1. Introduction
Mining tailings are residual suspensions that come from froth flotation stages and are made up of non-valuable minerals, such as silicates and phyllosilicates. These pulps have water contents close to 70 wt%; therefore, they must be thickened to recover part of the water resource and produce a thickened pulp that can be safely stored. For this, the pulp is fed to the feed-well of thickeners, where it is mixed with chemical reagents, especially soluble polymers of high molecular that can adhere to several particles simultaneously, forming aggregates that settle by gravity [
1]. The settling rate is a parameter of most interest to process engineers, since it is directly related to the amount of water recovered at this stage [
2]. The phenomena that determine the efficiency of particle flocculation depends on the physicochemical properties of the flocculant (molecular weight, structure, functionality) and its operational handling (points of dosage, dilution, dosage), water chemistry (pH, salinity), thickening technology (e.g., thickener type, feed well), and mineralogical characteristics (e.g., fines ore, clays) [
3,
4].
A particular concern for operators is dealing with clays that are frequently associated with valuable minerals (for example, copper ores). These particles have a complex structure and affect practically all the unit stages of mineral processing, including flocculation and sedimentation phenomena [
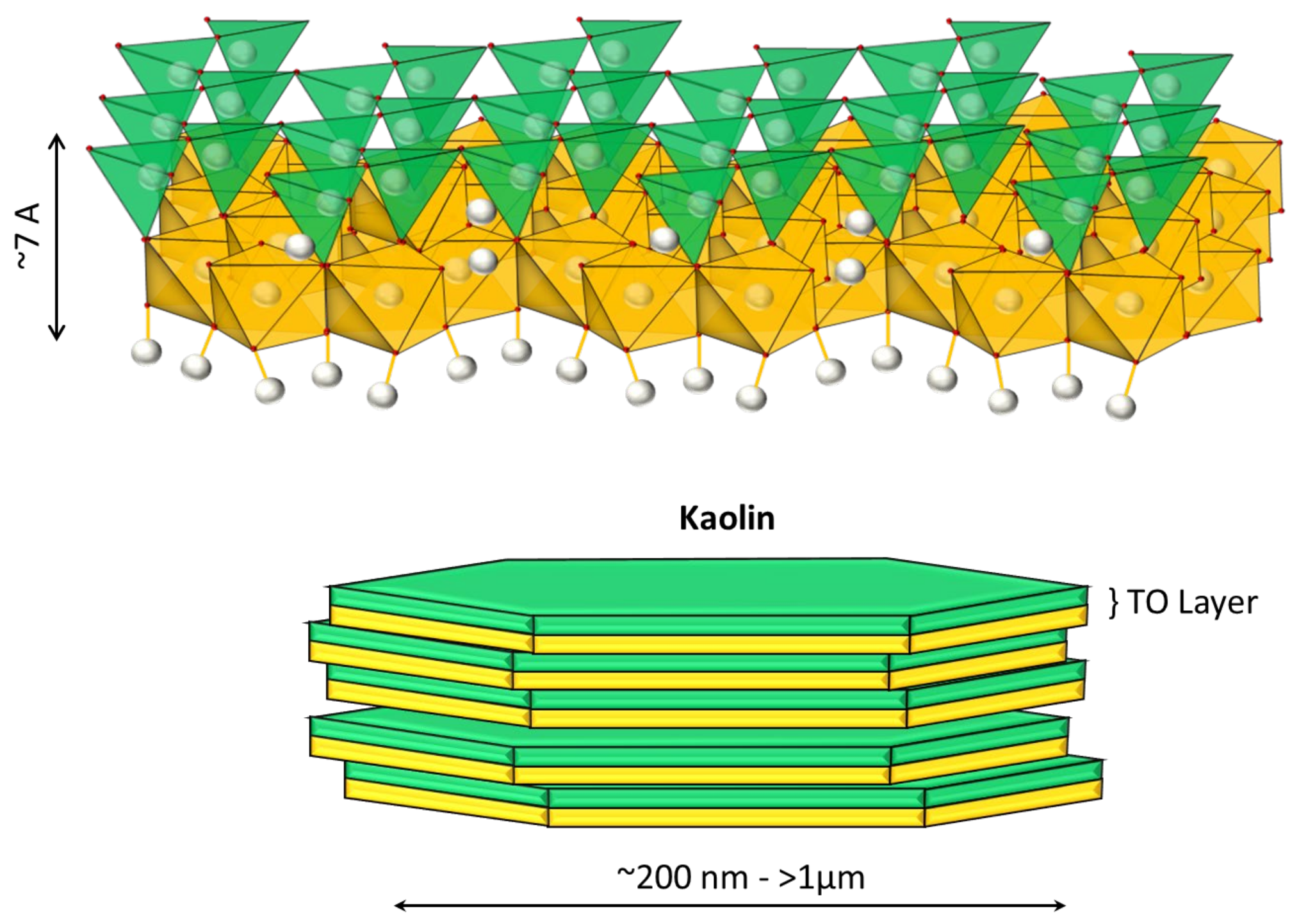
5]. Two phyllosilicates of interest in mining are kaolinite and Na-montmorillonite. Kaolinite of the composition Al
2Si
2O
5(OH)
4 is a non-swellable clay in freshwater, whose structure is composed of an octahedral sheet of aluminum hydroxide and a tetrahedral sheet of silica joined to form a basic unit of 1:1 repeat (see
Figure 1). This clay has two surfaces that are crystallographically different: the faces, which have an anionic charge, and the edges, which vary their charge (anionic or cationic) depending on the pH, product of the protonation or deprotonation of the aluminol groups (Al-OH), and silanol (Si-OH) in the exposed planes with hydroxyl termination [
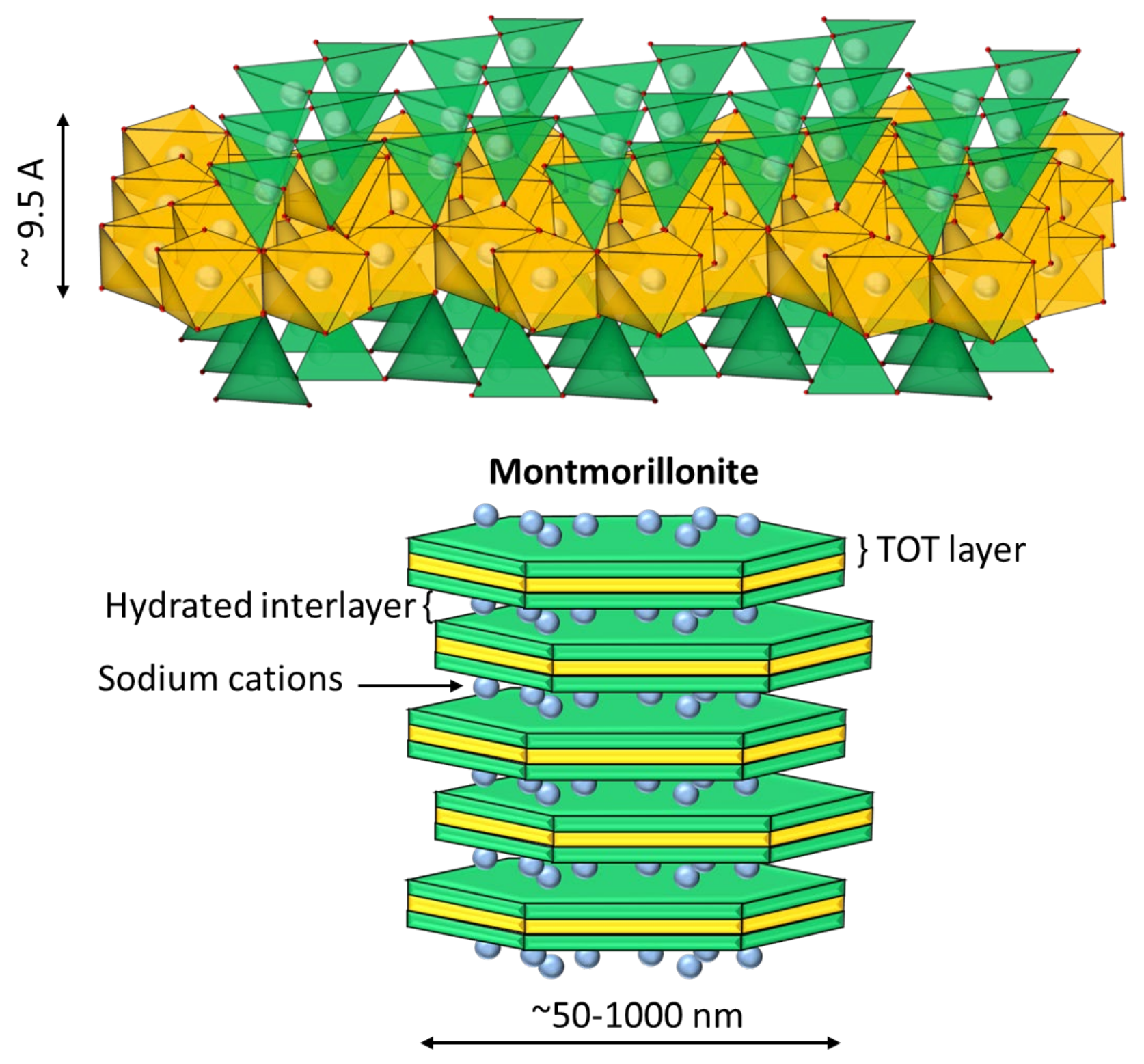
6]. On the other hand, Na-montmorillonite is swellable in freshwater. Its complex structure consists of an octahedral sheet of alumina and two tetrahedral sheets of silica, which are joined to form a basic unit of repeating layers in a 2:1 ratio (see
Figure 2) [
7]. The central layer contains octahedrally coordinated Al and Mg in oxides and hydroxides, and is surrounded by two outer layers formed by tetrahedrally coordinated silicon oxides. In this structure, the water molecules enter the middle of the layers and cause swelling and modification in the mineral structure [
8]. This clay has high chemical stability, a large surface area, and a high ion-exchange capacity, due to its weakly bound octahedral sheets. The increased swelling in fresh water makes sedimentation slow due to increased volumetric concentration. The swelling effect of sodium montmorillonite is reduced in a saline medium [
9]. Part of the cations present in the solution migrate towards the interior of the particles, being located inside the inter-laminar layers, which have an anionic charge. The subsequent reduction in electrostatic repulsion reduces the separation distance between sheets, which prevents the entry of water molecules into the clay [
10].
The particles’ electrical charge can be represented by the zeta potential, which indicates the degree of electrostatic repulsion between two surfaces. A high zeta potential value confers stability to the system, with particles that resist agglomeration. However, attractive forces can overcome repulsion when the magnitude of zeta potential is lowered and the pulp becomes unstable with flocculated particles. Empirical evidence has shown that the zeta potential of kaolin and montmorillonite is strongly influenced by the pH and salinity of the medium. In general, raising the salinity leads to a reduction in the zeta potential. This is due to the electrostatic shielding of the ions that compresses the ionic cloud surrounding the particles’ surface. On the other hand, the pH modifies the functional groups that are arranged on the particle surface, causing states in which there are no electrical charges (point of zero charge), as happens with kaolinite at a pH close to 3 [
6]. However, at copper sulfide pHs relevant for the mining industry (pH > 7), zeta potential measurements reflect that the surface’s charge is anionic; therefore, the stability of the particles prevents their agglomeration with slow sedimentation. In this sense, it is strictly necessary to add chemical reagents that favor the formation of large agglomerates with fast-settling structures.
The presence of cations can promote flocculation by forming extra bonds between the anionic polyelectrolyte and the anionic faces of quartz and/or clay minerals. This is at pH conditions typical in the mineral processing of copper ore, such as chalcopyrite (pH above 7). The cationic bridges improve the adsorption density of the reagent. However, high salinity reduces the radius of gyration of polyelectrolytes, since cations shield the electrical charge of anionic functional groups that keep molecules in solution extended, limiting their ability to form polymeric bridges [
11]. The competition of these phenomena can generate different results in flocculation, depending on the environment’s conditions. This could be beneficial or harmful. For example, Jeldres et al. [
12] studied the impact of sodium chloride on the yielding and viscoelastic behavior of flocculated kaolinite slurries at natural pH. The authors obtained optimal flocculation at 0.001 M NaCl, which generated a higher settling rate and yield stress. Castillo et al. [
13] used chemometric methods to analyze the impact of solids content, flocculant dose, shear rate, and kaolinite content on sedimentation of mining tailings and clarified water turbidity. Their results showed that the most critical variables to define turbidity were shear rate and flocculant dosage. At the same time, clay tailings were mainly influenced by solids concentration and shear rate during mixing. The lowest values of sedimentation rate were presented in saline media, which was explained by the coiling of the flocculant in the solution. Gumaste et al. [
14] investigated two types of electrolytes, NaCl and SrCl
2, in the sedimentation of sodium bentonite (90% Na-montmorillonite). The authors obtained an increase in suspension dehydration by a factor of 87 when the ionic strength of the sedimentation fluid increased from 0.1 to 250 mM. Similarly, Shaikh et al. [
15] showed that electrolytes and their valence improve the sedimentation of swelling clays. Liu et al. [
16] studied the kaolinite, bentonite, and illite sedimentation in saline media, using a negatively charged copolymer of acrylamide and sodium acrylate. The results showed increased salinity reduced kaolinite sedimentation, but the salinity slightly accelerated the settling rate for montmorillonite and illite. Ji et al. [
17] investigated the sedimentation of clay-rich coal tailings in saline solutions in the presence of three flocculants: polyacrylamide (PAM), Magnafloc 1011 (acrylamide-acrylate copolymer), and Al (OH) 3-PAM. The Magnafloc 1011 exhibited the best sedimentation performance in saline water. The reduction in electrostatic repulsion between the negatively charged particles and anionic flocculants led to more effective polymer-bridging interactions.
To date, few studies address the flocculation of clay-based tailings with relevant systems for the mining industry. Some works that introduce microscopic characterizations stand out, directly investigating properties of the aggregates, such as size and fractal dimension. Leiva et al. [
18] analyzed the temporal evolution of the aggregate structure of mining tailings with the presence of kaolin. The authors flocculated the mineral with a high molecular weight anionic polyacrylamide in seawater. Under more intense mixing and a prolonged flocculation period, the apparent decline in Df was concluded to reflect an increased contribution from the presence of aggregate fragments, i.e., broken-off branches of aggregates with a high-aspect ratio and a low effective porosity. In situ imaging of the aggregates at different reaction times supported this. Ramos et al. [
19] analyzed the effect of pH on the flocculation of synthetic tailings, finding that at highly alkaline conditions (pH> 10.3), the efficiency of the process drops drastically due to the precipitation of magnesium ions. Later, Jeldres et al. [
20] proposed to remove magnesium cations from seawater using a mixture of alkalizing agents, which significantly improved settling rates.
Currently, no systematic reports analyze the behavior of swelling clays, especially comparing the type of water, when these present low and high ionic charges. This research aims to analyze the structural properties of clay tailings aggregates, comparing systems with kaolin and sodium montmorillonite contents. The size distribution of the aggregates was obtained using the Focused Beam Reflectance Measurement (FBRM) technique, which obtained structural properties of aggregates, such as fractal dimension and density. This was developed by relating the size of the aggregates to the hindered settling rate [
21]. The numerical outcomes for floc structure were supported by imaging obtained with the Particle Vision and Measurement (PVM) probe. The study was conducted at pH 8, considering that this is a typical pH used in the copper mining industries that use seawater in their operations. The present results are helpful for the current knowledge on clay tailings flocculation, especially with complex, swelling clays. The scope of the study involves industries that use both freshwater and seawater in their operations.
2. Materials and Methods
2.1. Materials
The quartz particles were purchased from Donde Capo (Santiago, Chile). Firstly, 100% of the particles used were sieved under a # 270 mesh (ASTM standard). The quartz density was 2600 kg/m
3. According to X-ray diffraction (XRD) with TOPAS software, the quartz content (Q) was higher than 99% by weight. Kaolin (K) and montmorillonite (M) particles were purchased from Ward’s Science Clay Spur, WY, USA.
Figure 3 shows the chord-length distributions in unweighted (offering greater sensitivity in the range of fines) and square-weighted (influenced by the contribution of the aggregates of coarse particles) modes for kaolin (
Figure 3a) and montmorillonite (
Figure 3b). These data for seawater (SW) and industrial water (IW) were obtained through the FBRM technique. The change in water quality (from IW to SW) meant a shift to the right in all chord-length distributions (CLD) and growth in the maximum height weighted quadratically. This indicates that SW presents fewer fine particles (<20 µm) and more numerous aggregates between (20–90 µm) and (20–140 µm) for K and M suspensions, respectively. An increase in the salt concentration favors the coagulation of the particles, since the presence of cations reduces the ionic cloud surrounding the particles’ surface [
22].
Both clays had a density of 2600 kg/m
3. The mineralogical composition was determined by X-ray diffraction (XRD) using a Bruker brand X-ray diffractometer model D8 ADVANCE. The wavelength λ (Cukα) was 1.5406 Å. The 2θ angle studied was from 5° to 80°. The presence of mineralogical components was presented using the Powder Diffraction File of ICDD (International Center for Diffraction Data). The diffractogram shown in
Figure 4a confirms the existence of montmorillonite, quartz, and albite minerals, while the diffractogram presented in
Figure 4b confirms the presence of kaolinite, quartz, and albite minerals.
An anionic polyacrylamide of high molecular weight, provided by SNF Chile S.A., was used as a flocculant (SNF 704). This reagent has a molecular weight of 18 × 106. A stock solution of the solid flocculant was prepared by continuous stirring for 24 h at a concentration of 1 g/L. Subsequently, a part of the stock solution was diluted to a 0.1 g/L for settling tests and flocculation kinetics. The flocculant doses were expressed as grams of flocculant per tonne of dry solids (g/t).
Seawater (SW) and industrial water (IW) were synthetically prepared.
Table 1 shows the composition of SW. The IW was prepared by adding 0.005 M of CaCl
2, 0.01 M of NaCl, and 25 g/t of Al
2(SO
4)
3 as a coagulant. Both water qualities’ basic physical and chemical properties were determined using a HANNA H19829 multiparameter and an Anton Paar DMA 35 portable density (see
Table 2). Analytical grade sodium hydroxide (NaOH) (greater than 98%) was used as a pH regulator. All chemical reagents were of ultra-high purity (Merck, Darmstadt, Germany).
2.2. Flocculation of Clay-Based Tailings
Synthetic tailings were prepared using 300 g of the kaolin-quartz (KQ) and montmorillonite-quartz (MQ) mixture in SW and IW, with a total solids concentration of 8 wt% (mixture of 90 wt% quartz and 10 wt% clay). These colloidal suspensions were prepared in a flocculating vessel, with a conical end of 1 L capacity and 100 mm in diameter, with a valve in the lower part to pour the suspensions into test tubes. The suspensions were vigorously mixed for 15 min with the aid of an up-flow polytetrafluoroethylene (PTFE) blade located 20 mm above the bottom of the container, and with a control IKA® EuroStar 60 digital stirrer. Then the agitation rate was decreased to 180 rpm, and the flocculant dose of SNF 704 was added to the flocculating cup. The suspensions were prepared to develop batch sedimentation tests and flocculation kinetics.
The batch sedimentation tests were carried out after mixing the suspension with the flocculant for 20 s. This was conducted in a device constituted by a closed cylindrical column of 1000 mL volume with an internal diameter of 35 mm. The suspensions tested involved various doses of flocculant (8–34 g/t). After homogenization, the suspensions were immediately set to settle. After this, the suspension concentration was homogenized, and the initial settling rate was measured using a video camera and subsequent analysis of videos.
The particle Track G400 obtained the flocculation kinetics with FBRM technology from Mettler Toledo. The FBRM is a standard measurement technique used to measure and monitor aggregate formation and evolution in real time. This method makes it possible to analyze the mechanisms of growth, flocculation, breakage, and change in the shape of flocs through chord-length distribution conditioned by the particles’ concentration, shape, and size. The FBRM consists of a processing unit and a probe with a rotating lens that provides a laser beam focused on the external surface of a sapphire window (14 mm in diameter), responsible for scanning a circular path at a fixed speed (2 m/s for the experiments performed). When the laser beam encounters suspended solids, it produces a pulse of reflected light back to the probe window, inducing a signal peak until the particle leaves the beam’s path. The product of the tangential velocity of the spinning laser (2 m/s) and peak duration is defined as chord length, which can also be defined as an intercepted segment (or length) of a particle.
The FBRM probe was introduced vertically into the flocculator, 10 mm above the PTFE shaker and 20 mm off-axis. The suspensions tested contained a flocculant dose of 34 g/t. The total measurement time before and during flocculation corresponded to 5 min.
The images of the aggregates over time were also recorded at a flocculant dose of 34 g/t in a PVM probe (particle vision measurement) model V819 Mettler-Toledo, which has an outer diameter of 19 mm and a 14 mm diameter sapphire window, which registers the images on a scale of 1075 × 825 µm under a resolution of 2 µm. This probe can acquire up to ten images per second.
2.3. Fractal Dimension and Density of the Aggregates
The structure of the aggregates can be described by their fractal dimension [
23,
24]. This is when the total volume of an aggregate, simplified as a sphere (
), increases with the cube of its diameter, while the mass (
) increases at a lower fractal power (see Equation (1)), where
is the mass-length fractal dimension and takes a value between 1 and 3, considering 1 as a one-dimensional line and 3 as a solid sphere.
The fractal dimension
was calculated from the mathematical relationship used by Heath et al. [
21], where the diameter of the aggregate and the hindered sedimentation are related. Chord length is used as an indicator of diameter, valid within the size range considered in the present study [
25]. The relationship is expressed in Equation (2):
where
is the hindered settling rate
;
and
are the aggregate and particle diameters [m], respectively (approximated by the squared-weighted mean chord length),
is the acceleration of gravity
,
and
correspond to the density of solid and liquid, respectively
,
is the viscosity of the fluid
,
is the volumetric fraction of solids
, and
the fractal dimension.
The parameters
,
,
,
,
,
are constant and known for all experiments. The diameter of the aggregate
and
depend on the hydrodynamic conditions applied to the system. The hindered sedimentation rate is determined experimentally after 20 s of flocculation, as explained in
Section 2.2. The
is provided by the mean squared weighted chord length obtained through the FBRM probe. Finally, the estimate
is obtained from Equation (2) using least squares.
Obtaining
allows estimation of the density of the aggregates from the Kranenburg derivation [
26], Equation (3):
where
,
and
are the aggregate, solid, and liquid densities, respectively [kg/m
3],
and
are the aggregate and particle diameters, respectively
that approximate the squared-weighted mean chord length obtained with the FBRM probe, and
is the fractal dimension obtained from Equation (2).
4. Conclusions
The present research examined the phenomena of the flocculation and sedimentation of clay-based tailings, considering two types of clays: kaolin and montmorillonite. The assays were performed in two kinds of waters, prepared synthetically: seawater and industrial water.
The effect of the type of water on flocculation differs between tailings. In the first case, for quartz/kaolin, the highest performance was obtained in industrial water, with more significant aggregates and settling rates. The high salinity of the seawater rolled up the flocculant, reducing its ability to bridge particles, but at the same time, led to more compact aggregates and higher density. This was promoted by the differences in the adsorption modes of the flocculant on the mineral surfaces. Loop configurations are preferred in industrial water, while train configurations are favored in a highly saline medium like seawater.
On the other hand, the tailings composed of quartz and montmorillonite reversed this trend with respect to the type of water. The best flocculation was produced in seawater, although the flocculant was coiled. The swelling nature of this clay in industrial water harms flocculation, and high flocculant doses are required to promote particle aggregation. The high cation concentrations reduced the swelling phenomena, with a substantial improvement in the aggregation by the polymeric bridging.
The type of water impacted the efficiency of primary particles aggregation. The trials in industrial water generated a portion of non-flocculated particles, independent of the type of tailings observed through a bimodal distribution in the unweighted chord-length distribution. At the same time, by making the studies in seawater, there were no perceptible fractions of non-flocculated particles. The formation of additional bonds due to the increased cation concentration favored the agglomeration of finer primary particles.