Abstract
The utilization of nanotechnology has brought about notable advancements in the field of pediatric medicine, providing novel approaches for drug delivery, disease diagnosis, and tissue engineering. Nanotechnology involves the manipulation of materials at the nanoscale, resulting in improved drug effectiveness and decreased toxicity. Numerous nanosystems, including nanoparticles, nanocapsules, and nanotubes, have been explored for their therapeutic potential in addressing pediatric diseases such as HIV, leukemia, and neuroblastoma. Nanotechnology has also shown promise in enhancing disease diagnosis accuracy, drug availability, and overcoming the blood–brain barrier obstacle in treating medulloblastoma. It is important to acknowledge that while nanotechnology offers significant opportunities, there are inherent risks and limitations associated with the use of nanoparticles. This review provides a comprehensive summary of the existing literature on nanotechnology in pediatric medicine, highlighting its potential to revolutionize pediatric healthcare while also recognizing the challenges and limitations that need to be addressed.
1. Introduction
In the field of pediatric oncology, nanotechnology has emerged as a tool with significant potential to advance cancer treatment. It provides several advantages, including targeted drug delivery, reduced toxicity, and combined immunotherapy. These features offer promising benefits in the treatment of specific pediatric tumors such as neuroblastoma, retinoblastoma, CNS tumors, and musculoskeletal tumors [1,2]. Nanotechnology-based approaches, including tailored nanocarriers and liposomes, have shown promise in targeted drug delivery with reduced toxicity for pediatric cancers such as acute lymphoblastic leukemia (ALL) and acute myeloid leukemia [3,4,5,6]. Nanovesicles, peptide-functionalized liposomes, and tumor vascular-targeting liposomes have demonstrated effectiveness in neuroblastoma treatment [7,8]. Furthermore, nanomedicines, nanoparticle-based drug delivery systems, and nanotechnological-based miRNA interventions hold promise for addressing neuroblastoma [9,10,11,12,13,14]. Nanotechnology also shows potential in improving outcomes for osteosarcoma treatment through alpha-particle therapy, exosome mimetics, nanocarriers, and targeted drug delivery systems [15,16,17,18,19,20,21,22].
Nanotechnology extends its potential beyond cancer treatment, as it holds promise in pediatric infectious disease management. Nanomedicines enable targeted drug delivery for malaria treatment and leishmaniasis, reducing toxicity while maintaining efficacy [23,24,25]. Nanoparticles have been utilized in bioassays for detecting and controlling schistosomiasis [26]. Additionally, nanocarriers combat antibiotic resistance and enhance the performance of drugs in infectious diseases [27,28]. Nanofabricated biosensors show high sensitivity in detecting bacterial infections, contributing to innovative approaches in combatting pediatric infectious diseases [29]. Furthermore, nanotechnology has contributed in the diagnosis and treatment of tuberculosis (TB) and human immunodeficiency virus (HIV) infections, improving targeted drug delivery, diagnostics, and treatment outcomes [30,31,32,33,34,35,36,37,38,39].
Respiratory and pulmonary diseases also benefit from nanotechnology advancements. Nanoparticle-based technologies have demonstrated effectiveness in preventing biofilm formation and infection in ventilator-associated pneumonia (VAP) [40,41]. Nanotherapeutic approaches show potential in detecting and treating Respiratory Syncytial Virus (RSV) [42,43]. Nanotechnology has improved diagnostic methods for cystic fibrosis and offered pain management solutions [44,45]. These advancements offer opportunities to enhance disease diagnosis, treatment, and patient outcomes in respiratory and pulmonary diseases.
Nanotechnology plays a vital role in addressing critical issues in pediatric environmental health and infectious diseases. It has proven effective in detecting water-borne parasites and pathogens, providing solutions for public health challenges [46,47,48,49]. Nanotechnology offers potential strategies for addressing scorpion envenomation and controlling viral infections [50,51,52]. Coordinated efforts are needed to leverage nanotechnology’s potential in improving public health outcomes and addressing environmental health issues.
In the field of pediatric medicine, nanotechnology offers innovative solutions for the diagnosis and treatment of various conditions. It has shown promise in epilepsy, expanded newborn screening, cardiovascular diseases, neuroinflammation, neurodegenerative diseases, gestational diabetes, bone disorders, mosquito-borne diseases, micronutrient deficiency, vulvovaginitis, and more [53,54,55,56,57,58,59,60,61,62,63,64]. Nanotechnology holds potential for tissue engineering, personalized nanomedicine, scoliosis, respiratory tract disorders, neurosensory diseases, and infections [65,66,67,68,69,70,71,72,73,74,75,76,77].
The impact of nanotechnology extends to various branches of pediatric medicine. In pediatric dentistry, nanotechnology offers promising solutions, particularly in the treatment of occlusal cavities. Nanoparticles incorporated into resin coatings improve wear resistance, prolonging the lifespan of dental restorations [78]. Nanovectors delivering resveratrol oral sprays reduce plaque formation and inflammation, promoting oral health [79]. Furthermore, polysaccharide-based systems offer biocompatibility and drug delivery potential, enhancing treatment outcomes in pediatric dental care [80]. The incorporation of silver nanoparticles in dental sealants creates antibacterial and rechargeable sealants, preventing the onset of dental caries [81]. Additionally, biodegradable magnesium-alloy stents effectively manage pediatric airway obstruction, providing a promising solution for respiratory conditions [82]. While the benefits of nanotechnology in pediatric dentistry are evident, further research is needed to fully understand its benefits and drawbacks [83].
Pediatric dermatology also benefits from the application of nanotechnology, particularly in the management of atopic dermatitis. Nanocarriers improve drug delivery by enhancing solubility and skin permeation, reducing side effects associated with topical treatments [84]. Chitosan nanoparticles, for instance, enhance drug penetration, leading to improved therapeutic outcomes in atopic dermatitis [85,86]. Additionally, polydopamine nanoparticles have shown the ability to inhibit fibrosis in neonatal scleredema, providing new avenues for the treatment of this condition [87]. Nanotechnological carriers hold promise for improving the efficacy and safety of treatments for various pediatric skin disorders, addressing a significant unmet need in pediatric dermatology [84].
Another crucial area where nanotechnology holds potential is pediatric nutrition. It offers innovative solutions for addressing critical issues such as obesity, nutritional deficiencies, and food allergies in pediatric populations [88]. Nanotechnology-based food production can provide more nutritious and low-calorie options, contributing to improved pediatric dietary habits [89]. Furthermore, iron solid lipid nanoparticles offer an alternative to conventional iron supplements, enhancing the bioavailability of this essential nutrient [90]. Nanotechnology-based optical biosensors enhance food safety by enabling the rapid and accurate detection of harmful contaminants [91]. Additionally, nanotechnology-based diagnostics aid in personalized allergen immunotherapy, ensuring safer and more effective management of food allergies in children [92]. Collaborative efforts are crucial for advancing research and ensuring the safety and efficacy of nanotechnology in the field of pediatric nutrition [88].
One area where nanotechnology has made significant contributions is pediatric drug delivery. It has addressed key challenges such as solubility, taste, and stability, improving the effectiveness of drug therapies in children. Biomimetic nanovesicles incorporated into transdermal patches have been developed to enhance the delivery of micronutrients [93]. In situ self-assembly nanoparticles improve the oral delivery of solid dosage forms, increasing drug bioavailability and therapeutic efficacy [94,95]. Folic acid magnetic nanotheranostics have been developed to reduce cardiotoxicity and enhance targeted drug delivery [96]. Nanoparticle-based systems offer ease of administration and enhanced drug delivery across various routes [97]. Nanofibers and nanocapsules provide effective drug delivery approaches, improving the therapeutic outcomes of pediatric medications [98,99,100]. Nanopatch technology offers a needle-free and painless approach to vaccine delivery—particularly relevant for pediatric immunization [101]. Nanocosmeceuticals benefit from nanoformulations, enabling targeted delivery of skincare ingredients [102].
The integration of nanotechnology in pediatric medicine has ushered in a new era of possibilities. However, it is essential to acknowledge that, along with its tremendous potential, nanotechnology also raises concerns regarding potential health risks. Researchers have highlighted the impact of engineered nanoparticles on children’s health, emphasizing the need for thorough investigations into their safety profiles [103]. Studies have specifically examined the neurotoxicity of nanoparticles, shedding light on the importance of understanding their potential risks [104,105]. Carbon nanoparticles and ultrafine particles are areas that require further exploration to determine their impact on pediatric health [106,107,108].
It is crucial to approach nanotechnology with a cautious and responsible mindset. The unique properties of nanoparticles offer biomedical possibilities, but their safe and responsible use must be prioritized [108]. Public understanding of nanotechnology is also paramount, as it empowers individuals to make informed decisions and fosters trust in its applications. Education and awareness campaigns should be implemented to disseminate accurate information about nanotechnology, addressing both its potential benefits and risks. This paper aims to provide an overview of the advances in nanosystems and their potential applications in major pediatric disorders.
1.1. Pediatric Diseases
The pediatric population, i.e., children, are at high risk of various diseases and disorders such as malaria [109], iron deficiency [110], traumatic brain injury [111], pediatric cancer [112], respiratory syncytial virus [113], and inflammatory bowel disease [114], to name a few. Malaria is a parasitic disease that affects many children in developing countries, while iron deficiency anemia can lead to fatigue, weakness, and developmental delays. Traumatic brain injury is a significant cause of cognitive, emotional, and behavioral problems, and pediatric cancer can be challenging to treat in children. Respiratory syncytial virus can cause severe respiratory illness in infants and young children, and inflammatory bowel disease causes chronic inflammation in the digestive tract, leading to abdominal pain and diarrhea [113,114].
Other diseases and disorders that affect children include dental biofilm and gingival inflammation, vulvovaginitis, diffuse intrinsic pontine gliomas, neuroblastoma, acute myeloid leukemia, HIV, osteosarcoma chemotherapy, craniosynostosis, retinoblastoma, hereditary angioedema, epilepsy, neurodegenerative diseases, asthma, and liver diseases such as biliary atresia and hepatitis. Vaccines are essential for preventing infectious diseases and protecting the health of children, and routine childhood immunization schedules include vaccines against various diseases such as measles, mumps, rubella, and polio. Effective management and early diagnosis of these diseases are crucial for improving quality of life among children and reducing morbidity and mortality.
1.2. Nanosystems
Nanomedicines, a specific class of nanocarriers, have significantly advanced the field of medicine by allowing the targeted and efficient delivery of drugs, imaging agents, and genes to specific cells or tissues in the body [115]. Liposomes and polymeric nanoparticles are examples of nanocarriers that can encapsulate drugs and release them in response to specific triggers, offering controlled and targeted drug delivery [116]. Mesoporous silica nanoparticles, with their high surface area and pore volume, are well-suited for drug delivery, imaging, and biosensing applications [117]. Gold nanoparticles and iron oxide nanoparticles have also been employed for targeted drug delivery and imaging purposes [118]. Biodegradable and CO2-derivative cationic polymeric nanoparticles are emerging as promising nanocarriers in drug delivery due to their biocompatibility, biodegradability, and the ease with which their surface charge can be modified for efficient cellular uptake and targeted drug delivery. Lipid-based nanoparticles, such as solid lipid nanoparticles and nanostructured lipid carriers, possess unique properties that make them ideal for drug delivery and imaging applications. Composite scaffolds, which combine nanoparticles with natural or synthetic polymers, have found applications in tissue engineering and regenerative medicine, enabling the repair and regeneration of damaged tissues and organs [119]. Magnetic nanotheranostics are gaining prominence in the detection and treatment of various diseases, including cancer, cardiovascular diseases, and neurodegenerative diseases [120]. The nanopatch is a novel nanocarrier designed for transdermal drug delivery, providing a painless and convenient alternative to traditional injections [121]. Graphene and its derivatives, such as graphene oxide and reduced graphene oxide, possess unique mechanical, electrical, and optical properties that make them suitable for diverse biomedical applications, including drug delivery, imaging, and biosensing [122].
2. Pediatric Cancer Treatment and Research
2.1. Pediatric Cancers in General
Considerable advancements have been achieved in pediatric oncology, and nanotechnology has emerged as a valuable asset in the fight against cancer. The application of customized nanocarriers for drug delivery has demonstrated promising advantages when treating certain pediatric tumors such as neuroblastoma, retinoblastoma, CNS tumors, and musculoskeletal tumors [1]. Another notable advancement is the use of liposomes as delivery vehicles for anticancer agents in pediatric cancer treatment. This approach has demonstrated improved treatment efficacy while reducing toxic side effects [2].
Nanotechnology-based strategies offer substantial potential for enhancing clinical outcomes in pediatric oncology. These strategies aim to reduce toxicity, achieve targeted delivery, and combine with immunotherapeutic agents. Furthermore, nanotechnology holds promise in various areas, such as prevention, diagnosis, and treatment, encompassing tumor targeting and controlled release [123]. However, the field faces a significant challenge due to the limited availability of nanomedicines for pediatric cancer care [124].
Innovative nanotechnology-based approaches show promise in treating pediatric cancers such as diffuse midline gliomas [125], leukemia [126], osteosarcoma [127], and brain cancers [128]. Nanoparticle-based delivery systems have been found to inhibit tumor cell proliferation and migration in cholesteatoma and pediatric brain tumor cells [129,130]. Nanotechnology-based drug delivery enables the specific targeting of anticancer agents to leukemic cells, thereby reducing toxic side effects [131]. The development of nanotechnology has the potential to improve therapeutic efficiency, drug targeting, reduce toxicity, and mask the bitter taste of drugs, with anticancer drugs being the most frequently encountered therapeutic drug class [132].
For the molecular diagnosis of pediatric sarcomas, NanoString technology has proven to be a reliable approach. It can detect sarcoma-specific fusion transcripts in a single reaction with 100% concordance to RT-PCR [133]. In the proteomic analysis of pediatric ependymoma using high-resolution mass spectrometry, similarities with other pediatric brain tumor entities, such as astrocytomas and medulloblastomas, have been revealed [134]. Table 1 summarizes examples of nanocarrier systems utilized in pediatric medicine.

Table 1.
Nanosystems in general pediatric cancer medicine.
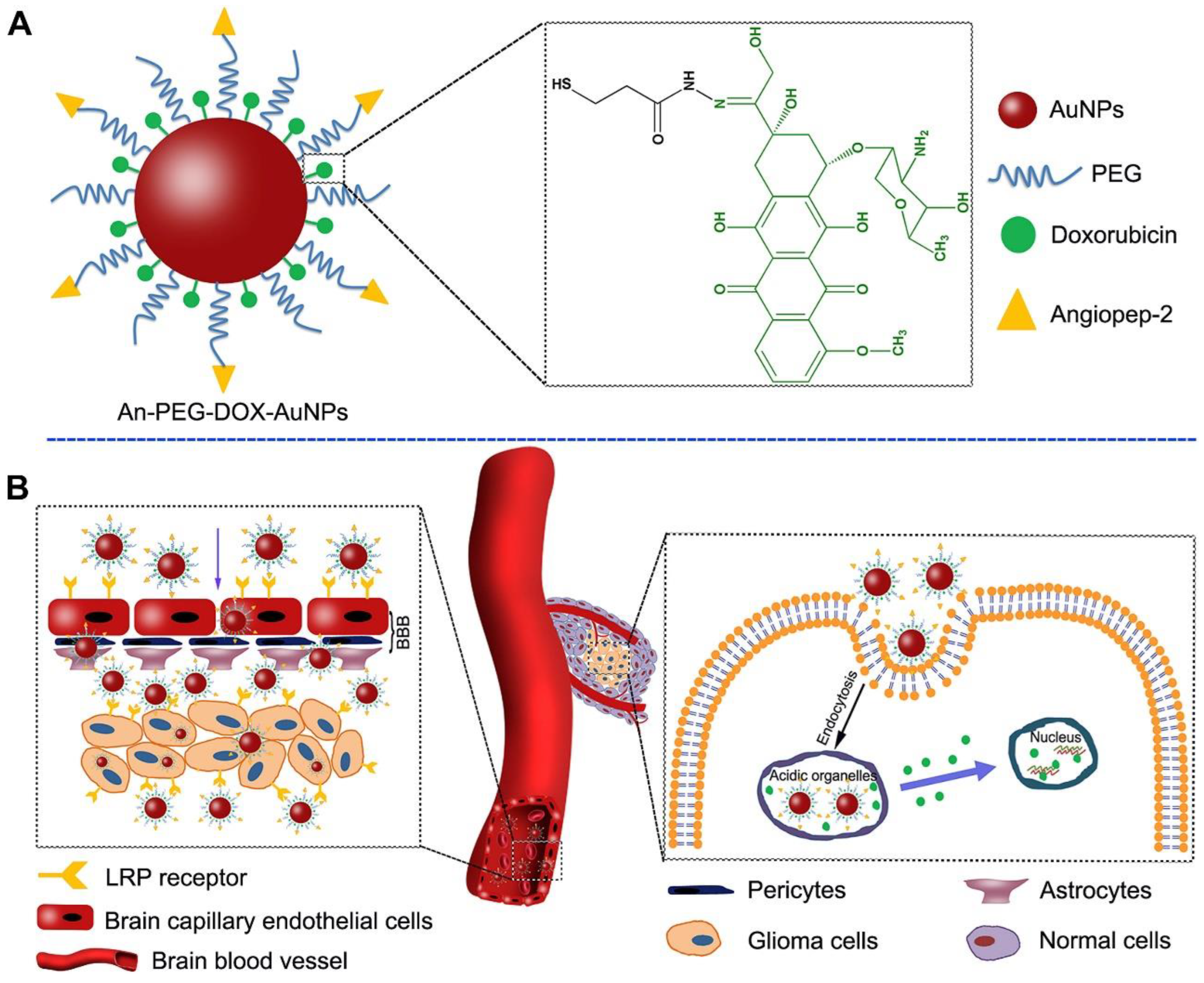
Nanotechnology has shown significant potential in addressing pediatric cancer [135]. Ongoing research in this field is expected to yield innovative and effective treatments for these devastating diseases. Figure 1 illustrates the utilization of two strategies employing gold nanoparticles for delivering doxorubicin (DOX) to gliomas. These strategies involve using Agiopeptide-2 as a targeting polymer and poly(ethylene glycol) (PEG) to evade immune recognition.
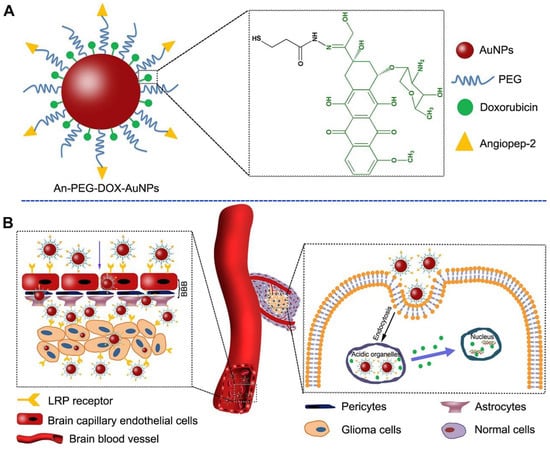
Figure 1.
(A) Elucidation of the An-PEG-DOX-AuNPs. (B) Elucidation of the delivery procedure of An-PEG-DOX-AuNPs. LRP1 receptor could mediate An-PEG-DOX-AuNPs and allow them to penetrate through BBB and target glioma cells, then DOX would be released at the tumor site or in tumor cells and enter into the nuclei to induce tumor cell apoptosis. Printed with permission from [136].
2.2. Leukemia
Numerous studies have investigated the application of nanotechnology in various types of pediatric leukemia, yielding encouraging results. For instance, the use of CHGNPs (carbon-encapsulated hollow gold nanoparticles) has been shown to selectively induce G1 cell cycle arrest by up-regulating the tumor suppressor protein P27. This advancement provides a cytotoxic drug for the clinical treatment of leukemia [3]. However, the efficacy of lipid-based cubosomal nanoformulations in treating Acute Lymphoblastic Leukemia (ALL) in children has yet to be established. This emphasizes the need for cautious consideration when utilizing nanotechnology to enhance drug efficacy [141].
Gold nanoparticle-based nanocarriers for antileukemic drugs have demonstrated potential in drug delivery, cancer diagnosis, and therapy for ALL. A comprehensive overview of conventional methods and nano-strategies for ALL treatment has highlighted the special focus on gold nanoparticle-based nanocarriers [4]. Similarly, polypeptide-based nanoparticles have shown promising outcomes in depleting CD22DeltaE12 through SiRNA-mediated treatment in B-cell Precursor Lymphoblastic Leukemia [5]. Furthermore, poly(lactide-co-glycolide) (PLGA) nanomedicines loaded with 6-mercaptopurine (6-MP) have exhibited enhanced oral bioavailability and tissue distribution. This has resulted in improved in vitro cytotoxicity of Jurkat cells and prolonged survival time in ALL model mice, offering a promising delivery strategy for clinical translation [142]. The Nessler method, employing ultraviolet-visible spectrophotometry, enables the quantification of PEGylated asparaginase activity in plasma for personalized nanomedicine in clinical settings [143]. Moreover, NanoString nCounter technology has demonstrated robust and cost-effective potential for the diagnosis of B-cell acute lymphoblastic leukemia, boasting high sensitivity and specificity [144].
Polymeric nanoparticles loaded with dexamethasone have been found to enhance therapeutic efficacy, leading to improved quality of life and survival in childhood leukemia [145]. Lastly, the use of siRNA-loaded lipid nanoparticles for LNP-si-LINC01257 treatment has proven to be a safe and effective therapeutic approach for pediatric acute myeloid leukemia [6]. While it is crucial to exercise caution when leveraging nanotechnology to enhance drug efficacy, the potential benefits are evident. The continued exploration and utilization of nanotechnology in the treatment of pediatric cancer holds promise for significant advancements in the field. Table 2 summarizes examples of nanocarrier systems utilized in leukemia treatment.

Table 2.
Nanosystems in pediatric Leukemia treatment.
2.3. Neuroblastoma
One highly promising development involves the utilization of nanovesicles coated with GASNGINAYLC peptide [7]. In vitro experiments have revealed that these nanovesicles exhibit exceptional biocompatibility and stability, making them a promising tool for actively targeted nanotherapy in the case of neuroblastoma. Additionally, studies have shown that peptide-functionalized liposomes hold great promise in enhancing tumor-homing properties, inducing tumor apoptosis, and reducing tumor glucose consumption. These unique properties make liposomal nanocarriers a valuable tool for multitargeted treatment of neuroblastoma [8].
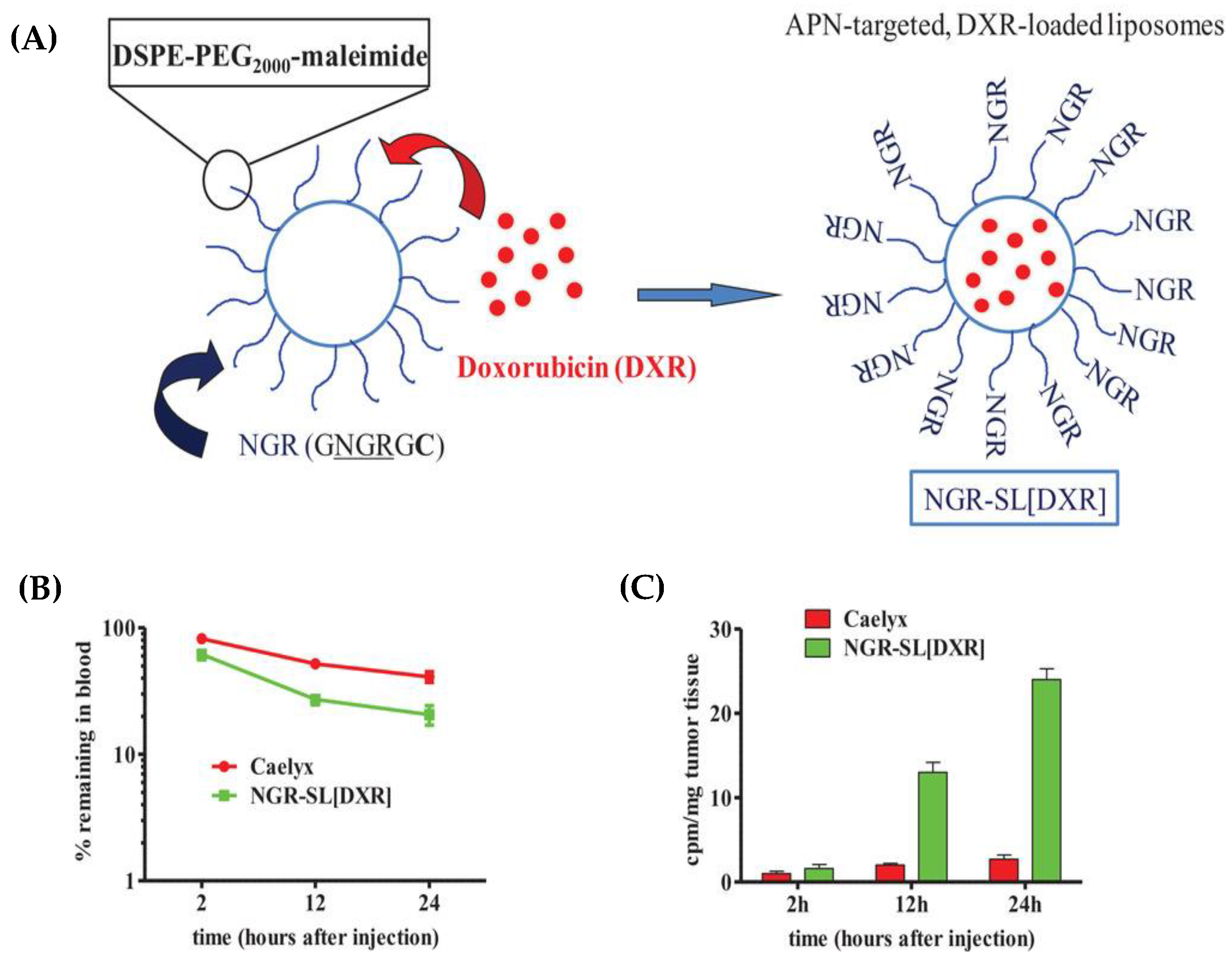
Furthermore, recent studies have showcased the development of tumor vascular-targeting liposomes, which allow for the targeted release of drugs. This approach has been successfully tested in mice, demonstrating positive results. Figure 2 visually presents the development of these liposomes and illustrates their release characteristics in mice.
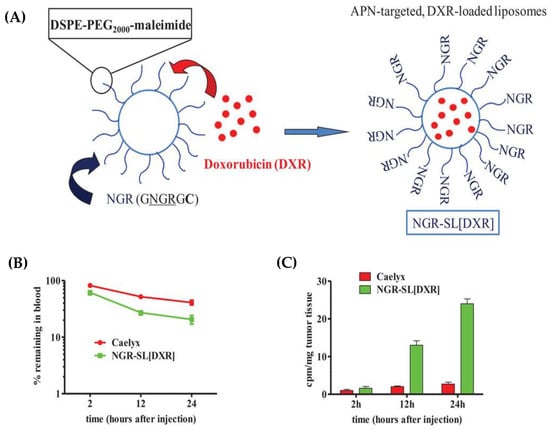
Figure 2.
Development of doxorubicin-loaded, tumor vascular-targeting liposomes. (A) Schematic representation of the NGR-containing peptide GNGRGGVRSSSRTPSDKYC (called GNGRGC)-targeted, doxorubicin-loaded stealth liposomes (NGR-SL[DXR]). In order to enable coupling of NGR-containing peptide to SL, a cysteine residue (C) was added to the peptide C-terminus. (B,C) Pharmacokinetic profiles and tumor accumulation of NGR-SL[DXR] in NB-bearing mice. Adapted with permission from [8].
These advancements in nanotechnology offer a potential avenue for the development of effective treatments for pediatric cancer, including neuroblastoma. The use of targeted nanocarriers and liposomes has shown promising results in both laboratory settings and animal studies, suggesting that there is potential for the creation of more efficient treatments for this aggressive form of cancer. Moreover, nanotechnological-based miRNA intervention has demonstrated promise in the therapeutic management of neuroblastoma, addressing challenges related to drug delivery and enhancing therapeutic success [9]. Nanomedicines, such as liposomes and doxorubicin-loaded nanocarriers targeted at nucleolin, have also displayed potential in overcoming the limitations of current diagnostic and therapeutic approaches, offering more effective and targeted options [10,11,12]. Additionally, nanoparticle-based drug delivery systems incorporating etoposide have synergized with alpha v integrin antagonists, improving patient care for high-risk neuroblastoma [13]. The co-assembly of amphiphilic antitumor agents has exhibited better antitumor profiles and controlled release behavior, representing a suitable pre-clinical candidate for childhood cancer therapy in neuroblastoma and osteosarcoma [14].
For anaplastic large cell lymphoma (ALCL), protamine nanomedicine with aptamers, dsDNA/drug payload, and siRNA has the potential to offer cell-selective chemotherapy and oncogene-specific gene therapy by targeting diagnostic biomarkers and therapeutic targets [146]. Additionally, nanomedicines and cell-based therapies are currently being investigated in phase I/II clinical trials for neuroblastoma and medulloblastoma, with the aim of reducing drug toxicity and improving efficacy [147]. Nanomedicine has demonstrated promise in overcoming the limitations of conventional chemotherapy for pediatric neuroblastoma [148]. It offers targeted drug delivery, reduces systemic side effects, and improves pharmacokinetic properties, thereby holding the potential to revolutionize the diagnosis and treatment of childhood cancer. The utilization of nanomedicines enables targeted drug delivery and improved pharmacokinetic properties, leading to a reduction in systemic side effects and the potential to revolutionize the diagnosis and treatment of childhood cancer. Further research in this field is of utmost importance to translate these promising advancements into clinical applications and ultimately improve outcomes for pediatric cancer patients. Table 3 summarizes examples of nanocarrier systems utilized in neuroblastoma treatment.

Table 3.
Nanosystems in pediatric neuroblastoma treatment.
2.4. Osteosarcoma
Osteosarcoma, a challenging form of cancer, is known for its resistance to chemotherapy and lack of effective targeted therapies. Researchers have made notable advancements in the development of diverse nanocarriers, drug delivery systems, and imaging agents. These innovations aim to improve the effectiveness of treatments while minimizing potential side effects. One particularly promising treatment approach involves alpha-particle therapy utilizing (227)Th and (223)Ra, which has demonstrated efficacy in treating multifocal osteosarcoma while exhibiting limited myelotoxicity and high relative biological effectiveness [15]. Additionally, exosome mimetics derived from BMSCs offer a natural platform for nano drug delivery, delivering potent tumor inhibition activity with reduced side effects [16].
Nanocarriers and targeted drug delivery systems also hold potential in overcoming drug resistance and minimizing side effects [17,18]. For instance, lipid nanoparticles loaded with edelfosine have been found to inhibit cell growth in vitro and prevent metastasis in vivo [19]. Furthermore, self-stabilized hyaluronate nanogels co-delivering doxorubicin and cisplatin have demonstrated enhanced antitumor efficacy and reduced side effects [20].
The use of near-infrared imaging and multifunctional graphene-based nano-drug delivery systems has exhibited highly selective anticancer efficiency by targeting mitochondria, offering synergistic phototherapy for drug-resistant osteosarcoma [21]. Moreover, IL-11Ralpha-targeted nanoparticles have shown superior efficacy in treating osteosarcoma by specifically targeting tumor cells. These nanoparticles have demonstrated strong anti-tumor effects in orthotopic and relapsed osteosarcoma models, as well as patient-derived osteosarcoma xenografts [22].
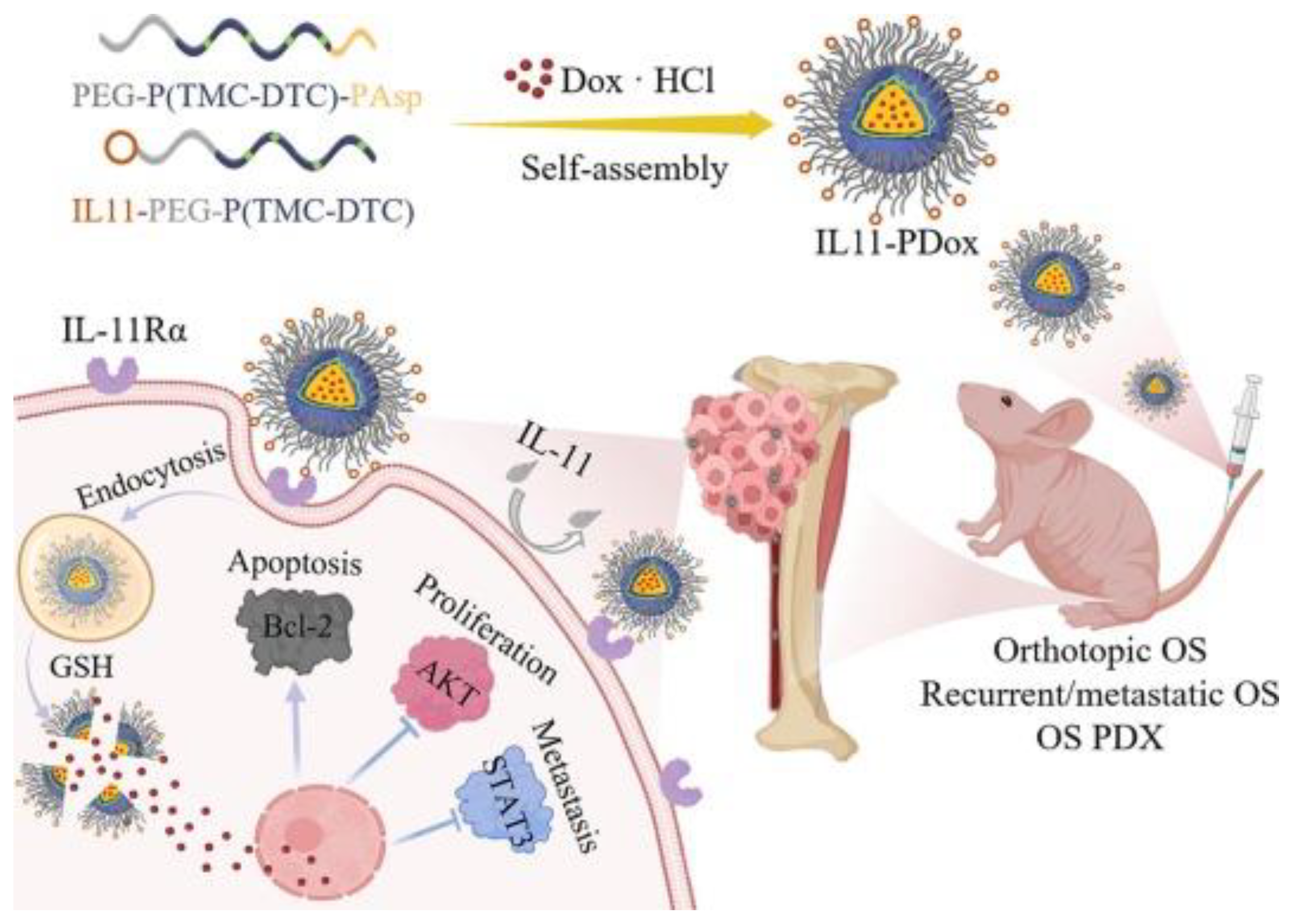
Figure 3 provides a schematic representation of the fabrication of IL-11Rα-targeting polymersomal Dox and its mechanism of inhibiting the growth, recurrence, and metastasis of malignant osteosarcoma. Additionally, Table 4 summarizes examples of nanocarrier systems employed in osteosarcoma treatment.
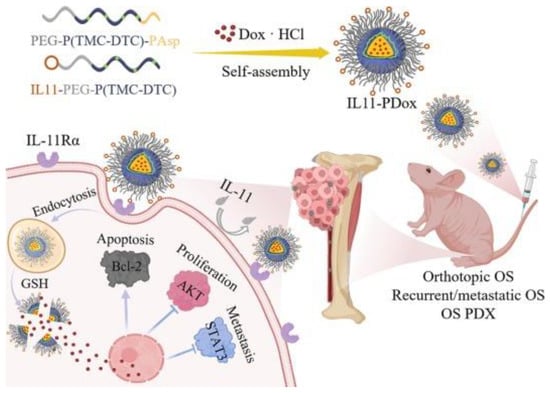
Figure 3.
Illustration of fabrication of IL-11Rα-targeting polymersomal Dox (IL11-PDox) and strong inhibition of growth, recurrence, and metastasis of malignant osteosarcoma. Adapted with permission from [22].

Table 4.
Nanosystems in pediatric osteosarcoma treatment.
2.5. Other Cancers and Cancer-Related Topics
One of the significant challenges in treating pediatric brain tumors, including brain tumors in children, is the blood–brain barrier, which limits effective drug delivery. However, nanotechnology has demonstrated potential with respect to overcoming this obstacle by facilitating drug delivery across the blood–brain barrier [154,155,156,157,158,159,160]. Moreover, nanotechnology offers the ability to selectively target pediatric brain tumors and enhance the bioavailability of phytoconstituents for treating medulloblastoma [158,159]. In the field of ophthalmology, photodynamic therapy utilizing mesoporous silica nanoparticles holds promise for the treatment of retinoblastoma [161]. In regenerative medicine and cancer treatment, nanomedicine-based therapies that combine stem cells with drug delivery systems have shown great potential for achieving improved results [162]. Additionally, gold nanoparticles have been studied for tumor diagnostics through imaging and as delivery devices for targeted therapy in adenoid cystic carcinoma [163]. These examples exemplify the potential of nanotechnology to overcome delivery challenges and provide effective treatments for various pediatric cancers, including osteosarcoma. Table 5 shows additional examples of nanocarrier systems utilized in the treatment of other pediatric cancers or cancer-related disorders.

Table 5.
Nanosystems in other pediatric cancer treatments.
3. Infectious Disease Management and Treatment
3.1. Antimalarial/Antibacterial Treatment
In the context of malaria treatment, nanomedicines have proven to be effective tools for targeted drug delivery against the disease [23]. Nanotechnology offers the capability to design strategies that specifically target drug molecules to different stages of the malaria parasite’s life cycle, address drug-resistant strains, and enhance vaccine effectiveness [24].
For the treatment of leishmaniasis, nanotechnology-based drug delivery systems have been developed to minimize toxicity while maintaining therapeutic efficacy [25]. Moreover, nanotechnology presents innovative solutions for administering drugs to pediatric patients affected by malaria, leishmaniasis, toxoplasmosis, and schistosomiasis [171].
Nanoparticles have also been utilized in bioassays for the detection and control of schistosomiasis, offering improved sensitivity, speed, and convenience [26]. In the case of Praziquantel (PZQ), nanocarriers have been developed to overcome the limitations of its low solubility and bioavailability, thereby enhancing its performance [27]. Nanotechnology has also been employed to combat antibiotic resistance by augmenting the antimicrobial efficacy of ceftriaxone against Gram-positive and Gram-negative bacteria using chitosan nanoparticles, providing an alternative to traditional antibiotics [28].
The application of nanotechnology extends to the treatment of pediatric infectious diseases and solid tumors, with its scope ranging from in vitro studies to clinical trials [172]. Additionally, a biosensor employing nano-fabricated structures and anti-E. coli antibodies has exhibited high sensitivity for clinical use in the detection of bacterial infections in human kidneys [29]. Table 6 provides a summary of examples of nanocarrier systems employed in the treatment of pediatric malaria and other bacterial diseases.

Table 6.
Nanosystems in pediatric malaria and other bacterial diseases.
3.2. COVID-19
Recent studies have showcased the potential of nanotechnology-based diagnostic methods in accurately detecting extracellular vesicles carrying SARS-CoV-2 RNA in plasma, presenting a promising alternative to traditional respiratory RNA level detection approaches. These advancements, encompassing CRISPR-based and optical-based sensing systems, hold significant promise for the development of efficient and rapid diagnostic techniques for COVID-19 [173,174].
Nanotechnology extends beyond diagnostics, offering promising prospects for treatment modalities, vaccination strategies, and the potential integration of artificial intelligence in the field of infectious diseases, including COVID-19 [175]. Moreover, the potential of electrochemical nano-biosensors, utilizing nanomaterials for signal amplification, has been demonstrated in detecting harmful DNA mutations in newborn infants with high sensitivity, a wide dynamic range, and exceptional specificity. This presents a valuable tool for newborn screening purposes [176]. Table 7 provides an overview of examples of nanocarrier systems employed in the treatment of COVID-19.

Table 7.
Nanosystems in pediatric COVID-19 treatment.
Overall, the integration of nanotechnology into the realm of infectious diseases holds potential for the development of innovative and effective diagnostic and treatment strategies. Ongoing research and development efforts in this field are anticipated to yield breakthroughs in the battle against infectious diseases, including malaria, bacterial infections, and viral infections such as SARS-CoV-2. The promising results reported in existing studies indicate that nanotechnology will play a significant role in the future of healthcare, revolutionizing the detection and treatment of various infectious diseases [173,174,175,176].
3.3. TB and HIV
Nanomedicine has paved the way for new treatment possibilities, including the delivery of antimicrobial host defense peptides, which have shown to enhance therapeutic effectiveness and reduce resistance in TB and HIV infections [30]. Nanotechnology-based diagnostic methods have exhibited potential in accurately diagnosing HIV in infants—a challenging population to detect [31]. Furthermore, the formulation of antiretroviral drugs with nanotechnology has improved their bioavailability, reduced dosage requirements, and enhanced treatment outcomes in HIV patients, particularly among pediatric populations [32,33,34].
Regarding TB, nanotechnology-based antigen testing and polymeric micelles have demonstrated high diagnostic accuracy and increased oral bioavailability of rifampicin, respectively, enabling early detection and effective treatment [35,36]. Various nanotechnology-based strategies have been proposed to develop more effective and patient-compliant medicines for TB treatment, including targeting infection reservoirs and overcoming drug resistance [37]. A child-friendly nanoemulsion containing rifampicin has shown promise in increasing drug bioavailability and reducing treatment failure in pediatric TB patients [39]. With increased institutional support, the integration of nanomedicine and genomic research holds the potential to achieve TB elimination by 2050 [38]. Table 8 provides a summary of examples of nanocarrier systems used in the treatment of pediatric TB and HIV.

Table 8.
Nanosystems in pediatric TB and HIV treatments.
3.4. Respiratory and Pulmonary Diseases
Nanoparticles have proven effective in preventing biofilm formation and colonization on endotracheal tubes in pediatric patients with VAP, thereby reducing the risk of infection [40]. Moreover, nanomodified endotracheal tubes have shown substantial reductions in the growth of P. aeruginosa, effectively combating VAP [41]. Nanotechnology-based therapeutic approaches hold promise for the detection and treatment of RSV with maximum efficacy and minimal side effects [42]. Gold nanorods, for instance, have demonstrated the potential to inhibit RSV by activating the immune response, making them a potential antiviral agent against RSV [43]. In cystic fibrosis, nanotechnology has facilitated the development of the Nanoduct sweat test system, which offers improved ease of use and higher diagnostic success rates in newborns compared to the Macroduct/Gibson and Cooke methods [44]. Furthermore, nanotechnology-based approaches have shown promise in managing the pain associated with cystic fibrosis, a common affliction for CF patients [45]. Table 9 provides an overview of nanocarrier systems utilized in the treatment of pediatric respiratory and pulmonary diseases.

Table 9.
Nanosystems in pediatric respiratory and pulmonary diseases.
3.5. Environmental Health and Infectious Diseases
In recent years, the application of nanotechnology in the field of environmental health and infectious diseases has garnered significant attention from scientists and researchers [46]. This emerging field has demonstrated remarkable potential in the detection and treatment of various diseases. However, it is important to acknowledge the associated risks. Studies using human placental perfusion models have indicated that nanoparticles have the ability to cross the placental barrier, raising concerns about potential risks to developing fetuses [46]. Nevertheless, nanotechnology has shown effectiveness in detecting water-borne parasites and mitigating biological contamination in drinking water, especially in areas with inadequate sanitation facilities in developing countries [47,48]. Nanotechnology-based assays and nanodevices have also exhibited promise in the identification of water-borne pathogens, which is crucial for safeguarding public health [49]. Given the escalating production of environmental pollutants and the health threats posed by climate change, concerted efforts are necessary to tackle these environmental health challenges [178]. Moreover, nanotechnology offers potential solutions for combatting the lethal effects of scorpion envenomation and presents strategies for the treatment and control of viral infections [50,51]. While PCR-based assays currently remain the gold standard for the detection of certain viruses, researchers are actively exploring the advantages of nanotechnology and advanced genetic platforms for therapeutic interventions [52]. Table 10 provides an overview of nanocarrier systems employed in the treatment of pediatric infectious diseases related to the environment.

Table 10.
Environmental health and pediatric infectious diseases.
4. Nanotechnology in Other Pediatric Related Areas
4.1. Medical Disorders
Liposome nanotechnology has enabled sustained delivery systems of glucocorticoids for epilepsy treatment, enhancing therapeutic efficacy [53]. The integration of mass spectrometry, genomics advancements, and nanotechnology has facilitated cost-effective expanded newborn screening, enabling the detection of a wider range of disorders in inborn errors of metabolism [54].
Saliva has shown potential as a diagnostic fluid for noninvasive and cost-effective detection of cardiovascular diseases and cancers, although there are still clinical challenges to overcome [55]. Core-cross-linked nanoparticles have demonstrated the ability to reduce neuroinflammation and limit secondary injury spread in a mouse model of traumatic brain injury [56]. Nanotechnology has also improved bioavailability and reduced side effects in the treatment of neurodegenerative diseases such as Parkinson’s, Alzheimer’s, multiple sclerosis, amyotrophic lateral sclerosis, Huntington’s, and Wilson’s diseases [57]. Additionally, nanotechnology-based approaches and biosensors hold promise for the high-performance diagnosis of gestational diabetes and jaundice, providing important monitoring tools during pregnancy [58].
Nanotechnology has also shown potential in aiding the diagnosis and treatment of pediatric bone conditions such as type III Osteogenesis Imperfecta, providing insights for effective management [59,60]. Nanofiltered C1 Esterase Inhibitor has been proven to effectively prevent hereditary angioedema attacks during dental, medical, or surgical procedures, with no reported adverse events [61]. Furthermore, the use of polymeric nanocapsules containing geraniol and icaridin has shown efficacy and safety in combating the Aedes aegypti mosquito—a major disease transmitter [62]. Nanoparticle coating techniques have demonstrated the ability to enhance the bioavailability of vitamin B(12) in food crops, addressing the risk of micronutrient deficiency and associated health issues [63]. Notably, the management of vulvovaginitis in girls has benefited from the use of bioyoghurt, probiotics, and petroleum jelly [64].
The integration of nanotechnology with pure sciences and the technologies of the fourth industrial revolution holds the potential for significant advancements in pediatric healthcare, including the development of nano-doctors that could eliminate the need for invasive surgeries and revolutionize diagnostics and therapeutics [65,66]. Albumin and liposome nanoparticles have also shown promise in the treatment of pediatric diseases [67]. Moreover, a thermosensitive liposome formulation combined with mild hyperthermia has improved the therapeutic index of vinorelbine for the treatment of Rhabdomyosarcoma [140]. Advanced microengraving technology has efficiently identified antigen-specific T-cell responses for T-cell immunology [68].
Nanotechnology-based delivery systems offer a potential solution to address the lack of pharmacokinetic data for pediatric drug development [69]. Additionally, these delivery strategies show promise in effectively treating traumatic brain injury (TBI) by bypassing biological barriers and enhancing target engagement [70]. Furthermore, nanomedicine holds potential with regard to developing urinary bladders for children with congenital bladder dysfunction, regenerating kidney, bladder, and urethra tissues using stem cell therapies, myoblasts, fibroblasts, and three-dimensional stem cell-derived organoids, addressing pediatric urological conditions [71]. Nanomedicine-based therapies have also demonstrated promise in managing pregnancy complications, improving outcomes for both mothers and unborn children while reducing the need for emergency caesarean sections [72]. Personalized nanomedicine has shown potential in the treatment of cerebral palsy using gold nanoparticles coated with targeted dendrimers in conjunction with CT imaging and transcranial magnetic stimulation, leading to improved motor function in affected children [73]. Nanomedicine and stem cell therapy offer possibilities for diagnosing and treating high-risk factors associated with cerebral palsy, such as prematurity and low birth weight [74]. Furthermore, nanofiltered human C1 inhibitor concentrate (C1-INH NF) has proven to be a safe and effective replacement for deficient plasma C1 inhibitor levels, reducing the incidence of angioedema attacks in hereditary angioedema [75]. Nanotechnology has also demonstrated its utility in scoliosis management, with ultra-low-dose full-spine protocols providing reliable and repeatable measurements of the Cobb angle with minimal radiation exposure [76]. Moreover, diagnostic and therapeutic applications of nanotechnology show promise in fetal, neonatal, and pediatric diseases affecting the respiratory tract, neurosensory system, and infectious conditions. However, the acquisition of further data is necessary to ascertain their safety and efficacy [77].
Table 11 provides a summary of examples of nanocarrier systems utilized in various medical treatments. In conclusion, nanotechnology-based treatments hold great potential for addressing a wide range of pediatric conditions, including tissue engineering, stem cell therapy, and personalized nanomedicine. These innovative approaches offer potential solutions for the development of new scaffolds for pediatric urological conditions, the management of pregnancy complications, cerebral palsy, hereditary angioedema, scoliosis, and other diseases affecting the respiratory tract, neurosensory system, and infectious conditions. However, further research is necessary to validate their efficacy and ensure their safety.

Table 11.
Nanosystems in medical treatments.
4.2. Dental Disorders
Nanotechnology has emerged as a promising field in pediatric dentistry, particularly in the treatment of occlusal cavities in children. One notable application of nanotechnology is the enhancement of wear resistance in dental materials through the use of resin coatings containing nanoparticles. This approach offers a more durable solution for young patients, as evidenced by studies demonstrating improved wear resistance [78].
Another development in pediatric dentistry is the use of nanovectors to deliver oral sprays containing resveratrol. These sprays have shown significant efficacy in reducing dental plaque and gingival inflammation in early childhood [79]. However, it is crucial to conduct further research to fully understand the potential drawbacks of nanoparticles in pediatric dentistry [83].
It is important to consider recent advancements in nanotechnology alongside the insights from the 2002 Pediatric Restorative Dentistry Consensus Conference [182]. Polysaccharide-based micro- and nano-sized drug delivery systems hold great promise for drug administration in pediatric dentistry due to their biocompatibility, biotolerance, biodegradability, and low toxicity [80]. Additionally, silver nanoparticles (AgNPs) exhibit potent antimicrobial properties and can potentially be incorporated into dental materials to improve their mechanical and antibacterial characteristics, potentially enhancing oral health outcomes in pediatric patients [81]. The integration of nanoparticles of amorphous calcium phosphate into dental sealants can also provide antibacterial and rechargeable sealants with desirable properties and elevated levels of calcium and phosphate ion release [183].
Furthermore, biodegradable airway stents made from magnesium alloys have demonstrated feasibility and efficacy in managing pediatric laryngotracheal stenosis. These stents offer a less invasive and more effective approach to managing pediatric airway obstruction [82]. Table 12 provides examples of nanocarrier systems utilized in the treatment of pediatric dental diseases.

Table 12.
Nanosystems in dental diseases.
4.3. Dermatological Disorders
Among the various skin conditions affecting children, atopic dermatitis has received significant attention in the development of nanocarrier-based drug delivery systems. These nanocarriers have demonstrated high efficacy in enhancing drug solubility, thermodynamic activity, and skin permeation, thereby reducing side effects and improving the management of atopic dermatitis [84]. Studies have specifically highlighted the effectiveness of nanomaterials such as chitosan nanoparticles in enhancing drug penetration and efficacy for the treatment of atopic dermatitis [85,86]. Furthermore, in the case of neonatal scleredema, the use of polydopamine nanoparticles coated with stem cell membrane fragments and doxorubicin has shown inhibitory effects on fibrosis, suggesting the potential of nanotechnology in treating this condition [87]. Although further advancements are necessary to fully exploit the potential of nanocarriers for anti-acne drugs as they hold significant promise in enhancing the effectiveness and safety of such treatments [184]. Table 13 provides a summary of examples highlighting nanocarrier systems employed in the treatment of pediatric dermatologic diseases.

Table 13.
Nanosystems in pediatric dermatologic diseases.
4.4. Nanotechnology in Pediatric Nutrition
The obesity epidemic, in particular, requires multidisciplinary collaborations between engineers, physical scientists, and nutrition experts to develop innovative technologies that support therapeutic advancements and promote behavioral changes [88]. By utilizing nanotechnology-based food production, it becomes possible to create more nutritious and lower-calorie food options that can help address nutritional deficiencies, obesity, and type 2 diabetes [89]. To combat common issues such as iron deficiency, researchers have developed iron solid lipid nanoparticles as an alternative to commercially available supplements, overcoming certain limitations [90]. Additionally, green-synthesized iron oxide nanoparticles show promise as antianemic preparations, although safety concerns must be thoroughly addressed [186].
Furthermore, nanotechnology-based optical biosensors, such as gold nanoclusters, have significant potential in ensuring food safety by detecting harmful pathogens and chemical substances [91]. In the context of food allergies and anaphylaxis, nanotechnology-based diagnostic methods can enable personalized allergen immunotherapy and avoidance diets to effectively manage the increasing prevalence of food allergies [92]. To drive progress in this field, collaborative efforts among food producers, policy makers, and health authorities are necessary to address safety concerns and provide adequate funding for research. Table 14 provides a summary of examples illustrating the utilization of nanocarrier systems in pediatric nutrition.

Table 14.
Nanosystems in pediatric nutrition.
4.5. Drug Delivery
Nanotechnology has played a pivotal role in advancing pediatric drug delivery, addressing critical issues such as poor water solubility, taste-masking, and drug stability. One notable application is the use of biomimetic nanovesicles incorporated into transdermal patches, which have demonstrated promising outcomes in enhancing micronutrient delivery for infants and mothers [93]. In situ self-assembly nanoparticle technology has emerged as a potent platform for drug delivery, improving the stability, palatability, and bioavailability of pediatric oral solid dosage forms [94,95]. Folic acid magnetic nanotheranostics hold the potential for safe and effective drug delivery, reducing cardiotoxicity and enhancing drug uptake [96]. Additionally, nanoparticle-based drug delivery systems have enabled the production of easily consumable spheroids, contributing to improved patient compliance [97]. Nanofibers composed of polycaprolactone and polyvinyl alcohol have shown promise in oromucosal drug delivery approaches for children and elderly patients [98,99]. The utilization of spironolactone-loaded nanocapsules has demonstrated a favorable outcome, with stable nanocapsules and high encapsulation efficiency, allowing for lower volumes of liquid preparation [100]. Nanopatch technology has garnered significant acceptability for vaccine delivery, particularly in low and middle-income countries [101]. Furthermore, nanoformulations are necessary for nanocosmeceuticals containing gallic acid and its derivatives due to their poor water solubility and biodegradability [102]. Examples of nanocarrier systems in drug delivery are summarized in Table 15.

Table 15.
Nanosystems in pediatric drug delivery.
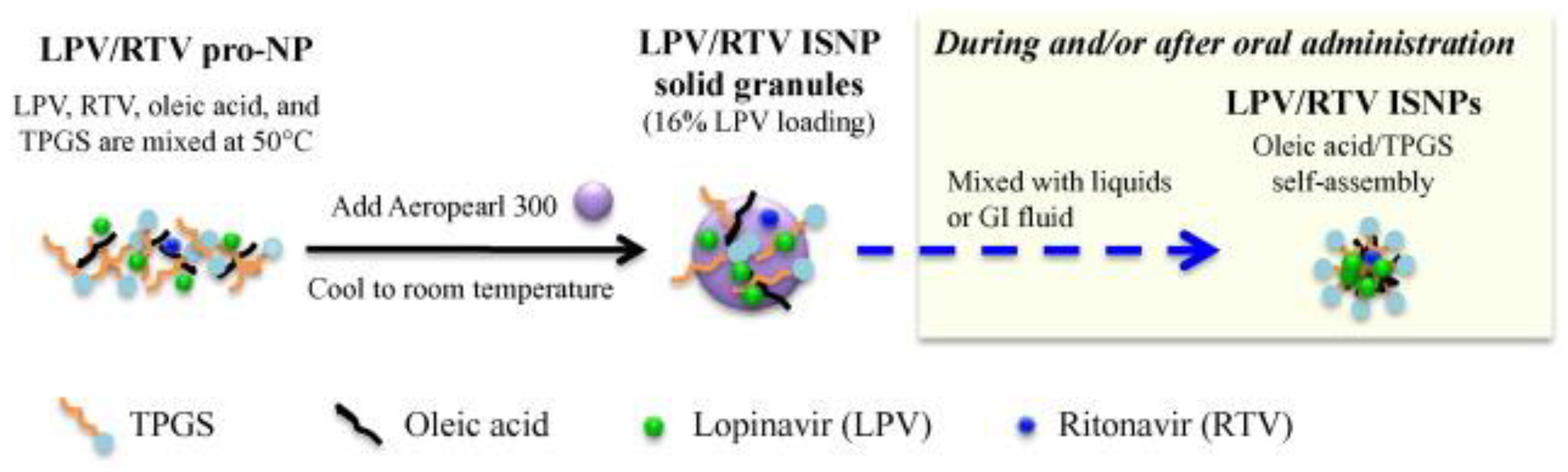
Nanotechnology-based drug delivery systems present novel approaches to enhance pediatric drug administration, minimize adverse effects, and improve the efficacy of existing therapies. Notably, the development of fixed-dose combinations, such as lopinavir/ritonavir, utilizing innovative in situ self-assembly nanoparticle technology, exemplifies the immense potential of nanotechnology in drug delivery (Figure 4). However, further research and development efforts are essential to fully harness the capabilities of nanotechnology in pediatric drug delivery.
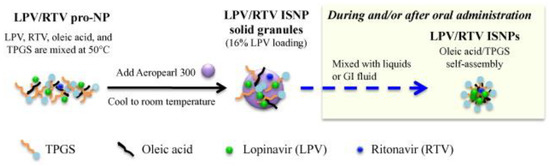
Figure 4.
Preparation of LPV/RTV ISNP granules and formation of LPV/RTV ISNPs. LPV and RTV completely dissolve in oleic acid and TPGS to form the pro-NP that is coated on the surface of Aeropearl 300. Once LPV/RTV ISNP solid granules are introduced into liquids (e.g., water during administration as a sachet or the fluid in the GI tract after administration as a sprinkle), oleic acid and TPGS form the ISNPs by a self-assembly process; meanwhile, LPV and RTV are entrapped into the ISNPs during the NP formation. Adapted with permission from [95].
5. Other Pediatric Applications
Highly sensitive assays using LC-MS/MS technology have identified melatonin and N-acetylserotonin as potential biomarkers for sleep-related disorders, providing valuable insights for diagnosis and treatment [188]. In audiology, nanotechnology research has shown promise in developing advanced sound and hearing implants, offering a potential breakthrough for individuals with profound deafness [189]. Moreover, the presence of metallic particles in human tonsil tissue and amniotic fluid has raised intriguing possibilities regarding their role in disease causation and emerging nanopathology [190].
The concern over nanoparticle exposure has led to investigations into resuspension rates, revealing variations depending on the product, flooring, and resuspension force. Products containing copper, silver, and zinc nanomaterials exhibited higher rates, highlighting the importance of further research and regulation in this area [191]. Advancements in drug delivery systems and nanomedicines hold promise for treating degenerative ocular diseases that manifest in childhood, offering the potential to significantly enhance the quality of life for affected pediatric patients [192]. Similarly, the use of nanocarrier-mediated drug delivery has garnered support and is clinically recommended for the treatment of atopic dermatitis [185].
Studies exploring microbial interactions have unveiled the strong binding of Streptococcus mutans-derived exoenzyme GtfB to Candida albicans, shedding light on the modulatory role of this interaction [193]. Additionally, the implementation of a nano-selenium reactive barrier approach has shown success in suppressing mercury release from compact fluorescent lamps, aiding in the identification of mercury contamination sources and achieving significant reductions in exposure scenarios [194]. A gold nanoparticle-based dynamic light scattering (DLS) probe has demonstrated potential for on-site monitoring of lead (Pb) levels in various samples, detecting concentrations as low as 100 ppt, which surpasses the EPA standard limit by nearly two orders of magnitude [195]. Table 16 provides an overview of nanocarrier systems employed in other areas of pediatric health.

Table 16.
Nanosystems in other pediatric disorders.
6. Potential Risks and Health Effects
Despite its potentially transformational role, it is crucial to carefully assess and address the potential risks associated with this rapidly advancing technology. Several studies have shed light on the potential health implications of nanotechnology, emphasizing the need for caution. For instance, one study [103] investigated the impact of engineered nanoparticles on children’s health, while others [104,105] focused on the neurotoxicity of nanoparticles. Additionally, the health effects of carbon nanoparticles and ultrafine particles warrant further investigation [106,107,108].
Although graphene is generally considered a safer alternative to carbon nanotubes, it is still essential to implement specific safety protocols when working with any type of nanomaterial. Studies have shown that nanoparticles can impose metabolic burden, oxidative stress, and potentially alter milk composition in breastfeeding systems, indicating potential risks [196]. Moreover, children are particularly vulnerable to the potential hazards associated with engineered nanoparticles, necessitating focused research on exposure levels and health consequences [197].
There is a significant lack of public understanding about nanotechnology, especially among middle-school children, despite its profound impact on various industries and society as a whole [198]. Therefore, it is crucial to educate the public about both the potential risks and benefits of nanotechnology. While research is needed to comprehend the disparities between children and adults in terms of harmful effects induced by exposure to ultrafine particulate matter, the unique physicochemical properties of nanoparticles offer promising opportunities for biomedical applications. Hence, it is essential to explore the potential benefits of nanoparticles within the field of nanotechnology [108]. Table 17 provides an overview of potential health risks associated with nanosystems in pediatric nanomedicine.

Table 17.
Nanosystems and their potential risks and impact on pediatric health.
In conclusion, while nanotechnology holds tremendous promise, it is imperative to carefully evaluate and mitigate its potential risks to ensure its safe and responsible utilization.
7. Conclusions
In conclusion, nanotechnology has been identified as a potential tool in the field of pediatric medicine, offering new possibilities for the diagnosis and treatment of various conditions. Its use in pediatric oncology shows promise, as it allows for targeted drug delivery, reduced toxicity, and combined immunotherapy, which may have benefits in treating specific pediatric tumors. Nanotechnology-based approaches have shown potential in delivering drugs directly to the affected areas in pediatric cancers such as leukemia and neuroblastoma. Additionally, nanotechnology has potential applications beyond cancer treatment, including the management of pediatric infectious diseases, respiratory and pulmonary conditions, and environmental health concerns. The integration of nanotechnology has led to advancements in drug delivery, diagnostics, and treatment outcomes. However, it is important to approach nanotechnology carefully and ensure the responsible use of nanoparticles. Thorough research is needed to understand their safety profiles, particularly in relation to potential health risks in children.
Author Contributions
Conceptualization, H.O.; writing—original draft preparation, H.O. and K.M.; writing—review and editing, H.O. and K.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors partly used OpenAI’s large-scale language-generation model. The authors reviewed, revised, and edited the document for accuracy and take full responsibility for the content of this publication. The authors declare no conflict of interest.
References
- Aleassa, E.M.; Xing, M.; Keijzer, R. Nanomedicine as an innovative therapeutic strategy for pediatric cancer. Pediatr. Surg. Int. 2015, 31, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Federman, N.; Denny, C.T. Targeting liposomes toward novel pediatric anticancer therapeutics. Pediatr. Res. 2010, 67, 514–519. [Google Scholar] [CrossRef]
- Xu, L.F.; Lu, Z.X.; Tan, G.H.; Huang, F.Y.; Cao, R.; He, N.Y.; Xu, T.F.; Zhang, L.M. Coomassie Bright Blue-Conjugated Human Serum Albumin Nanoparticles as a Tumor-Selective Weapon for Leukemia Therapy. Nanosci. Nanotechnol. Lett. 2019, 11, 1651–1660. [Google Scholar] [CrossRef]
- Tatar, A.S.; Nagy-Simon, T.; Tomuleasa, C.; Boca, S.; Astilean, S. Nanomedicine approaches in acute lymphoblastic leukemia. J. Control. Release 2016, 238, 123–138. [Google Scholar] [CrossRef]
- Uckun, F.M.; Qazi, S.; Ma, H.; Yin, L.; Cheng, J. A rationally designed nanoparticle for RNA interference therapy in B-lineage lymphoid malignancies. EBioMedicine 2014, 1, 141–155. [Google Scholar] [CrossRef]
- Connerty, P.; Moles, E.; de Bock, C.E.; Jayatilleke, N.; Smith, J.L.; Meshinchi, S.; Mayoh, C.; Kavallaris, M.; Lock, R.B. Development of siRNA-Loaded Lipid Nanoparticles Targeting Long Non-Coding RNA LINC01257 as a Novel and Safe Therapeutic Approach for t(8;21) Pediatric Acute Myeloid Leukemia. Pharmaceutics 2021, 13, 1681. [Google Scholar] [CrossRef] [PubMed]
- Haddad, Y.; Charousova, M.; Zivotska, H.; Splichal, Z.; Rodrigo, M.A.M.; Michalkova, H.; Krizkova, S.; Tesarova, B.; Richtera, L.; Vitek, P.; et al. Norepinephrine transporter-derived homing peptides enable rapid endocytosis of drug delivery nanovehicles into neuroblastoma cells. J. Nanobiotechnol. 2020, 18, 20. [Google Scholar] [CrossRef]
- Pastorino, F.; Brignole, C.; Di Paolo, D.; Perri, P.; Curnis, F.; Corti, A.; Ponzoni, M. Overcoming Biological Barriers in Neuroblastoma Therapy: The Vascular Targeting Approach with Liposomal Drug Nanocarriers. Small 2019, 15, 13. [Google Scholar] [CrossRef]
- Pottoo, F.H.; Barkat, M.A.; Harshita; Ansari, M.A.; Javed, M.N.; Sajid Jamal, Q.M.; Kamal, M.A. Nanotechnological based miRNA intervention in the therapeutic management of neuroblastoma. Semin. Cancer Biol. 2021, 69, 100–108. [Google Scholar] [CrossRef]
- Mobasheri, T.; Rayzan, E.; Shabani, M.; Hosseini, M.; Mahmoodi Chalbatani, G.; Rezaei, N. Neuroblastoma-targeted nanoparticles and novel nanotechnology-based treatment methods. J. Cell. Physiol. 2021, 236, 1751–1775. [Google Scholar] [CrossRef]
- Yan, H.; Zhai, B.; Yang, F.; Chen, Z.L.; Zhou, Q.; Paiva-Santos, A.C.; Yuan, Z.Q.; Zhou, Y. Nanotechnology-Based Diagnostic and Therapeutic Strategies for Neuroblastoma. Front. Pharmacol. 2022, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Brignole, C.; Bensa, V.; Fonseca, N.A.; Del Zotto, G.; Bruno, S.; Cruz, A.F.; Malaguti, F.; Carlini, B.; Morandi, F.; Calarco, E.; et al. Cell surface Nucleolin represents a novel cellular target for neuroblastoma therapy. J. Exp. Clin. Cancer Res. 2021, 40, 180. [Google Scholar] [CrossRef] [PubMed]
- Burgos-Panadero, R.; El Moukhtari, S.H.; Noguera, I.; Rodriguez-Nogales, C.; Martin-Vano, S.; Vicente-Munuera, P.; Canete, A.; Navarro, S.; Blanco-Prieto, M.J.; Noguera, R. Unraveling the extracellular matrix-tumor cell interactions to aid better targeted therapies for neuroblastoma. Int. J. Pharm. 2021, 608, 10. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Nogales, C.; Mura, S.; Couvreur, P.; Blanco-Prieto, M.J. Squalenoyl-gemcitabine/edelfosine nanoassemblies: Anticancer activity in pediatric cancer cells and pharmacokinetic profile in mice. Int. J. Pharm. 2020, 582, 8. [Google Scholar] [CrossRef]
- Baranowska-Kortylewicz, J.; Sharp, J.G.; McGuire, T.R.; Joshi, S.; Coulter, D.W. Alpha-Particle Therapy for Multifocal Osteosarcoma: A Hypothesis. Cancer Biother. Radiopharm. 2020, 35, 418–424. [Google Scholar] [CrossRef]
- Wang, J.K.; Li, M.J.; Jin, L.M.; Guo, P.; Zhang, Z.X.; Zhanghuang, C.H.; Tan, X.J.; Mi, T.; Liu, J.Y.; Wu, X.; et al. Exosome mimetics derived from bone marrow mesenchymal stem cells deliver doxorubicin to osteosarcoma in vitro and in vivo. Drug Deliv. 2022, 29, 3291–3303. [Google Scholar] [CrossRef]
- Wang, S.Y.; Hu, H.Z.; Qing, X.C.; Zhang, Z.C.; Shao, Z.W. Recent advances of drug delivery nanocarriers in osteosarcoma treatment. J. Cancer 2020, 11, 69–82. [Google Scholar] [CrossRef]
- Desai, S.A.; Manjappa, A.; Khulbe, P. Drug delivery nanocarriers and recent advances ventured to improve therapeutic efficacy against osteosarcoma: An overview. J. Egypt. Natl. Cancer Inst. 2021, 33, 14. [Google Scholar] [CrossRef]
- Gonzalez-Fernandez, Y.; Brown, H.K.; Patino-Garcia, A.; Heymann, D.; Blanco-Prieto, M.J. Oral administration of edelfosine encapsulated lipid nanoparticles causes regression of lung metastases in pre-clinical models of osteosarcoma. Cancer Lett. 2018, 430, 193–200. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, F.; Li, M.Q.; Yu, Z.Q.; Qi, R.G.; Ding, J.X.; Zhang, Z.Y.; Chen, X.S. Self-Stabilized Hyaluronate Nanogel for Intracellular Codelivery of Doxorubicin and Cisplatin to Osteosarcoma. Adv. Sci. 2018, 5, 12. [Google Scholar] [CrossRef]
- Zeng, W.N.; Yu, Q.P.; Wang, D.; Liu, J.L.; Yang, Q.J.; Zhou, Z.K.; Zeng, Y.P. Mitochondria-targeting graphene oxide nanocomposites for fluorescence imaging-guided synergistic phototherapy of drug-resistant osteosarcoma. J. Nanobiotechnol. 2021, 19, 19. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, R.; Yang, L.; Sha, Y.; Zhao, S.; Guo, J.; Chen, D.; Zhong, Z.; Meng, F. IL-11Ralpha-targeted nanostrategy empowers chemotherapy of relapsed and patient-derived osteosarcoma. J. Control. Release 2022, 350, 460–470. [Google Scholar] [CrossRef] [PubMed]
- Aditya, N.P.; Vathsala, P.G.; Vieira, V.; Murthy, R.S.R.; Souto, E.B. Advances in nanomedicines for malaria treatment. Adv. Colloid Interface Sci. 2013, 201, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Najer, A.; Palivan, C.G.; Beck, H.P.; Meier, W. Challenges in Malaria Management and a Glimpse at Some Nanotechnological Approaches. Adv. Exp. Med. Biol. 2018, 1052, 103–112. [Google Scholar] [CrossRef]
- Chavez-Fumagalli, M.A.; Ribeiro, T.G.; Castilho, R.O.; Fernandes, S.O.; Cardoso, V.N.; Coelho, C.S.; Mendonca, D.V.; Soto, M.; Tavares, C.A.; Faraco, A.A.; et al. New delivery systems for amphotericin B applied to the improvement of leishmaniasis treatment. Rev. Soc. Bras. Med. Trop. 2015, 48, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.Q.; Liu, J.Q.; Lin, Y.F.; Weng, R.N.; Chen, R.; Li, J.; Lv, Z.Y. Diagnosis, Monitoring, and Control of Schistosomiasis-An Update. J. Biomed. Nanotechnol. 2018, 14, 430–455. [Google Scholar] [CrossRef]
- Mengarda, A.C.; Iles, B.; João Paulo, F.L.; de Moraes, J. Recent trends in praziquantel nanoformulations for helminthiasis treatment. Expert Opin. Drug Deliv. 2022, 19, 383–393. [Google Scholar] [CrossRef]
- Duceac, L.D.; Calin, G.; Eva, L.; Marcu, C.; Bogdan Goroftei, E.R.; Dabija, M.G.; Mitrea, G.; Luca, A.C.; Hanganu, E.; Gutu, C.; et al. Third-Generation Cephalosporin-Loaded Chitosan Used to Limit Microorganisms Resistance. Materials 2020, 13, 4792. [Google Scholar] [CrossRef]
- Basu, M.; Seggerson, S.; Henshaw, J.; Jiang, J.; del A Cordona, R.; Lefave, C.; Boyle, P.J.; Miller, A.; Pugia, M.; Basu, S. Nano-biosensor development for bacterial detection during human kidney infection: Use of glycoconjugate-specific antibody-bound gold NanoWire arrays (GNWA). Glycoconj. J. 2004, 21, 487–496. [Google Scholar] [CrossRef]
- Dlozi, P.N.; Gladchuk, A.; Crutchley, R.D.; Keuler, N.; Coetzee, R.; Dube, A. Cathelicidins and defensins antimicrobial host defense peptides in the treatment of TB and HIV: Pharmacogenomic and nanomedicine approaches towards improved therapeutic outcomes. Biomed. Pharmacother. 2022, 151, 13. [Google Scholar] [CrossRef]
- Goel, N.; Sharma, R. Infant HIV and Nanotechnology. In Proceedings of the Nanotech 2009 Conference, Houston, TX, USA, 3–7 May 2009; p. 229. [Google Scholar]
- Kotta, S.; Khan, A.W.; Ansari, S.H.; Sharma, R.K.; Ali, J. Anti HIV nanoemulsion formulation: Optimization and in vitro-in vivo evaluation. Int. J. Pharm. 2014, 462, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Gogtay, J.A.; Malhotra, G. Reformulation of existing antiretroviral drugs. Curr. Opin. HIV AIDS 2013, 8, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Melo, R.; Lemos, A.; Preto, A.J.; Bueschell, B.; Matos-Filipe, P.; Barreto, C.; Almeida, J.G.; Silva, R.D.M.; Correia, J.D.G.; Moreira, I. An Overview of Antiretroviral Agents for Treating HIV Infection in Paediatric Population. Curr. Med. Chem. 2020, 27, 760–794. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.Y.; La Course, S.M.; Kim, S.; Liu, C.; Ning, B.; Bao, D.; Fan, J.; Lyon, C.J.; Sun, Z.Y.; Nachman, S.; et al. Evaluation of a serum-based antigen test for tuberculosis in HIV-exposed infants: A diagnostic accuracy study. BMC Med. 2021, 19, 12. [Google Scholar] [CrossRef]
- Moretton, M.A.; Hocht, C.; Taira, C.; Sosnik, A. Rifampicin-loaded ‘flower-like’ polymeric micelles for enhanced oral bioavailability in an extemporaneous liquid fixed-dose combination with isoniazid. Nanomedicine 2014, 9, 1635–1650. [Google Scholar] [CrossRef] [PubMed]
- Sosnik, A.; Carcaboso, A.M.; Glisoni, R.J.; Moretton, M.A.; Chiappetta, D.A. New old challenges in tuberculosis: Potentially effective nanotechnologies in drug delivery. Adv. Drug Deliv. Rev. 2010, 62, 547–559. [Google Scholar] [CrossRef] [PubMed]
- Jassal, M.S.; Aldrovandi, G.M. 2050: Ending the odyssey of the great white plague Part of a series on Pediatric Pharmacology, guest edited by Gianvincenzo Zuccotti, Emilio Clementi, and Massimo Molteni. Pharmacol. Res. 2011, 64, 176–179. [Google Scholar] [CrossRef] [PubMed]
- Halicki, P.C.B.; Hadrich, G.; Boschero, R.; Ferreira, L.A.; von Groll, A.; da Silva, P.E.A.; Dora, C.L.; Ramos, D.F. Alternative Pharmaceutical Formulation for Oral Administration of Rifampicin. Assay Drug Dev. Technol. 2018, 16, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.C.; Cheng, D.; Tarquinio, K.M.; Webster, T.J. Nanotechnology: Pediatric applications. Pediatr. Res. 2010, 67, 500–504. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.C.; Webster, T.J. Decreased Pseudomonas aeruginosa biofilm formation on nanomodified endotracheal tubes: A dynamic lung model. Int. J. Nanomed. 2016, 11, 3825–3831. [Google Scholar] [CrossRef]
- Subbarayan, P.; Informat Resources Management, A. Nanotechnology for the Management of Respiratory Disease; Igi Global: Hersey, PA, USA, 2018; pp. 927–940. [Google Scholar]
- Bawage, S.S.; Tiwari, P.M.; Singh, A.; Dixit, S.; Pillai, S.R.; Dennis, V.A.; Singh, S.R. Gold nanorods inhibit respiratory syncytial virus by stimulating the innate immune response. Nanomedicine 2016, 12, 2299–2310. [Google Scholar] [CrossRef] [PubMed]
- Vernooij-van Langen, A.; Dompeling, E.; Yntema, J.B.; Arets, B.; Tiddens, H.; Loeber, G.; Dankert-Roelse, J. Clinical evaluation of the Nanoduct sweat test system in the diagnosis of cystic fibrosis after newborn screening. Eur. J. Pediatr. 2015, 174, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Trandafir, L.M.; Leon, M.M.; Frasinariu, O.; Baciu, G.; Dodi, G.; Cojocaru, E. Current Practices and Potential Nanotechnology Perspectives for Pain Related to Cystic Fibrosis. J. Clin. Med. 2019, 8, 1023. [Google Scholar] [CrossRef] [PubMed]
- Grafmuller, S.; Manser, P.; Krug, H.F.; Wick, P.; von Mandach, U. Determination of the Transport Rate of Xenobiotics and Nanomaterials Across the Placenta using the ex vivo Human Placental Perfusion Model. J. Vis. Exp. 2013, 76, 50401. [Google Scholar] [CrossRef]
- Jain, S.; Melo, T.G.C.; Dolabella, S.S.; Liu, J.W. Current and emerging tools for detecting protozoan cysts and oocysts in water. TRAC-Trends Anal. Chem. 2019, 121, 11. [Google Scholar] [CrossRef]
- Kristanti, R.A.; Hadibarata, T.; Syafrudin, M.; Yilmaz, M.; Abdullah, S. Microbiological Contaminants in Drinking Water: Current Status and Challenges. Water Air Soil Pollut. 2022, 233, 17. [Google Scholar] [CrossRef]
- Theron, J.; Cloete, T.E.; de Kwaadsteniet, M. Current Molecular and Emerging Nanobiotechnology Approaches for the Detection of Microbial Pathogens; Caister Academic Press: Wymondham, UK, 2010; pp. 39–68. [Google Scholar]
- Mohamed, F.A.N.; Laraba-Djebari, F. Scorpion envenomation: A deadly illness requiring an effective therapy. Toxin Rev. 2021, 40, 592–605. [Google Scholar] [CrossRef]
- Melendez-Villanueva, M.A.; Moran-Santibanez, K.; Martinez-Sanmiguel, J.J.; Rangel-Lopez, R.; Garza-Navarro, M.A.; Rodriguez-Padilla, C.; Zarate-Trivino, D.G.; Trejo-Avila, L.M. Virucidal Activity of Gold Nanoparticles Synthesized by Green Chemistry Using Garlic Extract. Viruses 2019, 11, 1111. [Google Scholar] [CrossRef]
- Jeong, S.; Park, M.J.; Song, W.; Kim, H.S. Advances in laboratory assays for detecting human metapneumovirus. Ann. Transl. Med. 2020, 8, 17. [Google Scholar] [CrossRef]
- Holtman, L.; van Vliet, E.A.; Appeldoorn, C.; Gaillard, P.J.; de Boer, M.; Dorland, R.; Wadman, W.J.; Gorter, J.A. Glutathione pegylated liposomal methylprednisolone administration after the early phase of status epilepticus did not modify epileptogenesis in the rat. Epilepsy Res. 2014, 108, 396–404. [Google Scholar] [CrossRef]
- Mak, C.M.; Lee, H.C.H.; Chan, A.Y.W.; Lam, C.W. Inborn errors of metabolism and expanded newborn screening: Review and update. Crit. Rev. Clin. Lab. Sci. 2013, 50, 142–162. [Google Scholar] [CrossRef]
- Pfaffe, T.; Cooper-White, J.; Beyerlein, P.; Kostner, K.; Punyadeera, C. Diagnostic Potential of Saliva: Current State and Future Applications. Clin. Chem. 2011, 57, 675–687. [Google Scholar] [CrossRef]
- Yoo, D.; Magsam, A.W.; Kelly, A.M.; Stayton, P.S.; Kievit, F.M.; Convertine, A.J. Core-Cross-Linked Nanoparticles Reduce Neuroinflammation and Improve Outcome in a Mouse Model of Traumatic Brain Injury. ACS Nano 2017, 11, 8600–8611. [Google Scholar] [CrossRef]
- Jampilek, J.; Kralova, K.; Novak, P.; Novak, M. Nanobiotechnology in Neurodegenerative Diseases; Springer International Publishing Ag: Cham, Switzerland, 2019; pp. 65–138. [Google Scholar]
- Peng, S.Q.; Wang, Q.; Xiong, G.P.; Gopinath, S.C.B.; Lei, G. Biosensors and biomarkers for determining gestational diabetes mellitus and jaundice in children. Biotechnol. Appl. Biochem. 2022, 69, 1354–1364. [Google Scholar] [CrossRef]
- Fan, Z.; Smith, P.A.; Eckstein, E.C.; Harris, G.F. Mechanical properties of OI type III bone tissue measured by nanoindentation. J. Biomed. Mater. Res. Part A 2006, 79, 71–77. [Google Scholar] [CrossRef]
- Fan, Z.; Smith, P.A.; Harris, G.F.; Rauch, F.; Bajorunaite, R. Comparison of nanoindentation measurements between osteogenesis imperfecta Type III and Type IV and between different anatomic locations (femur/tibia versus iliac crest). Connect. Tissue Res. 2007, 48, 70–75. [Google Scholar] [CrossRef]
- Grant, J.A.; White, M.V.; Li, H.H.; Fitts, D.; Kalfus, I.N.; Uknis, M.E.; Lumry, W.R. Preprocedural administration of nanofiltered C1 esterase inhibitor to prevent hereditary angioedema attacks. Allergy Asthma Proc. 2012, 33, 348–353. [Google Scholar] [CrossRef]
- de Andrade, L.R.M.; Guilger-Casagrande, M.; Germano-Costa, T.; de Lima, R. Polymeric Nanorepellent Systems Containing Geraniol and Icaridin Aimed at Repelling Aedes aegypti. Int. J. Mol. Sci. 2022, 23, 8317. [Google Scholar] [CrossRef]
- Oh, S.; Cave, G.; Lu, C. Vitamin B(12) (Cobalamin) and Micronutrient Fortification in Food Crops Using Nanoparticle Technology. Front. Plant. Sci. 2021, 12, 668819. [Google Scholar] [CrossRef]
- Beyitler, I.; Kavukcu, S. Clinical presentation, diagnosis and treatment of vulvovaginitis in girls: A current approach and review of the literature. World J. Pediatr. 2017, 13, 101–105. [Google Scholar] [CrossRef]
- Bodunde, O.P.; Ikumapayi, O.M.; Akinlabi, E.T.; Oladapo, B.I.; Adeoye, A.O.M.; Fatoba, S.O. A futuristic insight into a ?nano-doctor?: A clinical review on medical diagnosis and devices using nanotechnology. In Proceedings of the 11th International Conference on Materials, Processing and Characterization (ICMPC), Indore, India, 15–17 December 2020; pp. 1144–1153. [Google Scholar]
- McCabe, E.R.B. Nanopediatrics: Enabling Personalized Medicine for Children. Pediatr. Res. 2010, 67, 453–457. [Google Scholar] [CrossRef]
- Nieto Gonzalez, N.; Obinu, A.; Rassu, G.; Giunchedi, P.; Gavini, E. Polymeric and Lipid Nanoparticles: Which Applications in Pediatrics? Pharmaceutics 2021, 13, 670. [Google Scholar] [CrossRef]
- Varadarajan, N.; Kwon, D.S.; Law, K.M.; Ogunniyi, A.O.; Anahtar, M.N.; Richter, J.M.; Walker, B.D.; Love, J.C. Rapid, efficient functional characterization and recovery of HIV-specific human CD8+ T cells using microengraving. Proc. Natl. Acad. Sci. USA 2012, 109, 3885–3890. [Google Scholar] [CrossRef]
- Yellepeddi, V.K.; Joseph, A.; Nance, E. Pharmacokinetics of nanotechnology-based formulations in pediatric populations. Adv. Drug Deliv. Rev. 2019, 151, 44–55. [Google Scholar] [CrossRef]
- Alam Bony, B.; Kievit, F.M. A Role for Nanoparticles in Treating Traumatic Brain Injury. Pharmaceutics 2019, 11, 473. [Google Scholar] [CrossRef]
- Roth, C.C. Urologic Tissue Engineering in Pediatrics: From Nanostructures to Bladders. Pediatr. Res. 2010, 67, 509–513. [Google Scholar] [CrossRef]
- Irvin-Choy, N.S.; Nelson, K.M.; Gleghorn, J.P.; Day, E.S. Design of nanomaterials for applications in maternal/fetal medicine. J. Mat. Chem. B 2020, 8, 6548–6561. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Luo, Y.Y.; Wu, X.L.; Yang, L.Q.; Cui, D.D.; Wei, C.Z.; Wang, W.Z. Gold Nanoparticle-Optimized Computed Tomography Imaging Combined with Transcranial Magnetic Stimulation on Rehabilitation Training of Children with Cerebral Palsy. Sci. Adv. Mater. 2021, 13, 1674–1684. [Google Scholar] [CrossRef]
- Paul, S.; Nahar, A.; Bhagawati, M.; Kunwar, A.J. A Review on Recent Advances of Cerebral Palsy. Oxidative Med. Cell. Longev. 2022, 2022, 20. [Google Scholar] [CrossRef]
- Lyseng-Williamson, K.A. Nanofiltered human C1 inhibitor concentrate (Cinryze(R)): In hereditary angioedema. BioDrugs 2011, 25, 317–327. [Google Scholar] [CrossRef]
- Pedersen, P.H.; Vergari, C.; Tran, A.; Xavier, F.; Jaeger, A.; Laboudie, P.; Housset, V.; Eiskjaer, S.P.; Vialle, R. A Nano-Dose Protocol For Cobb Angle Assessment in Children With Scoliosis: Results of a Phantom-based and Clinically Validated Study. Clin. Spine Surg. 2019, 32, E340–E345. [Google Scholar] [CrossRef]
- Nobile, S.; Nobile, L. Nanotechnology and Early Human Development. Appl. Sci. 2020, 10, 4323. [Google Scholar] [CrossRef]
- Diem, V.T.; Tyas, M.J.; Ngo, H.C.; Phuong, L.H.; Khanh, N.D. The effect of a nano-filled resin coating on the 3-year clinical performance of a conventional high-viscosity glass-ionomer cement. Clin. Oral Investig. 2014, 18, 753–759. [Google Scholar] [CrossRef]
- Berta, G.N.; Romano, F.; Vallone, R.; Abbadessa, G.; Di Scipio, F.; Defabianis, P. An Innovative Strategy for Oral Biofilm Control in Early Childhood Based on a Resveratrol-Cyclodextrin Nanotechnology Approach. Materials 2021, 14, 3801. [Google Scholar] [CrossRef]
- Katsarov, P.; Shindova, M.; Lukova, P.; Belcheva, A.; Delattre, C.; Pilicheva, B. Polysaccharide-Based Micro- and Nanosized Drug Delivery Systems for Potential Application in the Pediatric Dentistry. Polymers 2021, 13, 3342. [Google Scholar] [CrossRef]
- Khubchandani, M.; Thosar, N.R.; Dangore-Khasbage, S.; Srivastava, R. Applications of Silver Nanoparticles in Pediatric Dentistry: An Overview. Cureus J. Med. Sci. 2022, 14, 4. [Google Scholar] [CrossRef]
- Wu, J.; Mady, L.J.; Roy, A.; Aral, A.M.; Lee, B.; Zheng, F.; Catalin, T.; Chun, Y.; Wagner, W.R.; Yang, K.; et al. In-vivo efficacy of biodegradable ultrahigh ductility Mg-Li-Zn alloy tracheal stents for pediatric airway obstruction. Commun. Biol. 2020, 3, 787. [Google Scholar] [CrossRef]
- Acharya, S.; Godhi, B.S.; Saha, S.; Singh, M.; Dinsa, K.; Bhagchandani, J.; Gautam, A. Use of nanoparticles in pediatric dentistry: A narrative review. J. Int. Oral Health 2022, 14, 357–362. [Google Scholar] [CrossRef]
- Kahraman, E.; Aydilek, N.; Gungor, S. Recent Approaches on Novel Topical Delivery Systems for Atopic Dermatitis Treatment. Recent Pat. Drug Deliv. Formul. 2020, 14, 191–200. [Google Scholar] [CrossRef]
- Parekh, K.; Mehta, T.A.; Dhas, N.; Kumar, P.; Popat, A. Emerging Nanomedicines for the Treatment of Atopic Dermatitis. AAPS PharmSciTech 2021, 22, 55. [Google Scholar] [CrossRef]
- Md, S.; Kuldeep Singh, J.K.A.; Waqas, M.; Pandey, M.; Choudhury, H.; Habib, H.; Hussain, F.; Hussain, Z. Nanoencapsulation of betamethasone valerate using high pressure homogenization-solvent evaporation technique: Optimization of formulation and process parameters for efficient dermal targeting. Drug Dev. Ind. Pharm. 2019, 45, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.P.; Jin, D.M.; Jiang, X.S.; Qiu, Y.S. Preparation of Dopamine Nanoparticles and Its Application in the Treatment of Neonatal Scleredema. Sci. Adv. Mater. 2021, 13, 1048–1057. [Google Scholar] [CrossRef]
- Ershow, A.G.; Hill, J.O.; Baldwin, J.T. Novel engineering approaches to obesity, overweight, and energy balance: Public health needs and research opportunities. In Proceedings of the 26th Annual International Conference of the IEEE-Engineering-in-Medicine-and-Biology-Society, San Francisco, CA, USA, 1–5 September 2004; pp. 5212–5214. [Google Scholar]
- Eldaw, A. Nanotechnology in elevation of the worldwide impact of obesity and obesity-related diseases: Potential roles in human health and disease. J. Diabetes Sci. Technol. 2011, 5, 1005–1008. [Google Scholar] [CrossRef] [PubMed]
- Hosny, K.M.; Banjar, Z.M.; Hariri, A.H.; Hassan, A.H. Solid lipid nanoparticles loaded with iron to overcome barriers for treatment of iron deficiency anemia. Drug Des. Devel. Ther. 2015, 9, 313–320. [Google Scholar] [CrossRef]
- Khan, I.M.; Niazi, S.; Akhtar, W.; Yue, L.; Pasha, I.; Khan, M.K.I.; Mohsin, A.; Iqbal, M.W.; Zhang, Y.; Wang, Z.P. Surface functionalized AuNCs optical biosensor as an emerging food safety indicator: Fundamental mechanism to future prospects. Coord. Chem. Rev. 2023, 474, 28. [Google Scholar] [CrossRef]
- Escarrer-Jaume, M.; Julia-Benito, J.C.; Quevedo-Teruel, S.; del-Prado, A.P.; Sandoval-Ruballos, M.; Quesada-Sequeira, F.; Alvaro-Lozano, M. Changes in epidemiology and clinical practice in IgE-mediated allergy in children. An. Pediatr. 2021, 95, 8. [Google Scholar] [CrossRef] [PubMed]
- Kumari, D.; Bhatia, E.; Awasthi, L.; Banerjee, R. Phospholipid and menthol based nanovesicle impregnated transdermal patch for nutraceutical delivery to diminish folate and iron deficiency. Biomed. Mater. 2022, 17, 044101. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.J.; Pham, K.; Li, D.; Penzak, S.R.; Dong, X.W. Novel in situ self-assembly nanoparticles for formulating a poorly water-soluble drug in oral solid granules, improving stability, palatability, and bioavailability. Int. J. Nanomed. 2016, 11, 1451–1459. [Google Scholar] [CrossRef]
- Pham, K.; Li, D.; Guo, S.J.; Penzak, S.; Dong, X.W. Development and in vivo evaluation of child-friendly lopinavir/ritonavir pediatric granules utilizing novel in situ self-assembly nanoparticles. J. Control. Release 2016, 226, 88–97. [Google Scholar] [CrossRef]
- Igartua, D.E.; Azcona, P.L.; Martinez, C.S.; Alonso, S.D.V.; Lassalle, V.L.; Prieto, M.J. Folic acid magnetic nanotheranostics for delivering doxorubicin: Toxicological and biocompatibility studies on Zebrafish embryo and larvae. Toxicol. Appl. Pharmacol. 2018, 358, 23–34. [Google Scholar] [CrossRef]
- Dupont, G.; Flament, M.P.; Leterme, P.; Farah, N.; Gayot, A. Developing a study method for producing 400 microm spheroids. Int. J. Pharm. 2002, 247, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Potrc, T.; Baumgartner, S.; Roskar, R.; Planinsek, O.; Lavric, Z.; Kristl, J.; Kocbek, P. Electrospun polycaprolactone nanofibers as a potential oromucosal delivery system for poorly water-soluble drugs. Eur. J. Pharm. Sci. 2015, 75, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Kamble, R.N.; Mehtre, R.V.; Mehta, P.P.; Nangare, P.; Patil, S.S. Albendazole Electrospun Nanofiber Films: In-vitro and Ex-vivo Assessment. BioNanoScience 2019, 9, 625–636. [Google Scholar] [CrossRef]
- Limayem Blouza, I.; Charcosset, C.; Sfar, S.; Fessi, H. Preparation and characterization of spironolactone-loaded nanocapsules for paediatric use. Int. J. Pharm. 2006, 325, 124–131. [Google Scholar] [CrossRef]
- Guillermet, E.; Alfa, D.A.; Phuong Mai, L.T.; Subedi, M.; Demolis, R.; Giersing, B.; Jaillard, P. End-user acceptability study of the nanopatch; a microarray patch (MAP) for child immunization in low and middle-income countries. Vaccine 2019, 37, 4435–4443. [Google Scholar] [CrossRef] [PubMed]
- Khan, B.A.; Mahmood, T.; Menaa, F.; Shahzad, Y.; Yousaf, A.M.; Hussain, T.; Ray, S.D. New Perspectives on the Efficacy of Gallic Acid in Cosmetics & Nanocosmeceuticals. Curr. Pharm. Des. 2018, 24, 5181–5187. [Google Scholar] [CrossRef] [PubMed]
- Biskos, G.; Schmidt-Ott, A. Airborne Engineered Nanoparticles: Potential Risks and Monitoring Challenges for Assessing their Impacts on Children. Paediatr. Respir. Rev. 2012, 13, 79–83. [Google Scholar] [CrossRef]
- Calderon-Garciduenas, L.; Reynoso-Robles, R.; Gonzalez-Maciel, A. Combustion and friction-derived nanoparticles and industrial-sourced nanoparticles: The culprit of Alzheimer and Parkinson’s diseases. Environ. Res. 2019, 176, 19. [Google Scholar] [CrossRef]
- Sharma, A.; Muresanu, D.F.; Patnaik, R.; Sharma, H.S. Size- and age-dependent neurotoxicity of engineered metal nanoparticles in rats. Mol. Neurobiol. 2013, 48, 386–396. [Google Scholar] [CrossRef]
- Kolosnjaj-Tabi, J.; Szwarc, H.; Moussa, F. Carbon nanotubes: Culprit or witness of air pollution? Nano Today 2017, 15, 11–14. [Google Scholar] [CrossRef]
- Stone, V.; Johnston, H.; Clift, M.J.D. Air pollution, ultrafine and nanoparticle toxicology: Cellular and molecular interactions. IEEE Trans. Nanobiosci. 2007, 6, 331–340. [Google Scholar] [CrossRef]
- Schuepp, K.; Sly, P.D. The developing respiratory tract and its specific needs in regard to ultrafine particulate matter exposure. Paediatr. Respir. Rev. 2012, 13, 95–99. [Google Scholar] [CrossRef]
- World Health Organization. Malaria. 2021. Available online: https://www.who.int/health-topics/malaria (accessed on 28 February 2023).
- Micronutrition Facts, Iron Deficiency. Available online: https://www.cdc.gov/nutrition/micronutrient-malnutrition/micronutrients/#iron (accessed on 28 February 2023).
- National Institute of Neurological Disorders and Stroke. Traumatic Brain Injury. 2021. Available online: https://www.ninds.nih.gov/Disorders/All-Disorders/Traumatic-Brain-Injury-Information-Page (accessed on 28 February 2023).
- American Cancer Society. Cancer in Children. 2021. Available online: https://www.cancer.org/cancer/cancer-in-children.html (accessed on 28 February 2023).
- Centers for Disease Control and Prevention. Respiratory Syncytial Virus Infection (RSV). Available online: https://www.cdc.gov/rsv/index.html (accessed on 28 February 2023).
- Crohn’s & Colitis Foundation. What Is Inflammatory Bowel Disease? Available online: https://www.crohnscolitisfoundation.org/what-is-ibd (accessed on 28 February 2023).
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760. [Google Scholar] [CrossRef]
- Torchilin, V.P. Multifunctional, stimuli-sensitive nanoparticulate systems for drug delivery. Nat. Rev. Drug Discov. 2014, 13, 813–827. [Google Scholar] [CrossRef]
- Slowing, I.I.; Vivero-Escoto, J.L.; Wu, C.W.; Lin, V.S. Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev. 2008, 60, 1278–1288. [Google Scholar] [CrossRef]
- Dreaden, E.C.; Alkilany, A.M.; Huang, X.; Murphy, C.J.; El-Sayed, M.A. The golden age: Gold nanoparticles for biomedicine. Chem. Soc. Rev. 2012, 41, 2740–2779. [Google Scholar] [CrossRef]
- Bose, S.; Roy, M.; Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554. [Google Scholar] [CrossRef]
- Lee, N.; Yoo, D.; Ling, D.; Cho, M.H.; Hyeon, T.; Cheon, J. Iron oxide based nanoparticles for multimodal imaging and magnetoresponsive therapy. Chem. Rev. 2015, 115, 10637–10689. [Google Scholar] [CrossRef]
- Prausnitz, M.R. Engineering Microneedle Patches for Vaccination and Drug Delivery to Skin. Annu. Rev. Chem. Biomol. Eng. 2017, 8, 177–200. [Google Scholar] [CrossRef]
- Liu, Z.; Robinson, J.T.; Sun, X.; Dai, H. PEGylated nanographene oxide for delivery of water-insoluble cancer drugs. J. Am. Chem. Soc. 2008, 130, 10876–10877. [Google Scholar] [CrossRef]
- Zare-Zardini, H.; Amiri, A.; Shanbedi, M.; Taheri-Kafrani, A.; Sadri, Z.; Ghanizadeh, F.; Neamatzadeh, H.; Sheikhpour, R.; Keyvani Boroujeni, F.; Masoumi Dehshiri, R.; et al. Nanotechnology and Pediatric Cancer: Prevention, Diagnosis and Treatment. Iran. J. Ped. Hematol. Oncol. 2015, 5, 233–248. [Google Scholar]
- Rodriguez-Nogales, C.; Gonzalez-Fernandez, Y.; Aldaz, A.; Couvreur, P.; Blanco-Prieto, M.J. Nanomedicines for Pediatric Cancers. ACS Nano 2018, 12, 7482–7496. [Google Scholar] [CrossRef]
- Di Ruscio, V.; Del Baldo, G.; Fabozzi, F.; Vinci, M.; Cacchione, A.; de Billy, E.; Megaro, G.; Carai, A.; Mastronuzzi, A. Pediatric Diffuse Midline Gliomas: An Unfinished Puzzle. Diagnostics 2022, 12, 2064. [Google Scholar] [CrossRef] [PubMed]
- Basha, R.; Sabnis, N.; Heym, K.; Bowman, W.P.; Lacko, A.G. Targeted nanoparticles for pediatric leukemia therapy. Front. Oncol. 2014, 4, 101. [Google Scholar] [CrossRef] [PubMed]
- Mitxelena-Iribarren, O.; Lizarbe-Sancha, S.; Campisi, J.; Arana, S.; Mujika, M. Different Microfluidic Environments for In Vitro Testing of Lipid Nanoparticles against Osteosarcoma. Bioengineering 2021, 8, 77. [Google Scholar] [CrossRef] [PubMed]
- Guido, C.; Baldari, C.; Maiorano, G.; Mastronuzzi, A.; Carai, A.; Quintarelli, C.; De Angelis, B.; Cortese, B.; Gigli, G.; Palama, I.E. Nanoparticles for Diagnosis and Target Therapy in Pediatric Brain Cancers. Diagnostics 2022, 12, 173. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.W.; Wang, W.L.; Li, S.C.; Han, L. The Effect of Zbxz23ir-21 NANO (nanomaterials) Delivery Vector on Apoptosis and PTEN (phosphatase and tensin homolog deleted on chromosome ten)/PI3K(Intracellular phosphatidylinositol kinase)/AKT (related to the A and C kinase) in Children with CHOLESTEATOMA in Middle Ear. Bioengineered 2021, 12, 8809–8821. [Google Scholar] [CrossRef]
- Kievit, F.M.; Stephen, Z.R.; Wang, K.; Dayringer, C.J.; Sham, J.G.; Ellenbogen, R.G.; Silber, J.R.; Zhang, M. Nanoparticle mediated silencing of DNA repair sensitizes pediatric brain tumor cells to gamma-irradiation. Mol. Oncol. 2015, 9, 1071–1080. [Google Scholar] [CrossRef]
- Krishnan, V.; Rajasekaran, A.K. Clinical Nanomedicine: A Solution to the Chemotherapy Conundrum in Pediatric Leukemia Therapy. Clin. Pharmacol. Ther. 2014, 95, 168–178. [Google Scholar] [CrossRef]
- Marques, M.S.; Lima, L.A.; Poletto, F.; Contri, R.V.; Guerreiro, I.C.K. Nanotechnology for the treatment of paediatric diseases: A review. J. Drug Deliv. Sci. Technol. 2022, 75, 13. [Google Scholar] [CrossRef]
- Sheth, J.; Arnoldo, A.; Zhong, Y.; Marrano, P.; Pereira, C.; Ryall, S.; Thorner, P.; Hawkins, C.; Somers, G.R. Sarcoma Subgrouping by Detection of Fusion Transcripts Using NanoString nCounter Technology. Pediatr. Dev. Pathol. 2019, 22, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Tsangaris, G.T.; Papathanasiou, C.; Adamopoulos, P.G.; Scorilas, A.; Vorgias, C.E.; Prodromou, N.; Stathopoulou, F.T.; Stravopodis, D.J.; Anagnostopoulos, A.K. Pediatric Ependymoma: A Proteomics Perspective. Cancer Genom. Proteom. 2017, 14, 127–136. [Google Scholar] [CrossRef][Green Version]
- Yang, S.C.; Wallach, M.; Krishna, A.; Kurmasheva, R.; Sridhar, S. Recent Developments in Nanomedicine for Pediatric Cancer. J. Clin. Med. 2021, 10, 1437. [Google Scholar] [CrossRef] [PubMed]
- Ruan, S.; Yuan, M.; Zhang, L.; Hu, G.; Chen, J.; Cun, X.; Zhang, Q.; Yang, Y.; He, Q.; Gao, H. Tumor microenvironment sensitive doxorubicin delivery and release to glioma using angiopep-2 decorated gold nanoparticles. Biomaterials 2015, 37, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Spreafico, F.; Bongarzone, I.; Pizzamiglio, S.; Magni, R.; Taverna, E.; De Bortoli, M.; Ciniselli, C.M.; Barzano, E.; Biassoni, V.; Luchini, A.; et al. Proteomic analysis of cerebrospinal fluid from children with central nervous system tumors identifies candidate proteins relating to tumor metastatic spread. Oncotarget 2017, 8, 46177–46190. [Google Scholar] [CrossRef]
- Marsh, I.R.; Grudzinski, J.; Baiu, D.C.; Besemer, A.; Hernandez, R.; Jeffery, J.J.; Weichert, J.P.; Otto, M.; Bednarz, B.P. Preclinical Pharmacokinetics and Dosimetry Studies of (124)I/(131)I-CLR1404 for Treatment of Pediatric Solid Tumors in Murine Xenograft Models. J. Nucl. Med. 2019, 60, 1414–1420. [Google Scholar] [CrossRef]
- Sanchez-Molina, S.; Figuerola-Bou, E.; Sanchez-Margalet, V.; de la Cruz-Merino, L.; Mora, J.; de Alava Casado, E.; Garcia-Dominguez, D.J.; Hontecillas-Prieto, L. Ewing Sarcoma Meets Epigenetics, Immunology and Nanomedicine: Moving Forward into Novel Therapeutic Strategies. Cancers 2022, 14, 5473. [Google Scholar] [CrossRef]
- Regenold, M.; Kaneko, K.; Wang, X.; Peng, H.B.; Evans, J.C.; Bannigan, P.; Allen, C. Triggered release from thermosensitive liposomes improves tumor targeting of vinorelbine. J. Control. Release 2022, 354, 19–33. [Google Scholar] [CrossRef]
- Saber, M.M.; Al-Mahallawi, A.M.; Stork, B. Metformin dampens cisplatin cytotoxicity on leukemia cells after incorporation into cubosomal nanoformulation. Biomed. Pharmacother. 2021, 143, 112140. [Google Scholar] [CrossRef]
- Zou, Y.; Mei, D.; Yuan, J.; Han, J.; Xu, J.; Sun, N.; He, H.; Yang, C.; Zhao, L. Preparation, Characterization, Pharmacokinetic, and Therapeutic Potential of Novel 6-Mercaptopurine-Loaded Oral Nanomedicines for Acute Lymphoblastic Leukemia. Int. J. Nanomed. 2021, 16, 1127–1141. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Wang, R.; Shen, Y.; Xu, J.; Webster, T.J.; Fang, Y. Personalized nanomedicine: A rapid, sensitive, and selective UV-vis spectrophotometry method for the quantification of nanostructured PEG-asparaginase activity in children’s plasma. Int. J. Nanomed. 2018, 13, 6337–6344. [Google Scholar] [CrossRef]
- Zhong, Y.; Beimnet, K.; Alli, Z.; Arnoldo, A.; Kowalski, P.E.; Somers, G.R.; Hawkins, C.; Abdelhaleem, M. Multiplexed Digital Detection of B-Cell Acute Lymphoblastic Leukemia Fusion Transcripts Using the NanoString nCounter System. J. Mol. Diagn. 2020, 22, 72–80. [Google Scholar] [CrossRef]
- Krishnan, V.; Xu, X.; Barwe, S.P.; Yang, X.; Czymmek, K.; Waldman, S.A.; Mason, R.W.; Jia, X.; Rajasekaran, A.K. Dexamethasone-loaded block copolymer nanoparticles induce leukemia cell death and enhance therapeutic efficacy: A novel application in pediatric nanomedicine. Mol. Pharm. 2013, 10, 2199–2210. [Google Scholar] [CrossRef]
- Zeng, Z.H.; Tung, C.H.; Zu, Y.L. Aptamer-Equipped Protamine Nanomedicine for Precision Lymphoma Therapy. Cancers 2020, 12, 780. [Google Scholar] [CrossRef]
- El Moukhtari, S.H.; Garbayo, E.; Fernandez-Teijeiro, A.; Rodriguez-Nogales, C.; Couvreur, P.; Blanco-Prieto, M.J. Nanomedicines and cell-based therapies for embryonal tumors of the nervous system. J. Control. Release 2022, 348, 553–571. [Google Scholar] [CrossRef]
- Rodriguez-Nogales, C.; Noguera, R.; Couvreur, P.; Blanco-Prieto, M.J. Therapeutic Opportunities in Neuroblastoma Using Nanotechnology. J. Pharmacol. Exp. Ther. 2019, 370, 625–635. [Google Scholar] [CrossRef]
- Pereira-Silva, M.; Alvarez-Lorenzo, C.; Concheiro, A.; Santos, A.C.; Veiga, F.; Figueiras, A. Nanomedicine in osteosarcoma therapy: Micelleplexes for delivery of nucleic acids and drugs toward osteosarcoma-targeted therapies. Eur. J. Pharm. Biopharm. 2020, 148, 88–106. [Google Scholar] [CrossRef]
- Santos, A.; Domingues, C.; Jarak, I.; Veiga, F.; Figueiras, A. Osteosarcoma from the unknown to the use of exosomes as a versatile and dynamic therapeutic approach. Eur. J. Pharm. Biopharm. 2022, 170, 91–111. [Google Scholar] [CrossRef]
- Li, M.R.; Lin, Z.I.; Yang, J.Y.; Huang, H.Q.; Liu, G.L.; Liu, Q.Q.; Zhang, X.M.; Zhang, Y.; Xu, Z.R.; Lin, H.M.; et al. Biodegradable Carbon Dioxide-Derived Non-Viral Gene Vectors for Osteosarcoma Gene Therapy. Adv. Healthc. Mater. 2023, 12, 15. [Google Scholar] [CrossRef]
- Yu, W.; Zhu, J.; Wang, Y.; Wang, J.; Fang, W.; Xia, K.; Shao, J.; Wu, M.; Liu, B.; Liang, C.; et al. A review and outlook in the treatment of osteosarcoma and other deep tumors with photodynamic therapy: From basic to deep. Oncotarget 2017, 8, 39833–39848. [Google Scholar] [CrossRef]
- Gonzalez-Fernandez, Y.; Imbuluzqueta, E.; Patino-Garcia, A.; Blanco-Prieto, M.J. Antitumoral-Lipid-Based Nanoparticles: A Platform for Future Application in Osteosarcoma Therapy. Curr. Pharm. Design 2015, 21, 6104–6124. [Google Scholar] [CrossRef]
- Power, E.A.; Rechberger, J.S.; Gupta, S.; Schwartz, J.D.; Daniels, D.J.; Khatua, S. Drug delivery across the blood-brain barrier for the treatment of pediatric brain tumors-An update. Adv. Drug Deliv. Rev. 2022, 185, 19. [Google Scholar] [CrossRef]
- Bukchin, A.; Sanchez-Navarro, M.; Carrera, A.; Resa-Pares, C.; Castillo-Ecija, H.; Balaguer-Lluna, L.; Teixido, M.; Olaciregui, N.G.; Giralt, E.; Carcaboso, A.M.; et al. Amphiphilic Polymeric Nanoparticles Modified with a Protease-Resistant Peptide Shuttle for the Delivery of SN-38 in Diffuse Intrinsic Pontine Glioma. ACS Appl. Nano Mater. 2021, 4, 1314–1329. [Google Scholar] [CrossRef]
- Fahmy, S.A.; Dawoud, A.; Zeinelabdeen, Y.A.; Kiriacos, C.J.; Daniel, K.A.; Eltahtawy, O.; Abdelhalim, M.M.; Braoudaki, M.; Youness, R.A. Molecular Engines, Therapeutic Targets, and Challenges in Pediatric Brain Tumors: A Special Emphasis on Hydrogen Sulfide and RNA-Based Nano-Delivery. Cancers 2022, 14, 5244. [Google Scholar] [CrossRef]
- Joshi, P.; Katsushima, K.; Zhou, R.; Meoded, A.; Stapleton, S.; Jallo, G.; Raabe, E.; Eberhart, C.G.; Perera, R.J. The therapeutic and diagnostic potential of regulatory noncoding RNAs in medulloblastoma. Neuro-Oncol. Adv. 2019, 1, 14. [Google Scholar] [CrossRef]
- Mishra, P.; Tripathi, Y.B. Impact of Nano Preparation of Phytoconstituents in Medulloblastoma. Methods Mol. Biol. 2022, 2423, 115–122. [Google Scholar] [CrossRef]
- Lim, C.; Koo, J.; Oh, K.T. Nanomedicine approaches for medulloblastoma therapy. J. Pharm. Investig. 2022, 53, 213–233. [Google Scholar] [CrossRef]
- Bredlau, A.L.; Dixit, S.; Chen, C.; Broome, A.M. Nanotechnology Applications for Diffuse Intrinsic Pontine Glioma. Curr. Neuropharmacol. 2017, 15, 104–115. [Google Scholar] [CrossRef]
- Gallud, A.; Warther, D.; Maynadier, M.; Sefta, M.; Poyer, F.; Thomas, C.D.; Rouxel, C.; Mongin, O.; Blanchard-Desce, M.; Morere, A.; et al. Identification of MRC2 and CD209 receptors as targets for photodynamic therapy of retinoblastoma using mesoporous silica nanoparticles. RSC Adv. 2015, 5, 75167–75172. [Google Scholar] [CrossRef]
- Garbayo, E.; Pascual-Gil, S.; Rodriguez-Nogales, C.; Saludas, L.; Estella-Hermoso de Mendoza, A.; Blanco-Prieto, M.J. Nanomedicine and drug delivery systems in cancer and regenerative medicine. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1637. [Google Scholar] [CrossRef]
- Woiski, T.D.; de Castro Poncio, L.; de Moura, J.; Orsato, A.; Bezerra, A.G., Jr.; Minozzo, J.C.; de Figueiredo, B.C. Anti-hMC2RL1 Functionalized Gold Nanoparticles for Adrenocortical Tumor Cells Targeting and Imaging. J. Biomed. Nanotechnol. 2017, 13, 68–76. [Google Scholar] [CrossRef]
- Russo, E.; Spallarossa, A.; Tasso, B.; Villa, C.; Brullo, C. Nanotechnology for Pediatric Retinoblastoma Therapy. Pharmaceuticals 2022, 15, 1087. [Google Scholar] [CrossRef]
- Katti, K.V. Renaissance of nuclear medicine through green nanotechnology: Functionalized radioactive gold nanoparticles in cancer therapy-my journey from chemistry to saving human lives. J. Radioanal. Nucl. Chem. 2016, 309, 5–14. [Google Scholar] [CrossRef]
- Aldossary, A.M.; Tawfik, E.A.; Alomary, M.N.; Alsudir, S.A.; Alfahad, A.J.; Alshehri, A.A.; Almughem, F.A.; Mohammed, R.Y.; Alzaydi, M.M. Recent advances in mitochondrial diseases: From molecular insights to therapeutic perspectives. Saudi Pharm. J. 2022, 30, 1065–1078. [Google Scholar] [CrossRef]
- Fatima, M.; Fakhar-e-Alam, M.; Atif, M.; Shakoor, M.N.; Afzal, M.; Waseem, M.; Aziz, M.H. Apoptotic effect of alpha-Fe2O3 and SiO2 nanoparticles in human rhabdomyosarcoma cell line. Laser Phys. 2014, 24, 5. [Google Scholar] [CrossRef]
- Krishnan, V.; Xu, X.; Kelly, D.; Snook, A.; Waldman, S.A.; Mason, R.W.; Jia, X.Q.; Rajasekaran, A.K. CD19-Targeted Nanodelivery of Doxorubicin Enhances Therapeutic Efficacy in B-Cell Acute Lymphoblastic Leukemia. Mol. Pharm. 2015, 12, 2101–2111. [Google Scholar] [CrossRef]
- Mazur, J.; Roy, K.; Kanwar, J.R. Recent advances in nanomedicine and survivin targeting in brain cancers. Nanomedicine 2018, 13, 105–137. [Google Scholar] [CrossRef]
- Zoloth, L.; Backhus, L.; Woodruff, T. Waiting to be born: The ethical implications of the generation of “NUBorn” and “NUAge” mice from pre-pubertal ovarian tissue. Am. J. Bioeth. 2008, 8, 21–29. [Google Scholar] [CrossRef]
- Nawaz, M. Nanotechnology-Based Approaches in Pediatric Parasitic Infections. J. Pediatr. Infect. Dis. 2017, 12, 264–270. [Google Scholar] [CrossRef]
- Sosnik, A.; Carcaboso, A.M. Nanomedicines in the future of pediatric therapy. Adv. Drug Deliv. Rev. 2014, 73, 140–161. [Google Scholar] [CrossRef]
- Chauhan, N.; Soni, S.; Jain, U. Optimizing testing regimes for the detection of COVID-19 in children and older adults. Expert Rev. Mol. Diagn. 2021, 21, 999–1016. [Google Scholar] [CrossRef]
- Ning, B.; Huang, Z.; Youngquist, B.M.; Scott, J.W.; Niu, A.; Bojanowski, C.M.; Zwezdaryk, K.J.; Saba, N.S.; Fan, J.; Yin, X.M.; et al. Liposome-mediated detection of SARS-CoV-2 RNA-positive extracellular vesicles in plasma. Nat. Nanotechnol. 2021, 16, 1039–1044. [Google Scholar] [CrossRef]
- Chavda, V.P.; Vuppu, S.; Mishra, T.; Kamaraj, S.; Patel, A.B.; Sharma, N.; Chen, Z.S. Recent review of COVID-19 management: Diagnosis, treatment and vaccination. Pharmacol. Rep. 2022, 74, 1120–1148. [Google Scholar] [CrossRef]
- Seifati, S.M.; Nasirizadeh, N.; Azimzadeh, M. Nano-biosensor based on reduced graphene oxide and gold nanoparticles, for detection of phenylketonuria-associated DNA mutation. IET Nanobiotechnol. 2018, 12, 417–422. [Google Scholar] [CrossRef]
- Choudhary, R. Sepsis Management, Controversies, and Advancement in Nanotechnology: A Systematic Review. Cureus J. Med. Sci. 2022, 14, 15. [Google Scholar] [CrossRef]
- Zosky, G.R. Emerging issues in the Pacific Basin. Rev. Environ. Health 2011, 26, 39–44. [Google Scholar] [CrossRef]
- Kashii, M.; Hashimoto, J.; Nakano, T.; Umakoshi, Y.; Yoshikawa, H. Alendronate treatment promotes bone formation with a less anisotropic microstructure during intramembranous ossification in rats. J. Bone Miner. Metab. 2008, 26, 24–33. [Google Scholar] [CrossRef]
- Rathore, A.; Cleary, M.; Naito, Y.; Rocco, K.; Breuer, C. Development of tissue engineered vascular grafts and application of nanomedicine. Wiley Interdiscip. Rev.-Nanomed. Nanobiotechnol. 2012, 4, 257–272. [Google Scholar] [CrossRef]
- Sharma, S.; Gupta, D.K. Tissue Engineering and Stem Cell Therapy in Pediatric Urology. J. Indian Assoc. Pediatr. Surg. 2019, 24, 237–246. [Google Scholar] [CrossRef]
- Hackmyer, S.P.; Donly, K.J. Restorative dentistry for the pediatric patient. Tex. Dent. J. 2010, 127, 1165–1171. [Google Scholar]
- Ibrahim, M.S.; AlQarni, F.D.; Al-Dulaijan, Y.A.; Weir, M.D.; Oates, T.W.; Xu, H.H.K.; Melo, M.A.S. Tuning Nano-Amorphous Calcium Phosphate Content in Novel Rechargeable Antibacterial Dental Sealant. Materials 2018, 11, 1544. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Utreja, P.; Kumar, L. Nanotechnological Carriers for Treatment of Acne. Recent Pat. Antiinfect Drug Discov. 2018, 13, 105–126. [Google Scholar] [CrossRef] [PubMed]
- Hussain, Z.; Sahudin, S.; Thu, H.E.; Shuid, A.N.; Bukhari, S.N.; Kumolosasi, E. Recent Advances in Pharmacotherapeutic Paradigm of Mild to Recalcitrant Atopic Dermatitis. Crit. Rev. Ther. Drug Carrier. Syst. 2016, 33, 213–263. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.; Mohanty, N.; Kadam, N.N.; Swain, N.; Thakur, M. Green Synthesis to Develop Iron-Nano Formulations and Its Toxicity Assays. J. Pharmacopunct. 2020, 23, 165–172. [Google Scholar] [CrossRef]
- Vishvakarma, V.; Kaur, M.; Nagpal, M.; Arora, S. Role of Nanotechnology in Taste masking: Recent Updates. Curr. Drug Res. Rev. 2022, 15, 1–14. [Google Scholar] [CrossRef]
- Carter, M.D.; Calcutt, M.W.; Malow, B.A.; Rose, K.L.; Hachey, D.L. Quantitation of melatonin and n-acetylserotonin in human plasma by nanoflow LC-MS/MS and electrospray LC-MS/MS. J. Mass Spectrom. 2012, 47, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Clark, G.M. Personal reflections on the multichannel cochlear implant and a view of the future. J. Rehabil. Res. Dev. 2008, 45, 651–693. [Google Scholar] [CrossRef]
- Dvorackova, J.; Bielnikova, H.; Kukutschova, J.; Peikertova, P.; Filip, P.; Zelenik, K.; Kominek, P.; Uvirova, M.; Pradna, J.; Cermakova, Z.; et al. Detection of nano- and micro-sized particles in routine biopsy material—Pilot study. Biomed. Pap. 2015, 159, 87–92. [Google Scholar] [CrossRef]
- He, R.K.; Zhang, J.; Mainelis, G. Resuspension of particles deposited by nano-enabled consumer sprays: The role of product type, flooring material, and resuspension force. Indoor Air 2022, 32, 17. [Google Scholar] [CrossRef]
- Sheybani, N.D.; Yang, H. Pediatric ocular nanomedicines: Challenges and opportunities. Chin. Chem. Lett. 2017, 28, 1817–1821. [Google Scholar] [CrossRef]
- Hwang, G.; Marsh, G.; Gao, L.; Waugh, R.; Koo, H. Binding Force Dynamics of Streptococcus mutans-glucosyltransferase B to Candida albicans. J. Dent. Res. 2015, 94, 1310–1317. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Sarin, L.; Johnson, N.C.; Hurt, R.H. A nano-selenium reactive barrier approach for managing mercury over the life-cycle of compact fluorescent lamps. Environ. Sci. Technol. 2009, 43, 5915–5920. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Beqa, L.; Singh, A.K.; Khan, S.A.; Senapati, D.; Arumugam, S.R.; Ray, P.C. Gold Nanoparticle-Based Simple Colorimetric and Ultrasensitive Dynamic Light Scattering Assay for the Selective Detection of Pb(II) from Paints, Plastics, and Water Samples. ACS Appl. Mater. Interfaces 2011, 3, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Zang, X.W.; Wu, Z.Z.; Liu, J.X.; Wang, D.M. Altered protein S-glutathionylation depicts redox imbalance triggered by transition metal oxide nanoparticles in a breastfeeding system. NanoImpact 2021, 22, 14. [Google Scholar] [CrossRef]
- Tang, S.; Wang, M.; Germ, K.E.; Du, H.M.; Sun, W.J.; Gao, W.M.; Mayer, G.D. Health implications of engineered nanoparticles in infants and children. World J. Pediatr. 2015, 11, 197–206. [Google Scholar] [CrossRef]
- Waldron, A.M.; Spencer, D.; Batt, C.A. The current state of public understanding of nanotechnology. J. Nanopart. Res. 2006, 8, 569–575. [Google Scholar] [CrossRef]
- Bussy, C.; Ali-Boucetta, H.; Kostarelos, K. Safety Considerations for Graphene: Lessons Learnt from Carbon Nanotubes. Acc. Chem. Res. 2013, 46, 692–701. [Google Scholar] [CrossRef]
- Quadros, M.E.; Pierson, R.T.; Tulve, N.S.; Willis, R.; Rogers, K.; Thomas, T.A.; Marr, L.C. Release of silver from nanotechnology-based consumer products for children. Environ. Sci. Technol. 2013, 47, 8894–8901. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).