Abstract
Rotavirus remains one of the leading causes of severe gastroenteritis, particularly among infants and young children, despite the introduction of effective vaccines. Although the global burden of rotavirus-associated morbidity and mortality has decreased in recent years, significant challenges remain regarding accurate diagnosis, optimal clinical management, and equitable access to preventive measures. The aim of this narrative review is to provide a comprehensive synthesis of recent advances in the diagnosis and treatment of rotavirus infections. Particular emphasis is placed on post-guideline research emerging after the publication of the Evidence-Based Guidelines from the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) and the European Society for Pediatric Infectious Diseases (ESPID), offering updated perspectives on therapeutic strategies and clinical practices. In addition, this review discusses the expanding role of molecular diagnostic methods, which offer enhanced sensitivity and specificity in the detection of rotavirus, and evaluates novel antiviral agents under investigation. By integrating and analyzing the most relevant research published within the past decade, we aim to delineate key progress, identify persistent gaps in knowledge, and propose future directions for research and clinical application.
1. Introduction
Rotavirus (RV) is the primary cause of severe gastroenteritis in children under five years of age [1]. First identified in 1973 from duodenal biopsies and fecal specimens of individuals with acute gastroenteritis (AGE), RV remains a significant global health concern [2]. Before the advent of RV vaccines, RV infections were a leading cause of severe pediatric AGE, contributing to approximately 500,000 deaths among children each year. They were implicated in 30% to 50% of all hospital admissions for AGE in children under the age of five, underscoring their substantial global burden prior to widespread immunization efforts [3]. In high-income countries with established vaccination programs, the incidence of RV infection has declined significantly. However, in low- and middle-income countries where vaccine coverage is limited, RV remains a leading cause of life-threatening diarrhea in young children [4].
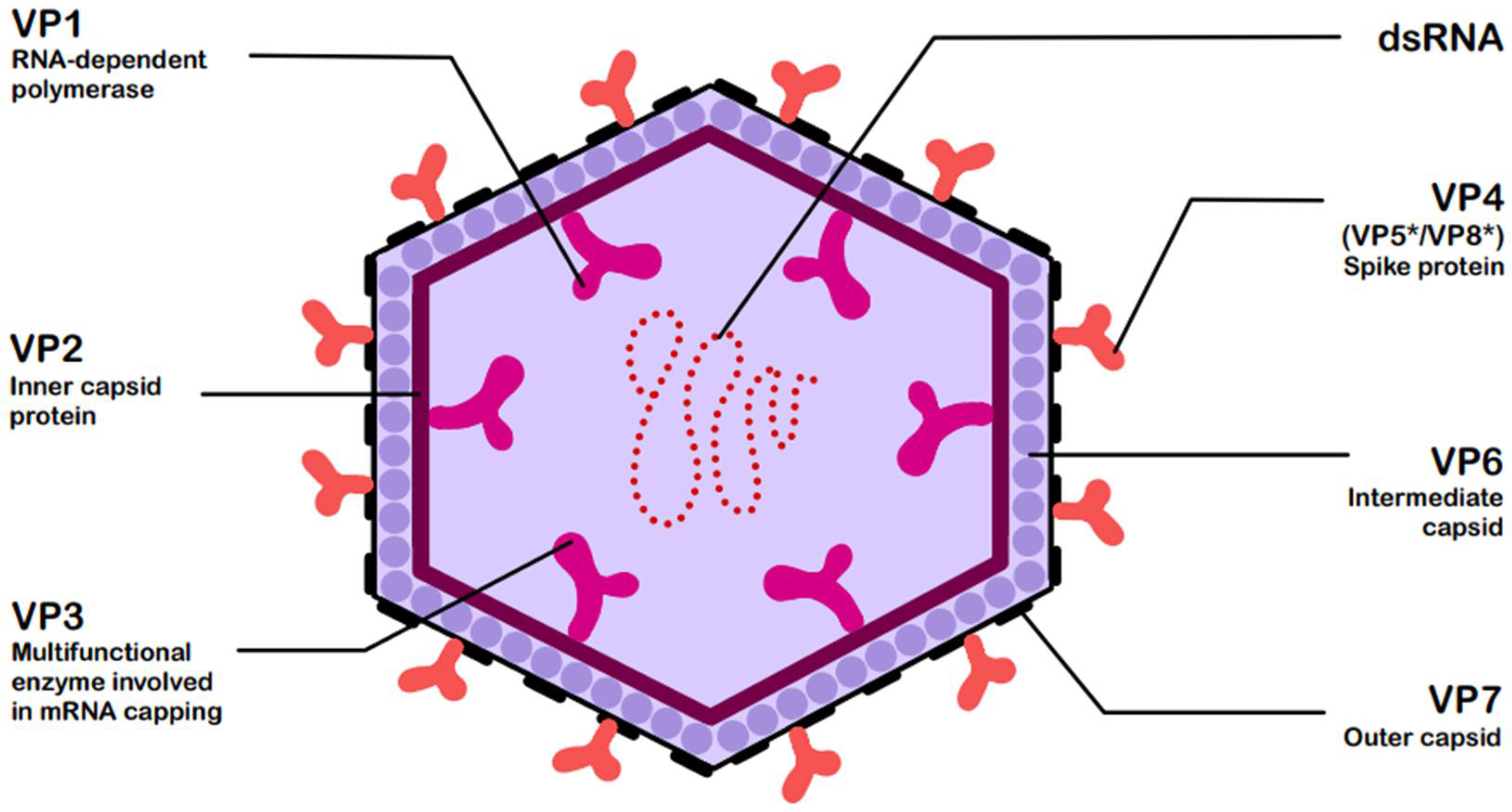
RVs are non-enveloped, double-stranded RNA (dsRNA) viruses classified by the current International Committee on Taxonomy of Viruses (ICTV) system within the genus Rotavirus and family Sedoreoviridae [5]. Based on antibody reactivity and VP6 protein sequence identity, the recognized rotavirus species include A, B, C, D, E, F, G, H, I, and J [2], among which species A is responsible for over 90% of infections, while species B and C are only sporadically detected in humans [6,7]. They are characterized by a structure consisting of three concentric capsids, enclosing a genome composed of 11 dsRNA segments. All segments encode 12 viral proteins: 6 structural (VP1–VP7) and 6 non-structural (NSP1–NSP6) [6]. Figure 1 presents the structure of rotavirus and the associated functions of its structural viral proteins (VPs) [6,8].
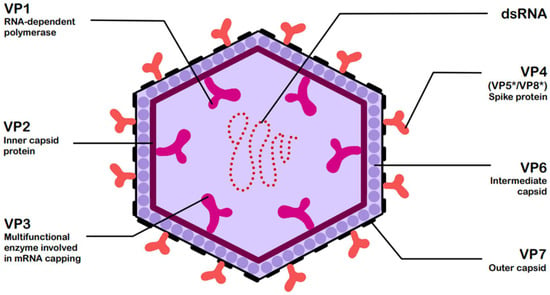
Figure 1.
Structure of rotavirus (RV) with functions of structural viral proteins (VPs) [6,8]. The asterisk * denotes these trypsin-cleaved, activated fragments.
Group A rotaviruses (RVA) are classified into distinct genotypes—42 for VP7 (G types) and 58 for VP4 (P types)—enabling the use of the binomial genotyping system for confirmatory vaccines design and characterization [7].
RVs are highly contagious and environmentally stable, facilitating their broad dissemination. They are transmitted primarily by the fecal–oral route, through both direct person-to-person contact and indirect exposure to contaminated surfaces or objects. Shedding of the virus in the feces can begin 2 days before the onset of symptoms and persist for several days after their resolution. Very high concentrations of the virus are present in the feces of infected individuals, making RV one of the most contagious pathogens causing diarrhea [1,4]. Clinical RV infection may involve the shedding of more than 1012 virus particles per gram of feces. The virus remains viable for months, contributing to its widespread transmission. In temperate climates, RV illness is most common during colder months, likely due to increased transmission within households and communities [2,9]. Hospital-acquired infections (HAIs) are common, and the virus can become endemic in neonatal hospital nurseries. While most human RV infections are not zoonotic—despite RVAs being widespread in young mammals—humans can occasionally be infected with reassortant strains originating from both human and animal sources. The role of such strains in driving new epidemics, either locally or globally, is still unclear [10].
Clinical manifestations typically include profuse diarrhea, vomiting, fever, and general malaise [1,11]. Moreover, emerging research suggests that RV may play a contributory role in a range of extraintestinal manifestations, including central nervous system complications, autoimmune conditions, biliary atresia, and certain respiratory tract disorders [8,12]. The combination of diarrhea and vomiting frequently leads to severe dehydration and reduced oral intake, often necessitating hospitalization and, if untreated, potentially leading to death [1,2].
RV antigens can be detected in stool samples using enzyme-linked immunosorbent assay (ELISA) and immunochromatographic (ICA) methods, while reverse transcription polymerase chain reaction (RT-PCR) assays are employed for the detection of viral RNA [1]. RT-PCR offers higher sensitivity and allows for viral genotyping. Confirmation of RV infection relies on stool sample testing through validated diagnostic assays [2,4]. Currently, RT-PCR is considered the reference standard for RV diagnosis. However, its widespread clinical application is limited by high costs, extended turnaround times, and the need for specialized laboratory infrastructure [13]. Antigen-based tests typically detect RV virions for up to seven days following symptom onset, whereas RT-PCR can identify viral RNA for extended periods—ranging from 4 to 57 days. Notably, RT-PCR may yield positive results in approximately 29% of asymptomatic children under one year of age. Given the simplicity, rapid turnaround, and low cost, ICA-based antigen detection is often recommended for routine use, especially in resource-limited settings [1,13]. However, false-positive antigen results are relatively common in recently vaccinated children, particularly outside hospital settings. These false positives can be misinterpreted as vaccine failure, potentially undermining public confidence in vaccine efficacy. As such, diagnostic testing plays a critical role not only in clinical management but also in disease surveillance and assessment of vaccine performance. To accurately distinguish vaccine-derived strains from wild-type viruses, routine RT-PCR testing is recommended in infants [2,4].
This review offers a comprehensive overview of the diagnostic and therapeutic approaches to RV AGE and is an in-depth analysis of recent advancements in RV diagnostics and treatment strategies. Importantly, this review emphasizes the role of molecular diagnostics in RV detection, showcasing advanced methods that markedly enhance sensitivity, specificity, and turnaround time. We also assess recent therapeutic studies published since the ESPGHAN/ESPID Evidence-Based Guidelines, highlighting the most up-to-date clinical evidence. Additionally, we examine the antiviral potential of investigational agents—ranging from natural compounds and host-directed molecules to pharmacological inhibitors—targeting distinct stages of the RV life cycle. This review synthesizes recent studies to map advances, outline remaining challenges, and suggest future directions in RV research. It serves two purposes—acquainting laboratory and diagnostic specialists with current molecular methods for RV detection and providing clinicians with evidence-based guidance on therapeutic options, ranging from established standards to emerging antivirals and supportive care.
2. Diagnostic Methods for RV Infections
2.1. Clinical Diagnosis
The clinical presentation of RV AGE varies widely, ranging from asymptomatic or mild, self-limiting watery diarrhea to severe illness characterized by frequent, profuse diarrhea accompanied by vomiting and fever. In severe cases, this may lead to dehydration, hypovolemic shock, electrolyte disturbances, and potentially death. Following an incubation period of 1 to 3 days, RV infection typically begins abruptly and presents with a range of clinical manifestations [14]. The most common clinical features include fever, diarrhea, and vomiting, which may occur individually or in combination. In one study of hospitalized children presenting with diarrhea, vomiting, and unexplained fever, the most frequent symptom combination was concurrent diarrhea, vomiting, and fever (63%). Overall, 97% of cases involved diarrhea and/or vomiting, with 91% presenting with diarrhea either alone or alongside other symptoms [14,15]. Approximately 30–40% of affected children develop moderate fever, with temperatures exceeding 39 °C. Vomiting generally subsides within one to two days, whereas gastrointestinal symptoms typically resolve within three to seven days [16].
Assessment of diarrhea-related dehydration relies on several validated clinical scoring systems, each characterized by distinct criteria, strengths, and limitations. As shown in Table 1, the Vesikari Scoring System (VSS), the Clark scale, and the Clinical Dehydration Scale (CDS) emphasize clinical features such as frequency of diarrhea, vomiting episodes, and degree of dehydration, whereas the Dehydration: Assessing Kids Accurately (DHAKA) and the Centre for Infectious Disease Research in Zambia (CIDRZ) scales were designed for rapid application in low-resource or high-burden settings [17,18,19,20,21,22,23].

Table 1.
Comparison of parameters used in scales (the Vesikari Scoring System (VSS), the Clark scale, the Clinical Dehydration Scale (CDS), the Dehydration: Assessing Kids Accurately (DHAKA) scale, the Centre for Infectious Disease Research in Zambia (CIDRZ) scale, the 10- and 4-point Gorelick scale for dehydration, the World Health Organization (WHO) scale for dehydration) assessing the severity of rotavirus (RV) acute gastroenteritis (AGE) in Children [17,18,19,20,21,22,23].
These distinctions have important implications for both clinical practice and research. For example, the VSS, which is frequently employed in clinical trials for RV vaccines, offers a comprehensive disease severity score based on symptom duration and treatment intensity; however, because it relies on meticulous clinical monitoring, it is less suitable for standard care [24,25]. In contrast, despite their moderate accuracy, the CDS and Gorelick scales are more convenient to use in hectic pediatric settings [26]. The Gorelick scales, which include both 10- and 4-point versions, emphasize practical bedside evaluation of dehydration severity. The WHO and Gorelick scales, however, have limited diagnostic utility according to systematic evidence, particularly in children with acute gastroenteritis [27].
The DHAKA and CIDRZ scales were developed and validated in low-resource clinical settings to improve dehydration assessment during acute pediatric diarrheal illness. Strong inter-rater reliability and positive likelihood ratios, along with empirical data from a cohort of patients with acute diarrhea in Dhaka, Bangladesh, supported the diagnostic accuracy of the DHAKA Dehydration Score and Decision Tree in under-resourced environments [20]. The CIDRZ diarrhea severity scoring tool was developed and evaluated through outpatient surveillance in Lusaka, Zambia. It performed similarly to the DHAKA score and exhibited high reliability and internal consistency [21]. When taken as a whole, these context-specific tools demonstrate the usefulness of locally developed, validated scales designed for environments with few clinical resources.
Therefore, Table 1 highlights the variety of dehydration assessment instruments as well as the crucial significance of selecting tools based on context. While frontline clinicians in resource-limited settings may benefit from simpler tools like the DHAKA scale, which maintain a balance between practicality and reasonable accuracy, providers in tertiary hospital or research settings may prefer the comprehensive and trial-aligned Vesikari or Clark scales.
Among them, the Vesikari scale [17] and the Clark scale [18] are widely adopted tools, initially developed to gauge the severity of acute AGE, particularly in the context of evaluating RV vaccine efficacy. Figure 2 presents a comparison of both scales scores [17,18].

Figure 2.
Comparison of Clark and Vesikari Scale Scores [17,18].
The Vesikari scale has become one of the most widely adopted tools for assessing pediatric gastroenteritis severity, supported by a 2011 manual that standardized its use and facilitated score calculation [28,29,30]. Its modified version—the Modified Vesikari Severity Score (MVSS)—has since been validated in large multicenter cohorts. Table 2 summarizes the key differences between the original VSS and the MVSS, providing a quick reference for clinical and research use. Wikswo et al. demonstrated in more than 14,000 US children that the MVSS strongly correlated with illness duration, hospitalization, and both school and parental work absenteeism [31]. Similarly, Schnadower et al., in a prospective study across five US emergency departments, confirmed the scale’s reliability, validity, and generalizability, with scores correlating with dehydration, hospitalization, and absenteeism [32], highlighting its utility even in acute care settings.

Table 2.
Comparison of parameters used in the Vesikari Score System (VSS) and the Modified Vesikari Severity Score (MVSS) [30,33].
When compared with the Clark scale, the Vesikari system appears more sensitive. A London study of 200 children with RV-induced gastroenteritis found that 57% were classified as severe by the Vesikari scale compared with only 1.5% by the Clark scale (p < 0.001), with 24% of cases labeled mild by the Clark scale identified as severe by the Vesikari scale [24]. This difference is likely due to the absence of a dehydration component in the Clark scale, which reduces its sensitivity. Consistent results from India also showed that the Clark scale underestimates clinically severe cases requiring intravenous rehydration [24,34]. Consequently, the Vesikari scale is a more effective endpoint measure in clinical trials evaluating severe RV gastroenteritis and vaccine efficacy than the Clark scale [24,35,36].
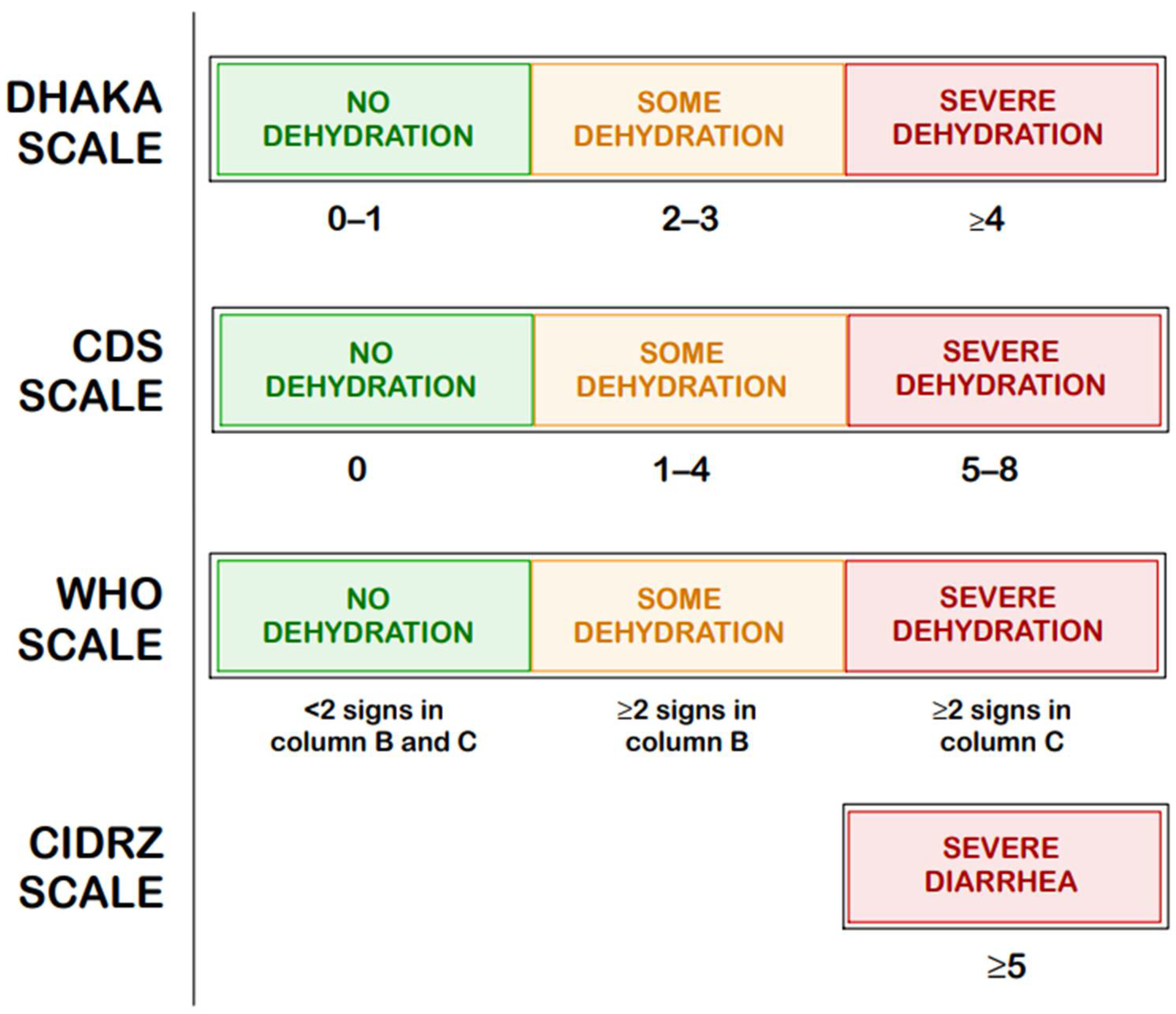
In contrast, the CDS [19], the DHAKA score [20], the more recently developed CIDRZ scale by St. Jean et al. [21], Gorelick scales (4-points and 10 points) [22], and the WHO dehydration scale [23] focus specifically on assessing the degree of dehydration regardless of its etiology; their scores are compared in Figure 3 [19,20,21,22,23].
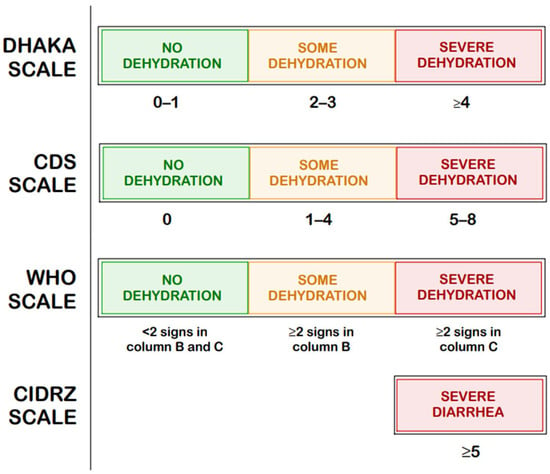
Figure 3.
Comparison of the Clinical Dehydration Scale (CDS), The Dehydration: Assessing Kids Accurately (DHAKA) Scale, Centre for Infectious Disease Research in Zambia (CIDRZ) Scale, and the dehydration scale by the World Health Organization (WHO) [19,20,21,22,23].
These tools play an important role in both clinical decision-making and research settings, especially in low-resource environments. However, as highlighted by the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition/ European Society for Pediatric Infectious Diseases (ESPGHAN/ESPID) Evidence-Based Guidelines Working Group, currently available scoring systems may offer clinical utility, yet no single tool has achieved universal acceptance or standardization in routine assessment [37]. St. Jean et al. summarizes the precision–recall area under the curve (PR-AUC) values for the Vesikari, Clark, DHAKA, and CIDRZ scales (the latter developed by the authors) collecting data from March 2019 to July 2019 in Luska, Zambia, as shown in Table 3 [21]. These results indicate that dehydration-focused tools, such as the DHAKA and CIDRZ scores outperform the Vesikari and Clark scales in predicting clinical outcomes in young children, particularly in resource-limited settings.

Table 3.
Comparative performance of dehydration scoring tools in predicting clinical outcomes in children under five years of age [21].
Hoxha et al. [38], in a prospective study, evaluated the diagnostic performance of three clinical dehydration assessment tools—the WHO scale, the Gorelick scales, and the CDS—in children aged 1 month to 5 years with acute diarrhea, using ROC curve analysis against the gold standard of percent weight change with rehydration, as presented in Table 4 [38]. In this study cohort, the CDS was the least effective tool for predicting dehydration, performing notably worse than both the WHO dehydration scale and the Gorelick scales.

Table 4.
Diagnostic performance of clinical dehydration scales in children aged 1 month to 5 years with diarrhea [38].
Despite continuous advancements in clinical practice, a universally endorsed and validated scale for reliably quantifying the severity of diarrheal disease and dehydration across diverse geographic and clinical settings is still lacking.
Hospital admission for AGE should be considered in the presence of profound dehydration exceeding 9% of body weight, neurologic impairment such as lethargy, stupor, or seizures, intractable or bilious vomiting, failure of oral rehydration therapy, clinical suspicion of surgical pathology, or when safe outpatient care and adequate home monitoring cannot be ensured [37].
2.2. Laboratory Diagnosis
Electron microscopy (EM) was the first method used for the diagnosis of RVs. Since the 1980s, ELISA tests have been commonly used, providing satisfactory results compared to EM. Recently, molecular techniques such as RT-PCR have replaced other diagnostic tests, and RT-PCR is now considered the gold-standard method for RV detection; however, these techniques require expensive equipment and advanced technical skills [39].
Traditional diagnostic methods are lengthy to operate and technically demanding [40]. Culturing the virus in cell lines is challenging and is not employed for diagnostic purposes. Additionally, serological tests lack clinical relevance because the majority of the population already possesses RV-specific antibodies [41]. EM is highly specific for RV detection and has a sensitivity comparable to some enzyme immunoassays (EIA), but lower than that of PCR. However, its use in routine testing of large numbers of stool samples is limited due to its time-consuming nature. Additionally, EM requires expensive equipment and highly trained personnel, and it cannot distinguish between different groups of RVs [42].
To detect viruses via EM, viral titers of approximately 106/mL are necessary; such levels of viral shedding are typically observed during the first 48 h of infection. The sensitivity of detection can be enhanced 10- to 100-fold with immune EM, which also improves specificity. However, the availability of specific reagents remains relatively limited [43].
The latex agglutination test is rapid (less than 15 min), easy to use, and does not require sophisticated technology to perform, which makes it a useful tool for detecting epidemic outbreaks [44].
ELISA is one of the most widely used tests due to its high sensitivity, specificity, and relatively low cost, with the ability to process up to 96 samples simultaneously. However, it is time-consuming because it requires multiple washing steps to remove non-specifically bound reactants [40,45]. The ELISA test is used to detect the RV VP6 antigen—part of the inner capsid—in stool samples, which can confirm RV infection. It is a method routinely used for certain groups, e.g., Group C RV in the United Kingdom [46].
ICA is characterized by a short detection time [43]. Commercially available kits, such as the SD Rota/Adeno Rapid test, are clinically useful for the rapid diagnosis of RV infections. A study conducted in Korea on the aforementioned test suggests that clinicians should be aware of the potential for false-positive results, especially when the ICA band intensity is weak [47]. Studies in Gabon on another rapid diagnostic test (RDT), the SD BIOLINE Rota/Adeno Ag RDT, have shown that it has lower diagnostic accuracy compared to RT-qPCR. Additionally, it was found that while this test is a good method for diagnosing infectious intestinal disease, it is not suitable for screening asymptomatic populations for the purpose of preventing RVA transmission [40].
Polyacrylamide Gel Electrophoresis (PAGE) is a well-established, ultrasensitive method for detecting RV infections, particularly in resource-limited settings [48,49]. This technique involves the separation of dsRNA segments of the virus by size, followed by visualization through silver staining or other staining methods [50]. Fecal samples from suspected cases are processed for RNA extraction, after which the RNA is subjected to PAGE and visualized using silver staining or equivalent techniques [45,46]. Silver staining enhances the detection of dsRNA bands, enabling identification of low viral loads with high sensitivity and specificity [51,52]. A study conducted in Thailand reported that PAGE detected RV in 96.7% of 1304 stool samples, demonstrating performance comparable to that of ELISA [52]. Additionally, PAGE facilitates the differentiation of RV strains by electropherotype, thereby contributing to insights into genetic diversity [53]. Compared to other molecular diagnostic methods, PAGE is relatively inexpensive and requires minimal specialized equipment, making it particularly useful for surveillance and outbreak investigations in low-resource settings [52,53].
A meta-analysis of 12 studies [54], involving a total of 4407 children with RV-induced AGE, indicates the high sensitivity and specificity of ICA for RV detection, albeit with lower sensitivity than RT-PCR. The reduced specificity in detecting RV can be compensated for by serial testing [1,54]. Subgroup analysis of children aged ≤5 years showed slightly lower sensitivity compared to the overall combined sensitivity, which can also be mitigated by serial testing [54].
RT-PCR is among the most sensitive and specific tests (90–95%) and allows for the detection of small quantities of the viral genome [55]. Although it requires special equipment, specialized technicians, and is time-consuming, it is widely used in research studies [56].
2.3. Radiological Diagnosis
Several alternative methods for assessing hydration status have also been explored, although these typically require specialized equipment. These include ultrasound-based measurements of the inferior vena cava (IVC) diameter, inferior-vena-cava-to-aorta-diameter ratio (IVC/Ao), aorta-to-IVC ratio, and IVC inspiratory collapse [57].
The initial research to evaluate pediatric dehydration with ultrasonographic assessment was conducted by Kosiak et al., who also initiated the trend of utilizing the IVC/Ao ratio as a routine diagnostic indicator [58]. Octavius et al. conducted a meta-analysis of five studies in total involving 461 children, evaluating the diagnostic accuracy of the IVC/Ao ratio for clinically significant dehydration. The overall sensitivity of these studies was 86% (95% confidence interval [CI]: 79–91%), and the specificity was 73% (95% CI: 59–84%), with a comparatively high diagnostic performance of the tested method [59]. In a prospective observational study, El Amrousy et al. validated the accuracy of IVC/Ao ratio for the detection of significant dehydration in 200 infants. By the gold standard of percent weight change, only 134 infants were indeed significantly dehydrated. Using a prehydration IVC/Ao ratio cut-off point of <0.75, the method had a sensitivity of 82%, specificity of 91%, and overall accuracy of 87% [60]. This difference underscores the need for more high-quality multicenter trials to establish ultrasonographic criteria in a standardized fashion and validate their applicability across diverse clinical settings. Most importantly, the research of El Amrousy et al. illustrates poor performance of traditional clinical judgment (sensitivity, 70%; specificity, 63%), which was significantly less accurate in diagnosis than objective ultrasound evaluation. Such observations justify the possible usefulness of incorporating sonographic assessment into routine assessment procedures for pediatric dehydration, especially in settings where quick, non-invasive, and accurate decision-making is crucial. Radiological evaluation is necessary if neurological symptoms such as seizures or encephalopathy occur. Magnetic resonance imaging (MRI) can reveal abnormalities, such as hyperintense lesions resulting from edema, when present [9,61].
3. Innovative Diagnosis Methods for RV Infections
Recently, several new diagnostic methods have been developed, which may allow for the detection of RV infection with less sample material and with greater sensitivity.
3.1. Enzyme Immunoassay (EIA)
The EIA is a rapid method developed for the detection of RV antigens in fecal specimens. It is designed to provide a quick and reliable diagnosis of RV AGE, particularly in clinical settings where timely results are crucial [62,63]. The assay delivers results in approximately 10 min, offering a significant advantage over traditional methods that may take several hours. In a study involving 137 patients, the new EIA exhibited 89.2% sensitivity and 90.0% specificity before resolution. After applying a blocking reagent, sensitivity increased to 100%, and specificity remained high at 98.9% [63]. When compared to the Pathfinder RV assay and EM, the new EIA demonstrated competitive performance. In a study with 100 fresh stool samples, the sensitivity and specificity were 95% and 90%, respectively, for new EIA, compared to 84% and 98% for Pathfinder, and 63% and 100% for EM [64]. Although TESTPACK established the utility of rapid immunoassays for rotavirus diagnosis in clinical and outbreak settings, most published evaluations are dated; recent performance assessments of contemporary rapid rotavirus antigen tests emphasize molecular confirmation (e.g., RT-PCR) as the reference standard [64,65].
Recent advances in EIA for RV detection aim to increase sensitivity, particularly in specimens with low viral shedding, such as those from vaccinated infants. In a study by Wang et al., researchers developed a new biotin–avidin sandwich EIA using a monoclonal antibody (1D4) targeting the VP6 antigen of RVAs [65]. The study evaluated 128 fecal samples from infants who had received the Rotarix vaccine in Mexico, comparing the new assay with two commercial EIA kits, using RT-PCR as the reference standard. The new assay detected the RV antigen in 36.7% of samples, nearly double the detection rate of the commercial kits—which detected 16.4% and 18.0%—without cross-reactivity to other enteric viruses. This demonstrates improved sensitivity at lower antigen levels, while retaining specificity, making it a promising tool for diagnostics in vaccine trials and surveillance, where existing EIAs may miss low-level shedding [65].
Recombinant-antigen- and peptide-based EIAs that use anti-VP6 capture and multi-epitope detector antibodies have similarly shown high diagnostic sensitivity and specificity across multiple host species, supporting VP6 as a robust target for RVAs antigen detection [66]. More recently developed assays have expanded beyond manual microplate formats to include VP7-specific potency EIAs for vaccine-related work and automated fluorescent immunoassays (e.g., AFIAS) that offer a rapid turnaround, strong concordance with ELISA/PCR, and workflow advantages for high-throughput laboratories [67,68]. Independent evaluations of contemporary rapid fecal antigen kits report variable sensitivity—often lower than molecular methods—but generally high specificity. This underscores that, while next-generation EIAs narrow the gap with molecular tests for many surveillance applications, RT-PCR remains the reference standard when maximal analytical sensitivity or genotyping is required [67,68].
3.2. Digital PCR (dPCR)
Digital PCR (dPCR) is an advanced molecular technique that offers high sensitivity and precision in detecting low-abundance nucleic acid targets. In the context of RV infections, dPCR has been explored as a diagnostic tool, particularly for environmental surveillance and wastewater-based epidemiology [69]. dPCR has been utilized to monitor RV presence in untreated sewage, providing insights into community-level viral loads and trends over time. This approach aids in assessing the prevalence of RV infections in populations, including asymptomatic individuals [70,71].
dPCR enables the detection of rare mutations and alleles within the RV genome, such as single nucleotide variants (SNVs). This capability is crucial for understanding genetic diversity and evolution of the virus, which can inform vaccine development and epidemiological studies [69]. dPCR can detect low-abundance targets, making it suitable for identifying infections in asymptomatic individuals or in environmental samples with low viral loads. The partitioning of samples in dPCR allows for accurate quantification of viral genomes, providing detailed insights into viral loads. dPCR can simultaneously detect multiple pathogens, facilitating comprehensive diagnostic panels [69,70].
dPCR represents a promising advancement in the molecular diagnosis of RV infections, offering enhanced sensitivity and quantitative capabilities. While its application in clinical diagnostics is still emerging, dPCR holds significant potential for improving detection accuracy and understanding the epidemiology of RV infections [70,71].
3.3. Sequencing Methods
Sequencing technologies have become integral to the diagnosis and molecular characterization of RV infections, offering insights into viral evolution, reassortment, and transmission dynamics. Sanger sequencing—historically considered the gold standard in viral genomics—is widely used to genotype RV strains based on partial sequencing of the VP7 (G type) and VP4 (P type) genes. This method provides high accuracy and reliable results for single-strain infections, with read lengths typically ranging from 500 to 800 base pairs (bp). However, its limitations include low throughput, limited ability to resolve mixed infections, and restricted genomic coverage, making it less suitable for large-scale epidemiological studies or detection of reassortant strains [72,73].
Next-Generation Sequencing (NGS) technologies have significantly advanced RV diagnostics by enabling comprehensive, high-throughput analysis of the viral genome. The Illumina MiSeq platform is one of the most commonly used short-read NGS systems in RV research. It allows for deep sequencing of all 11 genomic segments directly from clinical fecal specimens, often following reverse transcription and cDNA synthesis. Studies have demonstrated that MiSeq can produce near-complete RV genomes with >90% coverage in samples with a sufficient viral load (cycle threshold ≤25), enabling the detection of mixed-genotype infections, point mutations, and reassortment events with high sensitivity. The multiplexing capability of MiSeq facilitates the simultaneous processing of multiple samples, making it ideal for outbreak investigations and longitudinal surveillance [73,74].
Third-generation sequencing platforms, such as Oxford Nanopore Technologies’ MinION, have emerged as powerful tools for rapid, real-time genomic analysis of RVs. Unlike Illumina sequencing, MinION provides long-read capabilities and does not require prior amplification or complex library preparation, making it particularly advantageous in field and low-resource settings. The nanopore sequencing approach yielded near-complete genome coverage and enabled accurate genotyping within a matter of hours [75]. Additionally, large-scale metagenomic studies using nanopore sequencing have demonstrated its capacity to recover full-length RV genomes from over 90% of positive samples, with segment-level identity comparable to that of Sanger sequencing [76]. Nanopore-based methods have been extended to veterinary surveillance, allowing for the identification and complete genomic characterization of RV groups A, B, C, and H in pigs, underscoring the versatility of this platform in One Health applications [77]. Although nanopore sequencing currently has a higher per-read error rate compared to Illumina, ongoing improvements in basecalling algorithms and error correction methods are rapidly closing this gap.
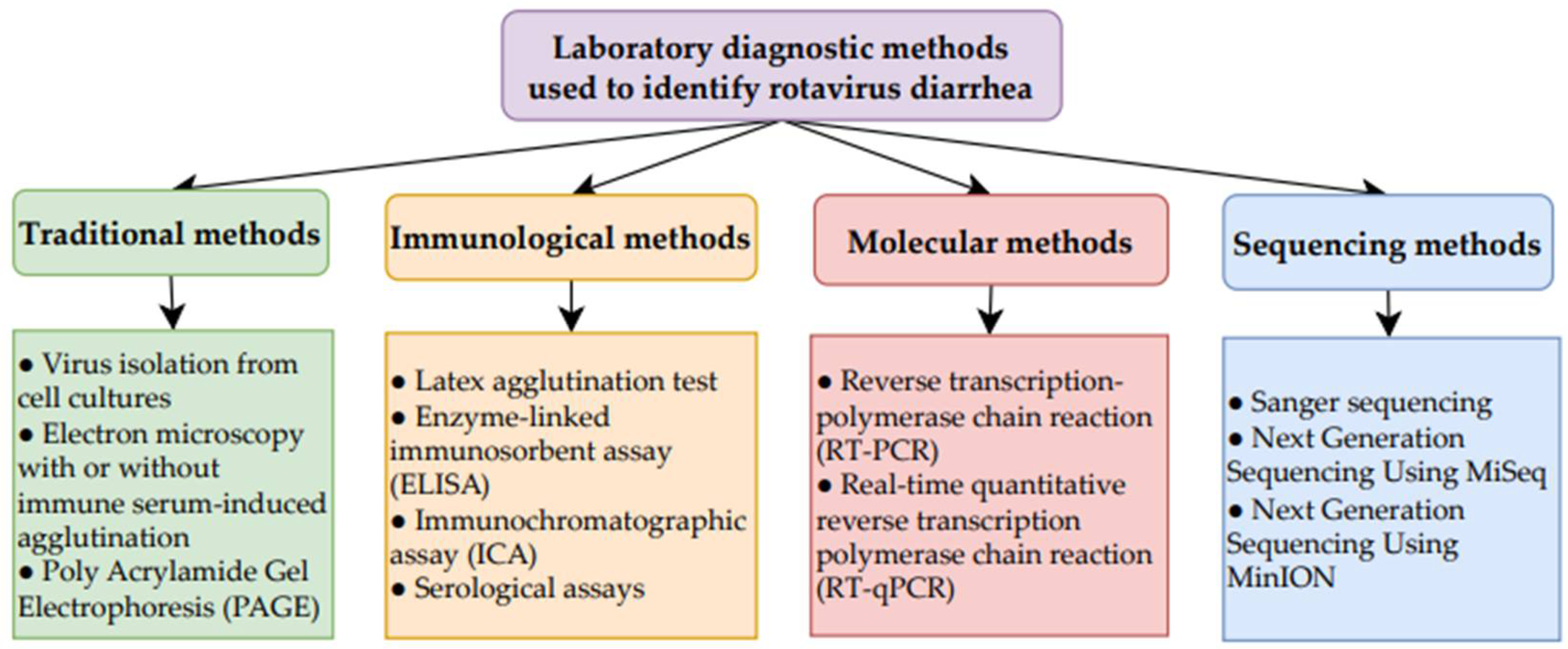
Table 5 presents the types of diagnostic test, the patient samples required, and the type of material detected for RV diagnosis [78,79,80]. Figure 4 provides a summary of the diagnostics tests used [78,79,80].

Table 5.
Types of diagnostic tests, including the type of specimen collected from the patient and the name of the detected RV particle [78,79,80].
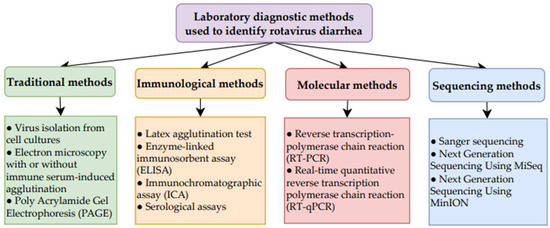
Figure 4.
Summary of laboratory diagnostic methods used to identify RV diarrhea [78,79,80].
5. Materials and Methods
This work was designed as a narrative review, drawing on a comprehensive analysis of research articles retrieved from major biomedical and multidisciplinary databases. Section 1, Section 2, Section 3 and Section 6 provide an in-depth analysis of studies available on PubMed, Google Scholar, Web of Science, Embase, and Scopus, identified using search terms related to: “rotavirus diarrhea”, “clinical diagnostic methods”, “innovative diagnostic methods”, “treatment of rotaviral diarrhea”, “prevention of viral infections”. While the primary focus was on the literature published within the past decade, the review spans a timeline from 1984 to 2025 to ensure a comprehensive, thorough perspective.
Section 4 presents a concise summary of the 2014 ESPGHAN/ESPID recommendations regarding the pharmacological management of acute diarrhea in children. This is followed by a critical appraisal of selected original research articles published between 2014 and 2025 (as shown in Table 6 and detailed in Section 4.2.1, Section 4.2.2, Section 4.2.3, Section 4.2.4 and Section 4.2.5). The selection of studies was based on a comprehensive and systematic literature search performed in PubMed and Scopus, targeting original research focused exclusively on RV-associated AGE in the pediatric population. Review articles were deliberately excluded to ensure analytical emphasis on primary clinical data. Date of searching: 31 July 2025.
Figure 1, Figure 2, Figure 3, Figure 4 and Figure 5 were created using GoodNotes 6 for iPad (v.6.3.55)—all based on the authors’ interpretation of synthesized data from multiple studies. Figure 4 was created using draw.io v27.0.5 (JGraph Ltd., Northampton, UK; draw.io AG, Zürich, Switzerland; operational headquarters: Wiesbaden, Germany).
6. Conclusions
RV is the leading cause of severe AGE in children under five, causing significant morbidity. While vaccination programs have reduced infection rates in high-income countries, RV remains a major health threat in low- and middle-income regions. Symptoms include diarrhea, vomiting, fever, and dehydration, with rare neurological complications.
Diagnosis primarily relies on stool testing using antigen detection methods like ELISA and ICA, and the more sensitive RT-PCR, which also enables viral genotyping. Although RT-PCR is the diagnostic gold standard, its cost and complexity limit its use in many settings. Antigen tests are favored for their short turnout time and affordability but can produce false positives, especially in recently vaccinated children. Accurate diagnosis is essential for clinical care, surveillance, and evaluating vaccine effectiveness. Recent advances in RV diagnosis include several highly sensitive and efficient methods. PAGE separates viral RNA segments and combined with silver staining, detects RV with high sensitivity and specificity. EIA offers rapid detection of RV antigens with improved sensitivity and specificity, providing results within minutes and performing well across different patient groups. dPCR is an emerging molecular technique that enables precise detection and quantification of low viral loads, useful for environmental surveillance and detection of viral mutations. Together, these innovations enhance RV detection accuracy and epidemiological monitoring.
We presented a comprehensive summary of current diagnostic and therapeutic approaches to RV AGE, with particular relevance to clinical practice. We outlined supportive and pharmacological treatments discussed in the recent literature and ESPGHAN guidelines, including hydration therapy, antiemetic drugs (ondansetron, dexamethasone, dimenhydrinate, granisetron, metoclopramide), antimotility and antiperistaltic agents (loperamide), adsorbents (diosmectite, diosmectite plus Lactobacillus rhamnosus GG, kaolin–pectin, attapulgite-activated charcoal), antisecretory agents (racecadotril, bismuth subsalicylate), zinc, probiotics, synbiotics, prebiotics, micronutrients (e.g., folic acid), gelatin tannate, and oral immunoglobulin therapy. Moreover, this review examines a broad spectrum of experimental antiviral agents with activity against distinct phases of the RV life cycle. These include natural bioactive compounds, immunomodulatory molecules, and targeted pharmacological inhibitors validated in preclinical settings. Among the most notable are Genipin, Cyclosporine, Cordycepin, Methyl-β-cyclodextrin, Genistein, Drebrin, ML241, Brequinar, Leflunomide, POL-P (Portulaca oleracea L.), Metformin hydrochloride, 6-Thioguanine (6-TG), MiR-525-3p, miRNA-7, Nitazoxanide, Ursolic acid, Molnupiravir, Deoxyshikonin, lipid metabolism regulators (e.g., TOFA, triacsin C, C75, A922500, betulinic acid, CI-976, PHB, isoproterenol/IBMX), proteasome inhibitors (MG132, bortezomib, and lactacystin), 18β-Glycyrrhetinic acid (18βGRA), RA839, and autophagy-inducing agents such as Rapamycin, LY294002, and BEZ235. Our work aims to support clinicians by providing an updated overview of diagnostic tools and therapeutic strategies, thereby contributing to improved patient care and evidence-based decision-making in the management of RV infections.
Author Contributions
Conceptualization, K.P. and A.F.; methodology, N.K., K.P., E.K., and T.B.; software, M.K.; validation, M.K., E.K., and N.K.; formal analysis, K.P.; investigation, K.P. and E.K.; resources, N.K.; data curation, M.K. and E.P.; writing—original draft preparation, K.P., N.K., E.P., A.F., and T.B.; writing—review and editing, E.P. and E.K.; visualization, E.K., K.P., and A.F.; supervision, E.P. and E.K.; project administration, E.P., K.P., and N.K. All authors have read and agreed to the published version of the manuscript.
Funding
This study did not receive any external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Crawford, S.E.; Ramani, S.; Tate, J.E.; Parashar, U.D.; Svensson, L.; Hagbom, M.; Franco, M.A.; Greenberg, H.B.; O’Ryan, M.; Kang, G.; et al. Rotavirus infection. Nat. Rev. Dis. Primers 2017, 3, 17083. [Google Scholar] [CrossRef]
- Maldonado, Y.A.; Yolken, R.H. Rotavirus. Baillieres Clin. Gastroenterol. 1990, 4, 609–625. [Google Scholar] [CrossRef]
- Parashar, U.D.; Nelson, E.A.; Kang, G. Diagnosis, management, and prevention of rotavirus gastroenteritis in children. BMJ 2013, 347, f7204. [Google Scholar] [CrossRef]
- Kim, J.S.; Lee, S.K.; Ko, D.H.; Hyun, J.; Kim, H.S. Performance evaluation of the automated fluorescent immunoassay system rotavirus assay in clinical samples. Ann. Lab. Med. 2019, 39, 50–57. [Google Scholar] [CrossRef]
- ICTV. Current ICTV Taxonomy Release: Rotavirus. Available online: https://ictv.global/taxonomy (accessed on 11 September 2025).
- Staat, M.A.; Azimi, P.H.; Berke, T.; Roberts, N.; Bernstein, D.I.; Ward, R.L.; Pickering, L.K.; Matson, D.O. Clinical presentations of rotavirus infection among hospitalized children. Pediatr. Infect. Dis. J. 2002, 21, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Le, X.; Tao, Y.; Wang, B.; Hou, Y.; Ning, Y.; Hou, J.; Wang, R.; Li, Q.; Xia, X. Diversity and potential cross-species transmission of rotavirus A in wild animals in Yunnan, China. Microorganisms 2025, 13, 145. [Google Scholar] [CrossRef]
- Pawłuszkiewicz, K.; Ryglowski, P.J.; Idzik, N.; Błaszczyszyn, K.; Kucharczyk, E.; Gaweł-Dąbrowska, D.; Siczek, M.; Widelski, J.; Paluch, E. Rotavirus infections: Pathophysiology, symptoms, and vaccination. Pathogens 2025, 14, 480. [Google Scholar] [CrossRef]
- Lee, K.Y. Rotavirus infection–associated central nervous system complications: Clinicoradiological features and potential mechanisms. Clin. Exp. Pediatr. 2022, 65, 484–491. [Google Scholar]
- Medycyna Praktyczna. Diarrhea. Available online: https://www.mp.pl/interna/chapter/B16.I.1.2 (accessed on 18 June 2025).
- LeClair, C.E.; McConnell, K.A. Rotavirus. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023; Volume 1. [Google Scholar]
- Xu, X.; Luo, Y.; He, C.; Dian, Z.; Mi, H.; Yang, J.; Feng, Y.; Miao, Z.; Xia, X. Increased Risk of Neurological Disease Following Pediatric Rotavirus Infection: A Two-Center Case-Control Study. J. Infect. Dis. 2023, 227, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Ramig, R.F. Pathogenesis of intestinal and systemic rotavirus infection. J. Virol. 2004, 78, 10213–10220. [Google Scholar] [CrossRef] [PubMed]
- Aghsaeifard, Z.; Heidari, G.; Alizadeh, R. Understanding the use of oral rehydration therapy: A narrative review from clinical practice to main recommendations. Health Sci. Rep. 2022, 5, e827. [Google Scholar] [CrossRef]
- Barr, W.; Smith, A. Acute diarrhea. Am. Fam. Physician 2014, 89, 180–189. [Google Scholar]
- Hahn, S.; Kim, Y.; Garner, P. Reduced osmolarity oral rehydration solution for treating dehydration due to diarrhoea in children: Systematic review. BMJ 2001, 323, 81–85. [Google Scholar] [CrossRef]
- Ruuska, T.; Vesikari, T. RV disease in Finnish children: Use of numerical scores for clinical severity of diarrhoeal episodes. Scand. J. Infect. Dis. 1990, 22, 259–267. [Google Scholar] [CrossRef]
- Clark, H.F.; Borian, F.E.; Bell, L.M.; Modesto, K.; Gouvea, V.; Plotkin, S.A. Protective effect of WC3 vaccine against RV diarrhea in infants during a predominantly serotype 1 RV season. J. Infect. Dis. 1988, 158, 570–587. [Google Scholar] [CrossRef]
- Friedman, J.N.; Goldman, R.D.; Srivastava, R.; Parkin, P.C. Development of a clinical dehydration scale for use in children between 1 and 36 months of age. J. Pediatr. 2004, 145, 201–207. [Google Scholar] [CrossRef]
- Levine, A.C.; Glavis-Bloom, J.; Modi, P.; Nasrin, S.; Rege, S.; Chu, C.; Schmid, C.H.; Alam, N.H. Empirically derived dehydration scoring and decision tree models for children with diarrhea: Assessment and internal validation in a prospective cohort study in Dhaka, Bangladesh. Glob. Health Sci. Pract. 2015, 3, 405–418. [Google Scholar] [CrossRef] [PubMed]
- St Jean, D.T.; Chilyabanyama, O.N.; Bosomprah, S.; Asombang, M.; Velu, R.M.; Chibuye, M.; Mureithi, F.; Sukwa, N.; Chirwa, M.; Mokha, P.; et al. Development of a diarrhoea severity scoring scale in a passive health facility-based surveillance system. PLoS ONE 2022, 17, e0272981. [Google Scholar] [CrossRef] [PubMed]
- Gorelick, M.; Shaw, K.; Murphy, K. Validity and reliability of clinical signs in the diagnosis of dehydration in children. Pediatrics 1997, 99, e6. [Google Scholar] [CrossRef]
- World Health Organization (WHO). The Treatment of Diarrhoea: A Manual for Physicians and Other Senior Health Workers, 4th ed.; World Health Organization: Geneva, Switzerland, 2005.
- Aslan, A.; Kurugol, Z.; Cetin, H.; Karakaşlilar, S.; Koturoğlu, G. Comparison of Vesikari and Clark scales regarding the definition of severe rotavirus gastroenteritis in children. Infect. Dis. 2015, 47, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Kang, G. Rotavirus genotypes and severity of diarrheal disease. Clin. Infect. Dis. 2006, 43, 315–316. [Google Scholar] [CrossRef][Green Version]
- Jauregui, J.; Nelson, D.; Choo, E.; Stearns, B.; Levine, A.C.; Liebmann, O.; Shah, S.P.; Carlo, W.A. External validation and comparison of three pediatric clinical dehydration scales. PLoS ONE 2014, 9, e95739. [Google Scholar] [CrossRef]
- Falszewska, A.; Szajewska, H.; Dziechciarz, P. Diagnostic accuracy of three clinical dehydration scales: A systematic review. Arch. Dis. Child. 2018, 103, 383–388. [Google Scholar] [CrossRef]
- Isanaka, S.; Guindo, O.; Langendorf, C.; Seck, A.M.; Plikaytis, B.D.; Sayinzoga-Makombe, N.; McNeal, M.M.; Meyer, N.; Adehossi, E.; Djibo, A.; et al. Efficacy of a Low-Cost, Heat-Stable Oral Rotavirus Vaccine in Niger. N. Engl. J. Med. 2017, 376, 1121–1130. [Google Scholar] [CrossRef]
- Schnadower, D.; I Tarr, P.; Casper, T.C.; Gorelick, M.H.; Dean, M.J.; O’cOnnell, K.J.; Mahajan, P.; Chun, T.H.; Bhatt, S.R.; Roskind, C.G.; et al. Randomised controlled trial of Lactobacillus rhamnosus (LGG) versus placebo in children presenting to the emergency department with acute gastroenteritis: The PECARN probiotic study protocol. BMJ Open 2017, 7, e018115. [Google Scholar] [CrossRef]
- Lewis, K. Vesikari Clinical Severity Scoring System Manual. 2011. Available online: https://www.path.org/our-impact/resources/vesikari-clinical-severity-scoring-system-manual/ (accessed on 3 September 2025).
- Wikswo, M.E.; Weinberg, G.A.; Szilagyi, P.G.; Selvarangan, R.; Harrison, C.J.; Klein, E.J.; A Englund, J.; Sahni, L.C.; A Boom, J.; Halasa, N.B.; et al. Evaluation of a Modified Vesikari Severity Score as a Research Tool for Assessing Pediatric Acute Gastroenteritis. J. Pediatr. Infect. Dis. Soc. 2024, 13, 547–550. [Google Scholar] [CrossRef] [PubMed]
- Schnadower, D.; Tarr, P.I.; Gorelick, M.H.; O’COnnell, K.; Roskind, C.G.; Powell, E.C.; Rao, J.; Bhatt, S.; Freedman, S.B. Validation of the modified Vesikari score in children with gastroenteritis in 5 US emergency departments. J. Pediatr. Gastroenterol. Nutr. 2013, 57, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.-L.; Chen, S.-Y.; Tsai, C.-N.; Chao, H.-C.; Lai, M.-W.; Chang, Y.-J.; Chen, C.-L.; Chiu, C.-H. Complicated norovirus infection and assessment of severity by a modified Vesikari disease score system in hospitalized children. BMC Pediatr. 2016, 16, 162. [Google Scholar] [CrossRef] [PubMed]
- Sowmyanarayanan, T.V.; Ramani, S.; Sarkar, R.; Arumugam, R.; Warier, J.P.; Moses, P.D.; Simon, A.; Agarwal, I.; Bose, A.; Arora, R.; et al. Severity of RV Gastroenteritis in Indian Children Requiring Hospitalization. Vaccine 2012, 30, 167–172. [Google Scholar] [CrossRef]
- Armah, G.E.; Sow, S.O.; Breiman, R.F.; Dallas, M.J.; Tapia, M.D.; Feikin, D.R.; Binka, F.N.; Steele, A.D.; Laserson, K.F.; Ansah, N.A.; et al. Efficacy of Pentavalent RV Vaccine against Severe RV Gastroenteritis in Infants in Developing Countries in Sub-Saharan Africa: A Randomised, Double-Blind, Placebo-Controlled Trial. Lancet 2010, 376, 606–614. [Google Scholar] [CrossRef]
- Madhi, S.A.; Cunliffe, N.A.; Steele, D.; Witte, D.; Kirsten, M.; Louw, C.; Ngwira, B.; Victor, J.C.; Gillard, P.H.; Cheuvart, B.B.; et al. Effect of Human RV Vaccine on Severe Diarrhea in African Infants. N. Engl. J. Med. 2010, 362, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Guarino, A.; Ashkenazi, S.; Gendrel, D.; Vecchio, A.L.; Shamir, R.; Szajewska, H. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition/European Society for Pediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe: Update 2014. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 132–152. [Google Scholar] [CrossRef] [PubMed]
- Hoxha, T.; Xhelili, L.; Azemi, M.; Avdiu, M.; IsmailiJaha, V.; EfendijaBeqa, U.; GrajcevciUka, V. Comparing the Accuracy of the Three Dehydration Scales in Children with Acute Diarrhea in a Developing Country of Kosovo. Mater. Sociomed. 2015, 27, 140–143. [Google Scholar] [CrossRef] [PubMed]
- Manouana, G.P.; Nguema-Moure, P.A.; Tomazatos, A.; Maloum, M.N.; Bock, C.-T.; Kremsner, P.G.; Velavan, T.P.; Adegnika, A.A.; Niendorf, S. RT-PCR-based assessment of the SD Bioline Rota/Adeno antigen-based test in infants with and without diarrhea. Virol. J. 2023, 20, 40. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, Y.; Li, Y.; Zhang, S.; Wang, X.; Zong, H.; Huang, W.; Kong, D.; Jiang, Y.; Liu, P.; et al. Development of a rapid homogeneous immunoassay for detection of rotavirus in stool samples. Front. Public Health 2022, 10, 975720. [Google Scholar] [CrossRef]
- Medycyna Praktyczna. Microbiological, Immunological, and Molecular Diagnostics of the Most Common Foodborne Infections in Children. Available online: https://www.mp.pl/pediatria/praktyka-kliniczna/badania-laboratoryjne/180277 (accessed on 16 June 2025).
- World Health Organization. Manual of Rotavirus Detection and Characterization Methods. Available online: https://iris.who.int/bitstream/handle/10665/342905/WER9628-301-319-eng-fre.pdf?sequence=1 (accessed on 16 June 2025).
- The Public Health Laboratory Network. Rotavirus—Laboratory Case Definition. Available online: https://www.health.gov.au/sites/default/files/documents/2022/06/rotavirus-laboratory-case-definition.pdf (accessed on 18 June 2025).
- Hughes, J.H.; Tuomari, A.V.; Mann, D.R.; Hamparian, V.V. Latex immunoassay for rapid detection of rotavirus. J. Clin. Microbiol. 1984, 20, 441–447. [Google Scholar] [CrossRef]
- Long, S.S.; Prober, C.G.; Fischer, M.; Kimberlin, D. Principles and Practice of Pediatric Infectious Diseases, 6th ed.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2022. [Google Scholar]
- James, V.L.A.; Lambden, P.R.; Caul, E.O.; Clarke, I.N. Enzyme-linked immunosorbent assay based on recombinant human group C rotavirus inner capsid protein (VP6) to detect human group C rotaviruses in fecal samples. J. Clin. Microbiol. 1998, 36, 3178–3181. [Google Scholar] [CrossRef]
- Hung, P.J.; Chen, C.C. Diagnostic accuracy of rotavirus antigen tests in children: A systematic review and meta-analysis. Trop. Med. Int. Health 2023, 28, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Merrill, C.R.; Goldman, D.; Sedman, S.A.; Ebert, M.H. Ultrasensitive stain for proteins in polyacrylamide gels shows regional variation in cerebrospinal fluid proteins. Science 1981, 211, 1437–1438. [Google Scholar] [CrossRef]
- Oakley, B.R.; Kirsch, D.R.; Morris, N.R. A simplified ultrasensitive silver stain for detecting proteins in polyacrylamide gels. Anal. Biochem. 1980, 105, 361–363. [Google Scholar] [CrossRef]
- Dash, S.K.; Kumar, K.; Tewari, A.; Varshney, P.; Goel, A.; Bhatia, A.K. Detection of rotavirus from hospitalized diarrheic children in Uttar Pradesh, India. Indian J. Microbiol. 2012, 52, 472–477. [Google Scholar] [CrossRef][Green Version]
- Hammami, S.; Castro, A.E.; Osburn, B.I. Comparison of polyacrylamide gel electrophoresis, an enzyme-linked immunosorbent assay, and an agglutination test for the direct identification of bovine rotavirus from feces and coelectrophoresis of viral RNAs. J. Vet. Diagn. Investig. 1990, 2, 184–190. [Google Scholar] [CrossRef]
- Kasempimolporn, S.; Louisirirotchanakul, S.; Sinarachatanant, P.; Wasi, C. Polyacrylamide gel electrophoresis and silver staining for detection of rotavirus in stools from diarrheic patients in Thailand. J. Clin. Microbiol. 1988, 26, 158–160. [Google Scholar] [CrossRef] [PubMed]
- Kusumakar, A.L.; Savita; Malik, Y.; Minakshi; Prasad, G. Detection of human rotavirus in hospitalized diarrheic children in central India. Indian J. Microbiol. 2007, 47, 373–376. [Google Scholar] [CrossRef][Green Version]
- Chen, C.C.; Hsiao, K.Y.; Bai, C.H.; Wang, Y.H. Investigation of the diagnostic performance of the SARS-CoV-2 saliva antigen test: A meta-analysis. J. Microbiol. Immunol. Infect. 2022, 55, 84–93. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Clinical Overview of Rotavirus. Rotavirus. Available online: https://www.cdc.gov/rotavirus/hcp/clinical-overview/?CDC_AAref_Val=https://www.cdc.gov/rotavirus/clinical.html (accessed on 16 June 2025).
- Carossino, M.; Barrandeguy, M.E.; Erol, E.; Li, Y.; Balasuriya, U.B.R. Development and evaluation of a one-step multiplex real-time TaqMan® RT-qPCR assay for the detection and genotyping of equine G3 and G14 rotaviruses in fecal samples. Virol. J. 2019, 16, 49. [Google Scholar] [CrossRef]
- Steyer, A.; Mičetić-Turk, D.; Fijan, S. The efficacy of probiotics as antiviral agents for the treatment of rotavirus gastrointestinal infections in children: An updated overview of literature. Microorganisms 2022, 10, 2392. [Google Scholar] [CrossRef]
- Kosiak, W.; Swieton, D.; Piskunowicz, M. Sonographic inferior vena cava/aorta diameter index, a new approach to the body fluid status assessment in children and young adults in emergency ultrasound—Preliminary study. Am. J. Emerg. Med. 2008, 26, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Octavius, G.S.; Imanuelly, M.; Wibowo, J.; Heryadi, N.K.; Widjaja, M. Inferior vena cava to aorta ratio in dehydrated pediatric patients: A systematic review and meta-analysis. Clin. Exp. Pediatr. 2023, 66, 477–484. [Google Scholar] [CrossRef]
- El Amrousy, D.; Gamal, R.; Elrifaey, S.; Hassan, S. Non-invasive Assessment of Significant Dehydration in Infants Using the Inferior Vena Cava to Aortic Ratio: Is it Useful? J. Pediatr. Gastroenterol. Nutr. 2018, 66, 882–886. [Google Scholar] [CrossRef]
- Paketçi, C.; Edem, P.; Okur, D.; Sarıoğlu, F.C.; Güleryüz, H.; Bayram, E.; Kurul, S.H.; Yiş, U. Rotavirus encephalopathy with concomitant acute cerebellitis: Report of a case and review of the literature. Turk. J. Pediatr. 2020, 62, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Dennehy, P.H.; Gauntlett, D.R.; Spangenberger, S.E. Choice of reference assay for the detection of rotavirus in fecal specimens: Electron microscopy versus enzyme immunoassay. J. Clin. Microbiol. 1990, 28, 1280–1283. [Google Scholar] [CrossRef] [PubMed]
- Marchlewicz, B.; Spiewak, M.; Lampinen, J. Evaluation of Abbott TESTPACK ROTAVIRUS with clinical specimens. J. Clin. Microbiol. 1988, 26, 2456–2458. [Google Scholar] [CrossRef]
- Brooks, R.G.; Brown, L.; Franklin, R.B. Comparison of a new rapid test (TestPack Rotavirus) with standard enzyme immunoassay and electron microscopy for the detection of rotavirus in symptomatic hospitalized children. J. Clin. Microbiol. 1989, 27, 775–777. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, M.; Sugata, K.; Wang, Y.; Hull, J.; Foytich, K.; Jiang, B. Development of a new enzyme immunoassay for improved detection of rotavirus in fecal specimens of vaccinated infants. J. Clin. Virol. 2018, 99–100, 44–49. [Google Scholar] [CrossRef]
- Kumar, N.; Malik, Y.S.; Kumar, S.; Sharma, K.; Sircar, S.; Saurabh, S.; Gulati, B.R.; Singh, N.; Singh, A.K.; Joshi, V.G.; et al. Peptide-Recombinant VP6 Protein Based Enzyme Immunoassay for the Detection of Group A Rotaviruses in Multiple Host Species. PLoS ONE 2016, 11, e0159027. [Google Scholar] [CrossRef]
- Moon, S.-S.; Wang, H.; Brown, K.; Wang, Y.; Bessy, T.; Greenberg, H.B.; Jiang, B. Development and validation of a VP7-specific EIA for determining the potency and stability of inactivated rotavirus vaccine. J. Virol. Methods 2025, 332. [Google Scholar] [CrossRef]
- Malik, Y.S.; Verma, A.K.; Kumar, N.; Touil, N.; Karthik, K.; Tiwari, R.; Bora, D.P.; Dhama, K.; Ghosh, S.; Hemida, M.G.; et al. Advances in diagnostic approaches for viral etiologies of diarrhea: From the lab to the field. Front. Microbiol. 2019, 10, 1957. [Google Scholar] [CrossRef]
- Mirabile, A.; Sangiorgio, G.; Bonacci, P.G.; Bivona, D.; Nicitra, E.; Bonomo, C.; Bongiorno, D.; Stefani, S.; Musso, N. Advancing pathogen identification: The role of digital PCR in enhancing diagnostic power in different settings. Diagnostics 2024, 14, 1598. [Google Scholar] [CrossRef]
- Kiulia, N.M.; Gonzalez, R.; Thompson, H.; Aw, T.G.; Rose, J.B. Quantification and trends of rotavirus and enterovirus in untreated sewage using reverse transcription droplet digital PCR. Food Environ. Virol. 2021, 13, 154–169. [Google Scholar] [CrossRef]
- Huge, B.J.; North, D.; Mousseau, C.B.; Bibby, K.; Dovichi, N.J.; Champion, M.M. Comparison of RT-dPCR and RT-qPCR and the effects of freeze-thaw cycle and glycine release buffer for wastewater SARS-CoV-2 analysis. Sci. Total Environ. 2022, 12, 20641. [Google Scholar] [CrossRef]
- Matthijnssens, J.; Ciarlet, M.; Heiman, E.; Arijs, I.; Delbeke, T.; McDonald, S.M.; Palombo, E.A.; Iturriza-GómAra, M.; Maes, P.; Patton, J.T.; et al. Full genome-based classification of rotaviruses reveals a common origin between human Wa-like and porcine rotavirus strains and human DS-1-like and bovine rotavirus strains. J. Virol. 2008, 82, 3204–3219. [Google Scholar] [CrossRef]
- Minami-Fukuda, F.; Nagai, M.; Takai, H.; Murakami, T.; Ozawa, T.; Tsuchiaka, S.; Okazaki, S.; Katayama, Y.; Oba, M.; Nishiura, N.; et al. Detection of bovine group A rotavirus using rapid antigen detection kits, RT-PCR and next-generation DNA sequencing. J. Vet. Med. Sci. 2013, 75, 1651–1655. [Google Scholar] [CrossRef][Green Version]
- Dung, T.T.N.; Duy, P.T.; Sessions, O.M.; Sangumathi, U.K.; Phat, V.V.; Tam, P.T.T.; To, N.T.N.; Phuc, T.M.; Chau, T.T.H.; Chau, N.N.M.; et al. A universal genome sequencing method for rotavirus A from human fecal samples which identifies segment reassortment and multi-genotype mixed infection. BMC Genom. 2017, 18, 324. [Google Scholar] [CrossRef] [PubMed]
- Kuba, Y.; Takemae, N.; Kawato, S.; Oba, K.; Taniguchi, K.; Kageyama, T. Evaluation of rapid amplicon-based nanopore sequencing using the latest chemistry for accurate whole genome analysis of influenza A virus in clinical samples. Jpn. J. Infect. Dis. 2025. [Google Scholar] [CrossRef] [PubMed]
- Yandle, Z.; Gonzalez, G.; Carr, M.; Matthijnssens, J.; De Gascun, C. A viral metagenomic protocol for nanopore sequencing of group A rotavirus. J. Virol. Methods 2023, 312, 114664. [Google Scholar] [CrossRef] [PubMed]
- Krasnikov, N.; Gulyukin, A.; Aliper, T.; Yuzhakov, A. Complete genome characterization by nanopore sequencing of rotaviruses A, B, and C circulating on large-scale pig farms in Russia. Virol. J. 2024, 21, 289. [Google Scholar] [CrossRef]
- Dunkin, M.A. Blood in Stool. WebMD. Available online: https://www.webmd.com/digestive-disorders/blood-in-stool (accessed on 18 June 2025).
- Spandorfer, P.R. Oral versus intravenous rehydration of moderately dehydrated children: A randomized, controlled trial. Pediatrics 2005, 115, 295–301. [Google Scholar] [CrossRef]
- Fonseca, B.K.; Holdgate, A.; Craig, J.C. Enteral vs intravenous rehydration therapy for children with gastroenteritis. Arch. Pediatr. Adolesc. Med. 2004, 158, 483–490. [Google Scholar] [CrossRef]
- Civra, A.; Francese, R.; Donalisio, M.; Tonetto, P.; Coscia, A.; Sottemano, S.; Balestrini, R.; Faccio, A.; Cavallarin, L.; Moro, G.E.; et al. Human Colostrum and Derived Extracellular Vesicles Prevent Infection by Human Rotavirus and Respiratory Syncytial Virus in Vitro. J. Hum. Lact. 2021, 37, 122–134. [Google Scholar] [CrossRef]
- Campo, J.J.; Seppo, A.E.; Randall, A.Z.; Pablo, J.; Hung, C.; Teng, A.; Shandling, A.D.; Truong, J.; Oberai, A.; Miller, J.; et al. Human milk antibodies to global pathogens reveal geographic and interindividual variations in IgA and IgG. J. Clin. Investig. 2024, 134, e168789. [Google Scholar] [CrossRef] [PubMed]
- Superti, F.; Ammendolia, M.G.; Valenti, P.; Seganti, L. Antirotaviral activity of milk proteins: Lactoferrin prevents rotavirus infection in the enterocyte-like cell line HT-29. Med. Microbiol. Immunol. 1997, 186, 83–91. [Google Scholar] [CrossRef]
- Valenti, P.; Antonini, G. Lactoferrin: An important host defence against microbial and viral attack. Cell Mol. Life Sci. 2005, 62, 2576–2587. [Google Scholar] [CrossRef]
- Jeong, Y.Y.; Lee, G.Y.; Yoo, Y.C. Bovine lactoferricin induces intestinal epithelial cell activation through phosphorylation of FAK and paxillin and prevents rotavirus infection. J. Microbiol. Biotechnol. 2021, 31, 1175–1182. [Google Scholar] [CrossRef]
- Owens, J.R.; Broadhead, R.; Hendrickse, R.G.; Jaswal, O.P.; Gangal, R.N. Loperamide in the 13ktreatment of acute gastroenteritis in early childhood: Report of a two-centre, double-blind, controlled clinical trial. Ann. Trop. Paediatr. 1981, 1, 135–141. [Google Scholar] [CrossRef]
- Hagbom, M.; Novak, D.; Ekström, M.; Khalid, Y.; Andersson, M.; Lindh, M.; Nordgren, J.; Svensson, L.; Mansur, A. Ondansetron treatment reduces rotavirus symptoms—A randomized double-blinded placebo-controlled trial. PLoS ONE 2017, 12, e0186824. [Google Scholar] [CrossRef]
- Kang, G.; Thuppal, S.V.; Srinivasan, R.; Sarkar, R.; Subashini, B.; Venugopal, S.; Sindhu, K.; Anbu, D.; Parez, N.; Svensson, L.; et al. Racecadotril in the management of rotavirus and non-rotavirus diarrhea in under-five children: Two randomized, double-blind, placebo-controlled trials. Indian Pediatr. 2016, 53, 595–600. [Google Scholar] [CrossRef]
- Dewi, M.R.; Soenarto, Y.; Karyana, I.P.G. Efficacy of synbiotic treatment in children with acute rotavirus diarrhea. Paediatr. Indones. 2015, 55, 74–78. [Google Scholar] [CrossRef][Green Version]
- Jiang, L.; Tang, A.; Song, L.; Tong, Y.; Fan, H. Advances in the development of antivirals for rotavirus infection. Front. Immunol. 2023, 14, 1041149. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.X.; Xu, C.D.; Yang, C.Q. Therapeutic effects of zinc supplement as adjunctive therapy in infants and young children with rotavirus enteritis. Zhongguo Dang Dai Er Ke Za Zhi 2016, 18, 826–830. [Google Scholar]
- Cai, Y.; Wang, X.; Li, C.; Li, F.; Yan, Z.; Ma, N.; Sun, M. Probiotics combined with zinc and selenium preparation in the treatment of child rotavirus enteritis. Am. J. Transl. Res. 2022, 14, 1043–1050. [Google Scholar]
- Xu, N.; Zhang, W.; Huo, J.; Tao, R.; Jin, T.; Zhang, Y.; Wang, Y.; Zhu, L.; Li, J.; Yao, Q.; et al. Characterization of changes in the intestinal microbiome following combination therapy with zinc preparation and conventional treatment for children with rotavirus enteritis. Front. Cell. Infect. Microbiol. 2023, 13, 1153701. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-F.; Liu, P.-Y.; Chen, Y.-Y.; Nong, B.-R.; Huang, I.-F.; Hsieh, K.-S.; Chen, K.-T. Three-combination probiotics therapy in children with salmonella and rotavirus gastroenteritis. J. Clin. Gastroenterol. 2014, 48, 37–42. [Google Scholar] [CrossRef]
- Aggarwal, S.; Upadhyay, A.; Shah, D.; Teotia, N.; Agarwal, A.; Jaiswal, V. Lactobacillus GG for treatment of acute childhood diarrhoea: An open labelled, randomized controlled trial. Indian J. Med. Res. 2014, 139, 379–385. [Google Scholar]
- Sindhu, K.N.C.; Sowmyanarayanan, T.V.; Paul, A.; Babji, S.; Ajjampur, S.S.R.; Priyadarshini, S.; Sarkar, R.; Balasubramanian, K.A.; Wanke, C.A.; Ward, H.D.; et al. Immune response and intestinal permeability in children with acute gastroenteritis treated with Lactobacillus rhamnosus GG: A randomized, double-blind, placebo-controlled trial. Clin. Infect. Dis. 2014, 58, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.K.; Park, J.E.; Kim, M.J.; Seo, J.G.; Lee, J.H.; Ha, N.J. Probiotic bacteria, B. longum and L. acidophilus inhibit infection by rotavirus in vitro and decrease the duration of diarrhea in pediatric patients. Clin. Res. Hepatol. Gastroenterol. 2015, 39, 237–244. [Google Scholar] [CrossRef]
- Sobouti, B.; Noorbakhsh, S.; Ashraf, H.; Ashraf-Talesh, S. Use of probiotic for the treatment of acute rotavirus diarrhea in children: A randomized single-blind controlled trial. Int. J. Child Adolesc. 2016, 2, 5–9. [Google Scholar]
- Das, S.; Gupta, P.K.; Das, R.R. Efficacy and safety of Saccharomyces boulardii in acute rotavirus diarrhea: Double-blind randomized controlled trial from a developing country. J. Trop. Pediatr. 2016, 62, 464–470. [Google Scholar] [CrossRef][Green Version]
- Park, M.S.; Kwon, B.; Ku, S.; Ji, G.E. The Efficacy of Bifidobacterium longum BORI and Lactobacillus acidophilus AD031 Probiotic Treatment in Infants with Rotavirus Infection. Nutrients 2017, 9, 887. [Google Scholar] [CrossRef] [PubMed]
- Hong Chau, T.T.; Minh Chau, N.N.; Hoang Le, N.T.; Chung The, H.; Voong Vinh, P.; Nguyen To, N.T.; Ngoc, N.M.; Tuan, H.M.; Chau Ngoc, T.L.; Kolader, M.-E.; et al. A Double-Blind, Randomized, Placebo-Controlled Trial of Lactobacillus acidophilus for the Treatment of Acute Watery Diarrhea in Vietnamese Children. Pediatr. Infect. Dis. J. 2018, 37, 35–42. [Google Scholar] [CrossRef]
- Shin, D.Y.; Yi, D.Y.; Jo, S.; Lee, Y.M.; Kim, J.-H.; Kim, W.; Park, M.R.; Yoon, S.M.; Kim, Y.; Yang, S.; et al. Effect of a New Lactobacillus plantarum Product, LRCC5310, on Clinical Symptoms and Virus Reduction in Children with Rotaviral Enteritis. Medicine 2020, 99, e22192. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Lan, L.; Chen, N.; Cheng, Y.; Gong, S. The efficacy of lactobacillus reuteri in conjunction with racecadotril in managing pediatric rotavirus enteritis and its impact on intestinal mucosa and immune function. Afr. Health Sci. 2025, 25, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Szymański, H.; Szajewska, H. Lack of Efficacy of Lactobacillus reuteri DSM 17938 for the Treatment of Acute Gastroenteritis: A Randomized Controlled Trial. Pediatr. Infect. Dis. J. 2019, 38, e237–e242. [Google Scholar] [CrossRef]
- Mourey, F.; Sureja, V.; Kheni, D.; Shah, P.; Parikh, D.; Upadhyay, U.; Satia, M.; Shah, D.; Troise, C.; Decherf, A. A Multicenter, Randomized, Double-Blind, Placebo-Controlled Trial of Saccharomyces boulardii in Infants and Children with Acute Diarrhea. Pediatr. Infect. Dis. J. 2020, 39, e347–e351. [Google Scholar] [CrossRef]
- Xie, Y.M.; Wang, L.Y.; Gao, S.; Wang, Z.L. Effect of ingested immunoglobulin on sIgA expression in pediatric rotavirus enteritis. Sichuan Da Xue Xue Bao Yi Xue Ban 2015, 46, 71–74. [Google Scholar]
- Williams, D. Treatment of Rotavirus-Associated Diarrhea Using Enteral Immunoglobulins for Pediatric Stem Cell Transplant Patients. J. Oncol. Pharm. Pract. 2015, 21, 238–240. [Google Scholar] [CrossRef]
- Flerlage, T.; Hayden, R.; Cross, S.J.; Dallas, R.; Srinivasan, A.; Tang, L.; Sun, Y.; Maron, G. Rotavirus Infection in Pediatric Allogeneic Hematopoietic Cell Transplant Recipients: Clinical Course and Experience Using Nitazoxanide and Enterally Administered Immunoglobulins. Pediatr. Infect. Dis. J. 2018, 37, 176–181. [Google Scholar] [CrossRef]
- Banerjee, S.; Sarkar, R.; Mukherjee, A.; Miyoshi, S.I.; Kitahara, K.; Halder, P.; Koley, H.; Chawla-Sarkar, M. Quercetin, a Flavonoid, Combats Rotavirus Infection by Deactivating Rotavirus-Induced Pro-Survival NF-κB Pathway. Front. Microbiol. 2022, 13, 951716. [Google Scholar] [CrossRef]
- Van Dycke, J.; Arnoldi, F.; Papa, G.; Vandepoele, J.; Burrone, O.R.; Mastrangelo, E.; Tarantino, D.; Heylen, E.; Neyts, J.; Rocha-Pereira, J. A Single Nucleoside Viral Polymerase Inhibitor Against Norovirus, Rotavirus, and Sapovirus-Induced Diarrhea. J. Infect. Dis. 2018, 218, 1753–1758. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Ding, S.; Yin, Y.; Xu, L.; Li, P.; Peppelenbosch, M.P.; Pan, Q.; Wang, W. Suppression of Pyrimidine Biosynthesis by Targeting DHODH Enzyme Robustly Inhibits Rotavirus Replication. Antivir. Res. 2019, 167, 35–44. [Google Scholar] [CrossRef]
- Zhou, X.; Li, Y.; Li, T.; Cao, J.; Guan, Z.; Xu, T.; Jia, G.; Ma, G.; Zhao, R. Portulaca oleracea L. Polysaccharide Inhibits Porcine Rotavirus In Vitro. Animals 2023, 13, 2306. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, K.; Kim, W. Genipin Inhibits Rotavirus-Induced Diarrhea by Suppressing Viral Replication and Regulating Inflammatory Responses. Sci. Rep. 2020, 10, 15836. [Google Scholar] [CrossRef]
- Eichwald, C.; De Lorenzo, G.; Schraner, E.M.; Papa, G.; Bollati, M.; Swuec, P.; de Rosa, M.; Milani, M.; Mastrangelo, E.; Ackermann, M.; et al. Identification of a Small Molecule That Compromises the Structural Integrity of Viroplasms and Rotavirus Double-Layered Particles. J. Virol. 2018, 92, e01943-17. [Google Scholar] [CrossRef]
- La Frazia, S.; Ciucci, A.; Arnoldi, F.; Coira, M.; Gianferretti, P.; Angelini, M.; Belardo, G.; Burrone, O.R.; Rossignol, J.-F.; Santoro, M.G. Thiazolides, a New Class of Antiviral Agents Effective Against Rotavirus Infection, Target Viral Morphogenesis, Inhibiting Viroplasm Formation. J. Virol. 2013, 87, 11096–11106. [Google Scholar] [CrossRef]
- Tohmé, M.J.; Giménez, M.; Peralta, A.; Colombo, M.; Delgui, L. Ursolic Acid: A Novel Antiviral Compound Inhibiting Rotavirus Infection In Vitro. Int. J. Antimicrob. Agents 2019, 54, 601–609. [Google Scholar] [CrossRef]
- Santos-Ferreira, N.; Van Dycke, J.; Chiu, W.; Neyts, J.; Matthijnssens, J.; Rocha-Pereira, J. Molnupiravir Inhibits Human Norovirus and Rotavirus Replication in 3D Human Intestinal Enteroids. Antivir. Res. 2024, 223, 105839. [Google Scholar] [CrossRef]
- Shen, Z.; He, H.; Wu, Y.; Li, J. Cyclosporin A Inhibits Rotavirus Replication and Restores Interferon-Beta Signaling Pathway In Vitro and In Vivo. PLoS ONE 2013, 8, e71815. [Google Scholar] [CrossRef]
- Chanda, S.D.; Banerjee, A.; Chawla, S.M. Cordycepin, an Adenosine Analogue, Executes Anti-Rotaviral Effect by Stimulating Induction of Type I Interferon. J. Virol. Antivir. Res. 2015, 4, 2. [Google Scholar] [CrossRef]
- Hardy, M.E.; Hendricks, J.M.; Paulson, J.M.; Faunce, N.R. 18 β-Glycyrrhetinic Acid Inhibits Rotavirus Replication in Culture. Virol. J. 2012, 9, 96. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hu, X.; Wu, J.; Lin, X.; Chen, R.; Lu, C.; Song, X.; Leng, Q.; Li, Y.; Kuang, X.; et al. ML241 Antagonizes ERK 1/2 Activation and Inhibits Rotavirus Proliferation. Viruses 2024, 16, 623. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Liao, D.; He, B.; Pu, R.; Cui, Y.; Zhou, G. Deoxyshikonin inhibited rotavirus replication by regulating autophagy and oxidative stress through SIRT1/FoxO1/Rab7 axis. Microb. Pathog. 2023, 178, 106065. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Feng, C.; Luo, D.; Zhao, R.; Kannan, P.R.; Yin, Y.; Iqbal, M.Z.; Hu, Y.; Kong, X. Metformin Hydrochloride significantly inhibits rotavirus infection in Caco2 cell line, intestinal organoids, and mice. Pharmaceuticals 2023, 16, 1279. [Google Scholar] [CrossRef]
- Cui, J.; Fu, X.; Xie, J.; Gao, M.; Hong, M.; Chen, Y.; Su, S.; Li, S. Critical role of cellular cholesterol in bovine rotavirus infection. Virol. J. 2014, 11, 98. [Google Scholar] [CrossRef]
- Dou, X.; Li, Y.; Han, J.; Zarlenga, D.S.; Zhu, W.; Ren, X.; Dong, N.; Li, X.; Li, G. Cholesterol of lipid rafts is a key determinant for entry and post-entry control of porcine rotavirus infection. BMC Vet. Res. 2018, 14, 45. [Google Scholar] [CrossRef]
- Patra, U.; Mukhopadhyay, U.; Sarkar, R.; Mukherjee, A.; Chawla-Sarkar, M. RA-839, a selective agonist of Nrf2/ARE pathway, exerts potent anti-rotaviral efficacy in vitro. Antivir. Res. 2019, 161, 53–62. [Google Scholar] [CrossRef]
- López, T.; López, S.; Arias, C.F. The tyrosine kinase inhibitor genistein induces the detachment of rotavirus particles from the cell surface. Virus Res. 2015, 210, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Ding, S.; Feng, N.; Mooney, N.; Ooi, Y.S.; Ren, L.; Diep, J.; Kelly, M.R.; Yasukawa, L.L.; Patton, J.T.; et al. Drebrin restricts rotavirus entry by inhibiting dynamin-mediated endocytosis. Proc. Natl. Acad. Sci. USA 2017, 114, E3642–E3651. [Google Scholar] [CrossRef]
- Yin, Y.; Chen, S.; Hakim, M.S.; Wang, W.; Xu, L.; Dang, W.; Qu, C.; Verhaar, A.P.; Su, J.; Fuhler, G.M.; et al. 6-Thioguanine inhibits rotavirus replication through suppression of Rac1 GDP/GTP cycling. Antivir. Res. 2018, 156, 92–101. [Google Scholar] [CrossRef]
- Tian, Z.; Zhang, J.; He, H.; Li, J.; Wu, Y.; Shen, Z. MiR-525-3p mediates antiviral defense to rotavirus infection by targeting nonstructural protein 1. Biochim. Biophys. Acta 2017, 1863, 3212–3225. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, L.; Du, J.; Hu, X.; Xie, Y.; Wu, J.; Lin, X.; Yin, N.; Sun, M.; Li, H. MicroRNA-7 inhibits rotavirus replication by targeting viral NSP5 in vivo and in vitro. Viruses 2020, 12, 209. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Dang, W.; Zhou, X.; Xu, L.; Wang, W.; Cao, W.; Chen, S.; Su, J.; Cai, X.; Xiao, S.; et al. PI3K-Akt-mTOR axis sustains rotavirus infection via the 4E-BP1 mediated autophagy pathway and represents an antiviral target. Virulence 2017, 9, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Crawford, S.E.; Desselberger, U. Lipid droplets form complexes with viroplasms and are crucial for rotavirus replication. Curr. Opin. Virol. 2016, 19, 11–15. [Google Scholar] [CrossRef]
- Contin, R.; Arnoldi, F.; Mano, M.; Burrone, O.R. Rotavirus replication requires a functional proteasome for effective assembly of viroplasms. J. Virol. 2011, 85, 2781–2792. [Google Scholar] [CrossRef]
- López, T.; Silva-Ayala, D.; López, S.; Arias, C.F. Replication of the rotavirus genome requires an active ubiquitin-proteasome system. J. Virol. 2011, 85, 11964–11971. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Wang, Y.; Li, P.; Yin, Y.; Bijvelds, M.J.; de Jonge, H.R.; Peppelenbosch, M.P.; Kainov, D.E.; Pan, Q. Drug screening identifies gemcitabine inhibiting rotavirus through alteration of pyrimidine nucleotide synthesis pathway. Antivir. Res. 2020, 180, 104823. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).