Heterogeneous Circulating Tumor Cells in Sarcoma: Implication for Clinical Practice
Abstract
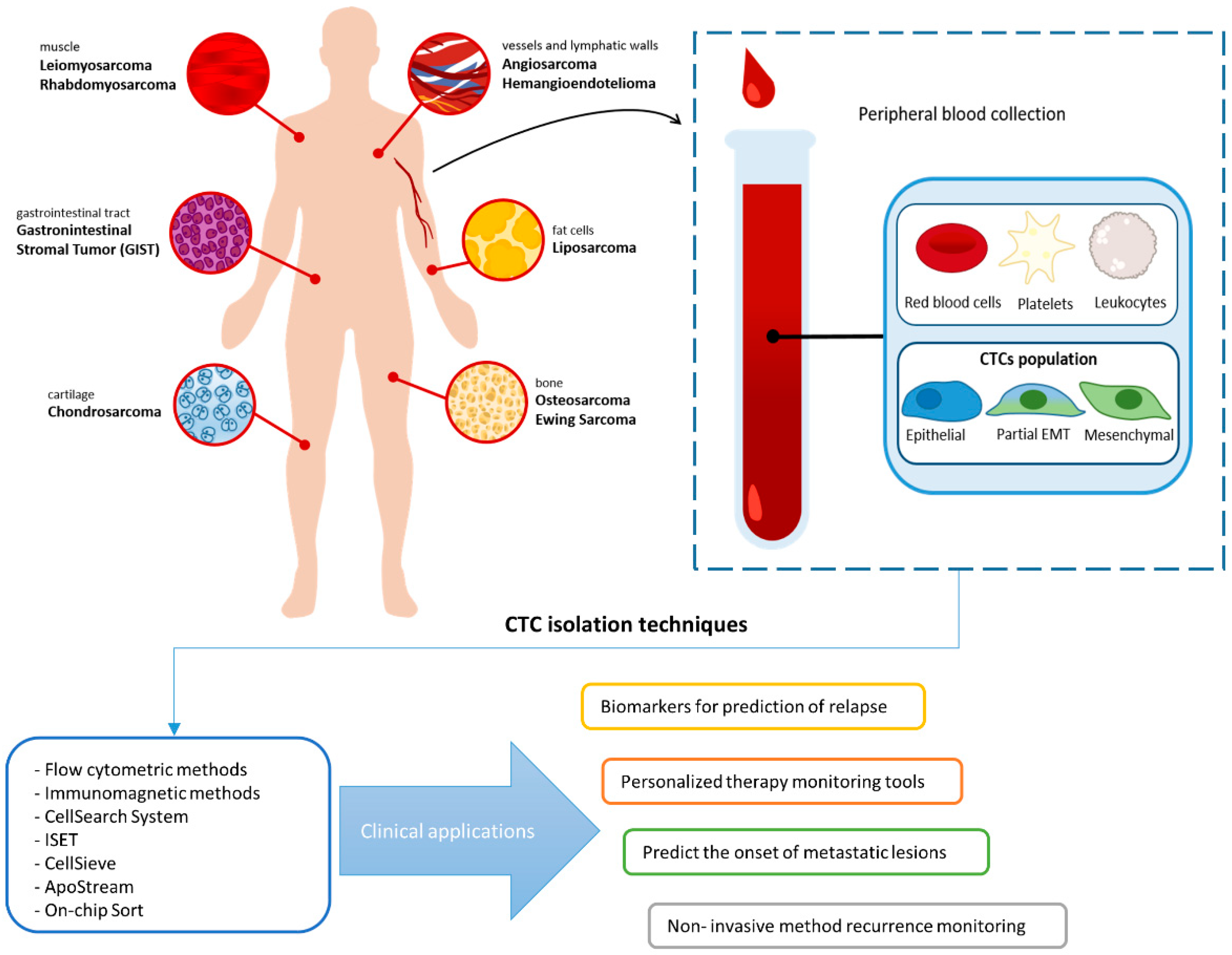
:Simple Summary
Abstract
1. Introduction
2. CTC Heterogeneity: Epithelial–Mesenchymal Transitioned and Stem Cell-Like CTCs
3. Technologies for Detection of CTCs in Sarcoma
4. Clinical Significance of CTC in Sarcoma Tumor
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Anderson, W.J.; Doyle, L.A. Updates from the 2020 World Health Organization Classification of Soft Tissue and Bone Tumours. Histopathology 2021. [Google Scholar] [CrossRef]
- Matushansky, I.; Charytonowicz, E.; Mills, J.; Siddiqi, S.; Hricik, T.; Cordon-Cardo, C. MFH Classification: Differentiating Undifferentiated Pleomorphic Sarcoma in the 21st Century. Expert Rev. Anticancer Ther. 2009, 9, 1135–1144. [Google Scholar] [CrossRef] [Green Version]
- De Vita, A.; Mercatali, L.; Recine, F.; Pieri, F.; Riva, N.; Bongiovanni, A.; Liverani, C.; Spadazzi, C.; Miserocchi, G.; Amadori, D.; et al. Current Classification, Treatment Options, and New Perspectives in the Management of Adipocytic Sarcomas. Onco Targets Ther. 2016, 9, 6233–6246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jo, V.Y.; Fletcher, C.D.M. WHO Classification of Soft Tissue Tumours: An Update Based on the 2013 (4th) Edition. Pathology 2014, 46, 95–104. [Google Scholar] [CrossRef]
- Amankwah, E.K.; Conley, A.P.; Reed, D.R. Epidemiology and Therapies for Metastatic Sarcoma. CLEP 2013, 5, 147. [Google Scholar] [CrossRef] [Green Version]
- Woll, P.J.; Reichardt, P.; Le Cesne, A.; Bonvalot, S.; Azzarelli, A.; Hoekstra, H.J.; Leahy, M.; Van Coevorden, F.; Verweij, J.; Hogendoorn, P.C.; et al. Adjuvant Chemotherapy with Doxorubicin, Ifosfamide, and Lenograstim for Resected Soft-Tissue Sarcoma (EORTC 62931): A Multicentre Randomised Controlled Trial. Lancet Oncol. 2012, 13, 1045–1054. [Google Scholar] [CrossRef]
- Caruso, C.; Garofalo, C. Pharmacogenomics Biomarkers of Soft Tissue Sarcoma Therapies. Front. Oncol. 2020, 10, 509. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casali, P.G.; Jost, L.; Sleijfer, S.; Verweij, J.; Blay, J.-Y. Soft Tissue Sarcomas: ESMO Clinical Recommendations for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2008, 19, ii89–ii93. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, N.; Hawkins, D.S.; Dirksen, U.; Lewis, I.J.; Ferrari, S.; Le Deley, M.-C.; Kovar, H.; Grimer, R.; Whelan, J.; Claude, L.; et al. Ewing Sarcoma: Current Management and Future Approaches Through Collaboration. JCO 2015, 33, 3036–3046. [Google Scholar] [CrossRef]
- Gennaro, N.; Reijers, S.; Bruining, A.; Messiou, C.; Haas, R.; Colombo, P.; Bodalal, Z.; Beets-Tan, R.; van Houdt, W.; van der Graaf, W.T.A. Imaging Response Evaluation after Neoadjuvant Treatment in Soft Tissue Sarcomas: Where Do We Stand? Crit. Rev. Oncol. Hematol. 2021, 160, 103309. [Google Scholar] [CrossRef] [PubMed]
- Brown, H.K.; Schiavone, K.; Gouin, F.; Heymann, M.-F.; Heymann, D. Biology of Bone Sarcomas and New Therapeutic Developments. Calcif. Tissue Int. 2018, 102, 174–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ségaliny, A.I.; Tellez-Gabriel, M.; Heymann, M.-F.; Heymann, D. Receptor Tyrosine Kinases: Characterisation, Mechanism of Action and Therapeutic Interests for Bone Cancers. J. Bone Oncol. 2015, 4, 1–12. [Google Scholar] [CrossRef]
- Heymann, M.-F.; Brown, H.K.; Heymann, D. Drugs in Early Clinical Development for the Treatment of Osteosarcoma. Expert Opin. Investig. Drugs 2016, 25, 1265–1280. [Google Scholar] [CrossRef] [PubMed]
- Womer, R.B.; West, D.C.; Krailo, M.D.; Dickman, P.S.; Pawel, B.R.; Grier, H.E.; Marcus, K.; Sailer, S.; Healey, J.H.; Dormans, J.P.; et al. Randomized Controlled Trial of Interval-Compressed Chemotherapy for the Treatment of Localized Ewing Sarcoma: A Report from the Children’s Oncology Group. JCO 2012, 30, 4148–4154. [Google Scholar] [CrossRef] [Green Version]
- Bielack, S.S.; Smeland, S.; Whelan, J.S.; Marina, N.; Jovic, G.; Hook, J.M.; Krailo, M.D.; Gebhardt, M.; Pápai, Z.; Meyer, J.; et al. Methotrexate, Doxorubicin, and Cisplatin (MAP) Plus Maintenance Pegylated Interferon Alfa-2b Versus MAP Alone in Patients with Resectable High-Grade Osteosarcoma and Good Histologic Response to Preoperative MAP: First Results of the EURAMOS-1 Good Response Randomized Controlled Trial. JCO 2015, 33, 2279–2287. [Google Scholar] [CrossRef]
- Kempf-Bielack, B.; Bielack, S.S.; Jürgens, H.; Branscheid, D.; Berdel, W.E.; Exner, G.U.; Göbel, U.; Helmke, K.; Jundt, G.; Kabisch, H.; et al. Osteosarcoma Relapse After Combined Modality Therapy: An Analysis of Unselected Patients in the Cooperative Osteosarcoma Study Group (COSS). JCO 2005, 23, 559–568. [Google Scholar] [CrossRef]
- In, G.K.; Hu, J.S.; Tseng, W.W. Treatment of Advanced, Metastatic Soft Tissue Sarcoma: Latest Evidence and Clinical Considerations. Ther. Adv. Med. Oncol. 2017, 9, 533–550. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Takahashi, S. Current Molecular Targeted Therapies for Bone and Soft Tissue Sarcomas. IJMS Int. J. Mol. Sci. 2018, 19, 739. [Google Scholar] [CrossRef] [Green Version]
- Ratan, R.; Patel, S.R. Chemotherapy for Soft Tissue Sarcoma: Chemotherapy for Soft Tissue Sarcoma. Cancer 2016, 122, 2952–2960. [Google Scholar] [CrossRef] [Green Version]
- Anninga, J.K.; Gelderblom, H.; Fiocco, M.; Kroep, J.R.; Taminiau, A.H.M.; Hogendoorn, P.C.W.; Egeler, R.M. Chemotherapeutic Adjuvant Treatment for Osteosarcoma: Where Do We Stand? Eur. J. Cancer 2011, 47, 2431–2445. [Google Scholar] [CrossRef]
- Dawson, S.-J.; Tsui, D.W.Y.; Murtaza, M.; Biggs, H.; Rueda, O.M.; Chin, S.-F.; Dunning, M.J.; Gale, D.; Forshew, T.; Mahler-Araujo, B.; et al. Analysis of Circulating Tumor DNA to Monitor Metastatic Breast Cancer. N. Engl. J. Med. 2013, 368, 1199–1209. [Google Scholar] [CrossRef] [Green Version]
- Falzone, L.; Scola, L.; Zanghì, A.; Biondi, A.; Di Cataldo, A.; Libra, M.; Candido, S. Integrated Analysis of Colorectal Cancer MicroRNA Datasets: Identification of MicroRNAs Associated with Tumor Development. Aging 2018, 10, 1000–1014. [Google Scholar] [CrossRef]
- Cheng, D.; Qiu, X.; Zhuang, M.; Zhu, C.; Zou, H.; Liu, Z. MicroRNAs with Prognostic Significance in Osteosarcoma: A Systemic Review and Meta-Analysis. Oncotarget 2017, 8, 81062–81074. [Google Scholar] [CrossRef] [PubMed]
- Galardi, A.; Colletti, M.; Di Paolo, V.; Vitullo, P.; Antonetti, L.; Russo, I.; Di Giannatale, A. Exosomal MiRNAs in Pediatric Cancers. Int. J. Mol. Sci. 2019, 20, 4600. [Google Scholar] [CrossRef] [Green Version]
- Masaoutis, C.; Korkolopoulou, P.; Theocharis, S. Exosomes in Sarcomas: Tiny Messengers with Broad Implications in Diagnosis, Surveillance, Prognosis and Treatment. Cancer Lett. 2019, 449, 172–177. [Google Scholar] [CrossRef]
- Li, X.; Seebacher, N.A.; Hornicek, F.J.; Xiao, T.; Duan, Z. Application of Liquid Biopsy in Bone and Soft Tissue Sarcomas: Present and Future. Cancer Lett. 2018, 439, 66–77. [Google Scholar] [CrossRef]
- Weiser, D.A.; West-Szymanski, D.C.; Fraint, E.; Weiner, S.; Rivas, M.A.; Zhao, C.W.T.; He, C.; Applebaum, M.A. Progress toward Liquid Biopsies in Pediatric Solid Tumors. Cancer Metastasis Rev. 2019, 38, 553–571. [Google Scholar] [CrossRef] [PubMed]
- Schatton, T.; Murphy, G.F.; Frank, N.Y.; Yamaura, K.; Waaga-Gasser, A.M.; Gasser, M.; Zhan, Q.; Jordan, S.; Duncan, L.M.; Weishaupt, C.; et al. Identification of Cells Initiating Human Melanomas. Nature 2008, 451, 345–349. [Google Scholar] [CrossRef] [Green Version]
- Brabletz, T. EMT and MET in Metastasis: Where Are the Cancer Stem Cells? Cancer Cell 2012, 22, 699–701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gold, B.; Cankovic, M.; Furtado, L.V.; Meier, F.; Gocke, C.D. Do Circulating Tumor Cells, Exosomes, and Circulating Tumor Nucleic Acids Have Clinical Utility? J. Mol. Diagn. 2015, 17, 209–224. [Google Scholar] [CrossRef] [Green Version]
- Pantel, K.; Brakenhoff, R.H. Dissecting the Metastatic Cascade. Nat. Rev. Cancer 2004, 4, 448–456. [Google Scholar] [CrossRef]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.J.; Nieto, M.A. Epithelial-Mesenchymal Transitions in Development and Disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef]
- Joosse, S.A.; Gorges, T.M.; Pantel, K. Biology, Detection, and Clinical Implications of Circulating Tumor Cells. EMBO Mol. Med. 2015, 7, 1–11. [Google Scholar] [CrossRef]
- Lambert, A.W.; Pattabiraman, D.R.; Weinberg, R.A. Emerging Biological Principles of Metastasis. Cell 2017, 168, 670–691. [Google Scholar] [CrossRef] [Green Version]
- Pantel, K.; Alix-Panabières, C.; Riethdorf, S. Cancer Micrometastases. Nat. Rev. Clin. Oncol. 2009, 6, 339–351. [Google Scholar] [CrossRef]
- Chaffer, C.L.; Weinberg, R.A. A Perspective on Cancer Cell Metastasis. Science 2011, 331, 1559–1564. [Google Scholar] [CrossRef] [PubMed]
- Castro-Giner, F.; Aceto, N. Tracking Cancer Progression: From Circulating Tumor Cells to Metastasis. Genome Med. 2020, 12, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alix-Panabières, C.; Pantel, K. Challenges in Circulating Tumour Cell Research. Nat. Rev. Cancer 2014, 14, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Stott, S.; Toner, M.; Maheswaran, S.; Haber, D.A. Circulating Tumor Cells: Approaches to Isolation and Characterization. J. Cell Biol. 2011, 192, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Kim, J.; Song, H.; Sohn, K.Y.; Jeon, M.; Han, K.-H. Microfluidic Technologies for Circulating Tumor Cell Isolation. Analyst 2018, 143, 2936–2970. [Google Scholar] [CrossRef]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.M.M.; et al. Circulating Tumor Cells, Disease Progression, and Survival in Metastatic Breast Cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janni, W.J.; Rack, B.; Terstappen, L.W.M.M.; Pierga, J.-Y.; Taran, F.-A.; Fehm, T.; Hall, C.; de Groot, M.R.; Bidard, F.-C.; Friedl, T.W.P.; et al. Pooled Analysis of the Prognostic Relevance of Circulating Tumor Cells in Primary Breast Cancer. Clin. Cancer Res. 2016, 22, 2583–2593. [Google Scholar] [CrossRef] [Green Version]
- Janni, W.; Vogl, F.D.; Wiedswang, G.; Synnestvedt, M.; Fehm, T.; Jückstock, J.; Borgen, E.; Rack, B.; Braun, S.; Sommer, H.; et al. Persistence of Disseminated Tumor Cells in the Bone Marrow of Breast Cancer Patients Predicts Increased Risk for Relapse—A European Pooled Analysis. Clin. Cancer Res. 2011, 17, 2967–2976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Müller, V.; Riethdorf, S.; Rack, B.; Janni, W.; Fasching, P.A.; Solomayer, E.; Aktas, B.; Kasimir-Bauer, S.; Pantel, K.; Fehm, T. Prognostic Impact of Circulating Tumor Cells Assessed with the CellSearch SystemTM and AdnaTest BreastTM in Metastatic Breast Cancer Patients: The DETECT Study. Breast Cancer Res. 2012, 14, R118. [Google Scholar] [CrossRef]
- Pantel, K.; Alix-Panabières, C. Circulating Tumour Cells in Cancer Patients: Challenges and Perspectives. Trends Mol. Med. 2010, 16, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Riethdorf, S.; Fritsche, H.; Müller, V.; Rau, T.; Schindlbeck, C.; Rack, B.; Janni, W.; Coith, C.; Beck, K.; Jänicke, F.; et al. Detection of Circulating Tumor Cells in Peripheral Blood of Patients with Metastatic Breast Cancer: A Validation Study of the CellSearch System. Clin. Cancer Res. 2007, 13, 920–928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, M.C.; Doyle, G.V.; Terstappen, L.W.M.M. Significance of Circulating Tumor Cells Detected by the CellSearch System in Patients with Metastatic Breast Colorectal and Prostate Cancer. J. Oncol. 2010, 2010, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Navin, N.E. Advances and Applications of Single-Cell Sequencing Technologies. Mol. Cell 2015, 58, 598–609. [Google Scholar] [CrossRef] [Green Version]
- Cheng, Y.-H.; Chen, Y.-C.; Lin, E.; Brien, R.; Jung, S.; Chen, Y.-T.; Lee, W.; Hao, Z.; Sahoo, S.; Min Kang, H.; et al. Hydro-Seq Enables Contamination-Free High-Throughput Single-Cell RNA-Sequencing for Circulating Tumor Cells. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Micalizzi, D.S.; Maheswaran, S.; Haber, D.A. A Conduit to Metastasis: Circulating Tumor Cell Biology. Genes Dev. 2017, 31, 1827–1840. [Google Scholar] [CrossRef]
- Lohr, J.G.; Adalsteinsson, V.A.; Cibulskis, K.; Choudhury, A.D.; Rosenberg, M.; Cruz-Gordillo, P.; Francis, J.M.; Zhang, C.-Z.; Shalek, A.K.; Satija, R.; et al. Whole-Exome Sequencing of Circulating Tumor Cells Provides a Window into Metastatic Prostate Cancer. Nat. Biotechnol. 2014, 32, 479–484. [Google Scholar] [CrossRef] [Green Version]
- Aceto, N.; Bardia, A.; Miyamoto, D.T.; Donaldson, M.C.; Wittner, B.S.; Spencer, J.A.; Yu, M.; Pely, A.; Engstrom, A.; Zhu, H.; et al. Circulating Tumor Cell Clusters Are Oligoclonal Precursors of Breast Cancer Metastasis. Cell 2014, 158, 1110–1122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aceto, N.; Toner, M.; Maheswaran, S.; Haber, D.A. En Route to Metastasis: Circulating Tumor Cell Clusters and Epithelial-to-Mesenchymal Transition. Trends Cancer 2015, 1, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Alix-Panabières, C.; Pantel, K. Circulating Tumor Cells: Liquid Biopsy of Cancer. Clin. Chem. 2013, 59, 110–118. [Google Scholar] [CrossRef]
- Agnoletto, C.; Corrà, F.; Minotti, L.; Baldassari, F.; Crudele, F.; Cook, W.; Di Leva, G.; d’Adamo, A.; Gasparini, P.; Volinia, S. Heterogeneity in Circulating Tumor Cells: The Relevance of the Stem-Cell Subset. Cancers 2019, 11, 483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Merino, D.; Weber, T.S.; Serrano, A.; Vaillant, F.; Liu, K.; Pal, B.; Di Stefano, L.; Schreuder, J.; Lin, D.; Chen, Y.; et al. Barcoding Reveals Complex Clonal Behavior in Patient-Derived Xenografts of Metastatic Triple Negative Breast Cancer. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G.; et al. Guidelines and Definitions for Research on Epithelial–Mesenchymal Transition. Nat. Rev. Mol. Cell Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tam, W.L.; Weinberg, R.A. The Epigenetics of Epithelial-Mesenchymal Plasticity in Cancer. Nat. Med. 2013, 19, 1438–1449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular Mechanisms of Epithelial–Mesenchymal Transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178–196. [Google Scholar] [CrossRef] [Green Version]
- Huang, R.Y.-J.; Wong, M.K.; Tan, T.Z.; Kuay, K.T.; Ng, A.H.C.; Chung, V.Y.; Chu, Y.-S.; Matsumura, N.; Lai, H.-C.; Lee, Y.F.; et al. An EMT Spectrum Defines an Anoikis-Resistant and Spheroidogenic Intermediate Mesenchymal State That Is Sensitive to e-Cadherin Restoration by a Src-Kinase Inhibitor, Saracatinib (AZD0530). Cell Death Dis. 2013, 4, e915. [Google Scholar] [CrossRef] [Green Version]
- Pastushenko, I.; Brisebarre, A.; Sifrim, A.; Fioramonti, M.; Revenco, T.; Boumahdi, S.; Van Keymeulen, A.; Brown, D.; Moers, V.; Lemaire, S.; et al. Identification of the Tumour Transition States Occurring during EMT. Nature 2018, 556, 463–468. [Google Scholar] [CrossRef]
- Brabletz, T.; Kalluri, R.; Nieto, M.A.; Weinberg, R.A. EMT in Cancer. Nat. Rev. Cancer 2018, 18, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Thompson, E.W.; Nagaraj, S.H. Transition States That Allow Cancer to Spread. Nature 2018, 556, 442–444. [Google Scholar] [CrossRef] [PubMed]
- Sikandar, S.S.; Kuo, A.H.; Kalisky, T.; Cai, S.; Zabala, M.; Hsieh, R.W.; Lobo, N.A.; Scheeren, F.A.; Sim, S.; Qian, D.; et al. Role of Epithelial to Mesenchymal Transition Associated Genes in Mammary Gland Regeneration and Breast Tumorigenesis. Nat. Commun. 2017, 8, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nieto, M.A.; Huang, R.Y.-J.; Jackson, R.A.; Thiery, J.P. EMT: 2016. Cell 2016, 166, 21–45. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.M.; Dedhar, S.; Kalluri, R.; Thompson, E.W. The Epithelial–Mesenchymal Transition: New Insights in Signaling, Development, and Disease. J. Cell Biol. 2006, 172, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Stemmler, M.P.; Eccles, R.L.; Brabletz, S.; Brabletz, T. Non-Redundant Functions of EMT Transcription Factors. Nat. Cell Biol. 2019, 21, 102–112. [Google Scholar] [CrossRef]
- Hong, T.; Watanabe, K.; Ta, C.H.; Villarreal-Ponce, A.; Nie, Q.; Dai, X. An Ovol2-Zeb1 Mutual Inhibitory Circuit Governs Bidirectional and Multi-Step Transition between Epithelial and Mesenchymal States. PLoS Comput. Biol. 2015, 11, e1004569. [Google Scholar] [CrossRef]
- Barrios-Rodiles, M. High-Throughput Mapping of a Dynamic Signaling Network in Mammalian Cells. Science 2005, 307, 1621–1625. [Google Scholar] [CrossRef] [Green Version]
- Ozdamar, B. Regulation of the Polarity Protein Par6 by TGF Receptors Controls Epithelial Cell Plasticity. Science 2005, 307, 1603–1609. [Google Scholar] [CrossRef]
- Janda, E.; Nevolo, M.; Lehmann, K.; Downward, J.; Beug, H.; Grieco, M. Raf plus TGFβ-Dependent EMT Is Initiated by Endocytosis and Lysosomal Degradation of E-Cadherin. Oncogene 2006, 25, 7117–7130. [Google Scholar] [CrossRef] [Green Version]
- Eckert, M.A.; Lwin, T.M.; Chang, A.T.; Kim, J.; Danis, E.; Ohno-Machado, L.; Yang, J. Twist1-Induced Invadopodia Formation Promotes Tumor Metastasis. Cancer Cell 2011, 19, 372–386. [Google Scholar] [CrossRef] [Green Version]
- Jung, H.-Y.; Fattet, L.; Tsai, J.H.; Kajimoto, T.; Chang, Q.; Newton, A.C.; Yang, J. Apical–Basal Polarity Inhibits Epithelial–Mesenchymal Transition and Tumour Metastasis by PAR-Complex-Mediated SNAI1 Degradation. Nat. Cell Biol. 2019, 21, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.H.; Yang, J. Epithelial-Mesenchymal Plasticity in Carcinoma Metastasis. Genes Dev. 2013, 27, 2192–2206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brabletz, T. To Differentiate or Not—Routes towards Metastasis. Nat. Rev. Cancer 2012, 12, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Chaffer, C.L.; San Juan, B.P.; Lim, E.; Weinberg, R.A. EMT, Cell Plasticity and Metastasis. Cancer Metastasis Rev. 2016, 35, 645–654. [Google Scholar] [CrossRef]
- Yu, M.; Bardia, A.; Wittner, B.S.; Stott, S.L.; Smas, M.E.; Ting, D.T.; Isakoff, S.J.; Ciciliano, J.C.; Wells, M.N.; Shah, A.M.; et al. Circulating Breast Tumor Cells Exhibit Dynamic Changes in Epithelial and Mesenchymal Composition. Science 2013, 339, 580–584. [Google Scholar] [CrossRef] [Green Version]
- Schliekelman, M.J.; Taguchi, A.; Zhu, J.; Dai, X.; Rodriguez, J.; Celiktas, M.; Zhang, Q.; Chin, A.; Wong, C.-H.; Wang, H.; et al. Molecular Portraits of Epithelial, Mesenchymal, and Hybrid States in Lung Adenocarcinoma and Their Relevance to Survival. Cancer Res. 2015, 75, 1789–1800. [Google Scholar] [CrossRef] [Green Version]
- Tan, T.Z.; Miow, Q.H.; Miki, Y.; Noda, T.; Mori, S.; Huang, R.Y.; Thiery, J.P. Epithelial-mesenchymal Transition Spectrum Quantification and Its Efficacy in Deciphering Survival and Drug Responses of Cancer Patients. EMBO Mol. Med. 2014, 6, 1279–1293. [Google Scholar] [CrossRef]
- Bierie, B.; Pierce, S.E.; Kroeger, C.; Stover, D.G.; Pattabiraman, D.R.; Thiru, P.; Liu Donaher, J.; Reinhardt, F.; Chaffer, C.L.; Keckesova, Z.; et al. Integrin-Β4 Identifies Cancer Stem Cell-Enriched Populations of Partially Mesenchymal Carcinoma Cells. Proc. Natl. Acad. Sci. USA 2017, 114, E2337–E2346. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Zhang, X.H.-F.; Massagué, J. Macrophage Binding to Receptor VCAM-1 Transmits Survival Signals in Breast Cancer Cells That Invade the Lungs. Cancer Cell 2011, 20, 538–549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seguin, L.; Kato, S.; Franovic, A.; Camargo, M.F.; Lesperance, J.; Elliott, K.C.; Yebra, M.; Mielgo, A.; Lowy, A.M.; Husain, H.; et al. An Integrin Β3–KRAS–RalB Complex Drives Tumour Stemness and Resistance to EGFR Inhibition. Nat. Cell Biol. 2014, 16, 457–468. [Google Scholar] [CrossRef] [Green Version]
- Slack-Davis, J.K.; Atkins, K.A.; Harrer, C.; Hershey, E.D.; Conaway, M. Vascular Cell Adhesion Molecule-1 Is a Regulator of Ovarian Cancer Peritoneal Metastasis. Cancer Res. 2009, 69, 1469–1476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Zhang, B.; Wu, H.; Cai, J.; Sui, X.; Wang, Y.; Li, H.; Qiu, Y.; Wang, T.; Chen, Z.; et al. CD51 Correlates with the TGF-Beta Pathway and Is a Functional Marker for Colorectal Cancer Stem Cells. Oncogene 2017, 36, 1351–1363. [Google Scholar] [CrossRef] [PubMed]
- Dalerba, P.; Dylla, S.J.; Park, I.-K.; Liu, R.; Wang, X.; Cho, R.W.; Hoey, T.; Gurney, A.; Huang, E.H.; Simeone, D.M.; et al. Phenotypic Characterization of Human Colorectal Cancer Stem Cells. Proc. Natl. Acad. Sci. USA 2007, 104, 10158–10163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Hajj, M.; Wicha, M.S.; Benito-Hernandez, A.; Morrison, S.J.; Clarke, M.F. Prospective Identification of Tumorigenic Breast Cancer Cells. Proc. Natl. Acad. Sci. USA 2003, 100, 3983–3988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, T.-Y.; Lu, R.-M.; Liao, M.-Y.; Yu, J.; Chung, C.-H.; Kao, C.-F.; Wu, H.-C. Epithelial Cell Adhesion Molecule Regulation Is Associated with the Maintenance of the Undifferentiated Phenotype of Human Embryonic Stem Cells. J. Biol. Chem. 2010, 285, 8719–8732. [Google Scholar] [CrossRef] [Green Version]
- Massoner, P.; Thomm, T.; Mack, B.; Untergasser, G.; Martowicz, A.; Bobowski, K.; Klocker, H.; Gires, O.; Puhr, M. EpCAM Is Overexpressed in Local and Metastatic Prostate Cancer, Suppressed by Chemotherapy and Modulated by MET-Associated MiRNA-200c/205. Br. J. Cancer 2014, 111, 955–964. [Google Scholar] [CrossRef] [Green Version]
- Sieuwerts, A.M.; Kraan, J.; Bolt, J.; van der Spoel, P.; Elstrodt, F.; Schutte, M.; Martens, J.W.M.; Gratama, J.-W.; Sleijfer, S.; Foekens, J.A. Anti-Epithelial Cell Adhesion Molecule Antibodies and the Detection of Circulating Normal-Like Breast Tumor Cells. JNCI J. Natl. Cancer Inst. 2009, 101, 61–66. [Google Scholar] [CrossRef]
- Kalluri, R.; Weinberg, R.A. The Basics of Epithelial-Mesenchymal Transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [Green Version]
- Satelli, A.; Li, S. Vimentin in Cancer and Its Potential as a Molecular Target for Cancer Therapy. Cell. Mol. Life Sci. 2011, 68, 3033–3046. [Google Scholar] [CrossRef] [Green Version]
- Satelli, A.; Mitra, A.; Cutrera, J.J.; Devarie, M.; Xia, X.; Ingram, D.R.; Dibra, D.; Somaiah, N.; Torres, K.E.; Ravi, V.; et al. Universal Marker and Detection Tool for Human Sarcoma Circulating Tumor Cells. Cancer Res. 2014, 74, 1645–1650. [Google Scholar] [CrossRef] [Green Version]
- Powell, A.A.; Talasaz, A.H.; Zhang, H.; Coram, M.A.; Reddy, A.; Deng, G.; Telli, M.L.; Advani, R.H.; Carlson, R.W.; Mollick, J.A.; et al. Single Cell Profiling of Circulating Tumor Cells: Transcriptional Heterogeneity and Diversity from Breast Cancer Cell Lines. PLoS ONE 2012, 7, e33788. [Google Scholar] [CrossRef] [Green Version]
- Satelli, A.; Mitra, A.; Brownlee, Z.; Xia, X.; Bellister, S.; Overman, M.J.; Kopetz, S.; Ellis, L.M.; Meng, Q.H.; Li, S. Epithelial–Mesenchymal Transitioned Circulating Tumor Cells Capture for Detecting Tumor Progression. Clin. Cancer Res. 2015, 21, 899–906. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thiery, J.P.; Lim, C.T. Tumor Dissemination: An EMT Affair. Cancer Cell 2013, 23, 272–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, X.; Carstens, J.L.; Kim, J.; Scheible, M.; Kaye, J.; Sugimoto, H.; Wu, C.-C.; LeBleu, V.S.; Kalluri, R. Epithelial-to-Mesenchymal Transition Is Dispensable for Metastasis but Induces Chemoresistance in Pancreatic Cancer. Nature 2015, 527, 525–530. [Google Scholar] [CrossRef] [Green Version]
- Fischer, K.R.; Durrans, A.; Lee, S.; Sheng, J.; Li, F.; Wong, S.T.C.; Choi, H.; El Rayes, T.; Ryu, S.; Troeger, J.; et al. Epithelial-to-Mesenchymal Transition Is Not Required for Lung Metastasis but Contributes to Chemoresistance. Nature 2015, 527, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Brabletz, T.; Kang, Y.; Longmore, G.D.; Nieto, M.A.; Stanger, B.Z.; Yang, J.; Weinberg, R.A. Upholding a Role for EMT in Breast Cancer Metastasis. Nature 2017, 547, E1–E3. [Google Scholar] [CrossRef]
- Diepenbruck, M.; Christofori, G. Epithelial–Mesenchymal Transition (EMT) and Metastasis: Yes, No, Maybe? Curr. Opin. Cell Biol. 2016, 43, 7–13. [Google Scholar] [CrossRef] [Green Version]
- Aiello, N.M.; Brabletz, T.; Kang, Y.; Nieto, M.A.; Weinberg, R.A.; Stanger, B.Z. Upholding a Role for EMT in Pancreatic Cancer Metastasis. Nature 2017, 547, E7–E8. [Google Scholar] [CrossRef]
- Liu, X.; Li, J.; Cadilha, B.L.; Markota, A.; Voigt, C.; Huang, Z.; Lin, P.P.; Wang, D.D.; Dai, J.; Kranz, G.; et al. Epithelial-Type Systemic Breast Carcinoma Cells with a Restricted Mesenchymal Transition Are a Major Source of Metastasis. Sci. Adv. 2019, 5, eaav4275. [Google Scholar] [CrossRef] [Green Version]
- Ye, X.; Weinberg, R.A. Epithelial–Mesenchymal Plasticity: A Central Regulator of Cancer Progression. Trends Cell Biol. 2015, 25, 675–686. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, A.; Settleman, J. EMT, Cancer Stem Cells and Drug Resistance: An Emerging Axis of Evil in the War on Cancer. Oncogene 2010, 29, 4741–4751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rumman, M.; Dhawan, J.; Kassem, M. Concise Review: Quiescence in Adult Stem Cells: Biological Significance and Relevance to Tissue Regeneration: ASC Quiescence: Role and Relevance to Tissue Regeneration. Stem Cells 2015, 33, 2903–2912. [Google Scholar] [CrossRef]
- Nieto, M.A. Epithelial Plasticity: A Common Theme in Embryonic and Cancer Cells. Science 2013, 342, 1234850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giuliano, M.; Shaikh, A.; Lo, H.C.; Arpino, G.; De Placido, S.; Zhang, X.H.; Cristofanilli, M.; Schiff, R.; Trivedi, M.V. Perspective on Circulating Tumor Cell Clusters: Why It Takes a Village to Metastasize. Cancer Res. 2018, 78, 845–852. [Google Scholar] [CrossRef] [Green Version]
- Murlidhar, V.; Reddy, R.M.; Fouladdel, S.; Zhao, L.; Ishikawa, M.K.; Grabauskiene, S.; Zhang, Z.; Lin, J.; Chang, A.C.; Carrott, P.; et al. Poor Prognosis Indicated by Venous Circulating Tumor Cell Clusters in Early-Stage Lung Cancers. Cancer Res. 2017, 77, 5194–5206. [Google Scholar] [CrossRef] [Green Version]
- Cheung, K.J.; Padmanaban, V.; Silvestri, V.; Schipper, K.; Cohen, J.D.; Fairchild, A.N.; Gorin, M.A.; Verdone, J.E.; Pienta, K.J.; Bader, J.S.; et al. Polyclonal Breast Cancer Metastases Arise from Collective Dissemination of Keratin 14-Expressing Tumor Cell Clusters. Proc. Natl. Acad. Sci. USA 2016, 113, E854–E863. [Google Scholar] [CrossRef] [Green Version]
- Padmanaban, V.; Krol, I.; Suhail, Y.; Szczerba, B.M.; Aceto, N.; Bader, J.S.; Ewald, A.J. E-Cadherin Is Required for Metastasis in Multiple Models of Breast Cancer. Nature 2019, 573, 439–444. [Google Scholar] [CrossRef]
- Hwang, P.Y.; Brenot, A.; King, A.C.; Longmore, G.D.; George, S.C. Randomly Distributed K14+ Breast Tumor Cells Polarize to the Leading Edge and Guide Collective Migration in Response to Chemical and Mechanical Environmental Cues. Cancer Res. 2019, 79, 1899–1912. [Google Scholar] [CrossRef] [Green Version]
- Adams, D.L.; Martin, S.S.; Alpaugh, R.K.; Charpentier, M.; Tsai, S.; Bergan, R.C.; Ogden, I.M.; Catalona, W.; Chumsri, S.; Tang, C.-M.; et al. Circulating Giant Macrophages as a Potential Biomarker of Solid Tumors. Proc. Natl. Acad. Sci. USA 2014, 111, 3514–3519. [Google Scholar] [CrossRef] [Green Version]
- Gundem, G.; Van Loo, P.; Kremeyer, B.; Alexandrov, L.B.; Tubio, J.M.C.; Papaemmanuil, E.; Brewer, D.S.; Kallio, H.M.L.; Högnäs, G.; Annala, M.; et al. The Evolutionary History of Lethal Metastatic Prostate Cancer. Nature 2015, 520, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Latil, M.; Nassar, D.; Beck, B.; Boumahdi, S.; Wang, L.; Brisebarre, A.; Dubois, C.; Nkusi, E.; Lenglez, S.; Checinska, A.; et al. Cell-Type-Specific Chromatin States Differentially Prime Squamous Cell Carcinoma Tumor-Initiating Cells for Epithelial to Mesenchymal Transition. Cell Stem Cell 2017, 20, 191–204.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mani, S.A.; Guo, W.; Liao, M.-J.; Eaton, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The Epithelial-Mesenchymal Transition Generates Cells with Properties of Stem Cells. Cell 2008, 133, 704–715. [Google Scholar] [CrossRef] [Green Version]
- Morel, A.-P.; Lièvre, M.; Thomas, C.; Hinkal, G.; Ansieau, S.; Puisieux, A. Generation of Breast Cancer Stem Cells through Epithelial-Mesenchymal Transition. PLoS ONE 2008, 3, e2888. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.B.; Chaffer, C.L.; Weinberg, R.A. Cancer Stem Cells: Mirage or Reality? Nat. Med. 2009, 15, 1010–1012. [Google Scholar] [CrossRef]
- Ginestier, C.; Hur, M.H.; Charafe-Jauffret, E.; Monville, F.; Dutcher, J.; Brown, M.; Jacquemier, J.; Viens, P.; Kleer, C.G.; Liu, S.; et al. ALDH1 Is a Marker of Normal and Malignant Human Mammary Stem Cells and a Predictor of Poor Clinical Outcome. Cell Stem Cell 2007, 1, 555–567. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Cong, Y.; Wang, D.; Sun, Y.; Deng, L.; Liu, Y.; Martin-Trevino, R.; Shang, L.; McDermott, S.P.; Landis, M.D.; et al. Breast Cancer Stem Cells Transition between Epithelial and Mesenchymal States Reflective of Their Normal Counterparts. Stem Cell Rep. 2014, 2, 78–91. [Google Scholar] [CrossRef]
- Kong, D.; Banerjee, S.; Ahmad, A.; Li, Y.; Wang, Z.; Sethi, S.; Sarkar, F.H. Epithelial to Mesenchymal Transition Is Mechanistically Linked with Stem Cell Signatures in Prostate Cancer Cells. PLoS ONE 2010, 5, e12445. [Google Scholar] [CrossRef]
- Shamir, E.R.; Pappalardo, E.; Jorgens, D.M.; Coutinho, K.; Tsai, W.-T.; Aziz, K.; Auer, M.; Tran, P.T.; Bader, J.S.; Ewald, A.J. Twist1-Induced Dissemination Preserves Epithelial Identity and Requires E-Cadherin. J. Cell Biol. 2014, 204, 839–856. [Google Scholar] [CrossRef]
- Schmidt, J.M.; Panzilius, E.; Bartsch, H.S.; Irmler, M.; Beckers, J.; Kari, V.; Linnemann, J.R.; Dragoi, D.; Hirschi, B.; Kloos, U.J.; et al. Stem-Cell-like Properties and Epithelial Plasticity Arise as Stable Traits after Transient Twist1 Activation. Cell Rep. 2015, 10, 131–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ocaña, O.H.; Córcoles, R.; Fabra, Á.; Moreno-Bueno, G.; Acloque, H.; Vega, S.; Barrallo-Gimeno, A.; Cano, A.; Nieto, M.A. Metastatic Colonization Requires the Repression of the Epithelial-Mesenchymal Transition Inducer Prrx1. Cancer Cell 2012, 22, 709–724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brabletz, T.; Jung, A.; Spaderna, S.; Hlubek, F.; Kirchner, T. Migrating Cancer Stem Cells—An Integrated Concept of Malignant Tumour Progression. Nat. Rev. Cancer 2005, 5, 744–749. [Google Scholar] [CrossRef] [PubMed]
- Theveneau, E.; Mayor, R. Can Mesenchymal Cells Undergo Collective Cell Migration? The Case of the Neural Crest: The Case of the Neural Crest. Cell Adhes. Migr. 2011, 5, 490–498. [Google Scholar] [CrossRef] [Green Version]
- Baccelli, I.; Schneeweiss, A.; Riethdorf, S.; Stenzinger, A.; Schillert, A.; Vogel, V.; Klein, C.; Saini, M.; Bäuerle, T.; Wallwiener, M.; et al. Identification of a Population of Blood Circulating Tumor Cells from Breast Cancer Patients That Initiates Metastasis in a Xenograft Assay. Nat. Biotechnol. 2013, 31, 539–544. [Google Scholar] [CrossRef]
- Celià-Terrassa, T.; Kang, Y. Distinctive Properties of Metastasis-Initiating Cells. Genes Dev. 2016, 30, 892–908. [Google Scholar] [CrossRef] [Green Version]
- Jolly, M.K. Implications of the Hybrid Epithelial/Mesenchymal Phenotype in Metastasis. Front. Oncol. 2015, 5. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.B.; Yeo, T.; Lee, W.D.; Bhagat, A.A.S.; Tan, S.J.; Tan, D.S.W.; Lim, W.-T.; Lim, C.T. Addressing Cellular Heterogeneity in Tumor and Circulation for Refined Prognostication. Proc. Natl. Acad. Sci. USA 2019, 116, 17957–17962. [Google Scholar] [CrossRef] [Green Version]
- Grosse-Wilde, A.; Fouquier d’Hérouël, A.; McIntosh, E.; Ertaylan, G.; Skupin, A.; Kuestner, R.E.; del Sol, A.; Walters, K.-A.; Huang, S. Stemness of the Hybrid Epithelial/Mesenchymal State in Breast Cancer and Its Association with Poor Survival. PLoS ONE 2015, 10, e0126522. [Google Scholar] [CrossRef]
- Papadaki, M.A.; Stoupis, G.; Theodoropoulos, P.A.; Mavroudis, D.; Georgoulias, V.; Agelaki, S. Circulating Tumor Cells with Stemness and Epithelial-to-Mesenchymal Transition Features Are Chemoresistant and Predictive of Poor Outcome in Metastatic Breast Cancer. Mol. Cancer Ther. 2019, 18, 437–447. [Google Scholar] [CrossRef] [Green Version]
- Gires, O.; Stoecklein, N.H. Dynamic EpCAM Expression on Circulating and Disseminating Tumor Cells: Causes and Consequences. Cell. Mol. Life Sci. 2014, 71, 4393–4402. [Google Scholar] [CrossRef]
- Korpal, M.; Ell, B.J.; Buffa, F.M.; Ibrahim, T.; Blanco, M.A.; Celià-Terrassa, T.; Mercatali, L.; Khan, Z.; Goodarzi, H.; Hua, Y.; et al. Direct Targeting of Sec23a by MiR-200s Influences Cancer Cell Secretome and Promotes Metastatic Colonization. Nat. Med. 2011, 17, 1101–1108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chao, Y.; Wu, Q.; Acquafondata, M.; Dhir, R.; Wells, A. Partial Mesenchymal to Epithelial Reverting Transition in Breast and Prostate Cancer Metastases. Cancer Microenviron. 2012, 5, 19–28. [Google Scholar] [CrossRef] [Green Version]
- De Wit, S.; Manicone, M.; Rossi, E.; Lampignano, R.; Yang, L.; Zill, B.; Rengel-Puertas, A.; Ouhlen, M.; Crespo, M.; Berghuis, A.M.S.; et al. EpCAMhigh and EpCAMlow Circulating Tumor Cells in Metastatic Prostate and Breast Cancer Patients. Oncotarget 2018, 9, 35705–35716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rack, B.; Schindlbeck, C.; Jückstock, J.; Andergassen, U.; Hepp, P.; Zwingers, T.; Friedl, T.W.P.; Lorenz, R.; Tesch, H.; Fasching, P.A.; et al. Circulating Tumor Cells Predict Survival in Early Average-to-High Risk Breast Cancer Patients. JNCI J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef] [PubMed]
- Folpe, A.L.; Goldblum, J.R.; Rubin, B.P.; Shehata, B.M.; Liu, W.; Dei Tos, A.P.; Weiss, S.W. Morphologic and Immunophenotypic Diversity in Ewing Family Tumors: A Study of 66 Genetically Confirmed Cases. Am. J. Surg. Pathol. 2005, 29, 1025–1033. [Google Scholar] [PubMed]
- Collini, P.; Sampietro, G.; Bertulli, R.; Casali, P.G.; Luksch, R.; Mezzelani, A.; Sozzi, G.; Pilotti, S. Cytokeratin Immunoreactivity in 41 Cases of ES/PNET Confirmed by Molecular Diagnostic Studies. Am. J. Surg. Pathol. 2001, 25, 273–274. [Google Scholar] [CrossRef]
- Machado, I.; López-Guerrero, J.A.; Navarro, S.; Alberghini, M.; Scotlandi, K.; Picci, P.; Llombart-Bosch, A. Epithelial Cell Adhesion Molecules and Epithelial Mesenchymal Transition (EMT) Markers in Ewing’s Sarcoma Family of Tumors (ESFTs). Do They Offer Any Prognostic Significance? Virchows Arch. 2012, 461, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Schuetz, A.N.; Rubin, B.P.; Goldblum, J.R.; Shehata, B.; Weiss, S.W.; Liu, W.; Wick, M.R.; Folpe, A.L. Intercellular Junctions in Ewing Sarcoma/Primitive Neuroectodermal Tumor: Additional Evidence of Epithelial Differentiation. Mod. Pathol. 2005, 18, 1403–1410. [Google Scholar] [CrossRef] [Green Version]
- Williamson, D.; Missiaglia, E.; de Reyniès, A.; Pierron, G.; Thuille, B.; Palenzuela, G.; Thway, K.; Orbach, D.; Laé, M.; Fréneaux, P.; et al. Fusion Gene–Negative Alveolar Rhabdomyosarcoma Is Clinically and Molecularly Indistinguishable from Embryonal Rhabdomyosarcoma. JCO 2010, 28, 2151–2158. [Google Scholar] [CrossRef] [Green Version]
- Tombolan, L.; Rossi, E.; Zin, A.; Santoro, L.; Bonvini, P.; Zamarchi, R.; Bisogno, G. Pediatric Sarcomas Display a Variable EpCAM Expression in a Histology-Dependent Manner. Transl. Oncol. 2020, 13, 100846. [Google Scholar] [CrossRef]
- Sannino, G.; Marchetto, A.; Kirchner, T.; Grünewald, T.G.P. Epithelial-to-Mesenchymal and Mesenchymal-to-Epithelial Transition in Mesenchymal Tumors: A Paradox in Sarcomas? Cancer Res. 2017, 77, 4556–4561. [Google Scholar] [CrossRef] [Green Version]
- Lv, Y.; Dai, H.; Yan, G.; Meng, G.; Zhang, X.; Guo, Q. Downregulation of Tumor Suppressing STF CDNA 3 Promotes Epithelial-Mesenchymal Transition and Tumor Metastasis of Osteosarcoma by the Wnt/GSK-3β/β-Catenin/Snail Signaling Pathway. Cancer Lett. 2016, 373, 164–173. [Google Scholar] [CrossRef]
- Thway, K.; Noujaim, J.; Zaidi, S.; Miah, A.B.; Benson, C.; Messiou, C.; Jones, R.L.; Fisher, C. Desmoplastic Small Round Cell Tumor: Pathology, Genetics, and Potential Therapeutic Strategies. Int. J. Surg. Pathol. 2016, 24, 672–684. [Google Scholar] [CrossRef]
- Devecchi, A.; De Cecco, L.; Dugo, M.; Penso, D.; Dagrada, G.; Brich, S.; Stacchiotti, S.; Sensi, M.; Canevari, S.; Pilotti, S. The Genomics of Desmoplastic Small Round Cell Tumor Reveals the Deregulation of Genes Related to DNA Damage Response, Epithelial-Mesenchymal Transition, and Immune Response. Cancer Commun. 2018, 38, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saito, T.; Nagai, M.; Ladanyi, M. SYT-SSX1 and SYT-SSX2 Interfere with Repression of E-Cadherin by Snail and Slug: A Potential Mechanism for Aberrant Mesenchymal to Epithelial Transition in Human Synovial Sarcoma. Cancer Res. 2006, 66, 6919–6927. [Google Scholar] [CrossRef] [Green Version]
- Franzetti, G.-A.; Laud-Duval, K.; van der Ent, W.; Brisac, A.; Irondelle, M.; Aubert, S.; Dirksen, U.; Bouvier, C.; de Pinieux, G.; Snaar-Jagalska, E.; et al. Cell-to-Cell Heterogeneity of EWSR1-FLI1 Activity Determines Proliferation/Migration Choices in Ewing Sarcoma Cells. Oncogene 2017, 36, 3505–3514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Yan, D.; Wu, W.; Zhu, J.; Ye, W.; Shu, Q. MicroRNA-130a Promotes the Metastasis and Epithelial-Mesenchymal Transition of Osteosarcoma by Targeting PTEN. Oncol. Rep. 2016, 35, 3285–3292. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Zhang, L.; Cui, W.; Du, J.; Li, Z.; Pang, Y.; Liu, Q.; Shang, H.; Meng, L.; Li, W.; et al. Epigenetically Upregulated GEFT-Derived Invasion and Metastasis of Rhabdomyosarcoma via Epithelial Mesenchymal Transition Promoted by the Rac1/Cdc42-PAK Signalling Pathway. EBioMedicine 2019, 50, 122–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossi, E.; Fassan, M.; Aieta, M.; Zilio, F.; Celadin, R.; Borin, M.; Grassi, A.; Troiani, L.; Basso, U.; Barile, C.; et al. Dynamic Changes of Live/Apoptotic Circulating Tumour Cells as Predictive Marker of Response to Sunitinib in Metastatic Renal Cancer. Br. J. Cancer 2012, 107, 1286–1294. [Google Scholar] [CrossRef] [Green Version]
- Visvader, J.E.; Lindeman, G.J. Cancer Stem Cells in Solid Tumours: Accumulating Evidence and Unresolved Questions. Nat. Rev. Cancer 2008, 8, 755–768. [Google Scholar] [CrossRef]
- Allard, W.J.; Matera, J.; Miller, M.C.; Repollet, M.; Connelly, M.C.; Rao, C.; Tibbe, A.G.J.; Uhr, J.W.; Terstappen, L.W.M.M. Tumor Cells Circulate in the Peripheral Blood of All Major Carcinomas but Not in Healthy Subjects or Patients with Nonmalignant Diseases. Clin. Cancer Res. 2004, 10, 6897–6904. [Google Scholar] [CrossRef] [Green Version]
- Königsberg, R.; Obermayr, E.; Bises, G.; Pfeiler, G.; Gneist, M.; Wrba, F.; de Santis, M.; Zeillinger, R.; Hudec, M.; Dittrich, C. Detection of EpCAM Positive and Negative Circulating Tumor Cells in Metastatic Breast Cancer Patients. Acta Oncol. 2011, 50, 700–710. [Google Scholar] [CrossRef] [PubMed]
- Lianidou, E.S. Circulating Tumor Cell Isolation: A Marathon Race Worth Running. Clin. Chem. 2014, 60, 287–289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lianidou, E.S.; Mavroudis, D.; Sotiropoulou, G.; Agelaki, S.; Pantel, K. What’s New on Circulating Tumor Cells? A Meeting Report. Breast Cancer Res. 2010, 12, 307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schleiermacher, G.; Peter, M.; Oberlin, O.; Philip, T.; Rubie, H.; Mechinaud, F.; Sommelet-Olive, D.; Landman-Parker, J.; Bours, D.; Michon, J.; et al. Increased Risk of Systemic Relapses Associated with Bone Marrow Micrometastasis and Circulating Tumor Cells in Localized Ewing Tumor. JCO 2003, 21, 85–91. [Google Scholar] [CrossRef]
- DuBois, S.G.; Epling, C.L.; Teague, J.; Matthay, K.K.; Sinclair, E. Flow Cytometric Detection of Ewing Sarcoma Cells in Peripheral Blood and Bone Marrow. Pediatr. Blood Cancer 2010, 54, 13–18. [Google Scholar] [CrossRef] [Green Version]
- Pfleiderer, C.; Zoubek, A.; Gruber, B.; Kronberger, M.; Ambros, P.F.; Lion, T.; Fink, F.-M.; Gadner, H.; Kovar, H. Detection of Tumour Cells in Peripheral Blood and Bone Marrow from Ewing Tumour Patients by Rt-Pcr. Int. J. Cancer 1995, 64, 135–139. [Google Scholar] [CrossRef]
- Peter, M.; Magdelenat, H.; Michon, J.; Melot, T.; Oberlin, O.; Zucker, J.; Thomas, G.; Delattre, O. Sensitive Detection of Occult Ewing’s Cells by the Reverse Transcriptase-Polymerase Chain Reaction. Br. J. Cancer 1995, 72, 96–100. [Google Scholar] [CrossRef] [Green Version]
- Avigad, S.; Cohen, I.J.; Zilberstein, J.; Liberzon, E.; Goshen, Y.; Ash, S.; Meller, I.; Kollender, Y.; Issakov, J.; Zaizov, R.; et al. The Predictive Potential of Molecular Detection in the Nonmetastatic Ewing Family of Tumors. Cancer 2004, 100, 1053–1058. [Google Scholar] [CrossRef]
- Sartori, F.; Alaggio, R.; Zanazzo, G.; Garaventa, A.; Di Cataldo, A.; Carli, M.; Rosolen, A. Results of a Prospective Minimal Disseminated Disease Study in Human Rhabdomyosarcoma Using Three Different Molecular Markers. Cancer 2006, 106, 1766–1775. [Google Scholar] [CrossRef] [PubMed]
- Wong, I.H.; Chan, A.T.; Johnson, P.J. Quantitative Analysis of Circulating Tumor Cells in Peripheral Blood of Osteosarcoma Patients Using Osteoblast-Specific Messenger RNA Markers: A Pilot Study. Clin. Cancer Res. 2000, 6, 2183–2188. [Google Scholar] [PubMed]
- Hoshino, M.; Ogose, A.; Kawashima, H.; Izumi, T.; Hotta, T.; Hatano, H.; Morita, T.; Otsuka, H.; Umezu, H.; Yanoma, S.; et al. Molecular Analyses of Cell Origin and Detection of Circulating Tumor Cells in the Peripheral Blood in Alveolar Soft Part Sarcoma. Cancer Genet. Cytogenet. 2009, 190, 75–80. [Google Scholar] [CrossRef]
- Kelly, K.M.; Womer, R.B.; Barr, F.G. Minimal Disease Detection in Patients with Alveolar Rhabdomyosarcoma Using a Reverse Transcriptase-Polymerase Chain Reaction Method. Cancer 1996, 78, 1320–1327. [Google Scholar] [CrossRef]
- Schleiermacher, G.; Delattre, O. Detection of micrometastases and circulating tumour cells using molecular biology technics in solid tumours. Bull. Cancer 2001, 88, 561–570. [Google Scholar] [PubMed]
- Gallego, S.; Llort, A.; Roma, J.; Sabado, C.; Gros, L.; de Toledo, J.S. Detection of Bone Marrow Micrometastasis and Microcirculating Disease in Rhabdomyosarcoma by a Real-Time RT-PCR Assay. J. Cancer Res. Clin. Oncol. 2006, 132, 356–362. [Google Scholar] [CrossRef]
- Hemming, M.L.; Klega, K.; Rhoades, J.; Ha, G.; Acker, K.E.; Andersen, J.L.; Thai, E.; Nag, A.; Thorner, A.R.; Raut, C.P.; et al. Detection of Circulating Tumor DNA in Patients with Leiomyosarcoma With Progressive Disease. JCO Precis. Oncol. 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Eastley, N.C.; Ottolini, B.; Neumann, R.; Luo, J.-L.; Hastings, R.K.; Khan, I.; Moore, D.A.; Esler, C.P.; Shaw, J.A.; Royle, N.J.; et al. Circulating Tumour-Derived DNA in Metastatic Soft Tissue Sarcoma. Oncotarget 2018, 9, 10549–10560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbosh, C.; Birkbak, N.J.; Swanton, C. Early Stage NSCLC—Challenges to Implementing CtDNA-Based Screening and MRD Detection. Nat. Rev. Clin. Oncol. 2018, 15, 577–586. [Google Scholar] [CrossRef]
- Krumbholz, M.; Hellberg, J.; Steif, B.; Bäuerle, T.; Gillmann, C.; Fritscher, T.; Agaimy, A.; Frey, B.; Juengert, J.; Wardelmann, E.; et al. Genomic EWSR1 Fusion Sequence as Highly Sensitive and Dynamic Plasma Tumor Marker in Ewing Sarcoma. Clin. Cancer Res. 2016, 22, 4356–4365. [Google Scholar] [CrossRef] [Green Version]
- Smolle, M.A.; Leithner, A.; Posch, F.; Szkandera, J.; Liegl-Atzwanger, B.; Pichler, M. MicroRNAs in Different Histologies of Soft Tissue Sarcoma: A Comprehensive Review. IJMS 2017, 18, 1960. [Google Scholar] [CrossRef] [Green Version]
- Kubota, D.; Kosaka, N.; Fujiwara, T.; Yoshida, A.; Arai, Y.; Qiao, Z.; Takeshita, F.; Ochiya, T.; Kawai, A.; Kondo, T. MiR-125b and MiR-100 Are Predictive Biomarkers of Response to Induction Chemotherapy in Osteosarcoma. Sarcoma 2016, 2016, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tellez-Gabriel, M.; Brown, H.K.; Young, R.; Heymann, M.-F.; Heymann, D. The Challenges of Detecting Circulating Tumor Cells in Sarcoma. Front. Oncol. 2016, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pecot, C.V.; Bischoff, F.Z.; Mayer, J.A.; Wong, K.L.; Pham, T.; Bottsford-Miller, J.; Stone, R.L.; Lin, Y.G.; Jaladurgam, P.; Roh, J.W.; et al. A Novel Platform for Detection of CK+ and CK− CTCs. Cancer Discov. 2011, 1, 580–586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yokobori, T.; Iinuma, H.; Shimamura, T.; Imoto, S.; Sugimachi, K.; Ishii, H.; Iwatsuki, M.; Ota, D.; Ohkuma, M.; Iwaya, T.; et al. Plastin3 Is a Novel Marker for Circulating Tumor Cells Undergoing the Epithelial–Mesenchymal Transition and Is Associated with Colorectal Cancer Prognosis. Cancer Res. 2013, 73, 2059–2069. [Google Scholar] [CrossRef] [Green Version]
- Bitting, R.L.; Boominathan, R.; Rao, C.; Kemeny, G.; Foulk, B.; Garcia-Blanco, M.A.; Connelly, M.; Armstrong, A.J. Development of a Method to Isolate Circulating Tumor Cells Using Mesenchymal-Based Capture. Methods 2013, 64, 129–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Werner, S.; Stenzl, A.; Pantel, K.; Todenhöfer, T. Expression of Epithelial Mesenchymal Transition and Cancer Stem Cell Markers in Circulating Tumor Cells. In Isolation and Molecular Characterization of Circulating Tumor Cells; Magbanua, M.J.M., Park, J.W., Eds.; Springer International Publishing: Cham, Switzerland, 2017; Volume 994, pp. 205–228. ISBN 978-3-319-55946-9. [Google Scholar]
- Bruland, O.S. Hematogenous Micrometastases in Osteosarcoma Patients. Clin. Cancer Res. 2005, 11, 4666–4673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorlick, R.; Anderson, P.; Andrulis, I.; Arndt, C.; Beardsley, G.P.; Bernstein, M.; Bridge, J.; Cheung, N.-K.; Dome, J.S.; Ebb, D.; et al. Biology of Childhood Osteogenic Sarcoma and Potential Targets for Therapeutic Development: Meeting Summary. Clin. Cancer Res. 2003, 9, 5442–5453. [Google Scholar]
- Bozzi, F.; Collini, P.; Aiello, A.; Barzanò, E.; Gambirasio, F.; Podda, M.; Meazza, C.; Ferrari, A.; Luksch, R. Flow Cytometric Phenotype of Rhabdomyosarcoma Bone Marrow Metastatic Cells and Its Implication in Differential Diagnosis with Neuroblastoma. Anticancer Res. 2008, 28, 1565–1569. [Google Scholar]
- Michelagnoli, M.P.; Burchill, S.A.; Cullinane, C.; Selby, P.J.; Lewis, I.J. Myogenin—A More Specific Target for RT-PCR Detection of Rhabdomyosarcoma than MyoD1. Med. Pediatr. Oncol. 2003, 40, 1–8. [Google Scholar] [CrossRef]
- Swerts, K.; De Moerloose, B.; Dhooge, C.; Brichard, B.; Benoit, Y.; Laureys, G.; Philippé, J. Detection of Residual Neuroblastoma Cells in Bone Marrow: Comparison of Flow Cytometry with Immunocytochemistry: Flow Cytometric Detection of Residual Neuroblastoma Cells. Cytometry 2004, 61B, 9–19. [Google Scholar] [CrossRef]
- Bian, Z.-Y.; Li, G.; Gan, Y.-K.; Hao, Y.-Q.; Xu, W.-T.; Tang, T.-T. Increased Number of Mesenchymal Stem Cell-like Cells in Peripheral Blood of Patients with Bone Sarcomas. Arch. Med. Res. 2009, 40, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Deans, R.J.; Moseley, A.B. Mesenchymal Stem Cells. Exp. Hematol. 2000, 28, 875–884. [Google Scholar] [CrossRef]
- Coltella, N.; Manara, M.C.; Cerisano, V.; Trusolino, L.; Di Renzo, M.F.; Scotlandi, K.; Ferracini, R. Role of the MET/HGF Receptor in Proliferation and Invasive Behavior of Osteosarcoma. FASEB J. 2003, 17, 1162–1164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, H.; Shabbir, A.; Molnar, M.; Yang, J.; Marion, S.; Canty, J.M.; Lee, T. Adenoviral Expression of Vascular Endothelial Growth Factor Splice Variants Differentially Regulate Bone Marrow-Derived Mesenchymal Stem Cells. J. Cell. Physiol. 2008, 216, 458–468. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, Y.; Nishimura, M.; Sekiya, K.; Suehiro, F.; Kanawa, M.; Nikawa, H.; Hamada, T.; Kato, Y. Comprehensive Analysis of Chemotactic Factors for Bone Marrow Mesenchymal Stem Cells. Stem Cells Dev. 2007, 16, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Van Mater, D.; Wagner, L. Management of Recurrent Ewing Sarcoma: Challenges and Approaches. Onco Targets Ther. 2019, 12, 2279–2288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dworzak, M.N.; Fritsch, G.; Buchinger, P.; Fleischer, C.; Printz, D.; Zellner, A.; Schöllhammer, A.; Steiner, G.; Ambros, P.F.; Gadner, H. Flow Cytometric Assessment of Human MIC2 Expression in Bone Marrow, Thymus, and Peripheral Blood. Blood 1994, 83, 415–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shah, V.O.; Civin, C.I.; Loken, M.R. Flow Cytometric Analysis of Human Bone Marrow. IV. Differential Quantitative Expression of T-200 Common Leukocyte Antigen during Normal Hemopoiesis. J. Immunol. 1988, 140, 1861–1867. [Google Scholar]
- Zietz, C.; Rössle, M.; Haas, C.; Sendelhofert, A.; Hirschmann, A.; Stürzl, M.; Löhrs, U. MDM-2 Oncoprotein Overexpression, P53 Gene Mutation, and VEGF Up-Regulation in Angiosarcomas. Am. J. Pathol. 1998, 153, 1425–1433. [Google Scholar] [CrossRef] [Green Version]
- Ladanyi, M.; Cha, C.; Lewis, R.; Jhanwar, S.C.; Huvos, A.G.; Healey, J.H. MDM2 Gene Amplification in Metastatic Osteosarcoma. Cancer Res. 1993, 53, 16–18. [Google Scholar]
- Yokoyama, R.; Schneider-Stock, R.; Radig, K.; Wex, T.; Roessner, A. Clinicopathologiclmplications of MDM2, P53 and K-Ras Gene Alterations in Osteosarcomas: MDM2 Amplification and P53 Mutations Found in Progressive Tumors. Pathol. Res. Pract. 1998, 194, 615–621. [Google Scholar] [CrossRef]
- Li, H.; Meng, Q.H.; Noh, H.; Batth, I.S.; Somaiah, N.; Torres, K.E.; Xia, X.; Wang, R.; Li, S. Detection of Circulating Tumor Cells from Cryopreserved Human Sarcoma Peripheral Blood Mononuclear Cells. Cancer Lett. 2017, 403, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Satelli, A.; Batth, I.S.; Brownlee, Z.; Rojas, C.; Meng, Q.H.; Kopetz, S.; Li, S. Potential Role of Nuclear PD-L1 Expression in Cell-Surface Vimentin Positive Circulating Tumor Cells as a Prognostic Marker in Cancer Patients. Sci Rep. 2016, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, S.; Asahara, H. The Myogenic Transcriptional Network. Cell. Mol. Life Sci. 2011, 68, 1843–1849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frascella, E.; Toffolatti, L.; Rosolen, A. Normal and Rearranged PAX3 Expression in Human Rhabdomyosarcoma. Cancer Genet. Cytogenet. 1998, 102, 104–109. [Google Scholar] [CrossRef]
- Schulte, T.W.; Toretsky, J.A.; Ress, E.; Helman, L.; Neckers, L.M. Expression of PAX3 in Ewing’s Sarcoma Family of Tumors. Biochem. Mol. Med. 1997, 60, 121–126. [Google Scholar] [CrossRef]
- Almazán-Moga, A.; Roma, J.; Molist, C.; Vidal, I.; Jubierre, L.; Soriano, A.; Segura, M.F.; Llort, A.; de Toledo, J.S.; Gallego, S. Optimization of Rhabdomyosarcoma Disseminated Disease Assessment by Flow Cytometry: MRD Assessment in Rhabdomyosarcoma. Cytometry 2014, 85, 1020–1029. [Google Scholar] [CrossRef]
- Bertucci, F.; Finetti, P.; Mamessier, E.; Pantaleo, M.A.; Astolfi, A.; Ostrowski, J.; Birnbaum, D. PDL1 Expression Is an Independent Prognostic Factor in Localized GIST. OncoImmunology 2015, 4, e1002729. [Google Scholar] [CrossRef] [Green Version]
- Sabatier, R.; Finetti, P.; Mamessier, E.; Adelaide, J.; Chaffanet, M.; Ali, H.R.; Viens, P.; Caldas, C.; Birnbaum, D.; Bertucci, F. Prognostic and Predictive Value of PDL1 Expression in Breast Cancer. Oncotarget 2015, 6, 5449–5464. [Google Scholar] [CrossRef] [Green Version]
- Hasan, A.; Ghebeh, H.; Lehe, C.; Ahmad, R.; Dermime, S. Therapeutic Targeting of B7-H1 in Breast Cancer. Expert Opin. Ther. Targets 2011, 15, 1211–1225. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Wu, D.; Li, L.; Chai, Y.; Huang, J. PD-L1 and Survival in Solid Tumors: A Meta-Analysis. PLoS ONE 2015, 10, e0131403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Gao, P.; Xiao, X.; Heger, M.; Geng, L.; Fan, B.; Yuan, Y.; Huang, C.; Chen, G.; Liu, Y.; et al. A Liquid Biopsy-Based Method for the Detection and Quantification of Circulating Tumor Cells in Surgical Osteosarcoma Patients. Int. J. Oncol. 2017, 50, 1075–1086. [Google Scholar] [CrossRef] [PubMed]
- Vo, K.T.; Edwards, J.V.; Epling, C.L.; Sinclair, E.; Hawkins, D.S.; Grier, H.E.; Janeway, K.A.; Barnette, P.; McIlvaine, E.; Krailo, M.D.; et al. Impact of Two Measures of Micrometastatic Disease on Clinical Outcomes in Patients with Newly Diagnosed Ewing Sarcoma: A Report from the Children’s Oncology Group. Clin. Cancer Res. 2016, 22, 3643–3650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loschi, S.; Dufour, C.; Oberlin, O.; Goma, G.; Valteau-Couanet, D.; Gaspar, N. Tandem High-Dose Chemotherapy Strategy as First-Line Treatment of Primary Disseminated Multifocal Ewing Sarcomas in Children, Adolescents and Young Adults. Bone Marrow Transplant. 2015, 50, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Smerage, J.B.; Barlow, W.E.; Hortobagyi, G.N.; Winer, E.P.; Leyland-Jones, B.; Srkalovic, G.; Tejwani, S.; Schott, A.F.; O’Rourke, M.A.; Lew, D.L.; et al. Circulating Tumor Cells and Response to Chemotherapy in Metastatic Breast Cancer: SWOG S0500. JCO 2014, 32, 3483–3489. [Google Scholar] [CrossRef]
- Vona, G.; Sabile, A.; Louha, M.; Sitruk, V.; Romana, S.; Schütze, K.; Capron, F.; Franco, D.; Pazzagli, M.; Vekemans, M.; et al. Isolation by Size of Epithelial Tumor Cells. Am. J. Pathol. 2000, 156, 57–63. [Google Scholar] [CrossRef]
- Khoja, L.; Backen, A.; Sloane, R.; Menasce, L.; Ryder, D.; Krebs, M.; Board, R.; Clack, G.; Hughes, A.; Blackhall, F.; et al. A Pilot Study to Explore Circulating Tumour Cells in Pancreatic Cancer as a Novel Biomarker. Br. J. Cancer 2012, 106, 508–516. [Google Scholar] [CrossRef]
- Hofman, V.; Bonnetaud, C.; Ilie, M.I.; Vielh, P.; Vignaud, J.M.; Fléjou, J.F.; Lantuejoul, S.; Piaton, E.; Mourad, N.; Butori, C.; et al. Preoperative Circulating Tumor Cell Detection Using the Isolation by Size of Epithelial Tumor Cell Method for Patients with Lung Cancer Is a New Prognostic Biomarker. Clin. Cancer Res. 2011, 17, 827–835. [Google Scholar] [CrossRef] [Green Version]
- Hofman, V.; Ilie, M.I.; Long, E.; Selva, E.; Bonnetaud, C.; Molina, T.; Vénissac, N.; Mouroux, J.; Vielh, P.; Hofman, P. Detection of Circulating Tumor Cells as a Prognostic Factor in Patients Undergoing Radical Surgery for Non-Small-Cell Lung Carcinoma: Comparison of the Efficacy of the CellSearch AssayTM and the Isolation by Size of Epithelial Tumor Cell Method. Int. J. Cancer 2011, 129, 1651–1660. [Google Scholar] [CrossRef]
- Chinen, L.; Mello, C.; Abdallah, E.; Ocea, L.; Buim, M.; Mingues, N.; Gasparini, J.L., Jr.; Fanelli, M.; Paterlini, P. Isolation, Detection, and Immunomorphological Characterization of Circulating Tumor Cells (CTCs) from Patients with Different Types of Sarcoma Using Isolation by Size of Tumor Cells: A Window on Sarcoma-Cell Invasion. OTT 2014, 1609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friedl, P.; Gilmour, D. Collective Cell Migration in Morphogenesis, Regeneration and Cancer. Nat. Rev. Mol. Cell Biol. 2009, 10, 445–457. [Google Scholar] [CrossRef]
- Krebs, M.G.; Hou, J.-M.; Sloane, R.; Lancashire, L.; Priest, L.; Nonaka, D.; Ward, T.H.; Backen, A.; Clack, G.; Hughes, A.; et al. Analysis of Circulating Tumor Cells in Patients with Non-Small Cell Lung Cancer Using Epithelial Marker-Dependent and -Independent Approaches. J. Thorac. Oncol. 2012, 7, 306–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hofman, V.J.; Ilie, M.I.; Bonnetaud, C.; Selva, E.; Long, E.; Molina, T.; Vignaud, J.M.; Fléjou, J.F.; Lantuejoul, S.; Piaton, E.; et al. Cytopathologic Detection of Circulating Tumor Cells Using the Isolation by Size of Epithelial Tumor Cell Method: Promises and Pitfalls. Am. J. Clin. Pathol. 2011, 135, 146–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rhim, A.D.; Mirek, E.T.; Aiello, N.M.; Maitra, A.; Bailey, J.M.; McAllister, F.; Reichert, M.; Beatty, G.L.; Rustgi, A.K.; Vonderheide, R.H.; et al. EMT and Dissemination Precede Pancreatic Tumor Formation. Cell 2012, 148, 349–361. [Google Scholar] [CrossRef] [Green Version]
- Hayashi, M.; Zhu, P.; McCarty, G.; Meyer, C.F.; Pratilas, C.A.; Levin, A.; Morris, C.D.; Albert, C.M.; Jackson, K.W.; Tang, C.-M.; et al. Size-Based Detection of Sarcoma Circulating Tumor Cells and Cell Clusters. Oncotarget 2017, 8, 78965–78977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, V.; Jafferji, I.; Garza, M.; Melnikova, V.O.; Hasegawa, D.K.; Pethig, R.; Davis, D.W. ApoStreamTM, a New Dielectrophoretic Device for Antibody Independent Isolation and Recovery of Viable Cancer Cells from Blood. Biomicrofluidics 2012, 6, 024133. [Google Scholar] [CrossRef] [Green Version]
- Gascoyne, P.; Shim, S. Isolation of Circulating Tumor Cells by Dielectrophoresis. Cancers 2014, 6, 545–579. [Google Scholar] [CrossRef] [Green Version]
- Gascoyne, P.R.C.; Shim, S.; Noshari, J.; Becker, F.F.; Stemke-Hale, K. Correlations between the Dielectric Properties and Exterior Morphology of Cells Revealed by Dielectrophoretic Field-Flow Fractionation: General. Electrophoresis 2013, 34, 1042–1050. [Google Scholar] [CrossRef]
- Balasubramanian, P.; Kinders, R.J.; Kummar, S.; Gupta, V.; Hasegawa, D.; Menachery, A.; Lawrence, S.M.; Wang, L.; Ferry-Galow, K.; Davis, D.; et al. Antibody-Independent Capture of Circulating Tumor Cells of Non-Epithelial Origin with the ApoStream® System. PLoS ONE 2017, 12, e0175414. [Google Scholar] [CrossRef] [Green Version]
- Foo, W.C.; Cruise, M.W.; Wick, M.R.; Hornick, J.L. Immunohistochemical Staining for TLE1 Distinguishes Synovial Sarcoma From Histologic Mimics. Am. J. Clin. Pathol. 2011, 135, 839–844. [Google Scholar] [CrossRef]
- Watanabe, M.; Serizawa, M.; Sawada, T.; Takeda, K.; Takahashi, T.; Yamamoto, N.; Koizumi, F.; Koh, Y. A Novel Flow Cytometry-Based Cell Capture Platform for the Detection, Capture and Molecular Characterization of Rare Tumor Cells in Blood. J. Transl. Med. 2014, 12, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasegawa, N.; Takeda Nakamura, I.; Ueno, T.; Kojima, S.; Kawazu, M.; Akaike, K.; Okubo, T.; Takagi, T.; Suehara, Y.; Hayashi, T.; et al. Detection of Circulating Sarcoma Tumor Cells Using a Microfluidic Chip-Type Cell Sorter. Sci. Rep. 2019, 9, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Lucci, A.; Hall, C.S.; Lodhi, A.K.; Bhattacharyya, A.; Anderson, A.E.; Xiao, L.; Bedrosian, I.; Kuerer, H.M.; Krishnamurthy, S. Circulating Tumour Cells in Non-Metastatic Breast Cancer: A Prospective Study. Lancet Oncol. 2012, 13, 688–695. [Google Scholar] [CrossRef]
- Pantel, K.; Speicher, M.R. The Biology of Circulating Tumor Cells. Oncogene 2016, 35, 1216–1224. [Google Scholar] [CrossRef] [PubMed]
- Alix-Panabières, C.; Pantel, K. Clinical Applications of Circulating Tumor Cells and Circulating Tumor DNA as Liquid Biopsy. Cancer Discov. 2016, 6, 479–491. [Google Scholar] [CrossRef] [Green Version]
- Pantel, K.; Alix-Panabières, C. Liquid Biopsy: Potential and Challenges. Mol. Oncol. 2016, 10, 371–373. [Google Scholar] [CrossRef] [Green Version]
- Chang, L.; Asatrian, G.; Dry, S.M.; James, A.W. Circulating Tumor Cells in Sarcomas: A Brief Review. Med. Oncol. 2015, 32, 430. [Google Scholar] [CrossRef]
- Martín-Broto, J.; Pousa, A.L.; Brohl, A.S.; Van Tine, B.A.; Powers, B.; Stacchiotti, S.; Blay, J.-Y.; Hu, J.S.; Oakley, G.J.; Wang, H.; et al. Circulating Tumor Cells and Biomarker Modulation with Olaratumab Monotherapy Followed by Olaratumab plus Doxorubicin: Phase Ib Study in Patients with Soft-Tissue Sarcoma. Mol. Cancer Ther. 2021, 20, 132–141. [Google Scholar] [CrossRef]
- Ye, Z.; Ding, Y.; Chen, Z.; Li, Z.; Ma, S.; Xu, Z.; Cheng, L.; Wang, X.; Zhang, X.; Ding, N.; et al. Detecting and Phenotyping of Aneuploid Circulating Tumor Cells in Patients with Various Malignancies. Cancer Biol. Ther. 2019, 20, 546–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giam, M.; Rancati, G. Aneuploidy and Chromosomal Instability in Cancer: A Jackpot to Chaos. Cell Div. 2015, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Dürrbaum, M.; Storchová, Z. Effects of Aneuploidy on Gene Expression: Implications for Cancer. FEBS J. 2016, 283, 791–802. [Google Scholar] [CrossRef] [PubMed]
- Alvegard, T.A.; Berg, N.O.; Baldetorp, B.; Fernö, M.; Killander, D.; Ranstam, J.; Rydholm, A.; Akerman, M. Cellular DNA Content and Prognosis of High-Grade Soft Tissue Sarcoma: The Scandinavian Sarcoma Group Experience. JCO 1990, 8, 538–547. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; Minelli, A.; Santini, D.; Tonini, G.; Vincenzi, B. Identification of Aneuploid Circulating Tumor Cells in Soft-Tissue Sarcoma Patients: A Pilot Study. Oncology 2020, 98, 893–896. [Google Scholar] [CrossRef] [PubMed]
- Chibon, F.; Lagarde, P.; Salas, S.; Pérot, G.; Brouste, V.; Tirode, F.; Lucchesi, C.; de Reynies, A.; Kauffmann, A.; Bui, B.; et al. Validated Prediction of Clinical Outcome in Sarcomas and Multiple Types of Cancer on the Basis of a Gene Expression Signature Related to Genome Complexity. Nat. Med. 2010, 16, 781–787. [Google Scholar] [CrossRef] [PubMed]
- Hynes, N.E.; Lane, H.A. ERBB Receptors and Cancer: The Complexity of Targeted Inhibitors. Nat. Rev. Cancer 2005, 5, 341–354. [Google Scholar] [CrossRef]
- Braun, A.C.; de Mello, C.A.L.; Corassa, M.; Abdallah, E.A.; Urvanegia, A.C.; Alves, V.S.; Flores, B.C.T.C.P.; Díaz, M.; Nicolau, U.R.; e Silva, V.S.; et al. EGFR Expression in Circulating Tumor Cells from High-Grade Metastatic Soft Tissue Sarcomas. Cancer Biol. Ther. 2018, 19, 454–460. [Google Scholar] [CrossRef] [Green Version]
- Sato, O.; Wada, T.; Kawai, A.; Yamaguchi, U.; Makimoto, A.; Kokai, Y.; Yamashita, T.; Chuman, H.; Beppu, Y.; Tani, Y.; et al. Expression of Epidermal Growth Factor Receptor, ERBB2 And KIT in Adult Soft Tissue Sarcomas: A Clinicopathologic Study of 281 Cases. Cancer 2005, 103, 1881–1890. [Google Scholar] [CrossRef]
- Yang, J.-L.; Hannan, M.T.; Russell, P.J.; Crowe, P.J. Expression of HER1/EGFR Protein in Human Soft Tissue Sarcomas. Eur. J. Surg. Oncol. 2006, 32, 466–468. [Google Scholar] [CrossRef]
- Benini, S.; Gamberi, G.; Cocchi, S.; Garbetta, J.; Alberti, L.; Righi, A.; Gambarotti, M.; Picci, P.; Ferrari, S. Detection of Circulating Tumor Cells in Liquid Biopsy from Ewing Sarcoma Patients. Cancer Manag. Res. 2018, 10, 49–60. [Google Scholar] [CrossRef] [Green Version]
- Jürgens, H.; Treuner, J.; Winkler, K.; Göbel, U. Ifosfamide in Pediatric Malignancies. Semin. Oncol. 1989, 16, 46–50. [Google Scholar] [PubMed]
- Chalopin, A.; Tellez-Gabriel, M.; Brown, H.K.; Vallette, F.; Heymann, M.-F.; Gouin, F.; Heymann, D. Isolation of Circulating Tumor Cells in a Preclinical Model of Osteosarcoma: Effect of Chemotherapy. J. Bone Oncol. 2018, 12, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Luetke, A.; Meyers, P.A.; Lewis, I.; Juergens, H. Osteosarcoma Treatment–Where Do We Stand? A State of the Art Review. Cancer Treat. Rev. 2014, 40, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Satelli, A.; Brownlee, Z.; Mitra, A.; Meng, Q.H.; Li, S. Circulating Tumor Cell Enumeration with a Combination of Epithelial Cell Adhesion Molecule– and Cell-Surface Vimentin–Based Methods for Monitoring Breast Cancer Therapeutic Response. Clin. Chem. 2015, 61, 259–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Meng, Q.H.; Noh, H.; Somaiah, N.; Torres, K.E.; Xia, X.; Batth, I.S.; Joseph, C.P.; Liu, M.; Wang, R.; et al. Cell-Surface Vimentin–Positive Macrophage-like Circulating Tumor Cells as a Novel Biomarker of Metastatic Gastrointestinal Stromal Tumors. OncoImmunology 2018, 7, e1420450. [Google Scholar] [CrossRef] [PubMed]
- Ries, C.H.; Cannarile, M.A.; Hoves, S.; Benz, J.; Wartha, K.; Runza, V.; Rey-Giraud, F.; Pradel, L.P.; Feuerhake, F.; Klaman, I.; et al. Targeting Tumor-Associated Macrophages with Anti-CSF-1R Antibody Reveals a Strategy for Cancer Therapy. Cancer Cell 2014, 25, 846–859. [Google Scholar] [CrossRef] [Green Version]
- Hamilton, G.; Rath, B.; Klameth, L.; Hochmair, M.J. Small Cell Lung Cancer: Recruitment of Macrophages by Circulating Tumor Cells. OncoImmunology 2016, 5, e1093277. [Google Scholar] [CrossRef] [Green Version]
- Cohen, S.J.; Punt, C.J.A.; Iannotti, N.; Saidman, B.H.; Sabbath, K.D.; Gabrail, N.Y.; Picus, J.; Morse, M.; Mitchell, E.; Miller, M.C.; et al. Relationship of Circulating Tumor Cells to Tumor Response, Progression-Free Survival, and Overall Survival in Patients with Metastatic Colorectal Cancer. JCO 2008, 26, 3213–3221. [Google Scholar] [CrossRef]
- Schulze, K.; Gasch, C.; Staufer, K.; Nashan, B.; Lohse, A.W.; Pantel, K.; Riethdorf, S.; Wege, H. Presence of EpCAM-Positive Circulating Tumor Cells as Biomarker for Systemic Disease Strongly Correlates to Survival in Patients with Hepatocellular Carcinoma: EpCAM-Positive CTCs and Hepatocellular Carcinoma. Int. J. Cancer 2013, 133, 2165–2171. [Google Scholar] [CrossRef]
- Heidrich, I.; Ačkar, L.; Mossahebi Mohammadi, P.; Pantel, K. Liquid Biopsies: Potential and Challenges. Int. J. Cancer 2021, 148, 528–545. [Google Scholar] [CrossRef]
| Clinical Trial ID. | Title | Status | Study Results | Tumor Types | Clinical Intervention | Phase | No. Subjects | Study Type |
|---|---|---|---|---|---|---|---|---|
| NCT02983539 | Detection of Circulating Tumor Cells in Patients with Sarcomas | Unknown status | No Results Available | Leiomyosarcoma, Pleomorphic Liposarcoma, Synovial Sarcoma, Liposarcoma | 20 | Observational | ||
| NCT03357315 | Mix Vaccine for Metastatic Sarcoma Patients | Completed | No Results Available | Metastatic Sarcoma | Biological: Mix vaccine | Phase 1, Phase 2 | 30 | Interventional |
| NCT02849366 | Combination of Cryosurgery and NK Immunotherapy for Recurrent Sarcoma | Completed | No Results Available | Recurrent Adult Soft Tissue Sarcoma | Device: Cryosurgery Biological: NK immunotherapy | Phase 1, Phase 2 | 30 | Interventional |
| NCT02783599 | A Study of Olaratumab (LY3012207) in Participants with Soft Tissue Sarcoma | Completed | Has Results | Soft Tissue Sarcoma | Drug: Olaratumab, Doxorubicin External Beam Radiotherapy | Phase 1 | 51 | Interventional |
| NCT04239443 | Clinical Study of PD-1 Monoclonal Antibody SHR-1210 and Apatinib in Advanced NSCLC, Soft Tissue Sarcoma, and Uterine Cancer | Recruiting | No Results Available | Advanced Non-Small Cell Lung Cancer, Uterine Cancer, Soft Tissue Sarcoma | Drug: PD-1 inhibitor, Apatinib | Phase 2 | 120 | Interventional |
| NCT01831609 | Biomarker Analysis of Solid Cancers Such as Gastrointestinal Cancer | Recruiting | No Results Available | Sarcoma | 1000 | Observational | ||
| NCT04628806 | Heat Shock Protein (HSP) 70 to Quantify and Characterize Circulating Tumor Cells | Not yet recruiting | No Results Available | Melanoma Stage IV, Sarcoma, Squamous Cell Carcinoma, Pancreatic Cancer Stage IV, Prostate Cancer, Breast Cancer Stage IV | Diagnostic Test: CTC isolation by HSP70 | 120 | Observational | |
| NCT03011528 | First-line Treatment of Ewing Tumors with Primary Extrapulmonary Dissemination in Patients From 2 to 50 Years | Recruiting | No Results Available | Ewing Sarcoma Family of Tumors | Drug: VDC-IE x2, VDC-IE, TEMIRI, BuMel Local treatment by surgery or radiotherapy | Phase 2 | 45 | Interventional |
| NCT03818412 | Circulating Tumor DNA in Soft Tissue Sarcoma | Recruiting | No Results Available | Soft Tissue Sarcoma | Procedure: tumor tissue collection and blood draws | Not Applicable | 40 | Interventional |
| NCT02859415 | Continuous 24h Intravenous Infusion of Mithramycin, an Inhibitor of Cancer Stem Cell Signaling, in People with Primary Thoracic Malignancies or Carcinomas, Sarcomas or Germ Cell Neoplasms with Pleuropulmonary Metastases | Recruiting | No Results Available | Esophageal Neoplasms, Lung Neoplasms, Mesothelioma, Thymus Neoplasms, Neoplasms, Germ Cell, and Embryonal | Drug: Mithramycin | Phase 1, Phase 2 | 60 | Interventional |
| NCT00474760 | Study of Anti-IGF-IR CP-751,871 In Patients With Solid Tumors | Completed | Has Results | Sarcoma, Ewing’s | Drug: CP-751,871 | Phase 1 | 65 | Interventional |
| NCT03085225 | Trabectedin Combined with Durvalumab in Patients with Advanced Pretreated Soft-tissue Sarcomas and Ovarian Carcinomas. | Active, not recruiting | No Results Available | Ovarian Carcinoma, Soft Tissue Sarcoma | Drug: Combination of trabectedin with durvalumab | Phase 1 | 50 | Interventional |
| NCT02636725 | Axitinib and Pembrolizumab in Subjects with Advanced Alveolar Soft Part Sarcoma and Other Soft Tissue Sarcomas | Active, not recruiting | No Results Available | Alveolar Soft Part Sarcoma, Soft Tissue Sarcomas | Drug: Axitinib, Pembrolizumab Blood Draw, Tumor Specimen Collection | Phase 2 | 33 | Interventional |
| NCT03946943 | Study of Anlotinib Hydrochloride and Toripalimab in Subjects with Unresectable or Metastatic Undifferentiated Pleomorphic Sarcoma | Not yet recruiting | No Results Available | Soft Tissue Sarcomas, Undifferentiated Pleomorphic Sarcoma | Drug: Anlotinib, Toripalimab Blood Draw, Tumor Specimen Collection | Phase 2 | 25 | Interventional |
| NCT01528774 | Culture and Characterization of Circulating Tumor Cells (CTC) in Melanoma and Other Cancers | Completed | No Results Available | Melanoma | Blood Draw | 150 | Observational | |
| NCT03600649 | Clinical Trial of SP-2577 (Seclidemstat) in Patients with Relapsed or Refractory Ewing Sarcoma | Recruiting | No Results Available | Ewing Sarcoma | Drug: SP-2577 | Phase 1 | 50 | Interventional |
| NCT04052334 | Lymphodepletion Plus Adoptive Cell Therapy with High Dose IL-2 in Adolescent and Young Adult Patients With Soft Tissue Sarcoma | Recruiting | No Results Available | Sarcoma | Drug: TIL, Interleukin-2, Fludarabine, Cyclophosphamide | Phase 1 | 15 | Interventional |
| NCT01222767 | Study of Zalypsis (PM00104) in Patients with Unresectable Locally Advanced and/or Metastatic Ewing Family of Tumors (EFT) Progressing After at Least One Prior Line of Chemotherapy | Completed | No Results Available | Ewing’s Sarcoma, Primitive Neuroectodermal Tumor (PNET), Askin’s Tumor of the Chest Wall, Extraosseous Ewing’s Sarcoma (EOE) | Drug: Zalypsis | Phase 2 | 17 | Interventional |
| NCT00588510 | Detection of Circulating Osteosarcoma Tumor Cells in the Blood of Patients Using the Polymerase Chain Reaction | Completed | No Results Available | Osteosarcoma | Blood draw | 59 | Observational | |
| NCT04512495 | Circulating “Cancer Cells/Macrophage” Hybrid Cells in Patients With Sarcoma? | Recruiting | No Results Available | Sarcoma | Blood draw | Not Applicable | 60 | Interventional |
| NCT03570437 | Does Cediranib With Paclitaxel, or Cediranib and Olaparib, Treat Advanced Endometrial Cancer Better Than Paclitaxel? | Recruiting | No Results Available | Carcinosarcoma, Endometrial Neoplasms | Drug: Paclitaxel, Cediranib, Olaparib | Phase 2 | 129 | Interventional |
| NCT01804634 | Reduced Intensity Haploidentical BMT for High-Risk Solid Tumors | Recruiting | No Results Available | Refractory and/or Relapsed Metastatic Solid Tumors | Drug: Cyclophosphamide, Fludarabine, Melphalan, Tacrolimus Low dose total body irradiation | Phase 2 | 60 | Interventional |
| NCT04214457 | Development of a Predictive Model for Early Differential Diagnosis of Uterine Leiomyomas and Leiomyosarcomas | Recruiting | No Results Available | Leiomyoma, Leiomyosarcoma | Biopsy and peripheral blood collection | 1000 | Observational | |
| NCT00898781 | Study of Circulating Cancer Cells in Patients with Metastatic Breast, Ovarian, Colon, or Pancreatic Cancer | Terminated | No Results Available | Breast Cancer, Colorectal Cancer, Ovarian Cancer, Pancreatic Cancer | 80 | Observational |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agnoletto, C.; Caruso, C.; Garofalo, C. Heterogeneous Circulating Tumor Cells in Sarcoma: Implication for Clinical Practice. Cancers 2021, 13, 2189. https://doi.org/10.3390/cancers13092189
Agnoletto C, Caruso C, Garofalo C. Heterogeneous Circulating Tumor Cells in Sarcoma: Implication for Clinical Practice. Cancers. 2021; 13(9):2189. https://doi.org/10.3390/cancers13092189
Chicago/Turabian StyleAgnoletto, Chiara, Chiara Caruso, and Cecilia Garofalo. 2021. "Heterogeneous Circulating Tumor Cells in Sarcoma: Implication for Clinical Practice" Cancers 13, no. 9: 2189. https://doi.org/10.3390/cancers13092189






