Genes Co-Expressed with ESR2 Influence Clinical Outcomes in Cancer Patients: TCGA Data Analysis
Abstract
:1. Introduction
2. Results
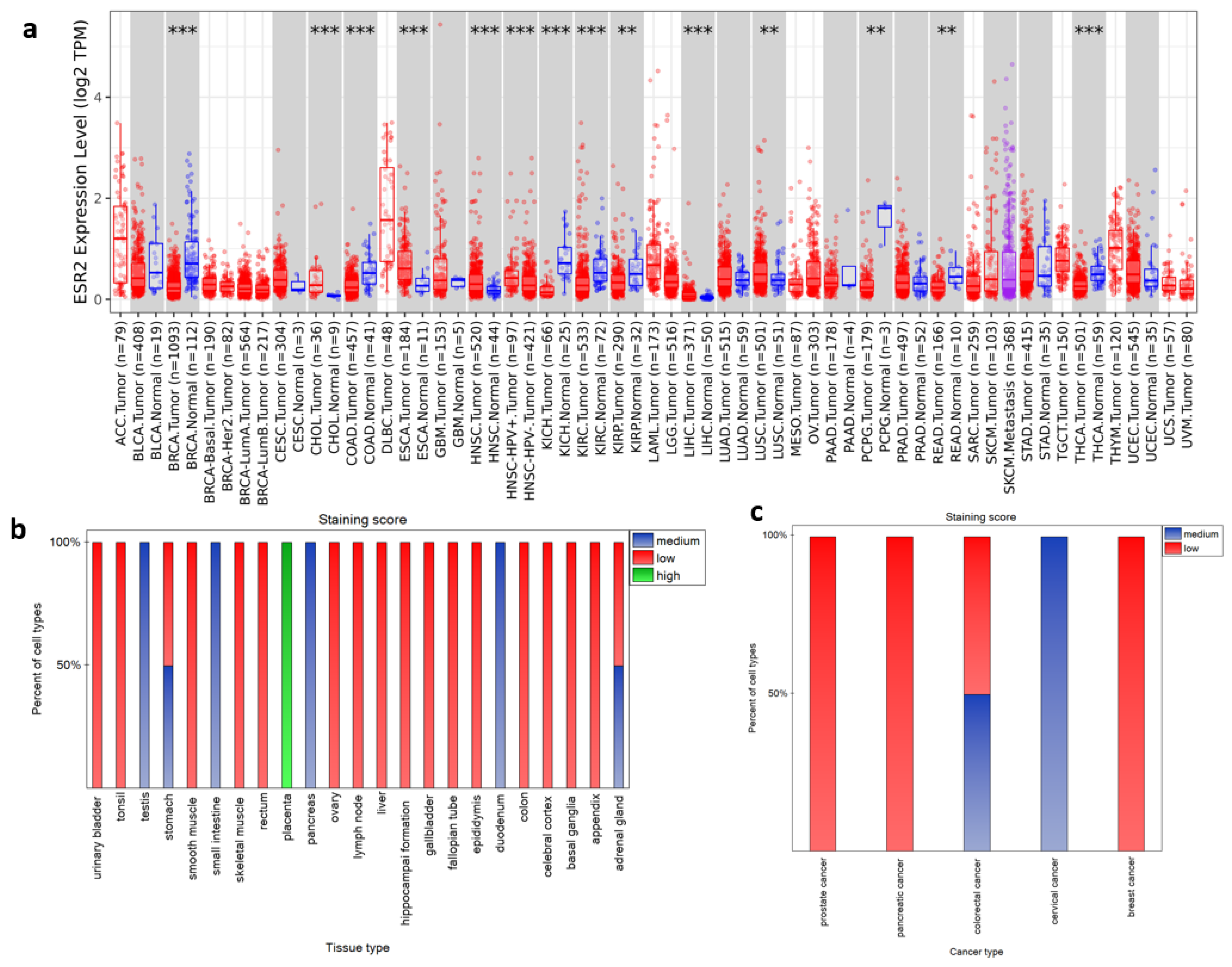
2.1. Cancer Tissue and Corresponding Healthy Tissue Present Different ESR2 Expression Levels in Selected Cancer Types
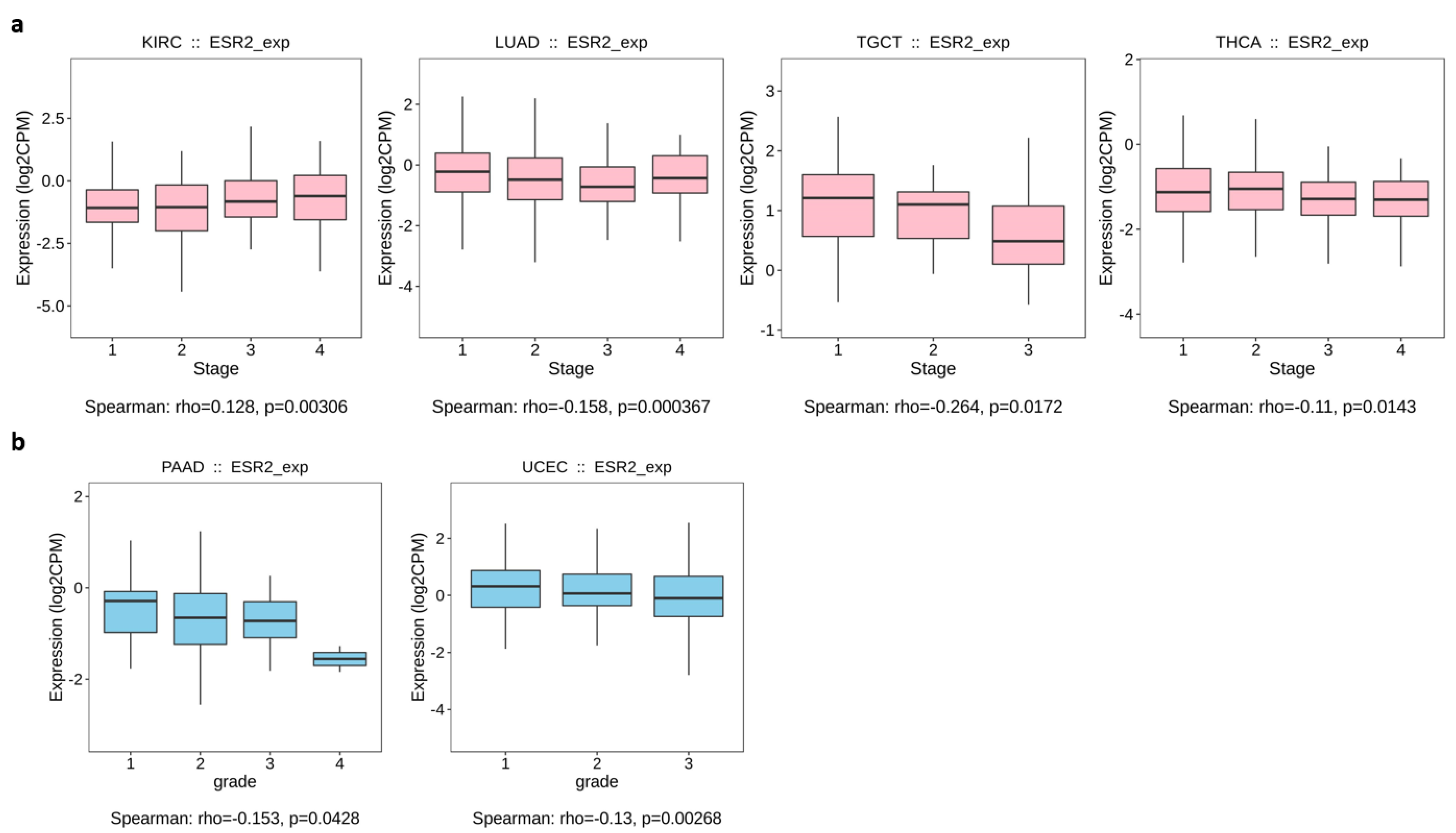
2.2. ESR2 Expression Correlates with the Grade and Stage of Several Tumor Types
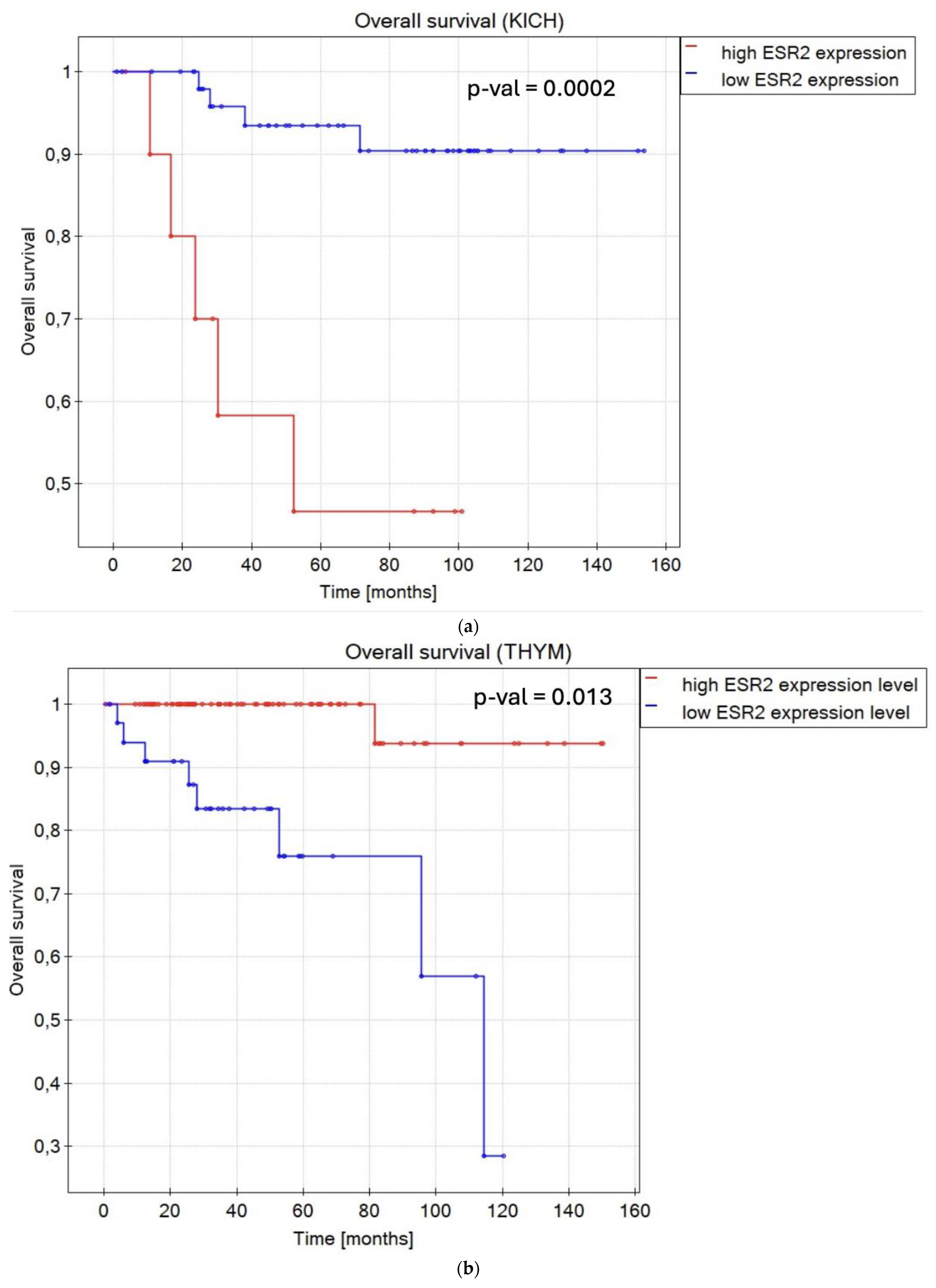
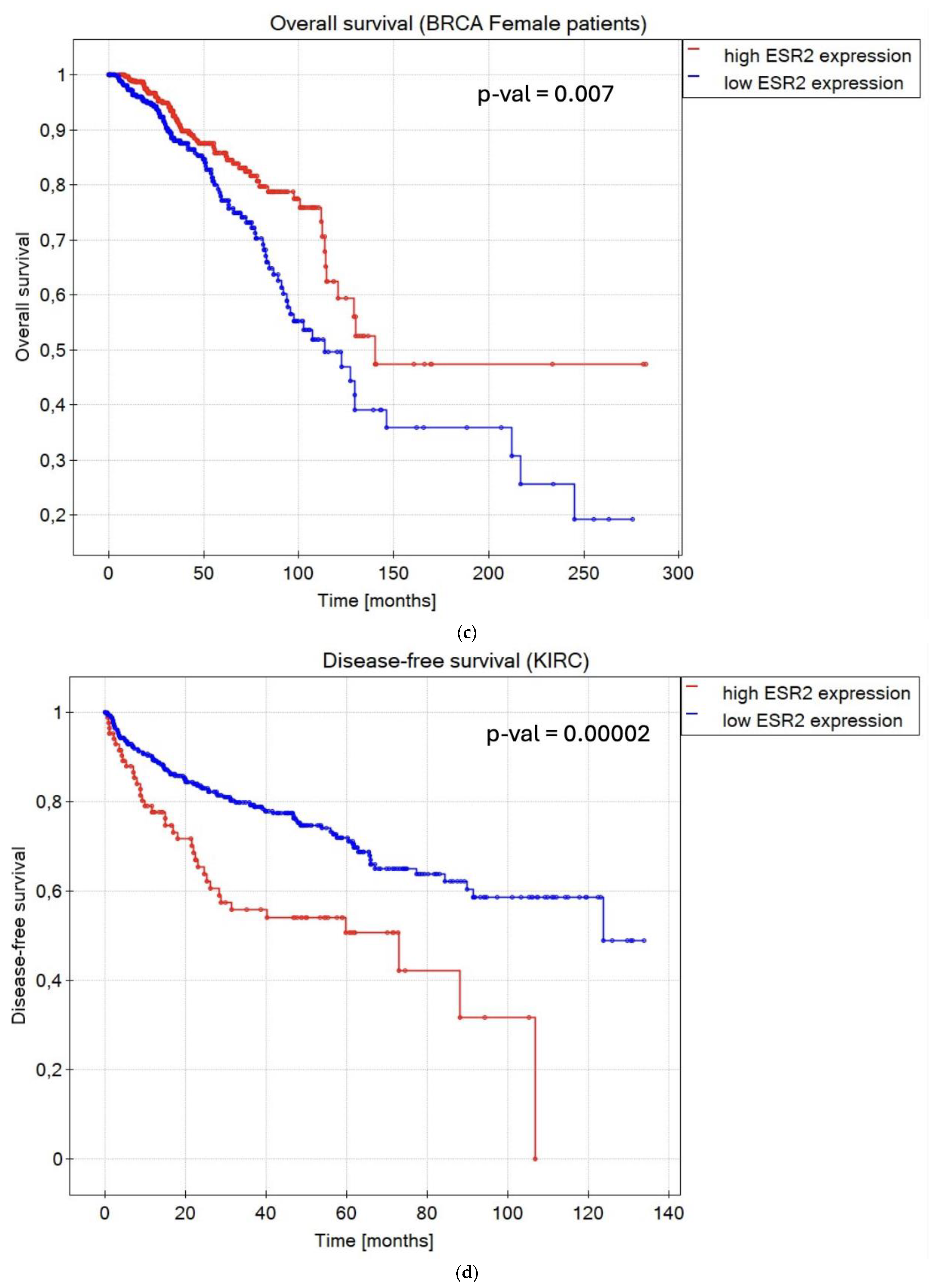
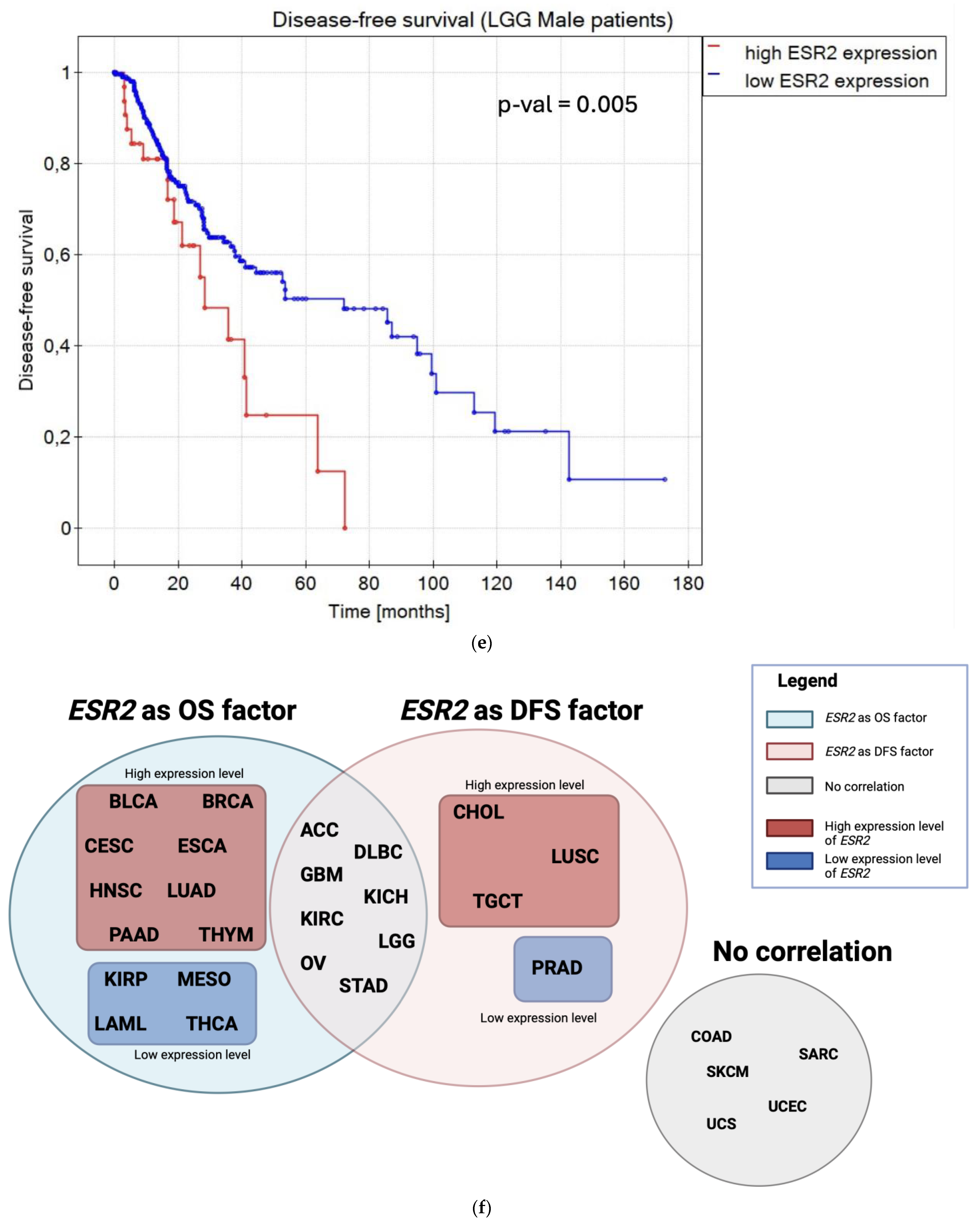
2.3. ESR2 Expression Level Affects OS and DFS in Various Cancer Types
2.4. ESR2 Affects Various Molecular Pathways in Cancer
2.5. Various Genes Present Expression Patterns Similar to That of ESR2 in Cancer
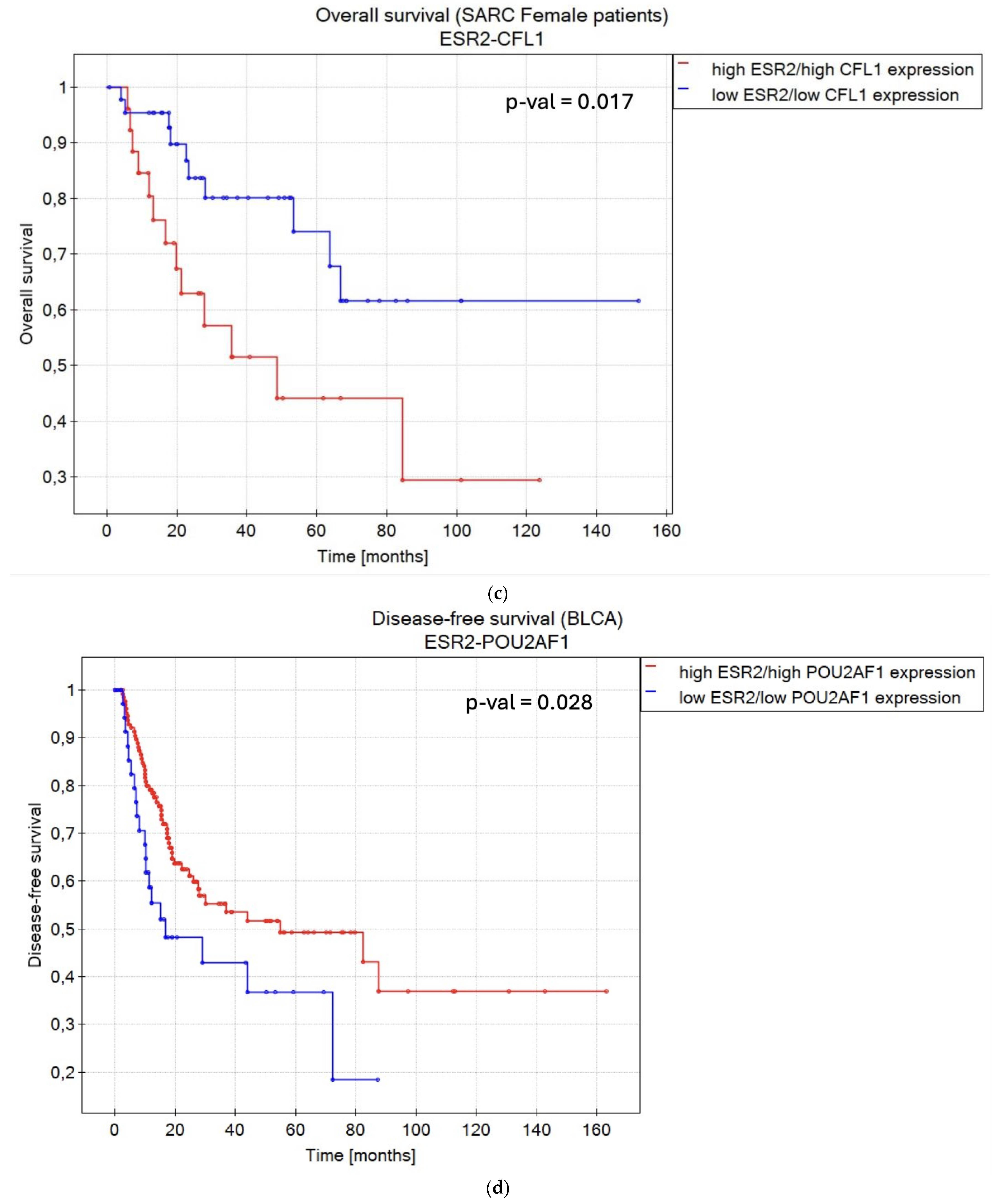
2.6. Selected Genes Present a Combined Effect with ESR2 on OS/DFS in Several Cancer Types
3. Discussion
4. Materials and Methods
4.1. Transcriptomic and Proteomic Data
4.2. Clinical Data
4.3. Databases and Bioinformatic Tools
4.3.1. cBioPortal
4.3.2. TIMER2.0
4.3.3. Human Protein Atlas
4.3.4. TISIDB
4.3.5. GSEA
4.3.6. UCSC Genome Browser
4.4. Statistical Analysis
5. Conclusions
- ESR2 mRNA expression differs between cancerous and normal tissue in various TCGA tumor types, including BRCA, COAD, KICH, CHOL, HNSC, and LUSC;
- ESR2 expression impacts patient survival in several TCGA tumor types, including BLCA, HNSC, THYM, KIRP, and THCA;
- GSEA analysis reveals ESR2 enrichment in gene sets related to epithelial–mesenchymal transition, hypoxia response, and cell cycle in cancers like PRAD and COAD;
- Twelve genes (ACIN1, CFL1, FNBP4, MDM4, NDUFB3, OCIAD2, PLIN3, POU2AF1, RAC1, SYNE2, TMEM141, TNFRSF13C) were identified as co-expressed with ESR2 and showing a combined effect with the receptor on patient survival in selected tumors, including BLCA, MESO, BRCA, COAD, and SKCM.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACC | Adrenocortical carcinoma |
| BLCA | Bladder urothelial carcinoma |
| BRCA | Breast invasive carcinoma |
| CESC | Cervical squamous cell carcinoma and endocervical adenocarcinoma |
| CHOL | Cholangio carcinoma |
| COAD | Colon adenocarcinoma |
| DLBC | Lymphoid neoplasm diffuse large B-cell lymphoma |
| ESCA | Esophageal carcinoma |
| GBM | Glioblastoma multiforme |
| HNSC | Head and neck squamous cell carcinoma |
| KICH | Kidney chromophobe |
| KIRC | Kidney renal clear cell carcinoma |
| KIRP | Kidney renal papillary cell carcinoma |
| LAML | Acute myeloid leukemia |
| LGG | Brain lower-grade glioma |
| LIHC | Liver hepatocellular carcinoma |
| LUAD | Lung adenocarcinoma |
| LUSC | Lung squamous cell carcinoma |
| MESO | Mesothelioma |
| OV | Ovarian serous cystadenocarcinoma |
| PAAD | Pancreatic adenocarcinoma |
| PCPG | Pheochromocytoma and paraganglioma |
| PRAD | Prostate adenocarcinoma |
| READ | Rectum adenocarcinoma |
| SARC | Sarcoma |
| SKCM | Skin cutaneous melanoma |
| STAD | Stomach adenocarcinoma |
| TGCT | Testicular germ cell tumor |
| THCA | Thyroid carcinoma |
| THYM | Thymoma |
| UCEC | Uterine corpus endometrial carcinoma |
| UCS | Uterine carcinosarcoma |
| UVM | Uveal melanoma |
References
- Jia, M.; Dahlman-Wright, K.; Gustafsson, J.Å. Estrogen receptor alpha and beta in health and disease. Best Pract. Res. Clin. Endocrinol. Metab. 2015, 29, 557–568. [Google Scholar] [CrossRef]
- Chen, P.; Li, B.; Ou-Yang, L. Role of estrogen receptors in health and disease. Front. Endocrinol. 2022, 13, 839005. [Google Scholar] [CrossRef] [PubMed]
- Božović, A.; Mandušić, V.; Todorović, L.; Krajnović, M. Estrogen Receptor Beta: The Promising Biomarker and Potential Target in Metastases. Int. J. Mol. Sci. 2021, 22, 1656. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, S.; Mäkelä, S.; Treuter, E.; Tujague, M.; Thomsen, J.; Andersson, G.; Enmark, E.; Pettersson, K.; Warner, M.; Gustafsson, J.A. Mechanisms of Estrogen Action. Physiol. Rev. 2001, 81, 1535–1565. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Zakharov, M.N.; Khan, S.H.; Miki, R.; Jang, H.; Toraldo, G.; Singh, R.; Bhasin, S.; Jasuja, R. The dynamic structure of the estrogen receptor. J. Amino Acids 2011, 2011, 812540. [Google Scholar] [CrossRef] [PubMed]
- Hwang, N.M.; Stabile, L.P. Estrogen Receptor ß in Cancer: To ß(e) or not to ß(e)? Endocrinology 2021, 162, bqab162. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-Y.; Vega, V.B.; Thomsen, J.S.; Zhang, T.; Kong, S.L.; Xie, M.; Chiu, K.P.; Lipovich, L.; Barnett, D.H.; Stossi, F.; et al. Whole-genome cartography of estrogen receptor α binding sites. PLoS Genet. 2007, 3, e87. [Google Scholar] [CrossRef] [PubMed]
- Heldring, N.; Pike, A.; Andersson, S.; Matthews, J.; Cheng, G.; Hartman, J.; Tujague, M.; Ström, A.; Treuter, E.; Warner, M.; et al. Estrogen receptors: How do they signal and what are their targets. Physiol. Rev. 2007, 87, 905–931. [Google Scholar] [CrossRef]
- Warner, M.; Huang, B.; Gustafsson, J.-A. Estrogen Receptor β as a Pharmaceutical Target. Trends Pharmacol. Sci. 2017, 38, 92–99. [Google Scholar] [CrossRef]
- Mal, R.; Magner, A.; David, J.; Datta, J.; Vallabhaneni, M.; Kassem, M.; Manouchehri, J.; Willingham, N.; Stover, D.; Vandeusen, J.; et al. Estrogen Receptor Beta (ERβ): A Ligand Activated Tumor Suppressor. Front. Oncol. 2020, 10, 587386. [Google Scholar] [CrossRef]
- Nelson, A.W.; Tilley, W.D.; Neal, D.E.; Carroll, J.S. Estrogen receptor beta in prostate cancer: Friend or foe? Endocr.-Relat. Cancer 2014, 21, T219–T234. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Omoto, Y.; Iwase, H.; Yamashita, H.; Toyama, T.; Coombes, R.C.; Filipovic, A.; Warner, M.; Gustafsson, J. Differential expression of estrogen receptor α, β1, and β2 in lobular and ductal breast cancer. Proc. Natl. Acad. Sci. USA 2014, 111, 1933–1938. [Google Scholar] [CrossRef] [PubMed]
- Treeck, O.; Juhasz-Boess, I.; Lattrich, C.; Horn, F.; Goerse, R.; Ortmann, O. Effects of exon-deleted estrogen receptor β transcript variants on growth, apoptosis and gene expression of human breast cancer cell lines. Breast Cancer Res. Treat. 2007, 110, 507–520. [Google Scholar] [CrossRef]
- Zhao, C.; Lam, E.W.-F.; Sunters, A.; Enmark, E.; De Bella, M.T.; Coombes, R.C.; Gustafsson, J.; Dahlman-Wright, K. Expression of estrogen receptor β isoforms in normal breast epithelial cells and breast cancer: Regulation by methylation. Oncogene 2003, 22, 7600–7606. [Google Scholar] [CrossRef] [PubMed]
- Markey, G.C.; Cullen, R.; Diggin, P.; Hill, A.D.K.; Mc Dermott, E.W.; O’Higgins, N.J.; Duffy, M.J. Estrogen receptor-β mRNA is associated with adverse outcome in patients with breast cancer. Tumor Biol. 2009, 30, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Increased Expression of Estrogen Receptor beta mRNA in Tamoxifen-Resistant Breast Cancer Patients. Available online: https://pubmed.ncbi.nlm.nih.gov/10554009/ (accessed on 11 March 2024).
- Giroux, V.; Bernatchez, G.; Carrier, J.C. Chemopreventive effect of ERβ-Selective agonist on intestinal tumorigenesis in ApcMin/+ mice. Mol. Carcinog. 2010, 50, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Giroux, V.; Lemay, F.; Bernatchez, G.; Robitaille, Y.; Carrier, J.C. Estrogen receptor β deficiency enhances small intestinal tumorigenesis in ApcMin/+ mice. Int. J. Cancer 2008, 123, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Wang, J.; Wang, L.; Shen, C.; Su, B.; Qi, M.; Hu, J.; Gao, W.; Tan, W.; Han, B. Estrogen induces androgen-repressed SOX4 expression to promote progression of prostate cancer cells. Prostate 2015, 75, 1363–1375. [Google Scholar] [CrossRef] [PubMed]
- Fixemer, T.; Remberger, K.; Bonkhoff, H. Differential expression of the estrogen receptor beta (ERβ) in human prostate tissue, premalignant changes, and in primary, metastatic, and recurrent prostatic adenocarcinoma. Prostate 2002, 54, 79–87. [Google Scholar] [CrossRef]
- Kuiper, G.G.; Enmark, E.; Pelto-Huikko, M.; Nilsson, S.; Gustafsson, J.A. Cloning of a novel receptor expressed in rat prostate and ovary. Proc. Natl. Acad. Sci. USA 1996, 93, 5925–5930. [Google Scholar] [CrossRef]
- Andersson, S.; Sundberg, M.; Pristovsek, N.; Ibrahim, A.; Jonsson, P.; Katona, B.; Clausson, C.-M.; Zieba, A.; Ramström, M.; Söderberg, O.; et al. Insufficient antibody validation challenges oestrogen receptor beta research. Nat. Commun. 2017, 8, 15840. [Google Scholar] [CrossRef]
- Nelson, A.W.; Groen, A.J.; Miller, J.L.; Warren, A.Y.; Holmes, K.A.; Tarulli, G.A.; Tilley, W.D.; Katzenellenbogen, B.S.; Hawse, J.R.; Gnanapragasam, V.J.; et al. Comprehensive assessment of estrogen receptor beta antibodies in cancer cell line models and tissue reveals critical limitations in reagent specificity. Mol. Cell. Endocrinol. 2017, 440, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-T.; Ouyang, B.; Ho, S.-M.; Leung, Y.-K. Differential expression of estrogen receptor beta isoforms in prostate cancer through interplay between transcriptional and translational regulation. Mol. Cell. Endocrinol. 2013, 376, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Leung, Y.-K.; Mak, P.; Hassan, S.; Ho, S.-M. Estrogen receptor (ER)-β isoforms: A key to understanding ER-β signaling. Proc. Natl. Acad. Sci. USA 2006, 103, 13162–13167. [Google Scholar] [CrossRef]
- de Cremoux, P.; Rosenberg, D.; Goussard, J.; Bremont-Weil, C.; Tissier, F.; Tran-Perennou, C.; Groussin, L.; Bertagna, X.; Bertherat, J.; Raffin-Sanson, M.-L. Expression of progesterone and estradiol receptors in normal adrenal cortex, adrenocortical tumors, and primary pigmented nodular adrenocortical disease. Endocr.-Relat. Cancer 2008, 15, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Ise, K.; Hata, S.; Yamashita, S.; Ito, A.; Sasano, H.; Nakamura, Y. Clinicopathological Significance of Estrogen Receptor β and Estrogen Synthesizing/Metabolizing Enzymes in Urothelial Carcinoma of Urinary Bladder. Pathol. Oncol. Res. 2021, 27, 589649. [Google Scholar] [CrossRef]
- Huang, D.; Berglund, M.; Damdimopoulos, A.; Antonson, P.; Lindskog, C.; Enblad, G.; Amini, R.-M.; Okret, S. Sex- and Female Age-Dependent Differences in Gene Expression in Diffuse Large B-Cell Lymphoma—Possible Estrogen Effects. Cancers 2023, 15, 1298. [Google Scholar] [CrossRef]
- Gul, Z.G.; Liaw, C.W.; Mehrazin, R. Gender Differences in Incidence, Diagnosis, Treatments, and Outcomes in Clinically Localized Bladder and Renal Cancer. Urology 2020, 151, 176–181. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, C.; Zhou, J.; Fang, H.; Wang, J. Overexpression of estrogen receptor β inhibits cellular functions of human hepatic stellate cells and promotes the anti-fibrosis effect of calycosin via inhibiting STAT3 phosphorylation. BMC Pharmacol. Toxicol. 2022, 23, 77. [Google Scholar] [CrossRef]
- Kim, M.; Kim, S.-J.; Ha, S.Y.; Xu, Z.; Han, Y.; Jee, H.-G.; Cho, S.W.; Park, Y.J.; Lee, K.E. BRAFV600E Mutation Enhances Estrogen-Induced Metastatic Potential of Thyroid Cancer by Regulating the Expression of Estrogen Receptors. Endocrinol. Metab. 2022, 37, 879–890. [Google Scholar] [CrossRef]
- Ogłuszka, M.; Orzechowska, M.; Jędroszka, D.; Witas, P.; Bednarek, A.K. Evaluate Cutpoints: Adaptable continuous data distribution system for determining survival in Kaplan-Meier estimator. Comput. Methods Programs Biomed. 2019, 177, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Sahara, S.; Aoto, M.; Eguchi, Y.; Imamoto, N.; Yoneda, Y.; Tsujimoto, Y. Acinus is a caspase-3-activated protein required for apoptotic chromatin condensation. Nature 1999, 401, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Welch, M.D.; DePace, A.H.; Verma, S.; Iwamatsu, A.; Mitchison, T.J. The human Arp2/3 complex is composed of evolutionarily conserved subunits and is localized to cellular regions of dynamic actin filament assembly. J. Cell Biol. 1997, 138, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Schrank, B.R.; Aparicio, T.; Li, Y.; Chang, W.; Chait, B.T.; Gundersen, G.G.; Gottesman, M.E.; Gautier, J. Nuclear ARP2/3 drives DNA break clustering for homology-directed repair. Nature 2018, 559, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Good, P.J.; Chen, Q.; Warner, S.J.; Herring, D.C. A Family of human RNA-binding proteins related to the drosophila bruno translational regulator. J. Biol. Chem. 2000, 275, 28583–28592. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.W.; Herrera-Abreu, M.T.; Rohn, J.L.; Racine, V.; Tajadura, V.; Suryavanshi, N.; Bechtel, S.; Wiemann, S.; Baum, B.; Ridley, A.J. Identification and characterization of a set of conserved and new regulators of cytoskeletal organization, cell morphology and migration. BMC Biol. 2011, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Chfr Regulates a Mitotic Stress Pathway through Its RING-Finger Domain with Ubiquitin Ligase Activity. Available online: https://pubmed.ncbi.nlm.nih.gov/11912157/ (accessed on 10 March 2024).
- Complete Primary Structure of the Human α 3(IV) Collagen Chain. Coexpression of the α 3(IV) and α 4(IV) Collagen Chains in Human Tissues. Available online: https://pubmed.ncbi.nlm.nih.gov/8083201/ (accessed on 4 February 2024).
- Maeshima, Y.; Colorado, P.C.; Torre, A.; Holthaus, K.A.; Grunkemeyer, J.A.; Ericksen, M.B.; Hopfer, H.; Xiao, Y.; Stillman, I.E.; Kalluri, R. Distinct antitumor properties of a type IV collagen domain derived from basement membrane. J. Biol. Chem. 2000, 275, 21340–21348. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhao, Y.; Hu, N.; Zhao, J.; Bai, Y. lncRNA KIAA0125 functions as a tumor suppressor modulating growth and metastasis of colorectal cancer via Wnt/β-catenin pathway. Cell Biol. Int. 2019, 43, 1463–1470. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, Y.; Weissman, S.M.; Bothwell, A.L. BXMAS1 identifies a cluster of homologous genes differentially expressed in B cells. Biochem. Biophys. Res. Commun. 2001, 285, 830–837. [Google Scholar] [CrossRef]
- Nagase, T.; Ishikawa, K.-I.; Suyama, M.; Kikuno, R.; Hirosawa, M.; Miyajima, N.; Tanaka, A.; Kotani, H.; Nomura, N.; Ohara, O. Prediction of the coding sequences of unidentified human genes. XIII. The complete sequences of 100 new cDNA clones from brain which code for large proteins in vitro. DNA Res. 1999, 6, 63–70. [Google Scholar] [CrossRef]
- Wende, H.; Volz, A.; Ziegler, A. Extensive gene duplications and a large inversion characterize the human leukocyte receptor cluster. Immunogenetics 2000, 51, 703–713. [Google Scholar] [CrossRef]
- Toufektchan, E.; Lejour, V.; Durand, R.; Giri, N.; Draskovic, I.; Bardot, B.; Laplante, P.; Jaber, S.; Alter, B.P.; Londono-Vallejo, J.-A.; et al. Germline mutation of MDM4, a major p53 regulator, in a familial syndrome of defective telomere maintenance. Sci. Adv. 2020, 6, eaay3511. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Dai, M.-S.; Lu, S.Z.; Xu, Y.; Luo, Z.; Zhao, Y.; Lu, H. 14-3-3γ binds to MDMX that is phosphorylated by UV-activated Chk1, resulting in p53 activation. EMBO J. 2006, 25, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- Jensen, L.; Nielsen, M.; Justesen, J.; Hansen, L. Assignment1 of human NADH dehydrogenase (ubiquinone) 1 beta subcomplex 3 (NDUFB3) and of its four pseudogenes to human chromosomes 2q31.3, 1p13.3→p13.1, 9q32→q34.1, 14q22.3→q23.1 and 14q32.2 by radiation hybrid mapping. Cytogenet. Genome Res. 2001, 93, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Chojnacka, K.J.; Elancheliyan, P.; Mussulini, B.H.M.; Mohanraj, K.; Callegari, S.; Gosk, A.; Banach, T.; Góral, T.; Szczepanowska, K.; Rehling, P.; et al. Ovarian carcinoma immunoreactive antigen–like protein 2 (OCIAD2) is a novel complex III–specific assembly factor in mitochondria. Mol. Biol. Cell 2022, 33, ar29. [Google Scholar] [CrossRef] [PubMed]
- Díaz, E.; Pfeffer, S.R. TIP47: A cargo selection device for mannose 6-phosphate receptor trafficking. Cell 1998, 93, 433–443. [Google Scholar] [CrossRef]
- Liu, R.; Lee, J.-H.; Li, J.; Yu, R.; Tan, L.; Xia, Y.; Zheng, Y.; Bian, X.-L.; Lorenzi, P.L.; Chen, Q.; et al. Choline kinase alpha 2 acts as a protein kinase to promote lipolysis of lipid droplets. Mol. Cell 2021, 81, 2722–2735.e9. [Google Scholar] [CrossRef] [PubMed]
- Strubin, M.; Newell, J.W.; Matthias, P. OBF-1, a novel B cell-specific coactivator that stimulates immunoglobulin promoter activity through association with octamer-binding proteins. Cell 1995, 80, 497–506. [Google Scholar] [CrossRef]
- Luo, Y.; Roeder, R.G. Cloning, functional characterization, and mechanism of action of the B-cell-specific transcriptional coactivator OCA-B. Mol. Cell. Biol. 1995, 15, 4115–4124. [Google Scholar] [CrossRef]
- Munro, S.; Ceulemans, H.; Bollen, M.; Diplexcito, J.; Cohen, P.T. A novel glycogen-targeting subunit of protein phosphatase 1 that is regulated by insulin and shows differential tissue distribution in humans and rodents. FEBS J. 2005, 272, 1478–1489. [Google Scholar] [CrossRef]
- Ridley, A.J.; Paterson, H.F.; Johnston, C.L.; Diekmann, D.; Hall, A. The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell 1992, 70, 401–410. [Google Scholar] [CrossRef]
- Reijnders, M.R.; Ansor, N.M.; Kousi, M.; Yue, W.W.; Tan, P.L.; Clarkson, K.; Clayton-Smith, J.; Corning, K.; Jones, J.R.; Lam, W.W.; et al. RAC1 Missense Mutations in Developmental Disorders with Diverse Phenotypes. Am. J. Hum. Genet. 2017, 101, 466–477. [Google Scholar] [CrossRef]
- Zhao, J.; Mialki, R.K.; Wei, J.; Coon, T.A.; Zou, C.; Chen, B.B.; Mallampalli, R.K.; Zhao, Y. SCF E3 ligase F-box protein complex SCFFBXL19regulates cell migration by mediating Rac1 ubiquitination and degradation. FASEB J. 2013, 27, 2611–2619. [Google Scholar] [CrossRef]
- Shimizu, H.; Toma-Fukai, S.; Saijo, S.; Shimizu, N.; Kontani, K.; Katada, T.; Shimizu, T. Structure-based analysis of the guanine nucleotide exchange factor SmgGDS reveals armadillo-repeat motifs and key regions for activity and GTPase binding. J. Biol. Chem. 2017, 292, 13441–13448. [Google Scholar] [CrossRef]
- Vikis, H.G.; Stewart, S.; Guan, K.-L. SmgGDS displays differential binding and exchange activity towards different Ras isoforms. Oncogene 2002, 21, 2425–2432. [Google Scholar] [CrossRef]
- Wallis, S.S.; Ventimiglia, L.N.; Otigbah, E.; Infante, E.; Cuesta-Geijo, M.A.; Kidiyoor, G.R.; Carbajal, M.A.; Fleck, R.A.; Foiani, M.; Garcia-Manyes, S.; et al. The ESCRT machinery counteracts Nesprin-2G-mediated mechanical forces during nuclear envelope repair. Dev. Cell 2021, 56, 3192–3202.e8. [Google Scholar] [CrossRef]
- Humphray, S.J.; Oliver, K.; Hunt, A.R.; Plumb, R.W.; Loveland, J.E.; Howe, K.L.; Andrews, T.D.; Searle, S.; Hunt, S.E.; Scott, C.E.; et al. DNA sequence and analysis of human chromosome. Nature 2004, 429, 369–374. [Google Scholar] [CrossRef]
- Kayagaki, N.; Yan, M.; Seshasayee, D.; Wang, H.; Lee, W.; French, D.M.; Grewal, I.S.; Cochran, A.G.; Gordon, N.C.; Yin, J.; et al. BAFF/BLyS receptor 3 binds the B cell survival factor BAFF ligand through a discrete surface loop and promotes processing of NF-κB. Immunity 2002, 17, 515–524. [Google Scholar] [CrossRef]
- Yan, M.; Brady, J.R.; Chan, B.; Lee, W.P.; Hsu, B.; Harless, S.; Cancro, M.; Grewal, I.S.; Dixit, V.M. Identification of a novel receptor for B lymphocyte stimulator that is mutated in a mouse strain with severe B cell deficiency. Curr. Biol. 2001, 11, 1547–1552. [Google Scholar] [CrossRef]
- Brandenberger, R.; Wei, H.; Zhang, S.; Lei, S.; Murage, J.; Fisk, G.J.; Li, Y.; Xu, C.; Fang, R.; Guegler, K.; et al. Transcriptome characterization elucidates signaling networks that control human ES cell growth and differentiation. Nat. Biotechnol. 2004, 22, 707–716. [Google Scholar] [CrossRef]
- Structures and Chromosomal Localizations of Two Human Genes Encoding Synaptobrevins 1 and 2. Available online: https://pubmed.ncbi.nlm.nih.gov/1976629/ (accessed on 4 February 2024).
- Isenmann, S.; Khew-Goodall, Y.; Gamble, J.; Vadas, M.; Wattenberg, B.W. A splice-isoform of vesicle-associated membrane protein-1 (VAMP-1) contains a mitochondrial targeting signal. Mol. Biol. Cell 1998, 9, 1649–1660. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-C.; Jeng, K.-S.; Lai, M.M.C. Zinc Finger-Containing Cellular Transcription Corepressor ZBTB25 Promotes Influenza Virus RNA Transcription and Is a Target for Zinc Ejector Drugs. J. Virol. 2017, 91, e00842-17. [Google Scholar] [CrossRef] [PubMed]
- Hanein, S.; Martin, E.; Boukhris, A.; Byrne, P.; Goizet, C.; Hamri, A.; Benomar, A.; Lossos, A.; Denora, P.; Fernandez, J.; et al. Identification of the SPG15 gene, encoding spastizin, as a frequent cause of complicated autosomal-recessive spastic paraplegia, including Kjellin syndrome. Am. J. Hum. Genet. 2008, 82, 992–1002. [Google Scholar] [CrossRef] [PubMed]
- Drewes, T.; Clairmont, A.; Klein-Hitpass, L.; Ryffel, G.U. Estrogen-inducible derivatives of hepatocyte nuclear factor-4, hepatocyte nuclear factor-3 and liver factor B1 are differently affected by pure and partial antiestrogens. Eur. J. Biochem. 1994, 225, 441–448. [Google Scholar] [CrossRef]
- A Shaw, J.; Udokang, K.; Mosquera, J.; Chauhan, H.; Jones, J.L.; A Walker, R. Oestrogen receptors alpha and beta differ in normal human breast and breast carcinomas. J. Pathol. 2002, 198, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Hartman, J.; Edvardsson, K.; Lindberg, K.; Zhao, C.; Williams, C.; Ström, A.; Gustafsson, J.-A. Tumor repressive functions of estrogen receptor β in SW480 colon cancer cells. Cancer Res. 2009, 69, 6100–6106. [Google Scholar] [CrossRef]
- Yu, C.-P.; Ho, J.-Y.; Huang, Y.-T.; Cha, T.-L.; Sun, G.-H.; Yu, D.-S.; Chang, F.-W.; Chen, S.-P.; Hsu, R.-J. Estrogen inhibits renal cell carcinoma cell progression through estrogen Receptor-β activation. PLoS ONE 2013, 8, e56667. [Google Scholar] [CrossRef]
- Chou, C.-K.; Chi, S.-Y.; Hung, Y.-Y.; Yang, Y.-C.; Fu, H.-C.; Wang, J.-H.; Chen, C.-C.; Kang, H.-Y. Decreased Expression of Estrogen Receptors Is Associated with Tumorigenesis in Papillary Thyroid Carcinoma. Int. J. Mol. Sci. 2022, 23, 1015. [Google Scholar] [CrossRef]
- Alvaro, D.; Barbaro, B.; Franchitto, A.; Onori, P.; Glaser, S.S.; Alpini, G.; Francis, H.; Marucci, L.; Sterpetti, P.; Ginanni-Corradini, S.; et al. Estrogens and insulin-like growth factor 1 modulate neoplastic cell growth in human Cholangiocarcinoma. Am. J. Pathol. 2006, 169, 877–888. [Google Scholar] [CrossRef]
- Al-Khyatt, W.; Tufarelli, C.; Khan, R.; Iftikhar, S.Y. Selective oestrogen receptor antagonists inhibit oesophageal cancer cell proliferation in vitro. BMC Cancer 2018, 18, 121. [Google Scholar] [CrossRef]
- Shatalova, E.G.; Klein-Szanto, A.J.; Devarajan, K.; Cukierman, E.; Clapper, M.L. Estrogen and cytochrome P450 1B1 contribute to both early- and late-stage head and neck carcinogenesis. Cancer Prev. Res. 2011, 4, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Wang, W.; Cheng, Y.; Lin, J.; Wang, M. Clinicopathological and Prognostic Significance of Klotho and Estrogen Receptors Expression in Human Hepatocellular Carcinoma. Turk. J. Gastroenterol. 2021, 32, 828–836. [Google Scholar] [CrossRef]
- The Expression of ERβ2, Bcl-xl and Bax in Non-Small Cell Lung Cancer and Associated with Prognosis. Available online: https://pubmed.ncbi.nlm.nih.gov/31966894/ (accessed on 17 February 2024).
- van Weelden, W.J.; Reijnen, C.; Küsters-Vandevelde, H.V.; Bulten, J.; Bult, P.; Leung, S.; Visser, N.C.; Santacana, M.; Bronsert, P.; Hirschfeld, M.; et al. The cutoff for estrogen and progesterone receptor expression in endometrial cancer revisited: A European Network for Individualized Treatment of Endometrial Cancer collaboration study. Hum. Pathol. 2020, 109, 80–91. [Google Scholar] [CrossRef]
- Trovik, J.; Wik, E.; Werner, H.M.; Krakstad, C.; Helland, H.; Vandenput, I.; Njolstad, T.S.; Stefansson, I.M.; Marcickiewicz, J.; Tingulstad, S.; et al. Hormone receptor loss in endometrial carcinoma curettage predicts lymph node metastasis and poor outcome in prospective multicentre trial. Eur. J. Cancer 2013, 49, 3431–3441. [Google Scholar] [CrossRef]
- Seeliger, H.; Pozios, I.; Assmann, G.; Zhao, Y.; Müller, M.H.; Knösel, T.; Kreis, M.E.; Bruns, C.J. Expression of estrogen receptor beta correlates with adverse prognosis in resected pancreatic adenocarcinoma. BMC Cancer 2018, 18, 1049. [Google Scholar] [CrossRef]
- Song, W.; He, D.; Chen, Y.; Yeh, C.; Hsu, I.; Huang, Q.; Zhang, X.; Chang, L.S.; Zuo, L.; Chen, J.; et al. Targeting newly identified ERβ/TGF-β1/SMAD3 signals with the FDA-approved anti-estrogen Faslodex or an ERβ selective antagonist in renal cell carcinoma. Mol. Oncol. 2018, 12, 2055–2071. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Xin, B.; Pang, H.; Han, L.; Shen, W.; Zhao, Z.; Duan, L.; Cao, P.; Liu, L.; Zhang, H. Downregulation of estrogen receptor β inhibits lung adenocarcinoma cell growth. Oncol. Rep. 2019, 41, 2967–2974. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, H.K.; Shin, B.K. Expression of female sex hormone receptors and its relation to clinicopathological characteristics and prognosis of lung adenocarcinoma. J. Pathol. Transl. Med. 2020, 54, 103–111. [Google Scholar] [CrossRef]
- Panza, S.; Santoro, M.; De Amicis, F.; Morelli, C.; Passarelli, V.; D’aquila, P.; Giordano, F.; Cione, E.; Passarino, G.; Bellizzi, D.; et al. Estradiol via estrogen receptor beta influences ROS levels through the transcriptional regulation of SIRT3 in human seminoma TCam-2 cells. Tumor Biol. 2017, 39, 1010428317701642. [Google Scholar] [CrossRef]
- Loss of Estrogen Receptor Beta Expression at Cancer Front Correlates with Tumor Progression and Poor Prognosis of Gallbladder Cancer. Available online: https://pubmed.ncbi.nlm.nih.gov/15492781/ (accessed on 27 February 2024).
- Dong, W.; Li, J.; Zhang, H.; Huang, Y.; He, L.; Wang, Z.; Shan, Z.; Teng, W. Altered Expression of Estrogen Receptor β2 is Associated with Different Biological Markers and Clinicopathological Factors in Papillary Thyroid Cancer. Available online: https://pubmed.ncbi.nlm.nih.gov/26261608/ (accessed on 27 February 2024).
- Goto, T.; Miyamoto, H. The Role of Estrogen Receptors in Urothelial Cancer. Front. Endocrinol. 2021, 12, 643870. [Google Scholar] [CrossRef]
- Dalal, H.; Dahlgren, M.; Gladchuk, S.; Brueffer, C.; Gruvberger-Saal, S.K.; Saal, L.H. Clinical associations of ESR2 (estrogen receptor beta) expression across thousands of primary breast tumors. Sci. Rep. 2022, 12, 4696. [Google Scholar] [CrossRef] [PubMed]
- Jensen, E.V.; Cheng, G.; Palmieri, C.; Saji, S.; Mäkelä, S.; Van Noorden, S.; Wahlström, T.; Warner, M.; Coombes, R.C.; Gustafsson, J. Estrogen receptors and proliferation markers in primary and recurrent breast cancer. Proc. Natl. Acad. Sci. USA 2001, 98, 15197–15202. [Google Scholar] [CrossRef] [PubMed]
- Skliris, G.P.; Leygue, E.; Curtis-Snell, L.; Watson, P.H.; Murphy, L.C. Expression of oestrogen receptor-β in oestrogen receptor-α negative human breast tumours. Br. J. Cancer 2006, 95, 616–626. [Google Scholar] [CrossRef]
- The Cervical Malignant Cells Display a down Regulation of ER-α But Retain the ER-β Expression. Available online: https://pubmed.ncbi.nlm.nih.gov/23923078/ (accessed on 13 February 2024).
- Fadiel, A.; Choi, S.D.; Park, B.; Kim, T.-H.; Buldo-Licciardi, J.; Ahmadi, M.; Arslan, A.; Mittal, K.; Naftolin, F. Expression of Ezrin and Estrogen Receptors During Cervical Carcinogenesis. Reprod. Sci. 2016, 24, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.-M.; Qi, Y.-J.; Jiang, Q.; Ma, Y.-F.; Wang, L.-D. Relevance of serum estradiol and estrogen receptor beta expression from a high-incidence area for esophageal squamous cell carcinoma in China. Med. Oncol. 2010, 28, 188–193. [Google Scholar] [CrossRef]
- Neto, C.P.D.O.; Brito, H.O.; GIL DA Costa, R.M.; Brito, L.M.O. Is There a Role for Sex Hormone Receptors in Head-and-neck Cancer? Links with HPV Infection and Prognosis. Anticancer Res. 2021, 41, 3707–3716. [Google Scholar] [CrossRef]
- Grsic, K.; Opacic, I.L.; Sitic, S.; Perisa, M.M.; Suton, P.; Sarcevic, B. The prognostic significance of estrogen receptor β in head and neck squamous cell carcinoma. Oncol. Lett. 2016, 12, 3861–3865. [Google Scholar] [CrossRef]
- Atef, A.; El-Rashidy, M.A.; Elzayat, S.; Kabel, A.M. The prognostic value of sex hormone receptors expression in laryngeal carcinoma. Tissue Cell 2019, 57, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, H.; Shi, J.; Fan, Q.; Zhou, Z.; Tang, X.; Wang, Y.; Liu, Y. ERβ overexpression may not be a direct prognostic factor in patients with NSCLC: A meta-analysis. Int. J. Biol. Markers 2022, 37, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Gu, D.; Qian, P. Prognostic value of ESR2 expression on adjuvant chemotherapy in completely resected NSCLC. PLoS ONE 2020, 15, e0243891. [Google Scholar] [CrossRef]
- Stabile, L.P.; Dacic, S.; Land, S.R.; Lenzner, D.E.; Dhir, R.; Acquafondata, M.; Landreneau, R.J.; Grandis, J.R.; Siegfried, J.M. Combined Analysis of Estrogen Receptor β-1 and Progesterone Receptor Expression Identifies Lung Cancer Patients with Poor Outcome. Clin. Cancer Res. 2011, 17, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hu, C.; Li, M.; An, J.; Zhou, W.; Guo, J.; Xiao, Y. Estrogen receptor beta promotes lung cancer invasion via increasing CXCR4 expression. Cell Death Dis. 2022, 13, 70. [Google Scholar] [CrossRef]
- De Stefano, I.; Zannoni, G.F.; Prisco, M.G.; Fagotti, A.; Tortorella, L.; Vizzielli, G.; Mencaglia, L.; Scambia, G.; Gallo, D. Cytoplasmic expression of estrogen receptor beta (ERβ) predicts poor clinical outcome in advanced serous ovarian cancer. Gynecol. Oncol. 2011, 122, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Konduri, S.; Schwarz, R.E. Estrogen Receptor β/α ratio predicts response of pancreatic cancer cells to estrogens and phytoestrogens. J. Surg. Res. 2007, 140, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-Y.; Wang, Y.-X.; Wang, L.; Qian, Z.-B.; Ji, M.-L. Cytoplasm estrogen receptor β5 as an improved prognostic factor in thymoma and thymic carcinoma progression. Oncol. Lett. 2015, 10, 2341–2346. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, H.; Suzuki, T.; Suzuki, S.; Moriya, T.; Kaneko, C.; Takizawa, T.; Sunamori, M.; Handa, M.; Kondo, T.; Sasano, H. Sex steroid hormone receptors in human thymoma. J. Clin. Endocrinol. Metab. 2003, 88, 2309–2317. [Google Scholar] [CrossRef]
- Taves, M.D.; Ashwell, J.D. Effects of sex steroids on thymic epithelium and thymocyte development. Front. Immunol. 2022, 13, 975858. [Google Scholar] [CrossRef]
- Wang, X.; Lin, P.; Li, Y.; Chen, G.; Yang, H.; He, Y.; Li, Q.; Liu, R. Identification of potential agents for thymoma by integrated analyses of differentially expressed tumour-associated genes and molecular docking experiments. Exp. Ther. Med. 2019, 18, 2001–2014. [Google Scholar] [CrossRef]
- Ishibashi, H.; Suzuki, T.; Suzuki, S.; Moriya, T.; Kaneko, C.; Nakata, T.; Sunamori, M.; Handa, M.; Kondo, T.; Sasano, H. Estrogen inhibits cell proliferation through in situ production in human thymoma. Clin. Cancer Res. 2005, 11, 6495–6504. [Google Scholar] [CrossRef]
- Mimae, T.; Tsuta, K.; Takahashi, F.; Yoshida, A.; Kondo, T.; Murakami, Y.; Okada, M.; Takeuchi, M.; Asamura, H.; Tsuda, H. Steroid receptor expression in thymomas and thymic carcinomas. Cancer 2011, 117, 4396–4405. [Google Scholar] [CrossRef]
- Collins, F.; MacPherson, S.; Brown, P.; Bombail, V.; Williams, A.R.; A Anderson, R.; Jabbour, H.N.; Saunders, P.T. Expression of oestrogen receptors, ERα, ERβ, and ERβ variants, in endometrial cancers and evidence that prostaglandin F may play a role in regulating expression of ERα. BMC Cancer 2009, 9, 330. [Google Scholar] [CrossRef]
- Lang, T.J. Estrogen as an immunomodulator. Clin. Immunol. 2004, 113, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Expression and Localization of Estrogen Receptors in Human Renal Cell Carcinoma and Their Clinical Significance. Available online: https://pubmed.ncbi.nlm.nih.gov/31938447/ (accessed on 16 February 2024).
- Ding, J.; Yeh, C.-R.; Sun, Y.; Lin, C.; Chou, J.; Ou, Z.; Chang, C.; Qi, J.; Yeh, S. Estrogen receptor β promotes renal cell carcinoma progression via regulating LncRNA HOTAIR-miR-138/200c/204/217 associated CeRNA network. Oncogene 2018, 37, 5037–5053. [Google Scholar] [CrossRef] [PubMed]
- Modarres, P.; Farsani, F.M.; Nekouie, A.A.; Vallian, S. Meta-analysis of gene signatures and key pathways indicates suppression of JNK pathway as a regulator of chemo-resistance in AML. Sci. Rep. 2021, 11, 12485. [Google Scholar] [CrossRef]
- Rodríguez-Cid, J.R.; García-Acevedo, O.; Benjamin-Contreras, J.; Bonilla-Molina, D.; Flores-Mariñelarena, R.R.; Martínez-Barrera, L.; Alatorre-Alexander, J.A.; Sanchez-Ríos, C.P.; Flores-Soto, M.d.R.; Santillan-Doherty, P.J.; et al. Expression of estrogen receptor beta (ERβ) and its prognostic value in pleural mesothelioma. J. Thorac. Dis. 2019, 11, 1456–1464. [Google Scholar] [CrossRef]
- Pinton, G.; Brunelli, E.; Murer, B.; Puntoni, R.; Puntoni, M.; Fennell, D.A.; Gaudino, G.; Mutti, L.; Moro, L. Estrogen receptor-β affects the prognosis of human malignant mesothelioma. Cancer Res. 2009, 69, 4598–4604. [Google Scholar] [CrossRef]
- Pillai, K.; Pourgholami, M.H.; Chua, T.C.; Morris, D.L. Oestrogen receptors are prognostic factors in malignant peritoneal mesothelioma. J. Cancer Res. Clin. Oncol. 2013, 139, 987–994. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Dong, W.; Li, J.; Zhang, H.; Shan, Z.; Teng, W. Differential expression patterns and clinical significance of estrogen receptor-α and β in papillary thyroid carcinoma. BMC Cancer 2014, 14, 383. [Google Scholar] [CrossRef]
- Jackson, S.S.; Pfeiffer, R.M.; Gabbi, C.; Anderson, L.; Gadalla, S.M.; Koshiol, J. Menopausal hormone therapy and risk of biliary tract cancers. Hepatology 2021, 75, 309–321. [Google Scholar] [CrossRef]
- Kilanczyk, E.; Ruminkiewicz, D.; Banales, J.M.; Milkiewicz, P.; Milkiewicz, M. DHEA Protects Human Cholangiocytes and Hepatocytes against Apoptosis and Oxidative Stress. Cells 2022, 11, 1038. [Google Scholar] [CrossRef]
- Alvaro, D.; Alpini, G.; Onori, P.; Franchitto, A.; Glaser, S.; Le Sage, G.; Folli, F.; Attili, A.; Gaudio, E. Alfa and beta estrogen receptors and the biliary tree. Mol. Cell. Endocrinol. 2002, 193, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, M.R.; Fanous, E.; Thaker, R.; Flory, M.J.; Seetharamu, N.; Dhar, M.; Starr, A.; Strange, T.J. Expression patterns and clinical significance of estrogen receptor in non-small cell lung cancer. Pathol.-Res. Pr. 2023, 241, 154298. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Xu, Y.; Zhang, T.; Han, Q.; Li, L.; Liu, M.; Li, N.; Shao, G. Cisplatin-activated ERβ/DCAF8 positive feedback loop induces chemoresistance in non-small cell lung cancer via PTEN/Akt axis. Drug Resist. Updat. 2023, 71, 101014. [Google Scholar] [CrossRef] [PubMed]
- Sareddy, G.R.; Nair, B.C.; Gonugunta, V.K.; Zhang, Q.-G.; Brenner, A.; Brann, D.W.; Tekmal, R.R.; Vadlamudi, R.K. Therapeutic significance of estrogen receptor β agonists in gliomas. Mol. Cancer Ther. 2012, 11, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Batistatou, A.; Stefanou, D.; Goussia, A.; Arkoumani, E.; Papavassiliou, A.G.; Agnantis, N.J. Estrogen receptor beta (ERβ) is expressed in brain astrocytic tumors and declines with dedifferentiation of the neoplasm. J. Cancer Res. Clin. Oncol. 2004, 130, 405–410. [Google Scholar] [CrossRef]
- Batistatou, A.; A Kyzas, P.; Goussia, A.; Arkoumani, E.; Voulgaris, S.; Polyzoidis, K.; Agnantis, N.J.; Stefanou, D. Estrogen receptor beta (ERβ) protein expression correlates with BAG-1 and prognosis in brain glial tumours. J. Neuro-Oncol. 2005, 77, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Zellweger, T.; Stürm, S.; Rey, S.; Zlobec, I.; Gsponer, J.R.; A Rentsch, C.; Terracciano, L.M.; Bachmann, A.; Bubendorf, L.; Ruiz, C. Estrogen receptor β expression and androgen receptor phosphorylation correlate with a poor clinical outcome in hormone-naïve prostate cancer and are elevated in castration-resistant disease. Endocr.-Relat. Cancer 2013, 20, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Bonkhoff, H.; Berges, R. The Evolving role of oestrogens and their receptors in the development and progression of prostate cancer. Eur. Urol. 2009, 55, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Grindstad, T.; Skjefstad, K.; Andersen, S.; Ness, N.; Nordby, Y.; Al-Saad, S.; Fismen, S.; Donnem, T.; Khanehkenari, M.R.; Busund, L.-T.; et al. Estrogen receptors α and β and aromatase as independent predictors for prostate cancer outcome. Sci. Rep. 2016, 6, 33114. [Google Scholar] [CrossRef]
- Leung, Y.-K.; Lam, H.-M.; Wu, S.; Song, D.; Levin, L.; Cheng, L.; Wu, C.-L.; Ho, S.-M. Estrogen receptor β2 and β5 are associated with poor prognosis in prostate cancer, and promote cancer cell migration and invasion. Endocr.-Relat. Cancer 2010, 17, 675–689. [Google Scholar] [CrossRef]
- Barzon, L.; Masi, G.; Pacenti, M.; Trevisan, M.; Fallo, F.; Remo, A.; Martignoni, G.; Montanaro, D.; Pezzi, V.; Palù, G. Expression of aromatase and estrogen receptors in human adrenocortical tumors. Virchows Arch. 2007, 452, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Eyster, K.M. The Estrogen Receptors: An Overview from Different Perspectives. Methods Mol. Biol. 2016, 1366, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yakimchuk, K.; Iravani, M.; Hasni, M.S.; Rhönnstad, P.; Nilsson, S.; Jondal, M.; Okret, S. Effect of ligand-activated estrogen receptor β on lymphoma growth in vitro and in vivo. Leukemia 2011, 25, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Zhang, Y.; Sun, Y.; Chen, J.; Chang, C.; Wang, X.; Yeh, S. ERβ-Mediated Alteration of circATP2B1 and miR-204-3p Signaling Promotes Invasion of Clear Cell Renal Cell Carcinoma. Cancer Res. 2018, 78, 2550–2563. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.Y.; Guo, J.L.; Jiang, Z.N.; Xie, S.D.; Shen, J.Y.; Wang, L.B. Prognostic role of estrogen receptor α and estrogen receptor β in gastric cancer. Ann. Surg. Oncol. 2010, 17, 2503–2509. [Google Scholar] [CrossRef] [PubMed]
- Mak, P.; Leav, I.; Pursell, B.; Bae, D.; Yang, X.; Taglienti, C.A.; Gouvin, L.M.; Sharma, V.M.; Mercurio, A.M. ERβ impedes prostate cancer EMT by destabilizing HIF-1α and inhibiting VEGF-mediated snail nuclear localization: Implications for gleason grading. Cancer Cell 2010, 17, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduct. Target. Ther. 2020, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Bae, T.; Hallis, S.P.; Kwak, M.-K. Hypoxia, oxidative stress, and the interplay of HIFs and NRF2 signaling in cancer. Exp. Mol. Med. 2024, 56, 501–514. [Google Scholar] [CrossRef] [PubMed]
- Dey, P.; Jonsson, P.; Hartman, J.; Williams, C.; Ström, A.; Gustafsson, J. Estrogen receptors β1 and β2 have opposing roles in regulating proliferation and bone metastasis genes in the prostate cancer cell line PC3. Mol. Endocrinol. 2012, 26, 1991–2003. [Google Scholar] [CrossRef]
- Rawłuszko-Wieczorek, A.A.; Lipowicz, J.; Nowacka, M.; Ostrowska, K.; Pietras, P.; Blatkiewicz, M.; Ruciński, M.; Jagodziński, P.P.; Nowicki, M. Estrogen receptor β affects hypoxia response in colorectal cancer cells. Biochim. Biophys. Acta-Mol. Basis Dis. 2024, 1870, 166894. [Google Scholar] [CrossRef]
- Refaat, B.; Aslam, A.; Idris, S.; Almalki, A.H.; Alkhaldi, M.Y.; Asiri, H.A.; Almaimani, R.A.; Mujalli, A.; Minshawi, F.; Alamri, S.A.; et al. Profiling estrogen, progesterone, and androgen receptors in colorectal cancer in relation to gender, menopausal status, clinical stage, and tumour sidedness. Front. Endocrinol. 2023, 14, 1187259. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Yan, Y.; Huang, Q.; Zheng, D. MDMX in Cancer: A Partner of p53 and a p53-Independent Effector. Biol. Targets Ther. 2024, 18, 61–78. [Google Scholar] [CrossRef] [PubMed]
- Toledo, F.; Wahl, G.M. MDM2 and MDM4: p53 regulators as targets in anticancer therapy. Int. J. Biochem. Cell Biol. 2007, 39, 1476–1482. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Lozano, G. Molecular Pathways: Targeting Mdm2 and Mdm4 in Cancer Therapy. Clin. Cancer Res. 2013, 19, 34–41. [Google Scholar] [CrossRef]
- Zhao, H.; Xie, Y.-Z.; Xing, R.; Sun, M.; Chi, F.; Zeng, Y.-C. MDMX is a prognostic factor for non-small cell lung cancer and regulates its sensitivity to cisplatin. Cell. Oncol. 2017, 40, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Shiba-Ishii, A.; Kim, Y.; Noguchi, M.; Sakamoto, N. Ovarian carcinoma immunoreactive antigen domain 2 controls mitochondrial apoptosis in lung adenocarcinoma. Cancer Sci. 2021, 112, 5114–5126. [Google Scholar] [CrossRef]
- Chen, T.-F.; Hao, H.-F.; Zhang, Y.; Chen, X.-Y.; Zhao, H.-S.; Yang, R.; Li, P.; Qiu, L.-X.; Sang, Y.-H.; Xu, C.; et al. HBO1 induces histone acetylation and is important for non-small cell lung cancer cell growth. Int. J. Biol. Sci. 2022, 18, 3313–3323. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhao, Q.; Wang, Z.; Xu, F.; Liu, Y.; Guo, Y.; Li, C.; Liu, T.; Zhao, Y.; Tang, X.; et al. Hepatocellular carcinoma cells downregulate NADH:Ubiquinone Oxidoreductase Subunit B3 to maintain reactive oxygen species homeostasis. Hepatol. Commun. 2024, 8, e0395. [Google Scholar] [CrossRef]
- Relationship of Intracellular Proteolysis with CAP1 and Cofilin1 in Non-Small-Cell Lung Cancer. Available online: https://pubmed.ncbi.nlm.nih.gov/34148878/ (accessed on 3 March 2024).
- Li, Y.; Wang, H.; Pan, Y.; Wang, S.; Zhang, Z.; Zhou, H.; Xu, M.; Liu, X. Identification of bicalutamide resistance-related genes and prognosis prediction in patients with prostate cancer. Front. Endocrinol. 2023, 14, 1125299. [Google Scholar] [CrossRef]
- Sousa-Squiavinato, A.C.M.; Vasconcelos, R.I.; Gehren, A.S.; Fernandes, P.V.; de Oliveira, I.M.; Boroni, M.; Morgado-Díaz, J.A. Cofilin-1, LIMK1 and SSH1 are differentially expressed in locally advanced colorectal cancer and according to consensus molecular subtypes. Cancer Cell Int. 2021, 21, 1–15. [Google Scholar] [CrossRef]
- Zheng, K.-W.; Zhang, C.-H.; Wu, W.; Zhu, Z.; Gong, J.-P.; Li, C.-M. FNBP4 is a Potential Biomarker Associated with Cuproptosis and Promotes Tumor Progression in Hepatocellular Carcinoma. Int. J. Gen. Med. 2023, 16, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-T.; Chang, Y.-L.; Shih, J.-Y.; Lee, Y.-C. The significance of estrogen receptor β in 301 surgically treated non–small cell lung cancers. J. Thorac. Cardiovasc. Surg. 2005, 130, 979–986. [Google Scholar] [CrossRef] [PubMed]
- Maki, M.; JeongMin, H.; Nakagawa, T.; Kawai, H.; Sakamoto, N.; Sato, Y.; Noguchi, M. Aberrant OCIAD2 demethylation in lung adenocarcinoma is associated with outcome. Pathol. Int. 2022, 72, 496–505. [Google Scholar] [CrossRef]
- King, S.J.; Nowak, K.; Suryavanshi, N.; Holt, I.; Shanahan, C.M.; Ridley, A.J. Nesprin-1 and nesprin-2 regulate endothelial cell shape and migration. Cytoskeleton 2014, 71, 423–434. [Google Scholar] [CrossRef] [PubMed]
- Warren, D.T.; Tajsic, T.; Mellad, J.A.; Searles, R.; Zhang, Q.; Shanahan, C.M. Novel nuclear nesprin-2 variants tether active extracellular signal-regulated MAPK1 and MAPK2 at promyelocytic leukemia protein nuclear bodies and act to regulate smooth muscle cell proliferation. J. Biol. Chem. 2010, 285, 1311–1320. [Google Scholar] [CrossRef]
- The ACIN1 Gene Is Hypermethylated in Early Stage Lung Adenocarcinoma. Available online: https://pubmed.ncbi.nlm.nih.gov/17409846/ (accessed on 2 March 2024).
- Chen, Y.-S.; Liu, C.-W.; Lin, Y.-C.; Tsai, C.-Y.; Yang, C.-H.; Lin, J.-C. The SRSF3-MBNL1-Acin1 circuit constitutes an emerging axis to lessen DNA fragmentation in colorectal cancer via an alternative splicing mechanism. Neoplasia 2020, 22, 702–713. [Google Scholar] [CrossRef]
- Valentin-Vega, Y.A.; Barboza, J.A.; Chau, G.P.; El-Naggar, A.K.; Lozano, G. High levels of the p53 inhibitor MDM4 in head and neck squamous carcinomas. Hum. Pathol. 2007, 38, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Men, X.; Zhu, W. Silencing of Perilipin 3 Inhibits Lung Adenocarcinoma Cell Immune Resistance by Regulating the Transcription of PD-L1 Through c-Myc. Immunol. Investig. 2023, 52, 815–831. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Y.; Yang, Y.; Huang, J.; Sun, Y.; Zhang, X.; Kong, X. A novel iTreg-related signature for prognostic prediction in lung adenocarcinoma. Cancer Sci. 2023, 115, 109–124. [Google Scholar] [CrossRef]
- Zhou, H.; Brekman, A.; Zuo, W.-L.; Ou, X.; Shaykhiev, R.; Agosto-Perez, F.J.; Wang, R.; Walters, M.S.; Salit, J.; Strulovici-Barel, Y.; et al. POU2AF1 Functions in the Human Airway Epithelium To Regulate Expression of Host Defense Genes. J. Immunol. 2016, 196, 3159–3167. [Google Scholar] [CrossRef]
- Luck, K.; Kim, D.-K.; Lambourne, L.; Spirohn, K.; Begg, B.E.; Bian, W.; Brignall, R.; Cafarelli, T.; Campos-Laborie, F.J.; Charloteaux, B.; et al. A reference map of the human binary protein interactome. Nature 2020, 580, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Haenig, C.; Atias, N.; Taylor, A.K.; Mazza, A.; Schaefer, M.H.; Russ, J.; Riechers, S.-P.; Jain, S.; Coughlin, M.; Fontaine, J.-F.; et al. Interactome Mapping Provides a Network of Neurodegenerative Disease Proteins and Uncovers Widespread Protein Aggregation in Affected Brains. Cell Rep. 2020, 32, 108050. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Yang, X.; Khan, A.; Yu, X.; Zhang, H.; Han, S.; Habulieti, X.; Sun, Y.; Wang, R.; Zhang, X. Panoramic variation analysis of a family with neurodevelopmental disorders caused by biallelic loss-of-function variants in TMEM141, DDHD2, and LHFPL. Front. Med. 2023, 18, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Woodland, R.T.; Fox, C.J.; Schmidt, M.R.; Hammerman, P.S.; Opferman, J.T.; Korsmeyer, S.J.; Hilbert, D.M.; Thompson, C.B. Multiple signaling pathways promote B lymphocyte stimulator–dependent B-cell growth and survival. Blood 2008, 111, 750–760. [Google Scholar] [CrossRef] [PubMed]
- Jellusova, J.; Miletic, A.V.; Cato, M.H.; Lin, W.-W.; Hu, Y.; Bishop, G.A.; Shlomchik, M.J.; Rickert, R.C. Context-specific BAFF-R signaling by the NF-κB and PI3K pathways. Cell Rep. 2013, 5, 1022–1035. [Google Scholar] [CrossRef]
- Wensveen, F.M.; Slinger, E.; van Attekum, M.H.; Brink, R.; Eldering, E. Antigen-affinity controls pre-germinal center B cell selection by promoting Mcl-1 induction through BAFF receptor signaling. Sci. Rep. 2016, 6, 35673. [Google Scholar] [CrossRef]
- Zeng, Q.; Qin, S.; Zhang, H.; Liu, B.; Qin, J.; Wang, X.; Zhang, R.; Liu, C.; Dong, X.; Zhang, S.; et al. Rapamycin attenuates BAFF-extended proliferation and survival via disruption of mTORC1/2 signaling in normal and neoplastic B-lymphoid cells. J. Cell. Physiol. 2017, 233, 516–529. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Huang, Z.; Li, P.; Sun, Z.; Hou, X.; Li, Z.; Sang, R.; Guo, Z.; Wu, S.; Cao, Y. Investigating the efficacy and mechanisms of Jinfu’ an decoction in treating non-small cell lung cancer using network pharmacology and in vitro and in vivo experiments. J. Ethnopharmacol. 2024, 321, 117518. [Google Scholar] [CrossRef]
- Marei, H.; Malliri, A. Rac1 in human diseases: The therapeutic potential of targeting Rac1 signaling regulatory mechanisms. Small GTPases 2016, 8, 139–163. [Google Scholar] [CrossRef]
- Park, B.-W.; Kim, K.-S.; Heo, M.-K.; Yang, W.-I.; Kim, S.I.; Kim, J.-H.; Kim, G.E.; Lee, K.S. The changes of estrogen receptor-β variants expression in breast carcinogenesis: Decrease of estrogen receptor-β2 expression is the key event in breast cancer development. J. Surg. Oncol. 2006, 93, 504–510. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio cancer genomics portal: An open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.E.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal. 2013, 6, pl1. [Google Scholar] [CrossRef]
- de Bruijn, I.; Kundra, R.; Mastrogiacomo, B.; Tran, T.N.; Sikina, L.; Mazor, T.; Li, X.; Ochoa, A.; Zhao, G.; Lai, B.; et al. Analysis and Visualization of Longitudinal Genomic and Clinical Data from the AACR Project GENIE Biopharma Collaborative in cBioPortal. Cancer Res. 2023, 83, 3861–3867. [Google Scholar] [CrossRef]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Proteomics. Tissue-Based Map of the Human Proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Uhlén, M.; Björling, E.; Agaton, C.; Szigyarto, C.A.-K.; Amini, B.; Andersen, E.; Andersson, A.-C.; Angelidou, P.; Asplund, A.; Asplund, C.; et al. A human protein atlas for normal and cancer tissues based on antibody proteomics. Mol. Cell. Proteom. MCP 2005, 4, 1920–1932. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Fu, J.; Zeng, Z.; Cohen, D.; Li, J.; Chen, Q.; Li, B.; Liu, X.S. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020, 48, W509–W514. [Google Scholar] [CrossRef]
- Li, T.; Fan, J.; Wang, B.; Traugh, N.; Chen, Q.; Liu, J.S.; Li, B.; Liu, X.S. TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res. 2017, 77, e108–e110. [Google Scholar] [CrossRef]
- Lasley, R.D.; Mentzer, R.M. Preconditioning and its potential role in myocardial protection during cardiac surgery. J. Card. Surg. 1995, 10, 349–353. [Google Scholar] [CrossRef]
- Ru, B.; Wong, C.N.; Tong, Y.; Zhong, J.Y.; Zhong, S.S.W.; Wu, W.C.; Chu, K.C.; Wong, C.Y.; Lau, C.Y.; Chen, I.; et al. TISIDB: An integrated repository portal for tumor–immune system interactions. Bioinformatics 2019, 35, 4200–4202. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [PubMed]
- Mootha, V.K.; Lindgren, C.M.; Eriksson, K.-F.; Subramanian, A.; Sihag, S.; Lehar, J.; Puigserver, P.; Carlsson, E.; Ridderstråle, M.; Laurila, E.; et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 2003, 34, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Mobashir, M.; Turunen, S.P.; Izhari, M.A.; Ashankyty, I.M.; Helleday, T.; Lehti, K. An Approach for Systems-Level Understanding of Prostate Cancer from High-Throughput Data Integration to Pathway Modeling and Simulation. Cells 2022, 11, 4121. [Google Scholar] [CrossRef] [PubMed]
- Nassar, L.R.; Barber, G.P.; Benet-Pagès, A.; Casper, J.; Clawson, H.; Diekhans, M.; Fischer, C.; Gonzalez, J.N.; Hinrichs, A.S.; Lee, B.T.; et al. The UCSC Genome Browser database: 2023 update. Nucleic Acids Res. 2022, 51, D1188–D1195. [Google Scholar] [CrossRef] [PubMed]




| Gene | Description | Source |
|---|---|---|
| ACIN1 [14q11.2] | Apoptotic chromatin condensation inducer 1 regulates chromatin condensation after activation by caspase-3 during apoptosis. Additionally, it may be involved in mRNA regulation after splicing. | [33] |
| ARPC2 [2q35] | Actin-related protein 2/3 complex subunit 2 mediates actin polymerization in the nucleus and therefore regulates transcription and homolog recombination in response to DNA damage. | [34,35] |
| CELF6 [15q23] | CUGBP elav-like family member 6 regulates pre-mRNA alternative splicing and may be involved in mRNA editing and translation. | [36] |
| CFL1 [11q13.1] | Cofilin 1 carries out F-actin depolymerization, thus regulating cell morphology through cytoskeletal organization in epithelial cells. | [37,38] |
| COL4A3 [2q36.3] | The collagen α-3(IV) chain is one of the structural components of glomerular basement membranes. Tumstatin within this domain presents anti-tumor activity. | [39,40] |
| FAM30A [14q32.33] | Family with sequence similarity to 30 member A and may be a non-coding RNA. | [41] |
| FCRL5 [1q23.1] | Fc receptor-like 5 is most likely involved in B-cell differentiation and may present an immunoregulatory role in marginal-zone B-cells. | [42] |
| FNBP4 [11p11.2] | Formin-binding protein 4 may be involved in the regulation of cytoskeletal dynamics during cell division and migration. | [43] |
| LENG8 [19q13.42] | Leukocyte receptor cluster member 8 encodes leukocyte-expressed receptors of the immunoglobulin superfamily. | [44] |
| MDM4 [1q32.1] | MDM4 regulator of p53 inhibits p53- and p73-mediated cell cycle arrest and apoptosis and inhibits the degradation of MDM2. | [45,46] |
| NDUFB3 [2q33.1] | NDAH–ubiquinone oxidoreductase subunit B3 is part of the electron transport chain of mitochondria on the inner membrane of the mitochondrion. | [47] |
| OCIAD2 [4p11] | Ovarian carcinoma immunoreactive antigen domain containing 2 in one of the mitochondrial respiratory chain complex assembly factors. | [48] |
| PLIN3 [19p13.3] | Perilipin 3 is a structural component of lipid droplets required for lipid storage in cells and is involved in mannose 6-phosphate receptor transport. | [49,50] |
| POU2AF1 [11q23.1] | POU class 2 homeobox associating factor 1 is a transcriptional coactivator associated with POU2F1/OCT1 or POU2F2/OCT2 complexes; likewise, it is essential for B-cells’ ability to respond to antigens. | [51,52] |
| PPP1R3E [14q11.2] | Protein phosphatase 1 regulatory subunit 3E is predicted to be involved in the positive regulation of the glycogen biosynthetic process and to be a part of the protein phosphatase type 1 complex. | [53] |
| RAC1 [7p22.1] | Rac family small GTPase 1 is a plasma membrane-associated protein binding to effector proteins involved in secretion, phagocytosis, migration, and differentiation. | [54,55,56] |
| RAP1GDS1 [4q23] | Rap1 GTPase-GDP dissociation stimulator 1 is a guanine nucleotide exchange factor in the GDP-GTP dissociation–binding sequence. | [57,58] |
| SYNE2 [14q23.2] | Spectrin repeat containing nuclear envelope protein 2 (Nesprin-2) is a component of the LInker of Nucleoskeleton and Cytoskeleton (LINC) and regulates the spatial organization of intracellular components. | [59] |
| TMEM141 [9q34.3] | Transmembrane protein 141 is predicted to be an integral component of the membrane. | [60] |
| TNFRSF13C [22q13.2] | Tumor necrosis factor receptor superfamily member 13C is a B-cell-specific receptor that promotes the survival of mature B-cells and the B-cell response. | [61,62] |
| TPTEP2 [22q13.1] | TPTE pseudogene 2 is a transmembrane phosphoinositide 3-phosphatase and tensin homolog 2 pseudogene. | [63] |
| VAMP1 [12p13.31] | Vesicle-associated membrane protein 1 is involved in the targeting and fusion of transport vesicles to their target membrane. | [64,65] |
| ZBTB25 [14q23.3] | Zinc finger- and BTB domain-containing 25 is predicted to be involved in the regulation of transcription by RNA polymerase II. | [66] |
| ZFYVE26 [14q24.1] | Zinc finger FYVE-type-containing 26 encodes protein targeted to membrane lipids through interaction with phospholipids in the membrane. | [67] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lipowicz, J.M.; Malińska, A.; Nowicki, M.; Rawłuszko-Wieczorek, A.A. Genes Co-Expressed with ESR2 Influence Clinical Outcomes in Cancer Patients: TCGA Data Analysis. Int. J. Mol. Sci. 2024, 25, 8707. https://doi.org/10.3390/ijms25168707
Lipowicz JM, Malińska A, Nowicki M, Rawłuszko-Wieczorek AA. Genes Co-Expressed with ESR2 Influence Clinical Outcomes in Cancer Patients: TCGA Data Analysis. International Journal of Molecular Sciences. 2024; 25(16):8707. https://doi.org/10.3390/ijms25168707
Chicago/Turabian StyleLipowicz, Julia Maria, Agnieszka Malińska, Michał Nowicki, and Agnieszka Anna Rawłuszko-Wieczorek. 2024. "Genes Co-Expressed with ESR2 Influence Clinical Outcomes in Cancer Patients: TCGA Data Analysis" International Journal of Molecular Sciences 25, no. 16: 8707. https://doi.org/10.3390/ijms25168707





