Abstract
Irisin is a myokine with potential effects on glucose metabolism and the development of diabetes in humans. We analysed irisin serum levels (ISL) in 47 patients without diabetes before and 1, 2, 3, 4 and 5 weeks after kidney transplantation (KTx). All measurements of irisin before KTx levels were lower than 25 ng/mL (median 8.4 ng/mL). We found an outstanding increase in ISL measured after KTx, reaching more than 1000 times in 44% of patients (HIL—high irisin level group). The increase appeared at the first measurement (one week after KTx). Factors connected to the large growth of ISL were, i.e., BMI > 30 (p = 0.04) and subsequent KTx—second and third (p < 0.001). The global mean blood glucose level during the first two weeks after KTx was significantly lower in the HIL group (p = 0.002), the same as the day-by-day analysed mean fasting and postprandial serum glucose in the first days after KTx. In 12 months of observation, diabetes requiring insulin therapy occurred in the HIL group at a rate of 19%, while in the rest of the patients, the rate was 27%, p = 0.526. Irisin levels increase significantly in some patients after kidney transplantation, accompanied by lower blood glucose levels in the early post-transplant period. Whether an increase in irisin levels results in better glycaemic control remains questionable and requires further research, as well as the relationship between irisin levels and the occurrence of PTDM.
1. Introduction
There is increasing evidence that skeletal muscle functions as an internal secretory organ [1]. Irisin is a myokine. It is mainly released from muscles in response to exercise and is widely distributed in the human body, and it binds to a yet undiscovered receptor on the surface of other tissues; however, in some tissues, it probably acts via a specific class of integrin receptors (αV/β5) [2]. Irisin affects the metabolism of the body and the communication between tissues. It was discovered in 2012 and was named after the ancient Greek goddess, the personification of the rainbow and messenger of the gods [3]. Irisin is a glycosylated protein hormone consisting of 112 amino acids, a PGC-1α (proliferator-activated receptor-γ coactivator-1α)-dependent myokine, a fragment of the extracellular domain of the FNDC5 (fibronectin type III domain-containing protein 5) transmembrane protein [3]. Irisin forms a continuous intersubunit β-sheet dimer [4]. The C-terminal fragment of FNDC5 is located in the cytoplasm, whereas the extracellular N-terminal portion is proteolytically cleaved to produce irisin. Irisin is completely conserved among vertebrates and has a 100% similarity between mice and humans [4].
The proposed beneficial effects of irisin include the browning of white adipose tissue and increased thermogenesis, which promotes insulin sensitivity, increased body weight, and glucose tolerance in mice [3,5,6]. Irisin has been proposed to improve glucose homeostasis by increasing fatty acid oxidation and utilising glucose via the AMPK signalling pathway in diabetic mice [7]. In human subjects, elevated circulating irisin levels are associated with a lower risk of insulin resistance in obese adults [8,9]. However, it was also reported that a high irisin level is associated with a higher risk of metabolic syndrome and cardiovascular disease, which could be the result of a compensatory increase in the secretion of irisin by muscle tissue and/or to overcome underlying irisin resistance in metabolically affected patients [10].
Glucose tolerance disorders are a common complication in the post-renal transplantation period, and they are characterised by a combination of insulin resistance and insulin hyposecretion. Risk factors for PTDM (post-transplant diabetes mellitus) include both general diabetes risk and some specific to transplant patients, such as immunosuppression. PTDM is associated with an increased risk of renal graft failure, cardiovascular disease and death [11,12,13,14]. A problem in daily practice in transplant centres is acute hyperglycaemia occurring in up to 90% of kidney recipients in the first weeks after transplantation [15,16]; most patients show an improvement in glucose tolerance after reducing the dose of immunosuppressive drugs, although early development of hyperglycaemia is a strong predictor of PTDM [17]. PTDM occurs in about 20–30% of kidney transplant recipients [11,18]. The role of irisin in glucose metabolism and the development of diabetes in humans is controversial. Irisin serum concentrations were not previously analysed in transplant patients and in post-transplant diabetes mellitus (PTDM). We also investigated the possible association between circulating irisin levels and glucose metabolism and diabetes after kidney transplantation.
2. Results
2.1. Irisin Serum Levels (ISLs) in Patients before and after KTx in the Studied Group
All measurements of ISL before KTx levels were lower than 25 ng/mL (median 8.4 ng/mL).
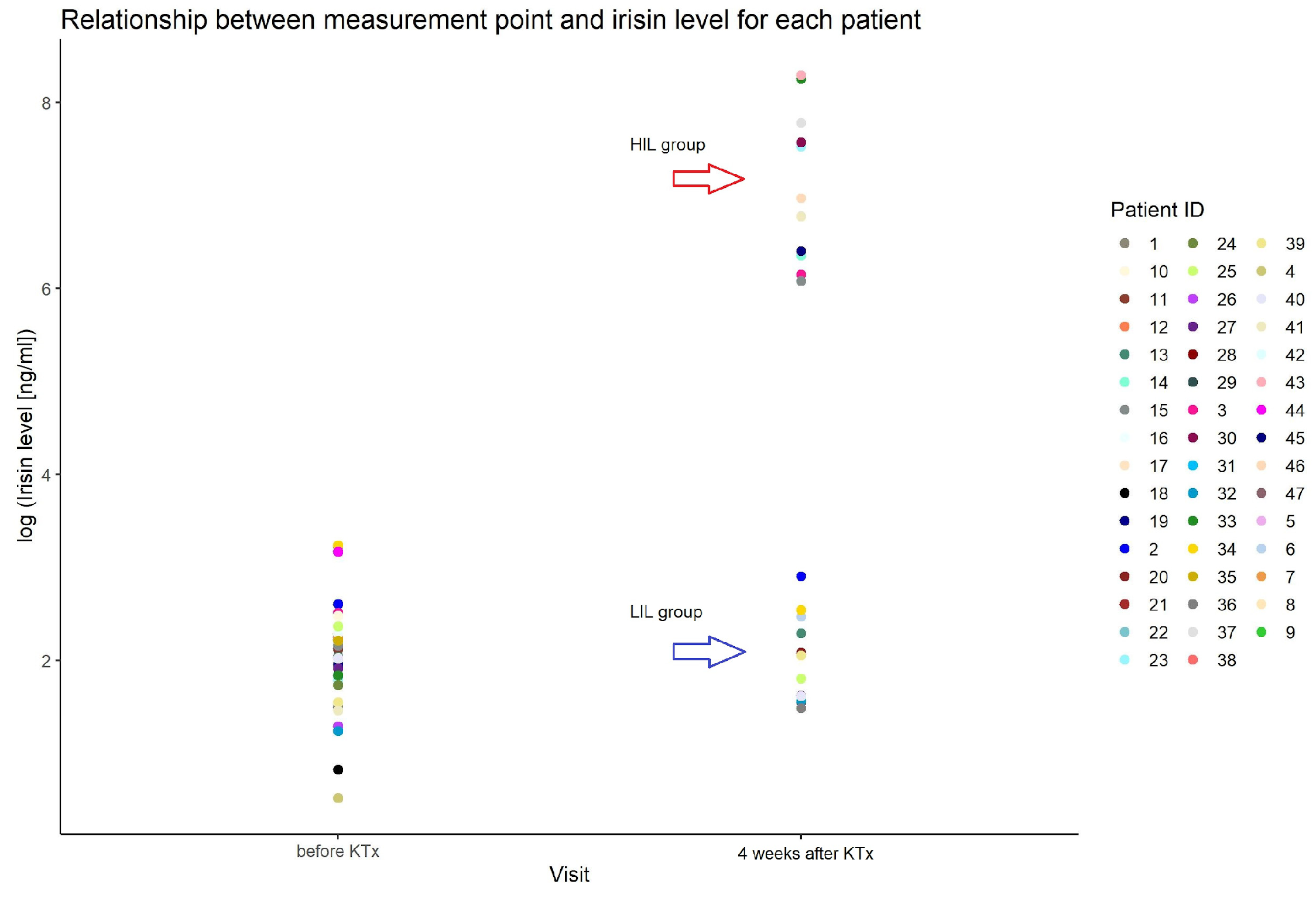
We found an outstanding increase in ISL measured after KTx, reaching more than 1000 times in 44% of patients (21/47) (HIL). The increase appeared at the first measurement (one week after KTx) and remained stable for at least a month. The median value of all measurements after the KTx procedure in the group with an increase in ISL was 1220 ng/mL (min–max: 158–5884), and in the group without irisin increase (LIL), it was 6 ng/mL (min–max: 2–20) (Figure 1).
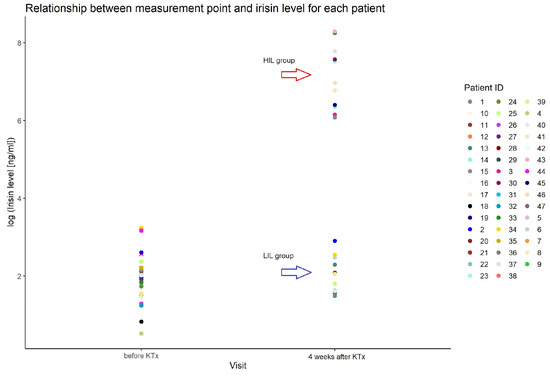
Figure 1.
Irisin serum level in patients before and after KTx in studied group.
2.2. Clinical and Biochemical Factors Analysed Comparatively in HIL and LIL of Patients after KTx
All the factors analysed are listed in Table 1.

Table 1.
Characteristics of the study group of patients after KTx and comparison of groups with and without an irisin increase (HIL and LIL group).
2.2.1. Factors That Are Statistically Significantly Associated with a Large Growth in ISL
- a.
- Obesity
In the HIL group, there were more obese patients with BMI > 30 kg/m2 (p = 0.04).
- b.
- Duration of renal replacement therapy and subsequent KTx—second and third
The duration of renal replacement therapy (total time spent on dialysis and transplantation, counted from the first dialysis) was longer in the HIL group, with 81.5 months in the HIL group and 26.0 months in the LIL group (p = 0.005).
Subsequent KTx—second and third—were connected to the large growth in ISL (p < 0.001). There were no patients in the LIL group after subsequent KTx.
- c.
- Cold ischaemia time
The cold ischaemic time of the transplanted kidney was longer in the HIL group (p = 0.05).
- d.
- Immunosuppression
Tacrolimus was part of immunosuppressive treatment in 89.4% of all patients; the rest of them received cyclosporin (10.6%). Tacrolimus was used equally often in both groups (HIL and LIL). The mean tacrolimus blood level on the 6th day after KTx was statistically significantly higher in patients in the HIL group, p = 0.001.
There was a positive correlation between tacrolimus and glycaemia levels by the analysis on the 6th day after KTx for fasting glucose (r(df) = 0.28; p = 0.05) and for postprandial glucose (r(df) = 0.05; p = 0.05).
In patients who received cyclosporin, its levels did not differ between the HIL and LIL groups.
All patients received only initial boluses of methylprednisolone within the first 3–4 days after KTx, followed by standard oral therapy. No patient in the entire study group had a diagnosis of acute rejection of the transplanted kidney, so none of the patients received additional doses of intravenous steroids.
The two-week cumulative dose of prednisone per kg body weight was 4.8 ± 1.1 mg/kg in the entire study group. The cumulative dose of prednisone was 4.68 ± 0.9 and 4.97 ± 1.2 mg/kg in the LIL and HIL groups, respectively (p = 0.36).
2.2.2. Factors Not Statistically Significant in the Comparison of HIL and LIL Groups
We did not find an influence of gender, age, cause of ESRD, dialysis modality before KTx or delayed graft function (DGF, defined as the need for dialysis during the first week after KTx) related to the increase in the irisin level.
The HIL and LIL groups did not differ in terms of kidney graft function one month after KTx, pre-transplant HbA1c, glucose, C-peptide, insulin, HOMA index, cholesterol and acute rejection (AR) in both groups (Table 1).
2.2.3. Glycaemic Disturbances in the Studied Group of Patients in the Time soon after KTx
The blood glucose analysis in patients after KTx in studied groups: HIL and LIL
Global mean blood glucose level during the first two weeks after kidney transplantation was statistically significantly higher in the LIL group (147.2 vs. 127.5 mg/dL, p = 0.002). Global mean blood glucose was calculated from all blood glucose measurements determined three times a day during hospitalisation for 2 weeks after kidney transplantation.
Glycaemic values above 200 mg/dL occurred in 8.9% of measurements in patients in the first two weeks after kidney transplantation in the HIL group, and this was statistically significantly less than in the LIL group (p = 0.007); in this group, 13.6% of measurements were above 200 mg/dL.
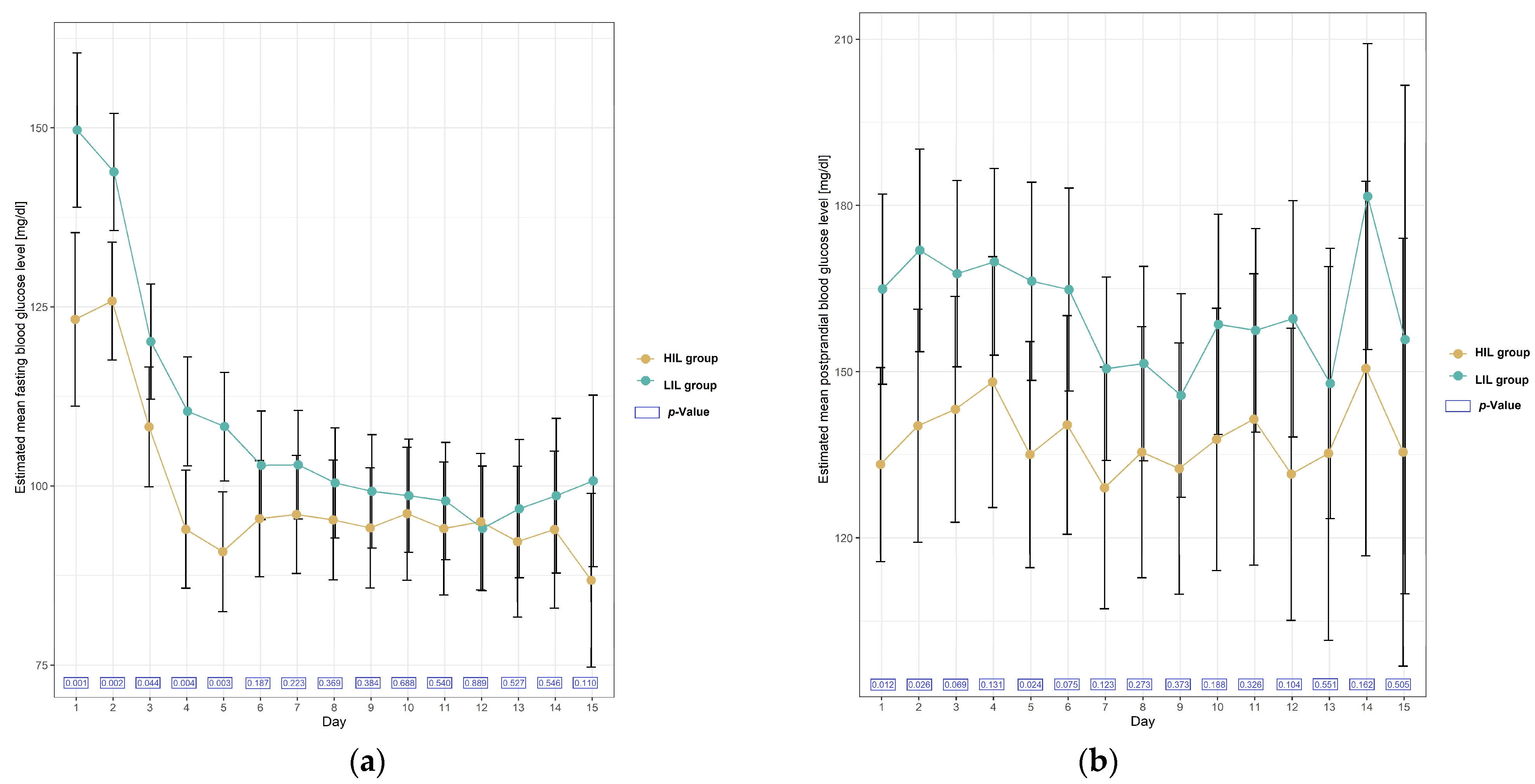
A day-by-day analysis of blood glucose levels showed that the mean fasting blood glucose level was higher almost every day in the LIL group, but the relationship was statistically significant on days 1–5 (Table 2, Figure 2).

Table 2.
Results of the linear mixed-effect model with repeated measurements of fasting blood glucose measurements for between-group differences regarding irisin subgroups (HIL and LIL).
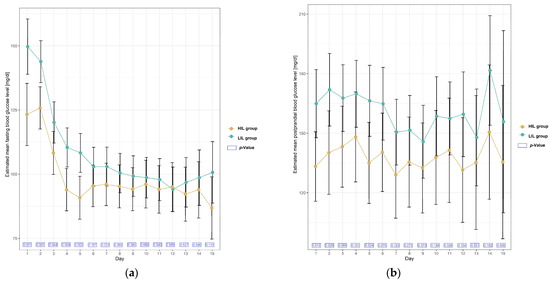
Figure 2.
Plots of estimated mean with 95% confidence interval in blood glucose level [mg/dL] in patients with and without irisin increase (HIL and LIL group) during 15 days after KTx. (a) Mean fasting blood glucose level; (b) mean postprandial blood glucose level.
A day-by-day analysis of postprandial blood glucose levels showed that mean glucose was also higher each day in the LIL group, and the relationship was statistically significant on days 1, 2 and 5 (Table 3, Figure 2).

Table 3.
Results of the linear mixed-effect model with repeated measurements of postprandial blood glucose measurements for between-group differences regarding irisin subgroups.
2.2.4. Diabetes Requiring Permanent Insulin Therapy (DM)
In the entire study group, glycaemic disturbances requiring permanent insulin therapy (DM), defined as the need for insulin therapy more than 3 months after KTx and continuing, occurred in 11 people (11/47)—23%. In the HIL group, DM occurred at a rate of 19% (4/21), while in the LIL group, it was 27% (7/26)—a difference not statistically significant (p = 0.526). All of these patients required insulin therapy at checkpoints 3 months and 12 months after kidney transplantation.
There was one more patient, aged 63, in the LIL group, who required pharmacotherapy; he was taking an oral diabetes treatment—metformin. The addition of this case does not change the result that there was no difference in the frequency of DM in the LIL and HIL groups, p = 0.36).
Gender, cause of end-stage renal disease (ESRD), dialysis method used before KTx, DGF, type of calcineurin inhibitor (tacrolimus or cyclosporin), glycaemia, HOMA and Quicki index, insulin level before KTx and BMI did not predispose to the development of DM. In the group of DM, HBA1c before transplantation was significantly higher (p = 0.021), as well as total cholesterol (p = 0.011) and LDL cholesterol levels (p = 0.017); these patients were also significantly older (58 vs. 45 years old (p = 0.05)).
2.2.5. Blood Pressure in the Studied Group of Patients after KTx
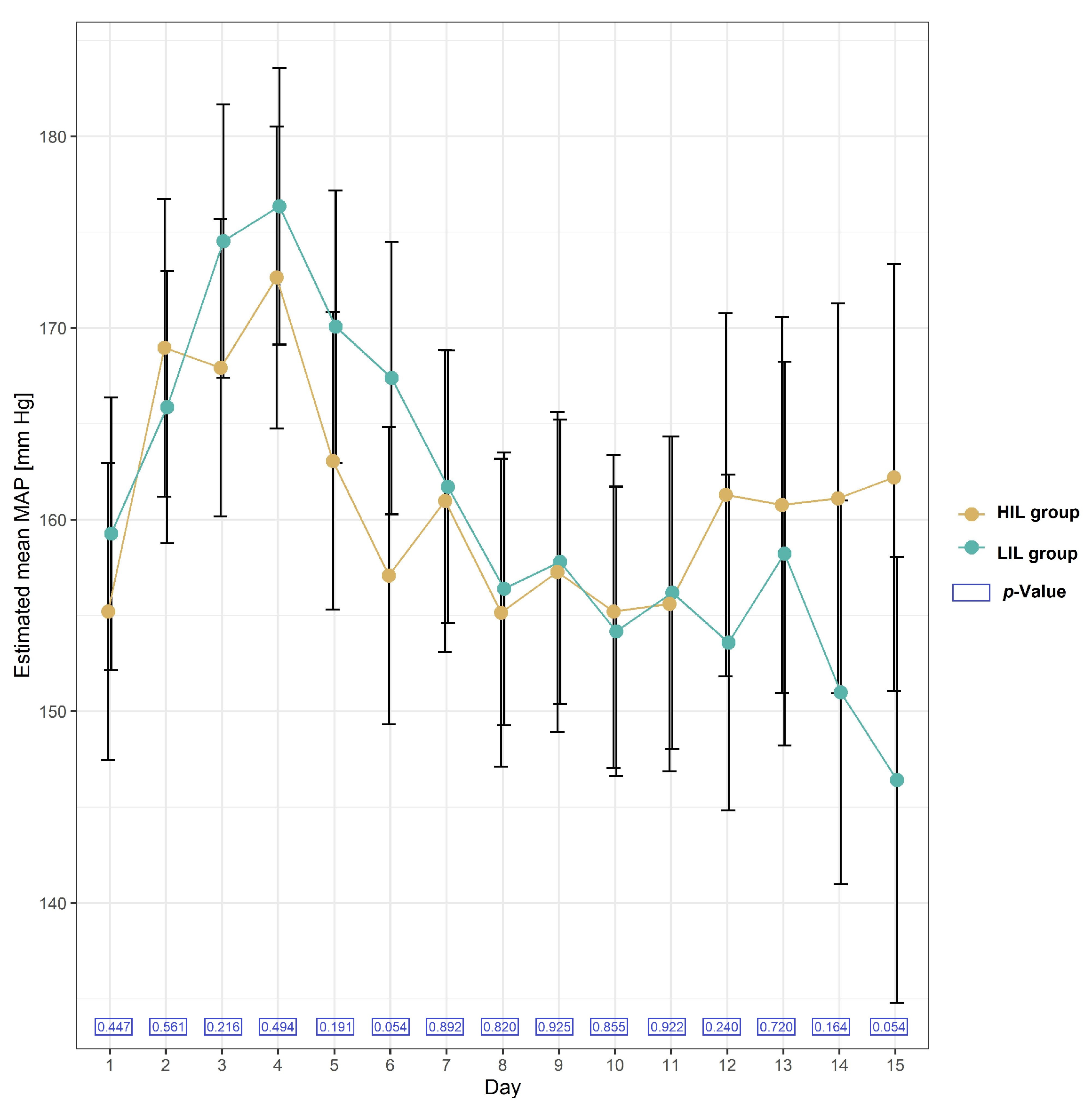
The mean values of Mean Arterial Pressure (MAP) were not significantly different in the HIL and LIL groups. The formula used to calculate MAP was as follows: MAP = DP + 1/3 (SP − DP), where DP is the diastolic blood pressure, and SP is the systolic blood pressure. The mean values of MAP on each day during the two-week follow-up after KTx in the HIL and LIL groups are presented in Figure 3.
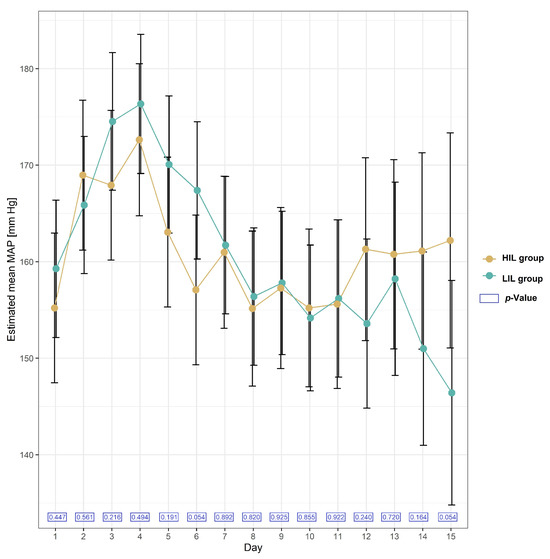
Figure 3.
Plot of estimated mean with 95% confidence interval in MAP (Mean Arterial Pressure) [mmHg] in patients with and without irisin increase (HIL and LIL group) during 15 days after KTx.
2.2.6. Obesity and Irisin Serum Levels
Irisin levels tested before kidney transplantation did not differ between patients with BMI above and below 30 kg/m2 (mean 9 vs. 8 ng/mL, p = 0.64).
Irisin levels measured one (mean 801 vs. 5303 ng/mL, p = 0.01), two (median 30 vs. 48, p = 0.06), three (mean 351 vs. 2793 ng/mL, p = 0.001) and four weeks after kidney transplantation (mean 547 vs. 1451; p = 0.12) were higher in the group with BMI above 30 kg/m2.
3. Discussion
The most important observation in our study is the surprisingly high increase in irisin levels, reaching more than 1000 times in some patients in response to the kidney transplant procedure and immunosuppression.
All measurements of ISL before KTx were lower than 25 ng/mL (median 8.4 ng/mL). Lower irisin concentrations have been previously observed in chronic kidney disease (CKD) patients [17,19,20]. The serum irisin level of patients in CKD stage 4 was significantly reduced compared with CKD stage 2 patients [21]. The lowest irisin concentrations were observed in patients with ESRD [19], and furthermore, irisin levels were lower in dialysis patients than in nondialysis patients and lower than in healthy controls [22]. Irisin is produced within muscles, and total muscle volume can affect the irisin level with the progression of kidney failure; a gradual decrease in muscle mass is observed, and it is one possible mechanism of the reduction of the irisin level.
Also, uremic toxins can contribute to decreased serum concentration of irisin in higher CKD stages. On the other hand, inflammation, oxidative stress, and activation of advanced protein glycation routes may be the causes of irisin reduction in patients with CKD [10,23]. Additionally, irisin is, in part, dialyzable; median irisin levels significantly decreased by 23% after haemodialysis as compared with predialysis concentrations [20]. In our material, irisin levels before transplantation were low in each group: peritoneal dialysis, haemodialysis and pre-emptive transplantation (without prior dialysis). We also did not find the influence of the DGF on the increase in the irisin level.
Before kidney transplantation, all patients, regardless of BMI (min–max, 18–34), had low levels of irisin in their serum. Only the transplantation procedure and the initiation of immunosuppressive treatment resulted in an outstanding increase in irisin levels, reaching more than 1000 times in 44% of patients (HIL group). Interestingly, surgery alone, for example, bariatric surgery, was not associated with a change in serum irisin levels [24,25].
This is the first report of that kind in post-transplant patients. The increase in irisine appeared at the first measurement (one week after KTx) and remained stable for at least a month.
In the HIL group, there were statistically significantly more people with BMI > 30. It remains questionable whether body composition, with BMI, muscle mass and fat mass as determinants of the nutritional status of patients with ESRD, influenced the large increase in serum irisin concentration after transplantation in the study group.
An interesting observation is that every patient who underwent a second or third KTx was characterised by a large increase in irisin. It is not surprising that the time these patients remained on renal replacement therapy (total time spent on dialysis and transplantation counted from the first dialysis) was longer—27.6 vs. 165.3 months—and included long periods of diabetogenic treatment—steroids and calcineurin inhibitors. On the other hand, in our study, second and third transplants did not predispose to the development of glycaemic disturbances requiring permanent insulin therapy. The group with and without irisin increase did not differ in the schema of immunosuppression, use of the type of calcineurin inhibitor or induction therapy (application of polyclonal or monoclonal antibodies). No patient in the entire study group had a diagnosis of acute rejection of the transplanted kidney, so all patients received only initial boluses of methylprednisolone within the first 3–4 days after KTx, followed by standard oral therapy.
In our study, the global mean blood glucose level during the first two weeks after KTx was statistically significantly higher in the LIL group. The mean fasting and postprandial blood glucose levels were higher almost every day in the LIL group; the relationship was statistically significant on days 1–5, on the days of the highest exposure to diabetogenic immunosuppressants, in particular boluses of methylprednisolone. In the following days, however, the relationship remained the same; the mean glucose concentration in the daily analysis was higher in the LIL group, although without statistical significance, probably due to the smaller number of observations, which is a limitation of the study. Whether an increase in irisin could be one of the mechanisms for managing post-transplant hyperglycaemia in renal transplant patients remains questionable. Post-transplant hyperglycaemia has a multifactorial aetiology and may be influenced by tacrolimus exposure. Tacrolimus results in not only insulin secretion deficiency but also insulin resistance. The mean tacrolimus level on day 6 after KTx was higher in the LIL group in our study. Thus, exposure to tacrolimus may be an interfering factor with glucose levels and interferes with the deduction of the effect of irisin, which is also a limitation of this study. On the other hand, it should be mentioned that although borderline significant, the correlation between tacrolimus levels and glycaemia on day 6th after KTx was weak.
In our study group, glycaemic disturbances requiring permanent insulin therapy (DM), defined as the need for insulin therapy 3 months after KTx and 12 months after KTx, were noticed at a rate of 19% and 27% in the HIL group and the LIL group, respectively. The difference was not statistically significant, most likely due to the small study group, which is a limitation of this study. Therefore, at this stage, no causal link can be made between irisin and post-transplant diabetes, as the aetiology of PTDM is multifactorial, depending on classic and specific risk factors for the period after organ transplantation.
Irisin is a myokine, and its level in the blood increases as a result of muscle activity. Assuming that an increase in irisin may be a protective mechanism against hyperglycaemia, in practice, physical exercise should be recommended to all patients after kidney transplantation, with particular emphasis on patients with risk factors for post-transplant diabetes or those with already developing glycaemic disorders.
Enlarging the study group and extending the follow-up period may allow for more far-reaching conclusions regarding the influence of irisin on glycaemic control and the development of PTDM. Additionally, the inclusion of an intervention element in the form of standardised physical activity will allow us to develop knowledge about the role of irisin in glucose metabolism.
Irisin seems to prevent obesity and insulin resistance in animal experiments [2]. In human subjects, elevated circulating irisin levels were described as associated with a lower risk of insulin resistance in obese adults [8,9]. It was also reported previously that circulating irisin concentration was negatively associated with obesity and insulin resistance and was lower in type 2 diabetes participants [26]; additionally, circulating irisin levels were significantly decreased in obese patients with insulin resistance compared to their controls [27]. In our study, obesity, with BMI > 30, was a factor connected to the significant growth of irisin in patients after KTx. In obese people, higher irisin levels may be the result of greater irisin production in the more developed adipose and muscle tissue or may reflect a compensatory increase in irisin levels to combat obesity and metabolic syndrome to overcome irisin resistance, just as elevated insulin and leptin levels occur in patients with insulin and leptin resistance.
In our study, irisin levels before transplantation were low in patients with BMI above and below 30; only after the kidney transplantation procedure and the introduction of diabetogenic immunosuppressive drugs did a large difference in irisin levels appear, levels significantly higher in obese patients, which seems to be consistent with the theory of an adaptive increase in irisin levels in patients with metabolic abnormalities associated with obesity [10].
In states of advanced obesity, irisin at physiological concentrations is unable to maintain the balance between energy storage and expenditure. In this situation, it is produced not only in muscle but also in adipose tissue [28]. Decreased irisin levels in people with T2DM may seem surprising because these people often are overweight or obese and, therefore, should rather tend to be inclined towards a higher level of myokines. Additionally, these patients develop insulin resistance, which also often correlates with elevated irisin levels. In turn, in non-obese and moderately obese patients with insulin resistance, irisin release from muscles is reduced as a result of muscle resistance to insulin. However, it remains unclear whether low irisin concentrations in T2DM describe a true cause-and-effect relationship.
The large increase in irisin level (in the HIL group) was not connected to pre-transplant HbA1c, glucose, C-peptide, insulin, HOMA index and cholesterol in our study.
Irisin stimulates nitric oxide production, which has been demonstrated in rats, and this may cause arterial relaxation and, as a consequence, lower blood pressure [8]. In our study, an increase in irisin level was not associated with the blood pressure level in patients in the early moments after KTx. Data on the relationship between irisin levels and blood pressure in human studies are conflicting [10,19,29,30,31].
4. Materials and Methods
4.1. Participants
We analysed irisin serum levels (ISL) in 47 patients without diabetes (M28, F19, mean age 48.5 years) before and after kidney transplantation (KTx) performed in our centre in 2021 and 2022. The study group (n = 47) is representative of the entire population of patients who underwent kidney transplantation at our centre (n = 232) during the study period.
All patients had end-stage renal failure before transplantation, were on haemodialysis, peritoneal dialysis or were on predialysis with GFR < 10 mL/min. None of the included patients had diabetes before kidney transplantation, and physical activity before and after transplantation was similar in all patients. Inclusion criteria: over 18 years of age, conscious and voluntary consent to participate in the study. Exclusion criteria: no informed consent to participate in this study and/or steroid therapy before transplantation. Only one patient received a kidney transplant from a living donor; the rest received kidney transplants from deceased donors (DBD (donation after brain death)).
4.2. Methods
4.2.1. Irisin Serum Level (ISL)
Blood samples (5 mL) were obtained before KTx and at the following time points: 1, 2, 3, 4 and 5 weeks after KTx.
The serum was separated after centrifugation, mixed with aprotinin (final concentration: 0.6 Unit/1 mL) in Eppendorf microtubes (GenoPlast Biotech S.A., Rokocin, Poland) and stored at −80 °C pending analysis. Quantification of serum irisin was based on competitive enzyme immunoassay kits (Phoenix Pharmaceuticals, Mannheim, Germany).
Due to post-transplant serum irisin concentration levels (SIL), the entire group was divided into subgroups with an increase in serum irisin concentration (high irisin level—HIL) and without an increase in serum irisin concentration (low irisin level—LIL).
4.2.2. Lipid Parameters, Glycaemia and Other Biochemical Parameters
Lipid parameters were measured by enzymatic methods using an Abbott Anility C analyser (Abbott, IL, USA). HbA1c was determined with the HPLC (high-performance liquid chromatography) method using a HLC 723 G11 analyser (Tosoh, Tokyo, Japan).
Glycaemic control during the post-transplant period was carried out after fasting (8 AM) and around 1 PM (non-fasting, postprandial), as taken in accordance with our centre’s procedures for the care of organ transplant patients. Some patients stayed in hospital for a shorter period, e.g., nine days; hence, systematic glycaemic monitoring was discontinued on that day. If the patient remained in hospital, measurements up to day 15 were included in the analysis.
Serum creatinine, glucose, C-peptide, insulin, homocysteine, albumin and uric acid were measured according to standard procedures in the certified laboratory of our university clinical centre directly before transplantation and before administration of immunosuppressive drugs.
4.2.3. Statistical Methodology
Statistical analysis was performed using the R statistical package (version 3.6.3).
The baseline characteristics of patients were presented as means, SD (standard deviation) and quartiles for numerical data, whereas qualitative variables were reported as counts and percentages. The differences in between-group baseline characteristics were evaluated using the unpaired t-test (or Mann–Whitney U test) for numerical data and the chi-square test for categorical variables.
A linear mixed-effect model was fitted to glucose level with time, irisin subgroups and time-by-irisin subgroup interactions as fixed effects and random intercepts. Based on this model, mean values for glucose level were estimated with 95% CI for specified measurement points and irisin subgroups. We also estimated mean within-group differences in change from measurement 1 (baseline) to each other measurement. The Kenward–Roger approximation was used to calculate denominator degrees of freedom in the t-test.
No formal adjustment for multiple testing was made. The two-tailed tests were carried out at a significant level of 0.05.
5. Conclusions
Irisin levels increase significantly in some patients after kidney transplantation, and this is accompanied by lower blood glucose levels in the early post-transplant period. Whether an increase in irisin levels results in better glycaemic control remains questionable and requires further research, as well as the relationship between irisin levels and the occurrence of PTDM.
Author Contributions
Conceptualization, B.B., A.K. (Agnieszka Kuchta) and K.S.-Z.; methodology, B.B., A.K. (Agnieszka Kuchta), K.S.-Z., A.K. (Aleksandra Krzesińska), G.C.-D., M.J. and A.D.-Ś.; software, B.B., A.K. (Agnieszka Kuchta) and K.S.-Z., validation, B.B., A.K. (Agnieszka Kuchta), K.S.-Z., M.J. and A.D.-Ś.; formal analysis, B.B., A.K. (Agnieszka Kuchta) and K.S.-Z.; investigation, B.B., A.K. (Agnieszka Kuchta), K.S.-Z., A.K. (Aleksandra Krzesińska), G.C.-D., M.J. and A.D.-Ś.; resources, B.B., A.K. (Agnieszka Kuchta), K.S.-Z., A.K. (Aleksandra Krzesińska), G.C.-D., M.J. and A.D.-Ś.; data curation, B.B., A.K. (Agnieszka Kuchta), K.S.-Z., A.K. (Aleksandra Krzesińska) and G.C.-D.; writing—original draft preparation, B.B., A.K. (Agnieszka Kuchta), K.S.-Z. and A.D.-Ś.; writing—review and editing, B.B., A.K. (Agnieszka Kuchta) and K.S.-Z.; visualization, B.B. and A.K. (Agnieszka Kuchta); supervision, M.J. and A.D.-Ś.; project administration, B.B., A.K. (Agnieszka Kuchta), K.S.-Z., A.K. (Aleksandra Krzesińska), G.C.-D., M.J. and A.D.-Ś.; funding acquisition, M.J. and A.D.-Ś. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Medical University of Gdańsk under grant nos. ST 02-20023/0004946, conducted in the Department of Nephrology, Transplantology and Internal Diseases, and ST 01-50023/0004938, conducted in the Department of Clinical Chemistry.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). All study procedures were reviewed and approved by the Independent Bioethics Committee for Scientific Research at the Medical University of Gdańsk (No. NKBBN/154/2021, approval date 26 April 2021).
Informed Consent Statement
Written informed consent was obtained from all participants involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
The authors would like to acknowledge Anna Safianowska for her contribution to the collection of material for the study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Pedersen, B.K.; Febbraio, M.A. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Wrann, C.D.; Jedrychowski, M.; Vidoni, S.; Kitase, Y.; Nagano, K.; Zhou, C.; Chou, J.; Parkman, V.A.; Novick, S.J.; et al. Irisin Mediates Effects on Bone and Fat via αV Integrin Receptors. Cell 2018, 175, 1756–1768. [Google Scholar] [CrossRef] [PubMed]
- Bostrom, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Bostrom, E.A.; Choi, J.H.; Long, J.Z.; et al. A pgc1-alpha-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- Schumacher, M.A.; Chinnam, N.; Ohashi, T.; Shah, R.S.; Erickson, H.P. The structure of irisin reveals a novel intersubunit beta-sheet fibronectin type III (FNIII) dimer: Implications for receptor activation. J. Biol. Chem. 2013, 288, 33738–33744. [Google Scholar] [CrossRef]
- Yang, X.; Enerback, S.; Smith, U. Reduced expression of FOXC2 and brown adipogenic genes in human subjects with insulin resistance. Obes. Res. 2003, 11, 1182–1191. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Y.; Wang, F.; Donelan, W.; Zona, M.C.; Li, S.; Reeves, W.; Ding, Y.; Tang, D.; Yang, L. Effects of irisin on the differentiation and browning of human visceral white adipocytes. Am. J. Transl. Res. 2019, 11, 7410–7421. [Google Scholar]
- Xin, C.; Liu, J.; Zhang, J.; Zhu, D.; Wang, H.; Xiong, L.; Lee, Y.; Ye, J.; Lian, K.; Xu, C.; et al. Irisin improves fatty acid oxidation and glucose utilization in type 2 diabetes by regulating the AMPK signaling pathway. Int. J. Obes. 2016, 40, 443–451. [Google Scholar] [CrossRef]
- Shi, X.; Lin, M.; Liu, C.; Xiao, F.; Liu, Y.; Huang, P.; Zeng, X.; Yan, B.; Liu, S.; Li, X.; et al. Elevated circulating irisin is associated with lower risk of insulin resistance: Association and path analyses of obese Chinese adults. BMC Endocr. Disord. 2016, 16, 44. [Google Scholar] [CrossRef]
- Belviranli, M.; Okudan, N.; Çelik, F. Association of Circulating Irisin with Insulin Resistance and Oxidative Stress in Obese Women. Horm. Metab. Res. 2016, 48, 653–657. [Google Scholar] [CrossRef]
- Park, K.H.; Zaichenko, L.; Brinkoetter, M.; Thakkar, B.; Sahin-Efe, A.; Joung, K.E.; Tsoukas, M.A.; Geladari, E.V.; Huh, J.Y.; Dincer, F.; et al. Circulating irisin in relation to insulin resistance and the metabolic syndrome. J. Clin. Endocrinol. Metab. 2013, 98, 4899–4907. [Google Scholar] [CrossRef] [PubMed]
- Kasiske, B.L.; Snyder, J.J.; Gilbertson, D.; Matas, A.J. Diabetes mellitus after kidney transplantation in the United States. Am. J. Transplant. 2003, 3, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Ojo, A.O. Cardiovascular complications after renal transplantation and their prevention. Transplantation 2006, 82, 603–611. [Google Scholar] [CrossRef]
- Hjelmesaeth, J.; Hartmann, A.; Leivestad, T.; Holdaas, H.; Sagedal, S.; Olstad, M.; Jenssen, T. The impact of early-diagnosed new-onset post-transplantation diabetes mellitus on survival and major cardiac events. Kidney Int. 2006, 69, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Boudreaux, J.P.; McHugh, L.; Canafax, D.M.; Ascher, N.; Sutherland, D.E.R.; Payne, W.; Simmons, R.L.; Najarian, J.S.; Fryd, D.S. The impact of cyclosporine and combination immunosuppression on the incidence of posttransplant diabetes in renal allograft recipients. Transplantation 1987, 44, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Hecking, M.; Haidinger, M.; Döller, D.; Werzowa, J.; Tura, A.; Zhang, J.; Tekoglu, H.; Pleiner, J.; Wrba, T.; Rasoul-Rockenschaub, S.; et al. Early basal insulin therapy decreases new-onset diabetes after renal transplantation. J. Am. Soc. Nephrol. 2012, 23, 739–749. [Google Scholar] [CrossRef]
- Chakkera, H.A.; Knowler, W.C.; Devarapalli, Y.; Weil, E.J.; Heilman, R.L.; Dueck, A.; Mulligan, D.C.; Reddy, K.S.; Moss, A.A.; Mekeel, K.L.; et al. Relationship between inpatient hyperglycemia and insulin treatment after kidney transplantation and future new onset diabetes mellitus. Clin. J. Am. Soc. Nephrol. 2010, 5, 1669–1675. [Google Scholar] [CrossRef]
- Chen, J.Q.; Huang, Y.Y.; Gusdon, A.M.; Qu, S. Irisin: A new molecular marker and target in metabolic disorder. Lipids Health Dis. 2015, 14, 2. [Google Scholar] [CrossRef]
- Porrini, E.L.; Díaz, J.M.; Moreso, F.; Mallén, P.I.D.; Torres, I.S.; Ibernon, M.; Bayés-Genís, B.; Benitez-Ruiz, R.; Lampreabe, I.; Lauzurrica, R.; et al. Clinical evolution of post-transplant diabetes mellitus. Nephrol. Dial. Transplant. 2016, 31, 495–505. [Google Scholar] [CrossRef]
- Ebert, T.; Focke, D.; Petroff, D.; Wurst, U.; Richter, J.; Bachmann, A.; Lössner, U.; Kralisch, S.; Kratzsch, J.; Beige, J.; et al. Serum levels of the myokine irisin in relation to metabolic and renal function. Eur. J. Endocrinol. 2014, 170, 501–506. [Google Scholar] [CrossRef]
- Wen, M.S.; Wang, C.Y.; Lin, S.L.; Hung, K.C. Decrease in irisin in patients with chronic kidney disease. PLoS ONE 2013, 8, e64025. [Google Scholar] [CrossRef]
- Sadeghi Shad, J.; Akbari, R.; Qujeq, D.; Hajian-Tilaki, K. Measurement of serum irisin in the different stages of chronic kidney disease. Casp. J. Intern. Med. 2019, 10, 314–319. [Google Scholar] [CrossRef]
- Gan, W.; Chen, W.; Li, T.; Shao, D.; Xu, F.; Huo, S.; Li, C.; Yang, Z.; Zeng, X. Circulating irisin level in chronic kidney disease patients: A systematic review and meta-analysis. Int. Urol. Nephrol. 2022, 54, 1295–1302. [Google Scholar] [CrossRef]
- Liu, J.J.; Wong, M.D.; Toy, W.C.; Tan, C.S.; Liu, S.; Ng, X.W.; Tavintharan, S.; Sum, C.F.; Lim, S.C. Lower circulating irisin is associated with type 2 diabetes mellitus. J. Diabetes Complicat. 2013, 27, 365–369. [Google Scholar] [CrossRef]
- Bidares, M.; Safari-Kish, B.; Abedi, M.; Malekzadeh-Shoushtari, H.; Jasemnezhad, M.; Azarbayejani, N.; Aziz, M.; Jahromi, S.P.; Fouladi, S.; Azizi-Soleiman, F. The Effect of Bariatric Surgery on Irisin Level: A Systematic Review and Meta-analysis. Obes. Surg. 2023, 33, 3256–3265. [Google Scholar] [CrossRef] [PubMed]
- Majorczyk, M.; Staszkiewicz, M.; Szklarczyk, J.; Major, P.; Pisarska, M.; Wysocki, M.; Stefura, T.; Kacprzyk, A.; Droś, J.; Hołda, M.K.; et al. The influence of bariatric surgery on serum levels of irisin and nesfatin-1. Acta Chir. Belg. 2019, 119, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Navarrete, J.M.; Ortega, F.; Serrano, M.; Guerra, E.; Pardo, G.; Tinahones, F.; Ricart, W.; Fernandez-Real, J.M. Irisin Is Expressed and Produced by Human Muscle and Adipose Tissue in Association With Obesity and Insulin Resistance. J. Clin. Endocrinol. Metab. 2013, 98, E769–E778. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Han, Y.; Wang, J.; Liu, Y.; Zheng, S.; Zhou, L.; Jose, P.A.; Zeng, C.; Zhang, Y.; Song, H.; et al. Irisin lowers blood pressure by improvement of endothelial dysfunction via AMPK-Akt-eNOS-NO pathway in the spontaneously hypertensive rat. J. Am. Heart Assoc. 2016, 5, e003433. [Google Scholar] [CrossRef] [PubMed]
- Pardo, M.; Crujeiras, A.B.; Amil, M.; Aguera, Z.; Jiménez-Murcia, S.; Baños, R.; Botella, C.; de la Torre, R.; Estivill, X.; Fagundo, A.B.; et al. Association of irisin with fat mass, resting energy expenditure, and daily activity in conditions of extreme body mass index. Int. J. Endocrinol. 2014, 2014, 857270. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Shi, X.; Zhang, H.; Pan, L.; Ma, Z.; Liu, S.; Liu, Y.; Li, X.; Yang, S.; Li, Z. Association of serum irisin with metabolic syndrome in obese Chinese adults. PLoS ONE 2014, 9, e94235. [Google Scholar] [CrossRef]
- Sesti, G.; Andreozzi, F.; Fiorentino, T.V.; Mannino, G.C.; Sciacqua, A.; Marini, M.A.; Perticone, F. High circulating irisin levels are associated with insulin resistance and vascular atherosclerosis in a cohort of nondiabetic adult subjects. Acta Diabetol. 2014, 51, 705–713. [Google Scholar] [CrossRef]
- Brondani, L.A.; Boelter, G.; Assmann, T.S.; Leitão, C.B.; Canani, L.H.; Crispim, D. Irisin-encoding gene (FNDC5) variant is associated with changes in blood pressure and lipid profile in type 2 diabetic women but not in men. Metabolism 2015, 64, 952–957. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).