Fungal Bioluminescence: Past, Present, and Future
Abstract
1. Introduction
1.1. Bioluminescence as a Common Term
1.2. Brief History of Studies on Bioluminescent Organisms
1.3. Aspects of Bioluminescent Fungi
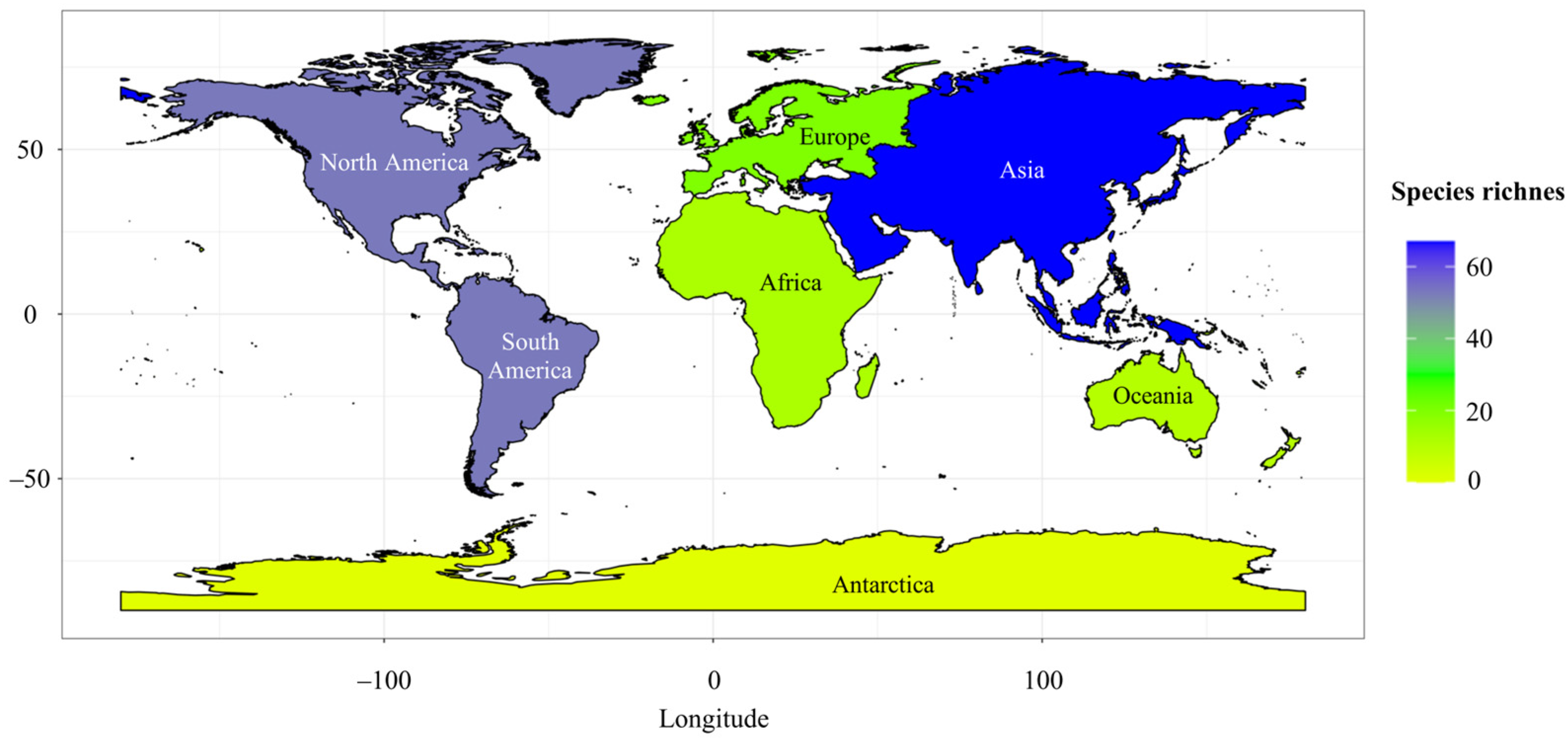
2. Diversity and Distribution of Bioluminescent Fungi
3. Evolution and Mechanisms of Bioluminescent Fungi
3.1. Evolution
3.2. Mechanisms
4. Importance of Bioluminescent Fungi in Ecology
5. Application of Fungal Bioluminescence
6. Conclusions and Future Directions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pandey, A.; Sharon, M. Bioluminescent organisms. BAOJ Chem. 2017, 3, e029. [Google Scholar]
- Ke, H.M.; Tsai, I.J. Understanding and using fungal bioluminescence-Recent progress and future perspectives. Curr. Opin. Green Sustain. Chem. 2022, 33, 100570. [Google Scholar] [CrossRef]
- Waldenmaier, H.E.; Oliveira, A.G.; Stevani, C.V. Thoughts on the diversity of convergent evolution of bioluminescence on earth. Int. J. Astrobiol. 2012, 11, 335–343. [Google Scholar] [CrossRef]
- Vieira, M.B.B.; Oliveira, I.C.; de Oliveira, M.D.D.A.; da Costa Júnior, J.S.; dos Santos, T.D.J.A.; Feitosa, C.M.; Rai, M.; Lima, N.M.; da Costa Silva, D. A review on bioluminescent fungus Neonothopanus gardneri. Res. Soc. Dev. 2022, 11, e16811528009. [Google Scholar] [CrossRef]
- Lau, E.S.; Oakley, T.H. Multi-level convergence of complex traits and the evolution of bioluminescence. Biol. Rev. Camb. Philos. Soc. 2020, 96, 673–691. [Google Scholar] [CrossRef] [PubMed]
- Syed, A.J.; Anderson, J.C. Applications of bioluminescence in biotechnology and beyond. Chem. Soc. Rev. 2021, 50, 5668–5705. [Google Scholar] [CrossRef] [PubMed]
- Delroisse, J.; Duchatelet, L.; Flammang, P.; Mallefet, J. Leaving the dark side? Insights into the evolution of luciferases. Front. Mar. Sci. 2021, 8, 673620. [Google Scholar] [CrossRef]
- Harvey, E.N. Bioluminescence; Academic Press: New York, NY, USA, 1952; pp. 1–649. [Google Scholar]
- Harvey, E.N. A History of Luminescence: From the Earliest Times until 1900; The American Philosophical Society: Philadelphia, PA, USA, 1957; pp. 1–692. [Google Scholar] [CrossRef]
- Lee, S.M.L. The status of bioluminescent fungal species in Singapore. Nat. Singap. 2022, 1, e2022124. [Google Scholar] [CrossRef]
- Mahish, P.K.; Chandrawanshi, N.K.; Kunjam, S.; Jadhav, S.K. Opportunities in the living lights: Special reference to bioluminescent fungi. Energy Cris. Chall. Solut. 2021, 10, 191–207. [Google Scholar]
- Lu, D. 71 species of macrofungi that bioluminescence. Edible Med. Mushrooms 2011, 19, 55–57. (In Chinese) [Google Scholar]
- Boyle, R. Observations and tryals about the resemblances and differences between a burning coal and shining wood. Phil. Trans. 1667, 2, 605–612. [Google Scholar] [CrossRef]
- Oba, Y.; Hosaka, K. The luminous fungi of Japan. J. Fungi 2023, 9, 615. [Google Scholar] [CrossRef] [PubMed]
- Kirk, P.M. World catalogue of 340K fungal names on-line. Mycol. Res. 2000, 104, 516–517. [Google Scholar]
- Crous, P.W.; Gams, W.; Stalpers, J.A.; Robert, V.; Stegehuis, G. MycoBank: An online initiative to launch mycology into the 21st century. Stud. Mycol. 2004, 50, 19–22. [Google Scholar]
- Taylor, J.W. One Fungus = One Name: DNA and fungal nomenclature twenty years after PCR. IMA Fungus 2011, 2, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Spouge, J.L.; Levesque, C.A.; Chen, W.; Fungal Barcoding Consortium; Fungal Barcoding Consortium Author List; Bolchacova, E.; et al. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA 2012, 109, 6241–6246. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Robbertse, B.; Robert, V.; Vu, D.; Cardinali, G.; Irinyi, L.; Meyer, W.; Nilsson, R.H.; Hughes, K.; Miller, A.N.; et al. Finding needles in haystacks: Linking scientific names, reference specimens and molecular data for Fungi. Database 2014, 2014, bau061. [Google Scholar] [CrossRef] [PubMed]
- McNeill, J.; Barrie, F.R.; Buck, W.R.; Demoulin, V.; Greuter, W.; Hawksworth, D.L.; Herendeen, P.S.; Knapp, S.; Marhold, K.; Prado, J.; et al. International Code of Nomenclature for algae, fungi, and plants. Regnum Veg. 2012, 154. Available online: http://www.iapt-taxon.org/nomen/main.php (accessed on 25 July 2024).
- Oliveira, A.G.; Desjardin, D.E.; Perry, B.A.; Stevani, C.V. Evidence that a single bioluminescent system is shared by all known bioluminescent fungal lineages. Photochem. Photobiol. Sci. 2012, 11, 848–852. [Google Scholar] [CrossRef] [PubMed]
- Karunarathna, S.C.; Mortimer, P.E.; Tibpromma, S.; Dutta, A.K.; Paloi, S.; Hu, Y.; Baurah, G.; Axford, S.; Marciniak, C.; Luangharn, T.; et al. Roridomyces phyllostachydis (Agaricales, Mycenaceae), a new bioluminescent fungus from Northeast India. Phytotaxa 2020, 459, 155–167. [Google Scholar] [CrossRef]
- Cortés-Pérez, A.; Guzmán-Dávalos, L.; Ramírez-Cruz, V.; Villalobos-Arámbula, A.R.; Ruiz-Sanchez, E.; Ramírez-Guillén, F. New Species of Bioluminescent Mycena Sect. Calodontes (Agaricales, Mycenaceae) from Mexico. J. Fungi 2023, 9, 902. [Google Scholar] [CrossRef]
- Labella, A.M.; Arahal, D.R.; Castro, D.; Lemos, M.L.; Borrego, J.J. Revisiting the genus Photobacterium: Taxonomy, ecology and pathogenesis. Int. Microbiol. 2017, 20, 1–10. [Google Scholar]
- Burtseva, O.; Kublanovskaya, A.; Baulina, O.; Fedorenko, T.; Lobakova, E.; Chekanov, K. The strains of bioluminescent bacteria isolated from the White Sea finfishes: Genera Photobacterium, Aliivibrio, Vibrio, Shewanella, and first luminous Kosakonia. J. Photoch. Photobio. B 2020, 208, 111895. [Google Scholar] [CrossRef]
- de Busserolles, F.; Marshall, N.J. Seeing in the deep-sea: Visual adaptations in lanternfishes. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2017, 372, 20160070. [Google Scholar] [CrossRef] [PubMed]
- Al-Handawi, M.B.; Polavaram, S.; Kurlevskaya, A.; Commins, P.; Schramm, S.; Carrasco-López, C.; Lui, N.M.; Solntsev, K.M.; Laptenok, S.P.; Navizet, I.; et al. Spectrochemistry of firefly bioluminescence. Chem. Rev. 2022, 122, 13207–13234. [Google Scholar] [CrossRef]
- Owens, A.; Van den Broeck, M.; De Cock, R.; Lewis, S.M. Behavioral responses of bioluminescent fireflies to artificial light at night. Front. Ecol. Evol. 2022, 10, 946640. [Google Scholar] [CrossRef]
- Elkhateeb, W.A.; Daba, G.M. Bioluminescent Mushrooms: Boon for environmental health. Environ. Sci. Arch. 2022, 1, 88–97. Available online: https://www.envsciarch.com/_files/ugd/4b6a78_9155754cbd4e4412b6bab4385f8ef2e1.pdf (accessed on 14 October 2022).
- Adams, C.A.; Donald, M.L.; Swearingen, C.; Escudero, E.; Sourell, S.; Landrein, S.; Seas, C.; Mueller, G.; Chaverri, P. Let there be nightlights: The ecological role of bioluminescence in a Costa Rican mushroom. bioRxiv 2023. [Google Scholar] [CrossRef]
- Oliveira, A.G.; Stevani, C.V. The enzymatic nature of fungal bioluminescence. Photochem. Photobiol. Sci. 2009, 8, 1416. [Google Scholar] [CrossRef] [PubMed]
- Kaskova, Z.M.; Dörr, F.A.; Petushkov, V.N.; Purtov, K.V.; Tsarkova, A.S.; Rodionova, N.S.; Mineev, K.S.; Guglya, E.B.; Kotlobay, A.; Baleeva, N.S.; et al. Mechanism and color modulation of fungal bioluminescence. Sci. Adv. 2017, 3, e1602847. [Google Scholar] [CrossRef]
- Kotlobay, A.A.; Sarkisyan, K.S.; Mokrushina, Y.A.; Marcet-Houben, M.; Serebrovskaya, E.O.; Markina, N.M.; Gonzalez, S.L.; Gorokhovatsky, A.Y.; Vvedensky, A.; Purtov, K.V.; et al. Genetically encodable bioluminescent system from fungi. Proc. Natl. Acad. Sci. USA 2018, 115, 12728–12732. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Chen, C.Y.; Li, W.W.; Ka, W. Mycena jingyinga, Mycena luguensis, and Mycena venus: Three new species of bioluminescent fungi from Taiwan. Taiwania 2020, 65, 396–406. Available online: https://taiwania.ntu.edu.tw/pdf/tai.2020.65.396.pdf (accessed on 27 July 2020).
- Mitiouchkina, T.; Mishin, A.S.; Somermeyer, L.G.; Markina, N.M.; Chepurnyh, T.V.; Guglya, E.B.; Karataeva, T.A.; Palkina, K.A.; Shakhova, E.S.; Fakhranurova, L.I.; et al. Plants with genetically encoded autoluminescence. Nat. Biotechnol. 2020, 38, 944–946. [Google Scholar] [CrossRef] [PubMed]
- Niskanen, T.; Lücking, R.; Dahlberg, A.; Gaya, E.; Suz, L.M.; Mikryukov, V.; Liimatainen, K.; Druzhinina, I.; Westrip, J.R.; Mueller, G.M.; et al. Pushing the frontiers of biodiversity research: Unveiling the global diversity, distribution, and conservation of fungi. Annu. Rev. Environ. Resour. 2023, 48, 149–176. [Google Scholar] [CrossRef]
- Rashhmi, M.; Kushveer, J.S.; Sarma, V.V. A worldwide list of endophytic fungi with notes on ecology and diversity. Mycosphere 2019, 10, 798–1079. [Google Scholar] [CrossRef]
- Van Der Heijden, M.G.; Martin, F.M.; Selosse, M.A.; Sanders, I.R. Mycorrhizal ecology, and evolution: The past, the present, and the future. New Phytol. 2015, 205, 1406–1423. [Google Scholar] [CrossRef] [PubMed]
- Stevani, C.V.; Zamuner, C.K.; Bastos, E.L.; de Nóbrega, B.B.; Soares, D.M.; Oliveira, A.G.; Bechara, E.J.; Shakhova, E.S.; Sarkisyan, K.S.; Yampolsky, I.V.; et al. The living light from fungi. J. Photochem. Photobiol. C Photochem. Rev. 2024, 58, 100654. [Google Scholar] [CrossRef]
- Vydryakova, G.A.; Gusev, A.A.; Medvedeva, S.E. Effect of organic and inorganic toxic compounds on luminescence of luminous fungi. Appl. Biochem. Microbiol. 2011, 47, 293–297. [Google Scholar] [CrossRef]
- Mihail, J.D. Bioluminescence patterns among North American Armillaria species. Fungal Biol. 2015, 119, 528–537. [Google Scholar] [CrossRef]
- Desjardin, D.E.; Oliveira, A.G.; Stevani, C.V. Fungi bioluminescence revisited. Photochem. Photobiol. Sci. 2008, 7, 170–182. [Google Scholar] [CrossRef]
- Tan, Z.J.; Xie, D.P.; Wang, Z.; Li, W.G.; Liu, S. The study on bioluminescence condition of Amillaria mellea. Acta Laser Biol. Sinica 2001, 10, 1007–7146. (In Chinese) [Google Scholar]
- Patil, S.R.; Yadav, S.V. Photographic record of Armillaria mellea a bioluminescent fungus from Lonavala in western Ghats, India. J. Threat. Taxa 2022, 14, 20692–20694. [Google Scholar] [CrossRef]
- BITB. Bioluminescence in the Bush: Glow in the Dark Mushrooms in Stewart Island. Available online: https://www.myconeer.com/p/bioluminescence-in-the-bush-glow (accessed on 24 April 2024).
- Ainsworth, M. Searching for luminous mushrooms of the marsh fungus Armillaria ectypa. Field Mycol. 2004, 5, 142–144. [Google Scholar] [CrossRef]
- Silva-Filho, A.G.; Mombert, A.; Nascimento, C.C.; Nóbrega, B.B.; Soares, D.M.; Martins, A.G.; Domingos, A.H.; Santos, I.; Della-Torre, O.H.; Perry, B.A.; et al. Eoscyphella luciurceolata gen. and sp. nov. (Agaricomycetes) shed light on Cyphellopsidaceae with a new lineage of bioluminescent fungi. J. Fungi 2023, 9, 1004. [Google Scholar] [CrossRef] [PubMed]
- Nimalrathna, T.; Tibpromma, S.; Nakamura, A.; Galappaththi, M.C.A.; Xu, J.; Mortimer, P.E.; Karunarathna, S.C. The case of the missing mushroom: A novel bioluminescent species discovered within Favolaschia in Southwestern China. Phytotaxa 2022, 539, 244–256. [Google Scholar] [CrossRef]
- Liu, P.G. Luminescent fungi. Chin. Biodivers. 1995, 3, 109–112. (In Chinese) [Google Scholar]
- Terashima, Y.; Neda, H.; Hiroi, M. Luminescent intensity of cultured mycelia of eight basidiomycetous fungi from Japan. Mushroom Sci. Biotechnol. 2016, 24, 176–181. [Google Scholar]
- Terashima, Y.; Takahashi, H.; Taneyama, Y. The Fungal Flora in Southwestern Japan: Agarics and Boletes; Tokai Daigaku: Kanagawa, Japan, 2016; p. 349. [Google Scholar]
- Liu, P.G.; Yang, Z.L. Studies of classification and geographic distribution of Laschia-complex from the Southern and Southeastern Yunnan, China. Acta Bot. Yunnanica 1994, 16, 47–52. (In Chinese) [Google Scholar]
- Chew, A.L.; Desjardin, D.E.; Tan, Y.S.; Musa, M.Y.; Sabaratnam, V. Bioluminescent fungi from peninsular Malaysia- A taxonomic and phylogenetic overview. Fungal Divers. 2014, 70, 149–187. [Google Scholar] [CrossRef]
- Yan, J.J.; Liu, X.R.; Xie, B.G.; Deng, Y.J. Isolation, identification, and characterization of Neonothopanus nambi (Basidiomycota, Fungi), a new record from China. Bull. Microbiol. 2015, 42, 1703–1709. [Google Scholar]
- Desjardin, D.E.; Perry, B.A.; Lodge, D.J.; Stevani, C.V.; Nagasawa, E. Luminescent Mycena: New and noteworthy species. Mycologia 2010, 102, 459–477. [Google Scholar] [CrossRef] [PubMed]
- Chew, A.L.; Tan, Y.S.; Desjardin, D.E.; Musa, M.Y.; Sabaratnam, V. Four new bioluminescent taxa of Mycena sect. Calodontes from Peninsular Malaysia. Mycologia 2014, 106, 976–988. [Google Scholar] [CrossRef]
- Oliveira, J.J.; Vargas-Isla, R.; Cabral, T.S.; Cardoso, J.S.; Andriolli, F.S.; Rodrigues, D.P.; Ikeda, T.; Clement, C.R.; Ishikawa, N.K. The amazonian luminescent Mycena cristinae sp. nov. from Brazil. Mycoscience 2021, 62, 395–405. [Google Scholar] [CrossRef]
- Heinzelmann, R.; Baggenstos, H.; Rudolf, A. Is the bioluminescence in many Mycena species overlooked?—A case study from M.crocata in Switzerland. Mycoscience 2024, 65, MYC633. [Google Scholar] [CrossRef]
- Aravindakshan, D.M.; Kumar, T.K.A.; Manimohan, P. A new bioluminescent species of Mycena sect. Exornatae from Kerala State, India. Mycosphere 2012, 3, 556–561. [Google Scholar] [CrossRef]
- Desjardin, D.E.; Perry, B.A.; Stevani, C.V. New luminescent Mycenoid fungi (Basidiomycota, Agaricales) from Sao Paulo state, Brazil. Mycologia 2016, 108, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Cortés-Pérez, A.; Desjardin, D.E.; Perry, B.A.; Ramírez-Cruz, V.; Ramírez-Guillén, F.; Villalobos-Arámbula, A.R.; Rockefeller, A. New species, and records of bioluminescent Mycena from Mexico. Mycologia 2019, 111, 319–573. [Google Scholar] [CrossRef]
- Soares, C.C.; Cabral, T.S.; Vargas-isla, R.U.B.Y.; Cardoso, J.S.; Rodrigues, D.P.; Ishikawa, N.K.; Oliveira, J.J. Mycena lamprocephala, a new luminescent species from the Brazilian Amazon. Phytotaxa 2024, 634, 187–203. [Google Scholar] [CrossRef]
- Alves, M.H.; do Nascimento, C.C. Mycena margarita (Murrill) Murrill, 1916 (Basidiomycota: Agaricales: Mycenaceae): A bioluminescent agaric first recorded in Brazil. Check List 2014, 10, 239–243. [Google Scholar] [CrossRef]
- Desjardin, D.E.; Capelari, M.; Stevani, C. Bioluminescent Mycena species from São Paulo, Brazil. Mycologia 2007, 99, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Corner, E.J.H. Descriptions of two luminous tropical agarics (Dictyopanus and Mycena). Mycologia 1950, 42, 423–431. [Google Scholar] [CrossRef]
- Corner, E.J.H. The agaric genus Panellus Karst. (including Dictyopanus Pat.) in Malaysia. Gard. Bull. 1986, 39, 103–147. Available online: https://biostor.org/reference/177690 (accessed on 25 July 2024).
- Horak, E. Mycena rorida (Fr.) Quél. and related species from the Southern Hemisphere. Berichte 1978, 88, 20–29. [Google Scholar] [CrossRef]
- Tolgor, B. Notes on Basidiomycetes of Jilin province (VIII). J. Fungal Res. 2007, 5, 72–74. [Google Scholar]
- Dauner, L.A.P.; Karunarathna, S.C.; Tibpromma, S.; Xu, J.; Mortimer, P.E. Bioluminescent fungus Roridomyces viridiluminus sp. nov. and the first Chinese record of the genus Roridomyces from Southwestern China. Phytotaxa 2021, 487, 233–250. [Google Scholar] [CrossRef]
- Yang, Z.L.; Feng, B. The genus Omphalotus (Omphalotaceae) in China. Mycosystema 2013, 32, 545–556. [Google Scholar]
- Li, J.Z.; Hu, X.W. A new species of Lampteromyces from Hunan. Acta Sci. Nat. Univ. Norm. Hunan 1993, 16, 2. [Google Scholar]
- OSSJ. Omphalotus Subilludens—Southern Jack O’ Lantern. Available online: https://www.texasmushrooms.org/en/omphalotus_subilludens.htm (accessed on 1 June 2024).
- Buller, A.H.R. Book: Researches on Fungi; Longmans: London, UK, 1958; Volume III, pp. 416–419. Available online: https://archive.org/details/researchesonfung03bull/page/418/mode/2up (accessed on 30 April 2008).
- Foerster, G.E.; Behrens, P.Q.; Airth, R.L. Bioluminescence and other characteristics of Collybia velutipes. Am. J. Bot. 1965, 52, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, V.; Hajirnis, S. A review on bioluminescent fungi: A torch of curiosity. Int. J. Life Sci. 2016, A7, 107–110. Available online: https://oaji.net/articles/2017/736-1518519635.pdf (accessed on 1 December 2016).
- Wassink, E.C. Observations on the luminescence in fungi I, including a critical review of the species mentioned as luminescent in literature. Recl. Des Trav. Bot. Néerlandais 1948, 4, 150–212. [Google Scholar]
- Desjardin, D.E.; Capelari, M.; Stevani, C.V. A new bioluminescent Agaric from São Paulo, Brazil. Fungal Divers. 2005, 18, 9–14. [Google Scholar]
- Stevani, C.V.; Oliveira, A.G.; Mendes, L.F.; Ventura, F.F.; Waldenmaier, H.E.; Carvalho, R.P.; Pereira, T.A. Current status of research on fungal bioluminescence: Biochemistry and prospects for ecotoxicological application. Photochem. Photobiol. 2013, 89, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Peterson, R.H.; Bermudes, D. Intercontinental compatibility in Panellus stypticus with a note on bioluminescence. Persoonia 1992, 14, 457–463. [Google Scholar]
- Arya, C.P.; Ratheesh, S.; Pradeep, C.K. New record of luminescent Mycena chlorophos (Mycenaceae) from Western Ghats of India. Stud. Fungi 2021, 6, 507–513. [Google Scholar] [CrossRef]
- Dutta, A.; Gupta, S.; Roy, J.K.; Ahmed, M.F. New distribution record of Roridomyces cf. phyllostachydis (Agaricales: Mycenaceae), a bioluminescent fungus from Namdapha National Park, Arunachal Pradesh, India. J. Threat. Taxa 2023, 15, 22920–22923. [Google Scholar] [CrossRef]
- Shih, Y.S.; Chen, C.Y.; Lin, W.W.; Kao, H.W. Mycena kentingensis, a new species of luminous mushroom in Taiwan, with reference to its culture method. Mycol. Prog. 2014, 13, 429–435. [Google Scholar] [CrossRef]
- Ke, H.M.; Lee, H.H.; Lin, C.I.; Liu, Y.C.; Lu, M.R.; Hsieh, J.A.; Chang, C.C.; Wu, P.H.; Lu, M.J.; Li, J.Y.; et al. Mycena genomes resolve the evolution of fungal bioluminescence. Proc. Natl. Acad. Sci. USA 2020, 117, 31267–31277. [Google Scholar] [CrossRef]
- Ke, H.M.; Lu, M.R.; Chang, C.C.; Hsiao, C.; Ou, J.H.; Taneyama, Y.; Tsai, I.J. Evolution and Diversity of Bioluminescent Fungi. In Evolution of Fungi and Fungal-Like Organisms; Springer International Publishing: Cham, Switzerland, 2023; pp. 275–294. [Google Scholar]
- Palkina, K.A.; Balakireva, A.V.; Belozerova, O.A.; Chepurnykh, T.V.; Markina, N.M.; Kovalchuk, S.I.; Tsarkova, A.S.; Mishin, A.S.; Yampolsky, I.V.; Sarkisyan, K.S. Domain truncation in hispidin synthase orthologs from non-bioluminescent fungi does not lead to hispidin biosynthesis. Int. J. Mol. Sci. 2023, 24, 1317. [Google Scholar] [CrossRef]
- Kahlke, T.; Umbers, K.D.L. Bioluminescence. Curr. Biol. 2016, 26, R313–R314. [Google Scholar] [CrossRef]
- Wilson, T.; Hastings, J.W. Bioluminescence: Living Lights, Lights for Living; Harvard University Press: Cambridge, MA, USA, 2013; p. 176. [Google Scholar] [CrossRef]
- Liu, X.; Wang, M.; Liu, Y. Chemistry in fungal bioluminescence: Theoretical studies on biosynthesis of luciferin from caffeic acid and regeneration of caffeic acid from oxidized luciferin. J. Fungi 2023, 9, 369. [Google Scholar] [CrossRef] [PubMed]
- Khakhar, A.; Starker, C.G.; Chamness, J.C.; Lee, N.; Stokke, S.; Wang, C.; Swanson, R.; Rizvi, F.; Imaizumi, T.; Voytas, D.F. Building customizable auto-luminescent luciferase-based reporters in plants. eLife 2020, 9, e52786. [Google Scholar] [CrossRef]
- Dubois, R. Note sur la fonction photogénique chez la Pholas dactylus. C. R. Seances Soc. Biol. 1887, 39, e564. [Google Scholar]
- Airth, R.L.; McElroy, W.D. Light emission from extract of luminous fungi. J. Bacteriol. 1959, 77, 249–250. Available online: https://journals.asm.org/doi/pdf/10.1128/jb.77.2.249-250.1959 (accessed on 25 July 2024). [CrossRef]
- Airth, R.L.; Foerster, G.E. The isolation of catalytic components required for cell-free fungal bioluminescence. Arch. Biochem. Biophys. 1962, 97, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Shimomura, O. Bioluminescence: Chemical Principles and Methods; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2006; p. 500. [Google Scholar] [CrossRef]
- Purtov, K.V.; Petushkov, V.N.; Baranov, M.S.; Mineev, K.S.; Rodionova, N.S.; Kaskova, Z.M.; Tsarkova, A.S.; Petunin, A.I.; Bondar, V.S.; Rodicheva, E.K.; et al. The chemical basis of fungal bioluminescence. Angew. Chem. Int. Ed. 2015, 54, 8124–8128. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.Y.; Liu, Y.J. Chemistry in fungal bioluminescence: A theoretical study from Luciferin to light emission. J. Org. Chem. 2021, 86, 1874–1881. [Google Scholar] [CrossRef] [PubMed]
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal secondary metabolism-from biochemistry to genomics. Nat. Rev. Microbiol. 2005, 3, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Rokas, A.; Wisecaver, J.H.; Lind, A.L. The birth, evolution and death of metabolic gene clusters in fungi. Nat. Rev. Microbiol. 2018, 16, 731–744. [Google Scholar] [CrossRef]
- Kim, J.; Park, M.J.; Shim, D.; Ryoo, R. De novo genome assembly of the bioluminescent mushroom Omphalotus guepiniiformis reveals an Omphalotus-specific lineage of the luciferase gene block. Genomics 2022, 114, e110514. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.G.; Stevani, C.V.; Waldenmaier, H.E.; Viviani, V.; Emerson, J.M.; Loros, J.J.; Dunlap, J.C. Circadian control sheds light on fungal bioluminescence. Curr. Biol. 2015, 25, 964–968. [Google Scholar] [CrossRef] [PubMed]
- Mishara, M.; Srivastava, D. Bioluminescent fungi: Reviewing nature’s riddle. J. Mycopathol. Res. 2021, 59, 199–206. [Google Scholar]
- Sivinski, J.M. Arthropods attracted to luminous fungi. Psyche A J. Entomol. 1981, 88, 383–390. [Google Scholar] [CrossRef]
- Bechara, E.J.H. Bioluminescence: A fungal nightlight with an internal timer. Curr. Biol. 2015, 25, R283–R285. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, P.; Delean, S.; Wood, T.; Austin, A.D. Bioluminescence in the ghost fungus Omphalotus nidiformis does not attract potential spore dispersing insects. IMA Fungus 2016, 7, 229–234. [Google Scholar] [CrossRef]
- Lingle, W.L. Bioluminescence and ligninolysis during secondary metabolism in the fungus Panellus stypticus. J. Biolum. Chemilum. 1993, 8, e100. [Google Scholar]
- Jabr, F. The Secret History of Bioluminescence. Hakai Magazine. 2016. Available online: https://hakaimagazine.com/features/secret-history-bioluminescence/ (accessed on 14 July 2023).
- Jain, N. In Meghalaya, Scientists Discover Glowing Mushrooms, Used by Locals as Natural Torches. 2020. Available online: https://scroll.in/article/978927/in-meghalaya-scientists-discover-glowing-mushrooms-used-by-locals-as-natural-torches (accessed on 14 July 2023).
- Lepp, H. Aboriginal Use of Fungi. 2013. Available online: https://www.anbg.gov.au/fungi/aboriginal.html (accessed on 14 July 2023).
- Araújo-Gomes, N.; Zambito, G.; Johnbosco, C.; Calejo, I.; Leijten, J.; Löwik, C.; Karperien, M.; Mezzanotte, L.; Teixeira, L.M. Bioluminescence imaging on-chip platforms for non-invasive high-content bioimaging. Biosens. Bioelectron. 2023, 237, e115510. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.A.; Welch, S.R.; Jain, S.; Sorvillo, T.E.; Coleman-McCray, J.D.; Montgomery, J.M.; Spiropoulou, C.F.; Albariño, C.; Spengler, J.R. Fluorescent and bioluminescent reporter mouse-adapted Ebola viruses maintain pathogenicity and can be visualized in vivo. J. Infect. Dis. 2023, 228, S536–S547. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Chen, R.; Zhu, C.; Kong, F. Glowing plants can light up the night sky? A review. Biotechnol. Bioeng. 2021, 118, 3706–3715. [Google Scholar] [CrossRef]
- Zheng, P.; Ge, J.; Ji, J.; Zhong, J.; Chen, H.; Luo, D.; Li, W.; Bi, B.; Ma, Y.; Tong, W.; et al. Metabolic engineering and mechanical investigation of enhanced plant autoluminescence. Plant Biotechnol. J. 2023, 21, 1671–1681. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, S.M.; Poziomek, E.J.; Engelmann, W.H.; Rogers, K.R. A review of environmental applications of bioluminescence measurements. Chemosphere 1995, 30, 2155–2197. [Google Scholar] [CrossRef]
- Gianfreda, L.; Rao, M.A. Interactions between xenobiotics and microbial and enzymatic soil activity. Crit. Rev. Environ. Sci. Technol. 2008, 3, 269–310. [Google Scholar] [CrossRef]
- Ventura, F.F.; Mendes, L.F.; Oliveira, A.G.; Bazito, R.C.; Bechara, E.J.; Freire, R.S.; Stevani, C.V. Evaluation of phenolic compound toxicity using a bioluminescent assay with the fungus Gerronema viridilucens. Environ. Toxicol. Chem. 2020, 39, 1558–1565. [Google Scholar] [CrossRef] [PubMed]
- Ventura, F.F.; Soares, D.M.; Bayle, K.; Oliveira, A.G.; Bechara, E.J.; Freire, R.S.; Stevani, C.V. Toxicity of metal cations and phenolic compounds to the bioluminescent fungus Neonothopanus gardneri. Environ. Adv. 2021, 4, 100044. [Google Scholar] [CrossRef]
- Endo, M.; Ozawa, T. Advanced bioluminescence system for in vivo imaging with brighter and red-shifted light emission. Int. J. Mol. Sci. 2020, 21, 6538. [Google Scholar] [CrossRef]
- Chen, M.; Zhou, K.; Dai, S.Y.; Tadepalli, S.; Balakrishnan, P.B.; Xie, J.; Rami, F.E.; Dai, T.; Cui, L.; Idoyaga, J.; et al. In vivo bioluminescence imaging of granzyme B activity in tumor response to cancer immunotherapy. Cell Chem. Biol. 2022, 29, 1556–1567. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Song, B.; Xu, Y.; Yang, Y.; Ji, J.; Cao, W.; Lu, J.; Ding, J.; Cao, H.; Chu, B.; et al. In vivo bioluminescence imaging of natural bacteria within deep tissues via ATP-binding cassette sugar transporter. Nat. Commun. 2023, 14, 2331. [Google Scholar] [CrossRef] [PubMed]
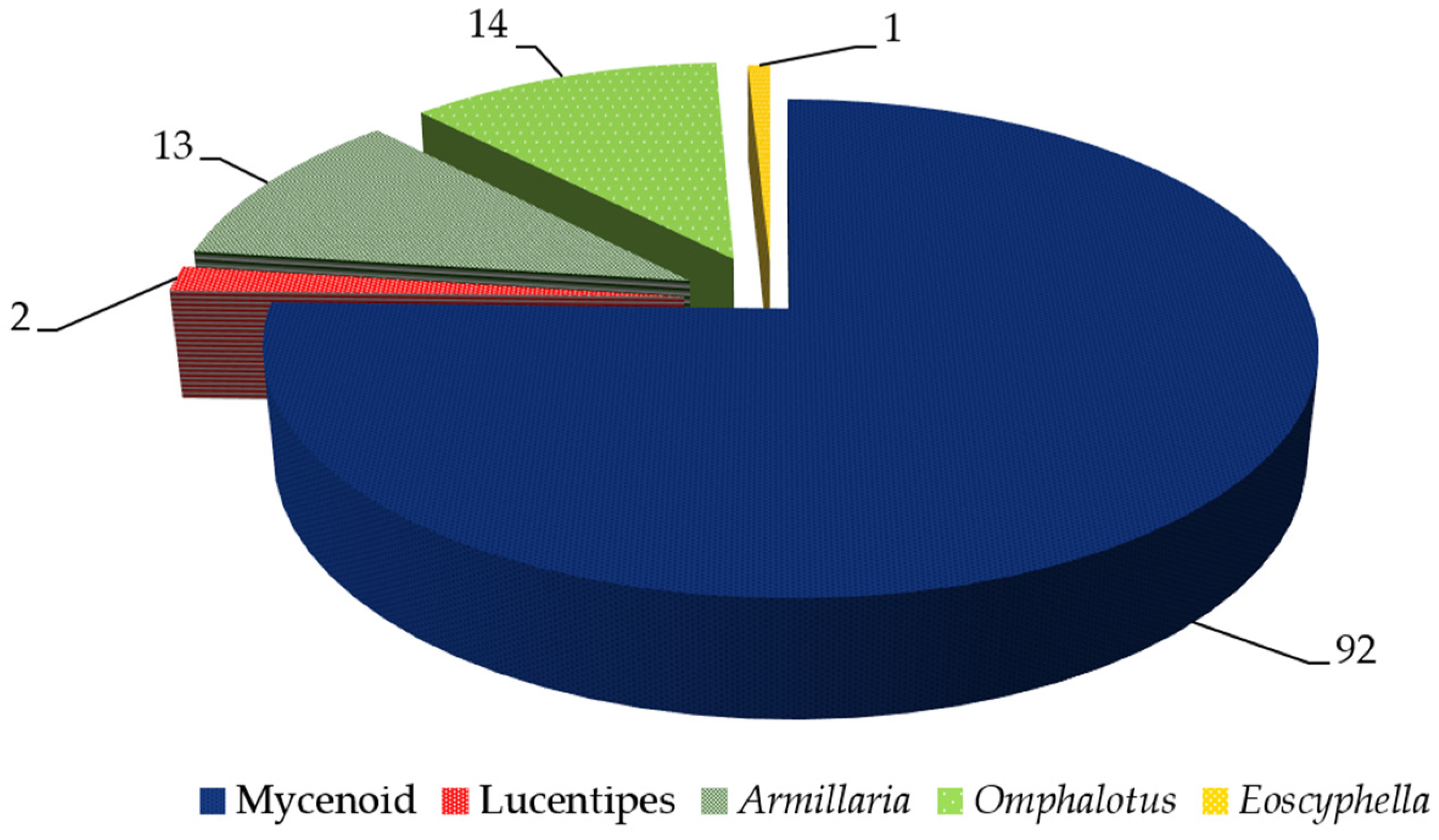

| Fungal Taxa | Distribution | Glowing Part | References | |||||
|---|---|---|---|---|---|---|---|---|
| Mycelium | Fruiting Bodies | Cap | Stipe | Spores | ||||
| Armillaria Lineage | Armillaria borealis | Russia | + | / | / | / | / | [40] |
| Armillaria calvescens | The USA | + | / | / | / | / | [41] | |
| Armillaria cepistipes | The USA | + | / | / | / | / | [41] | |
| Armillaria fuscipes | Malaysia | + | / | / | / | / | [42] | |
| Armillaria gallica | Europe and the USA | + | / | / | / | / | [42] | |
| Armillaria gemina | The USA | + | / | / | / | / | [42] | |
| Armillaria mellea | China, Europe, India, and the USA | + | / | / | / | / | [42,43,44] | |
| Armillaria nabsnona | The USA | + | / | / | / | / | [41] | |
| Armillaria novae-zelandiae | New Zealand | + | / | / | / | / | [45] | |
| Armillaria ostoyae | Europe and USA | + | / | / | / | / | [42] | |
| Armillaria sinapina | The USA | + | / | / | / | / | [41] | |
| Desarmillaria ectypa | Europe | + | + | + | / | / | [46] | |
| Desarmillaria tabescens | Europe and the USA | + | + | / | / | / | [42] | |
| Eoscyphella Lineage | Eoscyphella luciurceolata | Brazil | ? | + | ? | ? | ? | [47] |
| Lucentipes Lineage | Mycena lucentipes | South America and ♣ | + | + | ? | + | / | [42] |
| Gerronema viridilucens | South America | + | + | + | + | / | [42] | |
| Mycenoid Lineage | Cruentomycena orientalis | Japan | + | + | + | + | / | [14] |
| Dictyopanus foliicola | Japan | + | + | / | / | / | [42] | |
| Favolaschia xtbgensis | China | + | + | + | + | + | [48] | |
| Favolaschia tonkinensis | China | ? | + | + | + | / | [49] | |
| Favolaschia peziziformis | Japan | ? | + | + | + | / | [50,51] | |
| Filoboletus manipularis | Africa, China, Sri Lanka, Thailand, and ♪ | ? | + | / | + | / | [52,53,54] | |
| Filoboletus hanedae | Japan | ? | + | / | + | / | [42] | |
| Filoboletus pallescens | ♪ | ? | + | ? | ? | ? | [42] | |
| Filoboletus yunnanensis | China | ? | + | ? | ? | / | [52,54,55] | |
| Gerronema glutinipes | Africa and China | ? | + | / | / | / | [52] | |
| Mycena abieticola | Brazil | ? | + | + | + | / | [55] | |
| Mycena aspratilis | Brazil and Puerto Rico | / | + | / | + | / | [55] | |
| Mycena asterina | South America | + | + | + | / | / | [42] | |
| Mycena cahaya | ♪ | + | + | + | + | / | [56] | |
| Mycena chlorophos | China, Japan, the Pacific Islands, Sri Lanka, and ♪ | + | + | + | + | / | [42,53] | |
| Mycena citricolor | South America and the USA | + | / | / | / | / | [42] | |
| Mycena coralliformis | ♪ | + | / | / | / | / | [53] | |
| Mycena cristinae | Brazil | + | + | / | / | / | [57] | |
| Mycena crocata | Switzerland | + | / | / | / | / | [58] | |
| Mycena daisyogunensis | Japan | ? | + | ? | ? | / | [42] | |
| Mycena deeptha | India | + | / | / | / | / | [59] | |
| Mycena deformis | Brazil | + | / | / | / | / | [60] | |
| Mycena discobasis | Africa and South America | ? | + | + | + | / | [42] | |
| Mycena epipterygia | Europe, the USA, and Japan | + | / | / | / | / | [42] | |
| Mycena fera | South America | ? | + | + | + | / | [42] | |
| Mycena flammifera | Japan | + | + | + | + | / | [50,51] | |
| Mycena fulgoris | Mexico | / | + | / | + | / | [61] | |
| Mycena galopus | Europe, the USA, and Japan | + | / | / | / | / | [42] | |
| Mycena globulispora | Brazil and Mexico | ? | + | / | + | / | [60,61] | |
| Mycena gombakensis | ♪ | + | + | + | + | / | [53] | |
| Mycena guzmanii | Mexico | + | + | + | + | / | [61] | |
| Mycena haematopus | China, Europe, the USA, Japan, and South America | + | + | + | / | / | [42] | |
| Mycena illuminans | Japan and ♪ | ? | + | + | / | / | [42,53] | |
| Mycena inclinata | Africa, China, Europe, and the USA | + | / | / | / | / | [42] | |
| Mycena jingyinga | China | + | / | / | / | / | [34] | |
| Mycena kentingensis | China | + | + | + | / | / | [61] | |
| Mycena lacrimans | South America | ? | + | / | + | / | [42] | |
| Mycena lamprocephala | Brazil | + | + | + | + | ? | [62] | |
| Mycena lazulina | Japan | + | + | + | + | / | [50,51] | |
| Mycena luceata | Mexico | ? | + | + | / | ? | [23] | |
| Mycena luciferina | Mexico | ? | + | + | / | ? | [23] | |
| Mycena lucinieblae | Mexico | + | / | / | / | ? | [23] | |
| Mycena luguensis | China | + | / | / | / | / | [34] | |
| Mycena lumina | Mexico | + | + | + | + | / | [61] | |
| Mycena luxaeterna | Brazil | + | + | / | + | / | [55] | |
| Mycena luxarboricola | Brazil | ? | + | + | + | / | [55] | |
| Mycena lux-coeli | Japan | ? | + | + | + | / | [42] | |
| Mycena luxfoliata | Japan | + | / | / | / | / | [50,51] | |
| Mycena luxfoliicola | Mexico | + | + | + | + | / | [61] | |
| Mycena luxmanantlanensis | Mexico | + | + | + | / | ? | [23] | |
| Mycena luxperpetua | Puerto Rico | + | + | + | + | / | [42] | |
| Mycena maculata | Africa, Europe, and the USA | + | / | / | / | / | [42] | |
| Mycena margarita | Belize, Dominican Republic, Jamaica, Puerto Rico, and Brazil | / | + | + | + | / | [42,63] | |
| Mycena nebula | Mexico | ? | + | + | + | / | [61] | |
| Mycena nocticaelum | ♪ | + | + | + | / | / | [53] | |
| Mycena noctilucens | Pacific Islands and ♪ | ? | + | + | + | / | [42,53] | |
| Mycena oculisnymphae | Brazil | / | + | + | + | / | [60] | |
| Mycena olivaceomarginata | Europe and the USA | + | / | / | / | / | [42] | |
| Mycena perlae | Mexico | / | + | + | / | / | [61] | |
| Mycena polygramma | China, Europe, the USA, Japan, and Africa | + | + | / | / | / | [42] | |
| Mycena pseudostylobates | Japan | + | ? | ? | ? | / | [42] | |
| Mycena pura | China, Europe, the USA, Japan, and South America | + | / | / | / | / | [42] | |
| Mycena rosea | Europe | + | / | / | / | / | [42] | |
| Mycena roseoflava | New Zealand | + | + | / | + | / | [45] | |
| Mycena sanguinolenta | China, Europe, the USA, and Japan | + | / | / | / | / | [42] | |
| Mycena seminau | ♪ | + | + | + | / | / | [56] | |
| Mycena silvaelucens | ♪ | ? | + | + | + | / | [42,56] | |
| Mycena sinar | ♪ | + | + | + | + | / | [56] | |
| Mycena singeri | South America and ♣ | ? | + | + | + | / | [42] | |
| Mycena sophiae | Mexico | + | / | / | / | ? | [23] | |
| Mycena sp. (PDD 80772) | New Zealand | ? | + | / | / | / | [42] | |
| Mycena sp. (SP #380150) | South America | + | + | / | / | / | [42,64] | |
| Mycena sp. (SP #380281) | South America | ? | + | / | / | / | [42,64] | |
| Mycena stellaris | Japan | + | + | + | + | / | [50,51] | |
| Mycena stylobates | Africa, China, Europe, the USA, and Japan | + | / | / | / | / | [42] | |
| Mycena tintinnabulum | Europe | + | / | / | / | / | [42] | |
| Mycena venus | China | + | / | / | / | / | [34] | |
| Mycena zephirus | Europe | + | / | / | / | / | [42] | |
| Panellus luminescens | ♪ | + | + | + | + | ? | [65,66] | |
| Panellus luxfilamentus | Sri Lanka and ♪ | + | / | / | / | / | [56] | |
| Panellus pusillus | Africa, Australasia, China, Japan, the USA, South America, and ♪ | ? | + | ? | ? | / | [42,49] | |
| Panellus stipticus | Africa, Australasia, China, Europe, Japan, the USA, and South America | + | + | + | / | / | [42] | |
| Resinomycena fulgens | Japan | ? | + | + | + | / | [50,51] | |
| Resinomycena petarensis | Brazil | + | / | / | / | / | [60] | |
| Roridomyces irritans | Australasia | / | + | + | / | ? | [42] | |
| Roridomyces lamprosporus | Brazil, Ceylon, Malaysia, and Papua New Guinea, Singapore, and Trinidad | / | + | / | / | + | [67] | |
| Roridomyces phyllostachydis | India | ? | + | / | + | / | [10,22] | |
| Roridomyces pruinosoviscidus | Australasia and ♪ | + | + | + | + | ? | [42,53] | |
| Roridomyces roridus | China, Europe, the USA, South America, and Japan | + | / | / | / | / | [42,68] | |
| Roridomyces sublucens | Indonesia and ♪ | / | + | + | + | / | [42] | |
| Roridomyces viridiluminus | China | + | + | + | + | / | [69] | |
| Omphalotus Lineage | Marasmiellus venosus | Japan | + | + | / | / | / | [50,51] |
| Marasmiellus lucidus | Japan | ? | + | + | + | / | [50,51] | |
| Neonothopanus gardneri | South America | ? | + | + | + | / | [42] | |
| Neonothopanus nambi | Australasia, China, South America, Thailand, ♪, and ♣ | ? | + | + | + | / | [42,53,54] | |
| Nothopanus noctilucens | Japan | ? | + | / | / | / | [42] | |
| Omphalotus guepiniiformis | China and Japan | + | + | / | / | / | [42,54,70] | |
| Omphalotus illudens | Europe and the USA | + | + | + | / | / | [42] | |
| Omphalotus mangensis | China | ? | + | + | / | / | [42,54,71] | |
| Omphalotus nidiformis | Australasia | ? | + | + | + | / | [42] | |
| Omphalotus olearius | China and Europe | + | + | + | + | / | [42,54] | |
| Omphalotus olivascens | The USA | / | + | / | / | / | [42] | |
| Omphalotus subilludens | The USA | ? | + | / | / | ? | [72] | |
| Pleurotus decipiens | ♪ | ? | + | / | / | / | [42] | |
| Pleurotus nitidus | Japan | ? | + | / | / | / | [50,51] | |
| Ascomycota | Xylaria hypoxylon | Europe | ? | + | ? | ? | / | [73] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, W.; Priyashantha, A.K.H.; Galappaththi, M.C.A.; Tibpromma, S.; Dai, D.-Q.; Patabendige, N.M.; Premarathne, B.M.; Kulasuriya, D.M.; Ediriweera, A.N.; Nimalrathna, T.S.; et al. Fungal Bioluminescence: Past, Present, and Future. Diversity 2024, 16, 539. https://doi.org/10.3390/d16090539
Lu W, Priyashantha AKH, Galappaththi MCA, Tibpromma S, Dai D-Q, Patabendige NM, Premarathne BM, Kulasuriya DM, Ediriweera AN, Nimalrathna TS, et al. Fungal Bioluminescence: Past, Present, and Future. Diversity. 2024; 16(9):539. https://doi.org/10.3390/d16090539
Chicago/Turabian StyleLu, Wenhua, Alviti Kankanamalage Hasith Priyashantha, Mahesh C. A. Galappaththi, Saowaluck Tibpromma, Dong-Qin Dai, Nimesha M. Patabendige, Bhagya M. Premarathne, Dinesh M. Kulasuriya, Aseni N. Ediriweera, Thilina S. Nimalrathna, and et al. 2024. "Fungal Bioluminescence: Past, Present, and Future" Diversity 16, no. 9: 539. https://doi.org/10.3390/d16090539
APA StyleLu, W., Priyashantha, A. K. H., Galappaththi, M. C. A., Tibpromma, S., Dai, D.-Q., Patabendige, N. M., Premarathne, B. M., Kulasuriya, D. M., Ediriweera, A. N., Nimalrathna, T. S., Suwannarach, N., Lumyong, S., Tang, A., Shao, S.-C., & Karunarathna, S. C. (2024). Fungal Bioluminescence: Past, Present, and Future. Diversity, 16(9), 539. https://doi.org/10.3390/d16090539













