Abstract
Black chokeberry juice production results in a large amount of pomace, which is usually regarded as waste. Nevertheless, it contains significant amounts of anthocyanins, which can be utilized as health-promoting components, but also as food colorants. To take advantage of their benefits, green extraction methods such as microwave-assisted (MAE) and ultrasound-assisted extraction (UAE) are widely used for their isolation. This study aimed to evaluate the effects of MAE and UAE parameters (solvent, treatment time, temperature, or ultrasound amplitude) on the extraction yield of anthocyanins from black chokeberry pomace and to compare the effectiveness of these two green extraction methods with conventional reflux extraction, both in terms of total anthocyanins yield and effects on individual compounds. In both techniques, acidification of the extraction solvent did not show a significant effect on anthocyanin content. For MAE, a temperature increase from 40 to 60 °C positively affected the extraction yield, while 4 min was a substantial treatment time for the extraction. Conversely, UAE required 10 min of treatment time with no effect on amplitude. UPLC ESI-MS2 analysis confirmed the presence of 6 anthocyanins in the obtained extracts, with significantly higher levels of cyanidin-3-O-xyloside and cyanidin-3-O-arabinoside were in ones isolated by green extraction techniques.
1. Introduction
Aronia melanocarpa, or black chokeberry, is a shrub belonging to the Rosaceae family, originating from North America. Its fruit is a rich source of polyphenols, especially anthocyanins, which are responsible for its dark, deep purple-red color. Compared to other berries, chokeberry has been reported to have the highest polyphenol and anthocyanin contents, as well as antioxidant activity [1,2]. However, due to the high content of proanthocyanidins among black chokeberry polyphenols, it has an expressed astringency when consumed as a fresh berry [3]. Therefore, it is usually processed into a variety of products such as juice, jam, tea, wine, etc. Black chokeberry processing, especially the production of juice by pressing, results in large amounts of pomace, which is usually regarded as waste. Nevertheless, the remaining pomace contains significant amounts of polyphenols and anthocyanins. It has been reported that depending on the fruit characteristics and juice processing methodology, chokeberry pomace anthocyanin content ranges from 5 to 20 mg/g d.w., with exceptions reaching even 115 mg/g d.w. [4]. In order to achieve the minimization of waste generation and thereby fulfill the circular economy and sustainability concept, one of the main steps is the valorization of waste [5]. It has been reported that in Europe, juice production waste itself makes approximately 3% of total waste disposal, which makes it the fifth main contributor to total food waste [6]. In this context, chokeberry juice production residue, the pomace, makes a valuable by-product for the exploitation of its beneficial properties due to the high presence of anthocyanins.
Anthocyanins, as water soluble plant flavonoids, express numerous beneficial health effects through their antioxidant, antimicrobial, and anti-inflammatory action [7,8]. Thereby, they can be utilized as health-promoting components, but also as food colorants with preference to synthetic ones because of their beneficial effects [9]. However, their structure, characterized by unsaturated double bonds and easily oxidized groups, makes them prone to degradation changes caused by external factors such as pH, temperature, presence of oxygen, light, metal ions, etc. [10]. Therefore, their isolation from the food matrix, in this specific case the pomace, is a crucial step to their future application. Generally, anthocyanins are extracted by water or alcohol, such as ethanol and methanol, or water-alcohol mixtures and their acidified counterparts [11,12,13] due to the increased stability of anthocyanins in acidic media [14].
In terms of an environmentally friendly approach and green chemistry principle, nowadays, new, green extraction techniques have become the methods of choice for the isolation of bioactive molecules from plant material [15,16]. Among the green extraction techniques, microwave-assisted (MAE) and ultrasound-assisted extraction (UAE) are widely applied [17,18,19,20,21]. The MAE principle is based on the application of microwave energy, which is absorbed by the moisture present in the matrix, resulting in the swelling and rupture of plant cells, thereby enhancing the release of bioactives [22]. UAE applies ultrasound energy, which also causes cell wall disruption and enhanced bioactives release, but through a cavitation effect [18]. Although based on different mechanisms, both techniques provide the reduction of extraction time, lower solvent consumption, and minimized degradation of bioactives, and are therefore considered as green and more environmentally friendly than conventional extraction methods [23,24]. The majority of published studies emphasize the advantages of MAE and UAE for anthocyanins isolation in terms of enhancing the extraction efficiency while providing the shorter extraction time. For instance, UAE was shown to be more effective than conventional techniques for extraction of anthocyanins in red raspberry, blueberry wine pomace, and Hibiscus sabdariffa calyces [25,26,27]. Similarly, MAE showed to be more time and yield efficient in the isolation of anthocyanins from sour cherry, Chinese bayberry, blackberry, and black currant marc [22,28,29,30]. On the other hand, both microwaves and ultrasound can cause some adverse effects that could have negative effects on the overall quality of the extract. In the case of UAE, formation of OH-radicals during the cavitation phenomenon can cause degradation of active components [23], while MAE was reported to cause degradation of anthocyanin glycosides into their aglycone form [28]. Therefore, in order to make the most of their advantages and overcome the mentioned drawbacks, it is essential to carefully evaluate and select the extraction parameters, such as solvent type, temperature, time and microwave power, or ultrasound amplitude. For instance, elevated temperatures generally enhance the extraction process, but also can lead to selectivity decrease and degradation of thermolabile bioactives, similarly to prolonged irradiation or sonication time [22,31,32]. Higher microwave power and ultrasound amplitude favor the release of bioactives from plant tissue, but on the other hand, they have to be carefully considered in relation to treatment duration, as they can also increase the degradation rate. Thus, it is of an utmost importance to optimize each extraction process with regard to the material and target compound characteristics.
Several studies have been published on the extraction of various phenolic compounds from black chokeberry pomace, including the few focused on different extraction methods [33,34,35,36,37]. MAE was applied in terms of method optimization only for the isolation of phenolic compounds from the fresh berry and juice [38,39]. To the best of our knowledge, no study has reported the simultaneous optimization of both MAE and UAE for the isolation of black chokeberry pomace anthocyanins. Therefore, the aim of this study was to evaluate the effect of MAE and UAE parameters on the extraction yield of black chokeberry pomace anthocyanins and to compare the effectiveness of those two green extraction methods to conventional reflux extraction (CE) in terms of total anthocyanins yield, as well as the effect on individual compounds.
2. Materials and Methods
2.1. Chemicals and Reagents
Ethanol (96%) used for the extractions was obtained from Lach-ner (Neratovice, Czech Republic), HPLC-grade acetonitrile was obtained from J.T. Baker Chemicals (Deventer, The Netherlands), and formic acid (98–100%) was obtained from T.T.T. Ltd. (Sveta Nedjelja, Croatia). Distilled water was Milli-Q purity (Millipore, Bedford, MA, USA). Commercial standard for cyanidin-3-O-glucoside chloride was procured from Sigma-Aldrich (St. Louis, MO, USA). HPLC-grade methanol used for the dissolution of standards and preparation of stock and working solutions was from BDH Prolabo, UK.
2.2. Material
Chokeberry pomace used in the experiments was obtained as a by-product of juice pressing from Terra Food Ltd. company (Koprivnica, Croatia). Fruits of black chokeberry used for juice production were of cultivar Nero from organic cultivation, harvested in the middle of September when fully mature. Obtained pomace was frozen and stored at −18 °C. Prior to extraction, pomace was defrosted at room temperature and ground with an electric mill (GT11, Tefal, Rumilly, France).
2.3. Extraction of Anthocyanins
2.3.1. MAE
For MAE of anthocyanins from chokeberry pomace, the Ethos Easy (Milestone, Sorisole, Italy) microwave reactor was used. The following general extraction parameters were kept constant during the process: time to reach extraction temperature 2 min, agitation 50%, microwave power 400 W, and aeration after extraction 1 min. Extractions were performed according to a mixed level full factorial design, varying the extraction solvent (50% aqueous ethanol solution and 1% formic acid in 50% aqueous ethanol solution), temperature (40, 60, and 80 °C), and irradiation time (4, 6, 8, and 10 min). For each extraction, 1 g of the ground sample was mixed with the appropriate solvent in the extraction vessel with a magnetic stirrer and placed into the microwave reactor. The extractions were performed for the specified extraction time, while the temperature specified according to the experimental design was kept constant. Subsequently, the extracts were filtered through Whatman No. 40 filter paper into 50-mL volumetric flasks, made up with solvent, and stored in plastic Falcon tubes at −18 °C until analysis. All extractions were performed in duplicate.
2.3.2. UAE
The UP200Ht ultrasound processor (Dr. Hielscher GmbH, Teltow, Germany) with a maximum rated output power of 200 W and an ultrasound frequency of 26 kHz, equipped with a 154 mm2 titanium ultrasound probe, was used for UAE of chokeberry pomace anthocyanins. Extractions were performed using a mixed level full factorial design, varying the extraction solvent (50% aqueous ethanol solution and 1% formic acid in 50% aqueous ethanol solution), ultrasound amplitude (25, 50, and 75%), and sonication time (4, 6, 8, and 10 min). For extraction, the ground sample (1 g) and appropriate solvent were mixed in a 100-mL glass beaker. During extraction, the ultrasonic probe was immersed 1 cm deep in the extraction mixture, while the temperature was controlled with an infrared thermometer and kept below 30 °C with an ice cooling bath. Then, the extracts were filtered through Whatman No. 40 filter paper, made up to 50 mL in volumetric flasks, and stored in plastic Falcon tubes at −18 °C. All extractions were performed in duplicate.
2.3.3. CE
For the CE of the anthocyanins from chokeberry pomace, 1 g of the ground sample and 40 mL of the extraction solvent determined after evaluation of MAE and UAE were submitted to reflux in a flat bottom Erlenmeyer flask. Extraction was performed for 30 min, starting with the reaching of the solvent boiling point. Afterwards, the extract was filtered (Whatman No. 40 filter paper, Whatman International Ltd., Kent, UK), made up to volume with the extraction solvent in a 50 mL volumetric flask, and stored in a plastic Falcon tube at −18 °C. The extract was prepared in duplicate.
2.4. Determination of Total Monomeric Anthocyanins
The determination of total monomeric anthocyanins was performed using a spectrophotometric pH differential method (AOAC) [40]. Briefly, 1 mL of the extract was mixed with 4 mL of 0.025 M potassium chloride buffer (pH 1.0) and 1 mL with 4 mL of 0.4 M sodium acetate buffer (pH 4.5). The reaction mixture was left at room temperature for 20 min and absorbance was measured using a spectrophotometer at 520 and 700 nm, using distilled water as a blank. The concentration of monomeric anthocyanins in the sample was calculated as equivalent to cyanidin-3-glucoside (mg/L) according to the equation:
where: A = (A520nm − A700nm) pH = 1.0 − (A520nm − A700nm) pH = 4.5;
- MW = molecular weight (for cyanidin-3-glucoside 449.2 g/mol);
- DF = dilution factor;
- 103 = factor for conversion g to mg;
- ε = molar absorption extinction coefficient (for cyanidin-3-glucoside 26,900 L/mol cm);
- l = cuvette thickness (1 cm).
All measurements were performed in duplicate and results were expressed as mg/100 g of the sample.
2.5. UPLC ESI-MS2 Analysis of Individual Anthocyanins
Individual anthocyanins in extracts of chokeberry pomace were analyzed by the UPLC ESI-MS2 on Agilent 6430 Triple Quad LC/MS mass spectrometer (Agilent, Santa Clara, CA, USA) connected to the UPLC system (Agilent series 1290 RRLC instrument) and equipped with a binary pump, autosampler, and a column compartment thermostat, according to the method described by Elez Garofulić et al. (2018) [18]. Separation was carried out using the Zorbax Eclipse Plus C18 column (1.8 µm, 100 × 2.1 mm I.D.) kept at 35 °C with 2.5 µL injection volume. Ionization was done in ESI positive mode by nitrogen (99.999%, Messer, Croatia) (inducing cone and collision gas) under the following ionization source parameters: drying gas temperature 300 °C, flow rate 11 L/h, nebulizer pressure 40 psi, and positive/negative capillary voltage 4000/3500 V. Data were qualified and quantified in the MRM mode and analyzed using MassHunter software. All analyses were performed in duplicate.
The analytical method quality parameters, namely calibration curve, instrumental detection (LOD), and quantification (LOQ) limits, were determined using an external standard calibration methodology with cyanidin-3-O-glucoside chloride. A calibration curve was prepared by diluting the stock solution of cyanidin-3-O-glucoside (38 mg/L) in order to obtain five consecutive dilutions. Each concentration was injected in triplicate and analyzed by optimized parameters of retention time (3.005 min), precursor and product ion (m/z 485, 287), fragmentor voltage (120 V), and collision energy (19 V) in positive ionization mode. The regression equation thereby obtained was y = 2124.4x + 513.77 with the coefficient of determination R2 = 0.999 and area of linearity 0.02533–38.00 mg/L. The LOD was calculated at a signal to noise ratio of 3:1 and LOQ at 10:1 and were 0.0025 mg/L and 0.0077 mg/L, respectively.
The identification of individual chokeberry anthocyanins was done by comparison of the retention time of analyzed compounds (Rt) with the one of cyanidin-3-O-glucoside chloride standard, polarity, and comparing the characteristic mass spectral data and previously reported mass fragmentation patterns specific for each compound. Quantification of all identified compounds was performed according to the cyanidin-3-O-glucoside chloride calibration curve and the results were expressed as mg per 100 g of the sample.
2.6. Statistical Analysis
Statistical analysis was carried out using Statistica ver. 10.0 software (Statsoft Inc., Tulsa, OK, USA). All extractions and all analyses were performed in duplicate. A mixed level full factorial design comprising 24 experimental trials was employed for the evaluation of the effect of three independent variables, namely solvent, microwave power, and irradiation time on the TA content of chokeberry pomace during MAE, whereas during UAE, observed independent variables were solvent, ultrasound amplitude, and sonication time. The normality and homoscedasticity of the data were analyzed using the Shapiro–Wilk’s test and Levene’s test, respectively. Normally distributed data were analyzed using one-way and multifactorial analysis of variance (ANOVA), while marginal means between groups were compared with Tukey’s HSD multiple comparison test. Nonparametric data were analyzed by the Kruskal–Wallis test. The significance level for all tests was p ≤ 0.05, and the results of statistical analysis are presented as mean ± standard error (SE).
3. Results and Discussion
3.1. MAE of Black Chokeberry Pomace Anthocyanins
MAE of black chokeberry pomace anthocyanins was carried out varying the conditions of the acidification of the extraction solvent (50% EtOH or 1% HCOOH in 50% EtOH), temperature (40, 60, or 80 °C), and irradiation time (4, 6, 8, and 10 min). The extraction parameters span taken into consideration was selected upon previous research on MAE [17,21,22,32]. Based on the same studies, where microwave power was applied only for a fraction of the treatment time needed to achieve and maintain the temperature at a constant level and was usually found not significant for extraction yield, we have kept it constant for all treatments at 400 W. The extraction solvent, namely 50% aqueous ethanol solution, was chosen based on the report by Oancea et al. (2012) [41], Simic et al. (2016) [38], D’Alessandro et al. (2012) [42], and general conclusions regarding the suitability of water-alcohol mixtures for the recovery of anthocyanins [15,43]. However, the acidification of the solvent with 1% formic acid was taken into evaluation because of the effect on the stability of anthocyanin flavylium cations.
The results of the total anthocyanins (TA) content determination in black chokeberry pomace extract isolated under different MAE conditions are shown in Table 1. It can be observed that depending on the applied conditions, TA content ranged from 226.71 to 552.58 mg/100 g. The lowest values were obtained at the lowest applied temperature, while more intensive treatment resulted with higher TA concentrations, especially when observing the effect during longer treatment times (Supplementary Figures S1 and S2). The obtained range is in accordance with the values reported for pomace by Vásquez-Espinoza et al. (2019) [44] and those reported for the flesh and whole fruit by Kaloudi et al. (2022) [45].

Table 1.
Total anthocyanin (TA) content in black chokeberry pomace extracts isolated under different MAE conditions.
Statistical analysis (Table 2) showed the significant influence of temperature and irradiation time during MAE on TA content, while the acidification of the extraction solvent was not a significant factor in analysis, although acidified extracts had slightly higher amounts of TA. Nevertheless, although not influencing the extraction yield, acidification affects the stability of the obtained extracts, which could be crucial for future applications [15].

Table 2.
The effect of extraction solvent, temperature, and irradiation time on the TA content of black chokeberry pomace during MAE.
The elevation of temperature from 40 to 60 and 80 °C positively affected the content of TA, with no statistically significant difference between 60 and 80 °C, thereby implicating the moderate temperature of 60 °C to be optimal for MAE of black chokeberry pomace anthocyanins. Similar studies on MAE of anthocyanin-rich materials showed similar conclusions. For instance, our study on MAE of sour cherry anthocyanins [22] showed that a temperature increase up to 60 °C enhanced the extraction yield. Jafari et al. (2019) [46] concluded that 50 °C is the optimal temperature for MAE of saffron tepals’ anthocyanins, as further elevation resulted in their degradation. Temperature elevation generally enhances the extraction yield through an increase in anthocyanin solubility in extraction solvent and mass transfer acceleration [47,48]. However, the temperature effect is the most evident when observed in the context of treatment duration. Generally, it is considered that the loss of anthocyanins occurs at treatments longer than 10 min, regardless of the applied temperature [46]. Therefore, in this study, we have only included treatment up to 10 min, and it was shown that the highest extraction yield is obtained within the shortest irradiation time of 4 min. Liu et al. (2019) [49] reported that the anthocyanins from purple sweet potato pomace were fully extracted after approximately 8 min of irradiation time at 320 W, which could be correlated to our shorter irradiation time at higher microwave power/temperature. A short treatment of 4 min, like the one in our study, was reported as optimal for MAE of blueberry anthocyanins [29].
3.2. UAE of Black Chokeberry Pomace Anthocyanins
The UAE of black chokeberry pomace anthocyanins was performed varying the same solvent and treatment time parameters as in MAE under three different ultrasound amplitudes, namely 25, 50, and 75%, of ultrasound nominal power of 200 W. The results of TA content determination in the extracts obtained under different UAE conditions of solvent acidification, amplitude, and sonication time are shown in Table 3. The concentration range of TA was from 252.73 to 567.32 mg/100 g, which is similar to the range obtained during MAE experiments. Generally, there was an increase in TA content with prolongation of treatment time, which can be observed visually from 3D surface plots for each of the solvents used (Supplementary Figures S3 and S4).

Table 3.
Total anthocyanin (TA) content in black chokeberry pomace extracts isolated under different UAE conditions.
Statistical analysis of the effect of UAE parameters (Table 4) confirmed the significant influence of sonication time on TA content, while solvent composition and ultrasound amplitude had no effect.

Table 4.
The effect of extraction solvent, ultrasound amplitude, and sonication time on the content of black chokeberry pomace TA during UAE.
Acidification of the extraction solvent during UAE showed a similar trend of a slight increase in TA content like in MAE, although with no statistical significance. Furthermore, the increase in the applied ultrasound amplitude caused the slight increase in TA extraction yield, again with no statistically significant change, thereby implicating that the lowest applied amplitude is substantial for the effective isolation of black chokeberry pomace anthocyanins. As ultrasound amplitude represents a number of ultrasonic waves expansion and compression cycles, it indicates the intensity of sonication transmitted to the plant material [17,44]. Generally, the higher the sonication effect is, the extraction is more efficient. However, the characteristics of the plant material, of targeted compounds themselves, may determine different behavior of the extraction yield. Tiwari et al. (2008) [50] observed that the increase in ultrasound amplitude above 40% during treatment time longer than 5 min caused a decrease in the anthocyanin content of strawberry juice. The authors suggested that inadequacy of higher amplitudes may be attributed to the formation of free radicals [50,51]. Additionally, the application of higher amplitudes results in temperature elevation during the sonication process, which then consequently enhances the extraction yield. However, in our study, temperature was kept constant and below 30 °C by external cooling, thereby annulling the temperature effect, which could be the reason for the lack of significance of the amplitude increase. Sonication time was, on the other hand, shown to be the main factor affecting the UAE efficiency. Prolongation of the treatment time caused the increase in TA content, reaching the highest value at the longest time of 10 min. Ryu and Koh (2019) [52] reported that the effect of sonication time was greater than the one of amplitude on the TA content of black soybeans, causing a linear increase from 1 to 9 min, which is in accordance with our findings. Similarly, Sady et al. (2019) [35] concluded that the optimal sonication time for the extraction of black chokeberry anthocyanins is 13 min, while for total phenolics, treatment time is longer, 20 min. The results obtained in our study, as well as in mentioned literature reports, contribute to the fact that anthocyanins, due to their structure and proneness to degradation changes, require a moderate condition for their isolation even when novel “green” techniques are being applied.
3.3. Characterization and Comparison of Optimized MAE and UAE Extracts with CE
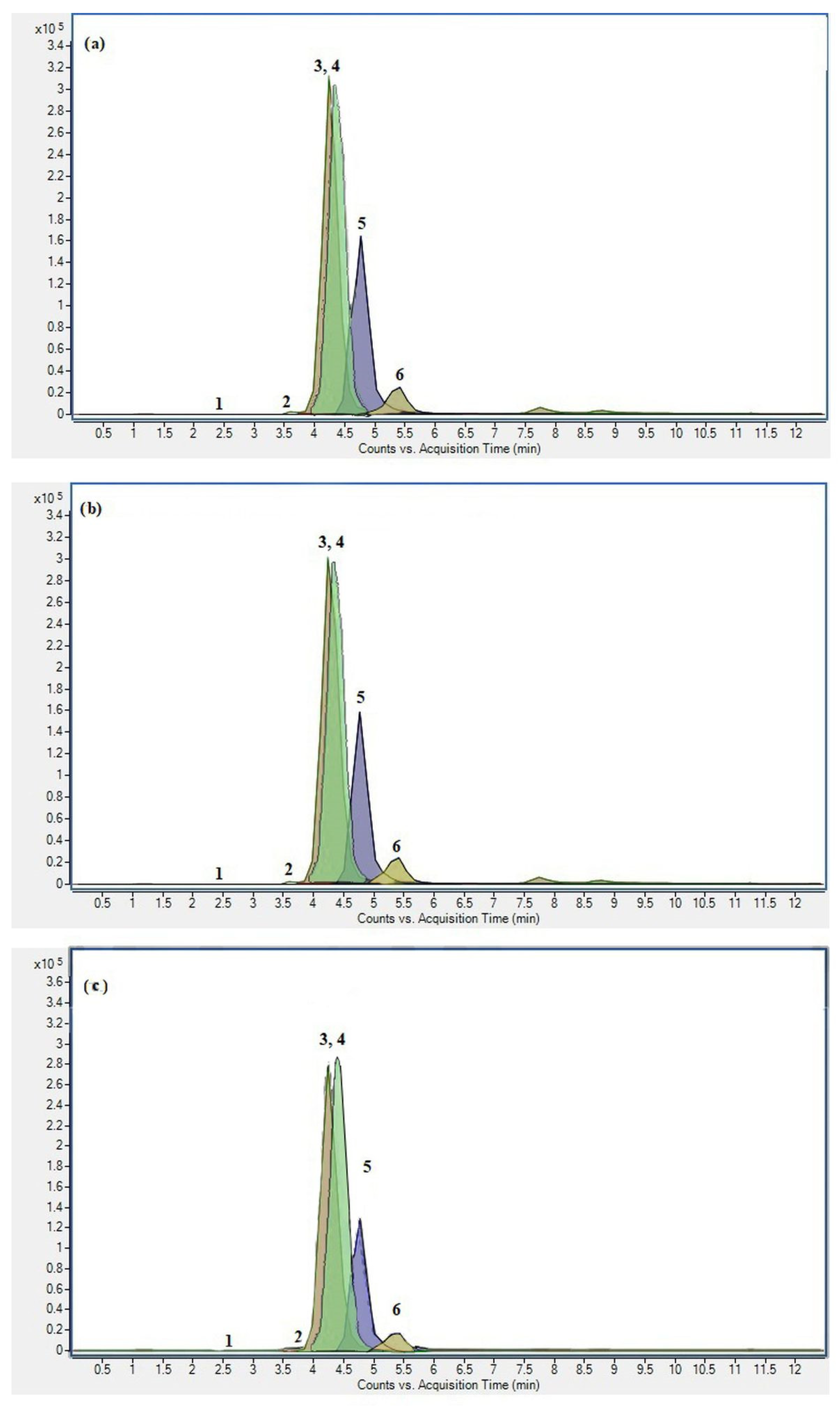
After evaluation of the effect of MAE and UAE parameters on the content of TA of black chokeberry pomace, optimal conditions were established for each technique, namely 60 °C and 4 min for MAE and 25% amplitude and 10 min for UAE, while 1% HCOOH in 50% EtOH was applied as the extraction solvent in both cases. Thus, optimized extracts were compared to conventional reflux extraction in a duration of 30 min for their TA content and, additionally, individual anthocyanin composition determined by UPLC ESI-MS2. Regardless of the extraction method, six anthocyanins were identified in pomace extracts (Figure 1).
Figure 1.
UPLC ESI-MS2 chromatogram in MRM acquisition mode of black chokeberry pomace extracts obtained under (a) optimized MAE, (b) optimized UAE, and (c) CE. (1) Cyanidin-3-hexoside-(epi)catechin, (2) Cyanidin-3-pentoside-(epi)catechin, (3) Cyanidin-3-O-galactoside, (4) Cyanidin-3-O-glucoside, (5) Cyanidin-3-O-arabinoside, and (6) Cyanidin-3-O-xyloside.
Among them, compound 4 was identified using an authentic standard as cyanidin-3-O-glucoside, while other compounds were tentatively assigned according to their characteristic MS2 spectra, fragmentation patterns, and literature reports. Compounds 1 and 2, although in low concentrations, were detected in all extracts. According to their precursor ion m/z 737 and 707, and obtained product ions, they were assigned as cyanidin-3-hexoside-(epi)catechin and cyanidin-3-pentoside-(epi)catechin, respectively, as the presence of the same compounds with the exact fragmentation pattern was reported previously by Oszmiański and Lachowicz (2016) in black chokeberry products [53]. Compounds 3, 5, and 6 were characterized by the same product ion (m/z 287), implicating the structure of cyanidin glycosides. As compound 4 was confirmed as glucoside by commercial standard, compound 3 was assigned as cyanidin hexoside due to the same fragmentation pattern and loss of −162 amu, characteristic for hexose moiety. As several reports confirmed the presence of two cyanidin hexosides in chokeberry, namely glucoside and galactoside, compound 3 was identified as cyanidin-3-O-galactoside [45,53,54]. Compounds 5 and 6 showed the same fragmentation pathway, precursor ion at m/z 419, and product ion at m/z 287, implicating the loss of pentose moiety and structure of cyanidin pentosides. Literature reports confirm the presence of two pentosides in chokeberry, cyanidin-3-O-arabinoside and cyanidin-3-O-xyloside [9,53,54,55], which were, according to their polarity, assigned as compounds 5 and 6, respectively.
The concentrations of individual anthocyanins in black chokeberry pomace isolated by MAE, UAE, and CE are shown in Table 5.

Table 5.
UPLC ESI-MS2 characterization of individual anthocyanins and total anthocyanin concentration in chokeberry pomace isolated under optimized MAE and UAE conditions compared to CE technique.
In all extracts, the most abundant anthocyanins were cyanidin-3-O-galactoside and cyanidin-3-O-glucoside, accounting for more than 60% of the TA content, followed by cyanidin-3-O-arabinoside and -xyloside. Several reports on the composition of chokeberry anthocyanins have confirmed cyanidin-3-O-galactoside to be the most abundant compound regardless of the type of chokeberry product (fruit, juice, pomace, etc.) [45,53,55]. However, in the same studies, cyanidin-3-O-arabinoside is reported as the second-most prevalent anthocyanin, which is not in agreement with our results showing cyanidin-3-O-glucoside as the second-most abundant compound. The concentration range of cyanidin-3-O-glucoside (124.20–145.60 mg/100 g) was similar to that of cyanidin-3-O-galactoside (125.76–149.06 mg/100 g), while concentrations of cyanidin-3-O-arabinoside and -xyloside were about two or even three-fold lower in the extracts obtained by CE. Vagiri and Jensen (2017) [9] reported a cyanidin-3-O-galactoside concentration in black chokeberry pomace obtained by cold pressing with no enzyme treatment of 114.9 mg/100 g, and the concentration of cyanidin-3-O-arabinoside of 55.8 mg/100 g, which is in line with our results. However, they found a significantly lower amount of cyanidin-3-O-glucoside and cyanidin-3-O-xyloside, 7.9 and 7.3 mg/100 g, respectively. Kapci et al. (2013) [56] reported higher concentrations of cyanidin-3-O-galactoside and -arabinoside (460.05 and 165.11 mg/100 g, respectively), but also low levels of cyanidin-3-O-glucoside and -xyloside (23.77 and 22.34 mg/100 g, respectively), in contrast to our results. These discrepancies in the respective amounts of each anthocyanin and their interrelation can be attributed to the characteristics of the plant itself, especially to the cultivar, stage of maturity at the harvest time, climatic and habitat conditions, and applied agrotechnical conditions during vegetation. It is well known that the concentration, distribution, and composition of plant bioactives change with plant maturity and are additionally influenced by various biotic and abiotic stress factors and plant physiological characteristics [57,58]. Only the authors Vagiri and Jensen (2017) [9] and Oszmiański and Lachowicz (2016) [53] reported the cultivar used in their study, Viking and Galicjanka, respectively, so differences in individual anthocyanin proportions can be attributed primarily to cultivar effect, followed by the effect of external factors.
Although the extraction technique did not influence the qualitative composition and interrelation of the anthocyanins, their concentrations differed within each extract. The significant differences were obtained regarding the concentration of cyanidin-3-O-arabinoside, where both green techniques exerted a higher extraction yield than CE and within cyanidin-3-O-xyloside, where MAE was found to positively affect the concentration. Those advantages in terms of higher individual yield reflected on the concentration of total identified compounds, implicating the excellence of both MAE and UAE in comparison to CE for the isolation of black chokeberry pomace anthocyanins. Previous studies on the employment of green techniques for black chokeberry anthocyanins isolation mostly did not include comparison of the extraction effectiveness to a conventional procedure, with the exception of the study by Jang and Koh (2021) [59] reporting no significant difference between optimized UAE and CE of fresh chokeberry anthocyanins. This is in agreement with our findings when observing the spectrophotometrically determined TA content; however, the insight into individual compounds showed significant differences between techniques. MAE and UAE, as green extraction procedures, not only provided shorter extraction times, 4 and 10 min, respectively, but also enabled higher extraction yields of individual anthocyanins than CE.
4. Conclusions
Two green extraction techniques were optimized and employed for the isolation of black chokeberry pomace anthocyanins, MAE and UAE. In both techniques, acidified 50% ethanol was selected as the extraction solvent, while optimized conditions were set as 60 °C and 4 min irradiation time for MAE and 25% amplitude and 10 min sonication time for UAE. Optimized procedures significantly shortened the extraction time while achieving a higher extraction yield than CE. The composition of individual anthocyanins remained the same, regardless of the extraction method; however, the concentrations of cyanidin-3-O-xyloside and cyanidin-3-O-arabinoside were higher in extracts obtained by green extraction techniques. The results of this study present a valuable insight into the possible exploitation of black chokeberry waste as a rich source of anthocyanins, which can be effectively isolated by environmentally friendly techniques such as MAE and UAE. Established procedures and obtained anthocyanin-rich extracts could be a tool to revalorize the black chokeberry pomace for further application in food and nutraceutical products, while at the same time fully complying with the circular economy principles.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/su15097047/s1, Figures S1 and S2: response surface diagrams of TA content during MAE with two different extraction solvents; Figures S3 and S4: response surface diagrams of TA content during UAE with two different extraction solvents.
Author Contributions
Conceptualization. I.E.G. and M.R.; methodology. I.E.G., M.R. and Z.Z.; formal analysis. I.E.G., M.R. and Z.Z.; investigation. I.E.G. and M.R.; data curation. I.E.G., M.R. and Z.Z.; writing—original draft preparation. I.E.G. and M.R.; writing—review and editing. V.D.-U.; funding acquisition. T.J. and V.D.-U. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by project “Biofracta”, “Strengthening the Economy by Applying Research and Innovation”, European Structural and Investment Funds, Operational Programme Competitiveness and Cohesion 2014–2020 (KK.01.2.1.02).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Authors would like to thank Zoran Herceg for support in providing laboratory equipment used for the experiments through the project “Equipping the Semi-Industrial Practice Laboratory for the Development of New Food Technologies” (KK.01.1.1.02.0001) financed by the European Union.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study, in the collection, analyses, interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
References
- Jakobek, L.; Šeruga, M.; Medvidović-Kosanović, M.; Novak, I.; Strossmayer, J.J. Antioxidant Activity and Poly-phenols of Aronia in Comparison to Other Berry Species. Agric. Conspec. Sci. 2007, 72, 301–306. [Google Scholar]
- Benvenuti, S.; Pellati, F.; Melegari, M.; Bertelli, D. Polyphenols, Anthocyanins, Ascorbic Acid, and Radical Scavenging Activity of Rubus, Ribes, and Aronia. J. Food Sci. 2006, 69, 164–169. [Google Scholar] [CrossRef]
- Huang, R.; Fang, W.; Xie, X.; Liu, Y.; Xu, C. Identification of key astringent compounds in aronia berry juice. Food Chem. 2022, 393, 133431. [Google Scholar] [CrossRef]
- Roda-Serrat, M.C.; Andrade, T.A.; Rindom, J.; Lund, P.B.; Norddahl, B.; Errico, M. Optimization of the Recovery of Anthocyanins from Chokeberry Juice Pomace by Homogenization in Acidified Water. Waste Biomass-Valorization 2021, 12, 1815–1827. [Google Scholar] [CrossRef]
- Ben-Othman, S.; Jõudu, I.; Bhat, R. Bioactives from Agri-Food Wastes: Present Insights and Future Challenges. Molecules 2020, 25, 510. [Google Scholar] [CrossRef]
- Fava, F.; Totaro, G.; Diels, L.; Reis, M.; Duarte, J.; Carioca, O.B.; Poggi-Varaldo, H.M.; Ferreira, B.S. Biowaste biorefinery in Europe: Opportunities and research & development needs. New Biotechnol. 2015, 32, 100–108. [Google Scholar] [CrossRef]
- Farooq, S.; Shah, M.A.; Siddiqui, M.W.; Dar, B.N.; Mir, S.A.; Ali, A. Recent trends in extraction techniques of anthocyanins from plant materials. J. Food Meas. Charact. 2020, 14, 3508–3519. [Google Scholar] [CrossRef]
- Mattioli, R.; Francioso, A.; Mosca, L.; Silva, P. Anthocyanins: A Comprehensive Review of Their Chemical Properties and Health Effects on Cardiovascular and Neurodegenerative Diseases. Molecules 2020, 25, 3809. [Google Scholar] [CrossRef]
- Vagiri, M.; Jensen, M. Influence of juice processing factors on quality of black chokeberry pomace as a future resource for colour extraction. Food Chem. 2017, 217, 409–417. [Google Scholar] [CrossRef]
- Tan, J.; Han, Y.; Han, B.; Qi, X.; Cai, X.; Ge, S.; Xue, H. Extraction and purification of anthocyanins: A review. J. Agric. Food Res. 2022, 8, 100306. [Google Scholar] [CrossRef]
- Bleve, M.; Ciurlia, L.; Erroi, E.; Lionetto, G.; Longo, L.; Rescio, L.; Schettino, T.; Vasapollo, G. An innovative method for the purification of anthocyanins from grape skin extracts by using liquid and sub-critical carbon dioxide. Sep. Purif. Technol. 2008, 64, 192–197. [Google Scholar] [CrossRef]
- Zuleta-Correa, A.; Chinn, M.S.; Alfaro-Córdoba, M.; Truong, V.-D.; Yencho, G.C.; Bruno-Bárcena, J.M. Use of unconventional mixed Acetone-Butanol-Ethanol solvents for anthocyanin extraction from Purple-Fleshed sweetpotatoes. Food Chem. 2020, 314, 125959. [Google Scholar] [CrossRef]
- Muangrat, R.; Williams, P.T.; Saengcharoenrat, P. Subcritical solvent extraction of total anthocyanins from dried purple waxy corn: Influence of process conditions. J. Food Process. Preserv. 2017, 41, e13252. [Google Scholar] [CrossRef]
- Castañeda-Ovando, A.; Pacheco-Hernandez, M.D.L.; Páez-Hernández, M.E.; Rodríguez, J.A.; Galán-Vidal, C.A. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Silva, S.; Costa, E.M.; Calhau, C.; Morais, R.M.; Pintado, M.E. Anthocyanin extraction from plant tissues: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 3072–3083. [Google Scholar] [CrossRef]
- Yang, S.; Mi, L.; Wu, J.; Liao, X.; Xu, Z. Strategy for anthocyanins production: From efficient green extraction to novel microbial biosynthesis. Crit. Rev. Food Sci. Nutr. 2022, 1–16. [Google Scholar] [CrossRef]
- Dobroslavić, E.; Elez Garofulić, I.; Zorić, Z.; Pedisić, S.; Dragović-Uzelac, V. Polyphenolic Characterization and Antioxidant Capacity of Laurus nobilis L. Leaf Extracts Obtained by Green and Conventional Extraction Techniques. Processes 2021, 9, 1840. [Google Scholar] [CrossRef]
- Garofulić, I.E.; Zorić, Z.; Pedisić, S.; Brnčić, M.; Dragović-Uzelac, V. UPLC-MS2 Profiling of Blackthorn Flower Polyphenols Isolated by Ultrasound-Assisted Extraction. J. Food Sci. 2018, 83, 2782–2789. [Google Scholar] [CrossRef]
- Dobrinčić, A.; Repajic, M.; Elez Garofulić, I.; Tuđen, L.; Dragović-Uzelac, V.; Levaj, B. Comparison of Different Extraction Methods for The Recovery of Olive Leaves Polyphenols. Processes 2020, 8, 1008. [Google Scholar] [CrossRef]
- Dent, M.; Dragovic-Uzelac, V.; Garofulic, I.E.; Bosiljkov, T.; Jezek, D.; Brncic, M. Comparison of Conventional and Ultrasound-assisted Extraction Techniques on Mass Fraction of Phenolic Compounds from Sage (Salvia officinalis L.). Chem. Biochem. Eng. Q. 2015, 29, 475–484. [Google Scholar] [CrossRef]
- Garofulić, I.E.; Kruk, V.; Martić, A.; Martić, I.; Zorić, Z.; Pedisić, S.; Dragović, S.; Dragović-Uzelac, V. Evaluation of Polyphenolic Profile and Antioxidant Activity of Pistacia lentiscus L. Leaves and Fruit Extract Obtained by Optimized Microwave-Assisted Extraction. Foods 2020, 9, 1556. [Google Scholar] [CrossRef] [PubMed]
- Garofulić, I.E.; Dragović-Uzelac, V.; Jambrak, A.R.; Jukić, M. The effect of microwave assisted extraction on the isolation of anthocyanins and phenolic acids from sour cherry Marasca (Prunus cerasus var. Marasca). J. Food Eng. 2013, 117, 437–442. [Google Scholar] [CrossRef]
- Ameer, K.; Shahbaz, H.M.; Kwon, J.-H. Green Extraction Methods for Polyphenols from Plant Matrices and Their Byproducts: A Review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 295–315. [Google Scholar] [CrossRef] [PubMed]
- Picot-Allain, C.; Mahomoodally, M.F.; Ak, G.; Zengin, G. Conventional versus green extraction techniques—A comparative perspective. Curr. Opin. Food Sci. 2021, 40, 144–156. [Google Scholar] [CrossRef]
- Chen, F.; Sun, Y.; Zhao, G.; Liao, X.; Hu, X.; Wu, J.; Wang, Z. Optimization of ultrasound-assisted extraction of anthocyanins in red raspberries and identification of anthocyanins in extract using high-performance liquid chromatography–mass spectrometry. Ultrason. Sonochem. 2007, 14, 767–778. [Google Scholar] [CrossRef]
- He, B.; Zhang, L.-L.; Yue, X.-Y.; Liang, J.; Jiang, J.; Gao, X.-L.; Yue, P.-X. Optimization of Ultrasound-Assisted Extraction of phenolic compounds and anthocyanins from blueberry (Vaccinium ashei) wine pomace. Food Chem. 2016, 204, 70–76. [Google Scholar] [CrossRef]
- Pinela, J.; Prieto, M.A.; Pereira, E.; Jabeur, I.; Barreiro, M.F.; Barros, L.; Ferreira, I.C.F.R. Optimization of heat- and ultrasound-assisted extraction of anthocyanins from Hibiscus sabdariffa calyces for natural food colorants. Food Chem. 2019, 275, 309–321. [Google Scholar] [CrossRef]
- Duan, W.; Jin, S.; Zhao, G.; Sun, P. Microwave-assisted extraction of anthocyanin from Chinese bayberry and its effects on anthocyanin stability. Food Sci. Technol. 2015, 35, 524–530. [Google Scholar] [CrossRef]
- Wen, Y.; Chen, H.; Zhou, X.; Deng, Q.; Zhao, Y.; Zhao, C.; Gong, X. Optimization of the microwave-assisted extraction and antioxidant activities of anthocyanins from blackberry using a response surface methodology. RSC Adv. 2015, 5, 19686–19695. [Google Scholar] [CrossRef]
- Pap, N.; Beszédes, S.; Pongrácz, E.; Myllykoski, L.; Gábor, M.; Gyimes, E.; Hodúr, C.; Keiski, R.L. Microwave-Assisted Extraction of Anthocyanins from Black Currant Marc. Food Bioprocess Technol. 2013, 6, 2666–2674. [Google Scholar] [CrossRef]
- Naczk, M.; Shahidi, F. Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar] [CrossRef]
- Elez Garofulić, I.; Malin, V.; Repajić, M.; Zorić, Z.; Pedisić, S.; Sterniša, M.; Smole Možina, S.; Dragović-Uzelac, V. Phenolic Profile, Antioxidant Capacity and Antimicrobial Activity of Nettle Leaves Extracts Obtained by Advanced Extraction Techniques. Molecules 2021, 26, 6153. [Google Scholar] [CrossRef]
- Grunovaitė, L.; Pukalskienė, M.; Pukalskas, A.; Venskutonis, P.R. Fractionation of black chokeberry pomace into functional ingredients using high pressure extraction methods and evaluation of their antioxidant capacity and chemical composition. J. Funct. Foods 2016, 24, 85–96. [Google Scholar] [CrossRef]
- D’alessandro, L.G.; Dimitrov, K.; Vauchel, P.; Nikov, I. Kinetics of ultrasound assisted extraction of anthocyanins from Aronia melanocarpa (black chokeberry) wastes. Chem. Eng. Res. Des. 2014, 92, 1818–1826. [Google Scholar] [CrossRef]
- Sady, S.; Matuszak, L.; Błaszczyk, A. Optimisation of ultrasonic-assisted extraction of bioactive compounds from chokeberry pomace using response surface methodology. Acta Sci. Pol. Technol. Aliment. 2019, 18, 249–256. [Google Scholar] [CrossRef]
- Ramić, M.; Vidović, S.; Zeković, Z.; Vladić, J.; Cvejin, A.; Pavlić, B. Modeling and optimization of ultrasound-assisted extraction of polyphenolic compounds from Aronia melanocarpa by-products from filter-tea factory. Ultrason. Sonochem. 2015, 23, 360–368. [Google Scholar] [CrossRef]
- Andrade, T.A.; Hamerski, F.; Fetzer, D.E.L.; Roda-Serrat, M.C.; Corazza, M.L.; Norddahl, B.; Errico, M. Ultrasound-assisted pressurized liquid extraction of anthocyanins from Aronia melanocarpa pomace. Sep. Purif. Technol. 2021, 276, 119290. [Google Scholar] [CrossRef]
- Simić, V.M.; Rajković, K.M.; Stojičević, S.S.; Veličković, D.T.; Nikolić, N.; Lazić, M.L.; Karabegović, I.T. Optimization of microwave-assisted extraction of total polyphenolic compounds from chokeberries by response surface methodology and artificial neural network. Sep. Purif. Technol. 2016, 160, 89–97. [Google Scholar] [CrossRef]
- Simić, V.M.; Stojičević, S.S.; Veličković, D.T.; Nikolić, N.Č.; Lazić, M.L.; Karabegović, I.T. RSM approach for modeling and optimization of microwave-assisted extraction of chokeberry. Adv. Technol. 2018, 7, 11–19. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E.; Barnes, K.W.; Eisele, T.; Giusti, M.M.; Haché, J.; Hofsommer, H.; Koswig, S.; Krueger, D.A.; et al. Determination of Total Monomeric Anthocyanin Pigment Content of Fruit Juices, Beverages, Natural Colorants, and Wines by the PH Differential Method: Collaborative Study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef]
- Oancea, S.; Stoia, M.; Coman, D. Effects of Extraction Conditions on Bioactive Anthocyanin Content of Vaccinium Corymbosum in the Perspective of Food Applications. Procedia Eng. 2012, 42, 489–495. [Google Scholar] [CrossRef]
- D’alessandro, L.G.; Kriaa, K.; Nikov, I.; Dimitrov, K. Ultrasound assisted extraction of polyphenols from black chokeberry. Sep. Purif. Technol. 2012, 93, 42–47. [Google Scholar] [CrossRef]
- Navas, M.J.; Moreno, A.M.J.; Bueno, J.M.; Sáez-Plaza, P.; Asuero, A.G. Analysis and Antioxidant Capacity of Anthocyanin Pigments. Part IV: Extraction of Anthocyanins. Crit. Rev. Anal. Chem. 2012, 42, 313–342. [Google Scholar] [CrossRef]
- Vázquez-Espinosa, M.; González-De-Peredo, A.V.; Espada-Bellido, E.; Ferreiro-González, M.; Toledo-Domínguez, J.J.; Carrera, C.; Palma, M.; Barbero, G.F. Ultrasound-Assisted Extraction of Two Types of Antioxidant Compounds (TPC and TA) from Black Chokeberry (Aronia melanocarpa L.): Optimization of the Individual and Simultaneous Extraction Methods. Agronomy 2019, 9, 456. [Google Scholar] [CrossRef]
- Kaloudi, T.; Tsimogiannis, D.; Oreopoulou, V. Aronia Melanocarpa: Identification and Exploitation of Its Phenolic Components. Molecules 2022, 27, 4375. [Google Scholar] [CrossRef]
- Jafari, S.M.; Khazaei, K.M.; Assadpour, E. Production of a natural color through microwave-assisted extraction of saffron tepal’s anthocyanins. Food Sci. Nutr. 2019, 7, 1438–1445. [Google Scholar] [CrossRef]
- Xue, H.; Tan, J.; Fan, L.; Li, Q.; Cai, X. Optimization microwave-assisted extraction of anthocyanins from cranberry using response surface methodology coupled with genetic algorithm and kinetics model analysis. J. Food Process. Eng. 2021, 44, e13688. [Google Scholar] [CrossRef]
- Jing, P.; Ruan, S.-Y.; Dong, Y.; Zhang, X.-G.; Yue, J.; Kan, J.-Q.; Slavin, M.; Yu, L. Optimization of purification conditions of radish (Raphanus sativus L.) anthocyanin-rich extracts using chitosan. LWT-Food Sci. Technol. 2011, 44, 2097–2103. [Google Scholar] [CrossRef]
- Liu, W.; Yang, C.; Zhou, C.; Wen, Z.; Dong, X. An improved microwave-assisted extraction of anthocyanins from purple sweet potato in favor of subsequent comprehensive utilization of pomace. Food Bioprod. Process. 2019, 115, 1–9. [Google Scholar] [CrossRef]
- Tiwari, B.K.; O’donnell, C.P.; Patras, A.; Cullen, P. Anthocyanin and Ascorbic Acid Degradation in Sonicated Strawberry Juice. J. Agric. Food Chem. 2008, 56, 10071–10077. [Google Scholar] [CrossRef]
- Mane, S.; Bremner, D.H.; Tziboula-Clarke, A.; Lemos, M.A. Effect of ultrasound on the extraction of total anthocyanins from Purple Majesty potato. Ultrason. Sonochem. 2015, 27, 509–514. [Google Scholar] [CrossRef]
- Ryu, D.; Koh, E. Optimization of Ultrasound-Assisted Extraction of Anthocyanins and Phenolic Compounds from Black Soybeans (Glycine max L.). Food Anal. Methods 2019, 12, 1382–1389. [Google Scholar] [CrossRef]
- Oszmiański, J.; Lachowicz, S. Effect of the Production of Dried Fruits and Juice from Chokeberry (Aronia melanocarpa L.) on the Content and Antioxidative Activity of Bioactive Compounds. Molecules 2016, 21, 1098. [Google Scholar] [CrossRef]
- Lin, S.; Meng, X.; Tan, C.; Tong, Y.; Wan, M.; Wang, M.; Zhao, Y.; Deng, H.; Kong, Y.; Ma, Y. Composition and antioxidant activity of anthocyanins from Aronia melanocarpa extracted using an ultrasonic-microwave-assisted natural deep eutectic solvent extraction method. Ultrason. Sonochem. 2022, 89, 106102. [Google Scholar] [CrossRef]
- Szopa, A.; Kokotkiewicz, A.; Kubica, P.; Banaszczak, P.; Wojtanowska-Krośniak, A.; Krosniak, M.; Marzec-Wróblewska, U.; Badura, A.; Zagrodzki, P.; Bucinski, A.; et al. Comparative analysis of different groups of phenolic compounds in fruit and leaf extracts of Aronia sp.: A. melanocarpa, A. arbutifolia, and A.× prunifolia and their antioxidant activities. Eur. Food Res. Technol. 2017, 243, 1645–1657. [Google Scholar] [CrossRef]
- Kapci, B.; Neradová, E.; Čížková, H.; Voldřich, M.; Rajchl, A.; Capanoglu, E. Investigating the antioxidant potential of chokeberry (Aronia melanocarpa) products. J. Food Nutr. Res. 2013, 52, 219–229. [Google Scholar]
- De Pascual-Teresa, S.; Sanchez-Ballesta, M.T. Anthocyanins: From plant to health. Phytochem. Rev. 2008, 7, 281–299. [Google Scholar] [CrossRef]
- Naing, A.H.; Kil Kim, C. Abiotic stress-induced anthocyanins in plants: Their role in tolerance to abiotic stresses. Physiol. Plant. 2021, 172, 1711–1723. [Google Scholar] [CrossRef]
- Jang, Y.; Koh, E. Sustainable Water Extraction of Anthocyanins in Aronia (Aronia melanocarpa L.) Using Conventional and Ultrasonic-Assisted Method. Korean J. Food Sci. Technol. 2021, 53, 527–534. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).