Abstract
The WRKY transcription factor family plays important regulatory roles in multiple biological processes in higher plants. They have been identified and functionally characterized in a number of plant species, but very little is known in Neolamarckia cadamba, a ‘miracle tree’ for its fast growth and potential medicinal resource in Southeast Asia. In this study, a total of 85 WRKY genes were identified in the genome of N. cadamba. They were divided into three groups according to their phylogenetic features, with the support of the characteristics of gene structures and conserved motifs of protein. The NcWRKY genes were unevenly distributed on 22 chromosomes, and there were two pairs of segmentally duplicated events. In addition, a number of putative cis-elements were identified in the promoter regions, of which hormone- and stress-related elements were shared in many NcWRKYs. The transcript levels of NcWRKY were analyzed using the RNA-seq data, revealing distinct expression patterns in various tissues and at different stages of vascular development. Furthermore, 16 and 12 NcWRKY genes were confirmed to respond to various hormone treatments and two different abiotic stress treatments, respectively. Moreover, the content of cadambine, the active metabolite used for the various pharmacological activities found in N. cadamba, significantly increased after Methyl jasmonate treatment. In addition, expression of NcWRKY64/74 was obviously upregulated, suggesting that they may have a potential function of regulating the biosynthesis of cadambine in response to MeJA. Taken together, this study provides clues into the regulatory roles of the WRKY gene family in N. cadamba.
1. Introduction
The WRKY proteins are one of the largest families of transcriptional regulators found throughout plants [1]. They share the defining feature WRKY domain which comprises the highly conserved WRKYGQK hepta peptide sequence at the N-terminal followed by a C2H2- or C2HC type of zinc-finger motif at the C-terminal. Both the WRKY domain and zinc-finger motif are required for the high binding affinity of WRKY transcription factor (TF) to the W-box cis-elements in the promoter regions of their target genes [2]. The WRKY proteins can be classified into three main groups (I–III) based on the number of WRKY domains and the category of zinc-finger motifs. Proteins with two WRKY domains belong to Group I, while Group II or III contain one WRKY domain with different zinc-finger motifs. Specifically, Group III has a zinc-finger structure of C2HC while Group II has a zinc-finger structure of C2H2 and can be further divided into five subgroups: IIa, IIb, IIc, IId, and IIe [3].
Many WRKY TFs have been experimentally identified from various plant species. They play vital regulatory roles in plant defense regulatory networks, including response to various abiotic stresses, which result from an interplay between WRKYs and a variety of plant hormones [4,5]. For instance, the synergistic interaction between OsWRKY51 and OsWRKY71 genes inhibited gibberellic acid (GA) signaling in the aleurone cells of rice seeds under ABA induction in rice [6,7]. Likewise, in Arabidopsis, AtWRKY18/40/60 were shown to participate in signaling pathways that are mediated by ABA, and AtWRKY60 might be a direct target gene of AtWRKY18 and AtWRKY40 in ABA signaling [8]. Moreover, PoWRKY13 in Populus tomentosa [9], PheWRKY86 in Moso bamboo [10] and AtWRKY25/26/39 [11,12] were involved in the response to heat stress and drought stress. DgWRKY5 in Chrysanthemum indicum [13], GbWRKY1 in cotton [14], and SlWRKY8 in Solanum lycopersicum [15] participated in regulating plant tolerance to salt stress.
WRKY TF also plays an activating or repressing role in the transcriptional regulation of key enzyme genes in plant secondary metabolites synthesis [4]. OpWRKY2 acted as a direct positive regulator of camptothecin biosynthesis by binding the central camptothecin pathway gene OpTDC [16]. Similarly, the total production of camptothecin was significantly upregulated in most overexpression lines of OpWRKY3 [17]. Overexpression of OpWRKY6 significantly reduced the accumulation of camptothecin. Conversely, camptothecin accumulation increased in OpWRKY6 knockout lines [18]. Agarwood sesquiterpene synthase 1 (ASS1) is one of the key enzymes responsible for the biosynthesis of sesquiterpenes, and AsWRKY44 directly binds to its promoter and represses ASS1 promoter activity [19]. Likewise, GaWRKY1 and AaWRKY1 activated the expression of key enzyme genes in the gossypol and artemisinin biosynthesis pathway, respectively, by binding to the W-box element in their promoter [20,21]; PgWRKY4X in ginseng interacted with the W-box in the squalene epoxidase (PgSE) promoter and overexpression of PgWRKY4X significantly upregulated PgSE and increased the accumulation of ginsenoside [22]. Paclitaxel, as a kind of terpenoid alkaloid, also has important clinical value in the treatment of cancer. Overexpression of TcWRKY8 and TcWRKY47 significantly increased the expression levels of genes related to paclitaxel biosynthesis [23]. Extracted from Catharanthus roseus, vinblastine is a monoterpenoid indole alkaloid (MIA) whose synthetic pathway has been fully resolved and is a natural antitumor drug widely used in cancer therapy. Overexpression of CrWRKY1 in C. roseus hairy root activated some key genes in the MIA pathway and the transcriptional repressors (ZCT1, ZCT2, and ZCT3). Interestingly, CrWRKY1 overexpression repressed the transcriptional activators ORCA2, ORCA3, and CrMYC2, yielding a higher level of serpentine accumulation [24]. To date, however, the regulation of WRKY in MIA synthesis is less understood.
Neolamarckia cadamba, from the Rubiaceae family, is widely distributed in tropical and subtropical regions of the world and is an important tree for the timber industry and traditional medicinal plants in southern China [25]. Cadambine, a kind of MIA only isolated from the Rubiaceae family, is the main component of total alkaloids in N. cadamba, accounting for about 50% of total alkalis, which has been shown to have clinical effects against malaria parasites and in the treatment of diabetes, and exhibits concentration-dependent inhibition on carcinoma cell and DNA topoisomerase [26,27,28]. With the completion of whole genome sequencing of N. cadamba [29], it enabled genome-wide identification and functional analysis of the gene families related to the development process, response to environmental change, and cadambine synthesis. Several reports suggested that WRKY participated in the regulation of MIA biosynthesis, but whether they were involved in cadambine biosynthesis was unknown. In this study, we identified 85 WRKY genes of N. cadamba at the genome-wide level and analyzed the gene structures, conserved domains, phylogenetic relationships and cis-element in the promoter of the WRKY genes. In addition, we performed a comprehensive analysis of spatiotemporal expression patterns of NcWRKY genes according to RNA-seq data and examined their expression profiles in response to hormone (MeJA, ABA, and GA) and abiotic stresses (high salinity and drought) by real time quantitative PCR (RT-qPCR). Moreover, the involvement of the NcWRKY in cadambine synthesis was further explored. Our genome-wide results identified all WRKY genes in the N. cadamba genome and provided valuable clues for further functional study.
2. Results
2.1. Identification of WRKY Genes in Neolamarckia Cadamba
In this study, a total of 85 WRKY gene sequences were identified in N. cadamba (Supplementary File S1). They were named based on the apparently complete WRKY domains and their position on the chromosome. Gene characteristics, including the length of the protein sequence, the protein molecular weight (MW), the isoelectric point (pI), and the subcellular localization, were analyzed using ExPaSy and Plant-mPLoc. The results showed that the length of all the identified WRKY proteins in N. cadamba ranged from 174 to 734 amino acids (aa), in which NcWRKY48 was the smallest protein and NcWRKY47 was the largest one. The molecular weight (MW) of the NcWRKY proteins ranged from 20.0 to 79.3 kDa, with the predicted isoelectric point values (pI) varying from 5.04 (NcWRKY6) to 9.69 (NcWRKY61). Subcellular localization prediction indicated that all the NcWRKYs were localized to nuclear, indicating that they may function as transcription factors.
2.2. Phylogenetic Analysis and Multiple Sequence Alignment of NcWRKYs
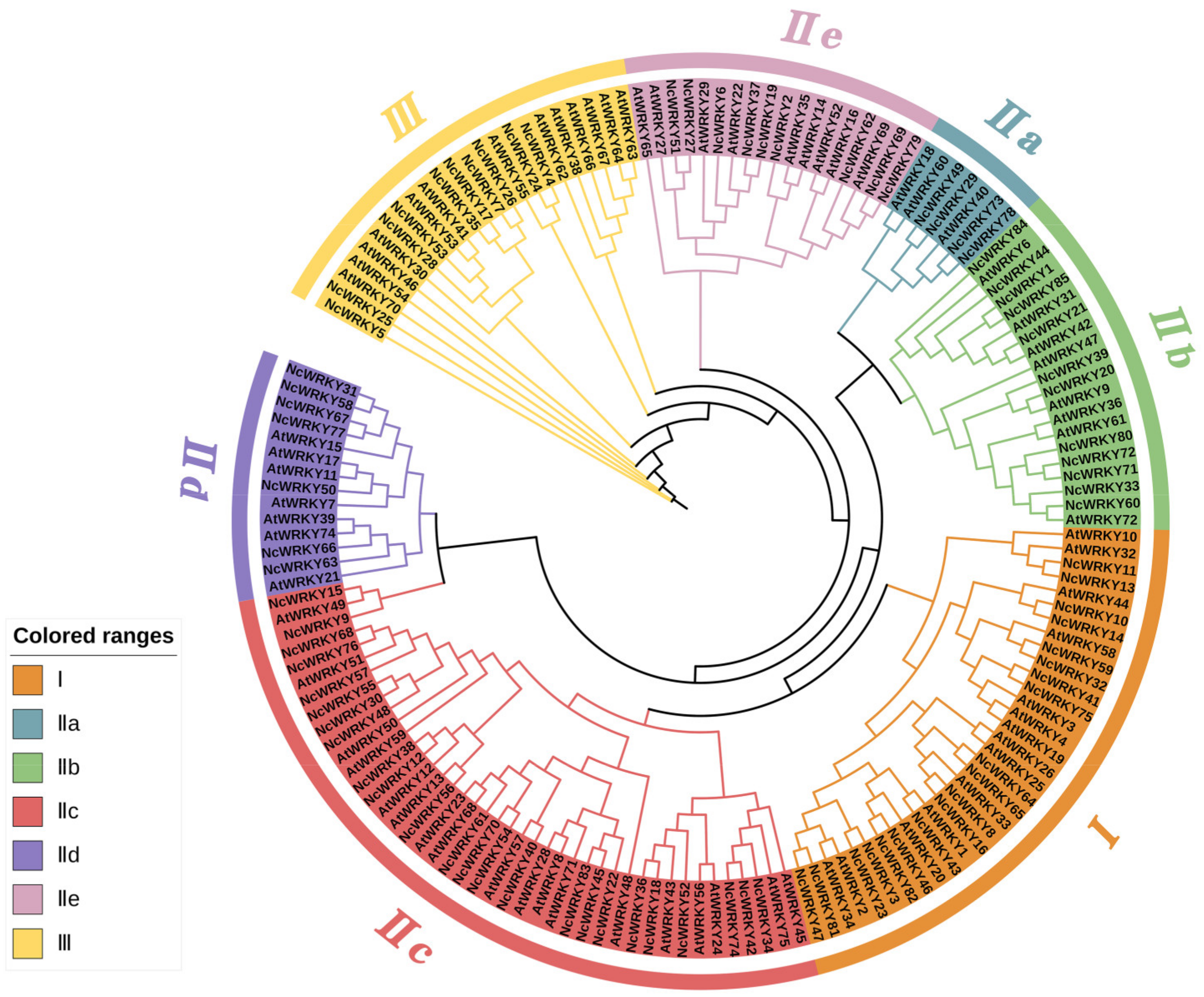
To investigate the phylogenetic relationships among WRKY family genes, we constructed an ML phylogenetic tree using the WRKY proteins from A. thaliana and N. cadamba. The result showed that the 85 NcWRKY proteins were classified into three groups (Figure 1), based on the classification described by a previous report on A. thaliana [3], indicating an evolutionary conservation between these two species. Group I contained 19 NcWRKY members, while 10 NcWRKYs belonged to Group Ⅲ. The largest group (Group II) consists of 56 NcWRKY proteins, which could be further divided into five subgroups, with 4, 12, 24, 7, and 9 members belonging to Group IIa–IIe, respectively. Multiple sequence alignment analysis (Figure S1) suggested that members from Group I contained two WRKY domains and C2H2-type zinc-finger motifs (C-X4-C-X22–23-H-X-H), except that NcWRKY23 only harbored two WRKY domains. All the 56 NcWRKYs from Group Ⅱ contained one WRKY domain, of which six harbored mutated WRKY domains (WRKYGKK). All members in Group III contained the C2HC-type zinc fingers (C-X7−C-X23-H-X-C).
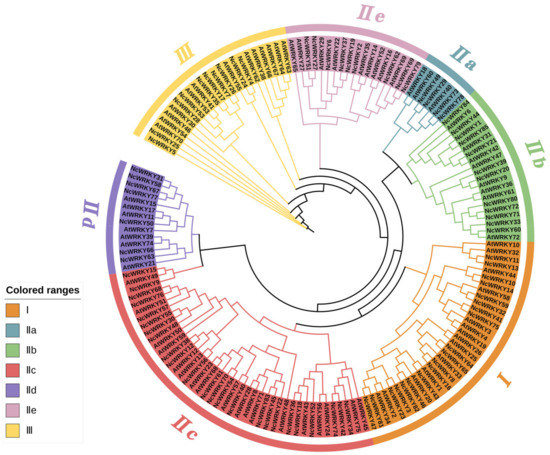
Figure 1.
Unrooted phylogenetic tree representing relationships among WRKY proteins of N. cadamba and A. thaliana. A maximum likelihood (ML) phylogenetic tree was constructed with 5000 bootstrap replicates. The different-colored arcs indicate different groups (or subgroups) of WRKY proteins. WRKY proteins from N. cadamba and A. thaliana were presented with the prefix ‘Nc’ and ‘At’, respectively.
2.3. Gene Structure and Conserved Motif Analysis
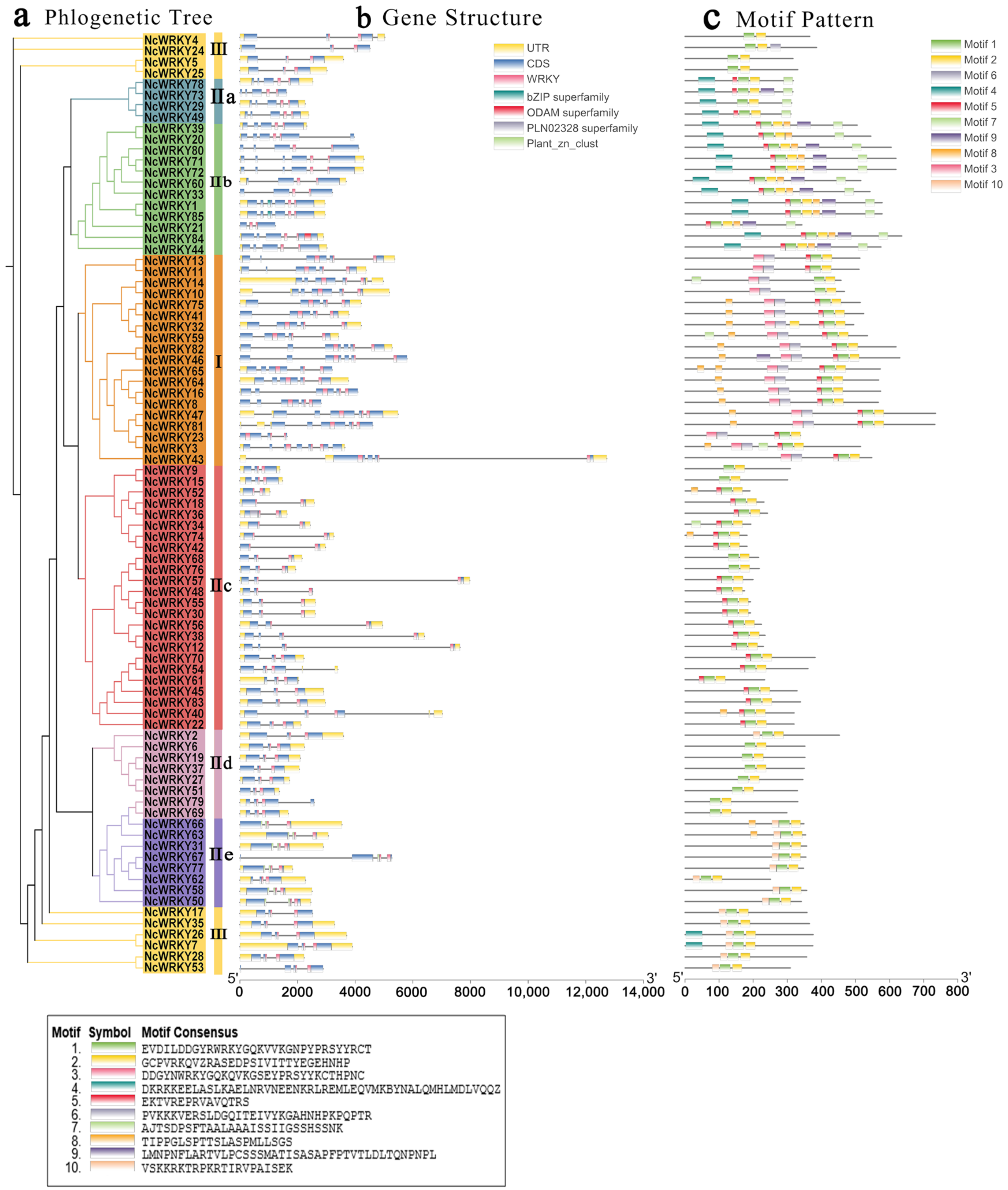
The exon–intron organizations of all the identified NcWRKYs were examined by TBtools. Another ML phylogenetic tree was constructed to align the 85 NcWRKY proteins (Figure 2a). As shown in Figure 2b, the number of introns in the NcWRKYs ranged from 1 to 6. Six NcWRKY genes from Group Ⅱc had only one intron, and NcWRKY3 had six introns. The majority of NcWRKY genes contained two to five introns (37 with two introns, 13 with three introns, 17 with four introns, and 11 with five introns). Interestingly, all NcWRKYs in Group I contained an intron in their respective WRKY domains, but no introns existed in the N-terminal WRKY domains (Figure 2b). Genes within the same group were usually similar in structure, slightly varying in length and distribution, and most genes had UTR regions. For example, all of Group IIa genes had four introns, and there were two UTR regions in all of them except for NcWRKY73. Most of Group IIc contained one to three introns, but NcWRKY40 and NcWRKY54 contained four introns and one of them was located in 3′ UTR.
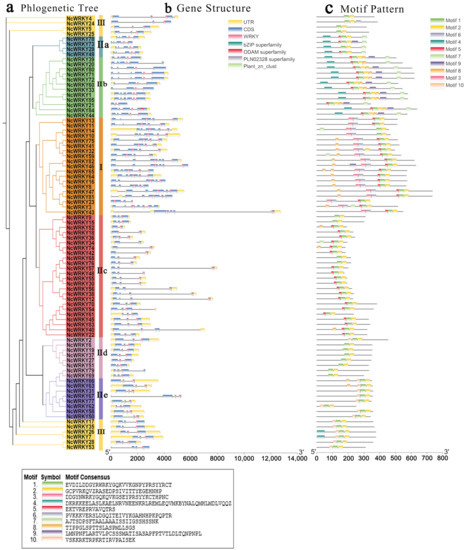
Figure 2.
Phylogenetic relationships, gene structure, and architecture of conserved protein motifs in WRKY genes from N. cadamba. (a) The phylogenetic tree was constructed based on the full-length sequences of NcWRKY proteins using TBtools. Details of clusters were shown in different colors as same of Figure 1. (b) Exon–intron structure of NcWRKYs. Yellow boxes indicated untranslated 5′- and 3′ regions, while blue boxes indicated coding sequence (CDS) and black lines represented introns. The WRKY domains were highlighted by pink boxes and other colors indicate different conserved domains which are found in CDD. The length of genes was estimated using the scale at the bottom. (c) The motif composition of NcWRKY proteins. The motifs, numbers 1–10, were displayed in different-colored boxes.
To further investigate the similarity and diversity of the motif composition of NcWRKY proteins, we annotated 10 conserved motifs predicted by MEME. As exhibited, NcWRKY members in the same subfamily were found to share similar motif profiles (Figure 2c). Motif 1 and motif 2 were highly conserved and distributed across all members of NcWRKY. Most members in Group I and Group IIb contained the highest number of motifs (n = 7), whereas three members in Group Ⅲ (NcWRKY4, NcWRKY5, and NcWRKY25) only have two motifs. In addition, we also found that some motifs were specific to different groups. For example, motif 3 and motif 6 were unique to Group I, whereas motif 9 was specific to Group IIa and IIb. The distinct motif composition might be contributed to the functional diversity among NcWRKYs.
2.4. Chromosomal Distribution and Synteny Analysis of NcWRKYs
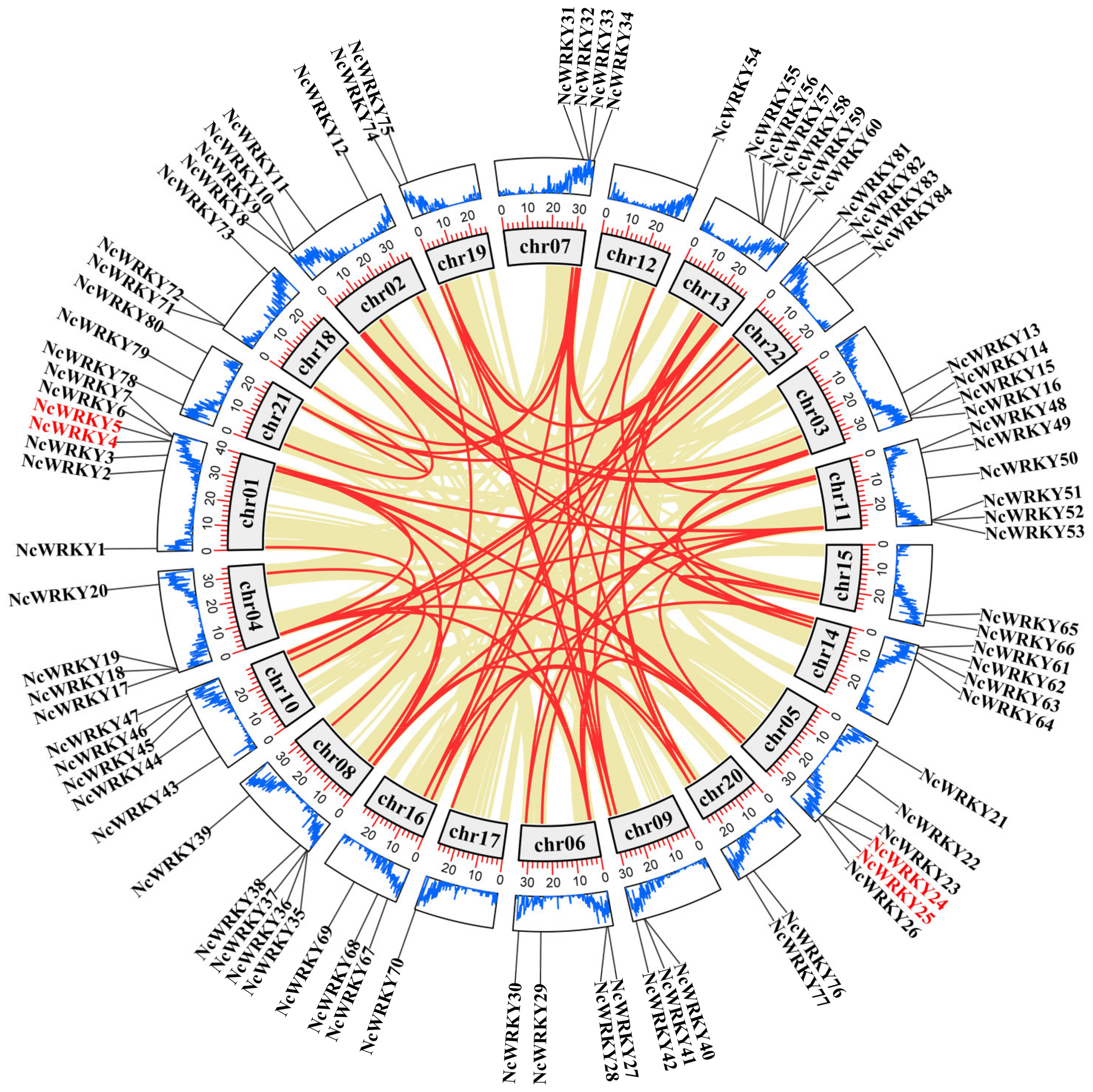
An analysis of genome chromosomal distribution revealed that 84 NcWRKYs were unevenly distributed on 22 chromosomes (Figure 3), while NcWRKY85 could not map to any chromosome. There was no evidence to show a correlation relationship between the number of NcWRKYs and the chromosome length. The investigation of gene duplication events was also performed to obtain insight into the expansion of the NcWRKY family. Four NcWRKYs (NcWRKY4/5 and NcWRKY24/25) were identified as tandem repeat gene pairs located on chr01 and chr05. In addition to tandem duplication, the fragment duplication events of the WRKY gene family were performed (Figure 3). The result showed that 76 segmental duplication events with 62 NcWRKYs were identified. All these results indicated that the NcWRKY family underwent an expansion in N. cadamba possibly generated by gene duplication, and the segmental duplication events played a major role as a driving force for NcWRKYs evolution. To better understand the evolutionary constraints acting on the NcWRKY family, the Ka/Ks ratios of the NcWRKY gene pairs were calculated. All segmental and tandem duplicated NcWRKY gene pairs had Ka/Ks < 1 (Supplementary File S2), suggesting that the N. cadamba WRKY gene family might have experienced strong purifying selective pressure during evolution.
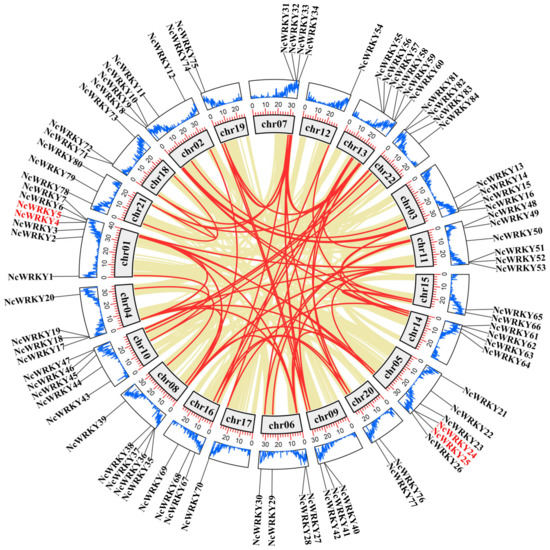
Figure 3.
Chromosome location and synteny analysis of WRKY genes within N. cadamba genome. Khaki lines indicated all synteny blocks in the N. cadamba genome, and the red lines indicated duplicated WRKY gene pairs. The gray boxes indicated the chromosomes of N. cadamba. The blue line in the boxes represented the gene abundance at that position on the chromosome, and the height of the line was proportional to the abundance. The outermost circle indicates the chromosomal locations of WRKY genes in N. cadamba. The genes in red font indicate tandem duplication.
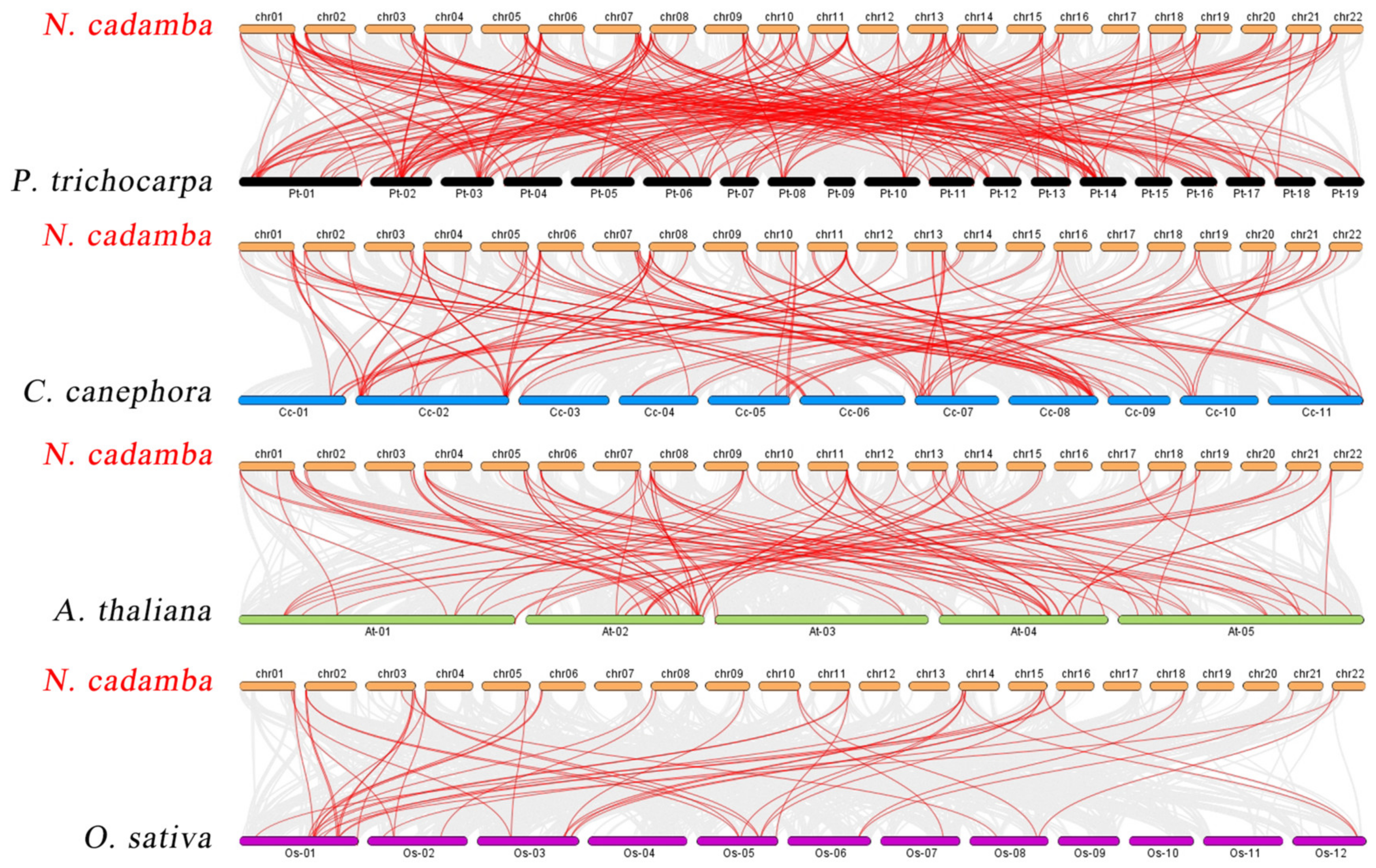
The phylogenetic mechanisms of the N. cadamba WRKY family were further explored by constructing comparative syntenic maps of N. cadamba associated with four representative species, including three represented dicots (A. thaliana, Coffea canephora, and Populus trichocarpa) and one model monocot (O. sativa) (Figure 4). On the whole, 78 NcWRKYs showed a syntenic relationship with those in P. trichocarpa, followed by C. canephora (74), A. thaliana (58), and O. sativa (30). The number of orthologous gene pairs between N. cadamba and the other species (P. trichocarpa, C. canephora, A. thaliana, and O. sativa) was 218, 114, 91, and 45, respectively. More than 90% of the NcWRKYs showed a syntenic relationship with WRKYs in P. trichocarpa, which is higher than C. canephora (87%), indicating that WRKY genes in N. cadamba and P. trichocarpa (both as tall arbor) evolved more closely related in evolution. Specifically, NcWRKY12/38/56 were found to be associated with at least two syntenic gene pairs between N. cadamba and P. trichocarpa, respectively. NcWRKY16 and NcWRKY64 were found to be associated with at least two syntenic gene pairs between N. cadamba and P. trichocarpa/A. thaliana/O. sativa, speculating that they may have played an important role in the WRKY gene family during evolution.
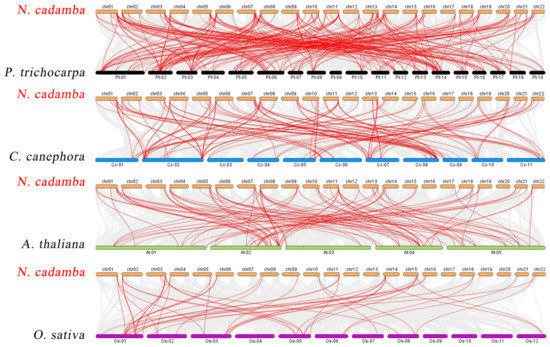
Figure 4.
Synteny analysis of WRKY genes between N. cadamba and four representative plant species. Gray lines in the background indicated all synteny blocks in the genome, while the red lines indicated the duplication of WRKY gene pairs.
2.5. Analysis of Promoter Cis-Acting Elements
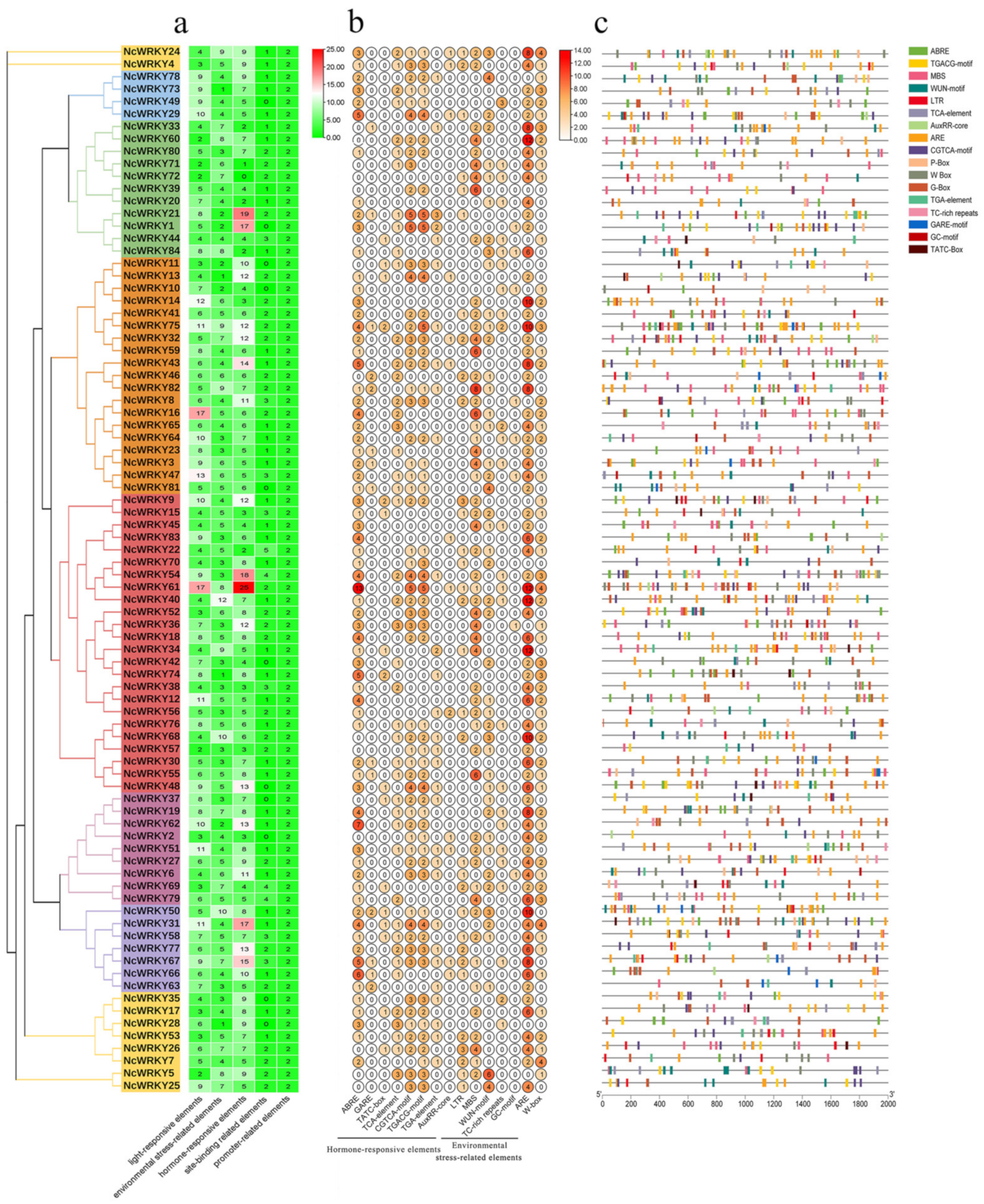
To further understand transcriptional regulation and the potential functions of NcWRKYs in N. cadamba, the cis-acting elements of NcWRKY promoters were predicted using PlantCARE. In addition to the well-characterized site-binding-related elements and promoter-related elements, three categories of cis-regulatory elements were found to be highly concentrated in the promoter region of NcWRKYs, including light-responsive, hormone-responsive, and environmental-stress-related elements. The most abundant elements in NcWRKYs promoters were hormone-responsive elements, which were represented by eight types (Figure 5a). Specifically, ABRE elements (response to ABA) were the most widely distributed and presented in over half of the identified NcWRKYs. Moreover, several environmental-stress-related cis-acting elements responsible for response to low-temperature (LTR), drought (MBS), wound (WUN motif), stress (TC-rich repeats and ARE), and anerobic (GC motif and ARE) were identified (Figure 5b). Specifically, ARE element was prevalent and displayed across most of the NcWRKY promoters, implying that NcWRKYs were responsive to anerobic stress. Taken together, these results suggested the potential role of NcWRKYs in response to stress and hormone signaling pathways. Interestingly, some NcWRKYs comprised more than 2 W-box (TTGACC) in their promoters, such as NcWRKY7, NcWRKY24, NcWRKY31, and NcWRKY61 have four W-box, indicating that these NcWRKYs have the potential function to regulate plant’s defensive response to stresses by self-regulating its expression and the cross-talk between different WRKY TFs.
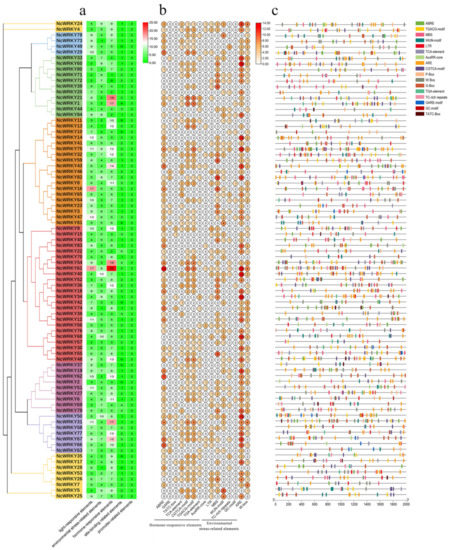
Figure 5.
Prediction of cis-acting elements in promoters of NcWRKYs. (a) The number of five types of cis-acting elements in each NcWRKYs promoter. (b) The number of different elements in hormone-responsive and environmental-stress-related elements. (c) Visualization of environmental-stress-related and hormone-responsive elements, W-box and G-box in NcWRKYs promoters.
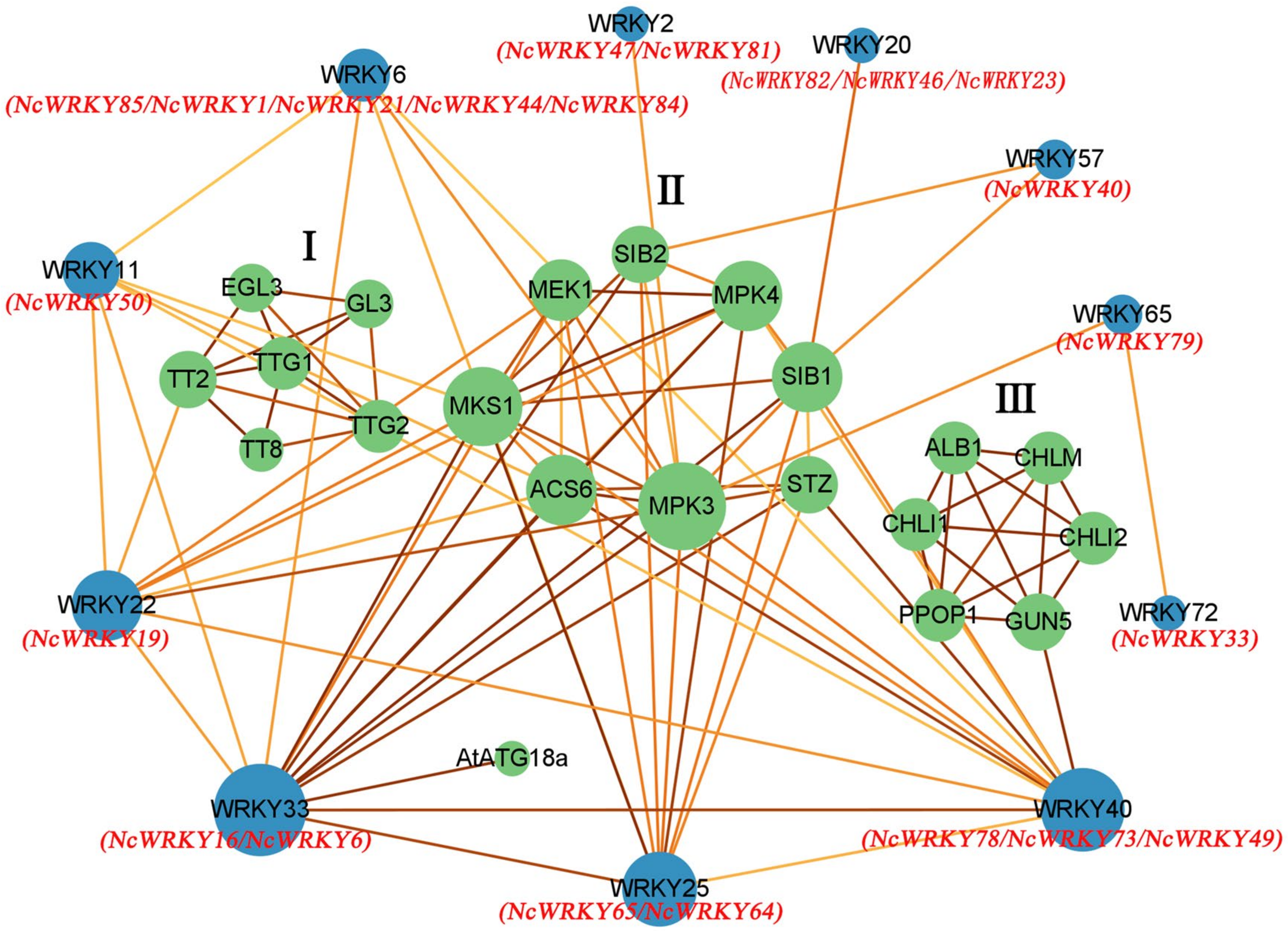
2.6. Protein Interaction Network of NcWRKY
To elucidate the biological functions and regulatory network of NcWRKY proteins, the 18 homologous WRKY proteins in A. thaliana were used to predict the protein–protein interaction network of NcWRKY proteins. The results of homologous similarity were shown in Supplementary File S3. The results indicated 11 AtWRKY proteins and 21 corresponding interacting functional proteins which can be divided into three groups (Figure 6). Most AtWRKY proteins interact with more than one protein, and eight proteins can interact with more than two other functional proteins. Some proteins that interact with each other in a group and only interact with one AtWRKY protein. Group 1 (EGL3, GL3, TT2, TT8, TTG1, and TTG2) only interact with AtWRKY22 by TT2 and Group 3 (CHLM, CHLI1, PPOP1, CHLI2, ALB1, and GUN5) only interact with AtWRKY40 by GUN5. According to the biological process analysis in the GO database, proteins of Group 1 were related to epidermal cell fate specification, positive regulation of anthocyanin biosynthetic process, trichome differentiation, and jasmonic-acid-mediated signaling pathway (Supplementary File S4). Proteins of Group 3 were related to the chlorophyll biosynthetic process and aromatic compound biosynthetic process. Proteins of Group 2 were mainly related to MAPK cascade which responds to abiotic and biotic stresses, such as defense and immune system processes, and responds to osmotic and extreme temperature stresses [30,31,32]. This group also included positive regulation of autophagy, cellular heat acclimation, and camalexin biosynthetic process. AtWRKY33 (orthologous with NcWRKY8 and NcWRKY16), an important TF in abiotic stress [12,33] and phytoalexin biosynthesis [34], played a key role in the whole protein interaction network, especially in MAPK cascade. AtWRKY22 (orthologous with NcWRKY19) and AtWRKY25 (orthologous with NcWRKY64 and NcWRKY65) participated in aging and response to cold, respectively.
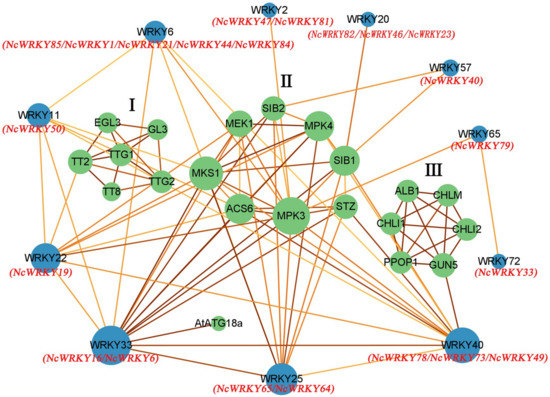
Figure 6.
Protein–protein interaction network for NcWRKY proteins based on their orthologs in A. thaliana. The green and blue circles represented the interacting proteins found in String and orthologs proteins in A. thaliana, respectively. The red name below the blue circle indicates the WRKY protein orthologous to this protein in N. cadamba. The abbreviated names were the genes of A. thaliana. The color of the line represents the strength of the interaction, in which a darker line indicated higher reliability.
2.7. Expression Patterns of NcWRKYs Gene in N. cadamba Tissues by RNA-Seq
The expression patterns of all 85 NcWRKYs in the transcriptome data, which was derived from different tissues, including bud, young leaves, old leaves, bark, cambium, phloem, root, and young fruit, were investigated in this study (Figure S2, Supplementary File S5). The results showed that all 85 NcWRKYs were expressed in different tissues (FPKM > 0), and they were generally highly expressed in roots and old leaves. Some of the NcWRKYs were not expressed in the tissues we tested due to special temporal and spatial expression patterns.
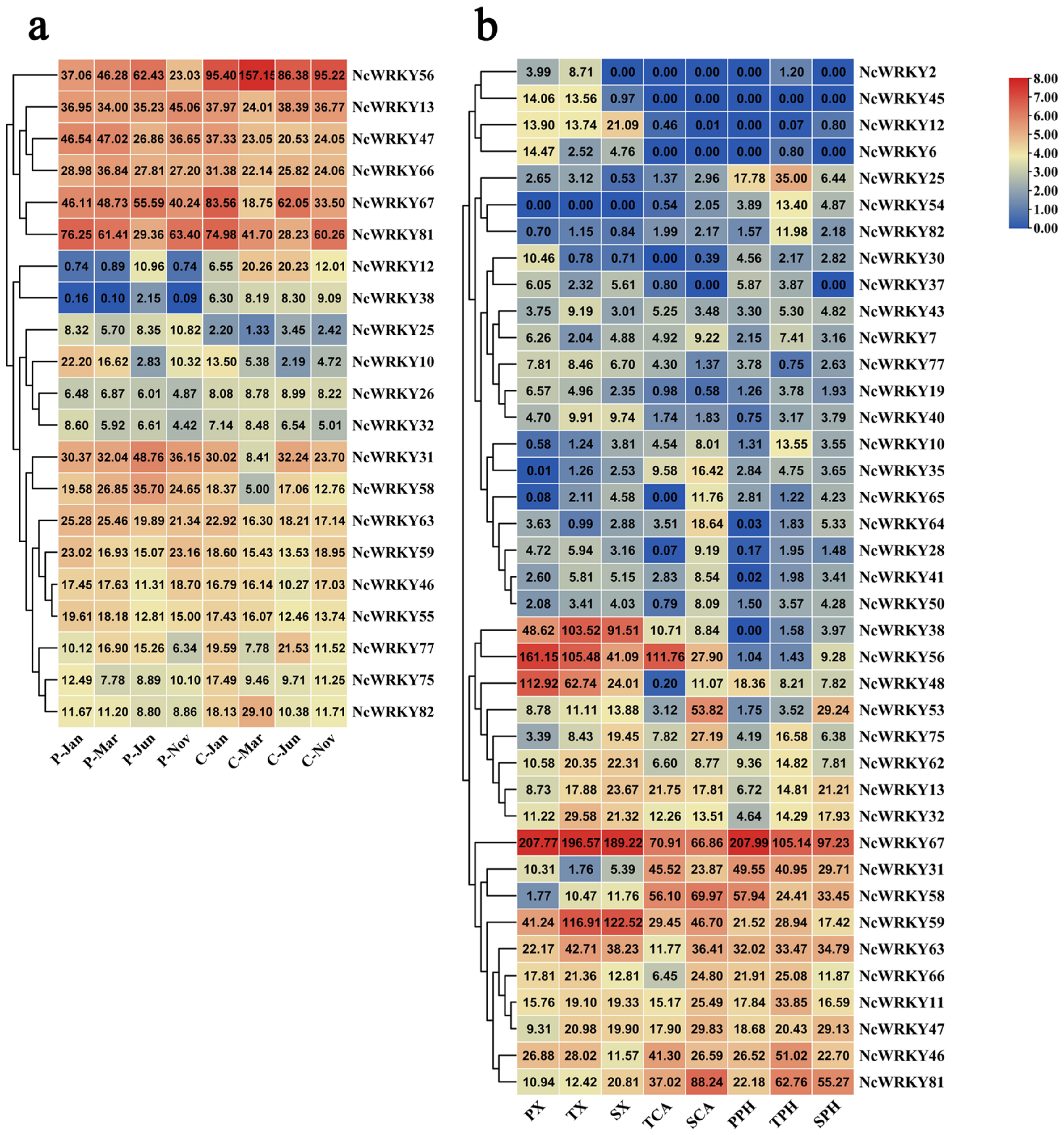
Since the wood formation is of great significance for perennial trees, we further focused on the analysis of the expression patterns in different seasonal stages of cambium and phloem. NcWRKY13/47/56/66/67/81 had high expression in both cambium and phloem in different seasons in the whole year (Figure 7). NcWRKY12 and NcWRKY38 had a similar expression mode, with higher expression in cambium but only expressed in phloem of June. To further explore the expression profiles of NcWRKY in different developmental vascular tissues, we isolated cambium, phloem, and xylem cells by laser microdissection at three stages, including primary growth, secondary growth, and the transition stage from primary to secondary growth, and the RNA-seq was subsequently carried out (data not published). The results showed that 39 NcWRKYs exhibited different expression levels during diverse phases of wood formation, suggesting that these NcWRKY genes played distinct roles during wood formation. For example, five NcWRKYs (NcWRKY38/48/56/59/67) had high expression in xylem of all three phases of wood formation, of which NcWRKY67 exhibited the highest expression, suggesting that these genes played important roles in regulating xylem development. In cambium, NcWRKY56, NcWRKY67, and NcWRKY58 showed the higher expression in the transition stage, while NcWRKY81 had the highest expression in the stage of secondary growth (Figure 7b). Moreover, some NcWRKYs had high expression in three different wood development stages, such as NcWRKY38/46/66/56/59/63/67, suggesting that these genes may participate in the development of stem of N. cadamba. Altogether, these results indicated that NcWRKY genes evolved diverse biological functions that were important for wood formation.
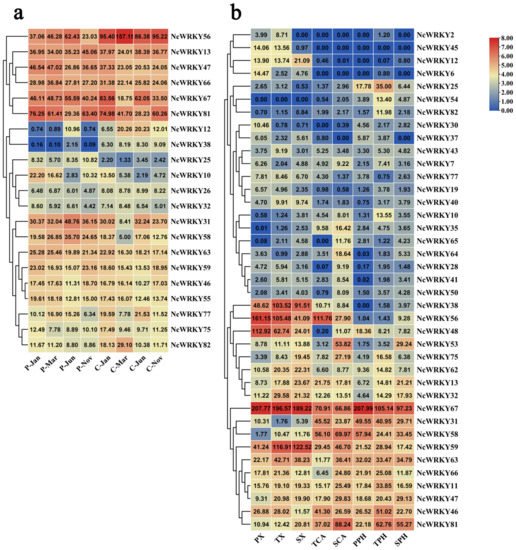
Figure 7.
NcWRKYs transcriptional levels at different developmental stages of cambium, phloem, and xylem and combined analysis by laser capture microdissection (LCM)-RNA-seq. The heatmaps were created by TBtools based on the transformed data of log2 (FPKM+1) values, and the cluster analysis was performed on gene expression level by row. (a) Expression of WRKY genes in cambium and phloem in different months. C and P indicated cambium and phloem, respectively, and suffixes ‘Jan, Mar, Jun, and Nov’ indicate January, March, June, and November, respectively. (b) Expression of WRKY genes in xylem, cambium, and phloem at the stages of primary growth, secondary growth, and transitional stages from primary to secondary growth. PX, primary xylem; TX, xylem at the transitional stage from primary to secondary growth; SX, secondary xylem; TCA, cambium at the transitional stage from primary to secondary growth; SCA, secondary cambium; PPH, primary phloem; TPH, phloem at the transitional stage from primary to secondary growth; SPH, secondary phloem.
2.8. Expression Patterns of NcWRKYs in Response to Different Treatments
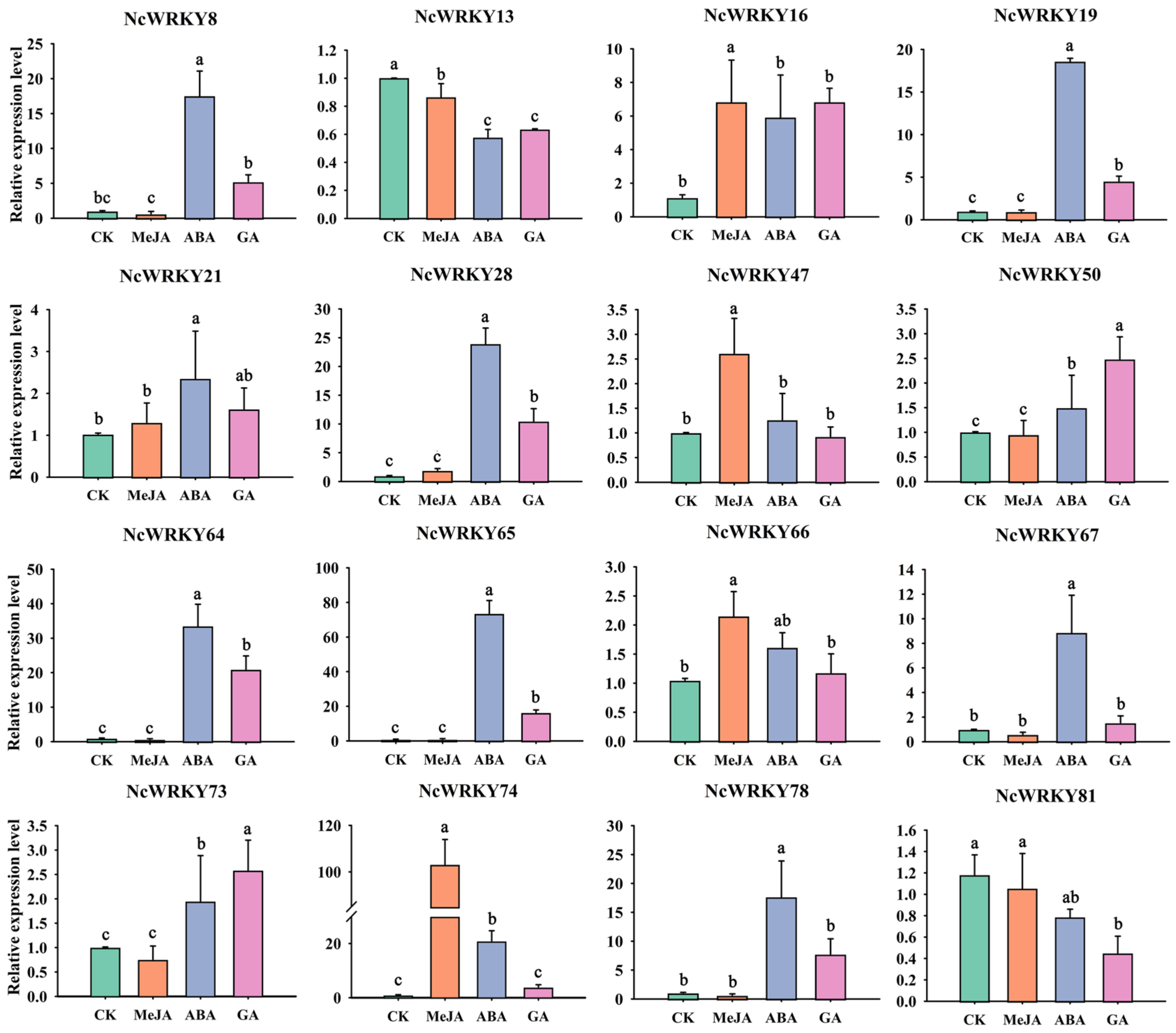
To further confirm whether the expression of NcWRKYs was influenced by different abiotic stresses and hormonal treatments, RT-qPCR was used to examine the expression patterns of the 16 selected NcWRKYs in different treatments, including ABA, MeJA, GA, PEG6000, and NaCl. As to various hormone treatments (Figure 8), most of the 16 NcWRKYs responded to multiple treatments at the transcriptional level. For instance, NcWRKY64 was induced by all tested treatments, except MeJA treatment. Some genes exhibited the same response pattern, such as NcWRKY28/64/78, which was strongly induced by ABA and GA, but slightly upregulated under MeJA treatment. On the contrary, NcWRKY50 and NcWRKY73 change little in different hormone treatments. In addition, several NcWRKYs were simultaneously induced by one treatment. For example, NcWRKY8/19/28/65/67/74/78 were induced by ABA treatment (the fold change was more than 8), NcWRKY8/16/28/64/65/78 were induced by GA3 treatment (fold change > 5), and five NcWRKY16/28/47/66/74 were induced by MeJA. Conversely, some genes were down-regulated after treatment, such as the transcript levels of NcWRKY78 were decreased to half of the control by MeJA treatment, and NcWRKY13 and NcWRKY81 were repressed after ABA and GA3 treatments.
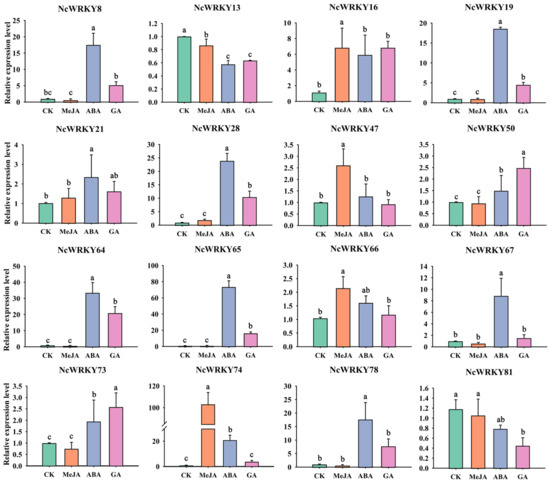
Figure 8.
Expression profiles of 16 selected NcWRKYs in response to various hormone treatments, including MeJA, ABA, and GA. NcUPL was used as the endogenous control. Error bars indicated the standard deviation of three biological replicates, each containing three technical replicates. The same letters indicate groups that were not significantly different from each other according to Duncan’s multiple range test, p = 0.05.
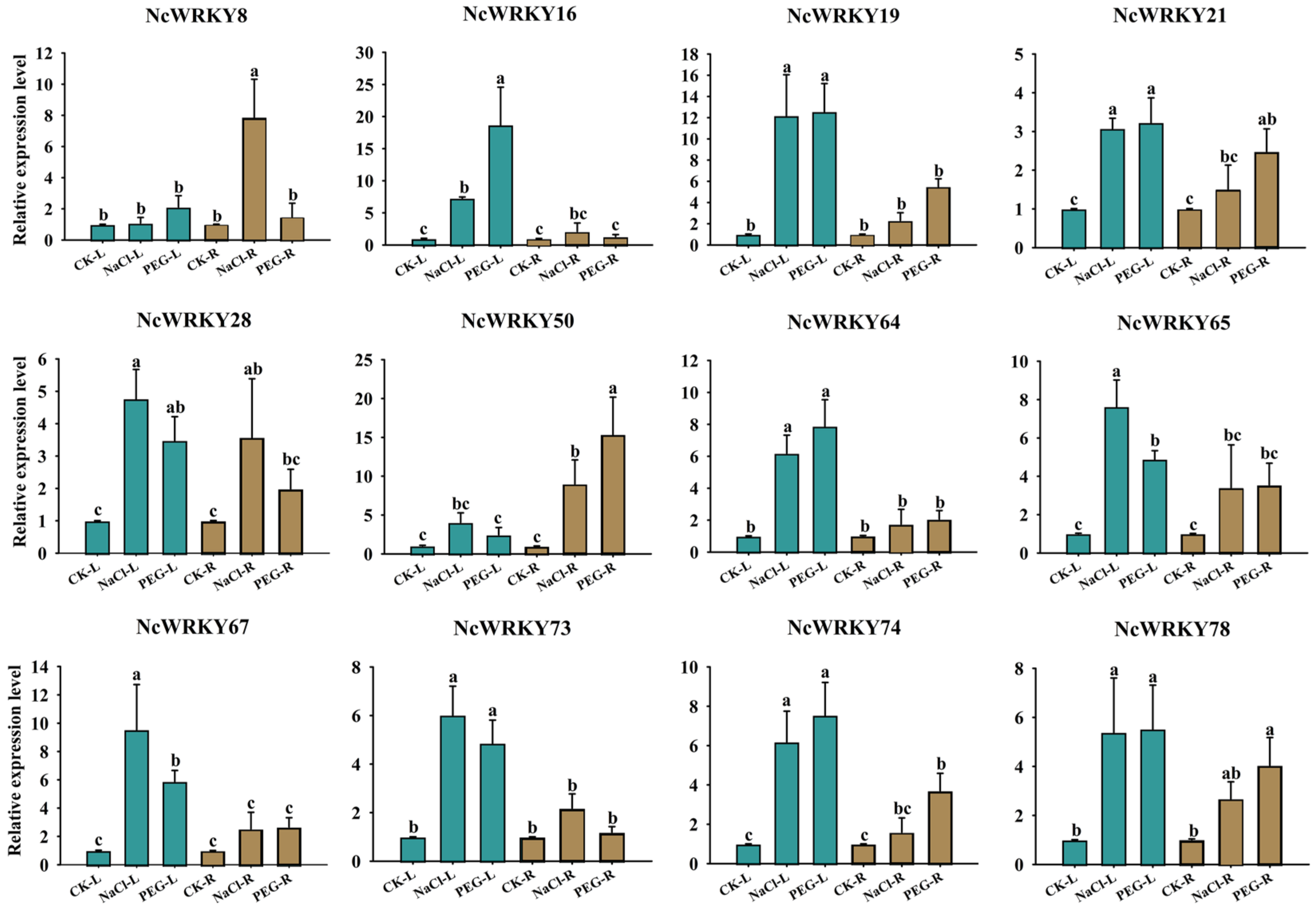
For PEG6000 and NaCl treatments, we selected 12 NcWRKY genes for detecting their expression by RT-qPCR. As shown in Figure 9, most genes were more strongly responsive in leaves compared to roots, such as NcWRKY16/19/64/65/73. On the contrary, NcWRKY50 had a stronger response intensity in roots. Four of the twelve genes (NcWRKY21/28/65/78) had little difference in expression between leaves and roots under PEG6000 and NaCl treatments. Interestingly, NcWRKY8 only had a comparatively large response intensity in the roots of NaCl treatment.
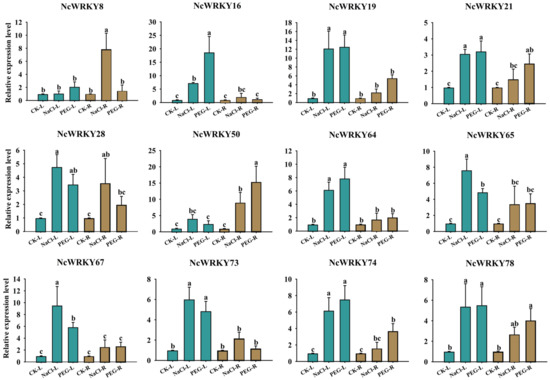
Figure 9.
Expression profiles of 12 selected NcWRKY genes in response to NaCl and PEG6000 treatments. The leaf and root were collected after NaCl and PEG stress treatments and used for the detection of NcWRKYs expression. The suffix ‘L’ and ‘R’ refers to the leaf and root, respectively. NcUPL was used as the endogenous control. Error bars indicated the standard deviation of three biological replicates, with each comprising three technical replicates. The same letters indicate groups that were not significantly different from each other according to Duncan’s multiple range test, p = 0.05.
2.9. MeJA Promotes Cadambine Biosynthesis and NcWRKYs Expression
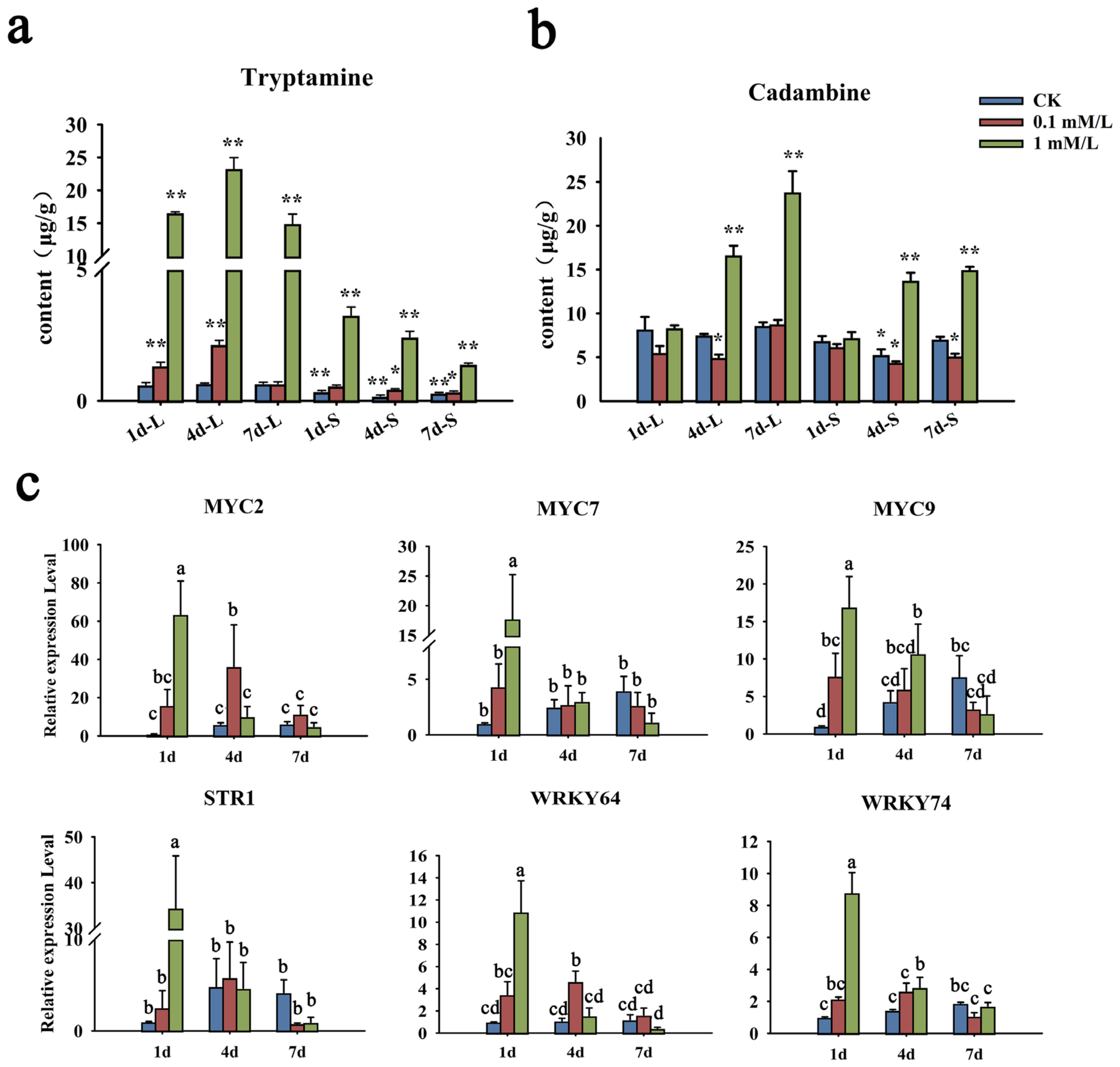
MeJA, as an exogenous hormone, can not only play a defensive role in plants, but also widely regulate the synthesis of a variety of secondary metabolites. The N. cadamba plants were treated with 0.1 mM/L and 1 mM/L of MeJA, and the contents of cadambine and tryptamine, the precursor of cadambine, were tested by UHPLC-MS-MS (Supplementary File S6). In leaves, the content of tryptamine was obviously elevated at 1 d and reached its highest at 4 d in the 1 mM/L MeJA treatment group. At 7 d after MeJA treatment, it dropped to the same level as 1 d (Figure 10a). However, the accumulation of cadambine gradually increased and reached the highest abundance at 7 d. In the stem, the content of tryptamine was slightly decreased from 1 d to 7 d. On the contrary, cadambine accumulation was obviously elevated at 4 d and slightly enhanced at 7 d (Figure 10b). As for the treatment of 0.1 mM/L, the changes of these two metabolites’ accumulation were similar to that of 1 mM/L, but with gentle enhancement in both leaves and the stem.
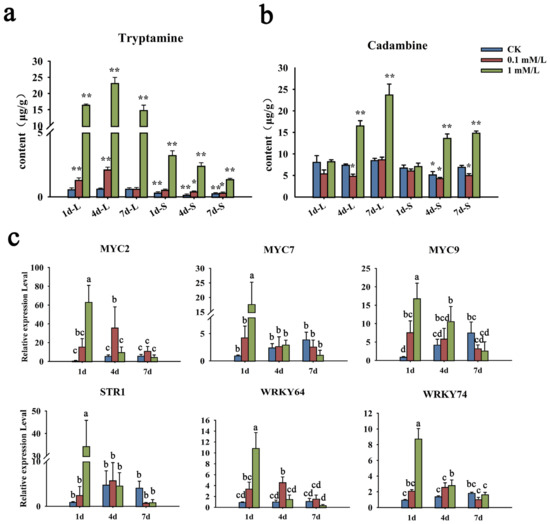
Figure 10.
Changes of metabolite content and gene expression after MeJA treatment. (a,b) The content change of tryptamine and cadambine under MeJA treatment. Quantitative determination of tryptamine and cadambine was performed by UHPLC-MS-MS. (c) Transcript levels of NcMYCs, NcSTR1, and NcWRKY64/74 in leaves of N. cadamba seedlings under MeJA treatments. The expression levels were determined by qRT-PCR. NcMYCs, as a kind of key transcriptional factor of MeJA signaling pathway, reflect the response of the jasmonic acid signal pathway after treatment. NcSTR1 can catalyze the synthesis of strictosidine in N. cadamba, reflecting whether MeJA treatment affects the expression of synthesis pathway genes. NcRPL was used as the reference gene. Error bars represented the standard deviation of three biological replicates. The same letters indicate groups that were not significantly different from each other according to Duncan’s multiple range test, p = 0.05. Statistically significant differences compared with the CK (untreated control) at 1 d were determined by t-tests (* p < 0.05, ** p < 0.01).
As the key intermediate of the cadambine biosynthetic pathway, NcSTR1 catalyzed the synthesis of strictosidine in N. cadamba [29]. Moreover, MYC is inhibited by JAZ protein, which leads to the downstream regulatory switch being turned off. When MeJA acted as an activation signal, JAZ protein would be degraded, and then the MYC transcription factor in the inhibited state was released, which thereby activates the target gene expression activity [35,36]. Accordingly, we further detected the expression patterns of NcMYCs, NcSTR1 (evm. model. Contig 69.90), and NcWRKYs after MeJA treatment by RT-qPCR (Figure 10c). For the 1 mM/L MeJA treatment group, three NcMYCs’ (NcMYC2, 7, and 9) expression was strongly induced by MeJA, with a peak after 1 d, and gradually decreased after 4 d and 7 d. The expression of the STR1 gene and two WRKY genes (NcWRKY64 and NcWRKY74) was similar to NcMYCs, with the same pattern. However, NcMYC2, STR1, NcARKY64, and NcWRKY74 were induced at later time points (i.e., 4 d) in the 0.1 mM/L MeJA treatment group, suggesting that a higher concentration of MeJA was essential for rapid induction of expression of these genes. A control time course of treatment with the solvent ethyl alcohol showed no effect on the expression of NcARKY64 and NcWRKY74, but promotes the expression of NcMYC and STR1 genes. More NcWRKYs were upregulated significantly after treatment with 1 Mm/L MeJA (Figure S3).
3. Discussion
The WRKY family is a large transcription factor family and plays pivotal roles in higher plants. Identification and characterization of WRKY genes have been performed in many plants, including the model plants, crops, and medicinal plants, such as Arabidopsis [3], corn [37], tomato [38], ginseng [39], and poplar [40]. In the present study, a total of 85 NcWRKY genes were identified in the genome of N. cadamba by bioinformatics analysis. Their features, expression patterns, and putative function were studied in detail.
3.1. Diverse Characterization of WRKY in N. cadamba
The uneven gene family size of WRKY was found in different plants. A previous report had indicated that the WRKY members ranged from 59 to 161 among seven species, of which Z. mays had the most prominent WRKY family members (genome size 2100 Mb), and a minor WRKY family was found in V. vinifera (genome size 427 Mb) [39]. N. cadamba had moderate WRKY family members (85), in accordance with the moderate genome size (744.5 Mb). These results indicated that the number of WRKY family members was correlated with genome size. However, an exception was found in ginseng, in which its genome size (2900 Mb) is larger than that of Z. mays, but with fewer WRKY family members [39]. Interestingly, N. cadamba possesses comparatively a greater number of WRKY genes compared with many dicotyledonous plants, including Solanum lycopersicum (81 WRKY genes), A. thaliana (72 WRKY genes), D. carota (67 WRKY genes), Salvia miltiorrhiza (61 WRKY genes), and C. roseus (47 WRKY genes) [41]. Gene duplication was found to play a very important role in the expansion of the WRKY gene family. In N. cadamba, a total of 76 segmental duplication events are identified in NcWRKYs, and the genes involved in segment duplication do not have distinct subgroup distributions, indicating that the WRKY family has no obvious evolutionary differences in the distribution of N. cadamba. Moreover, for all pairs, the Ka/Ks ratios are <1, indicating that the WRKY gene family in N. cadamba has undergone purifying selection rather than positive selection, and indicating that the NcWRKYs are highly conserved.
The WRKY domain is the key sequence that determines the specific binding of the WRKY protein to the cis-element W-box. According to previous studies, variations of the WRKYGQK motif in the WRKY domain might influence normal interactions of WRKY genes with downstream target genes [42]. The multiple sequence alignment results revealed that six NcWRKY proteins (NcWRKY30/48/55/57/68/76) in Group IIc had sequence variation in their WRKY domain (WRKYGKK), which binds specifically to the WK-box (TTTTCCAC) in tobacco [43]. According to transcriptome data, these six NcWRKYs had similar expression patterns, mainly expressed in old leaf, young fruit, and root (Additional Figure 2). The result indicated that the variation of the WRKY domain in N. cadamba may not affect its binding function, and the six NcWRKYs may have similar functions. It might be worth to further investigate whether the mutated domain confers a special function. The diversification of exon/intron patterns played a vital role in the evolution process of many gene families. We found that 85 NcWRKYs contained between one and six introns, consistent with that in pineapple [44], cucumber [45], and populus [46]. NcWRKY proteins from Group I have two WRKY domains, and no domain loss events were found in other NcWRKY proteins, which was similar to most of the dicotyledonous plants.
3.2. The Potential Function of NcWRKYs in Vascular Development and Response to Hormone and Abiotic Stress
Based on transcriptome data, some valuable clues about the functional role of NcWRKYs which are involved in the specific physiological process in N. cadamba were obtained. For different developmental stages of cambium, phloem, and xylem, some WRKY genes have high expression levels, and these genes were highly overlapping, such as NcWRKY12/38/56 specifically expressed in different developmental stages of cambium and phloem. These genes were orthologous with the MiWRKY12 and PtrWRKY19, which participate in pith secondary cell wall formation in Miscanthus and P. trichocarpa, respectively [47,48]. Moreover, in atwrky13 mutants, lignin-synthesis-related genes were repressed, and the number of sclerenchyma cells, stem diameter, and the number of vascular bundles were reduced [49]. NcWRKY12/38/56 were in the same subgroup as AtWRKY13, suggesting that they might involve in the stem development of N. cadamba. Accordingly, we inferred that these three NcWRKYs may participate in vascular development.
A variety of conserved cis-regulatory elements were shown in the promoter region of NcWRKYs, which were involved in a variety of functions, including hormone and abiotic stress responses (Figure 5). The response of NcWRKYs to hormone and abiotic stresses can provide valuable clues to reveal the potential role of WRKY genes in N. cadamba. In this study, 16 NcWRKYs chosen from different groups were subjected to hormones, salt, and drought stress treatments profiled by means of RT-qPCR. The result shows that all of NcWRKYs can be induced by at least two hormones, implying that a single WRKY gene can be regulated by various hormones, similar to that in Arabidopsis, AtWRKY18/40/60 were shown to participate in signaling pathways that were mediated by plant hormones SA, JA, and ABA [8], and Group III members of CsWRKYs in Cymbidium sinense were strongly induced in response to various hormone treatments [50]. Previous studies have revealed that WRKYs are important regulators in linking hormone signaling in response to environmental stresses [51,52]. FcWRKY40 from Fortunella crassifolia was involved in ABA signaling pathways and positively regulated salt tolerance by directly binding to and activating the promoters of FcSOS2 and FcP5CS1. The transcriptions of NcWRKY8/16/64/65 were promoted simultaneously by ABA and drought stress. In addition, several stress-related transcriptional regulatory elements, including an ABA-responsive element and MYB binding site involved in drought responses, were found in these NcWRKYs. These results suggested that these NcWRKYs may regulate drought responses through ABA-dependent signaling pathways, but this remains to be elucidated in further study. Moreover, these NcWRKYs have a phylogenetically closest relationship with AtWRKY33, a typical ABA and drought-responsive WRKY gene in A. thaliana [53,54], supporting that they play important roles in ABA signaling pathway and response to drought stress.
3.3. The Involvement of NcWRKYs in Cadambine Biosynthesis
Jasmonic acid (JA) is an important elicitor in plant secondary metabolism at the transcriptional level by altering the expression of a set of biosynthesis genes [55,56]. It has been shown that methyl-jasmonate (MeJA) induced the expression of the terpenoid indole alkaloid (TIA) biosynthesis genes, including the strictosidine synthase (STR) gene [55], resulting in promoting TIA metabolism in Catharanthus roseus [57]. In this study, cadambine and the key intermediate product tryptamine were both induced under MeJA treatment, suggesting that MeJA acted as an elicitor in cadambine biosynthesis. Our results facilitated that MeJA plays a significant role in plant metabolism. In addition, the MYC gene family is a kind of key TF in the MeJA signal pathway [58]. The expression levels of NcMYCs were highly induced after MeJA treatment (Figure 10c), indicating that the MeJA signal pathway was activated in N. cadamba seedlings.
Studies have indicated that WRKY proteins participated in transcriptional regulating biosynthesis of secondary metabolites, including alkaloids [24], volatile terpenes [59], and anthocyanin [60]. It has been reported that several WRKY proteins may regulate secondary metabolism biosynthesis in response to JA elicitation. In cotton, GaWRKY1 was strongly induced by MeJA and participated in the regulation of sesquiterpene phytoalexin biosynthesis by transactivating the promoter of the (1)-δ-cadinene synthase (CAD1) gene [20]. The expression pattern of NcWRKY64 and NcWRKY74 was consistent with that of NcMYCs and NcSTR1, which were upregulated after MeJA treatment. In addition, there were two WRKY TF binding site W-box (TTGACC) within 2000 bp upstream of the NcSTR1 promoter (Figure S4). We proposed that NcWRKY64/74 has the potential function of positively regulating the biosynthesis of cadambine by activating NcSTR1 in response to MeJA. The interaction between NcWRKY64/74 and NcSTR1 remained to be investigated in the future study.
Furthermore, AtWRKY33, OpWRKY6, CrWRKY1, and TcWRKYs were involved in the regulation of camalexin (Indole alkaloids), camptothecin (Quinoline alkaloid), vinblastine (Indole alkaloids), and taxol (Diterpene alkaloids) biosynthesis, respectively [18,23,24,61,62]. AtWRKY33 and NcWRKY64, OpWRKY6 and NcWRKY74 were close to each other in the phylogenetic tree (Figure S5), and they have the same WRKY domain (WRKYGQK) and zinc-finger motif (C-H4-X23-H-X-H) (Figure S6). Therefore, according to the high homology between AtWRKY33, NcWRKY64, and NcWRKY74, we inferred that NcWRKY64 and NcWRKY74 were essential for MeJA-responsive TIA accumulation in N. cadambia. No MeJA-responsive cis-acting element was found in the NcWRKY74 promoter, suggesting that the expression of NcWRKY74 was induced by MeJA in an indirect manner. Further functional characterization of NcWRKY64 and NcWRKY74 by overexpression or knockout of these NcWRKY genes may help to elucidate their function in TIA biosynthesis, which will provide crucial information for understanding the MeJA-WRKY-STR regulatory module in the cadambine biosynthesis.
4. Materials and Methods
4.1. Data Sources
All raw and processed sequencing data used in this study were derived from the previous research of our research group and can acquire in the NCBI BioProject database under accession number PRJNA650253. The raw sequencing data of the resequencing data and transcriptome were downloaded from the NCBI BioSample database under accession numbers SAMN15700860 and SAMN15700859, respectively. The genome annotation and assembled genome sequences were from the Figshare website (https://Figshare.com/s/ed20e0e82a4e7474396b) [29]. The AtWRKY protein sequences were downloaded from the A. thaliana Information Resource website (https://www.arabidopsis.org/index.jsp).
4.2. Identification of WRKY Genes in N. cadamba
To identify all candidate WRKY genes in N. cadamba, a BLASTP search with a threshold e-value of 1 × 10−5 was performed using A. thaliana WRKY protein sequences as query sequences. The hidden Markov model (HMM) file corresponding to the WRKY domain (PF03106) was downloaded from the Pfam database (ftp://ftp.ebi.ac.uk/pub/databases/Pfam/current_release/Pfam-A.hmm.gz). The Simple HMM Search program of TBtools [63] was used to search all the potential WRKY-domain-containing protein sequences in the N. cadamba genome. The unique potential WRKY proteins of N. cadamba based on the results of BLASTP and Simple HMM Search were further validated using the SMART (http://smart.embl.de/smart/batch.pl) and conserved domain database from NCBI (https://www.ncbi.nlm.nih.gov/cdd/) to determine that they indeed contained the core domain sequences. Eighty-five NcWRKY proteins in N. cadamba were finally obtained. The online tools from ExPASy (https://web.expasy.org/protparam/) were used to analyze the amino acid number, isoelectric point (pI), and MW of NcWRKY proteins. Subcellular localizations were predicted by the Plant-mPLoc website. (http://www.csbio.sjtu.edu.cn/bioinf/plant-multi/).
4.3. Phylogenetic Analysis of WRKY Family Members and Sequence Alignment
A phylogenetic tree with 72 AtWRKY protein sequences and the identified NcWRKY proteins sequences was constructed by One Step Bulid, an ML Tree program from TBtools [63] with default parameters, followed by visualization and optimization in iTOL [64]. The WRKY family members from A. thaliana were used as a reference for the classification of the WRKY family members in N. cadamba. All WRKY domain sequences of candidate NcWRKY proteins were aligned using DNAMAN software.
4.4. Analysis of Conserved Motifs, Conserved Domains, and Gene Structures
The conserved motifs of the NcWRKY proteins were analyzed using Simple MEME Wrapper from TBtools [63] with the following parameters: Num of Motifs was 10, Min Motif Width was 6, Max Motif Width was 50, and Max E-value was 10. The conserved domains of NcWRKY proteins were searched by CDD (https://www.ncbi.nlm.nih.gov/cdd/). The conserved motifs, conserved domains, and gene structures which were obtained from the gene structure annotation file were visualized by TBtools.
4.5. Promoter Cis-Regulatory Element Analysis and Chromosomal Localization
The 2000 bp gene sequence upstream of the initiation codon (ATG) of NcWRKYs was considered as the gene promoter sequence and deprived of the N. cadamba genome by TBtools [63]. Cis-acting elements in the promoter region were analyzed using the online software PlantCARE [65] and subsequently visualized using TBtools [63]. Location Visualize from GTF/GFF of TBtools was used to determine the chromosomal position of the identified NcWRKYs.
4.6. Colinear Analysis and Selective Pressure
To identify the pattern of gene duplication, One Step MCScanX from TBtools [63] with default parameters (E-value cut-off < 1 × 10−10 and Num of BlastHits with 5) was used to analyze WRKY genes in N. cadamba vs. itself and N. cadamba vs. A. thaliana/Coffea canephora/Populus trichocarpa/rice, respectively. The results were visualized using TBtools. To assess the selection pressure of genes encoding WRKY proteins, the ratio of nonsynonymous (Ka)/synonymous (Ks) (Ka/Ks is an indicator of selective pressure) was used to evaluate its evolutionary pressure. The values of Ka, Ks, and Ka/Ks were calculated by Simple KaKs Calculator in TBtools [63].
4.7. Homology Analyses and Protein Interaction Network Analysis
In order to obtain the orthologous gene pairs and paralogous gene pairs, all the individual NcWRKYs protein sequences were compared against AtWRKY protein sequences using BLASTP (https://blast.ncbi.nlm.nih.gov/Blast.cgi) with the following settings: Total Score >200, Query Cover >than 60%, E-value cut-off < 1 × 10−10, and Identity >45%. Using homolog A. thaliana WRKYs as a template, the WRKY protein interaction network of N. cadamba was analyzed using STRING (https://string-db.org/) with a threshold required score >0.4. The interaction network was further visualized and analyzed using the software Cytoscape 3.9.1.
4.8. Plant Material and Stress Treatments
For salinity and drought treatments, 7-week-old clonal plants of N. cadamba with uniform height were transferred into MS liquid medium supplied with 100 mM/L NaCl and 10% PEG6000 solution, respectively. The leaves and roots were collected at 8 h. For phytohormone analysis, the seedlings that were transplanted outdoors and grow up to 60 cm after 3 months were respectively sprayed 10 mg/L ABA, GA3, and MeJA 50 mL. Then wrap the leaves in fresh-keeping bags. The young leaves were collected 2 d after treatments. All the collected samples were immediately frozen in liquid nitrogen and stored at −80 °C for subsequent analysis.
The 4-month-old N. cadamba plants (about 100 cm in height) were sprayed with different MeJA concentrations (0.1 and 1 mM/L) that were mixed with pure water. The mother liquor is 1 mM/mL, which is prepared from MeJA stock solution and absolute ethanol. At about 8:30 every morning, the MeJA was sprayed on the leaf’s front and back surface till dripping for 7 consecutive days. The roots, stems, and leaves of the plants were sampled and stored at −80 °C on the first (1 d), the fourth (4 d), and the seventh (7 d) days after spraying was stopped. Each sample contained three biological replicates.
4.9. Expression Patterns of NcWRKYs
The transcriptome sequencing data of young leaves, old leaves, bud, bark, phloem, cambium, young fruit, and root from 5-year-old N. cadamba were collected for RNA extraction for RNA-seq in our previous study [29]. The vascular cells, including cambium, phloem, and xylem cells, at three developmental stages (primary growth, secondary growth, and the transition from primary to secondary growth) were captured by laser microdissection and used for subsequent RNA-seq [66]. The transcript abundance of N. cadamba WRKY genes was calculated as fragments per kilobase of exon model per million mapped reads (FPKM). The HeatMap program in TBtools was used to visualize the expression of the target gene obtained from the transcriptome. The HeatMap program in TBtools was used to visualize the expression of the target gene obtained from the transcriptome. Total RNA was isolated from each sample using the E.Z.N.A®Plant RNA Kit (Omega, GA, USA). RNA integrity was evaluated by 1% (v/v) agarose gel electrophoresis, and RNA purity was quantified using an IMPLEN NanoPhotometer. Total RNA was used to synthesize cDNA by using HiScript Ⅲ RT SuperMix for qPCR (+gDNA wiper) (Vazyme, Nanjing, China). The quantitative RT-qPCR was carried out with the Roche Lightcyler® 480 instrument using SYBR Green chemistry. The housekeeping UPL and RPL genes were used as an internal control of different treatments and MeJA treatment with different concentrations, respectively [67,68]. The RT-qPCR program was set as follows: 95 °C for 30 s, followed by 40 cycles of 95 °C/10 s, 60 °C/30 s, and the fluorescence signal was then read. Subsequently, the procedure was conducted as follows: 95 °C for 15 s, 60 °C for 60 s, and warming up to 95 °C then 50 °C for 30 s; after the fluorescence signal was read, the dissolution curve was analyzed. There were three biological replicates for each sample, and each biological replicate had three technical repetitions. Sequences of the primers used in this study were shown in detail in Supplementary File S7. The data obtained were visualized using SigmaPlot.
4.10. Extraction and Quantitative Determination of Tryptamine and Cadambine
The samples were ground at −80 °C using liquid nitrogen. We weighed 0.12 g of sample and added 5 mL of 70% ethanol, and the mixture was held overnight at −20 °C, followed by ultrasonic crushing for 30 min, vibrating every 5 min. After ultrasonic crushing, and centrifugation at 4 °C for 10 min at 7000 rpm, samples were filtered through 0.22 μm membrane filters before liquid chromatography–mass spectrometry (LC-MS) analysis.
Samples were analyzed by reversed-phase chromatography on an Agilent 1290 HPLC, using a 3.0 × 50 mm ECLIPS PLUS C18 column. Water with 0.1% formic acid (A) and methanol (B) was used as the mobile phase component at a flow rate of 0.3 mL/min with the following gradient: 0–3 min, 50% B; 3–6 min, 90% B; 5–6 min, 90% B; 6–10 min, 10% B. A coupled Agilent 6470 MS-QQQ mass spectrometer with ESI and Agilent Jet Stream was used to collect MS data in positive ion mode (parameters: Gas: 300 °C, 8 L/min; Nebulizer: 45 psi; Sheath Gas: 350 °C, 10 L/min; Capillary: 4000 V; VCharging: 1000). Scan type was MRM.
4.11. Statistical Analysis
Data from three biological and three technical replicates were used for statistical analysis. All data were indicated by an average of three biological replicate measurements and standard deviation. As for RT-qPCR, the relative gene expression levels were analyzed using the 2−ΔΔCt method. Significance was determined by pairwise comparison using t-tests or multiple comparison using Duncan’s multiple range test in SPSS software 26 (p = 0.05).
5. Conclusions
Overall, a total of 85 WRKY genes were identified from N. cadamba and divided into seven subfamilies according to their phylogenetic relationships, and members of the same subfamily had similar gene structures and conserved motifs and domains. The analyses of expression patterns based on RNA-seq data revealed their probable functions in different tissues and vascular development. In addition, the detection of remarkable expression profiles of NcWRKYs under hormones and abiotic stresses will provide clues for exploring the signaling pathways in N. cadambia in response to hormone and abiotic stresses. It is worth noting that MeJA promoted cadambine accumulation and induced the expression of the key intermediate gene STR1, NcWRKY64, and NcWRKY74, implying the MeJA-WRKY-STR regulatory module in the cadambine biosynthesis. This study suggests a basis for further functional research of the regulatory mechanism of NcWRKYs in hormone and stress responses and provides promising candidate genes for regulating cadambine synthesis in N. cadamba.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/ijms24087537/s1.
Author Contributions
C.P. and J.L. conceived the project; Z.X., Y.L. and H.F. performed the experiments and analyzed the data; Y.W. (Yanqiong Wen), Y.W. (Ying Wang) and J.Z. helped to prepare the plant materials and extracted metabolites; Z.X., J.L. and C.P. wrote and revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by funds from the National Natural Science Foundation of China (Grant No. 32271908); Natural Science Foundation of Guangdong Province of China (Grant Nos 2021A1515010816, 2023A1515030250, and 2023A1515010341); and Guangzhou Science and Technology Program (Grant No. 202102020505).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are provided within the article and Supplementary Materials.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ulker, B.; Somssich, I.E. WRKY transcription factors: From DNA binding towards biological function. Curr. Opin. Plant Biol. 2004, 7, 491–498. [Google Scholar] [CrossRef]
- Brand, L.H.; Fischer, N.M.; Harter, K.; Kohlbacher, O.; Wanke, D. Elucidating the evolutionary conserved DNA-binding specificities of WRKY transcription factors by molecular dynamics and in vitro binding assays. Nucleic Acids Res. 2013, 41, 9764–9778. [Google Scholar] [CrossRef] [PubMed]
- Eulgem, T.; Rushton, P.J.; Robatzek, S.; Somssich, I.E. The WRKY superfamily of plant transcription factors. Trends Plant Sci. 2000, 5, 199–206. [Google Scholar] [CrossRef]
- Jiang, J.J.; Ma, S.H.; Ye, N.H.; Jinag, M.; Cao, J.S.; Zhang, J.H. WRKY transcription factors in plant responses to stresses. J. Integr. Plant Biol. 2017, 59, 86–101. [Google Scholar] [CrossRef]
- Rushton, P.J.; Somssich, I.E.; Ringler, P.; Shen, Q.J. WRKY transcription factors. Trends Plant Sci. 2010, 15, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Zhang, Z.L.; Zou, X.; Yang, G.; Komatsu, S.; Shen, Q.J. Interactions of two abscisic-acid induced WRKY genes in repressing gibberellin signaling in aleurone cells. Plant J. 2006, 46, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.H.; Kwon, S.I.; Jang, J.Y.; Fang, I.L.; Lee, H.; Choi, C.; Park, S.; Ahn, I.; Bae, S.C.; Hwang, D.J. OsWRKY51, a rice transcription factor, functions as a positive regulator in defense response against Xanthomonas oryzae pv. oryzae. Plant Cell Rep. 2016, 35, 1975–1985. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lai, Z.; Shi, J.; Xiao, Y.; Chen, Z.; Xu, X. Roles of Arabidopsis WRKY18, WRKY40 and WRKY60 transcription factors in plant responses to abscisic acid and abiotic stress. BMC Plant Biol. 2010, 10, 281. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.X.; Ma, K.B.; Lu, Z.G.; Chen, G.; Cui, J.W.; Tong, X.P.; Wang, L.; Teng, N.J.; Jin, B. Transcriptomic and metabolomic analysis of the heat-stress response of Populus tomentosa Carr. Forests 2019, 10, 383. [Google Scholar] [CrossRef]
- Wu, M.; Zhang, K.; Xu, Y.; Wang, L.; Liu, H.; Qin, Z.; Xiang, Y. The moso bamboo WRKY transcription factor, PheWRKY86, regulates drought tolerance in transgenic plants. Plant Physiol. Biochem. 2022, 170, 180–191. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.P.; Ju, H.L.; Jae, H.Y.; Byeong, C.M.; Man, S.C.; Yun, H.K.; Sang, M.L.; Ho, S.K.; Kyu, Y.K.; Woo, S.C.; et al. WRKY group IId transcription factors interact with calmodulin. FEBS Lett. 2005, 579, 1545–1550. [Google Scholar] [CrossRef]
- Li, S.; Fu, Q.; Chen, L.; Huang, W.; Yu, D. Arabidopsis thaliana WRKY25, WRKY26, and WRKY33 coordinate induction of plant thermotolerance. Planta 2011, 233, 1237–1252. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Wu, Y.; Wang, K.; Bai, Z.; Liu, Q.; Pan, Y.; Zhang, L.; Jiang, B. Chrysanthemum WRKY gene DgWRKY5 enhances tolerance to salt stress in transgenic chrysanthemum. Sci. Rep. 2017, 7, 4799. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Li, C.; He, X.; Zhang, X.; Zhu, L. ABA signaling is negatively regulated by GbWRKY1 through JAZ1 and ABI1 to affect salt and drought tolerance. Plant Cell Rep. 2020, 39, 181–194. [Google Scholar] [CrossRef]
- Yong, F.G.; Ji, K.L.; Feng, M.Y.; Guo, Y.Z.; Dan, W.; Lin, Z.; Yong, B.O.; Yin, A.Y. The WRKY transcription factor WRKY8 promotes resistance to pathogen infection and mediates drought and salt stress tolerance in Solanum lycopersicum. Physiol. Plant. 2020, 168, 98–117. [Google Scholar] [CrossRef]
- Hao, X.; Xie, C.; Ruan, Q.; Zhang, X.; Wu, C.; Han, B.; Qian, J.; Zhou, W.; Nutzmann, H.W.; Kai, G. The transcription factor OpWRKY2 positively regulates the biosynthesis of the anticancer drug camptothecin in Ophiorrhiza pumila. Hortic. Res. 2021, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Can, W.; Chao, W.; Yao, W.; Chenhong, X.; Min, S.; Shivraj, N.; Zhigang, Z.; Guoyin, K. Transcription factor OpWRKY3 is involved in the development and biosynthesis of camptothecin and its precursors in Ophiorrhiza pumila hairy roots. Int. J. Mol. Sci. 2019, 20, 3996. [Google Scholar] [CrossRef]
- Wang, C.; Hao, X.; Wang, Y.; Maoz, I.; Zhou, W.; Zhou, Z.; Kai, G. Identification of WRKY transcription factors involved in regulating the biosynthesis of the anti-cancer drug camptothecin in Ophiorrhiza pumila. Hortic. Res. 2022, 9, uhac099. [Google Scholar] [CrossRef]
- Sun, P.W.; Xu, Y.H.; Yu, C.C.; Lv, F.F.; Tang, X.L.; Gao, Z.H.; Zhang, Z.; Wang, H.; Liu, Y.; Wei, J.H. WRKY44 represses expression of the wound-induced sesquiterpene biosynthetic gene ASS1 in Aquilaria sinensis. J. Exp. Bot. 2020, 71, 1128–1138. [Google Scholar] [CrossRef]
- Xu, Y.H.; Wang, J.W.; Wang, S.; Wang, J.Y.; Chen, X.Y. Characterization of GaWRKY1, a cotton transcription factor that regulates the sesquiterpene synthase gene (+)-delta-cadinene synthase-A. Plant Physiol. 2004, 135, 507–515. [Google Scholar] [CrossRef]
- Ma, D.; Pu, G.; Lei, C.; Ma, L.; Wang, H.; Guo, Y.; Chen, J.; Du, Z.; Wang, H.; Li, G.; et al. Isolation and characterization of AaWRKY1, an Artemisia annua transcription factor that regulates the amorpha-4,11-diene synthase gene, a key gene of artemisinin biosynthesis. Plant Cell Physiol. 2009, 50, 2146–2161. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Juan, W.; Jiachen, S.; Junping, H.; Kee-Yoeup, P.; So-Young, P.; Luqi, H.; Wenyuan, G. A WRKY transcription factor, PgWRKY4X, positively regulates ginsenoside biosynthesis by activating squalene epoxidase transcription in Panax ginseng. Ind. Crops Prod. 2020, 154, 112671. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, Y.; Nie, L.; Jin, X.; Liao, W.; Zhao, S.; Fu, C.; Yu, L. Transcriptome-wide identification and screening of WRKY factors involved in the regulation of taxol biosynthesis in Taxus chinensis. Sci. Rep. 2018, 8, 5197. [Google Scholar] [CrossRef] [PubMed]
- Suttipanta, N.; Pattanaik, S.; Kulshrestha, M.; Patra, B.; Singh, S.K.; Yuan, L. The transcription factor CrWRKY1 positively regulates the terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Plant Physiol. 2011, 157, 2081–2093. [Google Scholar] [CrossRef]
- Pandey, A.; Negi, P.S. Traditional uses, phytochemistry and pharmacological properties of Neolamarckia cadamba: A review. J. Ethnopharmacol. 2016, 181, 118–135. [Google Scholar] [CrossRef] [PubMed]
- Isao, K.; Hong, W.; Sanae, N.; Taifo, M.; Kazuyuki, H.; Motomasa, K.; Tahan, U.; Hirotaka, S. Indonesian Medicinal Plants. XIV. Characterization of 3′-0-Caffeoylswerosicle, a new secoiridoid glucoside, and kelampayosides A and B, two new phenolic apioglucosides, from the bark of Anthocephalus chinensis (Rubiaceae). Chem. Pharm. Bull. 1996, 44, 1162–1167. [Google Scholar] [CrossRef]
- Ashish, K.; Somenath, R.C.; Kumar, K.J.; Tulika, C.; Hemanta, K.M.; Tarun, J.; Sibabrata, M. Anthocephaline, a new indole alkaloid and cadambine, a potent inhibitor of DNA topoisomerase IB of leishmania donovani (LdTOP1LS), isolated from Anthocephalus cadamba. Nat. Prod. Commun. 2015, 10, 297–299. [Google Scholar] [CrossRef]
- Deepak, K.; Chilukuri, T.; Saiprasanna, R.; Sumana, M.; Bikas, C.P. Bio-assay guided isolation of anti-cancer compounds from Anthocephalus cadamba bark. Nat. Prod. Commun. 2015, 10, 1349–1350. [Google Scholar] [CrossRef]
- Zhao, X.; Hu, X.; OuYang, K.; Yang, J.; Que, Q.; Long, J.; Zhang, J.; Zhang, T.; Wang, X.; Gao, J.; et al. Chromosome-level assembly of the Neolamarckia cadamba genome provides insights into the evolution of cadambine biosynthesis. Plant J. 2022, 109, 891–908. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Wang, C.; Wang, H.; Li, L.; Wang, C. The function of MAPK cascades in response to various stresses in horticultural Plants. Front. Plant Sci. 2020, 11, 952. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Zhang, S. MAPK cascades in plant disease resistance signaling. Annu. Rev. Phytopathol. 2013, 51, 245–266. [Google Scholar] [CrossRef] [PubMed]
- Pitzschke, A.; Schikora, A.; Hirt, H. MAPK cascade signalling networks in plant defence. Curr. Opin. Plant Biol. 2009, 12, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, P.; Vishal, B.; Ho, W.J.; Lok, F.; Lee, F.; Kumar, P.P. Regulation of a cytochrome P450 gene CYP94B1 by WRKY33 transcription factor controls apoplastic barrier formation in roots to confer salt tolerance. Plant Physiol. 2020, 184, 2199–2215. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.; Meng, X.; Liu, Y.; Zheng, Z.; Chen, Z.; Zhang, S. Phosphorylation of a WRKY transcription factor by two pathogen-responsive MAPKs drives phytoalexin biosynthesis in Arabidopsis. Plant Cell 2011, 23, 1639–1653. [Google Scholar] [CrossRef]
- Hu, P.; Zhou, W.; Cheng, Z.; Fan, M.; Wang, L.; Xie, D. JAV1 controls jasmonate-regulated plant defense. Mol. Cell 2013, 50, 504–515. [Google Scholar] [CrossRef]
- Wasternack, C. Action of jasmonates in plant stress responses and development—Applied aspects. Biotechnol. Adv. 2014, 32, 31–39. [Google Scholar] [CrossRef]
- Tang, Y.; Guo, J.; Zhang, T.; Bai, S.; He, K.; Wang, Z. Genome-wide analysis of WRKY gene family and the dynamic responses of key WRKY genes involved in Ostrinia furnacalis attack in Zea mays. Int. J. Mol. Sci. 2021, 22, 13045. [Google Scholar] [CrossRef]
- Huang, S.; Gao, Y.; Liu, J.; Peng, X.; Niu, X.; Fei, Z.; Cao, S.; Liu, Y. Genome-wide analysis of WRKY transcription factors in Solanum lycopersicum. Mol. Genet. Genom. 2012, 287, 495–513. [Google Scholar] [CrossRef]
- Di, P.; Wang, P.; Yan, M.; Han, P.; Huang, X.; Yin, L.; Yan, Y.; Xu, Y.; Wang, Y. Genome-wide characterization and analysis of WRKY transcription factors in Panax ginseng. BMC Genom. 2021, 22, 834. [Google Scholar] [CrossRef]
- Jiang, Y.; Duan, Y.; Yin, J.; Ye, S.; Zhu, J.; Zhang, F.; Lu, W.; Fan, D.; Luo, K. Genome-wide identification and characterization of the Populus WRKY transcription factor family and analysis of their expression in response to biotic and abiotic stresses. J. Exp. Bot. 2014, 65, 6629–6644. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, X.; Xue, J.; Meng, L.; Li, R. Identification and expression analysis of WRKY transcription factors in medicinal plant Catharanthus roseus. Sheng Wu Gong Cheng Xue Bao Chin. J. Biotechnol. 2013, 29, 785–802. [Google Scholar]
- Baillo, E.H.; Kimotho, R.N.; Zhang, Z.; Xu, P. Transcription factors associated with abiotic and biotic stress tolerance and their potential for crops improvement. Genes 2019, 10, 771. [Google Scholar] [CrossRef] [PubMed]
- Van Verk, M.C.; Pappaioannou, D.; Neeleman, L.; Bol, J.F.; Linthorst, H.J. A novel WRKY transcription factor is required for induction of PR-1a gene expression by salicylic acid and bacterial elicitors. Plant Physiol. 2008, 146, 1983–1995. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Chen, C.; Li, C.; Liu, J.; Liu, C.; He, Y. Genome-wide investigation of WRKY gene family in pineapple: Evolution and expression profiles during development and stress. BMC Genom. 2018, 19, 490. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, X.; Han, J.; Lu, W.; Ren, Z. Genome-wide analysis of the WRKY gene family in the cucumber genome and transcriptome-wide identification of WRKY transcription factors that respond to biotic and abiotic stresses. BMC Plant Biol. 2020, 20, 443. [Google Scholar] [CrossRef]
- He, H.; Dong, Q.; Shao, Y.; Jiang, H.; Zhu, S.; Cheng, B.; Xiang, Y. Genome-wide survey and characterization of the WRKY gene family in Populus trichocarpa. Plant Cell Rep. 2012, 31, 1199–1217. [Google Scholar] [CrossRef]
- Yu, Y.; Hu, R.; Wang, H.; Cao, Y.; He, G.; Fu, C.; Zhou, G. MlWRKY12, a novel Miscanthus transcription factor, participates in pith secondary cell wall formation and promotes flowering. Plant Sci. 2013, 212, 1–9. [Google Scholar] [CrossRef]
- Yang, L.; Zhao, X.; Yang, F.; Fan, D.; Jiang, Y.; Luo, K. PtrWRKY19, a novel WRKY transcription factor, contributes to the regulation of pith secondary wall formation in Populus trichocarpa. Sci. Rep. 2016, 6, 18643. [Google Scholar] [CrossRef]
- Li, W.; Tian, Z.; Yu, D. WRKY13 acts in stem development in Arabidopsis thaliana. Plant Sci. 2015, 236, 205–213. [Google Scholar] [CrossRef]
- Wei, Y.; Jin, J.; Liang, D.; Gao, J.; Li, J.; Xie, Q.; Lu, C.; Yang, F.; Zhu, G. Genome-wide identification of Cymbidium sinense WRKY gene family and the importance of its Group III members in response to abiotic stress. Front. Plant Sci. 2022, 13, 969010. [Google Scholar] [CrossRef]
- Li, S.; Zhou, X.; Chen, L.; Huang, W.; Yu, D. Functional characterization of Arabidopsis thaliana WRKY39 in heat stress. Mol. Cells 2010, 29, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Rushton, D.L.; Tripathi, P.; Rabara, R.C.; Lin, J.; Ringler, P.; Boken, A.K.; Langum, T.J.; Smidt, L.; Boomsma, D.D.; Emme, N.J.; et al. WRKY transcription factors: Key components in abscisic acid signalling. Plant Biotechnol. J. 2012, 10, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Deyholos, M.K. Functional characterization of Arabidopsis NaCl-inducible WRKY25 and WRKY33 transcription factors in abiotic stresses. Plant Mol. Biol. 2009, 69, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Yu, D. Expression profiles of AtWRKY25, AtWRKY26 and AtWRKY33 under abiotic stresses. Heredity 2010, 32, 848–856. [Google Scholar] [CrossRef]
- Van der Fits, L.; Memelink, J. The jasmonate-inducible AP2/ERF-domain transcription factor ORCA3 activates gene expression via interaction with a jasmonate-responsive promoter element. Plant J. 2001, 25, 43–53. [Google Scholar] [CrossRef]
- De Geyter, N.; Gholami, A.; Goormachtig, S.; Goossens, A. Transcriptional machineries in jasmonate-elicited plant secondary metabolism. Trends Plant Sci. 2012, 17, 349–359. [Google Scholar] [CrossRef]
- Sibéril, Y.; Benhamron, S.; Memelink, J.; Giglioli-Guivarc’H, N.; Thiersault, M.; Boisson, B.; Doireau, P.; Gantet, P. Catharanthus roseus G-box binding factors 1 and 2 act as repressors of strictosidine synthase gene expression in cell cultures. Plant Mol. Biol. 2001, 45, 477–488. [Google Scholar] [CrossRef]
- Chini, A.; Boter, M.; Solano, R. Plant oxylipins: COI1/JAZs/MYC2 as the core jasmonic acid-signalling module. FEBS J. 2009, 276, 4682–4692. [Google Scholar] [CrossRef]
- Skibbe, M.; Qu, N.; Galis, I.; Baldwin, I.T. Induced plant defenses in the natural environment: Nicotiana attenuata WRKY3 and WRKY6 coordinate responses to herbivory. Plant Cell 2008, 20, 1984–2000. [Google Scholar] [CrossRef]
- Duan, S.; Wang, J.; Gao, C.; Jin, C.; Li, D.; Peng, D.; Du, G.; Li, Y.; Chen, M. Functional characterization of a heterologously expressed Brassica napus WRKY41-1 transcription factor in regulating anthocyanin biosynthesis in Arabidopsis thaliana. Plant Sci. 2018, 268, 47–53. [Google Scholar] [CrossRef]
- Kishi-Kaboshi, M.; Takahashi, A.; Hirochika, H. MAMP-responsive MAPK cascades regulate phytoalexin biosynthesis. Plant Signal. Behav. 2010, 5, 1653–1656. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.L.; Fiil, B.K.; Petersen, K.; Nielsen, H.B.; Botanga, C.J.; Thorgrimsen, S.; Palma, K.; Suarez-Rodriguez, M.C.; Sandbech-Clausen, S.; Lichota, J.; et al. Arabidopsis MAP kinase 4 regulates gene expression through transcription factor release in the nucleus. EMBO J. 2008, 27, 2214–2221. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Rui, X. TBtools: An integrative toolkit developed for interactive analyses of big biological data. Mol. Plant. 2020, 13, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef]
- Higo, K.; Ugawa, Y.; Iwamoto, M.; Korenaga, T. Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res. 1999, 27, 297–300. [Google Scholar] [CrossRef]
- Wang, X.; Long, J.; Dong, T.; Zheng, D.; Zhang, L.; Peng, C. Establishment of vascular tissue cells capture system by laser microdissection in Neolamarckia cadamba. Guihaia 2021, 41, 1226–1236, (In Chinese with English abstract). [Google Scholar]
- Huang, T.; Long, J.; Liu, S.W.; Yang, Z.; Zhu, Q.; Zhao, X.; Peng, C. Selection and validation of reference genes for mRNA expression by quantitative Real-Time PCR analysis in Neolamarckia cadamba. Sci. Rep. 2018, 8, 9931. [Google Scholar] [CrossRef]
- Zhang, D.; Li, J.J.; Zhang, M.J.; Bao, Y.T.; Yang, X.; Xu, W.Y.; Quyang, K.X.; Chen, X.Y. Selection and validation of reference genes for quantitative RT-PCR analysis in Neolamarckia cadamba. Chin. Bull. Bot. 2018, 53, 829–839. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).