Extracellular Vesicles as Mediators of Nickel-Induced Cancer Progression
Abstract
:1. Introduction
2. Results
2.1. Ni Exposure Alters EV Signature
2.2. EVs from Ni-Treated and Ni-Transformed Cells Increase Uptake in Target Epithelial Cells
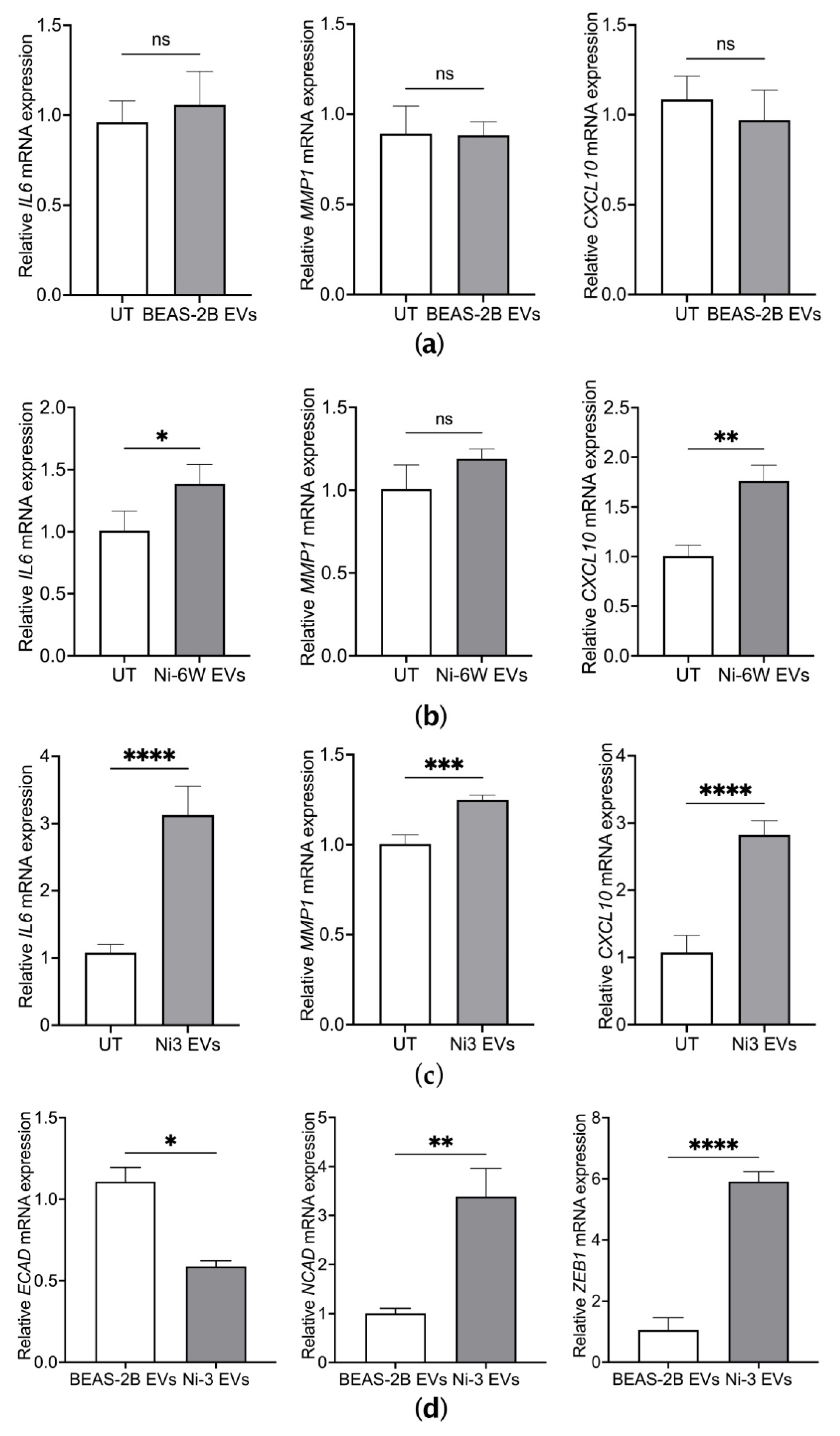
2.3. EVs from Ni-Transformed Cells Induce Inflammation Response in Target Epithelial Cells
2.4. Ni-Exposure Altered EVs from Epithelial Cells Increase Incorporation in Target Endothelial Cells
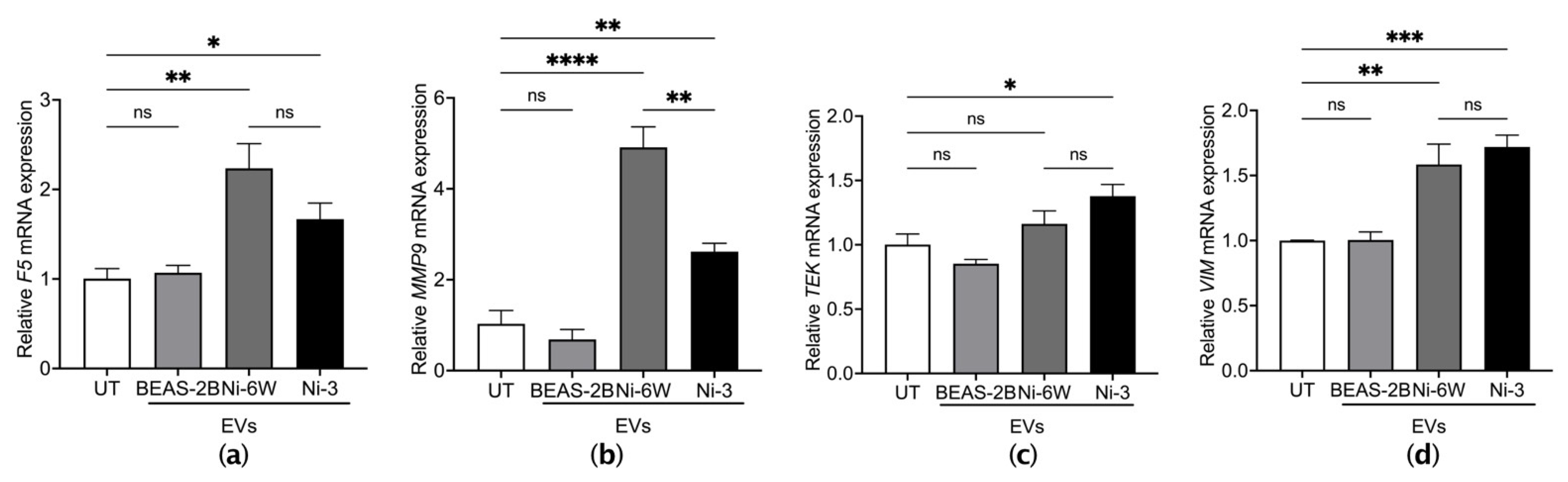
2.5. EVs from Ni-Exposed Epithelial Cells Regulate Inflammation and Coagulation in Endothelial Cells
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Ni Treatments
4.2. Isolation of Extracellular Vesicles
4.3. Characterization of Extracellular Vesicles
4.4. Extracellular Vesicles Incorporation Assays
4.5. Western Blot for EV Markers
4.6. RNA Isolation and qPCR
4.7. Graphical Depictions and Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Grimsrud, T.K.; Berge, S.R.; Haldorsen, T.; Andersen, A. Exposure to Different Forms of Nickel and Risk of Lung Cancer. Am. J. Epidemiol. 2002, 156, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Das, K.K.; Reddy, R.C.; Bagoji, I.B.; Das, S.; Bagali, S.; Mullur, L.; Khodnapur, J.P.; Biradar, M.S. Primary Concept of Nickel Toxicity—An Overview. J. Basic Clin. Physiol. Pharmacol. 2019, 30, 141–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Q.Y.; DesMarais, T.; Costa, M. Metals and Mechanisms of Carcinogenesis. Annu. Rev. Pharm. Toxicol. 2019, 59, 537–554. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, D.A.; Rodríguez-Sierra, C.J.; Jiménez-Velez, B.D. Concentrations of Ni and V, Other Heavy Metals, Arsenic, Elemental and Organic Carbon in Atmospheric Fine Particles (PM2.5) from Puerto Rico. Toxicol. Ind. Health 2006, 22, 87–99. [Google Scholar] [CrossRef] [Green Version]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Arsenic, Metals, Fibres, and Dusts. IARC Monogr. Eval. Carcinog. Risks Hum. 2012, 100, 11–465. [Google Scholar]
- Becker, A.; Thakur, B.K.; Weiss, J.M.; Kim, H.S.; Peinado, H.; Lyden, D. Extracellular Vesicles in Cancer: Cell-to-Cell Mediators of Metastasis. Cancer Cell 2016, 30, 836–848. [Google Scholar] [CrossRef] [Green Version]
- Jabalee, J.; Towle, R.; Garnis, C. The Role of Extracellular Vesicles in Cancer: Cargo, Function, and Therapeutic Implications. Cells 2018, 7, 93. [Google Scholar] [CrossRef] [Green Version]
- Han, L.; Lam, E.W.-F.; Sun, Y. Extracellular Vesicles in the Tumor Microenvironment: Old Stories, but New Tales. Mol. Cancer 2019, 18, 59. [Google Scholar] [CrossRef] [Green Version]
- Yáñez-Mó, M.; Siljander, P.R.-M.; Andreu, Z.; Bedina Zavec, A.; Borràs, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological Properties of Extracellular Vesicles and Their Physiological Functions. J. Extracell. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef] [Green Version]
- Jiao, Y.; Tang, Y.; Li, Y.; Liu, C.; He, J.; Zhang, L.-K.; Guan, Y.-Q. Tumor Cell-Derived Extracellular Vesicles for Breast Cancer Specific Delivery of Therapeutic P53. J. Control. Release 2022, 349, 606–616. [Google Scholar] [CrossRef]
- Stavrou, A.; Ortiz, A. Extracellular Vesicles: A Novel Tool in Nanomedicine and Cancer Treatment. Cancers 2022, 14, 4450. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Costa, M.; Ortiz, A. Chapter 12—Effects of Metals on Extracellular Vesicle Signaling. In Handbook on the Toxicology of Metals, 5th ed.; Nordberg, G.F., Costa, M., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 279–298. ISBN 978-0-12−823292−7. [Google Scholar]
- Gao, Y.; Qin, Y.; Wan, C.; Sun, Y.; Meng, J.; Huang, J.; Hu, Y.; Jin, H.; Yang, K. Small Extracellular Vesicles: A Novel Avenue for Cancer Management. Front. Oncol. 2021, 11, 638357. [Google Scholar] [CrossRef] [PubMed]
- Sódar, B.W.; Kittel, Á.; Pálóczi, K.; Vukman, K.V.; Osteikoetxea, X.; Szabó-Taylor, K.; Németh, A.; Sperlágh, B.; Baranyai, T.; Giricz, Z.; et al. Low-Density Lipoprotein Mimics Blood Plasma-Derived Exosomes and Microvesicles during Isolation and Detection. Sci. Rep. 2016, 6, 24316. [Google Scholar] [CrossRef] [Green Version]
- Ortiz, A.; Gui, J.; Zahedi, F.; Yu, P.; Cho, C.; Bhattacharya, S.; Carbone, C.J.; Yu, Q.; Katlinski, K.V.; Katlinskaya, Y.V.; et al. An Interferon-Driven Oxysterol-Based Defense against Tumor-Derived Extracellular Vesicles. Cancer Cell 2019, 35, 33–45.e6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clancy, H.A.; Sun, H.; Passantino, L.; Kluz, T.; Muñoz, A.; Zavadil, J.; Costa, M. Gene Expression Changes in Human Lung Cells Exposed to Arsenic, Chromium, Nickel or Vanadium Indicate the First Steps in Cancer. Metallomics 2012, 4, 784–793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal Information for Studies of Extracellular Vesicles 2018 (MISEV2018): A Position Statement of the International Society for Extracellular Vesicles and Update of the MISEV2014 Guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Zhang, H.; Gu, J.; Zhang, J.; Shi, H.; Qian, H.; Wang, D.; Xu, W.; Pan, J.; Santos, H.A. Engineered Extracellular Vesicles for Cancer Therapy. Adv. Mater. 2021, 33, 2005709. [Google Scholar] [CrossRef]
- Yang, D.; Guo, P.; He, T.; Powell, C.A. Role of Endothelial Cells in Tumor Microenvironment. Clin. Transl. Med. 2021, 11, e450. [Google Scholar] [CrossRef]
- Chennakrishnaiah, S.; Tsering, T.; Gregory, C.; Tawil, N.; Spinelli, C.; Montermini, L.; Karatzas, N.; Aprikian, S.; Choi, D.; Klewes, L.; et al. Extracellular Vesicles from Genetically Unstable, Oncogene-Driven Cancer Cells Trigger Micronuclei Formation in Endothelial Cells. Sci. Rep. 2020, 10, 8532. [Google Scholar] [CrossRef]
- Ko, S.Y.; Lee, W.; Kenny, H.A.; Dang, L.H.; Ellis, L.M.; Jonasch, E.; Lengyel, E.; Naora, H. Cancer-Derived Small Extracellular Vesicles Promote Angiogenesis by Heparin-Bound, Bevacizumab-Insensitive VEGF, Independent of Vesicle Uptake. Commun. Biol. 2019, 2, 386. [Google Scholar] [CrossRef] [Green Version]
- Jankovičová, J.; Sečová, P.; Michalková, K.; Antalíková, J. Tetraspanins, More than Markers of Extracellular Vesicles in Reproduction. Int. J. Mol. Sci. 2020, 21, 7568. [Google Scholar] [CrossRef] [PubMed]
- Phuyal, S.; Hessvik, N.P.; Skotland, T.; Sandvig, K.; Llorente, A. Regulation of Exosome Release by Glycosphingolipids and Flotillins. FEBS J. 2014, 281, 2214–2227. [Google Scholar] [CrossRef] [PubMed]
- Bebelman, M.P.; Janssen, E.; Pegtel, D.M.; Crudden, C. The Forces Driving Cancer Extracellular Vesicle Secretion. Neoplasia 2021, 23, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Logozzi, M.; Milito, A.D.; Lugini, L.; Borghi, M.; Calabrò, L.; Spada, M.; Perdicchio, M.; Marino, M.L.; Federici, C.; Iessi, E.; et al. High Levels of Exosomes Expressing CD63 and Caveolin-1 in Plasma of Melanoma Patients. PLoS ONE 2009, 4, e5219. [Google Scholar] [CrossRef] [Green Version]
- Riches, A.; Campbell, E.; Borger, E.; Powis, S. Regulation of Exosome Release from Mammary Epithelial and Breast Cancer Cells—A New Regulatory Pathway. Eur. J. Cancer 2014, 50, 1025–1034. [Google Scholar] [CrossRef]
- Lowry, M.C.; O’Driscoll, L. Can Hi-Jacking Hypoxia Inhibit Extracellular Vesicles in Cancer? Drug Discov. Today 2018, 23, 1267–1273. [Google Scholar] [CrossRef]
- Kanemoto, S.; Nitani, R.; Murakami, T.; Kaneko, M.; Asada, R.; Matsuhisa, K.; Saito, A.; Imaizumi, K. Multivesicular Body Formation Enhancement and Exosome Release during Endoplasmic Reticulum Stress. Biochem. Biophys. Res. Commun. 2016, 480, 166–172. [Google Scholar] [CrossRef]
- Savina, A.; Furlán, M.; Vidal, M.; Colombo, M.I. Exosome Release Is Regulated by a Calcium-Dependent Mechanism in K562 Cells. J. Biol. Chem. 2003, 278, 20083–20090. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharya, S.; Pal, K.; Sharma, A.K.; Dutta, S.K.; Lau, J.S.; Yan, I.K.; Wang, E.; Elkhanany, A.; Alkharfy, K.M.; Sanyal, A.; et al. GAIP Interacting Protein C-Terminus Regulates Autophagy and Exosome Biogenesis of Pancreatic Cancer through Metabolic Pathways. PLoS ONE 2014, 9, e114409. [Google Scholar] [CrossRef]
- Hedlund, M.; Nagaeva, O.; Kargl, D.; Baranov, V.; Mincheva-Nilsson, L. Thermal- and Oxidative Stress Causes Enhanced Release of NKG2D Ligand-Bearing Immunosuppressive Exosomes in Leukemia/Lymphoma T and B Cells. PLoS ONE 2011, 6, e16899. [Google Scholar] [CrossRef]
- Costa, M.; Davidson, T.L.; Chen, H.; Ke, Q.; Zhang, P.; Yan, Y.; Huang, C.; Kluz, T. Nickel Carcinogenesis: Epigenetics and Hypoxia Signaling. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2005, 592, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Di Bucchianico, S.; Gliga, A.R.; Åkerlund, E.; Skoglund, S.; Wallinder, I.O.; Fadeel, B.; Karlsson, H.L. Calcium-Dependent Cyto- and Genotoxicity of Nickel Metal and Nickel Oxide Nanoparticles in Human Lung Cells. Part. Fibre Toxicol. 2018, 15, 32. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Cui, H.; Peng, X.; Fang, J.; Zuo, Z.; Deng, J.; Wang, X.; Wu, B.; Chen, K.; Deng, J. Nickel Chloride (NiCl2) Induces Endoplasmic Reticulum (ER) Stress by Activating UPR Pathways in the Kidney of Broiler Chickens. Oncotarget 2016, 7, 17508–17519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mulcahy, L.A.; Pink, R.C.; Carter, D.R.F. Routes and Mechanisms of Extracellular Vesicle Uptake. J. Extracell. Vesicles 2014, 3, 24641. [Google Scholar] [CrossRef] [Green Version]
- Bielska, E.; Sisquella, M.A.; Aldeieg, M.; Birch, C.; O’Donoghue, E.J.; May, R.C. Pathogen-Derived Extracellular Vesicles Mediate Virulence in the Fatal Human Pathogen Cryptococcus Gattii. Nat. Commun. 2018, 9, 1556. [Google Scholar] [CrossRef] [Green Version]
- Rezaei, M.; Martins Cavaco, A.C.; Stehling, M.; Nottebaum, A.; Brockhaus, K.; Caliandro, M.F.; Schelhaas, S.; Schmalbein, F.; Vestweber, D.; Eble, J.A. Extracellular Vesicle Transfer from Endothelial Cells Drives VE-Cadherin Expression in Breast Cancer Cells, Thereby Causing Heterotypic Cell Contacts. Cancers 2020, 12, 2138. [Google Scholar] [CrossRef]
- Guo, H.; Liu, H.; Jian, Z.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L.; et al. Nickel Induces Inflammatory Activation via NF-ΚB, MAPKs, IRF3 and NLRP3 Inflammasome Signaling Pathways in Macrophages. Aging 2019, 11, 11659–11672. [Google Scholar] [CrossRef]
- Goebeler, M.; Roth, J.; Bröcker, E.B.; Sorg, C.; Schulze-Osthoff, K. Activation of Nuclear Factor-Kappa B and Gene Expression in Human Endothelial Cells by the Common Haptens Nickel and Cobalt. J. Immunol. 1995, 155, 2459–2467. [Google Scholar]
- Capasso, L.; Camatini, M.; Gualtieri, M. Nickel Oxide Nanoparticles Induce Inflammation and Genotoxic Effect in Lung Epithelial Cells. Toxicol. Lett. 2014, 226, 28–34. [Google Scholar] [CrossRef]
- Huang, H.; Zhu, J.; Li, Y.; Zhang, L.; Gu, J.; Xie, Q.; Jin, H.; Che, X.; Li, J.; Huang, C.; et al. Upregulation of SQSTM1/P62 Contributes to Nickel-Induced Malignant Transformation of Human Bronchial Epithelial Cells. Autophagy 2016, 12, 1687–1703. [Google Scholar] [CrossRef] [Green Version]
- Azmi, A.S. Unveiling the Role of Nuclear Transport in Epithelial-to-Mesenchymal Transition. Curr. Cancer Drug Targets 2013, 13, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Pastushenko, I.; Blanpain, C. EMT Transition States during Tumor Progression and Metastasis. Trends Cell Biol. 2019, 29, 212–226. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, S.; Shin, E.; Seong, K.M.; Jin, Y.W.; Youn, H.; Youn, B. The Emerging Roles of Exosomes as EMT Regulators in Cancer. Cells 2020, 9, 861. [Google Scholar] [CrossRef] [PubMed]
- Baghban, R.; Roshangar, L.; Jahanban-Esfahlan, R.; Seidi, K.; Ebrahimi-Kalan, A.; Jaymand, M.; Kolahian, S.; Javaheri, T.; Zare, P. Tumor Microenvironment Complexity and Therapeutic Implications at a Glance. Cell Commun. Signal. 2020, 18, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, D.X.; Vu, L.T.; Ismail, N.N.; Le, M.T.N.; Grimson, A. Landscape of Extracellular Vesicles in the Tumour Microenvironment: Interactions with Stromal Cells and with Non-Cell Components, and Impacts on Metabolic Reprogramming, Horizontal Transfer of Neoplastic Traits, and the Emergence of Therapeutic Resistance. Semin. Cancer Biol. 2021, 74, 24–44. [Google Scholar] [CrossRef]
- Sierko, E.; Wojtukiewicz, M.Z. Inhibition of Platelet Function: Does It Offer a Chance of Better Cancer Progression Control? Semin. Thromb. Hemost. 2007, 33, 712–721. [Google Scholar] [CrossRef]
- Liu, Y.; Liao, X.-W.; Qin, Y.-Z.; Mo, X.-W.; Luo, S.-S. Identification of F5 as a Prognostic Biomarker in Patients with Gastric Cancer. Biomed. Res. Int. 2020, 2020, 9280841. [Google Scholar] [CrossRef] [Green Version]
- Tinholt, M.; Stavik, B.; Tekpli, X.; Garred, Ø.; Borgen, E.; Kristensen, V.; Sahlberg, K.K.; Sandset, P.M.; Iversen, N. Coagulation Factor V Is a Marker of Tumor-Infiltrating Immune Cells in Breast Cancer. OncoImmunology 2020, 9, 1824644. [Google Scholar] [CrossRef]
- Tinholt, M.; Sandset, P.M.; Iversen, N. Polymorphisms of the Coagulation System and Risk of Cancer. Thromb. Res. 2016, 140 (Suppl. 1), S49–S54. [Google Scholar] [CrossRef]
- Schubert, S.Y.; Benarroch, A.; Monter-Solans, J.; Edelman, E.R. Primary Monocytes Regulate Endothelial Cell Survival Through Secretion of Angiopoietin-1 and Activation of Endothelial Tie2. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 870–875. [Google Scholar] [CrossRef] [Green Version]
- Dave, J.M.; Bayless, K.J. Vimentin as an Integral Regulator of Cell Adhesion and Endothelial Sprouting. Microcirculation 2014, 21, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.-F.; Ren, K.; Zha, J.-H.; Guo, Q.; Zhao, G.-J. Vimentin Promotes Endothelial Inflammation by Activating NLRP3. Int. J. Cardiol. 2020, 301, 155. [Google Scholar] [CrossRef]
- Quintero-Fabián, S.; Arreola, R.; Becerril-Villanueva, E.; Torres-Romero, J.C.; Arana-Argáez, V.; Lara-Riegos, J.; Ramírez-Camacho, M.A.; Alvarez-Sánchez, M.E. Role of Matrix Metalloproteinases in Angiogenesis and Cancer. Front. Oncol. 2019, 9, 1370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chandrasekar, B.; Mummidi, S.; Mahimainathan, L.; Patel, D.N.; Bailey, S.R.; Imam, S.Z.; Greene, W.C.; Valente, A.J. Interleukin-18-Induced Human Coronary Artery Smooth Muscle Cell Migration Is Dependent on NF-ΚB- and AP-1-Mediated Matrix Metalloproteinase-9 Expression and Is Inhibited by Atorvastatin. J. Biol. Chem. 2006, 281, 15099–15109. [Google Scholar] [CrossRef] [Green Version]
- Magid, R.; Murphy, T.J.; Galis, Z.S. Expression of Matrix Metalloproteinase-9 in Endothelial Cells Is Differentially Regulated by Shear Stress: ROLE OF c-Myc. J. Biol. Chem. 2003, 278, 32994–32999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jose, C.C.; Jagannathan, L.; Tanwar, V.S.; Zhang, X.; Zang, C.; Cuddapah, S. Nickel Exposure Induces Persistent Mesenchymal Phenotype in Human Lung Epithelial Cells through Epigenetic Activation of ZEB1. Mol. Carcinog. 2018, 57, 794–806. [Google Scholar] [CrossRef]
- Théry, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and Characterization of Exosomes from Cell Culture Supernatants and Biological Fluids. Curr. Protoc. Cell Biol. 2006, 30, 3–22. [Google Scholar] [CrossRef]



| Gene | Forward Primer (5′ ->3′) | Reverse Primer (5′ ->3′) |
|---|---|---|
| CD63 | ATG ATC ACG TTT GCC ATC TT | AGG GAT TTT CTC CCA ATC TG |
| CD82 | AAAGCAGAACCCGCAGAGTC | AAGACATAGGCCCCCATCCT |
| CXCL10 | TTCCTGCAAGCCAATTTTGTC | TCTTCTCACCCTTCTTTTTCATTGT |
| FLOT | CTCTCATCTCTGCGGTCAGT | TCAACCTCGGCTACTTCTTG |
| GAPDH | AGGGCTGCTTTTAACTCTGGT | CCCCACTTGATTTTGGAGGGA |
| IL6 | GGTACATCCTCGACGGCATCT | GTGCCTCTTTGCTGCTTTCAC |
| MMP1 | GAGATCATCGGGACAACTCTCCTT | GTTGGTCCACCTTTCATCTTCATCA |
| MMP9 | ATCCAGTTTGGTGTCGCGGAGC | GAAGGGGAAGACGCACAGCT |
| TEK | CTATCGGACTCCCTCCTCCAA | TCAAATTTAGAGCTGTCTGGCTTTT |
| F5 | CCAGGTAGCTGGCATGCAA | CCATTGGCATCTTACACTCTTTGT |
| VIM | GACGCCATCAACACCGAGTT | CTTTGTCGTTGGTTAGCTGGT |
| ZEB1 | AGCAGTGAAAGAGAAGGGAATGC | GGTCCTCTTCAGGTGCCTCAG |
| E-CAD | TGCCCAGAAAATGAAAAAGG | GTGTATGTGGCAATGCGTTC |
| N-CAD | TGGGAATCCGACGAATGG | TGCAGATCGGACCGGATACT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, S.; Ortiz, A.; Stavrou, A.; Talusan, A.R.; Costa, M. Extracellular Vesicles as Mediators of Nickel-Induced Cancer Progression. Int. J. Mol. Sci. 2022, 23, 16111. https://doi.org/10.3390/ijms232416111
Liu S, Ortiz A, Stavrou A, Talusan AR, Costa M. Extracellular Vesicles as Mediators of Nickel-Induced Cancer Progression. International Journal of Molecular Sciences. 2022; 23(24):16111. https://doi.org/10.3390/ijms232416111
Chicago/Turabian StyleLiu, Shan, Angelica Ortiz, Aikaterini Stavrou, Angela R. Talusan, and Max Costa. 2022. "Extracellular Vesicles as Mediators of Nickel-Induced Cancer Progression" International Journal of Molecular Sciences 23, no. 24: 16111. https://doi.org/10.3390/ijms232416111





