Abstract
Indoxyl sulfate (IxS) and p-cresyl sulfate (pCS) are toxic uremic compounds with documented pathological outcomes. This review critically and comprehensively analyzes the available liquid chromatography-mass spectrometry methods quantifying IxS and pCS in human matrices and the biological applications of these validated assays. Embase, Medline, PubMed, Scopus, and Web of Science were searched until December 2023 to identify assays with complete analytical and validation data (N = 23). Subsequently, citation analysis with PubMed and Scopus was utilized to identify the biological applications for these assays (N = 45). The extraction methods, mobile phase compositions, chromatography, and ionization methods were evaluated with respect to overall assay performance (e.g., sensitivity, separation, interference). Most of the assays focused on human serum/plasma, utilizing acetonitrile or methanol (with ammonium acetate/formate or formic/acetic acid), liquid–liquid extraction, reverse phase (e.g., C18) chromatography, and gradient elution for analyte separation. Mass spectrometry conditions were also consistent in the identified papers, with negative electrospray ionization, select multiple reaction monitoring transitions and deuterated internal standards being the most common approaches. The validated biological applications indicated IxS and/or pCS were correlated with renal disease progression and cardiovascular outcomes, with limited data on central nervous system disorders. Methods for reducing IxS and/or pCS concentrations were also identified (e.g., drugs, natural products, diet, dialysis, transplantation) where inconsistent findings have been reported. The clinical monitoring of IxS and pCS is gaining significant interest, and this review will serve as a useful compendium for scientists and clinicians.
1. Introduction
Kidneys are responsible for filtering the blood of waste products such as endogenous uremic compounds, which are accumulated in patients with impaired renal function [1,2]. Chronic kidney disease (CKD) can cause the elevation of uremic toxins (UTs) [2] and affects around 37 million people in the United States and 8–16% of the population worldwide [2,3]. Unfortunately, 90% of adults with CKD are not aware of their diagnosis [3]. CKD is pathologically identified as the presence of one or more kidney dysfunction markers for a duration of three or more months of (i) estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73 cm2, (ii) increased albumin excretion rates up to 30 mg/24 h, or more, (iii) increased albumin-to-creatinine ratio up to 30 mg/g, or more, and (iv) others (e.g., hematuria, structural abnormalities) [4,5]. eGFR is an important marker commonly used for the classification of CKD stages, where normal kidney function (“G1”) is defined as eGFR ≥ 90 mL/min/1.73 cm2, CKD stage G2 (eGFR = 60–89 mL/min/1.73 cm2), CKD stage G3a (eGFR = 45–59 mL/min/1.73 cm2), CKD stage G3b (eGFR = 30–44 mL/min/1.73 cm2), CKD stage G4 (eGFR = 15–29 mL/min/1.73 cm2), and CKD stage G5 (eGFR < 15 mL/min/1.73 cm2) [4].
The European Uremic Toxins Work Group (EUTox) identified a list of 130 UTs and provided a framework for classification [6,7,8,9]. The uremic toxins can be classified according to their physiochemical properties such as size and binding affinities [10]: (i) free water-soluble low molecular weight compounds (<0.5 kD), (ii) middle molecules (0.5–60 kD), and (iii) protein-bound uremic toxins (PBUTs) [7]. The latter class is known for its inefficient clearance during typical dialysis protocols [11,12]. In addition, Rosner et al. [13] suggested the use of a different classification system according to hemodialysis techniques, dialysis membranes, and patterns of solute removal [13]. Moreover, Rosner et al. [13] recommended the use of the 2018 classification, which relies on the biological outcomes of uremic toxins [6,13]. In this classification, the uremic toxins were given scores according to the reported experimental evidence on their toxicities [6]; for example, some small water-soluble solutes can have high toxicity scores [6]. Of interest to this review are the two sulfonated PBUTs, indoxyl sulfate (IxS) and p-cresyl sulfate (pCS), which have gained significant research interest in the last decade due to their toxicological impacts on serum accumulation (Figure 1). We refer the readers to several excellent reviews on the toxicology of IxS and pCS (e.g., [6,14,15,16,17,18,19]), which are known to be associated with cardiovascular outcomes in CKD patients [20], colonic cancer [15], oxidative stress [21], and the progression of CKD [17,19]. Recently, Vanholder et al. [6] have also ranked both IxS and pCS as uremic compounds of significant toxicological interest.
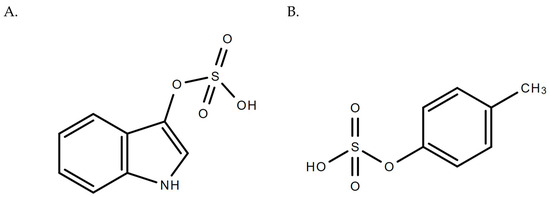
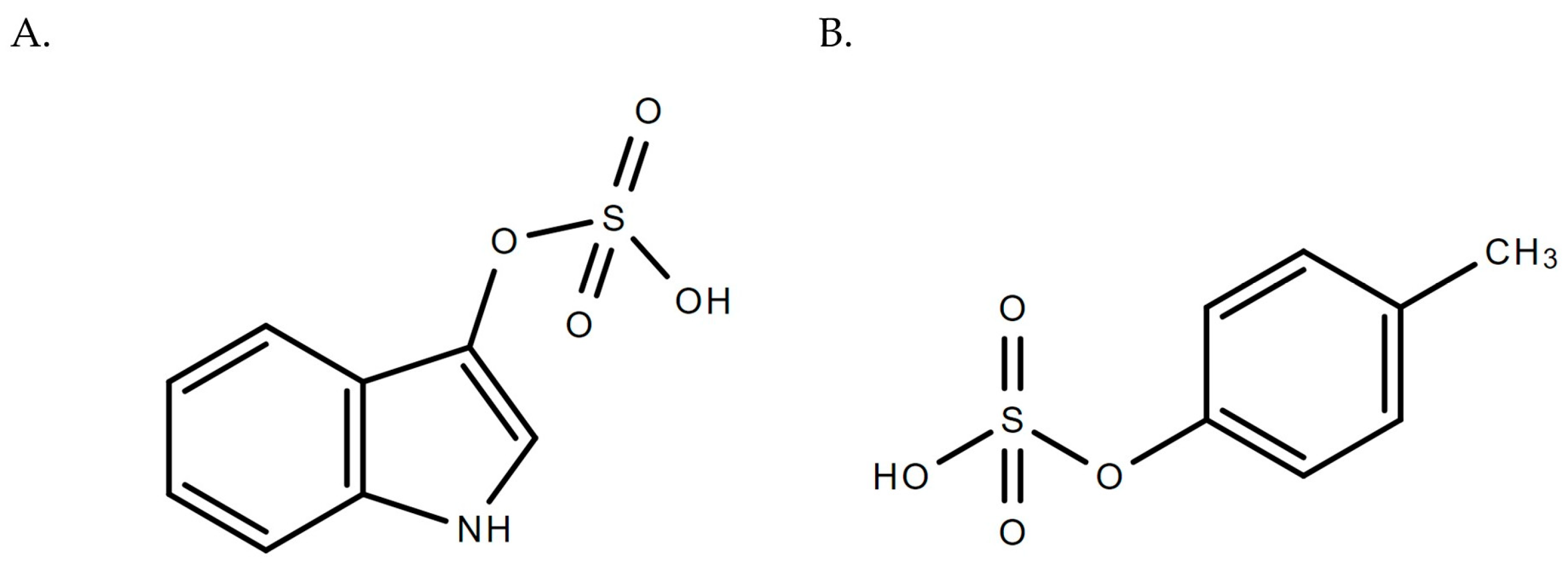
Figure 1.
Chemical structures for (A) indoxyl sulfate and (B) p-cresyl sulfate.
IxS, initially called “indican”, has a molecular weight of 213 g/mol, and its protein binding characteristics were reported to be 90% or more [19]. IxS remains at elevated concentrations even after dialysis due to its extensive protein-binding behavior [22]. IxS is produced endogenously from intestinal microflora that metabolizes tryptophan to form indole, which undergoes hepatic hydroxylation by CYP2E1 and conjugation by SULT1A1 to form IxS [14]. Similar to IxS, pCS (molecular weight 188.2 g/mol) is generated in the liver after the sulfonation of p-cresol (pC), a gut-derived metabolite of tyrosine and phenylalanine fermentation [17,23]. pCS binds to albumin (median percentage protein binding > 90%) and is extensively retained in the body in cases of abnormal low renal excretion [17,24]. Both IxS and pCS are excreted into the urine via renal tubular secretion [25]. In patients with renal dysfunction, the accumulation of IxS and pCS may lead to the manifestation of toxicities in various organ and tissue systems (e.g., [6,14,15,16,17,18,19]). Further examples include significant correlations observed between urine IxS concentrations and urinary oxidative stress markers such as 15-isoprostane F2t and pteridine in patients with type 2 diabetes mellitus [26] and elevated plasma pCS concentrations being associated with ischemic strokes in hemodialysis patients [27]. Therefore, monitoring IxS and pCS concentrations can potentially aid the diagnosis, prevention, and management of uremia-induced pathophysiology. The literature provides various analytical techniques to quantify IxS and pCS, such as ultra-performance liquid-chromatography (UPLC), LC-MS/MS, and enzyme-linked immunosorbent assays (ELISA) (e.g., [28,29,30]). However, LC-MS/MS assays are considered the most sensitive and selective, with the ability to quantify IxS and pCS in the nanomolar concentrations (e.g., [31,32]).
Several excellent reviews have been published around these subject areas. Fernandes et al. provided an overview of various analytical assays separating IxS, pCS, pC, and indole-3-acetic acid (IAA) [8], focusing on a wide array of sample preparation, chromatographic, and detection methods [8]. Heaney [33] provided discussions on the principles and examples of mass spectrometry-based analytical assays to quantify gut-derived bacterial metabolites and a variety of classes of uremic toxins [33]. Niwa provided general discussions on mass-spectrometry data for both IxS and pCS [34,35]. The current critical review provides a comprehensive update with a focus on LC-MS/MS assays validated specifically for IxS and pCS in human clinical samples. Our review discusses in further detail all relevant assay parameters such as sample handling, matrix, chromatography, mass-spectrometry, and validation. Additionally, our review provides a critical analysis of the clinical applications related to these validated assays, with a focus on potential diagnostic applications. This review is tailored to pharmaceutical analytical scientists with interest in assay development and also to clinicians who are interested in managing the pathophysiology of IxS and pCS-associated uremic toxicities.
2. Materials and Methods
Embase, Medline, PubMed, Scopus, and Web of Science were searched until December 2023 for articles included in this review using combinations of the following keywords: liquid chromatography-mass spectrometry (LC-MS/MS), high-pressure liquid chromatography (HPLC), and protein-bound uremic toxins (PBUT). The initial search resulted in 2,445,849 papers, which was reduced to 2,428,677 papers that only focused on PBUTs. Of these, 2,286,696 papers were in English, from which we included 9749 papers that analyzed human biological matrices. Further exclusion criteria were (i) database duplications (N = 9382 remaining); (ii) lack a clear description of validation parameters as outlined in the United States Food and Drug Administration guidance document [36] (N = 7161 remaining); (iii) lack of quantitative mass-spectrometry detection (N = 129 remaining), and (iv) PBUTs not deemed of clinical toxicological interest based on a recent critical review [6] (N = 47 remaining). Of these, we further narrowed the focus to 23 papers specifically tailored to IxS and/or pCS (Figure 2). Data pertaining to the biological matrix, sample processing, chromatography parameters, mass spectrometry detection, and validation were extracted for further analysis.

Figure 2.
Literature screening, exclusion, and inclusion. PBUTs: protein-bound uremic toxins. LC-MS/MS: high-performance liquid chromatography-mass spectrometry. IxS: indoxyl sulfate. pCS: p-cresyl sulfate. N: total number of papers remaining. PBUTs of the highest toxicological interest were determined according to a recent critical review [6].
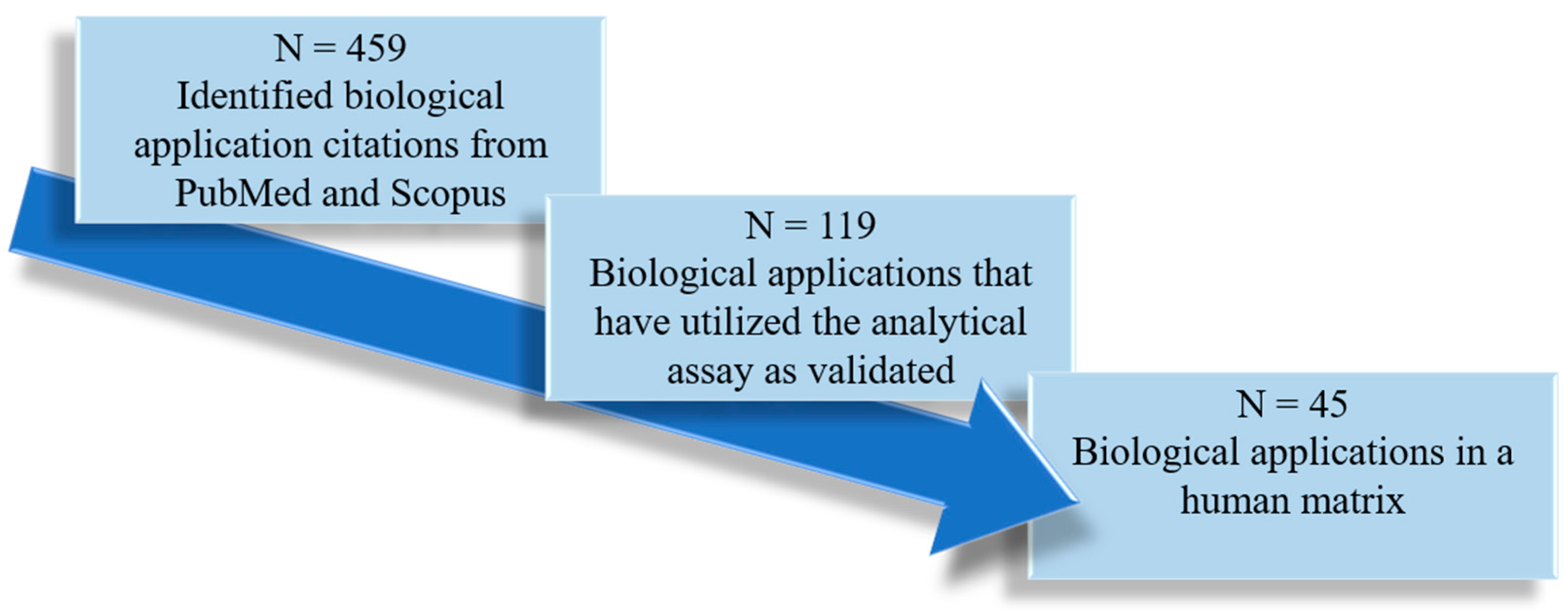
The biological applications reported within these analytical assays were also objectively summarized and discussed in this review. Additional applications of these analytical methods were identified in PubMed and Scopus by systematic citation analysis (i.e., searching for indexed peer-reviewed papers that have cited these 23 analytical assays as they were originally validated). A total of 45 biological applications were identified and further categorized based on the human biological matrix. If the biological applications did not use the cited analytical assay as originally developed/validated, they were excluded from our analysis (Figure 3). Data pertaining to experimental objectives, population characteristics, and the authors’ key findings were extracted for analysis.
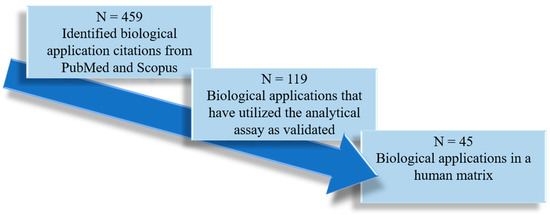
Figure 3.
Literature screening, inclusion, and exclusion for the human biological applications of the validated high-performance liquid chromatography-mass spectrometry assays quantifying indoxyl sulfate and p-cresyl sulfate (in reference to Figure 2). N: total number of biological applications remaining.
3. Results and Discussion
3.1. Analytical Assays in Serum and Plasma
Most of the methods that quantify IxS and/or pCS were validated in serum (N = 11), plasma (N = 5), or combined (N = 2). The majority of the assays quantified additional analytes (including IxS and/or pCS in the mix); however, some assays focused only on either IxS, pCS, or the combination of these two sulfonated metabolites [29,37,38,39] (Table 1).

Table 1.
Summary of validated LC-MS/MS assays for quantifying indoxyl sulfate and p-cresol sulfate in human biological matrices.
3.1.1. Sample Preparation
For assays in serum and/or plasma, the most common sample extraction procedure is liquid–liquid extraction (LLE), which includes the use of a protein precipitation solution typically containing the internal standards. The choice of the organic solvent depended mainly on the organic phase used in the liquid chromatography mobile phase and consisted primarily of acetonitrile (ACN) and/or methanol. Both IxS and pCS contain aromatic rings (i.e., indole and benzene) and a polar sulfonate functional group; therefore, methanol and ACN could possibly provide both dispersion [59] and electrostatic interactions [60], supporting their common usage in sample extraction (Table 1).
To our knowledge, the extraction efficiency of the different organic solvents for IxS and pCS has not been systematically tested. However, ACN appears to be utilized in more analytical assays (Table 1), and these assays had slightly longer run times (e.g., 2.5–20 min, Table 1) than assays that used methanol (e.g., 6–10.1 min, Table 1) when data were provided. With respect to assay sensitivity, the use of ACN for the extraction of serum and plasma was associated with LLOQs of ~20–500 ng/mL [37,46,50], whereas methanol was associated with LLOQs of the most sensitive assays being 1 ng/mL for IxS and pCS in serum [31,32] and ~50 ng/mL for IxS in plasma [51]. Different approaches were used to measure the matrix effects (e.g., with/without sample extraction, the choice of the blank matrix/neat solution) (Table 1) with inconsistencies across the studies; therefore, it may not be possible to compare the results directly between different papers. Based on Table 1, the use of ACN was accompanied by a matrix effect of <20% for IxS and pCS in most of the assays (e.g., [44,46]). However, a significant matrix effect of more than 20% was reported by André et al. [45], whereas methanol also had a matrix effect <20% for IxS and pCS [31,51], except for Monosik et al. [49]. Overall, there is no consistent pattern in the usage of a particular solvent for the quantification of IxS and/or pCS, and no clear trends in recovery efficiency were associated with a specific organic solvent, where 80–120% has been reported for IxS or pCS recovery in ACN and in methanol except for Wang et al. [47] and Zhu et al. [32] which reported the recovery outside of this range. On the other hand, both ACN- and methanol-processed samples can be placed at –20 °C or –80 °C for long-term storage, with most assays being stable up to three freeze–thaw cycles for both organic solvents (Table 1). An exception was reported for IxS stability after three freeze–thaw cycles performed by André et al. [45]. While there is no clear advantage of one organic solvent over the other, it should be cautioned, however, that these comparisons are only qualitative in nature as a properly controlled experiment is required to test the overall effects of ACN vs. methanol for extraction.
On the other hand, solid-phase extraction (SPE) was also used in some assays [43,51], which typically requires additional hardware, such as the Oasis WAX μElution plate [51] and the 3cc HLB cartridge [43]. In Oda et al. [51], the Oasis WAX μElution plate is pre-conditioned with methanol and water, the plasma sample is loaded into the plate after being mixed with the working solution, the plate is washed with 2% aqueous formic acid, and the analytes are eluted with 0.25% aqueous ammonia solution in methanol. This protocol generated a linearity range of 0.05–200 μg/mL for IxS with an extraction recovery corrected to the internal standards as 100.7%, 101.9%, and 101.6% for 0.15, 4.5, and 150 μg/mL, respectively [51]. In Zhang et al. [43], serum samples were loaded into a 3cc HLB cartridge conditioned with methanol and formic acid (1%), the cartridge washed with formic acid (1%), and the analytes eluted with ammonium peroxide (1%) in methanol: water (50:50 v/v%). The supernatant is then evaporated with an N2 stream and subsequently reconstituted with ammonium acetate (5 mM). This method generated a linearity range between 0.021–106.605 μg/mL for IxS and an extraction recovery of 108.7% and 115.3% for 33.3 and 10 µmol, respectively [43]. Based on the limited available data, it is difficult to determine which SPE protocol provided better assay performance, and it is not clear whether these protocols could also be applied to pCS. The LLE also does not require specific extraction cartridges, which may result in more cost savings.
To ensure efficient extraction, various mechanical means (e.g., incubation, vortex mixing, ultrasonication, and centrifugation) are utilized for sample cleanup and protein removal (Table 1). Most assays that used methanol for extraction tended to incubate the sample with the extraction solution (15–60 min) [32,38,49], but this approach was not commonly performed with ACN. This might be due to the C≡N group in ACN, which is a stronger nucleophile compared to the OH group, thereby minimizing the required contact time. On the other hand, some assays used a longer vortex time in place of having a separate incubation period, but it is not apparent whether this approach affects the extraction efficiency or sensitivity [37,49,52]. Likewise, the impact of ultrasonication is not clear since it was used only in one assay [50]. Centrifugation was a common step in all assays with a wide range of utilized speed and time, with no apparent advantage of any specific approach (Table 1). Drying/sample concentrating was typically conducted with a nitrogen stream or, less commonly, with vacuum evaporation (Table 1).
Both IxS and pCS are highly protein-bound [17,19], and it is more toxicologically relevant to quantify the unbound concentrations that are considered biologically active. However, only a few assays have quantified the free concentration (Table 1), likely due to the much longer sample processing time, higher costs, and the requirement for additional ultracentrifugation steps. Ultracentrifugation was the common approach to isolate the free fractions of IxS and pCS. As the molecular weights of the bound fraction exceed 60,000 Da [31], the filters commonly had a molecular weight cut-off (MWCO) of 3000–30,000 Da. In Boelaert et al. [40], the Millipore Centrifree device with an MWCO of 30,000 Da was used for ultracentrifugation at 1469× g for 25 min. The LLOQ was 75 and 95 ng/mL for IxS and pCS, respectively [40]. The same device was employed by Ma et al. [46] with higher centrifugation speed (3500× g) and longer time (30 min), with a corresponding LLOQ of 20 and 19 ng/mL for IxS and pCS, respectively [46]. In Lin et al. [29], the serum was centrifuged with a Merck Millipore filter with a MWCO of 3000 Da at 10,621× g for 30 min to provide an LLOQ of 50 ng/mL for both IxS and pCS [29]. On the other hand, in Shu et al. [37], serum samples were centrifuged at 20,000× g for 5 min with an unspecified device [37]. The LLOQ was 500 and 50 ng/mL for IxS and pCS, respectively [37]. Fabresse et al. [31] utilized the ultra-centrifugal 30,000 Da filter (Amicon, Merck) with centrifugation at 13,300× g for 20 min and reported relatively higher sensitivity with an LLOQ of 1 ng/mL for both IxS and pCS [31]. The sensitivity of the latter assay may also be attributed to their sample preparation technique (i.e., complete evaporation of the extracted sample and reconstitution with pure solvent (Table 1)) or more sensitive mass spectrometry parameters [31]. Generally, the quantification of unbound IxS and pCS does not only require extra sample preparation but needs assays with higher sensitivity as the serum/plasma free fraction for IxS and pCS are relatively low at 5–10% of the total concentration [61]. According to the literature, the IxS and pCS-free concentrations can be as low as 80 and 150 ng/mL in CKD patients, respectively (e.g., [29]).
3.1.2. Liquid Chromatography
The composition of the mobile phase depends on the instrument hardware, method of ionization, and utilized analytical column. In all assays discussed in this review, an aqueous phase containing additives (discussed below) is utilized (Table 1) due to the water solubility of IxS and pCS [62,63]. The organic solvent of choice is either methanol or ACN (Table 1) due to favorable chemical interactions with IxS and pCS already discussed above. As there was a lack of a systematic evaluation/comparison of different organic solvents in a single setting, it was not clear why most assays favored methanol or ACN. Likewise, data/descriptions on chromatography (i.e., resolution, background interference, retention factor, etc.) were not always available, which limited a direct comparison between methanol vs. ACN. However, ACN was used as the organic phase in more assays (Table 1), including a few assays that had used methanol in their sample extraction process [31,32,38]. Additionally, ACN was employed as the organic phase in assays with relatively shorter run times (i.e., <6 min) quantifying a mixture of IxS and pCS [29,37,39,44,45], compared to the run time (i.e., 6–22 min) achieved by assays utilizing methanol as the organic phase [40,42,43,49,52,53]. This is likely due to the potentially stronger interaction between ACN and IxS or pCS, as already discussed above. By comparing the available chromatograms, the majority of assays utilizing methanol as the organic phase exhibited relatively sharp/narrow peaks [40,42,43,53]. However, although most assays utilizing ACN illustrated sharp peaks, some exceptions were identified. For example, background interference and/or some asymmetry were observed in IxS and pCS peaks [45,46,51]. Notably, most assays using ACN and methanol, which reported less than ideal chromatographic peaks, were all tailored to analyte mixtures, perhaps due to the complexity of having to separate multiple compounds (e.g., [45,46,49]).
The mobile phase additives included the inorganic ammonium acetate or ammonium formate salts at concentrations between 2–10 mM and formic acid or acetic acid mainly at a concentration of 0.1% v/v (Table 1). The ammonium salts, acting as buffers, were commonly added to the aqueous phase but not the organic solvents, likely due to solubility (e.g., ammonium acetate is soluble in aqueous media up to ~148 g/100 cc and much less so in methanol ~7.89 g/100 cc [64]). Ammonium salts appear to affect the separation and sensitivity of IxS and pCS but not the total run time. Assays utilizing ammonium salts had an IxS-pCS separation between 0.58–1.7 min; total run between 4–20 min; IxS LLOQ between 21–500 ng/mL; and pCS LLOQ between 50–508 ng/mL (Table 1). Ammonium salt additives were utilized in both isocratic and gradient mobile phase conditions (Table 1). On the other hand, assays without ammonium salts had IxS–pCS separation of 0.04–0.47 min, at a total run time of between 2.5 and 22 min, and both IxS and pCS LLOQ ranged between 1 and 100 ng/mL. Few assays had chromatograms with asymmetric peaks or background interference (as discussed before). Of these assays, some utilized ammonium salts, such as Oda et al. [51], and some did not use ammonium salts, such as Ma et al. [46]. Therefore, other than the slightly longer separation between IxS and pCS, the impacts of ammonium salts on the overall chromatography (i.e., sensitivity, resolution, total run time…etc.) are not apparent. Furthermore, formic acid and acetic acid are added to adjust the pH of the mobile phase but not to all assays [37,43,52]. Assays with formic acid or acetic acid had wide LLOQ ranges between 1 and 405 ng/mL for IxS and 1 and 508 ng/mL for pCS. These assays provided an IxS–pCS separation of 0.04–1.4 min (Table 1). Similarly, the LLOQ values for assays that lacked acid additives were 21–500 ng/mL for IxS and 50 ng/mL for pCS [37,43,52], and the IxS–pCS separation was between 0.58–1.7 s. Therefore, the presence of formic or acetic acid does not appear to affect IxS and pCS sensitivity and separation (Table 1), which is consistent with the discussion provided by Fernandes et al. [8]. The assays utilizing formic or acidic acid in the mobile phase provided variable chromatogram shapes, where some assays provided asymmetrical peaks with tailing (e.g., [45,46,51]), but others did not show apparent abnormalities (e.g., [38,50]). Similarly, the assays with no formic or acidic acid also provided relatively sharp peaks with little fronting or tailing (e.g., [37,43,52]); therefore, the presence of these acids might not be essential for chromatography.
Isocratic elution was used by Prokopienko et al. [44] (10 mM ammonium formate [85%]: ACN [15%]), Shu et al. [37] (10 mM ammonium acetate [90%]: ACN [10%]), and Ma et al. [46] (acetic acid [0.1%] in water [60%]: ACN [40%]) [37,44,46]. It was common for the analytes to elute at higher aqueous compositions (Table 1). The run times were ~4, 5, and 8 min, respectively, where the IxS LLOQs were 200, 500, and 20 ng/mL, and the pCS LLOQs were 250, 50, and 19 ng/mL, respectively (Table 1). The columns and flow rates were BEH C18 (1.7 µm, 2.1 × 100 mm; 0.3 mL/min), SB-C18 (3.5 µm, 2.1 × 100 mm; 0.3 mL/min), and Poroshell 120 HILIC (4.6 µm, 2.7 × 100 mm; 0.6 mL/min), respectively. The use of higher organic compositions in Ma et al. [46] appeared to be associated with lower LLOQs for both IxS and pCS (20.14 ng/mL and 19.54 ng/mL, respectively) [46] compared to LLOQs higher than 50.00 ng/mL for both IxS and pCS in Shu et al. [37] and Prokopienko et al. [44]; whereas better separation (1.7 and 1.19 s) between IxS and pCS was evident at a lower flow rate (0.3 mL/min) in Shu et al. [37] and Prokopienko et al. [44] compared to 0.08 s in the Ma et al. [46] (0.6 mL/min). The higher IxS and pCS sensitivities at higher organic composition could be due to the interacting effects with ACN. Furthermore, all isocratic assays exhibited chromatograms with sharp peaks and negligible background interference except for Ma et al. [46] where minor asymmetrical peaks were evident for pCS [46], which might be attributed to the use of the HILIC column, which may exhibit different interactions with sulfonated analytes compared to C18 columns [50].
On the other hand, the majority of the assays utilized a gradient composition (Table 1), which typically starts with higher concentrations of the aqueous phase with increasing concentrations of the organic phase and re-equilibrium to baseline conditions over time. When gradients are used, both IxS and pCS are mostly eluted at relatively higher concentrations of the organic phase (e.g., ~60–95% [42,43,45]), with the flow rate ranging between 0.2–0.8 mL/min, total run times from 2.5–22 min, LLOQs between 1 and 405 ng/mL and 1 and 508 ng/mL for IxS and pCS, respectively, and the separation between IxS and pCS from 0.04 to 1.4 min (Table 1). Additionally, the chromatography was not ideal in some assays (e.g., [45,49,51]). As such, although gradient conditions were more commonly used in complex assays for quantifying multiple analytes, there does not appear to be a clear advantage over isocratic conditions for analyzing IxS and pCS with respect to chromatography, run time, or sensitivity (Table 1). However, in assays that quantify multiple analytes (in addition to IxS and pCS), gradient conditions may be better suited for separating IxS and pCS from other uremic toxins. For example, in de Loor et al. [42], IxS and pCS eluted at 95% of the organic phase, and five other analytes eluted at 3–16% of the organic phase [42].
Almost half of the analyzed gradient assays used a stable flow rate of ≤0.3 mL/min, with some assays reaching up to 0.8 mL/min (Table 1). On the other hand, two assays used a varying flow rate of up to 0.6 mL/min [49,50] (Table 1) and reported LLOQs of 19 and 405 ng/mL for IxS and 24 and 508 ng/mL for pCS. The separation between IxS and pCS was 0.14 and 1.15 s, which was achieved in a total run time of 6 and 20 min. Consistently, assays with fixed flow rates reported LLOQs between 1 and 156 ng/mL for IxS and 1 and 100 ng/mL for pCS. The separation between IxS and pCS in fixed-flow rate assays varied between 0.04–1.4 min achieved in a total run time between 2.5 and 22 min (Table 1). Therefore, it is not clear whether there is any utility for changing the flow rate during the run for analyzing IxS and pCS. However, a possible advantage is the separation of multiple analytes. For example, Monosik et al. [49] were able to elute three analytes at a flow rate of 0.55 mL/min, four additional analytes at 0.43 mL/min (including IxS and pCS), and the remaining analyte at 0.6 mL/min [49]. Additionally, varying flow rates can potentially shorten the total run time for multiple-analyte assays by controlling the timings of rate changes tailored to analyte elution and column re-equilibration.
The elution of IxS and pCS typically utilizes reverse-phase chromatography due to the lipophilic and charged interactions between the analytes and the column. Kanemitsu et al. [50] investigated different types of columns for the quantification of uremic toxins, including IxS and pCS [50]. The tested columns included the XSELECT CSH C18 (2.1 mm, 150 mm, 3.5 µm), Luna Phenyl-Hexyl (2 mm, 100 mm, 3 µm), SeQuant ZIC-HILIC (2.1 mm, 50 mm, 3.5 µm), Scherzo SM-C18 (2.1 mm, 100 mm, 3.5 µm), Scherzo SW-C18 (2.1 mm, 100 mm, 3.5 µm), and Scherzo SS-C18 (2.1 mm, 100 mm, 3.5 µm). According to the authors, C18 and Phenyl-Hexyl columns resulted in poor separation for higher polarity toxins such as trimethylamine-N-oxide (TMAO), whereas IxS and pCS could only be poorly resolved [50]. The ZIC-HILIC column produced shorter retention times for IxS and pCS [50], and the chromatograms showed some tailing was observed in the IxS and pCS peaks. Chromatograms generated by Scherzo SW-C18 produced evident tailing in pCS peaks, whereas chromatograms from Scherzo SM-C18 had narrower peaks for IxS and pCS [50]. According to Kanemitsu et al. [50], the Scherzo SS-C18 column produced better retention for IxS compared to pCS, resulting in ideal separation with a distinct order of elution, likely due to the ability of IxS to generate both “hydrophobic and ionic interactions” with this column [50]. Generally, broader but symmetric peaks were associated with Scherzo SS-C18 for IxS and pCS.
The particle size ranged between 1.7 and 5 µm (Table 1). Columns with particle sizes of 1.7–2.7 µm provided an IxS–pCS separation of 0.08–1.4 min, which was similar to the separation of 0.04–1.7 min from columns with particle sizes between 3 and 5 µm (Table 1). Furthermore, assays utilized 100 mm columns (eight assays) [29,31,37,40,44,46,47,49], 50 mm columns (six assays) [39,42,43,45,50,51], or longer columns (210 mm [38] and 150 mm [32,52,53]). IxS–pCS separation from columns ≥100 mm ranged between 0.08 and 1.7 min, which was only slightly longer than the 50 mm columns (0.04–1.15 min). On the other hand, the total run time for columns ≥100 mm ranged between 4–22 min, which was similar to 2.5–20 min associated with the 50 mm columns (Table 1). As mentioned before, asymmetric chromatography was associated with some assays [45,46,49,51] with no apparent correlation with column particle size and length (Table 1).
3.2. Analytical Assays in Other Matrices
Although the majority of methods quantifying IxS and/or pCS were validated for serum and plasma, assays in urine [32,49,54,55] and saliva [56,57,58] were also available (Table 1).
3.2.1. Sample Preparation
Extraction protocols are required in urine and saliva for sample cleanup (Table 1). For urine, centrifugation may be required for the initial sample processing before the addition of the precipitation solution [49,55], and sample dilution was common in all urine assays in this review (Table 1). The LLOQ for IxS and pCS ranged between 1 and 480 ng/mL and 1 and 24 ng/mL, respectively, whereas the ULOQ for IxS and pCS ranged between 9.86 and 48 µg/mL and 1 and 12.47 µg/mL, respectively (Table 1). According to the literature, the urine concentrations of IxS and pCS in kidney transplant patients were 48.4 ± 52.39 µM (10.32 ± 11.17 µg/mL) and 37.2 ± 65.91 µM (7.00 ± 12.40 µg/mL), respectively (e.g., [65]). Where the serum concentrations of IxS and pCS for the same population were 1.9 ± 2.9 µM (0.41 ± 0.62 µg/mL) and 4.1 ± 5.76 µM (0.77 ± 1.08 µg/mL), respectively [65]. Therefore, urine sample dilution may be required due to the significantly higher concentrations of IxS and pCS observed in the urine. The correlations between IxS and pCS serum concentrations and their respective urine concentrations were statistically significant (p < 0.0329) and (p < 0.0001), respectively [65]. Although urine sample collection is not invasive, it is not convenient (i.e., necessitating additional privacy) and requires various collection standards (e.g., clean location and appropriate transportation) to avoid sample contamination.
On the other hand, saliva requires additional assay steps for sample collection and preparation. It is important to consider that a collecting tool (e.g., cotton swab) is needed, and either stimulated or unstimulated saliva may be utilized [56,57,58]. Three saliva assays were identified in our review (Table 1), and similar to plasma/serum, both ACN and methanol were used in the extraction process. Nam et al. [58] studied the effects of three extraction solvents (methanol, ACN, and methanol–ACN mixture) and reported ACN to be superior as it resulted in the lowest intensities for large molecules (m/z > 550), suggesting better removal of proteins [58]. Giebułtowicz et al. [56] incubated saliva samples in ACN for 20 min for deproteinization and reported a comparable matrix effect compared to Fabresse et al. [57], where methanol was used for the extraction without an incubation step. The other sample preparation steps were generally comparable to those described in serum/plasma assays (Table 1).
The LLOQs for saliva assays were 1–1.5 for IxS and 1–3.7 ng/mL for pCS (Table 1), with ULOQ reaching 50 µg/mL for both IxS and pCS (Table 1). In the literature, the IxS saliva concentrations in kidney transplant patients are 21–33 ng/mL, whereas the pCS saliva concentrations are 46–57 ng/mL [66]; therefore, these assays exhibit sufficient sensitivities. The serum IxS and pCS concentrations for the same population were 1715–2075 ng/mL and 4697–7103 ng/mL, respectively [66], in which a significant univariate correlation was evident between saliva and serum concentrations of IxS and pCS [66]. The limitations associated with saliva samples include (i) contamination, (ii) interactions with food, beverages, and oral bacteria, (iii) multiple factors affecting the salivary flow rate such as the time of collection (e.g., day vs. night), gender, and hydration level, and (iv) interactions with patients’ concurrent medications (e.g., adverse effects such as mouth dryness from anticholinergics) and health conditions (sympathetic and parasympathetic systems) [67,68].
3.2.2. Liquid Chromatography Conditions
In the assays validated by Monosik et al. [49] and Zhu et al. [32], the same liquid chromatography conditions were used for serum/plasma as for urine within the same assay [32,49], indicating no additional specific requirements for urine after sample preparation. On the other hand, the reviewed saliva assays were only validated in this specific matrix. Overall, the chromatographic conditions were generally comparable between assays that have been specifically validated in urine or saliva and in serum or plasma (Table 1) with respect to mobile phase (both ACN and methanol were used) and column, leading to consistent assay performance parameters such as LLOQ (as low as 1 ng/mL for both IxS and pCS) (Table 1).
3.3. Mass Spectrometry
In addition to the high sensitivity provided by LC-MS/MS, it has significantly better selectivity compared to other analytical techniques (e.g., ultra-performance liquid chromatography). Electrospray negative ionization (ESI) with multiple reaction monitoring (MRM) was the most common method for detecting IxS and pCS (Table 1). Atmospheric pressure chemical ionization (APCI) requires the analyte to be volatile, and it was not utilized in the reviewed papers as it is not suitable for polar analytes such as IxS and pCS [69]. None of the assays used positive ionization to quantify IxS or pCS (Table 1), and this was expected since this approach is more suitable for amino groups, amides, or carbonyls [70], which are not associated with IxS and pCS. The reported mass-to-charge (m/z) transitions were consistent between the assays: IxS from 212 → 80 (the sulfate group) or 212 → 132 (the indoxyl group) and pCS from 187 → 80 (the sulfate group) or 187 → 107 (the p-cresol). The assays that used ammonium salts also reported comparable MRM transitions, reflecting a low probability for adduct formation (Table 1). Various internal standards were utilized, including hippuric acid–d5 [40,46], indole-3-acetic acid–d5 [40], tryptophan–d3 [53], p-toluene sulfonic acid [38,52], and 13C6 ([13C6-IxS]) [45] (Table 1), but IxS-d4 and pCS-d7 were most used (Table 1) and provided the ideal internal reference standard due to identical physiochemical characteristics as the original un-deuterated analytes.
Unique mass spectrometry parameters were evident in the reviewed assays (Table 1); however, some parameters were commonly observed (when data were provided). For example, nitrogen was frequently used as the nebulizing gas [31,37,42] and argon as a collision gas [31,39,40,42,49,50,51], with some exceptions where nitrogen was utilized for collision [37,47,58]. Across the assays (Table 1), the source temperature ranged between 105 and 600 °C. The LLOQ range for assays with a source temperature of <350 °C was 19–500 ng/mL for IxS and 1–95 ng/mL for pCS. The LLOQ for assays with a source temperature of ≥350 °C were 1–100 ng/mL for both IxS and pCS. The ion spray voltage was typically between 1500 and 5000 V. Overall, the assay development data for ionization parameters have generally not been provided in the reviewed papers (Table 1), making it difficult to assess their effects within each assay.
3.4. Assay Validation
A variety of guidelines were followed for assay validation, with the Food and Drug Administration (FDA) [71] and European Medicines Agency (EMA) [72], both previous versions, being the most commonly used (Table 1). Although the validation guidelines are generally similar, some differences can be observed; therefore, assays considered validated based on different standards may still exhibit inconsistent performances. For example, back conversion is not required by this version (i.e., year 2018) of the FDA document [71,73], and EMA provides more specific suggestions for the use of isotope-labeled internal standards in mass-spectrometry [72,73]. Furthermore, some validation criteria might not be completely consistent between these two versions of the guidance documents (e.g., matrix effect) [71,72], and indeed, different methods for detecting/reporting matrix interference have been noted in the reviewed papers, which may lead to inconsistencies in data interpretation. Overall, the majority of the reviewed assays have provided accuracy and precision, stability, recovery, and matrix effect, however, some assays did not provide full validation data (Table 1). Table 1 provides summaries of the assay validation results.
3.5. Analytical Assay Summary and Future Directions
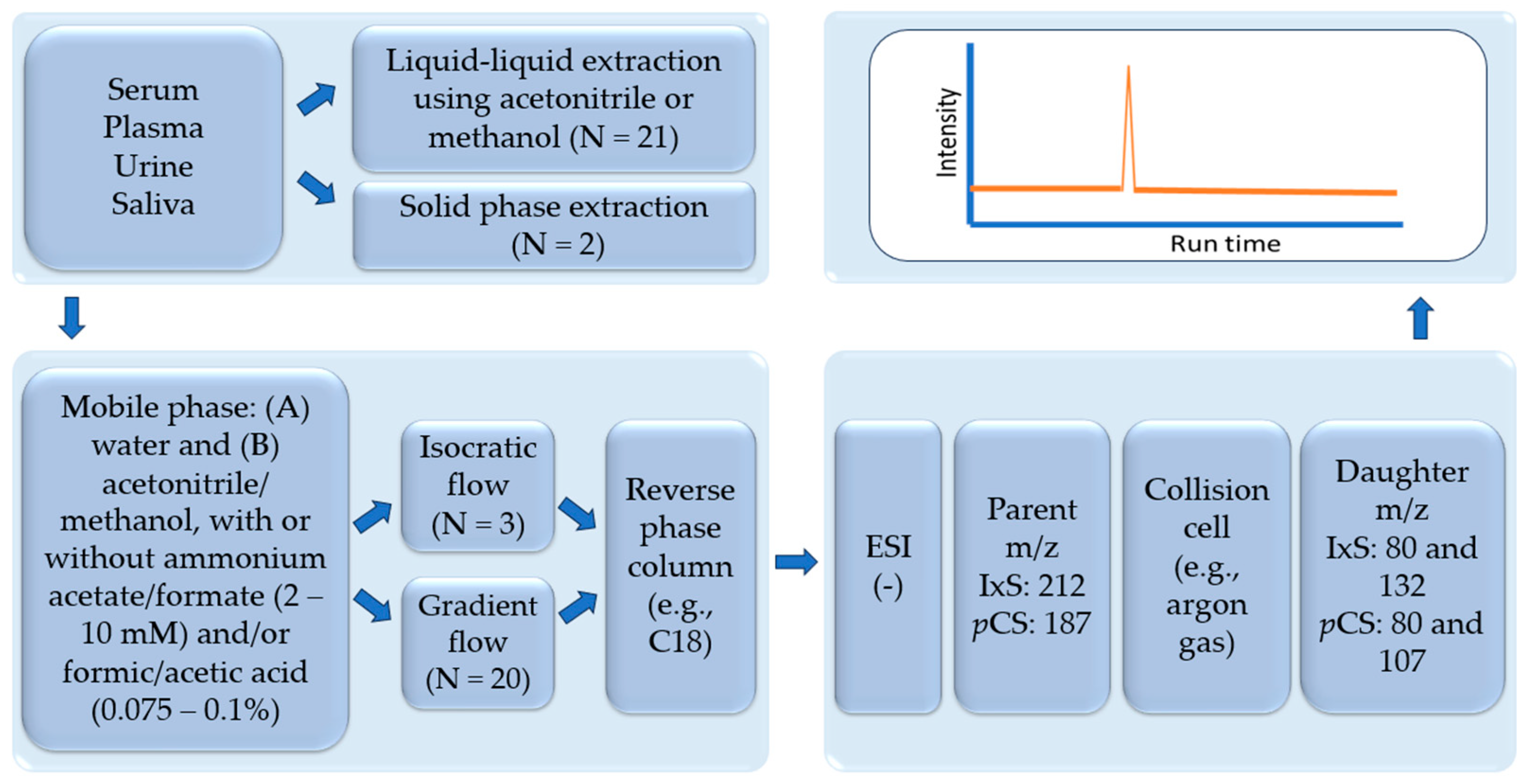
The review critically summarizes the published LC-MS/MS assays for quantifying IxS and pCS in human biological samples, including sample preparation protocols for both unbound and total concentrations (a summary figure of common workflow is presented in Figure 4; see Table 1 and text for specific differences). Liquid–liquid extraction is the most common approach and is typically conducted with methanol or ACN; however, a systematic comparison between a variety of organic extraction solvents is not yet available in the literature and would benefit future assays. The mobile phase of choice in all reviewed biological matrices consisted of water with either methanol or ACN with formic acid, acetic acid, and/or ammonium acetate. According to our analysis, these mobile phase additives generally do not affect the chromatography of IxS and pCS. The most commonly used analytical columns had a C18 stationary phase, which produced sufficient separations between IxS and pCS. A comparison between the different column parameters, in addition to the coating material, would also benefit future assays trying to analyze these uremic toxins. Gradient and isocratic conditions are both suited for separating IxS and pCS, although gradients are more commonly used if multiple toxins are being separated/analyzed. Negative electrospray ionization was used in all assays with consistent MRM transitions for IxS and pCS but variable ionization conditions are observed across the studies. Further controlled experiments are also needed to determine the most optimal ion source conditions for ionizing IxS and pCS. While the majority of assays have been developed in serum/plasma, analytical assays in saliva and urine are also available. The sample preparation procedures should be tailored to these specific biological samples, but the chromatographic and mass-spectrometry conditions remain comparable across the variety of matrices reviewed.
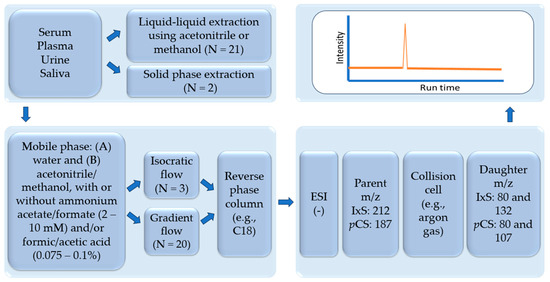
Figure 4.
Summary of common assay preparation, chromatography, and mass spectrometry conditions summarized in this review. ESI: electrospray ionization. IxS: indoxyl sulfate. pCS: p-cresyl sulfate. N: number of LC-MS/MS assays in each category. (–): negative ionization mode.
3.6. Clinical Data Obtained Using the IxS and pCS LCMS/MS Assays Validated in Human Biological Matrices
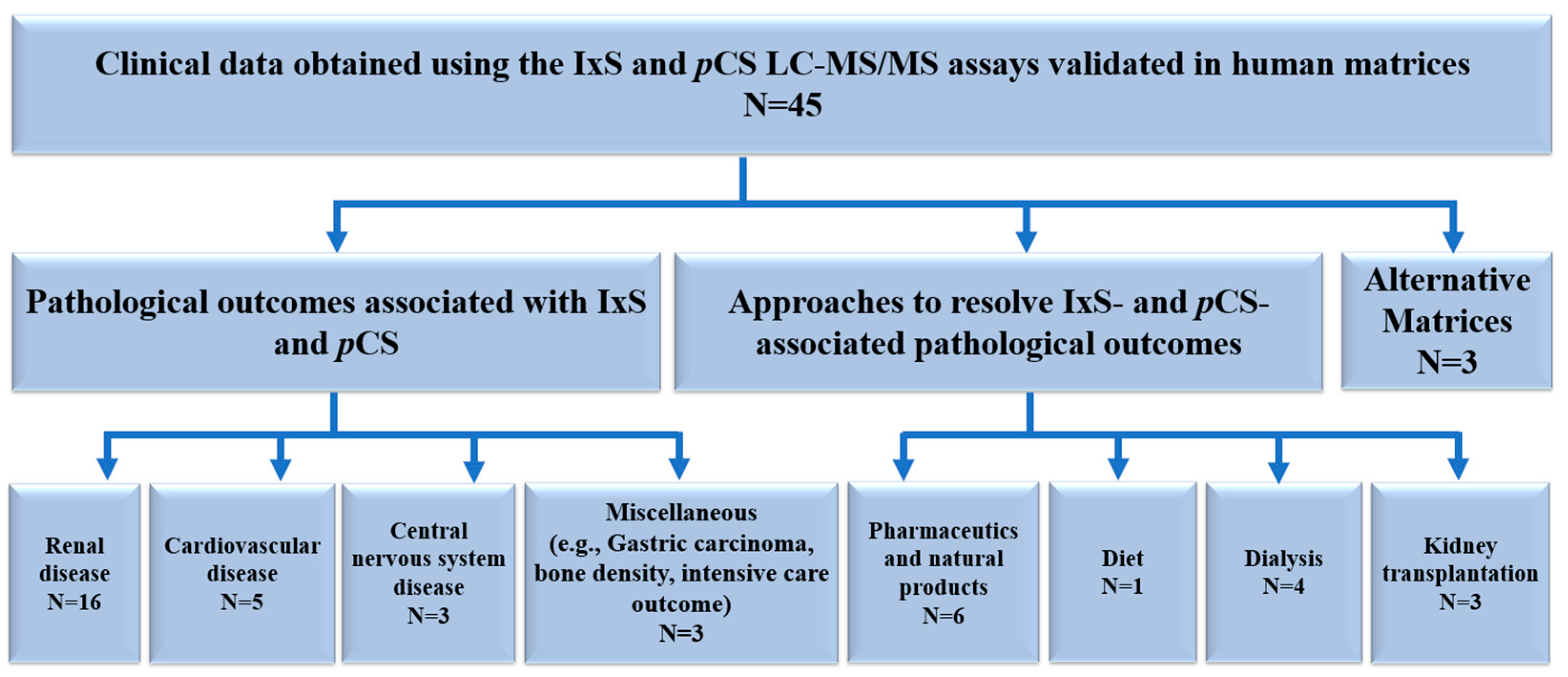
The accumulations of IxS and pCS are thought to be associated with organ and tissue malfunctions. The LC-MS/MS assays included in this review were employed in the quantification of IxS and pCS in various indications (Table 2). As discussed in the methods section, only the biological applications using the fully validated LC-MS/MS assays cited in Table 1 were further examined (Table 2). Figure 3 summarizes the biological application literature review process, citing the validated LC-MS/MS assays included in our paper. The data were categorized based on pathological outcomes associated with IxS and pCS, approaches used to mitigate these outcomes, and alternative matrices for quantifying IxS and pCS (Figure 5).

Table 2.
Associated biological applications for the validated LC-MS/MS assays for quantifying indoxyl sulfate and p-cresol sulfate.
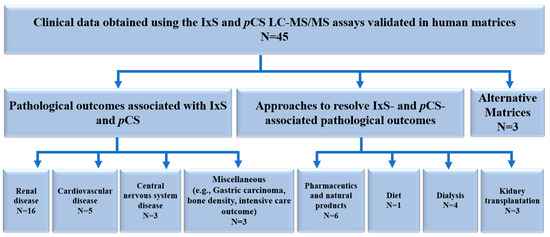
Figure 5.
Summary of the biological application data citing the validated LC-MS/MS assays included in the review. IxS: indoxyl sulfate. pCS: p-cresyl sulfate. LC-MS/MS: liquid chromatography-mass spectrometry. N: number of biological applications in each category.
3.6.1. Liquid Chromatography Conditions
The biological applications were classified according to organ systems (Figure 5, Table 2). The accumulations of IxS and pCS were mostly associated with renal insufficiency; however, additional organ pathologies have also been identified (Figure 5).
Renal Disease
Several studies have correlated the concentrations of IxS and pCS with symptoms of uremic syndrome secondary to CKD progression. IxS and pCS total serum concentrations in healthy individuals in the reviewed data were 0.25–3.02 µg/mL and 0.29–9.87 µg/mL, respectively (Table 2). IxS and pCS serum concentrations increase proportionally with CKD progression. This is evident in Boelaert et al. [40], who reported the progressive accumulations of IxS and pCS serum concentrations in pre-dialysis patients in CKD stages 1 to 5. Significant elevations were observed at stage 3 (IxS: 1.7 ± 1.1 µg/mL and pCS: 20.0 ± 7.3 µg/mL), reaching up to 4.6 ± 2.1 µg/mL and 26.5 ± 9.8 µg/mL, respectively, at stage 5 for patients not on dialysis [40]. These represent significant elevations compared to IxS (0.5 ± 0.3 µg/mL) and pCS (6.6 ± 3.7 µg/mL) serum concentrations in healthy controls within the same study [40]. Accumulations of IxS and pCS are also observed in serum and plasma samples at CKD stages 4 and 5 by Massy et al. [96] and Lin et al. [29] and at stages 3–5 in Oda et al. [51]. Although de Loor et al. [42] and Cuoghi et al. [38] (Table 2) did not provide detailed information pertaining to each CKD stage, the average data for pre-dialysis patients with CKD illustrated that IxS and pCS concentrations were generally elevated compared to healthy individual data in the literature.
Patients on dialysis were also reported to have accumulated concentrations of IxS and pCS. IxS and pCS serum/plasma concentrations in patients on hemodialysis were reported to be 3.5 ± 1.5 µg/mL to 23.74 µg/mL and 16.68 ± 8.8 µg/mL to 55.9 ± 15.1 µg/mL, respectively (Table 2). In patients on peritoneal dialysis, IxS and pCS serum/plasma concentrations were 17.6 ± 8.7 µg/mL to 32.33 ± 20.18 µg/mL and 4.6 ± 2.7 µg/mL to 23.90 ± 19.87 µg/mL, respectively (Table 2). Concentrations of IxS and pCS in hemodialysis patients were significantly higher than that reported in healthy volunteers and patients with chronic kidney disease (e.g., [40,51,82]) (more discussion regarding dialysis is available in Section Dialysis). Consistently, significant inverse correlations were observed in a CKD population between serum IxS or pCS concentrations and eGFR in de Loor et al. [42]. Similar data were reported for serum or salivary IxS or pCS and eGFR by Korytowska et al. [66] and serum IxS and eGFR by Wang et al. [85]. However, Shu et al. [37] only reported significant correlations between serum-free and total IxS, but not pCS, with eGFR.
The accumulation of IxS and pCS concentrations in different CKD stages is typically observed before the complete loss of kidney function, which suggests that monitoring IxS and pCS concentrations might be a potential approach for the detection of CKD progression. According to Lin et al. [29], IxS and pCS concentrations can be a predictable tool for CKD development and can be used as a therapeutic treatment for delaying the need for dialysis [29]. Furthermore, creatinine-based GFR calculations may not be the best marker for detecting early stages of CKD, and it may result in the overestimation of renal function, for example, in patients with liver diseases [99]. In addition, these sulfonated PBUTs could act as diagnostic markers for acute kidney injury (AKI) as suggested by André et al. [45], as the authors reported IxS and pCS serum concentrations to be elevated significantly in 8 patients who developed AKI within 72 h of acute care admission [45]. Therefore, IxS and pCS may potentially serve as better biomarkers for measuring renal function in patients with renal disease, but this would require extensive clinical and outcome validation in the clinic.
Various potential mechanisms by which IxS and pCS may lead to the worsening of CKD have been documented in the literature. As examples, IxS and pCS could generate oxidative stress and/or enhance free radical production, as evident by in vitro and in vivo experiments [100,101,102,103]. Mouse proximal renal tubular cells treated with IxS and pCS express genes related to inflammation response, which may be a compensatory response to oxidative stress injury [104]. Moreover, 5/6-nephrectomized rats administered IxS exhibit elevated mRNA of tubulointerstitial fibrosis-related genes [105], and IxS is also capable of generating the production of free radicals in rodent mesangial cells [106]. Additional pathological mechanisms include IxS and pCS activating both the renal renin-angiotensin-aldosterone system and the transforming growth factor β1 pathway, leading to an injured kidney [107]. Interactions with cellular transporters were also evident in Mutsaers et al. [108] in proximal renal tubular epithelial cells, where pCS was shown to inhibit multidrug resistance protein 4 and the breast cancer resistance protein [108]. These transporters are responsible for the renal uptake and therefore the excretion of drugs, metabolites, and endogenous molecules [108].
On the other hand, the impairment in kidney function can also lead to the accumulation of IxS and pCS, which may create a pathological feedback loop, resulting in further oxidative stress and the eventual worsening of kidney function. As IxS and pCS accumulations are significant around CKD stage 3 [40], this would indicate that earlier stages of CKD (i.e., initiation of disease state) were accompanied by normal serum levels of IxS and pCS, supporting the notion that renal dysfunction might possibly precede the accumulation of these PBUTs.
Cardiovascular Disease
IxS and pCS accumulation in CKD is potentially associated with the progression or development of cardiovascular disease (CVD), as evident by, for example, lipid profile [75], arterial stiffness [85], thrombosis [86], cardiovascular autonomic function [90], and fat deposits in skeletal muscle [83] (Table 2). Hobson et al. [75] reported that IxS and pCS were negatively correlated (univariately) with total cholesterol, low-density lipoprotein (LDL), and non-HDL in 611 patients with kidney failure [75] (Table 2). Wang et al. [85] demonstrated that IxS was an independent predictor of arterial stiffness in CKD patients using multiple regression, where categorical analysis also indicated CKD patients exhibiting arterial stiffness had significantly higher IxS concentrations [85]. Furthermore, the accumulation of IxS in CKD patients was associated with thrombosis, indicated by greater activities of aryl-hydrocarbon-receptor (AHR) and tissue factor (TF) as demonstrated by Kolachalama et al. [86] (Table 2). Cheng et al. [90] reported that significant reductions in IxS concentrations after peritoneal dialysis were associated with improvements in sympathetic/parasympathetic imbalance in 26 patients [90]. Keddar et al. [83] reported that a significant reduction in muscle radiation attenuation was associated with cardiovascular events in CKD that are also linked with the accumulation of pCS [83]. In this study, muscle radiation attenuation is an indicator of the amount of fat deposits in cardiac muscles, and higher values indicate lower fat deposits [83]. Moreover, the development of kidney failure was associated with lower muscle radiation attenuation, and kidney transplantation was able to elevate the muscle radiation attenuation [83]. Therefore, the relationship between muscle radiation attenuation and pCS concentrations may be mediated by renal function. However, the cause-effect relationships between these variables still remain to be established.
Potential mechanisms have been identified for the effects of IxS and pCS on cardiovascular outcomes in CKD patients. IxS is proposed to increase the uptake of oxidized LDL in the THP-1 macrophages, which facilitates the development of atherosclerosis [109]. IxS was reported to induce the proliferation of rat vascular smooth muscle cells and human aortic smooth muscle cells [110,111], potentially leading to vascular dysfunction. Consistent evidence is observed in rats where IxS contributes to the aortic wall thickness and aortic calcification [112], and pCS leads to vascular remodeling with oxidative stress induction, potentially leading to the development of cardiovascular events [113].
Central Nervous System Disease
CKD is also associated with nervous system complications [114]. For example, a study with 225 CKD patients on dialysis revealed that 16.4% of subjects experienced uremic neuropathy [115]. Table 2 included three studies reporting associations between IxS and pCS and central nervous system (CNS) complications in CKD patients. Lai et al. [87] reported that free IxS was independently associated with the composite amplitude score, which determines the severity of peripheral nerve dysfunctions in 54 stages 3–4 CKD patients (50% with diabetes; data not available with pCS), although no significant differences in IxS and pCS concentrations were reported in patients with diabetes compared to patients without diabetes [87] (Table 2). IxS and pCS can cause oxidative stress [100,101,102,103], and according to Lai et al. [87], IxS may exert similar pathophysiological effects resulting in neuropathy [87]. Furthermore, Olesova et al. [55] quantified urinary IxS and pCS (and other PBUTs) and reported significantly higher IxS concentrations in children 6–10 years old with autism spectrum disorder (ASD) compared to age-matched controls [55] (Table 2). According to the authors, a potential explanation might be that ASD is associated with changes in the microbiota and, therefore, may affect IxS production and concentrations [55]. However, further investigations and clinical studies are required before IxS can be considered as a diagnostic biomarker for ASD. On the other hand, te Linde et al. reported significant reductions in serum uremic toxins, including (IxS and pCS) with renal transplantation, but the improvement in cognitive function was not observed [76]. Therefore, further mechanistic experiments are warranted to determine how IxS and pCS affect cognitive functions in patients and whether the effects are clinically relevant.
Miscellaneous
CKD also impacts other organ systems or conditions. Table 2 summarizes the studies relating IxS and pCS concentrations to the gut microbiota, bone density, and ICU outcomes. Choi et al. [53] investigated the use of tryptophan metabolites as potential biomarkers for the development of gastric carcinoma [53] and reported plasma IxS to be significantly lower in gastric cancer patients [53]. However, the mechanisms and the cause-effect relationships between IxS and gastric cancer development remain to be further proven. Batteux et al. [94] characterized osteoporosis as measured by bone mineral density (BMD) and by bone fractures after transplantation [94] (Table 2). It was evident that serum IxS concentrations at the time of transplantation exhibited a positive relationship with BMD in the femoral neck and the total hip at one-month post-transplant [94], but pCS did not generate significant correlations. On the other hand, there were no associations between uremic toxins measured at the time of transplantation and BMD or bone fracture after up to 24 months post-transplantation [94]. According to the authors, the reduction in BMD after transplantation could be a result of multiple factors other than uremia, including changes in the body mass index, serum parathyroid hormone, and calcium concentrations in serum [94]. Kuo et al. [97] investigated the relationship between urine IxS concentrations and outcomes in the intensive care unit (ICU) [97]. It was evident that ICU patients who had lower urine IxS concentrations after 72 h of ICU admission were associated with reduced ICU-free days and higher mortality at one year [97]. However, the underlying mechanisms behind this association and whether serum concentrations held the same relationship remain unknown. Also, additional studies are needed to rule out all potential confounding effects that may be present in the ICU population.
3.6.2. Approaches to Mitigate IxS and pCS Associated Pathological Outcomes
Table 2 summarizes potential approaches to reduce the exposure of IxS and pCS, including pharmaceuticals and natural products, diet, dialysis, and kidney transplantation.
Pharmaceutics and Natural Products
Biruete et al. [77] investigated the effects of the prebiotic inulin for 4 weeks on gut microbiota composition and gut-derived metabolites in 12 CKD patients on hemodialysis in a cross-over study with maltodextrin as control [77] (Table 2). No significant effects on fecal p-cresol and indole or serum IxS and pCS were identified with inulin administration compared to maltodextrin [77]. However, both the experimental and the control interventions resulted in distinct patterns of microbiome changes [77]; therefore, maltodextrin may not be an appropriate control, as suggested by the authors [77]. Nazzal et al. [79] reported that the administration of oral vancomycin reduced the concentrations of IxS and pCS within 4 weeks of treatment [79]. According to the authors, this may be attributed to vancomycin modulating the microbiome, affecting the formation of IxS and pCS [79]. For example, they reported reductions in Clostridium and Bacteroides which could be responsible for the production of indole that is further metabolized to IxS [79]. However, the effects would have to be reproduced in a larger study with a balanced control group, as the authors indicated potential carryover effects of vancomycin, which precluded the use of half of their control samples [79].
Additional pharmacological approaches to mitigate IxS and pCS accumulation have been suggested (Table 2), including the use of adsorbents such as AST-120, which aim to adsorb the amino acids responsible for the final production of IxS and pCS [116]. Hsu et al. [91] investigated the effects of AST-120 in CKD patients at stages 4 and 5 [91] but did not observe significant changes in IxS and pCS concentrations despite trends of reducing pCS [91]. The literature data also provided inconsistent results for AST-120. For example, Schulman et al. reported AST-120 delayed the progression of composite CKD endpoints, including the need for dialysis, transplantation, and doubling of serum creatinine [117], but the study did not appear to have included uremic toxin concentrations. On the other hand, Cha et al. [118] reported that the administration of AST-120 did not delay the manifestation of CKD primary endpoints in patients receiving standard care [118] and that AST-120 did not affect serum IxS concentrations (despite a trend towards a reduction). Similarly, Akizawa et al. [119] also reported no effects of AST-120 on the progression of CKD [119] during 12 months of follow-up, but the study also did not measure uremic toxin concentrations. The potential inconsistent effects of AST-120 are very well-documented in the review article written by Asai et al. [120].
The Shen-Shuai-Ning tea was tested for its effects in reducing uremic toxins in CKD patients on peritoneal dialysis [84], where a significant decrease in serum total IxS, but not pCS, concentrations were reported [84]. The divergent effects on total vs. free concentrations may support distinct pharmacokinetic interacting mechanisms such as intrinsic clearance or binding displacement, but further investigations are needed to support these effects. According to the authors, this tea potentially increases bowel movement, which enhances the excretion of toxins [84], but limited data are available in the literature on its effects on IxS and pCS concentrations. On the other hand, Dai et al. [80] reported that sevelamer therapy in patients with ESRD was associated with significantly higher concentrations of IxS but not pCS [80]. The unexpected relationship with sevelamer, which is typically used to reduce phosphate concentrations in CKD patients, was proposed by the authors to be caused by interactions with the gut environment metabolism and transit times [80]. Biruete et al. [121] also reported in their review the inconsistent effects of sevelamer on IxS and pCS [121], indicating more studies are required to establish its efficacy on uremic toxin reduction. El Chamieh et al. [95] investigated the associations between proton pump inhibitors and uremic toxin concentrations in CKD patients [95]. Higher concentrations of total and free IxS and pCS were observed in patients receiving proton pump inhibitors, but a significant association with IxS was only observed using multiple linear regression [95]. According to the authors, proton pump inhibitors exhibit high affinities for OAT transporters, which are also responsible for the excretion of IxS and pCS, thus potentially explaining this observed interaction [95]. However, the observation was only correlational in nature, and baseline differences between the proton pump inhibitor groups vs. the comparator group were evident, which could potentially confound the findings.
Diet
Diet is also an approach to control serum exposure to IxS and pCS. In Table 2, Wu et al. [88] investigated the effects of low protein diet on gut microbiota and the generation of IxS and pCS in the CKD population [88]. Although significant changes were reported in gut microbiome composition between the experimental and control groups, the study did not demonstrate a difference in serum IxS and pCS concentrations in the low-protein diet group [88]. According to the authors, the small sample size can be a reason for the negative effects [88], but the general lack of difference in uremic toxin concentrations between CKD and health controls may also potentially mask their findings. In the literature, Yang et al. [122] suggested in their review that the intake of dietary fiber from vegetable and fruit sources could reduce IxS formation [122]. Takkavatakarn et al. [123] also discussed strategies to reduce IxS and pCS, including the use of a (very) low-protein diet [123], but reported inconsistent findings for IxS.
Dialysis
Dialysis is the artificial mechanism for removing toxins from the biological system; however, PBUTs are not effectively cleared with conventional dialysis. Ma et al. [46] reported the efficacy of hemodialysis in eliminating IxS and pCS to be 24.00 ± 18.29% and 34.28 ± 24.38%, respectively [46] (Table 2). According to Ma et al., the ineffective hemodialysis removal of these PBUTs is due to their high protein binding ratio and high affinity toward serum albumin [46]. Consistently, Ragi et al. [52] illustrated that IxS and pCS plasma concentrations pre- and post-hemodialysis were 3.5 ± 1.5 µg/mL to 2.34 ± 0.6 µg/mL and 16.68 ± 8.8 µg/mL to 12.59 ± 7.1 µg/mL, respectively [52]. Etinger et al. [81] hypothesized that dialysate pH affects the clearance of PBUTs and tested two conditions: isohydric dialysis with reduced bicarbonate buffer concentration for the first 90 min vs. standard dialysis with normal dialysate bicarbonate concentration for the full dialysis run [81] (Table 2). The overall reduction in IxS and pCS did not differ between the two conditions [81], but this could potentially be attributed to the lack of difference in IxS/pCS serum protein binding observed in the two dialysis methods. Furthermore, Vandenbosch et al. [78] tested the effects of asymmetric cellulose triacetate dialyzer with or without anticoagulants for clotting propensity [78]. Although they were not targeting the effects on IxS and pCS clearance, they reported no significant differences in the reduction ratios of these UTs without or without anticoagulants [78]. Furthermore, peritoneal dialysis was utilized by only a few studies in Table 2. Cheng et al. [90] demonstrated that IxS and pCS serum concentrations were reduced with peritoneal dialysis from 17.6 ± 8.7 µg/mL to 11.7 ± 5.3 µg/mL and from 4.6 ± 2.7 µg/mL to 3.7 ± 1.9 µg/mL, respectively, between baseline to 6-months into dialysis [90], suggesting that PD is also not efficient in removing both toxins. It is difficult to compare the efficiencies of IxS and pCS removal between hemodialysis and peritoneal dialysis due to the limited data.
Kidney Transplantation
The inefficient IxS and pCS removal by dialysis is likely attributed to their high percentage of protein binding; therefore, interventions such as organ transplantation might be required in patients with end-stage renal disease. Kidney transplantation was discussed in these studies in Table 2. te Linde et al. [76] reported significant decreases in serum IxS and pCS concentrations after adult kidney transplantation (as mentioned in the Section Central Nervous System Disease) [76] from 24.66 µg/mL to 0.86 µg/mL and 38.32 µg/mL to 5.45 µg/mL, respectively [76]. Korytowska et al. [66] documented salivary and serum IxS and pCS concentrations at baseline, ~6 months, and ~12 months after transplant and reported lower concentrations (although not statistically significant) at 6 and 12 months post-transplant compared to baseline [66]. Additionally, Korytowska et al. [66] investigated the correlations between saliva IxS or pCS and the deterioration of graft function in kidney transplant patients [66] and only found significant associations with IxS [66].
Furthermore, it is also important to consider the interacting effects of pharmacological agents that are usually administered to kidney transplant patients. André et al. [92] reported an association between higher serum pCS, but not IxS, concentrations and the usage of at least one OAT1/OAT3 inhibitor in adult kidney transplant patients [92]. The OAT1/OAT3 inhibitors used in this study included furosemide, acetylsalicylate, and pantoprazole [92], and these drugs likely exhibited different potency and mechanisms of interaction leading to the divergent findings observed with IxS and pCS. In addition, tacrolimus concentration-to-dose ratio, but not cyclosporine, was associated with significantly higher IxS and pCS serum concentrations in adult kidney transplant patients [93], but multiple regression, including the age variable, reduced the significance of pCS. The difference in the associations between the two calcineurin inhibitors might be attributed, according to the authors, to differences in their plasma protein binding properties [93]; however, additional pharmacokinetic interacting mechanisms may also be possible. From the literature, Rong et al. [124] reported a positive correlation between plasma pCS and mycophenolic acid trough concentrations [124], but this study did not find an association with tacrolimus. More studies are needed to characterize the interactions between immunosuppressants and IxS or pCS, which can potentially cause clinically significant pharmacodynamic effects in this already fragile population [124,125,126].
Alternative Matrices
Additional biological matrices, such as urine and saliva, have been utilized for the quantification of IxS and pCS (Table 2). Urinary IxS concentrations were correlated with ICU outcomes [97] and autism spectrum disorder in children [55], whereas Wijeyesekera et al. [54] established the reference ranges for urinary pCS concentrations in women and men and in two separate age groups (40–49 and 50–59 years old) [54]. These studies did not investigate whether urine was suitable for replacing serum/plasma for quantifying IxS and pCS. On the other hand, Giebułtowicz et al. [56] developed an analytical assay for characterizing IxS and pCS saliva concentrations in healthy volunteers at ages <30, 30–60, and >60 years (Table 2) [56], and this method was successfully applied to other studies in patients as described below. Additionally, Fabresse et al. [57] quantified the saliva IxS and pCS concentrations in healthy volunteers and reported Spearman’s correlations vs. free serum to be 0.78 (p < 0.0001) and 0.68 (p = 0.002), respectively [57]. Korytowska et al. [98] quantified saliva IxS and pCS in CKD patients and also reported significant correlations with free or total serum IxS and pCS concentrations (p < 0.00001) (Table 2) [98]. Consistent associations between salivary and serum concentrations were also observed in Korytowska et al. [66] in adult kidney transplant patients, where correlation coefficients were reported to be 0.81 (p < 0.00001) and 0.92 (p < 0.00001) for IxS and pCS, respectively [66]. However, despite these significant associations, additional characterizations on the precision and accuracy of saliva for predicting IxS or pCS concentrations in the serum are warranted (e.g., [127,128,129]). Although the use of saliva as an alternative matrix for measuring IxS and pCS concentrations may be promising, the matrix might be limited by potential contaminations, interactions with food and beverages, and physiological factors that may affect salivary flow (as discussed in Section 3.2.1).
3.7. Biological Applications Summary and Future Directions
The available clinical data illustrate the potential impacts of IxS and pCS on multiple organ systems. IxS and pCS are reported to accumulate with CKD progression, and the accumulation appears to become significant at CKD stage 3 and further elevated in patients on dialysis. These data suggest that CKD may lead to the accumulation of IxS and pCS; however, due to their propensity to cause tissue injury, IxS and pCS may also result in the progression and, hence, may serve as good biomarkers of CKD. Furthermore, IxS and/or pCS are potential predictors of cardiovascular events, as evidenced by associations with arterial stiffness, altered lipid profiles, thrombosis, and fatty tissue deposits. IxS is also associated with peripheral nerve dysfunction and the presence of ASD in children 6–10 years old. On the other hand, although kidney transplantation reduced IxS and pCS concentrations, no improvements in cognitive functions were observed. Likewise, several unexpected associations between IxS with gastric cancer, bone mineral density, or ICU outcomes are also observed, which would warrant further investigations. However, the majority of these data were obtained from correlational studies and should be further confirmed with cause-effect investigations.
Several therapeutic methods may be capable of reducing IxS and/or pCS concentrations, including drugs such as oral vancomycin, natural products such as Shen-Shuai-Ning, and kidney transplantation, although these are based on a limited amount of data. On the other hand, inconsistent or unexpected effects of AST-120, sevelamer, and diet on IxS and/or pCS concentrations are reported, and dialysis appears to be inefficient in reducing IxS and pCS. The data also suggest that there is potential for using saliva to monitor IxS and pCS concentrations, but further validation using prediction-error analyses is required. Overall, different outcomes/associations with IxS and pCS have been observed in various studies in different patient populations (Table 2), suggesting that each toxin may act by distinct interacting/toxicological/pathological mechanisms. As such, mitigation strategies to reduce toxin concentrations may also have to be tailored to each specific sulfonated uremic toxin and to the specific patient population. Further mechanistic investigations of the pathological mechanisms of IxS and pCS are also warranted. Finally, as a reflection of the significant interests in this review topic, we have identified further examples of biological applications (e.g., [130,131]) since stopping our systematic literature search (i.e., in the period from December 2023–May 2024). As this is a rapidly evolving field, we recommend revisiting the topic regularly, using this review as a foundation for further discussions.
4. Conclusions
This comprehensive review critically evaluated the LC-MS/MS assays that quantified IxS and/or pCS in different human biological matrices (Table 1) and presented the clinical applications using these validated assays (Table 2). The majority of the assays focused on human serum/plasma, utilizing acetonitrile or methanol, liquid–liquid extraction, reverse phase (e.g., C18) chromatography, and gradient elution for analyte separation. Mass spectrometry conditions were also consistent in the identified papers, with negative electrospray ionization, select MRM transitions and deuterated internal standards being the most common approaches. The available biological data illustrated the impacts of IxS and pCS on both renal and cardiovascular systems, whereas limited data were available on the other organ systems (e.g., CNS, bone density, gastrointestinal health, etc.). These data also supported that IxS and pCS might exhibit different toxicity mechanisms and different mitigation strategies might be warranted to detoxify these compounds. The clinical monitoring of IxS and pCS is gaining significant interest, and this review will serve as a useful compendium for both the scientific and clinical communities.
Author Contributions
Conceptualization, T.K.L.K.; methodology, T.K.L.K. and A.R.A.-D.; validation, T.K.L.K., A.R.A.-D. and Q.K.H.; formal analysis, T.K.L.K., A.R.A.-D. and Q.K.H.; investigation, T.K.L.K. and A.R.A.-D.; resources, T.K.L.K.; data curation, T.K.L.K. and A.R.A.-D.; writing—original draft preparation, A.R.A.-D.; writing—review and editing, T.K.L.K.; supervision, T.K.L.K.; project administration, T.K.L.K.; All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on Embase, Medline, PubMed, Scopus, and Web of Science.
Acknowledgments
We acknowledge the University of Alberta librarian, Janice Kung, for assisting with the initial literature search.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Denic, A.; Rule, A.D.; Glassock, R.J. Healthy and unhealthy aging on kidney structure and function: Human studies. Curr. Opin. Nephrol. Hypertens. 2022, 31, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.J.; Sidor, N.A.; Tonial, N.C.; Che, A.; Urquhart, B.L. Uremic Toxins in the Progression of Chronic Kidney Disease and Cardiovascular Disease: Mechanisms and Therapeutic Targets. Toxins 2021, 13, 142. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.; Mone, P.; Jankauskas, S.S.; Gambardella, J.; Santulli, G. Chronic kidney disease: Definition, updated epidemiology, staging, and mechanisms of increased cardiovascular risk. J. Clin. Hypertens. 2021, 23, 831–834. [Google Scholar] [CrossRef]
- Charles, C.; Ferris, A.H. Chronic Kidney Disease. Prim. Care 2020, 47, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.K.; Knicely, D.H.; Grams, M.E. Chronic Kidney Disease Diagnosis and Management: A Review. JAMA 2019, 322, 1294–1304. [Google Scholar] [CrossRef]
- Vanholder, R.; Pletinck, A.; Schepers, E.; Glorieux, G. Biochemical and clinical impact of organic uremic retention solutes: A comprehensive update. Toxins 2018, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- Duranton, F.; Cohen, G.; De Smet, R.; Rodriguez, M.; Jankowski, J.; Vanholder, R.; Argiles, A. Normal and pathologic concentrations of uremic toxins. J. Am. Soc. Nephrol. 2012, 23, 1258–1270. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, S.R.; Meireles, A.N.; Marques, S.S.; Silva, L.; Barreiros, L.; Sampaio-Maia, B.; Miró, M.; Segundo, M.A. Sample preparation and chromatographic methods for the determination of protein-bound uremic retention solutes in human biological samples: An overview. J. Chromatogr. B Analyt Technol. Biomed. Life Sci. 2023, 1215, 123578. [Google Scholar] [CrossRef]
- The European Uremic Toxins (EUTox) Database. Available online: www.uremic-toxins.org (accessed on 15 March 2024).
- Reis, T.; Hutchison, C.; de Assis Rocha Neves, F.; Zawadzki, B.; Zanella, M.; Ronco, C.; Rosner, M.H. Rationale for a New Classification of Solutes of Interest in Chronic Kidney Disease and Hemodialysis. Blood Purif. 2023, 52, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Graboski, A.L.; Redinbo, M.R. Gut-Derived Protein-Bound Uremic Toxins. Toxins 2020, 12, 590. [Google Scholar] [CrossRef] [PubMed]
- Daneshamouz, S.; Eduok, U.; Abdelrasoul, A.; Shoker, A. Protein-bound uremic toxins (PBUTs) in chronic kidney disease (CKD) patients: Production pathway, challenges and recent advances in renal PBUTs clearance. NanoImpact 2021, 21, 100299. [Google Scholar] [CrossRef] [PubMed]
- Rosner, M.H.; Reis, T.; Husain-Syed, F.; Vanholder, R.; Hutchison, C.; Stenvinkel, P.; Blankestijn, P.J.; Cozzolino, M.; Juillard, L.; Kashani, K.; et al. Classification of Uremic Toxins and Their Role in Kidney Failure. Clin. J. Am. Soc. Nephrol. 2021, 16, 1918–1928. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T. Overview of Uremic Toxins. In Uremic Toxins and Organ Failure; Saito, H., Abe, T., Eds.; Springer: Singapore, 2020; pp. 1–15. [Google Scholar] [CrossRef]
- Di Paola, R.; De, A.; Izhar, R.; Abate, M.; Zappavigna, S.; Capasso, A.; Perna, A.F.; La Russa, A.; Capasso, G.; Caraglia, M.; et al. Possible Effects of Uremic Toxins p-Cresol, Indoxyl Sulfate, p-Cresyl Sulfate on the Development and Progression of Colon Cancer in Patients with Chronic Renal Failure. Genes 2023, 14, 1257. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Wu, L.; Tang, M.Y.; Liu, Y.F.; Liu, L.; Liu, X.Y.; Zhang, C.; Huang, L. Indoxyl sulfate in atherosclerosis. Toxicol. Lett. 2023, 383, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Gryp, T.; Vanholder, R.; Vaneechoutte, M.; Glorieux, G. p-Cresyl sulfate. Toxins 2017, 9, 52. [Google Scholar] [CrossRef] [PubMed]
- Prokopienko, A.J.; Nolin, T.D. Microbiota-derived uremic retention solutes: Perpetrators of altered nonrenal drug clearance in kidney disease. Expert. Rev. Clin. Pharmacol. 2018, 11, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Leong, S.C.; Sirich, T.L. Indoxyl Sulfate-Review of Toxicity and Therapeutic Strategies. Toxins 2016, 8, 358. [Google Scholar] [CrossRef] [PubMed]
- Falconi, C.A.; Junho, C.; Fogaça-Ruiz, F.; Vernier, I.C.S.; da Cunha, R.S.; Stinghen, A.E.M.; Carneiro-Ramos, M.S. Uremic Toxins: An Alarming Danger Concerning the Cardiovascular System. Front. Physiol. 2021, 12, 686249. [Google Scholar] [CrossRef] [PubMed]
- Stockler-Pinto, M.B.; Fouque, D.; Soulage, C.O.; Croze, M.; Mafra, D. Indoxyl sulfate and p-cresyl sulfate in chronic kidney disease. Could these toxins modulate the antioxidant Nrf2-Keap1 pathway? J. Ren. Nutr. 2014, 24, 286–291. [Google Scholar] [CrossRef]
- Magnani, S.; Atti, M. Uremic Toxins and Blood Purification: A Review of Current Evidence and Future Perspectives. Toxins 2021, 13, 246. [Google Scholar] [CrossRef]
- Rong, Y.; Kiang, T.K.L. Characterization of human sulfotransferases catalyzing the formation of p-cresol sulfate and identification of mefenamic acid as a potent metabolism inhibitor and potential therapeutic agent for detoxification. Toxicol. Appl. Pharmacol. 2021, 425, 115553. [Google Scholar] [CrossRef] [PubMed]
- Deltombe, O.; Van Biesen, W.; Glorieux, G.; Massy, Z.; Dhondt, A.; Eloot, S. Exploring Protein Binding of Uremic Toxins in Patients with Different Stages of Chronic Kidney Disease and during Hemodialysis. Toxins 2015, 7, 3933–3946. [Google Scholar] [CrossRef] [PubMed]
- Suchy-Dicey, A.M.; Laha, T.; Hoofnagle, A.; Newitt, R.; Sirich, T.L.; Meyer, T.W.; Thummel, K.E.; Yanez, N.D.; Himmelfarb, J.; Weiss, N.S.; et al. Tubular Secretion in CKD. J. Am. Soc. Nephrol. 2016, 27, 2148–2155. [Google Scholar] [CrossRef]
- Wakabayashi, I.; Marumo, M. Evidence for Indoxyl Sulfate as an Inducer of Oxidative Stress in Patients with Diabetes. Vivo 2022, 36, 1790–1794. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Zou, J.; Xiang, F.; Zhang, P.; Shen, B.; Wang, Y.; Ding, X.; Cao, X. p-Cresyl Sulfate Predicts Ischemic Stroke among Patients on Hemodialysis: A Prospective Cohort Study. Dis. Markers 2022, 2022, 1358419. [Google Scholar] [CrossRef]
- Pretorius, C.J.; McWhinney, B.C.; Sipinkoski, B.; Johnson, L.A.; Rossi, M.; Campbell, K.L.; Ungerer, J.P. Reference ranges and biological variation of free and total serum indoxyl- and p-cresyl sulphate measured with a rapid UPLC fluorescence detection method. Clin. Chim. Acta 2013, 419, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.N.; Wu, I.W.; Huang, Y.F.; Peng, S.Y.; Huang, Y.C.; Ning, H.C. Measuring serum total and free indoxyl sulfate and p-cresyl sulfate in chronic kidney disease using UPLC-MS/MS. J. Food Drug Anal. 2019, 27, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Duan, S.; Pi, J.; Wang, C.H.; Hou, Y.C.; Andy Lee, C.Y.; Lin, C.J.; Shi, L.; Young, K.C.; Sun, H.Y. Assessment of ELISA-based method for the routine examination of serum indoxyl sulfate in patients with chronic kidney disease. Heliyon 2022, 8, e12220. [Google Scholar] [CrossRef] [PubMed]
- Fabresse, N.; Uteem, I.; Lamy, E.; Massy, Z.; Larabi, I.A.; Alvarez, J.C. Quantification of free and protein bound uremic toxins in human serum by LC-MS/MS: Comparison of rapid equilibrium dialysis and ultrafiltration. Clin. Chim. Acta 2020, 507, 228–235. [Google Scholar] [CrossRef]
- Zhu, W.; Stevens, A.P.; Dettmer, K.; Gottfried, E.; Hoves, S.; Kreutz, M.; Holler, E.; Canelas, A.B.; Kema, I.; Oefner, P.J. Quantitative profiling of tryptophan metabolites in serum, urine, and cell culture supernatants by liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2011, 401, 3249–3261. [Google Scholar] [CrossRef]
- Heaney, L.M. Applying mass spectrometry-based assays to explore gut microbial metabolism and associations with disease. Clin. Chem. Lab. Med. 2020, 58, 719–732. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T. Recent progress in the analysis of uremic toxins by mass spectrometry. J. Chromatogr. B Analyt Technol. Biomed. Life Sci. 2009, 877, 2600–2606. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T. Update of uremic toxin research by mass spectrometry. Mass. Spectrom. Rev. 2011, 30, 510–521. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Bioanalytical Method Validation, Guidance for Industry; FD Administration: Washington, DC, USA, 2018.
- Shu, C.; Chen, X.; Xia, T.; Zhang, F.; Gao, S.; Chen, W. LC-MS/MS method for simultaneous determination of serum p-cresyl sulfate and indoxyl sulfate in patients undergoing peritoneal dialysis. Biomed. Chromatogr. 2016, 30, 1782–1788. [Google Scholar] [CrossRef] [PubMed]
- Cuoghi, A.; Caiazzo, M.; Bellei, E.; Monari, E.; Bergamini, S.; Palladino, G.; Ozben, T.; Tomasi, A. Quantification of p-cresol sulphate in human plasma by selected reaction monitoring. Anal. Bioanal. Chem. 2012, 404, 2097–2104. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Sparidans, R.W.; Lu, J.; Mihaila, S.M.; Gerritsen, K.G.F.; Masereeuw, R. A robust, accurate, sensitive LC-MS/MS method to measure indoxyl sulfate, validated for plasma and kidney cells. Biomed. Chromatogr. 2022, 36, e5307. [Google Scholar] [CrossRef] [PubMed]
- Boelaert, J.; Lynen, F.; Glorieux, G.; Eloot, S.; Van Landschoot, M.; Waterloos, M.A.; Sandra, P.; Vanholder, R. A novel UPLC-MS-MS method for simultaneous determination of seven uremic retention toxins with cardiovascular relevance in chronic kidney disease patients. Anal. Bioanal. Chem. 2013, 405, 1937–1947. [Google Scholar] [CrossRef] [PubMed]
- Armbruster, D.A.; Tillman, M.D.; Hubbs, L.M. Limit of detection (LQD)/limit of quantitation (LOQ): Comparison of the empirical and the statistical methods exemplified with GC-MS assays of abused drugs. Clin. Chem. 1994, 40 Pt 1, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- de Loor, H.; Poesen, R.; De Leger, W.; Dehaen, W.; Augustijns, P.; Evenepoel, P.; Meijers, B. A liquid chromatography—Tandem mass spectrometry method to measure a selected panel of uremic retention solutes derived from endogenous and colonic microbial metabolism. Anal. Chim. Acta 2016, 936, 149–156. [Google Scholar] [CrossRef]
- Zhang, A.; Rijal, K.; Ng, S.K.; Ravid, K.; Chitalia, V. A mass spectrometric method for quantification of tryptophan-derived uremic solutes in human serum. J. Biol. Methods 2017, 4, e75. [Google Scholar] [CrossRef]
- Prokopienko, A.J.; West, R.E., 3rd; Stubbs, J.R.; Nolin, T.D. Development and validation of a UHPLC-MS/MS method for measurement of a gut-derived uremic toxin panel in human serum: An application in patients with kidney disease. J. Pharm. Biomed. Anal. 2019, 174, 618–624. [Google Scholar] [CrossRef] [PubMed]
- André, C.; Bennis, Y.; Titeca-Beauport, D.; Caillard, P.; Cluet, Y.; Kamel, S.; Choukroun, G.; Maizel, J.; Liabeuf, S.; Bodeau, S. Two rapid, accurate liquid chromatography tandem mass spectrometry methods for the quantification of seven uremic toxins: An application for describing their accumulation kinetic profile in a context of acute kidney injury. J. Chromatogr. B Analyt Technol. Biomed. Life Sci. 2020, 1152, 122234. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.R.; Xin, M.Y.; Li, K.; Wang, H.; Rao, Z.; Liu, T.X.; Wu, X.A. An LC-MS/MS analytical method for the determination of uremic toxins in patients with end-stage renal disease. J. Pharm. Biomed. Anal. 2020, 191, 113551. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jiang, H.; Chen, X.; Song, X.; Xu, F.; Chen, F.; Mao, Z.; Gao, S.; Chen, W. A rapid and sensitive method for simultaneous determination of eight protein-bound uremic toxins in human serum by UHPLC-MS/MS: Application in assessing peritoneal dialysis. J. Pharm. Biomed. Anal. 2020, 186, 113312. [Google Scholar] [CrossRef] [PubMed]
- Gürdeniz, G.; Kristensen, M.; Skov, T.; Dragsted, L.O. The Effect of LC-MS Data Preprocessing Methods on the Selection of Plasma Biomarkers in Fed vs. Fasted Rats. Metabolites 2012, 2, 77–99. [Google Scholar] [CrossRef] [PubMed]
- Monošík, R.; Dragsted, L.O. A versatile UHPLC-MSMS method for simultaneous quantification of various alcohol intake related compounds in human urine and blood. Anal. Methods 2016, 8, 6865–6871. [Google Scholar] [CrossRef]
- Kanemitsu, Y.; Asaji, K.; Matsumoto, Y.; Tsukamoto, H.; Saigusa, D.; Mukawa, C.; Tachikawa, T.; Abe, T.; Tomioka, Y. Simultaneous quantitative analysis of uremic toxins by LC-MS/MS with a reversed-phase/cation-exchange/anion-exchange tri-modal mixed-mode column. J. Chromatogr. B Analyt Technol. Biomed. Life Sci. 2017, 1068–1069, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Oda, A.; Suzuki, Y.; Sato, B.; Sato, H.; Tanaka, R.; Ono, H.; Ando, T.; Shin, T.; Mimata, H.; Itoh, H.; et al. Highly sensitive simultaneous quantification of indoxyl sulfate and 3-carboxy-4-methyl-5-propyl-2-furanpropanoic acid in human plasma using ultra-high-performance liquid chromatography coupled with tandem mass spectrometry. J. Sep. Sci. 2022, 45, 1672–1682. [Google Scholar] [CrossRef] [PubMed]
- Ragi, N.; Pallerla, P.; Babi Reddy Gari, A.R.; Lingampelly, S.S.; Ketavarapu, V.; Addipilli, R.; Chirra, N.; Kantevari, S.; Yadla, M.; Sripadi, P. Assessment of uremic toxins in advanced chronic kidney disease patients on maintenance hemodialysis by LC-ESI-MS/MS. Metabolomics 2023, 19, 14. [Google Scholar] [CrossRef]
- Choi, J.M.; Park, W.S.; Song, K.Y.; Lee, H.J.; Jung, B.H. Development of simultaneous analysis of tryptophan metabolites in serum and gastric juice—An investigation towards establishing a biomarker test for gastric cancer diagnosis. Biomed. Chromatogr. 2016, 30, 1963–1974. [Google Scholar] [CrossRef]
- Wijeyesekera, A.; Clarke, P.A.; Bictash, M.; Brown, I.J.; Fidock, M.; Ryckmans, T.; Yap, I.K.; Chan, Q.; Stamler, J.; Elliott, P.; et al. Quantitative UPLC-MS/MS analysis of the gut microbial co-metabolites phenylacetylglutamine, 4-cresyl sulphate and ippurate in human urine: INTERMAP Study. Anal. Methods 2012, 4, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Olesova, D.; Galba, J.; Piestansky, J.; Celusakova, H.; Repiska, G.; Babinska, K.; Ostatnikova, D.; Katina, S.; Kovac, A. A Novel UHPLC-MS Method Targeting Urinary Metabolomic Markers for Autism Spectrum Disorder. Metabolites 2020, 10, 443. [Google Scholar] [CrossRef] [PubMed]
- Giebułtowicz, J.; Korytowska, N.; Sankowski, B.; Wroczyński, P. Development and validation of a LC-MS/MS method for quantitative analysis of uraemic toxins p-cresol sulphate and indoxyl sulphate in saliva. Talanta 2016, 150, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Fabresse, N.; Larabi, I.A.; Abe, E.; Lamy, E.; Rigothier, C.; Massy, Z.A.; Alvarez, J.C. Correlation between Saliva Levels and Serum Levels of Free Uremic Toxins in Healthy Volunteers. Toxins 2023, 15, 150. [Google Scholar] [CrossRef]
- Nam, M.; Jo, S.R.; Park, J.H.; Kim, M.S. Evaluation of critical factors in the preparation of saliva sample from healthy subjects for metabolomics. J. Pharm. Biomed. Anal. 2023, 223, 115145. [Google Scholar] [CrossRef] [PubMed]
- Tsuzuki, S.; Wales, D. Intermolecular forces and clusters I. Struct. Bond. 2005, 115, 149. [Google Scholar]
- van der Lubbe, S.C.C.; Fonseca Guerra, C. The Nature of Hydrogen Bonds: A Delineation of the Role of Different Energy Components on Hydrogen Bond Strengths and Lengths. Chem. Asian J. 2019, 14, 2760–2769. [Google Scholar] [CrossRef] [PubMed]
- Eloot, S.; Schneditz, D.; Cornelis, T.; Van Biesen, W.; Glorieux, G.; Dhondt, A.; Kooman, J.; Vanholder, R. Protein-Bound Uremic Toxin Profiling as a Tool to Optimize Hemodialysis. PLoS ONE 2016, 11, e0147159. [Google Scholar] [CrossRef] [PubMed]
- PubChem. Indoxyl Sulfate. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Indoxyl-sulfate (accessed on 15 March 2024).
- PubChem. p-Cresol Sulfate. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/4615423 (accessed on 15 March 2024).
- PubChem. Ammonium Acetate. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Ammonium-acetate (accessed on 15 March 2024).
- Huang, S.T.; Shu, K.H.; Cheng, C.H.; Wu, M.J.; Yu, T.M.; Chuang, Y.W.; Chen, C.H. Serum total p-cresol and indoxyl sulfate correlated with stage of chronic kidney disease in renal transplant recipients. Transplant. Proc. 2012, 44, 621–624. [Google Scholar] [CrossRef]
- Korytowska, N.; Wyczałkowska-Tomasik, A.; Pączek, L.; Giebułtowicz, J. Evaluation of Salivary Indoxyl Sulfate with Proteinuria for Predicting Graft Deterioration in Kidney Transplant Recipients. Toxins 2021, 13, 571. [Google Scholar] [CrossRef]
- Celec, P.; Tóthová, Ľ.; Šebeková, K.; Podracká, Ľ.; Boor, P. Salivary markers of kidney function—Potentials and limitations. Clin. Chim. Acta 2016, 453, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Kiang, T.K.L.; Ranamukhaarachchi, S.A.; Ensom, M.H.H. Revolutionizing Therapeutic Drug Monitoring with the Use of Interstitial Fluid and Microneedles Technology. Pharmaceutics 2017, 9, 43. [Google Scholar] [CrossRef] [PubMed]
- SHIMADZU Excellence in Science. Interfaces for LC-MS. Available online: https://www.shimadzu.com/an/service-support/technical-support/analysis-basics/basics_of_lcms/interfaces_for_lcms.html (accessed on 15 March 2024).
- Shimadzu. Introduction to LC-MS Part4. Available online: https://www.shimadzu.com/an/service-support/technical-support/analysis-basics/lcms-intro/57intro.html#:~:text=M%20%2B%20BH%2B%20%E2%86%92MH%2B%20%2B%20B%20The%20positive,compounds%20with%20carboxyl%20groups%20or%20phenolic%20hydroxyl%20groups. (accessed on 15 March 2024).
- FDA. Bioanalytical Method Validation Guidance for Industry. 2018. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry (accessed on 15 March 2024).
- EMA. Guideline on Bioanalytical Method Validation. 2011. Available online: https://www.ema.europa.eu/en/bioanalytical-method-validation-scientific-guideline (accessed on 15 March 2024).
- Kaza, M.; Karaźniewicz-Łada, M.; Kosicka, K.; Siemiątkowska, A.; Rudzki, P.J. Bioanalytical method validation: New FDA guidance vs. EMA guideline. Better or worse? J. Pharm. Biomed. Anal. 2019, 165, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Jongers, B.; Hotterbeekx, A.; Bielen, K.; Vervliet, P.; Boddaert, J.; Lammens, C.; Fransen, E.; Baggerman, G.; Covaci, A.; Goossens, H.; et al. Identification of Potential Urinary Metabolite Biomarkers of Pseudomonas aeruginosa Ventilator-Associated Pneumonia. Biomark. Insights 2022, 17, 11772719221099131. [Google Scholar] [CrossRef] [PubMed]
- Hobson, S.; de Loor, H.; Kublickiene, K.; Beige, J.; Evenepoel, P.; Stenvinkel, P.; Ebert, T. Lipid Profile Is Negatively Associated with Uremic Toxins in Patients with Kidney Failure-A Tri-National Cohort. Toxins 2022, 14, 412. [Google Scholar] [CrossRef]
- Te Linde, E.; van Roij, C.J.M.; Meijers, B.K.I.; De Loor, H.; Kessels, R.P.C.; Wetzels, J.F.M. Cognitive Function and Uremic Toxins after Kidney Transplantation: An Exploratory Study. Kidney360 2020, 1, 1398–1406. [Google Scholar] [CrossRef] [PubMed]
- Biruete, A.; Cross, T.L.; Allen, J.M.; Kistler, B.M.; de Loor, H.; Evenepoel, P.; Fahey, G.C., Jr.; Bauer, L.; Swanson, K.S.; Wilund, K.R. Effect of Dietary Inulin Supplementation on the Gut Microbiota Composition and Derived Metabolites of Individuals Undergoing Hemodialysis: A Pilot Study. J. Ren. Nutr. 2021, 31, 512–522. [Google Scholar] [CrossRef]
- Vandenbosch, I.; Dejongh, S.; Claes, K.; Bammens, B.; De Vusser, K.; Van Craenenbroeck, A.; Kuypers, D.; Evenepoel, P.; Meijers, B. Strategies for asymmetrical triacetate dialyser heparin-free effective haemodialysis: The SAFE study. Clin. Kidney J. 2021, 14, 1901–1907. [Google Scholar] [CrossRef]
- Nazzal, L.; Soiefer, L.; Chang, M.; Tamizuddin, F.; Schatoff, D.; Cofer, L.; Aguero-Rosenfeld, M.E.; Matalon, A.; Meijers, B.; Holzman, R.; et al. Effect of Vancomycin on the Gut Microbiome and Plasma Concentrations of Gut-Derived Uremic Solutes. Kidney Int. Rep. 2021, 6, 2122–2133. [Google Scholar] [CrossRef]
- Dai, L.; Meijers, B.K.; Bammens, B.; de Loor, H.; Schurgers, L.J.; Qureshi, A.R.; Stenvinkel, P.; Evenepoel, P. Sevelamer Use in End-Stage Kidney Disease (ESKD) Patients Associates with Poor Vitamin K Status and High Levels of Gut-Derived Uremic Toxins: A Drug-Bug Interaction? Toxins 2020, 12, 351. [Google Scholar] [CrossRef]
- Etinger, A.; Kumar, S.R.; Ackley, W.; Soiefer, L.; Chun, J.; Singh, P.; Grossman, E.; Matalon, A.; Holzman, R.S.; Meijers, B.; et al. The effect of isohydric hemodialysis on the binding and removal of uremic retention solutes. PLoS ONE 2018, 13, e0192770. [Google Scholar] [CrossRef] [PubMed]
- Deltombe, O.; de Loor, H.; Glorieux, G.; Dhondt, A.; Van Biesen, W.; Meijers, B.; Eloot, S. Exploring binding characteristics and the related competition of different protein-bound uremic toxins. Biochimie 2017, 139, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Keddar, M.; Muylle, T.; Carrie, E.; Trefois, P.; Nachit, M.; Crott, R.; Christiaens, C.; Bammens, B.; Jadoul, M.; Goffin, E.; et al. Non-invasive Quantification of Fat Deposits in Skeletal Muscle Predicts Cardiovascular Outcome in Kidney Failure. Front. Physiol. 2020, 11, 130. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Gao, S.; Ruan, M.; Chen, S.; Xu, J.; Xing, X.; Pan, X.; Mei, C.; Mao, Z. Shen-Shuai-Ning granule decreased serum concentrations of indoxyl sulphate in uremic patients undergoing peritoneal dialysis. Biosci. Rep. 2018, 38, BSR2017164. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C.; Lai, Y.H.; Liu, C.H.; Wang, C.H.; Hsu, B.G.; Tsai, J.P. Association between serum indoxyl sulfate levels with carotid-femoral pulse wave velocity in patients with chronic kidney disease. Ren. Fail. 2021, 43, 796–802. [Google Scholar] [CrossRef] [PubMed]
- Kolachalama, V.B.; Shashar, M.; Alousi, F.; Shivanna, S.; Rijal, K.; Belghasem, M.E.; Walker, J.; Matsuura, S.; Chang, G.H.; Gibson, C.M.; et al. Uremic Solute-Aryl Hydrocarbon Receptor-Tissue Factor Axis Associates with Thrombosis after Vascular Injury in Humans. J. Am. Soc. Nephrol. 2018, 29, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.R.; Cheng, B.C.; Lin, C.N.; Chiu, W.C.; Lin, T.Y.; Chiang, H.C.; Kuo, C.A.; Huang, C.C.; Lu, C.H. The Effects of Indoxyl Sulfate and Oxidative Stress on the Severity of Peripheral Nerve Dysfunction in Patients with Chronic Kidney Diseases. Antioxidants 2022, 11, 2350. [Google Scholar] [CrossRef] [PubMed]
- Wu, I.W.; Lee, C.C.; Hsu, H.J.; Sun, C.Y.; Chen, Y.C.; Yang, K.J.; Yang, C.W.; Chung, W.H.; Lai, H.C.; Chang, L.C.; et al. Compositional and Functional Adaptations of Intestinal Microbiota and Related Metabolites in CKD Patients Receiving Dietary Protein Restriction. Nutrients 2020, 12, 2799. [Google Scholar] [CrossRef] [PubMed]
- Wu, I.W.; Lin, C.Y.; Chang, L.C.; Lee, C.C.; Chiu, C.Y.; Hsu, H.J.; Sun, C.Y.; Chen, Y.C.; Kuo, Y.L.; Yang, C.W.; et al. Gut Microbiota as Diagnostic Tools for Mirroring Disease Progression and Circulating Nephrotoxin Levels in Chronic Kidney Disease: Discovery and Validation Study. Int. J. Biol. Sci. 2020, 16, 420–434. [Google Scholar] [CrossRef]
- Cheng, B.C.; Lai, Y.R.; Huang, C.C.; Lu, C.H. Improved cardiovascular autonomic function and decreased protein-bound uremic toxins in patients with end-stage renal disease after peritoneal dialysis. J. Int. Med. Res. 2020, 48, 300060520933797. [Google Scholar] [CrossRef]
- Hsu, C.K.; Su, S.C.; Chang, L.C.; Yang, K.J.; Lee, C.C.; Hsu, H.J.; Chen, Y.T.; Sun, C.Y.; Wu, I.W. Oral Absorbent AST-120 Is Associated with Compositional and Functional Adaptations of Gut Microbiota and Modification of Serum Short and Medium-Chain Fatty Acids in Advanced CKD Patients. Biomedicines 2022, 10, 2234. [Google Scholar] [CrossRef] [PubMed]
- André, C.; Mernissi, T.; Choukroun, G.; Bennis, Y.; Kamel, S.; Liabeuf, S.; Bodeau, S. The Prescription of Drugs That Inhibit Organic Anion Transporters 1 or 3 Is Associated with the Plasma Accumulation of Uremic Toxins in Kidney Transplant Recipients. Toxins 2021, 14, 15. [Google Scholar] [CrossRef] [PubMed]
- André, C.; Choukroun, G.; Bennis, Y.; Kamel, S.; Lemaire-Hurtel, A.S.; Masmoudi, K.; Bodeau, S.; Liabeuf, S. Potential interactions between uraemic toxins and drugs: An application in kidney transplant recipients treated with calcineurin inhibitors. Nephrol. Dial. Transplant. 2022, 37, 2284–2292. [Google Scholar] [CrossRef] [PubMed]
- Batteux, B.; Bodeau, S.; André, C.; Hurtel-Lemaire, A.S.; Gras-Champel, V.; Desailly-Henry, I.; Masmoudi, K.; Bennis, Y.; Massy, Z.A.; Kamel, S.; et al. Association between Uremic Toxin Concentrations and Bone Mineral Density after Kidney Transplantation. Toxins 2020, 12, 715. [Google Scholar] [CrossRef] [PubMed]
- El Chamieh, C.; Larabi, I.A.; Laville, S.M.; Jacquelinet, C.; Combe, C.; Fouque, D.; Laville, M.; Frimat, L.; Pecoits-Filho, R.; Lange, C.; et al. Proton-Pump Inhibitors and Serum Concentrations of Uremic Toxins in Patients with Chronic Kidney Disease. Toxins 2023, 15, 276. [Google Scholar] [CrossRef] [PubMed]
- Massy, Z.A.; Chesnaye, N.C.; Larabi, I.A.; Dekker, F.W.; Evans, M.; Caskey, F.J.; Torino, C.; Porto, G.; Szymczak, M.; Drechsler, C.; et al. The relationship between uremic toxins and symptoms in older men and women with advanced chronic kidney disease. Clin. Kidney J. 2022, 15, 798–807. [Google Scholar] [CrossRef] [PubMed]
- Kuo, S.Z.; Dettmer, K.; Annavajhala, M.K.; Chong, D.H.; Uhlemann, A.C.; Abrams, J.A.; Oefner, P.J.; Freedberg, D.E. Associations between urinary 3-indoxyl sulfate, a gut microbiome-derived biomarker, and patient outcomes after intensive care unit admission. J. Crit. Care 2021, 63, 15–21. [Google Scholar] [CrossRef]
- Korytowska, N.; Sankowski, B.; Wyczałkowska-Tomasik, A.; Pączek, L.; Wroczyński, P.; Giebułtowicz, J. The utility of saliva testing in the estimation of uremic toxin levels in serum. Clin. Chem. Lab. Med. 2018, 57, 230–237. [Google Scholar] [CrossRef]
- Alaini, A.; Malhotra, D.; Rondon-Berrios, H.; Argyropoulos, C.P.; Khitan, Z.J.; Raj, D.S.C.; Rohrscheib, M.; Shapiro, J.I.; Tzamaloukas, A.H. Establishing the presence or absence of chronic kidney disease: Uses and limitations of formulas estimating the glomerular filtration rate. World J. Methodol. 2017, 7, 73–92. [Google Scholar] [CrossRef]
- Yu, M.; Kim, Y.J.; Kang, D.H. Indoxyl sulfate-induced endothelial dysfunction in patients with chronic kidney disease via an induction of oxidative stress. Clin. J. Am. Soc. Nephrol. 2011, 6, 30–39. [Google Scholar] [CrossRef]
- Dou, L.; Jourde-Chiche, N.; Faure, V.; Cerini, C.; Berland, Y.; Dignat-George, F.; Brunet, P. The uremic solute indoxyl sulfate induces oxidative stress in endothelial cells. J. Thromb. Haemost. 2007, 5, 1302–1308. [Google Scholar] [CrossRef] [PubMed]
- Schepers, E.; Meert, N.; Glorieux, G.; Goeman, J.; Van der Eycken, J.; Vanholder, R. P-cresylsulphate, the main in vivo metabolite of p-cresol, activates leucocyte free radical production. Nephrol. Dial. Transplant. 2007, 22, 592–596. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Miyamoto, Y.; Honda, D.; Tanaka, H.; Wu, Q.; Endo, M.; Noguchi, T.; Kadowaki, D.; Ishima, Y.; Kotani, S.; et al. p-Cresyl sulfate causes renal tubular cell damage by inducing oxidative stress by activation of NADPH oxidase. Kidney Int. 2013, 83, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.Y.; Hsu, H.H.; Wu, M.S. p-Cresol sulfate and indoxyl sulfate induce similar cellular inflammatory gene expressions in cultured proximal renal tubular cells. Nephrol. Dial. Transplant. 2013, 28, 70–78. [Google Scholar] [CrossRef]
- Miyazaki, T.; Ise, M.; Seo, H.; Niwa, T. Indoxyl sulfate increases the gene expressions of TGF-beta 1, TIMP-1 and pro-alpha 1(I) collagen in uremic rat kidneys. Kidney Int. Suppl. 1997, 62, S15–S22. [Google Scholar] [PubMed]
- Gelasco, A.K.; Raymond, J.R. Indoxyl sulfate induces complex redox alterations in mesangial cells. Am. J. Physiol. Renal Physiol. 2006, 290, F1551–F1558. [Google Scholar] [CrossRef]
- Sun, C.Y.; Chang, S.C.; Wu, M.S. Uremic toxins induce kidney fibrosis by activating intrarenal renin-angiotensin-aldosterone system associated epithelial-to-mesenchymal transition. PLoS ONE 2012, 7, e34026. [Google Scholar] [CrossRef] [PubMed]
- Mutsaers, H.A.; Caetano-Pinto, P.; Seegers, A.E.; Dankers, A.C.; van den Broek, P.H.; Wetzels, J.F.; van den Brand, J.A.; van den Heuvel, L.P.; Hoenderop, J.G.; Wilmer, M.J.; et al. Proximal tubular efflux transporters involved in renal excretion of p-cresyl sulfate and p-cresyl glucuronide: Implications for chronic kidney disease pathophysiology. Toxicol. Vitro 2015, 29, 1868–1877. [Google Scholar] [CrossRef] [PubMed]
- Cao, L. Indoxyl sulfate stimulates oxidized LDL uptake through up-regulation of CD36 expression in THP-1 macrophages. J. Appl. Biomed. 2014, 12, 203–209. [Google Scholar] [CrossRef]
- Yamamoto, H.; Tsuruoka, S.; Ioka, T.; Ando, H.; Ito, C.; Akimoto, T.; Fujimura, A.; Asano, Y.; Kusano, E. Indoxyl sulfate stimulates proliferation of rat vascular smooth muscle cells. Kidney Int. 2006, 69, 1780–1785. [Google Scholar] [CrossRef]
- Muteliefu, G.; Enomoto, A.; Niwa, T. Indoxyl sulfate promotes proliferation of human aortic smooth muscle cells by inducing oxidative stress. J. Ren. Nutr. 2009, 19, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Adijiang, A.; Goto, S.; Uramoto, S.; Nishijima, F.; Niwa, T. Indoxyl sulphate promotes aortic calcification with expression of osteoblast-specific proteins in hypertensive rats. Nephrol. Dial. Transplant. 2008, 23, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Gross, P.; Massy, Z.A.; Henaut, L.; Boudot, C.; Cagnard, J.; March, C.; Kamel, S.; Drueke, T.B.; Six, I. Para-cresyl sulfate acutely impairs vascular reactivity and induces vascular remodeling. J. Cell Physiol. 2015, 230, 2927–2935. [Google Scholar] [CrossRef] [PubMed]
- Arnold, R.; Issar, T.; Krishnan, A.V.; Pussell, B.A. Neurological complications in chronic kidney disease. JRSM Cardiovasc. Dis. 2016, 5, 2048004016677687. [Google Scholar] [CrossRef] [PubMed]
- Mambelli, E.; Barrella, M.; Facchini, M.G.; Mancini, E.; Sicuso, C.; Bainotti, S.; Formica, M.; Santoro, A. The prevalence of peripheral neuropathy in hemodialysis patients. Clin. Nephrol. 2012, 77, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Tomino, Y.; Lu, K.C. Impacts of Indoxyl Sulfate and p-Cresol Sulfate on Chronic Kidney Disease and Mitigating Effects of AST-120. Toxins 2018, 10, 367. [Google Scholar] [CrossRef] [PubMed]
- Schulman, G.; Berl, T.; Beck, G.J.; Remuzzi, G.; Ritz, E.; Shimizu, M.; Shobu, Y.; Kikuchi, M. The effects of AST-120 on chronic kidney disease progression in the United States of America: A post hoc subgroup analysis of randomized controlled trials. BMC Nephrol. 2016, 17, 141. [Google Scholar] [CrossRef] [PubMed]
- Cha, R.H.; Kang, S.W.; Park, C.W.; Cha, D.R.; Na, K.Y.; Kim, S.G.; Yoon, S.A.; Han, S.Y.; Chang, J.H.; Park, S.K.; et al. A Randomized, Controlled Trial of Oral Intestinal Sorbent AST-120 on Renal Function Deterioration in Patients with Advanced Renal Dysfunction. Clin. J. Am. Soc. Nephrol. 2016, 11, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Akizawa, T.; Asano, Y.; Morita, S.; Wakita, T.; Onishi, Y.; Fukuhara, S.; Gejyo, F.; Matsuo, S.; Yorioka, N.; Kurokawa, K. Effect of a carbonaceous oral adsorbent on the progression of CKD: A multicenter, randomized, controlled trial. Am. J. Kidney Dis. 2009, 54, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Asai, M.; Kumakura, S.; Kikuchi, M. Review of the efficacy of AST-120 (KREMEZIN®) on renal function in chronic kidney disease patients. Ren. Fail. 2019, 41, 47–56. [Google Scholar] [CrossRef]
- Biruete, A.; Hill Gallant, K.M.; Lindemann, S.R.; Wiese, G.N.; Chen, N.X.; Moe, S.M. Phosphate Binders and Nonphosphate Effects in the Gastrointestinal Tract. J. Ren. Nutr. 2020, 30, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Y.; Tarng, D.C. Diet, gut microbiome and indoxyl sulphate in chronic kidney disease patients. Nephrology 2018, 23 (Suppl. 4), 16–20. [Google Scholar] [CrossRef] [PubMed]
- Takkavatakarn, K.; Wuttiputinun, T.; Phannajit, J.; Praditpornsilpa, K.; Eiam-Ong, S.; Susantitaphong, P. Protein-bound uremic toxin lowering strategies in chronic kidney disease: A systematic review and meta-analysis. J. Nephrol. 2021, 34, 1805–1817. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Colbourne, P.; Gourishankar, S.; Kiang, T.K.L. Significant correlations between p-cresol sulfate and mycophenolic acid plasma concentrations in adult kidney transplant recipients. Clin. Drug Investig. 2022, 42, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Kiang, T.K.L. Mechanisms of Metabolism Interaction Between p-Cresol and Mycophenolic Acid. Toxicol. Sci. 2020, 173, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Kiang, T.K.L. Development and validation of a sensitive liquid-chromatography tandem mass spectrometry assay for mycophenolic acid and metabolites in HepaRG cell culture: Characterization of metabolism interactions between p-cresol and mycophenolic acid. Biomed. Chromatogr. 2019, 33, e4549. [Google Scholar] [CrossRef] [PubMed]
- Gallant, J.; Wichart, J.; Kiang, T.K.L. Predictability of Capillary Blood Spot Toward Venous Whole Blood Sampling for Therapeutic Drug Monitoring of Tacrolimus in Solid Organ Transplant Recipients. Eur. J. Drug Metab. Pharmacokinet. 2019, 44, 729–741. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Kiang, T.K.; Bring, P.; Ensom, M.H. Predictive Performance of the Winter-Tozer and Derivative Equations for Estimating Free Phenytoin Concentration. Can. J. Hosp. Pharm. 2016, 69, 269–279. [Google Scholar] [CrossRef]
- Kiang, T.K.; Ensom, M.H. A Comprehensive Review on the Predictive Performance of the Sheiner-Tozer and Derivative Equations for the Correction of Phenytoin Concentrations. Ann. Pharmacother. 2016, 50, 311–325. [Google Scholar] [CrossRef]
- Samaey, A.; Vázquez-Castellanos, J.F.; Caenepeel, C.; Evenepoel, P.; Vermeire, S.; Raes, J.; Knops, N. Effects of fecal microbiota transplantation for recurrent Clostridium difficile infection in children on kidney replacement therapy: A pilot study. Pediatr. Nephrol. 2024, 39, 1201–1212. [Google Scholar] [CrossRef]
- Zou, X.; Xiong, L.; Guo, Z.; Li, Y.; Ning, K.; Liu, X.; Xie, Y.; Peng, Y.; Liu, Y.; Cai, X.; et al. Trimethylamine-N-oxide (TMAO) and predicted risk of cardiovascular events after partial nephrectomy. Asian J. Surg. 2024, 47, 281–288. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).