Eugenol Reduced ΜPO, CD45 and HMGB1 Expression and Attenuated the Expression of Leukocyte Infiltration Markers in the Intestinal Tissue in Biliopancreatic Duct Ligation-Induced Pancreatitis in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animal Care and Handling
2.2. Experimental Design
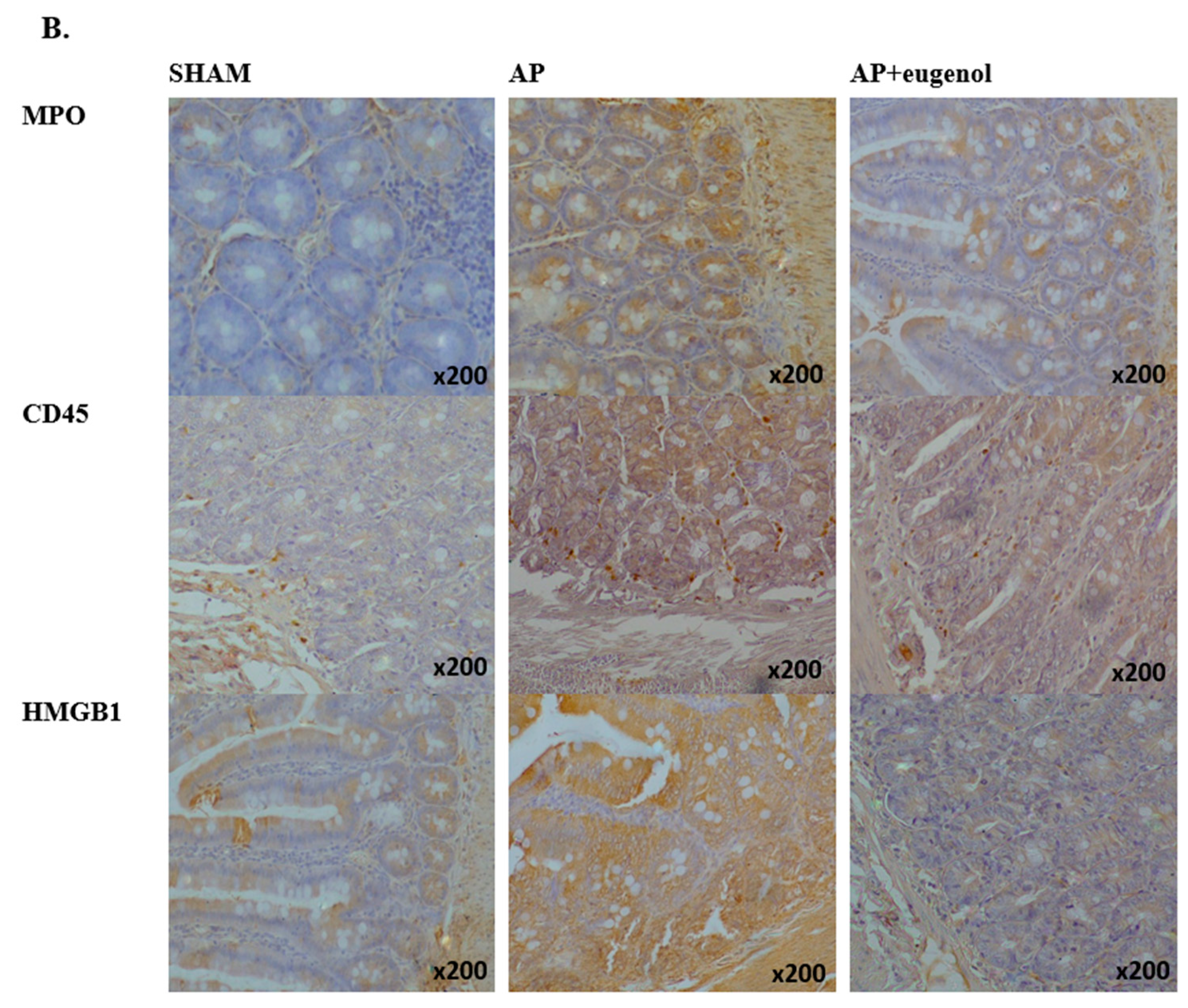
2.3. Histopathological Examination and Immunohistochemical Staining
2.4. Determination of Serum Protein Levels
2.5. Statistical Analysis
3. Results
3.1. Histopathological Findings
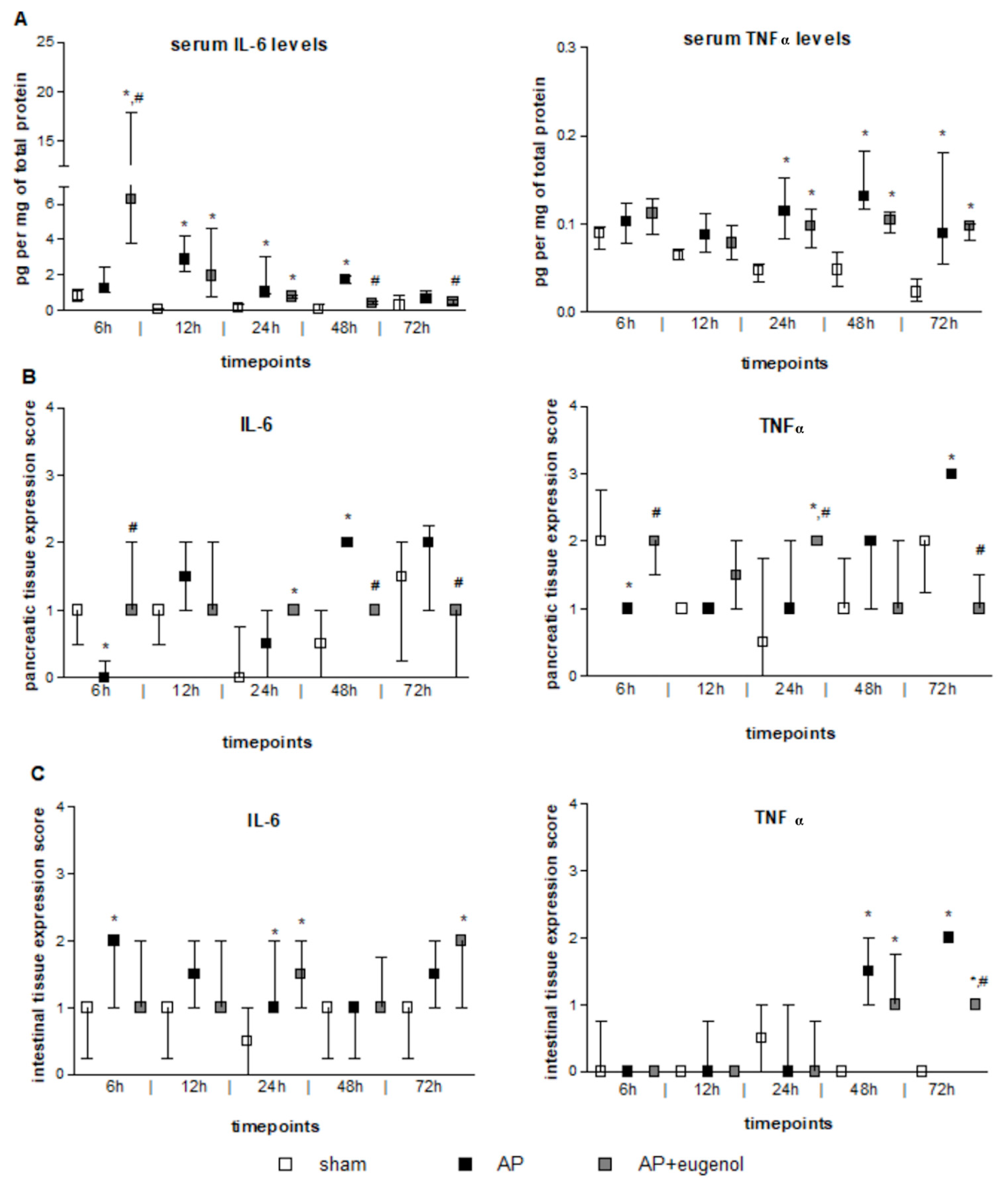
3.2. Eugenol Treatment Mainly Affects Serum IL-6 and Resistin Levels
3.3. Eugenol Affects IL-6 and TNFα Pancreatic Expression
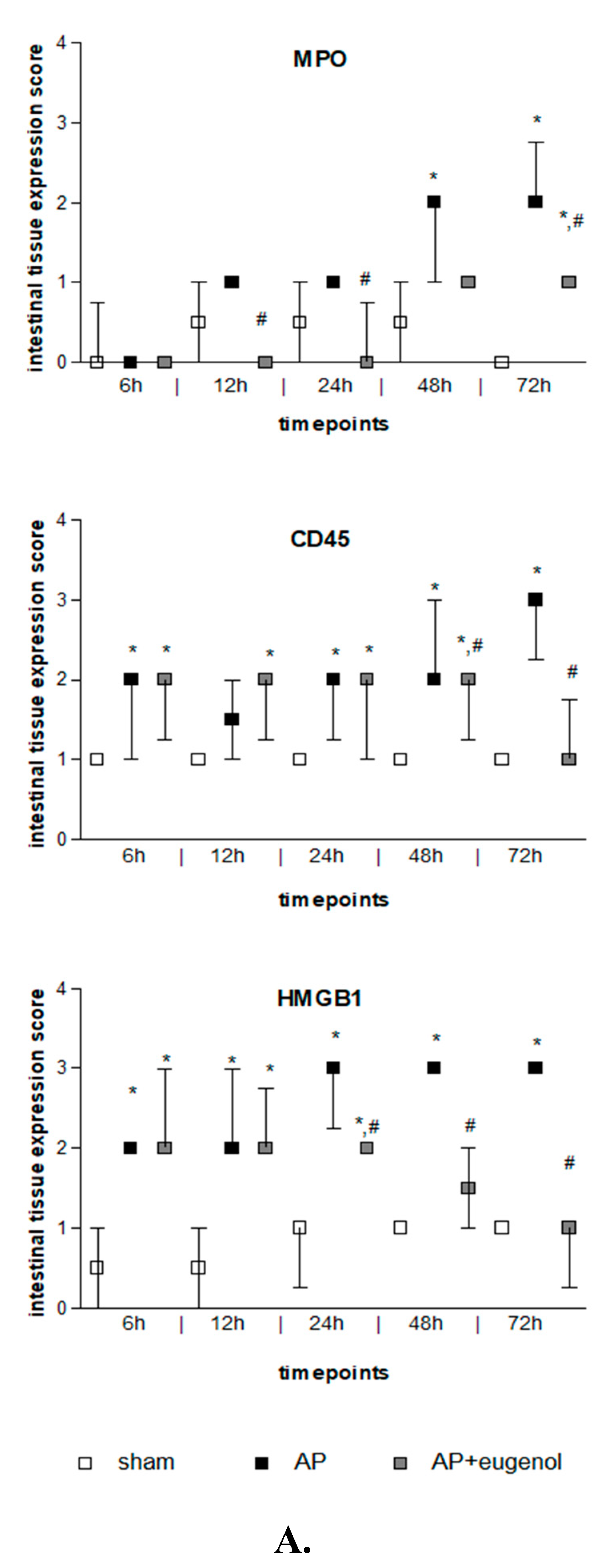
3.4. Eugenol Affects TNFα, MPO, CD45, and HMGB1 Expression in the Intestinal Tissue
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Banks, P.A.; Bollen, T.L.; Dervenis, C.; Gooszen, H.G.; Johnson, C.D.; Sarr, M.G.; Tsiotos, G.G.; Vege, S.S.; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis--2012: Revision of the Atlanta classification and definitions by international consensus. Gut 2013, 62, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Phillip, V.; Steiner, J.M.; Algül, H. Early phase of acute pancreatitis: Assessment and management. World J. Gastrointest. Pathophysiol. 2014, 5, 158–168. [Google Scholar] [CrossRef]
- Cicalese, L.; Sahai, A.; Sileri, P.; Rastellini, C.; Subbotin, V.; Ford, H.; Lee, K. Acute pancreatitis and bacterial translocation. Dig. Dis. Sci. 2001, 46, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kudo, M.; Strober, W. Immunopathogenesis of pancreatitis. Mucosal Immunol. 2017, 10, 283–298. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, M.; Wong, F.L.; Cao, Y.; Lau, H.Y.; Huang, J.; Puneet, P.; Chevali, L. Pathophysiology of acute pancreatitis. Pancreatol. Off. J. Int. Assoc. Pancreatol. (IAP) 2005, 5, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Leung, P.S.; Chan, Y.C. Role of oxidative stress in pancreatic inflammation. Antioxid. Redox Signal. 2009, 11, 135–165. [Google Scholar] [CrossRef] [PubMed]
- Bonior, J.; Warzecha, Z.; Ceranowicz, P.; Gajdosz, R.; Pierzchalski, P.; Kot, M.; Leja-Szpak, A.; Nawrot-Porąbka, K.; Link-Lenczowski, P.; Pędziwiatr, M.; et al. Capsaicin-Sensitive Sensory Nerves Are Necessary for the Protective Effect of Ghrelin in Cerulein-Induced Acute Pancreatitis in Rats. Int. J. Mol. Sci. 2017, 18, 1402. [Google Scholar] [CrossRef]
- Deng, W.; Abliz, A.; Xu, S.; Sun, R.; Guo, W.; Shi, Q.; Yu, J.; Wang, W. Severity of pancreatitis-associated intestinal mucosal barrier injury is reduced following treatment with the NADPH oxidase inhibitor apocynin. Mol. Med. Rep. 2016, 14, 3525–3534. [Google Scholar] [CrossRef]
- Heath, D.I.; Cruickshank, A.; Gudgeon, M.; Jehanli, A.; Shenkin, A.; Imrie, C.W. Role of interleukin-6 in mediating the acute phase protein response and potential as an early means of severity assessment in acute pancreatitis. Gut 1993, 34, 41–45. [Google Scholar] [CrossRef]
- Norman, J.; Franz, M.; Riker, A. Rapid elevation of systemic cytokines during acute pancreatitis and their origination within the pancreas. Surg. Forum. 1994, 45, 148–150. [Google Scholar]
- Mayer, J.; Rau, B.; Gansauge, F.; Beger, H.G. Inflammatory mediators in human acute pancreatitis: Clinical and pathophysiological implications. Gut 2000, 47, 546–552. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Wang, S.S.; Lu, R.H.; Lu, C.C.; Chang, F.Y.; Lee, S.D. Early changes of serum proinflammatory and anti-inflammatory cytokines after endoscopic retrograde cholangiopancreatography. Pancreas 2003, 26, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Lesina, M.; Wörmann, S.M.; Neuhöfer, P.; Song, L.; Algül, H. Interleukin-6 in inflammatory and malignant diseases of the pancreas. Sem. Immunol. 2014, 26, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Bokarewa, M.; Nagaev, I.; Dahlberg, L.; Smith, U.; Tarkowski, A. Resistin, an adipokine with potent proinflammatory properties. J. Immunol. 2005, 174, 5789–5795. [Google Scholar] [CrossRef] [PubMed]
- Filková, M.; Haluzík, M.; Gay, S.; Senolt, L. The role of resistin as a regulator of inflammation: Implications for various human pathologies. Clin. Immunol. 2009, 133, 157–170. [Google Scholar] [CrossRef]
- Chen, C.; Jiang, J.; Lü, J.M.; Chai, H.; Wang, X.; Lin, P.H.; Yao, Q. Resistin decreases expression of endothelial nitric oxide synthase through oxidative stress in human coronary artery endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H193–H201. [Google Scholar] [CrossRef]
- Raghuraman, G.; Zuniga, M.C.; Yuan, H.; Zhou, W. PKCε mediates resistin-induced NADPH oxidase activation and inflammation leading to smooth muscle cell dysfunction and intimal hyperplasia. Atherosclerosis 2016, 253, 29–37. [Google Scholar] [CrossRef]
- Acquarone, E.; Monacelli, F.; Borghi, R.; Nencioni, A.; Odetti, P. Resistin: A reappraisal. Mech. Ageing Dev. 2019, 178, 46–63. [Google Scholar] [CrossRef]
- Minn, A.H.; Patterson, N.B.; Pack, S.; Hoffmann, S.C.; Gavrilova, O.; Vinson, C.; Harlan, D.M.; Shalev, A. Resistin is expressed in pancreatic islets. Biochem. Biophys. Res. Commun. 2003, 310, 641–645. [Google Scholar] [CrossRef]
- Nogueiras, R.; Gallego, R.; Gualillo, O.; Caminos, J.E.; García-Caballero, T.; Casanueva, F.F.; Diéguez, C. Resistin is expressed in different rat tissues and is regulated in a tissue- and gender-specific manner. FEBS Lett. 2003, 548, 21–27. [Google Scholar] [CrossRef]
- Daniel, P.; Leśniowski, B.; Mokrowiecka, A.; Jasińska, A.; Pietruczuk, M.; Małecka-Panas, E. Circulating levels of visfatin, resistin and pro-inflammatory cytokine interleukin-8 in acute pancreatitis. Pancreatology 2010, 10, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.N.; Wang, X.Y.; Tan, Y.; Lin, M.; Zhang, W.; Xu, K.Q. Significance of resistin expression in acute pancreatitis. Exp. Ther. Med. 2015, 9, 1438–1442. [Google Scholar] [CrossRef] [PubMed]
- Kibar, Y.I.; Albayrak, F.; Arabul, M.; Dursun, H.; Albayrak, Y.; Ozturk, Y. Resistin: New serum marker for predicting severity of acute pancreatitis. J. Int. Med. Res. 2016, 44, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Dawra, S.; Rana, S.; Gupta, P.; Samanta, J.; Sinha, S.K.; Gupta, V.; Yadav, T.D.; Kochhar, R. Can serum resistin predict severity of acute pancreatitis? Biomarkers 2021, 26, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Altin, J.G.; Sloan, E.K. The role of CD45 and CD45-associated molecules in T cell activation. Immunol. Cell Biol. 1997, 75, 430–445. [Google Scholar] [CrossRef] [PubMed]
- Penninger, J.M.; Irie-Sasaki, J.; Sasaki, T.; Oliveira-dos-Santos, A.J. CD45: New jobs for an old acquaintance. Nat. Immunol. 2001, 2, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.L. The leukocyte common antigen family. Annu. Rev. Immunol. 1989, 7, 339–369. [Google Scholar] [CrossRef] [PubMed]
- De Dios, I.; Ramudo, L.; Alonso, J.R.; Recio, J.S.; Garcia-Montero, A.C.; Manso, M.A. CD45 expression on rat acinar cells: Involvement in pro-inflammatory cytokine production. FEBS Lett. 2005, 579, 6355–6360. [Google Scholar] [CrossRef]
- De Dios, I.; Ramudo, L.; García-Montero, A.C.; Manso, M.A. Redox-sensitive modulation of CD45 expression in pancreatic acinar cells during acute pancreatitis. J. Pathol. 2006, 210, 234–239. [Google Scholar] [CrossRef]
- Rayner, B.S.; Love, D.T.; Hawkins, C.L. Comparative reactivity of myeloperoxidase-derived oxidants with mammalian cells. Free Radic. Biol. Med. 2014, 71, 240–255. [Google Scholar] [CrossRef]
- Aratani, Y. Myeloperoxidase: Its role for host defense, inflammation, and neutrophil function. Arch. Biochem. Biophys. 2018, 640, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Chooklin, S.; Pereyaslov, A.; Bihalskyy, I. Pathogenic role of myeloperoxidase in acute pancreatitis. Hepatobiliary Pancreat. Dis. Int. 2009, 8, 627–631. [Google Scholar] [PubMed]
- Chen, H.; Su, H.; Yuan, L.; Miao, Y.F.; Zhang, Y.M.; Li, J.; Tang, W.F. Administration of electroacupuncture and Da-Cheng-Qi decoction has a synergetic effect in relieving injury of pancreas, lung and large intestine and inflammatory reactions in rats with acute pancreatitis. Zhen Ci Yan Jiu 2018, 43, 353–359. (In Chinese) [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Fang, X.; Wang, F.; Li, H.; Niu, W.; Liang, W.; Wu, C.; Li, J.; Tu, X.; Pan, L.L.; et al. Butyrate ameliorates caerulein-induced acute pancreatitis and associated intestinal injury by tissue-specific mechanisms. Br. J. Pharmcol. 2019, 176, 4446–4461. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, Y.; Dong, M.; Cui, J.; Rong, D.; Dong, Q. Oxymatrine ameliorates L-arginine-induced acute pancreatitis in rats. Inflammation 2012, 35, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Tracey, K.J. Targeting HMGB1 in inflammation. Biochim. Biophys. Acta 2010, 1799, 149–156. [Google Scholar] [CrossRef]
- Kang, R.; Chen, R.; Zhang, Q.; Hou, W.; Wu, S.; Cao, L.; Huang, J.; Yu, Y.; Fan, X.G.; Yan, Z.; et al. HMGB1 in health and disease. Mol. Asp. Med. 2014, 40, 1–116. [Google Scholar] [CrossRef]
- Yang, H.; Wang, H.; Andersson, U. Targeting Inflammation Driven by HMGB1. Front. Immunol. 2020, 11, 484. [Google Scholar] [CrossRef]
- Kocsis, A.K.; Szabolcs, A.; Hofner, P.; Takács, T.; Farkas, G.; Boda, K.; Mándi, Y. Plasma concentrations of high-mobility group box protein 1, soluble receptor for advanced glycation end-products and circulating DNA in patients with acute pancreatitis. Pancreatology 2009, 9, 383–391. [Google Scholar] [CrossRef]
- Zhang, Z.W.; Zhang, Q.Y.; Zhou, M.T.; Liu, N.X.; Chen, T.K.; Zhu, Y.F.; Wu, L. Antioxidant inhibits HMGB1 expression and reduces pancreas injury in rats with severe acute pancreatitis. Dig. Dis. Sci. 2010, 55, 2529–2536. [Google Scholar] [CrossRef]
- Yasuda, T.; Ueda, T.; Takeyama, Y.; Shinzeki, M.; Sawa, H.; Nakajima, T.; Ajiki, T.; Fujino, Y.; Suzuki, Y.; Kuroda, Y. Significant increase of serum high-mobility group box chromosomal protein 1 levels in patients with severe acute pancreatitis. Pancreas 2006, 33, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Qiu, C.; Liu, K.; Li, X.; Chen, W.; Zhang, S.; Huang, Y. Restoration of intestinal mucosa in euphorbia kansui-treated severe acute pancreatitis rats based on HMGB1/MFG-E8 expression. Curr. Pharm. Biotechnol. 2021, 22, 1672–1682. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhang, E.; Xiao, M.; Chen, C.; Xu, W. Study of anti-inflammatory activities of α-D-glucosylated eugenol. Arch. Pharm. Res. 2013, 36, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Barboza, J.N.; da Silva Maia Bezerra Filho, C.; Silva, R.O.; Medeiros, J.; de Sousa, D.P. An overview on the anti-inflammatory potential and antioxidant profile of eugenol. Oxid. Med. Cell Longev. 2018, 2018, 3957262. [Google Scholar] [CrossRef] [PubMed]
- Morsy, M.A.; Fouad, A.A. Mechanisms of gastroprotective effect of eugenol in indomethacin-induced ulcer in rats. Phytother. Res. 2008, 22, 1361–1366. [Google Scholar] [CrossRef] [PubMed]
- Daniel, A.N.; Sartoretto, S.M.; Schimidt, G.; Caparroz-Assef, S.M.; Bersani-Amado, C.A.; Cuman, R.K. Anti-inflammatory and antinociceptive activities of eugenol essential oil in experimental animal models. Rev. Bras. Farm. 2009, 19, 212–217. [Google Scholar] [CrossRef]
- Nagababu, E.; Rifkind, J.M.; Boindala, S.; Nakka, L. Assessment of antioxidant activity of eugenol in vitro and in vivo. Methods Mol. Biol. 2010, 610, 165–180. [Google Scholar] [CrossRef]
- Pramod, K.; Ansari, S.H.; Ali, J. Eugenol: A natural compound with versatile pharmacological actions. Nat. Prod. Commun. 2010, 5, 1999–2006. [Google Scholar] [CrossRef]
- Gülçin, İ. Antioxidant activity of eugenol: A structure-activity relationship study. J. Med. Food 2011, 14, 975–985. [Google Scholar] [CrossRef]
- Devi, K.P.; Sakthivel, R.; Nisha, S.A.; Suganthy, N.; Pandian, S.K. Eugenol alters the integrity of cell membrane and acts against the nosocomial pathogen Proteus mirabilis. Arch. Pharm. Res. 2013, 36, 282–292. [Google Scholar] [CrossRef]
- Huang, X.; Liu, Y.; Lu, Y.; Ma, C. Anti-inflammatory effects of eugenol on lipopolysaccharide-induced inflammatory reaction in acute lung injury via regulating inflammation and redox status. Int. Immunopharmacol. 2015, 26, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Barbieri, R.; Coppo, E.; Orhan, I.E.; Daglia, M.; Nabavi, S.F.; Izadi, M.; Abdollahi, M.; Nabavi, S.M.; Ajami, M. Antimicrobial activity of eugenol and essential oils containing eugenol: A mechanistic viewpoint. Crit. Rev. Microbiol. 2017, 43, 668–689. [Google Scholar] [CrossRef] [PubMed]
- Tsaroucha, A.; Kaldis, V.; Vailas, M.; Schizas, D.; Lambropoulou, M.; Papalois, A.; Tsigalou, C.; Gaitanidis, A.; Pitiakoudis, M.; Simopoulos, C. The positive effect of eugenol on acute pancreatic tissue injury: A rat experimental model. Pan. Afr. Med. J. 2021, 38, 132. [Google Scholar] [CrossRef] [PubMed]
- Sowjanya, J.; Sandhya, T.; Veeresh, B. Ameliorating effect of eugenol on L-arginine induced acute pancreatitis and associated pulmonary complications in rats. Pharmacologia 2012, 3, 657–664. [Google Scholar] [CrossRef]
- Hui, Q.; Ammeter, E.; Liu, S.; Yang, R.; Lu, P.; Lahaye, L.; Yang, C. Eugenol attenuates inflammatory response and enhances barrier function during lipopolysaccharide-induced inflammation in the porcine intestinal epithelial cells. J. Anim. Sci. 2020, 98, skaa245. [Google Scholar] [CrossRef] [PubMed]
- Markakis, C.; Tsaroucha, A.; Papalois, A.E.; Lambropoulou, M.; Spartalis, E.; Tsigalou, C.; Romanidis, C.; Simopoulos, C. The role of eugenol in the prevention of acute pancreatitis-induced acute kidney injury: Experimental study. HPB Surg. 2016, 2016, 3203147. [Google Scholar] [CrossRef]
- Pavlidis, E.T.; Lambropoulou, M.; Symeonidis, N.G.; Anagnostopoulos, C.; Tsaroucha, A.; Kotini, A.; Nikolaidou, C.; Kiziridou, A.; Simopoulos, C. The immunohistochemical expression MTA1 protein and its prognostic value in pancreatic cancer. J. Investig. Surg. 2018, 31, 142–150. [Google Scholar] [CrossRef]
- Kaiser, A.M.; Saluja, A.K.; Sengupta, A.; Saluja, M.; Steer, M.L. Relationship between severity, necrosis, and apoptosis in five models of experimental acute pancreatitis. Am. J. Physiol. 1995, 269 Pt 1, C1295–C1304. [Google Scholar] [CrossRef]
- Su, K.H.; Cuthbertson, C.; Christophi, C. Review of experimental animal models of acute pancreatitis. HPB 2006, 8, 264–286. [Google Scholar] [CrossRef]
- Inagaki, T.; Hoshino, M.; Hayakawa, T.; Ohara, H.; Yamada, T.; Yamada, H.; Iida, M.; Nakazawa, T.; Ogasawara, T.; Uchida, A.; et al. Interleukin-6 is a useful marker for early prediction of the severity of acute pancreatitis. Pancreas 1997, 14, 1–8. [Google Scholar] [CrossRef]
- Berney, T.; Gasche, Y.; Robert, J.; Jenny, A.; Mensi, N.; Grau, G.; Vermeulen, B.; Morel, P. Serum profiles of interleukin-6, interleukin-8, and interleukin-10 in patients with severe and mild acute pancreatitis. Pancreas 1999, 18, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Malmstrøm, M.L.; Hansen, M.B.; Andersen, A.M.; Ersbøll, A.K.; Nielsen, O.H.; Jørgensen, L.N.; Novovic, S. Cytokines and organ failure in acute pancreatitis: Inflammatory response in acute pancreatitis. Pancreas 2012, 41, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Yogalakshmi, B.; Viswanathan, P.; Anuradha, C.V. Investigation of antioxidant, anti-inflammatory and DNA-protective properties of eugenol in thioacetamide-induced liver injury in rats. Toxicology 2010, 268, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Lotze, M.T.; Tracey, K.J. High-mobility group box 1 protein (HMGB1): Nuclear weapon in the immune arsenal. Nat. Rev. Immunol. 2005, 5, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Zhang, Q.; Hou, W.; Yan, Z.; Chen, R.; Bonaroti, J.; Bansal, P.; Billiar, T.R.; Tsung, A.; Wang, Q.; et al. Intracellular HMGB1 inhibits inflammatory nucleosome release and limits acute pancreatitis in mice. Gastroenterology 2014, 146, 1097–1107. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhao, H.X.; Bai, C.; Zhou, X.Y. Blockade of high-mobility group box 1 attenuates intestinal mucosal barrier dysfunction in experimental acute pancreatitis. Sci. Rep. 2017, 7, 6799. [Google Scholar] [CrossRef]
- Sappington, P.L.; Yang, R.; Yang, H.; Tracey, K.J.; Delude, R.L.; Fink, M.P. HMGB1 B box increases the permeability of Caco-2 enterocytic monolayers and impairs intestinal barrier function in mice. Gastroenterology 2002, 123, 790–802. [Google Scholar] [CrossRef]
- Xu, G.F.; Guo, M.; Tian, Z.Q.; Wu, G.Z.; Zou, X.P.; Zhang, W.J. Increased of serum high-mobility group box chromosomal protein 1 correlated with intestinal mucosal barrier injury in patients with severe acute pancreatitis. World J. Emerg. Surg. 2014, 9, 61. [Google Scholar] [CrossRef]
- Pullar, J.M.; Vissers, M.C.; Winterbourn, C.C. Living with a killer: The effects of hypochlorous acid on mammalian cells. IUBMB Life 2000, 50, 259–266. [Google Scholar] [CrossRef]
- Tacchini-Cottier, F.; Zweifel, C.; Belkaid, Y.; Mukankundiye, C.; Vasei, M.; Launois, P.; Milon, G.; Louis, J.A. An immunomodulatory function for neutrophils during the induction of a CD4+ Th2 response in BALB/c mice infected with Leishmania major. J. Immunol. 2000, 165, 2628–2636. [Google Scholar] [CrossRef]
- Rehg, J.E.; Bush, D.; Ward, J.M. The utility of immunohistochemistry for the identification of hematopoietic and lymphoid cells in normal tissues and interpretation of proliferative and inflammatory lesions of mice and rats. Toxicol. Pathol. 2012, 40, 345–374. [Google Scholar] [CrossRef] [PubMed]
- Abd El Motteleb, D.M.; Selim, S.A.; Mohamed, A.M. Differential effects of eugenol against hepatic inflammation and overall damage induced by ischemia/re-perfusion injury. J. Immunotoxicol. 2014, 11, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Rau, B.; Baumgart, K.; Paszkowski, A.S.; Mayer, J.M.; Beger, H.G. Clinical relevance of caspase-1 activated cytokines in acute pancreatitis: High correlation of serum interleukin-18 with pancreatic necrosis and systemic complications. Crit. Care Med. 2001, 29, 1556–1562. [Google Scholar] [CrossRef] [PubMed]
- Wereszczynska-Siemiatkowska, U.; Mroczko, B.; Siemiatkowski, A. Serum profiles of interleukin-18 in different severity forms of human acute pancreatitis. Scand. J. Gastroenterol. 2002, 37, 1097–1102. [Google Scholar] [CrossRef]
- Corsini, E.; Mitjans, M.; Galbiati, V.; Lucchi, L.; Galli, C.L.; Marinovich, M. Use of IL-18 production in a human keratinocyte cell line to discriminate contact sensitizers from irritants and low molecular weight respiratory allergens. Toxicol. In Vitro 2009, 23, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zheng, S.; Wei, S.; Tian, Q.; Tao, Y.; Bo, R.; Liu, M.; Li, J. The protective effect and potential mechanisms of eugenol against Salmonella in vivo and in vitro. Poult. Sci. 2020, 101, 101801. [Google Scholar] [CrossRef]
- Grespan, R.; Paludo, M.; Lemos, H.; Barbosa, C.P.; Bersani-Amado, C.A.; Dalalio, M.M.; Cuman, R.K. Anti-arthritic effect of eugenol on collagen-induced arthritis experimental model. Biol. Pharm. 2012, 35, 1818–1820. [Google Scholar] [CrossRef]
- Ma, L.; Mu, Y.; Zhang, Z.; Sun, Q. Eugenol promotes functional recovery and alleviates inflammation, oxidative stress, and neural apoptosis in a rat model of spinal cord injury. Restor. Neurol. Neurosci. 2018, 36, 659–668. [Google Scholar] [CrossRef]
- Guenette, S.A.; Beaudry, F.; Marier, J.F.; Vachon, P. Pharmacokinetics and anesthetic activity of eugenol in male Sprague-Dawley rats. J. Vet. Pharmacol. Ther. 2006, 29, 265–270. [Google Scholar] [CrossRef]

| Hyperemia | Inflammatory Infiltrate in Mucosal Layer | Inflammatory Infiltrate in Submucosal Layer | Inflammatory Infiltrate in Muscular Layer | ||
|---|---|---|---|---|---|
| sham | 6 | 1 (0.25–1) | 0.50 (0–1) | 0.5 (0–1) | 0 (0) |
| 12 | 0.50 (0–1) | 0 (0) | 0 (0) | 0.5 (0–1) | |
| 24 | 1 (0.25–1) | 0 (0) | 0 (0) | 0 (0–0.75) | |
| 48 | 0.50 (0–1) | 0 (0–0.75) | 0 (0) | 0 (0) | |
| 72 | 1 (0.25–1) | 0 (0–0.75) | 0 (0) | 0 (0) | |
| AP | 6 | 0 (0–1) | 0 (0) | 0 (0) | 0 (0) |
| 12 | 1 (0.25–1) | 0 (0) | 0 (0) | 0 (0–1) | |
| 24 | 1 (1) | 0 (0–0.75) | 0.5 (0–1) | 0 (0) | |
| 48 | 1.50 (1–2) * | 0.5 (0–1) | 1 (0.25–1) | 0 (0–1) | |
| 72 | 2 (2) * | 1 (1) * | 1 (1) * | 1 (1) * | |
| AP + eugenol | 6 | 0.50 (0–1) | 0 (0) | 0 (0) | 0 (0) |
| 12 | 0 (0–1) | 0 (0) | 0 (0) | 0 (0) | |
| 24 | 0 (0–1) ** | 0 (0) | 0 (0) | 0 (0) | |
| 48 | 1 (1) | 0 (0–0.75) | 0 (0–0.75) | 1 (0–1) | |
| 72 | 1 (1–1.75) ** | 0.50 (0–1) ** | 1 (0–1) ** | 1 (0–1) ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oikonomou, P.; Nikolaou, C.; Papachristou, F.; Sovatzidis, A.; Lambropoulou, M.; Giouleka, C.; Kontaxis, V.; Linardoutsos, D.; Papalois, A.; Pitiakoudis, M.; et al. Eugenol Reduced ΜPO, CD45 and HMGB1 Expression and Attenuated the Expression of Leukocyte Infiltration Markers in the Intestinal Tissue in Biliopancreatic Duct Ligation-Induced Pancreatitis in Rats. Medicina 2024, 60, 74. https://doi.org/10.3390/medicina60010074
Oikonomou P, Nikolaou C, Papachristou F, Sovatzidis A, Lambropoulou M, Giouleka C, Kontaxis V, Linardoutsos D, Papalois A, Pitiakoudis M, et al. Eugenol Reduced ΜPO, CD45 and HMGB1 Expression and Attenuated the Expression of Leukocyte Infiltration Markers in the Intestinal Tissue in Biliopancreatic Duct Ligation-Induced Pancreatitis in Rats. Medicina. 2024; 60(1):74. https://doi.org/10.3390/medicina60010074
Chicago/Turabian StyleOikonomou, Panagoula, Christina Nikolaou, Fotini Papachristou, Apostolos Sovatzidis, Maria Lambropoulou, Charikleia Giouleka, Vasileios Kontaxis, Dimitrios Linardoutsos, Apostolos Papalois, Michael Pitiakoudis, and et al. 2024. "Eugenol Reduced ΜPO, CD45 and HMGB1 Expression and Attenuated the Expression of Leukocyte Infiltration Markers in the Intestinal Tissue in Biliopancreatic Duct Ligation-Induced Pancreatitis in Rats" Medicina 60, no. 1: 74. https://doi.org/10.3390/medicina60010074





