Mechanisms and Impact of Biofilms and Targeting of Biofilms Using Bioactive Compounds—A Review
Abstract
1. Introduction
2. Stages in Biofilm Formation and Its Development
2.1. Cellular Attachment
2.2. Microcolonies Formation
2.3. Biofilm Maturation
2.4. Detachment of Biofilm
3. Factors Contributing to Biofilm Formation
3.1. Structural Factors
3.1.1. EPS
3.1.2. Pili
3.1.3. Flagella
3.2. Environmental Factors
3.2.1. Nutritional Cues
3.2.2. Oxygen Levels
3.2.3. Temperature
3.2.4. pH Levels
3.2.5. Exposure to Antimicrobials
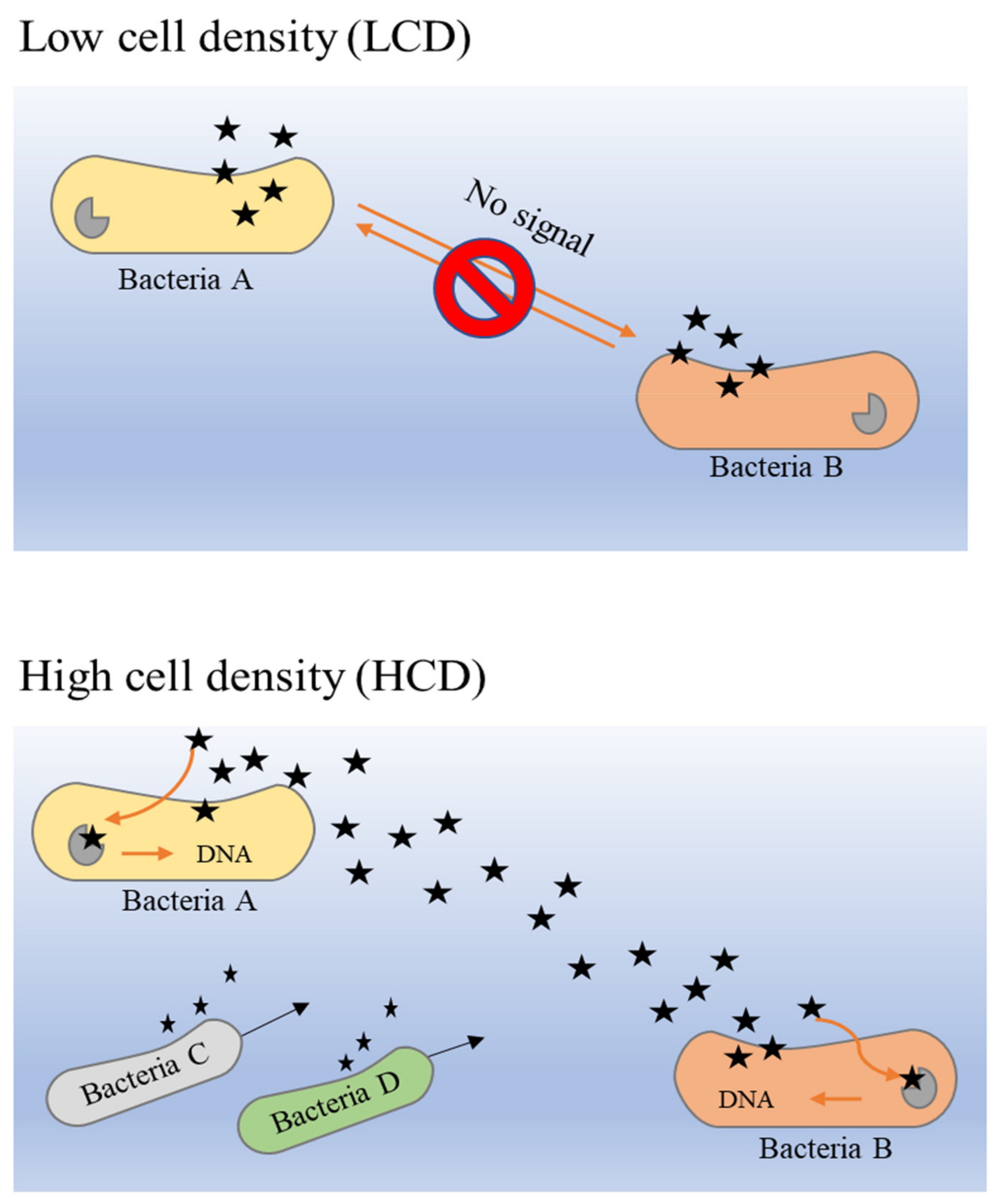
4. Quorum Sensing in Biofilm Formation
5. Consequences of Biofilm Formation
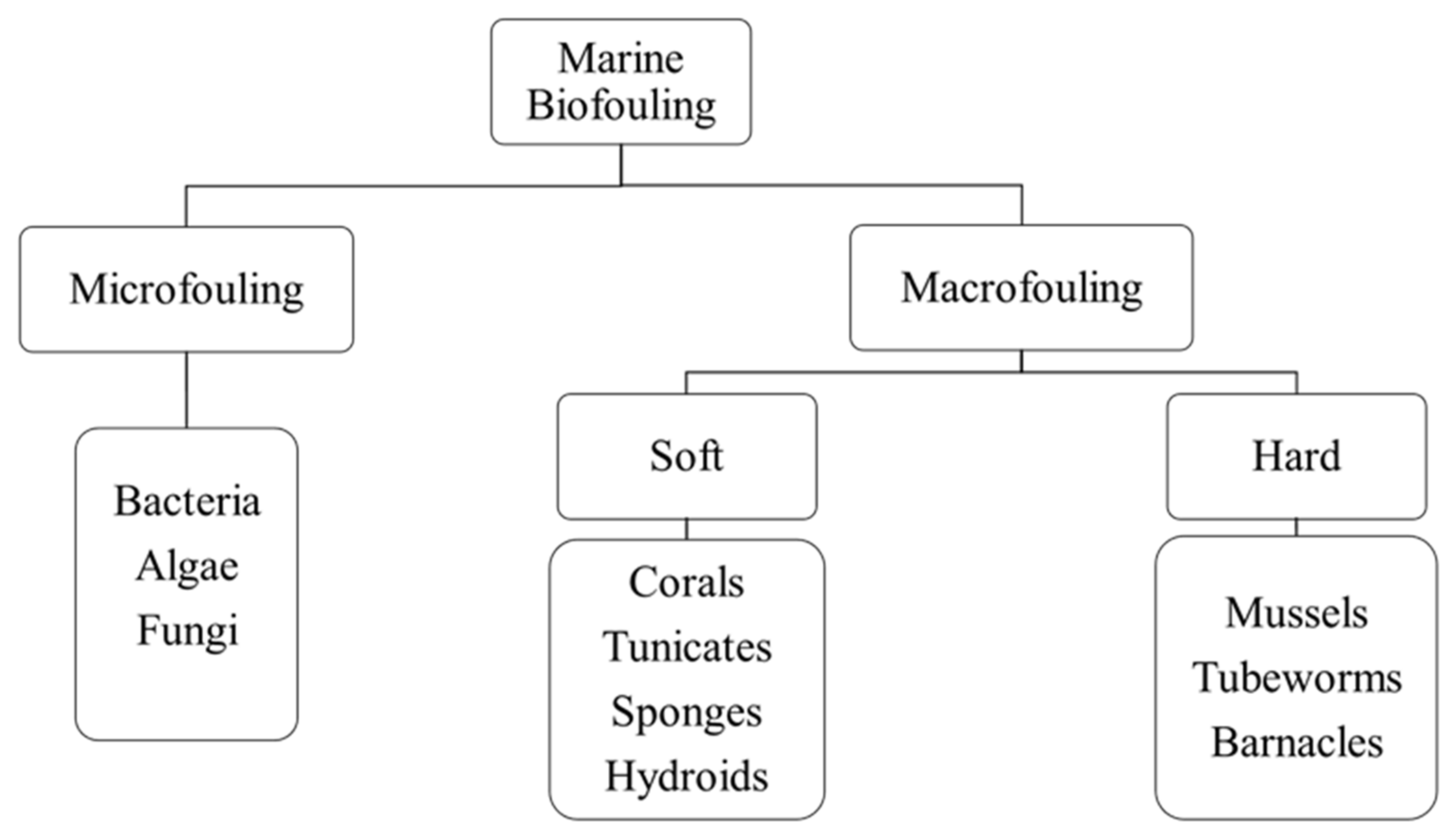
5.1. Biofouling
5.1.1. Biofouling in Marine Industries
5.1.2. Biofouling in Food and Beverage Industries
5.1.3. Biofouling in Medical Industries
5.2. Biofilm-Related Infections
5.2.1. Periodontitis
5.2.2. Rhinosinusitis
5.2.3. Cystic Fibrosis (CF)
6. Use of Bioactive Compounds as Antibiofilm Agents
6.1. Plant-Derived Bioactive Compounds
6.1.1. Phenolics and Polyphenolics
Flavonoids
Tannins
Phenolic Acids
Coumarins
6.1.2. Alkaloids
6.1.3. Terpenoids and Essential Oils
6.1.4. Lectins
6.1.5. Peptides
6.2. Animal-Derived Bioactive Compounds
6.2.1. Chitosan
6.2.2. Hyaluronic Acid
7. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Armbruster, C.R.; Parsek, M.R. New insight into the early stages of biofilm formation. Proc. Natl. Acad. Sci. USA 2018, 115, 4317–4319. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, R. Biofilms: Microbial cities of scientific significance. J. Microbiol. Exp. 2014, 1, 84–98. [Google Scholar] [CrossRef]
- Tasneem, U.; Yasin, N.; Nisa, I.; Shah, F.; Rasheed, U.; Momin, F.; Zaman, S.; Qasim, M. Biofilm producing bacteria: A serious threat to public health in developing countries. J. Food Sci. Nutr. 2018, 1, 25–31. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–663. [Google Scholar] [CrossRef] [PubMed]
- Karygianni, L.; Ren, Z.; Koo, H.; Thurnheer, T. Biofilm Matrixome: Extracellular components in structured microbial communities. Trends Microbiol. 2020, 28, 668–681. [Google Scholar] [CrossRef] [PubMed]
- Costa-Orlandi, C.B.; Sardi, J.; Pitangui, N.S.; de Oliveira, H.C.; Scozorni, L.; Galeane, M.C.; Medina-Alarcón, K.P.; Melo, W.; Marcelino, M.Y.; Braz, J.D.; et al. Fungal biofilms and polymicrobial diseases. J. Fungi 2017, 3, 22. [Google Scholar] [CrossRef]
- Bonaventura, G.D.; Piccolomini, R.; Paludi, D.; D’Orio, V.; Vergara, A.; Conter, M.; Ianieri, A. Influence of temperature on biofilm formation by Listeria monocytogenes on various food-contact surfaces: Relationship with motility and cell surface hydrophobicity. J. Appl. Microbiol. 2008, 104, 1552–1561. [Google Scholar] [CrossRef]
- Hostacká, A.; Ciznar, I.; Mária Štefkoviková, M. Temperature and pH Affect the Production of Bacterial Biofilm. Folia Microbiol. 2010, 55, 75–78. [Google Scholar] [CrossRef]
- Singh, R.; Shivaprakash, M.R.; Chakrabarti, A. Biofilm formation by zygomycetes: Quantification, structure and matrix composition. Microbiology 2011, 157, 2611–2618. [Google Scholar] [CrossRef]
- Fanning, S.; Mitchell, A.P. Fungal biofilms. PLoS Pathog. 2012, 8, e1002585. [Google Scholar] [CrossRef]
- D’Urzo, N.; Martinelli, M.; Pezzicoli, A.; De Cesare, V.; Pinto, V.; Margarit, I.; Telford, J.L.; Maione, D. Acidic pH strongly enhances in vitro biofilm formation by a subset of hypervirulent ST-17 Streptococcus agalactiae strains. Appl. Environ. Microbiol. 2014, 80, 2176–2185. [Google Scholar] [CrossRef]
- Sarah, L.S.; Pryjma, M.; Gaynor, E.C. Flagella-mediated adhesion and extracellular DNA release contribute to biofilm formation and stress tolerance of Campylobacter jejuni. PLoS ONE 2014, 9, e106063. [Google Scholar]
- Zhang, W.B.; Seminara, A.; Suaris, M.; Brenner, M.P.; Weitz, D.A.; Angelini, T.E. Nutrient depletion in Bacillus subtilis biofilms triggers matrix production. New J. Phys. 2014, 16, 15028. [Google Scholar] [CrossRef]
- Luo, L.M.; Wu, L.J.; Xiao, Y.L.; Zhao, D.; Chen, Z.X.; Kang, M.; Zhang, Q.; Xie, Y. Enhancing pili assembly and biofilm formation in Acinetobacter baumannii ATCC19606 using non-native acyl-homoserine lactones. BMC Microbiol. 2015, 15, 62. [Google Scholar] [CrossRef] [PubMed]
- Maldarelli, G.A.; Piepenbrink, K.H.; Scott, A.J.; Freiberg, J.A.; Song, Y.; Achermann, Y.; Ernst, R.K.; Shirtliff, M.E.; Sundberg, E.J.; Donnenberg, M.S.; et al. Type IV pili promote early biofilm formation by Clostridium difficile. Pathog. Dis. 2016, 74, 1–10. [Google Scholar] [CrossRef]
- Kirchhoff, L.; Olsowski, M.; Zilmans, K.; Dittmer, S.; Haase, G.; Sedlacek, L.; Steinmann, E.; Buer, J.; Rath, P.M.; Steinmann, J. Biofilm formation of the black yeast-like fungus Exophiala dermatitidis and its susceptibility to antiinfective agents. Sci. Rep. 2017, 7, 42886. [Google Scholar] [CrossRef] [PubMed]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Khatoon, Z.; McTiernan, C.; Suuronen, E.; Mah, T.; Alarcon, E. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018, 4, e01067. [Google Scholar] [CrossRef]
- Sharma, D.; Misba, L.; Khan, A.U. Antibiotics versus biofilm: An emerging battleground in microbial communities. Antimicrob. Resist. Infect. Control 2019, 8, 1–10. [Google Scholar] [CrossRef]
- Yin, W.; Wang, Y.; Liu, L.; He, J. Biofilms: The microbial “protective clothing” in extreme environments. Int. J. Mol. Sci. 2019, 20, 3423. [Google Scholar] [CrossRef]
- Rumbaugh, K.P.; Sauer, K. Biofilm dispersion. Nat. Rev. Microbiol. 2020, 18, 571–586. [Google Scholar] [CrossRef] [PubMed]
- Domenech, M.; Ramos-Sevillano, E.; García, E.; Moscoso, M.; Yuste, J. Biofilm formation avoids complement immunity and phagocytosis of Streptococcus pneumoniae. Infect. Immun. 2013, 81, 2606–2615. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.; Fiehn, N.E. Dental biofilm infections—An update. J. Pathol. Microbiol. Immunol. 2017, 125, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Crouzet, M.; Le Senechal, C.; Brözel, V.S.; Costaglioli, P.; Barthe, C.; Bonneu, M.; Garbay, B.; Vilain, S. Exploring early steps in biofilm formation: Set-up of an experimental system for molecular studies. BMC Microbiol. 2014, 14, 253. [Google Scholar] [CrossRef] [PubMed]
- Palmer, J.; Flint, S.; Brooks, J. Bacterial cell attachment, the beginning of a biofilm. J. Ind. Microbiol. Biotechnol. 2007, 34, 577–588. [Google Scholar] [CrossRef]
- Petrova, O.E.; Sauer, K. Sticky situations: Key components that control bacterial surface attachment. J. Bacteriol. 2012, 194, 2413–2425. [Google Scholar] [CrossRef]
- Caiazza, N.C.; O’Toole, G.A. SadB is required for the transition from reversible to irreversible attachment during biofilm formation by Pseudomonas aeruginosa PA14. J. Bacteriol. 2004, 186, 4476–4485. [Google Scholar] [CrossRef]
- Rabin, N.; Zheng, Y.; Opoku, T.C.; Du, Y.X.; Bonsu, F.; Sintim, H.O. Biofilm formation mechanisms and targets for developing antibiofilm agents. Future Med. Chem. 2015, 7, 493–512. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.; Neu, T.; Wozniak, D. The EPS matrix: The “house of biofilm cells”. J. Bacteriol. 2007, 189, 7945–7947. [Google Scholar] [CrossRef]
- Drescher, K.; Dunkel, J.; Nadell, C.D.; Teeffelen, S.; Grnja, I.; Wingreen, N.S.; Stone, H.A.; Bassler, B.L. Architectural transitions in Vibrio cholerae biofilms at single-cell resolution. Proc. Natl. Acad. Sci. USA 2016, 113, E2066–E2072. [Google Scholar] [CrossRef]
- Bowen, W.; Burne, R.; Wu, H.; Koo, H. Oral biofilms: Pathogens, matrix, and polymicrobial interactions in microenvironments. Trends Microbiol. 2018, 26, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Hobley, L.; Harkins, C.; MacPhee, C.; Stanley-Wall, N. Giving structure to the biofilm matrix: An overview of individual strategies and emerging common themes. FEMS Microbiol. Rev. 2015, 39, 649–669. [Google Scholar] [CrossRef]
- Preda, V.G.; Săndulescu, O. Communication is the key: Biofilms, quorum sensing, formation and prevention. Discoveries 2019, 7, e100. [Google Scholar] [CrossRef] [PubMed]
- Saxena, P.; Joshi, Y.; Rawat, K.; Bisht, R. Biofilms: Architecture, resistance, quorum sensing and control mechanisms. Indian J. Microbiol. 2019, 59, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Wilking, J.N.; Zaburdaev, V.; De Volder, M.; Losick, R.; Brenner, M.P.; Weitz, D.A. Liquid transport facilitated by channels in Bacillus subtilis biofilms. Proc. Natl. Acad. Sci. USA 2013, 110, 848–852. [Google Scholar] [CrossRef] [PubMed]
- Parsek, M.; Singh, P. Bacterial biofilms: An emerging link to disease pathogenesis. Annu. Rev. Microbiol. 2003, 57, 677–701. [Google Scholar] [CrossRef]
- Chua, S.L.; Liu, Y.; Yam, J.K.H.; Chen, Y.C.; Veiborg, R.M.; Tan, B.G.C.; Kjelleberg, S.; Tolker-Nielsen, T.; Givskov, M.; Yang, L. Dispersed cells represent a distinct stage in the transition from bacterial biofilm to planktonic lifestyles. Nat. Commun. 2014, 5, 4462. [Google Scholar] [CrossRef]
- Kaplan, J.B. Biofilm dispersal: Mechanisms, clinical implications, and potential therapeutic uses. J. Dent. Res. 2010, 89, 205–218. [Google Scholar] [CrossRef]
- Fleming, D.; Rumbaugh, K.P. Approaches to dispersing medical biofilms. Microorganisms 2017, 5, 15. [Google Scholar] [CrossRef]
- Kostakioti, M.; Hadjifrangiskou, M.; Hultgren, S.J. Bacterial biofilms: Development, dispersal, and therapeutic strategies in the dawn of the postantibiotic era. Cold Spring Harb. Perspect. Med. 2013, 3, a010306. [Google Scholar] [CrossRef]
- Guilhen, C.; Forestier, C.; Balestrino, D. Biofilm dispersal: Multiple elaborate strategies for dissemination of bacteria with unique properties. Mol. Microbiol. 2017, 105, 188–210. [Google Scholar] [CrossRef]
- Pirrone, M.; Pinciroli, R.; Berra, L. Microbiome, biofilms, and pneumonia in the ICU. Curr. Opin. Infect. Diseases 2016, 29, 160–166. [Google Scholar] [CrossRef]
- Kimura, K.R.; Nakata, M.; Sumitomo, T.; Kreikemeyer, B.; Podbielski, A.; Terao, Y.; Kawabata, S. Involvement of T6 pili in biofilm formation by serotype M6 Streptococcus pyogenes. J. Bacteriol. 2012, 194, 804–812. [Google Scholar] [CrossRef]
- Conrad, J.C. Physics of bacterial near-surface motility using flagella and type IV Pili: Implications for biofilm formation. Res. Microbiol. 2012, 163, 619–629. [Google Scholar] [CrossRef]
- Voběrková, S.; Hermanová, S.; Hrubanová, K.; Krzyžánek, V. Biofilm formation and extracellular polymeric substances (EPS) production by Bacillus subtilis depending on nutritional conditions in the presence of polyester film. Folia Microbiol. 2015, 61, 91–100. [Google Scholar] [CrossRef]
- Marvasi, M.; Visscher, P.T.; Martinez, L.C. Exopolymeric substances (EPS) from Bacillus subtilis:polymers and genes encoding their synthesis. FEMS Microbiol. Lett. 2010, 313, 1–9. [Google Scholar] [CrossRef]
- Czaczyk, K.; Myszka, K. Biosynthesis of extracellular polymeric substances (EPS) and its role in microbial biofilm formation. Pol. J. Environ. Stud. 2007, 16, 799–806. [Google Scholar]
- Parikh, A.; Madamwar, D. Partial characterization of extracellular polysaccharides from cyanobacteria. Bioresour. Technol. 2006, 97, 1822–1827. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, K.; Grigorova, D. Production and properties of exopolysaccharide by Rhodotorula acheniorum MC. Food Res. Int. 1999, 32, 473–477. [Google Scholar] [CrossRef]
- Hwang, H.J.; Kim, S.W.; Xu, C.P.; Choi, J.W.; Yun, J.W. Morphological and rheological properties of the three different species of basidiomycetes Phiellinus in submerged cultures. J. Appl. Microbiol. 2004, 96, 1296–1305. [Google Scholar] [CrossRef]
- Jain, R.; Raghukumar, S.; Tharanathan, R.; Bhosle, N.B. Extracellular polysaccharide production by Thraustochytrid protists. Mar. Biotechnol. 2005, 7, 184–192. [Google Scholar] [CrossRef]
- Li, Y.; Chen, W.; Liu, Q.; Xiang, W.; Zheng, X.; Qu, J. Effect on composition and quantity of extracellular polymeric substances in an innovative hybrid membrane bioreactor under different sludge retention times. Environ. Pollut. Bioavailab. 2019, 31, 103–111. [Google Scholar] [CrossRef]
- Roca, C.; Alves, V.; Freitas, F.; Reis, M. Exopolysaccharides enriched in rare sugars: Bacterial sources, production, and applications. Front. Microbiol. 2015, 6, 288. [Google Scholar] [CrossRef]
- Costa, O.; Raaijmakers, J.; Kuramae, E. Microbial extracellular polymeric substances: Ecological function and impact on soil aggregation. Front. Microbiol. 2018, 9, 1636. [Google Scholar] [CrossRef] [PubMed]
- Dragoš, A.; Kovács, Á. The peculiar functions of the bacterial extracellular matrix. Trends Microbiol. 2017, 25, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Hendrickx, L.; Hausner, M.; Wuertz, S. Natural genetic transformation in monoculture Acinetobacter sp. strain BD413 biofilms. Appl. Environ. Microbiol. 2003, 69, 1721–1727. [Google Scholar] [CrossRef][Green Version]
- Zeng, L.H.; Zhang, L.; Wang, P.R.; Meng, G.Y. Structural basis of host recognition and biofilm formation by Salmonella Saf pili. eLife 2017, 6, e28619. [Google Scholar] [CrossRef]
- Nallapareddy, S.R.; Singh, K.V.; Sillanpää, J.; Garsin, D.A.; Höök, M.; Erlandsen, S.L.; Murray, B.E. Endocarditis and biofilm-associated pili of Enterococcus faecalis. J. Clin. Investig. 2006, 116, 2799–2807. [Google Scholar] [CrossRef] [PubMed]
- Klausen, M.; Heydorn, A.; Ragas, P.; Lambertsen, L.; Aaes-Jorgensen, A.; Molin, S.; Tolker-Nielsen, T. Biofilm formation by Pseudomonas aeruginosa wild type, flagella and type IV pili mutants. Mol. Microbiol. 2003, 48, 1511–1524. [Google Scholar] [CrossRef]
- Serra, D.O.; Richter, A.M.; Klauck, G.; Mika, F.; Hengge, R. Microanatomy at cellular resolution and spatial order of physiological differentiation in a bacterial biofilm. mBio 2013, 4, e00103-13. [Google Scholar] [CrossRef]
- Du, B.; Gu, Y.; Chen, G.W.; Wang, G.; Liu, L. Flagellar motility mediates early-stage biofilm formation in oligotrophic aquatic environment. Ecotoxicol. Environ. Saf. 2020, 194, 110340. [Google Scholar] [CrossRef]
- Guttenplan, S.B.; Kearns, D.B. Regulation of flagellar motility during biofilm formation. FEMS Microbiol. Rev. 2013, 37, 849–871. [Google Scholar] [CrossRef] [PubMed]
- Valentini, M.; Filloux, A. Biofilms and cyclic di-GMP (c-di-GMP) signaling: Lessons from Pseudomonas aeruginosa and other bacteria. J. Biol. Chem. 2016, 291, 12547–12555. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Cole, S.; Badel-Berchoux, S.; Guillier, L.; Felix, B.; Krezdorn, N.; Hébraud, M.; Bernardi, T.; Sultan, I.; Piveteau, P. Biofilm formation of listeria monocytogenes strains under food processing environments and pan-genome-wide association study. Front. Microbiol. 2019, 10, 2698. [Google Scholar] [CrossRef] [PubMed]
- Mashruwala, A.A.; Guchte, A.; Boyd, J.M. Impaired respiration elicits SrrAB-dependent programmed cell lysis and biofilm formation in Staphylococcus aureus. eLife 2017, 6, e23845. [Google Scholar] [CrossRef]
- Ciofu, O.; Tolker-Nielsen, T. Tolerance and resistance of Pseudomonas aeruginosa biofilms to antimicrobial agents-how P. aeruginosa can escape antibiotics. Front. Microbiol. 2019, 10, 913. [Google Scholar] [CrossRef]
- Thomas, V.C.; Fey, P.D. Biofilms: Take my breath away. eLife 2017, 6, e25739. [Google Scholar] [CrossRef]
- An, S.; Wu, J.; Zhang, L.H. Modulation of Pseudomonas aeruginosa biofilm dispersal by a cyclic-Di-GMP phosphodiesterase with a putative hypoxia-sensing domain. Appl. Environ. Microbiol. 2010, 76, 8160–8173. [Google Scholar] [CrossRef]
- Cramton, S.E.; Ulrich, M.; Götz, F.; Döring, G. Anaerobic conditions induce expression of polysaccharide intercellular adhesin in Staphylococcus aureus and Staphylococcus epidermidis. Infect. Immun. 2001, 69, 4079–4085. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.F.; Weiss, E.C.; Otto, M.; Fey, P.D.; Smeltzer, M.S.; Somerville, G.A. Staphylococcus aureus biofilm metabolism and the influence of arginine on polysaccharide intercellular adhesin synthesis, biofilm formation, and pathogenesis. Infect. Immun. 2007, 75, 4219–4226. [Google Scholar] [CrossRef] [PubMed]
- Ghotaslou, R.; Salahi, B. Effects of oxygen on in-vitro biofilm formation and antimicrobial resistance of Pseudomonas aeruginosa. Pharm. Sci. 2013, 19, 96–99. [Google Scholar]
- Mizan, F.R.; Jahid, I.K.; Park, S.Y.; Myoung, J.J. Effects of temperature on biofilm formation and quorum sensing of Aeromonas hydrophila. Ital. J. Food Sci. 2018, 30, 456–466. [Google Scholar]
- Obana, N.; Nakamura, K.; Nomura, N. A Sporulation factor is involved in the morphological change of Clostridium perfringens biofilms in response to temperature. J. Bacteriol. 2014, 196, 1540–1550. [Google Scholar] [CrossRef]
- Speranza, B.; Sinigaglia, M.; Corbo, M.R. Effects of nutritional and environmental conditions on Salmonella sp biofilm formation. J. Food Sci. 2011, 76, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Mathlouthi, A.; Pennacchietti, E.; De Biase, D. Effect of temperature, pH and plasmids on in vitro biofilm formation in Escherichia coli. Acta Nat. 2018, 10, 129–132. [Google Scholar] [CrossRef]
- Balcázar, J.; Subirats, J.; Borrego, C.M. The role of biofilms as environmental reservoirs of antibiotic resistance. Front. Microbiol. 2015, 6, 1216. [Google Scholar] [CrossRef] [PubMed]
- Salcedo, D.E.; Lee, J.H.; Ha, U.H.; Kim, S.P. The effects of antibiotics on the biofilm formation and antibiotic resistance gene transfer. Desalination Water Treat. 2014, 54, 3582–3588. [Google Scholar] [CrossRef]
- Strelkova, E.A.; Zhurina, M.V.; Plakunov, V.K.; Beliaev, S.S. Antibiotics stimulation of biofilm formation. Mikrobiologiia 2012, 81, 282–285. [Google Scholar] [CrossRef]
- Shrikant, P.; Chandrajit, L. Quorum sensing: An imperative longevity weapon in bacteria. Afr. J. Microbiol. Res. 2018, 12, 96–104. [Google Scholar] [CrossRef][Green Version]
- Tommonaro, G. Quorum Sensing: Molecular Mechanism and Biotechnological Application; Elsevier: Amsterdam, The Netherlands, 2019; pp. 3–22. [Google Scholar]
- Zhang, J.; Feng, T.; Wang, J.; Wang, Y.; Zhang, X. The mechanisms and applications of quorum sensing (QS) and quorum quenching (QQ). J. Ocean Univ. China 2019, 18, 1427–1442. [Google Scholar] [CrossRef]
- Mangwani, N.; Dash, H.; Chauhan, A.; Das, S. Bacterial quorum sensing: Functional features and potential applications in biotechnology. J. Mol. Microbiol. Biotechnol. 2012, 22, 215–227. [Google Scholar] [CrossRef]
- Terwagne, M.; Mirabella, A.; Lemaire, J.; Deschamps, C.; De Bolle, X.; Letesson, J. Quorum sensing and self-quorum quenching in the intracellular pathogen brucellamelitensis. PLoS ONE 2013, 8, e82514. [Google Scholar] [CrossRef] [PubMed]
- Papenfort, K.; Bassler, B. Quorum sensing signal–response systems in Gram-negative bacteria. Nat. Rev. Microbiol. 2016, 14, 576–588. [Google Scholar] [CrossRef] [PubMed]
- Rémy, B.; Mion, S.; Plener, L.; Elias, M.; Chabrière, E.; Daudé, D. Interference in bacterial quorum sensing: A biopharmaceutical perspective. Front. Pharmacol. 2018, 9, 203. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Gámez, S.; Sorg, R.; Domenech, A.; Kjos, M.; Weissing, F.; van Doorn, G.; Veening, J. Quorum sensing integrates environmental cues, cell density and cell history to control bacterial competence. Nat. Commun. 2017, 8, 854. [Google Scholar] [CrossRef] [PubMed]
- Ng, W.; Bassler, B. Bacterial quorum-sensing network architectures. Annu. Rev. Genet. 2009, 43, 197–222. [Google Scholar] [CrossRef]
- Hence, B.; Schuster, M. Core principles of bacterial autoinducer systems. Microbiol. Mol. Biol. Rev. 2015, 79, 153–169. [Google Scholar] [CrossRef]
- Rutherford, S.; Bassler, B. Bacterial quorum sensing: Its role in virulence and possibilities for its control. Cold Spring Harb. Perspect. Med. 2012, 2, a012427. [Google Scholar] [CrossRef]
- Trajtenberg, F.; Albanesi, D.; Ruétalo, N.; Botti, H.; Mechaly, A.; Nieves, M.; Aguilar, P.; Cybulski, L.; Larrieux, N.; de Mendoza, D.; et al. Allosteric activation of bacterial response regulators: The role of the cognate histidine kinase beyond phosphorylation. mBio 2014, 5, e02105-14. [Google Scholar] [CrossRef]
- Della Sala, G.; Teta, R.; Esposito, G.; Costantino, V. The chemical language of gram-negative bacteria. Quor. Sens. 2019, 3–28. [Google Scholar] [CrossRef]
- Jacobi, C.; Grundler, S.; Hsieh, C.; Frick, J.; Adam, P.; Lamprecht, G.; Autenrieth, I.; Gregor, M.; Malfertheiner, P. Quorum sensing in the probiotic bacterium Escherichia coli Nissle 1917 (Mutaflor)—evidence that furanosyl borate diester (AI-2) is influencing the cytokine expression in the DSS colitis mouse model. Gut Pathog. 2012, 4, 8. [Google Scholar] [CrossRef]
- Asfour, H. Anti-quorum sensing natural compounds. J. Microsc. Ultrastruct. 2018, 6, 1. [Google Scholar] [CrossRef]
- Monnet, V.; Juillard, V.; Gardan, R. Peptide conversations in Gram-positive bacteria. Crit. Rev. Microbiol. 2016, 42, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Schuster, M.; Joseph Sexton, D.; Diggle, S.; Peter Greenberg, E. Acyl-homoserine lactone quorum sensing: From evolution to application. Annu. Rev. Microbiol. 2013, 67, 43–63. [Google Scholar] [CrossRef] [PubMed]
- Fuqua, C.; Winans, S.; Greenberg, E. Census and consensus in bacterial ecosystems: The LuxR-LuxI family of quorum-sensing transcriptional regulators. Annu. Rev. Microbiol. 1996, 50, 727–751. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Zhang, A.; Yin, H.; Chu, W. Bacillus sp. QSI-1 modulate quorum sensing signals reduce aeromonas hydrophila level and alter gut microbial community structure in fish. Front. Cell. Infect. Microbiol. 2016, 6, 184. [Google Scholar] [CrossRef] [PubMed]
- Achari, G.; Ramesh, R. Characterization of bacteria degrading 3-hydroxy palmitic acid methyl ester (3OH-PAME), a quorum sensing molecule of Ralstonia solanacearum. Lett. Appl. Microbiol. 2015, 60, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.C.; Murthy, P.S.; Venkatesan, R.; Cooksey, K. Marine and Industrial Biofouling; Springer: Los Angeles, CA, USA, 2009. [Google Scholar]
- Plouguerné, E.; Hellio, C.; Deslandes, E.; Véron, B.; Stiger-Pouvreau, V. Anti-microfouling activities in extracts of two invasive algae: Grateloupia turuturu and Sargassum muticum. Bot. Mar. 2008, 51, 202–208. [Google Scholar] [CrossRef]
- Flemming, H. Microbial biofouling: Unsolved problems, insufficient approaches, and possible solutions. In Springer Series on Biofilms; Springer: Berlin/Heidelberg, Germany, 2011; pp. 81–109. [Google Scholar]
- Cloete, E.; Molobela, I.; Van Der Merwe, A.; Richards, M. Biofilms in the food and beverage industries: An introduction. In Biofilms in the Food and Beverage Industries; Woodhead Publishing: Sawston, UK, 2009; pp. 3–41. [Google Scholar]
- Bixler, G.; Bhushan, B. Biofouling: Lessons from nature. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2012, 370, 2381–2417. [Google Scholar] [CrossRef]
- Gu, J. Biofouling and Prevention. In Handbook of Environmental Degradation of Materials; Elsevier: Amsterdam, The Netherlands, 2012; pp. 243–282. [Google Scholar]
- Dobretsov, S.; Dahms, H.; Qian, P. Inhibition of biofouling by marine microorganisms and their metabolites. Biofouling 2006, 22, 43–54. [Google Scholar] [CrossRef]
- Dobretsov, S. Inhibition and induction of marine biofouling by biofilms. In Springer Series on Biofilms; Springer: Berlin/Heidelberg, Germany, 2008; pp. 293–313. [Google Scholar]
- Bressy, C.; Lejars, M. Marine fouling: An overview. J. Ocean Technol. 2014, 9, 19–28. [Google Scholar]
- Daal, L.; de Vos, F.; Soons, J.; de Vries, T. Membrane technologies for water treatment and reuse in the power industries. In Advances in Membrane Technologies for Water Treatment; Woodhead Publishing: Sawston, UK, 2015; pp. 605–624. [Google Scholar]
- De Carvalho, C. Marine biofilms: A successful microbial strategy with economic implications. Front. Mar. Sci. 2018, 5, 5. [Google Scholar] [CrossRef]
- Doble, M.; Venkatesan, R.; Vijaya Kumar, N.; Kumar, R. Polymers in a Marine Environment; Smithers Information: Shawbury, UK, 2014. [Google Scholar]
- Salta, M.; Chambers, L.; Wharton, J.; Wood, R.; Briand, J.F.; Blache, Y.; Stokes, K.R. Marine fouling organisms and their use in antifouling bioassays. In Proceedings of the EUROCORR, Nice, France, 6–9 September 2009. [Google Scholar]
- Lebret, K.; Thabard, M.; Hellio, C. Algae as marine fouling organisms: Adhesion damage and prevention. In Advances in Marine Antifouling Coatings and Technologies; Woodhead Publishing: Sawston, UK, 2009; pp. 80–112. [Google Scholar]
- Gordon, D.P.; Mawatari, S.F. Atlas of marine-fouling Bryozoa of New-Zealand ports and harbours. Misc. Publ. N. Z. Oceanogr. Inst. 1992, 107, 1–52. [Google Scholar]
- Baciocco, A.J. ‘Keynote address’. In Marine Biodeterioration: An Interdisciplinary Study; Costlow, J.D., Tipper, R.C., Eds.; Naval Institute Press: Annapolis, MD, USA, 1984; pp. 9–21. [Google Scholar]
- Munk, T.; Kane, D.; Yebra, D. The effects of corrosion and fouling on the performance of ocean-going vessels: A naval architectural perspective. In Advances in Marine Antifouling Coatings and Technologies; Woodhead Publishing: Sawston, UK, 2009; pp. 148–176. [Google Scholar]
- Moreira, J.; Gomes, L.; Simões, M.; Melo, L.; Mergulhão, F. The impact of material properties, nutrient load and shear stress on biofouling in food industries. Food Bioprod. Process. 2015, 95, 228–236. [Google Scholar] [CrossRef]
- Verran, J. Biofouling in food processing: Biofilm or bio transfer potential? Food Bioprod. Process. 2002, 80, 292–298. [Google Scholar] [CrossRef]
- Gule, N.; Begum, N.; Klumperman, B. Advances in biofouling mitigation: A review. Crit. Rev. Environ. Sci. Technol. 2016, 46, 535–555. [Google Scholar] [CrossRef]
- Mizan, M.; Jahid, I.; Ha, S. Microbial biofilms in seafood: A food-hygiene challenge. Food Microbiol. 2015, 49, 41–55. [Google Scholar] [CrossRef]
- Galié, S.; García-Gutiérrez, C.; Miguélez, E.; Villar, C.; Lombó, F. Biofilms in the food industry: Health aspects and control methods. Front. Microbiol. 2018, 9, 898. [Google Scholar] [CrossRef] [PubMed]
- Bridier, A.; Sanchez-Vizuete, P.; Guilbaud, M.; Piard, J.; Naïtali, M.; Briandet, R. Biofilm-associated persistence of food-borne pathogens. Food Microbiol. 2015, 45, 167–178. [Google Scholar] [CrossRef]
- Srey, S.; Jahid, I.; Ha, S. Biofilm formation in food industries: A food safety concern. Food Control 2013, 31, 572–585. [Google Scholar] [CrossRef]
- Umaraw, P.; Prajapati, A.; Verma, A.; Pathak, V.; Singh, V. Control of campylobacter in poultry industry from farm to poultry processing unit: A review. Crit. Rev. Food Sci. Nutr. 2015, 57, 659–665. [Google Scholar] [CrossRef]
- Wang, H.; Ding, S.; Dong, Y.; Ye, K.; Xu, X.; Zhou, G. Biofilm formation of salmonella serotypes in simulated meat processing environments and its relationship to cell characteristics. J. Food Prot. 2013, 76, 1784–1789. [Google Scholar] [CrossRef]
- Rajkowski, K. Biofilms in fish processing. In Biofilms in the Food and Beverage Industries; Woodhead Publishing: Sawston, UK, 2009; pp. 499–516. [Google Scholar]
- Hickey, C.; Sheehan, J.; Wilkinson, M.; Auty, M. Growth and location of bacterial colonies within dairy foods using microscopy techniques: A review. Front. Microbiol. 2015, 6, 99. [Google Scholar] [CrossRef] [PubMed]
- Jessen, B.; Lammert, L. Biofilm and disinfection in meat processing plants. Int. Biodeterior. Biodegrad. 2003, 51, 265–269. [Google Scholar] [CrossRef]
- Marotta, F.; Garofolo, G.; Di Donato, G.; Aprea, G.; Platone, I.; Cianciavicchia, S.; Alessiani, A.; Di Giannatale, E. Population diversity of campylobacter jejuni poultry and its dynamic of contamination in chicken meat. BioMed Res. Int. 2015, 2015, 859845. [Google Scholar] [CrossRef] [PubMed]
- Ehling-Schulz, M.; Frenzel, E.; Gohar, M. Food–bacteria interplay: Pathometabolism of emetic Bacillus cereus. Front. Microbiol. 2015, 6, 704. [Google Scholar] [CrossRef] [PubMed]
- Gamazo, C.; Solano, C.; Lasa, I. Biofilm formation by Salmonella in food processing environments. In Biofilms in the Food and Beverage Industries; Woodhead Publishing: Sawston, UK, 2009; pp. 226–249. [Google Scholar]
- Linscott, A. Food-borne illnesses. Clin. Microbiol. Newsl. 2011, 33, 41–45. [Google Scholar] [CrossRef]
- Giaouris, E.; Heir, E.; Hébraud, M.; Chorianopoulos, N.; Langsrud, S.; Møretrø, T.; Habimana, O.; Desvaux, M.; Renier, S.; Nychas, G. Attachment and biofilm formation by foodborne bacteria in meat processing environments: Causes, implications, role of bacterial interactions and control by alternative novel methods. Meat Sci. 2014, 97, 298–309. [Google Scholar] [CrossRef]
- Pozo, J.L.D. Biofilm-related disease. Expert Rev. Anti-Infect. Ther. 2017, 16, 51–65. [Google Scholar] [CrossRef]
- Darouiche, R. Treatment of infections associated with surgical implants. N. Engl. J. Med. 2004, 350, 1422–1429. [Google Scholar] [CrossRef]
- LoVetri, K.; Gawande, P.V.; Yakandawala, N.; Madhyastha, S. Biofouling and anti-fouling of medical devices. In Biofouling: Types, Impact and Anti-Fouling; Nova Science Pub Inc.: Hauppauge, NY, USA, 2010; pp. 105–128. [Google Scholar]
- Trinidad, A.; Ibanez, A.; Gomez, D.; Garcia-Berrocal, J.R.; Ramirez-Camacho, R. Application of environmental scanning electron microscopy for study of biofilms in medical devices. Microsc. Sci. Technol. Appl. Educ. 2010, 1, 204–210. [Google Scholar]
- Mukherji, R.; Patil, A.; Prabhune, A. Role of extracellular proteases in biofilm disruption of gram positive bacteria with special emphasis on Staphylococcus aureus biofilms. Enz. Eng. 2015, 4. [Google Scholar] [CrossRef]
- Harding, J.; Reynolds, M. Combating medical device fouling. Trends Biotechnol. 2014, 32, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.; Ramage, G. Fungal Biofilms in Human Disease. Biofilm-Based Healthcare-Associated Infections, 2nd ed.; Springer: Cham, Switzerland, 2015. [Google Scholar]
- Strindberg, L.Z. The dependence of the results of pulp therapy on certain factors. Acta Odontol. Scand. 1956, 14, 1–75. [Google Scholar]
- Torabinejad, M.; Walton, R.E.; Fouad, A.F. Endodontics: Principles and Practice, 5th ed.; Elsevier: Beijing, China, 2015. [Google Scholar]
- Williams, C.; Rajendran, R.; Ramage, G. Aspergillus Biofilms in Human Disease. Fungal Biofilms and Related Infections, 3rd ed.; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Toretto, S.; Pignataro, L. The Role of Biofilms in Upper Respiratory Tract Infections. Infections of the Ears, Nose, Throat, and Sinuses; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Schurmann, M.; Oppel, F.; Gottaschalk, M.; Buker, B.; Jantos, C.A.; Knabbe, C.; Hutten, A.; Kaltschmidt, B.; Kaltschmidt, C.; Sudhoff, H. The therapeutic effect of 1, 8-cineol on pathogenic bacteria species present in chronic rhinosinusitis. Front. Microbiol. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Brussow, H. Pseudomonas biofilms, cystic fibrosis, and phage: A silver lining? mBio 2012, 3, 1–2. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, C.T.; Panmanee, W.; Hassett, D.J. An Overview of Infections in Cystic Fibrosis Airways and the Role of Environmental Conditions on Pseudomonas aeruginosa Biofilm Formation and Viability. Cystic Fibrosis in the Light of New Research; Intech Open: Rijeka, Croatia, 2015. [Google Scholar]
- Romling, U.; Fiedler, B.; Bosshammer, J.; Grothues, D.; Greipel, J.; von der Hardt, H.; Tummler, B. Epidemiology of chronic pseudomonas aeruginosa infections in cystic fibrosis. J. Infect. Dis. 1994, 170, 1616–1621. [Google Scholar] [CrossRef]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Del Pozo, J.; Patel, R. The challenge of treating biofilm-associated bacterial infections. Clin. Pharmacol. Ther. 2007, 82, 204–209. [Google Scholar] [CrossRef]
- Santajit, S.; Indrawattana, N. Mechanisms of antimicrobial resistance in ESKAPE pathogens. BioMed Res. Int. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Simões, M.; Simões, L.; Vieira, M. A review of current and emergent biofilm control strategies. LWT Food Sci. Technol. 2010, 43, 573–583. [Google Scholar] [CrossRef]
- Patel, R. Biofilms and antimicrobial resistance. Clin. Orthop. Relat. Res. NA 2005, 437, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Singh, S.; Chowdhury, I.; Singh, R. Understanding the mechanism of bacterial biofilms resistance to antimicrobial agents. Open Microbiol. J. 2017, 11, 53–62. [Google Scholar] [CrossRef]
- Olsen, I. Biofilm-specific antibiotic tolerance and resistance. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Donlan, R.M. Role of biofilms in antimicrobial resistance. ASAIO J. 2000, 46, S47–S52. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K. Persister cells. Annu. Rev. Microbiol. 2010, 64, 357–372. [Google Scholar] [CrossRef] [PubMed]
- Poole, K. Stress responses as determinants of antimicrobial resistance in Gram-negative bacteria. Trends Microbiol. 2012, 20, 227–234. [Google Scholar] [CrossRef]
- Rabin, N.; Zheng, Y.; Opoku-Temeng, C.; Du, Y.; Bonsu, E.; Sintim, H. Agents that inhibit bacterial biofilm formation. Future Med. Chem. 2015, 7, 647–671. [Google Scholar] [CrossRef]
- Borges, A.; Saavedra, M.; Simoes, M. Insights on antimicrobial resistance, biofilms and the use of phytochemicals as new antimicrobial agents. Curr. Med. Chem. 2015, 22, 2590–2614. [Google Scholar] [CrossRef]
- Savoia, D. Plant-derived antimicrobial compounds: Alternatives to antibiotics. Future Microbiol. 2012, 7, 979–990. [Google Scholar] [CrossRef]
- Samanta, A.; Das, G.; Das, S.K. Roles of flavonoids in plants. Int. J. Pharm. Sci. Technol. 2011, 6, 12–35. [Google Scholar]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Alvesalo, J.; Vuorela, H.; Tammela, P.; Leinonen, M.; Saikku, P.; Vuorela, P. Inhibitory effect of dietary phenolic compounds on Chlamydia pneumoniae in cell cultures. Biochem. Pharmacol. 2006, 71, 735–741. [Google Scholar] [CrossRef]
- Cushnie, T.P.; Lamb, A.J. Recent advances in understanding the antibacterial properties of flavonoids. Int. J. Antimicrob. Agents 2011, 38, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Vikram, A.; Jayaprakasha, G.K.; Jesudhasan, P.R.; Pillai, S.D.; Patil, B.S. Suppression of bacterial cell–cell signaling, biofilm formation and type III secretion system by citrus flavonoids. J. Appl. Microbiol. 2010, 109, 515–527. [Google Scholar] [CrossRef]
- Valsaraj, R.; Pushpangadan, P.; Smitt, U.W.; Adsersen, A.; Christensen, S.B.; Sittie, A.; Nyman, U.; Nielsen, C.; Olsen, C.E. New anti-HIV-1, antimalarial, and antifungal compounds from Terminalia bellerica. J. Nat. Prod. 1997, 60, 739–742. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Henika, P.R.; Levin, C.E.; Mandrell, R.E.; Kozukue, N. Antimicrobial activities of tea catechins and theaflavins and tea extracts against Bacillus cereus. J. Food Prot. 2006, 69, 354–361. [Google Scholar] [CrossRef]
- Lee, J.H.; Regmi, S.C.; Kim, J.A.; Cho, M.H.; Yun, H.; Lee, C.S.; Lee, J. Apple flavonoid phloretin inhibits Escherichia coli O157:H7 biofilm formation and ameliorates colon inflammation in rats. Infect. Immun. 2011, 79, 4819–4827. [Google Scholar] [CrossRef]
- Ukoha, P.O.; Cemaluk, E.A.; Nnamdi, O.L.; Madus, E.P. Tannins and other phytochemical of the Samanaea saman pods and their antimicrobial activities. Afr. J. Pure Appl. Chem. 2011, 5, 237–244. [Google Scholar]
- Lee, J.H.; Park, J.H.; Cho, H.S.; Joo, S.W.; Cho, M.H.; Lee, J. Anti-biofilm activities of quercetin and tannic acid against Staphylococcus aureus. Biofouling 2013, 29, 491–499. [Google Scholar] [CrossRef]
- Kiran, M.D.; Adikesavan, N.V.; Cirioni, O.; Giacometti, A.; Silvestri, C.; Scalise, G.; Ghiselli, R.; Saba, V.; Orlando, F.; Shoham, M. Discovery of a quorum-sensing inhibitor of drug-resistant staphylococcal infections by structure-based virtual screening. Mol. Pharmacol. 2008, 73, 1578–1586. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Sánchez-Maldonado, A.; Schieber, A.; Gänzle, M. Structure-function relationships of the antibacterial activity of phenolic acids and their metabolism by lactic acid bacteria. J. Appl. Microbiol. 2011, 111, 1176–1184. [Google Scholar] [CrossRef]
- Bor, T.; Aljaloud, S.; Gyawali, R.; Ibrahim, S. Antimicrobials from herbs, spices, and plants. In Fruits, Vegetables, and Herbs; Academic Press: Cambridge, MA, USA, 2016; pp. 551–578. [Google Scholar]
- Kalkhambkar, R.G.; Kulkarni, G.M.; Kamanavalli, C.M.; Premkumar, N.; Asdaq, S.M.B.; Sun, C.M. Synthesis and biological activities of some new fluorinated coumarins and 1-aza coumarins. Eur. J. Med. Chem. 2008, 43, 2178–2188. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, Y.G.; Cho, H.S.; Ryu, S.Y.; Cho, M.H.; Lee, J. Coumarins reduce biofilm formation and the virulence of Escherichia coli O157:H7. Phytomedicine 2014, 21, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Girennavar, B.; Cepeda, M.L.; Soni, K.A.; Vikram, A.; Jesudhasan, P.; Jayaprakasha, G.K.; Pillai, S.D.; Patil, B.S. Grapefruit juice and its furocoumarins inhibits autoinducer signaling and biofilm formation in bacteria. Int. J. Food Microbiol. 2008, 125, 204–208. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, S.J.; Lee, J.H.; Sun, W.S.; Kim, J.H. Antimicrobial activity of 9¬O¬acyl and 9¬O-alkylberberrubine derivatives. Planta Med. 2002, 68, 277–281. [Google Scholar] [CrossRef]
- Yi, Z.B.; Yu, Y.; Liang, Y.Z.; Zeng, B. Evaluation of the antimicrobial mode of berberine by LC/ESI-MS combined with principal component analysis. J. Pharm. Biomed. Anal. 2007, 44, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Kumar, P. Evaluation of antimicrobial activity of alkaloids of Terminalia chebula Retz. against some multidrugresistant microorganisms. Int. J. Green Pharm. IJGP 2012, 6. [Google Scholar] [CrossRef]
- Szczepanski, S.; Lipski, A. Essential oils show specific inhibiting effects on bacterial biofilm formation. Food Control 2014, 36, 224–229. [Google Scholar] [CrossRef]
- Bourne, Y.; Ayouba, A.; Rougé, P.; Cambillau, C. Interaction of a legume lectin with two components of the bacterial cell wall. A crystallographic study. J. Biol. Chem. 1994, 269, 9429–9435. [Google Scholar] [CrossRef]
- Paiva, P.M.G.; Gomes, F.S.; Napoleão, T.H.; Sá, R.A.; Correia, M.T.S.; Coelho, L.C.B.B. Current Research, Technology and Education Topics in Applied Microbiology and Microbial Biotechnology; Formatex Research Center: Badajoz, Spain, 2010; pp. 396–406. [Google Scholar]
- Karnchanatat, A. Antimicrobial Agents; IntechOpen: Rijeka, Croatia, 2012; pp. 145–178. [Google Scholar]
- Teixeira, E.H.; Napimoga, M.H.; Carneiro, V.A.; De Oliveira, T.M.; Nascimento, K.S.; Nagano, C.S.; Souza, J.B.; Havt, A.; Pinto, V.P.T.; Gonçalves, R.B.; et al. In vitro inhibition of oral streptococci binding to the acquired pellicle by algal lectins. J. Appl. Microbiol. 2007, 103, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Sá, R.A.; Gomes, F.S.; Napoleão, T.H.; Santos, N.D.L.; Melo, C.M.L.; Gusmão, N.B.; Coelho, L.C.B.B.; Paiva, P.M.G.; Bieber, L.W. Antibacterial and antifungal activities of Myracrodruon urundeuva heartwood. Wood Sci. Technol. 2009, 43, 85–95. [Google Scholar] [CrossRef]
- Mahlapuu, M.; Håkansson, J.; Ringstad, L.; Björn, C. Antimicrobial peptides: An emerging category of therapeutic agents. Front. Cell. Infect. Microbiol. 2016, 6, 194. [Google Scholar] [CrossRef] [PubMed]
- Pushpanathan, M.; Gunasekaran, P.; Rajendhran, J. Antimicrobial peptides: Versatile biological properties. Int. J. Pept. 2013, 2013, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Broekaert, W.F.; Cammue, B.P.A.; De Bolle, M.F.C.; Thevissen, K.; De Samblanx, G.W.; Osborn, R.W.; Nielson, K. Antimicrobial peptides from plants. Crit. Rev. Plant Sci. 1997, 16, 297–323. [Google Scholar] [CrossRef]
- Stotz, H.U.; Waller, F.; Wang, K. Antimicrobial Peptides and Innate Immunity; Springer: Berlin/Heidelberg, Germany, 2013; pp. 29–51. [Google Scholar]
- Batoni, G.; Maisetta, G.; Esin, S. Antimicrobial peptides and their interaction with biofilms of medically relevant bacteria. Biochim. Biophys. Acta 2016, 1858, 1044–1060. [Google Scholar] [CrossRef] [PubMed]
- Fjell, C.D.; Hiss, J.A.; Hancock, R.E.W.; Schneider, G. Designing antimicrobial peptides: Form follows function. Nat. Rev. Drug Discov. 2012, 11, 37–51. [Google Scholar] [CrossRef]
- Guilhelmelli, F.; Vilela, N.; Albuquerque, P.; Derengowski, L.; Silva-Pereira, I.; Kyaw, C. Antibiotic development challenges: The various mechanisms of action of antimicrobial peptides and of bacterial resistance. Front. Microbiol. 2013, 4, 353. [Google Scholar] [CrossRef]
- Shai, Y. Mode of action of membrane active antimicrobial peptides. Biopolymers 2002, 66, 236–248. [Google Scholar] [CrossRef]
- Takahashi, D.; Shukla, S.K.; Prakash, O.; Zhang, G. Structural determinants of host defense peptides for antimicrobial activity and target cell selectivity. Biochimie 2010, 92, 1236–1241. [Google Scholar] [CrossRef] [PubMed]
- Segev-Zarko, L.; Saar-Dover, R.; Brumfeld, V.; Mangoni, M.; Shai, Y. Mechanisms of biofilm inhibition and degradation by antimicrobial peptides. Biochem. J. 2015, 468, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Haney, E.; Mansour, S.; Hancock, R. Antimicrobial Peptides: An Introduction; Methods in Molecular Biology; Springer: Berlin/Heidelberg, Germany, 2016; pp. 3–22. [Google Scholar]
- Mandal, S.M.; Migliolo, L.; Franco, O.L.; Ghosh, A.K. Identification of an antifungal peptide from Trapa natans fruits with inhibitory effects on Candida tropicalis biofilm formation. Peptides 2011, 32, 1741–1747. [Google Scholar] [CrossRef]
- Tam, J.P.; Lu, Y.A.; Yang, J.L.; Chiu, K.W. An unusual structural motif of antimicrobial peptides containing end-to-end macrocycle and cystine-knot disulfides. Proc. Natl. Acad. Sci. USA 1999, 96, 8913–8918. [Google Scholar] [CrossRef] [PubMed]
- Daly, N.L.; Koltay, A.; Gustafson, K.R.; Boyd, M.R.; Casas-Finet, J.R.; Craik, D.J. Solution structure by NMR of circulin A: A macrocyclic knotted peptide having anti-HIV activity. J. Mol. Biol. 1999, 285, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Witherup, K.M.; Bogusky, M.J.; Anderson, P.S.; Ramjit, H.; Ransom, R.W.; Wood, T.; Sardana, M. Cyclopsychotride A, A biologically active, 31-residue cyclic peptide isolated from Psychotria longipes. J. Nat. Prod. 1994, 57, 1619–1625. [Google Scholar] [CrossRef]
- Barbosa, M.; Pêgo, A.; Amaral, I. Chitosan. In Comprehensive Biomaterials; Elsevier: Amsterdam, The Netherlands, 2011; pp. 221–237. [Google Scholar]
- Zhang, A.; Mu, H.; Zhang, W.; Cui, G.; Zhu, J.; Duan, J. Chitosan coupling makes microbial biofilms susceptible to antibiotics. Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Cobrado, L.; Azevedo, M.; Silva-Dias, A.; Ramos, J.; Pina-Vaz, C.; Rodrigues, A. Cerium, chitosan and hamamelitannin as novel biofilm inhibitors? J. Antimicrob. Chemother. 2012, 67, 1159–1162. [Google Scholar] [CrossRef]
- Goy, R.; Britto, D.; Assis, O. A review of the antimicrobial activity of chitosan. Polímeros 2009, 19, 241–247. [Google Scholar] [CrossRef]
- Orgaz, B.; Lobete, M.; Puga, C.; San Jose, C. Effectiveness of chitosan against mature biofilms formed by food related bacteria. Int. J. Mol. Sci. 2011, 12, 817–828. [Google Scholar] [CrossRef]
- Khan, F.; Pham, D.; Oloketuyi, S.; Manivasagan, P.; Oh, J.; Kim, Y. Chitosan and their derivatives: Antibiofilm drugs against pathogenic bacteria. Colloids Surf. B Biointerfaces 2020, 185, 110627. [Google Scholar] [CrossRef]
- Romanò, C.; De Vecchi, E.; Bortolin, M.; Morelli, I.; Drago, L. Hyaluronic acid and its composites as a local antimicrobial/antiadhesive barrier. J. Bone Jt. Infect. 2017, 2, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Drago, L.; Cappelletti, L.; De Vecchi, E.; Pignataro, L.; Torretta, S.; Mattina, R. Antiadhesive and antibiofilm activity of hyaluronic acid against bacteria responsible for respiratory tract infections. APMIS 2014, 122, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Carlson, G.A.; Dragoo, J.L.; Samimi, B.; Bruckner, D.A.; Bernard, G.W.; Hedrick, M.; Benhaim, P. Bacteriostatic properties of biomatrices against common orthopaedic pathogens. Biochem. Biophys. Res. Commun. 2004, 321, 472–478. [Google Scholar] [CrossRef]
- Pirnazar, P.; Wolinsky, L.; Nachnani, S.; Haake, S.; Pilloni, A.; Bernard, G.W. Bacteriostatic effects of hyaluronic acid. J. Periodontol. 1999, 70, 370–374. [Google Scholar] [CrossRef] [PubMed]



| Type of Bacteria | Optimum Temperature | References |
|---|---|---|
| Salmonella spp. | 30 °C | [74] |
| Listeria monocytogenes | 37 °C | [7] |
| Aeromonas hydrophila | 20–25 °C | [72] |
| Clostridium perfringens | 37 °C | [73] |
| Type of Food Industry | Prominent Bacteria | Effects | References |
|---|---|---|---|
| Dairy Industry | L. monocytogenes S. typhimurium and S. enteritidis E. coli (STEC) B. cereus | Gastroenteritis or listeriosis Gastroenteritis Enterohemorrhagic gastroenteritis or hemolytic uremic syndrome (HUS) Gastroenteritis or occasionally acute liver failure | [120,122,126,129,131] |
| Poultry Industry | S. enterica C. jejuni and C. coli | Gastroenteritis or septicemia Enterocolitis or gastroenteritis | [120,123,128] |
| Meat Industry | E. coli O157:H7 L. Monocytogenes Salmonella spp. | Hemorrhagic colitis or thrombotic thrombocytopenic purpura (TTP) Gastroenteritis or listeriosis Salmonellosis | [118,124,127,130,132] |
| Fish and Seafood Industry | Vibrio cholerae Aeromonas spp. Pseudomonas spp. | Cholera or gastroenteritis Epizootic ulcerative syndrome (EUS) | [119,121,122,125] |
| Type of Compound | Class/Subclass | Source | Target Microorganism | Antibiofilm Strategy | References |
|---|---|---|---|---|---|
| Flavonoids | Kaempferol and Naringenin | Citrus plants | E. coli O157:H7, | QS inhibition by interfering with AHL and their receptors | [164,165] |
| V. harveyi BB120 | |||||
| 7-hydroxy-3,4-(methylenedioxy) flavan | Terminalia bellirica fruits | C. albicans | Metabolism inhibition | [166] | |
| Catechin | Green tea | P. gingivalis | Forms complexes with bacterial cell walls of microorganisms | [167] | |
| Phloretin | Apple | E. coli O157:H7 | - | [168] | |
| Tannins | Tannic acid | Tea | S. aureus | Suppression of QS genes involved | [170] |
| Hamamelitannin | Hamamelis virginiana leaves | - | QS regulator RNAIII inhibition | [171] | |
| Phenolic Acids | Hydroxycinnamic and hydroxybenzoic acids | - | L. fermentum, L. plantaruma, L. brevi | Dependent on number of hydroxyl groups | [173] |
| Gallic and ferulic acids | - | P. aeruginosa, E. coli, L. monocytogenes, methicillin-resistant Staphylococcus aureus (MRSA) | - | [173] | |
| Coumarins | Umbelliferone | - | E. coli O157:H7 | - | [176] |
| Furocoumarins | Grape juice | Vibrio harveyi, E. coli O157:H7 | Inhibition of QS molecules, AI-1 and AI-2 | [177] | |
| Alkaloids | Indole alkaloids | Terminalia chebula | P. aeruginosa, E. coli, S. aureus, A. tumeficaens, B. subtilis, | - | [180] |
| Berberine | Berberis roots and stems | - | Target the RNA polymerases and nucleic acids of microorganisms | [178,179] | |
| Terpenoids and Essential Oils | Thymoquinone | Nigella sativa | S. aureus, L. monocytogenes | - | [181] |
| Gossypols | Cotton seeds | P. vulgaris, E. coli, P. aeruginosa, S. epidermidis, B. subtilis, B. cereus | - | [159] | |
| Lectins | - | Solieria filiformis | P. aeruginosa | - | [185] |
| - | B. triquetrum | Streptococcus spp. | Attachment to the pellicle | [185] | |
| Peptides | RsAFP2 | Rhapanus sativus seeds | C. albicans | - | [198] |
| TnAFP1 | Trapanatan fruits | C. tropicalis | |||
| Peptides | Circulins A-B | Chassalia parviflora | Klebsiella spp., E. coli, S. aureus, P. vulgaris, M. luteus | - | [199,200,201] |
| Cyclopsychotride A | Psychotria longipes | ||||
| Chitosan | Chitin | Shell of shrimp, lobster, crab and prawns | S. enterica, S. aureus, S. epidermidis, L. monocytogenes, B. cereus. | Inhibition of the protein synthesis, binding to the bacterial cell wall and suppression of bacterial growth by external barrier formation | [203,204,206] |
| Hyaluronic Acid | - | - | S. epidermidis | Inhibition of microbial adherence | [208] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samrot, A.V.; Abubakar Mohamed, A.; Faradjeva, E.; Si Jie, L.; Hooi Sze, C.; Arif, A.; Chuan Sean, T.; Norbert Michael, E.; Yeok Mun, C.; Xiao Qi, N.; et al. Mechanisms and Impact of Biofilms and Targeting of Biofilms Using Bioactive Compounds—A Review. Medicina 2021, 57, 839. https://doi.org/10.3390/medicina57080839
Samrot AV, Abubakar Mohamed A, Faradjeva E, Si Jie L, Hooi Sze C, Arif A, Chuan Sean T, Norbert Michael E, Yeok Mun C, Xiao Qi N, et al. Mechanisms and Impact of Biofilms and Targeting of Biofilms Using Bioactive Compounds—A Review. Medicina. 2021; 57(8):839. https://doi.org/10.3390/medicina57080839
Chicago/Turabian StyleSamrot, Antony V., Amira Abubakar Mohamed, Etel Faradjeva, Lee Si Jie, Chin Hooi Sze, Akasha Arif, Tan Chuan Sean, Emmanuel Norbert Michael, Chua Yeok Mun, Ng Xiao Qi, and et al. 2021. "Mechanisms and Impact of Biofilms and Targeting of Biofilms Using Bioactive Compounds—A Review" Medicina 57, no. 8: 839. https://doi.org/10.3390/medicina57080839
APA StyleSamrot, A. V., Abubakar Mohamed, A., Faradjeva, E., Si Jie, L., Hooi Sze, C., Arif, A., Chuan Sean, T., Norbert Michael, E., Yeok Mun, C., Xiao Qi, N., Ling Mok, P., & Kumar, S. S. (2021). Mechanisms and Impact of Biofilms and Targeting of Biofilms Using Bioactive Compounds—A Review. Medicina, 57(8), 839. https://doi.org/10.3390/medicina57080839









