Translational Relevance of Secondary Intracellular Signaling Cascades Following Traumatic Spinal Cord Injury
Abstract
1. Introduction
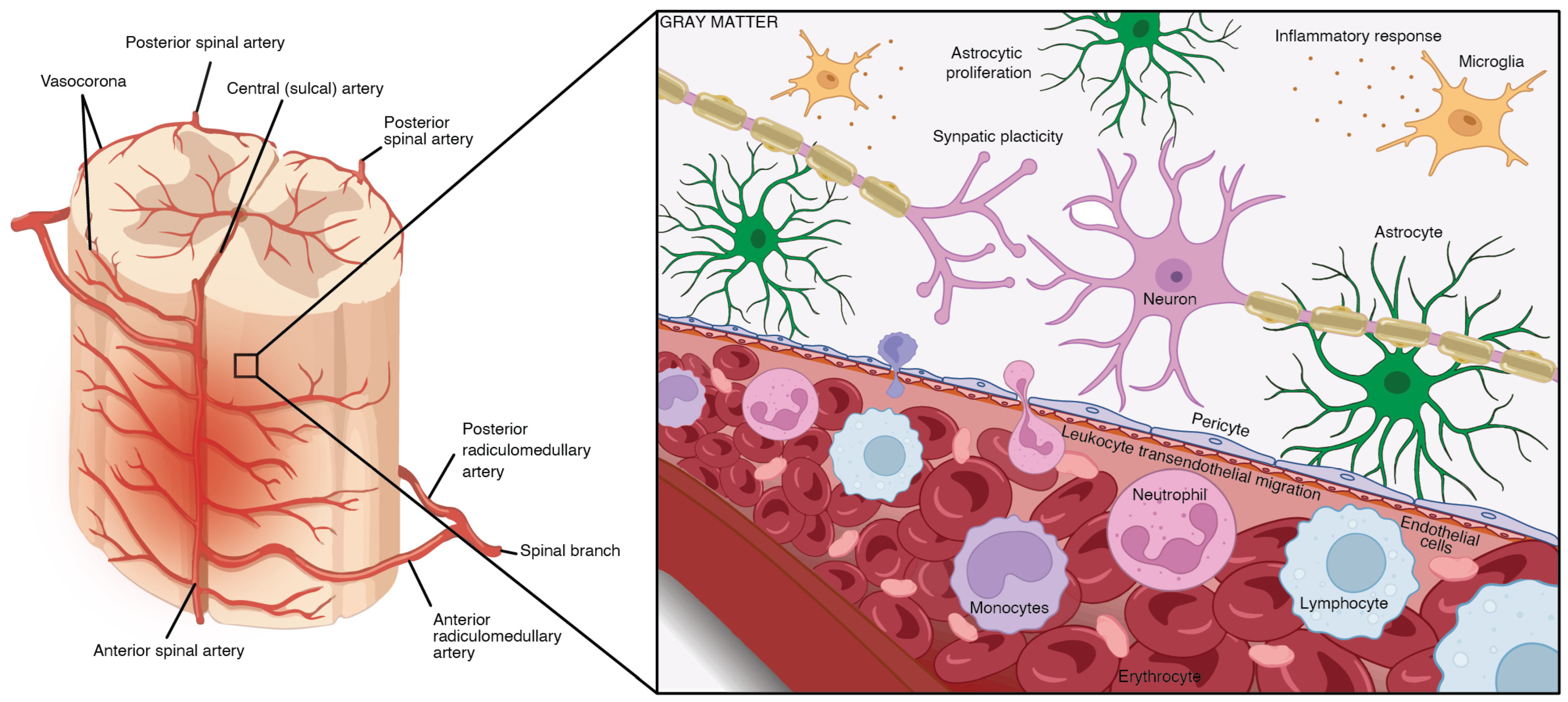
2. Secondary SCI Pathobiology
3. MAPK Signaling Network
3.1. ERK1/2 Pathway
3.2. SAPK/JNK Pathway
3.3. p38 Pathway
4. PI3K-AKT-mTOR Network
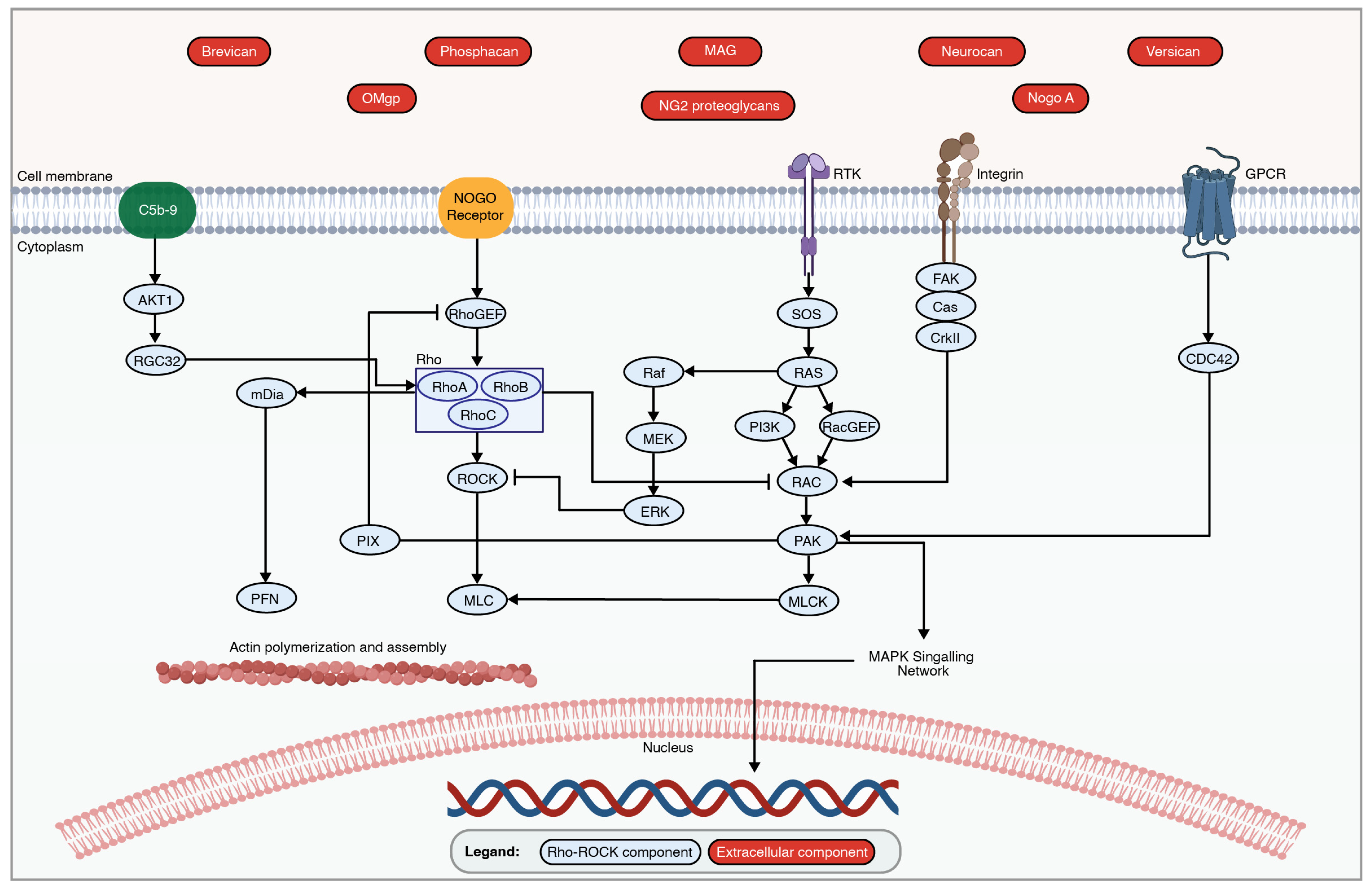
5. Rho-ROCK Pathway
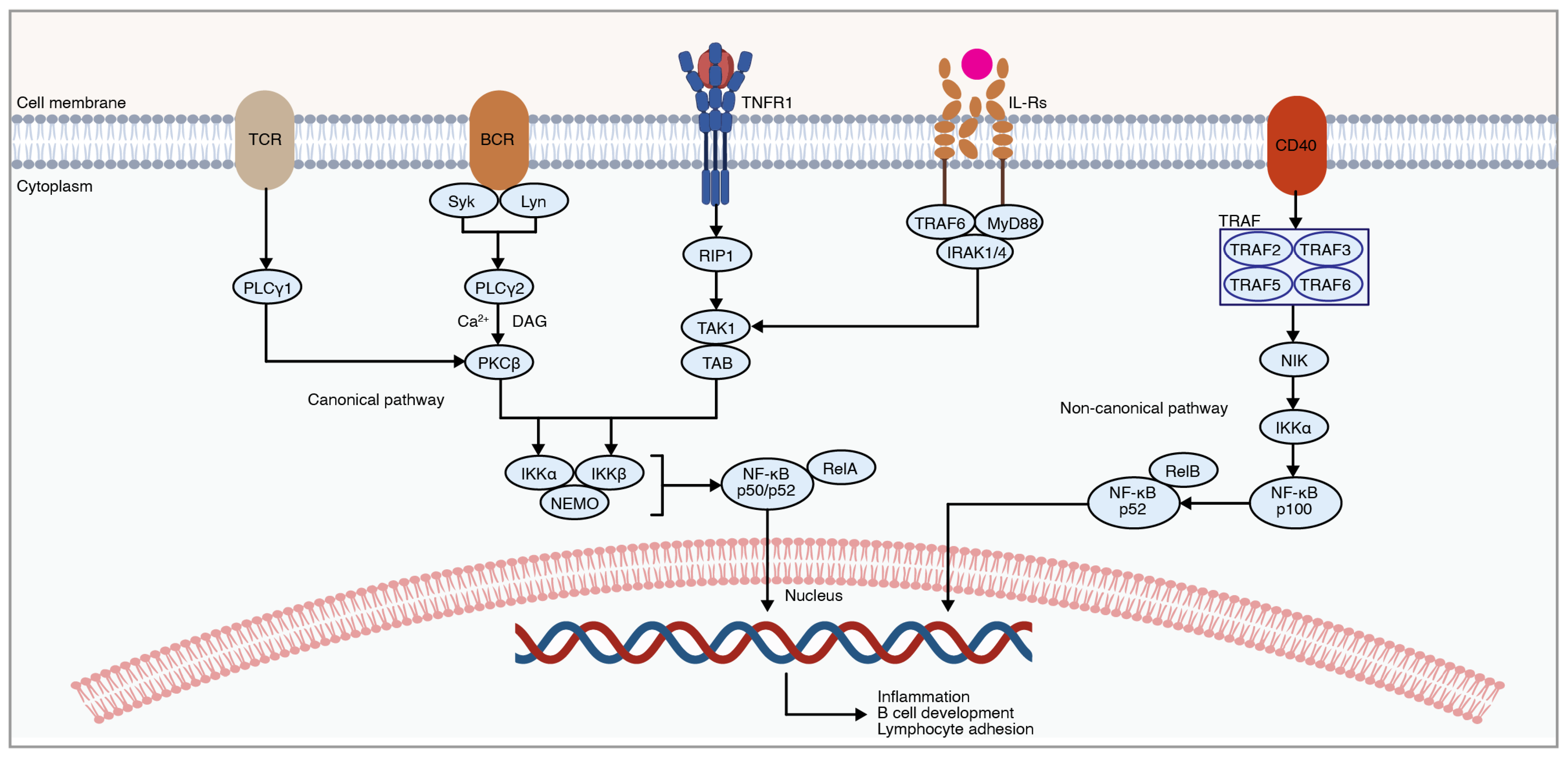
6. NF-κB Pathway
7. JAK-STAT Pathway

8. Translational Implications and Future Directions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baroudi, M.; Rezk, A.; Daher, M.; Balmaceno-Criss, M.; Gregoryczyk, J.G.; Sharma, Y.; McDonald, C.L.; Diebo, B.G.; Daniels, A.H. Management of Traumatic Spinal Cord Injury: A Current Concepts Review of Contemporary and Future Treatment. Injury 2024, 55, 111472. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Tetreault, L.; Kalsi-Ryan, S.; Nouri, A.; Fehlings, M. Global Prevalence and Incidence of Traumatic Spinal Cord Injury. Clin. Epidemiol. 2014, 6, 309–331. [Google Scholar] [CrossRef] [PubMed]
- National Spinal Cord Injury Statistical Center. Spinal Cord Injury Facts and Figures at a Glance; University of Alabama at Birmingham: Birmingham, AL, USA, 2021. [Google Scholar]
- Burnside, E.R.; De Winter, F.; Didangelos, A.; James, N.D.; Andreica, E.-C.; Layard-Horsfall, H.; Muir, E.M.; Verhaagen, J.; Bradbury, E.J. Immune-Evasive Gene Switch Enables Regulated Delivery of Chondroitinase after Spinal Cord Injury. Brain 2018, 141, 2362–2381. [Google Scholar] [CrossRef] [PubMed]
- Zavvarian, M.-M.; Hong, J.; Fehlings, M.G. The Functional Role of Spinal Interneurons Following Traumatic Spinal Cord Injury. Front. Cell. Neurosci. 2020, 14, 127. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.; Matson, K.J.E.; Li, L.; Hua, I.; Petrescu, J.; Kang, K.; Alkaslasi, M.R.; Lee, D.I.; Hasan, S.; Galuta, A.; et al. A Cellular Taxonomy of the Adult Human Spinal Cord. Neuron 2023, 111, 328–344.e7. [Google Scholar] [CrossRef] [PubMed]
- Sathyamurthy, A.; Johnson, K.R.; Matson, K.J.E.; Dobrott, C.I.; Li, L.; Ryba, A.R.; Bergman, T.B.; Kelly, M.C.; Kelley, M.W.; Levine, A.J. Massively Parallel Single Nucleus Transcriptional Profiling Defines Spinal Cord Neurons and Their Activity during Behavior. Cell Rep. 2018, 22, 2216–2225. [Google Scholar] [CrossRef] [PubMed]
- Vanlandewijck, M.; He, L.; Mäe, M.A.; Andrae, J.; Ando, K.; Del Gaudio, F.; Nahar, K.; Lebouvier, T.; Laviña, B.; Gouveia, L.; et al. A Molecular Atlas of Cell Types and Zonation in the Brain Vasculature. Nature 2018, 554, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hammel, G.; Shi, M.; Cheng, Z.; Zivkovic, S.; Wang, X.; Xu, P.; He, X.; Guo, B.; Ren, Y.; et al. Myelin Debris Stimulates NG2/CSPG4 Expression in Bone Marrow-Derived Macrophages in the Injured Spinal Cord. Front. Cell. Neurosci. 2021, 15, 651827. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cao, K.; Sun, X.; Chen, Y.; Duan, Z.; Sun, L.; Guo, L.; Bai, P.; Sun, D.; Fan, J.; et al. Macrophages in Spinal Cord Injury: Phenotypic and Functional Change from Exposure to Myelin Debris: Myelin Debris Altering Macrophage Function. Glia 2015, 63, 635–651. [Google Scholar] [CrossRef]
- Mautes, A.E.; Weinzierl, M.R.; Donovan, F.; Noble, L.J. Vascular Events After Spinal Cord Injury: Contribution to Secondary Pathogenesis. Phys. Ther. 2000, 80, 673–687. [Google Scholar] [CrossRef]
- Figley, S.A.; Khosravi, R.; Legasto, J.M.; Tseng, Y.-F.; Fehlings, M.G. Characterization of Vascular Disruption and Blood–Spinal Cord Barrier Permeability Following Traumatic Spinal Cord Injury. J. Neurotrauma 2014, 31, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.C.; Norenberg, M.D.; Ramsay, D.A.; Dekaban, G.A.; Marcillo, A.E.; Saenz, A.D.; Pasquale-Styles, M.; Dietrich, W.D.; Weaver, L.C. The Cellular Inflammatory Response in Human Spinal Cords after Injury. Brain 2006, 129, 3249–3269. [Google Scholar] [CrossRef] [PubMed]
- Chio, J.C.T.; Wang, J.; Badner, A.; Hong, J.; Surendran, V.; Fehlings, M.G. The Effects of Human Immunoglobulin G on Enhancing Tissue Protection and Neurobehavioral Recovery after Traumatic Cervical Spinal Cord Injury Are Mediated through the Neurovascular Unit. J. Neuroinflammation 2019, 16, 141. [Google Scholar] [CrossRef] [PubMed]
- Chio, J.C.T.; Wang, J.; Surendran, V.; Li, L.; Zavvarian, M.-M.; Pieczonka, K.; Fehlings, M.G. Delayed Administration of High Dose Human Immunoglobulin G Enhances Recovery after Traumatic Cervical Spinal Cord Injury by Modulation of Neuroinflammation and Protection of the Blood Spinal Cord Barrier. Neurobiol. Dis. 2021, 148, 105187. [Google Scholar] [CrossRef] [PubMed]
- Chio, J.C.T.; Xu, K.J.; Popovich, P.; David, S.; Fehlings, M.G. Neuroimmunological Therapies for Treating Spinal Cord Injury: Evidence and Future Perspectives. Exp. Neurol. 2021, 341, 113704. [Google Scholar] [CrossRef] [PubMed]
- Ulndreaj, A.; Tzekou, A.; Mothe, A.J.; Siddiqui, A.M.; Dragas, R.; Tator, C.H.; Torlakovic, E.E.; Fehlings, M.G. Characterization of the Antibody Response after Cervical Spinal Cord Injury. J. Neurotrauma 2017, 34, 1209–1226. [Google Scholar] [CrossRef] [PubMed]
- Zavvarian, M.-M.; Hong, J.; Chio, J.C.T.; Toossi, A.; Fehlings, M.G. Neurovascular Pathology Following Traumatic Spinal Cord Injury. In Cellular, Molecular, Physiological, and Behavioral Aspects of Spinal Cord Injury; Elsevier: Amsterdam, The Netherlands, 2022; pp. 119–132. ISBN 978-0-12-822427-4. [Google Scholar]
- Patel, F.; Mehdizadeh, C.; Amatya, P.; Bhakta, P.; Torrez Andia, E. A Case of Sudden-Onset Flaccid Paralysis In a Previously Healthy Person. Cureus 2023, 15, e37906. [Google Scholar] [CrossRef]
- Sharif, H.; Hou, S. Autonomic Dysreflexia: A Cardiovascular Disorder Following Spinal Cord Injury. Neural Regen. Res. 2017, 12, 1390–1400. [Google Scholar] [CrossRef]
- Guly, H.R.; Bouamra, O.; Lecky, F.E. The Incidence of Neurogenic Shock in Patients with Isolated Spinal Cord Injury in the Emergency Department. Resuscitation 2008, 76, 57–62. [Google Scholar] [CrossRef]
- Zavvarian, M.-M.; Zhou, C.; Kahnemuyipour, S.; Hong, J.; Fehlings, M.G. The MAPK Signaling Pathway Presents Novel Molecular Targets for Therapeutic Intervention after Traumatic Spinal Cord Injury: A Comparative Cross-Species Transcriptional Analysis. Int. J. Mol. Sci. 2021, 22, 12934. [Google Scholar] [CrossRef]
- Sinescu, C.; Popa, F.; Grigorean, V.T.; Onose, G.; Sandu, A.M.; Popescu, M.; Burnei, G.; Strambu, V.; Popa, C.; Davila, C.; et al. Molecular Basis of Vascular Events Following Spinal Cord Injury. J. Med. Life 2010, 3, 254–261. [Google Scholar] [PubMed]
- Fehlings, M.G.; Pedro, K.; Hejrati, N. Management of Acute Spinal Cord Injury: Where Have We Been? Where Are We Now? Where Are We Going? J. Neurotrauma 2022, 39, 1591–1602. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, C.S.; Wilson, J.R.; Nori, S.; Kotter, M.R.N.; Curt, A.; Fehlings, M.G. Traumatic Spinal Cord Injury. Nat. Rev. Primer 2017, 3, 17018. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, T.M.; Burda, J.E.; Sofroniew, M.V. Cell Biology of Spinal Cord Injury and Repair. J. Clin. Investig. 2017, 127, 3259–3270. [Google Scholar] [CrossRef]
- Freria, C.M.; Hall, J.C.E.; Wei, P.; Guan, Z.; McTigue, D.M.; Popovich, P.G. Deletion of the Fractalkine Receptor, CX3CR1, Improves Endogenous Repair, Axon Sprouting, and Synaptogenesis after Spinal Cord Injury in Mice. J. Neurosci. 2017, 37, 3568–3587. [Google Scholar] [CrossRef] [PubMed]
- Soderblom, C.; Luo, X.; Blumenthal, E.; Bray, E.; Lyapichev, K.; Ramos, J.; Krishnan, V.; Lai-Hsu, C.; Park, K.K.; Tsoulfas, P.; et al. Perivascular Fibroblasts Form the Fibrotic Scar after Contusive Spinal Cord Injury. J. Neurosci. 2013, 33, 13882–13887. [Google Scholar] [CrossRef] [PubMed]
- Almad, A.; Sahinkaya, F.R.; McTigue, D.M. Oligodendrocyte Fate after Spinal Cord Injury. Neurotherapeutics 2011, 8, 262–273. [Google Scholar] [CrossRef] [PubMed]
- Floriddia, E.M.; Lourenço, T.; Zhang, S.; Van Bruggen, D.; Hilscher, M.M.; Kukanja, P.; Gonçalves Dos Santos, J.P.; Altınkök, M.; Yokota, C.; Llorens-Bobadilla, E.; et al. Distinct Oligodendrocyte Populations Have Spatial Preference and Different Responses to Spinal Cord Injury. Nat. Commun. 2020, 11, 5860. [Google Scholar] [CrossRef] [PubMed]
- Dias, D.O.; Kim, H.; Holl, D.; Werne Solnestam, B.; Lundeberg, J.; Carlén, M.; Göritz, C.; Frisén, J. Reducing Pericyte-Derived Scarring Promotes Recovery after Spinal Cord Injury. Cell 2018, 173, 153–165.e22. [Google Scholar] [CrossRef]
- Dias, D.O.; Kalkitsas, J.; Kelahmetoglu, Y.; Estrada, C.P.; Tatarishvili, J.; Holl, D.; Jansson, L.; Banitalebi, S.; Amiry-Moghaddam, M.; Ernst, A.; et al. Pericyte-Derived Fibrotic Scarring Is Conserved across Diverse Central Nervous System Lesions. Nat. Commun. 2021, 12, 5501. [Google Scholar] [CrossRef]
- Donnelly, D.J.; Popovich, P.G. Inflammation and Its Role in Neuroprotection, Axonal Regeneration and Functional Recovery after Spinal Cord Injury. Exp. Neurol. 2008, 209, 378–388. [Google Scholar] [CrossRef]
- Quadri, S.A.; Farooqui, M.; Ikram, A.; Zafar, A.; Khan, M.A.; Suriya, S.S.; Claus, C.F.; Fiani, B.; Rahman, M.; Ramachandran, A.; et al. Recent Update on Basic Mechanisms of Spinal Cord Injury. Neurosurg. Rev. 2020, 43, 425–441. [Google Scholar] [CrossRef]
- Hellenbrand, D.J.; Quinn, C.M.; Piper, Z.J.; Morehouse, C.N.; Fixel, J.A.; Hanna, A.S. Inflammation after Spinal Cord Injury: A Review of the Critical Timeline of Signaling Cues and Cellular Infiltration. J. Neuroinflammation 2021, 18, 284. [Google Scholar] [CrossRef]
- Bradbury, E.J.; Burnside, E.R. Moving beyond the Glial Scar for Spinal Cord Repair. Nat. Commun. 2019, 10, 3879. [Google Scholar] [CrossRef]
- Tran, A.P.; Warren, P.M.; Silver, J. New Insights into Glial Scar Formation after Spinal Cord Injury. Cell Tissue Res. 2022, 387, 319–336. [Google Scholar] [CrossRef]
- Anderson, M.A.; Burda, J.E.; Ren, Y.; Ao, Y.; O’Shea, T.M.; Kawaguchi, R.; Coppola, G.; Khakh, B.S.; Deming, T.J.; Sofroniew, M.V. Astrocyte Scar Formation Aids Central Nervous System Axon Regeneration. Nature 2016, 532, 195–200. [Google Scholar] [CrossRef]
- Escartin, C.; Galea, E.; Lakatos, A.; O’Callaghan, J.P.; Petzold, G.C.; Serrano-Pozo, A.; Steinhäuser, C.; Volterra, A.; Carmignoto, G.; Agarwal, A.; et al. Reactive Astrocyte Nomenclature, Definitions, and Future Directions. Nat. Neurosci. 2021, 24, 312–325. [Google Scholar] [CrossRef]
- Li, Z.; Yu, S.; Hu, X.; Li, Y.; You, X.; Tian, D.; Cheng, L.; Zheng, M.; Jing, J. Fibrotic Scar After Spinal Cord Injury: Crosstalk With Other Cells, Cellular Origin, Function, and Mechanism. Front. Cell. Neurosci. 2021, 15, 720938. [Google Scholar] [CrossRef]
- Milhorat, T.H.; Capocelli, A.L.; Anzil, A.P.; Kotzen, R.M.; Milhorat, R.H. Pathological Basis of Spinal Cord Cavitation in Syringomyelia: Analysis of 105 Autopsy Cases. J. Neurosurg. 1995, 82, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Cregg, J.M.; DePaul, M.A.; Filous, A.R.; Lang, B.T.; Tran, A.; Silver, J. Functional Regeneration beyond the Glial Scar. Exp. Neurol. 2014, 253, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Burda, J.E.; Sofroniew, M.V. Reactive Gliosis and the Multicellular Response to CNS Damage and Disease. Neuron 2014, 81, 229–248. [Google Scholar] [CrossRef]
- Santillan, A.; Nacarino, V.; Greenberg, E.; Riina, H.A.; Gobin, Y.P.; Patsalides, A. Vascular Anatomy of the Spinal Cord. J. NeuroIntervent. Surg. 2012, 4, 67–74. [Google Scholar] [CrossRef]
- Wagner, F.C.; Van Gilder, J.C.; Dohrmann, G.J. The Development of Intramedullary Cavitation Following Spinal Cord Injury: An Experimental Pathological Study. Spinal Cord 1977, 14, 245–250. [Google Scholar] [CrossRef]
- Wozniewicz, B.; Filipowicz, K.; Swiderska, S.K.; Deraka, K. Pathophysiological Mechanism of Traumatic Cavitation of the Spinal Cord. Spinal Cord 1983, 21, 312–317. [Google Scholar] [CrossRef]
- Cao, X.; Feng, S.; Fu, C.; Gao, K.; Guo, J.; Guo, X.; He, X.; Huang, Z.; Li, Z.; Liu, L.; et al. Repair, Protection and Regeneration of Spinal Cord Injury. Neural Regen. Res. 2015, 10, 1953. [Google Scholar] [CrossRef]
- Wang, Q.-J.; Zhang, A.L.; Kang, Z.-Q.; Zhang, Z.-T.; Wang, Y.-S. Exogenous IL-19 Mediates Downregulation of TGF-β through Erk and P38 Pathway to Inhibit Epidural Fibrosis. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 7184–7190. [Google Scholar] [CrossRef]
- Plikus, M.V.; Wang, X.; Sinha, S.; Forte, E.; Thompson, S.M.; Herzog, E.L.; Driskell, R.R.; Rosenthal, N.; Biernaskie, J.; Horsley, V. Fibroblasts: Origins, Definitions, and Functions in Health and Disease. Cell 2021, 184, 3852–3872. [Google Scholar] [CrossRef]
- Duscher, D.; Maan, Z.N.; Wong, V.W.; Rennert, R.C.; Januszyk, M.; Rodrigues, M.; Hu, M.; Whitmore, A.J.; Whittam, A.J.; Longaker, M.T.; et al. Mechanotransduction and Fibrosis. J. Biomech. 2014, 47, 1997–2005. [Google Scholar] [CrossRef]
- Tang, X.; Davies, J.E.; Davies, S.J.A. Changes in Distribution, Cell Associations, and Protein Expression Levels of NG2, Neurocan, Phosphacan, Brevican, Versican V2, and Tenascin-C During Acute to Chronic Maturation of Spinal Cord Scar Tissue. J. Neurosci. Res. 2003, 71, 427–444. [Google Scholar] [CrossRef] [PubMed]
- Khakh, B.S.; Sofroniew, M.V. Diversity of Astrocyte Functions and Phenotypes in Neural Circuits. Nat. Neurosci. 2015, 18, 942–952. [Google Scholar] [CrossRef] [PubMed]
- Gadea, A.; Schinelli, S.; Gallo, V. Endothelin-1 Regulates Astrocyte Proliferation and Reactive Gliosis via a JNK/c-Jun Signaling Pathway. J. Neurosci. 2008, 28, 2394–2408. [Google Scholar] [CrossRef] [PubMed]
- Neary, J.T.; Kang, Y.; Willoughby, K.A.; Ellis, E.F. Activation of Extracellular Signal-Regulated Kinase by Stretch-Induced Injury in Astrocytes Involves Extracellular ATP and P2 Purinergic Receptors. J. Neurosci. 2003, 23, 2348–2356. [Google Scholar] [CrossRef] [PubMed]
- Sahni, V.; Mukhopadhyay, A.; Tysseling, V.; Hebert, A.; Birch, D.; Mcguire, T.L.; Stupp, S.I.; Kessler, J.A. BMPR1a and BMPR1b Signaling Exert Opposing Effects on Gliosis after Spinal Cord Injury. J. Neurosci. 2010, 30, 1839–1855. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, H.; Sakimoto, S.; Nakao, K.; Sugishita, A.; Konno, M.; Iida, S.; Kusano, A.; Hashimoto, E.; Nakagawa, T.; Kaneko, S. Transient Receptor Potential Canonical 3 (TRPC3) Mediates Thrombin-Induced Astrocyte Activation and Upregulates Its Own Expression in Cortical Astrocytes. J. Neurosci. 2010, 30, 13116–13129. [Google Scholar] [CrossRef] [PubMed]
- Sirko, S.; Behrendt, G.; Johansson, P.A.; Tripathi, P.; Costa, M.R.; Bek, S.; Heinrich, C.; Tiedt, S.; Colak, D.; Dichgans, M.; et al. Reactive Glia in the Injured Brain Acquire Stem Cell Properties in Response to Sonic Hedgehog. Cell Stem Cell 2013, 12, 426–439. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Buse, M.; Busuioc, C.; Drula, R.; Gulei, D.; Raduly, L.; Rusu, A.; Irimie, A.; Atanasov, A.G.; Slaby, O.; et al. A Comprehensive Review on MAPK: A Promising Therapeutic Target in Cancer. Cancers 2019, 11, 1618. [Google Scholar] [CrossRef] [PubMed]
- Soares-Silva, M.; Diniz, F.F.; Gomes, G.N.; Bahia, D. The Mitogen-Activated Protein Kinase (MAPK) Pathway: Role in Immune Evasion by Trypanosomatids. Front. Microbiol. 2016, 7, 183. [Google Scholar] [CrossRef] [PubMed]
- Kasuya, Y.; Umezawa, H.; Hatano, M. Stress-Activated Protein Kinases in Spinal Cord Injury: Focus on Roles of P38. Int. J. Mol. Sci. 2018, 19, 867. [Google Scholar] [CrossRef]
- Lin, B.; Xu, Y.; Zhang, B.; He, Y.; Yan, Y.; He, M.-C. MEK Inhibition Reduces Glial Scar Formation and Promotes the Recovery of Sensorimotor Function in Rats Following Spinal Cord Injury. Exp. Ther. Med. 2014, 7, 66–72. [Google Scholar] [CrossRef]
- Kaminska, B. MAPK Signalling Pathways as Molecular Targets for Anti-Inflammatory Therapy—From Molecular Mechanisms to Therapeutic Benefits. Biochim. Biophys. Acta BBA Proteins Proteom. 2005, 1754, 253–262. [Google Scholar] [CrossRef]
- Li, D.; Liu, N.; Zhao, H.-H.; Zhang, X.; Kawano, H.; Liu, L.; Zhao, L.; Li, H.-P. Interactions between Sirt1 and MAPKs Regulate Astrocyte Activation Induced by Brain Injury in Vitro and in Vivo. J. Neuroinflammation 2017, 14, 67. [Google Scholar] [CrossRef]
- Chi, H.; Barry, S.P.; Roth, R.J.; Wu, J.J.; Jones, E.A.; Bennett, A.M.; Flavell, R.A. Dynamic Regulation of Pro- and Anti-Inflammatory Cytokines by MAPK Phosphatase 1 (MKP-1) in Innate Immune Responses. Proc. Natl. Acad. Sci. USA 2006, 103, 2274–2279. [Google Scholar] [CrossRef] [PubMed]
- Diem, R.; Hobom, M.; Maier, K.; Weissert, R.; Storch, M.K.; Meyer, R.; Bähr, M. Methylprednisolone Increases Neuronal Apoptosis during Autoimmune CNS Inflammation by Inhibition of an Endogenous Neuroprotective Pathway. J. Neurosci. 2003, 23, 6993–7000. [Google Scholar] [CrossRef] [PubMed]
- Kassel, O. Glucocorticoids Inhibit MAP Kinase via Increased Expression and Decreased Degradation of MKP-1. EMBO J. 2001, 20, 7108–7116. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.H.; Morand, E.; Leech, M. Annexin A1: Potential for Glucocorticoid Sparing in RA. Nat. Rev. Rheumatol. 2013, 9, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Fan, G.; Chen, S.; Wu, Y.; Xu, X.M.; Hsu, C.Y. Methylprednisolone Inhibition of TNF-a Expression and NF-kB Activation after Spinal Cord Injury in Rats. Mol. Brain Res. 1998, 59, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wang, Y.; Zhan, X. The MAPK Pathway-Based Drug Therapeutic Targets in Pituitary Adenomas. Front. Endocrinol. 2019, 10, 330. [Google Scholar] [CrossRef] [PubMed]
- Aoki, K.; Yamada, M.; Kunida, K.; Yasuda, S.; Matsuda, M. Processive Phosphorylation of ERK MAP Kinase in Mammalian Cells. Proc. Natl. Acad. Sci. USA 2011, 108, 12675–12680. [Google Scholar] [CrossRef] [PubMed]
- Manna, P.R.; Stocco, D.M. The Role of Specific Mitogen-Activated Protein Kinase Signaling Cascades in the Regulation of Steroidogenesis. J. Signal Transduct. 2011, 2011, 821615. [Google Scholar] [CrossRef]
- Broom, O.J.; Widjaya, B.; Troelsen, J.; Olsen, J.; Nielsen, O.H. Mitogen Activated Protein Kinases: A Role in Inflammatory Bowel Disease? Clin. Exp. Immunol. 2009, 158, 272–280. [Google Scholar] [CrossRef]
- Feng, J.; Lu, S.; Ou, B.; Liu, Q.; Dai, J.; Ji, C.; Zhou, H.; Huang, H.; Ma, Y. The Role of JNk Signaling Pathway in Obesity-Driven Insulin Resistance. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 1399–1406. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, M.C.; Er, E.E.; Blenis, J. The Ras-ERK and PI3K-mTOR Pathways: Cross-Talk and Compensation. Trends Biochem. Sci. 2011, 36, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.P.E. The Interactions of Flavonoids within Neuronal Signalling Pathways. Genes Nutr. 2007, 2, 257–273. [Google Scholar] [CrossRef] [PubMed]
- Manning, A.M.; Davis, R.J. Targeting JNK for Therapeutic Benefit: From Junk to Gold? Nat. Rev. Drug Discov. 2003, 2, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Shi, L.Z.; Chi, H. Regulation of JNK and P38 MAPK in the Immune System: Signal Integration, Propagation and Termination. Cytokine 2009, 48, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Formstecher, E.; Ramos, J.W.; Fauquet, M.; Calderwood, D.A.; Hsieh, J.-C.; Canton, B.; Nguyen, X.-T.; Barnier, J.-V.; Camonis, J.; Ginsberg, M.H.; et al. PEA-15 Mediates Cytoplasmic Sequestration of ERK MAP Kinase. Dev. Cell 2001, 1, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Soloaga, A. MSK2 and MSK1 Mediate the Mitogen- and Stress-Induced Phosphorylation of Histone H3 and HMG-14. EMBO J. 2003, 22, 2788–2797. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Furumichi, M.; Sato, Y.; Kawashima, M.; Ishiguro-Watanabe, M. KEGG for Taxonomy-Based Analysis of Pathways and Genomes. Nucleic Acids Res. 2023, 51, D587–D592. [Google Scholar] [CrossRef] [PubMed]
- Chen, F. JNK-Induced Apoptosis, Compensatory Growth, and Cancer Stem Cells. Cancer Res. 2012, 72, 379–386. [Google Scholar] [CrossRef]
- Lei, K.; Davis, R.J. JNK Phosphorylation of Bim-Related Members of the Bcl2 Family Induces Bax-Dependent Apoptosis. Proc. Natl. Acad. Sci. USA 2003, 100, 2432–2437. [Google Scholar] [CrossRef]
- Pua, L.J.W.; Mai, C.-W.; Chung, F.F.-L.; Khoo, A.S.-B.; Leong, C.-O.; Lim, W.-M.; Hii, L.-W. Functional Roles of JNK and P38 MAPK Signaling in Nasopharyngeal Carcinoma. Int. J. Mol. Sci. 2022, 23, 1108. [Google Scholar] [CrossRef] [PubMed]
- Willoughby, E.A.; Collins, M.K. Dynamic Interaction between the Dual Specificity Phosphatase MKP7 and theJNK3 Scaffold Protein β-Arrestin2. J. Biol. Chem. 2005, 280, 25651–25658. [Google Scholar] [CrossRef] [PubMed]
- Ando-Suguimoto, E.S.; Benakanakere, M.R.; Mayer, M.P.A.; Kinane, D.F. Distinct Signaling Pathways Between Human Macrophages and Primary Gingival Epithelial Cells by Aggregatibacter Actinomycetemcomitans. Pathogens 2020, 9, 248. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.; Rangarajan, P.; Ling, E.-A.; Dheen, S.T. Dexamethasone Inhibits the Nox-Dependent ROS Production via Suppression of MKP-1-Dependent MAPK Pathways in Activated Microglia. BMC Neurosci. 2011, 12, 49. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.W.; Shin, M.-G.; Lee, S.; Kim, J.-R.; Park, W.S.; Cho, K.-H.; Meyer, T.; Do Heo, W. Cooperative Activation of PI3K by Ras and Rho Family Small GTPases. Mol. Cell 2012, 47, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.-S.; Tuscano, J.; Kehrl, J.H. Adaptor Proteins CRK and CRKL Associate with the Serine/Threonine Protein Kinase GCKR Promoting GCKR and SAPK Activation. Blood 2000, 95, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Cargnello, M.; Roux, P.P. Activation and Function of the MAPKs and Their Substrates, the MAPK-Activated Protein Kinases. Microbiol. Mol. Biol. Rev. 2011, 75, 50–83. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Shi, H.; Zhao, Y. Acetaldehyde Induces Tau Phosphorylation via Activation of P38 MAPK/JNK and ROS Production. Mol. Cell. Toxicol. 2022, 18, 311–320. [Google Scholar] [CrossRef]
- Meloche, S.; Pouysségur, J. The ERK1/2 Mitogen-Activated Protein Kinase Pathway as a Master Regulator of the G1- to S-Phase Transition. Oncogene 2007, 26, 3227–3239. [Google Scholar] [CrossRef]
- Kong, T.; Liu, M.; Ji, B.; Bai, B.; Cheng, B.; Wang, C. Role of the Extracellular Signal-Regulated Kinase 1/2 Signaling Pathway in Ischemia-Reperfusion Injury. Front. Physiol. 2019, 10, 1038. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, W.; Xu, S.; Wang, S.; Tu, Y.; Xiong, Y.; Mei, J.; Wang, C. The Role of MAPK Signaling Pathway in the Her-2-Positive Meningiomas. Oncol. Rep. 2016, 36, 685–695. [Google Scholar] [CrossRef]
- Hsu, J.L.; Hung, M.-C. The Role of HER2, EGFR, and Other Receptor Tyrosine Kinases in Breast Cancer. Cancer Metastasis Rev. 2016, 35, 575–588. [Google Scholar] [CrossRef]
- Harari, D.; Yarden, Y. Molecular Mechanisms Underlying ErbB2/HER2 Action in Breast Cancer. Oncogene 2000, 19, 6102–6114. [Google Scholar] [CrossRef]
- Citri, A.; Yarden, Y. EGF–ERBB Signalling: Towards the Systems Level. Nat. Rev. Mol. Cell Biol. 2006, 7, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Morrison, D.K. MAP Kinase Pathways. Cold Spring Harb. Perspect. Biol. 2012, 4, a011254. [Google Scholar] [CrossRef]
- Li, L.; Ni, L.; Eugenin, E.A.; Heary, R.F.; Elkabes, S. Toll-like Receptor 9 Antagonism Modulates Astrocyte Function and Preserves Proximal Axons Following Spinal Cord Injury. Brain Behav. Immun. 2019, 80, 328–343. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Zhang, Y.; Ning, B. Reactive Astrocytes in Central Nervous System Injury: Subgroup and Potential Therapy. Front. Cell. Neurosci. 2021, 15, 792764. [Google Scholar] [CrossRef] [PubMed]
- Qu, W.; Tian, D.; Guo, Z.; Fang, J.; Zhang, Q.; Yu, Z.; Xie, M.; Zhang, H.; Lü, J.; Wang, W. Inhibition of EGFR/MAPK Signaling Reduces Microglial Inflammatory Response and the Associated Secondary Damage in Rats after Spinal Cord Injury. J. Neuroinflammation 2012, 9, 642. [Google Scholar] [CrossRef] [PubMed]
- Nishina, H.; Wada, T.; Katada, T. Physiological Roles of SAPK/JNK Signaling Pathway. J. Biochem. 2004, 136, 123–126. [Google Scholar] [CrossRef]
- Schellino, R.; Boido, M.; Vercelli, A. JNK Signaling Pathway Involvement in Spinal Cord Neuron Development and Death. Cells 2019, 8, 1576. [Google Scholar] [CrossRef]
- Ha, J.; Kang, E.; Seo, J.; Cho, S. Phosphorylation Dynamics of JNK Signaling: Effects of Dual-Specificity Phosphatases (DUSPs) on the JNK Pathway. Int. J. Mol. Sci. 2019, 20, 6157. [Google Scholar] [CrossRef]
- Johnson, G.L.; Nakamura, K. The C-Jun Kinase/Stress-Activated Pathway: Regulation, Function and Role in Human Disease. Biochim. Biophys. Acta BBA Mol. Cell Res. 2007, 1773, 1341–1348. [Google Scholar] [CrossRef]
- Wada, T.; Penninger, J.M. Mitogen-Activated Protein Kinases in Apoptosis Regulation. Oncogene 2004, 23, 2838–2849. [Google Scholar] [CrossRef]
- Raman, M.; Chen, W.; Cobb, M.H. Differential Regulation and Properties of MAPKs. Oncogene 2007, 26, 3100–3112. [Google Scholar] [CrossRef]
- Hammouda, M.; Ford, A.; Liu, Y.; Zhang, J. The JNK Signaling Pathway in Inflammatory Skin Disorders and Cancer. Cells 2020, 9, 857. [Google Scholar] [CrossRef] [PubMed]
- Shiryaev, A.; Moens, U. Mitogen-Activated Protein Kinase P38 and MK2, MK3 and MK5: Ménage à Trois or Ménage à Quatre? Cell. Signal. 2010, 22, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Kostenko, S. Physiological Roles of Mitogen-Activated-Protein-Kinase-Activated P38-Regulated/Activated Protein Kinase. World J. Biol. Chem. 2011, 2, 73. [Google Scholar] [CrossRef]
- Katsogiannou, M.; Andrieu, C.; Rocchi, P. Heat Shock Protein 27 Phosphorylation State Is Associated with Cancer Progression. Front. Genet. 2014, 5, 97735. [Google Scholar] [CrossRef] [PubMed]
- Reyskens, K.M.S.E.; Arthur, J.S.C. Emerging Roles of the Mitogen and Stress Activated Kinases MSK1 and MSK2. Front. Cell Dev. Biol. 2016, 4, 56. [Google Scholar] [CrossRef] [PubMed]
- Corre, I.; Paris, F.; Huot, J. The P38 Pathway, a Major Pleiotropic Cascade That Transduces Stress and Metastatic Signals in Endothelial Cells. Oncotarget 2017, 8, 55684–55714. [Google Scholar] [CrossRef]
- Koul, H.K.; Pal, M.; Koul, S. Role of P38 MAP Kinase Signal Transduction in Solid Tumors. Genes Cancer 2013, 4, 342–359. [Google Scholar] [CrossRef]
- Horiuchi, H.; Ogata, T.; Morino, T.; Chuai, M.; Yamamoto, H. Continuous Intrathecal Infusion of SB203580, a Selective Inhibitor of P38 Mitogen-Activated Protein Kinase, Reduces the Damage of Hind-Limb Function after Thoracic Spinal Cord Injury in Rat. Neurosci. Res. 2003, 47, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.-E.; Cao, J.-L.; Zhang, L.-C.; Zeng, Y.-M. Activation of P38 Mitogen-Activated Protein Kinase in Spinal Cord Contributes to Chronic Constriction Injury-Induced Neuropathic Pain. Sheng Li Xue Bao 2005, 57, 545–551. [Google Scholar] [PubMed]
- Stirling, D.P.; Liu, J.; Plunet, W.; Steeves, J.D.; Tetzlaff, W. SB203580, a P38 Mitogen-Activated Protein Kinase Inhibitor, Fails to Improve Functional Outcome Following a Moderate Spinal Cord Injury in Rat. Neuroscience 2008, 155, 128–137. [Google Scholar] [CrossRef]
- Verma, P. Axonal Protein Synthesis and Degradation Are Necessary for Efficient Growth Cone Regeneration. J. Neurosci. 2005, 25, 331–342. [Google Scholar] [CrossRef]
- Boulton, T.G.; Nye, S.H.; Robbins, D.J.; Ip, N.Y.; Radzlejewska, E.; Morgenbesser, S.D.; DePinho, R.A.; Panayotatos, N.; Cobb, M.H.; Yancopoulos, G.D. ERKs: A Family of Protein-Serine/Threonine Kinases That Are Activated and Tyrosine Phosphorylated in Response to Insulin and NGF. Cell 1991, 65, 663–675. [Google Scholar] [CrossRef]
- Bachstetter, A.D.; Van Eldik, L.J. The P38 MAP Kinase Family as Regulators of Proinflammatory Cytokine Production in Degenerative Diseases of the CNS. Aging Dis. 2010, 1, 199–211. [Google Scholar]
- Thoman, K.; Hunt, S. The Regional Districution of Extracellularly Regulated Kinase-1 and Messenger RNA in the Adult Rat Central Nervous System. Neuroscience 1993, 56, 741–757. [Google Scholar] [CrossRef]
- Mai, L.; Zhu, X.; Huang, F.; He, H.; Fan, W. P38 Mitogen-Activated Protein Kinase and Pain. Life Sci. 2020, 256, 117885. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Fitzsimmons, B.; Mohan, A.; Zhang, L.; Terrando, N.; Kordasiewicz, H.; Ji, R.-R. Intrathecal Administration of Antisense Oligonucleotide against P38α but Not P38β MAP Kinase Isoform Reduces Neuropathic and Postoperative Pain and TLR4-Induced Pain in Male Mice. Brain Behav. Immun. 2018, 72, 34–44. [Google Scholar] [CrossRef]
- Shin, J.; Yin, Y.; Park, H.; Park, S.; Triantafillu, U.L.; Kim, Y.; Kim, S.R.; Lee, S.Y.; Kim, D.K.; Hong, J.; et al. P38 siRNA-Encapsulated PLGA Nanoparticles Alleviate Neuropathic Pain Behavior in Rats by Inhibiting Microglia Activation. Nanomedicine 2018, 13, 1607–1621. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ji, J.; Wang, Z.; Xu, T. Effects of Minocycline on Motor Function Recovery and Expression of Glial Fibrillary Acidic Protein and Brain-Derived Neurotrophic Factor after Spinal Cord Injury in Rats. J. Pharm. Pharmacol. 2021, 73, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Shultz, R.B.; Zhong, Y. Minocycline Targets Multiple Secondary Injury Mechanisms in Traumatic Spinal Cord Injury. Neural Regen. Res. 2017, 12, 702–713. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Yune, T.Y.; Kim, S.J.; Park, D.W.; Lee, Y.K.; Kim, Y.C.; Oh, Y.J.; Markelonis, G.J.; Oh, T.H. Minocycline Reduces Cell Death and Improves Functional Recovery after Traumatic Spinal Cord Injury in the Rat. J. Neurotrauma 2003, 20, 1017–1027. [Google Scholar] [CrossRef] [PubMed]
- Stirling, D.P.; Khodarahmi, K.; Liu, J.; McPhail, L.T.; McBride, C.B.; Steeves, J.D.; Ramer, M.S.; Tetzlaff, W. Minocycline Treatment Reduces Delayed Oligodendrocyte Death, Attenuates Axonal Dieback, and Improves Functional Outcome after Spinal Cord Injury. J. Neurosci. 2004, 24, 2182–2190. [Google Scholar] [CrossRef] [PubMed]
- Amin, A.R.; Attur, M.G.; Thakker, G.D.; Patel, P.D.; Vyas, P.R.; Patel, R.N.; Patel, I.R.; Abramson, S.B. A Novel Mechanism of Action of Tetracyclines: Effects on Nitric Oxide Synthases. Proc. Natl. Acad. Sci. USA 1996, 93, 14014–14019. [Google Scholar] [CrossRef] [PubMed]
- Casha, S.; Zygun, D.; McGowan, M.D.; Bains, I.; Yong, V.W.; Hurlbert, R.J. Results of a Phase II Placebo-Controlled Randomized Trial of Minocycline in Acute Spinal Cord Injury. Brain 2012, 135, 1224–1236. [Google Scholar] [CrossRef] [PubMed]
- Vanhaesebroeck, B.; Guillermet-Guibert, J.; Graupera, M.; Bilanges, B. The Emerging Mechanisms of Isoform-Specific PI3K Signalling. Nat. Rev. Mol. Cell Biol. 2010, 11, 329–341. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Li, Y.; Deng, B.; Lin, A.; Zhang, G.; Ma, M.; Wang, Y.; Yang, Y.; Kang, X. The PI3K/AKT Signalling Pathway in Inflammation, Cell Death and Glial Scar Formation after Traumatic Spinal Cord Injury: Mechanisms and Therapeutic Opportunities. Cell Prolif. 2022, 55, e13275. [Google Scholar] [CrossRef]
- Xiao, C.-L.; Yin, W.-C.; Zhong, Y.-C.; Luo, J.-Q.; Liu, L.-L.; Liu, W.-Y.; Zhao, K. The Role of PI3K/Akt Signalling Pathway in Spinal Cord Injury. Biomed. Pharmacother. 2022, 156, 113881. [Google Scholar] [CrossRef]
- Glaviano, A.; Foo, A.S.C.; Lam, H.Y.; Yap, K.C.H.; Jacot, W.; Jones, R.H.; Eng, H.; Nair, M.G.; Makvandi, P.; Geoerger, B.; et al. PI3K/AKT/mTOR Signaling Transduction Pathway and Targeted Therapies in Cancer. Mol. Cancer 2023, 22, 138. [Google Scholar] [CrossRef] [PubMed]
- Hennessy, B.T.; Smith, D.L.; Ram, P.T.; Lu, Y.; Mills, G.B. Exploiting the PI3K/AKT Pathway for Cancer Drug Discovery. Nat. Rev. Drug Discov. 2005, 4, 988–1004. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Kong, H.; Cai, H.; Chen, G.; Chen, H.; Ruan, W. Progression of the PI3K/Akt Signaling Pathway in Chronic Obstructive Pulmonary Disease. Front. Pharmacol. 2023, 14, 1238782. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri-Fard, S.; Khanbabapour Sasi, A.; Hussen, B.M.; Shoorei, H.; Siddiq, A.; Taheri, M.; Ayatollahi, S.A. Interplay between PI3K/AKT Pathway and Heart Disorders. Mol. Biol. Rep. 2022, 49, 9767–9781. [Google Scholar] [CrossRef]
- Janku, F.; Yap, T.A.; Meric-Bernstam, F. Targeting the PI3K Pathway in Cancer: Are We Making Headway? Nat. Rev. Clin. Oncol. 2018, 15, 273–291. [Google Scholar] [CrossRef]
- Abid, M.R.; Guo, S.; Minami, T.; Spokes, K.C.; Ueki, K.; Skurk, C.; Walsh, K.; Aird, W.C. Vascular Endothelial Growth Factor Activates PI3K/Akt/Forkhead Signaling in Endothelial Cells. Arter. Thromb. Vasc. Biol. 2004, 24, 294–300. [Google Scholar] [CrossRef] [PubMed]
- González-García, A.; Mérida, I.; Martinez, A.C.; Carrera, A.C. Intermediate Affinity Interleukin-2 Receptor Mediates Survival via a Phosphatidylinositol 3-Kinase-Dependent Pathway. J. Biol. Chem. 1997, 272, 10220–10226. [Google Scholar] [CrossRef]
- He, Y.; Sun, M.M.; Zhang, G.G.; Yang, J.; Chen, K.S.; Xu, W.W.; Li, B. Targeting PI3K/Akt Signal Transduction for Cancer Therapy. Sig. Transduct. Target. Ther. 2021, 6, 425. [Google Scholar] [CrossRef]
- Toulany, M.; Maier, J.; Iida, M.; Rebholz, S.; Holler, M.; Grottke, A.; Jüker, M.; Wheeler, D.L.; Rothbauer, U.; Rodemann, H.P. Akt1 and Akt3 but Not Akt2 through Interaction with DNA-PKcs Stimulate Proliferation and Post-Irradiation Cell Survival of K-RAS-Mutated Cancer Cells. Cell Death Discov. 2017, 3, 17072. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, L.-H.; Yang, L.; Tang, P.-F. The PI3K/Akt/FOXO3a Pathway Regulates Regeneration Following Spinal Cord Injury in Adult Rats through TNF-α and P27kip1 Expression. Int. J. Mol. Med. 2018, 41, 2832–2838. [Google Scholar] [CrossRef]
- Hill, R.; Kalathur, R.K.R.; Callejas, S.; Colaço, L.; Brandão, R.; Serelde, B.; Cebriá, A.; Blanco-Aparicio, C.; Pastor, J.; Futschik, M.; et al. A Novel Phosphatidylinositol 3-Kinase (PI3K) Inhibitor Directs a Potent FOXO-Dependent, P53-Independent Cell Cycle Arrest Phenotype Characterized by the Differential Induction of a Subset of FOXO-Regulated Genes. Breast Cancer Res. 2014, 16, 482. [Google Scholar] [CrossRef]
- Sehgal, S.N.; Baker, H.; Vézina, C. Rapamycin (AY-22,989), a New Antifungal Antibiotic. II. Fermentation, Isolation and Characterization. J. Antibiot. 1975, 28, 727–732. [Google Scholar] [CrossRef]
- Park, S.R.; Yoo, Y.J.; Ban, Y.-H.; Yoon, Y.J. Biosynthesis of Rapamycin and Its Regulation: Past Achievements and Recent Progress. J. Antibiot. 2010, 63, 434–441. [Google Scholar] [CrossRef]
- Li, X.-G.; Du, J.-H.; Lu, Y.; Lin, X.-J. Neuroprotective Effects of Rapamycin on Spinal Cord Injury in Rats by Increasing Autophagy and Akt Signaling. Neural Regen. Res. 2019, 14, 721. [Google Scholar] [CrossRef]
- Zhang, X.; Qin, C.; Jing, Y.; Yang, D.; Liu, C.; Gao, F.; Zhang, C.; Talifu, Z.; Yang, M.; Du, L.; et al. Therapeutic Effects of Rapamycin and Surgical Decompression in a Rabbit Spinal Cord Injury Model. Cell Death Dis. 2020, 11, 567. [Google Scholar] [CrossRef]
- Liu, J.; Li, R.; Huang, Z.; Lin, J.; Ji, W.; Huang, Z.; Liu, Q.; Wu, X.; Wu, X.; Jiang, H.; et al. Rapamycin Preserves Neural Tissue, Promotes Schwann Cell Myelination and Reduces Glial Scar Formation After Hemi-Contusion Spinal Cord Injury in Mice. Front. Mol. Neurosci. 2021, 13, 574041. [Google Scholar] [CrossRef]
- Kanno, H.; Ozawa, H.; Sekiguchi, A.; Yamaya, S.; Tateda, S.; Yahata, K.; Itoi, E. The Role of mTOR Signaling Pathway in Spinal Cord Injury. Cell Cycle 2012, 11, 3175–3179. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Chen, J.; He, L.; Stiles, B.L. PTEN: Tumor Suppressor and Metabolic Regulator. Front. Endocrinol. 2018, 9, 338. [Google Scholar] [CrossRef]
- Liu, K.; Lu, Y.; Lee, J.K.; Samara, R.; Willenberg, R.; Sears-Kraxberger, I.; Tedeschi, A.; Park, K.K.; Jin, D.; Cai, B.; et al. PTEN Deletion Enhances the Regenerative Ability of Adult Corticospinal Neurons. Nat. Neurosci. 2010, 13, 1075–1081. [Google Scholar] [CrossRef]
- Park, K.K.; Liu, K.; Hu, Y.; Smith, P.D.; Wang, C.; Cai, B.; Xu, B.; Connolly, L.; Kramvis, I.; Sahin, M.; et al. Promoting Axon Regeneration in the Adult CNS by Modulation of the PTEN/mTOR Pathway. Science 2008, 322, 963–966. [Google Scholar] [CrossRef]
- Bhowmick, S.; Abdul-Muneer, P.M. PTEN Blocking Stimulates Corticospinal and Raphespinal Axonal Regeneration and Promotes Functional Recovery After Spinal Cord Injury. J. Neuropathol. Exp. Neurol. 2021, 80, 169–181. [Google Scholar] [CrossRef]
- Duan, X.; Qiao, M.; Bei, F.; Kim, I.-J.; He, Z.; Sanes, J.R. Subtype-Specific Regeneration of Retinal Ganglion Cells Following Axotomy: Effects of Osteopontin and mTOR Signaling. Neuron 2015, 85, 1244–1256. [Google Scholar] [CrossRef]
- Zhang, D.; Yuan, Y.; Zhu, J.; Zhu, D.; Li, C.; Cui, W.; Wang, L.; Ma, S.; Duan, S.; Liu, B. Insulin-like Growth Factor 1 Promotes Neurological Functional Recovery after Spinal Cord Injury through Inhibition of Autophagy via the PI3K/Akt/mTOR Signaling Pathway. Exp. Ther. Med. 2021, 22, 1265. [Google Scholar] [CrossRef]
- Truebestein, L.; Elsner, D.J.; Fuchs, E.; Leonard, T.A. A Molecular Ruler Regulates Cytoskeletal Remodelling by the Rho Kinases. Nat. Commun. 2015, 6, 10029. [Google Scholar] [CrossRef]
- Monnier, P.P.; Sierra, A.; Schwab, J.M.; Henke-Fahle, S.; Mueller, B.K. The Rho/ROCK Pathway Mediates Neurite Growth-Inhibitory Activity Associated with the Chondroitin Sulfate Proteoglycans of the CNS Glial Scar. Mol. Cell. Neurosci. 2003, 22, 319–330. [Google Scholar] [CrossRef]
- Wu, X.; Xu, X. RhoA/Rho Kinase in Spinal Cord Injury. Neural Regen. Res. 2016, 11, 23. [Google Scholar] [CrossRef]
- Chambel, S.S.; Cruz, C.D. Axonal Growth Inhibitors and Their Receptors in Spinal Cord Injury: From Biology to Clinical Translation. Neural Regen. Res. 2023, 18, 2573–2581. [Google Scholar] [CrossRef]
- Jones, L.L.; Yamaguchi, Y.; Stallcup, W.B.; Tuszynski, M.H. NG2 Is a Major Chondroitin Sulfate Proteoglycan Produced after Spinal Cord Injury and Is Expressed by Macrophages and Oligodendrocyte Progenitors. J. Neurosci. 2002, 22, 2792–2803. [Google Scholar] [CrossRef]
- Eckenstaler, R.; Hauke, M.; Benndorf, R.A. A Current Overview of RhoA, RhoB, and RhoC Functions in Vascular Biology and Pathology. Biochem. Pharmacol. 2022, 206, 115321. [Google Scholar] [CrossRef]
- Mueller, B.K.; Mack, H.; Teusch, N. Rho Kinase, a Promising Drug Target for Neurological Disorders. Nat. Rev. Drug Discov. 2005, 4, 387–398. [Google Scholar] [CrossRef]
- Liu, H.W.; Halayko, A.J.; Fernandes, D.J.; Harmon, G.S.; McCauley, J.A.; Kocieniewski, P.; McConville, J.; Fu, Y.; Forsythe, S.M.; Kogut, P.; et al. The RhoA/Rho Kinase Pathway Regulates Nuclear Localization of Serum Response Factor. Am. J. Respir. Cell Mol. Biol. 2003, 29, 39–47. [Google Scholar] [CrossRef]
- Tabuchi, A.; Ihara, D. Regulation of Dendritic Synaptic Morphology and Transcription by the SRF Cofactor MKL/MRTF. Front. Mol. Neurosci. 2021, 14, 767842. [Google Scholar] [CrossRef]
- Lord-Fontaine, S.; Yang, F.; Diep, Q.; Dergham, P.; Munzer, S.; Tremblay, P.; McKerracher, L. Local Inhibition of Rho Signaling by Cell-Permeable Recombinant Protein BA-210 Prevents Secondary Damage and Promotes Functional Recovery Following Acute Spinal Cord Injury. J. Neurotrauma 2008, 25, 1309–1322. [Google Scholar] [CrossRef]
- Fehlings, M.G.; Theodore, N.; Harrop, J.; Maurais, G.; Kuntz, C.; Shaffrey, C.I.; Kwon, B.K.; Chapman, J.; Yee, A.; Tighe, A.; et al. A Phase I/IIa Clinical Trial of a Recombinant Rho Protein Antagonist in Acute Spinal Cord Injury. J. Neurotrauma 2011, 28, 787–796. [Google Scholar] [CrossRef]
- Fehlings, M.G.; Kim, K.D.; Aarabi, B.; Rizzo, M.; Bond, L.M.; McKerracher, L.; Vaccaro, A.R.; Okonkwo, D.O. Rho Inhibitor VX-210 in Acute Traumatic Subaxial Cervical Spinal Cord Injury: Design of the SPinal Cord Injury Rho INhibition InvestiGation (SPRING) Clinical Trial. J. Neurotrauma 2018, 35, 1049–1056. [Google Scholar] [CrossRef]
- Fehlings, M.G.; Chen, Y.; Aarabi, B.; Ahmad, F.; Anderson, K.D.; Dumont, T.; Fourney, D.R.; Harrop, J.S.; Kim, K.D.; Kwon, B.K.; et al. A Randomized Controlled Trial of Local Delivery of a Rho Inhibitor (VX-210) in Patients with Acute Traumatic Cervical Spinal Cord Injury. J. Neurotrauma 2021, 38, 2065–2072. [Google Scholar] [CrossRef]
- Jacobson, P.B.; Goody, R.; Lawrence, M.; Mueller, B.K.; Zhang, X.; Hooker, B.A.; Pfleeger, K.; Ziemann, A.; Locke, C.; Barraud, Q.; et al. Elezanumab, a Human Anti-RGMa Monoclonal Antibody, Promotes Neuroprotection, Neuroplasticity, and Neurorecovery Following a Thoracic Hemicompression Spinal Cord Injury in Non-Human Primates. Neurobiol. Dis. 2021, 155, 105385. [Google Scholar] [CrossRef]
- Mothe, A.J.; Tassew, N.G.; Shabanzadeh, A.P.; Penheiro, R.; Vigouroux, R.J.; Huang, L.; Grinnell, C.; Cui, Y.-F.; Fung, E.; Monnier, P.P.; et al. RGMa Inhibition with Human Monoclonal Antibodies Promotes Regeneration, Plasticity and Repair, and Attenuates Neuropathic Pain after Spinal Cord Injury. Sci. Rep. 2017, 7, 10529. [Google Scholar] [CrossRef]
- Hata, K.; Fujitani, M.; Yasuda, Y.; Doya, H.; Saito, T.; Yamagishi, S.; Mueller, B.K.; Yamashita, T. RGMa Inhibition Promotes Axonal Growth and Recovery after Spinal Cord Injury. J. Cell Biol. 2006, 173, 47–58. [Google Scholar] [CrossRef]
- Mothe, A.J.; Jacobson, P.B.; Caprelli, M.; Ulndreaj, A.; Rahemipour, R.; Huang, L.; Monnier, P.P.; Fehlings, M.G.; Tator, C.H. Delayed Administration of Elezanumab, a Human Anti-RGMa Neutralizing Monoclonal Antibody, Promotes Recovery Following Cervical Spinal Cord Injury. Neurobiol. Dis. 2022, 172, 105812. [Google Scholar] [CrossRef]
- Nakagawa, H.; Ninomiya, T.; Yamashita, T.; Takada, M. Treatment With the Neutralizing Antibody Against Repulsive Guidance Molecule-a Promotes Recovery From Impaired Manual Dexterity in a Primate Model of Spinal Cord Injury. Cereb. Cortex 2019, 29, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Forgione, N.; Fehlings, M.G. Rho-ROCK Inhibition in the Treatment of Spinal Cord Injury. World Neurosurg. 2014, 82, e535–e539. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Chen, Q. The NF-κB Pathway: A Focus on Inflammatory Responses in Spinal Cord Injury. Mol. Neurobiol. 2023, 60, 5292–5308. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Lin, L.; Zhang, Z.; Zhang, H.; Hu, H. Targeting NF-κB Pathway for the Therapy of Diseases: Mechanism and Clinical Study. Sig. Transduct. Target. Ther. 2020, 5, 209. [Google Scholar] [CrossRef]
- Taniguchi, K.; Karin, M. NF-κB, Inflammation, Immunity and Cancer: Coming of Age. Nat. Rev. Immunol. 2018, 18, 309–324. [Google Scholar] [CrossRef]
- Albensi, B.C. What Is Nuclear Factor Kappa B (NF-κB) Doing in and to the Mitochondrion? Front. Cell Dev. Biol. 2019, 7, 154. [Google Scholar] [CrossRef] [PubMed]
- Christian, F.; Smith, E.; Carmody, R. The Regulation of NF-κB Subunits by Phosphorylation. Cells 2016, 5, 12. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB Signaling in Inflammation. Sign. Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Bethea, J.R.; Castro, M.; Keane, R.W.; Lee, T.T.; Dietrich, W.D.; Yezierski, R.P. Traumatic Spinal Cord Injury Induces Nuclear Factor-kB Activation. J. Neurosci. 1998, 18, 3251–3260. [Google Scholar] [CrossRef]
- Xu, L.; Botchway, B.O.A.; Zhang, S.; Zhou, J.; Liu, X. Inhibition of NF-κB Signaling Pathway by Resveratrol Improves Spinal Cord Injury. Front. Neurosci. 2018, 12, 690. [Google Scholar] [CrossRef]
- Xia, M.; Zhang, Y.; Wu, H.; Zhang, Q.; Liu, Q.; Li, G.; Zhao, T.; Liu, X.; Zheng, S.; Qian, Z.; et al. Forsythoside B Attenuates Neuro-Inflammation and Neuronal Apoptosis by Inhibition of NF-κB and P38-MAPK Signaling Pathways through Activating Nrf2 Post Spinal Cord Injury. Int. Immunopharmacol. 2022, 111, 109120. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qiu, H.; Yao, S.; Li, Q.; Ding, Y.; Cao, Y.; Chen, X.; Zhu, X. Geniposide Exerts Protective Effects on Spinal Cord Injury in Rats by Inhibiting the IKKs/NF-κB Signaling Pathway. Int. Immunopharmacol. 2021, 100, 108158. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhu, H.; Wang, Z.; Gao, F.; Wang, J.; Zhang, W. Wogonoside Alleviates Inflammation Induced by Traumatic Spinal Cord Injury by Suppressing NF-κB and NLRP3 Inflammasome Activation. Exp. Ther. Med. 2017, 14, 3304–3308. [Google Scholar] [CrossRef]
- Feng, X.; Chen, X.; Zaeem, M.; Zhang, W.; Song, L.; Chen, L.; Mubwandarikwa, J.; Chen, X.; Xiao, J.; Xie, L.; et al. Sesamol Attenuates Neuroinflammation by Regulating the AMPK/SIRT1/NF-κB Signaling Pathway after Spinal Cord Injury in Mice. Oxidative Med. Cell. Longev. 2022, 2022, 8010670. [Google Scholar] [CrossRef]
- Jung, K.K.; Lee, H.S.; Cho, J.Y.; Shin, W.C.; Rhee, M.H.; Kim, T.G.; Kang, J.H.; Kim, S.H.; Hong, S.; Kang, S.Y. Inhibitory Effect of Curcumin on Nitric Oxide Production from Lipopolysaccharide-Activated Primary Microglia. Life Sci. 2006, 79, 2022–2031. [Google Scholar] [CrossRef]
- Huang, Y.; Zhu, N.; Chen, T.; Chen, W.; Kong, J.; Zheng, W.; Ruan, J. Triptolide Suppressed the Microglia Activation to Improve Spinal Cord Injury Through miR-96/IKKβ/NF-κB Pathway. Spine 2019, 44, E707–E714. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.-C. The Non-Canonical NF-κB Pathway in Immunity and Inflammation. Nat. Rev. Immunol. 2017, 17, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Jin, Y.; Chen, X.; Ye, X.; Shen, X.; Lin, M.; Zeng, C.; Zhou, T.; Zhang, J. NF-κB in Biology and Targeted Therapy: New Insights and Translational Implications. Sign. Transduct. Target. Ther. 2024, 9, 53. [Google Scholar] [CrossRef]
- Guo, X.; Jiang, C.; Chen, Z.; Wang, X.; Hong, F.; Hao, D. Regulation of the JAK/STAT Signaling Pathway in Spinal Cord Injury: An Updated Review. Front. Immunol. 2023, 14, 1276445. [Google Scholar] [CrossRef]
- Xue, C.; Yao, Q.; Gu, X.; Shi, Q.; Yuan, X.; Chu, Q.; Bao, Z.; Lu, J.; Li, L. Evolving Cognition of the JAK-STAT Signaling Pathway: Autoimmune Disorders and Cancer. Sign. Transduct. Target. Ther. 2023, 8, 204. [Google Scholar] [CrossRef]
- Jatiani, S.S.; Baker, S.J.; Silverman, L.R.; Reddy, E.P. JAK/STAT Pathways in Cytokine Signaling and Myeloproliferative Disorders: Approaches for Targeted Therapies. Genes Cancer 2010, 1, 979–993. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Bian, Q.; Rong, D.; Wang, L.; Song, J.; Huang, H.-S.; Zeng, J.; Mei, J.; Wang, P.-Y. JAK/STAT Pathway: Extracellular Signals, Diseases, Immunity, and Therapeutic Regimens. Front. Bioeng. Biotechnol. 2023, 11, 1110765. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, K.; Osuka, K.; Takayasu, M.; Usuda, N.; Nakazawa, A.; Nakahara, N.; Yoshida, M.; Aoshima, C.; Hara, M.; Yoshida, J. Activation of JAK/STAT Signalling in Neurons Following Spinal Cord Injury in Mice. J. Neurochem. 2006, 96, 1060–1070. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Yuan, W.; Liu, Y.; Zhang, Y.; Wang, Z.; Zhou, X.; Ning, G.; Zhang, L.; Yao, L.; Feng, S.; et al. The Role of the JAK-STAT Pathway in Neural Stem Cells, Neural Progenitor Cells and Reactive Astrocytes after Spinal Cord Injury. Biomed. Rep. 2015, 3, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Carow, B.; Rottenberg, M.E. SOCS3, a Major Regulator of Infection and Inflammation. Front. Immunol. 2014, 5, 58. [Google Scholar] [CrossRef] [PubMed]
- Kershaw, N.J.; Murphy, J.M.; Liau, N.P.D.; Varghese, L.N.; Laktyushin, A.; Whitlock, E.L.; Lucet, I.S.; Nicola, N.A.; Babon, J.J. SOCS3 Binds Specific Receptor–JAK Complexes to Control Cytokine Signaling by Direct Kinase Inhibition. Nat. Struct. Mol. Biol. 2013, 20, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Liu, Y.; Sun, F.; Wang, X.; Liu, X.; He, Z. Restoration of Skilled Locomotion by Sprouting Corticospinal Axons Induced by Co-Deletion of PTEN and SOCS3. Nat. Commun. 2015, 6, 8074. [Google Scholar] [CrossRef] [PubMed]
- Tapia, V.S.; Herrera-Rojas, M.; Larrain, J. JAK-STAT Pathway Activation in Response to Spinal Cord Injury in Regenerative and Non-Regenerative Stages of Xenopus Laevis. Regeneration 2017, 4, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Lee-Liu, D.; Moreno, M.; Almonacid, L.I.; Tapia, V.S.; Muñoz, R.; Von Marées, J.; Gaete, M.; Melo, F.; Larraín, J. Genome-Wide Expression Profile of the Response to Spinal Cord Injury in Xenopus Laevis Reveals Extensive Differences between Regenerative and Non-Regenerative Stages. Neural Dev. 2014, 9, 12. [Google Scholar] [CrossRef]
- Harrison, D.A. The JAK/STAT Pathway. Cold Spring Harb. Perspect. Biol. 2012, 4, a011205. [Google Scholar] [CrossRef]
- Shuai, K.; Liu, B. Regulation of JAK–STAT Signalling in the Immune System. Nat. Rev. Immunol. 2003, 3, 900–911. [Google Scholar] [CrossRef] [PubMed]
- Badhiwala, J.H.; Wilson, J.R.; Witiw, C.D.; Harrop, J.S.; Vaccaro, A.R.; Aarabi, B.; Grossman, R.G.; Geisler, F.H.; Fehlings, M.G. The Influence of Timing of Surgical Decompression for Acute Spinal Cord Injury: A Pooled Analysis of Individual Patient Data. Lancet Neurol. 2021, 20, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Fehlings, M.G.; Wilson, J.R.; Tetreault, L.A.; Aarabi, B.; Anderson, P.; Arnold, P.M.; Brodke, D.S.; Burns, A.S.; Chiba, K.; Dettori, J.R.; et al. A Clinical Practice Guideline for the Management of Patients With Acute Spinal Cord Injury: Recommendations on the Use of Methylprednisolone Sodium Succinate. Glob. Spine J. 2017, 7, 203S–211S. [Google Scholar] [CrossRef]
- Ahuja, C.S.; Badhiwala, J.H.; Fehlings, M.G. “Time Is Spine”: The Importance of Early Intervention for Traumatic Spinal Cord Injury. Spinal Cord 2020, 58, 1037–1039. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.-J.; Wang, Q.; Tang, H.-H.; Bai, J.-Z.; Wang, F.-Y.; Lv, Z.; Chen, S.-Z.; Liu, J.-S.; Hong, Y.; Zhang, J.-W. Heterogeneity among Traumatic Spinal Cord Injuries at the Thoracolumbar Junction: Helping Select Patients for Clinical Trials. Spinal Cord 2019, 57, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Badhiwala, J.H.; Wilson, J.R.; Kulkarni, A.V.; Kiss, A.; Harrop, J.S.; Vaccaro, A.R.; Aarabi, B.; Geisler, F.H.; Fehlings, M.G. A Novel Method to Classify Cervical Incomplete Spinal Cord Injury Based on Potential for Recovery: A Group-Based Trajectory Analysis. J. Neurotrauma 2022, 39, 1654–1664. [Google Scholar] [CrossRef] [PubMed]
- Rick Hansen Institute. A Look at Traumatic Spinal Cord Injury in Canada: Rick Hansen Spinal Cord Registry (RHSCIR). J. Spinal Cord Med. 2017, 40, 870–871. [Google Scholar] [CrossRef] [PubMed]
- Satkunendrarajah, K.; Karadimas, S.K.; Laliberte, A.M.; Montandon, G.; Fehlings, M.G. Cervical Excitatory Neurons Sustain Breathing after Spinal Cord Injury. Nature 2018, 562, 419–422. [Google Scholar] [CrossRef]
- Krassioukov, A.; Warburton, D.E.; Teasell, R.; Eng, J.J. A Systematic Review of the Management of Autonomic Dysreflexia After Spinal Cord Injury. Arch. Phys. Med. Rehabil. 2009, 90, 682–695. [Google Scholar] [CrossRef]
- Schwab, J.M.; Zhang, Y.; Kopp, M.A.; Brommer, B.; Popovich, P.G. The Paradox of Chronic Neuroinflammation, Systemic Immune Suppression, Autoimmunity after Traumatic Chronic Spinal Cord Injury. Exp. Neurol. 2014, 258, 121–129. [Google Scholar] [CrossRef]
- Zavvarian, M.-M.; Hong, J.; Khazaei, M.; Chio, J.C.T.; Wang, J.; Badner, A.; Fehlings, M.G. The Protein Kinase Inhibitor Midostaurin Improves Functional Neurological Recovery and Attenuates Inflammatory Changes Following Traumatic Cervical Spinal Cord Injury. Biomolecules 2021, 11, 972. [Google Scholar] [CrossRef] [PubMed]
- Zavvarian, M.-M.; Toossi, A.; Khazaei, M.; Hong, J.; Fehlings, M. Novel Innovations in Cell and Gene Therapies for Spinal Cord Injury. F1000Research 2020, 9, 279. [Google Scholar] [CrossRef] [PubMed]
- Kathe, C.; Skinnider, M.A.; Hutson, T.H.; Regazzi, N.; Gautier, M.; Demesmaeker, R.; Komi, S.; Ceto, S.; James, N.D.; Cho, N.; et al. The Neurons That Restore Walking after Paralysis. Nature 2022, 611, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Griffin, J.M.; Bradke, F. Therapeutic Repair for Spinal Cord Injury: Combinatory Approaches to Address a Multifaceted Problem. EMBO Mol. Med. 2020, 12, e11505. [Google Scholar] [CrossRef]
- High, K.A.; Roncarolo, M.G. Gene Therapy. N. Engl. J. Med. 2019, 381, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.A.; O’Shea, T.M.; Burda, J.E.; Ao, Y.; Barlatey, S.L.; Bernstein, A.M.; Kim, J.H.; James, N.D.; Rogers, A.; Kato, B.; et al. Required Growth Facilitators Propel Axon Regeneration across Complete Spinal Cord Injury. Nature 2018, 561, 396–400. [Google Scholar] [CrossRef]
- Slyper, M.; Porter, C.B.M.; Ashenberg, O.; Waldman, J.; Drokhlyansky, E.; Wakiro, I.; Smillie, C.; Smith-Rosario, G.; Wu, J.; Dionne, D.; et al. A Single-Cell and Single-Nucleus RNA-Seq Toolbox for Fresh and Frozen Human Tumors. Nat. Med. 2020, 26, 792–802. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.L.; Yaron, T.M.; Huntsman, E.M.; Kerelsky, A.; Song, J.; Regev, A.; Lin, T.-Y.; Liberatore, K.; Cizin, D.M.; Cohen, B.M.; et al. An Atlas of Substrate Specificities for the Human Serine/Threonine Kinome. Nature 2023, 613, 759–766. [Google Scholar] [CrossRef]
- Hutti, J.E.; Jarrell, E.T.; Chang, J.D.; Abbott, D.W.; Storz, P.; Toker, A.; Cantley, L.C.; Turk, B.E. A Rapid Method for Determining Protein Kinase Phosphorylation Specificity. Nat. Methods 2004, 1, 27–29. [Google Scholar] [CrossRef]
- Attwood, M.M.; Fabbro, D.; Sokolov, A.V.; Knapp, S.; Schiöth, H.B. Trends in Kinase Drug Discovery: Targets, Indications and Inhibitor Design. Nat. Rev. Drug Discov. 2021, 20, 839–861. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zavvarian, M.-M.; Modi, A.D.; Sadat, S.; Hong, J.; Fehlings, M.G. Translational Relevance of Secondary Intracellular Signaling Cascades Following Traumatic Spinal Cord Injury. Int. J. Mol. Sci. 2024, 25, 5708. https://doi.org/10.3390/ijms25115708
Zavvarian M-M, Modi AD, Sadat S, Hong J, Fehlings MG. Translational Relevance of Secondary Intracellular Signaling Cascades Following Traumatic Spinal Cord Injury. International Journal of Molecular Sciences. 2024; 25(11):5708. https://doi.org/10.3390/ijms25115708
Chicago/Turabian StyleZavvarian, Mohammad-Masoud, Akshat D. Modi, Sarah Sadat, James Hong, and Michael G. Fehlings. 2024. "Translational Relevance of Secondary Intracellular Signaling Cascades Following Traumatic Spinal Cord Injury" International Journal of Molecular Sciences 25, no. 11: 5708. https://doi.org/10.3390/ijms25115708
APA StyleZavvarian, M.-M., Modi, A. D., Sadat, S., Hong, J., & Fehlings, M. G. (2024). Translational Relevance of Secondary Intracellular Signaling Cascades Following Traumatic Spinal Cord Injury. International Journal of Molecular Sciences, 25(11), 5708. https://doi.org/10.3390/ijms25115708






