Inhaled Ivermectin-Loaded Lipid Polymer Hybrid Nanoparticles: Development and Characterization
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
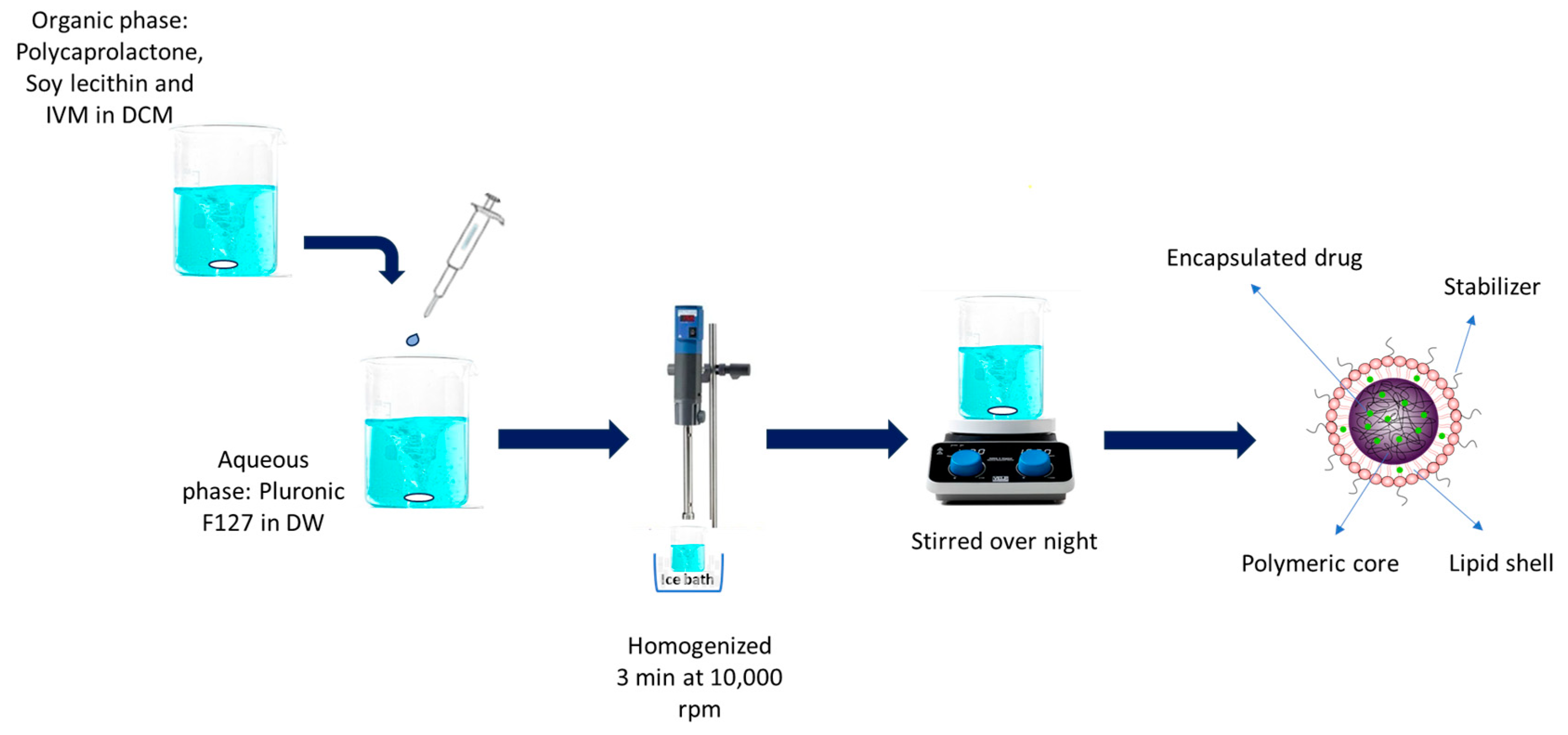
2.2. IVM-Loaded LPHNP Preparation
2.3. Physicochemical Characterization
2.4. Morphology of Nanoparticles
2.5. Drug Loading and Entrapment Efficiency
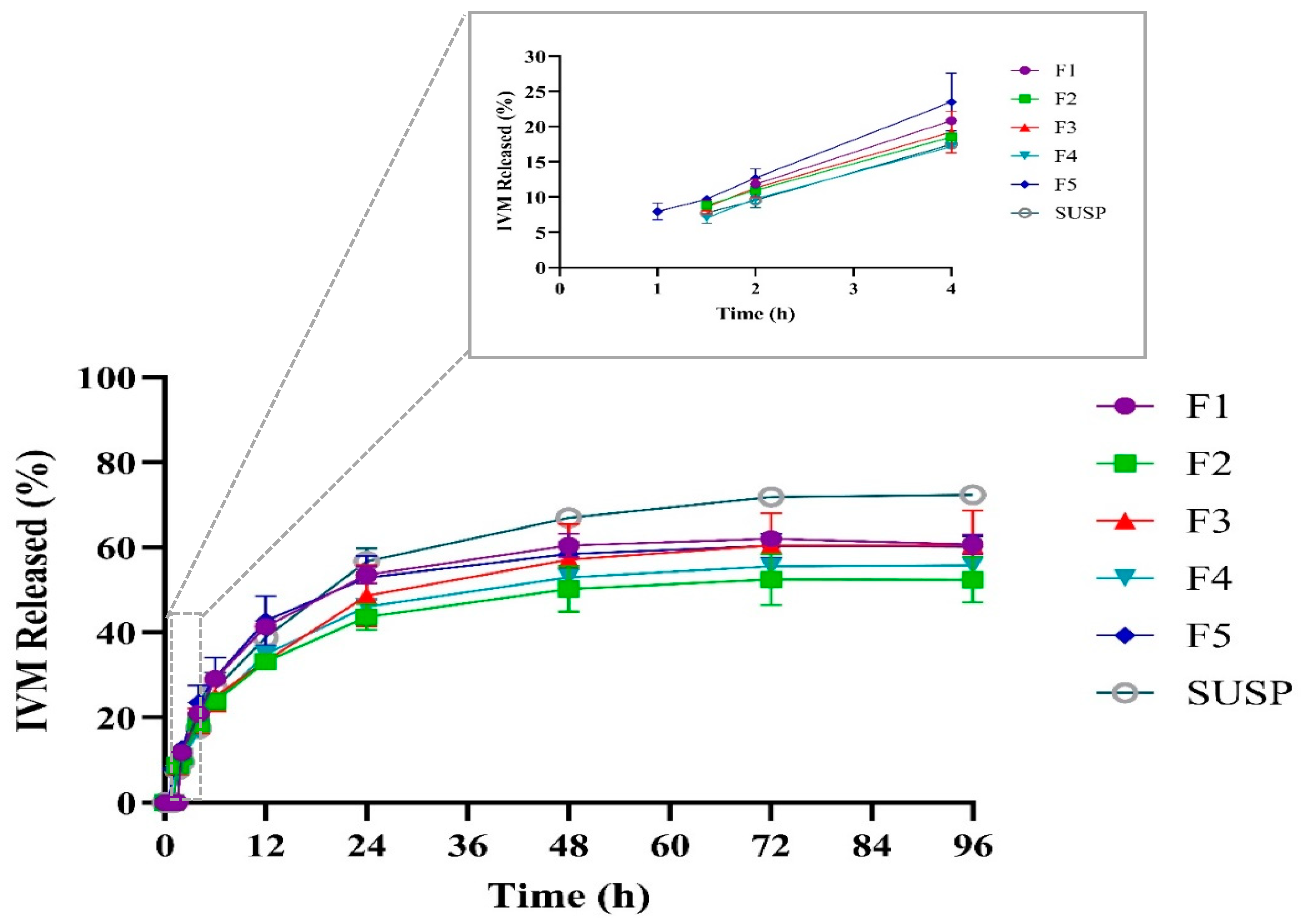
2.6. In Vitro Drug Release Study
2.7. Kinetics of IVM Release from LPHNPs
2.8. Solid-Phase Characterization
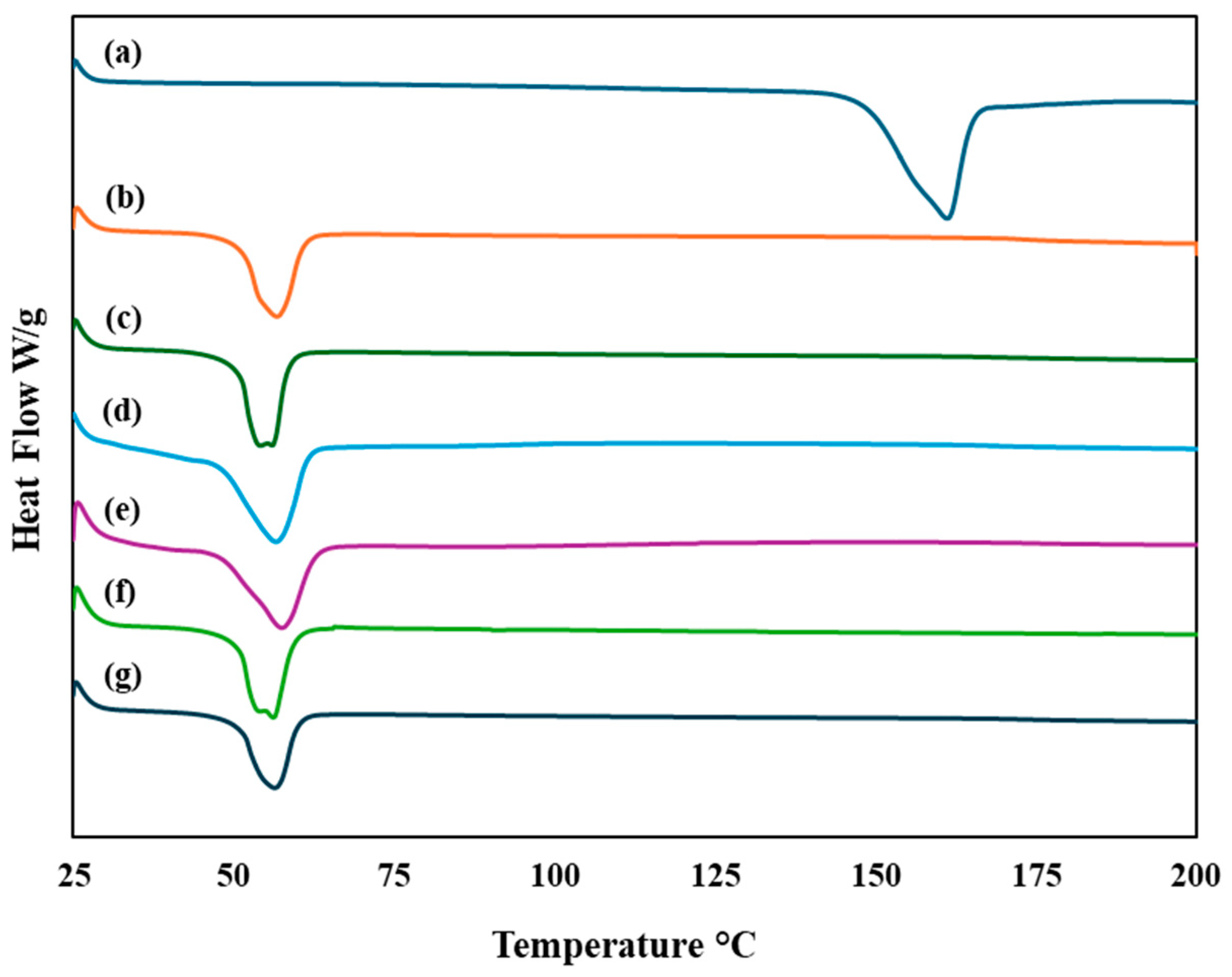
2.8.1. Differential Scanning Calorimetry (DSC)
2.8.2. Thermogravimetric Analysis (TGA)
2.8.3. Attenuated Total Reflection–Fourier Transform Infrared (ATR–FTIR)
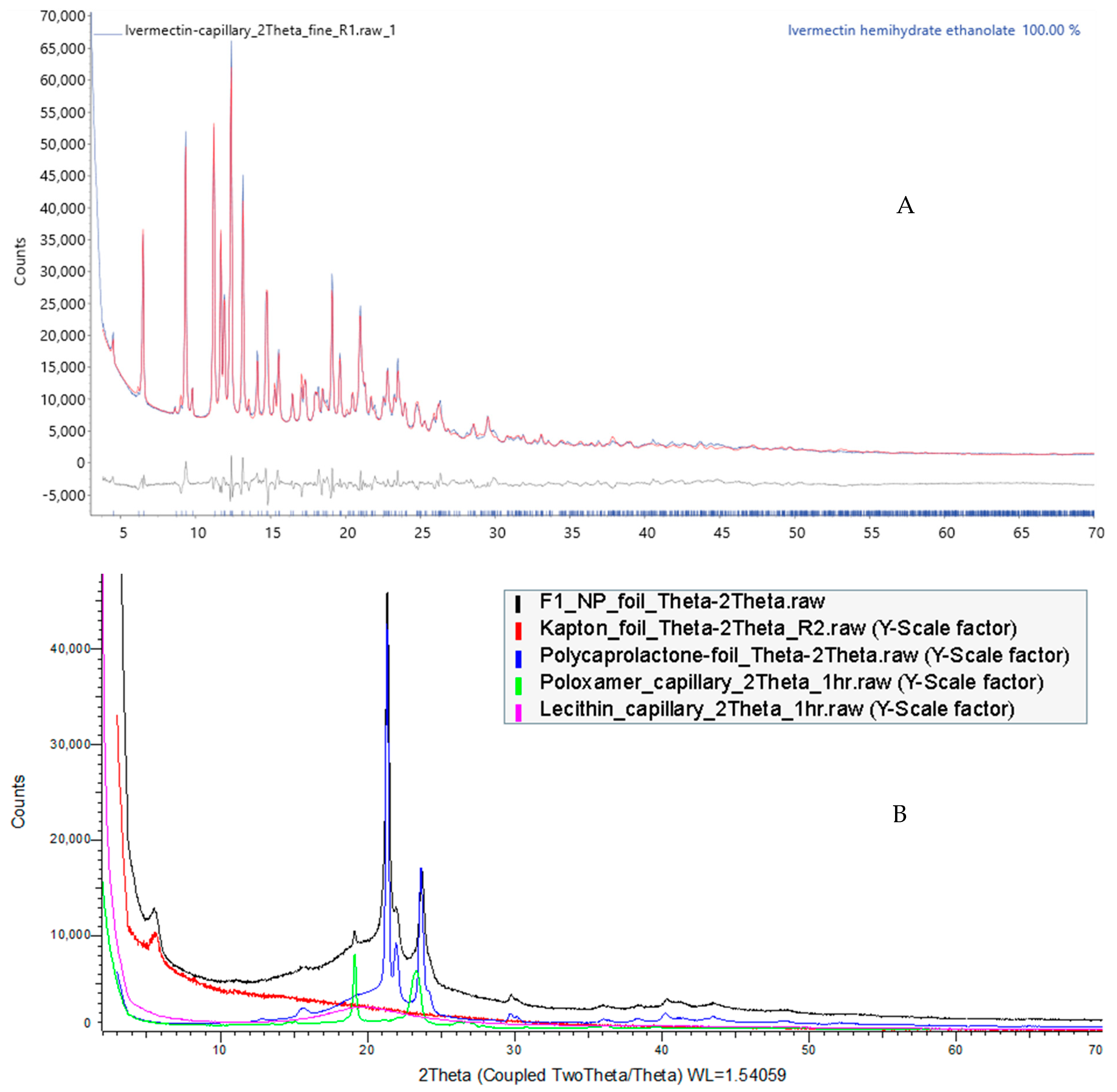
2.8.4. Powder X-ray Diffraction (PXRD)
- X-ray diffraction measurements—capillary transmission
- X-ray diffraction measurements—foil transmission
2.9. Particle Density and Flow Property
2.10. In Vitro Aerosolization Study
2.11. HPLC Assay
2.12. Statistical Analysis
3. Results and Discussion
3.1. IVM-Loaded LPHNP Preparation
3.2. Physicochemical Characterization
3.2.1. Particle Size and Size Distribution
3.2.2. Zeta Potential
3.3. Particle Shape and Morphology
3.4. Entrapment Efficiency and Drug Loading
3.5. In Vitro Drug Release Study and Kinetic of Release
3.6. Kinetics of IVM Release from LPHNPs
3.7. Solid-Phase Characterization
3.7.1. Differential Scanning Calorimetry (DSC)
3.7.2. Thermogravimetric Analysis (TGA)
3.7.3. Attenuated Total Reflection–Fourier Transform Infrared (ATR–FTIR)
3.7.4. Powder X-ray Diffraction (PXRD)
3.8. Particle Density and Flow Property
3.9. In Vitro Aerosolization Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thakur, A.K.; Chellappan, D.K.; Dua, K.; Mehta, M.; Satija, S.; Singh, I. Patented therapeutic drug delivery strategies for targeting pulmonary diseases. Expert Opin. Ther. Pat. 2020, 30, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Islam, N.; Rahman, S. Pulmonary drug delivery: Implication for new strategy for pharmacotherapy for neurodegenerative disorders. Drug Discov. Ther. 2008, 2, 264–276. [Google Scholar] [PubMed]
- Dong, L.; Zhuang, X. Insights into Inhalation Drug Disposition: The Roles of Pulmonary Drug-Metabolizing Enzymes and Transporters. Int. J. Mol. Sci. 2024, 25, 4671. [Google Scholar] [CrossRef]
- Gandhi, S.; Roy, I. Lipid-Based inhalable micro-and nanocarriers of active agents for treating non-small-cell lung cancer. Pharmaceutics 2023, 15, 1457. [Google Scholar] [CrossRef] [PubMed]
- Doroudian, M.; O’Neill, A.; Mac Loughlin, R.; Prina-Mello, A.; Volkov, Y.; Donnelly, S.C. Nanotechnology in pulmonary medicine. Curr. Opin. Pharmacol. 2021, 56, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cheng, W.; Xin, H.; Liu, R.; Wang, Q.; Cai, W.; Peng, X.; Yang, F.; Xin, H. Nanoparticles advanced from preclinical studies to clinical trials for lung cancer therapy. Cancer Nanotechnol. 2023, 14, 28. [Google Scholar] [CrossRef] [PubMed]
- Schabath, M.B.; Cote, M.L. Cancer progress and priorities: Lung cancer. Cancer Epidemiol. Biomark. Prev. 2019, 28, 1563–1579. [Google Scholar] [CrossRef] [PubMed]
- Majeed, U.; Manochakian, R.; Zhao, Y.; Lou, Y. Targeted therapy in advanced non-small cell lung cancer: Current advances and future trends. J. Hematol. Oncol. 2021, 14, 108. [Google Scholar] [CrossRef]
- Mandal, B.; Mittal, N.K.; Balabathula, P.; Thoma, L.A.; Wood, G.C. Development and in vitro evaluation of core–shell type lipid–polymer hybrid nanoparticles for the delivery of erlotinib in non-small cell lung cancer. Eur. J. Pharm. Sci. 2016, 81, 162–171. [Google Scholar] [CrossRef]
- Kumbhar, P.; Manjappa, A.; Shah, R.; Jha, N.K.; Singh, S.K.; Dua, K.; Disouza, J.; Patravale, V. Inhalation delivery of repurposed drugs for lung cancer: Approaches, benefits and challenges. J. Control. Release 2022, 341, 1–15. [Google Scholar] [CrossRef]
- Mohammed, S.W.; El-Megrab, N.A.; Hasan, A.A.; Gomaa, E. A remodeled ivermectin polycaprolactone-based nanoparticles for inhalation as a promising treatment of pulmonary inflammatory diseases. Eur. J. Pharm. Sci. 2024, 195, 106714. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Hu, X.; Wang, Y.; Yao, X.; Zhang, W.; Yu, C.; Cheng, F.; Li, J.; Fang, Q. Ivermectin, a potential anticancer drug derived from an antiparasitic drug. Pharmacol. Res. 2021, 163, 105207. [Google Scholar] [CrossRef] [PubMed]
- Johnson-Arbor, K. Ivermectin: A mini-review. Clin. Toxicol. 2022, 60, 571–575. [Google Scholar] [CrossRef] [PubMed]
- De Souza, Z.C.; Júnior, F.H.X.; Pinheiro, I.O.; de Souza Rebouças, J.; de Abreu, B.O.; Mesquita, P.R.R.; de Medeiros Rodrigues, F.; Quadros, H.C.; Mendes, T.M.F.; Nguewa, P. Ameliorating the antiparasitic activity of the multifaceted drug ivermectin through a polymer nanocapsule formulation. Int. J. Pharm. 2023, 639, 122965. [Google Scholar] [CrossRef] [PubMed]
- Esfahani, M.K.M.; Islam, N.; Cabot, P.J.; Izake, E.L. Development of thiabendazole-loaded mesoporous silica nanoparticles for cancer therapy. ACS Biomater. Sci. Eng. 2021, 8, 4153–4162. [Google Scholar] [CrossRef]
- Abdelaziz, H.M.; Gaber, M.; Abd-Elwakil, M.M.; Mabrouk, M.T.; Elgohary, M.M.; Kamel, N.M.; Kabary, D.M.; Freag, M.S.; Samaha, M.W.; Mortada, S.M. Inhalable particulate drug delivery systems for lung cancer therapy: Nanoparticles, microparticles, nanocomposites and nanoaggregates. J. Control. Release 2018, 269, 374–392. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Waters, A.K.; Kalyan, P.; Achrol, A.S.; Kesari, S.; Yenugonda, V.M. Lipid–polymer hybrid nanoparticles as a next-generation drug delivery platform: State of the art, emerging technologies, and perspectives. Int. J. Nanomed. 2019, 14, 1937–1952. [Google Scholar] [CrossRef] [PubMed]
- Kuperkar, K.; Atanase, L.I.; Bahadur, A.; Crivei, I.C.; Bahadur, P. Degradable polymeric bio (nano) materials and their biomedical applications: A comprehensive overview and recent updates. Polymers 2024, 16, 206. [Google Scholar] [CrossRef] [PubMed]
- Gajbhiye, K.R.; Salve, R.; Narwade, M.; Sheikh, A.; Kesharwani, P.; Gajbhiye, V. Lipid polymer hybrid nanoparticles: A custom-tailored next-generation approach for cancer therapeutics. Mol. Cancer 2023, 22, 160. [Google Scholar] [CrossRef]
- Anderson, C.F.; Grimmett, M.E.; Domalewski, C.J.; Cui, H. Inhalable nanotherapeutics to improve treatment efficacy for common lung diseases. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1586. [Google Scholar] [CrossRef]
- Hadinoto, K.; Sundaresan, A.; Cheow, W.S. Lipid–polymer hybrid nanoparticles as a new generation therapeutic delivery platform: A review. Eur. J. Pharm. Biopharm. 2013, 85, 427–443. [Google Scholar] [CrossRef]
- Dave, V.; Tak, K.; Sohgaura, A.; Gupta, A.; Sadhu, V.; Reddy, K.R. Lipid-polymer hybrid nanoparticles: Synthesis strategies and biomedical applications. J. Microbiol. Methods 2019, 160, 130–142. [Google Scholar] [CrossRef]
- Bardoliwala, D.; Patel, V.; Misra, A.; Sawant, K. Systematic development and characterization of inhalable dry powder containing Polymeric Lipid Hybrid Nanocarriers co-loaded with ABCB1 shRNA and docetaxel using QbD approach. J. Drug Deliv. Sci. Technol. 2021, 66, 102903. [Google Scholar] [CrossRef]
- Patel, V.; Bardoliwala, D.; Lalani, R.; Patil, S.; Ghosh, S.; Javia, A.; Misra, A. Development of a dry powder for inhalation of nanoparticles codelivering cisplatin and ABCC3 siRNA in lung cancer. Ther. Deliv. 2021, 12, 651–670. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, A.; Mehta, S.; Yadav, S.; Singh, S.K.; Grobler, A.; Goyal, A.K.; Mehta, A. Pulmonary delivery of antitubercular drugs using spray-dried lipid–polymer hybrid nanoparticles. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1544–1555. [Google Scholar] [CrossRef] [PubMed]
- Thanki, K.; van Eetvelde, D.; Geyer, A.; Fraire, J.; Hendrix, R.; Van Eygen, H.; Putteman, E.; Sami, H.; de Souza Carvalho-Wodarz, C.; Franzyk, H. Mechanistic profiling of the release kinetics of siRNA from lipidoid-polymer hybrid nanoparticles in vitro and in vivo after pulmonary administration. J. Control. Release 2019, 310, 82–93. [Google Scholar] [CrossRef]
- Vencken, S.; Foged, C.; Ramsey, J.M.; Sweeney, L.; Cryan, S.-A.; MacLoughlin, R.J.; Greene, C.M. Nebulised lipid–polymer hybrid nanoparticles for the delivery of a therapeutic anti-inflammatory microRNA to bronchial epithelial cells. ERJ Open Res. 2019, 5, 161–2018. [Google Scholar] [CrossRef]
- Conte, G.; Costabile, G.; Baldassi, D.; Rondelli, V.; Bassi, R.; Colombo, D.; Linardos, G.; Fiscarelli, E.V.; Sorrentino, R.; Miro, A. Hybrid lipid/polymer nanoparticles to tackle the cystic fibrosis mucus barrier in siRNA delivery to the lungs: Does PEGylation make the difference? ACS Appl. Mater. Interfaces 2022, 14, 7565–7578. [Google Scholar] [CrossRef] [PubMed]
- Craparo, E.F.; Cabibbo, M.; Scialabba, C.; Giammona, G.; Cavallaro, G. Inhalable Formulation Based on Lipid–Polymer Hybrid Nanoparticles for the Macrophage Targeted Delivery of Roflumilast. Biomacromolecules 2022, 23, 3439–3451. [Google Scholar] [CrossRef]
- Patel, M.; Karampuri, S.; Kansara, V.; Vyas, B. Inhalable dry powder containing lipid polymer hybrid nanoparticles of Nintedanib esylate: In vitro and in vivo evaluations. J. Drug Deliv. Sci. Technol. 2023, 86, 104716. [Google Scholar] [CrossRef]
- Wadhwa, A.; Bobak, T.R.; Bohrmann, L.; Geczy, R.; Sekar, S.; Sathyanarayanan, G.; Kutter, J.P.; Franzyk, H.; Foged, C.; Saatchi, K. Pulmonary delivery of siRNA-loaded lipid-polymer hybrid nanoparticles: Effect of nanoparticle size. OpenNano 2023, 13, 100180. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, L.; Sun, R.; Luo, B.; Zhou, Y.; Zhang, Y.; Liang, Y.; Xiao, B.; Wang, C. Pulmonary delivery of mucus-traversing PF127-modified silk fibroin nanoparticles loading with quercetin for lung cancer therapy. Asian J. Pharm. Sci. 2023, 18, 100833. [Google Scholar] [CrossRef]
- Pellosi, D.S.; d’Angelo, I.; Maiolino, S.; Mitidieri, E.; di Villa Bianca, R.d.E.; Sorrentino, R.; Quaglia, F.; Ungaro, F. In vitro/in vivo investigation on the potential of Pluronic® mixed micelles for pulmonary drug delivery. Eur. J. Pharm. Biopharm. 2018, 130, 30–38. [Google Scholar] [CrossRef]
- Godara, S.; Lather, V.; Kirthanashri, S.; Awasthi, R.; Pandita, D. Lipid-PLGA hybrid nanoparticles of paclitaxel: Preparation, characterization, in vitro and in vivo evaluation. Mater. Sci. Eng. C 2020, 109, 110576. [Google Scholar] [CrossRef]
- Guo, D.; Dou, D.; Li, X.; Zhang, Q.; Bhutto, Z.A.; Wang, L. Ivermection-loaded solid lipid nanoparticles: Preparation, characterisation, stability and transdermal behaviour. Artif. Cells Nanomed. Biotechnol. 2018, 46, 255–262. [Google Scholar] [CrossRef]
- Wang, H.; George, G.; Bartlett, S.; Gao, C.; Islam, N. Nicotine hydrogen tartrate loaded chitosan nanoparticles: Formulation, characterization and in vitro delivery from dry powder inhaler formulation. Eur. J. Pharm. Biopharm. 2017, 113, 118–131. [Google Scholar] [CrossRef]
- Das, S.; Lee, S.H.; Chia, V.D.; Chow, P.S.; Macbeath, C.; Liu, Y.; Shlieout, G. Development of microemulsion based topical ivermectin formulations: Pre-formulation and formulation studies. Colloids Surf. B Biointerfaces 2020, 189, 110823. [Google Scholar] [CrossRef]
- Ayoub, B.M.; Mowaka, S. LC–MS/MS determination of empagliflozin and metformin. J. Chromatogr. Sci. 2017, 55, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Qi, C.; Ruan, S.; Cao, G.; Ma, Z.; Zhang, X. Selenized polymer-lipid hybrid nanoparticles for oral delivery of tripterine with ameliorative oral anti-enteritis activity and bioavailability. Pharmaceutics 2023, 15, 821. [Google Scholar] [CrossRef]
- Gajra, B.; Dalwadi, C.; Patel, R. Formulation and optimization of itraconazole polymeric lipid hybrid nanoparticles (Lipomer) using box behnken design. DARU J. Pharm. Sci. 2015, 23, 3. [Google Scholar] [CrossRef]
- Khan, S.; Aamir, M.N.; Madni, A.; Jan, N.; Khan, A.; Jabar, A.; Shah, H.; Rahim, M.A.; Ali, A. Lipid poly (ɛ-caprolactone) hybrid nanoparticles of 5-fluorouracil for sustained release and enhanced anticancer efficacy. Life Sci. 2021, 284, 119909. [Google Scholar] [CrossRef] [PubMed]
- Tiyaboonchai, W.; Tungpradit, W.; Plianbangchang, P. Formulation and characterization of curcuminoids loaded solid lipid nanoparticles. Int. J. Pharm. 2007, 337, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Sabuj, M.Z.R.; Dargaville, T.R.; Nissen, L.; Islam, N. Inhaled ciprofloxacin-loaded poly (2-ethyl-2-oxazoline) nanoparticles from dry powder inhaler formulation for the potential treatment of lower respiratory tract infections. PLoS ONE 2021, 16, e0261720. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, A.; Savic, S.; Vuleta, G.; Mueller, R.H.; Keck, C.M. Polyhydroxy surfactants for the formulation of lipid nanoparticles (SLN and NLC): Effects on size, physical stability and particle matrix structure. Int. J. Pharm. 2011, 406, 163–172. [Google Scholar] [CrossRef]
- Honary, S.; Zahir, F. Effect of zeta potential on the properties of nano-drug delivery systems-a review (Part 2). Trop. J. Pharm. Res. 2013, 12, 265–273. [Google Scholar]
- Liu, Y.; Pan, J.; Feng, S.-S. Nanoparticles of lipid monolayer shell and biodegradable polymer core for controlled release of paclitaxel: Effects of surfactants on particles size, characteristics and in vitro performance. Int. J. Pharm. 2010, 395, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Tahir, N.; Madni, A.; Balasubramanian, V.; Rehman, M.; Correia, A.; Kashif, P.M.; Mäkilä, E.; Salonen, J.; Santos, H.A. Development and optimization of methotrexate-loaded lipid-polymer hybrid nanoparticles for controlled drug delivery applications. Int. J. Pharm. 2017, 533, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.; Chaves, L.L.; Lima, S.A.C.; Reis, S. Optimization of nanostructured lipid carriers loaded with methotrexate: A tool for inflammatory and cancer therapy. Int. J. Pharm. 2015, 492, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, G.; Jin, S.; Xu, L.; Zhao, C.X. Development of high-drug-loading nanoparticles. ChemPlusChem 2020, 85, 2143–2157. [Google Scholar] [CrossRef]
- Shen, S.; Wu, Y.; Liu, Y.; Wu, D. High drug-loading nanomedicines: Progress, current status, and prospects. Int. J. Nanomed. 2017, 12, 4085–4109. [Google Scholar] [CrossRef]
- Li, Y.; Wong, H.L.; Shuhendler, A.J.; Rauth, A.M.; Wu, X.Y. Molecular interactions, internal structure and drug release kinetics of rationally developed polymer–lipid hybrid nanoparticles. J. Control. Release 2008, 128, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Gan, Q.; Dai, D.; Yuan, Y.; Qian, J.; Sha, S.; Shi, J.; Liu, C. Effect of size on the cellular endocytosis and controlled release of mesoporous silica nanoparticles for intracellular delivery. Biomed. Microdevices 2012, 14, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Reddy, P. Effect of surfactant on azithromycin dihydrate loaded stearic acid solid lipid nanoparticles. Turk. J. Pharm. Sci. 2019, 16, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Sengel-Turk, C.T.; Hascicek, C. Design of lipid-polymer hybrid nanoparticles for therapy of BPH: Part I. Formulation optimization using a design of experiment approach. J. Drug Deliv. Sci. Technol. 2017, 39, 16–27. [Google Scholar] [CrossRef]
- Jahromi, L.P.; Ghazali, M.; Ashrafi, H.; Azadi, A. A comparison of models for the analysis of the kinetics of drug release from PLGA-based nanoparticles. Heliyon 2020, 6, e03451. [Google Scholar] [CrossRef] [PubMed]
- Soomherun, N.; Kreua-Ongarjnukool, N.; Niyomthai, S.T.; Chumnanvej, S. Lipid-Polymer Hybrid Nanoparticles Synthesized via Lipid-Based Surface Engineering for a robust drug delivery platform. Colloids Surf. B Biointerfaces 2024, 237, 113858. [Google Scholar] [CrossRef] [PubMed]
- Kassaee, S.N.; Nili-Ahmadabadi, A.; Mahboobian, M.M. Fabrication of poloxamer based besifloxacin thermosensitive in situ gelling nanoemulsions for ophthalmic delivery. J. Bioact. Compat. Polym. 2023, 38, 298–310. [Google Scholar] [CrossRef]
- Abou-Elnour, F.S.; El-Habashy, S.E.; Essawy, M.M.; Abdallah, O.Y. Codelivery of ivermectin and methyl dihydrojasmonate in nanostructured lipid carrier for synergistic antileukemia therapy. Int. J. Pharm. 2024, 656, 124086. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Z.; Zhu, S.; Cheng, Y.; Liu, J.; Ye, T.; Wang, S. Docetaxel liposomes for lung targeted delivery: Development and evaluation. Pharm. Dev. Technol. 2023, 28, 856–864. [Google Scholar] [CrossRef]
- Velho, M.C.; Funk, N.L.; Deon, M.; Benvenutti, E.V.; Buchner, S.; Hinrichs, R.; Pilger, D.A.; Beck, R.C.R. Ivermectin-Loaded Mesoporous Silica and Polymeric Nanocapsules: Impact on Drug Loading, In Vitro Solubility Enhancement, and Release Performance. Pharmaceutics 2024, 16, 325. [Google Scholar] [CrossRef]
- Valarini Junior, O.; Cardoso, F.A.R.; de Souza, G.B.M.; Machado Giufrida, W.; Cardozo-Filho, L. Single step encapsulation process of ivermectin in biocompatible polymer using a supercritical antisolvent system process. Asia-Pac. J. Chem. Eng. 2021, 16, e2672. [Google Scholar] [CrossRef]
- Lu, M.; Xiong, D.; Sun, W.; Yu, T.; Hu, Z.; Ding, J.; Cai, Y.; Yang, S.; Pan, B. Sustained release ivermectin-loaded solid lipid dispersion for subcutaneous delivery: In vitro and in vivo evaluation. Drug Deliv. 2017, 24, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Afzal, M.; Verma, M.; Misra-Bhattacharya, S.; Ahmad, F.J.; Dinda, A.K. Improved antifilarial activity of ivermectin in chitosan–alginate nanoparticles against human lymphatic filarial parasite, Brugia malayi. Parasitol. Res. 2013, 112, 2933–2943. [Google Scholar] [CrossRef] [PubMed]
- Noori, T.; Dehpour, A.R.; Alavi, S.D.; Hosseini, S.Z.; Korani, S.; Sureda, A.; Esmaeili, J.; Shirooie, S. Synthesis and evaluation of the effects of solid lipid nanoparticles of ivermectin and ivermectin on cuprizone-induced demyelination via targeting the TRPA1/NF-kB/GFAP signaling pathway. Iran. J. Basic Med. Sci. 2023, 26, 1272–1282. [Google Scholar] [PubMed]
- Othman, R.; Vladisavljevic, G.T.; Nagy, Z.K.; Holdich, R. Encapsulation and controlled release of rapamycin from polycaprolactone nanoparticles prepared by membrane micromixing combined with antisolvent precipitation. Langmuir 2016, 32, 10685–10693. [Google Scholar] [CrossRef] [PubMed]
- Macho, O.; Demková, K.; Gabrišová, Ľ.; Čierny, M.; Mužíková, J.; Galbavá, P.; Nižnanská, Ž.; Blaško, J.; Peciar, P.; Fekete, R. Analysis of static angle of repose with respect to powder material properties. Acta Polytech. 2020, 60, 73–80. [Google Scholar] [CrossRef]
- Hu, J.; Dong, Y.; Pastorin, G.; Ng, W.K.; Tan, R.B. Spherical agglomerates of pure drug nanoparticles for improved pulmonary delivery in dry powder inhalers. J. Nanopart. Res. 2013, 15, 1560. [Google Scholar] [CrossRef]
- Sinha, B.; Mukherjee, B. Development of an inhalation chamber and a dry powder inhaler device for administration of pulmonary medication in animal model. Drug Dev. Ind. Pharm. 2012, 38, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Marante, T.; Viegas, C.; Duarte, I.; Macedo, A.S.; Fonte, P. An overview on spray-drying of protein-loaded polymeric nanoparticles for dry powder inhalation. Pharmaceutics 2020, 12, 1032. [Google Scholar] [CrossRef]
- Wang, H.; Holgate, J.; Bartlett, S.; Islam, N. Assessment of nicotine release from nicotine-loaded chitosan nanoparticles dry powder inhaler formulations via locomotor activity of C57BL/6 mice. Eur. J. Pharm. Biopharm. 2020, 154, 175–185. [Google Scholar] [CrossRef]




| Formulation Code | PCL (mg) | Lecithin (mg) | Pluronic F127 (%w/v) | IVM (mg) |
|---|---|---|---|---|
| F1 | 45 | 30 | 0.25 | 6 |
| F2 | 45 | 45 | 0.25 | 6 |
| F3 | 45 | 60 | 0.25 | 6 |
| F4 | 45 | 30 | 0.5 | 6 |
| F5 | 45 | 30 | 0.75 | 6 |
| Before Freeze-Drying | After Freeze-Drying | |||||
|---|---|---|---|---|---|---|
| Particle Size (nm) | PDI | Zeta Potential (mV) | Particle Size (nm) | PDI | Zeta Potential (mV) | |
| F1 | 350.70 ± 16.04 | 0.32 ± 0.06 | −32.03 ± 1.36 | 438.39 ± 11.26 | 0.38 ± 0.01 | −36.92 ± 0.76 |
| F2 | 320.34 ± 14.81 | 0.28 ± 0.04 | −34.16 ± 1.25 | 382.12 ± 13.47 | 0.34 ± 0.03 | −38.38 ± 1.03 |
| F3 | 302.19 ± 19.27 | 0.32 ± 0.09 | −33.47 ± 1.63 | 346.83 ± 14.75 | 0.33 ± 0.04 | −37.24 ± 0.82 |
| F4 | 323.01 ± 7.24 | 0.42 ± 0.07 | −37.98 ± 1.94 | 368.60 ± 5.39 | 0.39 ± 0.02 | −43.15 ± 0.88 |
| F5 | 350.75 ± 27.57 | 0.38 ± 0.09 | −38.82 ± 1.69 | 465.17 ± 28.09 | 0.44 ± 0.04 | −43.8 ± 0.70 |
| Formulation Code | Entrapment Efficiency % | Drug Loading % |
|---|---|---|
| F1 | 80.59 ± 0.93 | 10.15 ± 0.22 |
| F2 | 75.40 ± 1.56 | 9.23 ± 0.24 |
| F3 | 71.65 ± 1.54 | 8.23 ± 0.25 |
| F4 | 74.64 ± 2.01 | 8.02 ± 0.35 |
| F5 | 68.32 ± 1.62 | 7.38 ± 0.31 |
| Formulation | Zero-Order | First-Order | Higuchi | Hixson–Crowell | Korsmeyer–Peppas | |
|---|---|---|---|---|---|---|
| r2 | r2 | r2 | r2 | r2 | n | |
| F1 | 0.6790 | 0.7517 | 0.8632 | 0.7284 | 0.8088 | 0.9112 |
| F2 | 0.7143 | 0.7778 | 0.8929 | 0.7573 | 0.7935 | 0.7858 |
| F3 | 0.7552 | 0.8285 | 0.9191 | 0.8054 | 0.8108 | 0.8108 |
| F4 | 0.7296 | 0.7950 | 0.9025 | 0.7739 | 0.8244 | 0.8138 |
| F5 | 0.6806 | 0.7541 | 0.8723 | 0.7304 | 0.7763 | 0.8146 |
| Blank LPHNP | F1 | F2 | F3 | F4 | F5 | |
|---|---|---|---|---|---|---|
| Bulk density | 0.21 ± 0.00 | 0.23 ± 0.01 | 0.19 ± 0.00 | 0.27 ± 0.01 | 0.15 ± 0.01 | 0.16 ± 0.01 |
| Tapped density | 0.25 ± 0.00 | 0.27 ± 0.01 | 0.23 ± 0.01 | 0.33 ± 0.01 | 0.20 ± 0.00 | 0.20 ± 0.00 |
| CI | 14.37 ± 2.3 | 13.31 ± 1.69 | 18.94 ± 3.43 | 16.37 ± 0.51 | 22.36 ± 3.88 | 20.86 ± 2.21 |
| HR | 1.17 ± 0.03 | 1.16 ± 0.01 | 1.24 ± 0.06 | 1.19 ± 0.01 | 1.29 ± 0.07 | 1.26 ± 0.03 |
| θ | 29.52 ± 1.89 | 28.26 ± 1.23 | 35.37 ± 0.95 | 31.31 ± 1.81 | 37.52 ± 0.91 | 36.99 ± 0.91 |
| Formulation | RD% | ED% | FPF% | FPD (µg) |
|---|---|---|---|---|
| Blank LPHNP | 95.20 ± 1.79 | 90.13 ± 0.59 | 24.26 ± 0.70 | - |
| F1 | 98.44 ± 1.16 | 90.55 ± 0.51 | 24.77 ± 1.69 | 438.09 ± 61.34 |
| F2 | 95.19 ± 1.82 | 87.59 ± 0.77 | 19.81 ± 1.19 | 331.01 ± 26.62 |
| F3 | 98.18 ± 0.97 | 89.90 ± 1.32 | 22.45 ± 1.83 | 410.68 ± 48.65 |
| F4 | 97.53 ± 0.84 | 87.79 ± 0.83 | 18.53 ± 0.87 | 314.32 ± 35.76 |
| F5 | 96.76 ± 0.59 | 89.66 ± 0.40 | 21.70 ± 1.51 | 379.86 ± 33.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kassaee, S.N.; Ayoko, G.A.; Richard, D.; Wang, T.; Islam, N. Inhaled Ivermectin-Loaded Lipid Polymer Hybrid Nanoparticles: Development and Characterization. Pharmaceutics 2024, 16, 1061. https://doi.org/10.3390/pharmaceutics16081061
Kassaee SN, Ayoko GA, Richard D, Wang T, Islam N. Inhaled Ivermectin-Loaded Lipid Polymer Hybrid Nanoparticles: Development and Characterization. Pharmaceutics. 2024; 16(8):1061. https://doi.org/10.3390/pharmaceutics16081061
Chicago/Turabian StyleKassaee, Seyedeh Negin, Godwin A. Ayoko, Derek Richard, Tony Wang, and Nazrul Islam. 2024. "Inhaled Ivermectin-Loaded Lipid Polymer Hybrid Nanoparticles: Development and Characterization" Pharmaceutics 16, no. 8: 1061. https://doi.org/10.3390/pharmaceutics16081061
APA StyleKassaee, S. N., Ayoko, G. A., Richard, D., Wang, T., & Islam, N. (2024). Inhaled Ivermectin-Loaded Lipid Polymer Hybrid Nanoparticles: Development and Characterization. Pharmaceutics, 16(8), 1061. https://doi.org/10.3390/pharmaceutics16081061






