: A review of the causes and effects. Arch. Biochem. Biophys. 2019, 662, 49–60. [Google Scholar] [CrossRef]
- Moriggi, M.; Vasso, M.; Fania, C.; Capitanio, D.; Bonifacio, G.; Salanova, M.; Blottner, D.; Rittweger, J.; Felsenberg, D.; Cerretelli, P.; et al. Long term bed rest with and without vibration exercise countermeasures: Effects on human muscle protein dysregulation. Proteomics 2010, 10, 3756–3774. [Google Scholar] [CrossRef]
- Dillon, E.L.; Soman, K.V.; Wiktorowicz, J.E.; Sur, R.; Jupiter, D.; Danesi, C.P.; Randolph, K.M.; Gilkison, C.R.; Durham, W.J.; Urban, R.J.; et al. Proteomic investigation of human skeletal muscle before and after 70 days of head down bed rest with or without exercise and testosterone countermeasures. PLoS ONE 2019, 14, e0217690. [Google Scholar] [CrossRef]
- Chopard, A.; Lecunff, M.; Danger, R.; Lamirault, G.; Bihouee, A.; Teusan, R.; Jasmin, B.J.; Marini, J.F.; Leger, J.J. Large-scale mRNA analysis of female skeletal muscles during 60 days of bed rest with and without exercise or dietary protein supplementation as countermeasures. Physiol. Genom. 2009, 38, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Alibegovic, A.C.; Sonne, M.P.; Hojbjerre, L.; Bork-Jensen, J.; Jacobsen, S.; Nilsson, E.; Faerch, K.; Hiscock, N.; Mortensen, B.; Friedrichsen, M.; et al. Insulin resistance induced by physical inactivity is associated with multiple transcriptional changes in skeletal muscle in young men. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E752–E763. [Google Scholar] [CrossRef] [PubMed]
- Trevino, M.B.; Zhang, X.; Standley, R.A.; Wang, M.; Han, X.; Reis, F.C.G.; Periasamy, M.; Yu, G.; Kelly, D.P.; Goodpaster, B.H.; et al. Loss of mitochondrial energetics is associated with poor recovery of muscle function but not mass following disuse atrophy. Am. J. Physiol. Endocrinol. Metab. 2019, 317, E899–E910. [Google Scholar] [CrossRef]
- Rullman, E.; Fernandez-Gonzalo, R.; Mekjavic, I.B.; Gustafsson, T.; Eiken, O. MEF2 as upstream regulator of the transcriptome signature in human skeletal muscle during unloading. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R799–R809. [Google Scholar] [CrossRef]
- Fernandez-Gonzalo, R.; Tesch, P.A.; Lundberg, T.R.; Alkner, B.A.; Rullman, E.; Gustafsson, T. Three months of bed rest induce a residual transcriptomic signature resilient to resistance exercise countermeasures. FASEB J. 2020, 34, 7958–7969. [Google Scholar] [CrossRef]
- Mahmassani, Z.S.; Reidy, P.T.; McKenzie, A.I.; Stubben, C.; Howard, M.T.; Drummond, M.J. Age-dependent skeletal muscle transcriptome response to bed rest-induced atrophy. J. Appl. Physiol. 2019, 126, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Wiggs, M.P.; Duarte, J.A.; Zergeroglu, A.M.; Demirel, H.A. Mitochondrial signaling contributes to disuse muscle atrophy. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E31–E39. [Google Scholar] [CrossRef]
- Max, S.R. Disuse atrophy of skeletal muscle: Loss of functional activity of mitochondria. Biochem. Biophys. Res. Commun. 1972, 46, 1394–1398. [Google Scholar] [CrossRef]
- Memme, J.M.; Slavin, M.; Moradi, N.; Hood, D.A. Mitochondrial Bioenergetics and Turnover during Chronic Muscle Disuse. Int. J. Mol. Sci. 2021, 22, 5179. [Google Scholar] [CrossRef] [PubMed]
- Hyatt, H.W.; Ozdemir, M.; Yoshihara, T.; Nguyen, B.L.; Deminice, R.; Powers, S.K. Calpains play an essential role in mechanical ventilation-induced diaphragmatic weakness and mitochondrial dysfunction. Redox Biol. 2021, 38, 101802. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.L.; Yeo, D. Mitochondrial dysregulation and muscle disuse atrophy. F1000Research 2019, 8. [Google Scholar] [CrossRef]
- Standley, R.A.; Distefano, G.; Trevino, M.B.; Chen, E.; Narain, N.R.; Greenwood, B.; Kondakci, G.; Tolstikov, V.V.; Kiebish, M.A.; Yu, G.; et al. Skeletal Muscle Energetics and Mitochondrial Function Are Impaired Following 10 Days of Bed Rest in Older Adults. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 1744–1753. [Google Scholar] [CrossRef]
- Buso, A.; Comelli, M.; Picco, R.; Isola, M.; Magnesa, B.; Pisot, R.; Rittweger, J.; Salvadego, D.; Simunic, B.; Grassi, B.; et al. Mitochondrial Adaptations in Elderly and Young Men Skeletal Muscle Following 2 Weeks of Bed Rest and Rehabilitation. Front. Physiol. 2019, 10, 474. [Google Scholar] [CrossRef]
- Jiroutkova, K.; Krajcova, A.; Ziak, J.; Fric, M.; Waldauf, P.; Dzupa, V.; Gojda, J.; Nemcova-Furstova, V.; Kovar, J.; Elkalaf, M.; et al. Mitochondrial function in skeletal muscle of patients with protracted critical illness and ICU-acquired weakness. Crit. Care 2015, 19, 448. [Google Scholar] [CrossRef]
- Theeuwes, W.F.; Gosker, H.R.; Langen, R.C.J.; Verhees, K.J.P.; Pansters, N.A.M.; Schols, A.; Remels, A.H.V. Inactivation of glycogen synthase kinase-3beta (GSK-3beta) enhances skeletal muscle oxidative metabolism. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 3075–3086. [Google Scholar] [CrossRef]
- Oishi, Y.; Ogata, T.; Yamamoto, K.I.; Terada, M.; Ohira, T.; Ohira, Y.; Taniguchi, K.; Roy, R.R. Cellular adaptations in soleus muscle during recovery after hindlimb unloading. Acta Physiol. 2008, 192, 381–395. [Google Scholar] [CrossRef]
- Handschin, C.; Chin, S.; Li, P.; Liu, F.; Maratos-Flier, E.; Lebrasseur, N.K.; Yan, Z.; Spiegelman, B.M. Skeletal muscle fiber-type switching, exercise intolerance, and myopathy in PGC-1alpha muscle-specific knock-out animals. J. Biol. Chem. 2007, 282, 30014–30021. [Google Scholar] [CrossRef]
- Tadaishi, M.; Miura, S.; Kai, Y.; Kano, Y.; Oishi, Y.; Ezaki, O. Skeletal muscle-specific expression of PGC-1alpha-b, an exercise-responsive isoform, increases exercise capacity and peak oxygen uptake. PLoS ONE 2011, 6, e28290. [Google Scholar] [CrossRef]
- Theeuwes, W.F.; Gosker, H.R.; Schols, A.; Langen, R.C.J.; Remels, A.H.V. Regulation of PGC-1alpha expression by a GSK-3beta-TFEB signaling axis in skeletal muscle. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118610. [Google Scholar] [CrossRef]
- Xu, M.; Chen, X.; Chen, D.; Yu, B.; Li, M.; He, J.; Huang, Z. MicroRNA-499–5p regulates skeletal myofiber specification via NFATc1/MEF2C pathway and Thrap1/MEF2C axis. Life Sci. 2018, 215, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Kozlovskaya, I.B.; Sayenko, I.V.; Sayenko, D.G.; Miller, T.F.; Khusnutdinova, D.R.; Melnik, K.A. Role of support afferentation in control of the tonic muscle activity. Acta Astronaut. 2007, 60, 285–294. [Google Scholar] [CrossRef]
- Mirzoev, T.M. Skeletal Muscle Recovery from Disuse Atrophy: Protein Turnover Signaling and Strategies for Accelerating Muscle Regrowth. Int. J. Mol. Sci. 2020, 21, 7940. [Google Scholar] [CrossRef]
- Clement, G. International roadmap for artificial gravity research. NPJ Microgravity 2017, 3, 29. [Google Scholar] [CrossRef] [PubMed]
- Layne, C.S.; Mulavara, A.P.; Pruett, C.J.; McDonald, P.V.; Kozlovskaya, I.B.; Bloomberg, J.J. The use of in-flight foot pressure as a countermeasure to neuromuscular degradation. Acta Astronaut. 1998, 42, 231–246. [Google Scholar] [CrossRef]
- Yarmanova, E.N.; Kozlovskaya, I.B.; Khimoroda, N.N.; Fomina, E.V. Evolution of Russian Microgravity Countermeasures. Aerosp. Med. Hum. Perform. 2015, 86, A32–A37. [Google Scholar] [CrossRef] [PubMed]
- Vitadello, M.; Germinario, E.; Ravara, B.; Libera, L.D.; Danieli-Betto, D.; Gorza, L. Curcumin counteracts loss of force and atrophy of hindlimb unloaded rat soleus by hampering neuronal nitric oxide synthase untethering from sarcolemma. J. Physiol. 2014, 592, 2637–2652. [Google Scholar] [CrossRef] [PubMed]
- Fomina, E.V.; Lysova, N.Y.; Chernova, M.V.; Khustnudinova, D.R.; Kozlovskaya, I.B. [Comparative Analisys of Efficacy of Countermeasure Provided by Different Modes of Locomotor Training in Space Flight.]. Fiziol. Cheloveka 2016, 42, 84–91. [Google Scholar] [PubMed]
- English, K.L.; Downs, M.; Goetchius, E.; Buxton, R.; Ryder, J.W.; Ploutz-Snyder, R.; Guilliams, M.; Scott, J.M.; Ploutz-Snyder, L.L. High intensity training during spaceflight: Results from the NASA Sprint Study. NPJ Microgravity 2020, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Kozlovskaya, I.B.; Grigoriev, A.I. Russian system of countermeasures on board of the International Space Station (ISS): The first results. Acta Astronaut. 2004, 55, 233–237. [Google Scholar] [CrossRef]
- Hotta, N.; Ishida, K.; Sato, K.; Koike, T.; Katayama, K.; Akima, H. The effect of intense interval cycle-training on unloading-induced dysfunction and atrophy in the human calf muscle. J. Physiol. Anthr. 2011, 30, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Ohira, Y.; Yoshinaga, T.; Ohara, M.; Nonaka, I.; Yoshioka, T.; Yamashita-Goto, K.; Shenkman, B.S.; Kozlovskaya, I.B.; Roy, R.R.; Edgerton, V.R. Myonuclear domain and myosin phenotype in human soleus after bed rest with or without loading. J. Appl. Physiol. 1999, 87, 1776–1785. [Google Scholar] [CrossRef] [PubMed]
- Usuki, F.; Fujimura, M.; Nakamura, A.; Nakano, J.; Okita, M.; Higuchi, I. Local Vibration Stimuli Induce Mechanical Stress-Induced Factors and Facilitate Recovery From Immobilization-Induced Oxidative Myofiber Atrophy in Rats. Front. Physiol. 2019, 10, 759. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.M.; Van Pelt, D.W.; Confides, A.L.; Hunt, E.R.; Hettinger, Z.R.; Laurin, J.L.; Reid, J.J.; Peelor, F.F., 3rd; Butterfield, T.A.; Dupont-Versteegden, E.E.; et al. Massage as a mechanotherapy promotes skeletal muscle protein and ribosomal turnover but does not mitigate muscle atrophy during disuse in adult rats. Acta Physiol. 2020, 229, e13460. [Google Scholar] [CrossRef]
- Symons, T.B.; Sheffield-Moore, M.; Chinkes, D.L.; Ferrando, A.A.; Paddon-Jones, D. Artificial gravity maintains skeletal muscle protein synthesis during 21 days of simulated microgravity. J. Appl. Physiol. 2009, 107, 34–38. [Google Scholar] [CrossRef]
- Caiozzo, V.J.; Haddad, F.; Lee, S.; Baker, M.; Paloski, W.; Baldwin, K.M. Artificial gravity as a countermeasure to microgravity: A pilot study examining the effects on knee extensor and plantar flexor muscle groups. J. Appl. Physiol. 2009, 107, 39–46. [Google Scholar] [CrossRef]
- D’Aunno, D.S.; Robinson, R.R.; Smith, G.S.; Thomason, D.B.; Booth, F.W. Intermittent acceleration as a countermeasure to soleus muscle atrophy. J. Appl. Physiol. 1992, 72, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Clement, G.; Paloski, W.H.; Rittweger, J.; Linnarsson, D.; Bareille, M.P.; Mulder, E.; Wuyts, F.L.; Zange, J. Centrifugation as a countermeasure during bed rest and dry immersion: What has been learned? J. Musculoskelet Neuronal. Interact. 2016, 16, 84–91. [Google Scholar] [PubMed]
- Mirzoev, T.; Tyganov, S.; Petrova, I.; Gnyubkin, V.; Laroche, N.; Vico, L.; Shenkman, B. Divergent Anabolic Signalling responses of Murine Soleus and Tibialis Anterior Muscles to Chronic 2G Hypergravity. Sci. Rep. 2017, 7, 3514. [Google Scholar] [CrossRef]
- Shenkman, B.S.; Podlubnaia, Z.A.; Vikhliantsev, I.M.; Litvinova, K.S.; Udal’tsov, S.N.; Nemirovskaia, T.L.; Lemesheva Iu, S.; Mukhina, A.M.; Kozlovskaia, I.B. Human soleus fibers contractile characteristics and sarcomeric cytoskeletal proteins after gravitational unloading. Contribution of support stimulus]. Biofizika 2004, 49, 881–890. [Google Scholar] [PubMed]
- Kyparos, A.; Feeback, D.L.; Layne, C.S.; Martinez, D.A.; Clarke, M.S. Mechanical stimulation of the plantar foot surface attenuates soleus muscle atrophy induced by hindlimb unloading in rats. J. Appl. Physiol. 2005, 99, 739–746. [Google Scholar] [CrossRef]
- Vanderthommen, M.; Crielaard, J.M. [Muscle electric stimulation in sports medicine]. Rev. Med. Liege 2001, 56, 391–395. [Google Scholar]
- Gerovasili, V.; Stefanidis, K.; Vitzilaios, K.; Karatzanos, E.; Politis, P.; Koroneos, A.; Chatzimichail, A.; Routsi, C.; Roussos, C.; Nanas, S. Electrical muscle stimulation preserves the muscle mass of critically ill patients: A randomized study. Crit. Care 2009, 13, R161. [Google Scholar] [CrossRef]
- Toth, M.J.; Voigt, T.B.; Tourville, T.W.; Prior, S.M.; Guigni, B.A.; Schlosberg, A.V.; Smith, I.B.; Forest, T.J.; Kaufman, P.A.; Wood, M.E.; et al. Effect of neuromuscular electrical stimulation on skeletal muscle size and function in patients with breast cancer receiving chemotherapy. J. Appl. Physiol. 2020, 128, 1654–1665. [Google Scholar] [CrossRef]
- Morrissey, M.C.; Brewster, C.E.; Shields, C.L., Jr.; Brown, M. The effects of electrical stimulation on the quadriceps during postoperative knee immobilization. Am. J. Sports Med. 1985, 13, 40–45. [Google Scholar] [CrossRef]
- Kwong, W.H.; Vrbova, G. Effects of low-frequency electrical stimulation on fast and slow muscles of the rat. Pflug. Arch. 1981, 391, 200–207. [Google Scholar] [CrossRef]
- Scott, O.M.; Vrbova, G.; Hyde, S.A.; Dubowitz, V. Effects of chronic low frequency electrical stimulation on normal human tibialis anterior muscle. J. Neurol. Neurosurg. Psychiatry 1985, 48, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Petrie, M.; Suneja, M.; Shields, R.K. Low-frequency stimulation regulates metabolic gene expression in paralyzed muscle. J. Appl. Physiol. 2015, 118, 723–731. [Google Scholar] [CrossRef]
- Nuhr, M.J.; Pette, D.; Berger, R.; Quittan, M.; Crevenna, R.; Huelsman, M.; Wiesinger, G.F.; Moser, P.; Fialka-Moser, V.; Pacher, R. Beneficial effects of chronic low-frequency stimulation of thigh muscles in patients with advanced chronic heart failure. Eur. Heart J. 2004, 25, 136–143. [Google Scholar] [CrossRef]
- Cherepakhin, M.A.; Kakurin, L.I.; Il’ina-Kakueva, E.I.; Fedorenko, G.T. [Evaluation of the effectiveness of electrostimulation of the muscles in preventing disorders related to prolonged limited motor activity in man]. Kosm. Biol. Aviakosm. Med. 1977, 11, 64–68. [Google Scholar]
- Shenkman, B.S.; Liubaeva, E.V.; Popov, D.V.; Netreba, A.I.; Tarasova, O.S.; Vdovina, A.B.; Tarakin, P.P.; Lemesheva Iu, S.; Belichenko, O.I.; Sinitsyn, V.E. [Effects of chronic low-frequency electrical stimulation of human knee extensor muscles exposed to static passive stretching]. Fiziol. Cheloveka 2006, 32, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Conjard, A.; Peuker, H.; Pette, D. Energy state and myosin heavy chain isoforms in single fibres of normal and transforming rabbit muscles. Pflug. Arch. 1998, 436, 962–969. [Google Scholar] [CrossRef] [PubMed]
- Ausoni, S.; Gorza, L.; Schiaffino, S.; Gundersen, K.; Lomo, T. Expression of myosin heavy chain isoforms in stimulated fast and slow rat muscles. J. Neurosci. 1990, 10, 153–160. [Google Scholar] [CrossRef]
- Kubis, H.P.; Scheibe, R.J.; Meissner, J.D.; Hornung, G.; Gros, G. Fast-to-slow transformation and nuclear import/export kinetics of the transcription factor NFATc1 during electrostimulation of rabbit muscle cells in culture. J. Physiol. 2002, 541, 835–847. [Google Scholar] [CrossRef]
- Canon, F.; Goubel, F.; Guezennec, C.Y. Effects of chronic low frequency stimulation on contractile and elastic properties of hindlimb suspended rat soleus muscle. Eur. J. Appl. Physiol. Occup. Physiol. 1998, 77, 118–124. [Google Scholar] [CrossRef]
- Leterme, D.; Falempin, M. Compensatory effects of chronic electrostimulation on unweighted rat soleus muscle. Pflug. Arch. 1994, 426, 155–160. [Google Scholar] [CrossRef]
- Stein, T.P.; Blanc, S. Does protein supplementation prevent muscle disuse atrophy and loss of strength? Crit. Rev. Food Sci. Nutr. 2011, 51, 828–834. [Google Scholar] [CrossRef]
- Stuart, C.A.; Shangraw, R.E.; Peters, E.J.; Wolfe, R.R. Effect of dietary protein on bed-rest-related changes in whole-body-protein synthesis. Am. J. Clin. Nutr. 1990, 52, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D. Interplay of stress and physical inactivity on muscle loss: Nutritional countermeasures. J. Nutr. 2006, 136, 2123–2126. [Google Scholar] [CrossRef]
- Brooks, N.; Cloutier, G.J.; Cadena, S.M.; Layne, J.E.; Nelsen, C.A.; Freed, A.M.; Roubenoff, R.; Castaneda-Sceppa, C. Resistance training and timed essential amino acids protect against the loss of muscle mass and strength during 28 days of bed rest and energy deficit. J. Appl. Physiol. 2008, 105, 241–248. [Google Scholar] [CrossRef]
- Powers, S.K.; Kavazis, A.N.; McClung, J.M. Oxidative stress and disuse muscle atrophy. J. Appl. Physiol. 2007, 102, 2389–2397. [Google Scholar] [CrossRef]
- Appell, H.J.; Duarte, J.A.; Soares, J.M. Supplementation of vitamin E may attenuate skeletal muscle immobilization atrophy. Int. J. Sports Med. 1997, 18, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Desaphy, J.F.; Pierno, S.; Liantonio, A.; Giannuzzi, V.; Digennaro, C.; Dinardo, M.M.; Camerino, G.M.; Ricciuti, P.; Brocca, L.; Pellegrino, M.A.; et al. Antioxidant treatment of hindlimb-unloaded mouse counteracts fiber type transition but not atrophy of disused muscles. Pharm. Res. 2010, 61, 553–563. [Google Scholar] [CrossRef]
- Powers, S.K. Can antioxidants protect against disuse muscle atrophy? Sports Med. 2014, 44 (Suppl. 2), S155–S165. [Google Scholar] [CrossRef]
- Whitley, K.C.; Hamstra, S.I.; Baranowski, R.W.; Watson, C.J.F.; MacPherson, R.E.K.; MacNeil, A.J.; Roy, B.D.; Vandenboom, R.; Fajardo, V.A. GSK3 inhibition with low dose lithium supplementation augments murine muscle fatigue resistance and specific force production. Physiol. Rep. 2020, 8, e14517. [Google Scholar] [CrossRef] [PubMed]
- Mirzoev, T.M.; Sharlo, K.A.; Shenkman, B.S. The Role of GSK-3β in the Regulation of Protein Turnover, Myosin Phenotype, and Oxidative Capacity in Skeletal Muscle under Disuse Conditions. Int. J. Mol. Sci. 2021, 22, 5081. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).