Abstract
Resveratrol is a natural polyphenolic compound that may have multiple influences on human health, including antiaging, anti-inflammatory, anti-neoplastic, antioxidant, insulin-sensitizing, cardioprotective and vasodilating activities. Growing evidence also suggests a potential positive effect of resveratrol on female fertility. The aim of the present study was to collate and appraise the scientific literature on the relationship between resveratrol and female fertility. We systematically searched Medline, PubMed, Web of Science and Embase from the databases’ inception (1951, 1951, 1947 and 1900, respectively) until 9th May 2024. All in vivo or in vitro retrospective or prospective studies reporting the effects of resveratrol interventions on women’s fertility were included. We ultimately incorporated twenty-four studies into a systematic review with a narrative summary of the results; of those studies, nine were performed on women seeking natural or assisted fertility, and fifteen were in vitro studies performed on human cells and tissues in different stages of the reproductive cascade. The current literature, though limited, suggests that resveratrol may play a role in female infertility. Specifically, it may significantly and positively impact reproductive outcomes, owing to its potential therapeutic effects improving ovarian function. Further studies are now needed to better understand resveratrol’s effects and define the optimal dosage and periods of intake to maximize beneficial effects, as well as to prevent adverse outcomes on implantation, subsequent pregnancy and the fetus.
1. Introduction
Resveratrol (3,5,4-trihydroxystilbene) is a natural polyphenolic compound present in a variety of plants, foods and drinks [1]. In plants, it is produced in response to ultraviolet radiation, injury, fungal or bacterial infection and it is predominantly found in the skin of grapes, blueberries, raspberries, mulberries and peanuts [2]. Resveratrol may have many effects on human health, including antiaging, anti-inflammatory, anti-neoplastic, antioxidant, insulin-sensitizing, cardioprotective and vasodilating activities [3]. In particular, this antiaging activity is carried out through resveratrol’s ability to improve cellular mitochondrial activity and trigger a series of molecular mediators capable of counteracting some of the most important metabolic mechanisms of aging [4,5,6]. Moreover, resveratrol exhibits antioxidant properties by scavenging free radicals and enhancing the activity of antioxidant enzymes. Additionally, it exerts anti-inflammatory effects through a variety of signaling pathways inhibiting pro-inflammatory cytokines [7]. Growing evidence suggests a potential positive effect of resveratrol on the infertile population, particularly females, by potentially enhancing ovarian function, improving oocyte quality and exerting protective effects against age-related fertility decline and polycystic ovary syndrome (PCOS) [8]. In particular, in vitro, resveratrol inhibits proliferation and androgen production by theca–interstitial cells, exerting a cytostatic but not cytotoxic effect on granulosa cells, while decreasing aromatization and vascular endothelial growth factor expression [8]. Interestingly, in vivo, resveratrol treatment has been found to reduce the size of adipocytes and improve estrus cyclicity in an acyclic rat model of polycystic ovary syndrome (PCOS), also increasing the ovarian follicular reserve and prolonging the ovarian life span [9]. Another study found that after resveratrol treatment, mitochondrial membrane potential and ATP content in oocytes of aging mice was increased, resulting in the restoration of oocyte quality without adverse effects in the animals or their offspring [10]. At low doses, resveratrol activates SIRT1, an NADH-dependent deacetylase, able to program cellular energy metabolism through transcriptional regulation of the pGC-1α gene, a master regulator of mitochondrial metabolic activity, suggesting the ability of resveratrol to optimize such activity [11]. SIRT-1 can activate LKB1 by deacetylation; LKB1 phosphorylates and activates AMP-dependent kinase (AMPK) in a reciprocal positive feedback relationship. Indeed, activated AMPK itself can increase the concentration of NAD+ by promoting further activation of SIRT-1. Both AMPK and SIRT-1 are involved in the activation of PGC-1α, which is responsible for the co-activation of nuclear respiratory factors (NRF-1 and NRF-2), which induce transcription of genes involved in mitochondrial biogenesis and mitochondrial transcription factor A (TFAM). This process is directly involved in mitochondrial DNA replication [11]. Ragonese et al. confirmed in vitro and ex vivo the mitochondrial activity of resveratrol on granulosa cells, showing that activation of SIRT-1 and AMPK by resveratrol promotes increased mitochondrial membrane potential, ATP production and mitogenesis. This suggests the impact of resveratrol as an energy enhancer for granulosa cells, which are essential for oocyte development and maturation [12].
Interestingly, a limited body of literature also suggests a role of resveratrol in enhancing male fertility by improving testicular function [13] and sperm quality [14]. However, there is no consensus on the usage and posology of resveratrol. Moreover, literature on the relationship between resveratrol and female fertility is contrasting. For these reasons, the aim of this systematic review was to explore the impact of resveratrol on female fertility, examining data extracted from both in vivo and in vitro studies performed in females and human cells or tissues.
2. Methods
This systematic review with a narrative summary of the results adhered to the PRISMA [15] and MOOSE [16] statements and followed a structured protocol available under reasonable request from the corresponding author.
2.1. Search Strategy
Two investigators (AB and DP) independently conducted a literature search using the Medline, PubMed, Web of Science and Embase databases from their inception (1951, 1951, 1947 and 1900, respectively) to the 9th of May 2024. The following search strategy was used: “(3,5,4′-Trihydroxystilbene” OR “3,4′,5-Stilbenetriol” OR “trans-Resveratrol-3-O-sulfate” OR “SRT 501” OR “cis-Resveratrol” OR “Resveratrol” OR “trans-Resveratrol” OR “Resveratrol-3-sulfate”) AND (“oocytes” OR “oocytes development competency” OR “fertilization” OR “blastulation” OR “pregnancy” OR “live birth” OR “oocyte quality” OR “embryo” OR “embryo quality” OR “in vitro fertilization” OR “fertilisation in vitro” OR “IVF” OR “ICSI” OR “assisted reproduct*” OR “reproduct* medic*”). The references of the retrieved articles together with the proceedings of relevant conferences were hand-searched to identify other potentially eligible studies for inclusion in the analysis missed by the initial search or any unpublished data. The literature search, assessment of inclusion and exclusion criteria, quality assessment of studies and extraction of data were independently undertaken and verified by two investigators (AB, DP). The results were then compared, and in the case of a discrepancy, a consensus was reached with the involvement of a third senior investigator (LS). There was no language restriction applied.
2.2. Types of Studies; Inclusion and Exclusion Criteria
Studies included in the present review had to meet the following criteria: (1) the study design was retrospective, cross-sectional, prospective, a randomized clinical trial (RCT) or another experimental design; (2) the population included women seeking fertility; (3) resveratrol supplementation was included as the intervention or an exposure group; (4) the study contained a female control group not treated with resveratrol supplementation; (5) the study reported effects of resveratrol on women’s fertility.
Studies were excluded if they had a study design different from those stated in the inclusion criteria, focused on males, contained no data on resveratrol supplementation or did not investigate any aspect of fertility.
2.3. Data Extraction and Statistical Analyses
For each eligible study, two independent investigators (AB, DP) extracted the following data: name of the first author and year of publication, article type, study design, sample size, sample characteristics, intervention, outcome measures and findings.
2.4. Outcomes
The primary outcome was the effect of resveratrol supplementation on female fertility. Secondary outcomes included in vitro findings related to the effect of resveratrol on fertility cascade processes.
2.5. Assessment of Study Quality
For human studies two independent authors (DP, AB) assessed study quality using the Newcastle–Ottawa Scale (NOS) [17]. The NOS assigns a maximum of 9 points based on three quality parameters: selection, comparability and outcome. As per the NOS grading in past reviews, we graded studies as having a high (<5 stars), moderate (5–7 stars) or low risk of bias (≥8 stars) [18]. Notably, for in vitro studies, there is no general consensus and standard tool for assessing study quality.
2.6. Assessment of the Certainty of Evidence
To ascertain the certainty of the evidence, the Grading of Recommendations Assessment, Development, and Evaluations (GRADE) framework was used [19].
3. Results
As shown in Figure 1, we initially found 395 possibly eligible articles. After removing 366 papers through title/abstract screening, 29 were retrieved as full text. Of the twenty-nine full-text articles, five studies were excluded—four because they focused on male fertility and one because it assessed resveratrol levels without supplementation—leaving twenty-four studies published between 2010 and 2024 to be included in the systematic review [12,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42]. Among these, nine were performed on women seeking natural or assisted fertility [20,21,22,23,24,25,26,27,28], and fifteen were in vitro studies performed on human cells and tissues in different stages of the reproductive cascade [12,29,30,31,32,33,34,35,36,37,38,39,40,41,42]. Most of the studies (n = 10) were conducted in Europe, nine in Asia, three in South America, one in the Middle East and one in Oceania. The nine in vivo studies included a total of 9563 participants, and descriptions of the characteristics of these studies and their main findings are reported in Table 1. The main results are varied and not always consistent. Resveratrol induced a reduction in the expression of the vascular endothelial growth factor and hypoxia-inducible factor 1 genes in the granulosa cells. The number of mature oocytes, cleavage rate, fertilization rate and fertility rate were not significantly different, but the high-quality oocyte rate and high-quality embryo rate were higher in the resveratrol group [20]. It also induced modification in the miRNome reflecting transcriptomic and proteomic modification in granulosa cells. The number of fertilized good-quality oocytes increased in treated women, and a significant anticorrelation between miR-125 fold change values and biochemical pregnancy was present [21]. Resveratrol treatment was associated with statistically significant increases in the follicle output rate and follicle-to-oocyte index with no difference in the number of oocytes retrieved, biochemical pregnancy, clinical pregnancy or live birth rates [22]. The time needed to control blood pressure in resveratrol-treated women was significantly reduced, while time before a new crisis was extended. The number of treatment doses needed to control blood pressure was lower in treated women. No differences in maternal or neonatal adverse effects were observed between the two groups [23]. Again, resveratrol supplementation was associated with significantly higher numbers of oocytes and MII oocytes retrieved, a higher fertilization rate, more cleavage embryos per patient, more blastocytes per patient and more cryopreserved embryos. No significant differences in biochemical or clinical pregnancy, live birth or miscarriage rates were revealed, but a trend toward a higher live birth rate was revealed in the resveratrol group [24]. Resveratrol treatment promoted remodeling of the scarred uterus, regeneration of the endometrium and muscular cells and vascularization. It also improved the pregnancy rate compared with patients receiving placebo [25]. No difference in systolic or diastolic parameters between treated and control-group obese women were reported. All blood chemistry parameters improved compared to placebo at 30 days and significantly improved at 60 days with respect to placebo. Resveratrol also significantly improved lipid and glucose parameters at 30 to 60 days of treatment [26]. No difference in the treatment of pain in endometriosis was observed [27]. Resveratrol intake was also reported to be strongly associated with a decrease in the clinical pregnancy rate and an increased risk of miscarriage [28].
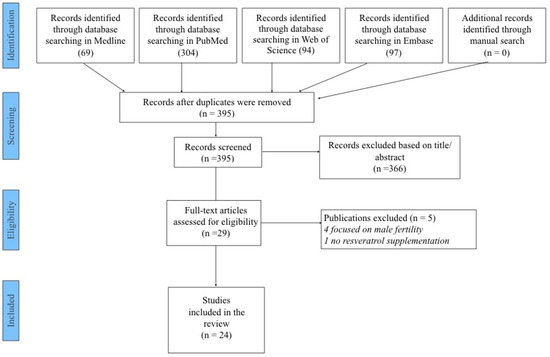
Figure 1.
PRISMA flowchart.

Table 1.
Main information and findings of included studies on fertility and reproductive outcomes.
Table 2 reports the characteristics and the main findings of the in vitro studies.

Table 2.
Main information and findings of included studies on in vitro outcomes.
No publication bias test was performed. The median quality of the studies performed on human studies was 5.9 (range: 4–9), indicating an overall satisfactory quality. Because of the high heterogeneity, the certainly of this evidence has been rated as moderate.
4. Discussion
The findings of our systematic review suggest a general positive impact of resveratrol on female reproductive health. Although the wide range of aspects considered in the included studies does not allow robust conclusions, the results do suggest a potential positive effect of resveratrol on multiple domains of the female reproductive system. The number and quality of matured oocytes were investigated by four and two studies, respectively. The number was reported as being increased in two [21,24] and having no significant differences in the others [20,22]. Both studies assessing quality reported an increased quality in women who underwent resveratrol supplementation. A possible explanation is the direct action of resveratrol in reducing oxidative stress, protecting mitochondrial DNA from damage and mutations, while enhancing telomerase activity and reducing cellular aging. Moreover, it activates sirtuin 1 (SIRT1), a key molecule in aging, which is typically reduced in aged oocytes, making them vulnerable to oxidative stress [43]. By compensating for this decreased SIRT1 expression, resveratrol may inhibit ovarian aging and extend ovarian lifespan [8,44]. Additionally, it potentially exerts positive effects on PCOS and obesity-related infertility by inhibiting pathways involved in androgen production and reducing inflammation and oxidative stress [45,46]. Moreover, in a rodent premature ovarian insufficiency model induced by chemotherapy or radiotherapy, resveratrol inhibited oxidative stress and inflammatory events in the ovaries by activating the PI3K/Akt/mTOR and NF-κB signaling pathways [47], improving loss of the oogonial stem cells through antiapoptotic effects [48]. In in vitro fertilization treatments, it was observed that resveratrol enhanced oocyte maturation and embryo development to the blastocyst stage in both animals and humans [31] and protected against postovulatory oocyte aging [49]. Fertilization and fertility rates were reported as being improved by Gerli et al. [24] and as having no difference by Bahramrezaie and colleagues [20]. The latter study also reported no difference in cleavage rate but found an increased embryo quality [20]. The follicle output rate and follicle-to-oocyte index were reported as improved in one study [22], and two studies reported no difference in terms of live birth [22,24]. Importantly, there are contrasting data regarding pregnancy rates and miscarriage indicators. The pregnancy rate was reported by five studies, with two indicating an increase [21,25], two no difference [22,24] and Ochiai et al. reporting a decrease [28]. Ochiai and colleagues also reported an increase in the miscarriage rate [28], while Gerli et al. observed no difference [24]. Moreover, during spontaneous pregnancies in overweight patients, resveratrol was found to significantly improve lipid and glucose parameters [26], and the literature suggests that it may be beneficial for the treatment of preeclampsia in combination with oral nifedipine [23]. This contrasting evidence could be explained by the fact that, since implantation requires an inflammatory response with local secretion of proinflammatory cytokines and prostaglandins from the decidualized endometrium, the anti-inflammatory properties of resveratrol might directly suppress embryo implantation. In turn, this could imply an optimization of the supplementation scheme in terms of period, dosage and duration. Finally, no effect in pain relief in women suffering endometriosis was observed [27], although in vitro studies showed antiapoptotic and antiproliferative effects with a possible role in inhibiting the progression of ectopic endometrium [9]. Thus, resveratrol may have therapeutic benefits for treating infertility associated with endometriosis, even without symptomatic effects. Taken together, all these findings do not allow putative conclusions to be drawn, but they spark optimism for the possible use of resveratrol for women seeking fertility. Many questions still need to be addressed, including the dosage, treatment duration, optimal time window and possible side and teratogenic effects. However, additional encouraging results are coming from in vitro studies that also shed light on possible mechanisms of action. Importantly, in vitro studies included in the present review assessed different aspects of the impact of resveratrol on human cells, indicating an overall potential beneficial effect of supplementation. Interestingly, its antioxidant activity was reported by multiple studies [29,30,34,39,40]. Granulosa cells’ viability was also found to be improved by supplementation [12,34,38], and other observed effects included an anti-inflammatory response [30]; oocyte maturation [31]; myometrium relaxation [35]; reduction of dehydroepiandrosterone, androstenedione and 11-deoxicortisol [37]; and neuroprotection [41]. Contrasting results were reported on decidualization. Three studies showed enhancement of in vitro decidualization [32,33,42], while Ochiai and colleagues reported anti-deciduogenic properties [36]. In this case, different methodological approaches, resveratrol doses and timing of administration may explain the contrasting results, highlighting the need to increase efforts in studying this multi-potential supplement. Despite some contrasting evidence, it seems clear that resveratrol may play an important role in female fertility management. Moreover, a limited body of literature also suggests a role of resveratrol in improving testicular function and sperm quality through enhanced protection from reactive oxygen species (ROS), which negatively impacts sperm quality by damaging mitochondrial membranes, impairing sperm motility and increasing sperm DNA damage [13,14].
The considerations drawn in this systemic review should be interpreted in the light of some limitations including the limited number of in vivo studies; the different parameters taken into account by each study and the lack of robust data on dosage, side effects and teratogenic effects. Moreover, although the quality of the studies was satisfactory overall, due to the high heterogeneity, the certainly of this evidence has been rated as moderate. Finally, the present review was not registered in the PROSPERO database.
Resveratrol is generally considered safe and well tolerated when consumed in moderate amounts through diet and in supplemental doses up to 5 g/day for a month [50,51]; however, the safety of high-dose supplementation, particularly over long periods, remains unclear. Potential side effects include gastrointestinal disturbances and interactions with medications. Therefore, it is crucial to conduct further and larger studies to determine the safe and effective dosage of resveratrol for improving female fertility. Considering its limited absorption after oral administration, particular attention should also be paid to resveratrol’s bioavailability and, in particular, to the different resveratrol-based technologies that can ameliorate resveratrol’s pharmacokinetic characteristics, exploiting its biopharmaceutical potential [52].
5. Conclusions
In conclusion, although it is not possible to define conclusive indications on resveratrol supplementation, the current evidence suggests that its utilization for women seeking fertility and during pregnancy could significantly and positively impact reproductive outcomes, particularly because of its potential therapeutic effects in improving ovarian function. Further studies are needed to better understand resveratrol’s potential in women in order to define the optimal doses and periods of intake to maximize beneficial effects and to prevent adverse effects on implantation, subsequent pregnancy and the fetus.
Author Contributions
Conceptualization, A.B. and M.C.; methodology, D.K.Y.; validation, V.G., J.S. and S.C.; formal analysis, D.P.; investigation, P.C.; data curation, P.C.; writing—original draft preparation, L.B. and A.B.; writing—review and editing, F.R.; visualization, J.G.; supervision, L.S.; project administration, A.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
Authors Damiano Pizzol and Susanna Caminada were employed by Eni SpA, but the paper is not reflecting a company involvement. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Hasan, M.; Bae, H. An Overview of Stress-Induced Resveratrol Synthesis in Grapes: Perspectives for Resveratrol-Enriched Grape Products. Molecules 2017, 22, 294. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.; Yokota, T.; Ashihara, H.; Lean, M.E.; Crozier, A. Plant foods and herbal sources of resveratrol. J. Agric. Food Chem. 2002, 50, 3337–3340. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, A.; Kuroda, K. Preconception resveratrol intake against infertility: Friend or foe? Reprod. Med. Biol. 2019, 19, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Pervaiz, S.; Holme, A.L. Resveratrol: Its biologic targets and functional activity. Antioxid. Redox Signal. 2009, 11, 2851–2897. [Google Scholar] [CrossRef]
- Gusman, J.; Malonne, H.; Atassi, G. A reappraisal of the potential chemopreventive and chemotherapeutic properties of resveratrol. Carcinogenesis 2001, 22, 1111–1117. [Google Scholar] [CrossRef]
- Park, S.J.; Ahmad, F.; Philp, A.; Baar, K.; Williams, T.; Luo, H.; Ke, H.; Rehmann, H.; Taussig, R.; Brown, A.L.; et al. Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell 2012, 148, 421–433. [Google Scholar] [CrossRef]
- Meng, T.; Xiao, D.; Muhammed, A.; Deng, J.; Chen, L.; He, J. Anti-Inflammatory Action and Mechanisms of Resveratrol. Molecules 2021, 26, 229. [Google Scholar] [CrossRef]
- Ortega, I.; Duleba, A.J. Ovarian actions of resveratrol. Ann. N. Y. Acad. Sci. 2015, 1348, 86–96. [Google Scholar] [CrossRef]
- Kolahdouz, R.; Arablou, T. Resveratrol and endometriosis: In vitro and animal studies and underlying mechanisms. Biomed. Pharmacother. 2017, 91, 220–228. [Google Scholar] [CrossRef]
- Okamoto, N.; Sato, Y.; Kawagoe, Y.; Shimizu, T.; Kawamura, K. Short-term resveratrol treatment restored the quality of oocytes in aging mice. Aging 2022, 14, 5628–5640. [Google Scholar] [CrossRef]
- Price, N.L.; Gomes, A.P.; Ling, A.J.; Duarte, F.V.; Martin-Montalvo, A.; North, B.J.; Agarwal, B.; Ye, L.; Ramadori, G.; Teodoro, J.S.; et al. SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell Metab. 2012, 15, 675–690. [Google Scholar] [CrossRef] [PubMed]
- Ragonese, F.; Monarca, L.; De Luca, A.; Mancinelli, L.; Mariani, M.; Corbucci, C.; Gerli, S.; Iannitti, R.G.; Leonardi, L.; Fioretti, B. Resveratrol depolarizes the membrane potential in human granulosa cells and promotes mitochondrial biogenesis. Fertil. Steril. 2021, 115, 1063–1073. [Google Scholar] [CrossRef] [PubMed]
- Mongioì, L.M.; Perelli, S.; Condorelli, R.A.; Barbagallo, F.; Crafa, A.; Cannarella RLa Vignera, S.; Calogero, A.E. The Role of Resveratrol in Human Male Fertility. Molecules 2021, 26, 2495. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S. DNA damage in human sperm: The sperm chromosome assay. Reprod. Med. Biol. 2022, 21, e12461. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, W-65. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin Ja Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Wells, G.A.; Tugwell, P.; O’Connell, D.; Peterson, J.; Welch, V.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. In Proceedings of the 3rd Symposium on Systematic Reviews: Beyond the Basics, Oxford, UK, 3–5 July 2000. [Google Scholar]
- Luchini, C.; Stubbs, B.; Solmi, M.; Veronese, N. Assessing the quality of studies in meta-analysis: Advantages and limitations of the Newcastle Ottawa Scale. World J. Meta-Anal. 2017, 5, 80–84. [Google Scholar] [CrossRef]
- Siemieniuk, R.; Guyatt, G. What Is GRADE. Available online: https://bestpractice.bmj.com/info/toolkit/learn-ebm/what-is-grade/ (accessed on 12 November 2022).
- Bahramrezaie, M.; Amidi, F.; Aleyasin, A.; Saremi, A.; Aghahoseini, M.; Brenjian, S.; Khodarahmian, M.; Pooladi, A. Effects of resveratrol on VEGF & HIF1 genes expression in granulosa cells in the angiogenesis pathway and laboratory parameters of polycystic ovary syndrome: A triple-blind randomized clinical trial. J. Assist. Reprod. Genet. 2019, 36, 1701–1712. [Google Scholar]
- Battaglia, R.; Caponnetto, A.; Caringella, A.M.; Cortone, A.; Ferrara, C.; Smirni, S.; Iannitti, R.; Purrello, M.; D’Amato, G.; Fioretti, B.; et al. Resveratrol Treatment Induces Mito-miRNome Modification in Follicular Fluid from Aged Women with a Poor Prognosis for In Vitro Fertilization Cycles. Antioxidants 2022, 21, 1019. [Google Scholar] [CrossRef]
- Conforti, A.; Iorio, G.G.; Di Girolamo, R.; Rovetto, M.Y.; Picarelli, S.; Cariati, F.; Gentile, R.; D’Amato, A.; Gliozheni, O.; Fioretti, B.; et al. The impact of resveratrol on the outcome of the in vitro fertilization: An exploratory randomized placebo-controlled trial. J. Ovarian Res. 2024, 17, 81. [Google Scholar] [CrossRef]
- Ding, J.; Kang, Y.; Fan, Y.; Chen, Q. Efficacy of resveratrol to supplement oral nifedipine treatment in pregnancy-induced preeclampsia. Endocr. Connect. 2017, 6, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Gerli, S.; Della Morte, C.; Ceccobelli, M.; Mariani, M.; Favilli, A.; Leonardi, L.; Lanti, A.; Iannitti, R.G.; Fioretti, B. Biological and clinical effects of a resveratrol-based multivitamin supplement on intracytoplasmic sperm injection cycles: A single-center, randomized controlled trial. J. Matern. Fetal Neonatal Med. 2022, 35, 7640–7648. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Qiao, Z. Analysis of the efficacy of resveratrol treatment in patients with scarred uterus. Exp. Ther. Med. 2018, 15, 5410–5414. [Google Scholar] [CrossRef] [PubMed]
- Malvasi, A.; Kosmas, I.; Mynbaev, O.A.; Sparic, R.; Gustapane, S.; Guido, M.; Tinelli, A. Can trans resveratrol plus d-chiro-inositol and myo-inositol improve maternal metabolic profile in overweight pregnant patients? Clin. Ther. 2017, 168, 240–247. [Google Scholar]
- Mendes da Silva, D.; Gross, L.A.; Neto, E.P.G.; Lessey, B.A.; Savaris, R.F. The Use of Resveratrol as an Adjuvant Treatment of Pain in Endometriosis: A Randomized Clinical Trial. J. Endocr. Soc. 2017, 15, 359–369. [Google Scholar] [CrossRef]
- Ochiai, A.; Kuroda, K.; Ikemoto, Y.; Ozaki, R.; Nakagawa, K.; Nojiri, S.; Takeda, S.; Sugiyama, R. Influence of resveratrol supplementation on IVF-embryo transfer cycle outcomes. Reprod. Biomed. Online 2019, 39, 205–210. [Google Scholar] [CrossRef]
- Caldeira-Dias, M.; Montenegro, M.F.; Bettiol, H.; Barbieri, M.A.; Cardoso, V.C.; Cavalli, R.C.; Sandrim, V.C. Resveratrol improves endothelial cell markers impaired by plasma incubation from women who subsequently develop preeclampsia. Hypertens. Res. 2019, 42, 1166–1174. [Google Scholar] [CrossRef]
- Hannan, N.J.; Brownfoot, F.C.; Cannon, P.; Deo, M.; Beard, S.; Nguyen, T.V.; Palmer, K.R.; Tong, S.; Kaitu’u-Lino, T.J. Resveratrol inhibits release of soluble fms-like tyrosine kinase (sFlt-1) and soluble endoglin and improves vascular dysfunction—Implications as a preeclampsia treatment. Sci. Rep. 2017, 12, 1819. [Google Scholar] [CrossRef]
- Liu, M.J.; Sun, A.G.; Zhao, S.G.; Liu, H.; Ma, S.Y.; Li, M.; Huai, Y.X.; Zhao, H.; Liu, H.B. Resveratrol improves in vitro maturation of oocytes in aged mice and humans. Fertil. Steril. 2018, 109, 900–907. [Google Scholar] [CrossRef]
- Long, C.; Li, Z.; Liang, S.; Yao, S.; Zhu, S.; Lu, L.; Cao, R.; Chen, Y.; Huang, Y.; Ma, Y.; et al. Resveratrol reliefs DEHP-induced defects during human decidualization. Ecotoxicol. Environ. Saf. 2023, 15, 258. [Google Scholar] [CrossRef]
- Mestre Citrinovitz, A.C.; Langer, L.; Strowitzki, T.; Germeyer, A. Resveratrol enhances decidualization of human endometrial stromal cells. Reproduction 2020, 159, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Pinto, B.; Costa, L.; Felgueira, E.; Fonseca, B.M.; Rebelo, I. Low Doses of Resveratrol Protect Human Granulosa Cells from Induced-Oxidative Stress. Antioxidants 2021, 4, 561. [Google Scholar] [CrossRef] [PubMed]
- Novaković, R.; Radunović, N.; Marković-Lipkovski, J.; Ćirović, S.; Beleslin-Čokić, B.; Ilić, B.; Ivković, B.; Heinle, H.; Živanović, V.; Gojković-Bukarica, L.J. Effects of the polyphenol resveratrol on contractility of human term pregnant myometrium. Mol. Hum. Reprod. 2015, 21, 545–551. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ochiai, A.; Kuroda, K.; Ozaki, R.; Ikemoto, Y.; Murakami, K.; Muter, J.; Matsumoto, A.; Itakura, A.; Brosens, J.J.; Takeda, S. Resveratrol inhibits decidualization by accelerating downregulation of the CRABP2-RAR pathway in differentiating human endometrial stromal cells. Cell Death Dis. 2019, 20, 276–281. [Google Scholar] [CrossRef]
- Savchuk, I.; Morvan, M.L.; Søeborg, T.; Antignac, J.P.; Gemzell-Danielsson, K.; Le Bizec, B.; Söder, O.; Svechnikov, K. Resveratrol inhibits steroidogenesis in human fetal adrenocortical cells at the end of first trimester. Mol. Nutr. Food Res. 2017, 61, 1600522. [Google Scholar] [CrossRef]
- Schube, U.; Nowicki, M.; Jogschies, P.; Blumenauer, V.; Bechmann, I.; Serke, H. Resveratrol and desferoxamine protect human OxLDL-treated granulosa cell subtypes from degeneration. J. Clin. Endocrinol. Metab. 2014, 99, 229–239. [Google Scholar] [CrossRef]
- Viana-Mattioli, S.; Cinegaglia, N.; Bertozzi-Matheus, M.; Bueno-Pereira, T.O.; Caldeira-Dias, M.; Cavalli, R.C.; Sandrim, V.C. SIRT1-dependent effects of resveratrol and grape juice in an in vitro model of preeclampsia. Biomed. Pharmacother. 2020, 131, 110659. [Google Scholar] [CrossRef]
- Wang, P.; Huang, C.X.; Gao, J.J.; Shi, Y.; Li, H.; Yan, H.; Yan, S.J.; Zhang, Z. Resveratrol induces SIRT1-Dependent autophagy to prevent H2O2-Induced oxidative stress and apoptosis in HTR8/SVneo cells. Placenta 2020, 91, 11–18. [Google Scholar] [CrossRef]
- Wang, Y.; Wei, T.; Wang, Q.; Zhang, C.; Li, K.; Deng, J. Resveratrol’s neural protective effects for the injured embryoid body and cerebral organoid. BMC Pharmacol. Toxicol. 2022, 12, 47–55. [Google Scholar] [CrossRef]
- Yao, S.; Wei, W.; Cao, R.; Lu, L.; Liang, S.; Xiong, M.; Zhang, C.; Liang, X.; Ma, Y. Resveratrol alleviates zea-induced decidualization disturbance in human endometrial stromal cells. Ecotoxicol. Environ. Saf. 2021, 1, 207–2014. [Google Scholar] [CrossRef]
- Di Emidio, G.; Falone, S.; Vitti, M.; D’Alessandro, A.M.; Vento, M.; Di Pietro, C.; Amicarelli, F.; Tatone, C. SIRT1 signalling protects mouse oocytes against oxidative stress and is deregulated during aging. Hum. Reprod. 2014, 29, 2006–2017. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Yin, Y.; Ye, X.; Zeng, M.; Zhao, Q.; Keefe, D.L.; Liu, L. Resveratrol protects against age-associated infertility in mice. Hum. Reprod. 2013, 28, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Furat Rencber, S.; Kurnaz Ozbek, S.; Eraldemır, C.; Sezer, Z.; Kum, T.; Ceylan, S.; Guzel, E. Effect of resveratrol and metformin on ovarian reserve and ultrastructure in PCOS: An experimental study. J. Ovarian Res. 2018, 11, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Di Emidio, G.; Santini, S.J.; D’Alessandro, A.M.; Vetuschi, A.; Sferra, R.; Artini, P.G.; Carta, G.; Falone, S.; Amicarelli, F.; Tatone, C. SIRT1 participates in the response to methylglyoxal-dependent glycative stress in mouse oocytes and ovary. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1389–1401. [Google Scholar] [CrossRef]
- Li, N.; Liu, L. Mechanism of resveratrol in improving ovarian function in a rat model of premature ovarian insufficiency. J. Obs. Gynaecol. Res. 2018, 44, 1431–1438. [Google Scholar] [CrossRef]
- Wu, M.; Ma, L.; Xue, L.; Ye, W.; Lu, Z.; Li, X.; Jin, Y.; Qin, X.; Chen, D.; Tang, W.; et al. Resveratrol alleviates chemotherapy-induced oogonial stem cell apoptosis and ovarian aging in mice. Aging 2019, 11, 1030–1044. [Google Scholar] [CrossRef]
- Liang, Q.-X.; Lin, Y.-H.; Zhang, C.-H.; Sun, H.-M.; Zhou, L.; Schatten, H.; Sun, Q.-Y.; Qian, W.-P. Resveratrol increases resistance of mouse oocytes to postovulatory aging in vivo. Aging 2018, 10, 1586–1596. [Google Scholar] [CrossRef]
- Park, E.J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta 2015, 1852, 1071–1113. [Google Scholar] [CrossRef]
- Patel, K.R.; Scott, E.; Brown, V.A.; Gescher, A.J.; Steward, W.P.; Brown, K. Clinical trials of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 161–169. [Google Scholar] [CrossRef]
- Walle, T. Bioavailability of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 9–15. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).