Simple Summary
Approximately 60% of all melanomas are associated with a constitutive activating BRAF mutation. Inhibition of BRAF downstream signaling by targeted therapies significantly improved patient outcomes. However, most patients eventually develop resistance. Here we identified miR-129-5p as a novel tumor suppressor in BRAF mutated melanoma, which expression is increased during response to BRAF inhibition, but repressed in an EZH2 dependent manner during activated BRAF signaling. Overexpression of miR-129-5p decreases melanoma cell proliferation and improves response to BRAF inhibition by targeting SOX4. Taken together our results emphasize SOX4 as a potential therapeutic target in BRAF driven melanoma which could be attacked by pharmaceutically.
Abstract
Many melanomas are associated with activating BRAF mutation. Targeted therapies by inhibitors of BRAF and MEK (BRAFi, MEKi) show marked antitumor response, but become limited by drug resistance. The mechanisms for this are not fully revealed, but include miRNA. Wishing to improve efficacy of BRAFi and knowing that certain miRNAs are linked to resistance to BRAFi, we wanted to focus on miRNAs exclusively associated with response to BRAFi. We found increased expression of miR-129-5p during BRAFi treatment of BRAF- mutant melanoma cells. Parallel to emergence of resistance we observed mir-129-5p expression to become suppressed by BRAF/EZH2 signaling. In functional analyses we revealed that miR-129-5p acts as a tumor suppressor as its overexpression decreased cell proliferation, improved treatment response and reduced viability of BRAFi resistant melanoma cells. By protein expression analyses and luciferase reporter assays we confirmed SOX4 as a direct target of mir-129-5p. Thus, modulation of the miR-129-5p-SOX4 axis could serve as a promising novel strategy to improve response to BRAFi in melanoma.
1. Introduction
Melanoma is the most lethal form of skin cancer with an increasing incidence [1]. Stage IV melanoma has a poor prognosis for patients, with a 5-year survival probability of less than 5–25%, if untreated [2,3]. Progression of a majority of cutaneous melanomas depends on oncogenic, partially mutually exclusive somatic mutations involving BRAF (50–60%), NRAS (20–26%), TP53 (19%), or PTEN (12%) [4,5,6,7,8]. The most common BRAF mutation is the V600E (90%) substitution, which constitutively activates aberrant BRAF signaling [9].
During the last decade, BRAF-mutant melanomas have become amenable to new targeted therapies based on initially BRAF inhibitor (BRAFi) and now on its combination with a MEK inhibitor (MEKi). Introduction of combinatory BRAFi/MEKi therapies improved patient outcomes significantly [10,11,12]; however, most patients eventually develop resistance mechanisms towards these targeted therapies [13]. Several molecular mechanisms are involved in the acquisition of BRAFi resistance. The most frequent one encompasses reactivation of the MAPK pathway, commonly through NRAS mutations [14], alterations in BRAF splicing [15] or amplification [16,17] and less often by alterations of MEK1/2 [18]. Alternatively, in some patients, the PI3K/AKT pathway, a secondary signaling route becomes hyper-activated [19]. Adaptive resistance to MEKi likewise is acquired by reactivation of MAPK signaling (e.g., ERK) or activation of parallel signaling pathways (e.g., PI3K, STAT and Hippo signaling pathways) [20]. Yet, a considerable proportion of BRAFi resistant tumors (40%) displays mechanisms of resistance that have not been fully revealed [21,22].
To identify novel strategies for improving efficacy of BRAFi in melanoma, it would be helpful to find molecular mechanisms of resistance, which can be modified or reverted.
There is evidence that miRNAs are involved in the development of resistance in melanoma treatment to targeted therapies [23]. MicroRNAs are small noncoding RNAs (20–22 nt) which post-transcriptionally regulate protein expression. To this end miRNAs guide the RNA-induced silencing complex (RISC) to a complementary seed sequence in the 3′untranslated region (3′UTR) of target mRNAs [24]. Binding of the miRNA-RISC reduces the efficacy of protein translation and induces destabilization and cleavage of the targeted mRNA [25]. MicroRNAs are involved in development or progression of cancer, but obviously also in mechanisms of drug resistance in leukaemia and in various solid cancers [26,27,28,29,30]. They could present potential therapeutic targets, by the use of chemically modified complementary RNA molecules, such as locked nucleic acids (LNAs) or miRNA mimics [31,32,33,34].
Thus, there is promise to investigate this group of molecular factors in more detail to define novel mechanisms of resistance, which could later on serve as potential therapeutic targets.
In melanoma, the expression of miR-7 [35], miR-126-3p [36] and miR-579-3p [37] is significantly reduced in BRAFi resistant cell lines, while their overexpression increases the sensitivity to BRAFi. In contrast, miR-34a, miR-100 and miR-125b were highly expressed in resistant melanoma cells and inhibition of those miRNAs restores the sensitivity of BRAFi resistant cells [38]. The miRNA family miR-204-5p and miR-211-5p are the most investigated, but most controversially discussed miRNAs associated with tumor progression and BRAFi resistance in melanoma. Both are induced by BRAFi treatment and their expression remains increased in cells resistant to BRAFi [39,40,41]. Moreover, enforced expression miR-211-5p contributes to BRAFi resistance by directly targeting DUSP6, which modulates the DUSP6-ERK5 signaling axis and promotes BRAFV600E driven melanoma growth and BRAFi/MEKi inhibitor resistance [40]. Vitiello et al. reported a context dependent function of miR-204 and miR-211. They show that in melanotic melanoma miR-211 targets EDEM1 [41], which potentiates Vemurafenib induced pigmentation, resulting in a limited Vemurafenib efficacy. miR-204 was shown to inhibit melanoma cell motility by targeting AP1S in amelanotic melanoma [41].
These findings illustrate that specific miRNAs are involved in alternative mechanisms of BRAFi resistance and that their manipulation can partially restore sensitivity to BRAF targeted therapies.
While microRNAs have been linked mostly to resistance to BRAFi in melanoma, we wanted to identify miRNAs associated specifically with response to BRAFi. We found that miR-129-5p was most strongly induced after Vemurafenib treatment in parental BRAF mutated melanoma cells, but not in the corresponding BRAFi resistant cells, normal melanocytes or BRAF wildtype melanoma cells. Subsequently, we investigated the transcriptional regulation of miR-129-5p downstream of constitutive active BRAF signaling and the biological function of miR-129-5p in BRAF mutated melanoma, as well as BRAFi resistance. Further, we wanted to identify a direct target of miR-129-5p, which mediates melanoma progression and BRAFi resistance.
The results of our study demonstrate that EZH2 dependent repression of miR-129-5p is solved by BRAFi/MEKi treatment thereby modulating BRAFi resistance and melanoma progression via targeting SOX4.
2. Materials and Methods
2.1. Data Sets
The published datasets GSE94423 [41], GSE98314 and GSE50509 [22] were obtained from GEO DataSets (https://www.ncbi.nlm.nih.gov/gds accessed on 22 April 2021). For GSE98314 and GSE50509 gene expression data were normalized using the cubic spline function. TCGA expression data (log2(RPM + 1) of miR-129-5p and EZH2 mutation status of melanoma samples were used from the cancer genome atlas (https://www.cancer.gov/tcga accessed on 22 April 2021).
2.2. Cell Culture
Normal human epidermal melanocytes (NHEM) were isolated from foreskin tissues and cultured in medium 254 (Cascade Biologics®) including human melanocyte growth supplement (HMGS) and 1% penicillin-streptomycin. Melanoma cell lines (A375, WM35, WM902B, WM9, MV3 and SK-Mel30) were cultured in DMEM supplemented with 10% fetal calve serum (FCS) and 1% penicillin-streptomycin. All cells were incubated at 37 °C and 5% CO2. Resistant cell lines A375R, WM35R, WM902BR and WM9R were generated by treating parental BRAFV600E mutant melanoma cell lines with increasing concentrations of Vemurafenib (PLX4032; LC-Laboratories, Woburn, MA, USA). Cells with the ability to grow in 2 µM or in 10 µM Vemurafenib were described as resistant and maintained in continuous presence of 2 µM or 10 µM Vemurafenib. For MEK inhibitor treatment we used Trametinib (LC-Laboratories, Woburn, MA, USA) with indicated concentrations. EZH2 inhibition was performed by Tazemetostat (EPZ-6438) (Selleckchem, Houston, TX, USA).
2.3. miRNA Detection by Quantitative Real-Time PCR
Total RNA was extracted from cells using the TriFast™ reagent (Peqlab). The miRNA quantification was performed by qRT-PCR using TaqMan® MicroRNA Reverse Transcription Kit and TaqMan® Universal Master Mix II following the manufacturer’s instructions. For normalization RNUB6 expression was used. TaqMan® miRNA assays for RNUB6 and hsa-miR-129-5p were obtained from Thermo Fisher Scientific.
2.4. Transcriptional Analysis by qRT-PCR
The total RNA was isolated using TriFast™ reagent (Peqlab). Reverse transcription was performed by M-MLV Reverse Transcriptase using oligo(dT)18-primer (Thermo Fisher Scientific, Waltham, MA, USA). For quantitative RT-PCR we used PowerUp™ SYBR™Green master mix (Thermo Fisher Scientific, Waltham, MA, USA) following the manufacturer’s instructions. qPCRs were performed on QuantStudio™ 5 Real-Time PCR Systems. Real-time quantitative PCR assay was performed to detect the relative expression level of SOX4 (forward 5′-GGTCTCTAGTTCTTGCACGCTC-3′ and reverse 5′-CGGAATCGGCACTAAGGAG-3′) with GAPDH (forward 5′-ACCACAGTCCATGCCATCAC-3′ and reverse 5′-TCCACCACCCTGTTGCTGTA-3′) as endogenous control. The relative expression levels were calculated with the 2−∆∆Ct method and experiments were repeated at least in three independent triplicates.
2.5. DNA Constructs and siRNA
For miR-129-5p overexpression miR-129 sequence was cloned into the pcDNA6.2-GW/EmGFP-miR plasmid using the BLOCK-iT Pol II miR RNAi Expression Vector Kit (Invitrogen). The following oligonucleotide sequences were used: miR-129-top, 5′-TGCTGCTTTTTGCGGTCTGGGCTTGCGTTTTGGCCAC TGACTGACGCAAGCCCAGAGCAAAAAG-3′ and miR-129-bottom, 5′-CCTGCTTTTTGCTCTGGGCTT GCGTCAGTCAGTGGCCAAAACGCAAGCCCAGACCGCAAAAAG-3′. The correct assembly of the vectors was verified by sequencing. pmiRZip-129-5p construct (Cat# MZIP129-5p-PA-1) for miR-129-5p inhibition and control vector pmiRZip-scr (Cat#MZIP000-PA-1) were obtained from System Biosciences (Mountain View, CA, USA). SOX4 siRNA was obtained from Qiagen (Hilden, Germany).
2.6. Transfections
Transfection to A375 and WM35 cells was performed with Lipofectamine 3000 reagent (Invitrogen) or Amaxa Cell line Nucleofector Kit V (LONZA) following the manufacturer’s instructions. Positive transfected cells were sorted by FACS, blasticidin (pCDNA6.2-constructs) or puromycin (miRZIP-constructs) selection.
2.7. Immunoblot Snalyses
Cells were lysed using a RIPA buffer for 30 min at 4 °C. Protein extracts were resolved by SDS–PAGE, blotted to nitrocellulose membranes and probed with the following antibodies: anti-GAPDH (Cat#2118), anti-EZH2 (Cat#5246), anti-H3 (Cat#4499), anti-H3K27me3 (Cat#9733), anti-ERK1/2 (Cat#4695) and anti-phosphoERK1/2 (Cat#4377) all from Cell Signaling Technology (Danvers, MA, USA) and anti-SOX4 (Cat#LS-C499849) from LSBio (Eching, Germany). For antibody detection we used anti-rabbit IgG-HRP (Cat#7074, Cell Signaling Technology, Danvers, MA, USA).
2.8. Cell Viability Assay
The number of viable cells was determined by CellTiter-Glo® Luminescent Cell Viability Assay (Promega). Cells (5000 per well) were seeded in 96-weell plates. After 24 h the cells were treated with 0.4 µM Vemurafenib or DMSO as control for at least 72 h. Cell viability was documented by TECAN plate reader following the manufacturer’s instructions.
2.9. Cell Growth Assay
For growth curves, 1 × 105 of respective cells were seeded in each well of a 6-well plate and cell numbers were determined over the time periods indicated by using a Neubauer counting chamber.
2.10. Cell Cycle Analysis
For cell cycle analysis, cells were trypsinized and fixed with 70% ice-cold ethanol for 30 min on 4 °C, washed twice with PBS and resuspended in 200 µL PI (from 50 µg/mL stock solution, abcam). Stained cells were analyzed on a BD FACS Scan cytometer using CellQuest software (BD Biosciences, Franklin Lakes, NJ, USA).
2.11. 3D Spheroid Growth Assay
For melanosphere formation, 5000 cells were seeded in a 3D culture-qualified 96-well spheroid formation plate (Cat#650970, Greiner). Spheroids were formed for 96 h before treatment. The growth of 3D spheroid cultures was assessed 96 h after seeding (0 h) and a following initial Vemurafenib treatment (1 µM) for 72 h, 96 h and 144 h. Melanospheres were photographed at indicated time points with a Keyence BZ-X810 fluorescence microscope and sphere areas were determined using ImageJ software.
2.12. Luciferase Reporter Assay
To confirm miR-129-5p binding to SOX4 3′UTR we used luciferase reporter clone for human SOX4 3′UTR (HmiT017630-MT06, Genecopoeia) and luciferase control reporter construct (CmiT000001-MT06, Genecopoeia). Constructs were transfected in A375 (ctrl) and A375 miR129-5p (overexpression) cells 24 h after seeding. The relative Luciferase activity was measured 48 h after transfection using Luc-Pair Duo-Luciferase Assay Kits 2.0 (Genecopoeia) following the manufacturer’s instructions.
2.13. Statistical Analyses
Statistical analyses were performed with GraphPad Prism. Student’s t-test was used for independent or paired samples to determine the statistical significance of experimental results. A p-value of 0.05 or less was considered significant. The results were represented as the average ± standard deviation from at least three independent experiments.
3. Results
3.1. Expression of miR-129-5p Increases during BRAFi and MEKi Treatment
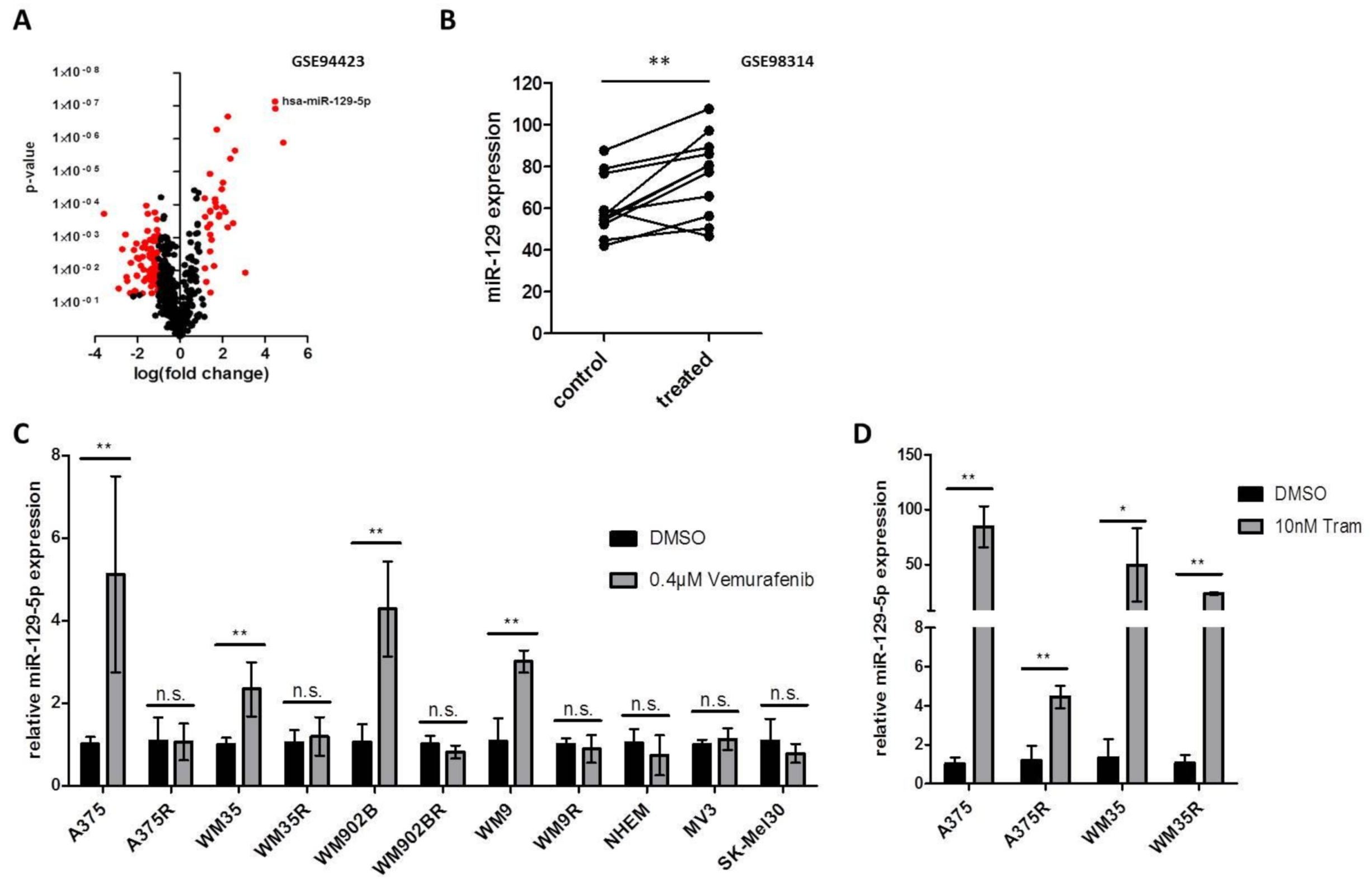
To identify miRNAs associated with response or resistance to BRAFi and MEKi treatment we reanalyzed a published next generation data set (GSE94423) [41]. To this end, we compared the miRNA expression of parental A375 cells treated with Vemurafenib to three different conditions: parental A375 cells treated with DMSO, a resistant clone of A375 cells (A375R) treated with Vemurafenib and A375R treated with DMSO. In our analysis we found 71 miRNAs upregulated (log2 fold change >1; p ≤ 0.05) and 137 miRNAs downregulated (log2 fold change <−1; p ≤ 0.05) in parental A375 cells treated with Vemurafenib (Figure 1A). One of the miRNAs which were induced the most after Vemurafenib treatment was miR-129-5p (Figure 1A). We could confirm our data analysis by virtue of a second published dataset (GSE98314) encompassing 11 BRAF mutated cell lines treated with BRAFi (Dabrafenib) or BRAFi/MEKi (Dabrafenib/Trametinib) and DMSO treated controls: when we performed a comparative analysis, we found that miR-129 expression is induced under BRAFi or BRAFi/MEKi in 10 out of 11 cell lines (Figure 1B).
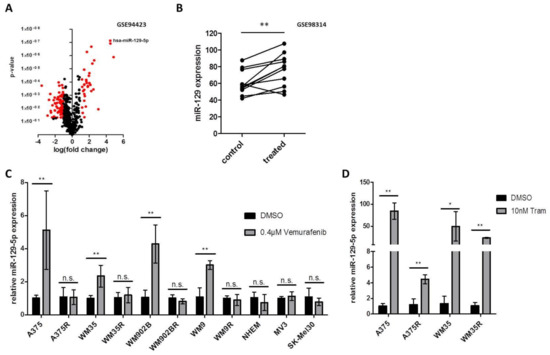
Figure 1.
miR-129-5p expression is increased during BRAFi and MEKi treatment. (A) Volcano plot of miRNAs differentially expressed in BRAFi treated A375 cells vs. a group of three different conditions: A375 treated with DMSO and a resistant clone of A375 cells (A375R) treated with Vemurafenib or DMSO (GSE94423). Red dots represent significant differentially expressed miRNAs (log2 fold change: ≥1; ≤−1; p ≤ 0.05). (B) Analyses of the miR-129 expression in eleven BRAF mutated melanoma cell lines treated with DMSO (control), Dabrafenib or a combination of Dabrafenib and Trametinib (treated). Expression data are shown as cubic spline function normalized values. (C) qRT-PCR analyses of miR-129-5p after BRAFi (0.4 µM Vemurafenib, 24 h) in BRAF mutation associated cell lines (A375, WM35, WM902B, WM9), BRAFi resistant cell lines (A375R, WM35R, WM902BR, WM9R), normal human epidermal melanocytes (NHEM) and BRAF wildtype melanoma cell lines (MV3, SK-MEL30). (D) qRT-PCR for miR-129-5p expression after MEK inhibition by 10 nM Trametinib for 48 h on parental sensitive (A375, WM35) and the corresponding resistant BRAF mutated cell lines (A375R, WM35R). Bars represent average ± standard deviation of at least three independent experiments. * p ≤ 0.05; ** p ≤ 0.01.
In order to confirm these in silico findings we performed qRT-PCR of BRAF mutated, treatment sensitive melanoma cell lines (A375, WM9, WM35 and WM902B) and found that miR-129-5p expression was increased after Vemurafenib treatment. The respective BRAF resistant cell lines (A375R, WM9R, WM35R and WM902BR (Supplementary Figure S1), as well as BRAF wildtype melanoma cell lines (MV3, SK-Mel30) and normal human epidermal melanocytes (NHEM) showed no significant change in miR-129-5p expression after Vemurafenib treatment (Figure 1C).
Further analyses demonstrated that expression of miR-129-5p was additionally increased during treatment with MEKi (Trametinib) (Figure 1D) also in BRAFi resistant cell lines (A375R, WM35R) (Figure 1D). Combinatory treatment with Vemurafenib and Trametinib also induces the expression of miR-129-5p (Supplementary Figure S2).
Taken together these results strongly suggest that miR-129-5p expression is mediated by BRAF/MEK pathway signaling.
3.2. miR-129-5p Expression Decreases During Emergence of Resistance to BRAFi
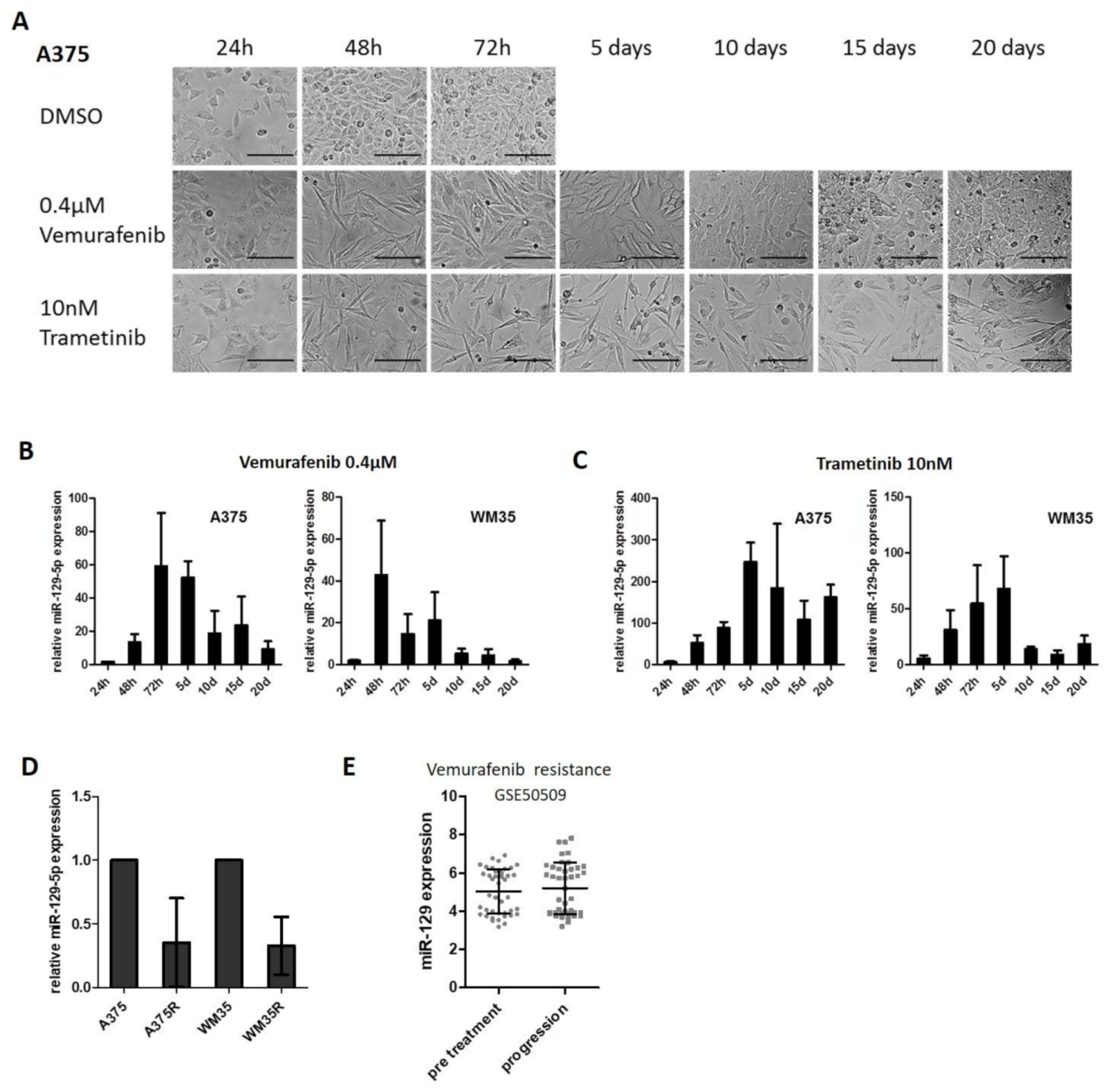
Having demonstrated that miR-129-5p expression is mediated by constitutive active BRAF/MEK signaling in BRAF mutated melanoma, we investigated its expression during long term BRAFi or MEKi treatment. Therefore, A375 and WM35 cell lines were treated with Vemurafenib or Trametinib for a periode of 20 days. Culture medium containing the inhibitors was exchanged every 48 h.
The treatment initially resulted in morphological changes of the melanoma cells: Initially both BRAFi and MEKi treated cells became spindle-shaped and ceased to proliferate, reflecting the antineplastic response. Then cells treated with BRAFi regained initial morphology and increased proliferation at days 10–15, indicating emerging resistance to treatment. Trametinib treated cells showed a prolonged treatment response (Figure 2A, Supplementary Figure S3). Next, we analyzed the expression of miR-129-5p at specific time points by qRT-PCR. Expression of miR-129-5p strongly increased in the first days during treatment with Vemurafenib and declined to almost the initial expression levels at day 20 (Figure 2B); this time course correlated with the morphological changes and the initially interrupted and finally resumed proliferation of the cells reflecting emergence of resistance.
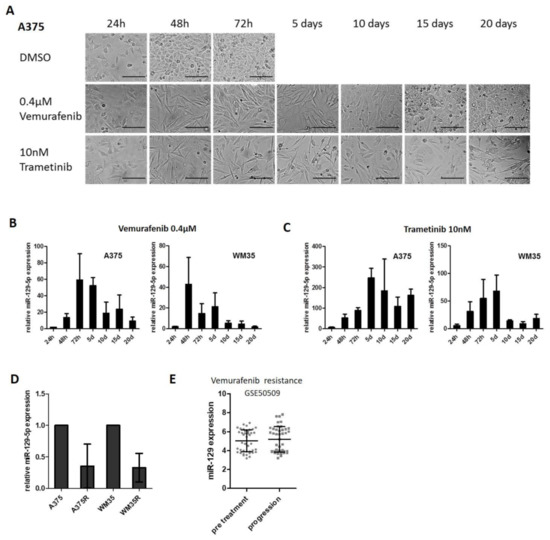
Figure 2.
miR-129-5p expression decreases during emergence of resistance to BRAFi. (A) A375 cells were treated with Vemurafenib or Trametinib every second day for a period of 20 days. Images were taken at indicated time points. Scale bars represent 200 µm. (B,C) Expression of miR-129-5p was analysed by qRT-PCR during permanent BRAF (B) or MEK (C) inhibition over 20 days in two BRAF mutated melanoma cell lines (A375 and WM35). (D) qRT-PCR expression analyses of miR-129-5p in A375/WM35 compared to the corresponding BRAFi resistant cell line (A375R/WM35R). (E) Analysis of miR-129-5p expression in the data set GSE50509 comparing untreated melanoma samples and samples at tumor progression during BRAFi treatment. Bars represent average ± standard deviation of at least three independent experiments.
Trametinib treatment delivered similar results, only that induction of miR-129-5p expression was stronger compared to BRAFi treatment and that the decrease of miR-129-5p expression did not reach the initial level by day 20 of treatment (Figure 2C). When we investigated Vemurafenib resistant cell lines (A375R, WM35R), we observed a reduced expression of miR-129-5p compared to parental cell lines (A375, WM35) (Figure 2D). We could evaluate this association of miR-129-5p levels with the response to Dabrafenib or Vemurafenib also found in the published data set (GSE50509) of melanoma tumor samples from patients before starting Dabrafenib or Vemurafenib and at the time of tumor progression: here, the levels of miR-129-5p in untreated melanoma samples were equal to samples of treatment resistant and progressive melanoma (Figure 2E). Our results demonstrate that miR-129-5p expression is induced in cells responsive to Vemurafenib or Trametinib treatment, but inhibited during treatment resistance.
3.3. EZH2 Suppresses miR-129-5p Expression Downstream of Constitutive Active BRAF Signaling
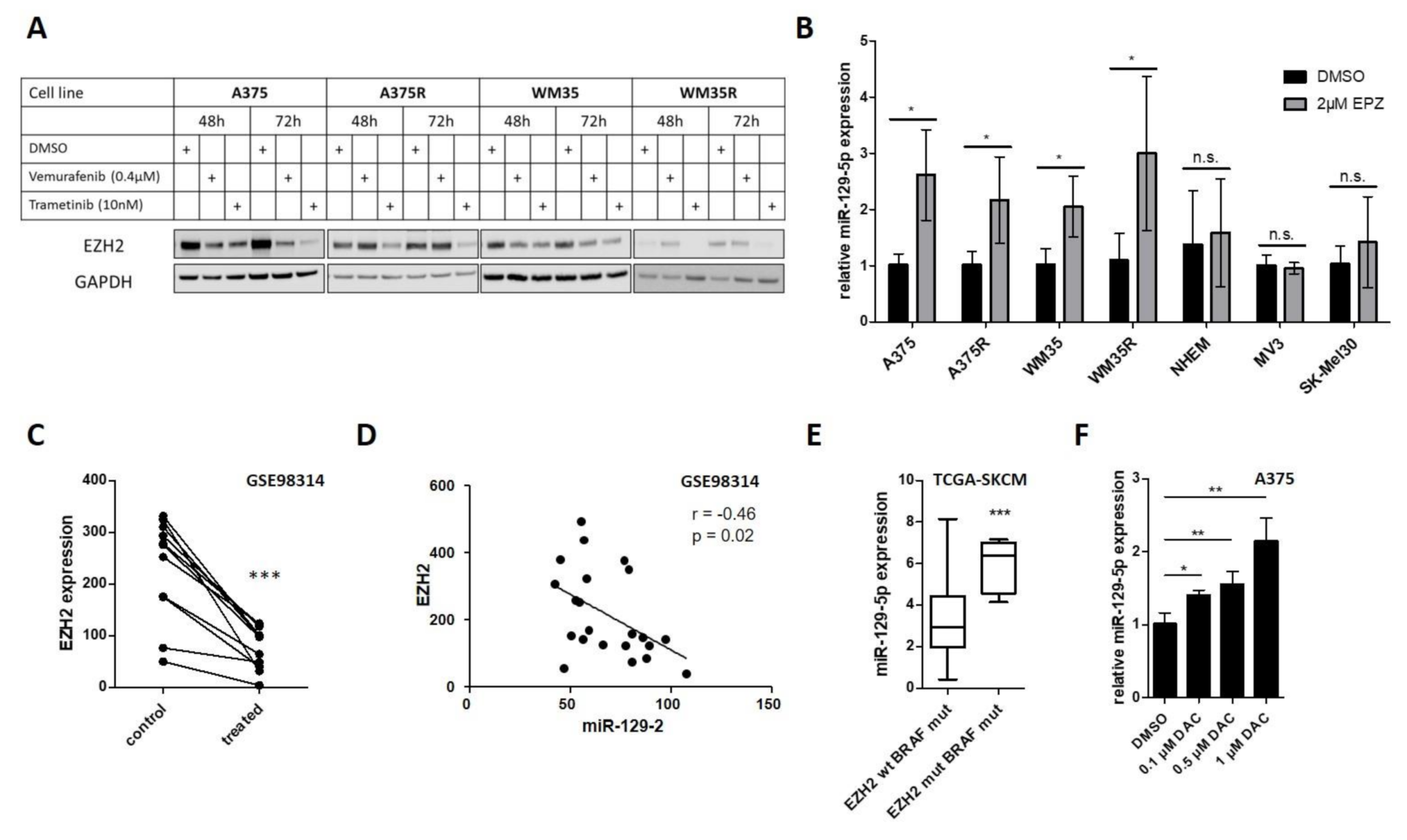
Since miR-129-5p expression in various cancer entities (e.g., endometrial cancer, breast cancer and gastric cancer) is mediated by EZH2 [42,43,44], an epigenetic modulator of H3K27me3 and DNA methylation [45,46], we investigated if EZH2 is mediated by BRAF/MEK pathway inhibition. Western blot analyses revealed that BRAFi (Vemurafenib), as well as MEKi (Trametinib), treatment decreased EZH2 protein levels in BRAF mutated cell lines (A375, WM35) (Figure 3A). We also demonstrated that even in BRAFi resistant cell lines (A375R, WM35R), MEK inhibition reduced EZH2 protein expression, while BRAF inhibition did not (Figure 3A). Consequently, we analyzed if inhibition of EZH2 induces miR-129-5p expression in BRAF mutated cell lines. Inhibition of EZH2 with the specific inhibitor EPZ-6438 (EPZ) significantly increased miR-129-5p expression in BRAF mutated melanoma cell lines (A375, WM35), even in BRAFi resistant cell lines (A375R, WM35R). In comparison, normal melanocytes (NHEM) and BRAF wildtype melanoma cell lines (MV3, SK-Mel30) displayed no induction of miR-129-5p expression (Figure 3B). Reanalysis of a published dataset (GSE98314) revealed that treatment with the BRAFi Dabrafenib decreased EZH2 expression in 11 different BRAF mutated melanoma cell lines (Figure 3C) and we found a significant inverse correlation of EZH2 and miR-129 expression (r = −0.46; p = 0.029) (Figure 3D). Additionally, the subset of the BRAF mutated melanoma cohort of the cancer genome atlas (TCGA) harboring an EZH2 silent mutation, showed a highly significant enforced miR-129-5p expression compared to BRAF mutated melanoma patients with EZH2 wildtype (Figure 3E).
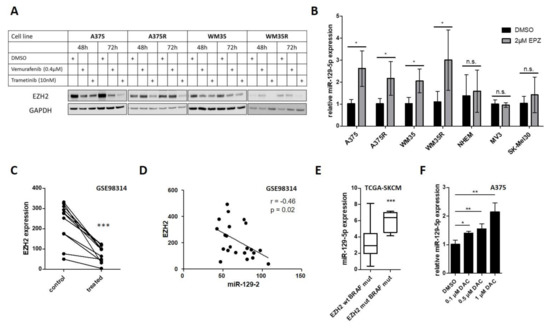
Figure 3.
miR-129-5p is repressed by BRAF/EZH2 pathway. (A) Western blot analyses of EZH2 in sensitive and resistant A375 and WM35 cells after BRAF and MEK inhibition. GAPDH immunoblot was used for normalization. (B) miR-129-5p expression of BRAF mutated cell lines (A375, WM35), corresponding BRAFi resistant cell lines (A375R, WM35R), normal human epidermal melanocytes (NHEM) and BRAF wildtype melanoma cell lines (MV3, SK-Mel30) after inhibition of EZH2 by EPZ (48 h). (C) Analysis of EZH2 expression in 11 BRAF mutated melanoma cell lines after BRAFi or BRAFi/MEKi compared to DMSO treated cells (control) (GSE98314). (D) Pearson correlation of EZH2 and miR-129-5p expression (GSE98314). Data are presented as cubic spline function normalized values. (E) Comparison of miR-129-5p expression in BRAF mutated melanoma patients with and without EZH2 mutation (TCGA). Values are presented as log2(RPM+1). (F) qRT-PCR analyses of miR-129-5p expression in A375 cells after DNA methylation inhibition by decitabine for 72 h. Bars represent average ± standard deviation of at least three independent experiments. n.s., not significant; * p ≤ 0.05; ** p ≤ 0.01; *** p ≤ 0.001.
To confirm that EZH2 is involved in transcriptional regulation of miR-129-5p, we analyzed H3K27me3 of Vemurafenib treated A375 and WM35 cells. Because we observed no changes in H3K27me3 after BRAFi treatment (Supplementary Figure S4), we investigated if miR-129-5p expression is controlled by DNA methylation, an epigenetic mechanism mediated by EZH2. Therefore, we analyzed miR-129-5p expression after DNA methylation inhibition by decetabine (DAC) and found a significant miR-129-5p induction in BRAF mutated A375 cells (Figure 3F).
Taken together these results indicate that EZH2 mediates miR-129-5p expression downstream of constitutive active BRAF signaling.
3.4. miR-129-5p Acts as Tumor Suppressor In Vitro and in a 3D Spheroid Model
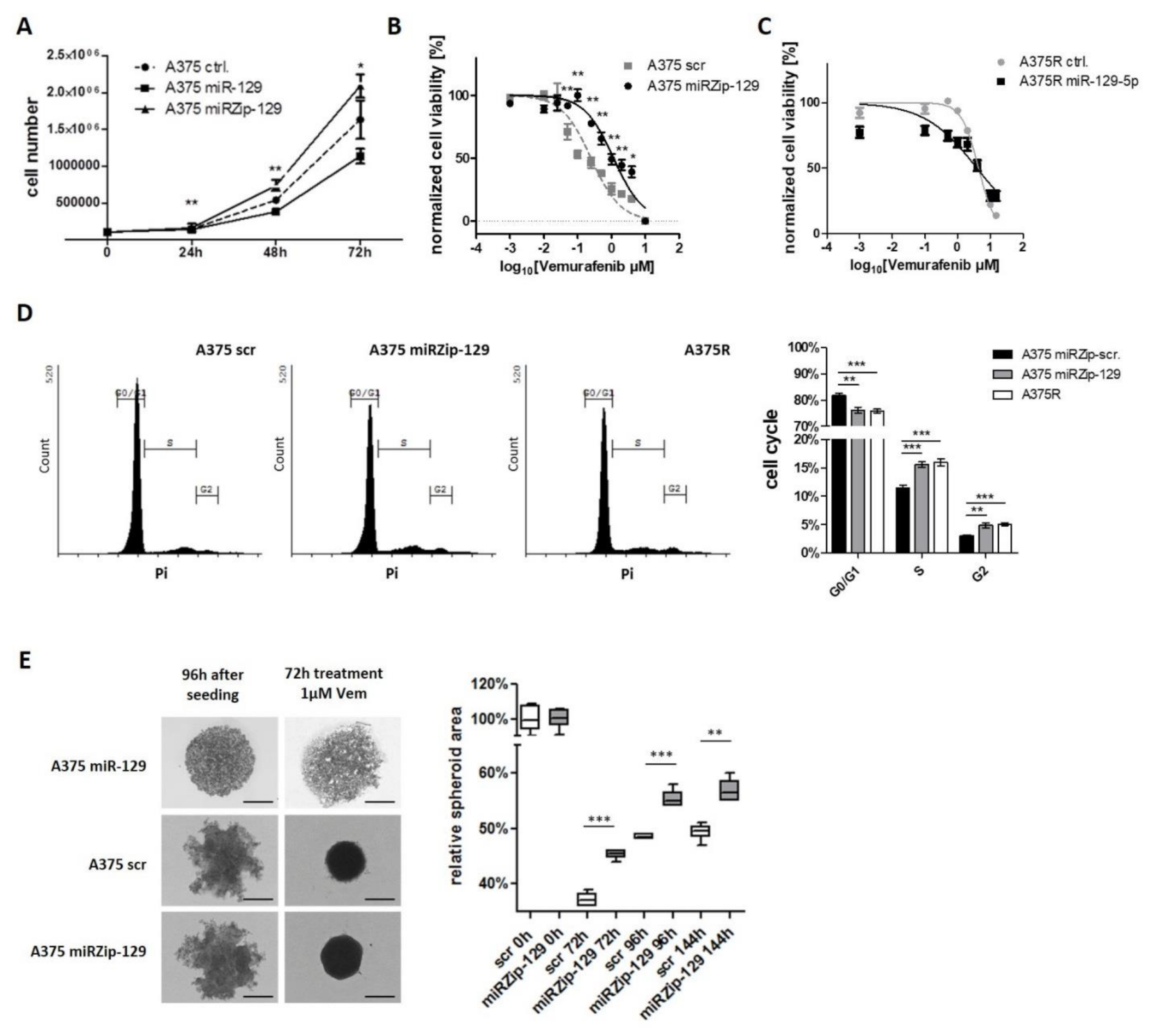
Having established that miR-129-5p is induced during BRAFi and MEKi response and mediated by EZH2 downstream of constitutive active BRAF signaling, we subsequently studied the biological function of miR-129-5p on proliferation of BRAF mutated melanoma cells: miR-129-5p overexpression reduced proliferation of melanoma cell line A375, whereas knockdown of miR-129-5p by a miRZip-129-5p construct significantly increased proliferation (Figure 4A). Since, miR-129-5p expression was upregulated during BRAFi or MEKi treatment in sensitive, but not in resistant cells, we wondered if regulation of miR-129-5p could mediate potentially therapeutic effects in terms of overcoming resistance. Therefore, we performed viability assays for A375 cells with either knockdown or overexpression of miR-129-5p during BRAFi treatment. MiR-129-5p knockdown increased viability of Vemurafenib treated A375 cells (A375 scr IC50: 0.25 µM vs. A375 miRZip-129 IC50: 1.15 µM) (Figure 4B), while its overexpression improved the treatment response of BRAFi resistant A375R cells (A375R scr IC50: 4.37 µM vs. A375R miR-129-5p IC50: 3.87 µM) (Figure 4C). In cell cycle analyses we found, that inhibition of miR-129-5p function through expression of a miRZip-129 construct in A375 cells increased the number of cells in the S/G2 phase during Vemurafenib treatment compared to A375 scr (Figure 4D). Interestingly, the cell cycle distribution of A375 miRZip-129 under BRAFi treatment cells was similar to A375R cells (Figure 4D). In contrast, untreated A375 miRZip-129 and scr cells showed an equivalent cell cycle that was different from A375R cells (Supplementary Figure S5A).
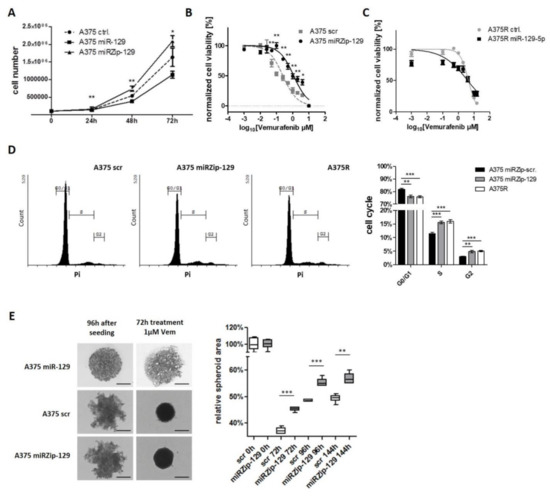
Figure 4.
miR-129-5p acts as a tumor suppressor. (A) Proliferation Assay of A375 cells overexpressing miR-129-5p (A375 miR-129) and knockdown miR-129-5p (A375 miRZip-129) compared to parental A375 (control). (B) BRAFi treatment response of miR-129-5p knockdown cells (A375 miRZip-129) and (C) BRAFi resistant cells overexpressing miR-129-5p (A375R miR-129) using cell viability assay. (D) Cell cycle distribution of sensitive parental A375 cells and their corresponding Vemurafenib resistant clone (A375R) compared to miR-129-5p knockdown cells (A375 miRZip-129). (E) Representative photographs of spheroid growth after initial Vemurafenib treatment (1 µM) were taken at indicated time points. Scale bar represents 500 µm. Spheroid area was measured of at least 5 spheroids using ImageJ software. Bars represent average of at least three independent experiments. * p ≤ 0.05; ** p ≤ 0.01; *** p ≤ 0.001.
To approach the question if such effects are relevant in situ we used a 3D spheroid model. While A375 scr control cells and miR-129-5p knockdown cells formed intermediate ragged spheroids, cells overexpressing miR-129-5p lost the ability to form spheroids and loosely accumulated (Figure 4E). When treated with Vemurafenib the spheroid size of A375 scr control cells and A375 miR-129-5p knockdown cells shrank and the surface became smoother. Spheroid area was reanalyzed 72 h, 96 h and 144 h after Vemurafenib treatment. A375 cells with miR-129-5p knockdown showed a significant stronger and faster spheroid growth compared to A375 control cells (Figure 4E; Supplementary Figure S5B,C).
Our results reveal miR-129-5p as a tumor suppressor in melanoma and that its repression attenuates BRAFi response.
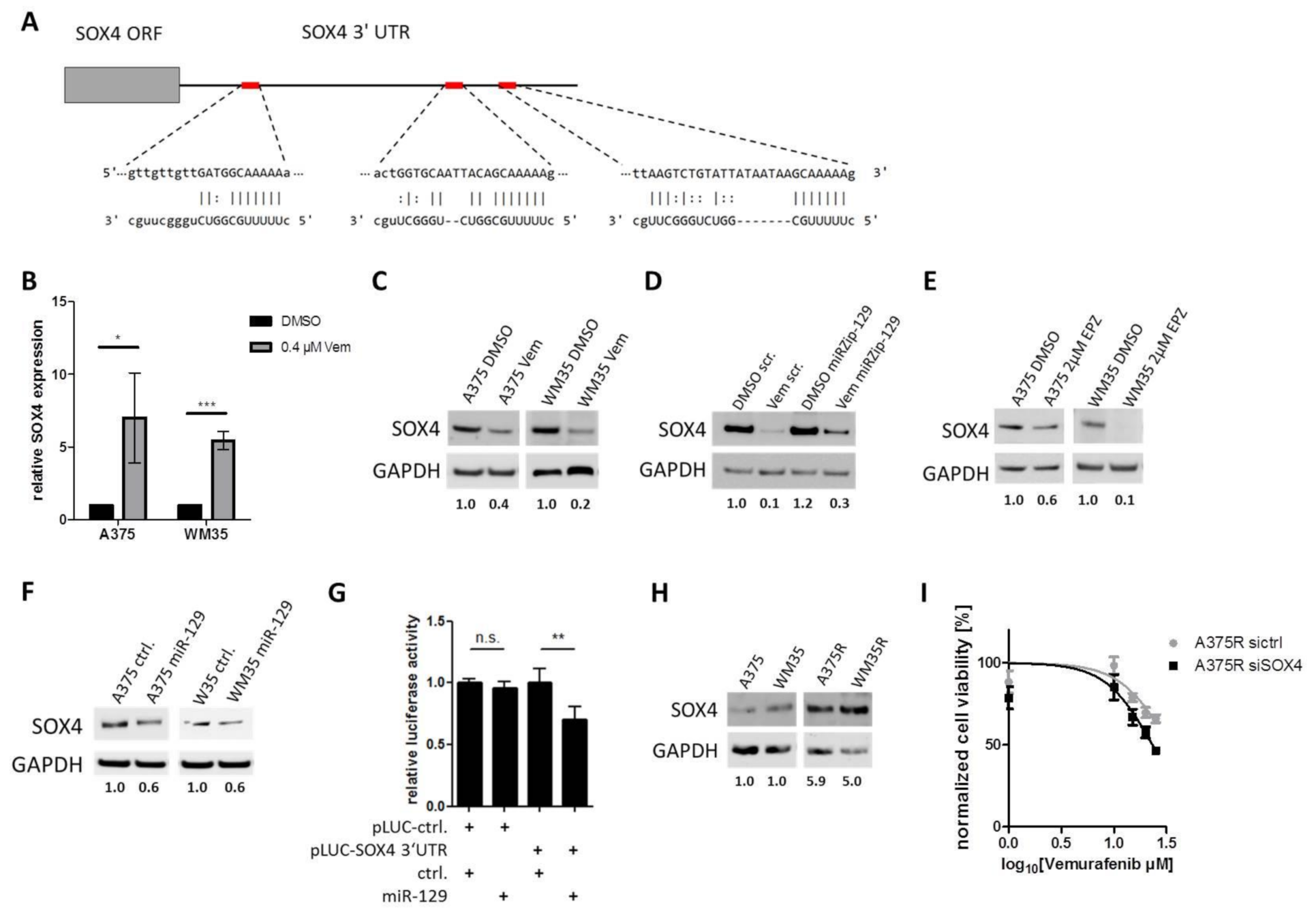
3.5. SOX4 is a Targeted by miR-129-5p During BRAFi Response
To understand the molecular function of miR-129-5p, we performed in silico analyses to predict potential targets mRNAs of genes, which were known to be involved in melanoma progression and drug resistance. One potential target of miR-129-5p is SOX4, which was reported to induce cell proliferation [47] and to mediate BRAF inhibitor resistance in melanoma [48]. SOX4 harbors three putative miR-129-5p binding sites in its mRNA 3′ UTR (Figure 5A). When we analyzed SOX4 expression after BRAF inhibitor treatment, we found an inverse correlation of SOX4 mRNA and SOX4 protein levels. SOX4 mRNA was significantly increased (Figure 5B), while the protein levels were decreased after BRAF inhibition in A375 and WM35 cell lines (Figure 5C). To verify that SOX4 protein decrease caused by Vemurafenib was mediated by induction of miR-129-5p, we transfected A375 cells with a miRZip-129 construct, to block miR-129-5p function. Treatment of those cells with Vemurafenib still decreased SOX4 protein, however blocking miR-129-5p through miRZip-129 partially attenuated SOX4 reduction caused by Vemurafenib (Figure 5D). Since EZH2 inhibition induced miR-129-5p expression, we hypothesize that EZH2 inhibition mediates SOX4 protein level. In support of this hypothesis we found that treatment of A375 and WM35 cells with EZH2 inhibitor resulted in decreased SOX4 protein levels (Figure 5E). To identify the function of miR-129-5p in post-transcriptional SOX4 regulation, we overexpressed miR-129-5p in A375 and WM35 cells. In Western blot analyses, we determined a decreased SOX4 protein level in miR-129-5p overexpressing melanoma cell lines (Figure 5F).
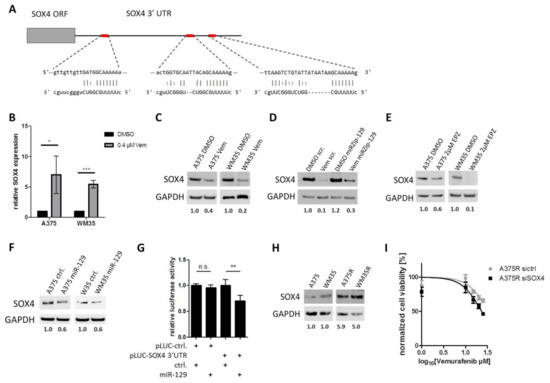
Figure 5.
miR-129-5p targets SOX4. (A) miR-129-5p and its three predicted binding sites at the 3’UTR in SOX4. (B) SOX4 mRNA expression and (C) protein expression after BRAFi treatment in A375 and WM35 cells. (D) SOX4 protein level was compared in BRAFi treated (Vem) or untreated (DMSO) A375 cells with miR-129-5p knockdown (miRZip-129) or scramble control (scr). (E) Western blot of SOX4 protein in A375 or WM35 cells after EZH2 inhibition by EPZ (48 h; 2 µM). (F) SOX4 protein levels in A375 and WM35 cells overexpressing miR-129-5p or scramble control. (G) Luciferase reporter assay was performed in A375 cells. Cells were co-transfected with SOX4 3′UTR luciferase reporter construct (pLuc-SOX4 3′UTR) or control (pLuc-ctrl.) and miR-129-5p expression construct (miR-129) or control (ctrl.) respectively. Luciferase activity was analysed after 24 h. (H) Western blot analyses of SOX4 in parental and corresponding BRAFi resistant A375 and WM35 cells. (I) Viability assay of BRAFi treated A375R cells after SOX4 knockdown by siRNA or control. Cells were treated for 96 h. Bars represent average of at least three independent experiments. * p ≤ 0.05; ** p ≤ 0.01; *** p ≤ 0.001.
To prove the direct binding of miR-129-5p to the SOX4 3′UTR we performed luciferase reporter assays in A375 cells. Luciferase activity decreased after miR-129-5p overexpression (Figure 5G), which indicates the direct binding of miR-129-5p to the SOX4 3′UTR.
To validate that SOX4 influences emergence of BRAFi resistance, we measured SOX4 protein levels in BRAFi sensitive (A375, WM35) and resistant (A375R, WM35R) cells. In western blot analyses BRAFi resistant cells showed an increased SOX4 protein expression compared to the parental cells (Figure 5H). Furthermore, SOX4 knockdown by siRNA resulted in an improved BRAF inhibitor response of BRAFi resistant cells (A375R sictrl IC50: 33.8 µM vs. A375R siSOX4 IC50: 23.2 µM) (Figure 5I).
Thus, these results demonstrate that SOX4 is a direct target of miR-129-5p during melanoma response to BRAFi and they indicate that SOX4 mediates resistance to BRAFi treatment.
4. Discussion
We showed by both in silico and in vitro analyses that miR-129-5p is induced by BRAFi or MEKi treatment exclusively in melanoma cell lines with BRAF mutations, and not in primary normal human epidermal melanocytes (NHEMs), BRAF wildtype melanoma cells or BRAFi resistant melanoma cells. This BRAFi-mediated increase of miR-129-5p expression is then dynamically reduced with the emergence of resistance. Mechanistically, we were able to show that the miR-129-5p repression was mediated by EZH2, a downstream effector of BRAF. Our study provides evidences that miR-129-5p acts as a tumor suppressor improving the response to BRAFi and inhibiting the proliferation of melanoma cells by targeting SOX4. Due to these results we conclude that miR-129-5p is an important molecular regulator of response to BRAFi, which is repressed by downstream signaling pathways of constitutively active BRAF in melanoma.
The expression of several miRNAs in melanoma is altered by BRAFi and MEKi treatment [41,49,50] and reprogramming of miRNA expression is involved in the emergence of drug resistance in BRAF mutated melanoma [51]. Thus far, the miR-204, miR-211, as well as miR-410-3p, were shown to be induced by BRAF or MEK inhibitor and to contribute to resistance to BRAF inhibitor in melanoma by enhancing the activity of downstream pathways, such as MEK or ERK [39,40,50]. In contrast to these three miRNAs, miR-129-5p, was not only induced during response to BRAFi or MEKi treatment, but also decreased when the cells developed resistance. In addition without treatment miR-129-5p expression was reduced in BRAFi resistant cell lines compared to parental cells. These results were also supported by our in silico findings, that miR-129 has comparable expression levels in samples of progressive melanomas both prior to treatment with Dabrafenib or Vemurafenib and at time of tumor progression (GSE50509 dataset). Our results thus reveal that miR-129-5p expression correlates strongly with the response to BRAFi/MEKi in melanoma.
Previous studies showed that miR-129 is downregulated in melanoma tissues [52] and several different cancer entities compared to normal tissues [43,53,54,55,56,57]. In breast cancer [43], endometrial cancer [42] and gastric cancer [44] repression was mediated by epigenetically modifications of H3K27 trimethylation or DNA methylation. Both processes are regulated by the epigenetic modifier EZH2 [45,46]. EZH2 expression is associated with high proliferation rates and aggressive tumor subgroups of cutaneous melanoma [58]. It controls melanoma growth and metastasis through silencing of distinct tumor suppressors [59,60]. Of note EZH2 was also shown as a mediator of treatment resistance in BRAF mutated melanoma [61,62]. We have now demonstrated that, in melanoma, EZH2 represses miR-129-5p, dependent on constitutive active BRAF signaling. After treatment of BRAF mutated melanoma cells with BRAFi or MEKi, we observed a reduction of EZH2 protein as well as mRNA, as shown previously for melanoma [62,63]. Additionally, specific inhibition of EZH2 induces miR-129-5p expression in BRAF mutated melanoma cell lines, independent from their BRAFi response status, while BRAF wildtype cells and normal melanocytes show no changes of expression. In silico, miR-129 and EZH2 expression during treatment correlated inversely with administration of BRAF inhibitor (GSE98314). This correlation could not be determined in the melanoma cohort of the cancer genome atlas (TCGA, https://www.cancer.gov/tcga, accessed on 22 April 2021), because these patients had not received BRAFi or MEKi. However, miR-129-5p expression was elevated in BRAF mutated melanoma patients harboring EZH2 missense or silencing mutations compared to wildtype EZH2. This provides further evidence that, in melanoma, EZH2 mediates miR-129-5p expression downstream of constitutive active BRAF signaling. Although previous studies showed a reduction of H3K27me3 during treatment with BRAFi or MEKi in melanoma [62,63] we could not confirm these changes in our experiments. These different results may be explained by using low doses of treatment (0.4 µM Vemurafenib or 10 nM Trametinib) or shorter treatment times (48 h). Treatment with the cytosine analogue Cytarabine (AraC), which inhibits DNA methylation, a process also mediated by EZH2 [45,59], induces miR-129-5p expression. Our results and the previously described repression of miR-129 via EZH2 in different entities by DNA methylation or histone modification [43,64] let us conclude, that in BRAF mutated melanoma suppression of miR-129-5p is mediated via EZH2 downstream of constitutive active BRAF signaling.
Our functional analyses demonstrate that miR-129-5p inhibits cell proliferation, cell cycle progression and that it mediates BRAFi response in BRAF mutated melanoma. This is in line with previous in vitro studies reporting that miR-129-5p acts as tumor suppressor in lung cancer [65], gastrointestinal cancer [55,57,66,67], esophageal squamous cell carcinoma [68,69], hepatocellular cancer [70], cervical cancer [71], breast cancer [43,72], and glioblastoma [73].
We reveal that miR-129-5p overexpression improves response to BRAFi of resistant melanoma cells, while knockdown of miR-129-5p in parental cells shows the opposite effect. These results are supported by our finding that miR-129-5p expression is decreased during emergence of resistance. While other studies also found that repression of miR-129-5p modulates resistance to multiple drugs (e.g., chemotherapeutic, such as 5-Fluorouracil, Gemcitabine and Adriamycine, as well as antibody therapy, such as Trastuzumab) in several cancers, e.g., gastric cancer [44], breast cancer [43,74,75], ovarian cancer [76], and bladder cancer [77], our study, for the first time, reports on miR-129-5p as mediator of BRAFi response in melanoma.
Having elaborated that miR-129-5p functions as tumor suppressor and mediates BRAF inhibitor treatment response, we investigated potential target genes, known to be involved in melanoma progression and drug resistance. SOX4 seemed a likely fitting candidate, since it is not only induced during melanoma progression and promotes melanoma proliferation by AKT signaling activation [47], but also mediates to BRAFi in melanoma through regulation of IGF-1R [48]. Our results reveal that BRAFi treatment induces SOX4 mRNA, but reduces SOX4 protein, which implicates a posttranscriptional regulation. By a luciferase reporter assay, we confirmed SOX4 as direct target of miR-129-5p. SOX4 was also observed to be a target of miR-129-5p in breast cancer [43], esophageal carcinoma [69], chondrosarcoma [54] or cervical cancer [71], in which miR-129-5p mediated repression of SOX4 was associated with reduced cancer cell progression.
We then demonstrated that knockdown of SOX4 by applying a siRNA resulted in an improved response of BRAFi resistant cells towards Vemurafenib treatment, comparable to miR-129-5p overexpression. This result indicates that miRNA-mediated mechanisms of resistance could indeed be amenable to therapeutic modifications, e.g., by agomiRs or mimics. However, tissue distribution and targeted cell delivery is still an obstacle for the systemic therapeutic approach [31,32].
Additional putative targets of miR-129-5p that are associated with drug resistance in cancers are ABC transporters (ABCB1, ABCC5, ABCG1) [44] and RUNX1 [78]. For RUNX1 we could not prove a posttranscriptional regulation by miR-129-5p in BRAF associated melanoma (data not shown). Further research should investigate if ABC transporters act as additional targets of miR-129-5p in the context of BRAF/MEK inhibitor resistance in melanoma.
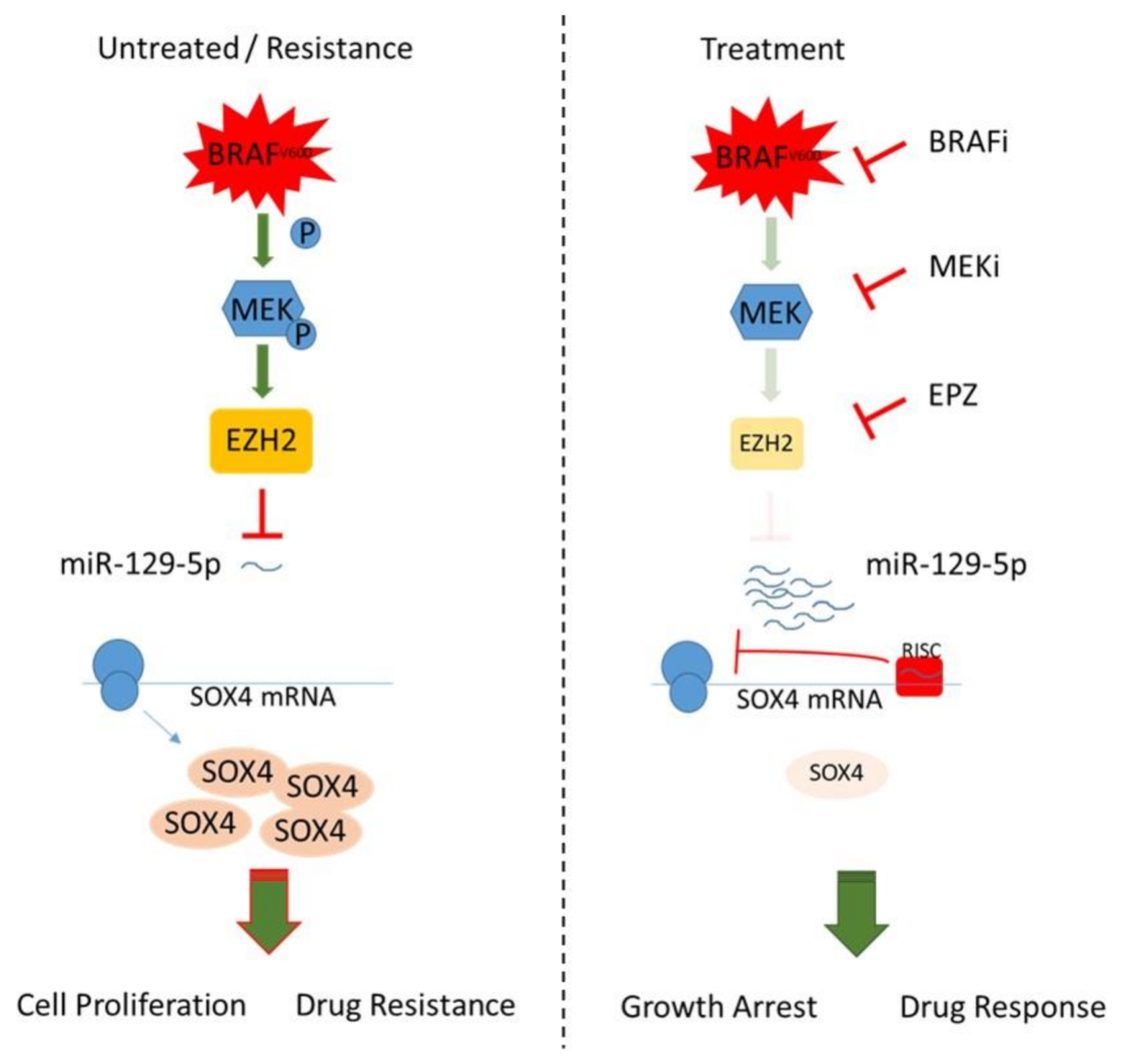
Since SOX4 controls EZH2 expression by direct promotor binding [79] and SOX4/EZH2 are shown to interact as co-repressors on tumor suppressive miR-31 in invasive esophageal cancer cells [80], we hypothesize a regulatory network in BRAF mutated melanoma, in which constitutively activated BRAF signaling induces SOX4 and EZH2 expression, resulting in miR-129-5p repression (Figure 6). During BRAF/MEK inhibition, EZH2 is reduced, which releases the epigenetic repression of miR-129-5p. The elevated miR-129-5p inhibits SOX4 protein translation, resulting in a reduction of proliferation and improved treatment response (Figure 6). The emergence of resistance by reactivation of BRAF downstream or bypass pathways reinforces EZH2 resulting in repression of miR-129-5p.
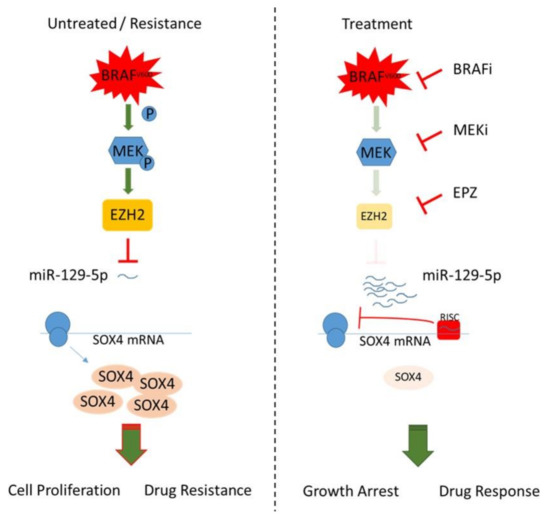
Figure 6.
BRAF/EZH2 signaling represses miR-129-5p inhibition of SOX4 thereby modulating BRAFi resistance in melanoma.
Taken together our results emphasize SOX4 as a potential therapeutic target in BRAF driven melanoma which could be attacked by pharmaceutically, e.g., by miR-129-5p mimics.
5. Conclusions
Targeted therapies, e.g., BRAFi improved patient outcomes in BRAF mutated melanoma. Emergence of resistance to these therapies is an obstacle urgent to overcome. To this end, we wanted to investigate whether miRNAs enhance response to BRAF inhibition. In this study we identified the tumor suppressor miR-129-5p to be induced during BRAF inhibition. Finally, we found that miR-129-5p decreases melanoma cell proliferation and improves response to BRAFi by targeting SOX4.
Taken together our results emphasize miR-129-5p, as well as SOX4, as potential therapeutic targets in BRAF-driven melanoma.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/cancers13102393/s1, Supplemental Figure S1. Establishment of Vemurafenib resistant cell lines; Supplemental Figure S2. miR-129-5p expression is increased after BRAFi/MEKi combinatory treatment; Supplemental Figure S3. Cell morphology of WM35 during long term BRAFi and MEKi treatment; Supplemental Figure S4. H3K27me3 after BRAFi and MEKi; Supplemental Figure S5. miR-129-5p acts as tumor suppressor. Supplemental Materials: original western blot pictures.
Author Contributions
K.G., C.S. and D.G. designed the research. K.G., D.G. and E.G. performed the experiments and analyzed the data. B.E. supported the experiments with resources. D.G., K.G. and C.S. wrote the manuscript. L.N., S.K.-H., R.K.C.M., B.E. and E.G. commented on the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
We acknowledge the financial support within the funding program Open Access Publishing by the German Research Foundation (DFG).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in this article and supplementary material.
Acknowledgments
We would like to thank Anja Sobisch, Sylke Fasshauer and Claudia Bruhne for excellent technical assistance.
Conflicts of Interest
The authors declare they have no conflict of interests.
References
- Markovic, S.N.; Erickson, L.A.; Flotte, T.J.; Kottschade, L.A.; McWilliams, R.R.; Jakub, J.W.; Farley, D.R.; Tran, N.V.; Schild, S.E.; Olivier, K.R.; et al. Metastatic malignant melanoma. Gancer Ital. Derm. Venereol. 2009, 144, 1–26. [Google Scholar]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in globocan 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Sini, M.C.; Doneddu, V.; Paliogiannis, P.; Casula, M.; Colombino, M.; Manca, A.; Botti, G.; Ascierto, P.A.; Lissia, A.; Cossu, A.; et al. Genetic alterations in main candidate genes during melanoma progression. Oncotarget 2018, 9, 8531–8541. [Google Scholar] [CrossRef]
- Hayward, N.K.; Wilmott, J.S.; Waddell, N.; Johansson, P.A.; Field, M.A.; Nones, K.; Patch, A.M.; Kakavand, H.; Alexandrov, L.B.; Burke, H.; et al. Whole-genome landscapes of major melanoma subtypes. Nature 2017, 545, 175–180. [Google Scholar] [CrossRef]
- Rabbie, R.; Ferguson, P.; Molina-Aguilar, C.; Adams, D.J.; Robles-Espinoza, C.D. Melanoma subtypes: Genomic profiles, prognostic molecular markers and therapeutic possibilities. J. Pathol. 2019, 247, 539–551. [Google Scholar] [CrossRef]
- Garman, B.; Anastopoulos, I.N.; Krepler, C.; Brafford, P.; Sproesser, K.; Jiang, Y.; Wubbenhorst, B.; Amaravadi, R.; Bennett, J.; Beqiri, M.; et al. Genetic and genomic characterization of 462 melanoma patient-derived xenografts, tumor biopsies, and cell lines. Cell Rep. 2017, 21, 1936–1952. [Google Scholar] [CrossRef]
- Hodis, E.; Watson, I.R.; Kryukov, G.V.; Arold, S.T.; Imielinski, M.; Theurillat, J.P.; Nickerson, E.; Auclair, D.; Li, L.; Place, C.; et al. A landscape of driver mutations in melanoma. Cell 2012, 150, 251–263. [Google Scholar] [CrossRef]
- Sullivan, R.J.; Flaherty, K.T. Resistance to braf-targeted therapy in melanoma. Eur. J. Cancer 2013, 49, 1297–1304. [Google Scholar] [CrossRef]
- Larkin, J.; Ascierto, P.A.; Dreno, B.; Atkinson, V.; Liszkay, G.; Maio, M.; Mandala, M.; Demidov, L.; Stroyakovskiy, D.; Thomas, L.; et al. Combined vemurafenib and cobimetinib in braf-mutated melanoma. N. Engl. J. Med. 2014, 371, 1867–1876. [Google Scholar] [CrossRef]
- Robert, C.; Karaszewska, B.; Schachter, J.; Rutkowski, P.; Mackiewicz, A.; Stroiakovski, D.; Lichinitser, M.; Dummer, R.; Grange, F.; Mortier, L.; et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N. Engl. J. Med. 2015, 372, 30–39. [Google Scholar] [CrossRef]
- Long, G.V.; Weber, J.S.; Infante, J.R.; Kim, K.B.; Daud, A.; Gonzalez, R.; Sosman, J.A.; Hamid, O.; Schuchter, L.; Cebon, J.; et al. Overall survival and durable responses in patients with braf v600 mutant metastatic melanoma receiving dabrafenib combined with trametinib (vol 34, pg 871, 2016). J. Clin. Oncol. 2019, 37, 355-355. [Google Scholar]
- Chapman, P.B.; Hauschild, A.; Robert, C.; Haanen, J.B.; Ascierto, P.; Larkin, J.; Dummer, R.; Garbe, C.; Testori, A.; Maio, M.; et al. Improved survival with vemurafenib in melanoma with braf v600e mutation. N. Engl. J. Med. 2011, 364, 2507–2516. [Google Scholar] [CrossRef]
- Nazarian, R.; Shi, H.B.; Wang, Q.; Kong, X.J.; Koya, R.C.; Lee, H.; Chen, Z.G.; Lee, M.K.; Attar, N.; Sazegar, H.; et al. Melanomas acquire resistance tob-raf(v600e) inhibition by rtk or n-ras upregulation. Nature 2010, 468, 973–977. [Google Scholar] [CrossRef]
- Poulikakos, P.I.; Persaud, Y.; Janakiraman, M.; Kong, X.J.; Ng, C.; Moriceau, G.; Shi, H.B.; Atefi, M.; Titz, B.; Gabay, M.T.; et al. Raf inhibitor resistance is mediated by dimerization of aberrantly spliced braf(v600e). Nature 2011, 480, 387–390. [Google Scholar] [CrossRef]
- Shi, H.; Moriceau, G.; Kong, X.; Lee, M.K.; Lee, H.; Koya, R.C.; Ng, C.; Chodon, T.; Scolyer, R.A.; Dahlman, K.B.; et al. Melanoma whole-exome sequencing identifies (v600e)b-raf amplification-mediated acquired b-raf inhibitor resistance. Nat. Commun. 2012, 3, 724. [Google Scholar] [CrossRef]
- Johannessen, C.M.; Boehm, J.S.; Kim, S.Y.; Thomas, S.R.; Wardwell, L.; Johnson, L.A.; Emery, C.M.; Stransky, N.; Cogdill, A.P.; Barretina, J.; et al. Cot drives resistance to raf inhibition through map kinase pathway reactivation. Nature 2010, 468, 968–972. [Google Scholar] [CrossRef]
- Emery, C.M.; Vijayendran, K.G.; Zipser, M.C.; Sawyer, A.M.; Niu, L.; Kim, J.J.; Hatton, C.; Chopra, R.; Oberholzer, P.A.; Karpova, M.B.; et al. Mek1 mutations confer resistance to mek and b-raf inhibition. Proc. Natl. Acad. Sci. USA 2009, 106, 20411–20416. [Google Scholar] [CrossRef]
- Lito, P.; Rosen, N.; Solit, D.B. Tumor adaptation and resistance to raf inhibitors. Nat. Med. 2013, 19, 1401–1409. [Google Scholar] [CrossRef]
- Kun, E.; Tsang, Y.T.M.; Ng, C.W.; Gershenson, D.M.; Wong, K.K. Mek inhibitor resistance mechanisms and recent developments in combination trials. Cancer Treat. Rev. 2021, 92, 102137. [Google Scholar] [CrossRef]
- Johnson, D.B.; Menzies, A.M.; Zimmer, L.; Eroglu, Z.; Ye, F.; Zhao, S.; Rizos, H.; Sucker, A.; Scolyer, R.A.; Gutzmer, R.; et al. Acquired braf inhibitor resistance: A multicenter meta-analysis of the spectrum and frequencies, clinical behaviour, and phenotypic associations of resistance mechanisms. Eur. J. Cancer 2015, 51, 2792–2799. [Google Scholar] [CrossRef] [PubMed]
- Rizos, H.; Menzies, A.M.; Pupo, G.M.; Carlino, M.S.; Fung, C.; Hyman, J.; Haydu, L.E.; Mijatov, B.; Becker, T.M.; Boyd, S.C.; et al. Braf inhibitor resistance mechanisms in metastatic melanoma: Spectrum and clinical impact. Clin. Cancer Res. 2014, 20, 1965–1977. [Google Scholar] [CrossRef] [PubMed]
- Varrone, F.; Caputo, E. The mirnas role in melanoma and in its resistance to therapy. Int. J. Mol. Sci. 2020, 21, 878. [Google Scholar] [CrossRef]
- Bartel, D.P. Micrornas: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Ambros, V. The functions of animal micrornas. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Krakowsky, R.H.E.; Wurm, A.A.; Gerloff, D.; Katzerke, C.; Brauer-Hartmann, D.; Hartmann, J.U.; Wilke, F.; Thiede, C.; Muller-Tidow, C.; Niederwieser, D.; et al. Mir-451a abrogates treatment resistance in flt3-itd-positive acute myeloid leukemia. Blood Cancer J. 2018, 8, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.W.; Zhao, W.J.; Zhu, Y.G.; Zhao, X.D. Mir-185 inhibits tumor growth and enhances chemo-resistance via targeting sry-related high mobility group box transcription factor 13 in non-small-cell carcinoma. Am. J. Transl. Res. 2018, 10, 2600. [Google Scholar]
- Gao, Y.W.; Liu, Z.Y.; Ding, Z.H.; Hou, S.C.; Li, J.; Jiang, K.H. Microrna-155 increases colon cancer chemoresistance to cisplatin by targeting forkhead box o3. Oncol. Lett. 2018, 15, 4781–4788. [Google Scholar] [CrossRef]
- Wurm, A.A.; Zjablovskaja, P.; Kardosova, M.; Gerloff, D.; Brauer-Hartmann, D.; Katzerke, C.; Hartmann, J.U.; Benoukraf, T.; Fricke, S.; Hilger, N.; et al. Disruption of the c/ebpalpha-mir-182 balance impairs granulocytic differentiation. Nat. Commun. 2017, 8, 46. [Google Scholar] [CrossRef]
- Gerloff, D.; Grundler, R.; Wurm, A.A.; Brauer-Hartmann, D.; Katzerke, C.; Hartmann, J.U.; Madan, V.; Muller-Tidow, C.; Duyster, J.; Tenen, D.G.; et al. Nf-kappab/stat5/mir-155 network targets pu.1 in flt3-itd-driven acute myeloid leukemia. Leukemia 2015, 29, 535–547. [Google Scholar] [CrossRef]
- Medarova, Z.; Pantazopoulos, P.; Yoo, B. Screening of potential mirna therapeutics for the prevention of multi-drug resistance in cancer cells. Sci. Rep. 2020, 10, 1970. [Google Scholar] [CrossRef]
- Linck-Paulus, L.; Hellerbrand, C.; Bosserhoff, A.K.; Dietrich, P. Dissimilar appearances are deceptive-common micrornas and therapeutic strategies in liver cancer and melanoma. Cells 2020, 9, 114. [Google Scholar] [CrossRef]
- Gerloff, D.; Sunderkotter, C.; Wohlrab, J. Importance of micrornas in skin oncogenesis and their suitability as agents and targets for topical therapy. Ski. Pharm. Physiol. 2020, 33, 270–279. [Google Scholar] [CrossRef]
- Janssen, H.L.; Reesink, H.W.; Lawitz, E.J.; Zeuzem, S.; Rodriguez-Torres, M.; Patel, K.; van der Meer, A.J.; Patick, A.K.; Chen, A.; Zhou, Y.; et al. Treatment of hcv infection by targeting microrna. N. Engl. J. Med. 2013, 368, 1685–1694. [Google Scholar] [CrossRef]
- Sun, X.; Li, J.; Sun, Y.; Zhang, Y.; Dong, L.; Shen, C.; Yang, L.; Yang, M.; Li, Y.; Shen, G.; et al. Mir-7 reverses the resistance to brafi in melanoma by targeting egfr/igf-1r/craf and inhibiting the mapk and pi3k/akt signaling pathways. Oncotarget 2016, 7, 53558–53570. [Google Scholar] [CrossRef]
- Caporali, S.; Amaro, A.; Levati, L.; Alvino, E.; Lacal, P.M.; Mastroeni, S.; Ruffini, F.; Bonmassar, L.; Antonini Cappellini, G.C.; Felli, N.; et al. Mir-126-3p down-regulation contributes to dabrafenib acquired resistance in melanoma by up-regulating adam9 and vegf-a. J. Exp. Clin. Cancer Res. Cr. 2019, 38, 272. [Google Scholar] [CrossRef]
- Fattore, L.; Mancini, R.; Acunzo, M.; Romano, G.; Lagana, A.; Pisanu, M.E.; Malpicci, D.; Madonna, G.; Mallardo, D.; Capone, M.; et al. Mir-579-3p controls melanoma progression and resistance to target therapy. Proc. Natl. Acad. Sci. USA 2016, 113, E5005–E5013. [Google Scholar] [CrossRef]
- Vergani, E.; Di Guardo, L.; Dugo, M.; Rigoletto, S.; Tragni, G.; Ruggeri, R.; Perrone, F.; Tamborini, E.; Gloghini, A.; Arienti, F.; et al. Overcoming melanoma resistance to vemurafenib by targeting ccl2-induced mir-34a, mir-100 and mir-125b. Oncotarget 2016, 7, 4428–4441. [Google Scholar] [CrossRef]
- Diaz-Martinez, M.; Benito-Jardon, L.; Alonso, L.; Koetz-Ploch, L.; Hernando, E.; Teixido, J. Mir-204-5p and mir-211-5p contribute to braf inhibitor resistance in melanoma. Cancer Res. 2018, 78, 1017–1030. [Google Scholar] [CrossRef]
- Lee, B.; Sahoo, A.; Sawada, J.; Marchica, J.; Sahoo, S.; Layng, F.; Finlay, D.; Mazar, J.; Joshi, P.; Komatsu, M.; et al. Microrna-211 modulates the dusp6-erk5 signaling axis to promote braf(v600e)-driven melanoma growth in vivo and braf/mek inhibitor resistance. J. Investig. Derm. 2020, 141, 385–394. [Google Scholar] [CrossRef]
- Vitiello, M.; Tuccoli, A.; D’Aurizio, R.; Sarti, S.; Giannecchini, L.; Lubrano, S.; Marranci, A.; Evangelista, M.; Peppicelli, S.; Ippolito, C.; et al. Context-dependent mir-204 and mir-211 affect the biological properties of amelanotic and melanotic melanoma cells. Oncotarget 2017, 8, 25395–25417. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.W.; Liu, J.C.; Deatherage, D.E.; Luo, J.Q.; Mutch, D.G.; Goodfellow, P.J.; Miller, D.S.; Huang, T.H.M. Epigenetic repression of microrna-129-2 leads to overexpression of sox4 oncogene in endometrial cancer. Cancer Res. 2009, 69, 9038–9046. [Google Scholar] [CrossRef] [PubMed]
- Luan, Q.X.; Zhang, B.G.; Li, X.J.; Guo, M.Y. Mir-129-5p is downregulated in breast cancer cells partly due to promoter h3k27m3 modification and regulates epithelial-mesenchymal transition and multi-drug resistance. Eur. Rev. Med. Pharm. 2016, 20, 4257–4265. [Google Scholar]
- Wu, Q.; Yang, Z.; Xia, L.; Nie, Y.; Wu, K.; Shi, Y.; Fan, D. Methylation of mir-129-5p cpg island modulates multi-drug resistance in gastric cancer by targeting abc transporters. Oncotarget 2014, 5, 11552–11563. [Google Scholar] [CrossRef] [PubMed]
- Vire, E.; Brenner, C.; Deplus, R.; Blanchon, L.; Fraga, M.; Didelot, C.; Morey, L.; Van Eynde, A.; Bernard, D.; Vanderwinden, J.M.; et al. The polycomb group protein ezh2 directly controls DNA methylation. Nature 2006, 439, 871–874. [Google Scholar] [CrossRef]
- Mahmoud, F.; Shields, B.; Makhoul, I.; Hutchins, L.F.; Shalin, S.C.; Tackett, A.J. Role of ezh2 histone methyltrasferase in melanoma progression and metastasis. Cancer Biol. 2016, 17, 579–591. [Google Scholar] [CrossRef]
- Dai, W.; Xu, X.; Li, S.; Ma, J.; Shi, Q.; Guo, S.; Liu, L.; Guo, W.; Xu, P.; He, Y.; et al. Sox4 promotes proliferative signals by regulating glycolysis through akt activation in melanoma cells. J. Investig. Derm. 2017, 137, 2407–2416. [Google Scholar] [CrossRef]
- Liu, X.; Mi, J.; Qin, H.-H.; He, S.; Li, Z.; Chai, J.-X.; Li, M.; Xu, J.-H.; Wu, J.-F. Sox4 mediates braf inhibitor resistance in melanoma through regulation of igf-1r signaling: In vitro study. Int. J. Dermatol. Venereol. 2020, 3, 156–165. [Google Scholar] [CrossRef]
- Kozar, I.; Cesi, G.; Margue, C.; Philippidou, D.; Kreis, S. Impact of braf kinase inhibitors on the mirnomes and transcriptomes of melanoma cells. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 2980–2992. [Google Scholar] [CrossRef]
- Grzywa, T.M.; Klicka, K.; Paskal, W.; Dudkiewicz, J.; Wejman, J.; Pyzlak, M.; Wlodarski, P.K. Mir-410-3p is induced by vemurafenib via er stress and contributes to resistance to braf inhibitor in melanoma. PLoS ONE 2020, 15, e0234707. [Google Scholar] [CrossRef]
- Fattore, L.; Ruggiero, C.F.; Pisanu, M.E.; Liguoro, D.; Cerri, A.; Costantini, S.; Capone, F.; Acunzo, M.; Romano, G.; Nigita, G.; et al. Reprogramming mirnas global expression orchestrates development of drug resistance in braf mutated melanoma. Cell Death Differ. 2019, 26, 1267–1282. [Google Scholar] [CrossRef] [PubMed]
- Long, J.; Menggen, Q.; Wuren, Q.; Shi, Q.; Pi, X. Long noncoding rna taurine-upregulated gene1 (tug1) promotes tumor growth and metastasis through tug1/mir-129-5p/astrocyte-elevated gene-1 (aeg-1) axis in malignant melanoma. Med. Sci. Monit. 2018, 24, 1547–1559. [Google Scholar] [CrossRef]
- Gao, Y.; Feng, B.; Han, S.; Lu, L.; Chen, Y.; Chu, X.; Wang, R.; Chen, L. Microrna-129 in human cancers: From tumorigenesis to clinical treatment. Cell Physiol. Biochem. 2016, 39, 2186–2202. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Li, J.; Song, Y.; Wang, X. Mir-129-5p inhibits proliferation and invasion of chondrosarcoma cells by regulating sox4/wnt/beta-catenin signaling pathway. Cell Physiol. Biochem. 2017, 42, 242–253. [Google Scholar] [CrossRef]
- Wan, P.; Bai, X.; Yang, C.; He, T.; Luo, L.; Wang, Y.; Fan, M.; Wang, Z.; Lu, L.; Yin, Y.; et al. Mir-129-5p inhibits proliferation, migration, and invasion in rectal adenocarcinoma cells through targeting e2f7. J. Cell Physiol. 2020, 235, 5689–5701. [Google Scholar] [CrossRef]
- Gao, G.; Xiu, D.; Yang, B.; Sun, D.; Wei, X.; Ding, Y.; Ma, Y.; Wang, Z. Mir-129-5p inhibits prostate cancer proliferation via targeting etv1. Onco Targets 2019, 12, 3531–3544. [Google Scholar] [CrossRef]
- Feng, J.; Guo, J.; Wang, J.P.; Chai, B.F. Mir-129-5p inhibits proliferation of gastric cancer cells through targeted inhibition on hmgb1 expression. Eur. Rev. Med. Pharm. Sci. 2020, 24, 3665–3673. [Google Scholar]
- Bachmann, I.M.; Halvorsen, O.J.; Collett, K.; Stefansson, I.M.; Straume, O.; Haukaas, S.A.; Salvesen, H.B.; Otte, A.P.; Akslen, L.A. Ezh2 expression is associated with high proliferation rate and aggressive tumor subgroups in cutaneous melanoma and cancers of the endometrium, prostate, and breast. J. Clin. Oncol. 2006, 24, 268–273. [Google Scholar] [CrossRef]
- Tiffen, J.; Gallagher, S.J.; Filipp, F.; Gunatilake, D.; Emran, A.A.; Cullinane, C.; Dutton-Register, K.; Aoude, L.; Hayward, N.; Chatterjee, A.; et al. Ezh2 cooperates with DNA methylation to downregulate key tumor suppressors and ifn gene signatures in melanoma. J. Investig. Derm. 2020, 140, 2442–2454 e2445. [Google Scholar] [CrossRef]
- Zingg, D.; Debbache, J.; Schaefer, S.M.; Tuncer, E.; Frommel, S.C.; Cheng, P.; Arenas-Ramirez, N.; Haeusel, J.; Zhang, Y.; Bonalli, M.; et al. The epigenetic modifier ezh2 controls melanoma growth and metastasis through silencing of distinct tumour suppressors. Nat. Commun. 2015, 6, 6051. [Google Scholar] [CrossRef]
- Tiffen, J.C.; Gallagher, S.J.; Tseng, H.Y.; Filipp, F.V.; Fazekas de St. Groth, B.; Hersey, P. Ezh2 as a mediator of treatment resistance in melanoma. Pigment. Cell Melanoma Res. 2016, 29, 500–507. [Google Scholar] [CrossRef]
- Grigore, F.; Yang, H.N.; Hanson, N.D.; VanBrocklin, M.W.; Sarver, A.L.; Robinson, J.P. Braf inhibition in melanoma is associated with the dysregulation of histone methylation and histone methyltransferases. Neoplasia 2020, 22, 376–389. [Google Scholar] [CrossRef]
- Qu, Y.; Yang, Q.; Liu, J.; Shi, B.; Ji, M.; Li, G.; Hou, P. C-myc is required for braf(v600e)-induced epigenetic silencing by h3k27me3 in tumorigenesis. Theranostics 2017, 7, 2092–2107. [Google Scholar] [CrossRef]
- Han, H.; Li, W.; Shen, H.; Zhang, J.; Zhu, Y.; Li, Y. Microrna-129-5p, a c-myc negative target, affects hepatocellular carcinoma progression by blocking the warburg effect. J. Mol. Cell Biol. 2016, 8, 400–410. [Google Scholar] [CrossRef]
- Xiao, Y.; Li, X.; Wang, H.; Wen, R.; He, J.; Tang, J. Epigenetic regulation of mir-129-2 and its effects on the proliferation and invasion in lung cancer cells. J. Cell Mol. Med. 2015, 19, 2172–2180. [Google Scholar] [CrossRef]
- Fesler, A.; Zhai, H.; Ju, J. Mir-129 as a novel therapeutic target and biomarker in gastrointestinal cancer. Onco Targets 2014, 7, 1481–1485. [Google Scholar]
- Yuan, C.; Yang, L. Long non-coding rna pitpna-as1 accelerates the progression of colorectal cancer through mir-129-5p/hmgb1 axis. Cancer Manag. Res. 2020, 12, 12497–12507. [Google Scholar] [CrossRef]
- Wang, H.; Li, H.; Yu, Y.; Jiang, Q.; Zhang, R.; Sun, H.; Xing, W.; Li, Y. Long non-coding rna xist promotes the progression of esophageal squamous cell carcinoma through sponging mir-129-5p and upregulating ccnd1 expression. Cell Cycle 2020, 20, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Li, Y.; Liu, W.; Wang, R.; Tang, A.; Hao, H.; Liu, Z.; Ou, H. Mir-129-2 suppresses proliferation and migration of esophageal carcinoma cells through downregulation of sox4 expression. Int. J. Mol. Med. 2013, 32, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.; Qu, S.; Li, X.; Zhong, J.; Chen, X.; Qu, Z.; Wu, D. Mir-129 suppresses tumor cell growth and invasion by targeting pak5 in hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 2015, 464, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhu, H.; Ma, H.; Yuan, L.; Hu, Q.; Yang, L. Linc01305 inhibits malignant progression of cervical cancer via mir-129-5p/sox4 axis. Am. J. Transl. Res. 2020, 12, 7581–7592. [Google Scholar]
- Tang, X.; Tang, J.; Liu, X.; Zeng, L.; Cheng, C.; Luo, Y.; Li, L.; Qin, S.L.; Sang, Y.; Deng, L.M.; et al. Downregulation of mir-129-2 by promoter hypermethylation regulates breast cancer cell proliferation and apoptosis. Oncol. Rep. 2016, 35, 2963–2969. [Google Scholar] [CrossRef]
- Zeng, A.; Yin, J.; Li, Y.; Li, R.; Wang, Z.; Zhou, X.; Jin, X.; Shen, F.; Yan, W.; You, Y. Mir-129-5p targets wnt5a to block pkc/erk/nf-kappab and jnk pathways in glioblastoma. Cell Death Dis. 2018, 9, 394. [Google Scholar] [CrossRef]
- Zeng, H.; Wang, L.; Wang, J.; Chen, T.; Li, H.; Zhang, K.; Chen, J.; Zhen, S.; Tuluhong, D.; Li, J.; et al. Microrna-129-5p suppresses adriamycin resistance in breast cancer by targeting sox2. Arch. Biochem. Biophys. 2018, 651, 52–60. [Google Scholar] [CrossRef]
- Lu, X.; Ma, J.; Chu, J.; Shao, Q.; Zhang, Y.; Lu, G.; Li, J.; Huang, X.; Li, W.; Li, Y.; et al. Mir-129-5p sensitizes the response of her-2 positive breast cancer to trastuzumab by reducing rps6. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2017, 44, 2346–2356. [Google Scholar] [CrossRef]
- Wang, J.; Ye, C.; Liu, J.; Hu, Y. Uca1 confers paclitaxel resistance to ovarian cancer through mir-129/abcb1 axis. Biochem. Biophys. Res. Commun. 2018, 501, 1034–1040. [Google Scholar] [CrossRef]
- Cao, J.; Wang, Q.; Wu, G.; Li, S.; Wang, Q. Mir-129-5p inhibits gemcitabine resistance and promotes cell apoptosis of bladder cancer cells by targeting wnt5a. Int. Urol. Nephrol. 2018, 50, 1811–1819. [Google Scholar] [CrossRef]
- Giricz, O.; Mo, Y.; Dahlman, K.B.; Cotto-Rios, X.M.; Vardabasso, C.; Nguyen, H.; Matusow, B.; Bartenstein, M.; Polishchuk, V.; Johnson, D.B.; et al. The runx1/il-34/csf-1r axis is an autocrinally regulated modulator of resistance to braf-v600e inhibition in melanoma. JCI Insight 2018, 3, 14. [Google Scholar] [CrossRef]
- Tiwari, N.; Tiwari, V.K.; Waldmeier, L.; Balwierz, P.J.; Arnold, P.; Pachkov, M.; Meyer-Schaller, N.; Schubeler, D.; van Nimwegen, E.; Christofori, G. Sox4 is a master regulator of epithelial-mesenchymal transition by controlling ezh2 expression and epigenetic reprogramming. Cancer Cell 2013, 23, 768–783. [Google Scholar] [CrossRef]
- Koumangoye, R.B.; Andl, T.; Taubenslag, K.J.; Zilberman, S.T.; Taylor, C.J.; Loomans, H.A.; Andl, C.D. Sox4 interacts with ezh2 and hdac3 to suppress microrna-31 in invasive esophageal cancer cells. Mol. Cancer 2015, 14, 24. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).