An Overview on the Effect of Neonicotinoid Insecticides on Mammalian Cholinergic Functions through the Activation of Neuronal Nicotinic Acetylcholine Receptors
Abstract
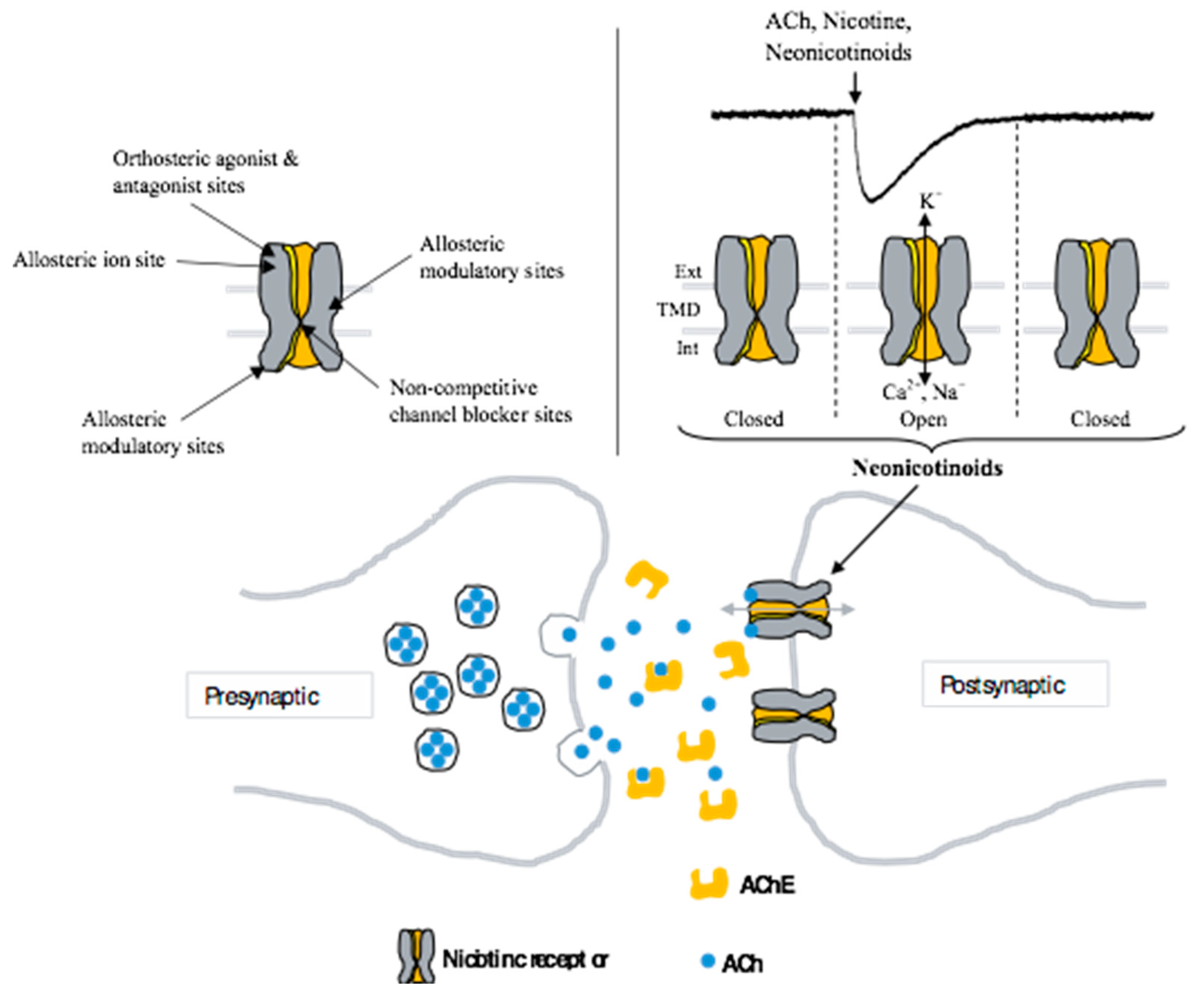
:1. Introduction
2. Diversity of Mammalian Neuronal Nicotinic Acetylcholine Receptors
2.1. Multiple Receptor Subtypes and Different Pharmacological Properties
2.2. Neonicotinoids Are Poor Activators of Mammalian Neuronal nAChRs
3. Detoxification Mechanisms in Mammals in Regards to Neonicotinoid Sensitivity
4. Alterations of Cholinergic Functions
4.1. Modulation of Mammalian Neuronal nAChR Function
4.2. Are Neonicotinoid Insecticides Able to Interact with Mechanisms Involved in Neurodegenerative Diseases?
4.3. Neonicotinoid Insecticides Affect Other Mammalian Neuronal Mechanisms
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Changeux, J.-P.; Edelstein, S.J. Allosteric Receptors after 30 Years. Neuron 1998, 21, 959–980. [Google Scholar] [CrossRef] [Green Version]
- Changeux, J.-P. The concept of allosteric modulation: An overview. Drug Discov. Today Technol. 2013, 10, e223–e228. [Google Scholar] [CrossRef] [PubMed]
- Changeux, J.-P.; Edelstein, S.J. Allosteric Mechanisms of Signal Transduction. Science 2005, 308, 1424–1428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arora, K.; Cheng, J.; Nichols, R.A. Nicotinic Acetylcholine Receptors Sensitize a MAPK-linked Toxicity Pathway on Prolonged Exposure to β-Amyloid. J. Boil. Chem. 2015, 290, 21409–21420. [Google Scholar] [CrossRef] [Green Version]
- Barykin, E.P.; Garifulina, A.I.; Kryukova, E.V.; Spirova, E.N.; Anashkina, A.; Adzhubei, A.A.; Shelukhina, I.V.; Kasheverov, I.E.; Mitkevich, V.A.; Кoзин, С.А.; et al. Isomerization of Asp7 in Beta-Amyloid Enhances Inhibition of the α7 Nicotinic Receptor and Promotes Neurotoxicity. Cells 2019, 8, 771. [Google Scholar] [CrossRef] [Green Version]
- Tanimura, A.; Du, Y.; Kondapalli, J.; Wokosin, D.L.; Surmeier, D.J. Cholinergic Interneurons Amplify Thalamostriatal Excitation of Striatal Indirect Pathway Neurons in Parkinson’s Disease Models. Neuron 2019, 101, 444–458. [Google Scholar] [CrossRef] [Green Version]
- Leino, S.; Koski, S.; Hanninen, R.; Tapanainen, T.; Rannanpää, S.; Salminen, O. Attenuated dopaminergic neurodegeneration and motor dysfunction in hemiparkinsonian mice lacking the α5 nicotinic acetylcholine receptor subunit. Neuropharmacology 2018, 138, 371–380. [Google Scholar] [CrossRef]
- Le Novère, N.; Corringer, P.-J.; Changeux, J.-P. The diversity of subunit composition in nAChRs: Evolutionary origins, physiologic and pharmacologic consequences. J. Neurobiol. 2002, 53, 447–456. [Google Scholar] [CrossRef]
- Lukas, R.J.; Changeux, J.P.; Le Novère, N.; Albuquerque, E.X.; Balfour, D.J.; Berg, D.K.; Bertrand, D.; Chiappinelli, V.A.; Clarke, P.B.; Collins, A.C.; et al. International Union of Pharmacology. XX. Current status of the nomenclature for nicotinic acetylcholine receptors and their subunits. Pharmacol. Rev. 1999, 51, 397–401. [Google Scholar]
- Lindstrom, J.; Schoepfer, R.; Conroy, W.; Whiting, P.; Das, M.; Saedi, M.; Anand, R. The Nicotinic Acetylcholine Receptor Gene Family: Structure of Nicotinic Receptors from Muscle and Neurons and Neuronal α-Bungarotoxin-Binding Proteins. Results Probl. Cell Differ. 1991, 287, 255–278. [Google Scholar] [CrossRef]
- Gotti, C.; Zoli, M.; Clementi, F. Brain nicotinic acetylcholine receptors: Native subtypes and their relevance. Trends Pharmacol. Sci. 2006, 27, 482–491. [Google Scholar] [CrossRef]
- Anand, R.; Conroy, W.G.; Schoepfer, R.; Whiting, P.; Lindstrom, J. Neuronal nicotinic acetylcholine receptors expressed in Xenopus oocytes have a pentameric quaternary structure. J. Boil. Chem. 1991, 266, 11192–11198. [Google Scholar]
- Nelson, M.E.; Kuryatov, A.; Choi, C.H.; Zhou, Y.; Lindstrom, J.; Kohout, T.A.; Lefkowitz, R.J. Alternate Stoichiometries of α4β2 Nicotinic Acetylcholine Receptors. Mol. Pharmacol. 2003, 63, 332–341. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Nelson, M.E.; Kuryatov, A.; Choi, C.; Cooper, J.; Lindstrom, J. Human alpha4beta2 acetylcholine receptors formed from linked subunits. J. Neurosci. 2003, 23, 9004–9015. [Google Scholar] [CrossRef] [Green Version]
- DeDominicis, K.E.; Sahibzada, N.; Olson, T.T.; Xiao, Y.; Wolfe, B.B.; Kellar, K.J.; Yasuda, R.P. The (alpha4)3(beta2)2 Stoichiometry of the Nicotinic Acetylcholine Receptor Predominates in the Rat Motor Cortex. Mol. Pharmacol. 2017, 92, 327–337. [Google Scholar] [CrossRef] [Green Version]
- Mazzaferro, S.; Bermudez, I.; Sine, S.M. alpha4beta2 Nicotinic Acetylcholine Receptors: Relationships Between Subunit Stoichiometry and Function at the Single Channel Level. J. Biol. Chem. 2017, 292, 2729–2740. [Google Scholar] [CrossRef] [Green Version]
- Maurer, J.; Sandager-Nielsen, K.; Schmidt, H.D. Attenuation of nicotine taking and seeking in rats by the stoichiometry-selective alpha4beta2 nicotinic acetylcholine receptor positive allosteric modulator NS9283. Psychopharmacology 2016, 234, 475–484. [Google Scholar] [CrossRef]
- Dani, J.A.; Radcliffe, K.A.; Pidoplichko, V.I. Variations in desensitization of nicotinic acetylcholine receptors from hippocampus and midbrain dopamine areas. Eur. J. Pharmacol. 2000, 393, 31–38. [Google Scholar] [CrossRef]
- Séguéla, P.; Wadiche, J.; Dineley-Miller, K.; Dani, J.; Patrick, J. Molecular cloning, functional properties, and distribution of rat brain alpha 7: A nicotinic cation channel highly permeable to calcium. J. Neurosci. 1993, 13, 596–604. [Google Scholar] [CrossRef] [Green Version]
- Bertrand, D.; Galzi, J.L.; Devillérs-Thiery, A.; Bertrand, S.; Changeux, J.P. Mutations at two distinct sites within the channel domain M2 alter calcium permeability of neuronal alpha 7 nicotinic receptor. Proc. Natl. Acad. Sci. USA 1993, 90, 6971–6975. [Google Scholar] [CrossRef] [Green Version]
- Maienfisch, P.; Huerlimann, H.; Rindlisbacher, A.; Gsell, L.; Dettwiler, H.; Haettenschwiler, J.; Sieger, E.; Walti, M. The discovery of thiamethoxam: A second-generation neonicotinoid. Pest Manag. Sci. 2001, 57, 165–176. [Google Scholar] [CrossRef]
- Wakita, T.; Yasui, N.; Yamada, E.; Kishi, D. Development of a novel insecticide, Dinotefuran. J. Pestic. Sci. 2005, 30, 112–123. [Google Scholar] [CrossRef] [Green Version]
- Kagabu, S. Discovery of Imidacloprid and Further Developments from Strategic Molecular Designs. J. Agric. Food Chem. 2011, 59, 2887–2896. [Google Scholar] [CrossRef]
- Tan, J.; Galligan, J.J.; Hollingworth, R.M. Agonist actions of neonicotinoids on nicotinic acetylcholine rececptors expressed by cockroach neurons. NeuroToxicology 2007, 28, 829–842. [Google Scholar] [CrossRef]
- Thany, S.H. Thiamethoxam, a poor agonist of nicotinic acetylcholine receptors expressed on isolated cell bodies, acts as a full agonist at cockroach cercal afferent/giant interneuron synapses. Neuropharmacology 2011, 60, 587–592. [Google Scholar] [CrossRef] [Green Version]
- Nagata, K.; Song, J.H.; Shono, T.; Narahashi, T. Modulation of the neuronal nicotinic acetylcholine receptor-channel by the nitromethylene heterocycle imidacloprid. J. Pharmacol. Exp. Ther. 1998, 285, 731–738. [Google Scholar]
- Ihara, M.; Matsuda, K.; Otake, M.; Kuwamura, M.; Shimomura, M.; Komai, K.; Akamatsu, M.; Raymond, V.; Sattelle, D.B. Diverse actions of neonicotinoids on chicken alpha7, alpha4beta2 and Drosophila-chicken SADbeta2 and ALSbeta2 hybrid nicotinic acetylcholine receptors expressed in Xenopus laevis oocytes. Neuropharmacology 2003, 45, 133–144. [Google Scholar] [CrossRef]
- Clements, J.; Olson, J.M.; Sanchez-Sedillo, B.; Bradford, B.; Groves, R.L. Changes in emergence phenology, fatty acid composition, and xenobiotic-metabolizing enzyme expression is associated with increased insecticide resistance in the Colorado potato beetle. Arch. Insect Biochem. Physiol. 2019, 103, e21630. [Google Scholar] [CrossRef]
- Chen, X.; Tang, C.; Ma, K.; Xia, J.; Song, D.; Gao, X. Overexpression of UDP-glycosyltransferase potentially involved in insecticide resistance in Aphis gossypii Glover collected from Bt cotton fields in China. Pest Manag. Sci. 2019, 76, 1371–1377. [Google Scholar] [CrossRef]
- Tang, B.; Dai, W.; Qi, L.; Zhang, Q.; Zhang, C. Identification and Functional Analysis of a Delta Class Glutathione S-Transferase Gene Associated with Insecticide Detoxification in Bradysia odoriphaga. J. Agric. Food Chem. 2019, 67, 9979–9988. [Google Scholar] [CrossRef]
- Jin, R.; Mao, K.; Liao, X.; Xu, P.; Li, Z.; Ali, E.; Wan, H.; Li, J.-H. Overexpression of CYP6ER1 associated with clothianidin resistance in Nilaparvata lugens (Stål). Pestic. Biochem. Physiol. 2019, 154, 39–45. [Google Scholar] [CrossRef]
- Sun, X.; Gong, Y.; Ali, S.; Hou, M. Mechanisms of resistance to thiamethoxam and dinotefuran compared to imidacloprid in the brown planthopper: Roles of cytochrome P450 monooxygenase and a P450 gene CYP6ER1. Pestic. Biochem. Physiol. 2018, 150, 17–26. [Google Scholar] [CrossRef]
- Elzaki, M.E.A.; Miah, M.A.; Wu, M.; Zhang, H.; Pu, J.; Jiang, L.; Han, Z. Imidacloprid is degraded by CYP353D1v2, a cytochrome P450 overexpressed in a resistant strain of Laodelphax striatellus. Pest Manag. Sci. 2017, 73, 1358–1363. [Google Scholar] [CrossRef]
- Bao, H.; Gao, H.; Zhang, Y.; Fan, D.; Fang, J.; Liu, Z. The roles of CYP6AY1 and CYP6ER1 in imidacloprid resistance in the brown planthopper: Expression levels and detoxification efficiency. Pestic. Biochem. Physiol. 2016, 129, 70–74. [Google Scholar] [CrossRef]
- He, C.; Xie, W.; Yang, X.; Wang, S.-L.; Wu, Q.-J.; Zhang, Y. Identification of glutathione S-transferases in Bemisia tabaci (Hemiptera: Aleyrodidae) and evidence that GSTd7 helps explain the difference in insecticide susceptibility between B. tabaci Middle East-Minor Asia 1 and Mediterranean. Insect Mol. Boil. 2017, 27, 22–35. [Google Scholar] [CrossRef]
- Li, Z.; Li, M.; He, J.; Zhao, X.; Chaimanee, V.; Huang, W.-F.; Nie, H.; Zhao, Y.; Su, S. Differential physiological effects of neonicotinoid insecticides on honey bees: A comparison between Apis mellifera and Apis cerana. Pestic. Biochem. Physiol. 2017, 140, 1–8. [Google Scholar] [CrossRef]
- Dick, R.A.; Kanne, D.B.; Casida, J.E. Identification of Aldehyde Oxidase as the Neonicotinoid Nitroreductase. Chem. Res. Toxicol. 2005, 18, 317–323. [Google Scholar] [CrossRef]
- Dick, R.A.; Kanne, D.B.; Casida, J.E. Substrate Specificity of Rabbit Aldehyde Oxidase for Nitroguanidine and Nitromethylene Neonicotinoid Insecticides. Chem. Res. Toxicol. 2006, 19, 38–43. [Google Scholar] [CrossRef]
- Ford, K.A.; Casida, J.E. Unique and Common Metabolites of Thiamethoxam, Clothianidin, and Dinotefuran in Mice. Chem. Res. Toxicol. 2006, 19, 1549–1556. [Google Scholar] [CrossRef]
- Schulz-Jander, D.A.; Casida, J.E. Imidacloprid insecticide metabolism: Human cytochrome P450 isozymes differ in selectivity for imidazolidine oxidation versus nitroimine reduction. Toxicol. Lett. 2002, 132, 65–70. [Google Scholar] [CrossRef]
- Schulz-Jander, D.A.; Leimkuehler, W.M.; Casida, J.E. Neonicotinoid Insecticides: Reduction and Cleavage of Imidacloprid Nitroimine Substituent by Liver Microsomal and Cytosolic Enzymes. Chem. Res. Toxicol. 2002, 15, 1158–1165. [Google Scholar] [CrossRef]
- Shi, X.; Dick, R.A.; Ford, K.A.; Casida, J.E. Enzymes and Inhibitors in Neonicotinoid Insecticide Metabolism. J. Agric. Food Chem. 2009, 57, 4861–4866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swenson, T.L.; Casida, J.E. Aldehyde Oxidase Importance In Vivo in Xenobiotic Metabolism: Imidacloprid Nitroreduction in Mice. Toxicol. Sci. 2013, 133, 22–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swenson, T.L.; Casida, J.E. Neonicotinoid formaldehyde generators: Possible mechanism of mouse-specific hepatotoxicity/hepatocarcinogenicity of thiamethoxam. Toxicol. Lett. 2013, 216, 139–145. [Google Scholar] [CrossRef]
- Green, T.; Toghill, A.; Lee, R.; Waechter, F.; Weber, E.; Noakes, J. Thiamethoxam induced mouse liver tumors and their relevance to humans. Part 1: mode of action studies in the mouse. Toxicol. Sci. 2005, 86, 36–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Green, T.; Toghill, A.; Lee, R.; Waechter, F.; Weber, E.; Peffer, R.; Noakes, J.; Robinson, M. Thiamethoxam induced mouse liver tumors and their relevance to humans. Part 2: species differences in response. Toxicol. Sci. 2005, 86, 48–55. [Google Scholar] [CrossRef]
- Saeed, N.; Tonina, L.; Battisti, A.; Mori, N. Temperature Alters the Response to Insecticides in Drosophila suzukii (Diptera: Drosophilidae). J. Econ. Èntomol. 2018, 111, 1306–1312. [Google Scholar] [CrossRef]
- Camp, A.; Buchwalter, D.B. Can’t take the heat: Temperature-enhanced toxicity in the mayfly Isonychia bicolor exposed to the neonicotinoid insecticide imidacloprid. Aquat. Toxicol. 2016, 178, 49–57. [Google Scholar] [CrossRef]
- Mao, K.; Jin, R.; Li, W.; Ren, Z.; Qin, X.; He, S.; Li, J.; Wan, H. The influence of temperature on the toxicity of insecticides to Nilaparvata lugens (Stål). Pestic. Biochem. Physiol. 2019, 156, 80–86. [Google Scholar] [CrossRef]
- Li, P.; Ann, J.; Akk, G. Activation and modulation of human alpha4beta2 nicotinic acetylcholine receptors by the neonicotinoids clothianidin and imidacloprid. J. Neurosci. Res. 2011, 89, 1295–1301. [Google Scholar] [CrossRef] [Green Version]
- Cartereau, A.; Martin, C.; Thany, S.H. Neonicotinoid insecticides differently modulate acetycholine-induced currents on mammalian α7 nicotinic acetylcholine receptors. Br. J. Pharmacol. 2017, 175, 1987–1998. [Google Scholar] [CrossRef] [PubMed]
- Toshima, K.I.M.; Kanaoka, S.; Tarumoto, K.; Yamada, A.; Sattelle, D.B.; Matsuda, K. Potentiating and blocking actions of neonicotinoids on the response to acetylcholine of the neuronal alpha4beta2 nicotinic acetylcholine receptor. J. Pestic. Sci. 2008, 33, 146–151. [Google Scholar] [CrossRef] [Green Version]
- Matsuda, K.; Buckingham, S.D.; Freeman, J.C.; Squire, M.D.; Baylis, H.; Sattelle, D.B. Effects of the α subunit on imidacloprid sensitivity of recombinant nicotinic acetylcholine receptors. Br. J. Pharmacol. 1998, 123, 518–524. [Google Scholar] [CrossRef] [Green Version]
- Toshima, K.; Kanaoka, S.; Yamada, A.; Tarumoto, K.; Akamatsu, M.; Sattelle, D.B.; Matsuda, K. Combined roles of loops C and D in the interactions of a neonicotinoid insecticide imidacloprid with the alpha4beta2 nicotinic acetylcholine receptor. Neuropharmacology 2009, 56, 264–272. [Google Scholar] [CrossRef]
- Hirano, T.; Yanai, S.; Takada, T.; Yoneda, N.; Omotehara, T.; Kubota, N.; Minami, K.; Yamamoto, A.; Mantani, Y.; Yokoyama, T.; et al. NOAEL-dose of a neonicotinoid pesticide, clothianidin, acutely induce anxiety-related behavior with human-audible vocalizations in male mice in a novel environment. Toxicol. Lett. 2018, 282, 57–63. [Google Scholar] [CrossRef]
- Bal, R.; Erdogan, S.; Theophilidis, G.; Baydas, G.; Nazıroğlu, M.; Nazıroğlu, M. Assessing the effects of the neonicotinoid insecticide imidacloprid in the cholinergic synapses of the stellate cells of the mouse cochlear nucleus using whole-cell patch-clamp recording. NeuroToxicology 2010, 31, 113–120. [Google Scholar] [CrossRef]
- Kagawa, N.; Nagao, T. Neurodevelopmental toxicity in the mouse neocortex following prenatal exposure to acetamiprid. J. Appl. Toxicol. 2018, 38, 1521–1528. [Google Scholar] [CrossRef]
- Giordano, G.; Afsharinejad, Z.; Guizzetti, M.; Vitalone, A.; Kavanagh, T.J.; Costa, L.G. Organophosphorus insecticides chlorpyrifos and diazinon and oxidative stress in neuronal cells in a genetic model of glutathione deficiency. Toxicol. Appl. Pharmacol. 2007, 219, 181–189. [Google Scholar] [CrossRef]
- Lee, J.E.; Park, J.H.; Shin, I.C.; Koh, H.C. Reactive oxygen species regulated mitochondria-mediated apoptosis in PC12 cells exposed to chlorpyrifos. Toxicol. Appl. Pharmacol. 2012, 263, 148–162. [Google Scholar] [CrossRef]
- Vidau, C.; Polo, R.-A.G.; Niso-Santano, M.; Gómez-Sánchez, R.; Pedro, J.M.B.-S.; Pizarro-Estrella, E.; Blasco, R.; Brunet, J.-L.; Belzunces, L.P.; Fuentes, J.M. Fipronil is a powerful uncoupler of oxidative phosphorylation that triggers apoptosis in human neuronal cell line SHSY5Y. NeuroToxicology 2011, 32, 935–943. [Google Scholar] [CrossRef]
- Dhouib, I.E.; Annabi, A.; Doghri, R.; Rejeb, I.; Dallagi, Y.; Bdiri, Y.; Lasram, M.M.; Elgaaied, A.; Marrakchi, R.; Fazaa, S.; et al. Neuroprotective effects of curcumin against acetamiprid-induced neurotoxicity and oxidative stress in the developing male rat cerebellum: Biochemical, histological, and behavioral changes. Environ. Sci. Pollut. Res. 2017, 24, 27515–27524. [Google Scholar] [CrossRef]
- Barger, S.; Harmon, A.D. Microglial activation by Alzheimer amyloid precursor protein and modulation by apolipoprotein E. Nature 1997, 388, 878–881. [Google Scholar] [CrossRef]
- McDonald, U.R.; Brunden, K.R.; Landreth, G.E. Amyloid Fibrils Activate Tyrosine Kinase-Dependent Signaling and Superoxide Production in Microglia. J. Neurosci. 1997, 17, 2284–2294. [Google Scholar] [CrossRef]
- Kamkwalala, A.; Newhouse, P. Beyond Acetylcholinesterase Inhibitors: Novel Cholinergic Treatments for Alzheimer’s Disease. Curr. Alzheimer Res. 2016, 13, 1. [Google Scholar] [CrossRef]
- Sun, J.L.; Stokoe, S.A.; Roberts, J.P.; Sathler, M.F.; Nip, K.A.; Shou, J.; Ko, K.; Tsunoda, S.; Kim, S. Co-activation of selective nicotinic acetylcholine receptors is required to reverse beta amyloid-induced Ca2+ hyperexcitation. Neurobiol. Aging 2019, 84, 166–177. [Google Scholar] [CrossRef]
- Thomsen, M.S.; Arvaniti, M.; Jensen, M.M.; Shulepko, M.A.; Dolgikh, D.A.; Pinborg, L.H.; Hartig, W.; Lyukmanova, E.N.; Mikkelsen, J.D. Lynx1 and Abeta1-42 bind competitively to multiple nicotinic acetylcholine receptor subtypes. Neurobiol. Aging 2016, 46, 13–21. [Google Scholar] [CrossRef] [Green Version]
- Hellström-Lindahl, E.; Gorbounova, O.; Seiger, Å.; Mousavi, M.; Nordberg, A. Regional distribution of nicotinic receptors during prenatal development of human brain and spinal cord. Dev. Brain Res. 1998, 108, 147–160. [Google Scholar] [CrossRef]
- Miwa, J.; Freedman, R.; Lester, H.A. Neural systems governed by nicotinic acetylcholine receptors: Emerging hypotheses. Neuron 2011, 70, 20–33. [Google Scholar] [CrossRef] [Green Version]
- Lozada, A.F.; Wang, X.; Gounko, N.; Massey, K.A.; Duan, J.; Liu, Z.; Berg, D.K. Glutamatergic synapse formation is promoted by α7-containing nicotinic acetylcholine receptors. J. Neurosci. 2012, 32, 7651–7661. [Google Scholar] [CrossRef] [Green Version]
- Faro, L.R.F.; Tak-Kim, H.; Alfonso, M.; Duran, R. Clothianidin, a neonicotinoid insecticide, activates alpha4beta2, alpha7 and muscarinic receptors to induce in vivo dopamine release from rat striatum. Toxicology 2019, 426, 152285. [Google Scholar] [CrossRef]
- Kawahata, I.; Yamakuni, T. Imidacloprid, a neonicotinoid insecticide, facilitates tyrosine hydroxylase transcription and phenylethanolamine N-methyltransferase mRNA expression to enhance catecholamine synthesis and its nicotine-evoked elevation in PC12D cells. Toxicology 2017, 394, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, K.; De Santana, M.B.; Nascimento, J.D.; Picanço-Diniz, D.; Maués, L.; Santos, S.; Ferreira, V.; Alfonso, M.; Duran, R.; Faro, L. Behavioral and biochemical effects of neonicotinoid thiamethoxam on the cholinergic system in rats. Ecotoxicol. Environ. Saf. 2010, 73, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Amaraneni, M.; Pang, J.; Mortuza, T.B.; Muralidhara, S.; Cummings, B.S.; White, C.A.; Vorhees, C.V.; Zastre, J.; Bruckner, J.V. Brain uptake of deltamethrin in rats as a function of plasma protein binding and blood–brain barrier maturation. NeuroToxicology 2017, 62, 24–29. [Google Scholar] [CrossRef]
- Mortuza, T.B.; Edwards, G.L.; White, C.A.; Patel, V.; Cummings, B.S.; Bruckner, J. Age Dependency of Blood-Brain Barrier Penetration by cis- and trans-Permethrin in the Rat. Drug Metab. Dispos. 2018, 47, 234–237. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, I.; Tomizawa, M.; Saito, T.; Miyamoto, T.; Walcott, E.C.; Sumikawa, K. Structural factors contributing to insecticidal and selective actions of neonicotinoids. Arch. Insect Biochem. Physiol. 1998, 37, 24–32. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Houchat, J.-N.; Cartereau, A.; Le Mauff, A.; Taillebois, E.; Thany, S.H. An Overview on the Effect of Neonicotinoid Insecticides on Mammalian Cholinergic Functions through the Activation of Neuronal Nicotinic Acetylcholine Receptors. Int. J. Environ. Res. Public Health 2020, 17, 3222. https://doi.org/10.3390/ijerph17093222
Houchat J-N, Cartereau A, Le Mauff A, Taillebois E, Thany SH. An Overview on the Effect of Neonicotinoid Insecticides on Mammalian Cholinergic Functions through the Activation of Neuronal Nicotinic Acetylcholine Receptors. International Journal of Environmental Research and Public Health. 2020; 17(9):3222. https://doi.org/10.3390/ijerph17093222
Chicago/Turabian StyleHouchat, Jean-Noël, Alison Cartereau, Anaïs Le Mauff, Emiliane Taillebois, and Steeve H. Thany. 2020. "An Overview on the Effect of Neonicotinoid Insecticides on Mammalian Cholinergic Functions through the Activation of Neuronal Nicotinic Acetylcholine Receptors" International Journal of Environmental Research and Public Health 17, no. 9: 3222. https://doi.org/10.3390/ijerph17093222




