Efficacy of Long-Term Use of Azithromycin in the Management of Cystic Fibrosis in Pediatric Patients with or Without Pseudomonas aeruginosa: A Systematic Review and Meta-Analysis Article
Abstract
:1. Introduction
2. Materials and Methods
2.1. Database Searching and Screening
2.2. Eligibility Criteria
2.3. Screening
2.4. Data Extraction
2.5. Quality and Risk of Bias Assessments
2.6. Statistical Analysis
3. Results
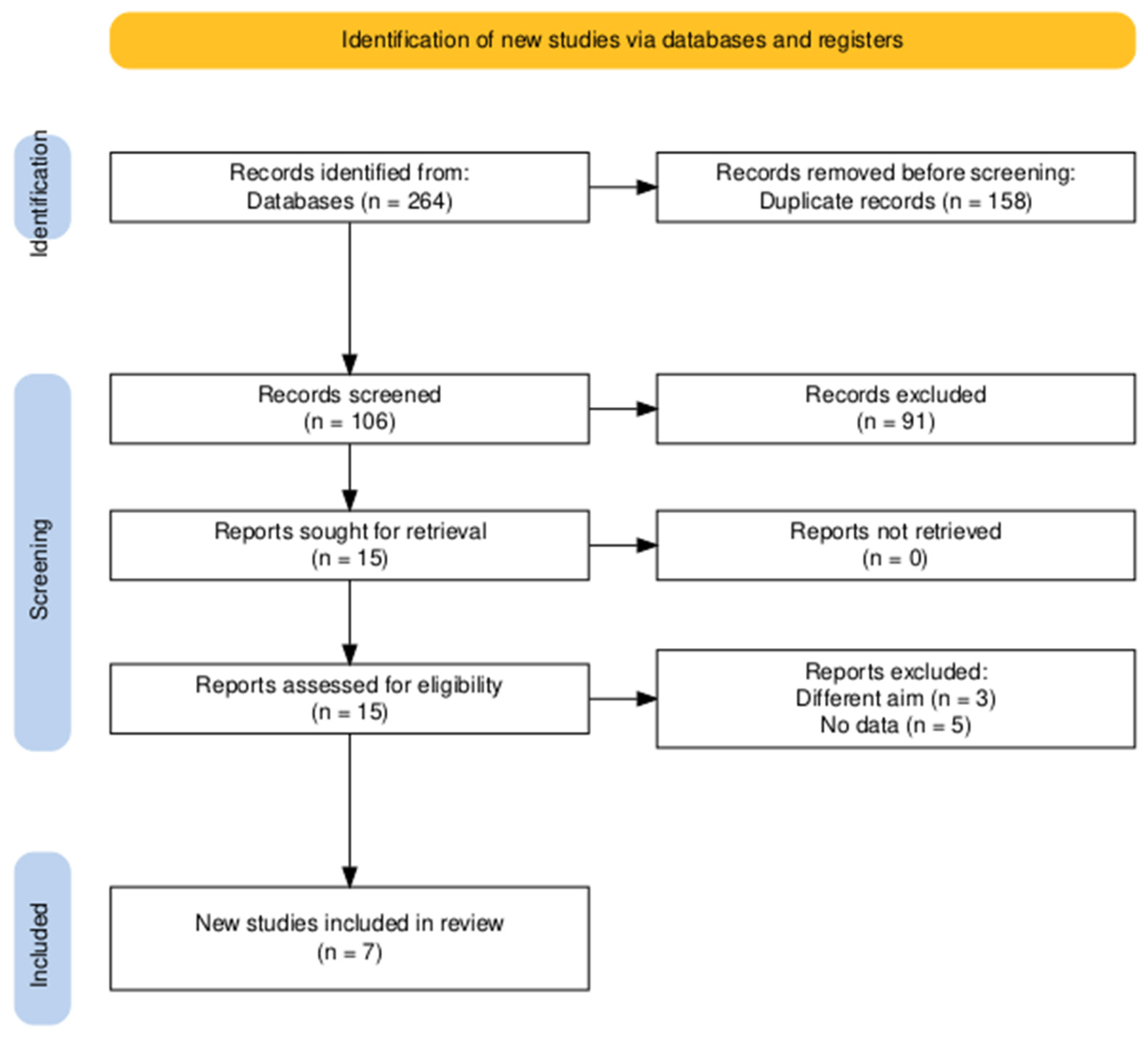
3.1. Search Results and Screening
3.2. Quality and Risk of Bias Assessments
3.3. Baseline Characteristics
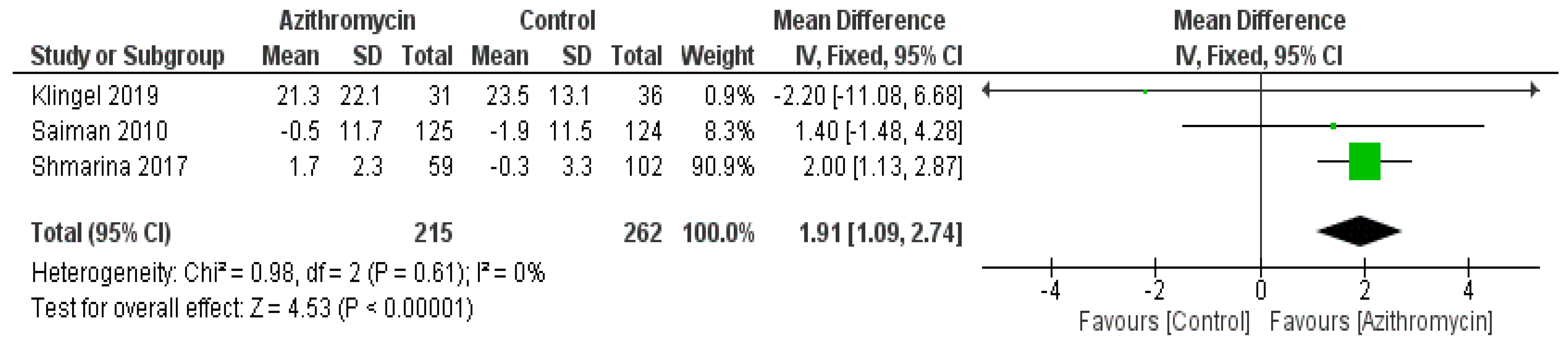
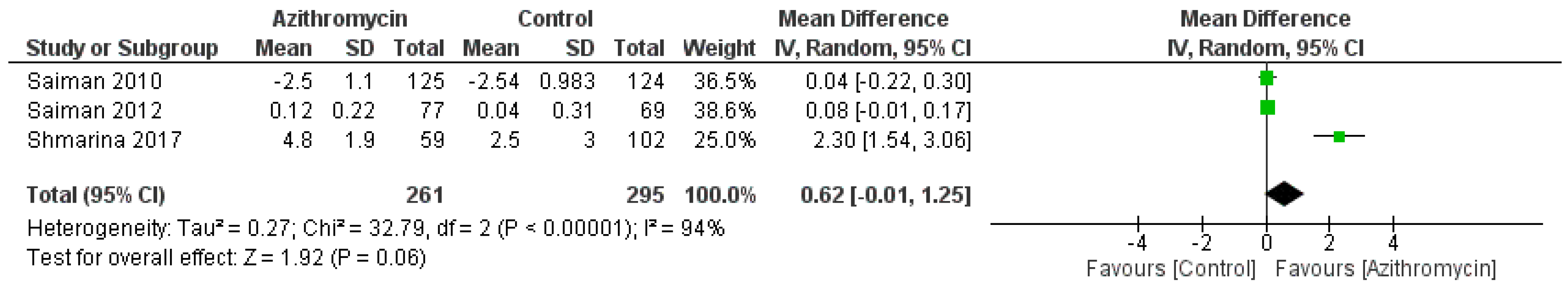
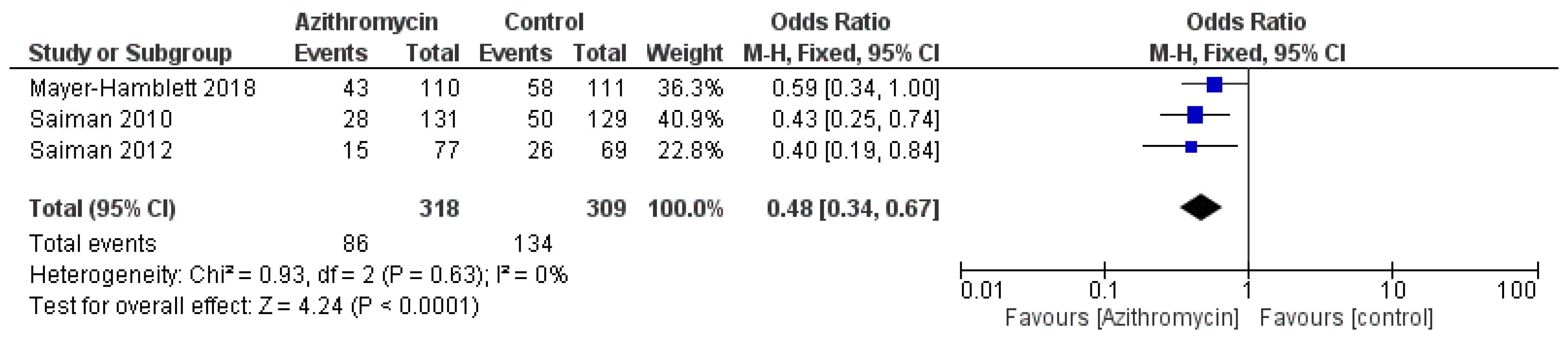
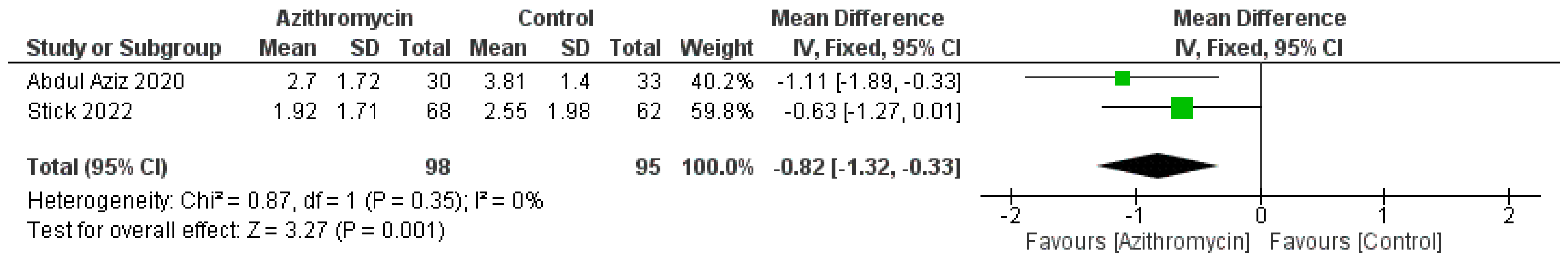
3.4. Statistical Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Cogen, J.D.; Oron, A.P.; Gibson, R.L.; Hoffman, L.R.; Kronman, M.P.; Ong, T.; Rosenfeld, M. Characterization of Inpatient Cystic Fibrosis Pulmonary Exacerbations. Pediatrics 2017, 139, e20162642. [Google Scholar] [CrossRef] [PubMed]
- Aziz, D.A.; Billoo, A.G.; Qureshi, A.; Khalid, M.; Kirmani, S. Clinical and laboratory profile of children with Cystic Fibrosis: Experience of a tertiary care center in Pakistan. Pak. J. Med. Sci. 2017, 33, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Sanders, D.B.; Fink, A.K. Background and Epidemiology. Pediatr. Clin. N. Am. 2016, 63, 567–584. [Google Scholar] [CrossRef]
- Cant, N.; Pollock, N.; Ford, R.C. CFTR structure and cystic fibrosis. Int. J. Biochem. Cell Biol. 2014, 52, 15–25. [Google Scholar] [CrossRef]
- Berkebile, A.R.; McCray, P.B., Jr. Effects of airway surface liquid pH on host defense in cystic fibrosis. Int. J. Biochem. Cell Biol. 2014, 52, 124–129. [Google Scholar] [CrossRef]
- Stoltz, D.A.; Meyerholz, D.K.; Welsh, M.J. Origins of cystic fibrosis lung disease. N. Engl. J. Med. 2015, 372, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Cantin, A.M.; Hartl, D.; Konstan, M.W.; Chmiel, J.F. Inflammation in cystic fibrosis lung disease: Pathogenesis and therapy. J. Cyst. Fibros. 2015, 14, 419–430. [Google Scholar] [CrossRef]
- Nichols, D.P.; Chmiel, J.F. Inflammation and its genesis in cystic fibrosis. Pediatr. Pulmonol. 2015, 50 (Suppl. 40), S39–S56. [Google Scholar] [CrossRef]
- Goss, C.H.; Burns, J.L. Exacerbations in cystic fibrosis. 1: Epidemiology and pathogenesis. Thorax 2007, 62, 360–367. [Google Scholar] [CrossRef]
- Boucher, R.C. New concepts of the pathogenesis of cystic fibrosis lung disease. Eur. Respir. J. 2004, 23, 146–158. [Google Scholar] [CrossRef]
- Bonfield, T.L.; Panuska, J.R.; Konstan, M.W.; Hilliard, K.A.; Hilliard, J.B.; Ghnaim, H.; Berger, M. Inflammatory cytokines in cystic fibrosis lungs. Am. J. Respir. Crit. Care Med. 1995, 152 Pt 1, 2111–2118. [Google Scholar] [CrossRef] [PubMed]
- Saiman, L.; Marshall, B.C.; Mayer-Hamblett, N.; Burns, J.L.; Quittner, A.L.; Cibene, D.A.; Macrolide Study Group. Azithromycin in patients with cystic fibrosis chronically infected with Pseudomonas aeruginosa: A randomized controlled trial. JAMA 2003, 290, 1749–1756. [Google Scholar] [CrossRef] [PubMed]
- Saiman, L.; Anstead, M.; Mayer-Hamblett, N.; Lands, L.C.; Kloster, M.; Hocevar-Trnka, J.; AZ0004 Azithromycin Study Group. Effect of azithromycin on pulmonary function in patients with cystic fibrosis uninfected with Pseudomonas aeruginosa: A randomized controlled trial. JAMA 2010, 303, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Ratjen, F.; Saiman, L.; Mayer-Hamblett, N.; Lands, L.C.; Kloster, M.; Thompson, V.; Anstead, M. Effect of azithromycin on systemic markers of inflammation in patients with cystic fibrosis uninfected with Pseudomonas aeruginosa. Chest 2012, 142, 1259–1266. [Google Scholar] [CrossRef]
- Ichimiya, T.; Takeoka, K.; Hiramatsu, K.; Hirai, K.; Yamasaki, T.; Nasu, M. The influence of azithromycin on the biofilm formation of Pseudomonas aeruginosa in vitro. Chemotherapy 1996, 42, 186–191. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Rayyan. Available online: https://rayyan.ai/ (accessed on 1 September 2024).
- Goebell, P.J.; Kamat, A.M.; Sylvester, R.J.; Black, P.; Droller, M.; Godoy, G.; Hudson, M.A.; Junker, K.; Kassouf, W.; Knowles, M.A.; et al. Assessing the quality of studies on the diagnostic accuracy of tumor markers. Urol Oncol. 2014, 32, 1051–1060. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Flemyng, E.; Moore, T.H.; Boutron, I.; Higgins, J.P.; Hróbjartsson, A.; Nejstgaard, C.H.; Dwan, K. Using Risk of Bias 2 to assess results from randomised controlled trials: Guidance from Cochrane. BMJ Evid. Based Med. 2023, 28, 260–266. [Google Scholar] [CrossRef]
- RevMan: Systematic Review and Meta-Analysis Tool for Researchers Worldwide | Cochrane RevMan. Available online: https://revman.cochrane.org/info (accessed on 1 September 2024).
- Mayer-Hamblett, N.; Retsch-Bogart, G.; Kloster, M.; Accurso, F.; Rosenfeld, M.; Albers, G.; Ramsey, B.W. Azithromycin for Early Pseudomonas Infection in Cystic Fibrosis. The OPTIMIZE Randomized Trial. Am. J. Respir. Crit. Care Med. 2018, 198, 1177–1187. [Google Scholar] [CrossRef]
- Aziz, D.A.; Shahid, S.; Iftikhar, H.; Mir, F. Role of Long-Term Intermittent Use of Oral Azithromycin On Pulmonary Exacerbations In Cystic Fibrosis Children. J. Ayub Med. Coll. Abbottabad 2020, 32, 429–434. [Google Scholar]
- Shmarina, G.; Pukhalsky, A.; Avakian, L.; Semykin, S.; Pukhalskaya, D.; Alioshkin, V. Steady-State Therapy with Azithromycin or Low-Dose Prednisolone in Paediatric Cystic Fibrosis Patients: Inflammatory Markers and Disease Progression. Int. Arch. Allergy Immunol. 2017, 172, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Stick, S.M.; Foti, A.; Ware, R.S.; Tiddens, H.A.; Clements, B.S.; Armstrong, D.S.; Sly, P.D. The effect of azithromycin on structural lung disease in infants with cystic fibrosis (COMBAT CF): A phase 3, randomised, double-blind, placebo-controlled clinical trial. Lancet Respir. Med. 2022, 10, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Klingel, M.; Stanojevic, S.; Tullis, E.; Ratjen, F.; Waters, V. Oral Azithromycin and Response to Pulmonary Exacerbations Treated with Intravenous Tobramycin in Children with Cystic Fibrosis. Ann. Am. Thorac. Soc. 2019, 16, 861–867. [Google Scholar] [CrossRef]
- Saiman, L.; Mayer-Hamblett, N.; Anstead, M.; Lands, L.C.; Kloster, M.; Goss, C.H.; AZ0004 Macrolide Study Team. Open-label, follow-on study of azithromycin in pediatric patients with CF uninfected with Pseudomonas aeruginosa. Pediatr. Pulmonol. 2012, 47, 641–648. [Google Scholar] [CrossRef]
- Equi, A.; Balfour-Lynn, I.M.; Bush, A.; Rosenthal, M. Long term azithromycin in children with cystic fibrosis: A randomised, placebo-controlled crossover trial. Lancet 2002, 360, 978–984. [Google Scholar] [CrossRef]
- Clement, A.; Tamalet, A.; Leroux, E.; Ravilly, S.; Fauroux, B.; Jais, J.P. Long term effects of azithromycin in patients with cystic fibrosis: A double blind, placebo controlled trial. Thorax 2006, 61, 895–902. [Google Scholar] [CrossRef]
- Saiman, L. The use of macrolide antibiotics in patients with cystic fibrosis. Curr. Opin. Pulm. Med. 2004, 10, 515–523. [Google Scholar] [CrossRef]
- Tateda, K.; Ishii, Y.; Kimura, S.; Horikawa, M.; Miyairi, S.; Yamaguchi, K. Suppression of Pseudomonas aeruginosa quorum-sensing systems by macrolides: A promising strategy or an oriental mystery? J. Infect. Chemother. 2007, 13, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.C.; Rodriguez, M.L.; Young, K.S.; Deng, J.C.; Thannickal, V.J.; Tateda, K.; Standiford, T.J. Azithromycin blocks neutrophil recruitment in Pseudomonas endobronchial infection. Am. J. Respir. Crit. Care Med. 2004, 170, 1331–1339. [Google Scholar] [CrossRef]
- Verleden, G.M.; Vanaudenaerde, B.M.; Dupont, L.J.; Van Raemdonck, D.E. Azithromycin reduces airway neutrophilia and interleukin-8 in patients with bronchiolitis obliterans syndrome. Am. J. Respir. Crit. Care Med. 2006, 174, 566–570. [Google Scholar] [CrossRef]
- Cigana, C.; Nicolis, E.; Pasetto, M.; Assael, B.M.; Melotti, P. Anti-inflammatory effects of azithromycin in cystic fibrosis airway epithelial cells. Biochem. Biophys. Res. Commun. 2006, 350, 977–982. [Google Scholar] [CrossRef]
- Legssyer, R.; Huaux, F.; Lebacq, J.; Delos, M.; Marbaix, E.; Lebecque, P.; Leal, T. Azithromycin reduces spontaneous and induced inflammation in DeltaF508 cystic fibrosis mice. Respir. Res. 2006, 7, 134. [Google Scholar] [CrossRef]
- Parnham, M.J.; Haber, V.E.; Giamarellos-Bourboulis, E.J.; Perletti, G.; Verleden, G.M.; Vos, R. Azithromycin: Mechanisms of action and their relevance for clinical applications. Pharmacol. Ther. 2014, 143, 225–245. [Google Scholar] [CrossRef] [PubMed]
- Venditto, V.J.; Haydar, D.; Abdel-Latif, A.; Gensel, J.C.; Anstead, M.I.; Pitts, M.G.; Feola, D.J. Immunomodulatory Effects of Azithromycin Revisited: Potential Applications to COVID-19. Front. Immunol. 2021, 12, 574425. [Google Scholar] [CrossRef]
- Čulić, O.; Eraković, V.; Čepelak, I.; Barišić, K.; Brajša, K.; Ferenčić, Ž.; Parnham, M.J. Azithromycin modulates neutrophil function and circulating inflammatory mediators in healthy human subjects. Eur. J. Pharmacol. 2002, 450, 277–289. [Google Scholar] [CrossRef]
- Davis, T.A.; Miller, A.; Hachem, C.; Velez, C.; Patel, D. The current state of gastrointestinal motility evaluation in cystic fibrosis: A comprehensive literature review. Transl. Gastroenterol. Hepatol. 2024, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Shakir, A.K.; Altaf, M.A. Azithromycin Induces Migrating Motor Complexes in Pediatric Patients Undergoing Antroduodenal Motility Studies. J. Pediatr. Pharmacol. Ther. 2018, 23, 390–394. [Google Scholar] [CrossRef]
- McDonnell, L.; Gilkes, A.; Ashworth, M.; Rowland, V.; Harries, T.H.; Armstrong, D.; White, P. Association between antibiotics and gut microbiome dysbiosis in children: Systematic review and meta-analysis. Gut Microbes 2021, 13, 1–18. [Google Scholar] [CrossRef]
- Heidary, M.; Ebrahimi Samangani, A.; Kargari, A.; Kiani Nejad, A.; Yashmi, I.; Motahar, M.; Khoshnood, S. Mechanism of action, resistance, synergism, and clinical implications of azithromycin. J. Clin. Lab. Anal. 2022, 36, e24427. [Google Scholar] [CrossRef]
- Albert, R.K.; Connett, J.; Bailey, W.C.; Casaburi, R.; Cooper, J.A.D., Jr.; Criner, G.J.; Anthonisen, N.R. Azithromycin for prevention of exacerbations of COPD. N. Engl. J. Med. 2011, 365, 689–698. [Google Scholar] [CrossRef]
- Grayston, J.T.; Kronmal, R.A.; Jackson, L.A.; Parisi, A.F.; Muhlestein, J.B.; Cohen, J.D.; Knirsch, C. Azithromycin for the secondary prevention of coronary events. N. Engl. J. Med. 2005, 352, 1637–1645. [Google Scholar] [CrossRef] [PubMed]
- Ray, W.A.; Murray, K.T.; Hall, K.; Arbogast, P.G.; Stein, C.M. Azithromycin and the risk of cardiovascular death. N. Engl. J. Med. 2012, 366, 1881–1890. [Google Scholar] [CrossRef] [PubMed]
- Nichols, D.P.; Happoldt, C.L.; Bratcher, P.E.; Caceres, S.M.; Chmiel, J.F.; Malcolm, K.C.; Nick, J.A. Impact of azithromycin on the clinical and antimicrobial effectiveness of tobramycin in the treatment of cystic fibrosis. J. Cyst. Fibros. 2017, 16, 358–366. [Google Scholar] [CrossRef]
- Nick, J.A.; Moskowitz, S.M.; Chmiel, J.F.; Forssén, A.V.; Kim, S.H.; Saavedra, M.T.; Nichols, D.P. Azithromycin may antagonize inhaled tobramycin when targeting Pseudomonas aeruginosa in cystic fibrosis. Ann. Am. Thorac. Soc. 2014, 11, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, D.H.; Chen, L.L.; Zhao, Q.; Yu, Y.Z.; Ding, J.J.; Xie, C.M. Meta-analysis of the adverse effects of long-term azithromycin use in patients with chronic lung diseases. Antimicrob. Agents Chemother. 2014, 58, 511–517. [Google Scholar] [CrossRef]
- Harman, K.; Dobra, R.; Davies, J.C. Disease-modifying drug therapy in cystic fibrosis. Paediatr. Respir. Rev. 2018, 26, 7–9. [Google Scholar] [CrossRef]


| Study Name | Representativeness of the Exposed Cohort (★) | Selection of the Non-Exposed Cohort (★) | Ascertainment of Exposure (★) | Demonstration That Outcome of Interest Was Not Present at Start of Study (★) | Comparability of Cohorts Based on the Design or Analysis (Max.: ★★) | Was Follow-Up Long Enough for Outcomes to Occur? (★) | Assessment of Outcome (★) | Adequacy of Follow-Up of Cohorts (★) | Quality Level |
|---|---|---|---|---|---|---|---|---|---|
| Shmarina 2017 [23] | ✩ | ✩ | ✩ | ✩ | ✩ | ✩ | ✩ | ✩ | High |
| Klingel 2019 [25] | ✩ | ✩ | ✩ | ✩ | ✩ | ✩ | ✩ | - | High |
| Abdul Aziz 2020 [22] | ✩ | - | ✩ | ✩ | ✩ | ✩ | ✩ | ✩ | High |
| Study ID | Design | Population | Follow-Up (Months) | Sample Size | Age, Mean (SD) Years | Male, n (%) | Other Drugs | |||
|---|---|---|---|---|---|---|---|---|---|---|
| AZN | No AZN | AZN | No AZN | AZN | No AZN | |||||
| Saiman 2010 [13] | RCT | Patients with cystic fibrosis uninfected with Pseudomonas aeruginosa | 5.6 | 131 | 129 | 10.7 (3.25) | 10.6 (3.1) | 77 (59) | 70 (71) | Dornase alfa, tobramycin, ibuprofen, and hypertonic saline |
| Stick 2022 [24] | RCT | Infants with cystic fibrosis | 36 | 68 | 62 | 3.6 (1.2) months | 3.6 (1.3) months | 38 (56) | 40 (65) | NR |
| Mayer-Hamblett 2018 [21] | RCT | Early Pseudomonas infection in cystic fibrosis | 11 | 110 | 111 | 7.1 (5.1) | 6.8 (5) | 55 (50) | 62 (55.9) | Ivacaftor/lumacaftor |
| Shmarina 2017 [23] | Cohort | Pediatric patients with cystic fibrosis | 12 | 59 | 102 | 14.3 (0.4) | 13 (0.4) | 28 (47.5) | 59 (57.8) | NR |
| Klingel 2019 [25] | Cohort | Children with cystic fibrosis with exacerbations who are on tobramycin | 12 | 31 | 36 | 13.6 (10.6 to 15.4) median (IQR) | 14.9 (12.3 to 16.1) | NR | NR | Tobramycin, colistimethate, amikacin, and aztreonam |
| Abdul Aziz 2020 [22] | Cohort | Children with cystic fibrosis with exacerbations who are on tobramycin | 12 | 30 | 33 | 14.4 (3) | 9.96 (3.57) | 18 (60%) | 21 (63.6) | NR |
| Saiman 2012 [26] | RCT | Patients with cystic fibrosis uninfected with Pseudomonas aeruginosa | 5.6 | 77 | 69 | 6–18 years | NR | NR | NR | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-shehri, H.; Albassam, D. Efficacy of Long-Term Use of Azithromycin in the Management of Cystic Fibrosis in Pediatric Patients with or Without Pseudomonas aeruginosa: A Systematic Review and Meta-Analysis Article. Medicina 2025, 61, 653. https://doi.org/10.3390/medicina61040653
Al-shehri H, Albassam D. Efficacy of Long-Term Use of Azithromycin in the Management of Cystic Fibrosis in Pediatric Patients with or Without Pseudomonas aeruginosa: A Systematic Review and Meta-Analysis Article. Medicina. 2025; 61(4):653. https://doi.org/10.3390/medicina61040653
Chicago/Turabian StyleAl-shehri, Hassan, and Dana Albassam. 2025. "Efficacy of Long-Term Use of Azithromycin in the Management of Cystic Fibrosis in Pediatric Patients with or Without Pseudomonas aeruginosa: A Systematic Review and Meta-Analysis Article" Medicina 61, no. 4: 653. https://doi.org/10.3390/medicina61040653
APA StyleAl-shehri, H., & Albassam, D. (2025). Efficacy of Long-Term Use of Azithromycin in the Management of Cystic Fibrosis in Pediatric Patients with or Without Pseudomonas aeruginosa: A Systematic Review and Meta-Analysis Article. Medicina, 61(4), 653. https://doi.org/10.3390/medicina61040653










