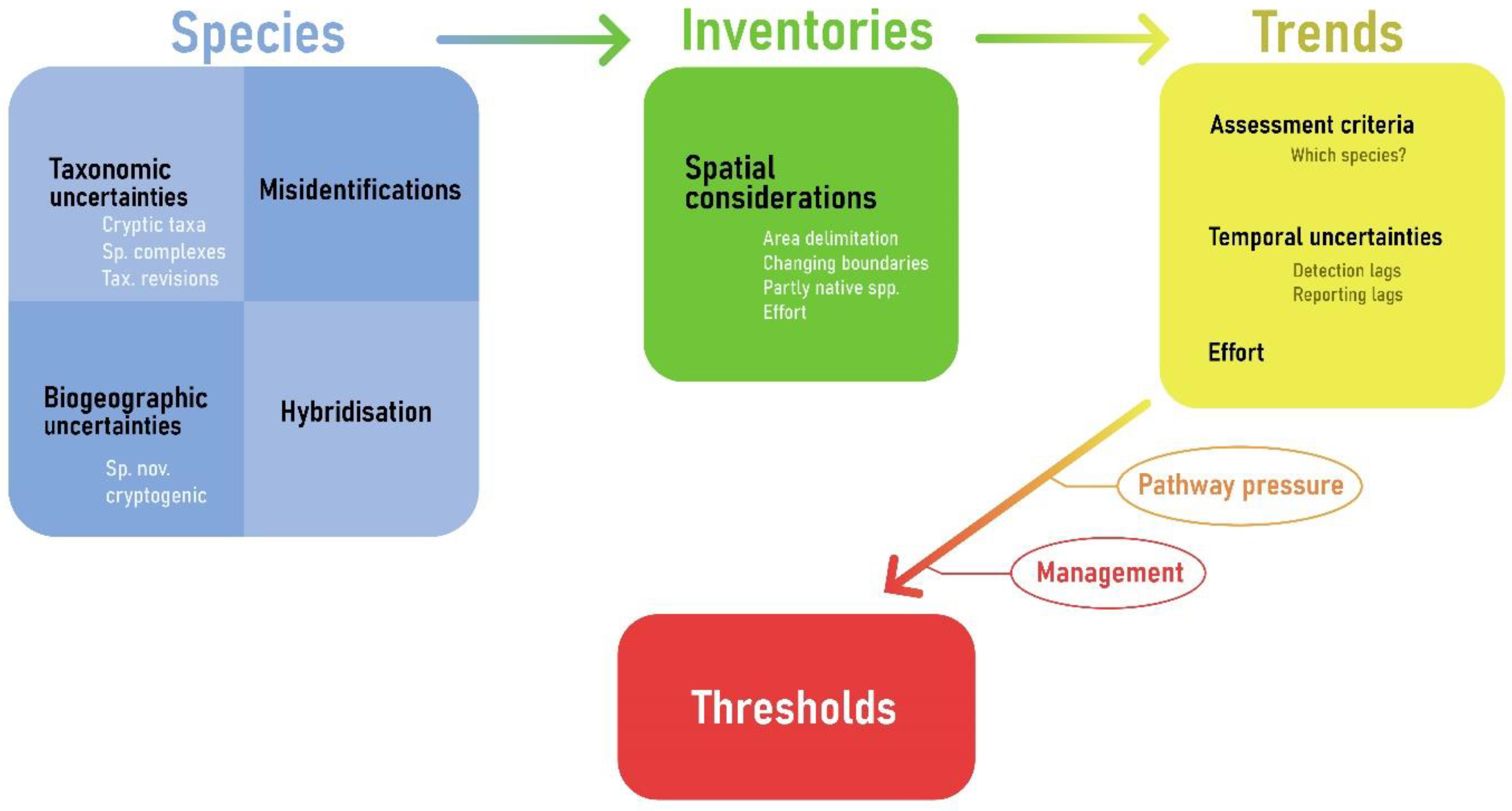
Abstract
Invasive alien species are a major worldwide driver of biodiversity change. The current study lists verified records of non-indigenous species (NIS) in European marine waters until 2020, with the purpose of establishing a baseline, assessing trends, and discussing appropriate threshold values for good environmental status (GES) according to the relevant European legislation. All NIS records were verified by national experts and trends are presented in six-year assessment periods from 1970 to 2020 according to the European Union Marine Strategy Framework Directive. Altogether, 874 NIS have been introduced to European marine waters until 2020 with the Mediterranean Sea and North-East Atlantic Ocean hosting most of the introductions. Overall, the number of new introductions has steadily increased since 2000. The annual rate of new introductions reached 21 new NIS in European seas within the last six-year assessment period (2012–2017). This increase is likely due to increased human activities and research efforts that have intensified during the early 21st century within European Seas. As Europe seas are not environmentally, nor geographically homogenous, the setting of threshold values for assessing GES requires regional expertise. Further, once management measures are operational, pathway-specific threshold values would enable assessing the effectiveness of such measures.
1. Introduction
The introduction of marine Non-Indigenous Species (NIS) is widely perceived as one of the main threats to biological diversity next to habitat destruction at a global scale [1,2]. Invasive Alien Species (IAS) are a subset of NIS, which are of particular concern due to their ability to naturally reproduce in the recipient areas, spread rapidly, and threaten biological diversity in various ways, from reducing genetic variation and modifying gene pools, displacing, hybridizing or competing with local endemic or native species to altering habitat and ecosystem functioning [3,4,5,6,7]. It is essential to note that the term “invasive” may have various implications depending on the context. From a scientific perspective, “invasive” refers to the ability of the species to survive, reproduce and spread in the invaded region [8], whereas political frameworks, such as the EU Regulation (No 1143/2014) on the prevention and management of the introduction and spread of invasive alien species (IAS Regulation) often connect invasiveness to impact.
Marine NIS, and IAS in particular, are addressed by European Union (EU) policies, such as the EU Biodiversity Strategy 2020 (COM (2011) 244) target 5; the European Water Framework Directive (WFD) (2000/60/EC); the EU Marine Strategy Framework Directive (MSFD) (2008/56/EC) with a dedicated descriptor (D2 “Non-indigenous species introduced by human activities are at levels that do not adversely alter the ecosystems”) and the IAS Regulation (No 1143/2014). Non-indigenous species is one of the 11 descriptors in the MSFD that refer to anthropogenic pressures on the marine environment of the EU [9]. In the latest MSFD update [9] among the criteria for assessing descriptor D2 on marine NIS, primary criterion D2C1 concerning new NIS introductions states that: “The number of non-indigenous species which are newly introduced via human activity into the wild, per assessment period (6 years), measured from the reference year (2011) as reported for initial assessment under Article 8(1) of Directive 2008/56/EC, is minimised and where possible reduced to zero”. Efforts to make this target more quantitative are ongoing [10,11,12], further encouraged by Target 6 of the first draft of the Convention on Biological Diversity (CBD) Post-2020 Global Biodiversity Framework, which stipulates at least a 50% reduction in the rate of new introductions [13]. However, to date, only the Baltic Marine Environment Protection Commission (Helsinki Convention, HELCOM) has set a numerical threshold of zero new NIS introductions through anthropogenic activities in the Baltic Sea [10]. At the EU level, Tsiamis et al. [14] suggested that the most suitable approach for setting the Good Environmental Status (GES) thresholds for criterion D2C1 would be a percentage reduction of new NIS introductions for an assessment period compared to the previous six-year assessment period (baseline). Preferably, the more previous six-year cycles that are included in the assessment, the better (e.g., starting from the 1970s) since the inclusion of earlier assessment periods enables tracking down how management measures have changed the result of the assessment over time. Thus, as qualitative GES descriptions turn into quantitative targets, it is now more imperative than ever that information on NIS in European seas is as accurate and complete as possible to provide a sound baseline for future management.
The first compilation of marine NIS inventory in Europe was conducted by Streftaris et al. [15] and followed by an update in 2009 toward the SEBI2010 report [16]. In the same period, comprehensive data collection from a wide range of taxonomic groups through the EU-funded project Delivering Alien Species Inventories for Europe resulted in a European database [17]. The DAISIE database, which included recorded information on the impacts, pathways of introduction, and associated references, was integrated into the information system on Aquatic Non-Indigenous and Cryptogenic Species (AquaNIS) [18]. In parallel, the European Alien Species Information Network (EASIN) [19] has been developed by the European Commission’s Joint Research Centre (JRC) aiming to facilitate the exploration of existing alien species information from a variety of distributed information sources through freely available tools and interoperable web services, compliant with internationally recognized standards. Updated information on NIS is provided by data partners and the editorial board of EASIN [20]. AquaNIS stores and disseminates information on NIS introduction histories, recipient regions, taxonomy, biological traits, impacts, and other relevant documented data. The system is continuously updated with new NIS records provided by registered data providers.
With the digital infrastructure in place and prompted by the increased demands placed by legislation, there is an increasing availability of national (e.g., Portugal) [21] and regional inventories of NIS (e.g., Baltic [22], Mediterranean [23], Black Sea [24]), which have been instrumental for analyzing trends and pathways of NIS introductions at national (e.g., Italy [25], Greece [26], Denmark [27], Belgium [28]), subregional (Macaronesia [29]), regional (Mediterranean [30], Baltic [22]), and global scales [31]. All these assessments have the shared ambition to assess the most updated status of NIS and provide a robust baseline for understanding trends in new NIS arrivals and pathways. Such knowledge is essential for the optimal implementation of existing policies and for evaluating policy effectiveness. Furthermore, knowledge is important to evaluate the need for new policies and management strategies. Updated and validated NIS inventories constitute a milestone for the implementation of the MSFD D2. Based on refined baseline inventories of NIS set by each EU Member State (MS), in the context of the MSFD and the updated data of EASIN, Tsiamis et al. [32] estimated that 787 non-indigenous taxa were found in EU marine and partially transitional waters (including Macaronesia) by the end of 2011. Further, Tsiamis et al. [14] updated the EASIN marine data at the national and MSFD subregional levels up to 31 December 2017. In the period of 2018–2020, not only have new NIS been identified in the European seas, but also new information has emerged on the taxonomic identity (e.g., as a consequence of recent taxonomic revision efforts), biogeographic origin, and distribution of NIS records, resulting in significant changes in both the status and distribution of several species. Now more than ever, it is crucial to reassess, revise and update the NIS inventories at all spatial assessment levels. In this context, the present work presents the most updated list of marine NIS introduced in the EU and surrounding waters validated by national experts and examines trends in these NIS introductions at European, regional, and subregional levels paving the way for the setting of threshold values for new NIS introductions in the context of the MSFD, and particularly of the primary criterion D2C1.
2. Methodology
The national inventories of EU countries submitted to JRC for the purposes of the 2012–2017 assessment cycle [33] formed the starting point for the revision process. They were updated with published data from biodiversity and hot-spot campaigns, academic surveys, and citizen science project observations until December 2020 (reported until June 2022). For Norway, Albania, and Montenegro, local experts were invited. The subsequent validation of the revised lists with the contribution of national experts included several rounds of communication whereby many discrepancies were resolved, and several controversial species were agreed upon. Subsequently, the national data were aggregated at subregional, regional, and Pan-European levels. The species list includes every first novel report of species introduction, irrespective of the establishment status. In our analysis, we only considered the first new record of a NIS within a region/subregion. Duplicate records for any given species were removed to avoid overestimating new NIS records at all spatial levels. The number of species detected/observed per six-year cycles since 1970 was analyzed from these datasets.
2.1. Geographic Coverage
The study area included European marine waters surrounding EU countries, EU candidate countries (Albania, Montenegro), and Norway a country of the European Economic Area (EEA) all divided into regions and subregions (Figure 1, Table 1) as per the MSFD delineation [33]. Marine waters of the United Kingdom (UK), Turkey, and Russian Federation were not considered in this work, meaning that NIS records from these countries are not included.
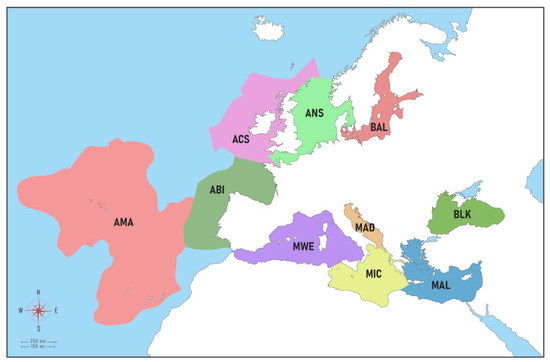
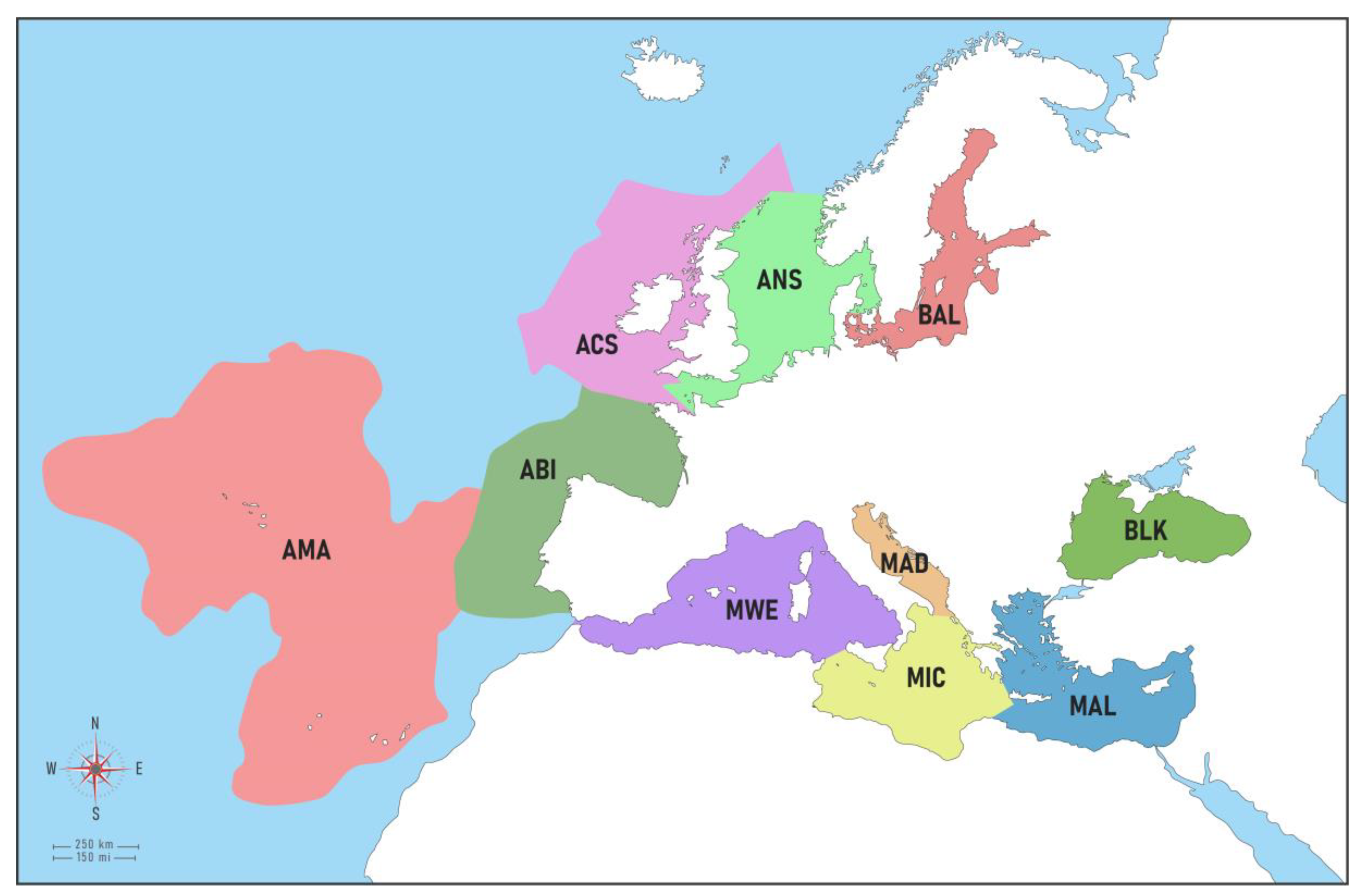
Figure 1.
European subregions (modified from Jensen et al. [34]). BAL = Baltic Sea, ANS = Greater North Sea, ACS = Celtic Seas, ABI = Bay of Biscay-Iberian Shelf, AMA = Macaronesia, MWE = Western Mediterranean, MIC = Central Mediterranean, MAD = Adriatic Sea, MAL = Eastern Mediterranean, BLK = Black Sea.

Table 1.
Geographic coverage of new NIS introductions in the present study at regional and subregional levels. Abbreviation: ABI = Bay of Biscay and the Iberian Coast, ACS = Celtic Seas, ANS = Greater North Sea, AMA = Macaronesia, MWE = Western Mediterranean Sea, MIC = Ionian Sea and the Central Mediterranean Sea, MAD = Adriatic Sea, MAL = Aegean-Levantine Sea (Eastern Mediterranean Sea).
The Baltic Sea (BAL) is here regarded as both a region and a subregion according to the MSFD delineation, and the same applies to the Black Sea (BLK). The North-East Atlantic (NEA) comprises four MSFD subregions, namely: (a) Greater North Sea (ANS) (b) Celtic Seas (ACS), (c) the Bay of Biscay and the Iberian Coast (ABI), and (d) Macaronesia (AMA). The ANS spans the Kattegat, the eastern English Channel, and a small part of the Western English Channel. It covers NIS in coastal and estuarine waters from seven countries including Norway (an EEA country). The Celtic Seas (ACS) are represented only by Ireland and the western English Channel waters of France. Macaronesia (AMA) is a complex of oceanic islands located in the NEA. The region comprises the archipelagos of the Azores (Portugal), Madeira (Portugal), Canary Islands (Spain), and Cabo Verde. For the present paper exclusively European Macaronesia (i.e., the Azores, Madeira, and Canary Islands), which.h is the European marine ecoregion within the Lusitanian province following the proposed classification in [35], was considered. The Mediterranean Sea (MED) includes four MSFD subregions: (a) the Western Mediterranean Sea (MWE); (b) the Ionian Sea and the Central Mediterranean Sea (MIC); (c) the Adriatic Sea (MAD); and (d) the Eastern Mediterranean Sea (MAL), encompassing the Aegean and Levantine basins.
2.2. Data Included
The most recent MSFD D2 evaluation recommendations [13] were largely followed for the inclusion of marine NIS in the present analyses. Accordingly, cryptogenic, and crypto-expanding species for the regions considered were removed from NIS lists and subsequent analyses. The terms cryptogenic and crypto expanding refer to uncertainties in the status of a species in relation to either their true native range [36] or true dispersion pathway (i.e., natural range expansion vs. human-mediated expansion) [14].
Species with insufficient information or new records unverified by experts or NIS with unresolved taxonomic status [32] were included in this study only after detailed scrutiny by different experts and a general agreement that there is a strong indication that their presence and distribution pattern implies an introduction event. It is worth mentioning the case of the annelid Laonome xeprovala, by Bick and Bastrop in Bick et al., 2018, a species described from the Netherlands and subsequently found in other Dutch rivers, canals, and estuaries [37], as well as in the eastern part of the Baltic Sea, and identified originally as Laonome calida Capa, 2007 [38]. Previous literature suggests that North America’s eastern coast is a potential native origin for Laonome xeprovala, although further clarification is still required [39].
It has been heavily debated in recent years whether parasitic NIS and pathogens (including disease agents) should be omitted from MSFD D2 since they are managed under the Aquatic Animal Health Directive (2006/88/EC) [32]. Overall, the JRC group agreed that these NIS should be reported in D2 criteria, but not considered when assessing against a GES threshold [14]. Aiming to produce results that are as representative and comparable as possible with future GES assessments, parasites and pathogens are listed in Table 2 but were not considered in the D2 trend and status analyses.
There are contrasting opinions among national NIS experts with regard to microscopic algae (phytoplankton) and to their native, cryptogenic, or NIS status, which is reflected in the literature [40] but also in the information systems of EASIN and AquaNIS. However, due to the high reproductive potential of phytoplankton and thus the high potential of spreading, it is important to have a gauge on phytoplankton expansion. The JRC invited the D2 NIS experts’ network to contact phytoplankton experts across Europe, to set up a working group that could deliver a consolidated revision of phytoplankton NIS in European seas [14]. Given that further clarification is yet to be provided regarding the status of microalgae in Europe, they are listed in Table 2 but were not considered in the D2 trend and status analyses.
Oligohaline species are included if such species were found in estuarine or coastal systems of the marine region.
NIS spreading from one region/subregion to another through natural dispersal mechanisms (secondary introduction) is included in our analyses. Their introduction pathway was classified as UNAIDED. Such is the case of many Red Sea species that have invaded the eastern Mediterranean (known as Lessepsian immigrants) and are progressively moving to the central and western Mediterranean as well as to the Adriatic Sea. However, species that have undergone tropicalization processes (i.e., shifts in range distribution induced by climate change) [41] were not included as NIS, and thus not considered in these analyses.
With regards to partly native and partly cryptogenic species, here defined as species that are native or cryptogenic in one EU region while they are non-indigenous (i.e., introduced by humans), in another EU region, they were included in the analyses at regional and/or subregional level but not at the pan-European level. Such NIS notably include Mediterranean molluscan transported with shellfish movements to the North-East Atlantic and vice versa, as well as also sessile biota, such as tunicates. Species native within a subregion (e.g., North Sea) that have been anthropogenically transferred to another country within the same subregion, were not included in the subregional analysis, although they are regarded as NIS in the countries they have invaded. This also applies to countries with coastal areas in more than one regional sea (Denmark, France, Germany, Spain, and Sweden).
2.3. Detection Year
The year of introduction was based on the reported date of the first collection/detection. However, it is important to point out that this date does not necessarily reflect the actual year of introduction which may have occurred years or even decades earlier since most species are often overlooked in the early stages of the invasion process, e.g., the green alga Codium fragile that has spread rapidly throughout the globe from its native range in Japan and the North Pacific was first detected in Europe c. 1900 in the Netherlands but reported in 1955 [42]. In addition, the date of first detection/collection is not always documented. In such cases, the publication date was accepted as the first record date. Moreover, in cases where only a time range has been supplied (e.g., 1986–1994), or the first record refers to a decade (e.g., the 1970s), the introduction date was set approximately as the average year for that given period (1990 and 1975, respectively).
3. Results
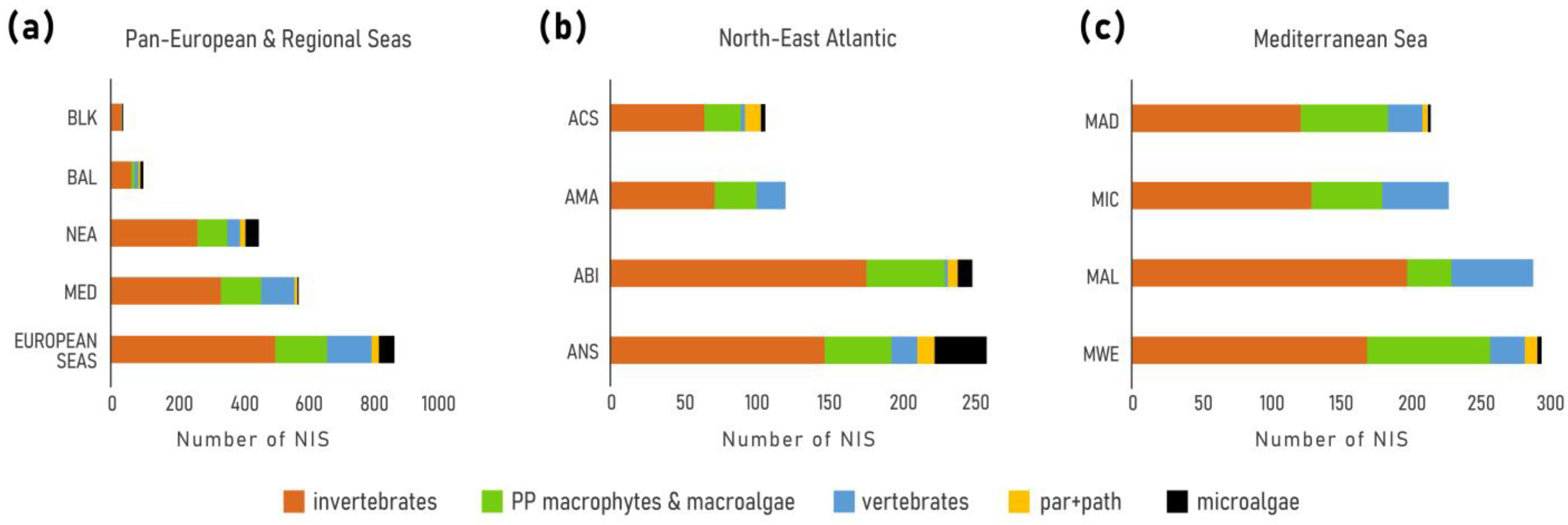
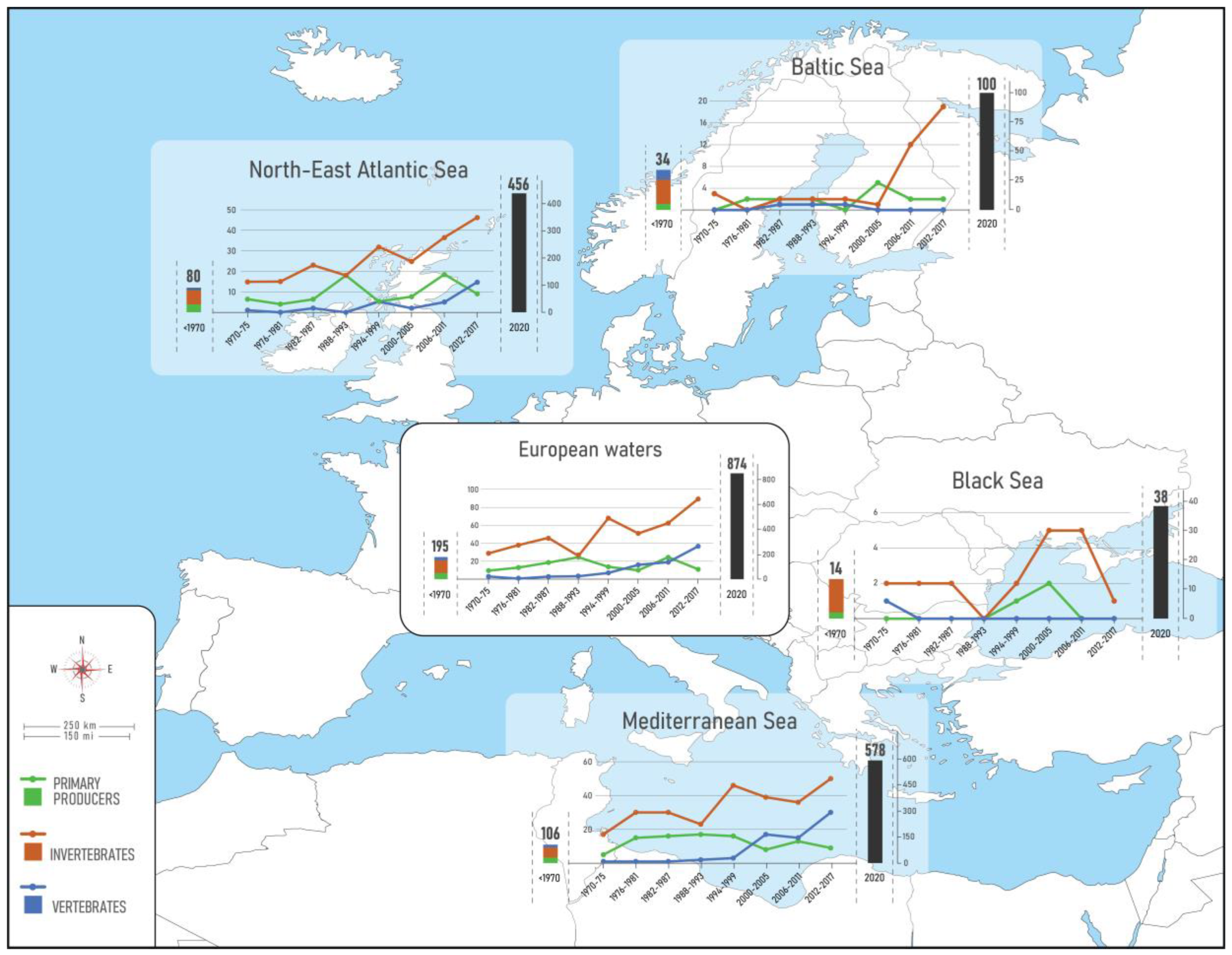
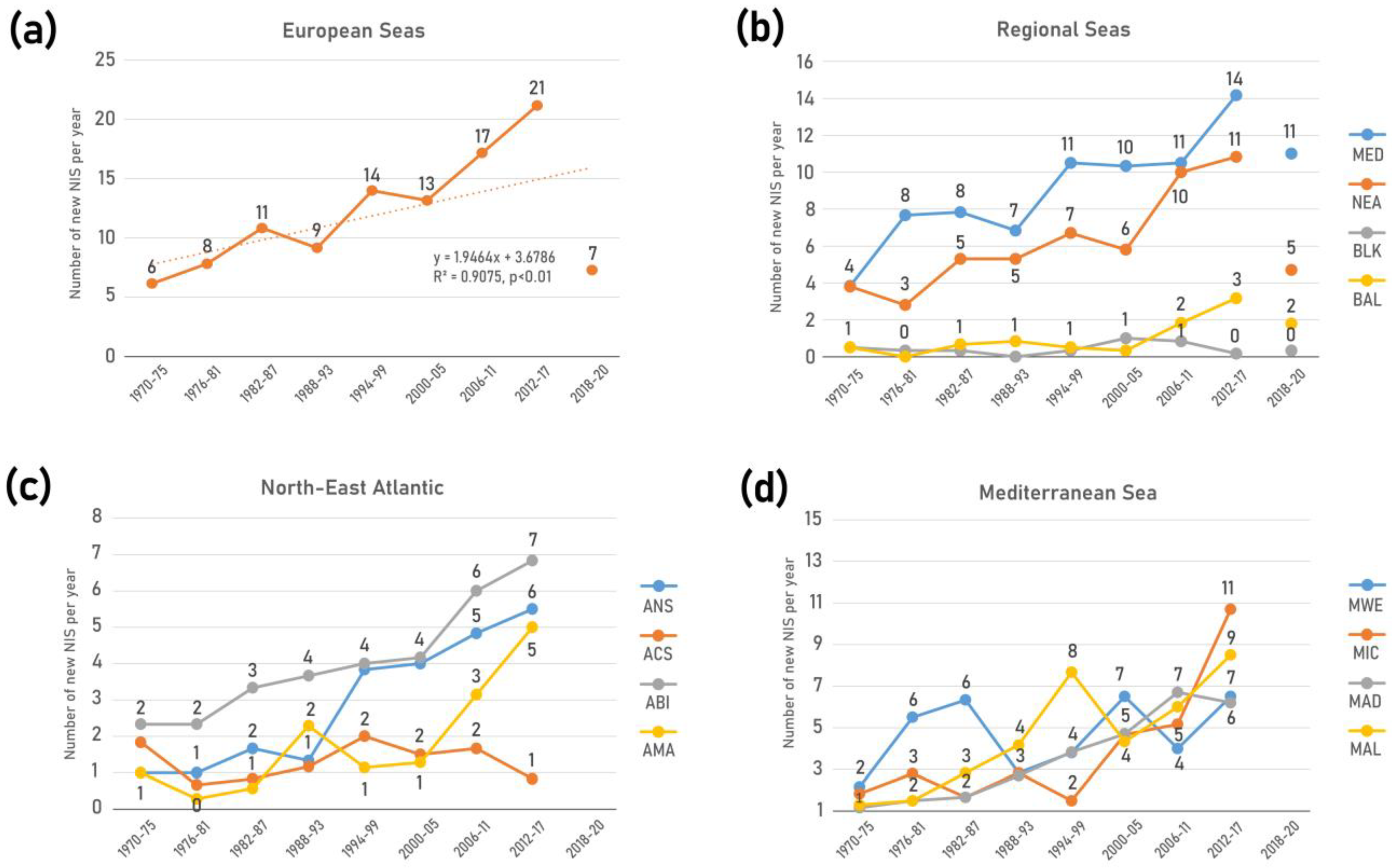
In total, 874 NIS were identified across European seas by December 2020 including 22 species of parasites and pathogens, and 50 species of microalgae (Table 2, Figure 2a). Of these 80% (701 taxa) were first reported in 1970. The vast majority of NIS are invertebrates (59%), followed by primary producers (algae and plants) (25%) and vertebrates (16%). Dissimilar proportions of all mentioned groups were evidenced across regions and subregions (Figure 3). While invertebrates dominate at all regional seas, the contribution of vertebrates (fishes) at the pan-European level is largely driven by the high contribution of Red Sea fish species in the Mediterranean Sea (Lessepsian immigrants) as opposed to their low presence in the NEA and Black Sea. Primary producers have a higher share in the NEA (29%) than the other regional seas (14–22%).
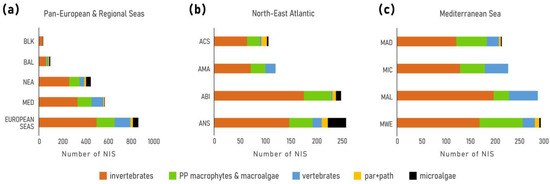
Figure 2.
Number of NIS detected by December 2020. (a) European waters and regional Seas, (b) North-East Atlantic subregions: ANS = Greater North Sea, ABI = Bay of Biscay-Iberian Shelf, AMA = Macaronesia, ACS = Celtic Seas; (c) Mediterranean subregions: MWE = Western Mediterranean, MAL = Eastern Mediterranean, MIC = Central Mediterranean, MAD = Adriatic Sea.
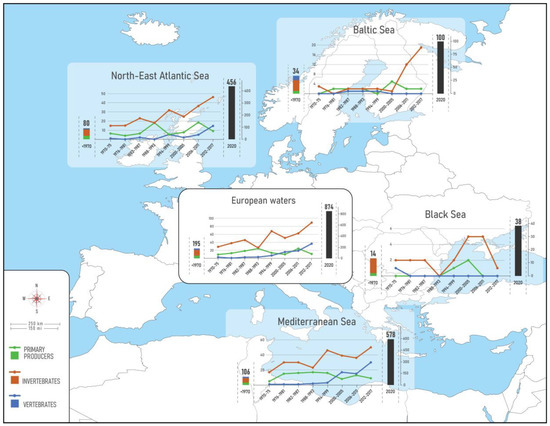
Figure 3.
Status and trends in introduction of NIS in European seas. Bars depict the cumulative number of NIS, from historical times to 2020. Details for the status in 2020 (black bar) as in Figure 2. Lines show the trends in new NIS introductions per 6-year intervals from 1970 to 2017. Note: parasites/pathogens and microalgae were excluded from the trend analyses.

Table 2.
List of NIS and their first year of detection at pan-European and regional levels. Group: VER = vertebrate, INV = invertebrate, PP = primary producer, INV/par = parasite, PP/micro = microalgae. BAL = Baltic Sea, NEA = North-East Atlantic Sea, MED = Mediterranean Sea, BLK = Black Sea. In bold, species detected since 1970. Asterisk denotes freshwater species detected in marine/estuarine environments.
Table 2.
List of NIS and their first year of detection at pan-European and regional levels. Group: VER = vertebrate, INV = invertebrate, PP = primary producer, INV/par = parasite, PP/micro = microalgae. BAL = Baltic Sea, NEA = North-East Atlantic Sea, MED = Mediterranean Sea, BLK = Black Sea. In bold, species detected since 1970. Asterisk denotes freshwater species detected in marine/estuarine environments.
| Group | Species | Pan-European | BAL | NEA | MED | BLK |
|---|---|---|---|---|---|---|
| VER | Ablennes hians (Valenciennes, 1846) | 2018 | 2018 | |||
| VER | Abudefduf sexfasciatus (Lacepède, 1801) | 2017 | 2017 | |||
| VER | Abudefduf vaigiensis (Quoy & Gaimard, 1825) | 2005 | 2005 | |||
| VER | Abudefduf hoefleri (Steindachner, 1881) | 2014 | 2014 | |||
| INV | Acanthaster planci (Linnaeus, 1758) | 2006 | 2006 | |||
| VER | Acanthopagrus bifasciatus (Forsskål, 1775) | 2019 | 2019 | |||
| PP | Acanthosiphonia echinata (Harvey) A.M.Savoie & G.W.Saunders | 2018 | 2018 | |||
| VER | Acanthurus bahianus Castelnau, 1855 | 2013 | 2013 | |||
| VER | Acanthurus cfr gahhm (Forsskål, 1775) | 2019 | 2019 | |||
| VER | Acanthurus coeruleus Bloch & Schneider, 1801 | 2011 | 2013 | 2011 | ||
| VER | Acanthurus sohal (Forsskål, 1775) | 2017 | 2017 | |||
| VER | Acanthurus chirurgus (Bloch, 1787) | 2012 | 2013 | 2012 | ||
| INV | Acartia (Acanthacartia) tonsa Dana, 1849 | 1921 | 1921 | 1921 | 1986 | 1976 |
| INV | Acartia (Acartiura) omorii Bradford, 1976 | 2004 | 2004 | |||
| INV | Achelia sawayai Marcus, 1940 | 2016 | 2016 | |||
| VER | Acipenser baerii Brandt, 1869 | 1960 | 1960 | 1985 | ||
| VER | Acipenser gueldenstaedtii Brandt & Ratzeburg, 1833* | 1962 | 1962 | 2010 | ||
| VER | Acipenser ruthenus Linnaeus, 1758* | 1887 | 1887 | |||
| VER | Acipenser stellatus Pallas, 1771 | 1999 | 1999 | |||
| VER | Acipenser transmontanus Richardson, 1836 | 1999 | 1999 | |||
| PP | Acrochaetium catenulatum M.A.Howe | 1967 | 1967 | |||
| PP | Acrothamnion preissii (Sonder) E.M.Wollaston | 1968 | 2009 | 1968 | ||
| INV | Actaeodes tomentosus (H. Milne Edwards, 1834) | 2013 | 2013 | |||
| INV | Acteocina mucronata (Philippi, 1849) | 1991 | 1991 | |||
| INV | Actumnus globulus Heller, 1861 | 1978 | 1978 | |||
| PP | Adelosina carinatastriata (Wiesner) | 2004 | 2004 | |||
| Pathogen | Aerococcus viridans Williams, Hirch & Cowan | 1961 | 1961 | |||
| PP | Agardhiella subulata (C.Agardh) Kraft & M.J.Wynne | 1984 | 1989 | 1984 | ||
| PP | Agarophyton vermiculophyllum (Ohmi) Gurgel, J.N.Norris & Fredericq | 1989 | 2003 | 1989 | 2008 | |
| PP | Aglaothamnion halliae (Collins) Aponte, D.L.Ballantine & J.N.Norris | 1960 | 1960 | 2016 | ||
| VER | Agonus cataphractus (Linnaeus, 1758) | 2005 | 2005 | |||
| PP | Ahnfeltiopsis flabelliformis (Harvey) Masuda, 1993 | 1994 | 1994 | |||
| PP/micro | Akashiwo sanguinea (K.Hirasaka) G.Hansen & Ø.Moestrup | 1982 | 1982 | |||
| VER | Alepes djedaba (Forsskål, 1775) | 1960 | 1960 | |||
| PP/micro | Alexandrium ostenfeldii (Paulsen) Balech & Tangen | 1986 | 1986 | |||
| PP/micro | Alexandrium affine (H.Inoue & Y.Fukuyo) Balech | 1987 | 1987 | |||
| PP/micro | Alexandrium leei Balech | 1991 | 1991 | |||
| PP/micro | Alexandrium margalefii Balech | 2006 | 2006 | |||
| PP/micro | Alexandrium taylori Balech | 1994 | 1994 | |||
| INV | Aliculastrum cylindricum (Helbling, 1779) | 2020 | 2020 | |||
| INV/par | Allolepidapedon fistulariae Yamaguti, 1940 | 2005 | 2005 | |||
| INV | Alpheus rapacida de Man, 1908 | 1998 | 1998 | |||
| INV | Amathina tricarinata (Linnaeus, 1767) | 2012 | 2012 | |||
| INV | Ammothea hilgendorfi (Böhm, 1879) | 1979 | 2013 | 1979 | ||
| INV | Ampelisca cavicoxa Reid, 1951 | 2005 | 2005 | |||
| INV | Ampelisca heterodactyla Schellenberg, 1925 | 1986 | 1986 | |||
| INV | Amphibalanus eburneus (Gould, 1841) | 1818 | 1872 | 1818 | 1933 | |
| INV | Amphibalanus reticulatus (Utinomi, 1967) | 1977 | 1997 | 1977 | ||
| INV | Amphibalanus variegatus (Darwin, 1854) | 1997 | 1997 | |||
| INV | Amphinome rostrata (Pallas, 1766) | 1900 | 1900 | |||
| PP | Amphistegina cf. papillosa Said, 1949 | 2005 | 2005 | |||
| PP | Amphistegina lessonii d’Orbigny in Guérin-Méneville, 1832 | 2001 | 2001 | |||
| PP | Amphistegina lobifera Larsen, 1976 | 1959 | 1959 | |||
| INV | Ampithoe valida Smith, 1873 | 1985 | 1985 | 2000 | ||
| INV | Anadara kagoshimensis (Tokunaga, 1906) | 1966 | 1993 | 1966 | 1981 | |
| INV | Anadara transversa (Say, 1822) | 1975 | 2016 | 1975 | ||
| INV/par | Anguillicola crassus (Kuwahara, Niimi & Itagaki, 1974) | 1980 | 1988 | 1982 | 1980 | |
| INV | Anomia chinensis Philippi, 1849 | 1974 | 1974 | |||
| INV | Anoplodactylus californicus Hall, 1912 | 1965 | 1965 | |||
| PP | Anotrichium furcellatum (J.Agardh) Baldock | 1950 | 1950 | |||
| PP | Antithamnion densum (Suhr) M.Howe | 1964 | 1964 | |||
| PP | Antithamnion diminuatum Wollaston | 1989 | 1989 | |||
| PP | Antithamnion hubbsii E.Y.Dawson | 1987 | 1989 | 1987 | ||
| PP | Antithamnion amphigeneum A.J.K.Millar | 1992 | 1995 | 1992 | ||
| PP | Antithamnionella ternifolia (Hooker fil. & Harvey) Lyle | 1910 | 2014 | 1910 | 1981 | |
| INV | Aoroides curvipes Ariyama, 2004 | 2009 | 2009 | |||
| INV | Aoroides semicurvatus Ariyama, 2004 | 2009 | 2009 | |||
| INV | Aoroides longimerus Ren & Zheng, 1996 | 2013 | 2013 | 2015 | ||
| INV | Apanthura addui Wägele, 1981 | 1998 | 1998 | |||
| INV | Aplidium antillense (Gravier, 1955) | 2004 | 2004 | |||
| INV | Aplidium accarense (Millar, 1953) | 2012 | 2012 | |||
| VER | Apogonichthyoides pharaonis (Bellotti, 1874) | 1964 | 1964 | |||
| INV | Aquilonastra burtoni (Gray, 1840) | 2003 | 2003 | |||
| INV | Arachnidium lacourti d’Hondt & Faasse, 2006 | 1999 | 2015 | 1999 | ||
| INV | Arachnoidella protecta Harmer, 1915 | 1992 | 1992 | |||
| INV | Arbopercula tenella (Hincks, 1880) | 1990 | 1990 | |||
| INV | Arctapodema australis (Vanhöffen, 1912) | 1967 | 1967 | |||
| INV | Arcuatula senhousia (Benson, 1842) | 1982 | 2002 | 1982 | 2002 | |
| INV | Argopecten gibbus (Linnaeus, 1758) | 2016 | 2016 | |||
| INV | Arhynchite arhynchite (Ikeda, 1924) | 2001 | 2001 | |||
| INV | Arietellus pavoninus Sars G.O., 1905 | 1967 | 1967 | |||
| VER | Arothron hispidus (Linnaeus, 1758) | 2018 | 2018 | |||
| INV | Artemia monica Verrill, 1869 | 1972 | 1987 | 1972 | ||
| INV | Ascidia curvata (Traustedt, 1882) | 2014 | 2014 | |||
| INV | Ascidia interrupta Heller, 1878 | 1990 | 1990 | |||
| INV | Asclerocheilus ashworthi Blake, 1981 | 2005 | 2005 | |||
| PP | Ascophyllum nodosum (Linnaeus) Le Jolis | 2009 | 2009 | |||
| PP | Asparagopsis taxiformis (Delile) Trevisan de Saint-Léon (lineage 2) | 1928 | 1928 | 1992 | ||
| PP | Asparagopsis armata Harvey | 1880 | 1922 | 1880 | ||
| INV | Asterocarpa humilis (Heller, 1878) | 2005 | 2005 | |||
| PP/micro | Asteromphalus sarcophagus Wallich, 1860 | 1993 | 1993 | |||
| INV | Atactodea striata (Gmelin, 1791) | 1977 | 1977 | |||
| INV | Atergatis roseus (Rüppell, 1830) | 2009 | 2009 | |||
| VER | Atherinomorus forskalii (Rüppell, 1838) | 1929 | 1929 | |||
| INV | Atys angustatus E. A. Smith, 1872 | 2017 | 2017 | |||
| INV | Atys ehrenbergi (Issel, 1869) | 2016 | 2016 | |||
| INV | Aurelia coerulea von Lendenfeld, 1884 | 2002 | 2002 | |||
| INV | Aurelia solida Browne, 1905 | 2000 | 2000 | |||
| INV | Austrominius modestus (Darwin, 1854) | 1944 | 1944 | 1990 | ||
| INV | Axionice medusa (Savigny in Lamarck, 1818) | 1976 | 1976 | |||
| INV | Baeolidia moebii Bergh, 1888 | 2017 | 2017 | |||
| INV | Balanus glandula Darwin, 1854 | 2015 | 2015 | |||
| INV | Balanus trigonus Darwin, 1854 | 1887 | 1887 | 1927 | ||
| VER | Balistoides conspicillum (Bloch & Schneider, 1801) | 2012 | 2012 | |||
| INV | Bankia fimbriatula Moll & Roch, 1931 | 1847 | 1847 | |||
| INV | Barentsia ramosa (Robertson, 1900) | 1962 | 1962 | |||
| PP | Batophora occidentalis var. largoensis (Harvey) S.Berger & Kaever ex M.J.Wynne | 2020 | 2020 | |||
| INV | Beania maxilladentata Ramalho, Muricy & Taylor, 2010 | 2013 | 2013 | |||
| INV | Bemlos leptocheirus (Walker, 1909) | 2015 | 2015 | |||
| INV | Beroe ovata Bruguière, 1789 | 1997 | 2011 | 2013 | 2004 | 1997 |
| INV | Berthellina citrina (Rüppell & Leuckart, 1828) | 2019 | 2019 | |||
| PP/micro | Biddulphia rhombus (Ehrenberg) W.Smith | 1983 | 1983 | |||
| PP/micro | Biddulphia sinensis Greville | 1903 | 1904 | 1903 | ||
| INV | Biflustra grandicella (Canu & Bassler, 1929) | 2016 | 2016 | |||
| INV | Bispira polyomma Giangrande & Faasse, 2012 | 2010 | 2010 | 2014 | ||
| INV | Biuve fulvipunctata (Baba, 1938) | 1993 | 1993 | |||
| INV | Boccardia proboscidea Hartman, 1940 | 1996 | 1996 | 2014 | ||
| INV | Boccardia semibranchiata Guérin, 1990 | 1999 | 1999 | |||
| INV | Boccardiella hamata (Webster, 1879) | 2001 | 2001 | |||
| Pathogen | Bonamia exitiosa Hine, Cochennac & Berthe | 2006 | 2006 | 2007 | ||
| Pathogen | Bonamia ostreae Pichot, Comps, Tigé, Grizel & Rabouin | 1978 | 1978 | 1990 | ||
| PP | Bonnemaisonia hamifera Hariot | 1898 | 1900 | 1898 | 1932 | |
| INV | Bostrycapulus odites Collin, 2005 | 1973 | 1973 | |||
| INV | Botrylloides diegensis Ritter & Forsyth, 1917 | 1999 | 1999 | 2004 | ||
| INV | Botrylloides giganteum (Pérès, 1949) | 2003 | 2003 | |||
| INV | Botrylloides niger Herdman, 1886 | 2013 | 2013 | 2014 | ||
| INV | Botrylloides violaceus Oka, 1927 | 1991 | 1999 | 1991 | ||
| PP | Botryocladia wrightii (Harvey) W.E.Schmidt, D.L.Ballantine & Fredericq | 1978 | 2005 | 1978 | ||
| PP | Botryocladia madagascariensis G.Feldmann | 1978 | 1978 | |||
| PP | Botrytella parva (Takamatsu) H.S.Kim | 1996 | 1996 | |||
| INV | Bougainvillia macloviana Lesson, 1830 | 1895 | 1895 | |||
| INV | Brachidontes exustus (Linnaeus, 1758) | 1977 | 1977 | |||
| INV | Brachidontes pharaonis (P. Fischer, 1870) | 1960 | 1960 | |||
| INV | Branchiomma bairdi (McIntsosh, 1885) | 1998 | 2012 | 1998 | ||
| INV | Branchiomma boholense (Grube, 1878) | 2004 | 2004 | |||
| INV | Branchiomma luctuosum (Grube, 1870) | 1978 | 2015 | 1978 | ||
| VER | Bregmaceros nectabanus Whitley, 1941 | 2014 | 2014 | |||
| INV | Bugulina simplex (Hincks, 1886) | 1982 | 1982 | |||
| INV | Bugulina stolonifera (Ryland, 1960) | 1976 | 1976 | |||
| INV | Bulla arabica Malaquias & Reid, 2008 | 1998 | 1998 | |||
| INV | Bursatella leachii Blainville, 1817 | 1969 | 1969 | |||
| INV | Calanopia elliptica (Dana, 1849) | 1891 | 1891 | |||
| INV | Callinectes danae Smith, 1869 | 1981 | 1981 | |||
| INV | Callinectes pallidus (de Rochebrune, 1883) | 2013 | 2013 | |||
| INV | Callinectes sapidus Rathbun, 1896 | 1901 | 1951 | 1901 | 1947 | 1967 |
| VER | Callionymus filamentosus Valenciennes, 1837 | 2003 | 2003 | |||
| INV | Calyptospadix cerulea Clarke, 1882 | 1940 | 2014 | 1978 | 1940 | |
| VER | Cantherhines pullus (Ranzani, 1842) | 2015 | 2015 | |||
| INV | Caprella mutica Schurin, 1935 | 1985 | 2017 | 1985 | ||
| INV | Caprella scaura Templeton, 1836 | 1985 | 1985 | 1994 | ||
| VER | Carassius auratus (Linnaeus, 1758) | 2012 | 2012 | |||
| VER | Carassius gibelio (Bloch, 1782)* | 1800 | 1800 | |||
| INV | Carijoa riisei (Duchassaing & Michelotti, 1860) | 2016 | 2016 | |||
| INV | Carupa tenuipes Dana, 1852 | 2009 | 2009 | |||
| INV | Cassiopea andromeda (Forsskål, 1775) | 1903 | 1903 | |||
| PP | Caulacanthus okamurae Yamada | 1999 | 1999 | 2002 | ||
| PP | Caulerpa cylindracea Sonder | 1991 | 1997 | 1991 | ||
| PP | Caulerpa lamourouxii (Turner) C.Agardh | 1956 | 1956 | |||
| PP | Caulerpa taxifolia (M.Vahl) C.Agardh | 1984 | 1984 | |||
| PP | Caulerpa taxifolia var. distichophylla (Sonder) Verlaque, Huisman & Procaccini | 2007 | 2007 | |||
| PP | Caulerpa webbiana Montagne | 2002 | 2002 | |||
| INV | Caulibugula zanzibariensis (Waters, 1913) | 2003 | 2003 | |||
| INV | Cellana rota (Gmelin, 1791) | 2007 | 2007 | |||
| INV | Celleporaria inaudita Tilbrook, Hayward & Gordon, 2001 | 2007 | 2007 | |||
| INV | Celleporaria aperta (Hincks, 1882) | 1975 | 1975 | |||
| INV | Celleporaria brunnea (Hincks, 1884) | 2007 | 2007 | 2010 | ||
| INV | Celleporaria vermiformis (Waters, 1909) | 2015 | 2015 | |||
| INV | Celleporella carolinensis Ryland, 1979 | 1993 | 1993 | |||
| INV | Celtodoryx ciocalyptoides (Burton, 1935) | 1996 | 1996 | |||
| INV | Centropages furcatus (Dana, 1849) | 1988 | 1988 | |||
| VER | Cephalopholis hemistiktos (Rüppell, 1830) | 2009 | 2009 | |||
| VER | Cephalopholis taeniops (Valenciennes, 1828) | 2009 | 2009 | |||
| VER | Cephalopholis nigri (Günther, 1859) | 2016 | 2016 | |||
| INV | Cephalothrix simula Iwata, 1952 | 2012 | 2012 | |||
| PP | Ceramium atrorubescens Kylin | 1988 | 1988 | |||
| PP | Ceramium sungminbooi Hughey & Boo | 2018 | 2018 | |||
| PP | Ceramium tenuicorne (Kützing) Waern | 2011 | 2011 | |||
| PP | Ceramium bisporum D.L.Ballantine | 1980 | 1980 | |||
| PP | Ceramium strobiliforme G.W.Lawson & D.M.John | 1991 | 1991 | |||
| INV | Ceratonereis mirabilis Kinberg, 1865 | 1997 | 1997 | |||
| INV | Cerithidium perparvulum (Watson, 1886) | 1995 | 1995 | |||
| INV | Cerithiopsis pulvis (Issel, 1869) | 1985 | 1985 | |||
| INV | Cerithiopsis tenthrenois (Melvill, 1896) | 1985 | 1985 | |||
| INV | Cerithium scabridum Philippi, 1848 | 1972 | 1972 | |||
| PP/micro | Chaetoceros peruvianus Brightwell | 1981 | 1981 | |||
| PP/micro | Chaetoceros rostratus Ralfs | 2003 | 2003 | |||
| PP/micro | Chaetoceros bacteriastroides G.H.H.Karsten | 1996 | 1996 | |||
| PP/micro | Chaetoceros concavicornis Mangin | 2011 | 2011 | |||
| PP/micro | Chaetoceros pseudosymmetricus Nielsen | 2015 | 2015 | |||
| VER | Chaetodipterus faber (Broussonet, 1782) | 2019 | 2019 | |||
| VER | Chaetodon sanctaehelenae Günther, 1868 | 1993 | 1993 | |||
| VER | Chaetodon auriga Forsskål, 1775 | 2015 | 2015 | |||
| VER | Chaetodontoplus septentrionalis (Temminck & Schlegel, 1844) | 2015 | 2015 | |||
| INV | Chaetopleura angulata (Spengler, 1797) | 1850 | 1850 | |||
| INV | Chaetozone corona Berkeley & Berkeley, 1941 | 1982 | 1996 | 1982 | ||
| INV | Chama asperella Lamarck, 1819 | 2007 | 2007 | |||
| INV | Chama pacifica Broderip, 1835 | 1998 | 1998 | |||
| VER | Champsodon nudivittis (Ogilby, 1895) | 2012 | 2012 | |||
| INV | Charybdis (Charybdis) japonica (A. Milne-Edwards, 1861) | 2006 | 2006 | |||
| INV | Charybdis (Charybdis) feriata (Linnaeus, 1758) | 2004 | 2004 | |||
| INV | Charybdis (Charybdis) hellerii (A. Milne-Edwards, 1867) | 1998 | 1998 | |||
| INV | Charybdis (Charybdis) lucifera (Fabricius, 1798) | 2006 | 2006 | |||
| INV | Charybdis (Goniohellenus) longicollis Leene, 1938 | 1969 | 1969 | |||
| PP/micro | Chattonella marina (Subrahmanyan) Hara & Chihara | 1974 | 1974 | |||
| VER | Cheilodipterus novemstriatus (Rüppell, 1838) | 2015 | 2015 | |||
| INV | Chelicorophium robustum (G.O. Sars, 1895) | 2018 | 2018 | |||
| INV | Chelicorophium curvispinum (G.O. Sars, 1895) | 1912 | 1921 | 1912 | ||
| VER | Chlorurus rhakoura Randall & Anderson, 1997 | 2017 | 2017 | |||
| PP | Chondria pygmaea Garbary & Vandermeulen | 1974 | 1974 | |||
| PP | Chondria curvilineata F.S.Collins & Hervey | 1981 | 1981 | |||
| PP | Chondrus giganteus f. flabellatus Mikami | 1994 | 1994 | |||
| VER | Chromis multilineata (Guichenot, 1853) | 2015 | 2015 | |||
| INV | Chromodoris quadricolor (Rüppell & Leuckart, 1830) | 1982 | 1982 | |||
| INV | Chrysaora achlyos Martin, Gershwin, Burnett, Cargo & Bloom, 1997 | 2018 | 2018 | |||
| VER | Chrysiptera cyanea (Quoy & Gaimard, 1825) | 2013 | 2013 | |||
| VER | Chrysiptera hemicyanea (Weber, 1913) | 2017 | 2017 | |||
| PP | Chrysonephos lewisii (W.R.Taylor) W.R.Taylor | 1988 | 1988 | |||
| INV | Cingulina isseli (Tryon, 1886) | 1998 | 1998 | |||
| INV | Ciona robusta Hoshino & Tokioka, 1967 | 1901 | 2007 | 1901 | ||
| VER | Cirrhitus atlanticus Osório, 1893 | 2018 | 2018 | |||
| PP | Cladophora patentiramea (Montagne) Kützing | 1991 | 1991 | |||
| INV | Clavelina oblonga Herdman, 1880 | 1929 | 1971 | 1929 | ||
| Pathogen | Claviceps purpurea (Fr.:Fr.)Tul. | 1960 | 1960 | |||
| PP | Clavulina cf. multicamerata Chapman, 1907 | 2012 | 2012 | |||
| INV | Clementia papyracea (Gmelin, 1791) | 1985 | 1985 | |||
| INV | Clymenella torquata (Leidy, 1855) | 1977 | 1977 | |||
| INV | Clytia gregaria (Agassiz, 1862) | 2017 | 2017 | |||
| INV | Clytia hummelincki (Leloup, 1935) | 1996 | 1996 | |||
| INV | Clytia linearis (Thorneley, 1900) | 1951 | 1983 | 1951 | ||
| PP | Codium arabicum Kützing | 2006 | 2006 | |||
| PP | Codium fragile subsp. fragile (Suringar) Hariot | 1895 | 1919 | 1895 | 1946 | |
| PP | Colaconema codicola (Børgesen) H.Stegenga, J.J. Bolton & R.J.Anderson | 1926 | 1926 | 1952 | ||
| PP | Colaconema dasyae (F.S.Collins) Stegenga, I.Mol, Prud’homme van Reine & Lokhorst | 1951 | 1951 | |||
| INV | Coleusia signata (Paul’son, 1875) | 2005 | 2005 | |||
| PP | Colpomenia peregrina Sauvageau | 1905 | 1905 | 1918 | ||
| INV | Conomurex persicus (Swainson, 1821) | 1983 | 1983 | |||
| INV | Corambe obscura (A.E. Verrill, 1870) | 1879 | 1879 | 1986 | ||
| INV | Corbicula fluminea (O. F. Müller, 1774) | 1978 | 1978 | |||
| INV | Corella eumyota Traustedt, 1882 | 2002 | 2002 | |||
| PP/micro | Corymbellus aureus J.C.Green | 1992 | 1992 | |||
| PP | Corynomorpha prismatica (J.Agardh) J.Agardh | 1990 | 1990 | |||
| PP | Corynophlaea verruculiformis (Y.-P.Lee & I.K.Lee) Y.-P.Lee | 1994 | 1994 | |||
| INV | Coryphellina rubrolineata O’Donoghue, 1929 | 2008 | 2008 | |||
| INV | Crassostrea rhizophorae (Guilding, 1828) | 1976 | 1976 | |||
| INV | Crassostrea virginica (Gmelin, 1791) | 1861 | 1861 | 1974 | ||
| INV | Crepidacantha poissonii (Audouin, 1826) | 1982 | 1982 | |||
| INV | Crepidula fornicata (Linnaeus, 1758) | 1902 | 1902 | 1957 | ||
| INV | Crepipatella dilatata (Lamarck, 1822) | 2005 | 2005 | 2014 | ||
| INV | Crisularia plumosa (Pallas, 1766) | 1937 | 1937 | |||
| INV | Crisularia serrata (Lamarck, 1816) | 1902 | 1902 | |||
| PP | Cryptonemia hibernica Guiry & L.M.Irvine | 1911 | 1911 | |||
| PP | Cushmanina striatopunctata (Parker & Jones, 1865) | 1913 | 1913 | |||
| INV | Cuthona perca (Er. Marcus, 1958) | 1976 | 1976 | |||
| INV | Cycloscala hyalina (G. B. Sowerby II, 1844) | 1992 | 1992 | |||
| INV | Cymodoce fuscina Schotte & Kensley, 2005 | 2015 | 2015 | |||
| VER | Cynoscion regalis (Bloch & Schneider, 1801) | 2009 | 2009 | |||
| VER | Cyprinus carpio (Linnaeus, 1758)* | 1200 | 1200 | 1879 | ||
| PP | Dasya sessilis Yamada | 1984 | 1989 | 1984 | ||
| PP | Dasysiphonia japonica (Yendo) H.-S.Kim | 1984 | 1984 | 1998 | ||
| INV | Dendostrea frons (Linnaeus, 1758) | 1983 | 1983 | |||
| INV | Dendostrea folium (Linnaeus, 1758) | 2005 | 2005 | |||
| PP | Derbesia rhizophora Yamada | 1984 | 1984 | |||
| INV | Desdemona ornata Banse, 1957 | 1983 | 1993 | 1983 | ||
| INV | Diadema setosum (Leske, 1778) | 2010 | 2010 | |||
| INV | Diadumene lineata (Verrill, 1869) | 1925 | 2011 | 1963 | 1925 | 1945 |
| PP/micro | Dicroerisma psilonereiella F.J.R.Taylor & S.A. Cattell | 1998 | 1998 | |||
| PP | Dictyota cyanoloma Tronholm, De Clerck, A.Gómez-Garreta & Rull Lluch in Tronholm et al. | 1935 | 2006 | 1935 | ||
| INV | Didemnum perlucidum Monniot F., 1983 | 2006 | 2006 | |||
| INV | Didemnum vexillum Kott, 2002 | 1968 | 1968 | 2007 | ||
| INV | Dikerogammarus villosus (Sowinsky, 1894) | 2015 | 2015 | |||
| INV | Dikoleps micalii Agamennone, Sbrana, Nardi, Siragusa & Germanà, 2020 | 2016 | 2016 | |||
| PP/micro | Dinophysis sacculus Stein | 2004 | 2004 | |||
| INV | Diodora funiculata (Reeve, 1850) | 2013 | 2013 | |||
| INV | Diplosoma listerianum (Milne Edwards, 1841) | 1877 | 1877 | |||
| INV | Dipolydora quadrilobata (Jacobi, 1883) | 2003 | 2003 | |||
| INV | Dipolydora socialis (Schmarda, 1861) | 2006 | 2006 | |||
| INV | Dipolydora tentaculata (Blake & Kudenov, 1978) | 2005 | 2005 | |||
| PP | Dipterosiphonia dendritica (C.Agardh) F.Schmitz | 1961 | 1961 | |||
| INV | Dispio magna (Day, 1955) | 1982 | 1982 | |||
| PP/micro | Dissodinium pseudocalani (Gonnert) Drebes ex Elbrachter & Drebes | 2003 | 2003 | |||
| INV | Distaplia magnilarva (Della Valle, 1881) | 1929 | 1929 | |||
| INV | Distaplia bermudensis Van Name, 1902 | 1953 | 2006 | 1953 | ||
| INV | Distaplia corolla Monniot F., 1974 | 1971 | 1971 | |||
| INV | Dodecaceria capensis Day, 1961 | 1976 | 1976 | |||
| INV | Dorvillea similis (Crossland, 1924) | 2014 | 2014 | |||
| INV | Dreissena rostriformis bugensis (Andrusov, 1897) | 2014 | 2014 | |||
| VER | Dussumieria elopsoides Bleeker, 1849 | 2005 | 2005 | |||
| INV | Dyspanopeus texanus (Stimpson, 1859) | 2015 | 2015 | |||
| INV | Dyspanopeus sayi (Smith, 1869) | 1992 | 2007 | 1992 | ||
| INV | Echinogammarus trichiatus (Martynov, 1932) | 2014 | 2014 | |||
| INV | Ecteinascidia styeloides (Traustedt, 1882) | 1983 | 1983 | |||
| INV | Ectopleura crocea (Agassiz, 1862) | 1895 | 1989 | 1895 | ||
| INV | Edwardsiella lineata (Verrill in Baird, 1873) | 2010 | 2010 | |||
| PP | Elachista spp mentioned as E. flaccida | 1993 | 1993 | |||
| VER | Elates ransonnettii (Steindachner, 1876) | 2005 | 2005 | |||
| PP | Elodea canadensis Michx.* | 1873 | 1873 | |||
| PP | Elodea nuttallii (Planch.) H.St.John | 1991 | 1991 | 2006 | ||
| PP | Elphidium striatopunctatum (Fichtel & Moll, 1798) | 1911 | 1911 | |||
| INV | Elysia nealae (Ostergaard, 1955) | 2018 | 2018 | |||
| PP/micro | Emiliania huxleyi (Lohmann) W.W.Hay & H.P.Mohler | 1989 | 1989 | |||
| INV | Endeis biseriata Stock, 1968 | 1979 | 1979 | |||
| INV | Ensis leei M. Huber, 2015 | 1978 | 1991 | 1978 | ||
| INV | Eocuma dimorphum Fage, 1928 | 1992 | 1992 | |||
| INV | Eocuma sarsii (Kossmann), 1880 | 1901 | 1901 | |||
| VER | Epinephelus fasciatus (Forsskål, 1775) | 2018 | 2018 | |||
| VER | Epinephelus coioides (Hamilton, 1822) | 1998 | 1998 | |||
| VER | Epinephelus malabaricus (Bloch & Schneider, 1801) | 2011 | 2011 | |||
| VER | Epinephelus merra Bloch, 1793 | 2004 | 2004 | |||
| VER | Equulites klunzingeri (Steindachner, 1898) | 1955 | 1955 | |||
| INV | Ergalatax junionae Houart, 2008 | 1993 | 1993 | |||
| INV | Eriocheir sinensis H. Milne Edwards, 1853* | 1912 | 1921 | 1912 | 1959 | 1997 |
| INV | Erugosquilla massavensis (Kossmann, 1880) | 1956 | 1956 | |||
| PP/micro | Ethmodiscus punctiger Castracane | 1800 | 1979 | 1800 | ||
| VER | Etrumeus golanii DiBattista, Randall & Bowen, 2012 | 1999 | 1999 | |||
| INV | Euchaeta concinna Dana, 1849 | 1987 | 1987 | |||
| INV | Eucheilota paradoxica Mayer, 1900 | 1967 | 1967 | |||
| INV | Euchone limnicola Reish, 1959 | 2015 | 2015 | |||
| INV | Eucidaris tribuloides (Lamarck, 1816) | 1998 | 1998 | |||
| INV | Eudendrium carneum Clarke, 1882 | 1950 | 1950 | |||
| INV | Eudendrium merulum Watson, 1985 | 1969 | 1969 | |||
| INV | Eunaticina papilla (Gmelin, 1791) | 2020 | 2020 | |||
| INV | Euplana gracilis Girard, 1853 | 2002 | 2002 | |||
| INV | Euplokamis dunlapae Mills, 1987 | 2011 | 2011 | |||
| PP/micro | Eupyxidicula turris (Greville) S.Blanco & C.E. Wetzel | 1983 | 1983 | |||
| INV | Eurypanopeus depressus (Smith, 1869) | 2009 | 2009 | |||
| INV | Eurytemora americana Williams, 1906 | 1938 | 1938 | |||
| INV | Eurytemora carolleeae Alekseev & Souissi, 2011 | 2011 | 2012 | 2011 | ||
| INV | Eurytemora pacifica Sato, 1913 | 2014 | 2014 | |||
| INV | Eurythoe laevisetis Fauvel, 1914 | 2011 | 2011 | |||
| INV | Eusarsiella zostericola (Cushman, 1906) | 2012 | 2012 | |||
| INV | Eusyllis kupfferi Langerhans, 1879 | 1998 | 1998 | |||
| INV | Euthymella colzumensis (Jousseaume, 1898) | 2017 | 2017 | |||
| PP/micro | Eutintinnus lusus-undae (Entz) | 2001 | 2001 | |||
| INV | Fauveliopsis glabra (Hartman, 1960) | 2007 | 2007 | |||
| INV | Favorinus ghanensis Edmunds, 1968 | 2020 | 2020 | |||
| INV | Faxonius limosus (Rafinesque, 1817) | 2015 | 2015 | |||
| INV | Fenestrulina malusii (Audouin, 1826) | 2011 | 2011 | |||
| INV | Fenestrulina delicia Winston, Hayward & Craig, 2000 | 2002 | 2002 | |||
| INV | Ferosagitta galerita (Dallot, 1971) | 2011 | 2011 | |||
| PP/micro | Fibrocapsa japonica S.Toriumi & H.Takano | 1924 | 1924 | |||
| INV | Ficopomatus enigmaticus (Fauvel, 1923) | 1919 | 1939 | 1921 | 1919 | 1935 |
| INV | Finella pupoides A. Adams, 1860 | 1996 | 1996 | |||
| VER | Fistularia petimba Lacepède, 1803 | 2018 | 2018 | |||
| VER | Fistularia commersonii Rüppell, 1838 | 1999 | 1999 | |||
| INV | Fistulobalanus albicostatus (Pilsbry, 1916) | 1973 | 1973 | |||
| INV | Fulvia fragilis (Forsskål in Niebuhr, 1775) | 1983 | 1983 | |||
| VER | Fundulus heteroclitus heteroclitus (Linnaeus, 1766) | 1970 | 1970 | 2005 | ||
| INV | Gafrarium savignyi (Jonas, 1846) | 2005 | 2005 | |||
| INV | Gammarus tigrinus Sexton, 1939 | 1931 | 1975 | 1931 | ||
| PP | Gelidium microdonticum W.R.Taylor | 2017 | 2017 | |||
| PP | Gelidium vagum Okamura | 2010 | 2010 | |||
| VER | Genyatremus cavifrons (Cuvier, 1830) | 2015 | 2015 | |||
| INV | Glabropilumnus laevis (Dana, 1852) | 1956 | 1956 | |||
| INV | Glycinde bonhourei Gravier, 1904 | 2007 | 2007 | |||
| VER | Gobiosoma bosc (Lacepède, 1800) | 2009 | 2009 | |||
| INV | Godiva quadricolor (Barnard, 1927) | 1985 | 1985 | |||
| INV | Goniadella gracilis (Verrill, 1873) | 1968 | 1968 | |||
| INV | Goniobranchus annulatus (Eliot, 1904) | 2004 | 2004 | |||
| INV | Goniobranchus obsoletus (Rüppell & Leuckart, 1830) | 2018 | 2018 | |||
| INV | Gonioinfradens giardi (Nobili, 1905) | 2010 | 2010 | |||
| INV | Gonionemus vertens A. Agassiz, 1862 | 1700 | 1700 | 1918 | ||
| PP | Goniotrichopsis sublittoralis G.M.Smith | 1975 | 1975 | 1989 | ||
| PP | Gracilariopsis chorda (Holmes) Ohmi | 2010 | 2010 | |||
| INV | Grandidierella japonica Stephensen, 1938 | 2010 | 2010 | 2010 | 2013 | |
| PP | Grateloupia imbricata Holmes | 2005 | 2005 | |||
| PP | Grateloupia asiatica S.Kawaguchi & H.W.Wang | 1984 | 1984 | |||
| PP | Grateloupia patens (Okamura) S.Kawaguchi & H.W.Wang | 1994 | 1994 | |||
| PP | Grateloupia subpectinata Holmes | 1978 | 1978 | 1990 | ||
| PP | Grateloupia turuturu Yamada | 1982 | 1989 | 1982 | ||
| PP | Grateloupia yinggehaiensis H.W.Wang & R.X.Luan | 2008 | 2008 | |||
| INV | Guinearma alberti (Rathbun, 1921) | 2016 | 2016 | |||
| VER | Gymnomuraena zebra (Shaw, 1797) | 2002 | 2002 | |||
| PP | Gymnophycus hapsiphorus Huisman & Kraft | 2011 | 2011 | |||
| INV/par | Gyrodactylus salaris Malmberg, 1957 | 1975 | 1975 | |||
| PP/micro | Gyrodinium corallinum Kofoid & Swezy | 2001 | 2001 | |||
| INV | Halgerda willeyi Eliot, 1904 | 1988 | 1988 | |||
| INV | Haliclona (Halichoclona) vansoesti de Weerdt, de Kluijver & Gómez, 1999 | 2019 | 2019 | |||
| INV | Haliclystus tenuis Kishinouye, 1910 | 2010 | 2010 | |||
| PP | Halimeda incrassata (J.Ellis) J.V.Lamouroux | 2011 | 2011 | |||
| INV | Haliotis discus hannai Ino, 1953 | 1985 | 1985 | |||
| INV | Haloa japonica (Pilsbry, 1895) | 1992 | 1992 | 1992 | ||
| PP | Halophila stipulacea (Forsskål) Ascherson | 1894 | 1894 | |||
| INV | Haminella solitaria (Say, 1822) | 2016 | 2016 | 2020 | ||
| Pathogen | Haplosporidium nelsoni Haskin, Stauber & Mackin | 1975 | 1975 | |||
| INV | Heleobia charruana (d’Orbigny, 1841) | 2014 | 2014 | |||
| INV | Heliacus implexus (Mighels, 1845) | 2019 | 2019 | |||
| INV | Hemigrapsus sanguineus (De Haan, 1835) | 1999 | 1999 | 1999 | 2008 | |
| INV | Hemigrapsus takanoi Asakura & Watanabe, 2005 | 1993 | 2014 | 1993 | ||
| INV | Hemimysis anomala G.O. Sars, 1907* | 1962 | 1962 | 1999 | 2007 | |
| VER | Hemiramphus far (Forsskål, 1775) | 1943 | 1943 | |||
| VER | Heniochus acuminatus (Linnaeus, 1758) | 2014 | 2014 | |||
| VER | Heniochus intermedius Steindachner, 1893 | 2013 | 2013 | 2014 | ||
| INV | Herbstia nitida Manning & Holthuis, 1981 | 2002 | 2002 | |||
| INV | Herdmania momus (Savigny, 1816) | 1998 | 1998 | |||
| PP | Herposiphonia parca Setchell | 1997 | 2006 | 1997 | ||
| INV | Hesperibalanus fallax (Broch, 1927) | 1976 | 1976 | 1976 | ||
| PP | Heterostegina depressa d’Orbigny, 1826 | 1988 | 1988 | |||
| INV | Heterotentacula mirabilis (Kramp, 1957) | 1997 | 1997 | |||
| PP | Hildenbrandia occidentalis Setch. | 2011 | 2011 | |||
| VER | Hippocampus kuda Bleeker, 1852 | 2014 | 2014 | |||
| INV | Hippopodina feegeensis (Busk, 1884) | 1996 | 1996 | |||
| VER | Holacanthus africanus Cadenat, 1951 | 2017 | 2018 | 2017 | ||
| VER | Holacanthus ciliaris (Linnaeus, 1758) | 2011 | 2011 | |||
| VER | Holocentrus adscensionis (Osbeck, 1765) | 2016 | 2016 | |||
| INV | Homarus americanus H. Milne Edwards, 1837 | 1961 | 2007 | 1961 | 2018 | |
| VER | Huso huso (Linnaeus, 1758)* | 1962 | 1962 | |||
| PP | Hydroclathrus tilesii (Endlicher) Santiañez & M.J.Wynne | 2006 | 2006 | |||
| INV | Hydroides brachyacantha Rioja, 1941 | 2015 | 2015 | |||
| INV | Hydroides dirampha Mörch, 1863 | 1981 | 1982 | 1981 | ||
| INV | Hydroides elegans (Haswell, 1883) | 1868 | 1973 | 1868 | ||
| INV | Hydroides ezoensis Okuda, 1934 | 1968 | 1968 | |||
| INV | Hydroides heterocera (Grube, 1868) | 1998 | 1998 | |||
| INV | Hymeniacidon gracilis (Hentschel, 1912) | 2017 | 2017 | |||
| INV | Hypania invalida (Grube, 1860) | 1995 | 1995 | |||
| INV | Hypereteone heteropoda (Hartman, 1951) | 2017 | 2017 | |||
| PP | Hypnea musciformis (Wulfen) J.V.Lamouroux | 2005 | 2005 | |||
| PP | Hypnea anastomosans Papenfuss, Lipkin & P.C.Silva | 2008 | 2008 | |||
| PP | Hypnea cervicornis J.Agardh | 2009 | 2009 | |||
| PP | Hypnea cornuta (Kützing) J.Agardh | 1894 | 1894 | |||
| PP | Hypnea spinella (C.Agardh) Kützing | 1977 | 1977 | |||
| PP | Hypnea valentiae (Turner) Montagne | 1996 | 2006 | 1996 | ||
| INV | Hypselodoris infucata (Rüppell & Leuckart, 1830) | 2002 | 2002 | |||
| INV | Ianiropsis serricaudis Gurjanova, 1936 | 2000 | 2000 | 2012 | ||
| INV | Incisocalliope aestuarius (Watling & Maurer, 1973) | 1975 | 1975 | |||
| INV | Indothais lacera (Born, 1778) | 1983 | 1983 | |||
| INV | Isognomon aff. australicus (Reeve, 1858) | 2016 | 2016 | |||
| INV | Isognomon legumen (Gmelin, 1791) | 2016 | 2016 | |||
| INV | Isognomon radiatus (Anton, 1838) | 1996 | 1996 | |||
| INV | Isolda pulchella Müller in Grube, 1858 | 1994 | 1994 | |||
| INV | Ixa monodi Holthuis & Gottlieb, 1956 | 1999 | 1999 | |||
| INV | Jasus lalandii (H. Milne Edwards, 1837) | 1980 | 1980 | |||
| PP | Kapraunia schneideri (Stuercke & Freshwater) A.M.Savoie & G.W.Saunders | 2010 | 2010 | 2016 | ||
| PP/micro | Karenia longicanalis Z.B.Yang, I.J.Hodgkiss & Gerd Hansen | 2008 | 2008 | |||
| PP/micro | Karenia mikimotoi (Miyake & Kominami ex Oda) Gert Hansen & Ø.Moestrup | 1968 | 1980 | 1968 | ||
| PP/micro | Karenia papilionacea A.J.Haywood & K.A.Steidinger | 1994 | 1994 | |||
| INV | Koinostylochus ostreophagus (Hyman, 1955) | 1970 | 1970 | |||
| Pathogen | Labyrinthula zosterae D. Porter & Muehlst. in Muehlstein & Short | 1930 | 1930 | |||
| VER | Lactophrys triqueter (Linnaeus, 1758) | 1909 | 1909 | |||
| VER | Lagocephalus guentheri Miranda Ribeiro, 1915 | 1952 | 1952 | |||
| VER | Lagocephalus sceleratus (Gmelin, 1789) | 2004 | 2004 | |||
| VER | Lagocephalus suezensis Clark & Gohar, 1953 | 2003 | 2003 | |||
| INV | Lamprohaminoea ovalis (Pease, 1868) | 2001 | 2001 | |||
| INV | Laonome xeprovala Bick & Bastrop, in Bick et al., 2018 | 2012 | 2012 | 2016 | 2018 | |
| INV | Latopilumnus malardi (De Man, 1914) | 1910 | 1910 | |||
| PP/micro | Lauderia pumila Castracane | 1995 | 1995 | |||
| PP | Laurencia brongniartii J.Agardh | 1989 | 1989 | |||
| PP | Laurencia caduciramulosa Masuda & Kawaguchi | 1991 | 1991 | |||
| PP | Laurencia okamurae Yamada | 1984 | 1984 | |||
| PP | Leathesia marina (Lyngbye) Decaisne | 1905 | 1905 | |||
| INV | Leiocapitellides analis Hartmann-Schröder, 1960 | 2000 | 2000 | |||
| INV | Leiochrides australis Augener, 1914 | 2002 | 2002 | |||
| PP/micro | Lennoxia faveolata H.A.Thomsen & K.R.Buck | 2007 | 2007 | |||
| INV | Leonnates persicus Wesenberg-Lund, 1949 | 2013 | 2013 | |||
| INV | Lepidonotus tenuisetosus (Gravier, 1902) | 2007 | 2007 | |||
| INV | Lepidonotus carinulatus (Grube, 1870) | 1984 | 1984 | |||
| INV | Leucotina natalensis E. A. Smith, 1910 | 1996 | 1996 | |||
| INV | Limnodrilus profundicola (Verrill, 1871) | 2014 | 2014 | |||
| INV | Limulus polyphemus (Linnaeus, 1758) | 1866 | 1866 | |||
| INV | Linguimaera caesaris Krapp-Schickel, 2003 | 1997 | 1997 | |||
| INV | Linopherus canariensis Langerhans, 1881 | 1997 | 1997 | |||
| INV | Lioberus ligneus (Reeve, 1858) | 2019 | 2019 | |||
| PP | Lithophyllum yessoense Foslie | 1994 | 1994 | |||
| PP | Lomentaria flaccida Tanaka | 2002 | 2002 | |||
| PP | Lomentaria hakodatensis Yendo | 1978 | 1984 | 1978 | ||
| PP | Lophocladia lallemandii (Montagne) F.Schmitz | 1908 | 1908 | |||
| INV | Lottia sp. | 2015 | 2015 | |||
| INV | Lovenella assimilis (Browne, 1905) | 2007 | 2007 | |||
| INV | Lumbrinerides crassicephala (Hartman, 1965) | 1994 | 1994 | |||
| INV | Lumbrinerides neogesae Miura, 1981 | 2002 | 2002 | |||
| INV | Lumbrineris perkinsi Carrera-Parra, 2001 | 1973 | 1973 | |||
| VER | Lutjanus argentimaculatus (Forsskål, 1775) | 2019 | 2019 | |||
| VER | Lutjanus griseus (Linnaeus, 1758) | 2018 | 2018 | |||
| VER | Lutjanus jocu (Bloch & Schneider, 1801) | 2005 | 2005 | |||
| VER | Lutjanus sebae (Cuvier, 1816) | 2010 | 2010 | |||
| VER | Lutjanus fulviflamma (Forsskål, 1775) | 2013 | 2013 | |||
| INV | Lysidice collaris Grube, 1870 | 1961 | 1961 | |||
| PP | Macrocystis pyrifera (Linnaeus) C.Agardh | 1972 | 1972 | |||
| INV | Macromedaeus voeltzkowi (Lenz, 1905) | 1910 | 1910 | |||
| INV | Macrophthalmus (Macrophthalmus) indicus Davie, 2012 | 2009 | 2009 | |||
| INV | Macrorhynchia philippina Kirchenpauer, 1872 | 1982 | 1982 | |||
| INV | Magallana angulata (Lamarck, 1819) | 1700 | 1700 | |||
| INV | Magallana gigas (Thunberg, 1793) | 1700 | 2019 | 1700 | 1850 | 2010 |
| INV | Magallana rivularis (Gould, 1861) | 1994 | 1994 | |||
| INV | Magallana sikamea (Amemiya, 1928) | 1994 | 1994 | |||
| INV | Malleus regula (Forsskål in Niebuhr, 1775) | 1970 | 1970 | |||
| INV | Marenzelleria arctia (Chamberlin, 1920) | 2004 | 2004 | |||
| INV | Marenzelleria neglecta Sikorski & Bick, 2004 | 1983 | 1983 | 1985 | ||
| INV | Marenzelleria viridis (Verrill, 1873) | 1983 | 1985 | 1983 | ||
| INV | Marginella glabella (Linnaeus, 1758) | 2009 | 2009 | |||
| INV | Maritigrella fuscopunctata (Prudhoe, 1978) | 2014 | 2014 | |||
| INV | Marivagia stellata Galil & Gershwin, 2010 | 2019 | 2019 | |||
| INV | Marphysa victori Lavesque, Daffe, Bonifácio & Hutchings, 2017 | 1975 | 1975 | |||
| Pathogen | Marteilia refringens Grizel, Comps, Bonami, Cousserans, Duthoit & Le Pennec | 1975 | 1975 | 1992 | ||
| INV | Matuta victor (J.C. Fabricius, 1781) | 2018 | 2018 | |||
| PP/micro | Mediopyxis helysia Kühn, Hargreaves & Halliger | 2003 | 2003 | |||
| INV | Megabalanus tintinnabulum (Linnaeus, 1758) | 1764 | 1764 | 1971 | ||
| INV | Megabalanus coccopoma (Darwin, 1854) | 1851 | 1851 | |||
| INV | Melanella orientalis Agamennone, Micali & Siragusa, 2020 | 2016 | 2016 | |||
| PP | Melanothamnus flavimarinus (M.-S.Kim & I.K.Lee) Díaz-Tapia & Maggs | 2010 | 2010 | |||
| PP | Melanothamnus harveyi (Bailey) Díaz-Tapia & Maggs | 1958 | 1982 | 2015 | 1958 | |
| PP | Melanothamnus japonicus (Harvey) Díaz-Tapia & Maggs | 2016 | 2016 | |||
| INV | Melibe viridis (Kelaart, 1858) | 1970 | 1970 | |||
| INV | Melita nitida S.I. Smith in Verrill, 1873 | 1996 | 2010 | 1996 | ||
| INV | Menaethius monoceros (Latreille, 1825) | 1978 | 1978 | |||
| INV | Mercenaria mercenaria (Linnaeus, 1758) | 1861 | 1861 | 1964 | ||
| INV | Mesanthura cfr. romulea Poore & Lew Ton, 1986 | 2000 | 2000 | |||
| INV | Metacalanus acutioperculum Ohtsuka, 1984 | 1995 | 1995 | |||
| INV | Metacirolana rotunda (Bruce & Jones, 1978) | 1998 | 1998 | |||
| INV | Metapenaeopsis aegyptia Galil & Golani, 1990 | 1996 | 1996 | |||
| INV | Metapenaeopsis mogiensis consobrina (Nobili, 1904) | 1995 | 1995 | |||
| INV | Metapenaeus monoceros (Fabricius, 1798) | 1961 | 1961 | |||
| INV | Metaxia bacillum (Issel, 1869) | 1995 | 1995 | |||
| INV | Microcosmus anchylodeirus Traustedt, 1883 | 1980 | 1980 | |||
| INV | Microcosmus squamiger Michaelsen, 1927 | 1971 | 1992 | 1971 | ||
| INV | Microcosmus exasperatus Heller, 1878 | 2005 | 2005 | 2014 | ||
| VER | Micropogonias undulatus (Linnaeus, 1766) | 1998 | 1998 | |||
| PP | Miliolinella fichteliana (d’Orbigny, 1839) | 1911 | 1911 | |||
| INV | Millepora alcicornis Linnaeus, 1758 | 2004 | 2004 | |||
| PP | Mimosina affinis Millett, 1900 | 2012 | 2012 | |||
| INV | Mitrella psilla (Duclos, 1846) | 2016 | 2016 | |||
| INV | Mizuhopecten yessoensis (Jay, 1857) | 1979 | 1979 | |||
| INV | Mnemiopsis leidyi A. Agassiz, 1865 | 1986 | 2006 | 2001 | 1990 | 1986 |
| INV | Mnestia girardi (Audouin, 1826) | 1990 | 1990 | |||
| INV | Moerisia inkermanica Paltschikowa-Ostroumowa | 1959 | 2018 | 1959 | ||
| INV | Molgula occidentalis Traustedt, 1883 | 2010 | 2010 | |||
| INV | Monocorophium uenoi (Stephensen, 1932) | 2007 | 2007 | |||
| VER | Morone saxatilis x Morone chrysops | 2019 | 2019 | |||
| INV | Mulinia lateralis (Say, 1822) | 2017 | 2017 | |||
| INV | Murchisonellidae T. L. Casey, 1904 | 2013 | 2013 | |||
| INV | Mycale (Carmia) senegalensis Lévi, 1952 | 2002 | 2002 | |||
| VER | Mycteroperca tigris (Valenciennes, 1833) | 2018 | 2018 | |||
| INV/par | Myicola ostreae Hoshina & Sugiura, 1953 | 1972 | 1972 | 1972 | ||
| INV | Myra subgranulata Kossmann, 1877 | 2004 | 2004 | |||
| INV/par | Mytilicola orientalis Mori, 1935 | 1977 | 2018 | 1977 | 1977 | |
| INV | Mytilopsis leucophaeata (Conrad, 1831) | 1835 | 1928 | 1835 | ||
| INV | Naineris setosa (Verrill, 1900) | 2010 | 2010 | |||
| INV | Namanereis littoralis (Grube, 1872) | 1991 | 1991 | |||
| INV | Neanthes agulhana (Day, 1963) | 2007 | 2007 | |||
| PP | Nemalion vermiculare Suringar | 2005 | 2005 | |||
| VER | Nemipterus randalli Russell, 1986 | 2014 | 2014 | |||
| INV | Nemopsis bachei L. Agassiz, 1849 | 1905 | 1905 | |||
| INV | Neodexiospira brasiliensis (Grube, 1872) | 1982 | 1982 | |||
| PP | Neogastroclonium subarticulatum (Turner) L.Le Gall, Dalen & G.W.Saunders | 2017 | 2017 | |||
| VER | Neogobius melanostomus (Pallas, 1814) | 1990 | 1990 | 2004 | ||
| PP | Neoizziella divaricata (C.K.Tseng) S.-M.Lin, S.-Y.Yang & Huisman | 1989 | 1989 | |||
| INV | Neomysis americana (S.I. Smith, 1873) | 2010 | 2010 | |||
| INV | Nereis jacksoni Kinberg, 1865 | 1964 | 1964 | |||
| INV | Nerita sanguinolenta Menke, 1829 | 1969 | 1969 | |||
| INV | Nippoleucon hinumensis (Gamô, 1967) | 2019 | 2019 | |||
| PP | Nitophyllum stellato-corticatum Okamura | 1984 | 1984 | |||
| PP | Nonionella sp. T1/Nonionella stella | 2012 | 2012 | |||
| INV | Notocochlis gualtieriana (Récluz, 1844) | 1978 | 1978 | |||
| INV | Notomastus aberans Day, 1957 | 1964 | 1964 | |||
| INV | Notomastus mossambicus (Thomassin, 1970) | 1997 | 1997 | |||
| INV | Novafabricia infratorquata (Fitzhugh, 1973) | 1985 | 2013 | 1985 | ||
| INV/par | Nybelinia africana Dollfus, 1960 | 2005 | 2005 | |||
| INV | Obesogammarus crassus (Sars G.O., 1894)* | 1962 | 1962 | 2016 | ||
| INV | Ocinebrellus inornatus (Récluz, 1851) | 1993 | 1993 | |||
| INV | Odontodactylus scyllarus (Linnaeus, 1758) | 2009 | 2009 | |||
| INV | Oithona davisae Ferrari F.D. & Orsi, 1984 | 2000 | 2002 | 2000 | 2009 | |
| VER | Oncorhynchus gorbuscha (Walbaum, 1792) | 1958 | 1958 | 1958 | ||
| VER | Oncorhynchus kisutch (Walbaum, 1792)* | 1905 | 1984 | 1905 | ||
| VER | Oncorhynchus mykiss (Walbaum, 1792)* | 1882 | 1882 | 1899 | ||
| PP | Operculina ammonoides (Gronovius, 1781) | 1911 | 1911 | |||
| INV | Ophiactis macrolepidota Marktanner-Turneretscher, 1887 | 1998 | 1998 | |||
| INV | Ophiactis savignyi (Müller & Troschel, 1842) | 1968 | 1968 | |||
| VER | Ophioblennius atlanticus (Valenciennes, 1836) | 2017 | 2017 | |||
| INV | Ophryotrocha japonica Paxton & Åkesson, 2010 | 1999 | 1999 | |||
| INV | Ophryotrocha diadema Åkesson, 1976 | 2006 | 2006 | |||
| VER | Oplegnathus fasciatus (Temminck & Schlegel, 1844) | 2009 | 2009 | |||
| VER | Orthopristis chrysoptera (Linnaeus, 1766) | 2020 | 2020 | |||
| INV | Oscilla galilae Bogi, Karhan & Yokeş, 2012 | 2016 | 2016 | |||
| VER | Ostorhinchus fasciatus (White, 1790) | 2014 | 2014 | |||
| Pathogen | Ostracoblabe implexa Born & Flahault | 1951 | 1951 | |||
| INV | Ostraea angasi G. B. Sowerby II, 1871 | 1985 | 1985 | |||
| INV | Ostrea equestris Say, 1834 | 1995 | 1995 | |||
| INV | Ostrea denselamellosa Lischke, 1869 | 1982 | 1982 | |||
| INV | Ostrea puelchana d’Orbigny, 1842 | 1989 | 1989 | |||
| INV | Oulastrea crispata (Lamarck, 1816) | 2012 | 2012 | |||
| INV | Oxydromus humesi (Pettibone, 1961) | 2009 | 2009 | |||
| PP/micro | Oxytoxum criophilum Balech | 2003 | 2003 | |||
| VER | Oxyurichthys papuensis (Valenciennes, 1837) | 2010 | 2010 | |||
| INV | Pachygrapsus gracilis (de Saussure, 1857) | 2013 | 2013 | |||
| PP | Pachymeniopsis gargiuli S.Y.Kim, Manghisi, Morabito & S.M.Boo | 1968 | 2001 | 1968 | ||
| PP | Pachymeniopsis lanceolata (K.Okamura) Y.Yamada ex S.Kawabata | 1982 | 2006 | 1982 | ||
| INV | Pacifastacus leniusculus (Dana, 1852) | 2014 | 2014 | |||
| INV | Pacificincola perforata (Okada & Mawatari, 1937) | 2001 | 2001 | |||
| PP | Padina boergesenii Allender & Kraft | 1965 | 1965 | |||
| VER | Pagrus major (Temminck & Schlegel, 1843) | 2004 | 2004 | |||
| INV | Pagurus longicarpus (Say, 1817) | 2020 | 2020 | |||
| INV | Palaemon macrodactylus Rathbun, 1902 | 1998 | 2014 | 1998 | 2005 | 2002 |
| INV | Palola valida (Gravier, 1900) | 2014 | 2014 | |||
| VER | Pampus argenteus (Euphrasen, 1788) | 1896 | 1896 | |||
| INV | Panopeus occidentalis Saussure, 1857 | 2015 | 2015 | |||
| PP | Papenfussiella kuromo (Yendo) Inagaki | 1990 | 1990 | |||
| INV | Paracalanus quasimodo Bowman, 1971 | 2017 | 2017 | |||
| INV | Paracaprella pusilla Mayer, 1890 | 2010 | 2010 | 2011 | ||
| INV | Paracerceis sculpta (Holmes, 1904) | 1981 | 1988 | 1981 | ||
| INV | Paradella dianae (Menzies, 1962) | 1985 | 1988 | 1985 | ||
| INV | Paradyte cf. crinoidicola (Potts, 1910) | 1968 | 1968 | |||
| INV | Paraleucilla magna Klautau, Monteiro & Borojevic, 2004 | 2000 | 2006 | 2000 | ||
| INV | Paralithodes camtschaticus (Tilesius, 1815) | 2008 | 2008 | |||
| INV | Parametopella cypris Holmes, 1905 | 2014 | 2014 | |||
| INV | Paramysis (Mesomysis) intermedia (Czerniavsky, 1882) | 2008 | 2008 | |||
| INV | Paramysis (Serrapalpisis) lacustris (Czerniavsky, 1882) | 1962 | 1962 | |||
| INV | Paranais frici Hrabĕ, 1941 | 1970 | 1970 | |||
| VER | Paranthias furcifer (Valenciennes, 1828) | 2011 | 2014 | 2011 | ||
| INV | Paranthura japonica Richardson, 1909 | 2005 | 2007 | 2005 | ||
| INV | Parasmittina alba Ramalho, Muricy & Taylor, 2011 | 2014 | 2014 | |||
| INV | Parasmittina multiaviculata Souto, Ramalhosa & Canning-Clode, 2016 | 2016 | 2016 | |||
| INV | Parasmittina egyptiaca (Waters, 1909) | 2016 | 2016 | |||
| PP | Parasorites orbitolitoides Hofker, 1930 | 2016 | 2016 | |||
| INV | Paratapes textilis (Gmelin, 1791) | 2004 | 2004 | |||
| INV/par | Paratenuisentis ambiguus (Van Cleave, 1921) | 2001 | 2001 | |||
| VER | Parexocoetus mento (Valenciennes, 1847) | 1955 | 1955 | |||
| VER | Parupeneus forsskali (Fourmanoir & Guézé, 1976) | 2014 | 2014 | |||
| INV | Parvocalanus crassirostris (Dahl F., 1894) | 2009 | 2009 | |||
| PP | Pegidia lacunata McCulloch, 1977 | 2010 | 2010 | |||
| VER | Pempheris rhomboidea Kossmann & Räuber, 1877 | 1983 | 1983 | |||
| INV | Penaeus aztecus Ives, 1891 | 2012 | 2018 | 2012 | ||
| INV | Penaeus hathor (Burkenroad, 1959) | 2012 | 2012 | |||
| INV | Penaeus monodon Fabricius, 1798 | 2011 | 2011 | |||
| INV | Penaeus japonicus Spence Bate, 1888 | 1972 | 1980 | 1972 | ||
| INV | Penaeus pulchricaudatus Stebbing, 1914 | 1961 | 1982 | 1961 | ||
| INV | Penaeus semisulcatus De Haan, 1844 [in De Haan, 1833–1850] | 2016 | 2016 | |||
| PP/micro | Peridiniella catenata (Levander) Balech | 1987 | 1987 | |||
| PP/micro | Peridiniella danica (Paulsen) Y.B.Okolodkov & J.D.Dodge | 1901 | 1901 | |||
| PP/micro | Peridinium quadridentatum (F.Stein) Gert Hansen | 2005 | 2008 | 2005 | ||
| INV | Perinereis linea (Treadwell, 1936) | 2012 | 2012 | |||
| Pathogen | Perkinsus chesapeaki McLaughlin, Tall, Shaheen, El Sayed & Faisal | 1992 | 1992 | 1992 | ||
| Pathogen | Perkinsus olsenii R.J.G.Lester & G.H.G.Davis | 1983 | 1983 | |||
| INV | Perophora multiclathrata (Sluiter, 1904) | 1983 | 1983 | |||
| INV | Perophora viridis Verrill, 1871 | 1971 | 1971 | |||
| INV | Perophora japonica Oka, 1927 | 1982 | 1982 | |||
| PP | Petalonia binghamiseae (J.Agardh) K.L.Vinogradova | 1985 | 1985 | |||
| INV | Petricolaria pholadiformis (Lamarck, 1818) | 1896 | 1927 | 1896 | 1985 | |
| VER | Petroscirtes ancylodon Rüppell, 1835 | 2004 | 2004 | |||
| INV | Phallusia nigra Savigny, 1816 | 2008 | 2008 | |||
| INV | Phascolion convestitum Sluiter, 1902 | 1977 | 1977 | |||
| INV | Phascolosoma (Phascolosoma) scolops (Selenka & de Man, 1883) | 1975 | 1975 | |||
| INV | Photis lamellifera Schellenberg, 1928 | 1990 | 1990 | |||
| Pathogen | Photobacterium damsela Love, Teebken-Fisher, Hose, Farmer III, Hickman & Fanning | 1992 | 1992 | |||
| PP | Phrix spatulata (E.Y.Dawson) M.J.Wynne, M.Kamiya & J.A.West | 1992 | 1992 | |||
| INV | Phyllorhiza punctata Lendenfeld, 1884 | 2005 | 2018 | 2005 | ||
| VER | Piaractus brachypomus (Cuvier, 1818) | 2013 | 2013 | |||
| PP | Pikea californica Harvey | 1991 | 1991 | |||
| INV | Pileolaria berkeleyana (Rioja, 1942) | 1977 | 2007 | 1977 | ||
| INV | Pilumnopeus africanus (de Man, 1902) | 2013 | 2013 | |||
| INV | Pilumnopeus vauquelini (Audouin, 1826) | 1963 | 1963 | |||
| INV | Pilumnus minutus De Haan, 1835 [in De Haan, 1833–1850] | 2017 | 2017 | |||
| INV | Pinctada fucata (A. Gould, 1850) | 2018 | 2018 | |||
| INV | Pinctada radiata (Leach, 1814) | 1899 | 1998 | 1899 | ||
| VER | Pinguipes brasilianus Cuvier, 1829 | 1990 | 1990 | |||
| INV/par | Piscicola pojmanskae Bielecki, 1994 | 2008 | 2008 | |||
| INV | Pista unibranchia Day, 1963 | 1997 | 2005 | 1997 | ||
| INV | Plagusia squamosa (Herbst, 1790) | 1906 | 1906 | |||
| VER | Planiliza haematocheila (Temminck & Schlegel, 1845) | 1972 | 1995 | 1972 | ||
| PP | Planispirinella exigua (Brady, 1879) | 1910 | 1910 | |||
| PP | Planogypsina acervalis (Brady, 1884) | 1909 | 1909 | |||
| VER | Platycephalus indicus (Linnaeus, 1758) | 1978 | 1978 | |||
| PP | Plocamium secundatum (Kützing) Kützing | 1991 | 1991 | |||
| INV | Plocamopherus ocellatus Rüppell & Leuckart, 1828 | 2015 | 2015 | |||
| VER | Poecilopsetta beanii (Goode, 1881) | 1995 | 1995 | |||
| INV | Polyandrocarpa zorritensis (Van Name, 1931) | 1974 | 1974 | |||
| INV | Polycera hedgpethi Er. Marcus, 1964 | 1986 | 2001 | 1986 | ||
| INV | Polycerella emertoni A. E. Verrill, 1880 | 1964 | 1981 | 1964 | ||
| INV | Polycirrus twisti Potts, 1928 | 1983 | 1983 | |||
| INV | Polyclinum constellatum Savigny, 1816 | 2014 | 2014 | |||
| INV | Polydora colonia Moore, 1907 | 1983 | 2018 | 1983 | ||
| INV | Polydora triglanda Radashevsky & Hsieh, 2000 | 2014 | 2014 | |||
| INV | Polydora websteri Hartman in Loosanoff & Engle, 1943 | 2014 | 2014 | |||
| PP | Polyopes lancifolius (Harvey) Kawaguchi & Wang | 2008 | 2008 | |||
| PP | Polysiphonia paniculata Montagne | 1967 | 1967 | |||
| PP | Polysiphonia forfex Harvey | 2011 | 2011 | |||
| PP | Polysiphonia morrowii Harvey | 1975 | 1975 | 1997 | ||
| PP | Polysiphonia senticulosa Harvey | 1993 | 1993 | |||
| VER | Pomacanthus imperator (Bloch, 1787) | 2016 | 2016 | |||
| VER | Pomacanthus paru (Bloch, 1787) | 2015 | 2015 | |||
| VER | Pomacanthus maculosus (Forsskål, 1775) | 1994 | 1994 | 2012 | ||
| VER | Pomadasys stridens (Forsskål, 1775) | 1968 | 1968 | |||
| INV | Pontogammarus robustoides (Sars, 1894)* | 1962 | 1962 | |||
| PP | Porphyra umbilicalis Kützing | 1989 | 1989 | |||
| INV | Portunus segnis (Forskål, 1775) | 1958 | 1958 | |||
| INV | Potamocorbula amurensis (Schrenck, 1862) | 2018 | 2018 | |||
| INV | Potamopyrgus antipodarum (Gray, 1843)* | 1801 | 1801 | 1887 | ||
| INV | Potamothrix moldaviensis Vejdovský & Mrázek, 1903 | 2008 | 2008 | |||
| INV | Potamothrix bavaricus (Oschmann, 1913) | 2015 | 2015 | |||
| INV | Potamothrix bedoti (Piguet, 1913) | 1915 | 1915 | |||
| INV | Potamothrix heuscheri (Bretscher, 1900)* | 1960 | 1960 | |||
| INV | Potamothrix vejdovskyi (Hrabĕ, 1941)* | 1967 | 1967 | |||
| inv | Prionospio aluta Maciolek, 1985 | 1994 | 1994 | |||
| INV | Prionospio depauperata Imajima, 1990 | 2018 | 2018 | |||
| INV | Prionospio pulchra Imajima, 1990 | 1989 | 1989 | 1991 | ||
| INV | Proasellus coxalis (Dollfus, 1892) | 2011 | 2011 | |||
| INV | Procambarus clarkii (Girard, 1852)* | 2000 | 2000 | |||
| INV | Prokelisia marginata (Van Duzee, 1897) | 2011 | 2011 | |||
| PP/micro | Prorocentrum gracile Schütt | 1989 | 1989 | |||
| INV | Prosphaerosyllis longipapillata (Hartmann-Schröder, 1979) | 1997 | 1997 | |||
| VER | Proterorhinus nasalis (De Filippi, 1863) | 2020 | 2020 | |||
| INV | Protodorvillea biarticulata Day, 1963 | 1975 | 1975 | |||
| INV | Protoreaster nodosus (Linnaeus, 1758) | 1981 | 1981 | |||
| INV | Psammacoma gubernaculum (Hanley, 1844) | 2009 | 2009 | |||
| PP/micro | Pseudochattonella farcimen (Riisberg I.) | 1998 | 2001 | 1998 | ||
| PP/micro | Pseudochattonella verruculosa (Y.Hara & M.Chihara) S.Tanabe-Hosoi, D.Honda, S.Fukaya, Y.Inagaki & Y.Sako | 1998 | 2015 | 1998 | ||
| INV/par | Pseudodactylogyrus anguillae (Yin & Sproston, 1948) | 1982 | 1985 | 1982 | ||
| INV/par | Pseudodactylogyrus bini (Kikuchi, 1929) | 1985 | 1985 | 1997 | ||
| INV | Pseudodiaptomus marinus Sato, 1913 | 2007 | 2010 | 2007 | ||
| INV | Pseudonereis anomala Gravier, 1899 | 1969 | 1969 | |||
| PP/micro | Pseudo-nitzschia australis Frenguelli | 1995 | 1995 | 2000 | ||
| PP/micro | Pseudo-nitzschia multistriata (Takano) Takano | 1985 | 1985 | |||
| INV | Pseudopolydora paucibranchiata (Okuda, 1937) | 1977 | 1982 | 1977 | ||
| VER | Pteragogus trispilus Randall, 2013 | 1992 | 1992 | |||
| VER | Pterois miles (Bennett, 1828) | 2009 | 2009 | |||
| INV | Ptilohyale littoralis (Stimpson, 1853) | 2009 | 2009 | |||
| INV | Purpuradusta gracilis notata (Gill, 1858) | 1988 | 1988 | |||
| INV | Pyrgulina pupaeformis (Souverbie, 1865) | 1995 | 1995 | |||
| INV | Pyromaia tuberculata (Lockington, 1877) | 2016 | 2016 | |||
| PP | Pyropia yezoensis (Ueda) M.S.Hwang & H.G.Choi | 1975 | 1984 | 1975 | ||
| PP | Pyropia suborbiculata (Kjellman) J.E.Sutherland, H.G.Choi, M.S.Hwang & W.A.Nelson | 2010 | 2010 | 2014 | ||
| INV | Pyrunculus fourierii (Audouin, 1826) | 1995 | 1995 | |||
| INV | Rangia cuneata (G. B. Sowerby I, 1832) | 1997 | 2011 | 1997 | ||
| INV | Rapana venosa (Valenciennes, 1846) | 1956 | 1997 | 1973 | 1956 | |
| VER | Rastrelliger kanagurta (Cuvier, 1816) | 2018 | 2018 | |||
| INV | Rhinoclavis kochi (Philippi, 1848) | 1976 | 1976 | |||
| INV | Rhithropanopeus harrisii (Gould, 1841) | 1936 | 1936 | 1950 | 1994 | 1948 |
| PP/micro | Rhizosolenia calcar-avis Schultze | 2009 | 2009 | |||
| INV | Rhopilema nomadica Galil, 1990 | 1995 | 1995 | |||
| INV | Ringicula minuta H. Adams, 1872 | 2019 | 2019 | |||
| INV | Rissoina bertholleti Issel, 1869 | 1985 | 1985 | |||
| INV | Ruditapes philippinarum (Adams & Reeve, 1850) | 1973 | 1973 | 1980 | ||
| PP | Rugulopteryx okamurae (E.Y.Dawson) I.K.Hwang, W.J.Lee & H.S.Kim | 2002 | 2015 | 2002 | ||
| PP | Saccharina japonica (J.E. Areschoug) C.E.Lane, C.Mayes, Druehl & G.W.Saunders | 1976 | 1980 | 1976 | ||
| INV | Saccostrea cuccullata (Born, 1778) | 2007 | 2007 | |||
| INV | Saccostrea glomerata (Gould, 1850) | 1984 | 1984 | |||
| VER | Salvelinus fontinalis (Mitchill, 1814)* | 1916 | 1916 | |||
| PP | Sarconema filiforme (Sonder) Kylin | 1990 | 1990 | |||
| PP | Sarconema scinaioides Børgesen | 1980 | 1980 | |||
| PP | Sargassum muticum (Yendo) Fensholt | 1972 | 1972 | 1980 | ||
| VER | Sargocentron rubrum (Forsskål, 1775) | 1943 | 1943 | |||
| VER | Saurida lessepsianus Russell, Golani & Tikochinski, 2015 | 1960 | 1960 | |||
| PP | Scageliopsis patens Wollaston | 1989 | 1989 | |||
| VER | Scarus ghobban Forsskål, 1775 | 2010 | 2010 | |||
| VER | Scatophagus argus (Linnaeus, 1766) | 2007 | 2007 | |||
| INV | Schizoporella japonica Ortmann, 1890 | 1976 | 1976 | |||
| INV | Schizoporella pungens Canu & Bassler, 1928 | 2010 | 2010 | |||
| VER | Sciaenops ocellatus (Linnaeus, 1766) | 2016 | 2016 | |||
| INV | Scolelepis (Parascolelepis) gilchristi (Day, 1961) | 1977 | 1977 | |||
| INV | Scolelepis korsuni Sikorski, 1994 | 1994 | 1994 | |||
| INV | Scolionema suvaense (Agassiz & Mayer, 1899) | 1950 | 1950 | |||
| VER | Scomberomorus commerson (Lacepède, 1800) | 2008 | 2008 | |||
| INV | Scyllarus caparti Holthuis, 1952 | 1977 | 1977 | |||
| PP | Scytosiphon dotyi M.J.Wynne | 1968 | 1991 | 1968 | ||
| VER | Sebastes schlegelii Hilgendorf, 1880 | 2008 | 2008 | |||
| INV | Sebastiscus marmoratus (Cuvier, 1829) | 2016 | 2016 | |||
| INV | Sepioteuthis lessoniana Férussac [in Lesson], 1831 | 2009 | 2009 | |||
| INV | Septifer cumingii Récluz, 1848 | 2005 | 2005 | |||
| VER | Siganus fuscescens (Houttuyn, 1782) | 2020 | 2020 | |||
| VER | Siganus virgatus (Valenciennes, 1835) | 1975 | 1975 | |||
| VER | Siganus luridus (Rüppell, 1829) | 1964 | 1964 | |||
| VER | Siganus rivulatus Forsskål & Niebuhr, 1775 | 1925 | 1925 | |||
| PP | Sigmamiliolinella australis (Parr, 1932) | 2001 | 2001 | |||
| VER | Sillago suezensis Golani, Fricke & Tikochinski, 2013 | 2009 | 2009 | |||
| INV | Sinelobus vanhaareni Bamber, 2014 | 2006 | 2010 | 2006 | ||
| INV | Sinezona plicata (Hedley, 1899) | 2019 | 2019 | |||
| INV | Smaragdia souverbiana (Montrouzier in Souverbie & Montrouzier, 1863) | 1993 | 1993 | |||
| INV | Smittina nitidissima (Hincks, 1880) | 2014 | 2014 | |||
| INV | Smittoidea prolifica Osburn, 1952 | 1995 | 1995 | |||
| PP | Solieria filiformis (Kützing) P.W.Gabrielson | 1922 | 1922 | |||
| PP | Sorites variabilis Lacroix, 1941 | 1996 | 1996 | |||
| PP | Spartina anglica C.E. Hubbard | 1924 | 1924 | |||
| PP | Spartina densiflora Brongn. | 1986 | 1986 | |||
| PP | Spartina patens (Aiton) Muhl. | 1986 | 1986 | |||
| PP | Spartina alterniflora Loisel | 1806 | 1806 | |||
| PP | Spermothamnion cymosum (Harvey) De Toni | 2010 | 2010 | |||
| INV | Sphaeroma walkeri Stebbing, 1905 | 1977 | 2015 | 1977 | 2004 | |
| PP | Sphaerotrichia firma (E.S.Gepp) A.D.Zinova | 1981 | 1981 | |||
| INV | Sphaerozius nitidus Stimpson, 1858 | 2013 | 2013 | |||
| VER | Sphyraena chrysotaenia Klunzinger, 1884 | 1964 | 1964 | |||
| VER | Sphyraena flavicauda Rüppell, 1838 | 2003 | 2003 | |||
| INV | Spirobranchus tetraceros (Schmarda, 1861) | 1970 | 1970 | |||
| PP | Spiroloculina angulata Cushman, 1917 | 1996 | 1996 | |||
| PP | Spiroloculina antillarum d’Orbigny, 1839 | 1911 | 1911 | |||
| INV | Spirorbis (Spirorbis) marioni Caullery & Mesnil, 1897 | 1974 | 1974 | 1977 | ||
| INV | Spondylus spinosus Schreibers, 1793 | 2001 | 2001 | |||
| PP | Spongoclonium caribaeum (Børgesen) M.J.Wynne | 1967 | 1967 | 1974 | ||
| VER | Spratelloides delicatulus (Bennett, 1832) | 2014 | 2014 | |||
| VER | Stegastes variabilis (Castelnau, 1855) | 2014 | 2014 | |||
| INV | Stenothoe georgiana Bynum & Fox, 1977 | 2010 | 2011 | 2010 | ||
| VER | Stephanolepis diaspros Fraser-Brunner, 1940 | 1935 | 1935 | |||
| INV | Sternodromia spinirostris (Miers, 1881) | 1969 | 1969 | |||
| INV | Sticteulima lentiginosa (A. Adams, 1861) | 1995 | 1995 | |||
| INV | Stomatella sp. | 2011 | 2011 | |||
| INV | Streblospio gynobranchiata Rice & Levin, 1998 | 2011 | 2011 | |||
| INV | Streblospio benedicti Webster, 1879 | 1982 | 1982 | |||
| INV | Styela plicata (Lesueur, 1823) | 1877 | 1989 | 1877 | ||
| INV | Styela canopus (Savigny, 1816) | 2006 | 2006 | |||
| INV | Styela clava Herdman, 1881 | 1968 | 2017 | 1968 | 2005 | 2002 |
| PP | Stypopodium schimperi (Kützing) M.Verlaque & Boudouresque | 1990 | 1990 | |||
| INV | Syllis hyllebergi (Licher, 1999) | 1972 | 1972 | |||
| INV | Syllis pectinans Haswell, 1920 | 1982 | 1982 | 2013 | ||
| PP | Symphyocladia marchantioides (Harvey) Falkenberg | 1971 | 1971 | 1984 | ||
| PP | Symphyocladiella dendroidea (Montagne) D.Bustamante, B.Y.Won, S.C.Lindstrom & T.O.Cho | 1993 | 2005 | 1993 | ||
| INV | Symplegma rubra Monniot C., 1972 | 2014 | 2014 | |||
| INV | Symplegma brakenhielmi (Michaelsen, 1904) | 2003 | 2003 | |||
| VER | Synagrops japonicus (Döderlein, 1883) | 1987 | 1987 | |||
| INV | Synaptula reciprocans (Forsskål, 1775) | 1967 | 1967 | |||
| VER | Synchiropus sechellensis Regan, 1908 | 2014 | 2014 | |||
| INV | Synidotea laticauda Benedict, 1897 | 1975 | 1975 | |||
| INV | Syphonota geographica (A. Adams & Reeve, 1850) | 1999 | 1999 | |||
| INV | Syrnola fasciata Jickeli, 1882 | 1995 | 1995 | |||
| INV/par | Taeniastrotos sp. | 1993 | 1993 | |||
| PP/micro | Takayama tasmanica de Salas, Bolch & Hallegraeff | 2008 | 2008 | |||
| INV | Telmatogeton japonicus Tokunaga, 1933 | 1962 | 1962 | 1979 | ||
| INV | Tenellia adspersa (Nordmann, 1845) | 2001 | 2001 | |||
| VER | Terapon theraps (Cuvier, 1829) | 2007 | 2007 | |||
| INV | Terebella ehrenbergi Gravier, 1906 | 1952 | 1952 | |||
| INV | Teredo bartschi Clapp, 1923 | 2003 | 2003 | 2007 | ||
| INV | Thalamita gloriensis Crosnier, 1962 | 1977 | 1977 | |||
| INV | Thalamita poissonii (Audouin, 1826) | 1969 | 1969 | |||
| PP/micro | Thalassiosira nordenskioeldii Cleve | 1967 | 1967 | |||
| PP/micro | Thalassiosira hendeyi Hasle & G.Fryxell | 1978 | 1978 | |||
| PP/micro | Thalassiosira tealata Takano | 1968 | 1968 | |||
| PP/micro | Thecadinium yashimaense S.Yoshimatsu, S.Toriumi & J.D.Dodge | 2002 | 2002 | |||
| INV | Thelepus japonicus Marenzeller, 1884 | 2017 | 2017 | |||
| INV | Theora lubrica Gould, 1861 | 2001 | 2010 | 2001 | ||
| INV | Timarete punctata (Grube, 1859) | 2006 | 2006 | |||
| INV | Tonicia atrata (G.B. Sowerby II, 1840) | 1978 | 1978 | |||
| VER | Torquigener flavimaculosus Hardy & Randall, 1983 | 2006 | 2006 | |||
| INV | Trachysalambria palaestinensis (Steinitz, 1932) | 1995 | 1995 | |||
| INV | Tremoctopus gracilis (Souleyet, 1852) | 1937 | 1937 | |||
| INV | Tricellaria inopinata d’Hondt & Occhipinti Ambrogi, 1985 | 1982 | 1996 | 1982 | ||
| INV | Triconia rufa (Boxshall & Böttger, 1987) | 2004 | 2004 | |||
| INV | Triconia umerus (Böttger-Schnack & Boxshall, 1990) | 2004 | 2004 | |||
| INV | Tridentata marginata (Kirchenpauer, 1864) | 1980 | 1980 | 1990 | ||
| VER | Tridentiger barbatus (Günther, 1861) | 2016 | 2016 | |||
| PP/micro | Trieres mobiliensis (J.W.Bailey) Ashworth & Theriot | 1983 | 1983 | |||
| PP/micro | Trieres regia (M.Schultze) M.P.Ashworth & E.C.Theriot | 1989 | 1989 | |||
| VER | Trinectes maculatus (Bloch & Schneider, 1801) | 1984 | 1984 | |||
| PP/micro | Tripos arietinus (Cleve) F.Gómez | 1992 | 1992 | |||
| PP/micro | Tripos macroceros (Ehrenberg) F.Gómez | 1983 | 1983 | |||
| INV | Trochus erithreus Brocchi, 1821 | 1985 | 1985 | |||
| INV | Tubastraea tagusensis Wells, 1982 | 2017 | 2017 | |||
| INV | Turbonilla edgarii (Melvill, 1896) | 1996 | 1996 | |||
| VER | Tylerius spinosissimus (Regan, 1908) | 2004 | 2004 | |||
| VER | Tylosurus crocodilus crocodilus (Péron & Lesueur, 1821) | 2003 | 2003 | |||
| PP | Ulva australis Areschoug | 1984 | 1990 | 1984 | ||
| PP | Ulva californica Wille | 2006 | 2006 | 2011 | ||
| PP | Ulva gigantea (Kützing) Bliding | 2015 | 2015 | |||
| PP | Ulva ohnoi M.Hiraoka & S.Shimada | 2011 | 2015 | 2011 | ||
| PP | Ulvaria obscura (Kützing) P.Gayral ex C.Bliding | 1985 | 1985 | |||
| PP | Umbraulva dangeardii M.J.Wynne & G.Furnari | 2014 | 2014 | |||
| PP | Undaria pinnatifida (Harvey) Suringar | 1971 | 1975 | 1971 | ||
| PP | Undella hadai Balech | 2004 | 2004 | |||
| VER | Upeneus moluccensis (Bleeker, 1855) | 1947 | 1947 | |||
| VER | Upeneus pori Ben-Tuvia & Golani, 1989 | 2003 | 2003 | |||
| INV | Urocaridella pulchella Yokes & Galil, 2006 | 2018 | 2018 | |||
| PP | Uronema marinum Womersley | 1989 | 1989 | |||
| INV | Urosalpinx cinerea (Say, 1822) | 1960 | 1960 | |||
| INV | Vallicula multiformis Rankin, 1956 | 1998 | 1998 | |||
| VER | Vanderhorstia mertensi Klausewitz, 1974 | 2019 | 2019 | |||
| VER | Variola louti (Forsskål, 1775) | 2018 | 2018 | |||
| PP | Vaucheria longicaulis Hoppaugh | 2020 | 2020 | |||
| INV | Viriola sp.[cf. bayani] Jousseaume, 1884 | 2016 | 2016 | |||
| INV | Watersipora aterrima (Ortmann, 1890) | 1983 | 1983 | |||
| INV | Watersipora subatra (Ortmann, 1890) | 1987 | 1987 | |||
| INV | Watersipora arcuata Banta, 1969 | 1990 | 1990 | 2013 | ||
| PP | Womersleyella setacea (Hollenberg) R.E.Norris | 1986 | 1986 | |||
| INV | Xanthias lamarckii (H. Milne Edwards, 1834) | 2013 | 2013 | |||
| INV | Xenostrobus securis (Lamarck, 1819) | 1991 | 2005 | 1991 | ||
| INV | Yoldia limatula (Say, 1831) | 2019 | 2019 | |||
| INV | Zafra savignyi (Moazzo, 1939) | 1995 | 1995 | |||
| INV | Zafra selasphora (Melvill & Standen, 1901) | 1995 | 1995 | |||
| VER | Zebrasoma flavescens (Bennett, 1828) | 2008 | 2008 | |||
| VER | Zebrasoma xanthurum (Blyth, 1852) | 2015 | 2015 |
The Baltic Sea dataset encompasses 100 NIS introductions (including 6 parasites and 9 microalgae), 34 of which were introduced before 1970. The major proportion of the introductions since 1970 have been invertebrates (42 species, ~83%), followed by primary producers (5 species, ~10%), and vertebrates (4 species, ~8%). Invertebrates consist of a wide range of benthic crustaceans, as well as pelagic zooplanktonic taxa, whereas primary producers include both, phytoplankton, and phytobenthic species. Vertebrate species include Ponto-Caspian sturgeons and gobies, as well as cultured salmonids.
456 NIS are known from the North-East Atlantic (NEA), 372 of which have been detected since 1970 (81%). The Greater North Sea (ANS) hosts 260 NIS including parasites and pathogens (Figure 4b), 193 of which (74%) have been observed since 1970. The NIS biota is dominated by invertebrates (154 taxa = 59%) and primary producers (macroalgae, microalgae, pathogens) 88 taxa (34%). The proportion of vertebrates (fish) is low (18 taxa = 7%), and mostly related to freshwater NIS expanding their distribution into estuarine coastal waters.
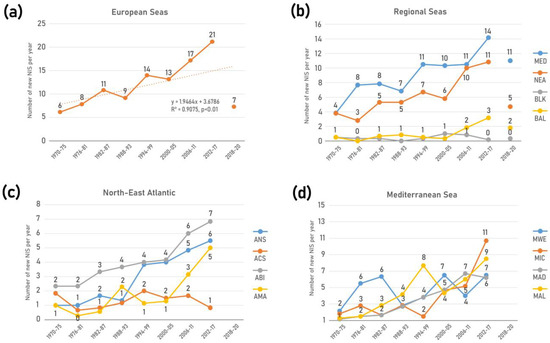
Figure 4.
Annual rate of NIS introductions (6-year average) at different geographic levels: (a) European waters; (b) regional seas, (c) North-East Atlantic subregions: ABI = Bay of Biscay-Iberian Shelf, ACS = Celtic Seas, ANS = Greater North Sea, AMA = Macaronesia (d) Mediterranean subregions. MWE = Western Mediterranean, MIC = Central Mediterranean, MAD = Adriatic Sea, MAL = Eastern Mediterranean. Dotted line for the EU trend (Figure 4a) is a linear regression line. Note that the annual average for the final interval has been calculated for three years only.
The Celtic Seas (ACS) host 107 NIS including parasites and pathogens (Figure 4b), 72 of which (67%) have been detected since 1970. The vast majority (69 taxa = 64%) are invertebrates, followed by primary producers (35 taxa = 33%) while vertebrates are represented only by three freshwater fishes that have been observed in Irish estuarine waters.
The Bay of Biscay and Iberian Shelf (ABI) subregion hosts 250 NIS, 215 of which (86%) have been introduced since 1970. Most of them are invertebrates (180 taxa = 72%), followed by primary producers (68 taxa = 27%) and vertebrates (2 taxa = 1%).
The Macaronesia (AMA) hosts 121 species, 109 (90%) introduced since 1970. Invertebrates dominate (72 taxa = 59%), followed by primary producers (29 taxa = 24%) and vertebrates (20 taxa = 17%).
The Mediterranean NIS list includes 578 species (473 = 83% since 1970) dominated by invertebrates (59%) (Figure 4a). Primary producers follow with approximately 23% of species among which macroalgae and Rhodophyta prevail. Vertebrates (103 taxa = 18%) are dominated by Red Sea (Lessepsian) fishes. The contribution of NIS groups varies among the Mediterranean subregions (Figure 2c). Primary producers have their largest representation in MWE and MAD (31–32%), introduced as contaminants in shellfish consignments in the major shellfish culture areas of the northern Adriatic and the French coast. On the other hand, the percentage of vertebrates is higher in MAL where they mostly arrived through the Suez Canal, and in MIC which receives naturally dispersing fish from MAL than all other subregions.
The EU part of the Black Sea (Bulgaria and Romania) hosts only 38 validated NIS out of a total of more than 110 NIS reported for the whole Black Sea. These are mostly invertebrates (33 species) with crustacean and molluscan species dominating. Only 24 NIS have been reported since 1970 including two microalgae.
In addition to the 874 NIS in European waters, 57 NIS detected in one regional sea are native or cryptogenic in at least one other regional Sea (Supplementary Table S1). These include macroalgae (18 taxa), mollusks (13 taxa), crustaceans (11 taxa), cnidarian (5 taxa), polychaetes (5 taxa), tunicates (2 taxa), bryozoan (1 taxon), Fish (1 taxon), and microalgae (1 taxon). They have been transferred from the NEA to the MED and BLK Seas (more than 27 taxa), but also from the MED to the NEA (more than 22 taxa). Finally, six species have been transferred from the EU BLK waters to the BAL.
Species classified as NIS in a country but partly native or cryptogenic within the subregion/region of the country were not included in the analyses, with some examples provided in Table 3. In contrast, species native in one subregion, but NIS in another subregion within the same MSFD region were not listed in Table 2 but are considered as NIS at the subregional level (Supplementary Table S2). They are mostly widespread native or cryptogenic species in the MED and NEA that have been classified as NIS in Macaronesia.

Table 3.
Examples of partly native/cryptogenic species within the same region/subregion excluded from the analyses. For regions/subregions’ abbreviations see Table 1.
The trend in new NIS introductions per 6 year assessment periods varies among groups and regional seas (Figure 3). The upward trend observed for invertebrates at the pan-European level is evident in the BAL, NEA, and MED Seas but not in the BLK Sea.
Overall, the rate of new NIS introductions (excluding parasites, pathogens, and microalgae) at the Pan-European level has increased at what appears to be a linear trend since 1970 from six to 21 NIS per year (Figure 4a). While evident in most regional seas, the increase also obscures large regional differences such as the steep increase from the early 2000s to 2017 in the Baltic Sea (Figure 4b) and a decreasing trend in the Black Sea (Figure 4b) and the Celtic Seas (Figure 4c). Comparison with the latest assessment period (2018–2020) shows a decline in the annual average rate of new NIS introductions compared to the preceding trends in many regional seas. Thus, while the annual rate of NIS in the North-East Atlantic steadily increased since 1970, although with subregional differences, reaching 11 new NIS per year in the 2012–2017 period, the latest assessment period (2018–2020) indicated a decline to an average of five NIS per year (Figure 4b). The annual rate of new NIS in the Greater North Sea (ANS) increased rapidly in the 1994–1999 period and maintained the upward trend in the last assessment period reaching six new NIS per year (Figure 4c). In the Bay of Biscay and Iberian Shelf (ABI), a steady upward trend was observed until 2005, followed by a sharp increase in the following periods, reaching seven new NIS per year in the 2012–2017 period. A similar pattern to that of ABI was observed in Macaronesia where the annual rate reached five NIS/per year in the 2012–2017 period. The highest number of new NIS introductions in the Celtic Seas occurred in the assessment period 1994–2005 with two new NIS per year. A declining trend was observed in the last assessment periods. Only five invertebrates were detected in the 2012–2017 period, and none since 2017.
All analyses in the Mediterranean Sea are based on 460 NIS taxa observed for the first time since 1970. On an annual basis, the number of newly introduced NIS has increased in the Mediterranean since the late 1990′s reaching 14 species per year in the period 2012–2017 (Figure 4b). This increasing trend is also observed at a subregional level for all regions but the MWE. Specifically, the annual new NIS rate calculated in the assessment period (2012–2017) reached 11 new NIS per year in the MIC, followed by nine in the MAL, seven in the MWE and six in the MAD (Figure 4d). In the MWE, the annual rate of NIS introductions fluctuates between two and seven species per year without any pronounced peaks or temporal trends. In contrast, a slight leveling off in the introductions rate appears in the MAD, while the rate of new NIS introductions presents a steeper increase in the MAL and MIC after the mid-2000s.
The rate of introductions in the BLK peaked in the 1994–2006 period reaching one new NIS per year but dropped in the following periods (Figure 4b). As many as six species (18%) have expanded the geographic range from neighboring areas surrounding the Black Sea where they first invaded, while the presence of two NIS namely the oysters Crassostrea virginica and Magallana gigas is attributed to escape from confinement (oyster culture facilities).
4. Discussion
With the current work, we aimed at establishing an updated status of NIS in European waters to provide a robust baseline for understanding trends in new NIS arrivals. The presented analyses documented an increasing trend in the annual rate of new NIS at all spatial levels until 2017 while highlighting some major regional differences both in the composition of xenodiversity and the temporal evolution of new NIS introductions at the subregional level, that can prove useful in further steps of setting thresholds for NIS trends indicators. Our findings are discussed in the context of spatial, temporal, species-specific and effort-related sources of uncertainty (Figure 5), which are primarily epistemic in nature (sensu [43,44]) i.e., they relate to measurement or systematic error, be it in species taxonomy, identification, and origin, in the spatial aspects of inventories or the temporal uncertainties associated with trends estimation. Subjective judgment may introduce additional uncertainty in determining species to include/exclude from management actions, such as cryptogenic species or functional groups addressed with different policy instruments. Finally, we provided an explicit account of partly native species in different management units, helping to resolve linguistic uncertainties stemming from a context-dependent definition of the terms alien/native.
Figure 5.
Schematic diagram of the process of NIS trends calculation identifying sources of uncertainty (outlined in rectangles) as they propagate from species to inventories to trends. Additional considerations for threshold setting are indicated by oval outlines. Sp. complexes = species complexes, Tax. Revisions = taxonomic revisions. Sp.nov. = species novae.
4.1. Validation of European NIS: A Challenging and Dynamic Task
One of the main challenges in establishing a robust and accurate baseline is addressing taxonomic or biogeographic uncertainties and incorporating new taxonomic information. To maintain a conservative viewpoint and avoid potential false positives, the authors agreed to exclude species that have raised uncertainties regarding (i) the known existence of cryptic species, (ii) recent taxonomic revisions, (iii) suspicions of possible errors for taxa belonging to species complex, and/or (iv) species that are possibly non-native but only recently described and thus requiring further clarification about their status. Issues arising from cryptic species, taxonomic revision, and occurrence of species complexes were noticed in the NEA for the ascidians Botrylloides schlosseri, Ciona intestinalis, and the mussels Mytilus galloprovincialis and Mytilus trossulus.
Botrylloides schlosseri is an example of the problems associated with the identification of cryptic species complexes, which are common among widely distributed marine taxa [45]. An extensive study by Bock et al. [46] showed that several cryptic species of B. schlosseri coexist at a regional scale in northwestern Europe. Some are probably native (e.g., clade E in Brittany, France) while others are likely to be introduced, considering their near-global distribution (e.g., clade A in Brittany, France). The specimens of B. schlosseri, reported in the North-East Atlantic, could thus be either NIS or native species. Thus, overall, it seems more reasonable to assign B. schlosseri a cryptogenic status.
In the case of Ciona intestinalis, uncertainties stem from a recent extensive taxonomic revision [47]. Based on a series of morphological and molecular investigations (references in 47), this species name was shown to bring together two distinct species, namely Ciona intestinalis and Ciona robusta, which had previously been described as two distinct species but unfortunately synonymized in 1985. Until a recent taxonomic revision, C. robusta was known as C. intestinalis type A and C. intestinalis as C. intestinalis type B although the type was not always reported. Furthermore, since the taxonomic revision was announced in 2017, the use of the correct species name is questionable for our dataset ending in 2020. C. robusta, native to Asia, is the only Ciona species introduced, so far, to the North-East Atlantic (in the early 2000s) [48,49]. We, therefore, excluded records of C. intestinalis and retained only records of C. robusta or C. intestinalis type A, as the use of these names refers to the Pacific-origin species.
The situation is even more complicated with the Mytilus edulis species complex, which obscures three European accepted species M. edulis, M. galloprovincialis and M. trossulus that still hybridize and exchange genes at contact zones. In our list, we have two species reported as introduced in the North-East Atlantic, for which reports are questionable: M.e galloprovincialis and M. trossulus. The use of the species name M. galloprovincialis is insufficient to determine native vs. introduced status, as it covers two distinct lineages, one present in the Mediterranean Sea, and the other in the Atlantic [50]. As with C. intestinalis prior to its taxonomic revision, the name M. galloprovincialis does not allow us to determine the native or introduced status of specimens reported from the North-East Atlantic. In addition, the natural presence of the Atlantic lineage as enclosed population patches in Brittany, Wales, Scotland, and Northern Ireland is not always recognized by some specialists and is debated. In the case of M. trossulus, identification has most often been established using barcoding or metabarcoding based on the COI mitochondrial marker. However, in the absence of details regarding the reference sequence that was used for the taxonomic assignment, we face another problem here. Some of the reference data available in public databases are indeed from specimens collected in the Baltic Sea, where the mitochondrial genome of M. trossulus has been extensively introgressed (i.e., replaced) by that of M. edulis, which may lead to a false taxonomic assignment of a M. edulis specimen to M. trossulus [51]. In addition, recent work has shown that M. edulis carries a transmissible cancer of M. trossulus origin. Thus, molecular-based identification may lead to the assignment of M. trossulus or edulis-trossulus hybrids for M. edulis specimens with this cancer [52,53]. The so-called “Baltic Mytilus trossulus” actually differs distinctly in morphology, ecology and genetic characters from M. trossulus, i.e., a species described from the NE Pacific [54]. To resolve this, Mytilus edulis balthicus by Gittenberger and Gittenberger, 2021, has recently been described. In addition, to further the nomenclatorial stability within the M. edulis complex, the locus typicus restrictus of the nominal taxon M. edulis has been restricted to the North Sea off the Dutch coast [54].
The improvement of molecular methods in ecological studies has helped to shed some light on species’ origins and their actual distribution, (see for instance the case of Tritia neritea detailed in the next Section). However, at the same time, this may give rise to some controversies until further studies finally provide unequivocal confirmation of status with more data. This is the case, for example, of the oyster Ostrea stentina, which was recently found to encompass two different genotypes, one of them belonging to the newly described Ostrea neostentina with type locality in Hong Kong [55]. A new distribution map of this genus has thus been constructed, with O. stentina present in both the MED and NEA regions, and O. neostentina only in the MED. New studies are taking place to confirm the native range, but, so far, regarding the present knowledge of historical records and taxonomical studies, the population of O. stentina present in the ABI subregion is considered introduced.
In addition, systematics is a dynamic field of research, as novel species are continuously being described; some of them possibly being novel introduced species. However, in the absence of further verification regarding their status, we did not include some of these species in our list. A case in point is that of the spaghetti worm Terebella banksyi nov. sp [56] newly described following its collection in 2017 in Arcachon Bay and found in farms or reefs of the Pacific oyster Magallana gigas. Similar uncertainty is occurring for the newly described colonial tunicate Didemnum pseudovexillum nov. sp [57], distinctive from the well-known invader D. vexillum by morphological traits and genetic characteristics and found only in marinas in the Celtic Seas (Brittany, France) and NW Mediterranean Sea (Spain). Considering the habitats (farms, marinas) and extensive range of D. pseudovexillum nov., it is likely that it had been introduced. However, further clarification would be needed to ascertain its introduced or cryptogenic status.
We included in the list of accepted species that arose following hybridization between a NIS and a native species. Hybridization between native and introduced species is very common in plants [58,59]. It has also been documented in marine species although being still poorly examined, and yet an important issue to consider for marine NIS management [6]. In coastal systems, this process is well-illustrated by cordgrass species from the Spartina genus [60,61]. For instance, S. alterniflora hybridized with the native species S. maritima after its introduction in the United Kingdom. This hybridization gave rise to S. townsendii, a sterile species, which then gave rise through polyploidization to S. anglica. The latter species is highly successful, displacing the native S. maritima, and is present in most of the ANS and locally in the western BAL. Thus, S. anglica is not per se introduced but is included in our list, because it would have never existed without the introduction of S. alterniflora in Europe.
Another cordgrass species, Spartina versicolor Fabre, has also a controversial taxonomic status. Although it was recorded as NIS in several European countries in the 19th and 20th centuries, it was considered synonymous with Spartina patens, due to morphological similarities [62,63] sampled several populations of S. versicolor in the Mediterranean, Atlantic, and North Africa saltmarshes and conducted cytogenetic and molecular analysis (microsatellite, nuclear and chloroplast DNA sequences) and compared it to North American Spartina species. Their results supported the hypothesis that all European and African populations of S. versicolor are, in fact, North American S. patens, introduced before or at the beginning of the nineteenth century. Due to potential hybridization within Spartina species, further investigations are needed to clarify any potential hybridizations between introduced species with the native ones (e.g., S. maritima).
4.2. Issues with Assessing the Spatial Distribution of NIS in Europe’s Seas
The NIS data-gathering process is not standardized (there is no consistent methodology) among EU Member States, which is a drawback and likely to generate bias and uncertainty in the assessment itself. In addition, biases may arise from the lack of dedicated surveillance programs. Not only studies focused on NIS introduction hot spots, such as ports and marinas or aquaculture facilities, but also the increment of monitoring programs to give responses to other MSFD descriptors increased the probability of finding newly introduced NIS during the surveys. However, it must be highlighted that several new records are introductions that most probably either went unnoticed in previous surveys or from areas that were never previously investigated. Monitoring programs are also not equally implemented in all subregions, and only a few have specifically focused on NIS and cryptogenic species detection [14].
Therefore, data need to be updated continuously from other monitoring programs or scientific literature reporting NIS. For example, in the NEA region, subregions such as ANS or ACS have historically received more attention than ABI [64]. In several countries such as Spain, Portugal, and Denmark, there were no baseline studies for NIS until very recently and the list included in the last assessment period (2012–2017), can therefore be considered as a baseline for some countries.
Boundaries between sub-regions established for MSFD reports are also challenging. In particular, the ABI subregion boundaries, as the boundaries between ANS and ACS, very often raise questions when establishing the status of some species because the natural borders between water masses are not static at these human-established borders (Figure 1). The same holds for the MWE subregion. Its western limit finishes a few kilometers after the strait of Gibraltar making it difficult to establish proper frontiers between Mediterranean and Atlantic waters since the Mediterranean shows a high influence even until central Atlantic waters [65]. In this sense in the southern extension of the ABI subregion, being highly influenced by Mediterranean waters, some species whose native range extends in both NEA and MED regions can be found, giving them the category of partly native species in a subregion, but being NIS in a country of this same subregion (Table 3) or in another subregion of the same region. This is the case, for example, of the gastropod Cymbium olla, whose native range includes Algarve (southern Portugal) and the Gulf of Cadiz (southern Spain—Atlantic coast), which are part of the ABI subregion, but also Cadiz in the Alboran Sea site, which is in the MWE subregion. Therefore, Cymbium olla, which is partly native in the MWE subregion even in some other localities in the MWE, might be locally classified as NIS [66].
Species distribution and their possible expansion, are never contained within any human delineation of marine borders, making it difficult to categorize their status when it comes to classification at any bordering level (subregion, region, or Pan-European). This issue is particularly important for species spreading gradually, which might be considered either as a natural expansion or introduced by human activities. For example, the nassariid gastropod Tritia neritea’s native range includes the Mediterranean and the Black Sea, as well as all around the Iberian Peninsula (Hidalgo [67] as Cyclops neriteum), but since the 1970s, this gastropod has been extending its range along the coast of Frances since its first record in 1976 in Arcachon Bay [68]. Its presence almost exclusively in oyster farming areas and the genetic characteristics of the French populations (e.g., admixture of lineages found in different locations of the Mediterranean Sea that indicated multiple introductions [69]) finally concur to report this nassariid gastropod as a NIS, probably introduced by oyster cultures in France [70]. Therefore, it is considered partly native to the ABI subregion because of its native range in Portugal and Spain, and its later introduction in France (Table 3). Some cases such as Tritia neritea, exemplify the difficulty of sometimes categorizing species as either NIS, cryptogenic or native because of their life history, migratory and demographic history, influenced by paleoclimatic events in a longer time scale and more recently by human activities [69,71]. These processes determine the species’ contemporary distribution, showing a patched map of native and introduced localities, even at local small scales [72].
Another example of a partly native species is that of the amphipod Ericthonius didymus (Krapp-Schickel, 2013), which was described in the Adriatic Sea from the Venice Lagoon (Italy). This recent description was rapidly followed by new records in Europe both in the Mediterranean and the Atlantic between 2013 and 2017 [73]. These observations, some of which date back to the year of description of the species, do not allow an unequivocal designation of the species as non-indigenous in the Bay of Biscay. However, the species is considered NIS in the ANS and the AMA, due to its presence in anthropogenically stressed sites, such as harbors/marinas and shellfish grounds [73].
4.3. Trends Indicator
Across all taxonomic groups, the rate of new NIS introductions in EU waters has increased gradually since 1970 and reached an average of 21 NIS per year in the period 2012–2017. The same upward trend was noticed for the Baltic, North-East Atlantic, and the Mediterranean Sea, but was more evident in the Mediterranean and Baltic Seas. In contrast, a decreasing trend was seen in the Black Sea with only one new species detected in the last assessment periods (0.2–0.3 NIS per year). Low figures noticed in the periods of 1988–1993 and 2000–2005 are likely an artifact of varying monitoring and reporting efforts between the regions over these periods.
The high rate of annual Introductions from 2000–2005 was very likely associated with a growing research interest in NIS, rather than discrete episodic events leading to high levels of new introductions during these years. Indeed, the development of several dedicated projects (AquaNIS, DAISIE, EASIN) produced outputs with updates on the list of NIS.
The decreased annual rate of new NIS introductions in the period 2018–2020 at almost all geographic levels examined has recently been attributed to time lags in reporting [74] rather than a result of NIS intervention programs. Also, there are fewer sampling years in this last interval analyzed, which might entail larger variability in the annual rate. This provisional reduction of new NIS registered is furthermore not likely to be associated with the implementation of measures since no new programs of measures have been implemented yet (e.g., only three marine NIS, the fish Plotosus lineatus, the seaweed Rugulopteryx okamurae, and the crab Eriocheir sinensis (only partly marine), are in the EU list of Invasive Alien Species of Union concern) and the implementation of the Ballast Water Management Convention at the European level is still in progress [75]. The only exception is the Council Regulation (EC) No 708/2007 of 11 June 2007 concerning the use of alien and locally absent species in aquaculture that may have decreased the risk of novel species introduced for cultivation purposes, although not preventing transfer within each EU country’s borders. A decrease in new NIS records in the last assessment period (2018–2020) for most regions might furthermore be explained by the homogeneity of marine NIS fauna since more and more species previously found exclusively in one of the countries are now found in more countries. Probably many species are expanding naturally from previously invaded countries.
The present upward trend in new NIS introductions to the Baltic Sea contradicts the previous D2C1 assessment, which indicated that the trend was decreasing since 2011 [76]. The discrepancy is very likely due to updated NIS records from several countries around the Baltic Sea. The latest assessment period in the present study covered only three years (2018–2020), but already five new NIS were recorded from the EU marine waters of the Baltic Sea during this time, suggesting that the ultimate HELCOM goal of zero new NIS introductions will not be reached, even though the rate of new NIS introductions has dropped to less than two new NIS per year. Overall, the current Baltic Sea analysis indicated that the number of new introductions has had a steep increase from the early 2000s to 2017. The increase may be due to growing scientific interest and promotion of citizen science projects [77], but it is evident that anthropogenic pressure through intensified shipping has steadily increased toward the marine environment of the Baltic Sea [78].
The NEA region encompasses several ecoregions, 4 sub-regions, and 10 different countries, making this region a very complex one for analyzing trends because of the heterogeneity in surveys and ecosystems. It is thus not surprising that quite a large number of species are reported as NIS within the region, and subregions (Figure 4b,c). Altogether the number of novel NIS has always been increasing, at least for invertebrates that are the most numerous NIS in this region (Figure 3). This is likely attributed to the continued increased maritime traffic in the region. Indeed, overall shipping density increased across the North-East Atlantic by 33.6% between 2013 and 2017 [79].
In comparison to the previous assessment [3,15,32], this work does not consider data from the UK waters. This leads to differences not only in the total number of NIS but also in the trends indicator as first detection dates may be years earlier in neighboring non-EU countries.
An earlier assessment (over the period of 2003–2014) of NIS in the ANS, ACS, and the ABI subregions showed that the number of newly recorded NIS varied by year and region showing a relatively constant linear increase in the ANS only, but not so in the ACS and ABI [80]. In this study, an increasing trend was observed in all subregions but the ACS. The high number of NIS in the ABI in the 2012–2017 period (7 NIS/year) is partly attributed to intensive studies in port areas and marinas [81,82,83] in the framework of the implementation of the MSFD descriptor 2 or research projects dedicated to NIS surveys. Furthermore, the increase of studies based on genetic analyses within this last decade has helped to rapidly and accurately detect newly introduced species reassess some species that have been misidentified, and elaborate an updated checklist of NIS [84,85,86]. In addition to traditional genetic approaches, in recent years metabarcoding of environmental DNA had been proposed and is increasingly used as a new tool to improve NIS detection [87]. The approach is promising and effective although it needs to be used cautiously to avoid both false negatives (i.e., present, but undetected NIS) and false positives (i.e., NIS erroneously detected) [51]. NIS detection by these methods requires fit-to-purpose protocols and should not be based on molecular data obtained for general biodiversity assessments [88]. Either way, the data show that the increase seems to be stabilizing, indicating that it is a good time to set the baseline.
The increasing trend in introductions in ANS, which culminated in the 2012–2017 period with six NIS per year, appears to be slowing down in the last assessment period (2018–2020) with four new NIS per year, although future publications are expected to bring to light more NIS. During the period 2018–2020, in France, the number of records increased. However, this is the only French subregion with such an increase, thanks to dedicated surveys programs carried out in the Normandy region [86]; these reports are not new either for France or for ANS [89] (and references therein), suggesting a decrease of new species but an important dispersal between subregions.
In the ACS, the decrease is even more pronounced than in the ANS, with no novel NIS reported after 2017. As for the ANS, the difference from the previous assessment can be partly attributed to the geographic areas involved. In the previous assessment [76] the NIS of the United Kingdom in ACS were included in the analysis. Moreover, pathogens were also included. Additionally, in the Western English Channel (French and UK coastline), a research project (Interreg Marinexus project) dedicated to rapid assessment surveys of NIS in marinas, well-known introduction hotspots, was carried out over 2010–2017 [78], and provided novel reports for European waters (e.g., the ascidian Asterocarpa humilis [90]).
The AMA NIS list presented here represents an updated version of the list reported by Castro et al. [29] following similar criteria. As opposed to the current study, species that underwent tropicalization processes (see 29, 41) were considered one of the criteria for NIS attributes in Castro et al. [29] inventory. Most changes were made on macroalgae records for the Azores as more information on records, taxonomy, and distributional updates have been gathered and led to some changes. In addition, a few new records have been added as [29] included records only until 2020 whereas the present account includes records reported until summer 2022.
Comparisons with the full NIS inventory of the MED are somewhat hampered by the geographic coverage of the current study, which is limited to the EU waters of the Mediterranean (plus Albania and Montenegro). As a result, total numbers of new NIS, as well as annual introduction rates, appear to be reduced in comparison to, e.g., [30], especially for the eastern Mediterranean, as primary Lessepsian introductions restricted to the Levantine were outside the spatial scope of this study. Indicatively, the whole Mediterranean Sea hosts upward of 1000 validated NIS, 786 of which are in the MAL [12,23,91], compared with the 579 NIS present in the EU parts of these waters. As such, it is not surprising that the annual introduction rate in the central Mediterranean in the 2012–2017 period exceeds that of the eastern Mediterranean, as the accelerated sea warming rates favor the spread of Indo-Pacific species already present in the Levantine [92]. On the other hand, the reduction in Transport-Contaminant species [76], which are more prevalent in the Adriatic and the western Mediterranean, may have contributed to the observed leveling off or decreasing NIS trends in these two subregions. For the Mediterranean Sea as a whole, there appear to be two “stepwise” increases in new NIS introductions, the first one in the late 1990′s, mostly driven by introductions in the MAL and likely related to sea surface warming [30,93], and the second in the 2012–2017 period. This last peak could partly reflect intensified research efforts, which the whole basin has undoubtedly experienced in the last decade [94] as already suggested for other regions and subregions of the NEA, and in line with comments by Bailey et al. [31]. In Slovenia, for example, the number of detected NIS has increased from 17 in 2012 to 57 in 2021, which is due to increased targeted research, mainly founded by the Ministry of Agriculture, Forestry and Food for the implementation of D2 in the country [95]. It also coincides with a sharp increase in the introduction rate of fouling species, notably in marinas and on leisure boats, at least in their detection and reporting [96,97]. Hence it is difficult to really evaluate the significance of these trends without considering a measure of “effort”, which again starkly exemplifies the need for standardized monitoring for any assessments to be meaningful.
Some of the earlier invading NIS in the BLK such as the blue crab Callinectes sapidus (Rathbun, 1896) appear to be established and spreading in the area over the years. Callinectes sapidus was first found on the Bulgarian coast of the Black Sea in 1967 [98], most likely transferred in ballast water but could have been spreading via the Marmara Sea from an invasive population in the northern Aegean. Six new records of the blue crab have been documented near the Bulgarian Black Sea coast since 2010. This is evidence of a recent expansion of the species in this part of the Black Sea. This expansion could be explained by the existence of an established population in the area and is confirmed by the capturing of an egg-bearing female in Varna Bay in 2005 [99]. It is anticipated that in the face of climate change the number of NIS in the EU areas of the BLK will increase in the near future due to the spreading (Unaided pathway) of NIS from the North Aegean Sea that has already invaded the BLK via the Sea of Marmara such as the marbled pine foot Siganus rivulatus [100,101]. Moreover, NIS recently introduced via vessels in the northeastern and southern Black Sea could spread unaided in the study area [102,103]. Such is the case of the polychaetes Laonome xeprovala that spread in the Danube Delta–Black Sea Ecosystem and Marenzelleria neglecta that was detected in 2021 in the same area [103].
4.4. Uncertainties in Trends
Uncertainties in trends first rely on the uncertainty of the first date of the report (if not consistent across periods). The true introduction year of NIS may be different from the detection year. As an example, the Terebellid polychaete Marphysa victori was detected in 2016 and described in 2017 from French waters in the Arcachon Bay, with doubts already surrounding its true origin due to its presence in and close to oyster farms where Magallana gigas is cultivated [104]. This possibility was verified several years later. Marphysa victori is native to the Northwest Pacific [105], and it has undoubtedly been introduced as a contaminant with oyster transfers. However, it remains unproven if its introduction is a consequence of oyster importation from Japan. Between 1971 and 1975, about 1200 t of Magallana gigas spat collected from Sendai Bay (Japan) were introduced into Arcachon Bay. Marphysa victori has a substantial economic value as bait and is widely collected by recreational and professional fishermen. The number of worms collected in the lagoon (13 companies) could reach 1 million per year [104]. Reaching such densities within a year would be impossible. Thus 1975 was set as the most plausible year of its introduction.
Other examples include Mollusca species observations in EU waters around 80 years after their first detection in neighboring non-EU waters. Such are the cases of the gastropod Berthellina citrina (Rüppell & Leuckart, 1828), which was first reported in the MED from the Gaza Strip in 1940 [106], but only in 2019 in EU Mediterranean waters: Cyprus [107] or of the bivalve Gafrarium savignyi (Jonas, 1846) with a first Mediterranean record in 1905 from Egypt [108] but an EU record in 2005 from Cyprus [109].
Various policy measures relevant to the Baltic Sea countries can result in uncertainties regarding the emergent reports of new NIS introductions. Trend analyses on new NIS introductions to the Baltic Sea, such as [22,27,110] may differ mainly due to the applied assessment principles, e.g., area of interest, and species included in the analyses. Baltic Sea delineation determined according to the EU MSFD differs from HELCOM delineation, and this often leads to NIS being reported, for example, from the Kattegat area, which is BAL according to the HELCOM delineation, but at the same time a part of the ANS according to the EU MSFD delineation. In addition, Russian coastal waters outside of St. Petersburg and Kaliningrad are obviously part of the Baltic Sea but are not included in assessments that refer to the marine waters of the EU.
Even more, pronounced discrepancies may be observed with pan-Mediterranean assessments due to the exclusion of non-EU Mediterranean countries in this study (see above). Regardless of administrative boundaries for EU policies, it is crucial that the marine environment is managed with sufficient harmonization between regional policies. Toward that end, the Contracting Parties to the Barcelona Convention—21 Mediterranean countries and the European Union—have recently developed and adopted the Integrated Monitoring and Assessment Programme for the Mediterranean region (IMAP) [111]. Within its framework and in accordance with the MSFD [9], GES for NIS in the Mediterranean was defined as the minimization of the introduction and spread of NIS linked to human activities, in particular for potential IAS, with the reduction in human-mediated introductions as the proposed State Target [112], a target that clearly needs to be further refined but seems far from achieved based on our latest data.
4.5. Threshold Values
Qualitative GES assessments to date have been based on directional trends and, despite ongoing efforts [110], threshold values for the NIS trend indicator have not been set yet and neither have more specific recommendations been made for the magnitude of this reduction or the number of reporting cycles that will define the reference conditions [113].
Waiting for a value of the percentage reduction to be established at a European level, as suggested by [14], the French decree relating to the definition of GES states that GES is achieved if there is a significant decrease in the number of new NIS over two cycles at minimum. As visible in this work, the number of new NIS increased in all French marine subregions during the previous cycle (2012–2017), and the goal has therefore not been reached to date.
The identification and comprehension of impact thresholds on ABI marine native communities is required. ABI countries must collaborate more closely to implement common methodologies for MSFD implementation, particularly regarding non-indigenous species (D2) [114]. Furthermore, good coordination is required for the creation of an effective alert system. It is worth mentioning the risk-based approaches to good environmental status (RAGES) project, which attempted to establish reproducible, transparent, and standardized risk management decision procedures based on international best practices. The increase in the number of new NIS introductions in the period 2006–2017 seems to be stabilizing, indicating that it is a good time to set the baseline. This decrease in new NIS records might be explained by a biotic homogenization of the ABI marine NIS fauna since more and more species previously found exclusively in one of the countries are now found in all three ABI countries. Probably many species are expanding naturally from previously invaded countries.
In the Mediterranean Sea, preliminary analyses [12] indicated that threshold values should be established separately for each subregion and should be sought by examining the data of the last two decades, if not an even more recent period. Further work by Galanidi and Zenetos [30], based on breakpoint analysis of 1970–2017 NIS data, corroborated the validity of a subregional approach, demonstrating different temporal breakpoints in the rate of NIS introductions per subregion, ranging from 1997 in the MAL, to 2000 in the MAD, 2003 in the MWE and 2012 in the MIC. They suggest that the mean introduction rate of these periods can be used to define threshold values but stress that GES target refinement and percentage reduction cannot proceed without careful consideration of management objectives and pathway pressure, as also pointed out in Tsiamis et al. [14].
Trends in the arrival of new NIS is a core indicator of the Baltic Marine Environment Protection Commission (Helsinki Convention, HELCOM), and the primary criterion D2C1 was assessed for the first time for a six-year assessment period (2011–2016) in 2018 [10]. The report listed new NIS and cryptogenic species for BAL over the assessment period. Contracting Parties of HELCOM have set a precautionary threshold to assess GES in relation to NIS. Zero new NIS introductions through anthropogenic activities to the Baltic Sea per six-year assessment period has been defined as the GES for NIS [10], and therefore one or more introductions to BAL would result in GES not being reached. Furthermore, it has been argued whether a reduction in new NIS introductions could be set as an intermediate objective if the goal of no new introductions cannot be reached. Even though a proportional reduction of new NIS introductions between the assessment periods would indicate temporary improvement of GES, the “zero tolerance policy” was chosen as the GES threshold to the BAL, because it is pragmatic, independent of earlier assessment periods, applicable even with uncertainties in relation to taxonomy and introduction pathways, and efficiently reflecting management measures [10,110].
4.6. Concluding Remarks—The Way forward
Considering how dynamic biological invasions are, NIS inventories should be curated regularly, especially when used to inform policy, in order to minimize errors and avoid over- or under-estimating the state of invasions in a region [44]. While the validation process in this work explicitly addressed many of the taxonomic and spatial components of uncertainty in the EU NIS baseline, other issues remain unresolved, among which the lack of standardized monitoring needs to be urgently rectified both for the meaningful interpretation of results and for the refinement of the relevant indicators.
Regional and sub-regional analyses revealed that there are relatively strong variations in the number of new NIS introductions between the European seas, as well as among the subregions within the same region. Hence, it is natural that GES threshold values for the primary criterion D2C1 are discussed and decided under regional cooperation, as some regions have preferable conditions for a wider variety of species and thus tend to suffer from a higher number of introductions. In addition, NIS pathways are region-specific (e.g., the Suez Canal in the MED, shipping in the NEA). Shipping was found to be a likely vector for over half of NIS in European waters both through biofouling and ballast discharges [2], while biofouling, particularly of recreational vessels, appears to be an important driver for the homogenization of the alien biota in the Mediterranean. As such, a more detailed focus on quantitative measures of pathway pressure would help better elucidate the observed NIS patterns, inform target setting and evaluate GES achievement in relation to management. Considering that currently only aquaculture-related introductions are addressed with EU-wide legislation and that the BWMC is not expected to be fully implemented until 2024 at the earliest, expectations for percentage reduction should have a realistic temporal horizon and, if possible, promote management implementation for the remaining major introduction pathways. More specific national or local measures may be put in place to protect sectors or sensitive habitats, e.g., see [115] for additional measures related to shellfish culture in the Wadden Sea), pathways of species introductions however operate globally and should be managed at appropriate scales.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/d14121077/s1, Table S1: Partly native or cryptogenic (CRY) species in European seas; Table S2: Species native/cryptogenic in one subregion, but NIS in another subregion.
Author Contributions
A.Z. and O.O.: conceptualization, review, and data collection. All authors: providing and validating national data, contribution with taxonomic expertise, interpretation of data. A.Z., O.O., M.G.: data analysis, writing the first draft of the manuscript. F.V. made significant contributions to early drafts of the manuscript. All authors wrote, revised, and contributed to the editing of the final manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
A.Z. and O.O. were partially funded by the European Environment Agency, through ETC/ICM 2021. The authors from the National Institute of Biology (Slovenia) acknowledge the financial support of the Slovenian Research Agency (Research Core Funding No. P1-0237) and of the Ministry of Agriculture, Forestry and Food. F.V. is supported by the CNRS Institute for Ecology and Environment. This work benefited from results obtained during surveys carried out in the Interreg IVa Marinexus programme and the Aquanis2.0 (TOTAL Foundation) and MarEEE (i-site MUSE; French National Research Agency under the “Investissements d’Avenir” programme ANR-16-IDEX-0006) projects allocated to FV. This is publication ISEM 2022-296. A.C.C.’s work was partially funded by FEDER funds through the Operational Programme for Competitiveness Factors—COMPETE and by Portuguese National Funds through FCT (Foundation for Science and Technology) under the UID/BIA/50027/2020 and POCI-01-0145-FEDER-006821. P.C. is supported by 2020.01797.CEECIND and (UIDB/04292/2020), granted by Fundação para a Ciência (FCT) e Tecnologia. RR’s work id funded by GI4Sado—IPS RD project. C.B.’s work is partially supported by programme MarBIS—Marine Biodiversity Information System financed through the Portuguese Government. Other support was provided by the Marine and Environmental Sciences Centre (MARE) financed by Portuguese National Funds through FCT/MCTES (UIDB/04292/2020), and by the project LA/P/0069/2020 granted to the Associate Laboratory ARNET. The Portuguese assessment benefited from the contribution of all the Portuguese experts working group on marine NIS. Authors from SLU acknowledge funding from The Swedish Agency for Sea and Water Management.
Institutional Review Board Statement
Not Applicable.
Data Availability Statement
The data availability statement for this manuscript is already described in the results section and the Supplementary Materials.
Acknowledgments
The outcome of the present study was improved through cooperation within the Joint Working Group on Ballast and Other Ship Vectors under the International Council for the Exploration of the Seas (ICES), Intergovernmental Oceanographic Commission of Unesco and International Maritime Organization, as well as the ICES Working Group on Introductions and Transfers of Marine Organisms. The authors thank Nicholas Jason Xentidis for preparing the figures.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Díaz, S.; Settele, J.; Brondízio, E.S.; Ngo, H.T.; Agard, J.; Arneth, A.; Zayas, C.N. Pervasive human-driven decline of life on Earth points to the need for transformative change. Science 2019, 366, eaax3100. [Google Scholar] [CrossRef] [PubMed]
- Korpinen, S.; Klančnik, K.; Peterlin, M.; Nurmi, M.; Laamanen, L.; Zupančič, G.; Popit, A.; Murray, C.; Harvey, T.; Andersen, J.H.; et al. Multiple Pressures and Their Combined Effects in Europe’s Seas; ETC/ICM Technical Report 4/2019: European Topic Centre on Inland, Coastal and Marine Waters; ETC/ICM: Magdeburg, Germany, 2019. [Google Scholar]
- Simberloff, D.; Martin, J.-L.; Genovesi, P.; Maris, V.; Wardle, D.A.; Aronson, J.; Courchamp, F.; Galil, B.; Garcia-Berthou, E.; Pascal, M.; et al. Impacts of biological invasions: What’s what and the way forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Ojaveer, H.; Galil, B.S.; Campbell, M.L.; Carlton, J.T.; Canning-Clode, J.; Cook, E.J.; Davidson, A.D.; Hewitt, C.L.; Jelmert, A.; Marchini, A.; et al. Classification of Non-Indigenous Species Based on Their Impacts: Considerations for Application in Marine Management. PLoS Biol. 2015, 13, e1002130. [Google Scholar] [CrossRef] [PubMed]
- Anton, A.; Geraldi, N.R.; Lovelock, C.E.; Apostolaki, E.T.; Bennett, S.; Cebrian, J.; Krause-Jensen, D.; Marbà, N.; Martinetto, P.; Pandolfi, J.M. Global ecological impacts of marine exotic species. Nat. Ecol. Evol. 2019, 3, 787–800. [Google Scholar] [CrossRef]
- Viard, F.; Riginos, C.; Bierne, N. Anthropogenic Hybridization at Sea: Three evolutionary questions relevant to invasive species management. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2020, 375, 20190547. [Google Scholar] [CrossRef]
- Tsirintanis, K.; Azzurro, E.; Crocetta, F.; Dimiza, M.; Froglia, C.; Gerovasileiou, V.; Langeneck, J.; Mancinelli, G.; Rosso, A.; Stern, N. Bioinvasion impacts on biodiversity, ecosystem services, and human health in the Mediterranean Sea. Aquat Invasions 2022, 3, 308–352. [Google Scholar] [CrossRef]
- Blackburn, T.M.; Pyšek, P.; Bacher, S.; Carlton, J.T.; Duncan, R.P.; Jarošík, V.; Wilson, J.R.; Richardson, D.M. A proposed unified framework for biological invasions. Trends Ecol. Evol. 2011, 26, 333–339. [Google Scholar] [CrossRef]
- European Commission. Commission Decision (EU) 2017/848 of May 2017 laying down criteria and methodological standards on good environmental status of marine waters and specifications and standardised methods for monitoring and assessment, and repealing Decision 2010/477/EU. Off. J. Eur. Union L 2017, 125, 43–74. [Google Scholar]
- HELCOM. Trends in Arrival of New Non-Indigenous Species. HELCOM Core Indicator Report. Available online: https://helcom.fi/media/core%20indicators/Trends-in-arrival-of-new-non-indigenous-species-HELCOM-core-indicator-2018.pdf (accessed on 10 August 2022).
- OSPAR CEMP Guidelines Common Indicator: Changes to Non-Indigenous Species Communities (NIS3) (OSPAR Agreement2018-04). Available online: https://www.ospar.org/documents?v=38992 (accessed on 10 June 2022).
- UNEP/MED WG. 500/Monitoring and Assessment Scales, Assessment Criteria and Thresholds Values for the IMAP Common Indicator Related to Non-Indigenous Species. In Proceedings of the CORMON Meeting, Online, 10 June 2021. [Google Scholar]
- UNEP First Draft of the Post-Biodiversity Framework. In Proceedings of the Open Ended Working Group on the Post-Global Biodiversity Framework, Online, 23 August–3 September 2021; Convention on Biological Diversity: Montreal, QC, Canada, 2021.
- Tsiamis, K.; Palialexis, A.; Connor, D.; Antoniadis, S.; Bartilotti, C.; Bartolo, G.A.; Berggreen, U.C.; Boschetti, S.; Buschbaum, C.; Canning-Clode, J.; et al. Marine Strategy Framework Directive-Descriptor 2, Non-Indigenous Species, Delivering Solid Recommendations for Setting Threshold Values for Non-Indigenous Species Pressure on European Seas; Publications Office of the European Union: Luxembourg, 2021. [Google Scholar] [CrossRef]
- Streftaris, N.; Zenetos, A.; Papathanassiou, E. Globalisation in marine ecosystems—The story of non indigenous marine species across European Seas. Oceanogr. Mar. Biol. 2005, 43, 419–453. [Google Scholar]
- Zenetos, A.; Streftaris, N.; Micu, D.; Todorova, V.; Joseffson, M.; Gollasch, S.; Zaiko, A.; Olenin, S. Harmonisation of European alien species databases: A 2009 update of marine alien species towards the forthcoming SEBI2010 report. In Proceedings of the Poster Presented at BIOLIEF, World Conference on Biological Invasions and Ecosystem Functioning, Porto, Portugal, 27–30 October 2009. [Google Scholar]
- DAISIE. Handbook on Alien Species in Europe; Springer: Berlin, Germany, 2009; p. 399. [Google Scholar]
- AquaNIS. Editorial Board, Information System on Aquatic Non-Indigenous and Cryptogenic Species. World Wide Web Electronic Publication. Available online: https://www.corpi.ku.lt/databases/aquanis (accessed on 1 June 2021).
- EASIN, European Commission—Joint Research Centre—European Alien Species Information Network (EASIN). 2022. Available online: https://easin.jrc.ec.europa.eu/ (accessed on 16 August 2022).
- Tsiamis, K.; Gervasini, E.; D’Amico, F.; Backeljau, T. The EASIN editorial board: Quality assurance, exchange and sharing of alien species information in Europe. Manag. Biol. Invasions 2016, 7, 321–328. [Google Scholar] [CrossRef]
- Chainho, P.; Fernandes, A.; Amorim, A.; Ávila, S.P.; Canning-Clode, J.; Castro, J.J.; Costa, A.C.; Costa, J.L.; Cruz, T.; Gollasch, S.; et al. Non-indigenous species in Portuguese coastal areas, coastal lagoons, estuaries and islands. Estuar. Coast. Shelf Sci. 2015, 167, 199–211. [Google Scholar] [CrossRef]
- Ojaveer, H.; Olenin, S.; Narščius, A.; Florin, A.-B.; Ezhova, E.; Gollasch, S.; Jensen, K.R.; Lehtiniemi, M.; Minchin, D.; Normant Saremba, M.; et al. Dynamics of Biological Invasions and Pathways over Time: A Case Study of a Temperate Coastal Sea. Biol. Invasions 2017, 19, 799–813. [Google Scholar] [CrossRef]
- Zenetos, A.; Albano, P.G.; Garcia, E.L.; Stern, N.; Tsiamis, K.; Galanidi, M. Established non-indigenous species increased by 40% in 11 years in the Mediterranean Sea. Meditter. Mar. Sci. 2022, 23, 196–212. [Google Scholar] [CrossRef]
- Băncilă, R.I.; Skolka, M.; Ivanova, P.; Surugiu, V.; Stefanova, K.; Todorova, V.; Zenetos, A. Alien species of the Romanian and Bulgarian Black Sea coast: State of knowledge, uncertainties, and needs for future research. Aquat Invasions 2022, 17, 353–373. [Google Scholar] [CrossRef]
- Servello, G.; Andaloro, F.; Azzurro, E.; Castriota, L.; Catra, M.; Chiarore, A.; Crocetta, F.; D’Alessandro, M.; Denitto, F.; Froglia, C.; et al. Marine alien species in Italy: A contribution to the implementation of descriptor D2 of the marine strategy framework directive. Mediterr. Mar. Sci. 2019, 20, 1–48. [Google Scholar] [CrossRef]
- Zenetos, A.; Karachle, P.K.; Corsini-Foka, M.; Gerovasileiou, V.; Simboura, N.; Xentidis, N.J.; Tsiamis, K. Is the trend in new introductions of marine non-indigenous species a reliable criterion for assessing good environmental status? The case study of Greece. Meditter. Mar. Sci. 2020, 21, 775–793. [Google Scholar] [CrossRef]
- Staehr, P.A.; Jakobsen, H.H.; Hansen, J.L.S.; Andersen, P.; Christensen, J.; Göke, C.; Thomsen, M.S.; Stebbing, P.D. Trends in records and contribution of non-indigenous and cryptogenic species to marine communities in Danish waters: Potential indicators for assessing impact. Aquat. Invasions 2020, 15, 217–244. [Google Scholar] [CrossRef]
- Verleye, T.J.; De Raedemaecker, F.; Vandepitte, L.; Fockedey, N.; Lescrauwaet, A.-K.; De Pooter, D.; Mees, J. (Eds.) Niet-inheemse Soorten in Het Belgisch Deel van de Noordzee en Aanpalende Estuaria Anno 2020; VLIZ Special Publication, 86; Vlaams Instituut voor de Zee (VLIZ): Oostende, België, 2020; p. 623. ISBN 9789464206005. [Google Scholar]
- Castro, N.; Carlton, J.T.; Costa, A.C.; Marques, C.; Hewitt, C.L.; Cacabelos, E.; Gizzi, F.; Gestoso, I.; Monteiro, J.G.; Costa, J.L.; et al. Diversity and patterns of marine non-native species in the archipelagos of Macaronesia. Divers. Distrib. 2022, 28, 667–684. [Google Scholar] [CrossRef]
- Galanidi, M.; Zenetos, A. Data-Driven Recommendations for Establishing Threshold Values for the NIS Trend Indicator in the Mediterranean Sea. Diversity 2022, 14, 57. [Google Scholar] [CrossRef]
- Bailey, S.A.; Brown, L.; Campbell, M.L.; Canning-Clode, J.; Carlton, J.T.; Castro, N.; Chainho, P.; Chan, F.T.; Creed, J.C.; Curd, A.; et al. Trends in the Detection of Aquatic Non-indigenous Species across Global Marine, Estuarine and Freshwater Ecosystems: A 50-year Perspective. Divers. Distrib. 2020, 26, 1780–1797. [Google Scholar] [CrossRef]
- Tsiamis, K.; Palialexis, A.; Stefanova, K.; Gladan, Ž.N.; Skejić, S.; Despalatović, M.; Cvitković, I.; Dragičević, B.; Dulčić, J.; Vidjak, O.; et al. Non-indigenous species refined national baseline inventories: A synthesis in the context of the European Union’s Marine Strategy Framework Directive. Mar. Pollut. Bull. 2019, 145, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Tsiamis, K.; Boschetti, S.; Palialexis, A.; Somma, F.; Cardoso, A.C. Marine Strategy Framework Directive—Review and Analysis of EU Member States’ 2018 Reports—Descriptor 2: Non-Indigenous Species; Assessment (Art. 8), Good Environmental Status (Art. 9) and Targets (Art. 10); Publications Office of the European Union: Luxembourg, 2021; EUR EN. [Google Scholar] [CrossRef]
- Jensen, H.M.; Panagiotidis, P.; Reker, J. Delineation of the MSFD Article Marine Regions and Subregions, Version 1.0; European Environment Agency: Kopenhagen, Denmark. Available online: https://data.europa.eu/euodp/data/dataset/data_msfdregions-and-subregions (accessed on 15 September 2021).
- Spalding, M.D.; Fox, H.E.; Allen, G.R.; Davidson, N.; Ferdaña, A.Z.; Finlayson, M.; Halpern, B.S.; Jorge, M.A.; Lombana, A.; Lourie, S.A. Marine ecoregions of the world: A bioregionalization of coastal and shelf areas. BioScience 2007, 57, 573–583. [Google Scholar] [CrossRef]
- Carlton, J.T. Biological invasions and cryptogenic species. Ecology 1996, 77, 1653–1655. [Google Scholar] [CrossRef]
- Bick, A.; Bastrop, R.; Kotta, J.; Meißner, K.; Meyer, M.; Syomin, V. Description of a new species of Sabellidae (Polychaeta, Annelida) from fresh and brackish waters in Europe, with some remarks on the branchial crown of Laonome. Zootaxa 2018, 4483, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Capa, M.; van Moorsel, G.; Tempelman, D. The Australian feather-duster worm Laonome calida Capa, 2007 (Annelida: Sabellidae) introduced into European inland waters? Bioinvasions Rec. 2014, 3, 1–11. [Google Scholar] [CrossRef]
- Tamulyonis, A.Y.; Gagaev, S.Y.; Stratanenko, E.A.; Zuyev, Y.A.; Potin, V.V. Invasion of the Polychaeta Laonome xeprovala Bick & Bastrop, 2018 (Sabellidae, Polychaeta) into the estuary of the Luga and Khabolovka Rivers (Luga Bay, Gulf of Finland). Russ. J. Biol. Invasions 2020, 11, 148–154. [Google Scholar] [CrossRef]
- Gómez, F. Comments on the non-indigenous microalgae in the European seas. Mar Pollut Bull. 2019, 148, 1–2. [Google Scholar] [CrossRef]
- Canning-Clode, J.; Carlton, J.T. Refining and expanding global climate change scenarios in the sea: Poleward creep complexities, range termini, and setbacks and surges. Divers. Distrib. 2017, 23, 463–473. [Google Scholar] [CrossRef]
- Provan, J.; Booth, D.; Todd, N.P.; Beatty, G.E.; Maggs, C.A. Tracking biological invasions in space and time: Elucidating the invasive history of the green alga Codium fragile using old DNA. Divers. Distrib. 2008, 14, 343–354. [Google Scholar] [CrossRef]
- Regan, H.M.; Colyman, M.; Burgman, M.A. A taxonomy and treatment of uncertainty for ecology and conservation biology. Ecol. Appl. 2002, 12, 618–628. [Google Scholar] [CrossRef]
- Latombe, G.; Canavan, S.; Hirsch, H.; Hui, C.; Kumschick, S.; Nsikani, M.M.; Potgieter, L.J.; Robinson, T.B.; Saul, W.-C.; Turner, S.C.; et al. A four-component classification of uncertainties in biological invasions: Implications for management. Ecosphere 2019, 10, e02669. [Google Scholar] [CrossRef]
- Appeltans, W.; Ahyong, S.T.; Anderson, G.; Angel, M.V.; Artois, T.; Bailly, N.; Bamber, R.; Barber, A.; Bartsch, I.; Berta, A. The Magnitude of Global Marine Species Diversity. Curr. Biol. 2012, 22, 2189–2202. [Google Scholar] [CrossRef] [PubMed]
- Bock, D.G.; MacIsaac, H.J.; Cristescu, M.E. Multilocus genetic analyses differentiate between widespread and spatially restricted cryptic species in a model ascidian. Proc. R. Soc. B Biol. Sci. 2012, 279, 2377–2385. [Google Scholar] [CrossRef]
- Gissi, C.; Hastings, K.E.M.; Gasparini, F.; Stach, T.; Pennati, R.; Manni, L. An unprecedented taxonomic revision of a model organism: The paradigmatic case of Ciona robusta and Ciona intestinalis. Zool. Scr. 2017, 46, 521–522. [Google Scholar] [CrossRef]
- Nydam, M.; Harrison, R. Genealogical relationships within and among shallow-water Ciona species (Ascidiacea). Mar. Biol. 2007, 151, 1839–1847. [Google Scholar] [CrossRef]
- Bouchemousse, S.; Bishop, J.; Viard, F. Contrasting global genetic patterns in two biologically similar, widespread and invasive Ciona species (Tunicata, Ascidiacea). Sci. Rep. 2016, 6, 24875. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.; Fraïsse, C.; El Ayari, T.; Liautard-Haag, C.; Strelkov, P.; Welch, J.J.; Bierne, N. How do species barriers decay? Concordance and local introgression in mosaic hybrid zones of mussels. J. Evol. Biol. 2021, 34, 208–223. [Google Scholar] [CrossRef]
- Couton, M.; Lévêque, L.; Daguin-Thiébaut, C.; Comtet, T.; Viard, F. Water eDNA metabarcoding is effective in detecting non-native species in marinas, but detection errors still hinder its use for passive monitoring. Biofouling 2022, 38, 367–383. [Google Scholar] [CrossRef]
- Riquet, F.; Simon, A.; Bierne, N. Weird genotypes? Don’t discard them, transmissible cancer could be an explanation. Evol. Appl. 2017, 10, 140–145. [Google Scholar] [CrossRef]
- Hammel, M.; Simon, A.; Arbiol, C.; Villalba, A.; Burioli, E.A.V.; Pepin, J.F.; Lamy, J.B.; Benabdelmouna, A.; Bernard, I.; Houssin, M.; et al. Prevalence and polymorphism of a mussel transmissible cancer in Europe. Mol. Ecol. 2022, 31, 736–751. [Google Scholar] [CrossRef]
- Gittenberger, A.; Gittenberger, E. Polytypic Mytilus edulis, with a name for the Baltic subspecies. Basteria 2021, 85, 116–125. [Google Scholar]
- Hu, L.; Wang, H.; Zhang, Z.; Li, C.; Guo, X. Classification of small flat oysters of Ostrea stentina species complex and a new specie Ostrea neostentina sp. nov. (Bivalvia: Ostreidae). J. Shellfish Res. 2019, 38, 295–308. [Google Scholar] [CrossRef]
- Lavesque, N.; Daffe, G.; Londono-Mesa, M.H.; Hutchings, P. Revision of the French Terebellidae sensu stricto (Annelida, Terebelliformia), with descriptions of nine new species. Zootaxa 2021, 5038, 1–63. [Google Scholar] [CrossRef] [PubMed]
- Turon, X.; Casso, M.; Pascual, M.; Viard, F. Looks can be deceiving: Didemnum pseudovexillum sp. nov. (Ascidiacea) in European harbours. Mar. Biodivers. 2020, 50, 48. [Google Scholar] [CrossRef]
- Abbott, R.J. Plant invasions, interspecific hybridization and the evolution of new plant taxa. Trends Ecol. Evol. 1992, 7, 401–405. [Google Scholar] [CrossRef]
- Preston, C.D.; Pearman, D.A. Plant hybrids in the wild: Evidence from biological recording. Biol. J. Linn. Soc. 2015, 115, 555–572. [Google Scholar] [CrossRef]
- Ainouche, M.L.; Fornute, M.P.; Salmon, A.; Parisod, C.; Grandbastien, M.-A.; Fukunaga, K.; Ricou, M.; Misset, M.-T. Hybridization, polyploidy and invasion: Lessons from Spartina (Poaceae). Biol. Invasions 2009, 11, 1159–1173. [Google Scholar] [CrossRef]
- Ferreira de Carvalho, J.; Poulain, J.; Da Silva, C.; Wincker, P.; Michon-Coudouel, S.; Dheilly, A.; Naquin, D.; Boutte, J.; Salmon, A.; Ainouche, M. Transcriptome de novo assembly from next-generation sequencing and comparative analyses in the hexaploid salt marsh species Spartina maritima and Spartina alterniflora (Poaceae). Heredity 2013, 110, 181–193. [Google Scholar] [CrossRef]
- Mobberley, D. Taxonomy and distribution of the genus Spartina. Iowa State Coll J. Sci. 1956, 30, 471–574. [Google Scholar]
- Baumel, A.; Rousseau-Gueutin, M.; Sapienza-Bianchi, C.; Gareil, A.; Duong, N.; Rousseau, H.; Coriton, O.; Amirouche, R.; Sciandrello, S.; Duarte, B.; et al. Spartina versicolor Fabre: Another case of Spartina trans-Atlantic introduction? Biol. Invasions 2016, 18, 2123–2135. [Google Scholar] [CrossRef]
- Patrício, J.; Little, S.; Mazik, K.; Papadopoulou, K.N.; Smith, C.J.; Teixeira, H.; Hoffmann, H.; Uyarra, M.C.; Solaun, O.; Zenetos, A. European marine biodiversity monitoring networks: Strengths, weaknesses, opportunities and threats. Front. Mar. Sci. 2016, 3, 161. [Google Scholar] [CrossRef]
- Mauritzen, C.; Morel, Y.; Paillet, J. On the influence of Mediterranean Water on the Central Waters of the North Atlantic Ocean. Deep Sea Res. Part I Oceanogr. Res. Pap. 2001, 48, 347–381. [Google Scholar] [CrossRef]
- Carrasco, J.F. Excepcional presencia de Cymbium papillatum Schumacher (Grastropoda, Volutidae) en la costa catalana. Butll. Centre d’Est. Natura B-N 2000, 5, 67–68. [Google Scholar]
- Hidalgo, J. Fauna malacológica de España, Portugal y las Baleares: Moluscos Testáceos Marinos. Trab. del Mus. Nac. Ciencias Nat. Ser. Zool. 1917, 30, 1–752. [Google Scholar]
- Bachelet, G.; Cazaux, C.; Gantès, H.; Labourg, P. Contribution à l’étude de la faune marine de la région d’Arcachon, IX. Bull. Cent. Etud. Rech. Sci. Biarritz 1980, 13, 45–64. [Google Scholar]
- Simon-Bouhet, B.; Garcia-Meunier, P.; Viard, F. Multiple introductions promote range expansion of the mollusc Cyclope neritea (Nassariidae) in France: Evidence from mitochondrial sequence data. Mol. Ecol. 2006, 15, 1699–1711. [Google Scholar] [CrossRef]
- Sauriau, P.-G. Spread of Cyclope neritea (Mollusca: Gastropoda) along the north-eastern Atlantic coasts in relation to oyster culture and to climatic fluctuations. Mar. Biol. 1991, 109, 299–309. [Google Scholar] [CrossRef]
- Boissin, E.; Neglia, V.; Baksay, S.; Micu, D.; Bat, L.; Topaloglu, B.; Todorova, V.; Panayotova, M.; Kruschel, C.; Milchakova, N.; et al. Chaotic genetic structure and past demographic expansion of the invasive gastropod Tritia neritea in its native range, the Mediterranean Sea. Sci. Rep. 2020, 10, 21624. [Google Scholar] [CrossRef]
- Couceiro, L.; López, L.; Ruiz, J.M.; Barreiro, R. Population structure and range expansion: The case of the invasive gastropod Cyclope neritea in northwest Iberian Peninsula. Int. Zoo. 2012, 7, 286–298. [Google Scholar] [CrossRef]
- Gouillieux, B.; Ariyama, H.; Costa, A.C.; Daffe, G.; Marchini, A.; Micael, J.; Ulman, A. New records of Ericthonius didymus Krapp-Schickel, 2013 (Crustacea: Amphipoda: Ischyroceridae) in European waters with a focus in Arcachon Bay, France and key to Ericthonius species. J. Mar. Biol. Assoc. UK 2020, 100, 401–412. [Google Scholar] [CrossRef]
- Zenetos, A.; Gratsia, E.; Cardoso, A.; Tsiamis, K. Time lags in reporting of biological invasions: The case of Mediterranean Sea. Meditter. Mar. Sci. 2019, 20, 469–475. [Google Scholar] [CrossRef]
- Outinen, O.; Bailey, S.A.; Broeg, K.; Chasse, J.; Clarke, S.; Daigle, R.M.; Gollasch, S.; Kakkonen, J.E.; Lehtiniemi, M.; Normant-Saremba, M.; et al. Exceptions and exemptions under the ballast water management convention–Sustainable alternatives for ballast water management? J. Environ. Manag. 2021, 293, 112823. [Google Scholar] [CrossRef] [PubMed]
- EEA Trends in Marine Non-Indigenous Species. European Environment Agency. 2019. Available online: https://www.eea.europa.eu/data-and-maps/indicators/trends-in-marine-alien-species-mas-3/assessment (accessed on 1 September 2022).
- Lehtiniemi, M.; Outinen, O.; Puntila-Dodd, R. Citizen science provides added value in the monitoring for coastal non-indigenous species. J. Environ. Manag. 2020, 267, 110608. [Google Scholar] [CrossRef] [PubMed]
- Jalkanen, J.-P.; Johansson, L.; Wilewska-Bien, M.; Granhag, L.; Ytreberg, E.; Eriksson, K.M.; Yngsell, D.; Hassellöv, I.-M.; Magnusson, K.; Raudsepp, U.; et al. Modelling of Discharges from Baltic Sea Shipping. Ocean Sci. 2021, 17, 699–728. [Google Scholar] [CrossRef]
- Robbins, J.R.; Bouchet, P.J.; Miller, D.L.; Evans, P.G.; Waggitt, J.; Ford, A.T.; Marley, S.A. Shipping in the north-east Atlantic: Identifying spatial and temporal patterns of change. Mar. Pollut. Bull. 2022, 179, 113681. [Google Scholar] [CrossRef]
- OSPAR NIS3: Trends in New Records of Non-Indigenous Species Introduced by Human Activities, in: OSPAR (Ed.), OSPAR Intermediate Assessment OSPAR London UK. Available online: https://oap.ospar.org/en/ospar-assessments/intermediateassessment-2017/pressures-human-activities/non-indigenous/ (accessed on 10 August 2022).
- Bishop, J.; Wood, C.A.; Lévêque, L.; Yunnie, A.L.E.; Viard, F. Repeated rapid assessment surveys reveal contrasting trends in occupancy of marinas by non-indigenous species on opposite sides of the western English Channel. Mar. Pollut. Bull. 2015, 95, 699–706. [Google Scholar] [CrossRef]
- Ibabe, A.; Miralles, L.; Carleos, C.E.; Soto-López, V.; Menéndez-Teleña, D.; Bartolomé, M.; Montes, H.J.; González, M.; Dopico, E.; Garcia-Vazquez, E.; et al. Building on gAMBI in ports for a challenging biological invasions scenario: Blue-gNIS as a proof of concept. Mar. Environ. Res. 2021, 169, 105340. [Google Scholar] [CrossRef]
- Miralles, L.; Ibabe, A.; González, M.; García-Vázquez, E.; Borrell, Y.J. “If you know the enemy and know yourself”: Addressing the problem of biological invasions in ports through a new NIS invasion threat score, routine monitoring, and preventive action plans. Front. Mar. Sci. 2021, 8, 633118. [Google Scholar] [CrossRef]
- Pejovic, I.; Ardura, A.; Miralles, L.; Arias, A.; Borrell, Y.J.; Garcia-Vazquez, E. DNA barcoding for assessment of exotic molluscs associated with maritime ports in northern Iberia. Mar. Bio. Res. 2015, 12, 168–176. [Google Scholar] [CrossRef]
- Miralles, L.; Ardura, A.; Arias, A.; Borrell, Y.J.; Clusa, L.; Dopico, E.; de Rojas, A.; Lopez, B.; Muñoz-Colmenero, M.; Roca, A.; et al. Barcodes of marine invertebrates from north Iberian ports: Native diversity and resistance to biological invasions. Mar. Pollut. Bull. 2016, 112, 183–188. [Google Scholar] [CrossRef]
- Viard, F.; Roby, C.; Turon, X.; Bouchemousse, S.; Bishop, J. Cryptic diversity and database errors challenge non-indigenous species surveys: An illustration with Botrylloides spp. in the English Channel and Mediterranean Sea. Front. Mar. Sci. 2019, 6, 615. [Google Scholar] [CrossRef]
- Duarte, S.; Vieira, P.E.; Lavrador, A.S.; Costa, F.O. Status and prospects of marine NIS detection and monitoring through (e)DNA metabarcoding. Sci. Total Environ. 2021, 751, 141729. [Google Scholar] [CrossRef]
- Darling, J.A.; Pochon, X.; Abbott, C.L.; Inglis, G.J.; Zaiko, A.; Leroy, B. The risks of using molecular biodiversity data for incidental detection of species of concern. Divers. Distrib. 2020, 26, 1116–1121. [Google Scholar] [CrossRef]
- Pezy, J.-P.; Baffreau, A.; Raoux, A.; Rusig, A.-M.; Mussio, I.; Dauvin, J.-C. Non-indigenous species in marine and brackish waters along the Normandy coast. BioInvasions Rec. 2021, 10, 755–774. [Google Scholar] [CrossRef]
- Bishop, J.D.D.; Roby, C.; Yunnie, A.L.E.; Wood, C.A.; Leveque, L.; Turon, X.; Viard, F. The southern hemisphere ascidian Asterocarpa humilis is unrecognised but widely established in NW France and Great Britain. Biol. Invasions 2013, 15, 253–260. [Google Scholar] [CrossRef][Green Version]
- Zenetos, A.; Albano, P.G.; Garcia, E.L.; Stern, N.; Tsiamis, K.; Galanidi, M. Corrigendum to the Review Article. Meditter. Mar. Sci. 2022, 23, 196–212, Erratum in 2022, 23, 876–878. [Google Scholar] [CrossRef]
- Zenetos, A.; Galanidi, M. Mediterranean non indigenous species at the start of the 2020s: Recent changes. Mar. Biodivers. Rec. 2020, 13, 10. [Google Scholar] [CrossRef]
- Raitsos, D.E.; Beaugrand, G.; Georgopoulos, D.; Zenetos, A.; Pancucci-Papadopoulou, A.M.; Theocharis, A.; Papathanassiou, E. Global climate change amplifies the entry of tropical species into the Eastern Mediterranean Sea. Limnol. Oceanogr. 2010, 55, 1478–1484. [Google Scholar] [CrossRef]
- Zenetos, A. Mediterranean Sea: 30 years of biological invasions (1988–2017). In Proceedings of the 1st Mediterranean Symposium on the Non-Indigenous Species, Antalya, Turkey, 17–18 January 2019; Langar, H., Ouerghi, A., Eds.; SPA/RAC Publiction: Tunis, Tunis, 2019. 116p. [Google Scholar]
- Mavrič, B.; Orlando-Bonaca, M.; Fortič, A.; Francé, J.; Mozetič, P.; Slavinec, P.; Pitacco, V.; Trkov, D.; Vascotto, I.; Zamuda, L.L.; et al. Monitoring Species Diversity and Abundance of Non-Indigenous Species in the Slovenian Sea (in Slovenian). Final Project report. Marine Biology Station Piran, National Institute of Biology, Report 195, p. 83. Available online: http://www.ribiski-sklad.si/f/docs/Dokumenti_1/koncno_porocilo_NIS_2018-2021_s_CIP.pdf (accessed on 15 May 2022).
- Ferrario, J.; Caronni, S.; Occhipinti-Ambrogi, A.; Marchini, A. Role of commercial harbours and recreational marinas in the spread of non-indigenous fouling species. Biofouling 2017, 33, 651–660. [Google Scholar] [CrossRef]
- Ulman, A.; Ferrario, J.; Occhipinti-Ambrogi, A.; Arvanitidis, C.; Bandi, A.; Bertolino, M.; Bogi, C.; Chatzigeorgiou, G.; Çiçek, B.A.; Deidun, A.; et al. A massive update of non-indigenous species records in Mediterranean marinas. PeerJ 2017, 5, 1–59. [Google Scholar]
- Bulgurkov, K. Occurrence of Callinectes sapidus Rathbun (Crustacea—Decapoda) in Black Sea. Bull. Natl.Inst. Ocean. Fish Varna 1968, 9, 97–99. (In Bulgarian) [Google Scholar]
- Stefanov, T. Recent expansion of the alien invasive blue crab Callinectes sapidus (Rathbun, 1896)(Decapoda, Crustacea) along the Bulgarian coast of the Black Sea. Hist. Nat. Bulg. 2021, 42, 49–53. [Google Scholar] [CrossRef]
- Gücü, A.C.; Ünal, V.; Ulman, A.; Morello, E.B.; Bernal, M. Management responses to non-indigenous species in response to climate change. In Adaptive Management to Fisheries FAO Fisheries and Aquaculture Technical Paper 667; Bahri, T., Vasconcellos, M., Welch, D.J., Johnson, J., Perry, R.I., Ma, X., Eds.; FAO: Rome, Italy, 2021; pp. 161–176. [Google Scholar]
- Maltsev, V.I.; Kulish, A.V.; Beletskaya, M.A. The First Find of the Marbled Spinefoot Siganus rivulatus (Siganidae) in the Black Sea. J. Ichthyol. 2022, 62, 514–516. [Google Scholar] [CrossRef]
- Boltachova, N.A.; Lisitskaya, E.V.; Podzorova, D.V. Distribution of alien polychaetes in biotopes of the northern part of the Black Sea. Russ. J. Biol. Invasions 2021, 12, 11–26. [Google Scholar] [CrossRef]
- Teacă, A.; Begun, T.; Menabit, S.; Mureșan, M. The First Record of Marenzelleria neglecta and the Spread of Laonome xeprovala in the Danube Delta–Black Sea Ecosystem. Diversity 2022, 14, 423. [Google Scholar] [CrossRef]
- Lavesque, N.; Daffe, G.; Bonifácio, P.; Hutchings, P. A new species of the Marphysa sanguinea complex from French waters (Bay of Biscay, NE Atlantic) (Annelida, Eunicidae). Zookeys 2017, 716, 1–17. [Google Scholar] [CrossRef]
- Lavesque, N.; Hutchings, P.; Abe, H.; Daffe, G.; Gunton, L.M.; Glasby, C.J. Confirmation of the exotic status of Marphysa victori Lavesque, Daffe, Bonifácio & Hutchings, 2017 (Annelida) in French waters and synonymy of Marphysa bulla Liu, Hutchings & Kupriyanova. Aquat. Invasions 2018, 15, 355–366. [Google Scholar] [CrossRef]
- O’Donoghue, C.H.; White, K.M. A collection of marine molluscs, mainly opisthobranchs, from Palestina. Proc. Malacol. Soc. Lond. 1940, 24, 92–96. [Google Scholar]
- Bariche, M.; Al-Mabruk, S.A.; Ates, M.A.; Büyük, A.; Crocetta, F.; Dritsas, M.; Edde, D.; Fortic, A.; Gavriil, E.; Gerovasileiou, V.; et al. New Alien Mediterranean Biodiversity Records Meditter. Mar. Sci. 2020, 21, 129–145. [Google Scholar] [CrossRef]
- Tillier, L.; Bavay, A. Les mollusques testacés du Canal de Suez. Bull. de la Soc. Zool. de France 1905, 30, 170–181. [Google Scholar]
- Zenetos, A.; Konstantinou, F.; Konstantinou, G. Towards homogenization of the Levantine alien biota: Additions to the alien molluscan fauna along the Cypriot coast. Mar. Biodivers. Rec. 2009, 2, E156. [Google Scholar] [CrossRef]
- Olenin, S.; Narščius, A.; Gollasch, S.; Lehtiniemi, M.; Marchini, A.; Minchin, D.; Srėbalienė, G. New arrivals: An indicator for non-indigenous species introductions at different geographical scales. Front. Mar. Sci. 2016, 3, 208. [Google Scholar] [CrossRef]
- UNEP/MAP. Integrated Monitoring and Assessment Programme of the Mediterranean Sea and Coast and Related Assessment Criteria; UN Environment/MAP: Athens, Greece, 2017; Available online: https://wedocs.unep.org/bitstream/handle/20.500.11822/17012/imap_2017_eng.pdf?sequence=5&isAllowed=y (accessed on 28 October 2021).
- UNEP(DEPI)/MED. Decision IG.21/3 on the Ecosystems Approach Including Adopting Definitions of Good Environmental Status (GES) and Targets; UNEP(DEPI)/MED IG.21/9, Annex II—Thematic Decisions; UNEP(DEPI)/MED: Istanbul, Turkey, 2013. [Google Scholar]
- Vasilakopoulos, P.; Palialexis, A.; Boschetti, S.T.; Cardoso, A.C.; Druon, J.-N.; Konrad, C.; Kotta, M.; Magliozzi, C.; Palma, M.; Piroddi, C.; et al. Marine Strategy Framework Directive, Thresholds for MSFD Criteria: State of Play and Next Steps; EUR 31131 EN; Publications Office of the European Union: Luxembourg, 2022; ISBN 978-92- 76-53689-5. [Google Scholar] [CrossRef]
- Cavallo, M.; Elliott, M.; Quintino, V.; Touza, J. Can National Management Measures Achieve Good Status across International Boundaries? A Case Study of the Bay of Biscay and Iberian Coast Sub-Region. Ocean Coast. Manag. 2018, 160, 93–102. [Google Scholar] [CrossRef]
- WG-AS; Gittenberger, A. Trilateral Wadden Sea Alien Species Management and Action Plan; Busch, J.A., Lüerssen, G., de Jong, F., Eds.; Common Wadden Sea Secretariat (CWSS): Wilhelmshaven, Germany, 2018. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).