Int. J. Mol. Sci. 2023, 24(6), 5385; https://doi.org/10.3390/ijms24065385 - 11 Mar 2023
Cited by 2 | Viewed by 1616
Abstract
Crayfish axons contain a system of parallel membranous cisternae spaced by ~2 μm and oriented perpendicularly to the axon’s long axis. Each cisterna is composed of two roughly parallel membranes, separated by a 150–400 Å wide space. The cisternae are interrupted by 500–600
[...] Read more.
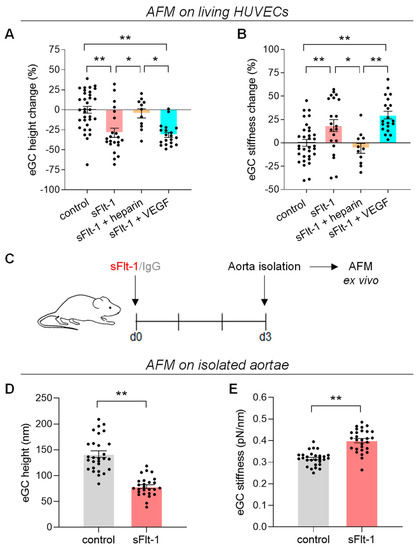
Crayfish axons contain a system of parallel membranous cisternae spaced by ~2 μm and oriented perpendicularly to the axon’s long axis. Each cisterna is composed of two roughly parallel membranes, separated by a 150–400 Å wide space. The cisternae are interrupted by 500–600 Å pores, each occupied by a microtubule. Significantly, filaments, likely made of kinesin, often bridge the gap between the microtubule and the edge of the pore. Neighboring cisternae are linked by longitudinal membranous tubules. In small axons, the cisternae seem to be continuous across the axon, while in large axons they are intact only at the axon’s periphery. Due to the presence of pores, we have named these structures “Fenestrated Septa” (FS). Similar structures are also present in vertebrates, including mammals, proving that they are widely expressed in the animal kingdom. We propose that FS are components of the “anterograde transport” mechanism that moves cisternae of the Golgi apparatus (GA) toward the nerve ending by means of motor proteins, likely to be kinesins. In crayfish lateral giant axons, we believe that vesicles that bud off FS at the nerve ending contain gap junction hemichannels (innexons) for gap junction channel and hemichannel formation and function.
Full article
(This article belongs to the Special Issue Connexin Hemichannels: Structure, Function, and Dysfunction)
►
Show Figures