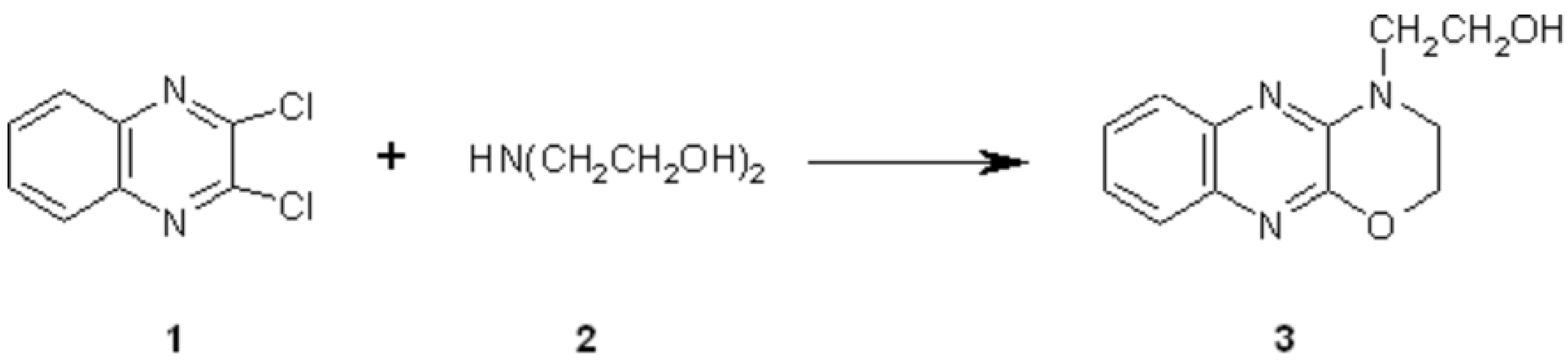
A mixture of 2,3-dichloroquinoxaline 1[1] (5.0 g, 25 mmol) and diethanolamine 2 (12 mL, 125 mmol) was heated to 130° C for 3 h, with magnetic stirring. The reaction mixture was cooled to room temperature and then poured into 300 mL of water. The resulting solid was filtered and then recrystallized from water to give white crystals of 2-(1,4-oxazino[2,3-b]quinoxalin-4-yl)ethanol 3 (5.3 g, 91%).
Melting point: 159 – 161°C. (Ref. [2]: 88%, m.p. 158°C)
IR (nmax, KBr, cm-1): 3295 (OH), 1530, 1350, 1205, 1060
1H-NMR (400 MHz, CDCl3, d (ppm): 7.58 – 7.52 (m, 2H, Ar-H), 7.41 (t, 1H, Ar-H), 7.29 (t, 1H, Ar-H), 4.86 (t, 1H, OH, D2O exchangeable), 4.48 (t, 2H, CH2), 3.80 – 3.72 (m, 6H, (3 x CH2) ).
13C-NMR (100 MHz, CDCl3, d (ppm): 147.5, 142.7, 139.2, 134.9, 126.7, 126.1, 124.7, 123.8, 64.3, 58.1, 49.9, 46.2
MS (m/z, %): 231 (M+, 35.7), 212 ([M-H3O.]+, 6.0), 201 ([M - HCHO]+, 26.4), 200 ([M - CH2OH]+, 100), 187 ([M - CH2 = CHOH]+, 70.9), 129 (24.8), 90 (15.2), 56 (41.5).
Supplementary Materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgment
We thank the Alexander von Humboldt foundation for a post-doctoral fellowship (CAO).
References
- Obafemi, C.A.; Pfleiderer, Wolfgang. Helv. Chim. Acta 1994, 77, 1549–1556. [CrossRef]
- Goncharova, I.N.; Postovskii, I. Ya. J. J. Gen. Chem. USSR 1962, 32, 3271–3278, 1H-NMR, 13C-NMR and mass spectra data are not given in this paper.
© 2006 MDPI. All rights reserved.
