Abstract
Post-transcriptional regulation of gene expression plays a key role in cellular proliferation, differentiation, migration, and apoptosis. Increasing evidence suggests dysregulated post-transcriptional gene expression as an important mechanism in the pathogenesis of cancer. The tristetraprolin family of RNA-binding proteins (RBPs), which include Zinc Finger Protein 36 (ZFP36; commonly referred to as tristetraprolin (TTP)), Zinc Finger Protein 36 like 1 (ZFP36L1), and Zinc Finger Protein 36 like 2 (ZFP36L2), play key roles in the post-transcriptional regulation of gene expression. Mechanistically, these proteins function by binding to the AU-rich elements within the 3′-untranslated regions of their target mRNAs and, in turn, increasing mRNA turnover. The TTP family RBPs are emerging as key regulators of multiple biological processes relevant to cancer and are aberrantly expressed in numerous human cancers. The TTP family RBPs have tumor-suppressive properties and are also associated with cancer prognosis, metastasis, and resistance to chemotherapy. Herein, we summarize the various hallmark molecular traits of cancers that are reported to be regulated by the TTP family RBPs. We emphasize the role of the TTP family RBPs in the regulation of trait-associated mRNA targets in relevant cancer types/cell lines. Finally, we highlight the potential of the TTP family RBPs as prognostic indicators and discuss the possibility of targeting these TTP family RBPs for therapeutic benefits.
1. Introduction
In healthy cells, expression of mRNAs for genes associated with cell survival pathways is maintained at normal levels through tight transcriptional and post-transcriptional mechanisms. In contrast, tumor cells possess abnormally stable mRNAs for various categories of pro-survival genes, including protooncogenes, tumor suppressors, and cytokines. A large number of these tumor-associated pro-survival mRNAs possess AU-rich elements (AREs) in their 3′-untranslated regions (3′UTRs). Specific ARE-binding proteins, such as the tristetraprolin family of RNA-binding proteins, are known to post-transcriptionally regulate the expression of these mRNAs.
The tristetraprolin family of RNA-binding proteins (TTP family RBPs) are characterized by the presence of one or more CCCH zinc finger domain(s) that contain three cysteine (C) and one histidine (H) residues. There are three human members in this family, including Zinc Finger Protein 36 (ZFP36) or TTP itself, encoded by the ZFP36 gene; Zinc Finger Protein 36 Like 1 (ZFP36L1), encoded by the ZFP36L1 gene; and Zinc Finger Protein 36 Like 2 (ZFP36L2), encoded by the ZFP36L2 gene [1] (Table 1). A fourth member, Zinc Finger Protein 36 Like 3 (ZFP36L3), is restricted to rodents. Orthologues of the TTP family RBPs have been found in many vertebrates with the only exception in birds [2].

Table 1.
Human tristetraprolin (TTP) family RNA-binding proteins.
Through a highly conserved zinc finger domain, the TTP family RBPs bind to AREs at the 3′UTRs of their target mRNAs in a sequence- and structure-specific manner and catalyze the removal of the poly (A) tail, thus resulting in their mRNA decay. The consensus sequence of AREs in the 3′UTRs of the target mRNAs is UUAUUUAUU, although some variations of this sequence still mediate high affinity binding [3]. All the mammalian TTP family members appear to act similarly in biochemical studies involving RNA binding and decay. Interestingly, germline gene knockouts of the three TTP family RBPs in mice resulted in vastly different phenotypes [4,5,6,7]. For instance, while germline deletion of TTP resulted in a systemic inflammatory syndrome [4], germline deletion of ZFP36L1 was embryonically lethal [5], and germline deletion of ZFP36L2 resulted in post-natal mortality within two weeks post-birth due to defects in hematopoiesis [6]. These experiments clearly demonstrated that the TTP family RBPs may have differential target and cell/tissue-type specificity under varying physiological and pathological conditions. Furthermore, the TTP family RBPs may be expressed at different times during pre- and post-natal life. Some studies, including our unpublished observations, have also reported on the redundant functions of the TTP family RBPs [8].
Although the TTP family proteins were discovered more than 20 years ago, most of the studies investigating their role in carcinogenesis have been reported within the last decade. The TTP family RBPs have tumor-suppressor properties, which are directly related to their ability to post-transcriptionally regulate oncogenic mRNAs. For instance, oncogenes, including NOTCH1, MYC, BCL-2, and COX-2, contain 3′UTR AREs, and have been identified as direct TTP family RBP targets [8,9,10,11]. Conversely, TTP expression is also directly suppressed by certain oncoproteins [9]. TTP has also been shown to complement the function of tumor suppressors, such as p53, through downregulation of the oncogenes [12]. In fact, TTP expression is induced by p53 in cancer cells [12]. In rare instances, the TTP family RBPs are also known to directly target tumor suppressors. For instance, TTP has been shown to target the mRNA encoding the tumor suppressor LATS2 [13]. In sum, while increasing evidence suggests a protective role of the TTP proteins in tumorigenesis, some mechanisms seem to exist that counter the beneficial aspects of the TTP family RBPs.
Alterations in the expression/activity of the TTP family RBPs have been reported to be associated with multiple cancers [14] (Table 2). Numerous studies have specifically reported a loss of TTP family RBP expression in a variety of cancers [15,16,17,18]. Loss of expression/activity of the TTP family RBPs is expected to result in increased stability of their target mRNAs. Three different mechanisms for loss of expression/activity of the TTP family RBPs have been reported: (1) MicroRNA-mediated regulation; (2) epigenetic silencing via DNA methylation; and (3) modulation of protein activity through post-translational modifications, particularly phosphorylation. Regardless of the mechanisms involved, owing to the regulation of a broad range of target mRNAs concurrently, the TTP family RBPs loss can result in significant changes in gene expression and can have dramatic consequences for the development and progression of cancer.

Table 2.
TTP family of RNA-binding proteins in various cancers.
In this review, we will discuss the key molecular traits of cancer that the TTP family RBPs regulate; the molecular mechanisms of the regulation; and the TTP family RBP mRNA targets that have been identified in various cancer cells and tissues. Specifically, the molecular traits of cancers, including uncontrolled cellular proliferation in the absence of external growth signals, resistance to apoptosis, sustained angiogenesis, as well as tissue invasion and metastasis, will be discussed. We will also discuss the potential of the TTP family RBPs as prognostic biomarkers and the possibility of targeting the TTP family RBPs for therapeutic purposes. The outstanding questions that remain will also be highlighted.
2. TTP Family Proteins and Cell Cycle Control
Dysregulation of the cell cycle is a characteristic feature of all cancers. The cell cycle involves four distinct phases, i.e., G1 (Gap 1 or first growth phase), S (DNA replication phase), G2 (Gap 2 or second growth phase), and M (mitosis phase). Regulatory mechanisms are in place to ensure that cells in the G1 phase that acquire DNA damage are prohibited from entering the S phase, and that errors during DNA replication in the S phase are repaired in the G2 phase before the cells enter the M phase. Several oncogenic processes function by dysregulating the normal controls and checkpoints and enforcing the cells into cell cycle progression in a mitogen-independent manner.
TTP’s role in regulating the cell cycle is linked to its ability to bind and destabilize critical cell cycle regulators. For instance, critical cell cycle regulators, namely c-Myc and cyclin D1, possess 3′UTR AU-rich elements and have been shown to be regulated by TTP [73]. C-Myc is a member of the Myc oncogene family of transcription factors that regulate cellular proliferation, differentiation, metabolism, and apoptosis, and are frequently dysregulated in human cancers [74]. Cyclin D1 is a proto-oncogene that regulates G1-S phase progression and is frequently overexpressed in cancer. Another example of a cell cycle checkpoint protein regulated by TTP is E2F transcription factor 1 (E2F1). E2F1 regulates G1-S phase progression and is frequently overexpressed in many types of human cancers. Aberrant expression of E2F1 is associated with high-grade tumors, metastases, and unfavorable patient prognosis. TTP was shown to post-transcriptionally regulate E2F1, suggesting that TTP controls cellular proliferation through the regulation of E2F1 mRNA stability [75]. Along similar lines, Xu et al. reported that TTP inhibits cellular proliferation in breast tumor cells in vitro and breast tumor growth in vivo by inducing cell cycle arrest at the S phase. TTP was found to inhibit c-Jun expression through blocking the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) p65 nuclear translocation, which resulted in increased expression of Wee1, a regulatory molecule that controls cell cycle transition from the S to the G2 phase [26].
Interestingly, resveratrol (3,5,4′-trihydroxystilbene), a naturally occurring polyphenol compound found in natural sources, including grape skin and red wine, was shown to activate TTP, resulting in downregulation of E2F1, inhibitor of apoptosis 2 (cIAP2), large tumor suppressor kinase 2 (LATS2), and lin-28 homolog A (Lin28)—all downstream targets of TTP—thus suppressing the proliferation and invasion/metastasis of colon cancer cells [39]. Hitti et al. performed a systematic analysis of ARE-mRNA expression across multiple cancer types, including invasive breast cancer, and showed that ARE-mRNAs were overrepresented and correlate with TTP expression. A cluster of 11 overexpressed ARE-mRNAs that are involved in the mitotic (M phase) cell cycle phase was found to negatively correlate with TTP expression. These ARE-mRNAs also physically interacted with TTP, indicating direct regulation. Furthermore, breast cancer patients with a high mean expression of this cluster showed poor survival [27]. These studies suggested an anti-mitotic role of TTP. TTP was also shown to directly bind NEDD9, a protein that has a potential role in prostate cancer cell growth regulation [63].
Chen et al. showed that cyclin-dependent kinase 6 (CDK6) is post-transcriptionally regulated by ZFP36L1. These authors demonstrated that ZFP36L1 functions as a positive regulator of monocyte/macrophage differentiation by regulating CDK6. Accordingly, levels of ZFP36L1 were found to be significantly reduced in acute myeloid leukemia patients [52]. ZFP36L1 has also been suggested to be a post-transcriptional regulator of the cell cycle signaling genes, including E2F1 and CCND1 (cyclin D1). In a recent study, ZFP36L1 was particularly shown to regulate hypoxia signaling through direct binding and degradation of HIF1A. This study indicated a tumor-suppressor role for ZFP36L1 through regulation of hypoxia, cell cycle, and angiogenesis [15]. Furthermore, these authors found that ZFP36L1 is epigenetically silenced through hypermethylation of the second exon and is downregulated in several patient cohorts of bladder and breast cancers. Functionally, silencing ZFP36L1 enhanced tumor cell growth while overexpression of ZFP36L1 suppressed cell proliferation and migration in bladder and breast cancer cell lines [15]. Finally, both ZFP36L1 and ZFP36L2 were shown to inhibit cellular proliferation through downregulation of cyclin D expression, resulting in cell cycle arrest at the G1 phase [76]. All these studies indicate that TTP, ZFP36L1, and ZFP36L2 are critical regulators of the cell cycle.
3. TTP Family Proteins and Control of Apoptosis
One of the hallmark characteristics of cancer is to evade apoptosis or resist cell death [77]. Apoptosis occurs through two distinct pathways: the intrinsic (mitochondrial) pathway and the extrinsic (death receptor) pathway. While the two pathways are distinct, both involve activating caspases in the final steps.
The TTP family RBPs modulate tumor cell apoptosis by directly regulating the apoptotic mediators within both pathways. Johnson et al. demonstrated that TTP results in apoptotic cell death in various cell types, likely through direct regulation of the TTP targets [78]. This was one of the earliest studies that indicated a role of TTP proteins in cell survival and apoptosis. The authors suggested that TTP was unique and somewhat different from the two other family members, ZFP36L1 and ZFP36L2, because TTP, but not ZFP36L1 and ZFP36L2, could also sensitize the cells to apoptosis by inducing TNFα. However, it remained unclear whether TTP was inactive upon ectopic expression in these studies. Hydroquinone, an aromatic organic compound, induces apoptosis in human leukemia U937, human leukemia HL-60, and Jurkat cells through a TTP-dependent mechanism. Mechanistically, this study showed that TTP phosphorylation and inactivation through the p38 MAPK pathway resulted in increased TNFα-induced apoptosis [50]. Along similar lines, albendazole, a microtube-targeting anthelmintic, was demonstrated to induce apoptosis in human leukemia cells through the p38–TTP–TNFα axis [51].
Conversely, resveratrol was able to induce TTP expression in human glioma cells that resulted in apoptosis and suppression of cell growth through destabilization of the urokinase plasminogen activator (uPA) and urokinase plasminogen activator receptor (uPAR). Both, uPA and uPAR are overexpressed in glioblastomas and play a role in invasion [47].
PIM1, an oncogenic serine-threonine kinase, functions by repressing apoptosis and is a direct target of TTP [61,79]. In fact, ectopic TTP expression impaired the viability and invasiveness of glioblastoma multiforme cancer cells by destabilizing the PIM1, PIM2, and X-linked inhibitor of apoptosis proteins (XIAP) in these cells [45]. Park et al. recently reported that TTP enhances cisplatin sensitivity in head and neck squamous cell carcinoma (SCCHN) cells by reducing the levels of BCL-2, an anti-apoptotic protein, which is overexpressed in cancer and confers resistance to cisplatin [70]. While, earlier, Lee et al. had showed that ZFP36L1 enhanced cisplatin sensitivity in SCCHN cells by inhibiting the human inhibitor of apoptosis protein-2 (cIAP2) and resulting in increased caspase-3 activity [71]. ZFP36L1 has also been shown to mediate its pro-apoptotic effects on malignant B-cells by regulating BCL-2 [10]. The BCL-2 family of proteins control the permeabilization of the mitochondrial outer membrane, thus regulating commitment to apoptosis. The role of ZFP36L2 in modulating apoptosis remains unexplored. Together, all these studies indicate an important role of the TTP family RBPs in regulating apoptosis.
4. TTP Family Proteins and Regulation of Pro-Tumorigenic Inflammatory Mediators
TTP is a known regulator of inflammation. This critical function of TTP first became evident when germline TTP knockout mice were generated [4]. TTP knockout mice appeared normal at birth; however, within 2–3 weeks, they developed a systemic inflammatory syndrome characterized by cachexia, arthritis, dermatitis, conjunctivitis, myeloid hyperplasia, and autoimmunity [4]. This phenotype was largely attributed to overexpression of TNFα, a potent pro-inflammatory cytokine, as evidenced by the prevention of the development of the syndrome by anti-TNFα antibody injections [4]. Further, it was demonstrated that TTP binding to AREs within the 3′UTR of TNFα mRNA results in an increased turnover, an effect that was abrogated in TTP deficiency [80]. Subsequent studies showed that a number of other pro-inflammatory cytokines and chemokines, including IL-23, IL-17, IL-1β, CXCL1, and CXCL2, are also directly regulated by TTP [81,82]. These and other mRNA targets of the TTP family RBPs are reviewed elsewhere [83]. Furthermore, germline overexpression of TTP using its endogenous promoter resulted in protection against immune-mediated inflammatory diseases, including arthritis, psoriasis, and autoimmune encephalomyelitis [84]. These critical studies indicate that TTP directly regulates inflammation and that loss of TTP expression/activity results in enhanced inflammation.
Inflammation is also a critical component of the tumor progression and tumor microenvironment, and many tumors are known to arise at the site of chronic inflammation. TTP is a well-established post-transcriptional regulator of pro-inflammatory cytokines and chemokines and, due to this function, TTP is an important modulator of tumor development and progression. Twizere et al. showed that TTP physically interacts with viral protein Tax, thus reverting the inhibition of pro-inflammatory cytokine TNFα [48]. The authors indicated that this may be of importance in cell transformation caused by leukemogenic viruses. Stoecklin et al. demonstrated the tumor-suppressor role of TTP in a v-H-ras-dependent mast cell tumor model through regulation of interleukin-3 (IL-3) [56]. Tumor cells in this model exhibit abnormally stable expression of IL-3 mRNA as part of an oncogenic autocrine loop. TTP delayed tumor progression by four weeks by enhancing IL-3 mRNA degradation in this model. Interestingly, TTP expression was lost in tumors that managed to appear at or after four weeks. Additionally, TTP reduced the cloning efficiency in vitro when transfected into a fully established tumor cell line and the growth of the inoculated cells in vivo. These studies indicate a critical role of mRNA stabilization in oncogenesis and suggest that tumor suppression is achievable by interfering with mRNA turnover.
Sawaoka et al. showed an interesting mechanism of regulation of cyclooxygenase-2 (COX-2) by TTP in colon adenocarcinoma cells. These cells express two distinct transcript variants of COX-2: a full length, 4465nt mRNA; and a truncated 2577nt polyadenylation variant, in which the terminal 1888 nt 3′untranslated region is absent. During cellular growth, the levels of the full-length transcript reduced, whereas the levels of the truncated variant increased. Most importantly, TTP levels were inversely correlated with the levels of the full-length transcript, and TTP transfection resulted in a reduction in the levels of the full-length transcript, indicating TTP regulation of COX-2 in these cells [11]. COX-2 is a product of an immediate early gene that is induced by growth factors and cytokines and plays a role in cellular proliferation [85,86,87]. COX-2 expression is increased in a number of cancers, including human colorectal [88], esophageal [89], pancreatic [90], lung [91], prostate [92], and mammary [93].
Similarly, TTP was shown to destabilize interleukin 8 (IL-8) and vascular endothelial growth factor (VEGF) mRNAs in malignant glioma cells, resulting in a dose-dependent decrease in cellular proliferation, loss of cell viability, and apoptosis. TTP was, in fact, ubiquitously expressed in primary gliomas and benign astrogliotic tissues; however, hyperphosphorylated/inactive TTP was present in malignant glioma tumors. It is generally accepted that the TTP activity is repressed upon phosphorylation [46]. Al-Souhibani et al. showed that TTP expression is significantly lower in invasive breast cancer cells compared to normal breast cells, and that the genes involved in cellular growth, invasion, and metastasis, namely matrix metalloproteinase 1 (MMP1), urokinase-type plasminogen activator (uPA), and urokinase plasminogen activator receptor (uPAR), were directly regulated by TTP in breast cancer cells [21]. Along the same lines, TTP was found to be weakly expressed in melanoma cells. These cells express high levels of the C-X-C motif chemokine ligand 8 (CXCL8), which plays a role in cellular growth and angiogenesis. These authors further demonstrated that extracellular signal-regulated kinase (ERK) inhibition restored TTP, which destabilized and inhibited CXCL8, suppressed cellular proliferation, and induced apoptosis [57]. TTP was also shown to directly regulate hypoxia-inducible factor 1 (HIF-1), a factor critically required for survival in hypoxic conditions, indicating that a low TTP poses a significant advantage to cancer cells by increasing HIF-1 and allowing adaptation to hypoxia [94]. Interestingly, latent membrane protein 1, a viral oncoprotein, was found to significantly enhance HIF-1A expression in nasopharyngeal carcinoma cells by inhibiting TTP [95].
In yet another study, TTP was shown to post-transcriptionally regulate interleukin 23 (IL-23) in mouse colon cancer cells [35]. IL-23 is highly expressed in many tumors and its levels correlate with tumor progression. Squamous cell carcinoma of the head and neck (SCCHN) patients with low interleukin 6 (IL-6) and high MMP9, or with high IL-6 and low MMP9, were found to have the poorest outcomes followed by patients with both high IL-6 and high MMP9. In comparison, patients with low IL-6 and low MMP9 had the best outcomes with respect to tumor recurrence, surgery, or death. Functionally, TTP suppression enhanced cellular invasiveness in vitro in an oral-cancer-equivalent 3D model and in vivo in chick chorioallantoic membrane models, resulting from increased secretion of IL-6, MMP2, and MMP9 [68].
TTP was found to be remarkably reduced in gastric cancer and inversely correlated with interleukin 33 (IL-33) expression. Furthermore, low TTP expression contributed to gastric cancer progression and was associated with depth of invasion, lymph node metastasis, advanced TNM stage, and poor survival. Conversely, elevated TTP expression was shown to inhibit the proliferation, migration, and invasion of gastric cancer cells through suppression of IL-33, a tumor promoting cytokine [43]. Similar results were found in human glioma tissues and cells where TTP was significantly downregulated and associated with reduced survival. In this particular study, TTP inversely correlated with IL-13 levels in glioma tissues and TTP inhibited the growth, migration, and invasion of glioma cells through downregulation of IL-13 and attenuation of the PI3K/Akt/mTOR pathway [96].
TTP knockout mice has increased numbers of cytotoxic T-cells due to direct regulation of interleukin 27 (IL-27), a CD8 + T-cell regulatory cytokine. Interestingly, in a mouse mammary gland tumor model, TTP knockout mice showed retracted tumor growth due to increased tumor-infiltrating CD8+ T cells [29]. Kratochvill et al. showed that TTP is constitutively highly expressed in tumor-associated macrophages. However, the effects of TTP on mRNA stability were blocked by the constitutively active p38 in the tumor microenvironment, which drove the production of inflammatory cytokines [97]. A very elegant study by Coelho et al. showed innately immunoresistant RAS mutant tumors are characterized by the upregulation of immunosuppressive protein programmed death-ligand 1 (PD-L1) through RAS–MEK–MK2-induced TTP phosphorylation/inactivation, resulting in increased PD-L1 mRNA stability. In humans, RAS activation was associated with PD-L1 upregulation in human lung and colon adenocarcinoma [37]. TTP has also been identified as one of the eight genes functionally related to the NF-κB pathway that were highly downregulated in lethal prostate cancer [63]. Gambogic acid, a polyprenylated xanthone, was demonstrated to significantly inhibit cancer stem cells in colorectal carcinoma, both in vitro and in vivo, by inhibiting EGFR–ERK signaling, resulting in upregulation of TTP [38]. TTP has also been demonstrated to be a post-transcriptional regulator of aryl hydrocarbon receptor repressor (AHRR) in breast cancer cells [23].
Similar to TTP, ZFP36L1 phosphorylation and inactivation by the p38–MK2 axis has been shown to stabilize Nanog and Klf4 in triple-negative breast cancer cells, resulting in breast cancer stem cell phenotype, a feature of chemotherapy-resistance in triple-negative breast cancer [31].
5. TTP Family Proteins and Cellular Senescence
Cellular senescence is characterized by cells undergoing growth arrest in response to a wide variety of extrinsic and intrinsic insults, including DNA damage, loss of telomeres, and oncogenic activation. Cells undergoing senescence secrete a collective set of proteins that includes cytokines, chemokines, and growth factors, among others. While under basal conditions, cellular senescence may be beneficial in maintaining tissue homeostasis, cellular senescence is potentially detrimental in aging. Cellular senescence has dynamic roles in cancer: beneficial in tumor cells by improving the therapeutic outcomes, and detrimental in non-tumor cells by causing relapse and secondary tumors. Selected studies have shown a role for the TTP family proteins in regulating senescence. For instance, human papilloma virus-18 (HPV-18)-positive HeLa cells were used to show that TTP promoted cellular senescence through rapid decay of E6-associated protein (E6-AP) mRNA, resulting in p53 stabilization and inhibition of human telomerase reverse transcription gene (hTERT) transcription. E6 is a viral protein that HPV uses for cellular transformation. Association of E6 with E6-AP facilitates cell transformation by p53 degradation and activation of hTERT. This study linked the TTP-mediated post-transcriptional regulation to HPV-associated cervical carcinogenesis [34]. In fact, TTP was found to be consistently absent in cervical carcinomas compared to normal human cervixes. These studies also suggested the tumor suppressive role of TTP in cervical cancer.
In a recent study, ZFP36L1 was demonstrated as a key regulator of cellular senescence by directly regulating components of the senescence-associated secretory protein (SASP) through post-transcriptional regulation. In this study, ZFP36L1 was found to signal downstream of mTOR in regulating SASP mRNAs and phosphorylation inhibited ZFP36L1 activity [98]. The role of ZFP36L2 in cellular senescence remains undetermined.
6. TTP Family Proteins and Regulation of Angiogenesis
One of the hallmarks of cancer is angiogenesis, the formation of new blood vessels, which are required for supplying nutrition and overcome a hypoxic microenvironment in rapidly growing tumor masses. VEGF is an angiogenic cytokine that plays a key role in tumor angiogenesis. VEGF and IL-6 are markedly increased in squamous cell carcinoma of the head and neck (SCCHN) and are associated with poor survival. GALR2, a pro-survival G-protein coupled receptor promoted angiogenesis via p38-mediated phosphorylation/inactivation of TTP, resulting in increased VEGF and IL-6 levels both in vitro in SCCHN cancer cells and in vivo in murine tumor xenografts and chorioallantoic membrane models [69].
Importantly, ZFP36L1 has been specifically shown to post-transcriptionally regulate VEGF [99]. Interestingly, a single intratumoral injection of a ZFP36L1 fusion protein was shown to be effective at decreasing VEGF, acidic FGF, TNFα, IL-1α, and IL-6, as well as at reducing tumor growth [100]. Both TTP and ZFP36L1 have been demonstrated to regulate HIF1α, a member of the family of transcription factors that are the primary effectors of the adaptive response of tumor cells to hypoxia [15,101]. The role of ZFP36L2 in regulating modulators of angiogenesis has not been explored yet.
7. TTP Family Proteins and Epithelial Mesenchymal Transition
Epithelial–mesenchymal transition (EMT) is a reversible process whereby epithelial cells transition to the mesenchymal phenotype by repressing epithelial-specific traits, i.e., intercellular adhesion and proliferation, and acquisition of mesenchymal traits, i.e., migration and invasion. EMT is a crucial step in metastasis and drug resistance, and epithelial tumors are well known to undergo EMT. TTP has been shown to directly regulate EMT regulators, including ZEB1 (zinc finger E-box binding homeobox 1), SOX9 (sex-determining region Y box 9), and MACC1 (metastasis associated in colon cancer 1), all of which are known to be downregulated in colorectal carcinomas (CRC). Re-expressing TTP reverted the EMT phenotype in this study [36]. Two other EMT regulators, TWIST1 (twist-related protein 1) and SNAIL1 (zinc finger protein snail 1), are also known targets of TTP [36]. Interestingly, TTP was identified as a target of a microRNA, miR-29a, and miR-29a-mediated downregulation of TTP was associated with EMT and metastasis in breast cancer [25].
Rataj et al. demonstrated that ZFP36L1 was markedly suppressed in breast cancer cells and patient tissues and that a derivative of ZFP36L1 fused to cell-penetrating peptide inhibited the proliferation, migration, invasion, and anchorage-independent growth in vitro and impaired the tumor growth and EMT markers, including Snail, Vimentin, and N-cadherin, in vivo [32]. Interestingly, ZFP36L1 was identified as a key regulator of neural progenitor cell-fate transition from oligodendrocyte to astrocytes and through this process it is a key regulator of processes such as myelination and gliomagenesis [49]. This study showed that while the loss of ZFP36L1 in the neural lineage resulted in myelination deficits due to the oligodendrocyte–astrocyte switch, in tumorigenesis this process was in fact beneficial by preventing gliomagenesis, thus enhancing survival. The role of ZFP36L2 in EMT has not been explored yet.
8. TTP Family Proteins and Tumor Suppressor and Oncogenic Roles
Tumorigenesis is a consequence of mutations in oncogenes and tumor-suppressor genes that frequently result in either an overexpression of oncogenes or loss of tumor suppressors. A key study in 2012 discovered TTPs important role as a tumor suppressor. The MYC oncoprotein was found to directly suppress TTP transcription, and TTP repression appeared to be a hallmark of malignancies with MYC involvement. Furthermore, enforced expression of TTP impaired the development of lymphoma and abolished the maintenance of the malignant state. ZFP36L1 was also repressed by MYC; however, it was not suggested to be a tumor suppressor in this model [9].
TTP has also been shown to function as a tumor suppressor through downregulation of estrogen receptor alpha (ER-α) transactivation, resulting in reduced cellular proliferation and reduced potential of the cells to form tumors in a mouse model. In this study, TTP was shown to be associated with ER-α and was recruited to the promoter region, indicating that TTP may be a bona fide nuclear receptor corepressor [24]. TTP has also been shown to be downregulated in hepatocellular carcinoma (HCC) cells and tumors through an epigenetic mechanism that involves hypermethylation of a single CpG site within the TGFβ1 responsive region of the TTP promoter. The epigenetic inactivation of TTP resulted in an increased half-life of c-Myc, causing cancer cells to undergo selective resistance to TGFβ1 antiproliferative signaling [53]. TTP is significantly downregulated in liver tumors. During tumor progression, TTP functions as a tumor suppressor and inhibits proliferation and migration, reduces expression of several oncogenes, and increases chemo sensitivity [54]. The anti-proliferative properties of metformin, an anti-diabetic drug, in breast cancer cells were mediated by induction of TTP through c-Myc downregulation [28].
Interestingly, ZFP36L1 was found to be downregulated due to enhancer hypermethylation within the second exon in myelofibrosis, which conversely led to an increased expression of its target mRNAs. Functionally, ZFP36L1 expression induced apoptosis in leukemia cells, indicating a tumor-suppressor role of ZFP36L1 in myelofibrosis [59]. Similar to TTP and ZFP36L1, ZFP36L2 also functions as a tumor suppressor. Hypermethylation of a super-enhancer site in ZFP36L2 resulted in epigenetic silencing in a large data set of esophageal squamous cell carcinoma (SCC) whole-exome sequenced tissues. This phenomenon was also found in other SCCs analyzed from the cancer genome atlas (TCGA) and resulted in reduced mRNA expression in all SCCs [41].
The strongest evidence for the role of ZFP36L1 and ZFP36L2 as tumor suppressors came from studies done by Hodson et al. [8]. These authors demonstrated that loss of both ZFP36L1 and ZFP36L2 in mouse thymocytes resulted in the development of T cell acute lymphoblastic leukemia (T-ALL) due to stabilization of an oncogenic transcriptional regulator, Notch 1 [8]. Interestingly, both ZFP36L1 and ZFP36L2 were found to function in a redundant manner in this study. Recently, genomic mutation in ZFP36L1 was identified as a potential driver of tumorigenesis in patients with concomitant diffuse large B-cell lymphoma and hepatitis B virus (HBV) infection [55]. One study in particular suggested an oncogenic role for ZFP36L2. The authors showed that tandem duplication induced amplification of the super enhancers and were associated with an increase in ZFP36L2 expression in ~10% of gastric cancers. Functionally, ZFP36L2 promoted the growth of gastric cancer cells in this study [44]. Together, these studies indicate that all three members of the TTP family RBPs function as tumor suppressors in various types of cancers.
9. TTP Family Proteins and Regulation of Tumor Metastasis
Tumor metastasis is defined by cancer cells acquiring features of motility, invasion, plasticity, and ability to colonize secondary organs/tissues, and is the primary cause of cancer morbidity and mortality. Interestingly, microRNA-29a (miR-29a) was found to promote tumor progression and invasion by downregulating TTP both in vitro and in vivo in pancreatic cancer. miR-29a was upregulated and TTP was downregulated in pancreatic cancer cells and tissues [60]. In another study, two main clusters of breast cancers that differed on their lymph node status were identified from the breast cancer serial analysis of gene expression. Interestingly, ZFP36L1 was upregulated only in lymph node positive primary breast cancer, indicating that patterns of gene expression in primary tumors at the time of surgical removal could discriminate those that have lymph node metastasis [30]. Finally, ZFP36L2 was identified as an NME1, a metastatic suppressor, regulated gene in a screen of two metastatic cancer cells: melanoma and follicular thyroid carcinoma [42].
10. TTP Family Proteins as Potential Biomarkers
A very elegant study in 2010 showed that TTP is widely suppressed in a number of human cancers, including those of the thyroid, lung, ovary, uterus, and breast, as well as in a number of cancer cell lines, including those of lung and cervical cancer. Here, suppressed TTP was a negative prognostic indicator in breast cancer where more advanced tumors exhibited the weakest TTP expression [16]. Moreover, restoring TTP expression in cancer cells resulted in suppression of the tumorigenic phenotypes while reducing the TTP levels promoted the neoplastic phenotype [16]. Another similar study showed that among breast cancer types, higher grade tumors showed the weakest TTP expression at the protein level compared to low grade tumors, suggesting that the TTP protein levels correlate with prognosis [17].
TTP has also been suggested as a promising biomarker for prostate cancer risk assessment. TTP expression was markedly reduced in metastatic prostate cancer compared to primary tumors [64]. Men with low TTP-expressing primary prostate cancer had significantly increased chances of biochemical reoccurrence in this study. Induction of TTP inhibited the growth, proliferation, and tumorigenic potential of prostate cancer cells in a mouse xenograft model of prostate cancer [64]. Another study that also investigated prostate cancers, showed that low-TTP tumors had faster reoccurrence or metastasis versus high-TTP tumors [66]. Additionally, the low time to reoccurrence in low-TTP tumors was more pronounced in low-grade tumors. This study suggested that TTP is a promising prostate cancer biomarker for predicting the low-grade radical prostatectomy prostate cancer patients that will have poor outcomes [66]. Another study similarly showed low TTP expression in prostate cancer compared to non-cancerous tissues [65].
Low TTP expression in tumors versus adjacent normal tissues was also shown in pancreatic cancer [61]. Here, TTP expression was almost negative in poorly differentiated cancer, weakly positive in moderately differentiated, and highly positive in well differentiated pancreatic cancers. Low TTP expression was associated with age, tumor size, tumor differentiation, postoperative T stage, postoperative N stage, and TNM stage. Low TTP expression correlated with low patient survival rates and poor prognosis, suggesting that TTP could act as a prognostic indicator in pancreatic cancer [61]. Components of the AP-1 transcription factor, including JUN, JUNB, FOS, FOSB, were enriched in association with TTP as a conserved co-regulated group of genes and were significantly downregulated in breast, liver, lung, kidney, and thyroid carcinomas. Patients with low expression of these genes displayed poor prognosis [18].
Furthermore, TCGA datasets for breast cancer, lung adenocarcinoma, lung squamous cell carcinoma, and colon adenocarcinoma revealed a shared signature of 50 genes that were differentially expressed between the low- and high-TTP-expressing tumors [19]. The TTP-low gene signature was also a feature of several other cancers, including pancreatic, bladder, and prostate from non-TCGA datasets. Low TTP expression was a poor prognostic indicator in breast cancer and lung adenocarcinoma patients and was associated with decreased survival and more aggressive necrotic tumors. A TTP-low signature was characterized by perturbation of several inflammatory pathways in this study [19].
ZFP36L1 was found to be one of the genes with variants that was associated with an increased risk of subtype-specific epithelial ovarian cancers [40]. ZFP36L2 was overexpressed in pancreatic ductal adenocarcinoma (PDAC) tissues and cells as a result of suppression of microRNA-375, indicating the involvement of ZFP36L2-regulated pathways in PDAC pathogenesis. This was further supported by silencing ZFP36L2 in vitro, which inhibited cancer cell aggressiveness in PDAC cells. High ZFP36L2 expression also predicted shorter survival in PDAC, indicating that ZFP36L2 expression could be used as a prognostic marker in PDAC [62]. ZFP36L2 was also identified as a potential candidate for prediction of bone metastasis of breast cancer [33]. Finally, ZFP36L2 has been shown as a reoccurrence-associated gene in bladder cancer [20].
11. TTP Family Proteins and Response to Treatment
TTP proteins have also been associated with response to treatment in cancer in a few selective studies. Griseri et al. showed the presence of a synonymous polymorphism (rs3746083) in the TTP gene in an aggressive TTP-negative breast cancer cell line [22]. Interestingly, this mutation did not change the corresponding amino acid but affected the protein translation and was significantly associated with a lack of response to Herceptin treatment in HER2-positive breast cancer patients [22]. Whole genome microarray profiling of peripheral blood mononuclear cells of locally advanced rectal cancer patients revealed that, among other genes, TTP was differentially expressed between responders and non-responders to chemoradiotherapy [67]. Finally, the antitumor activity of curcumin analogue DM-1 in melanoma was shown to be mediated by multiple targets, which included TTP, ZFP36L1, and ZFP36L2, among others [58].
12. Outstanding Questions
A few outstanding questions remain regarding the role of the TTP family RBPs in carcinogenesis. For instance, much of our current knowledge regarding the role of TTP family RBPs in human cancer comes from the gene and protein expression data on patient tumor samples. Therefore, it is not clear whether the loss of TTP family RBPs is an early event that initiates tumor development or is a consequence of tumor development. Hence, there is an utmost need to develop transgenic animal models to understand the mechanisms by which the TTP family RBPs modulate the initiation and progression of cancer.
13. Conclusions
Together, the studies discussed in this review indicate that the TTP family RBPs are critical regulators of multiple cancer traits (Figure 1). Given the increasing number of cancers in which TTP family proteins have been reported to be dysregulated, it appears that this family of proteins are common and important regulators of many, if not all, cancer types. It also appears that the TTP family RBPs are frequently silenced with loss of function in a majority of cancers, indicating their role as tumor suppressors. Silencing of TTP family RBP expression occurs at the epigenetic, post-transcriptional, and post-translational levels. Moreover, TTP family proteins appear to regulate multiple pathways involved in cancer development and progression and present poor prognostic outcomes for patients. While conventional cancer therapies target single genes or pathways at one time, therapeutically targeting TTP family RBPs, the master regulators of multiple cancer-relevant genes, would target multiple cancer-relevant pathways simultaneously. Therefore, targeting these proteins and restoring their function may represent an effective and novel therapeutic approach.
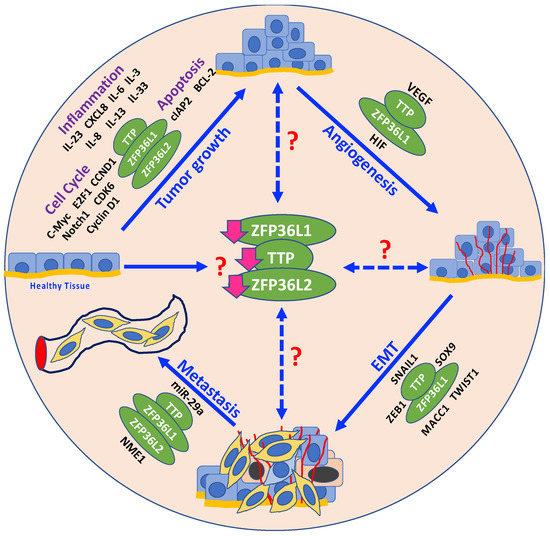
Figure 1.
Schematic representation of the various molecular cancer traits that are potentially regulated by TTP family RBPs: TTP family RBPs regulate mRNA targets (shown in black around TTP family RBPs, represented in green ovals) at various stages during carcinogenesis, including tumor growth, angiogenesis, epithelial-mesenchymal transition (EMT), and metastasis. At what stage during the process of carcinogenesis does the loss of expression (downward pink arrows) of TTP family RBPs occur remains unknown (red question mark alongside downregulated expression of TTP family RBPs in the center of the figure). For example, it is possible that normal cells undergo loss of TTP family RBPs expression under the influence of an unknown stressor, thus driving tumor initiation or complementing the effects of the driver genes. Conversely, it is possible that loss of TTP family RBP expression occurs at subsequent stages after initial tumor formation, thus accelerating tumor progression and promoting tumor aggressiveness. Broken bidirectional arrows with red question marks represent this possibility.
Author Contributions
S.P. conceived the article; S.P. and Y.S. wrote the article; J.C. prepared Table 2 and edited the article; Y.S., J.C., and S.P. proofread and approved the submitted version. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by grants from the National Institutes of Health and Louisiana Board of Regents.
Conflicts of Interest
The authors have no conflict of interest to report.
Abbreviations
| 3′UTR | 3′-untranslated region |
| AHRR | aryl hydrocarbon receptor-repressor |
| ARES | au-rich elements |
| BCL2 | B-cell lymphoma-2 |
| BERG36 | 36-Kda Zinc finger protein |
| BRF | Butyrate response factor |
| C-MYC | Myc protooncogene |
| CDK6 | cyclin-dependent kinase 6 |
| CIAP2 | cellular inhibitors of apoptosis 2 |
| COX2 | cyclooxygenase 2 |
| CXCL8 | chemokine (c-x-c motif) ligand 8 |
| E2F1 | E2F transcription factor 1 |
| EMT | epithelial-mesenchymal transition |
| ERA | estrogen receptor alpha |
| ERF | EGF-response factor 1 |
| GALR2 | galanin receptor type 2 |
| GOS24 | G0/G1 switch regulatory protein 24 |
| HBV | hepatitis b virus |
| HIF-1A | hypoxia-inducible factor 1 alpha |
| hTERT | human telomerase reverse transcription gene |
| IL-13 | interleukin 13 |
| IL-23 | interleukin 23 |
| IL-27 | interleukin 27 |
| IL-3 | interleukin 3 |
| IL-33 | interleukin 33 |
| IL-6 | interleukin 6 |
| IL-8 | interleukin 8 |
| LATS2 | large tumor suppressor kinase 2 |
| MACC1 | metastasis-associated in colon cancer 1 |
| MMP1 | matrix metalloproteinase 1 |
| MMP2 | matrix metalloproteinase 2 |
| MMP9 | matrix metalloproteinase 9 |
| mTOR | mammalian target of rapamycin |
| NEDD9 | neural precursor cell expressed developmentally downregulated protein 9 |
| NF-KB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| NME1 | nucleoside diphosphate kinase 1 |
| NUP475 | growth factor-inducible nuclear protein nup475 |
| PD-L1 | programmed death-ligand 1 |
| PI3K | phosphatidylinositol-3-kinase |
| PIM1 | proto-oncogene serine/threonine-protein kinase 1 |
| PIM2 | proto-oncogene serine/threonine-protein kinase 2 |
| RB | retinoblastoma 1 |
| SASP | senescence associated secretory protein |
| SNAI1 | zinc finger protein snail 1 |
| SOX9 | sry-box transcription factor 9 |
| TGF-B1 | transforming growth factor beta 1 |
| TIS11 | tpa-inducible sequence 11 |
| TNFA | tumor necrosis factor alpha |
| TTP | tristetraprolin |
| TWIST1 | twist-related protein 1 |
| UPA | urokinase-type plasminogen activator |
| UPAR | urokinase plasminogen activator receptor |
| VEGF | vascular endothelial growth factor |
| XIAP | x-linked inhibitor of apoptosis |
| ZEB1 | zinc finger e-box binding homeobox 1 |
| ZEB2 | zinc finger e-box binding homeobox 2 |
| ZFP36 | zinc finger protein 36 |
| ZFP36L1 | zinc finger protein 36 like 1 |
| ZFP36L2 | zinc finger protein 36 like 2 |
| ZFP36L3 | zinc finger protein 36 like 3 |
References
- Fu, M.; Blackshear, P.J. RNA-binding proteins in immune regulation: A focus on CCCH zinc finger proteins. Nat. Rev. Immunol. 2016, 17, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.S.; Stumpo, D.J.; Kennington, E.A.; Burkholder, A.; Ward, J.; Fargo, D.L.; Blackshear, P.J. Life without TTP: Apparent absence of an important anti-inflammatory protein in birds. Am. J. Physiol. Integr. Comp. Physiol. 2013, 305, R689–R700. [Google Scholar] [CrossRef]
- Brewer, B.Y.; Malicka, J.; Blackshear, P.J.; Wilson, G. RNA Sequence Elements Required for High Affinity Binding by the Zinc Finger Domain of Tristetraprolin. J. Biol. Chem. 2004, 279, 27870–27877. [Google Scholar] [CrossRef]
- Taylor, G.A.; Carballo, E.; Lee, D.M.; Lai, W.S.; Thompson, M.J.; Patel, D.D.; I Schenkman, D.; Gilkeson, G.S.; E Broxmeyer, H.; Haynes, B.F.; et al. A Pathogenetic Role for TNFα in the Syndrome of Cachexia, Arthritis, and Autoimmunity Resulting from Tristetraprolin (TTP) Deficiency. Immunity 1996, 4, 445–454. [Google Scholar] [CrossRef]
- Stumpo, D.J.; Byrd, N.A.; Phillips, R.S.; Ghosh, S.; Maronpot, R.R.; Castranio, T.; Meyers, E.N.; Mishina, Y.; Blackshear, P.J. Chorioallantoic Fusion Defects and Embryonic Lethality Resulting from Disruption of Zfp36L1, a Gene Encoding a CCCH Tandem Zinc Finger Protein of the Tristetraprolin Family. Mol. Cell. Biol. 2004, 24, 6445–6455. [Google Scholar] [CrossRef]
- Stumpo, D.J.; Broxmeyer, H.E.; Ward, T.; Cooper, S.; Hangoc, G.; Chung, Y.J.; Shelley, W.C.; Richfield, E.K.; Ray, M.K.; Yoder, M.C.; et al. Targeted disruption of Zfp36l2, encoding a CCCH tandem zinc finger RNA-binding protein, results in defective hematopoiesis. Blood 2009, 114, 2401–2410. [Google Scholar] [CrossRef]
- Stumpo, D.J.; Trempus, C.S.; Tucker, C.J.; Huang, W.; Li, L.; Kluckman, K.; Bortner, D.M.; Blackshear, P.J. Deficiency of the placenta- and yolk sac-specific tristetraprolin family member ZFP36L3 identifies likely mRNA targets and an unexpected link to placental iron metabolism. Development 2016, 143, 1424–1433. [Google Scholar] [CrossRef]
- Hodson, D.J.; Janas, M.L.; Galloway, A.; Bell, S.E.; Andrews, S.; Li, C.M.; Pannell, R.; Siebel, C.W.; Macdonald, H.R.; De Keersmaecker, K.; et al. Deletion of the RNA-binding proteins ZFP36L1 and ZFP36L2 leads to perturbed thymic development and T lymphoblastic leukemia. Nat. Immunol. 2010, 11, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Rounbehler, R.J.; Fallahi, M.; Yang, C.; Steeves, M.A.; Li, W.; Doherty, J.R.; Schaub, F.X.; Sanduja, S.; Dixon, D.A.; Blackshear, P.J.; et al. Tristetraprolin impairs myc-induced lymphoma and abolishes the malignant state. Cell 2012, 150, 563–574. [Google Scholar] [CrossRef]
- Zekavati, A.; Nasir, A.; Alcaraz, A.; Aldrovandi, M.; Marsh, P.; Norton, J.D.; Murphy, J.J. Post-Transcriptional Regulation of BCL2 mRNA by the RNA-Binding Protein ZFP36L1 in Malignant B Cells. PLoS ONE 2014, 9, e102625. [Google Scholar] [CrossRef]
- Sawaoka, H.; Dixon, D.A.; Oates, J.A.; Boutaud, O. Tristetraprolin Binds to the 3′-Untranslated Region of Cyclooxygenase-2 mRNA. J. Biol. Chem. 2003, 278, 13928–13935. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, H.J.; Yoon, N.A.; Lee, W.H.; Min, Y.J.; Ko, B.K.; Lee, B.J.; Lee, A.; Cha, H.J.; Cho, W.J.; et al. Tumor suppressor p53 plays a key role in induction of both tristetraprolin and let-7 in human cancer cells. Nucleic Acids Res. 2013, 41, 5614–5625. [Google Scholar] [CrossRef]
- Lee, H.H.; Vo, M.-T.; Kim, H.J.; Lee, U.H.; Kim, C.W.; Kim, H.K.; Ko, M.S.; Lee, W.H.; Cha, S.J.; Min, Y.J.; et al. Stability of the LATS2 Tumor Suppressor Gene Is Regulated by Tristetraprolin. J. Biol. Chem. 2010, 285, 17329–17337. [Google Scholar] [CrossRef]
- Wang, H.; Ding, N.; Guo, J.; Xia, J.; Ruan, Y. Dysregulation of TTP and HuR plays an important role in cancers. Tumor Biol. 2016, 37, 14451–14461. [Google Scholar] [CrossRef]
- Loh, X.-Y.; Sun, Q.-Y.; Ding, L.-W.; Mayakonda, A.; Venkatachalam, N.; Yeo, M.-S.; Silva, T.C.; Xiao, J.-F.; Doan, N.B.; Said, J.W.; et al. RNA-Binding Protein ZFP36L1 Suppresses Hypoxia and Cell-Cycle Signaling. Cancer Res. 2019, 80, 219–233. [Google Scholar] [CrossRef]
- Brennan, S.E.; Kuwano, Y.; Alkharouf, N.; Blackshear, P.J.; Gorospe, M.; Wilson, G. The mRNA-destabilizing protein tristetraprolin is suppressed in many cancers, altering tumorigenic phenotypes and patient prognosis. Cancer Res. 2009, 69, 5168–5176. [Google Scholar] [CrossRef]
- Goddio, M.V.; Gattelli, A.; Slomiansky, V.; Lacunza, E.; Gingerich, T.; Tocci, J.M.; Facchinetti, M.M.; Curino, A.C.; Lamarre, J.; Abba, M.; et al. Mammary differentiation induces expression of Tristetraprolin, a tumor suppressor AU-rich mRNA-binding protein. Breast Cancer Res. Treat. 2012, 135, 749–758. [Google Scholar] [CrossRef]
- Canzoneri, R.; Naipauer, J.; Stedile, M.; Peña, A.R.; Lacunza, E.; Gandini, N.A.; Curino, A.C.; Facchinetti, M.M.; Coso, O.A.; Kordon, E.; et al. Identification of an AP1-ZFP36 Regulatory Network Associated with Breast Cancer Prognosis. J. Mammary Gland. Biol. Neoplasia 2020, 1–10. [Google Scholar] [CrossRef]
- Fallahi, M.; Amelio, A.L.; Cleveland, J.L.; Rounbehler, R.J. CREB Targets Define the Gene Expression Signature of Malignancies Having Reduced Levels of the Tumor Suppressor Tristetraprolin. PLoS ONE 2014, 9, e115517. [Google Scholar] [CrossRef]
- Han, Y.; Zheng, Q.; Tian, Y.; Ji, Z.; Ye, H. Identification of a nine-gene panel as a prognostic indicator for recurrence with muscle-invasive bladder cancer. J. Surg. Oncol. 2019, 119, 1145–1154. [Google Scholar] [CrossRef]
- Al-Souhibani, N.; Al-Ahmadi, W.; Hesketh, J.E.; Blackshear, P.J.; Khabar, K.S.A. The RNA-binding zinc-finger protein tristetraprolin regulates AU-rich mRNAs involved in breast cancer-related processes. Oncogene 2010, 29, 4205–4215. [Google Scholar] [CrossRef]
- Griseri, P.; Bourcier, C.; Hieblot, C.; Essafi-Benkhadir, K.; Chamorey, E.; Touriol, C.; Pagès, G. A synonymous polymorphism of the Tristetraprolin (TTP) gene, an AU-rich mRNA-binding protein, affects translation efficiency and response to Herceptin treatment in breast cancer patients. Hum. Mol. Genet. 2011, 20, 4556–4568. [Google Scholar] [CrossRef]
- Lee, H.H.; Kim, W.-T.; Kim, N.H.; Park, J.W.; Kang, T.-H.; Chung, J.W.; Leem, S.-H. Tristetraprolin suppresses AHRR expression through mRNA destabilization. FEBS Lett. 2013, 587, 1518–1523. [Google Scholar] [CrossRef]
- Barrios-Garcia, T.; Tecalco-Cruz, A.; Gómez-Romero, V.; Reyes-Carmona, S.; Meneses-Morales, I.; León-Del-Río, A. Tristetraprolin Represses Estrogen Receptor α Transactivation in Breast Cancer Cells. J. Biol. Chem. 2014, 289, 15554–15565. [Google Scholar] [CrossRef]
- Gebeshuber, C.A.; Zatloukal, K.; Martínez, J. miR-29a suppresses tristetraprolin, which is a regulator of epithelial polarity and metastasis. EMBO Rep. 2009, 10, 400–405. [Google Scholar] [CrossRef]
- Xu, L.; Ning, H.; Gu, L.; Wang, Q.; Lu, W.; Peng, H.; Cui, W.; Ying, B.; Ross, C.R.; Wilson, G.M.; et al. Tristetraprolin induces cell cycle arrest in breast tumor cells through targeting AP-1/c-Jun and NF-kappaB pathway. Oncotarget 2015, 6, 41679. [Google Scholar] [CrossRef]
- Hitti, E.; Bakheet, T.; Al-Souhibani, N.; Moghrabi, W.; Al-Yahya, S.; Al-Ghamdi, M.; Al-Saif, M.; Shoukri, M.M.; Lánczky, A.; Grepin, R.; et al. Systematic Analysis of AU-Rich Element Expression in Cancer Reveals Common Functional Clusters Regulated by Key RNA-Binding Proteins. Cancer Res. 2016, 76, 4068–4080. [Google Scholar] [CrossRef]
- Pandiri, I.; Chen, Y.; Joe, Y.; Kim, H.J.; Park, J.; Chung, H.T.; Park, J.W. Tristetraprolin mediates the anti-proliferative effects of metformin in breast cancer cells. Breast Cancer Res. Treat. 2016, 156, 57–64. [Google Scholar] [CrossRef][Green Version]
- Wang, Q.; Ning, H.; Peng, H.; Wei, L.; Hou, R.; Hoft, D.F.; Liu, J. Tristetraprolin inhibits macrophage IL-27-induced activation of antitumour cytotoxic T cell responses. Nat. Commun. 2017, 8, 867. [Google Scholar] [CrossRef]
- Abba, M.; Sun, H.; Hawkins, K.A.; Drake, J.A.; Hu, Y.; Nunez, M.I.; Gaddis, S.; Shi, T.; Horvath, S.; Sahin, A.; et al. Breast cancer molecular signatures as determined by SAGE: Correlation with lymph node status. Mol. Cancer Res. 2007, 5, 881–890. [Google Scholar] [CrossRef]
- Lu, H.; Tran, L.; Park, Y.; Chen, I.; Lan, J.; Xie, Y.; Semenza, G.L. Reciprocal Regulation of DUSP9 and DUSP16 Expression by HIF1 Controls ERK and p38 MAP Kinase Activity and Mediates Chemotherapy-Induced Breast Cancer Stem Cell Enrichment. Cancer Res. 2018, 78, 4191–4202. [Google Scholar] [CrossRef]
- Rataj, F.; Planel, S.; Denis, J.; Roelants, C.; Filhol, O.; Guyon, L.; Feige, J.-J.; Cherradi, N. Targeting AU-rich element-mediated mRNA decay with a truncated active form of the zinc-finger protein TIS11b/BRF1 impairs major hallmarks of mammary tumorigenesis. Oncogene 2019, 38, 5174–5190. [Google Scholar] [CrossRef]
- Park, S.B.; Chung, C.K.; Gonzalez, E.; Yoo, C. Causal Inference Network of Genes Related with Bone Metastasis of Breast Cancer and Osteoblasts Using Causal Bayesian Networks. J. Bone Metab. 2018, 25, 251–266. [Google Scholar] [CrossRef]
- Sanduja, S.; Kaza, V.; Dixon, D.A. The mRNA decay factor tristetraprolin (TTP) induces senescence in human papillomavirus-transformed cervical cancer cells by targeting E6-AP ubiquitin ligase. Aging 2009, 1, 803–817. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Yang, S.S.; Vo, M.-T.; Cho, W.J.; Lee, B.J.; Leem, S.-H.; Lee, S.-H.; Cha, H.J.; Park, J.W. Tristetraprolin down-regulates IL-23 expression in colon cancer cells. Mol. Cells 2013, 36, 571–576. [Google Scholar] [CrossRef]
- Montorsi, L.; Guizzetti, F.; Alecci, C.; Caporali, A.; Martello, A.; Atene, C.G.; Parenti, S.; Pizzini, S.; Zanovello, P.; Bortoluzzi, S.; et al. Loss of ZFP36 expression in colorectal cancer correlates to wnt/ ss-catenin activity and enhances epithelial-to-mesenchymal transition through upregulation of ZEB1, SOX9 and MACC1. Oncotarget 2016, 7, 59144–59157. [Google Scholar] [CrossRef]
- Coelho, M.; Trécesson, S.D.C.; Rana, S.; Zecchin, D.; Moore, C.; Molina-Arcas, M.; East, P.; Spencer-Dene, B.; Nye, E.; Barnouin, K.; et al. Oncogenic RAS Signaling Promotes Tumor Immunoresistance by Stabilizing PD-L1 mRNA. Immunity 2017, 47, 1083–1099. [Google Scholar] [CrossRef]
- Wei, F.; Zhang, T.; Yang, Z.; Wei, J.-C.; Shen, H.-F.; Xiao, N.; Wang, Q.; Yang, P.; Chen, H.-C.; Hu, H.; et al. Gambogic Acid Efficiently Kills Stem-Like Colorectal Cancer Cells by Upregulating ZFP36 Expression. Cell. Physiol. Biochem. 2018, 46, 829–846. [Google Scholar] [CrossRef]
- Lee, S.-R.; Jin, H.; Kim, W.-T.; Kim, W.-J.; Kim, S.Z.; Leem, S.-H.; Kim, S.M. Tristetraprolin activation by resveratrol inhibits the proliferation and metastasis of colorectal cancer cells. Int. J. Oncol. 2018, 53, 1269–1278. [Google Scholar] [CrossRef]
- Earp, M.A.; Kelemen, L.E.; Magliocco, A.M.; Swenerton, K.D.; Chenevix-Trench, G.; Lu, Y.; Hein, A.; Ekici, A.B.; Beckmann, M.W.; Fasching, P.A.; et al. Genome-wide Association Study of Subtype-Specific Epithelial Ovarian Cancer Risk Alleles Using Pooled DNA. Qual. Life Res. 2013, 133, 481–497. [Google Scholar] [CrossRef]
- Lin, D.-C.; Dinh, H.Q.; Xie, J.-J.; Mayakonda, A.; Silva, T.C.; Jiang, Y.-Y.; Ding, L.-W.; He, J.-Z.; Xu, X.-E.; Hao, J.-J.; et al. Identification of distinct mutational patterns and new driver genes in oesophageal squamous cell carcinomas and adenocarcinomas. Gut 2017, 67, 1769–1779. [Google Scholar] [CrossRef]
- McCorkle, J.R.; Leonard, M.K.; Kraner, S.D.; Blalock, E.M.; Ma, D.; Zimmer, S.G.; Kaetzel, D.M.; Deqin, M. The metastasis suppressor NME1 regulates expression of genes linked to metastasis and patient outcome in melanoma and breast carcinoma. Cancer Genom. Proteom. 2014, 11, 175–194. [Google Scholar]
- Deng, K.; Wang, H.; Shan, T.; Chen, Y.; Zhou, H.; Zhao, Q.; Xia, J. Tristetraprolin inhibits gastric cancer progression through suppression of IL-33. Sci. Rep. 2016, 6, 24505. [Google Scholar] [CrossRef] [PubMed]
- Xing, R.; Zhou, Y.; Yu, J.; Yu, Y.; Nie, Y.; Luo, W.; Yang, C.; Xiong, T.; Wu, W.K.K.; Li, Z.; et al. Whole-genome sequencing reveals novel tandem-duplication hotspots and a prognostic mutational signature in gastric cancer. Nat. Commun. 2019, 10, 2037. [Google Scholar] [CrossRef] [PubMed]
- Selmi, T.; Martello, A.; Vignudelli, T.; Ferrari, E.; Grande, A.; Atene, C.G.; Salomoni, P.; Ferrari, S.; Zanocco-Marani, T. ZFP36 expression impairs glioblastoma cell lines viability and invasiveness by targeting multiple signal transduction pathways. Cell Cycle 2012, 11, 1977–1987. [Google Scholar] [CrossRef] [PubMed]
- Suswam, E.; Li, Y.; Zhang, X.; Gillespie, G.Y.; Li, X.; Shacka, J.J.; Lu, L.; Zheng, L.; King, P.H. Tristetraprolin Down-regulates Interleukin-8 and Vascular Endothelial Growth Factor in Malignant Glioma Cells. Cancer Res. 2008, 68, 674–682. [Google Scholar] [CrossRef]
- Ryu, J.; Yoon, N.A.; Seong, H.; Jeong, J.Y.; Kang, S.; Park, N.; Choi, J.; Lee, N.H.; Roh, G.S.; Kim, H.J.; et al. Resveratrol Induces Glioma Cell Apoptosis through Activation of Tristetraprolin. Mol. Cells 2015, 38, 991–997. [Google Scholar]
- Twizere, J.-C.; Kruys, V.; Lefèbvre, L.; Vanderplasschen, A.; Collete, D.; Debacq, C.; Lai, W.S.; Jauniaux, J.-C.; Bernstein, L.R.; Semmes, O.J.; et al. Interaction of Retroviral Tax Oncoproteins With Tristetraprolin and Regulation of Tumor Necrosis Factor-Alpha Expression. J. Natl. Cancer Inst. 2003, 95, 1846–1859. [Google Scholar] [CrossRef]
- Weng, Q.; Wang, J.; Wang, J.; He, D.; Cheng, Z.; Zhang, F.; Verma, R.; Xu, L.; Dong, X.; Liao, Y.; et al. Single-Cell Transcriptomics Uncovers Glial Progenitor Diversity and Cell Fate Determinants during Development and Gliomagenesis. Cell Stem Cell 2019, 24, 707–723. [Google Scholar] [CrossRef]
- Liu, W.H.; Chou, W.M.; Chang, L.S. p38 MAPK/PP2Acalpha/TTP pathway on the connection of TNF-alpha and caspases activation on hydroquinone-induced apoptosis. Carcinogenesis 2013, 34, 818–827. [Google Scholar] [CrossRef]
- Wang, L.J.; Lee, Y.C.; Huang, C.H.; Shi, Y.J.; Chen, Y.J.; Pei, S.N.; Chou, Y.W.; Chang, L.S. Non-mitotic effect of albendazole triggers apoptosis of human leukemia cells via SIRT3/ROS/p38 MAPK/TTP axis-mediated TNF-alpha upregulation. Biochem. Pharm. 2019, 162, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-T.; Dong, L.; Zhang, X.-H.; Yin, X.-L.; Ning, H.-M.; Shen, C.; Su, R.; Li, F.; Song, L.; Ma, Y.-N.; et al. ZFP36L1 promotes monocyte/macrophage differentiation by repressing CDK6. Sci. Rep. 2015, 5, 16229. [Google Scholar] [CrossRef]
- Sohn, B.H.; Park, I.Y.; Lee, J.J.; Yang, S.; Jang, Y.J.; Park, K.C.; Kim, D.J.; Lee, D.C.; Sohn, H.A.; Kim, T.W.; et al. Functional Switching of TGF-β1 Signaling in Liver Cancer via Epigenetic Modulation of a Single CpG Site in TTP Promoter. Gastroenterology 2010, 138, 1898–1908. [Google Scholar] [CrossRef] [PubMed]
- Kröhler, T.; Kessler, S.M.; Hosseini, K.; List, M.; Barghash, A.; Patial, S.; Laggai, S.; Gemperlein, K.; Haybaeck, J.; Mueller, R.; et al. The mRNA-binding Protein TTP/ZFP36 in Hepatocarcinogenesis and Hepatocellular Carcinoma. Cancers 2019, 11, 1754. [Google Scholar] [CrossRef]
- Ren, W.; Ye, X.; Su, H.; Li, W.; Liu, D.-B.; Pirmoradian, M.; Wang, X.; Zhang, B.; Zhang, Q.; Chen, L.; et al. Genetic landscape of hepatitis B virus-associated diffuse large B-cell lymphoma. Blood 2018, 131, 2670–2681. [Google Scholar] [CrossRef] [PubMed]
- Stoecklin, G.; Gross, B.; Ming, X.; Moroni, C. A novel mechanism of tumor suppression by destabilizing AU-rich growth factor mRNA. Oncogene 2003, 22, 3554–3561. [Google Scholar] [CrossRef] [PubMed]
- Bourcier, C.; Griseri, P.; Grepin, R.; Bertolotto, C.; Mazure, N.; Pages, G. Constitutive ERK activity induces downregulation of tristetraprolin, a major protein controlling interleukin8/CXCL8 mRNA stability in melanoma cells. Am. J. Physiol. Cell Physiol. 2011, 301, C609–C618. [Google Scholar] [CrossRef]
- De Oliveira, É.A.; De Lima, D.S.; Cardozo, L.E.; De Souza, G.F.; De Souza, N.; Alves-Fernandes, D.K.; Faião-Flores, F.; Quincoces, J.A.P.; Barros, S.B.D.M.; Nakaya, H.I.; et al. Toxicogenomic and bioinformatics platforms to identify key molecular mechanisms of a curcumin-analogue DM-1 toxicity in melanoma cells. Pharmacol. Res. 2017, 125, 178–187. [Google Scholar] [CrossRef]
- Martínez-Calle, N.; Pascual, M.; Ordoñez, R.; José-Enériz, E.S.; Kulis, M.; Miranda, E.; Guruceaga, E.; Segura, V.; Larráyoz, M.J.; Bellosillo, B.; et al. Epigenomic profiling of myelofibrosis reveals widespread DNA methylation changes in enhancer elements and ZFP36L1 as a potential tumor suppressor gene that is epigenetically regulated. Haematologica 2019, 104, 1572–1579. [Google Scholar] [CrossRef]
- Sun, X.-J.; Liu, B.-Y.; Yan, S.; Jiang, T.-H.; Cheng, H.-Q.; Jiang, H.-S.; Cao, Y.; Mao, A.-W. MicroRNA-29a Promotes Pancreatic Cancer Growth by Inhibiting Tristetraprolin. Cell. Physiol. Biochem. 2015, 37, 707–718. [Google Scholar] [CrossRef]
- Wei, Z.-R.; Liang, C.; Feng, D.; Cheng, Y.-J.; Wang, W.-M.; Yang, D.-J.; Wang, Y.; Cai, Q.-P. Low tristetraprolin expression promotes cell proliferation and predicts poor patients outcome in pancreatic cancer. Oncotarget 2016, 7, 17737–17750. [Google Scholar] [CrossRef] [PubMed]
- Yonemori, K.; Seki, N.; Kurahara, H.; Osako, Y.; Idichi, T.; Arai, T.; Koshizuka, K.; Kita, Y.; Maemura, K.; Natsugoe, S. ZFP36L2 promotes cancer cell aggressiveness and is regulated by antitumor microRNA-375 in pancreatic ductal adenocarcinoma. Cancer Sci. 2017, 108, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Bornigen, D.; Tyekucheva, S.; Wang, X.; Rider, J.R.; Lee, G.S.; Mucci, L.A.; Sweeney, C.; Huttenhower, C. Computational Reconstruction of NFkappaB Pathway Interaction Mechanisms during Prostate Cancer. PLoS Comput. Biol. 2016, 12, e1004820. [Google Scholar] [CrossRef] [PubMed]
- Berglund, A.E.; Scott, K.E.; Li, W.; Yang, C.; Fernandez, M.R.; Schaub, F.X.; Cleveland, J.L.; Rounbehler, R.J. Tristetraprolin disables prostate cancer maintenance by impairing proliferation and metabolic function. Oncotarget 2016, 7, 83462–83475. [Google Scholar] [CrossRef]
- Zhu, J.-G.; Yuan, D.-B.; Chen, W.-H.; Han, Z.-D.; Liang, Y.-X.; Chen, G.-X.; Fu, X.; Sun, Z.-L.; Liu, Z.-Z.; Chen, J.-H.; et al. Prognostic value of ZFP36 and SOCS3 expressions in human prostate cancer. Clin. Transl. Oncol. 2015, 18, 782–791. [Google Scholar] [CrossRef]
- Rounbehler, R.J.; Berglund, A.E.; Gerke, T.A.; Takhar, M.M.; Awasthi, S.; Li, W.; Davicioni, E.; Erho, N.; Ross, A.E.; Schaeffer, E.M.; et al. Tristetraprolin Is a Prognostic Biomarker for Poor Outcomes among Patients with Low-Grade Prostate Cancer. Cancer Epidemiol. Biomark. Prev. 2018, 27, 1376–1383. [Google Scholar] [CrossRef]
- Palma, P.; Cuadros, M.; Muíño, R.C.; Olmedo, C.; Cano, C.; Segura-Jiménez, I.; Blanco, A.; Bueno, P.; Ferrón, J.A.; Medina, P.P. Microarray Profiling of Mononuclear Peripheral Blood Cells Identifies Novel Candidate Genes Related to Chemoradiation Response in Rectal Cancer. PLoS ONE 2013, 8, e74034. [Google Scholar] [CrossRef]
- Van Tubergen, E.A.; Banerjee, R.; Liu, M.; Broek, R.V.; Light, E.; Kuo, S.; Feinberg, S.E.; Willis, A.L.; Wolf, G.; E Carey, T.; et al. Inactivation or loss of TTP promotes invasion in head and neck cancer via transcript stabilization and secretion of MMP9, MMP2, and IL-6. Clin. Cancer Res. 2013, 19, 1169–1179. [Google Scholar] [CrossRef]
- Banerjee, R.; Van Tubergen, E.A.; Scanlon, C.S.; Broek, R.V.; Lints, J.P.; Liu, M.; Russo, N.; Inglehart, R.C.; Wang, Y.; Polverini, P.J.; et al. The G protein-coupled receptor GALR2 promotes angiogenesis in head and neck cancer. Mol. Cancer Ther. 2014, 13, 1323–1333. [Google Scholar] [CrossRef]
- Park, S.B.; Lee, J.H.; Jeong, W.W.; Kim, Y.H.; Cha, H.J.; Joe, Y.; Chung, H.T.; Cho, W.J.; Do, J.W.; Lee, B.J.; et al. TTP mediates cisplatin-induced apoptosis of head and neck cancer cells by down-regulating the expression ofBcl-2. J. Chemother. 2015, 27, 174–180. [Google Scholar] [CrossRef]
- Lee, S.K.; Kim, S.B.; Kim, J.S.; Moon, C.H.; Han, M.S.; Lee, B.J.; Chung, D.K.; Min, Y.J.; Park, J.H.; Choi, D.H.; et al. Butyrate response factor 1 enhances cisplatin sensitivity in human head and neck squamous cell carcinoma cell lines. Int. J. Cancer 2005, 117, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Reale, C.; Russo, F.; Credendino, S.C.; Cuomo, D.; De Vita, G.; Mallardo, M.; Pennino, F.; Porreca, I.; Triassi, M.; De Felice, M.; et al. A Toxicogenomic Approach Reveals a Novel Gene Regulatory Network Active in In Vitro and In Vivo Models of Thyroid Carcinogenesis. Int. J. Environ. Res. Public Health 2019, 16, 122. [Google Scholar] [CrossRef]
- Marderosian, M.; Sharma, A.; Funk, A.P.; Vartanian, R.; Masri, J.; Jo, O.D.; Gera, J. Tristetraprolin regulates Cyclin D1 and c-Myc mRNA stability in response to rapamycin in an Akt-dependent manner via p38 MAPK signaling. Oncogene 2006, 25, 6277–6290. [Google Scholar] [CrossRef]
- Dang, C.V. MYC on the Path to Cancer. Cell 2012, 149, 22–35. [Google Scholar] [CrossRef]
- Lee, H.H.; Lee, S.-R.; Leem, S.-H. Tristetraprolin regulates prostate cancer cell growth through suppression of E2F1. J. Microbiol. Biotechnol. 2014, 24, 287–294. [Google Scholar] [CrossRef]
- Suk, F.-M.; Chang, C.-C.; Lin, R.-J.; Lin, S.-Y.; Liu, S.-C.; Jau, C.-F.; Liang, Y.-C. ZFP36L1 and ZFP36L2 inhibit cell proliferation in a cyclin D-dependent and p53-independent manner. Sci. Rep. 2018, 8, 2742. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.A.; Geha, M.; Blackwell, T.K. Similar but distinct effects of the tristetraprolin/TIS11 immediate-early proteins on cell survival. Oncogene 2000, 19, 1657–1664. [Google Scholar] [CrossRef]
- Kim, H.K.; Kim, C.W.; Vo, M.-T.; Lee, H.H.; Lee, J.Y.; Yoon, N.A.; Lee, C.Y.; Moon, C.H.; Min, Y.J.; Park, J.W.; et al. Expression of Proviral Integration Site for Moloney Murine Leukemia Virus 1 (Pim-1) Is Post-transcriptionally Regulated by Tristetraprolin in Cancer Cells. J. Biol. Chem. 2012, 287, 28770–28778. [Google Scholar] [CrossRef]
- Lai, W.S.; Carballo, E.; Strum, J.R.; Kennington, E.A.; Phillips, R.S.; Blackshear, P.J. Evidence that Tristetraprolin Binds to AU-Rich Elements and Promotes the Deadenylation and Destabilization of Tumor Necrosis Factor Alpha mRNA. Mol. Cell. Biol. 1999, 19, 4311–4323. [Google Scholar] [CrossRef]
- Molle, C.; Zhang, T.; De Lendonck, L.Y.; Gueydan, C.; Andrianne, M.; Sherer, F.; Van Simaeys, G.; Blackshear, P.J.; Leo, O.; Goriely, S. Tristetraprolin regulation of interleukin 23 mRNA stability prevents a spontaneous inflammatory disease. J. Exp. Med. 2013, 210, 1675–1684. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.-Q.; Lai, W.S.; Bradbury, A.; Zeldin, D.C.; Blackshear, P.J. Tristetraprolin (TTP) coordinately regulates primary and secondary cellular responses to proinflammatory stimuli. J. Leukoc. Biol. 2015, 97, 723–736. [Google Scholar] [CrossRef]
- Brooks, S.A.; Blackshear, P.J. Tristetraprolin (TTP): Interactions with mRNA and proteins, and current thoughts on mechanisms of action. Biochim. Biophys. Acta 2013, 1829, 666–679. [Google Scholar] [CrossRef] [PubMed]
- Patial, S.; Curtis, A.D., 2nd; Lai, W.S.; Stumpo, D.J.; Hill, G.D.; Flake, G.P.; Mannie, M.D.; Blackshear, P.J. Enhanced stability of tristetraprolin mRNA protects mice against immune-mediated inflammatory pathologies. Proc. Natl. Acad. Sci. USA 2016, 113, 1865–1870. [Google Scholar] [CrossRef] [PubMed]
- Dubois, R.N.; Tsujii, M.; Bishop, P.; Awad, J.A.; Makita, K.; Lanahan, A. Cloning and characterization of a growth factor-inducible cyclooxygenase gene from rat intestinal epithelial cells. Am. J. Physiol. Liver Physiol. 1994, 266, G822–G827. [Google Scholar] [CrossRef] [PubMed]
- Dubois, R.N.; Awad, J.; Morrow, J.; Roberts, L.J.; Bishop, P.R. Regulation of eicosanoid production and mitogenesis in rat intestinal epithelial cells by transforming growth factor-alpha and phorbol ester. J. Clin. Investig. 1994, 93, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Ristimäki, A.; Garfinkel, S.; Wessendorf, J.; Maciag, T.; Hla, T. Induction of cyclooxygenase-2 by interleukin-1 alpha. Evidence for post-transcriptional regulation. J. Biol. Chem. 1994, 269, 11769–11775. [Google Scholar]
- Eberhart, C.E.; Coffey, R.J.; Radhika, A.; Giardiello, F.M.; Ferrenbach, S.; Dubois, R.N. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 1994, 107, 1183–1188. [Google Scholar] [CrossRef]
- Zimmermann, K.C.; Sarbia, M.; A Weber, A.; Borchard, F.; E Gabbert, H.; Schrör, K. Cyclooxygenase-2 expression in human esophageal carcinoma. Cancer Res. 1999, 59, 198–204. [Google Scholar]
- A Molina, M.; Sitja-Arnau, M.; Lemoine, M.G.; Frazier, M.L.; A Sinicrope, F. Increased cyclooxygenase-2 expression in human pancreatic carcinomas and cell lines: Growth inhibition by nonsteroidal anti-inflammatory drugs. Cancer Res. 1999, 59, 4356–4362. [Google Scholar]
- Wolff, H.; Saukkonen, K.; Anttila, S.; Karjalainen, A.; Vainio, H.; Ristimäki, A. Expression of cyclooxygenase-2 in human lung carcinoma. Cancer Res. 1998, 58, 4997–5001. [Google Scholar] [PubMed]
- Yoshimura, R.; Sano, H.; Masuda, C.; Kawamura, M.; Tsubouchi, Y.; Chargui, J.; Yoshimura, N.; Hla, T.; Wada, S. Expression of cyclooxygenase-2 in prostate carcinoma. Cancer 2000, 89, 589–596. [Google Scholar] [CrossRef]
- Soslow, R.A.; Dannenberg, A.J.; Rush, D.; Woerner, B.M.; Khan, K.N.; Masferrer, J.; Koki, A.T. COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer 2000, 89, 2637–2645. [Google Scholar] [CrossRef]
- Kim, T.W.; Yim, S.; Choi, B.J.; Jang, Y.; Lee, J.J.; Sohn, B.H.; Yoo, H.-S.; Yeom, Y.I.; Park, K.C. Tristetraprolin regulates the stability of HIF-1α mRNA during prolonged hypoxia. Biochem. Biophys. Res. Commun. 2010, 391, 963–968. [Google Scholar] [CrossRef]
- Sung, W.-W.; Chu, Y.-C.; Chen, P.-R.; Liao, M.-H.; Lee, J.-W. Positive regulation of HIF-1A expression by EBV oncoprotein LMP1 in nasopharyngeal carcinoma cells. Cancer Lett. 2016, 382, 21–31. [Google Scholar] [CrossRef]
- Zeng, B.; Zhu, D.; Su, Z.; Li, Z.; Yu, Z. Tristetraprolin exerts tumor suppressive functions on the tumorigenesis of glioma by targeting IL-13. Int. Immunopharmacol. 2016, 39, 63–70. [Google Scholar] [CrossRef]
- Kratochvill, F.; Gratz, N.; Qualls, J.; Van De Velde, L.-A.; Chi, H.; Kovarik, P.; Murray, P.J. Tristetraprolin Limits Inflammatory Cytokine Production in Tumor-Associated Macrophages in an mRNA Decay-Independent Manner. Cancer Res. 2015, 75, 3054–3064. [Google Scholar] [CrossRef]
- Herranz, N.; Gallage, S.; Mellone, M.; Wuestefeld, T.; Klotz, S.; Hanley, C.J.; Raguz, S.; Acosta, J.C.; Innes, A.J.; Banito, A.; et al. mTOR regulates MAPKAPK2 translation to control the senescence-associated secretory phenotype. Nat. Cell Biol. 2015, 17, 1205–1217. [Google Scholar] [CrossRef] [PubMed]
- Ciais, D.; Cherradi, N.; Bailly, S.; Grenier, E.; Berra, E.; Pouysségur, J.; Lamarre, J.; Feige, J.-J. Destabilization of vascular endothelial growth factor mRNA by the zinc-finger protein TIS11b. Oncogene 2004, 23, 8673–8680. [Google Scholar] [CrossRef]
- Planel, S.; Salomon, A.; Jalinot, P.; Feige, J.-J.; Cherradi, N. A novel concept in antiangiogenic and antitumoral therapy: Multitarget destabilization of short-lived mRNAs by the zinc finger protein ZFP36L1. Oncogene 2010, 29, 5989–6003. [Google Scholar] [CrossRef]
- Chamboredon, S.; Ciais, D.; Desroches-Castan, A.; Savi, P.; Bono, F.; Feige, J.-J.; Cherradi, N. Hypoxia-inducible factor-1α mRNA: A new target for destabilization by tristetraprolin in endothelial cells. Mol. Biol. Cell 2011, 22, 3366–3378. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).