Abstract
Long-term continuous monoculture cultivation harms soil physicochemical and microbial communities in agricultural practices. However, little has been reported on the effect of continuous cropping of industrial hemp on bacterial community and diversity in the rhizosphere soil. Our study investigated the changes in physicochemical properties and bacterial communities of industrial hemp rhizosphere soils in different continuous cropping years. The results showed that continuous cropping would reduce soil pH and available phosphorus (AP), while electrical conductivity (EC), available nitrogen (AN), and available potassium (AK) would increase. Soil bacterial diversity and richness index decreased with continuous cropping years. At the same time, continuous cropping marked Acidobacteria, Bacteroidetes, and Gemmatimonadetes increase, and the Proteobacteria and Actinobacteria decreased. Moreover, we found that pH, AK, and AP were the critical factors associated with the changes in the abundance and structure of the bacterial community. Overall, our study first reported the effect of continuous cropping on the rhizosphere soil microflora of industrial hemp. The results can provide a theoretical basis for revealing the obstacle mechanism of continuous cropping of industrial hemp and contribute to the sustainable cultivation of industrial hemp in the future.
1. Introduction
Industrial hemp (Cannabis sativa L.) refers to the type of cannabis with ∆9-tetrahydrocannabinol (THC) less than 0.3% and non-toxic use-value [1]. As an annual herb, it has a wide geographical range of cultivation, relatively low production input, and many industrial uses—mainly textile, medical, food, cosmetics, building materials, and other fields, making it a multifunctional and sustainable cash crop [1,2,3,4]. In the last ten years, with the expansion of global planting area and the increase of continuous cropping area, industrial hemp is also facing the same continuous cropping obstacles as traditional crops, including the decline of yield and quality, plant death, and a decreased or non-existent harvest and so on [5,6,7].
The root cause of continuous cropping obstacles may be that monoculture reduces crops and biological diversity, resulting in the degradation of the soil ecosystem, including vulnerability to erosion, weed invasion, an increase of soil-borne bacteria, soil acidification, and imbalance of soil microbial flora [8,9]. Rhizosphere soil is directly affected by plant roots, and its microbial composition is closely related to the transformation and utilization of soil nutrients, which directly affects crop growth [10]. Bacteria are the most abundant flora in soil microorganisms, and they play a vital role in water transport, organic matter decomposition, nutrient cycling, and maintaining sustainable soil productivity to promote plant growth [11,12]. Some omics-based studies have also proved the crucial role of rhizosphere bacteria in inhibiting soil diseases [13]. In addition, some previous studies have shown that continuous cropping can reduce beneficial bacteria in rhizosphere soil and increase harmful bacteria [14,15]. The α-diversity of the rhizosphere soil bacteria decreased with continuous cropping years, and pH and organic matter had significant effects on the structure of the bacterial community [16]. Therefore, it is of great significance to explore the diversity, community composition, and influencing factors of rhizosphere bacteria in the process of continuous cropping of industrial hemp for improving crop soil biological environment, reducing continuous cropping obstacles, and enhancing crop productivity [17,18].
The adverse effects of continuous cropping on rhizosphere soil microorganisms have been confirmed in many crops, including traditional cash crops such as tomato, soybean, corn, and wheat [19,20,21]. In contrast, due to people’s views on the toxicity of cannabis for a long time, there is less information related to cannabis cultivation, mainly focusing on the identification of subspecies [22], the germination ability of seeds [23], and the adsorption of soil pollutants [24]. Therefore, based on high throughput sequencing technology, we studied the changes of soil bacterial community in the rhizosphere of industrial hemp after continuous cropping for the first time and analyzed the correlation between soil physical and chemical properties and bacterial community. Our findings help provide theoretical insights into the barrier mechanism of continuous cropping and sustainable planting of industrial hemp.
2. Materials and Methods
2.1. Site Description
The experimental site was established at the scientific research farm of Daqing branch, Academy of Agricultural Sciences in Heilongjiang Province, China (46°40′ N, 125°13′ E). This region belongs to the northern temperate continental monsoon climate zone, with cold and snowy winter and many spring and autumn monsoons. The annual average temperature is 4.2 °C, the average annual frost-free period is 143 days, the average annual precipitation is 427.5 mm, and the altitude is 145 m. Before planting industry hemp, the land use type of the experimental site was a cornfield. The soil type is chernozem (Haplic Chernozem, FAO).
2.2. Experimental Design and Sample Collection
Industrial hemp was planted in May every year for five consecutive years (2016–2021), and the harvest period was the end of the September of the year. Industrial hemp was conventionally cultivated for fiber, and the field management method (only apply 20% nitrogen fertilizer of 450 kg ha−1 at each sowing time) was the same within five years to keep the nitrogen available for plants constant according to our previous study [25]. The industrial hemp variety was “Qinghemp No. 1”. The soil within 2 mm of the cannabis root surface as the rhizosphere soil samples were collected after the first [26], second, third, fourth, and fifth years of continuous monoculture, respectively. The planting area of five treatments was 0.12 ha, respectively, and each planting area was divided into three sample plots. Ten rhizosphere soil samples were collected from the sample plot using the S-sampling method and mixed as biological replicates. The collected samples were immediately put into sterile bags, transferred into portable refrigerators, and returned to the laboratory. Then, all samples were homogenized through a 2 mm sieve and further divided into two parts: one was dried to determine soil physical and chemical properties, while the other was stored in a refrigerator at −20 °C for DNA determination within one week.
2.3. Soil DNA Extraction and PCR Amplification of 16S rRNA
The genomic DNA was extracted from 0.5 g rhizosphere soil samples using the PowerSoil DNA Isolation Kit (MoBio, Carlsbad, CA USA) following the manufacturer’s instructions. The final DNA concentration and purification were quantified by a NanoDrop spectrophotometer (Thermo Fisher Scientific, Wilmington, NC, USA), and DNA quality was checked by 1% agarose gel electrophoresis. The V3-V4 hypervariable region of the 16S rRNA (341 F 5′-ACTCCTACGGGAGGCAGCA-3′ and 806R F 5′-GGACTACHVGGGTWTCTAAT-3′) was amplified for assessment of the bacteria [27]. The PCR product was excised from the 1.5% agarose gel and purified using a QIAquick Gel Extraction Kit (Qiagen, Hilden, Germany), quantified using real-time PCR, and sequenced at Sangon Biotech Co., Ltd., Shanghai, China.
2.4. Illumina MiSeq Sequencing and Processing of Sequencing Date
High-throughput sequencing was commissioned on an illumine HiSeq 400 platform (Illumina, San Diego, CA, USA) by Sangon Biotech Co., Ltd., Shanghai, China. The raw sequence data were first screened, and the high-quality reads, containing at least 200 bp length and having a minimum average quality of 25 Phred score, were remained. Chimeric sequences were checked using UCHIME in de novo mode, as implemented in MOTHUR and removed from the datasets. The cleaned sequences were clustered into OTUs at a similarity level of 97% to sequence identity and assigned taxonomy using the SILVA 132 database for bacterial 16S rRNA gene. The raw sequences were deposited in the National Center for Biotechnology Information (NCBI) Sequence Read Archive with BioProject accession number: PRJNA809891.
2.5. Determination of Soil Physicochemical Properties
After the soil samples were passed through a 2 mm sieve, the soil pH and soil electrical conductivity (EC) was measured by the glass electrode method in a 1:5 (w/v) suspension of soil in water. The soil organic matter (SOM) content was determined by the potassium dichromate oxidation external heating method. The available soil nitrogen (AN) content was determined by the alkali hydrolyzable diffusion method. The available soil phosphorus (AP) content was extracted with 0.5 M NaHCO3 (pH 8.5) and determined by molybdenum blue colorimetry. An ammonium acetate extraction flame photometer determined the soil available potassium (AK) content.
2.6. Data Analysis and Statistics
The physicochemical data of rhizosphere soil were analyzed by one-way analysis of variance (ANOVA) followed by Duncan’s multiple range tests (p < 0.05) in SPSS v24.0 (BM Corporation, New York, USA). Chao1, Shannon, Simpson, and Coverage indexes were calculated with Mothur v.1.21.1 [27]. The system clustering method was used to classify the samples at the genus level with the R Stats package. The degree of bacterial differences between samples was analyzed based on principal component analysis (PCA) with the R Vegan package. Redundancy analysis (RDA) and spearman’s correlation heat map were performed to analyze the correlations between soil physicochemical properties and bacterial community composition with the R Vegan package.
3. Results
3.1. Soil Physical and Chemical Properties of Industrial Hemp in Different Continuous Cropping Years
The rhizosphere soil nutrient content in the soil of industrial hemp was shown in Table 1. With the increase of continuous cropping years, soil pH, and AP gradually decreased, while EC and AN gradually increased, and soil AK first increased and then decreased. In addition, there was no difference in the change of SOM (p < 0.05).

Table 1.
Effect of different continuous cropping years on rhizosphere soil nutrient in industrial hemp.
3.2. Effects of Different Continuous Cropping Years on Bacterial Diversity of Rhizosphere Soil
The indexes of soil bacterial richness (ACE and Chao1) and diversity (Shannon and Simpson) are shown in Table 2. In general, Chao1, ACE, Shannon and Simpson indexes showed a downward trend with the increase of continuous cropping years. In addition, the coverage index of soil bacteria in different continuous cropping years was 0.99, indicating that the sequencing depth was good and could reflect the real situation of microorganisms in the sample.

Table 2.
Effect of different continuous cropping years on bacterial diversity index of rhizosphere soil in industrial hemp.
3.3. Effects of Different Continuous Cropping Years on the Bacterial Community of Rhizosphere Soil
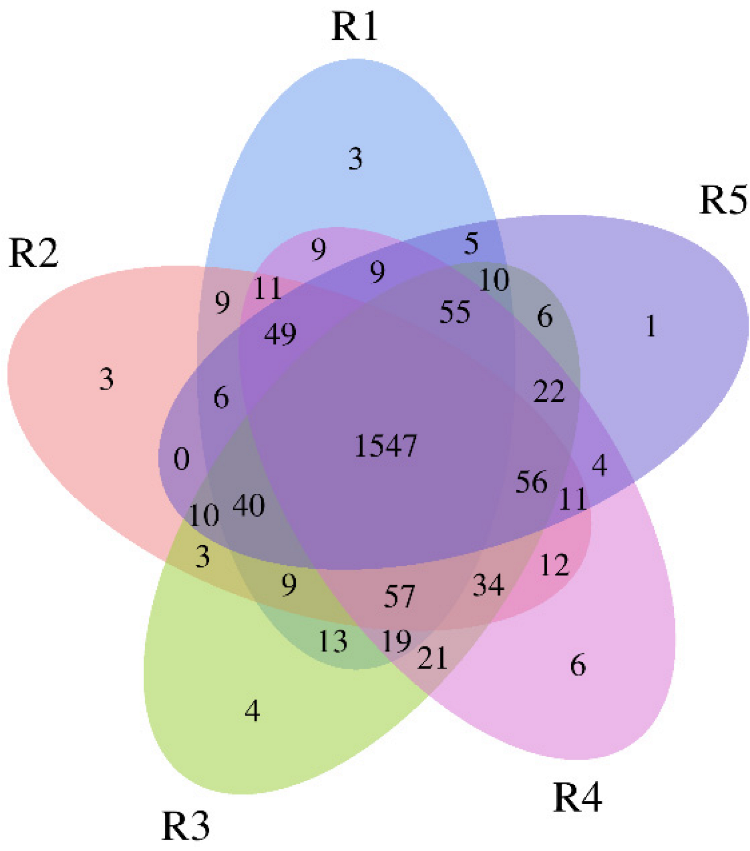
In our study, a total of 2044 bacterial OTUs at 97% similarity were obtained, and the R1, R2, R3, R4, and R5 groups obtained 1851, 1857, 1906, 1922, and 1831 bacterial OTUs, respectively (Figure 1). The number of shared OTUs in the five different groups was 1547, accounting for 75.68% of the fungal OTUs in all groups, indicating the bacterial groups in the five different continuous cropping years were more similar.
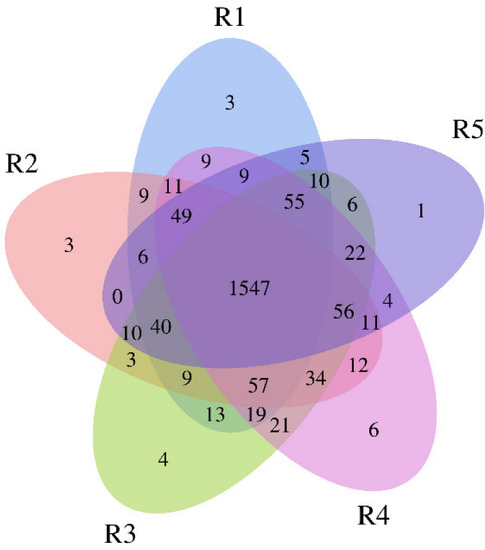
Figure 1.
Venn diagram of bacterial OTUs for the soil samples under continuous cropping of industrial hemp. R1, R2, R3, R4, and R5 represent planting years for 1, 2, 3, 4, and 5 years, respectively.
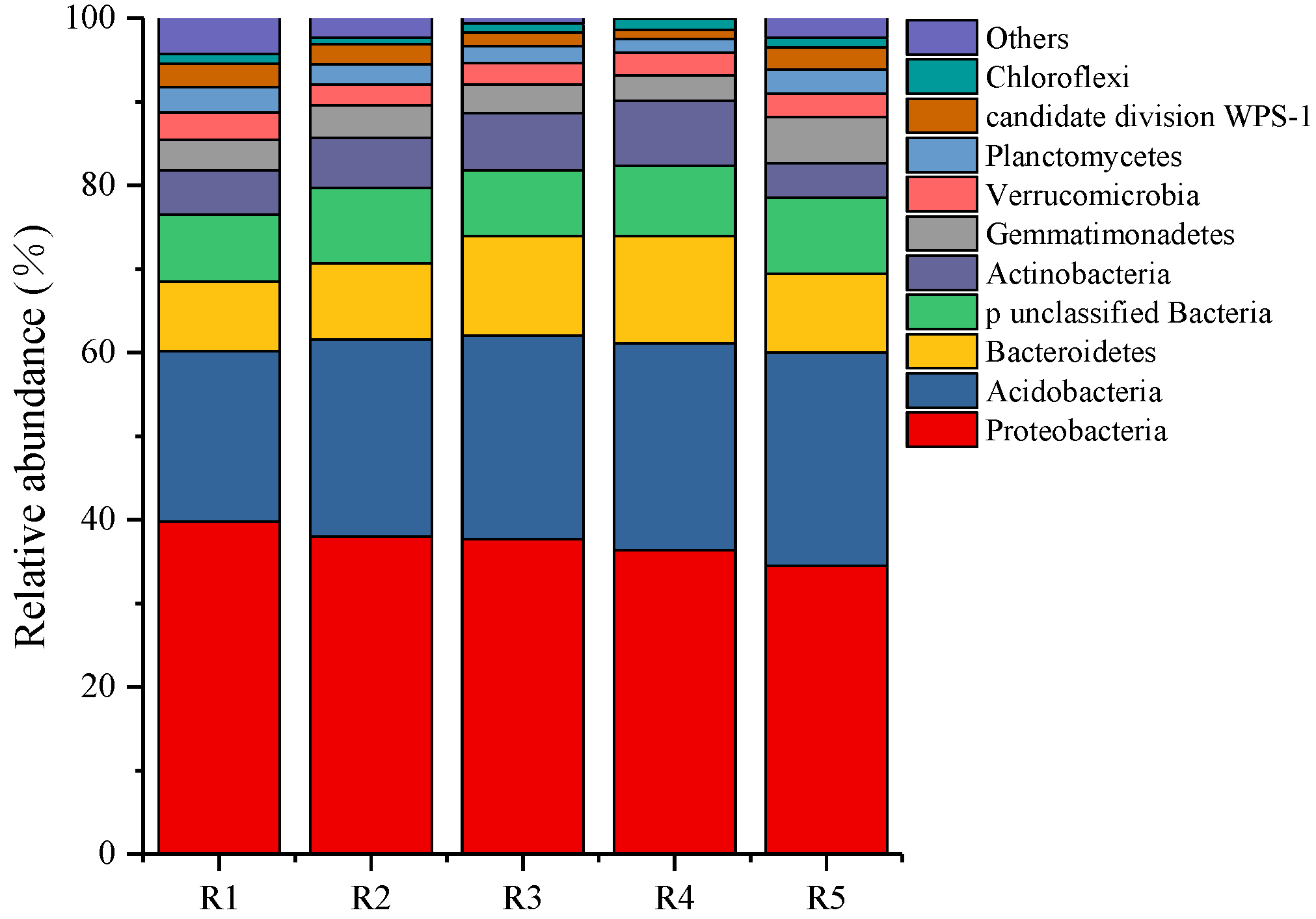
At the bacterial phylum level (Figure 2), the dominant phyla of all samples were Proteobacteria (34.47–39.81%), Acidobacteria (20.34–25.58%), Bacteroidetes (8.37–12.87%), Actinobacteria (4.18–7.77%), and Gemmatimonadetes (3.04–5.53%). Among them, with the increase of continuous cropping years, the relative abundance of Proteobacteria and Actinobacteria decreased gradually, while the relative abundance of Acidobacteria, Bacteroidetes, and Gemmatimonadetes increased gradually.
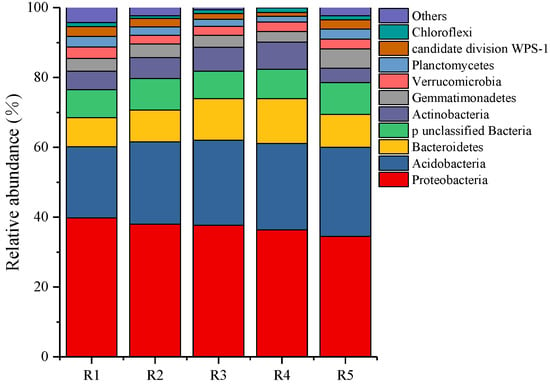
Figure 2.
The relative abundance of bacterial community composition in the phylum level. R1, R2, R3, R4, and R5 represent planting years for 1, 2, 3, 4, and 5 years, respectively.
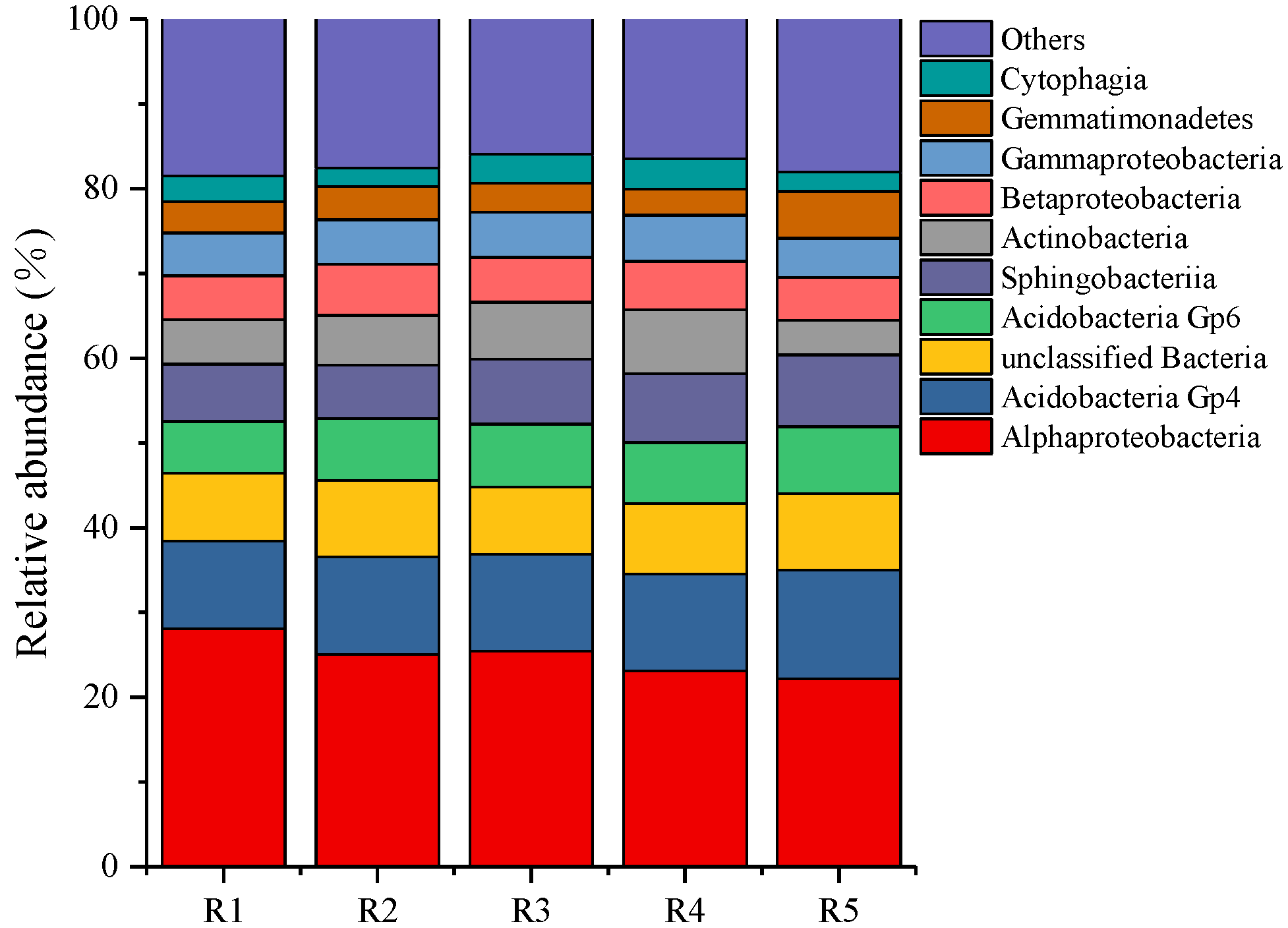
At the bacterial class level (Figure 3), the dominant bacteria of all samples were Alphaproteobacteria (22.20–28.06%), Acidobacteria Gp4 (10.37–12.79%), Acidobacteria GP6 (6.11–7.87%), Sphingobacteriia (6.31–8.49%), Actinobacteria (4.09–7.54%), Betaproteobacteria (5.08–6.05%), Gammaproteobacteria (4.62–5.47%), Gemmatimonadetes (3.04–5.53%). and Cytophagia (2.22–3.61%).
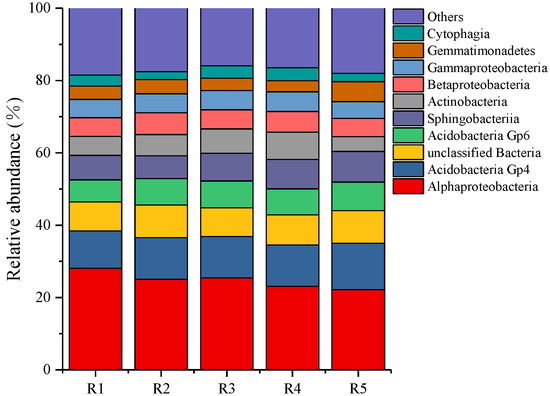
Figure 3.
The relative abundance of bacterial community composition in classes level. R1, R2, R3, R4, and R5 represent planting years for 1, 2, 3, 4, and 5 years, respectively.
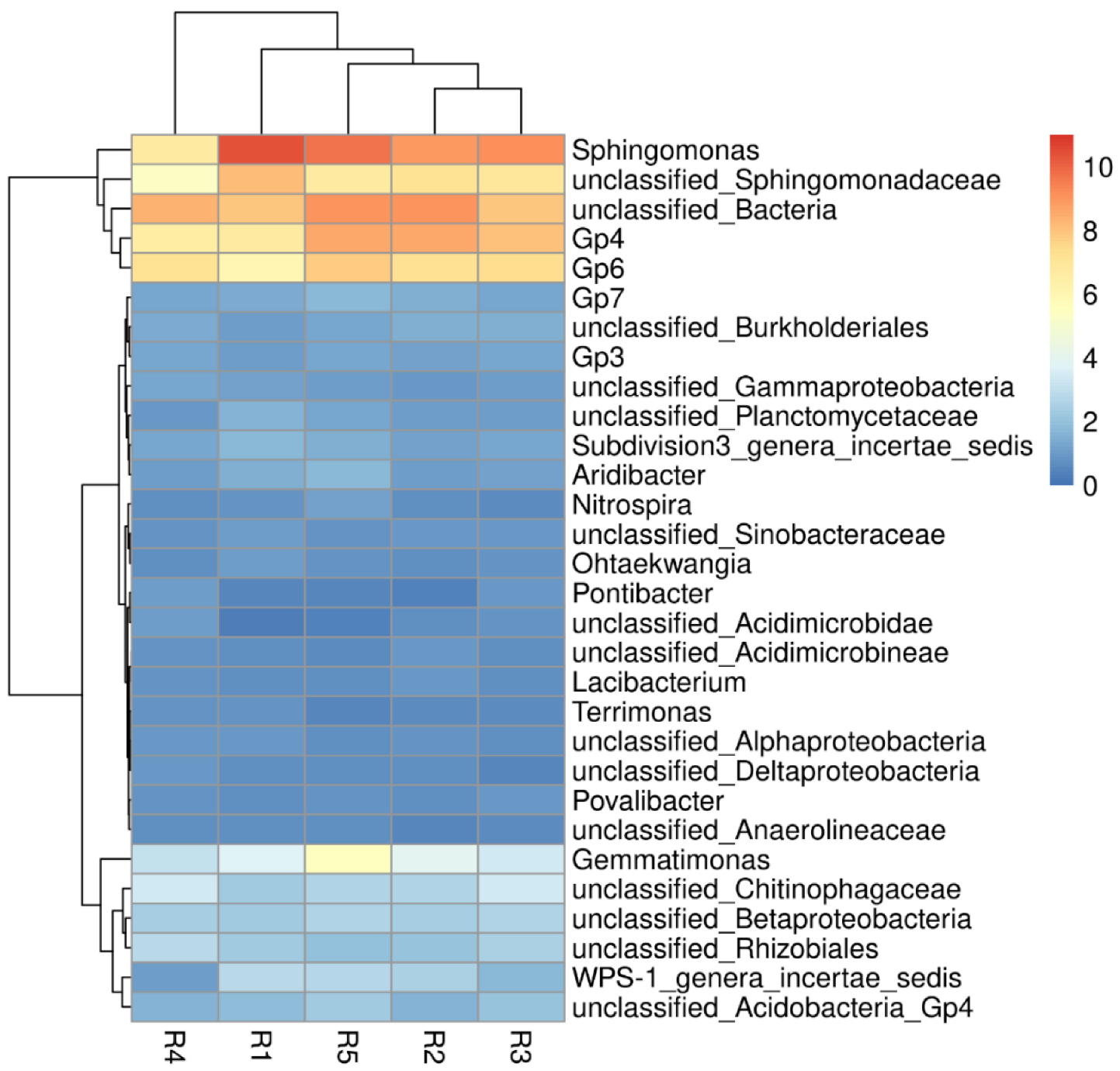
At the bacterial genus level (Figure 4), Sphingomonas (6.68–10.43%), Gp4 (6.61–8.58%) and GP6 (6.11–7.87%) were the top three bacterial genera in all rhizosphere soils. The other major bacterial genera were Gemmatimonas (3.04–5.53%), WPS-1 genera incertae sedis (1.10–2.81%), Gp7 (1.25–1.76%), Subdivision3 genera incertae Sedis (1.28–1.69%), and Aridibacter (1.02–1.72%). With the increase of continuous cropping years, the relative abundance of Sphingomonas decreased gradually, while the relative abundance of Gp4 and GP6 increased gradually.
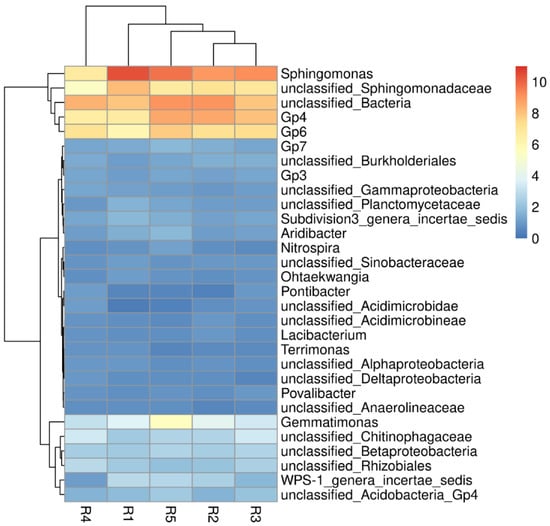
Figure 4.
Microbial community heatmap and cluster analysis of the bacterial community composition in genus level. R1, R2, R3, R4, and R5 represent planting years for 1, 2, 3, 4, and 5 years, respectively.
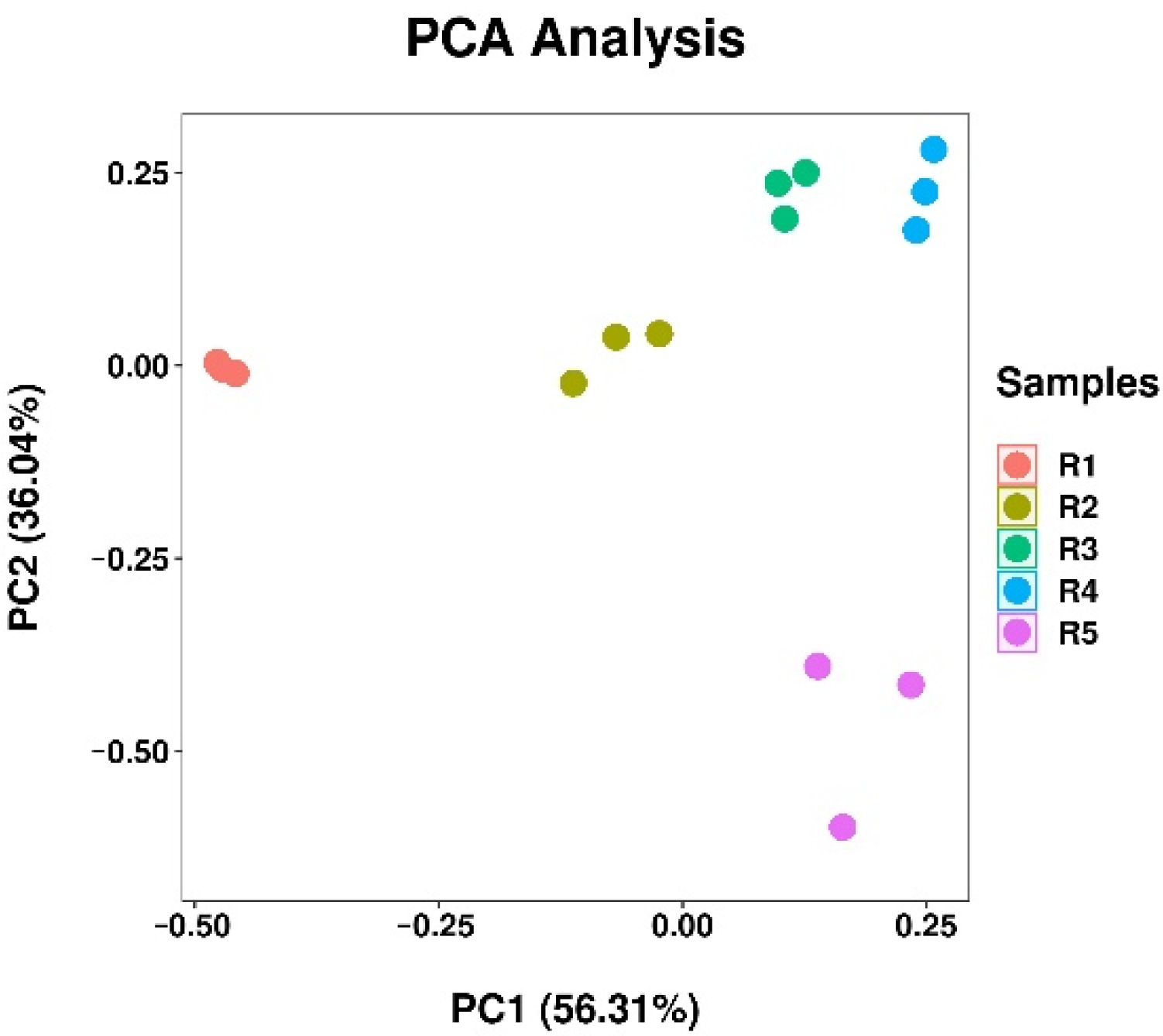
PCA analysis was carried out on rhizosphere soil bacteria out in different continuous cropping years (Figure 5). The results showed that the first principal component explained 56.31% of the variation, the second principal component explained 36.04% of the variation, and the two principal components explained 92.35% of the total variation. With the increase of continuous cropping time, the distance between rhizosphere soil samples from different times was getting farther and farther, which shows that with the increase of continuous cropping years, the difference between bacterial communities was also increasing, and continuous cropping leads to the change of bacterial community structure in cannabis rhizosphere soil.
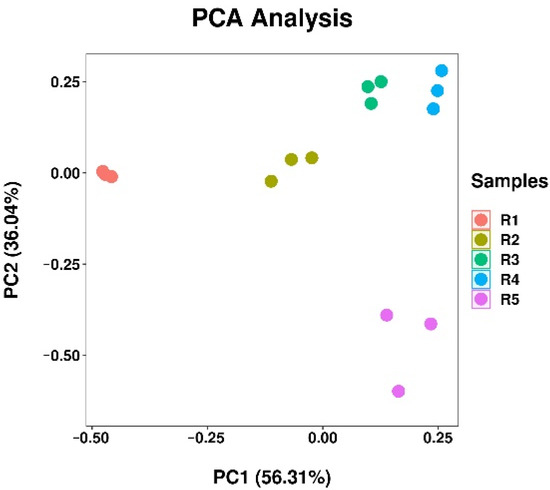
Figure 5.
Principal component analysis of soil bacterial community. R1, R2, R3, R4, and R5 represent planting years for 1, 2, 3, 4, and 5 years, respectively.
3.4. Effect of Soil Factors on Bacterial Communities
The RDA results of the top ten bacterial phyla and soil environmental factors in the rhizosphere soil of industrial hemp with different continuous cropping years are shown in Figure 6. RDA1 and RDA2 explained 39.82% and 26.33% of the total variation, respectively. The effects of soil properties on bacterial community structure were pH > AK > AP > EC > AN > SOM. The results showed that pH was a strong predictor of bacterial community composition in rhizosphere soil of continuous cropping cannabis.
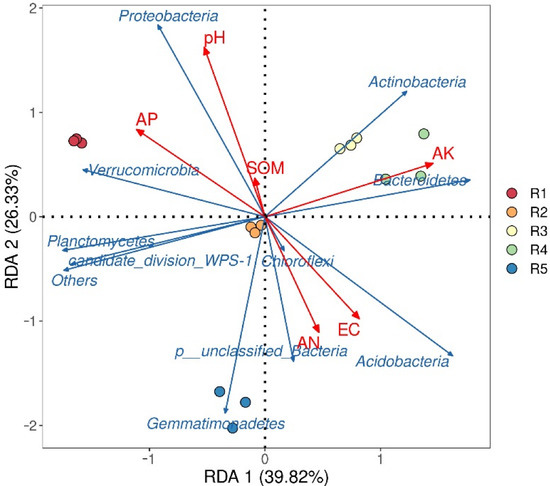
Figure 6.
Redundancy analysis of the 10 dominant bacterial phyla and soil physicochemical properties. R1, R2, R3, R4, and R5 represent planting years for 1, 2, 3, 4, and 5 years, respectively. EC, electrical conductivity; SOM, soil organic matter; AN, available nitrogen; AP, available phosphorus; AK, available potassium.
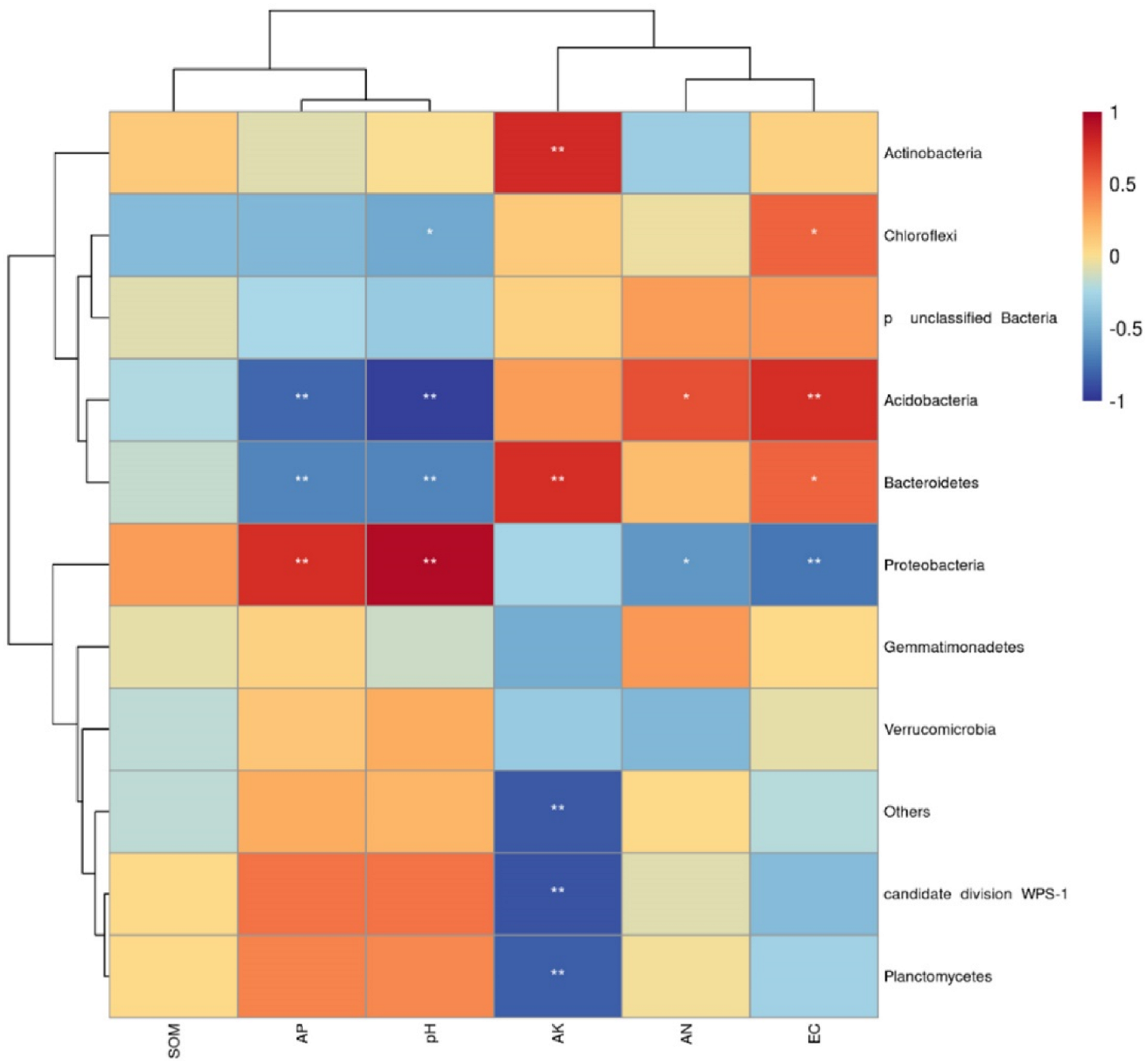
In addition, the results of Spearman correlation coefficient analysis are shown in Figure 7. pH has a significant positive correlation with Proteobacteria and a significant negative correlation with Bacteroidetes and acidobacteria. AK was significantly positively correlated with Bacteroides and actinobacteria, and with candidate division. There was a significant negative correlation between WPS-1 and Planctomycetes. At the same time, it can be seen from Figure 7 that the physical and chemical properties of soil can be divided into two groups, SOM, AP and pH were one group, and the rest were divided into another group, indicating that the effects of SOM, AP and pH on bacterial community structure were similar, but different from other groups.
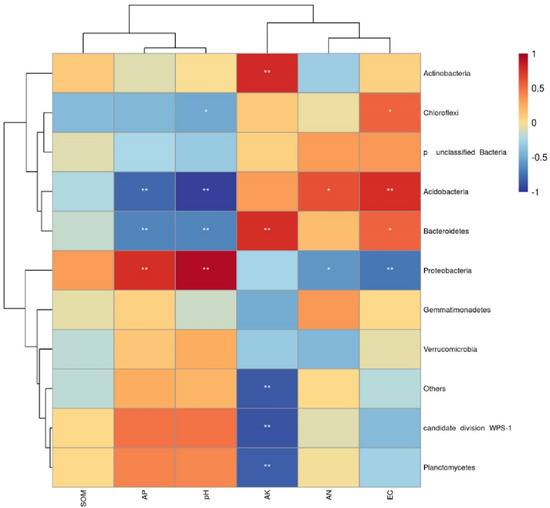
Figure 7.
Correlations between the 10 dominant bacterial phyla and soil properties. * p < 0.05; ** p < 0.01. EC, electrical conductivity; SOM, soil organic matter; AN, available nitrogen; AP, available phosphorus; AK, available potassium.
4. Discussion
A continuous cropping obstacle is widely defined as the fact that planting a single crop on the same land for 3 years or more will reduce crop yield [28,29]. However, due to the differences between crops, the causes of continuous cropping obstacles have not been fully revealed [5,16,18]. Traditional continuous monoculture will lead to a series of problems, such as the deterioration of soil physical and chemical properties, the reduction of soil biological enzyme activity, the accumulation of autotoxic substances, and the imbalance of soil microbial community [28,30], which will eventually lead to the decline of crop yield and quality and the reduction of disease resistance [31].
Previous studies have shown that continuous cropping will affect rhizosphere soil nutrients, thus reducing the ability of plant roots to obtain nutrients [32]. In this study, with the increase of continuous cropping years, pH and AP gradually decreased (Table 1), while EC, AN, and AK gradually increased, which may be caused by an unreasonable fertilization system. Although the use of nitrogen fertilizer alleviates the demand for AN of industrial hemp to a certain extent, the input of excessive nitrogen will lead to soil acidification [33]. With the increase of continuous cropping years, the utilization ability of rhizosphere microorganisms to potassium and phosphorus decreased [34,35]. In addition, after the 4th and 5th years of continuous cropping, the EC Value in soil increased significantly, indicating that continuous cropping increased the risk of secondary salinization of soil [36]. In conclusion, the changes in soil physicochemical properties are likely to limit the growth of industrial hemp, resulting in the decline of the yield and quality of industrial hemp after continuous cropping.
The diversity and composition of soil bacteria show different responses to soil physical and chemical properties, which are often related to crops’ health [37,38]. Many studies showed significant differences in soil microbial biomass and community composition during continuous cropping [13,19,20,32]. In our study, the soil bacterial diversity index and richness index decreased with the increase of continuous cropping years (Table 2). Recent research firstly reported that industrial hemp root rot was closely related to Pythium in soil [39]. Therefore, it can be speculated that in our study, after 5 years of continuous cropping treatment, a certain amount of autotoxic substances accumulated in the rhizosphere soil of industrial hemp, which inhibited the growth of soil bacteria, reduced the diversity and richness of the bacterial community, and there may be the risk of plant diseases. Furthermore, we found Proteobacteria and Acidobacteria were the dominant bacterial phyla in the bacterial communities at all stages of monoculture (Figure 2), which was consistent with published studies [17,33]. Proteobacteria are enriched in the rhizosphere soil of many plants, which can decompose organic matter and promote plant growth [17,33]. Acidobacteria usually exists in nutrient-poor and highly acidic soil environments and have the ability to degrade complex and stubborn carbon sources [40]. In our study, the relative abundance of Proteobacteria decreased with continuous cropping years, while the relative abundance of Acidobacteria increased, indicating that the soil bacterial community found changes to adapt to the changes in soil properties. In addition, our study also showed that Actinomycetes could produce antibiotics to inhibit plant pathogens in the soil, and play an essential role in the decomposition of organic matter [41], decreased with continuous cropping (Figure 2). Conversely, Gemmatimonadetes, a harmful bacteria that can lead to N loss and reduce crop growth [17], increased with continuous cropping.
At the genus level (Figure 4), Sphingomonas, Gp4, and GP6 had higher relative abundances in our study. Gp4 and GP6 belong to Acidobacteria and they could be used as indicators of a poor soil environment, while the relative abundance of Gp4 and GP6 increases significantly with continuous cropping years, indicating that the nutritional status of continuous cropping soil deteriorates [40]. However, Sphingomonas shows the ability of nitrogen fixation and denitrification in the soil nitrogen cycling, and can reduce the accumulation of toxic substances in soil [33,42]. Therefore, even under the condition of single cultivation, increasing the relative abundance of Sphingomonas may contribute to the healthy growth of industrial hemp in poor soil. Future research must consider screening beneficial bacterial species for inoculation and cultivation of industrial hemp and further study the interaction mechanism between beneficial bacteria and host.
The correlation between specific microbial communities and soil physicochemical properties can provide information on how microbial community composition affects soil nutrient cycling. In this study, the results of RDA showed that pH was the most important factor affecting the structure of the soil bacterial community (Figure 6). Soil pH is closely related to microbial nutrient availability [43]. Bacteria can effectively use amino acids and carbohydrates and have a wider metabolic capacity at higher pH [44]. When the pH decreases, it will decline bacterial diversity because most bacterial groups have poor tolerance to growth [19]. In addition, in the bacterial community, we found that Acidobacteria and Bacteroidetes had a significant negative correlation with pH and a significant positive correlation with Proteobacteria (Figure 7), which further supported the results of bacterial community succession. Therefore, the changes in soil physicochemical properties caused by long-term continuous cropping may be the main factor driving the success of the bacterial community and may also be one of the main reasons hindering the continuous cropping of industrial hemp.
5. Conclusions
The 5-year continuous cropping of industrial hemp led to a decrease in biodiversity indices linked to soil bacterial OTUs, which means that the rhizosphere soil has changed, despite having kept the parameters related to the cultivation of industrial hemp constant and field management. At the same time, the relative abundance of beneficial bacteria was significantly reduced, while the increase of bacteria taxons, including potentially harmful bacteria to plant growth, resulting in changes in the bacterial community structure. Continuous cropping reduced the soil pH, resulting in a decrease in soil bacterial richness and diversity, adaptively changed the composition of bacterial community structure, and broke the original soil micro ecological balance, which may be one of the important reasons for the obstacles of continuous cropping of industrial hemp. Furthermore, it is not easy to artificially put into the soil what it lacks after a crop to remain in equilibrium over time, especially in a monoculture, because there are no other plants that could compensate for the shortcomings.
Author Contributions
Conceptualization, L.G. and X.C.; methodology, L.G.; software, X.C.; validation, L.G.; formal analysis, Z.L.; investigation, M.W. and L.Z.; resources, X.C.; Y.C.; curation, L.G.; writing—original draft preparation, X.C.; writing—review and editing, L.G.; visualization, Z.J.; supervision, S.J.; project administration, L.G.; funding acquisition, L.G. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the China Agriculture Research System for Bast and Leaf Fiber Crops (CARS-16-S4).
Institutional Review Board Statement
No animal interventions were used in this research.
Data Availability Statement
All data are available on request.
Acknowledgments
The authors wish to thank the anonymous reviewers for their constructive comments on the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Rehman, M.; Fahad, S.; Du, G.; Cheng, X.; Yang, Y.; Tang, K.; Liu, L.; Liu, F.-H.; Deng, G. Evaluation of hemp (Cannabis sativa L.) as an industrial crop: A review. Environ. Sci. Pollut. Res. 2021, 28, 52832–52843. [Google Scholar] [CrossRef] [PubMed]
- Ceyhan, V.; Türkten, H.; Yıldırım, Ç.; Canan, S. Economic viability of industrial hemp production in Turkey. Ind. Crops Prod. 2022, 176, 114354. [Google Scholar] [CrossRef]
- Papstylianou, P.; Kakabouki, I.; Travlos, I. Effect of Nitrogen Fertilization on Growth and Yield of Industrial Hemp (Cannabis sativa L.). Not. Bot. Horti Agrobot. Cluj-Napoca 2018, 46, 197–201. [Google Scholar] [CrossRef] [Green Version]
- Duque Schumacher, A.G.; Pequito, S.; Pazour, J. Industrial hemp fiber: A sustainable and economical alternative to cotton. J. Clean. Prod. 2020, 268, 122180. [Google Scholar] [CrossRef]
- Parada-Rojas, C.H.; Pecota, K.; Almeyda, C.; Yencho, G.C.; Quesada-Ocampo, L.M. Sweetpotato Root Development Influences Susceptibility to Black Rot Caused by the Fungal Pathogen Ceratocystis fimbriata. Phytopathology 2021, 111, 1660–1669. [Google Scholar] [CrossRef]
- Han, G.; Lan, J.; Chen, Q.; Yu, C.; Bie, S. Response of soil microbial community to application of biochar in cotton soils with different continuous cropping years. Sci. Rep. 2017, 7, 10184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, Y.; Zhou, Y.; He, H. Effects of rehabilitation through afforestation on soil aggregate stability and aggregate-associated carbon after forest fires in subtropical China. Geoderma 2020, 376, 114548. [Google Scholar] [CrossRef]
- Li, Y.; Chi, J.; Ao, J.; Gao, X.; Liu, X.; Sun, Y.; Zhu, W. Effects of Different Continuous Cropping Years on Bacterial Community and Diversity of Cucumber Rhizosphere Soil in Solar-Greenhouse. Curr. Microbiol. 2021, 78, 2380–2390. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wu, C.; Cheng, Z.; Meng, H.; Zhang, M.; Zhang, H. Soil Chemical Property Changes in Eggplant/Garlic Relay Intercropping Systems under Continuous Cropping. PLoS ONE 2014, 9, e111040. [Google Scholar] [CrossRef] [Green Version]
- Ramesha, G.K.; Leno, N.; Radhika, N.S. Linking root phenomics, nutrient acquisition and utilisation in amaranthus with thermochemical organic fertilizer from biowaste. Rhizosphere 2021, 20, 100426. [Google Scholar] [CrossRef]
- Bukhat, S.; Imran, A.; Javaid, S.; Shahid, M.; Majeed, A.; Naqqash, T. Communication of plants with microbial world: Exploring the regulatory networks for PGPR mediated defense signaling. Microbiol. Res. 2020, 238, 126486. [Google Scholar] [CrossRef] [PubMed]
- Ortíz-Castro, R.; Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; López-Bucio, J. The role of microbial signals in plant growth and development. Plant Signal. Behav. 2009, 4, 701–712. [Google Scholar] [CrossRef] [Green Version]
- Gómez Expósito, R.; de Bruijn, I.; Postma, J.; Raaijmakers, J.M. Current Insights into the Role of Rhizosphere Bacteria in Disease Suppressive Soils. Front. Microbiol. 2017, 8, 2529. [Google Scholar] [CrossRef]
- Wu, J.; Jiao, Z.; Zhou, J.; Guo, F.; Ding, Z.; Qiu, Z. Analysis of bacterial communities in rhizosphere soil of continuously cropped healthy and diseased konjac. World J. Microbiol. Biotechnol. 2017, 33, 134. [Google Scholar] [CrossRef]
- Xiong, W.; Zhao, Q.; Zhao, J.; Xun, W.; Li, R.; Zhang, R.; Wu, H.; Shen, Q. Different Continuous Cropping Spans Significantly Affect Microbial Community Membership and Structure in a Vanilla-Grown Soil as Revealed by Deep Pyrosequencing. Microb. Ecol. 2015, 70, 209–218. [Google Scholar] [CrossRef]
- Tan, Y.; Cui, Y.; Li, H.; Kuang, A.; Li, X.; Wei, Y.; JI, X. Diversity and composition of rhizospheric soil and root endogenous bacteria in Panax notoginseng during continuous cropping practices. J. Basic Microbiol. 2017, 57, 337–344. [Google Scholar] [CrossRef]
- Gao, Z.; Hu, Y.; Han, M.; Xu, J.; Wang, X.; Liu, L.; Tang, Z.; Jiao, W.; Jin, R.; Liu, M.; et al. Effects of continuous cropping of sweet potatoes on the bacterial community structure in rhizospheric soil. BMC Microbiol. 2021, 21, 102. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Liu, T.; Zheng, C.; Kang, C.; Yang, Z.; Yao, X.; Song, F.; Zhang, R.; Wang, X.; Xu, N.; et al. Changes in soil bacterial community structure as a result of incorporation of Brassica plants compared with continuous planting eggplant and chemical disinfection in greenhouses. PLoS ONE 2017, 12, e0173923. [Google Scholar] [CrossRef]
- Ren, N.; Wang, Y.; Ye, Y.; Zhao, Y.; Huang, Y.; Fu, W.; Chu, X. Effects of Continuous Nitrogen Fertilizer Application on the Diversity and Composition of Rhizosphere Soil Bacteria. Front. Microbiol. 2020, 11, 1948. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.-Y.; Li, H.; Hao, M.-M.; Ren, Y.-N.; Zhang, M.-K.; Liu, R.-Y.; Zhang, Y.; Li, G.; Chen, J.-S.; Ning, T.-Y.; et al. Nitrogen fixation and crop productivity enhancements co-driven by intercrop root exudates and key rhizosphere bacteria. J. Appl. Ecol. 2021, 58, 2243–2255. [Google Scholar] [CrossRef]
- Jin, L.; Lyu, J.; Jin, N.; Xie, J.; Wu, Y.; Zhang, G.; Feng, Z.; Tang, Z.; Liu, Z.; Luo, S.; et al. Effects of different vegetable rotations on the rhizosphere bacterial community and tomato growth in a continuous tomato cropping substrate. PLoS ONE 2021, 16, e0257432. [Google Scholar] [CrossRef] [PubMed]
- Horne, M.; Mastrianni, K.R.; Amick, G.; Hardy, R.; Renneker, E.; Miller, K.W.P. Fast Discrimination of Marijuana using Automated High-throughput Cannabis Sample Preparation and Analysis by Gas Chromatography–Mass Spectrometry. J. Forensic Sci. 2020, 65, 1709–1715. [Google Scholar] [CrossRef]
- Small, E.; Brookes, B. Temperature and Moisture Content for Storage Maintenance of Germination Capacity of Seeds of Industrial Hemp, Marijuana, and Ditchweed Forms of Cannabis sativa. J. Nat. Fibers 2012, 9, 240–255. [Google Scholar] [CrossRef]
- Wu, Y.; Trejo, H.X.; Chen, G.; Li, S. Phytoremediation of contaminants of emerging concern from soil with industrial hemp (Cannabis sativa L.): A review. Environ. Dev. Sustain. 2021, 23, 14405–14435. [Google Scholar] [CrossRef]
- Tang, K.; Wang, J.; Yang, Y.; Deng, G.; Yu, J.; Hu, W.; Guo, L.; Du, G.; Liu, F. Fiber hemp (Cannabis sativa L.) yield and its response to fertilization and planting density in China. Ind. Crops Prod. 2022, 177, 114542. [Google Scholar] [CrossRef]
- DeAngelis, K.M.; Brodie, E.L.; DeSantis, T.Z.; Andersen, G.L.; Lindow, S.E.; Firestone, M.K. Selective progressive response of soil microbial community to wild oat roots. ISME J. 2009, 3, 168–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, N.; Tan, G.; Wang, H.; Gai, X. Effect of biochar additions to soil on nitrogen leaching, microbial biomass and bacterial community structure. Eur. J. Soil Biol. 2016, 74, 1–8. [Google Scholar] [CrossRef]
- Ashworth, A.J.; Owens, P.R.; Allen, F.L. Long-term cropping systems management influences soil strength and nutrient cycling. Geoderma 2020, 361, 114062. [Google Scholar] [CrossRef]
- Crookston, R.; Kurle, J.; Copeland, P.; Ford, J.; Lueschen, W. Rotational Cropping Sequence Affects Yield of Corn and Soybean. Agron. J. 1991, 83, 108–113. [Google Scholar] [CrossRef]
- Gentry, L.F.; Ruffo, M.L.; Below, F.E. Identifying Factors Controlling the Continuous Corn Yield Penalty. Agron. J. 2013, 105, 295–303. [Google Scholar] [CrossRef] [Green Version]
- Sun, K.; Fu, L.; Song, Y.; Yuan, L.; Zhang, H.; Wen, D.; Yang, N.; Wang, X.; Yue, Y.; Li, X.; et al. Effects of continuous cucumber cropping on crop quality and soil fungal community. Environ. Monit. Assess. 2021, 193, 436. [Google Scholar] [CrossRef]
- Alami, M.M.; Xue, J.; Ma, Y.; Zhu, D.; Abbas, A.; Gong, Z.; Wang, X. Structure, Function, Diversity, and Composition of Fungal Communities in Rhizospheric Soil of Coptis chinensis Franch under a Successive Cropping System. Plants 2020, 9, 244. [Google Scholar] [CrossRef] [Green Version]
- Pang, Z.; Dong, F.; Liu, Q.; Lin, W.; Hu, C.; Yuan, Z. Soil Metagenomics Reveals Effects of Continuous Sugarcane Cropping on the Structure and Functional Pathway of Rhizospheric Microbial Community. Front. Microbiol. 2021, 12, 369. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Gao, Q.; Xu, S.; Ma, L.; Tian, C. Long-term effect of residue return and fertilization on microbial biomass and community composition of a clay loam soil. J. Agric. Sci. 2016, 154, 1051–1061. [Google Scholar] [CrossRef]
- Zu, C.; Li, Z.; Yang, J.; Yu, H.; Sun, Y.; Tang, H.; Yost, R.; Wu, H. Acid Soil Is Associated with Reduced Yield, Root Growth and Nutrient Uptake in Black Pepper ( Piper nigrum L.). J. Agric. Sci. 2014, 5, 466–473. [Google Scholar] [CrossRef] [Green Version]
- Mu, Y.; Tang, D.; Mao, L.; Zhang, D.; Zhou, P.; Zhi, Y.; Zhang, J. Phytoremediation of secondary saline soil by halophytes with the enhancement of γ-polyglutamic acid. Chemosphere 2021, 285, 131450. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Zhou, Y.; Gong, J. Physiological mechanisms of the tolerance response to manganese stress exhibited by Pinus massoniana, a candidate plant for the phytoremediation of Mn-contaminated soil. Environ. Sci. Pollut. Res. 2021, 28, 45422–45433. [Google Scholar] [CrossRef]
- Wallenstein, M.D. Managing and manipulating the rhizosphere microbiome for plant health: A systems approach. Rhizosphere 2017, 3, 230–232. [Google Scholar] [CrossRef]
- Hu, J.; Masson, R. First Report of Crown and Root Rot Caused by Pythium aphanidermatum on Industrial Hemp (Cannabis sativa) in Arizona. Plant Dis. 2021, 105, 2257. [Google Scholar] [CrossRef]
- Sikorski, J.; Baumgartner, V.; Birkhofer, K.; Boeddinghaus, R.S.; Bunk, B.; Fischer, M.; Fösel, B.U.; Friedrich, M.W.; Göker, M.; Hölzel, N.; et al. The Evolution of Ecological Diversity in Acidobacteria. Front. Microbiol. 2022, 13, 715637. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, R.; Chen, S.; Qi, G.; He, Z.; Zhao, X. Microbial taxa and functional genes shift in degraded soil with bacterial wilt. Sci. Rep. 2017, 7, 39911. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Gao, S.; Jiang, Y.; Lin, Z.; Luo, J.; Li, M.; Guo, J.; Su, Y.; Xu, L.; Que, Y. The Physiological and Agronomic Responses to Nitrogen Dosage in Different Sugarcane Varieties. Front. Plant Sci. 2019, 10, 406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Sui, Y.; Yu, Z.; Shi, Y.; Chu, H.; Jin, J.; Liu, X.; Wang, G. High throughput sequencing analysis of biogeographical distribution of bacterial communities in the black soils of northeast China. Soil Biol. Biochem. 2014, 70, 113–122. [Google Scholar] [CrossRef]
- van der Bom, F.; Nunes, I.; Raymond, N.S.; Hansen, V.; Bonnichsen, L.; Magid, J.; Nybroe, O.; Jensen, L.S. Long-term fertilisation form, level and duration affect the diversity, structure and functioning of soil microbial communities in the field. Soil Biol. Biochem. 2018, 122, 91–103. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).