Abstract
The BTB (Broad-complex, tramtrack, and bric-a-brac) gene family, characterized by a highly conserved BTB domain, is implicated in a spectrum of biological processes, encompassing growth and development, as well as stress responses. Characterization and functional studies of BTB genes in poplar are still limited, especially regarding their response to hormones and biotic/abiotic stresses. In this study, we conducted an HMMER search in conjunction with BLASTp and identified 95 BTB gene models in Populus trichocarpa. Through domain motif and phylogenetic relationship analyses, these proteins were classified into eight families, NPH3, TAZ, Ankyrin, only BTB, BACK, Armadillo, TPR, and MATH. Collinearity analysis of poplar BTB genes with homologs in six other species elucidated evolutionary relationships and functional conservations. RNA-seq analysis of five tissues of poplar identified BTB genes as playing a pivotal role during developmental processes. Comprehensive RT-qPCR analysis of 11 BTB genes across leaves, roots, and xylem tissues revealed their responsive expression patterns under diverse hormonal and biotic/abiotic stress conditions, with varying degrees of regulation observed in the results. This study marks the first in-depth exploration of the BTB gene family in poplar, providing insights into the potential roles of BTB genes in hormonal regulation and response to stress.
1. Introduction
The BTB (Broad-complex, tramtrack, and bric-a-brac) protein was initially discovered in Drosophila, and the BTB proteins contain a highly conserved BTB domain of approximately 115 amino acids, also known as the POZ (Pox virus and zinc) finger domain [1]. The core structure of the BTB domain comprises an α-helix hairpin structure and a β-fold structure [2]. In addition to the core domain, variations exist in both the C-terminal and N-terminal extension regions of the BTB domain, which facilitate distinct functional roles for different types of BTB proteins. DNA-binding domains within BTB proteins enable gene expression regulation, while interactions between BTB domains and other domains enhance the protein functionality [1]. BTB proteins are classified into various subfamilies based on other domains, including BTB-only, BTB–BACK, BTB–Kelch, BTB–back–Kelch, BTB–Arm, BTB–TAZ, BTB–ANK, BTB–PHR and Rho–BTB [3]. Among these subfamilies, most members of the BTB–BACK subfamily contain both the BTB domain and Kelch repeats. The Kelch repeats in the BACK proteins form a beta propeller that can interact with actin filaments [4]. The Arm, a 42-amino acid protein–protein interaction motif, was first identified in the Drosophila segment polarity gene, armadillo [5], and a subset of these proteins is conserved across the eukaryotic kingdoms. BTB–TAZ proteins are presented in plants. BTB–ANK is a key transcriptional co-regulator of immunity and physiology in many plant species [2,3].
BTB proteins have been extensively investigated in animals [6,7]. In recent years, the BTB proteins have been identified in many plants, including Arabidopsis thaliana [8], rice [9], tomato [10], Paulownia fortunei [11], and sugar beet [12]. BTB/POZ proteins play a very important role in various processes of plant growth and development, such as plant morphogenesis [13,14,15], secondary metabolite synthesis [16,17], plant hormone signal transduction [18,19], and stress resistance [20,21]. In Arabidopsis, the BTB protein can interact with RBX1 and CUL3 to assemble the CRL3 (E3 ubiquitin ligase complex), demonstrating the role of BTB proteins in plant growth/development and response to stress through mediating protein ubiquitination [22]. In addition, the BTB protein in conjunction with the MATH domain protein, MATH–BTB, is involved in the abscisic acid (ABA) signaling pathways and abiotic stress response [23]. In Arabidopsis, six MATH–BTB proteins (called BPM1-6) are involved in regulating seed germination as well as stomatal opening and closing [19]. BTB domain-containing proteins, ATEOL1, ATEOL2, and ATETO1, participate in the regulation of ethylene biosynthesis in plants, while the HT1 protein negatively regulates hormone-mediated inhibition of Arabidopsis germination [24]. The ATBOP1 protein containing both the BTB and ANK domains play roles in plant morphogenesis control [12].
During plant growth and development, plants are highly susceptible to a range of biotic/abiotic stresses, such as salinity, drought, high temperatures, insect pests, fungal infections, and so on. These dynamic environmental factors adversely influence plant normal growth and development. BTB family proteins play a pivotal role in the regulation of stress-related genes, which is integral to the plant’s adaptive response to various stressors [21]. In Arabidopsis, the BTB–TAZ protein BT2 has been observed to be responsive to a range of environmental and hormonal cues [25]. Notably, light exposure, indole-3-acetic acid (IAA), ABA, and low temperatures have been found to suppress the expression of BT2, whereas cytokinins (CK), methyl jasmonate (Me-JA), and hydrogen peroxide (H2O2) stimulate its expression [20]. The NPR1 protein harbors two unique protein–protein interaction motifs, an ankyrin (ANK) repeat, and a BTB/POZ domain [26]. The Sl ANK protein, characterized by its ankyrin repeats, is pivotal in responding to both biotic and abiotic stresses and is indispensable for plant growth and development in tomato [27]. In Arabidopsis, the NPR1 protein with a BTB domain exerts regulatory effects on systemic acquired resistance (SAR) and induced systemic resistance (ISR), and this dual functionality is crucial for its role in the plant stress response [28]. Overexpression of the Arabidopsis NPR1 gene in tomatoes confers a wide-ranging resistance to various pathogens [29]. As a close relative of NPR1, the OsNPR1/NH1 protein in rice has been identified to enhance plant resistance to bacterial blight, which is caused by Xanthomonas oryzae pv [30]. These studies show the integral role of BTB proteins in modulating plant stress responses.
As the main worldwide afforestation tree species, especially in Northern China, poplar is the first perennial woody plant for which the whole genome sequence has been sequenced [31], and it has become a model plant for forest tree studies. Currently, the members of the BTB gene in poplar have not yet been reported. In this study, we conducted comprehensive bioinformatics and expression analysis of BTB gene in P. trichocarpa. A total of 95 PtrBTB genes were identified in the genome, and their chromosomal locations, gene pair relationships, phylogenetic comparison, and promoter cis-acting elements were analyzed. RT-qPCR was employed to investigate the expression pattern of the selected BTB genes in hormone treatments and biotic/abiotic stress conditions across different tissues. Altogether, our research details the BTB gene family in poplar and provides valuable information for the functional characterization of BTB proteins.
2. Results
2.1. Chromosomal Location and Homologous Gene Analysis of PtrBTB Genes
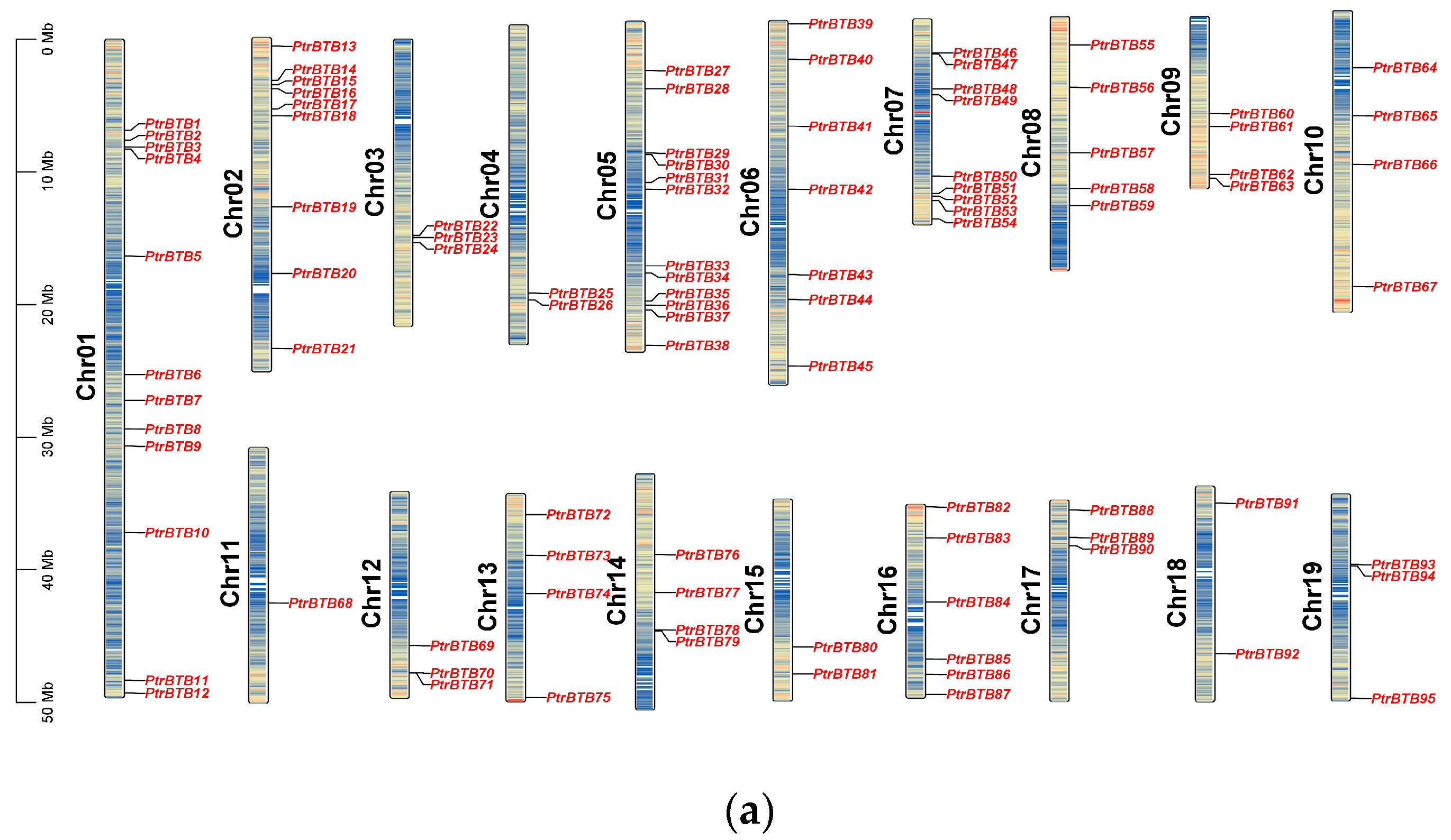
The PtrBTB gene family members were identified from the P. trichocarpa genome (v4.1) using the hidden Markov models algorithm, with the conserved BTB domain model (PF00651) as an inquiry. Using the BLASTp software (version 2.14.0) to compare with BTB proteins in Arabidopsis, a total of 95 PtrBTB gene models were identified in P. trichocarpa. For consistency, we detected the distribution of the identified BTB genes in chromosomes and named these genes as PtrBTB1 to PtrBTB95 according to their distributions and relative linear orders among their respective chromosomes. These genes were graphically aligned to the respective chromosomes, as depicted in Figure 1a. The resulting distribution map revealed an uneven distribution of 95 PtrBTB genes across the full spectrum of the 19 chromosomes. Among them, chromosome 11 was distinguished by having the lowest number of PtrBTB genes, featuring merely a single gene. Notably, chromosomes 1 and 5 each contained twelve PtrBTB genes, which remarkably exceeded the count on other chromosomes.
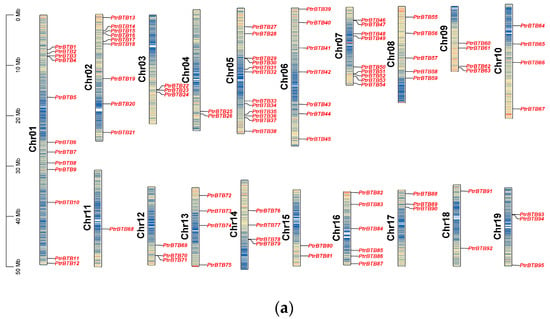
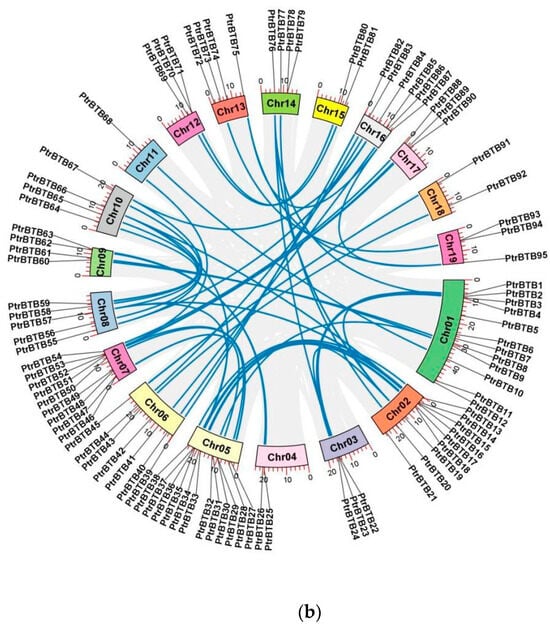
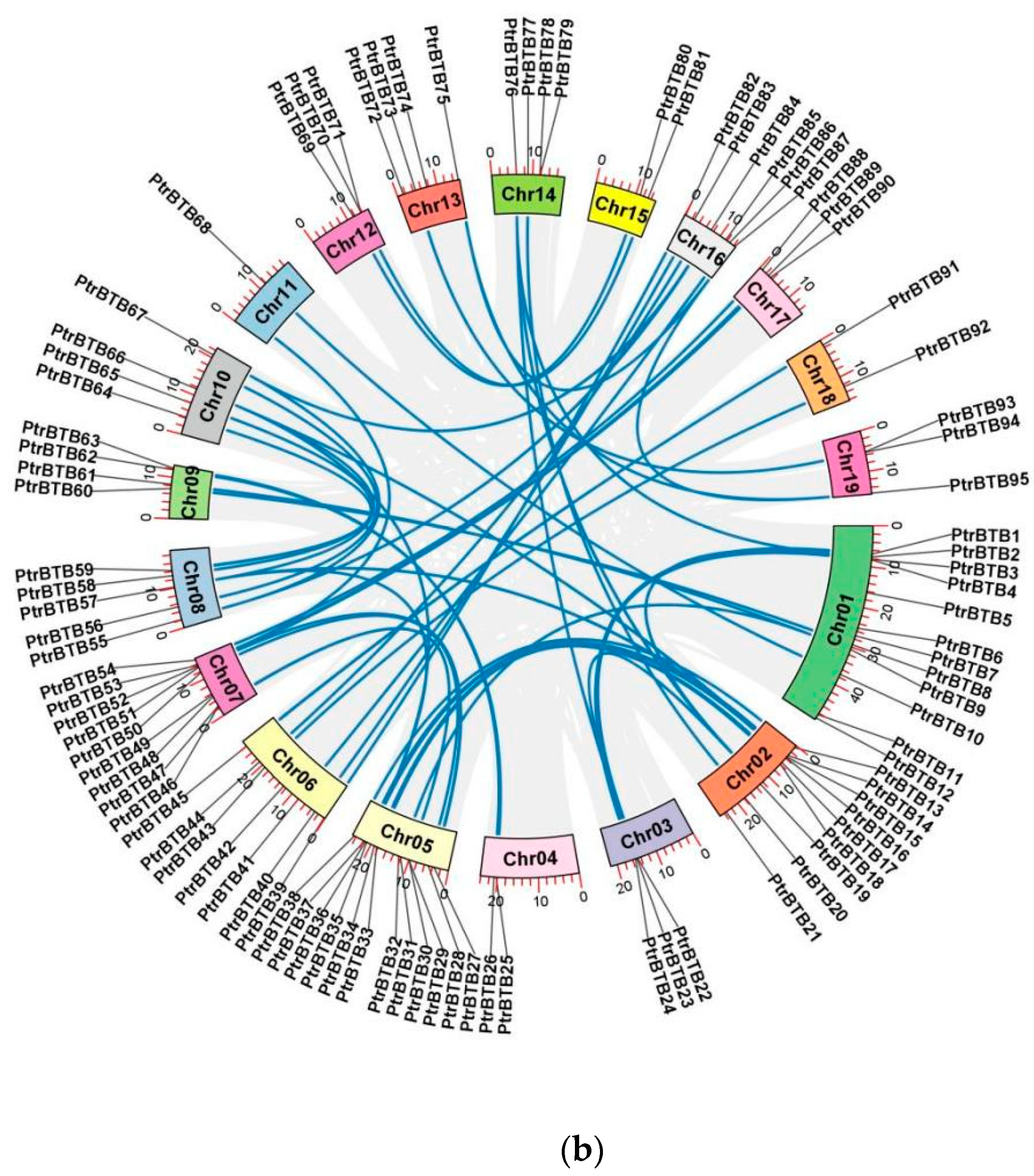
Figure 1.
Chromosome distribution and synteny analysis of BTB genes in P. trichocarpa. (a) The chromosomal mapping of PtrBTB genes across all 19 chromosomes of P. trichocarpa. (b) Comparative analysis of the distribution and syntenic relationships within the PtrBTB gene family, with syntenic gene pairs visually represented by blue connecting lines.
Detailed information of each PtrBTB protein, including gene name, gene ID, chromosome location (Chr), numbers of amino acid (AA), molecular weight (MW), theoretical isoelectric point (pI), and putative subcellular localization were presented in supplementary Table S1. These 95 PtrBTB proteins had divergent lengths, resulting in diverse isoelectric points and molecular weights. Subcellular localization analysis showed that most PtrBTB proteins were localized in both the nucleus and cytoplasm, and a few might be localized in other subcellular compartments, such as the chloroplast and mitochondria (Supplementary Table S1). No discernible pattern was observed linking the length of the chromosomes to the distribution density of PtrBTB genes along them. Utilizing the MCScanX software (version 2019), we successfully identified homologous genes within the PtrBTB gene family (Figure 1b). Our findings indicate that the BTB gene family in P. trichocarpa comprises 48 homologous gene pairs. Specifically, we observed six pairs of homologous genes between chromosomes 2 and 5 and five pairs between chromosomes 8 and 10. The non-synonymous (Ka)/synonymous (Ks) ratios of these gene pairs were both less than one, indicating that a strong purifying selection was experienced in the process of evolution (Supplementary Table S2). Among them, the Ka and Ks values for PtrBTB75 and PtrBTB87 of the NPH3 subfamily were the highest, while the Ankyrin subfamily’s PtrBTB16 and PtrBTB35 exhibited the maximal ratio of Ka/Ks.
2.2. Evolution Relationship of BTB Genes in Various Species
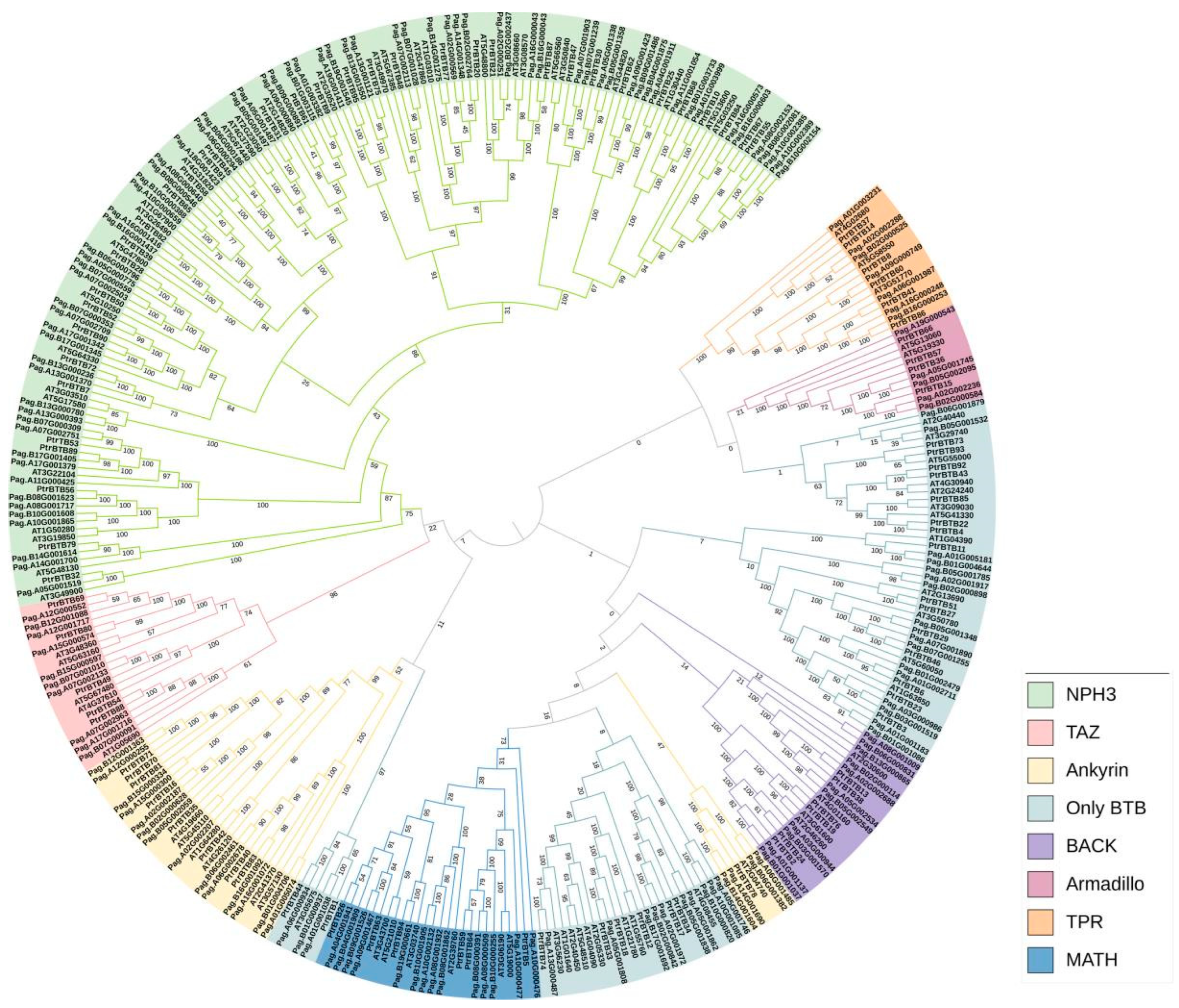
We also used the same above-mentioned method to identify the BTB family in a hybrid poplar P. alba × P. glandulosa and obtained 165 genes. A phylogenetic tree was constructed to explore the evolutionary relationships within the BTB gene family. Utilizing MEGA-X 10.2, we applied the Neighbor-Joining (NJ) method to analyze BTB proteins from P. trichocarpa (95 members), P. alba × P. glandulosa (165 members), and Arabidopsis (80 members) (Figure 2). The phylogenetic tree showed that a total of 340 BTB proteins were classified into eight subfamilies, NPH3, TAZ, Ankyrin, only BTB, BACK, Armadillo, TPR, and MATH. Among them, the NPH3 subfamily had the largest number, including 36 BTB proteins in P. trichocarpa, 71 in P. alba × P. glandulosa, and 32 in Arabidopsis. In contrast, the Armadillo subfamily was characterized by a lower number of BTB proteins, specifically four, each featuring eight Armadillo repeats at the C-terminus and a BTB domain at the N-terminus (Supplementary Figure S1). The number of BTB proteins between poplar and Arabidopsis varied greatly in the same subfamily. The poplar BTB proteins were distributed in all eight subfamilies, and most of the poplar BTB proteins had higher homologies with Arabidopsis, suggesting a conserved functional role across these two plant species.
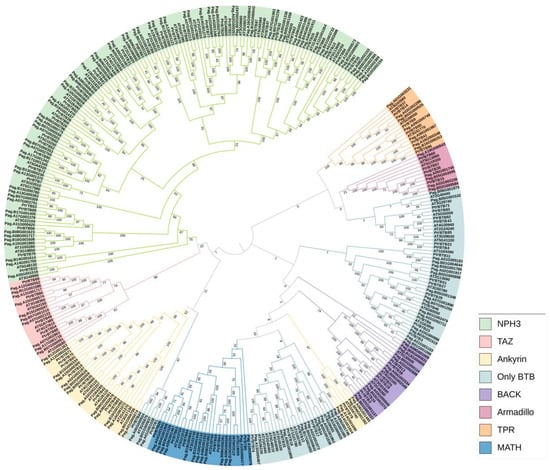
Figure 2.
A phylogenetic tree of BTB proteins from three plant species, P. trichocarpa, P. alba × P. glandulosa, and Arabidopsis. The analysis shows that 340 BTB proteins are classified into eight subgroups: MATH, Armadillo, BTB-only, TAZ, BACK, Ankyrin, TPR, and NPH3. The phylogenetic tree was constructed using MEGA-X 10.2 software using the neighbor-joining method at 1000 bootstrap replicates.
2.3. Gene Structure and Conserved Motif Analysis of PtrBTB Genes
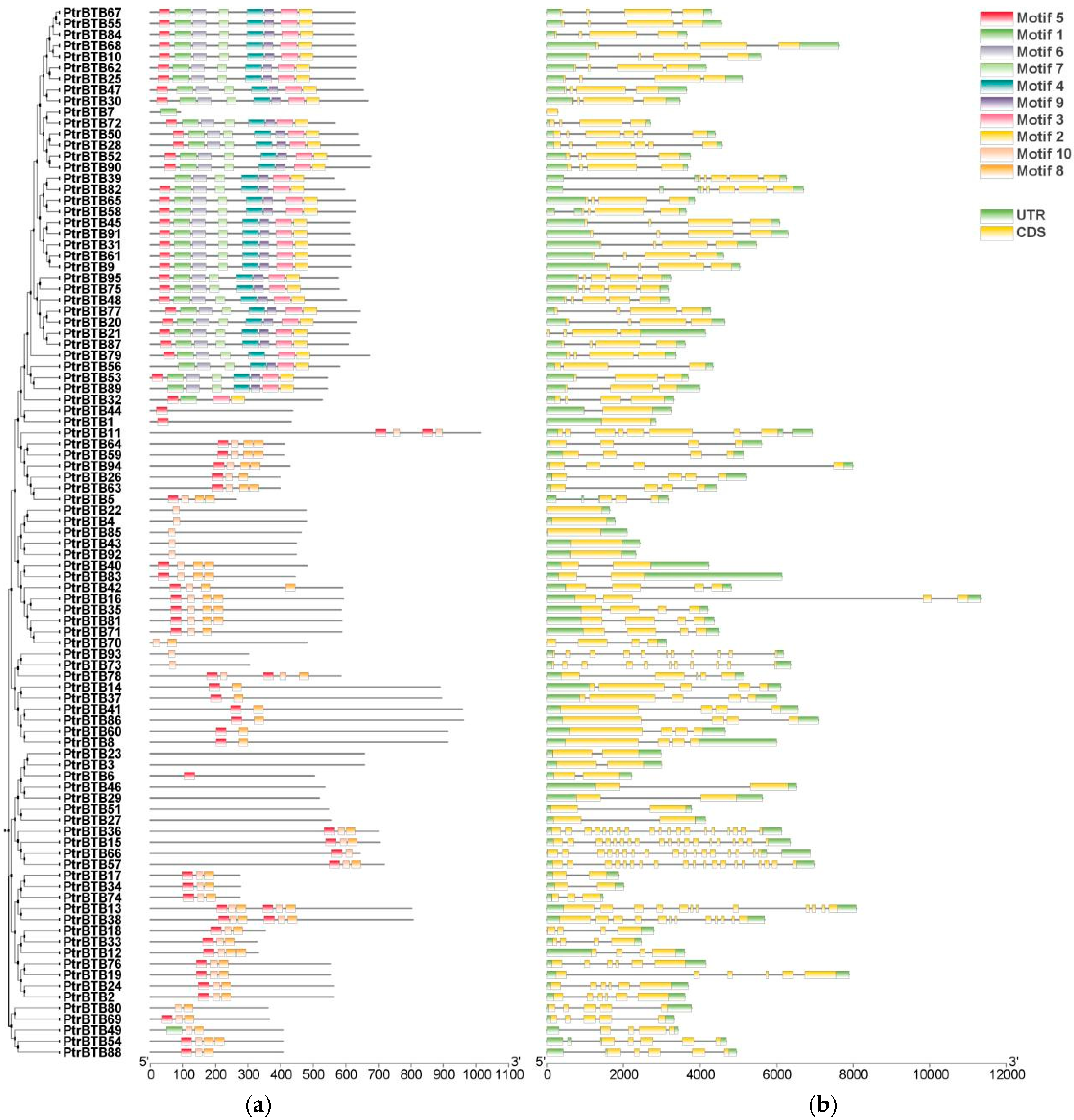
To gain insight into the identified 95 PtrBTB genes, we conducted the gene structure and motif distribution analyses using the MEME suite. Up to 10 motifs were obtained and were designated motif 1 through 10 (Figure 3a). The three most conserved motifs, consisting of 41, 50, and 50 amino acids, respectively, overlapped with the BTB domain sequence (Supplementary Figure S2). Conserved motif analysis revealed that various PtrBTB proteins exhibited similar types and arrangements of motif. Among them, almost all members contained motif 2, indicating that the structures of BTB are conserved. The largest subfamily NPH3 members contained eight motifs, including motifs 1, 2, 3, 4, 5, 6, 7, and 9. However, variations within the subfamilies suggest that genes among members of the same subfamily may exhibit functional diversity.
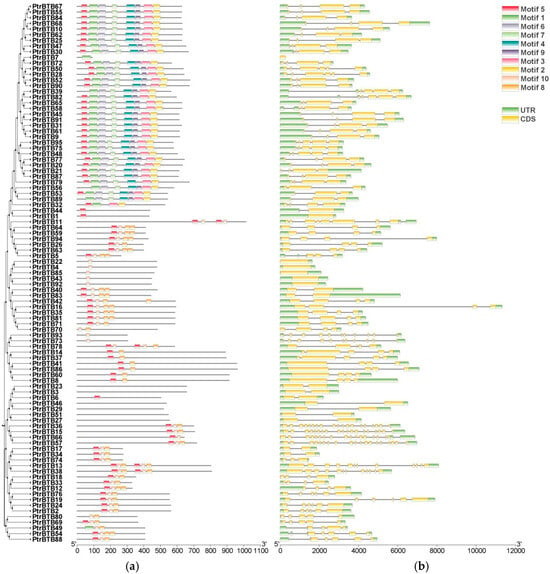
Figure 3.
Analysis of the conserved motifs and gene structures within the PtrBTB gene family. The clustering is performed according to the results of phylogenetic analysis. (a) Conserved motifs within PtrBTB proteins, identified by the MEME tool (version 5.5.6), yielding 10 distinct motifs, labeled as Motif 1 through 10. 100 aa is indicated by the scale bar. (b) Gene structure analysis of PtrBTB genes revealed the organization of UTR, intron, and exon regions, with UTR in green, exons in yellow, and introns in grey. The scale bar corresponds to 2 kb.
The exon–intron pattern is a crucial feature influencing gene function diversification; therefore, the gene structure of PtrBTB genes was characterized accordingly (Figure 3b). The analysis revealed variability in exon numbers ranging from 1 to 19 and intron numbers ranging from 0 to 18 within the PtrBTB gene family. Most members of the largest subfamily NPH3 contained 4 exons and 3 introns, whereas the Armadillo subfamily, which was characterized by the highest exon–intron count, contained 19 exons and 18 introns. The genes clustered into the same clade in the phylogenetic tree had similar gene structure, suggesting a relatively recent evolutionary relationship.
2.4. Cis-Acting Element Prediction of PtrBTB Genes
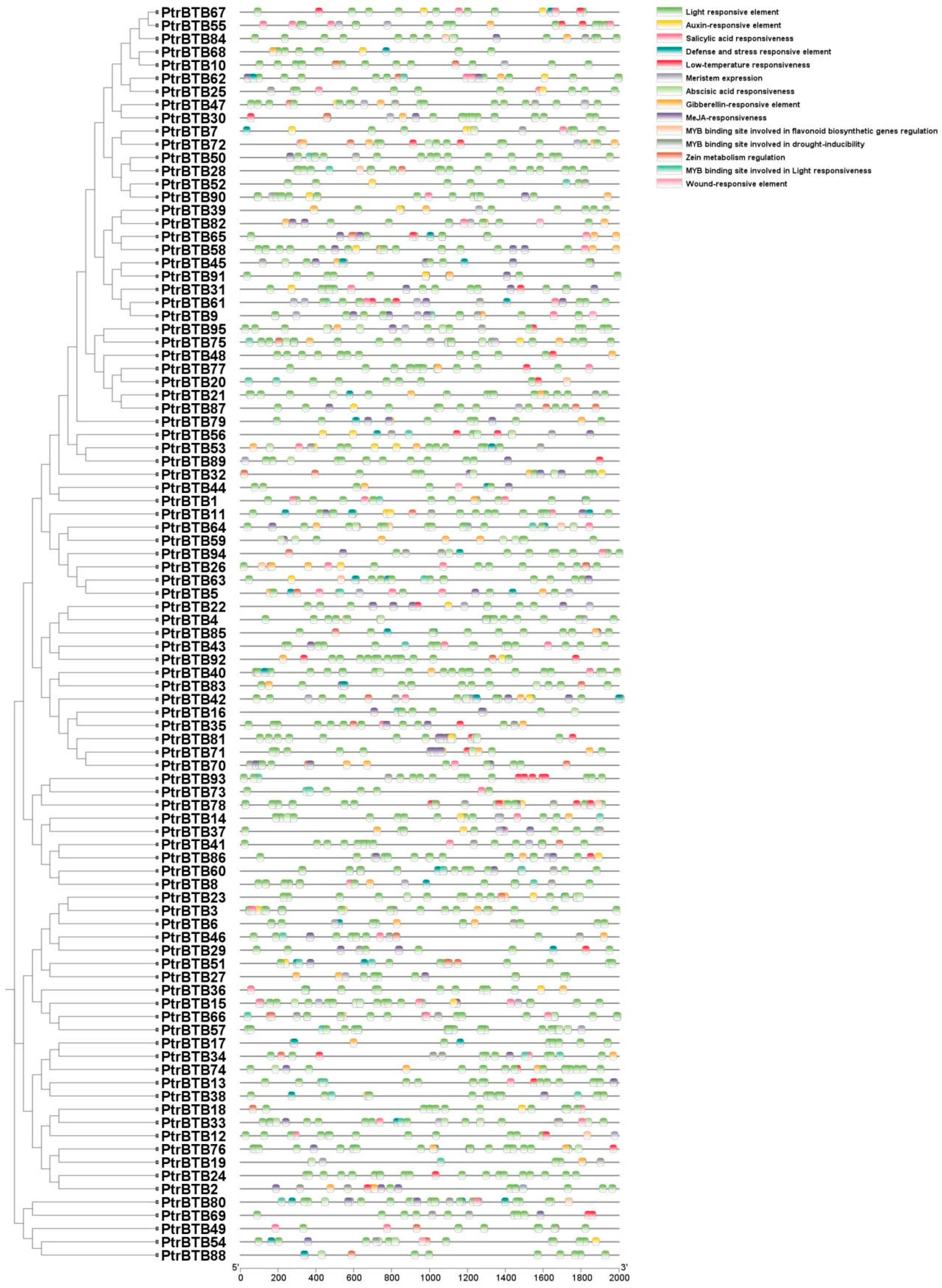
Cis-acting elements are necessary clues in predicting gene functions. By attaching to the cis-acting component of target genes in particular organic processes, transcription elements may affect the level of gene expression [32]. The promoter regions of various PtrBTB genes display a variety of cis-acting elements, predominantly categorized into two classes. One class of these elements was implicated in the regulation of hormonal pathways, encompassing ABA, methyl jasmonate (MeJA), gibberellin (GA), and IAA, which are crucial for plant growth and development. The other class was associated with the plant response to environmental stressors, such as exposure to light, low temperatures, arid conditions, and the activation of defense mechanisms. Among them, the largest numbers of cis-elements were the light-response elements, and they were found in all the PtrBTB family members. Notably, the numbers of ABA response elements (151) and MeJA response elements (200) were the second highest in the promoters of PtrBTB genes, implying their potential involvement in plant resistance mediated by ABA and MeJA signaling pathways. Additionally, 46 of these promoters displayed a responsive nature to salicylic acid (SA), while 53 showed a GA-responsive element, and 39 were found to be responsive to auxin. Specifically, among the 63 PtrBTB promoters, a significant presence of MYB binding sites that were crucial for drought-inducible elements was identified (Figure 4). Collectively, these findings suggest that PtrBTB proteins may play a role in coordinating both biotic and abiotic responses in plants through hormonal signaling pathways.
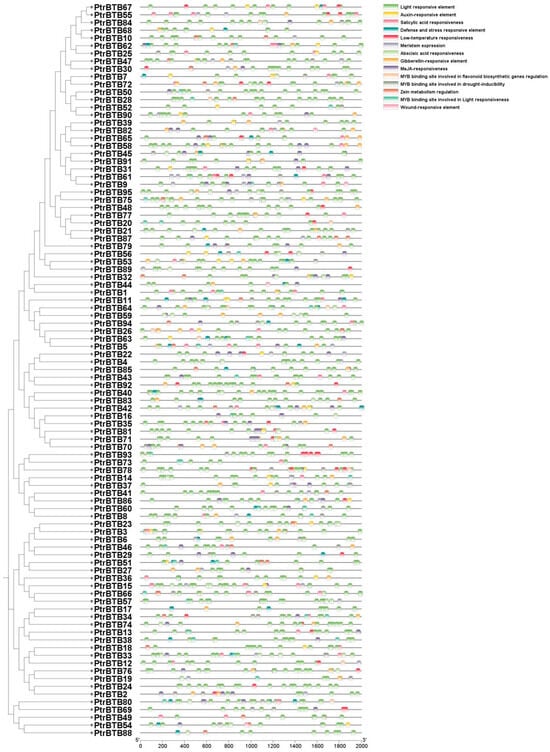
Figure 4.
Analysis of cis-elements in the 2000 bp upstream promoter of PtrBTB genes. The clustering is performed according to the results of phylogenetic analysis. Within the predicted promoter region of the PtrBTB genes, a total of 14 distinct regulatory motifs have been identified. These motifs are distinguished by a spectrum of colors, each representing a unique class of transcription factor binding sites.
2.5. Collinear Analysis of the PtrBTB Gene Family in Different Species
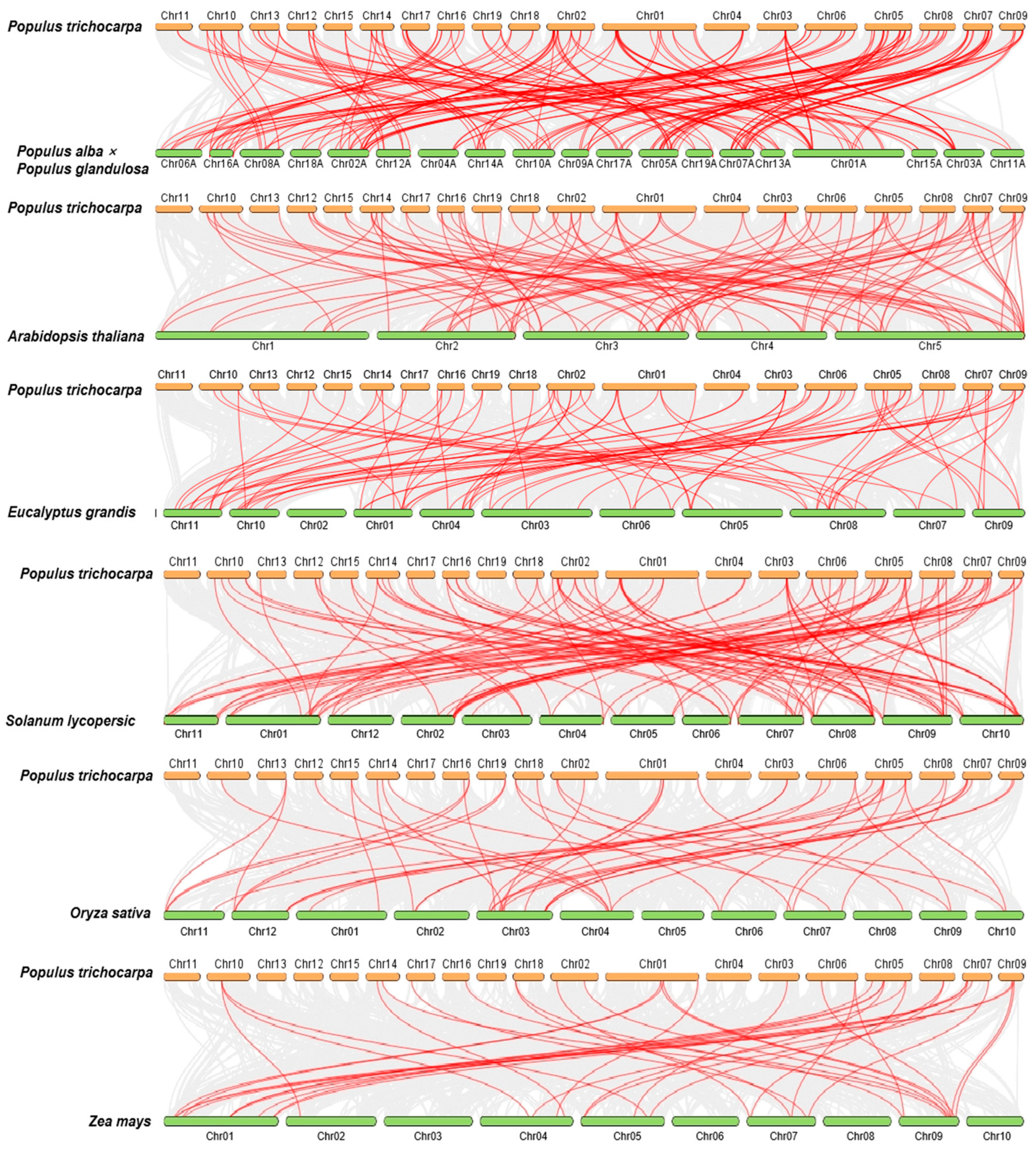
To further investigate the collinearity among the BTB genes, we examined the genomic synteny of P. trichocarpa with those of six other plant species. This group included four dicotyledons—P. alba × P. glandulosa, A. thaliana, Solanum lycopersicum, and E. grandis [33]—as well as two monocotyledons, Oryza sativa and Zea mays. As depicted in Figure 5, the number of homologous pairs between P. trichocarpa and these species were as follows: 87 with P. alba × P. glandulosa, 69 with E. grandis, 69 with A. thaliana, 68 with S. lycopersicum, 34 with O. sativa, and 26 with Z. mays (Figure 5). The observed phenomenon can mainly be attributed to the divergent phylogenetic relationships that distinguish dicotyledons from monocotyledons. Among them, the PtrBTB genes exhibited the greatest collinearity with those of P. alba × P. glandulosa and E. grandis, both of which are also dicotyledonous woody plants. Notably, PtrBTB5, PtrBTB71, PtrBTB81, and PtrBTB86 were not detected in either P. alba × P. glandulosa or E. grandis, which imply that these genes might have undergone species-specific evolution. In addition, we found that a large number of PtrBTB genes, such as PtrBTB19, PtrBTB24, PtrBTB31, PtrBTB61, and PtrBTB76, had collinear relationships with three Arabidopsis genes, suggesting that these genes may play an important role in the evolution of gene families.
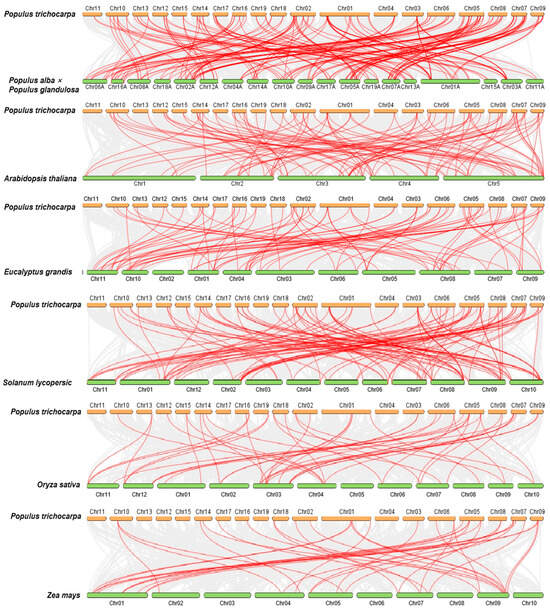
Figure 5.
Collinear analysis of PtrBTB genes from P. trichocarpa with four dicotyledons (P. alba × P. glandulosa, A. thaliana, S. lycopersicum, and E. grandis) and two monocotyledons (O. sativa and Z. mays).
2.6. Expression Patterns of PtrBTB Genes in Different Tissues
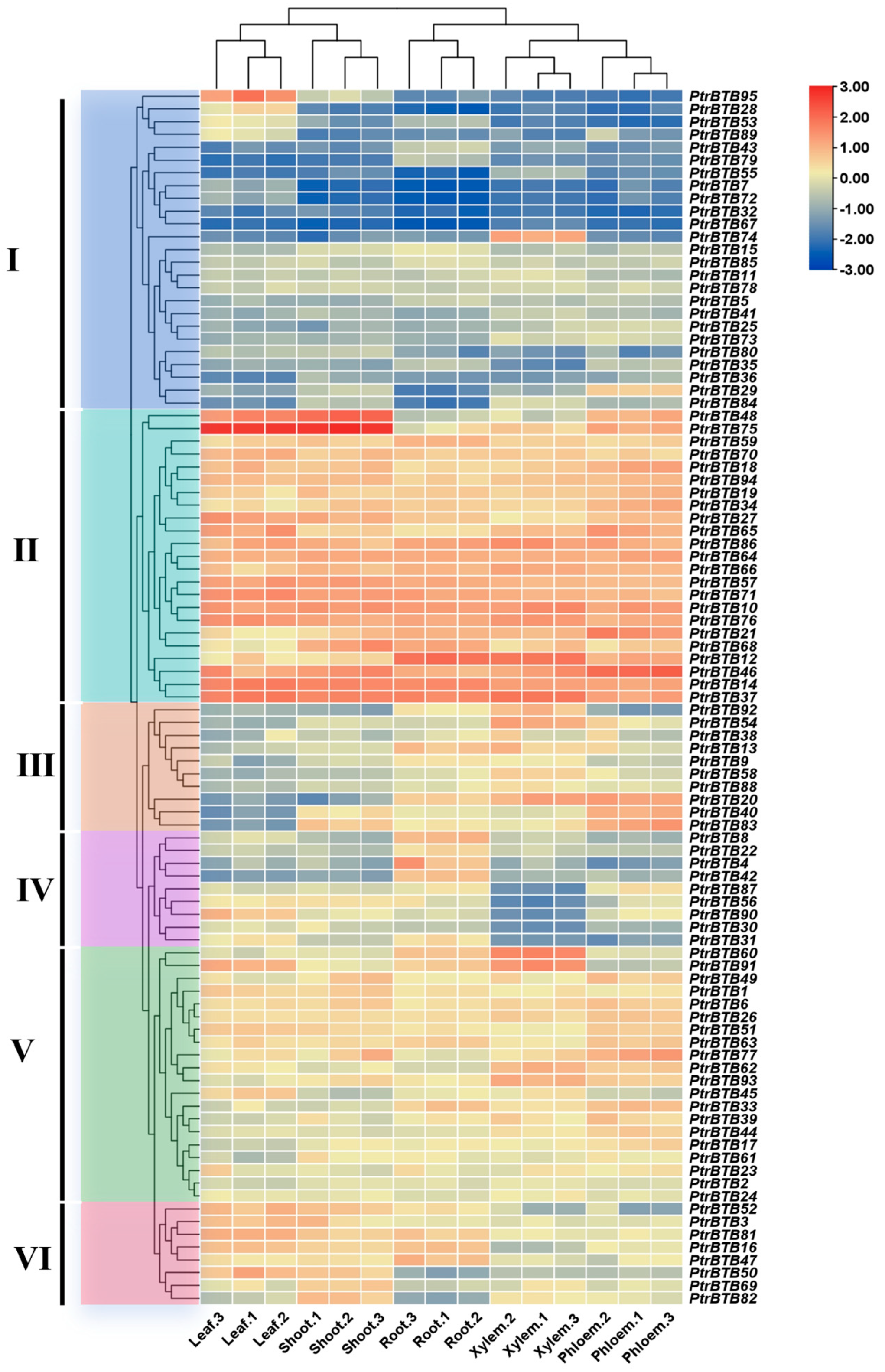
To investigate the expression pattern of the PtrBTB gene family in different tissues, we used the RNA-seq to analyze the BTB gene expression in poplar leaves, shoots, roots, xylem, and phloem. Meanwhile, in order to ensure the accuracy of the data, we selected 11 PtrBTB genes for RT-qPCR validation and found that the results were consistent with the RNA-seq (Supplementary Figure S3). As shown in Figure 6, the cluster analysis revealed that these genes were divided into six groups with specific expression patterns. In group Ⅰ, PtrBTB95 was highly expressed in leaves, PtrBTB74 was highly expressed in xylem, and PtrBTB29 was higher in phloem, while the remaining gene had lower expression level across all tissues. In contrast, the expression level of all the PtrBTB genes in group Ⅱ was high, especially PtrBTB48 and PtrBTB75, which had the highest expression level in leaves and shoots. A majority of the genes in group III were preferentially expressed in roots and xylem. Furthermore, PtrBTB20, PtrBTB40, and PtrBTB83 were noted for their pronounced expression in the phloem as well. Genes in group Ⅳ had high expression levels in the roots, among which PtrBTB90 was also highly expressed in the leaves, indicating its potential involvement in the leaf development process. Most genes in group Ⅴ exhibited high expression level in both xylem and phloem, while genes in group VI had high expression levels in the leaves and shoots, indicating these genes may function in distinct biological processes.
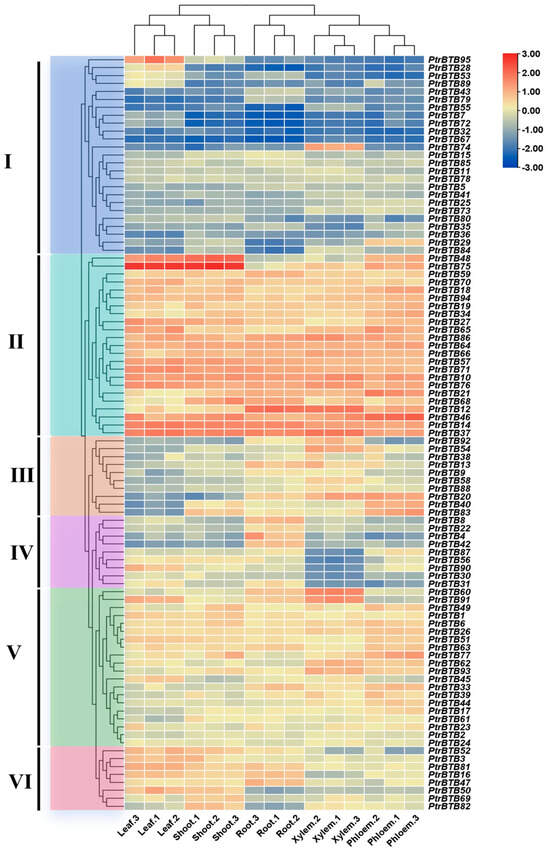
Figure 6.
Expression profiles of PtrBTB genes in different tissues, shoots, roots, leaves, xylem and phloem. Ⅰ−VI represent different clusters. All data of RNA−seq analysis was deposited in GEO (accession number: GSE81077). The color scale represents the FPKM values normalized by log2FPKM. Red represents highly expressed genes and blue represents low expressed genes.
2.7. Expression Patterns of Poplar BTB Genes in Response to Exogenous Hormones
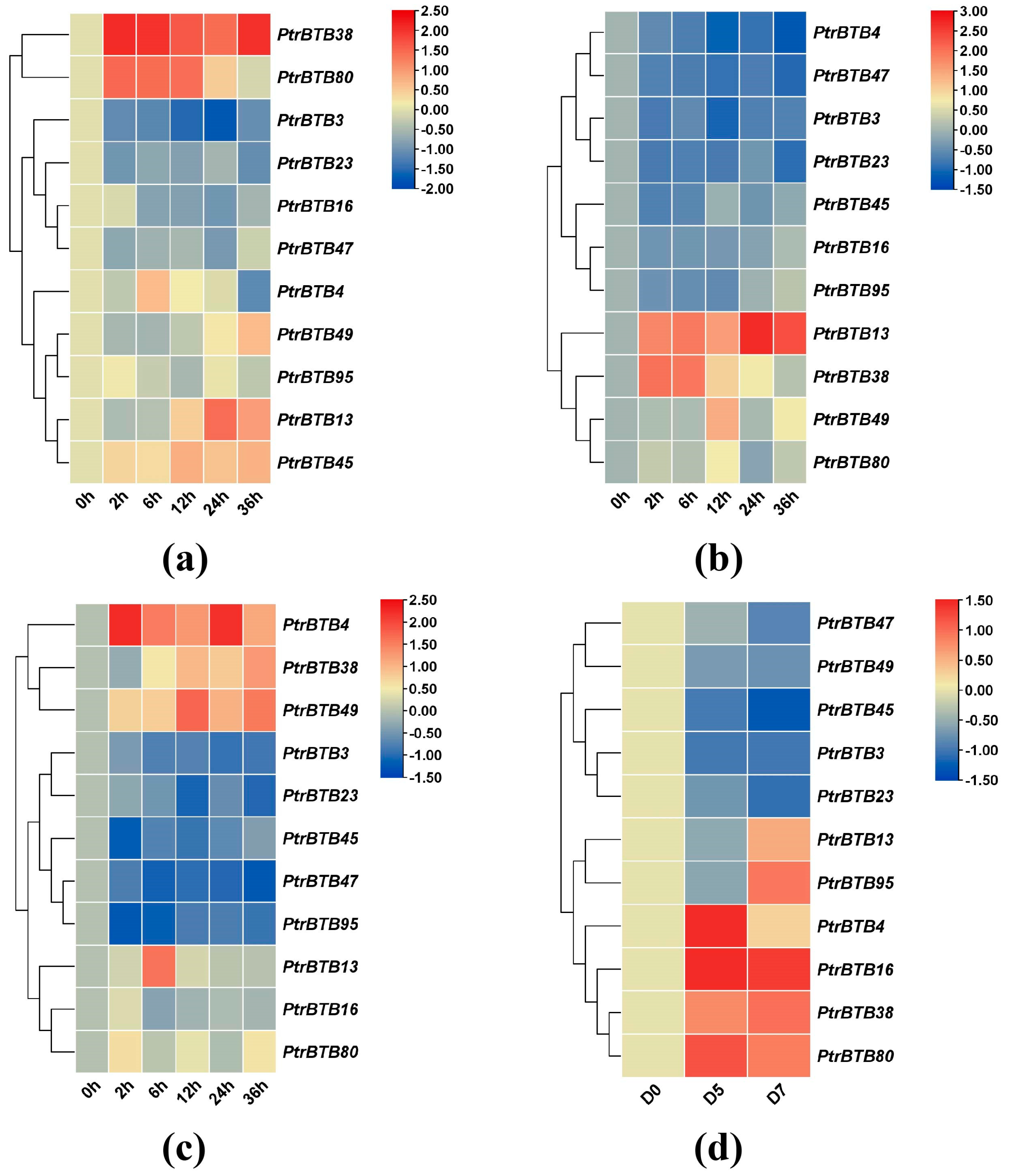
Based on RNA-seq data from previous hormone treatments and stress treatments, we identified eleven genes with significant differences, namely PtrBTB3, PtrBTB4, PtrBTB13, PtrBTB16, PtrBTB23, PtrBTB38, PtrBTB45, PtrBTB47, PtrBTB49, PtrBTB80 and PtrBTB95, which belong to five distinct subfamilies (Supplementary Figure S4). Since P. trichocarpa plants cannot grow well in the area of Beijing, China, we analyzed the expression of these eleven BTB genes in leaf tissues of P. alba × P. glandulosa in response to five hormone treatments (GA, ABA, SA, NAA, and JA), comparing their expression levels with those in untreated leaves of wild-type poplar. The results show that the expression levels of these PagBTB genes were distinctly affected by various treatments, revealing both connections and distinctions in the expression pattern among these PagBTB members.
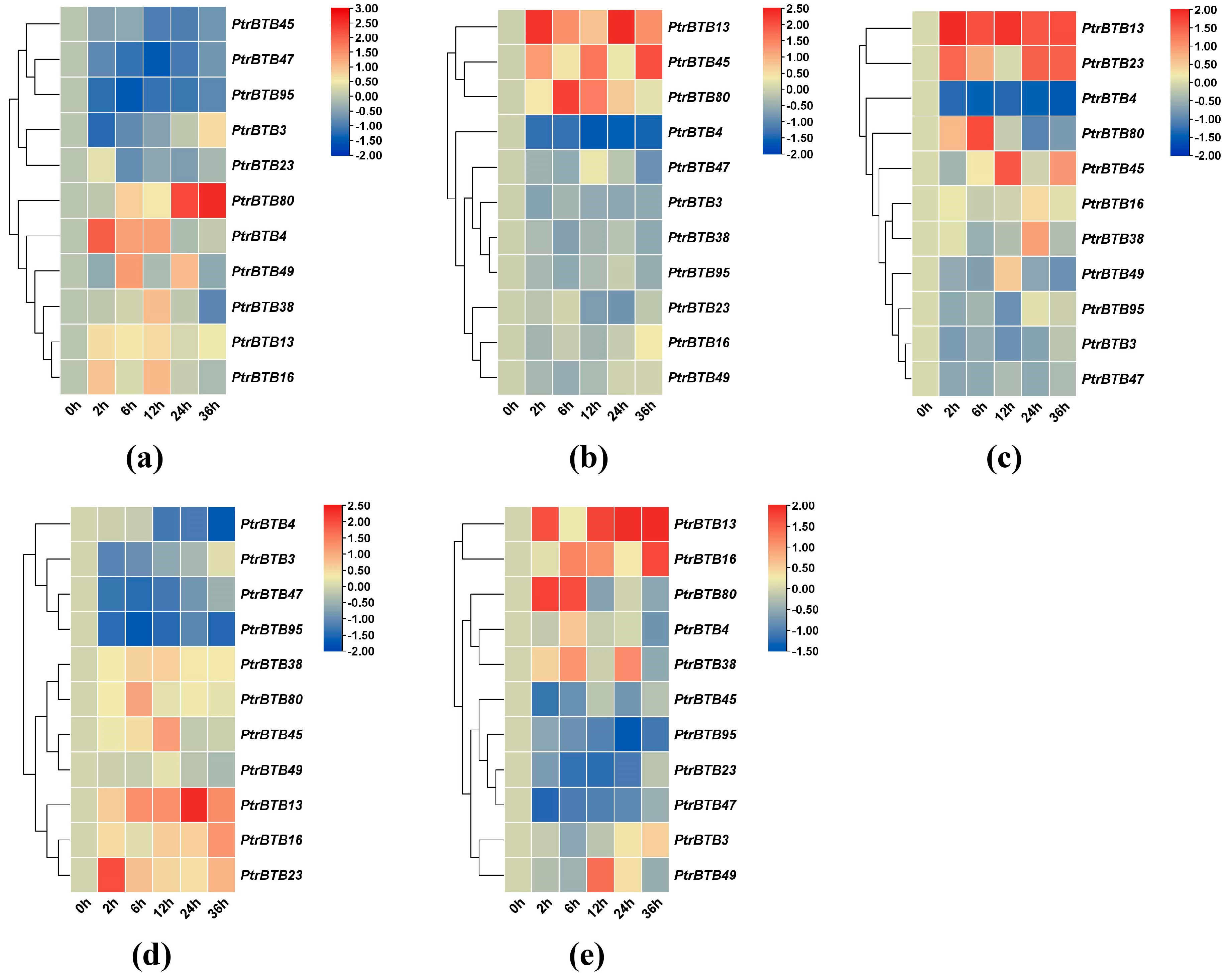
Figure 7a illustrates the expression levels of eleven PagBTB genes in response to JA treatment, with PagBTB4, PagBTB13, PagBTB16, PagBTB38, PagBTB49, and PagBTB80 exhibiting markedly upregulated expressions. In contrast, the expression of five other PagBTB genes, including PagBTB3, PagBTB23, PagBTB45, PagBTB47, and PagBTB95, was remarkably downregulated. Upon treatment with ABA, with the exception of PagBTB16, PagBTB23, and PagBTB49, the expression of the other PagBTB genes underwent notable alterations. As depicted in Figure 7b, PagBTB13, PagBTB45, and PagBTB80 exhibited pronounced upregulation in expression, whereas the expression levels of PagBTB3, PagBTB4, PagBTB38, PagBTB47, and PagBTB95 were considerably downregulated. In response to GA treatment, only PagBTB3, PagBTB4, PagBTB47, and PagBTB95 showed significant downregulation in expression, while the remaining seven PagBTB genes displayed a considerable increase in their transcript abundance (Figure 7c). The gene expression changes induced by GA were found to parallel those seen with the NAA treatment (Figure 7d), although PagBTB3, PagBTB47, and PagBTB95 exhibited a more pronounced reduction in response to NAA. Notably, PagBTB13 and PagBTB23 showed obviously higher upregulation in response to GA treatment, indicating a distinct sensitivity to various plant hormones. Simultaneously, SA treatment induced the expression of PagBTB3, PagBTB4, PagBTB13, PagBTB16, PagBTB38, PagBTB49, and PagBTB80 genes in leaves, while the expressions of PagBTB23, PagBTB45, PagBTB47, and PagBTB95 genes were inhibited by SA treatment (Figure 7e).
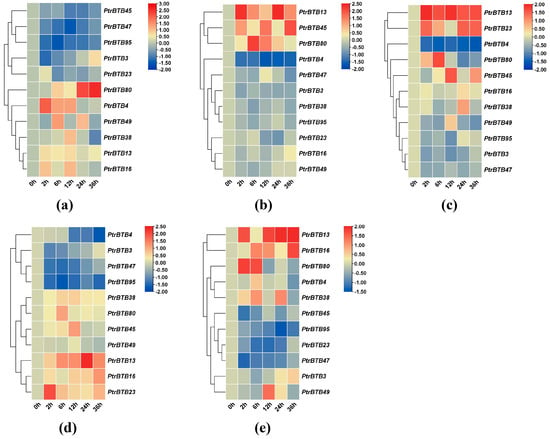
Figure 7.
Expression profiles of BTB genes under exogenous hormones in P. alba × P. glandulosa. Color scale represents log2 expression values, red represents highly expressed genes and blue represents low expressed genes. (a) JA treatment, (b) ABA treatment, (c) GA treatment, (d) NAA treatment, (e) SA treatment. All samples were collected from leaf tissues at specified time points, with three biological replicates for each treatment. Error bars indicate ± SE of the means (n = 3).
2.8. Expression Patterns of Poplar BTB Genes in Response to Different Biotic/Abiotic Stress
To investigate the expression patterns of the poplar BTB genes under biotic and abiotic stresses, we treated leaves and roots with sodium chloride, and treated leaves with F. solani, while the xylem was subjected to drought stress, followed by the examination of expression changes in these above-mentioned 11 BTB genes in P. alba × P. glandulosa. In the roots treated with NaCl, the expression levels of PagBTB3, PagBTB16, PagBTB23, and PagBTB47 genes were obviously suppressed, while the expression levels of the remaining genes were induced (Figure 8a). In the leaves, PagBTB13, PagBTB38, PagBTB49, and PagBTB80 showed a notable increase in expression levels, whereas PagBTB3, PagBTB4, PagBTB16, PagBTB23, PagBTB45, PagBTB47, and PagBTB95 were obviously downregulated (Figure 8b). Likewise, in response to F. solani-induced stress, there was significant suppression in the expression levels of PagBTB3, PagBTB23, PagBTB45, PagBTB47, and PagBTB95, while PagBTB4, PagBTB13, PagBTB16, PagBTB38, PagBTB49, and PagBTB80 displayed a marked increase in their expression in leaves (Figure 8c). During the course of drought stress, eleven PagBTB genes exhibited dynamic changes in their transcriptional levels. Specifically, PagBTB4, PagBTB16, PagBTB38, and PagBTB80 exhibited increased expression levels, whereas PagBTB3, PagBTB23, PagBTB45, PagBTB47, and PagBTB49 displayed decreased expression levels. Notably, the expression of PagBTB13 and PagBTB95 exhibited a biphasic response, initially decreasing at D5 of the drought treatment and subsequently upregulating at D7 (Figure 8d). Overall, PagBTBs have different expression patterns under different types of treatments, and they may play important functions in plant resistance to hormones and biotic/abiotic stresses.
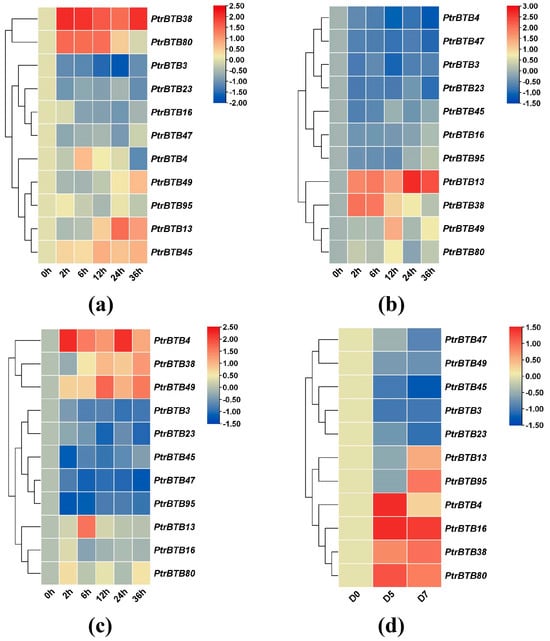
Figure 8.
Expression profiles of PagBTB genes under different biotic/abiotic stress in poplar 84K. Color scale represents log2 expression values, red represents highly expressed genes and blue represents low expressed genes. (a) NaCl−root, (b) NaCl−leaf, (c) F. solani stress−leaf, (d) Drought treatment−xylem. All samples were collected at the indicated time intervals from three biological replicates of each treatment. Error bars indicate ± SE of the means (n = 3).
3. Discussion
BTB/POZ family proteins, commonly found in animals and plants, have been extensively investigated in animals. However, recent evidence suggests their essential roles in plant growth and development, encompassing diverse functions such as resistance modulation, phototropic growth regulation, ubiquitination-mediated degradation, cell cycle control, and ion channel regulation [6,34]. Nevertheless, the functional characterization of BTB/POZ proteins in poplar remains limited compared to Arabidopsis. In this study, we integrated the HMMER search method and the BLASTp method and identified 95 BTB genes in P. trichocarpa that were classified into eight subfamilies based on their conserved domains, including NPH3, TAZ, Ankyrin, only BTB, BACK, Armadillo, TPR, and MATH (Supplementary Figure S1). The BTB protein family, characterized by their DNA-binding domains, is hypothesized to regulate gene expression. Moreover, the interaction of BTB domain with additional domains expands the functional range of the BTB proteins [1]. Among them, NPH3 is the largest subfamily and contains the most BTB members in poplar. Recently, the NPH3, exclusive to higher plants, has been found to engage with the blue light receptor phot1, acting as a key substrate adapter for the CULLIN3-dependent E3 ubiquitin ligase [35]. It is evident that the NPH3 subfamily plays a pivotal role in the blue light-mediated phototropic responses, serving as an essential element in this biological process. We also found that the high degree of conservation of BTB family members allows genes localized in the same subfamily to have the same or similar functions. Gene structure and motif analysis further confirmed a close relationship among the poplar BTB genes within the same subgroup, indicating consistent exons counts and motif types that align with observed distribution patterns in the phylogenetic tree. This intriguing aspect warrants further investigation to elucidate its underlying mechanisms and potential applications.
The expression of genes is mostly related to the cis-elements in the promoter region [36]. In Arabidopsis, the cis-acting element DRE (Dehydration-responsive element) plays a crucial role in regulating gene expression under conditions of dehydration and low temperature [37]. Analysis of cis-elements in the promoter region revealed a significant presence of plant hormone-related and stress-responsive elements within the promoters of the BTB genes in P. trichocarpa, indicating their potential involvement in related functional expression. Notably, this investigation has uncovered a range of cis-elements within PtrBTB promoters involved in responses to light exposure, hormonal stimuli, and environmental stress (Figure 4). It is worth noting that light-responsive elements are ubiquitous and most abundant, aligning with the findings from various species including grape [38], sugar beet [12], and tomato [10]. This result suggests that the transcriptional control of PtrBTB is likely influenced by light signals across a broad spectrum of plant species. In addition, four distinct poplar BTB genes, namely PtrBTB4, PtrBTB44, PtrBTB54, and PtrBTB82, have been identified to contain cis-elements associated with wound response. This discovery is novel as these elements have not been previously reported in species such as soybeans, apples, and grapes, suggesting a potential role for these genes in the self-healing process and enhanced vitality of poplar trees.
The cross-species collinearity analysis indicated a closer evolutionary relationship between P. trichocarpa and dicotyledons compared to monocotyledons (Figure 5). This observation hints at a potential divergence in the evolution of BTB genes in conjunction with the evolutionary split between dicotyledons and monocotyledons. Collectively, the bioinformatic assessments indicate congruence in phylogenetic and structural characteristics of the PtrBTB gene family across various species, suggesting analogous functional roles of BTB genes and underscoring their conserved biological significance. In addition, previous reports have shown that the BTB protein can promote plant resistance to various abiotic and biological stresses. The expression of the AtBT4 gene in Arabidopsis is upregulated, thereby enhancing the plant resistance to Botrytis griseus through the ethylene (ET)/JA pathway [39]. Additionally, under drought conditions, the expression of the MdBT2 gene in apple is downregulated, resulting in the activation of NAM, ATAF1/2, CUC1/2 (NAC). These genes, originally inhibited by MdBT2 protein, subsequently pass through acid signaling pathways that regulate reactive oxygen species (ROS) levels and participate in plant stress response [40]. Furthermore, following inoculation with potato virus Y (PVY), tobacco initiates a defense response involving active hormones such as IAA, SA, and JA, along with ROS, suggesting the potential regulatory roles of the BTB/POZ family members in the response of tobacco to PVY [41]. Therefore, we subjected the plant material to drought, salt, F. solani stress, and various hormones treatments. Based on prior RNA-seq data, we chose eleven BTB genes that exhibited significant expression changes under various treatment conditions. Specifically, the genes PtrBTB45, PtrBTB47, and PtrBTB95 belong to the NPH3–BTB subfamily, PtrBTB13 and PtrBTB38 are members of the BACK–BTB subfamily, and PtrBTB49 and PtrBTB80 are associated with the TAZ–BTB subfamily. PtrBTB16 is a part of the Ank–BTB subfamily, whereas PtrBTB3, PtrBTB4, and PtrBTB23 are classified under the only-BTB subfamily, which lacks additional domains.
The RT-qPCR analysis across the three treatments revealed that a majority of these PagBTB genes demonstrated a significant response to the applied treatments. Following hormonal stimuli and environmental perturbations, the genes exhibited diverse temporal expression patterns, and the individual genes showed tissue-specific modulation, emphasizing the complexity of gene expression in response to stimuli. Upon hormonal intervention, PagBTB13 and PagBTB80 were characterized by a significant upregulation in their expression levels, in contrast with the diminished expression levels of PagBTB47 and PagBTB95, illustrating a divergent gene expression response to hormonal stimuli (Figure 7). Furthermore, following NaCl treatment, PagBTB4, PagBTB45, and PagBTB95 showed a significant upregulation in the root tissues but notable downregulation in the leaf tissues (Figure 8). These observations suggest that poplar BTB genes are responsive to stress stimuli and exhibit tissue-specific expression patterns, with distinct responses in roots compared to leaves. Collectively, our findings from promoter analysis and gene expression studies under stress conditions suggest a pivotal role for the BTB proteins in plant response to both biotic and abiotic stressors [42].
4. Materials and Methods
4.1. Plant Materials and Treatment
The experimental materials used in this study were from the State Key Laboratory of Tree Genetics and Breeding, Chinese Academy of Forestry, Beijing, China. Sterile Populus alba × Populus glandulosa (84K) plants were grown on one-half Murashige and Skoog (Sigma-Aldrich, Darmstadt, Germany) media containing 3% sucrose and 0.6% agar and maintained at 23 °C under a 16 h light/8 h dark photoperiod. The plantlets were grown on humus soil with a 16/8 h day/night cycle at 25 °C in the greenhouse. For the gene expression profiling analysis, the P. alba × P. glandulosa plants were simultaneously subjected to various stresses or hormone treatments. The culture seedlings of 1-month-old were transplanted into 1/2 MS liquid medium containing 3% (w/v) sucrose and pre-cultured for 16 h. Samples were then treated with 1/2 MS liquid medium containing 100 μM 1-Naphthaleneacetic acid (NAA), 100 μM ABA, 100 μM gibberellic acid (GA), 100 μM SA, 100 μM GA, 200 mM NaCl, and 107 spores/mL Fusarium solani (Mart.) Sacc (F. solani), respectively.
Following these treatments, the leaves and roots were collected as experimental materials under the NaCl treatment; the leaves were collected as experimental materials in the other treatments. Then, the tissues were harvested after various treatment time points (0, 2, 4, 6, 12, 24 h), immediately frozen in liquid nitrogen and then stored at −80 °C for RNA preparation. Pilot studies on drought stress have indicated that applying both 5-day (D5) and 7-day (D7) drought treatments could potentially induce the most significant and extensive genetic alterations [43]. Consequently, we cultivated 3-month-old poplar 84K in a controlled environment to a stage of maturity suitable for experimentation and subjected them to the aforementioned drought conditions. Subsequently, samples of differentiating xylem from the stems of both well-irrigated (control group, devoid of drought stress) and the drought-affected plants were harvested. These samples were immediately frozen in liquid nitrogen and stored at −80 °C for RNA extraction. Each sample was collected in triplicate as biological replicates.
4.2. Identification and Annotation of BTB Proteins in P. trichocarpa
Genome data and genome annotation files of P. trichocarpa (v4.1), Arabidopsis (TAIR10) were extracted from the Phytozome database (https://phytozome-next.jgi.doe.gov/ (accessed on 24 December 2023)), and the files of P. alba × P. glandulosa (v3.1) were downloaded from the website (https://doi.org/10.6084/m9.figshare.12369209 (accessed on 24 December 2023)) [44]. The Arabidopsis BTB protein sequences were utilized as queries for BLASTp searches against the P. trichocarpa and the P. alba × P. glandulosa genomes. The following parameters were used: −evalue 10−5, −best hit overhang 0.25, and −max target seqs 5, with the searches conducted in the NCBI database (https://www.ncbi.nlm.nih.gov/ (accessed on 24 December 2023)). Furthermore, the HMM file (PF00651) of the conserved domain was downloaded from the Pfam database (http://pfam-legacy.xfam.org/ (accessed on 24 December 2023)). A Hidden Markov Model profile of the HMMER 3.0 software was employed to search for putative BTB in P. trichocarpa and P. alba × P. glandulosa, with an E-value threshold of 10−5. The results obtained by the two methods were integrated with those of previous studies to avoid repetition. After the protein sequences were aligned and trimmed by ClustalW, the protein sequences of 95 BTB proteins in P. trichocarpa, 165 BTB proteins in poplar 84K, and 80 BTB proteins in Arabidopsis were used to construct the phylogenetic tree by the MEGA-X 10.2 Neighbor-Joining (NJ) method, and the parameters were p-distance model and 1000 bootstrap replicates. Subsequently, a phylogeny of the BTB proteins was generated on the iTOL web browser (https://itol.embl.de/ (accessed on 30 December 2023)). Furthermore, the physical and chemical parameters of PtrBTB proteins were determined using the ExPASy website (http://web.expasy.org/protparam/ (accessed on 30 December 2023)). Subcellular locations of PtrBTB proteins were predicted using the WoLF PSORT ProtParam online analysis tool (https://www.genscript.com/wolf-psort.html (accessed on 30 December 2023)).
4.3. Chromosomal Location and Collinearity Analysis
GTF annotations of the P. trichocarpa genome were mapped to each PtrBTB gene and to its corresponding chromosomal location using the Gene Location Visualize module of TBtools software (version 2.019) [45]. The genome sequence files of P. trichocarpa, P. alba × P. glandulosa, Eucalyptus grandis, A. thaliana, Oryza sativa, Solanum lycopersicum, and Zea mays were aligned and analyzed using the MCScanX software (version 2019). Gene collinearity analysis was used to determine their conservative relationships and visualized by TBtools (version 2.019). We also used the KaKs_Calculator software (version 3.0) to calculate the ratio of non-synonymous substitution and synonymous substitution (Ka/Ks) for duplication gene pairs with the NG method [46].
4.4. Analysis of Domains, Gene Structures, Motifs and Cis-Acting Elements
Conserved domains and motifs of BTB proteins were identified using the SMART (http://smart.embl-heidelberg.de/ (accessed on 14 March 2024)), GSDS (https://gsds.gao-lab.org/Gsds_help.php (accessed on 18 March 2024)), and MEME (http://meme-suite.org/tools/meme (accessed on 25 March 2024)) websites. The gff3 file from the P. trichocarpa whole genome database was utilized to identify and annotate gene structures, including the intron, exon, and UTR regions. The 2000 bp upstream regions of all the PtrBTB genes were extracted for analysis of promoter cis-acting elements through the PlantCARE online database (http://bioinformatics.psb.ugent.be/webtools/plantcare/html/ (accessed on 26 March 2024)) [47]. All those generated files were visualized using the TBtools software (version 2.019) [45].
4.5. Expression Pattern Analysis of PtrBTB Genes
The RNA-seq data under different treatments in P. trichocarpa were obtained from previous studies [43,48,49]. The corresponding expression data were downloaded from the National Center for Biotechnology Information Gene Expression Omnibus database (https://www.ncbi.nlm.nih.gov/geo/ (accessed on 18 March 2024)) (accession number: GSE81048, GSE81077, GSE109609) and the BIGD Genome Sequence Archive (https://bigd.big.ac.cn (accessed on 18 March 2024)) under accession number CRA006695. Transcript levels were normalized based on fragments per kilobase of transcript per million fragments (FPKM) with Cufflinks (v. 2.1.1) with default options. Heatmaps were plotted using TBtools (version 2.019) [45].
4.6. RNA Isolation and Quantitative Real-Time PCR
Total RNAs were isolated using the RNeasy Plant Mini Kit (QIAGEN, Redwood, CA, USA), and mRNAs were reverse-transcribed into cDNA using the Prime Script TM RT Reagent Kit with a gDNA Eraser (TaKaRa, Dalian, China). RT-qPCR was conducted using the CFX96 Real-Time PCR (Bio-Rad, Hercules, CA, USA) with SYBR Green Premix ExTagII (TaKaRa, Dalian, China). The PCR program was as follows: initial incubation at 94 °C for 5 min; 40 cycles of 94 °C for 10 s, 58 °C for 15 s, and 72 °C for 30 s. Data were gathered during the extension step. Melting-curve acquisition and analyses were performed on the cycler, with relative expression changes of genes analyzed using the 2−ΔΔCT method. Actin was used as the internal reference. The gene-specific primers are listed in Supplementary Table S3. Reactions were performed with three biological replicates, each with three technical replicates.
5. Conclusions
In our research, we have successfully identified 95 PtrBTB genes in P. trichocarpa genome and classified them as eight subfamilies. The chromosomal mapping of these genes exposed an uneven distribution, with 95 PtrBTB genes scattered across nineteen chromosomes. Structural and motif analyses revealed remarkable consistency in gene architecture and motif composition across all members within each cluster. Furthermore, collinearity analysis uncovered a greater prevalence of collinear genes between the woody plant poplar and E. grandis, suggesting a potential gene divergence coinciding with the evolution of herbaceous and woody species. A detailed examination of the gene promoters revealed a significant presence of the cis-elements associated with hormones and biotic/abiotic stress responses, indicating a probable involvement of these genes in the stress adaptation mechanisms. RT-qPCR analysis further substantiated the responsiveness of the eleven PagBTB genes to a spectrum of hormonal and stress stimuli. This comprehensive analysis enhances our understanding of the BTB gene family in poplar and paves the way for future studies aimed at enhancing stress resilience in plants.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms25169048/s1.
Author Contributions
Formal analysis, X.D.; Methodology, Q.L.; Software, J.Y.; Validation, J.Y.; Writing—Original Draft, J.Y.; Writing—Review and Editing, Q.L. and M.W. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Fundamental Research Funds of the Chinese Academy of Forestry (CAFYBB2022QC001-2).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data are contained within the article and Supplementary Materials.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Chaharbakhshi, E.; Jemc, J.C. Broad-complex, tramtrack, and bric-à-brac (BTB) proteins: Critical regulators of development. Genesis 2016, 54, 505–518. [Google Scholar] [CrossRef]
- Stogios, P.J.; Downs, G.S.; Jauhal, J.J.; Nandra, S.K.; Privé, G.G. Sequence and structural analysis of BTB domain proteins. Genome Biol. 2005, 6, R82. [Google Scholar] [CrossRef]
- Cheng, D.; Qian, W.; Meng, M.; Wang, Y.; Peng, J.; Xia, Q. Identification and expression profiling of the BTB domain-containing protein gene family in the silkworm, Bombyx mori. Int. J. Genom. 2014, 2014, 865065. [Google Scholar] [CrossRef]
- Stogios, P.J.; Privé, G.G. The BACK domain in BTB-kelch proteins. Trends Biochem. Sci. 2004, 29, 634–637. [Google Scholar] [CrossRef]
- Michaely, P.; Bennett, V. The ANK repeat: A ubiquitous motif involved in macromolecular recognition. Trends Cell Biol. 1992, 2, 127–129. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, K.F.; Melnick, A.; Lax, S.; Bouchard, D.; Liu, J.; Kiang, C.L.; Mayer, S.; Takahashi, S.; Licht, J.D.; Privé, G.G. Mechanism of SMRT corepressor recruitment by the BCL6 BTB domain. Mol. Cell 2003, 12, 1551–1564. [Google Scholar] [CrossRef]
- Melnick, A.; Ahmad, K.F.; Arai, S.; Polinger, A.; Ball, H.; Borden, K.L.; Carlile, G.W.; Prive, G.G.; Licht, J.D. In-depth mutational analysis of the promyelocytic leukemia zinc finger BTB/POZ domain reveals motifs and residues required for biological and transcriptional functions. Mol. Cell. Biol. 2000, 20, 6550–6567. [Google Scholar] [CrossRef] [PubMed]
- Weber, H.; Bernhardt, A.; Dieterle, M.; Hano, P.; Mutlu, A.; Estelle, M.; Genschik, P.; Hellmann, H. Arabidopsis AtCUL3a and AtCUL3b form complexes with members of the BTB/POZ-MATH protein family. Plant Physiol. 2005, 137, 83–93. [Google Scholar] [CrossRef]
- Gingerich, D.J.; Hanada, K.; Shiu, S.H.; Vierstra, R.D. Large-scale, lineage-specific expansion of a bric-a-brac/tramtrack/broad complex ubiquitin-ligase gene family in rice. Plant Cell 2007, 19, 2329–2348. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Su, X.; Wang, Y.; Yang, W.; Pan, Y.; Su, C.; Zhang, X. Genome-wide identification and expression analysis of the BTB domain-containing protein gene family in tomato. Genes Genom. 2018, 40, 1–15. [Google Scholar] [CrossRef]
- Zhu, P.; Fan, Y.; Xu, P.; Fan, G. Bioinformatic analysis of the BTB gene family in Paulownia fortunei and functional characterization in response to abiotic and biotic stresses. Plants 2023, 12, 4144. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Liu, Y.; Zhao, C.; Wang, X.; Ding, G.; Li, Y.; Chen, L. Genome-wide identification and expression analysis of the BTB domain-containing protein gene family in sugar beet. Agronomy 2022, 12, 253. [Google Scholar] [CrossRef]
- Ren, Y.R.; Zhao, Q.; Yang, Y.Y.; Zhang, R.; Wang, X.F.; Zhang, T.E.; You, C.X.; Huo, H.Q.; Hao, Y.J. Interaction of BTB-TAZ protein MdBT2 and DELLA protein MdRGL3a regulates nitrate-mediated plant growth. Plant Physiol. 2021, 186, 750–766. [Google Scholar] [CrossRef]
- Xu, L.; Wei, Y.; Reboul, J.; Vaglio, P.; Shin, T.H.; Vidal, M.; Elledge, S.J.; Harper, J.W. BTB proteins are substrate-specific adaptors in an SCF-like modular ubiquitin ligase containing CUL-3. Nature 2003, 425, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Pintard, L.; Willis, J.H.; Willems, A.; Johnson, J.L.; Srayko, M.; Kurz, T.; Glaser, S.; Mains, P.E.; Tyers, M.; Bowerman, B.; et al. The BTB protein MEL-26 is a substrate-specific adaptor of the CUL-3 ubiquitin-ligase. Nature 2003, 425, 311–316. [Google Scholar] [CrossRef]
- An, J.P.; Liu, Y.J.; Zhang, X.W.; Bi, S.Q.; Wang, X.F.; You, C.X.; Hao, Y.J. Dynamic regulation of anthocyanin biosynthesis at different light intensities by the BT2-TCP46-MYB1 module in apple. J. Exp. Bot. 2020, 71, 3094–3109. [Google Scholar] [CrossRef] [PubMed]
- An, J.P.; Zhang, X.W.; You, C.X.; Bi, S.Q.; Wang, X.F.; Hao, Y.J. MdWRKY40 promotes wounding-induced anthocyanin biosynthesis in association with MdMYB1 and undergoes MdBT2-mediated degradation. New Phytol. 2019, 224, 380–395. [Google Scholar] [CrossRef]
- Julian, J.; Coego, A.; Lozano-Juste, J.; Lechner, E.; Wu, Q.; Zhang, X.; Merilo, E.; Belda-Palazon, B.; Park, S.Y.; Cutler, S.R.; et al. The MATH-BTB BPM3 and BPM5 subunits of Cullin3-RING E3 ubiquitin ligases target PP2CA and other clade A PP2Cs for degradation. Proc. Natl. Acad. Sci. USA 2019, 116, 15725–15734. [Google Scholar] [CrossRef]
- Lechner, E.; Leonhardt, N.; Eisler, H.; Parmentier, Y.; Alioua, M.; Jacquet, H.; Leung, J.; Genschik, P. MATH/BTB CRL3 receptors target the homeodomain-leucine zipper ATHB6 to modulate abscisic acid signaling. Dev. Cell 2011, 21, 1116–1128. [Google Scholar] [CrossRef]
- Mandadi, K.K.; Misra, A.; Ren, S.; McKnight, T.D. BT2, a BTB protein, mediates multiple responses to nutrients, stresses, and hormones in Arabidopsis. Plant Physiol. 2009, 150, 1930–1939. [Google Scholar] [CrossRef]
- Boyle, P.; Le Su, E.; Rochon, A.; Shearer, H.L.; Murmu, J.; Chu, J.Y.; Fobert, P.R.; Després, C. The BTB/POZ domain of the Arabidopsis disease resistance protein NPR1 interacts with the repression domain of TGA2 to negate its function. Plant Cell 2009, 21, 3700–3713. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, P.; Gusmaroli, G.; Serino, G.; Habashi, J.; Ma, L.; Shen, Y.; Feng, S.; Bostick, M.; Callis, J.; Hellmann, H.; et al. Arabidopsis has two redundant Cullin3 proteins that are essential for embryo development and that interact with RBX1 and BTB proteins to form multisubunit E3 ubiquitin ligase complexes in vivo. Plant Cell 2005, 17, 1180–1195. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, S.H.; Seo, D.H.; Chung, S.; Kim, S.W.; Lee, J.S.; Kim, W.T.; Lee, J.H. ABA-HYPERSENSITIVE BTB/POZ PROTEIN 1 functions as a negative regulator in ABA-mediated inhibition of germination in Arabidopsis. Plant Mol. Biol. 2016, 90, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Christians, M.J.; Gingerich, D.J.; Hansen, M.; Binder, B.M.; Kieber, J.J.; Vierstra, R.D. The BTB ubiquitin ligases ETO1, EOL1 and EOL2 act collectively to regulate ethylene biosynthesis in Arabidopsis by controlling type-2 ACC synthase levels. Plant J. 2009, 57, 332–345. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.S.; Quint, A.; Brand, D.; Vivian-Smith, A.; Offringa, R. BTB and TAZ domain scaffold proteins perform a crucial function in Arabidopsis development. Plant J. 2009, 58, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhang, D.; Chu, J.Y.; Boyle, P.; Wang, Y.; Brindle, I.D.; De Luca, V.; Després, C. The Arabidopsis NPR1 protein is a receptor for the plant defense hormone salicylic acid. Cell Rep. 2012, 1, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Zhang, S.; Qing, X.; Sun, M.; Liu, S.; Su, H.; Shu, H.; Li, X. Superfamily of ankyrin repeat proteins in tomato. Gene 2013, 523, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Withers, J.; Dong, X. Posttranslational Modifications of NPR1: A single protein playing multiple roles in plant immunity and physiology. PLoS Pathog. 2016, 12, e1005707. [Google Scholar] [CrossRef]
- Lin, W.C.; Lu, C.F.; Wu, J.W.; Cheng, M.L.; Lin, Y.M.; Yang, N.S.; Black, L.; Green, S.K.; Wang, J.F.; Cheng, C.P. Transgenic tomato plants expressing the Arabidopsis NPR1 gene display enhanced resistance to a spectrum of fungal and bacterial diseases. Transgenic Res. 2004, 13, 567–581. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhong, S.; Li, Q.; Zhu, Z.; Lou, Y.; Wang, L.; Wang, J.; Wang, M.; Li, Q.; Yang, D.; et al. Functional analysis of rice NPR1-like genes reveals that OsNPR1/NH1 is the rice orthologue conferring disease resistance with enhanced herbivore susceptibility. Plant Biotechnol. J. 2007, 5, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Tuskan, G.A.; Difazio, S.; Jansson, S.; Bohlmann, J.; Grigoriev, I.; Hellsten, U.; Putnam, N.; Ralph, S.; Rombauts, S.; Salamov, A.; et al. The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 2006, 313, 1596–1604. [Google Scholar] [PubMed]
- Li, Y.; Qi, S.; Chen, S.; Li, H.; Zhang, T.; Bao, F.; Zhan, D.; Pang, Z.; Zhang, J.; Zhao, J. Genome-wide identification and expression analysis of late embryogenesis abundant (LEA) genes reveal their potential roles in somatic embryogenesis in hybrid sweetgum (Liquidambar styraciflua × Liquidambar formosana). For. Reserach 2023, 3, 12. [Google Scholar] [CrossRef]
- Zhou, F.; Zhang, K.; Zheng, X.; Wang, G.; Cao, H.; Xing, J.; Dong, J. BTB and TAZ domain protein BT4 positively regulates the resistance to Botrytis cinerea in Arabidopsis. Plant Signal. Behav. 2022, 17, 2104003. [Google Scholar] [CrossRef] [PubMed]
- Collins, T.; Stone, J.R.; Williams, A.J. All in the family: The BTB/POZ, KRAB, and SCAN domains. Mol. Cell. Biol. 2001, 21, 3609–3615. [Google Scholar] [CrossRef]
- Roberts, D.; Pedmale, U.V.; Morrow, J.; Sachdev, S.; Lechner, E.; Tang, X.; Zheng, N.; Hannink, M.; Genschik, P.; Liscum, E. Modulation of phototropic responsiveness in Arabidopsis through ubiquitination of phototropin 1 by the CUL3-Ring E3 ubiquitin ligase CRL3(NPH3). Plant Cell 2011, 23, 3627–3640. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Yamaguchi-Shinozaki, K. Promoters and transcription factors in abiotic stress-responsive gene expression. In Abiotic Stress Adaptation in Plants: Physiological, Molecular and Genomic Foundation; Springer: Dordrecht, the Netherlands, 2009; pp. 199–216. [Google Scholar]
- Liu, Q.; Kasuga, M.; Sakuma, Y.; Abe, H.; Miura, S.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Two transcription factors, DREB1 and DREB2, with an EREBP/AP2 DNA binding domain separate two cellular signal transduction pathways in drought and low-temperature-responsive gene expression, respectively, in Arabidopsis. Plant Cell 1998, 10, 1391–1406. [Google Scholar] [CrossRef] [PubMed]
- Goyal, N.; Bhuria, M.; Verma, D.; Garewal, N.; Singh, K. Genome-Wide Identification of BTB domain-containing gene family in grapevine (Vitis vinifera L.). Agronomy 2023, 13, 252. [Google Scholar] [CrossRef]
- Zhou, T.; Lin, Y.; Lin, Y.; Luo, J.; Ding, J. Regeneration and Agrobacterium-mediated genetic transformation of twelve Eucalyptus species. For. Reserach 2022, 2, 15. [Google Scholar] [CrossRef]
- Tran, L.S.; Nakashima, K.; Sakuma, Y.; Simpson, S.D.; Fujita, Y.; Maruyama, K.; Fujita, M.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Isolation and functional analysis of Arabidopsis stress-inducible NAC transcription factors that bind to a drought-responsive cis-element in the early responsive to dehydration stress 1 promoter. Plant Cell 2004, 16, 2481–2498. [Google Scholar] [CrossRef]
- Zhu, P.; Li, H.; Lu, T.; Liang, R.; Wan, B. Combined analysis of mRNA and miRNA transcriptomes reveals the regulatory mechanism of Xanthomonas arboricola pv pruni resistance in Prunus persica. BMC Genom. 2024, 25, 214. [Google Scholar] [CrossRef]
- Manivannan, A.; Han, K.; Lee, S.Y.; Lee, H.E.; Hong, J.P.; Kim, J.; Lee, Y.R.; Lee, E.S.; Kim, D.S. Genome-wide analysis of MYB10 transcription factor in fragaria and identification of QTLs associated with fruit color in octoploid strawberry. Int. J. Mol. Sci. 2021, 22, 12587. [Google Scholar] [CrossRef]
- Li, S.; Lin, Y.J.; Wang, P.; Zhang, B.; Li, M.; Chen, S.; Shi, R.; Tunlaya-Anukit, S.; Liu, X.; Wang, Z.; et al. The AREB1 transcription factor influences histone acetylation to regulate drought responses and tolerance in Populus trichocarpa. Plant Cell 2019, 31, 663–686. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Chen, S.; Peng, X.; Bae, E.-K.; Dai, X.; Liu, G.; Qu, G.; Ko, J.-H.; Lee, H.; Chen, S.; et al. An improved draft genome sequence of hybrid Populus alba × Populus glandulosa. J. For. Res. 2021, 32, 1663–1672. [Google Scholar] [CrossRef]
- Chen, C.; Wu, Y.; Li, J.; Wang, X.; Zeng, Z.; Xu, J.; Liu, Y.; Feng, J.; Chen, H.; He, Y.; et al. TBtools-II: A “one for all, all for one” bioinformatics platform for biological big-data mining. Mol. Plant 2023, 16, 1733–1742. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, Y.; Zhang, Z.; Zhu, J.; Yu, J. KaKs_Calculator 2.0: A toolkit incorporating gamma-series methods and sliding window strategies. Genom. Proteom. Bioinform. 2010, 8, 77–80. [Google Scholar] [CrossRef]
- Lescot, M.; Déhais, P.; Thijs, G.; Marchal, K.; Moreau, Y.; Van de Peer, Y.; Rouzé, P.; Rombauts, S. PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res. 2002, 30, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Wang, Y.; Xie, J.; Tan, S.; Wang, H.; Zhao, Y.; Liu, Q.; El-Kassaby, Y.A.; Zhang, D. Growth-regulating factor 15-mediated gene regulatory network enhances salt tolerance in poplar. Plant Physiol. 2023, 191, 2367–2384. [Google Scholar] [CrossRef]
- Xing, H.; Fu, X.; Yang, C.; Tang, X.; Guo, L.; Li, C.; Xu, C.; Luo, K. Genome-wide investigation of pentatricopeptide repeat gene family in poplar and their expression analysis in response to biotic and abiotic stresses. Sci. Rep. 2018, 8, 2817. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).