Vasopressin as a Possible Link between Sleep-Disturbances and Memory Problems
Abstract
:1. Introduction
2. Vasopressin
2.1. Receptor Subtypes
2.2. Anatomy
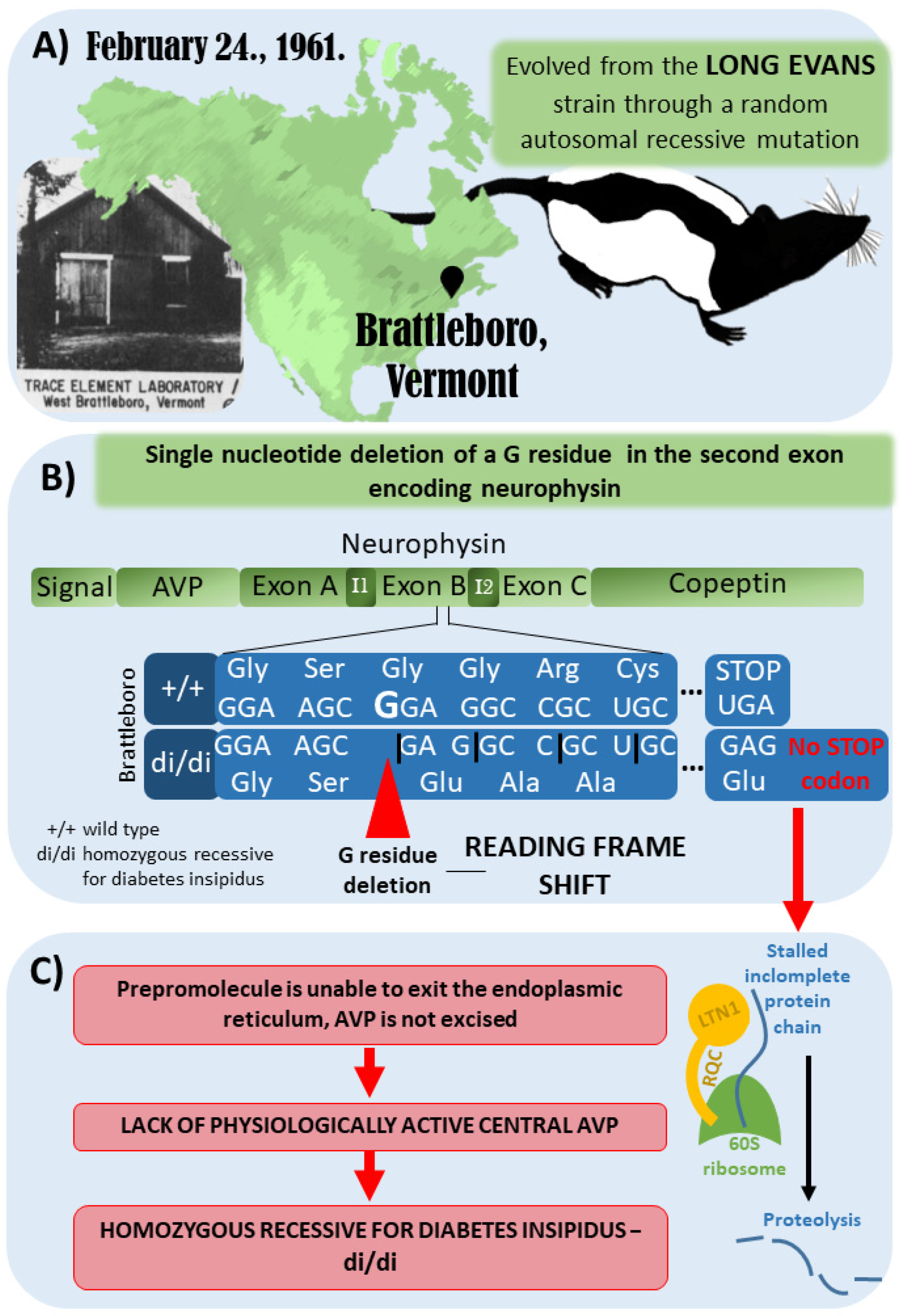
3. The Brattleboro Rat as a Model of AVP-Deficiency
4. Circadian Rhythm
The Circadian Rhythms of Brattleboro Rats
5. Suprachiasmatic Nucleus (SCN), the Endogenous Clock
5.1. Suprachiasmatic Nucleus and Vasopressin
5.2. The Suprachiasmatic Nucleus of the Brattleboro Rat
6. Importance of Sleep in Physiology and Disease
6.1. Electroencephalographic (EEG) Stages of Sleep
6.2. Theta Waves
6.3. Vasopressin and Sleep (Figure 3)
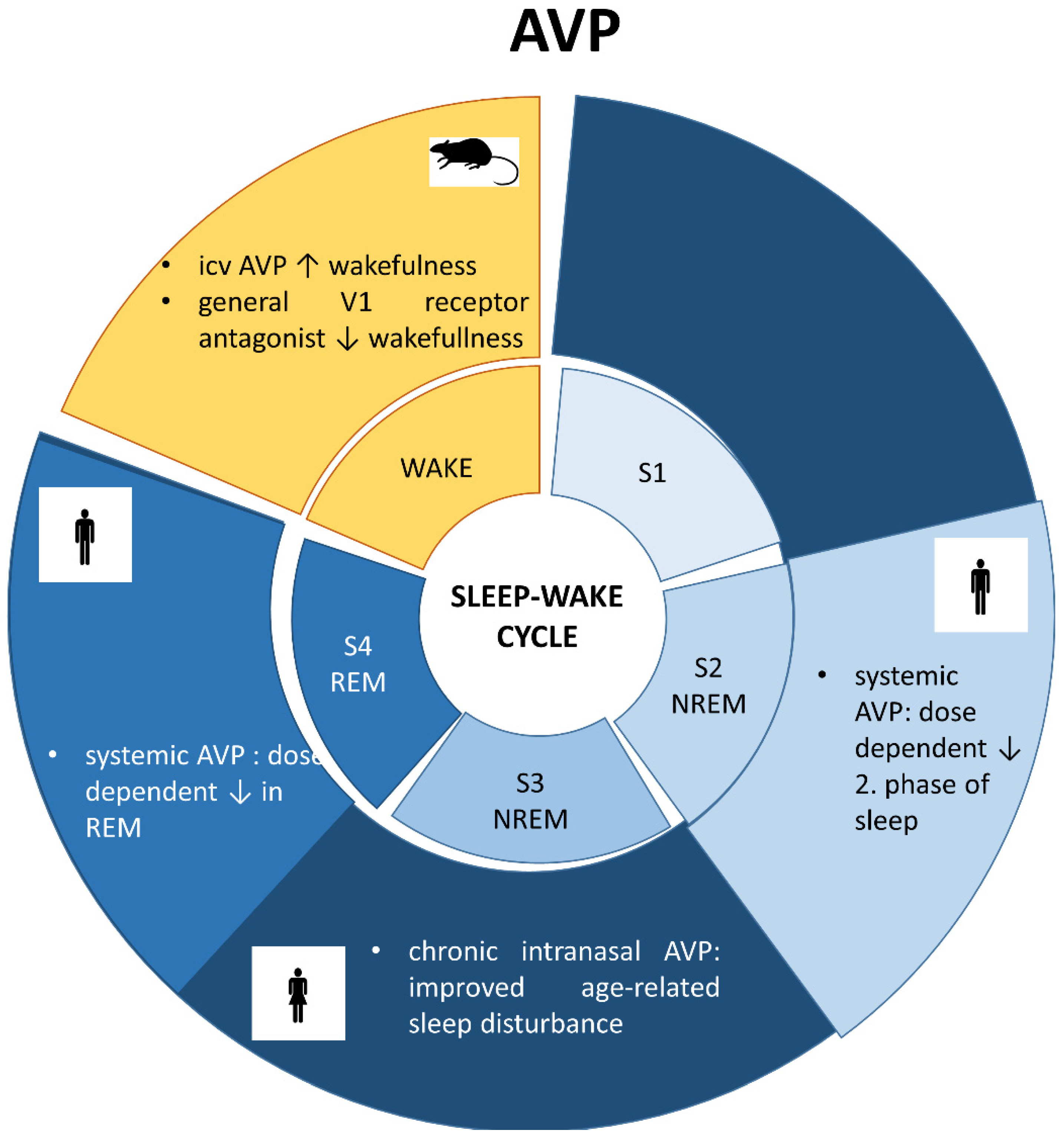
6.4. Sleep in Brattleboro Rats
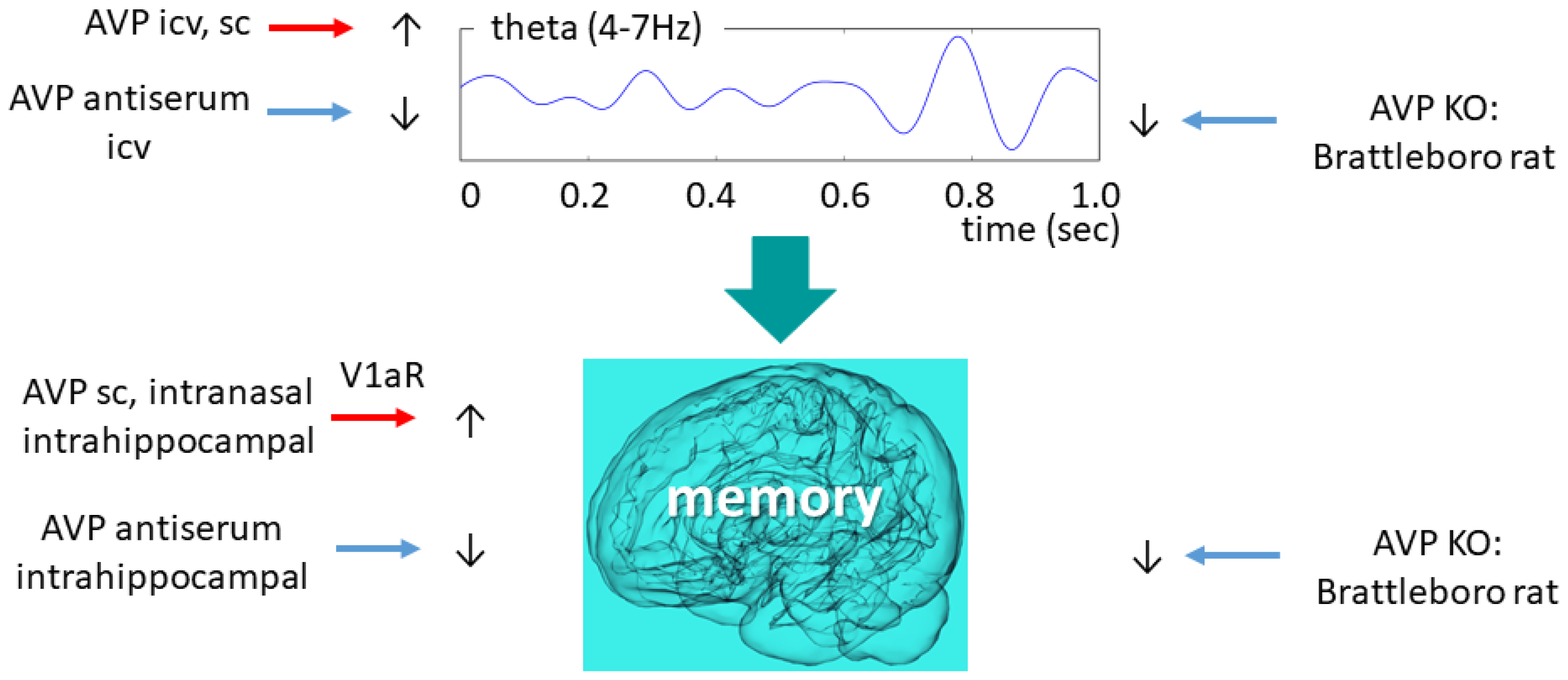
6.5. Vasopressin and Theta Oscillation with Focus on Brattleboro Rat
7. Memory Disturbances Associated with Improper Sleep
7.1. Sleep-Dependent Memory Consolidation
7.2. Vasopressin Function Related to Memory
7.3. Memory Problems in Brattleboro Rats
8. Sex Differences
9. Discussion
10. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Manoach, D.S.; Stickgold, R. Abnormal Sleep Spindles, Memory Consolidation, and Schizophrenia. Annu. Rev. Clin. Psychol. 2019, 15, 451–479. [Google Scholar] [CrossRef]
- Kotagal, S.; Broomall, E. Sleep in children with autism spectrum disorder. Pediatr. Neurol. 2012, 47, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Ben Shalom, D. Memory in autism: Review and synthesis. Cortex 2003, 39, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Singareddy, R.K.; Balon, R. Sleep in posttraumatic stress disorder. Ann. Clin. Psychiatry 2002, 14, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Matsuzaki, T. Regulation of aquaporins by vasopressin in the kidney. Vitam. Horm. 2015, 98, 307–337. [Google Scholar]
- Zelena, D.; Pinter, O.; Balazsfi, D.G.; Langnaese, K.; Richter, K.; Landgraf, R.; Makara, G.B.; Engelmann, M. Vasopressin signaling at brain level controls stress hormone release: The vasopressin-deficient Brattleboro rat as a model. Amino Acids 2015, 47, 2245–2253. [Google Scholar] [CrossRef]
- Born, J.; Kellner, C.; Uthgenannt, D.; Kern, W.; Fehm, H.L. Vasopressin regulates human sleep by reducing rapid-eye-movement sleep. Am. J. Physiol. 1992, 262, E295–E300. [Google Scholar] [CrossRef]
- Perras, B.; Pannenborg, H.; Marshall, L.; Pietrowsky, R.; Born, J.; Lorenz Fehm, H. Beneficial treatment of age-related sleep disturbances with prolonged intranasal vasopressin. J. Clin. Psychopharmacol. 1999, 19, 28–36. [Google Scholar] [CrossRef]
- Perras, B.; Wagner, U.; Born, J.; Fehm, H.L. Improvement of sleep and pituitary-adrenal inhibition after subchronic intranasal vasopressin treatment in elderly humans. J. Clin. Psychopharmacol. 2003, 23, 35–44. [Google Scholar] [CrossRef]
- Bittman, E.L. Vasopressin: More than just an output of the circadian pacemaker? Focus on “Vasopressin receptor V1a regulates circadian rhythms of locomotor activity and expression of clock-controlled genes in the suprachiasmatic nuclei”. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R821–R823. [Google Scholar] [CrossRef] [Green Version]
- Croiset, G.; Nijsen, M.J.; Kamphuis, P.J. Role of corticotropin-releasing factor, vasopressin and the autonomic nervous system in learning and memory. Eur. J. Pharmacol. 2000, 405, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Abramova, O.; Zorkina, Y.; Ushakova, V.; Zubkov, E.; Morozova, A.; Chekhonin, V. The role of oxytocin and vasopressin dysfunction in cognitive impairment and mental disorders. Neuropeptides 2020, 83, 102079. [Google Scholar] [CrossRef]
- Urban, I.J. Effects of vasopressin and related peptides on neurons of the rat lateral septum and ventral hippocampus. Prog. Brain Res. 1998, 119, 285–310. [Google Scholar] [PubMed]
- Torok, B.; Fodor, A.; Klausz, B.; Varga, J.; Zelena, D. Ameliorating schizophrenia-like symptoms in vasopressin deficient male Brattleboro rat by chronic antipsychotic treatment. Eur. J. Pharmacol. 2021, 909, 174383. [Google Scholar] [CrossRef] [PubMed]
- Demeter, K.; Torok, B.; Fodor, A.; Varga, J.; Ferenczi, S.; Kovacs, K.J.; Eszik, I.; Szegedi, V.; Zelena, D. Possible contribution of epigenetic changes in the development of schizophrenia-like behavior in vasopressin-deficient Brattleboro rats. Behav. Brain Res. 2016, 300, 123–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varga, J.; Klausz, B.; Domokos, A.; Kalman, S.; Pakaski, M.; Szucs, S.; Garab, D.; Zvara, A.; Puskas, L.; Kalman, J.; et al. Increase in Alzheimer’s related markers preceeds memory disturbances: Studies in vasopressin-deficient Brattleboro rat. Brain Res. Bull. 2014, 100, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Zelena, D. Comparison of Natural and Artificial Vasopressin Deficiency: Why is the Latter Lethal. Russ. J. Genet. Appl. Res. 2017, 7, 243–248. [Google Scholar] [CrossRef]
- Kuenzel, W.J.; Kang, S.W.; Jurkevich, A. The vasotocinergic system and its role in the regulation of stress in birds. Vitam. Horm. 2020, 113, 183–216. [Google Scholar]
- Glavas, M.; Gitlin-Domagalska, A.; Debowski, D.; Ptaszynska, N.; Legowska, A.; Rolka, K. Vasopressin and Its Analogues: From Natural Hormones to Multitasking Peptides. Int. J. Mol. Sci. 2022, 23, 3068. [Google Scholar] [CrossRef]
- Csikota, P.; Fodor, A.; Balazsfi, D.; Pinter, O.; Mizukami, H.; Weger, S.; Heilbronn, R.; Engelmann, M.; Zelena, D. Vasopressinergic control of stress-related behavior: Studies in Brattleboro rats. Stress 2016, 19, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Koshimizu, T.A.; Nakamura, K.; Egashira, N.; Hiroyama, M.; Nonoguchi, H.; Tanoue, A. Vasopressin V1a and V1b receptors: From molecules to physiological systems. Physiol. Rev. 2012, 92, 1813–1864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalsbeek, A.; Fliers, E.; Hofman, M.A.; Swaab, D.F.; Buijs, R.M. Vasopressin and the output of the hypothalamic biological clock. J. Neuroendocrinol. 2010, 22, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Ingram, C.D.; Ciobanu, R.; Coculescu, I.L.; Tanasescu, R.; Coculescu, M.; Mihai, R. Vasopressin neurotransmission and the control of circadian rhythms in the suprachiasmatic nucleus. Prog. Brain Res. 1998, 119, 351–364. [Google Scholar] [PubMed]
- Bredewold, R.; Veenema, A.H. Sex differences in the regulation of social and anxiety-related behaviors: Insights from vasopressin and oxytocin brain systems. Curr. Opin. Neurobiol. 2018, 49, 132–140. [Google Scholar] [CrossRef]
- Bao, A.M.; Swaab, D.F. Corticotropin-releasing hormone and arginine vasopressin in depression focus on the human postmortem hypothalamus. Vitam. Horm. 2010, 82, 339–365. [Google Scholar] [PubMed]
- Insel, T.R. The challenge of translation in social neuroscience: A review of oxytocin, vasopressin, and affiliative behavior. Neuron 2010, 65, 768–779. [Google Scholar] [CrossRef] [Green Version]
- Young, L.J.; Toloczko, D.; Insel, T.R. Localization of vasopressin (V1a) receptor binding and mRNA in the rhesus monkey brain. J. Neuroendocrinol. 1999, 11, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, R.S.; Chen, Q.; Schreiber, S.S.; Brinton, R.D. Localization of V1a vasopressin receptor mRNA expression in cultured neurons, astroglia, and oligodendroglia of rat cerebral cortex. Brain Res. Mol. Brain Res. 1997, 45, 138–140. [Google Scholar] [CrossRef]
- Bakker, R.; Tiesinga, P.; Kotter, R. The Scalable Brain Atlas: Instant Web-Based Access to Public Brain Atlases and Related Content. Neuroinformatics 2015, 13, 353–366. [Google Scholar] [CrossRef] [Green Version]
- Iovino, M.; Giagulli, V.A.; Licchelli, B.; Iovino, E.; Guastamacchia, E.; Triggiani, V. Synaptic Inputs of Neural Afferent Pathways to Vasopressin- and Oxytocin-Secreting Neurons of Supraoptic and Paraventricular Hypothalamic Nuclei. Endocr. Metab. Immune Disord. Drug Targets 2016, 16, 276–287. [Google Scholar] [CrossRef]
- Ludwig, M.; Bull, P.M.; Tobin, V.A.; Sabatier, N.; Landgraf, R.; Dayanithi, G.; Leng, G. Regulation of activity-dependent dendritic vasopressin release from rat supraoptic neurones. J. Physiol. 2005, 564 Pt 2, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Douglas, A.J.; Johnstone, L.E.; Neumann, I.; Leng, G.; Russell, J.A. Oxytocin neurones in the supraoptic nucleus (SON) are inhibited by endogenous opioids in late pregnant rats. Gene Ther. 1994, 1 (Suppl. S1), 84. [Google Scholar]
- Ludwig, M.; Leng, G. Intrahypothalamic vasopressin release. An inhibitor of systemic vasopressin secretion? Adv. Exp. Med. Biol. 1998, 449, 163–173. [Google Scholar] [PubMed]
- Albers, H.E. Species, sex and individual differences in the vasotocin/vasopressin system: Relationship to neurochemical signaling in the social behavior neural network. Front. Neuroendocrinol. 2015, 36, 49–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engelmann, M.; Ludwig, M. The activity of the hypothalamo-neurohypophysial system in response to acute stressor exposure: Neuroendocrine and electrophysiological observations. Stress 2004, 7, 91–96. [Google Scholar] [CrossRef]
- Landgraf, R.; Neumann, I.D. Vasopressin and oxytocin release within the brain: A dynamic concept of multiple and variable modes of neuropeptide communication. Front. Neuroendocrinol. 2004, 25, 150–176. [Google Scholar] [CrossRef]
- Ludwig, M.; Stern, J. Multiple signalling modalities mediated by dendritic exocytosis of oxytocin and vasopressin. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Raadsheer, F.C.; Sluiter, A.A.; Ravid, R.; Tilders, F.J.; Swaab, D.F. Localization of corticotropin-releasing hormone (CRH) neurons in the paraventricular nucleus of the human hypothalamus; age-dependent colocalization with vasopressin. Brain Res. 1993, 615, 50–62. [Google Scholar] [CrossRef] [Green Version]
- Rood, B.D.; De Vries, G.J. Vasopressin innervation of the mouse (Mus musculus) brain and spinal cord. J. Comp. Neurol. 2011, 519, 2434–2474. [Google Scholar] [CrossRef] [Green Version]
- Zink, C.F.; Stein, J.L.; Kempf, L.; Hakimi, S.; Meyer-Lindenberg, A. Vasopressin modulates medial prefrontal cortex-amygdala circuitry during emotion processing in humans. J. Neurosci. 2010, 30, 7017–7022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sawyer, W.H.; Valtin, H.; Sokol, H.W. Neurohypophysial Principles in Rats with Familial Hypothalamic Diabetes Insipidus (Brattleboro Strain). Endocrinology 1964, 74, 153–155. [Google Scholar] [CrossRef]
- Valtin, H. The discovery of the Brattleboro rat, recommended nomenclature, and the question of proper controls. Ann. N. Y. Acad. Sci. 1982, 394, 1–9. [Google Scholar] [CrossRef]
- Schmale, H.; Richter, D. Single base deletion in the vasopressin gene is the cause of diabetes insipidus in Brattleboro rats. Nature 1984, 308, 705–709. [Google Scholar] [CrossRef]
- Schmale, H.; Ivell, R.; Breindl, M.; Darmer, D.; Richter, D. The mutant vasopressin gene from diabetes insipidus (Brattleboro) rats is transcribed but the message is not efficiently translated. EMBO J. 1984, 3, 3289–3293. [Google Scholar] [CrossRef]
- Defenouillere, Q.; Zhang, E.; Namane, A.; Mouaikel, J.; Jacquier, A.; Fromont-Racine, M. Rqc1 and Ltn1 Prevent C-terminal Alanine-Threonine Tail (CAT-tail)-induced Protein Aggregation by Efficient Recruitment of Cdc48 on Stalled 60S Subunits. J. Biol. Chem. 2016, 291, 12245–12253. [Google Scholar] [CrossRef] [Green Version]
- Danguir, J. Sleep deficits in rats with hereditary diabetes insipidus. Nature 1983, 304, 163–164. [Google Scholar] [CrossRef]
- Vitaterna, M.H.; Takahashi, J.S.; Turek, F.W. Overview of circadian rhythms. Alcohol Res. Health 2001, 25, 85–93. [Google Scholar]
- Williams, A.R.; Carey, R.J.; Miller, M. Behavioral differences between vasopressin-deficient (Brattleboro) and normal Long-Evans rats. Peptides 1983, 4, 711–716. [Google Scholar] [CrossRef]
- Ambrogi Lorenzini, C.; Bucherelli, C.; Giachetti, A.; Tassoni, G. The behavior of the homozygous and heterozygous sub-types of rats which are genetically-selected for diabetes insipidus: A comparison with Long Evans and Wistar stocks. Experientia 1991, 47, 1019–1026. [Google Scholar] [CrossRef]
- Zelena, D.; Mergl, Z.; Makara, G.B. Postnatal development in vasopressin deficient Brattleboro rats with special attention to the hypothalamo-pituitary-adrenal axis function: The role of maternal genotype. Int. J. Dev. Neurosci. 2009, 27, 175–183. [Google Scholar] [CrossRef]
- Schank, J.C. Early locomotor and social effects in vasopressin deficient neonatal rats. Behav. Brain Res. 2009, 197, 166–177. [Google Scholar] [CrossRef]
- Schatz, K.C.; Kyne, R.F.; Parmeter, S.L.; Paul, M.J. Investigation of social, affective, and locomotor behavior of adolescent Brattleboro rats reveals a link between vasopressin’s actions on arousal and social behavior. Horm. Behav. 2018, 106, 1–9. [Google Scholar] [CrossRef]
- Mlynarik, M.; Zelena, D.; Bagdy, G.; Makara, G.B.; Jezova, D. Signs of attenuated depression-like behavior in vasopressin deficient Brattleboro rats. Horm. Behav. 2007, 51, 395–405. [Google Scholar] [CrossRef]
- Varga, J.; Domokos, A.; Barna, I.; Jankord, R.; Bagdy, G.; Zelena, D. Lack of vasopressin does not prevent the behavioural and endocrine changes induced by chronic unpredictable stress. Brain Res. Bull. 2011, 84, 45–52. [Google Scholar] [CrossRef]
- Fodor, A.; Klausz, B.; Pinter, O.; Daviu, N.; Rabasa, C.; Rotllant, D.; Balazsfi, D.; Kovacs, K.B.; Nadal, R.; Zelena, D. Maternal neglect with reduced depressive-like behavior and blunted c-fos activation in Brattleboro mothers, the role of central vasopressin. Horm. Behav. 2012, 62, 539–551. [Google Scholar] [CrossRef]
- Fodor, A.; Kovacs, K.B.; Balazsfi, D.; Klausz, B.; Pinter, O.; Demeter, K.; Daviu, N.; Rabasa, C.; Rotllant, D.; Nadal, R.; et al. Depressive- and anxiety-like behaviors and stress-related neuronal activation in vasopressin-deficient female Brattleboro rats. Physiol. Behav. 2016, 158, 100–111. [Google Scholar] [CrossRef]
- Zuena, A.R.; Zinni, M.; Giuli, C.; Cinque, C.; Alema, G.S.; Giuliani, A.; Catalani, A.; Casolini, P.; Cozzolino, R. Maternal exposure to environmental enrichment before and during gestation influences behaviour of rat offspring in a sex-specific manner. Physiol. Behav. 2016, 163, 274–287. [Google Scholar] [CrossRef]
- Zelena, D.; Langnaese, K.; Domokos, A.; Pinter, O.; Landgraf, R.; Makara, G.B.; Engelmann, M. Vasopressin administration into the paraventricular nucleus normalizes plasma oxytocin and corticosterone levels in Brattleboro rats. Endocrinology 2009, 150, 2791–2798. [Google Scholar] [CrossRef] [Green Version]
- Stephan, F.K.; Zucker, I. Circadian rhythms in drinking behavior and locomotor activity of rats are eliminated by hypothalamic lesions. Proc. Natl. Acad. Sci. USA 1972, 69, 1583–1586. [Google Scholar] [CrossRef] [Green Version]
- Moore, R.Y.; Eichler, V.B. Loss of a circadian adrenal corticosterone rhythm following suprachiasmatic lesions in the rat. Brain Res. 1972, 42, 201–206. [Google Scholar] [CrossRef]
- Welsh, D.K.; Logothetis, D.E.; Meister, M.; Reppert, S.M. Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 1995, 14, 697–706. [Google Scholar] [CrossRef] [Green Version]
- Reghunandanan, V.; Reghunandanan, R.; Singh, P.I. Neurotransmitters of the suprachiasmatic nucleus: Role in the regulation of circadian rhythms. Prog. Neurobiol. 1993, 41, 647–655. [Google Scholar] [CrossRef]
- Morin, L.P. Neuroanatomy of the extended circadian rhythm system. Exp. Neurol. 2013, 243, 4–20. [Google Scholar] [CrossRef] [Green Version]
- Trbovic, S.M. Schizophrenia as a possible dysfunction of the suprachiasmatic nucleus. Med. Hypotheses 2010, 74, 127–131. [Google Scholar] [CrossRef]
- Feifel, D.; Mexal, S.; Melendez, G.; Liu, P.Y.; Goldenberg, J.R.; Shilling, P.D. The brattleboro rat displays a natural deficit in social discrimination that is restored by clozapine and a neurotensin analog. Neuropsychopharmacology 2009, 34, 2011–2018. [Google Scholar] [CrossRef] [Green Version]
- Hastings, M.H.; Maywood, E.S.; Reddy, A.B. Two decades of circadian time. J. Neuroendocrinol. 2008, 20, 812–819. [Google Scholar] [CrossRef]
- Begemann, K.; Neumann, A.M.; Oster, H. Regulation and function of extra-SCN circadian oscillators in the brain. Acta Physiol. 2020, 229, e13446. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Zhang, H.; Su, Y. New Insights into the Diurnal Rhythmicity of Gut Microbiota and Its Crosstalk with Host Circadian Rhythm. Animals 2022, 12, 1677. [Google Scholar] [CrossRef]
- Bedont, J.L.; Rohr, K.E.; Bathini, A.; Hattar, S.; Blackshaw, S.; Sehgal, A.; Evans, J.A. Asymmetric vasopressin signaling spatially organizes the master circadian clock. J. Comp. Neurol. 2018, 526, 2048–2067. [Google Scholar] [CrossRef]
- Reppert, S.M.; Artman, H.G.; Swaminathan, S.; Fisher, D.A. Vasopressin exhibits a rhythmic daily pattern in cerebrospinal fluid but not in blood. Science 1981, 213, 1256–1257. [Google Scholar] [CrossRef]
- Li, J.D.; Burton, K.J.; Zhang, C.; Hu, S.B.; Zhou, Q.Y. Vasopressin receptor V1a regulates circadian rhythms of locomotor activity and expression of clock-controlled genes in the suprachiasmatic nuclei. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R824–R830. [Google Scholar] [CrossRef] [Green Version]
- Buijs, R.M.; Hurtado-Alvarado, G.; Soto-Tinoco, E. Vasopressin: An output signal from the suprachiasmatic nucleus to prepare physiology and behaviour for the resting phase. J. Neuroendocrinol. 2021, 33, e12998. [Google Scholar] [CrossRef]
- Rood, B.D.; Stott, R.T.; You, S.; Smith, C.J.; Woodbury, M.E.; De Vries, G.J. Site of origin of and sex differences in the vasopressin innervation of the mouse (Mus musculus) brain. J. Comp. Neurol. 2013, 521, 2321–2358. [Google Scholar] [CrossRef]
- Hume, C.; Allchorne, A.; Grinevich, V.; Leng, G.; Ludwig, M. Effects of optogenetic stimulation of vasopressinergic retinal afferents on suprachiasmatic neurones. J. Neuroendocrinol. 2019, 31, e12806. [Google Scholar] [CrossRef] [PubMed]
- Rohr, K.E.; Telega, A.; Savaglio, A.; Evans, J.A. Vasopressin regulates daily rhythms and circadian clock circuits in a manner influenced by sex. Horm. Behav. 2021, 127, 104888. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Suzuki, T.; Mizoro, Y.; Kori, H.; Okada, K.; Chen, Y.; Fustin, J.M.; Yamazaki, F.; Mizuguchi, N.; Zhang, J.; et al. Mice genetically deficient in vasopressin V1a and V1b receptors are resistant to jet lag. Science 2013, 342, 85–90. [Google Scholar] [CrossRef] [Green Version]
- Yamaguchi, Y. Arginine vasopressin signaling in the suprachiasmatic nucleus on the resilience of circadian clock to jet lag. Neurosci. Res. 2018, 129, 57–61. [Google Scholar] [CrossRef]
- Shan, Y.; Abel, J.H.; Li, Y.; Izumo, M.; Cox, K.H.; Jeong, B.; Yoo, S.H.; Olson, D.P.; Doyle, F.J., 3rd; Takahashi, J.S. Dual-Color Single-Cell Imaging of the Suprachiasmatic Nucleus Reveals a Circadian Role in Network Synchrony. Neuron 2020, 108, 164–179.e7. [Google Scholar] [CrossRef]
- Liu, D.; Stowie, A.; de Zavalia, N.; Leise, T.; Pathak, S.S.; Drewes, L.R.; Davidson, A.J.; Amir, S.; Sonenberg, N.; Cao, R. mTOR signaling in VIP neurons regulates circadian clock synchrony and olfaction. Proc. Natl. Acad. Sci. USA 2018, 115, E3296–E3304. [Google Scholar] [CrossRef] [Green Version]
- Cugini, P.; Lucia, P.; Di Palma, L.; Re, M.; Leone, G.; Battisti, P.; Canova, R.; Gasbarrone, L.; Cianetti, A. Vasoactive intestinal peptide fluctuates in human blood with a circadian rhythm. Regul. Pept. 1991, 34, 141–148. [Google Scholar] [CrossRef]
- Reghunandanan, V. Vasopressin in circadian function of SCN. J. Biosci. 2020, 45, 1–11. [Google Scholar] [CrossRef]
- Harmar, A.J. An essential role for peptidergic signalling in the control of circadian rhythms in the suprachiasmatic nuclei. J. Neuroendocrinol. 2003, 15, 335–338. [Google Scholar] [CrossRef]
- Rohr, K.E.; Inda, T.; Evans, J.A. Vasopressin Resets the Central Circadian Clock in a Manner Influenced by Sex and Vasoactive Intestinal Polypeptide Signaling. Neuroendocrinology 2022, 112, 904–916. [Google Scholar] [CrossRef]
- Ono, D.; Honma, K.I.; Honma, S. Roles of Neuropeptides, VIP and AVP, in the Mammalian Central Circadian Clock. Front. Neurosci. 2021, 15, 650154. [Google Scholar] [CrossRef]
- Ingram, C.D.; Snowball, R.K.; Mihai, R. Circadian rhythm of neuronal activity in suprachiasmatic nucleus slices from the vasopressin-deficient Brattleboro rat. Neuroscience 1996, 75, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J. Sleep health: Can we define it? Does it matter? Sleep 2014, 37, 9–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leemburg, S.; Vyazovskiy, V.V.; Olcese, U.; Bassetti, C.L.; Tononi, G.; Cirelli, C. Sleep homeostasis in the rat is preserved during chronic sleep restriction. Proc. Natl. Acad. Sci. USA 2010, 107, 15939–15944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irwin, M.R. Why sleep is important for health: A psychoneuroimmunology perspective. Annu. Rev. Psychol. 2015, 66, 143–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jagannath, A.; Peirson, S.N.; Foster, R.G. Sleep and circadian rhythm disruption in neuropsychiatric illness. Curr. Opin. Neurobiol. 2013, 23, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Besedovsky, L.; Lange, T.; Haack, M. The Sleep-Immune Crosstalk in Health and Disease. Physiol. Rev. 2019, 99, 1325–1380. [Google Scholar] [CrossRef] [Green Version]
- Axelsson, J.; Rehman, J.U.; Akerstedt, T.; Ekman, R.; Miller, G.E.; Hoglund, C.O.; Lekander, M. Effects of sustained sleep restriction on mitogen-stimulated cytokines, chemokines and T helper 1/ T helper 2 balance in humans. PLoS ONE 2013, 8, e82291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, S.; Doyle, W.J.; Alper, C.M.; Janicki-Deverts, D.; Turner, R.B. Sleep habits and susceptibility to the common cold. Arch. Intern. Med. 2009, 169, 62–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kripke, D.F.; Garfinkel, L.; Wingard, D.L.; Klauber, M.R.; Marler, M.R. Mortality associated with sleep duration and insomnia. Arch. Gen. Psychiatry 2002, 59, 131–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stickgold, R. Sleep-dependent memory consolidation. Nature 2005, 437, 1272–1278. [Google Scholar] [CrossRef] [PubMed]
- Klinzing, J.G.; Niethard, N.; Born, J. Mechanisms of systems memory consolidation during sleep. Nat. Neurosci. 2019, 22, 1598–1610. [Google Scholar] [CrossRef]
- Ackermann, S.; Rasch, B. Differential effects of non-REM and REM sleep on memory consolidation? Curr. Neurol. Neurosci. Rep. 2014, 14, 430. [Google Scholar] [CrossRef]
- Hu, X.; Cheng, L.Y.; Chiu, M.H.; Paller, K.A. Promoting memory consolidation during sleep: A meta-analysis of targeted memory reactivation. Psychol Bull 2020, 146, 218–244. [Google Scholar] [CrossRef]
- Cousins, J.N.; Fernandez, G. The impact of sleep deprivation on declarative memory. Prog. Brain Res. 2019, 246, 27–53. [Google Scholar]
- Brownlow, J.A.; Miller, K.E.; Gehrman, P.R. Insomnia and Cognitive Performance. Sleep Med. Clin. 2020, 15, 71–76. [Google Scholar] [CrossRef]
- Raven, F.; Van der Zee, E.A.; Meerlo, P.; Havekes, R. The role of sleep in regulating structural plasticity and synaptic strength: Implications for memory and cognitive function. Sleep Med. Rev. 2018, 39, 3–11. [Google Scholar] [CrossRef]
- Xia, Z.; Storm, D. Role of circadian rhythm and REM sleep for memory consolidation. Neurosci. Res. 2017, 118, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Erwin, C.W.; Somerville, E.R.; Radtke, R.A. A review of electroencephalographic features of normal sleep. J. Clin. Neurophysiol. 1984, 1, 253–274. [Google Scholar] [CrossRef]
- Battaglia, F.P.; Benchenane, K.; Sirota, A.; Pennartz, C.M.; Wiener, S.I. The hippocampus: Hub of brain network communication for memory. Trends Cogn. Sci. 2011, 15, 310–318. [Google Scholar] [CrossRef]
- Girardeau, G.; Lopes-Dos-Santos, V. Brain neural patterns and the memory function of sleep. Science 2021, 374, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Zielinski, M.C.; Tang, W.; Jadhav, S.P. The role of replay and theta sequences in mediating hippocampal-prefrontal interactions for memory and cognition. Hippocampus 2020, 30, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Buzsaki, G.; Moser, E.I. Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat. Neurosci. 2013, 16, 130–138. [Google Scholar] [CrossRef] [Green Version]
- Jutras, M.J.; Buffalo, E.A. Oscillatory correlates of memory in non-human primates. Neuroimage 2014, 85 Pt 2, 694–701. [Google Scholar] [CrossRef] [Green Version]
- Bastiaansen, M.C.; van der Linden, M.; Ter Keurs, M.; Dijkstra, T.; Hagoort, P. Theta responses are involved in lexical-semantic retrieval during language processing. J. Cogn. Neurosci. 2005, 17, 530–541. [Google Scholar] [CrossRef] [Green Version]
- Cavanagh, J.F.; Frank, M.J. Frontal theta as a mechanism for cognitive control. Trends Cogn. Sci. 2014, 18, 414–421. [Google Scholar] [CrossRef] [Green Version]
- Koster, M.; Finger, H.; Graetz, S.; Kater, M.; Gruber, T. Theta-gamma coupling binds visual perceptual features in an associative memory task. Sci. Rep. 2018, 8, 17688. [Google Scholar] [CrossRef] [Green Version]
- Arnauld, E.; Bibene, V.; Meynard, J.; Rodriguez, F.; Vincent, J.D. Effects of chronic icv infusion of vasopressin on sleep-waking cycle of rats. Am. J. Physiol. 1989, 256, R674–R684. [Google Scholar] [CrossRef] [PubMed]
- Solomonova, E.; Lee, Y.E.A.; Robins, S.; King, L.; Feeley, N.; Gold, I.; Hayton, B.; Libman, E.; Nagy, C.; Turecki, G.; et al. Sleep quality is associated with vasopressin methylation in pregnant and postpartum women with a history of psychosocial stress. Psychoneuroendocrinology 2019, 107, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Haller, J.; Aliczki, M.; Pelczer, K.G.; Spitzer, K.; Balogh, Z.; Kantor, S. Effects of the fatty acid amide hydrolase inhibitor URB597 on coping behavior under challenging conditions in mice. Psychopharmacology 2014, 231, 593–601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, M.H.; Nunez, A.A. Vasopressin-deficient rats show a reduced amplitude of the circadian sleep rhythm. Physiol. Behav. 1989, 46, 759–762. [Google Scholar] [CrossRef]
- Porcu, A.; Vaughan, M.; Nilsson, A.; Arimoto, N.; Lamia, K.; Welsh, D.K. Vulnerability to helpless behavior is regulated by the circadian clock component CRYPTOCHROME in the mouse nucleus accumbens. Proc. Natl. Acad. Sci. USA 2020, 117, 13771–13782. [Google Scholar] [CrossRef]
- Ehlers, C.L.; Reed, T.K.; Wang, M.; Lebrun, C.J.; Koob, G.F. EEG effects of subcutaneous and intracerebroventricular injections of arginine vasopressin in the rat. Psychopharmacology 1985, 87, 430–433. [Google Scholar] [CrossRef]
- Li, Q.; Yang, C.; Zhang, X.; Yang, Z.; Zhang, T. Arginine vasopressin attenuates dysfunction of hippocampal theta and gamma oscillations in chronic cerebral hypoperfusion via V1a receptor. Brain Res. Bull. 2019, 153, 84–92. [Google Scholar] [CrossRef]
- Urban, I.; De Wied, D. Neuropeptides: Effects on paradoxical sleep and theta rhythm in rats. Pharmacol. Biochem. Behav. 1978, 8, 51–59. [Google Scholar] [CrossRef]
- Urban, I.J. Intraseptal administration of vasopressin and oxytocin affects hippocampal electroencephalogram in rats. Exp. Neurol. 1981, 74, 131–147. [Google Scholar] [CrossRef]
- Leung, L.W.; Msi, V.; Desborough, K.A. Hippocampal electrical activity in the diabetes insipidus (Brattleboro) rat. Exp. Neurol. 1987, 97, 672–685. [Google Scholar] [CrossRef]
- Diekelmann, S. Sleep for cognitive enhancement. Front. Syst. Neurosci. 2014, 8, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Y.; Xi, Y.; Sun, J.; Guo, F.; Xu, Y.; Fei, N.; Zhang, X.; Yang, X.; Yin, H.; Qin, W. Neural correlates of dynamic changes in working memory performance during one night of sleep deprivation. Hum. Brain Mapp. 2019, 40, 3265–3278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Dongen, H.P.; Maislin, G.; Mullington, J.M.; Dinges, D.F. The cumulative cost of additional wakefulness: Dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep 2003, 26, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Schmid, D.; Erlacher, D.; Klostermann, A.; Kredel, R.; Hossner, E.J. Sleep-dependent motor memory consolidation in healthy adults: A meta-analysis. Neurosci. Biobehav. Rev. 2020, 118, 270–281. [Google Scholar] [CrossRef]
- Landfield, P.W. Computer-determined EEG patterns associated with memory-facilitating drugs and with ECS. Brain Res. Bull. 1976, 1, 9–17. [Google Scholar] [CrossRef]
- Landfield, P.W.; McGaugh, J.L.; Tusa, R.J. Theta rhythm: A temporal correlate of memory storage processes in the rat. Science 1972, 175, 87–89. [Google Scholar] [CrossRef]
- Empson, J.A.; Clarke, P.R. Rapid eye movements and remembering. Nature 1970, 227, 287–288. [Google Scholar] [CrossRef]
- Parmeggiani, P.L.; Zanocco, G. A Study on the Bioelectrical Rhythms of Cortical and Subcortical Structures during Activated Sleep. Arch. Ital. Biol. 1963, 101, 385–412. [Google Scholar]
- Page, J.M.; Wakschlag, L.S.; Norton, E.S. Nonrapid eye movement sleep characteristics and relations with motor, memory, and cognitive ability from infancy to preadolescence. Dev. Psychobiol. 2021, 63, e22202. [Google Scholar] [CrossRef]
- van Dongen, E.V.; Takashima, A.; Barth, M.; Zapp, J.; Schad, L.R.; Paller, K.A.; Fernandez, G. Memory stabilization with targeted reactivation during human slow-wave sleep. Proc. Natl. Acad. Sci. USA 2012, 109, 10575–10580. [Google Scholar] [CrossRef] [Green Version]
- Dastgheib, M.; Kulanayagam, A.; Dringenberg, H.C. Is the role of sleep in memory consolidation overrated? Neurosci. Biobehav. Rev. 2022, 140, 104799. [Google Scholar] [CrossRef] [PubMed]
- Muraro, N.I.; Pirez, N.; Ceriani, M.F. The circadian system: Plasticity at many levels. Neuroscience 2013, 247, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Scott, V.; Brown, C.H. State-dependent plasticity in vasopressin neurones: Dehydration-induced changes in activity patterning. J. Neuroendocrinol. 2010, 22, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhao, F.; Wang, C.; Zhang, J.; Bai, Y.; Zhou, F.; Wang, Z.; Wu, M.; Yang, W.; Guo, J.; et al. AVP(4-8) Improves Cognitive Behaviors and Hippocampal Synaptic Plasticity in the APP/PS1 Mouse Model of Alzheimer’s Disease. Neurosci. Bull. 2020, 36, 254–262. [Google Scholar] [CrossRef] [PubMed]
- De Wied, D. The Influence of the Posterior and Intermediate Lobe of the Pituitary and Pituitary Peptides on the Maintenance of a Conditioned Avoidance Response in Rats. Int. J. Neuropharmacol. 1965, 4, 157–167. [Google Scholar] [CrossRef]
- Jiang, S.; Wang, Y.Q.; Tang, Y.; Lu, X.; Guo, D. Environmental Enrichment Protects Against Sepsis-Associated Encephalopathy-Induced Learning and Memory Deficits by Enhancing the Synthesis and Release of Vasopressin in the Supraoptic Nucleus. J. Inflamm. Res. 2022, 15, 363–379. [Google Scholar] [CrossRef]
- Gaffori, O.J.; De Wied, D. Time-related memory effects of vasopressin analogues in rats. Pharmacol. Biochem. Behav. 1986, 25, 1125–1129. [Google Scholar] [CrossRef]
- Finton, C.J.; Ophir, A.G. Developmental exposure to intranasal vasopressin impacts adult prairie vole spatial memory. Psychoneuroendocrinology 2022, 141, 105750. [Google Scholar] [CrossRef]
- Metzger, D.; Alescio-Lautier, B.; Bosler, O.; Devigne, C.; Soumireu-Mourat, B. Effect of changes in the intrahippocampal vasopressin on memory retrieval and relearning. Behav. Neural. Biol. 1993, 59, 29–48. [Google Scholar] [CrossRef] [PubMed]
- Lei, S.; Boyle, C.A.; Mastrud, M. PLCbeta-Mediated Depletion of PIP2 and ATP-Sensitive K(+) Channels Are Involved in Arginine Vasopressin-Induced Facilitation of Neuronal Excitability and LTP in the Dentate Gyrus. eNeuro 2022, 9. [Google Scholar] [CrossRef]
- Williams Avram, S.K.; Lee, H.J.; Fastman, J.; Cymerblit-Sabba, A.; Smith, A.; Vincent, M.; Song, J.; Granovetter, M.C.; Lee, S.H.; Cilz, N.I.; et al. NMDA Receptor in Vasopressin 1b Neurons Is Not Required for Short-Term Social Memory, Object Memory or Aggression. Front. Behav. Neurosci. 2019, 13, 218. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhu, D.; Zhang, P.; Li, W.; Qin, W.; Liu, F.; Xu, J.; Xu, Q.; Wang, J.; Ye, Z.; et al. Neural mechanisms of AVPR1A RS3-RS1 haplotypes that impact verbal learning and memory. Neuroimage 2020, 222, 117283. [Google Scholar] [CrossRef]
- Plasencia, G.; Luedicke, J.M.; Nazarloo, H.P.; Carter, C.S.; Ebner, N.C. Plasma oxytocin and vasopressin levels in young and older men and women: Functional relationships with attachment and cognition. Psychoneuroendocrinology 2019, 110, 104419. [Google Scholar] [CrossRef]
- Leng, G.; Leng, R.I.; Maclean, S. The vasopressin-memory hypothesis: A citation network analysis of a debate. Ann. N. Y. Acad. Sci. 2019, 1455, 126–140. [Google Scholar] [CrossRef]
- Kawakami, T.; Fujisawa, H.; Nakayama, S.; Yoshino, Y.; Hattori, S.; Seino, Y.; Takayanagi, T.; Miyakawa, T.; Suzuki, A.; Sugimura, Y. Vasopressin escape and memory impairment in a model of chronic syndrome of inappropriate secretion of antidiuretic hormone in mice. Endocr. J. 2021, 68, 31–43. [Google Scholar] [CrossRef]
- Morrison, T.R.; Kulkarni, P.; Cai, X.; Iriah, S.; Aggarwal, D.; Lu, S.F.; Simon, N.G.; Madularu, D.; Ferris, C.F. Treating head injury using a novel vasopressin 1a receptor antagonist. Neurosci. Lett. 2020, 714, 134565. [Google Scholar] [CrossRef]
- Kecel-Gunduz, S.; Budama-Kilinc, Y.; Cakir-Koc, R.; Zorlu, T.; Bicak, B.; Kokcu, Y.; Ozel, A.E.; Akyuz, S. In Silico design of AVP (4-5) peptide and synthesis, characterization and in vitro activity of chitosan nanoparticles. Daru 2020, 28, 139–157. [Google Scholar] [CrossRef]
- Zenina, T.A.; Gudasheva, T.A.; Bukreyev, Y.S.; Seredenin, S.B. Neuroprotective effect of dipeptide AVP(4-5)-NH2 is associated with nerve growth factor and heat shock protein HSP70. Bull. Exp. Biol. Med. 2007, 144, 543–545. [Google Scholar] [CrossRef]
- Celestian, J.F.; Carey, R.J.; Miller, M. Unimpaired maintenance of a conditioned avoidance response in the rat with diabetes insipidus. Physiol. Behav. 1975, 15, 707–711. [Google Scholar] [CrossRef]
- de Weid, D.; Bohus, B.; van Wimersma Greidanus, T.B. Memory deficit in rats with hereditary diabetes insipidus. Brain Res. 1975, 85, 152–156. [Google Scholar] [CrossRef]
- Carey, R.J.; Miller, M. Absence of learning and memory deficits in the vasopressin-deficient rat (Brattleboro strain). Behav. Brain Res. 1982, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Makara, G.B.; Mergl, Z.; Zelena, D. The role of vasopressin in hypothalamo-pituitary-adrenal axis activation during stress: An assessment of the evidence. Ann. N. Y. Acad. Sci. 2004, 1018, 151–161. [Google Scholar] [CrossRef]
- Zelena, D.; Domokos, A.; Jain, S.K.; Jankord, R.; Filaretova, L. The stimuli-specific role of vasopressin in the hypothalamus-pituitary-adrenal axis response to stress. J. Endocrinol. 2009, 202, 263–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engelmann, M.; Landgraf, R. Microdialysis administration of vasopressin into the septum improves social recognition in Brattleboro rats. Physiol. Behav. 1994, 55, 145–149. [Google Scholar] [CrossRef]
- Fodor, A.; Barsvari, B.; Aliczki, M.; Balogh, Z.; Zelena, D.; Goldberg, S.R.; Haller, J. The effects of vasopressin deficiency on aggression and impulsiveness in male and female rats. Psychoneuroendocrinology 2014, 47, 141–150. [Google Scholar] [CrossRef] [Green Version]
- Torok, B.; Csikota, P.; Fodor, A.; Balazsfi, D.; Ferenczi, S.; Demeter, K.; Toth, Z.E.; Konczol, K.; Perna, J.C.; Farkas, I.; et al. Rescue of Vasopressin Synthesis in Magnocellular Neurons of the Supraoptic Nucleus Normalises Acute Stress-Induced Adrenocorticotropin Secretion and Unmasks an Effect on Social Behaviour in Male Vasopressin-Deficient Brattleboro Rats. Int. J. Mol. Sci. 2022, 23, 1357. [Google Scholar] [CrossRef]
- Mao, M.J.; Gao, Y.Z.; Yang, J.J.; Zhou, Z.Q.; Ji, M.H. Abnormal theta oscillation aggravated by chronic stress in the CA1 may mediate the deterioration of fear memory impairment induced by lipopolysaccharide. Brain Res. Bull. 2021, 171, 172–182. [Google Scholar] [CrossRef]
- Nirogi, R.; Daripelli, S.; Benade, V.; Tirumalasetty, C.; Bhyrapuneni, G.; Jayarajan, P. Simultaneous monitoring of electroencephalographic characteristics in animals subjected to behavioral tests: A preclinical investigation. Behav. Pharmacol. 2017, 28, 661–669. [Google Scholar] [CrossRef]
- Kinley, B.L.; Kyne, R.F.; Lawton-Stone, T.S.; Walker, D.M.; Paul, M.J. Long-term consequences of peri-adolescent social isolation on social preference, anxiety-like behaviour, and vasopressin neural circuitry of male and female rats. Eur. J. Neurosci. 2021, 54, 7790–7804. [Google Scholar] [CrossRef]
- De Vries, G.J.; Best, W.; Sluiter, A.A. The influence of androgens on the development of a sex difference in the vasopressinergic innervation of the rat lateral septum. Brain Res. 1983, 284, 377–380. [Google Scholar] [CrossRef]
- de Vries, G.J.; Buijs, R.M.; Swaab, D.F. Ontogeny of the vasopressinergic neurons of the suprachiasmatic nucleus and their extrahypothalamic projections in the rat brain—presence of a sex difference in the lateral septum. Brain Res. 1981, 218, 67–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murray, E.K.; Varnum, M.M.; Fernandez, J.L.; de Vries, G.J.; Forger, N.G. Effects of neonatal treatment with valproic acid on vasopressin immunoreactivity and olfactory behaviour in mice. J. Neuroendocrinol. 2011, 23, 906–914. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lonstein, J.S.; De Vries, G.J. Sex differences in the parental behaviour of adult virgin prairie voles: Independence from gonadal hormones and vasopressin. J. Neuroendocrinol. 1999, 11, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, D.P. Sexual dimorphism in the central nervous system of marsupials. Int. Rev. Cytol. 2002, 214, 193–224. [Google Scholar]
- Silver, R.; LeSauter, J. Efferent signals of the suprachiasmatic nucleus. J. Biol. Rhythms 1993, 8, S89–S92. [Google Scholar] [PubMed]
- Bartness, T.J.; Song, C.K.; Demas, G.E. SCN efferents to peripheral tissues: Implications for biological rhythms. J. Biol. Rhythms 2001, 16, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Geovanini, G.R.; Vasques, E.R.; de Oliveira Alvim, R.; Mill, J.G.; Andreao, R.V.; Vasques, B.K.; Pereira, A.C.; Krieger, J.E. Age and Sex Differences in Heart Rate Variability and Vagal Specific Patterns—Baependi Heart Study. Glob. Heart 2020, 15, 71. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Török, B.; Varga, J.; Zelena, D. Vasopressin as a Possible Link between Sleep-Disturbances and Memory Problems. Int. J. Mol. Sci. 2022, 23, 15467. https://doi.org/10.3390/ijms232415467
Török B, Varga J, Zelena D. Vasopressin as a Possible Link between Sleep-Disturbances and Memory Problems. International Journal of Molecular Sciences. 2022; 23(24):15467. https://doi.org/10.3390/ijms232415467
Chicago/Turabian StyleTörök, Bibiána, János Varga, and Dóra Zelena. 2022. "Vasopressin as a Possible Link between Sleep-Disturbances and Memory Problems" International Journal of Molecular Sciences 23, no. 24: 15467. https://doi.org/10.3390/ijms232415467





