Simple Summary
Immune-modulating cancer treatments have proved to be highly effective in a wide range of tumour types. They interrupt the usual communication between cells in the immune system, encouraging them to become more active in identifying and destroying cancer cells. Although these therapies are very successful in treating cancer, patients frequently experience liver injury as a side effect related to over activation of the immune system. If cancer patients develop this side effect, they need to stop their cancer therapy and be given strong immunosuppressants. Researchers are now working on understanding the mechanisms involved in the development of liver inflammation. In this review we will summarise findings identifying classes of immune cells that are of particular importance in this context and highlight ways in which we can use this knowledge to improve the safety of these new cancer drugs.
Abstract
Drug-related hepatotoxicity is an emerging clinical challenge with the widening use of immunotherapeutic agents in the field of oncology. This is an important complication to consider as more immune oncological targets are being identified to show promising results in clinical trials. The application of these therapeutics may be complicated by the development of immune-related adverse events (irAEs), a serious limitation often requiring high-dose immunosuppression and discontinuation of cancer therapy. Hepatoxicity presents one of the most frequently encountered irAEs and a better understanding of the underlying mechanism is crucial for the development of alternative therapeutic interventions. As a novel drug side effect, the immunopathogenesis of the condition is not completely understood. In the liver, myeloid cells play a central role in the maintenance of homeostasis and promotion of inflammation. Recent research has identified myeloid cells to be associated with hepatic adverse events of various immune modulatory monoclonal antibodies. In this review article, we provide an overview of the role of myeloid cells in the immune pathogenesis during hepatoxicity related to cancer immunotherapies and highlight potential treatment options.
1. Introduction
Drug-related hepatotoxicity in the context of cancer therapy is a frequently encountered adverse event. Immunotherapy is a class of novel cancer treatment utilising the host’s immune system with the aim of re-programming effector cells to enhance their anti-tumour immune responses [1,2,3]. These include, for example: blocking immune cell checkpoints such as CTLA-4, PD-1 and its ligand PD-L1; the activation of co-stimulatory pathways (e.g., CD40, ICOS, OX40 and 4-1BB agonist); and manipulation of immunometabolism (IDO1 inhibitors). This therapeutic strategy has proven efficacy in a number of solid organ and haematological malignancies [1,4,5].
Unfortunately, the efficacy of these agents is associated with autoimmune-like inflammatory side effects, termed immune-related adverse events (irAEs) in a large proportion of patients [6,7,8]. These novel drug side effects manifest as tissue destructive immune-mediated toxicity, which can affect any organ [9]. Common irAEs include colitis, dermatitis and hepatitis [7,8,9,10] and are classified according to the Common Terminology Criteria for Adverse Events (CTCAE) grading system ranging from 1–5 in ascending severity, with 5 being fatality. Hepatotoxicity is among the most frequently encountered irAEs (see Figure 1 for main immunotherapy regimens and their targets associated with hepatotoxicity) [7,10,11,12,13]. The development of drug-related hepatoxicity frequently requires the interruption or permanent cessation of immunotherapy. While the immunopathogenesis is not completely understood, there is emerging evidence for the involvement of myeloid cells, in particular monocytes and macrophages [14,15,16,17].
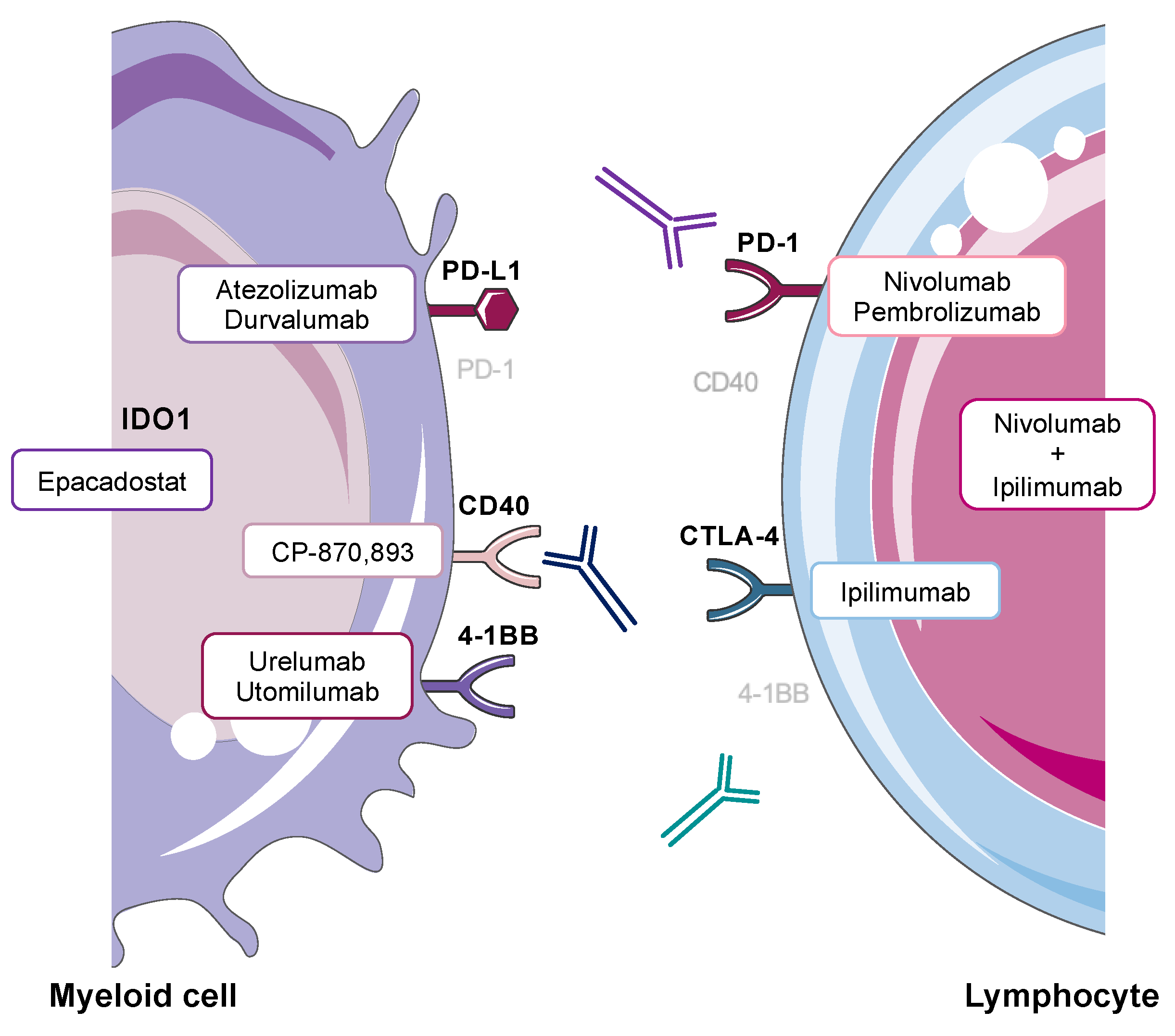
Figure 1.
Myeloid and lymphoid cell expression of key targets of cancer immunotherapy associated with hepatotoxicity [5,18,19,20,21,22,23]. Antagonist monoclonal antibody therapies include Nivolumab and Pembrolizumab (anti-PD-1), Atezolizumab and Durvalumab (anti-PD-L1), Ipilimumab (anti-CTLA-4) and combination Nivolumab and Ipilimumab (anti-PD-1 + anti-CTLA-4). Agonist monoclonal antibody therapies are CP-870,893 (anti-CD40), Urelumab and Utomilumab (anti-4-1BB). Small molecule inhibitors targeting IDO1 include Epacadostat.
In this review we will summarise our current understanding of the maintenance of immune tolerance in liver homeostasis, recognised pathways to liver inflammation when tolerance is broken, the role of immunotherapy in mediating breakdown of tolerance and the involvement of myeloid cells in related hepatotoxicity.
2. Liver Function during Homeostasis
The liver is uniquely perfused with mixed arterio-venous blood. The dual blood supply exposes the liver to high levels of microbial and dietary products coming from the gastrointestinal tract via the portal vein. This exposure, coupled with tissue remodelling and metabolic functions of the liver, necessitates a distinct immune privileged environment. In order to prevent excessive activation of immune cells triggered by this tonic exposure, the liver is biased towards immune unresponsiveness [24,25].
Mechanisms of Liver Immune Tolerance
During homeostasis, tolerance suppresses the initiation of inflammation against self and non-self antigenic proteins [26]. Hepatic tolerance is mediated by various suppressive mechanisms, including reprogramming of immune cell function and the presence of immunosuppressive cells including regulatory T cells (Tregs), cytokines (e.g., interleukin 10 (IL-10), transforming growth factor β (TGFβ)) and inhibitory receptor/ligand interactions (e.g., PD-1/PD-L1) [27,28,29]. The liver is enriched with a variety of liver-resident and circulating myeloid cells including infiltrating monocytes, monocyte-derived macrophages (MoMF), liver-resident Kupffer cells (KC) and neutrophils [30,31]. These cells, together with lymphocytes, play crucial roles in promoting immune tolerance during homeostasis and liver inflammation following injury or infection (see Table 1 for key features).

Table 1.
Key features of myeloid subsets within the liver.
As the liver is exposed to a constant presence of low levels of microbial peptides such as lipopolysaccharide (LPS) coming from the gut microbiome, parenchymal and non-parenchymal cells are often refractory to stimulation by toll-like receptor 4 (TLR4) [41,42,43]. This state is termed ‘endotoxin tolerance’. Endotoxin tolerance leads to a fairly weak response of hepatocytes to TLR stimulation [44,45]. Liver-resident Kupffer cells (KCs), which comprise approximately 80% of the body’s tissue-resident macrophages and 35% of non-parenchymal liver cells [46], produce predominantly anti-inflammatory cytokines (e.g., IL-10 and TGFβ) in response to low-level LPS exposure [47,48].
To further promote hepatic tolerance, KCs downregulate co-stimulatory molecules such as CD80/86 and have reduced expression of major histocompatibility complex (MHC) molecules required for the activation of the adaptive immune compartment [41]. Although they still express low levels of MHC molecules for T cell activation, the reduced levels of co-stimulatory molecules lead to an incomplete activation of T cells. This subsequently leads to an initial proliferation of T cells followed by clonal exhaustion and anergy, characterised by the upregulation of negative regulatory immune checkpoints (e.g., T cell immunoglobulin and mucin domain 3 (TIM-3), PD-1 and CTLA-4), and ultimately apoptosis [49,50]. In contrast, Tregs constitutively express immune checkpoint receptors and their interaction with ligands induces Treg activation [51,52]. Tregs are essential for maintenance of peripheral tolerance and they enhance the immunosuppressive milieu of the liver either via cell-to-cell contact (e.g., CTLA-4/CD80 and CD86 interaction) or through the secretion of the suppressive cytokines (e.g., IL-10 and TGFβ) [53,54]. Even though Tregs play a major role in promoting liver tolerance, research by Kido et al. demonstrated the importance of PD-1/PD-L1-mediated immune regulation in the liver in the absence of Tregs [55]. They report that, in experimental autoimmune hepatitis (AIH), liver inflammation could only be induced following neonatal thymectomy for the depletion of Tregs in combination with genetic deletion of PD-1 [55]. This concomitant loss of Tregs and PD-1 regulation was characterised by liver infiltration of autoreactive CD4+ and CD8+ T cells and severe hepatitis and the progression to fatal AIH [55].
While liver-resident cells such as KC and the hepatic endothelium constitutively express ligands for inhibitory immune receptors (e.g., PD-L1), receptor expression is usually induced in effector cells by the hepatic environment and engagement with their ligands leads to further suppression of immune function [49,56,57]. PD-L1 expression on liver sinusoidal endothelial cells has been shown to be required for the local induction of CD8+ T cell tolerance [58]. In 2004, Dong et al. showed that genetic deletion of PD-L1 in mice causes the spontaneous infiltration and accumulation of previously activated CD8+ T cells within the liver [59]. Dong et al. further showed a rapid and more severe progression of liver injury during a model of T cell-mediated hepatitis using Concanavalin A in PD-L1 knockout mice compared to wild-type mice [59]. This suggests a potential role of PD-L1 in the deletion of CD8+ T cells to protect the liver from activated cytotoxic T cells.
Maintenance of this balance between immune activation and tolerance is essential for a healthy hepatic environment and its dysregulation can cause tissue damaging inflammatory responses.
3. Hepatoxicity Related to Cancer Immunotherapies
Improved understanding of regulatory and activating pathways and their role in cancer immunology has led to a therapeutic breakthrough in oncology treatment [60,61]. The use of immunotherapy generally aims to re-programme immune cells to stimulate anti-tumour immune responses (see Figure 2 for key effects of monoclonal antibody cancer immunotherapy on myeloid and lymphoid cells) [1,2,3,62]. However, the manipulation of the balance between immune activation and suppression/tolerance may result in off-target initiation of inflammation in a number of organs, including the liver (Table 2) [1,7,13,22,63]. Liver toxicity during checkpoint inhibitor treatment generally presents as an asymptomatic elevation in serum liver enzymes, typically alanine transaminase (ALT) and aspartate transaminase (AST) levels indicative of hepatocellular damage, though rarer biliary patterns of injury have been described [13,64,65,66]. The clinical course can range from mild, self-limiting inflammation to fulminant hepatic failure and death. The main histological pattern of liver injury is lymphocyte-rich lobular inflammation with spotty or confluent necrosis, hepatocyte apoptosis, ballooning degeneration of hepatocytes and immune aggregates which may form ring granulomas [13,66,67,68].
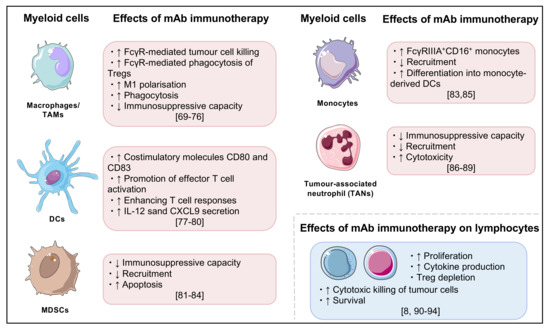
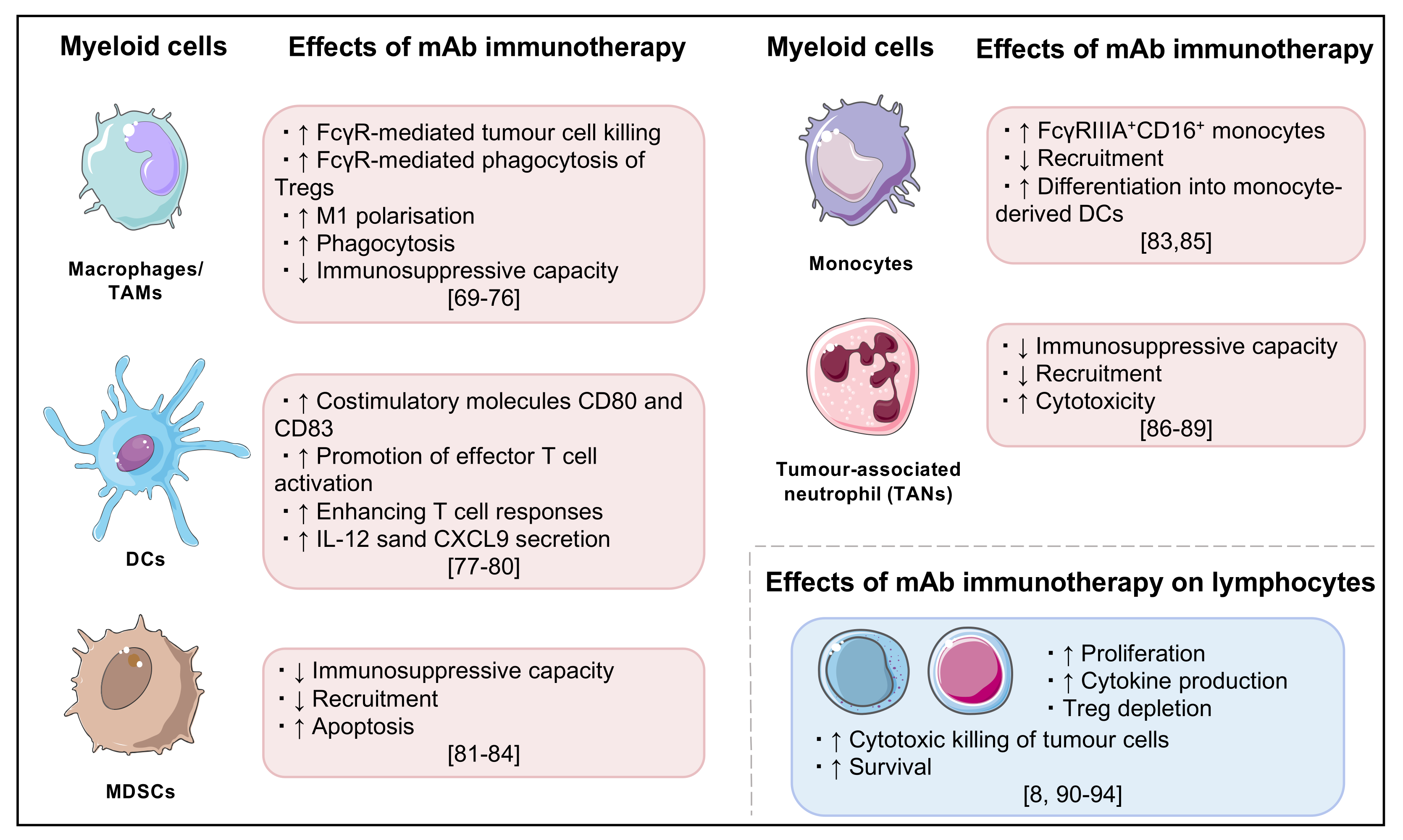
Figure 2.
Summary of key effects of monoclonal antibody cancer immunotherapy, particularly on myeloid cells and lymphocytes [69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94].

Table 2.
Prevalence of hepatic irAEs in major immunotherapy clinical trials.
This form of liver toxicity is mechanistically distinct from other forms of DILI, as it is a result of global immune reprogramming, that in a proportion of individuals leads to hepatocyte-targeted, immune-mediated toxicity [95], as opposed to direct hepatocellular damage by drug or their metabolites. CPI-induced hepatitis is also pathologically distinct from idiopathic AIH. In CPI-induced hepatitis, very few patients test positive for antinuclear antibodies or display hypergammaglobulinemia and discontinuation of cancer therapy and administration of immunosuppressants usually resolves liver inflammation [13]. Histologically CPI-induced hepatitis can be distinguished from both AIH and DILI [67], suggesting that immunotherapy induced hepatoxicity differs pathologically as well as clinically from other recognised forms of liver injury.
3.1. Breaking Hepatic Tolerance
As discussed above, due to the liver’s high tonic exposure to dietary and gut microbial antigen, it contains an abundance of immunosuppressive molecules, in particular immune checkpoints, which are constitutively expressed by myeloid cells in the liver [31,47]. These regulatory molecules are important in immune regulation and maintenance of tolerance and protect the liver from spontaneous induction of inflammation by non-pathogenic cues [47,99,100,101,102]. Under inflammatory conditions, this tolerance can be overcome but requires a high threshold of antigenic, TLR and cytokine stimulations to induce robust immune responses [103]. Following an inflammatory response, these immunosuppressive pathways in the liver are crucial for the resolution of inflammation and promotion of tissue repair [104]. Thus, blocking these pivotal regulatory pathways has the potential to lower the high activation threshold of the liver and render it susceptible to an acute inflammatory response. While the exact mechanism of hepatoxicity related to immunotherapy is incompletely understood, increasing evidence describes a role of myeloid cells and in particular monocytes and macrophages in the disease pathogenesis, either by directly causing tissue damage or by the activation of cytotoxic cells (Table 3).

Table 3.
Summary of the effect of different classes of immunotherapy on hepatic myeloid cells and their crosstalk with other subsets.
3.2. Checkpoint Inhibitors (CPIs)
CPI-induced hepatitis represents one of the most common irAEs associated with CPI therapy. CPIs target immune cell checkpoints such as CTLA-4, PD-1 and PD-L1 that are not only important for T cell regulation, but also control innate inflammatory responses [107,108]. Approximately 5–10% of patients treated with single-agent CPIs and 25–30% on combination CPI therapy [7,11,12,95] experience CPI-induced hepatitis and it usually occurs within one and three months of treatment [109].
Our group recently reported a description of the peripheral and intra-hepatic immune phenotype of monocytes/macrophages and CD8+ T cells in patients with CPI-induced hepatitis [14] and provided evidence for the involvement of myeloid cells in the pathogenesis. In CPI-induced hepatitis, circulating classical monocytes were expanded and showed an activated, tissue homing phenotype (CD163highCCR2highCCR7low), in which the proportion of classical monocytes and CD163 expression correlated positively with disease severity. This was accompanied by high levels of soluble CD163 in sera from patients with CPI-induced hepatitis, which has been shown to be a biomarker of monocyte/macrophage activation in other acute liver injury syndromes [110,111,112]. The transcriptional profile of circulating monocytes from patients with CPI-induced hepatitis demonstrated an increased expression of genes associated with activation and survival factors and reduced expression of negative regulators. Monocytes from patients with CPI-induced hepatitis showed further elevation of activation (CD40highCD163high) and increased secretion of pro-inflammatory cytokines (IL-1β, IL-6, IFNγ, IL-12p70, TNFα), once differentiated into macrophages in vitro.
The CCR2+ tissue-homing inflammatory phenotype of circulating monocytes correlated positively with activation markers of cytotoxic CD8+ T cells [14]. Parallel to these systemic changes, liver biopsies from CPI-induced hepatitis patients show focal immune aggregates composed of cytotoxic granzyme B+CD8+ T cells co-localising with CD163+ and CCR2+ expressing MoMFs. While further investigations are necessary, the presence of CCR2high monocytes in blood and CCR2+ MoMF in liver biopsies suggests monocyte recruitment to the liver from the circulating monocyte pool may be, together with their interaction with cytotoxic CD8+ T cells, mechanistically important in the pathogenesis of CPI-induced hepatitis (Figure 3a–c).
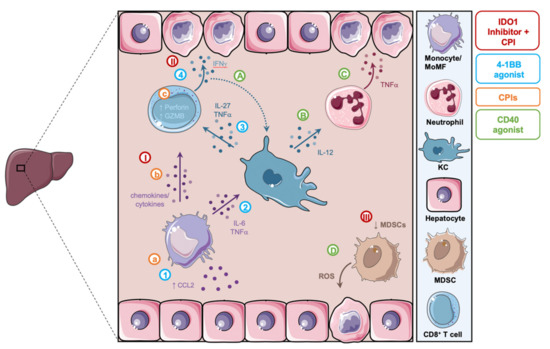
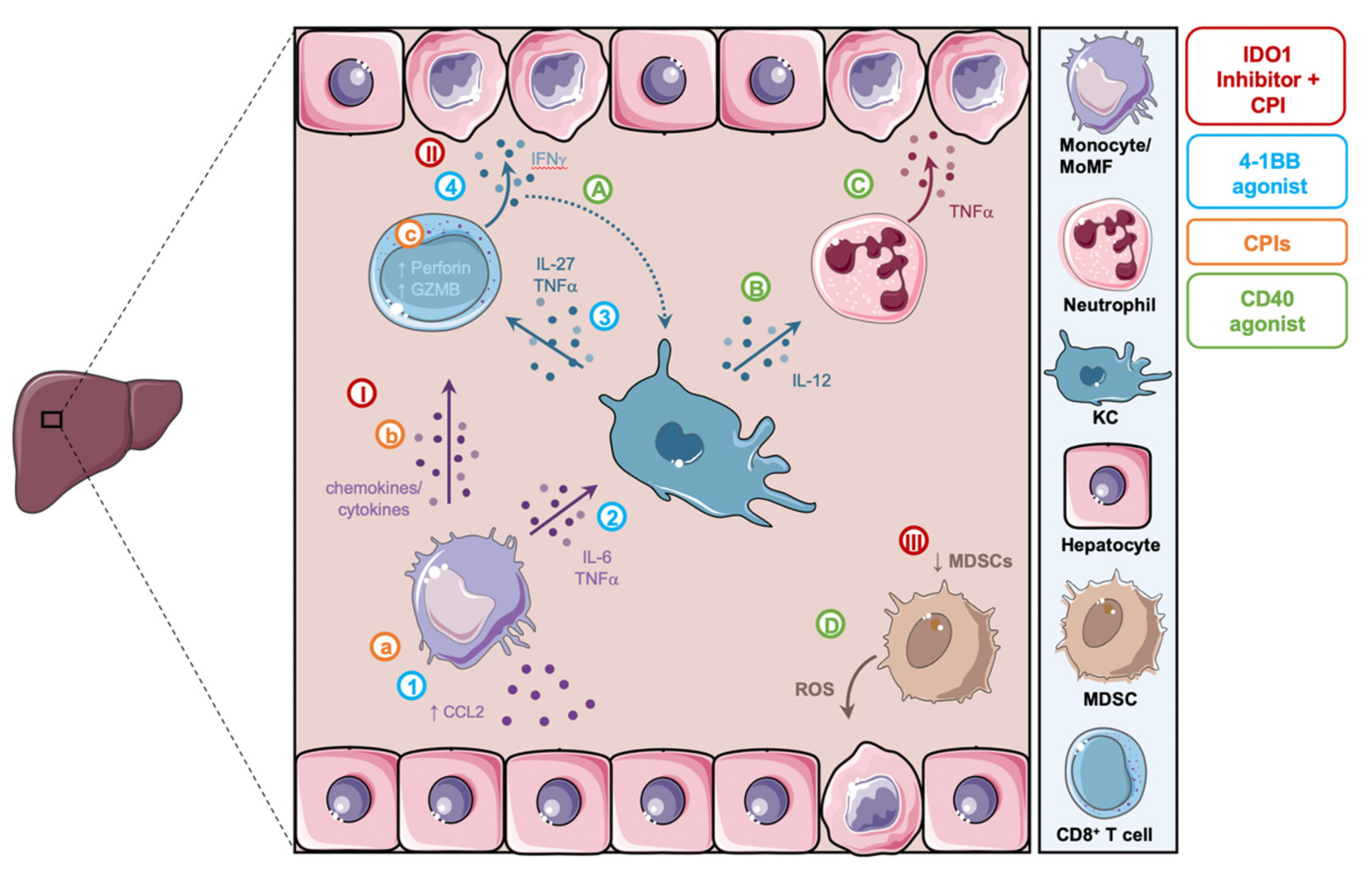
Figure 3.
Summary of proposed mechanistic models of hepatoxicity related to cancer immunotherapy. (A,B) Following CD40 agonism, KCs sense IFNγ secreted by CD8+ T cells and activate neutrophils in an IL-12 dependent manner. (C) These neutrophils in turn secrete TNFα leading to hepatocyte damage. (D) CD40 agonism reprograms MDSCs and causes the release of ROS and hepatocyte death. (a–c) CPIs induce the activation of monocytes and highly cytotoxic CD8+ T cells and lead to the formation of inflammatory aggregates associated with hepatotoxicity. (I–III) IDO1 inhibitors in combination with CPIs lead to the activation of CD8+ T cells by MoMFs, their secretion of IFNγ and subsequent hepatocellular injury, as well as the reduction in MDSCs. (1–4) 4-1BB agonism leads to the activation and CCL2/CCR2-dependent liver recruitment of MoMFs. MoMFs promote the activation of KCs, which in turn activate tissue damaging IFNγ secreting CD8+ T cells. MDSC, myeloid-derived suppressor cells; ROS, reactive oxygen species; KC, kupffer cells; MoMF, monocyte-derived macrophages; CPIs, checkpoint inhibitors; GZMB, granzyme B.
3.3. Agonistic Anti-CD40
Another immunotherapy that has received recent attention is agonistic anti-CD40 treatment. CD40 is expressed on antigen presenting cells (APCs) and stimulates CD8+ T cell activation and pro-inflammatory Th1-polarisation [113,114]. In response, T cells are able to overcome tumour-induced tolerance and produce IFNγ and IL-12 for the initiation of anti-tumour immunity [15,115,116]. However, several clinical trials reported the development of severe adverse events, most commonly cytokine release syndrome and hepatotoxicity, in response to agonistic anti-CD40 therapy [3,10,23,117]. Here, hepatoxicity, similarly to other forms of immunotherapy-induced liver injury, is associated with increased liver enzymes such as ALT and AST [10].
Early research reported a myeloid-derived suppressor cell (MDSC) associated phenotype in CD40 agonist-induced hepatoxicity [105]. CD40 plays an important role in the maturation of immunosuppressive MDSCs. In murine livers and in patients with HCC, accumulations of tumour-induced MDSCs have been observed [118,119]. Moreover, hepatic MDSCs have also been associated with the generation of hepatic metastases [120]. The treatment with CD40 agonist monoclonal antibodies activates tumour-induced myeloid cells and reduces the suppressive function of murine and human MDSCs [105]. Medina-Echeverz et al. proposed that anti-CD40 treatment caused reprogramming of CD11b+Gr-1+ MDSCs to a proinflammatory phenotype, lacking a suppressor function which ultimately leads to the release of reactive oxygen species (ROS) and hepatocyte death (Figure 3D) [105].
In contrast, recent animal studies investigating the underlying mechanism of anti-CD40 induced hepatoxicity reported a complex interplay between Th1 and various subsets of myeloid cells that dictate hepatotoxicity [15,64]. For example, in 2020 Bonnans et al. showed that CD40 agonist induced an inflammatory network of TNFα, IFNγ and IL-12, in which only deficiency in IL-12 was protective of liver injury and led to the decreased activation and frequency of CD11b+CD14+F4/80+MHCII+ hepatic macrophages [64]. Similarly, in 2021 Siwicki et al. show the induction of pathology by IFNγ and IL-12 in tumour free tissues, which was dependent on macrophages and neutrophils [15]. In contrast, IL-12 and IFNγ producing DC and cytotoxic CD8+ T cells mediated anti-tumour immunity but were not necessary for tissue pathology. In the liver, KCs were able to sense IFNγ secreted by T cells and in turn produced IL-12. Using transgenic mice treated with anti-CD40 in which KCs lack the receptor for IFNγ (Clec4f-cr+/0 Ifngr1fl/fl), the authors showed that IL-12 production and liver necrosis were nearly diminished. They further showed that the IL-12 response of KC mediated by IFNγ acted as a positive feedback loop promoting local IFNγ production. IFNγ sensing subsequently induced the increased presence and Tnf expression of tissue damaging neutrophils in the liver (Figure 3A–C). Neutrophils were the main source of Tnf as they contributed approximately 92% of all Tnf expression in the inflamed liver. Neutrophil and TNFɑ neutralisation in mice treated with CD40 agonist led to the protection from liver necrosis and inflammation. Siwicki et al. further demonstrated similarities between these findings to the pathology of CPI-induced hepatitis. They describe neutrophil liver infiltrates to be associated with severity of inflammation in human CPI-induced hepatitis. Mice treated with combination anti-CTLA-4 and anti-PD-1 present with elevated levels of IL-12 in tumour free tissues, an activated MHCIIhigh phenotype of KCs and an increase in liver neutrophils.
In this study, neutrophil based interventions were able to suppress CD40 agonist associated liver damage without negatively impacting on the tumour response [15]. Not only may targeting neutrophils have the potential to treat hepatoxicity in this context and potentially other immunotherapy related hepatotoxicities, but neutrophil based therapies are currently being trialled as cancer therapy, making this an attractive adjunct to CD40 agonism [121,122].
3.4. 4-1BB Activation
4-1BB (CD137) in an activation-induced costimulatory receptor and is expressed by a wide range of activated lymphocyte and myeloid subsets [62]. The interaction with its ligand stimulates activation of these cells and promotes CD8 driven anti-tumour responses [123]. However, despite the effectiveness of this treatment approach, the activation of the 4-1BB pathway was associated with dose-limiting severe hepatocellular injury in pre-clinical trials and no trial has advanced beyond early phase II [62].
Research by Bartkowiak et al. showed that 4-1BB agonist-associated hepatotoxicity is not triggered by activated CD8+ T cell responses but is initiated through the activation of KCs and their secretion of TNFα and IL-27, which in turn promoted the cytotoxic function of CD8+ T cells and hepatocyte damage [17]. They show that 4-1BB activated bone marrow-derived monocytes home to the liver and cause an inflammatory environment which stimulated the upregulation of 4-1BB in KCs. KCs then respond to 4-1BB agonistic therapy by increasing their antigen presentation capacity (MHC II upregulation) and producing IL-27 for the attraction of CD8+ T cells. This leads to the activation of CD8+ T cells with elevated IFNγ secretion and ultimately local hepatocyte damage (Figure 3 1–4). In the pathogenesis, CD8+ T cells directly mediate tissue injury, as mice deficient in CD8+ T cells are protected from hepatotoxicity. However, the activation of CD8+ T cells is highly dependent on the presence of KC-derived IL-27. They further found that CCR2−/− mice, which are deficient in bone marrow-derived monocytes, were also protected from hepatotoxicity but showed an intact CD8 anti-tumour response. The authors speculate that 4-1BB activated monocytes initiate an immune cascade to trigger off target liver injury and that CCR2 inhibitors are a potential target to treat liver inflammation in this condition.
3.5. Combined Agents
The combination of immunotherapeutic agents has shown to be more effective than single agent therapy and to improve overall survival [2,124,125]. For example, in metastatic melanoma, five-year overall survival rates were increased to 52% in Ipilimumab and Nivolumab combination therapy [1]. In contrast, single agent CPI treated patients showed an overall survival of 44%. Moreover, studies showed that combining CPIs with agonists of immune stimulatory molecules as well as various other inhibitors (e.g., small molecules) has beneficial effects in boosting the anti-tumour response [16,17,106].
3.5.1. Checkpoint Inhibitor Combination Therapy
While the combination of CPIs (e.g., Ipilimumab and Nivolumab) was associated with increased survival, the frequency and severity of CPI-induced hepatitis was also higher in patients on dual therapy [1]. In fact, up to 15% of those patients develop high grade hepatitis (grade 3–4, defined by an ALT level of >5 or >20 times the upper limit of normal (ULN)) [11,12]. In contrast, the development of severe, grade 3–4 hepatitis in patients on single therapy is less frequent, with an incidence rate of 1–2%.
The histological features and peripheral immune phenotype associated with anti-CTLA-4 and/or anti-PD-1 mediated liver injury in CPI-induced hepatitis are also distinct [13,126]. For example, liver biopsies showed a more equal CD4:CD8 ratio following single agent anti-PD-1 therapy compared with combination CPI [13]. This could also be observed in the circulation of patients with CPI-induced hepatitis [14]. All treatment regimens were described to have inflammatory liver infiltrates that consisted predominantly of lymphocytes and macrophages; however, the occurrence of those immune aggregates is particularly pronounced in dual agent therapy containing anti-CTLA-4 and anti-PD-1 CPI [13,127]. In contrast, the phenotype of monocytes/macrophages in CPI-induced hepatitis were similar between single and dual agent CPI [14].
3.5.2. Small Molecule Indoleamine 2,3-Dioxygenase 1 (IDO1) Inhibitors and CPIs
IDO1 is a cytosolic enzyme involved in the suppression of cytotoxic cells such as CD8 T cells and natural killer (NK) cells by depletion of tryptophan and the promotion of regulatory T cells, MDSCs and tumour-associated macrophages (TAMs) [128]. In preclinical studies, IDO1 was shown to be involved in the tumour escape from immune surveillance and its activation in human cancers was associated with poor prognosis [129]. Activation of IDO1 can be observed in tumour cells themselves as well as stromal and vascular cells and innate immune cells [130]. The suppressive tumour microenvironment recruits cells expressing IDO1, which in turn recruit and tolerise further immune cells [130]. This created a positive feedback loop for the reprogramming of cells and exacerbating a suppressive milieu. Due to this role in immune-oncology, small molecule inhibitors of IDO1 present a promising concept in cancer therapies [4].
In the liver, hepatic stellate cells were shown to promote tolerogenic DCs by inducing IDO1 expression, enhancing hepatic tolerance [131]. In contrast, in Ido1 deficient mice (Ido1−/−), inflammatory stimuli within the liver can overcome the suppressive environment and induce liver inflammation, suggesting a lowered threshold for activation [132,133]. IDO inhibitors such as Epacadostat are currently being trialled as cancer treatment, in particular in combination with CPIs [4]. Murine studies on the use of an IDO1 inhibitor with CPIs demonstrated the development of hepatocyte injury and liver infiltration of primarily CD8+ T cells with a reduction in MDSC frequencies (Figure 3 III) [106]. In turn, the number of liver infiltrating MoMF was increased [16]. Similarly to other drug related hepatoxicities, CPI+IDO1 inhibitor induced liver injury was abolished when CD8+ T cells were depleted [16]. Single cell and bulk RNA sequencing of these mice revealed that a central role of IFNγ in the disease pathogenesis and MoMF promoting this pro-inflammatory T cell response (Figure 3 I,II). Interestingly, this study also reported on CPI combination therapy together with 4-1BB agonism, which has a similar expression profile [16].
3.5.3. Immunotherapy in Combination with Conventional Chemotherapeutics
To further enhance the effectiveness of immunotherapy and to promote anti-tumour responses in immunotherapy refractory patients, numerous clinical trials are currently exploring immunotherapy combined with chemotherapy [134,135,136,137,138,139,140,141]. Certain chemotherapy agents can be immunostimulatory and increase antigenicity of cancer cells leading to the activation of effector cells such as DCs, M1-like TAMs and cytotoxic cells, and depletion of immunosuppressive cells including MDSCs and Tregs [142,143,144,145,146,147]. This is particularly mediated by agents inducing immunogenic cell death of malignant cells, during which dying cells accumulate nucleic acids in their cytosol and release cytoplasmic and nuclear proteins in their extracellular space to activate the innate and subsequently adaptive immune compartment [148,149]. These agents include, for example, cyclophosphamide, dactinomycin, gemcitabine and cisplatin [149]. Preclinical and clinical studies investigating chemotherapy in combination with anti-PD-1 and anti-CTLA-4 checkpoint inhibitors show promising results with higher response rates and overall survival [142]. In addition, chemotherapies were also demonstrated to promote the upregulation of PD-L1 on various cells such as cancer cells and TAMs [150,151]. Combination of those therapies with anti-PD-(L)1 treatment could further increase anti-tumour responses in patients [152]. While the majority of trials are still ongoing, the increased risk of hepatotoxicity has been associated with the use of combination therapy in a few studies (Table 4) [134,135,136,137,138,139,140,141]. A recent meta-analysis by Guo et al., assessing the risk of hepatoxicity following different treatment regimens involving anti-PD-(L)1 and chemotherapy, showed an elevated risk of all-grade and high-grade hepatitis with use of anti-PD-(L)1 with and without chemotherapy, compared to chemotherapy alone [153]. Thus far, the role of myeloid cell involvement in this is unclear and warrants further investigation.

Table 4.
Reported hepatoxicity related to cancer immunotherapy combined with chemotherapy.
3.6. In Vivo Experimental Models of Immune-Related Adverse Events
Various studies are aiming to establish clinically relevant murine models for the investigation of the underlying immunological mechanisms of liver irAEs and to lay the foundation for in vivo testing of therapeutic agents. One of the major challenges to date is that mice do not spontaneously develop hepatic inflammation in the absence of checkpoint signalling, either through blockade (e.g., anti-CTLA-4, anti-PD-1) or genetic deletion (e.g., PD-1−/−) [17,104,106].
In an effort to develop a murine model of drug-induced liver injury (DILI), Metushi et al. reported the development of more severe and persistent DILI when hepatic tolerance is broken by interruption of CPI signalling [104,106,154]. In PD-1−/− mice, the co-treatment of the hepatotoxin Amodiaquine with anti-CTLA-4 resulted in liver infiltration of macrophages and cytotoxic CD8+ T cells and the formation of immune cell aggregates in areas of necrosis [104]. This evidence suggested that breakdown of immune checkpoints, normally mediating hepatic immune tolerance mechanisms, promotes liver damage caused by other triggers. This need for a priming event for the initiation of hepatic inflammation in the context of immunotherapy seems to be a common feature in murine models [15,16,17,106] and raises questions about the induction of human pathology.
In 2021, Adam et al. showed that treatment with anti-CTLA-4 + anti-PD-1 in tumour bearing mice from a genetic background (B6/lpr mice) prone to autoimmune responses, induced multiple organ irAEs [155]. Using this model, they demonstrated that most effective anti-tumour responses in mice were associated with the development of irAEs. They postulate that the reduction in tumour size could be used as a predictive marker for irAEs. In fact, early data from patients with melanoma and lung cancer shows correlations of anti-tumour responses following CPI therapy with irAE severity [156,157,158]. Adam et al. further showed that prednisolone treatment in this model prevented the development of irAEs in mice treated with combination CPI [155]. However, it also demolished anti-tumour immunity. While limited due to the mice’ genetic susceptibility to autoimmunity, this informative study shows the importance of establishing effective murine models to inform early detection and treatment regimens of human irAEs.
4. Future Perspective
The management of immune-mediated hepatitis in the context of cancer immunotherapies, in a way that does not compromise the anti-tumour response, presents a clinical challenge. New technologies such as single cell RNA sequencing (scRNAseq) make it possible to study highly heterogeneous tumour cells, the immune landscape of the tumour microenvironment and classify new immune subpopulations [159,160,161,162,163]. Such techniques are proving crucial to identify effective diagnostic and prognostic biomarkers, develop new tumour immunotherapy and unravel the complexity of immune interactions during drug toxicity [160,161,162,164,165,166,167]. Novel prediction platforms such as Beyondcell using scRNAseq datasets and drug response profiles have the potential to indicate targetable pathways with very high response rates [168]. Moreover, machine learning algorithms utilising irAE datasets collected from patient symptom questionnaires and Common Terminology Criteria for Adverse Events (CTCAE) have been shown to predict the presence and onset of irAEs with high accuracy and have the potential to aid early detection of irAEs [169]. While early detection systems and biomarkers are important factors for the safety of immunotherapy in patients, the development of alternative treatment strategies that do not require cessation of cancer treatment nor negatively impact the anti-tumour response are paramount. Understanding the underlying mechanisms of these hepatoxicities will inform as to the type and timing of immune-based interventions to resolve liver toxicity. Research to date has demonstrated that targeting myeloid cells such as monocytes and neutrophils presents a promising approach. Although evidence across a number of agents and models suggests that lymphocytes contribute to hepatoxicity, their inhibition would significantly compromise anti-tumour immunity [170,171,172,173]. In contrast, targeted inhibition or depletion of involved myeloid subsets may not directly affect T cell licensing for anti-tumour responses but has the potential to improve hepatotoxicity significantly.
Author Contributions
Conceptualization and writing—original draft preparation, C.L.C.G. and L.A.P.; writing—review and editing, C.L.C.G. and L.A.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
L.A.P. recognizes the support of the NIHR Imperial College Biomedical Research Centre. Figures were generated using Microsoft PowerPoint and smart.servier.com illustrations.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.-J.; Rutkowski, P.; Lao, C.D.; Cowey, C.L.; Schadendorf, D.; Wagstaff, J.; Dummer, R.; et al. Five-Year Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2019, 381, 1535–1546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolchok, J.D.; Chiarion-Sileni, V.; Gonzalez, R.; Rutkowski, P.; Grob, J.-J.; Cowey, C.L.; Lao, C.D.; Wagstaff, J.; Schadendorf, D.; Ferrucci, P.F.; et al. Overall Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2017, 377, 1345–1356. [Google Scholar] [CrossRef] [PubMed]
- Rüter, J.; Antonia, S.J.; Burris, H.A.; Huhn, R.D.; Vonderheide, R.H. Immune modulation with weekly dosing of an agonist CD40 antibody in a phase I study of patients with advanced solid tumors. Cancer Biol. Ther. 2010, 10, 983–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, K.; Wu, Y.-H.; Song, Y.; Yu, B. Indoleamine 2,3-dioxygenase 1 (IDO1) inhibitors in clinical trials for cancer immunotherapy. J. Hematol. Oncol. 2021, 14, 68. [Google Scholar] [CrossRef]
- Segal, N.H.; Logan, T.F.; Hodi, F.S.; McDermott, D.; Melero, I.; Hamid, O.; Schmidt, H.; Robert, C.; Chiarion-Sileni, V.; Ascierto, P.A.; et al. Results from an Integrated Safety Analysis of Urelumab, an Agonist Anti-CD137 Monoclonal Antibody. Clin. Cancer Res. 2017, 23, 1929–1936. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Wahab, N.; Shah, M.; Suarez-Almazor, M.E. Adverse Events Associated with Immune Checkpoint Blockade in Patients with Cancer: A Systematic Review of Case Reports. PLoS ONE 2016, 11, e0160221. [Google Scholar] [CrossRef]
- Fessas, P.; Possamai, L.A.; Clark, J.; Daniels, E.; Gudd, C.; Mullish, B.H.; Alexander, J.L.; Pinato, D.J. Immunotoxicity from checkpoint inhibitor therapy: Clinical features and underlying mechanisms. Immunology 2020, 159, 167–177. [Google Scholar] [CrossRef]
- Postow, M.A.; Sidlow, R.; Hellmann, M.D. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N. Engl. J. Med. 2018, 378, 158–168. [Google Scholar] [CrossRef]
- Kottschade, L.A. Incidence and Management of Immune-Related Adverse Events in Patients Undergoing Treatment with Immune Checkpoint Inhibitors. Curr. Oncol. Rep. 2018, 20, 24. [Google Scholar] [CrossRef]
- Vonderheide, R.H.; Flaherty, K.T.; Khalil, M.; Stumacher, M.S.; Bajor, D.L.; Hutnick, N.A.; Sullivan, P.; Mahany, J.J.; Gallagher, M.; Kramer, A.; et al. Clinical Activity and Immune Modulation in Cancer Patients Treated With CP-870,893, a Novel CD40 Agonist Monoclonal Antibody. J. Clin. Oncol. 2007, 25, 876–883. [Google Scholar] [CrossRef]
- Haanen, J.B.A.G.; Carbonnel, F.; Robert, C.; Kerr, K.M.; Peters, S.; Larkin, J.; Jordan, K.; Committee, on behalf of the E.G. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up†. Ann. Oncol. 2017, 28, iv119–iv142. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R.; Lacchetti, C.; Schneider, B.J.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; Ernstoff, M.S.; Gardner, J.M.; Ginex, P.; et al. Management of Immune-Related Adverse Events in Patients Treated with Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 1714–1768. [Google Scholar] [CrossRef] [PubMed]
- De Martin, E.; Michot, J.M.; Papouin, B.; Champiat, S.; Mateus, C.; Lambotte, O.; Roche, B.; Antonini, T.M.; Coilly, A.; Laghouati, S.; et al. Characterization of liver injury induced by cancer immunotherapy using immune checkpoint inhibitors. J. Hepatol. 2018, 68, 1181–1190. [Google Scholar] [CrossRef]
- Gudd, C.L.C.; Au, L.; Triantafyllou, E.; Shum, B.; Liu, T.; Nathwani, R.; Kumar, N.; Mukherjee, S.; Dhar, A.; Woollard, K.J.; et al. Activation and transcriptional profile of monocytes and CD8+ T cells are altered in checkpoint inhibitor-related hepatitis. J. Hepatol. 2021, 75, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Siwicki, M.; Gort-Freitas, N.A.; Messemaker, M.; Bill, R.; Gungabeesoon, J.; Engblom, C.; Zilionis, R.; Garris, C.; Gerhard, G.M.; Kohl, A.; et al. Resident Kupffer cells and neutrophils drive liver toxicity in cancer immunotherapy. Sci. Immunol. 2021, 6, eabi7083. [Google Scholar] [CrossRef] [PubMed]
- Llewellyn, H.P.; Arat, S.; Gao, J.; Wen, J.; Xia, S.; Kalabat, D.; Oziolor, E.; Virgen-Slane, R.; Affolter, T.; Ji, C. T cells and monocyte-derived myeloid cells mediate immunotherapy-related hepatitis in a mouse model. J. Hepatol. 2021, 75, 1083–1095. [Google Scholar] [CrossRef] [PubMed]
- Bartkowiak, T.; Jaiswal, A.R.; Ager, C.R.; Chin, R.; Chen, C.H.; Budhani, P.; Ai, M.; Reilley, M.J.; Sebastian, M.M.; Hong, D.S.; et al. Activation of 4-1BB on liver myeloid cells triggers hepatitis via an interleukin-27–dependent pathway. Clin. Cancer Res. 2018, 24, 1138–1151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robert, C.; Schachter, J.; Long, G.V.; Arance, A.; Grob, J.J.; Mortier, L.; Daud, A.; Carlino, M.S.; McNeil, C.; Lotem, M.; et al. Pembrolizumab versus Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2015, 372, 2521–2532. [Google Scholar] [CrossRef]
- Eggermont, A.M.M.; Blank, C.U.; Mandala, M.; Long, G.V.; Atkinson, V.; Dalle, S.; Haydon, A.; Lichinitser, M.; Khattak, A.; Carlino, M.S.; et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma. N. Engl. J. Med. 2018, 378, 1789–1801. [Google Scholar] [CrossRef]
- Wolchok, J.D.; Neyns, B.; Linette, G.; Negrier, S.; Lutzky, J.; Thomas, L.; Waterfield, W.; Schadendorf, D.; Smylie, M.; Guthrie, T., Jr.; et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: A randomised, double-blind, multicentre, phase 2, dose-ranging study. Lancet Oncol. 2010, 11, 155–164. [Google Scholar] [CrossRef]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved Survival with Ipilimumab in Patients with Metastatic Melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.J.; Cowey, C.L.; Lao, C.D.; Schadendorf, D.; Dummer, R.; Smylie, M.; Rutkowski, P.; et al. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N. Engl. J. Med. 2015, 373, 23–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beatty, G.L.; Torigian, D.A.; Chiorean, E.G.; Saboury, B.; Brothers, A.; Alavi, A.; Troxel, A.B.; Sun, W.; Teitelbaum, U.R.; Vonderheide, R.H.; et al. A Phase I Study of an Agonist CD40 Monoclonal Antibody (CP-870,893) in Combination with Gemcitabine in Patients with Advanced Pancreatic Ductal Adenocarcinoma. Clin. Cancer Res. 2013, 19, 6286–6295. [Google Scholar] [CrossRef] [Green Version]
- Calne, R.Y.; Sells, R.A.; Pena, J.R.; Davis, D.R.; Millard, P.R.; Herbertson, B.M.; Binns, R.M.; Davies, D.A.L. Induction of Immunological Tolerance by Porcine Liver Allografts. Nature 1969, 223, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Orlando, G.; Soker, S.; Wood, K. Operational tolerance after liver transplantation. J. Hepatol. 2009, 50, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.W.; Harmon, C.; O’Farrelly, C. Liver immunology and its role in inflammation and homeostasis. Cell. Mol. Immunol. 2016, 13, 267–276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, M.; Xu, S.; Han, Y.; Cao, X. Apoptotic cells attenuate fulminant hepatitis by priming Kupffer cells to produce interleukin-10 through membrane-bound TGF-β. Hepatology 2011, 53, 306–316. [Google Scholar] [CrossRef]
- Wu, K.; Kryczek, I.; Chen, L.; Zou, W.; Welling, T.H. Kupffer cell suppression of CD8+ T cells in human hepatocellular carcinoma is mediated by B7-H1/programmed death-1 interactions. Cancer Res. 2009, 69, 8067–8075. [Google Scholar] [CrossRef] [Green Version]
- Erhardt, A.; Biburger, M.; Papadopoulos, T.; Tiegs, G. IL-10, regulatory T cells, and Kupffer cells mediate tolerance in concanavalin A-induced liver injury in mice. Hepatology 2007, 45, 475–485. [Google Scholar] [CrossRef]
- Heymann, F.; Tacke, F. Immunology in the liver-from homeostasis to disease. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 88–110. [Google Scholar] [CrossRef]
- Crispe, I.N. The Liver as a Lymphoid Organ. Annu. Rev. Immunol. 2009, 27, 147–163. [Google Scholar] [CrossRef] [PubMed]
- Triantafyllou, E.; Pop, O.T.; Possamai, L.A.; Wilhelm, A.; Liaskou, E.; Singanayagam, A.; Bernsmeier, C.; Khamri, W.; Petts, G.; Dargue, R.; et al. MerTK expressing hepatic macrophages promote the resolution of inflammation in acute liver failure. Gut 2018, 67, 333–347. [Google Scholar] [CrossRef]
- Mills, C.D.; Kincaid, K.; Alt, J.M.; Heilman, M.J.; Hill, A.M. M-1/M-2 Macrophages and the Th1/Th2 Paradigm. J. Immunol. 2000, 164, 6166–6173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bilzer, M.; Roggel, F.; Gerbes, A.L. Role of Kupffer cells in host defense and liver disease. Liver Int. 2006, 26, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Ju, C.; Tacke, F. Hepatic macrophages in homeostasis and liver diseases: From pathogenesis to novel therapeutic strategies. Cell. Mol. Immunol. 2016, 13, 316–327. [Google Scholar] [CrossRef] [Green Version]
- Hammerich, L.; Tacke, F. Emerging roles of myeloid derived suppressor cells in hepatic inflammation and fibrosis. World J. Gastrointest. Pathophysiol. 2015, 6, 43. [Google Scholar] [CrossRef]
- Tang, J.; Yan, Z.; Feng, Q.; Yu, L.; Wang, H. The Roles of Neutrophils in the Pathogenesis of Liver Diseases. Front. Immunol. 2021, 12, 625472. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil Extracellular Traps Kill Bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Lukacs-Kornek, V.; Schuppan, D. Dendritic cells in liver injury and fibrosis: Shortcomings and promises. J. Hepatol. 2013, 59, 1124–1126. [Google Scholar] [CrossRef] [Green Version]
- Goddard, S.; Youster, J.; Morgan, E.; Adams, D.H. Interleukin-10 Secretion Differentiates Dendritic Cells from Human Liver and Skin. Am. J. Pathol. 2004, 164, 511–519. [Google Scholar] [CrossRef] [Green Version]
- Thomson, A.W.; Knolle, P.A. Antigen-presenting cell function in the tolerogenic liver environment. Nat. Rev. Immunol. 2010, 10, 753–766. [Google Scholar] [CrossRef] [PubMed]
- McDonald, B.; Jenne, C.N.; Zhuo, L.; Kimata, K.; Kubes, P. Kupffer cells and activation of endothelial TLR4 coordinate neutrophil adhesion within liver sinusoids during endotoxemia. Am. J. Physiol. Liver Physiol. 2013, 305, G797–G806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Wang, Z.; Zou, Y.; Lu, E.; Duan, J.; Yang, H.; Wu, Q.; Zhao, X.; Wang, Y.; You, L.; et al. Pretreatment with lipopolysaccharide attenuates diethylnitrosamine-caused liver injury in mice via TLR4-dependent induction of Kupffer cell M2 polarization. Immunol. Res. 2015, 62, 137–145. [Google Scholar] [CrossRef]
- Liu, S.; Gallo, D.J.; Green, A.M.; Williams, D.L.; Gong, X.; Shapiro, R.A.; Gambotto, A.A.; Humphris, E.L.; Vodovotz, Y.; Billiar, T.R. Role of toll-like receptors in changes in gene expression and NF-kappa B activation in mouse hepatocytes stimulated with lipopolysaccharide. Infect. Immun. 2002, 70, 3433–3442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsumura, T.; Degawa, T.; Takii, T.; Hayashi, H.; Okamoto, T.; Inoue, J.; Onozaki, K. TRAF6-NF-kappaB pathway is essential for interleukin-1-induced TLR2 expression and its functional response to TLR2 ligand in murine hepatocytes. Immunology 2003, 109, 127–136. [Google Scholar] [CrossRef]
- Gomez Perdiguero, E.; Klapproth, K.; Schulz, C.; Busch, K.; Azzoni, E.; Crozet, L.; Garner, H.; Trouillet, C.; de Bruijn, M.F.; Geissmann, F.; et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 2015, 518, 547–551. [Google Scholar] [CrossRef]
- Crispe, I.N. Immune tolerance in liver disease. Hepatology 2014, 60, 2109–2117. [Google Scholar] [CrossRef] [Green Version]
- Jenne, C.N.; Kubes, P. Immune surveillance by the liver. Nat. Immunol. 2013, 14, 996–1006. [Google Scholar] [CrossRef]
- Knolle, P.A.; Uhrig, A.; Hegenbarth, S.; Löser, E.; Schmitt, E.; Gerken, G.; Lohse, A.W. IL-10 down-regulates T cell activation by antigen-presenting liver sinusoidal endothelial cells through decreased antigen uptake via the mannose receptor and lowered surface expression of accessory molecules. Clin. Exp. Immunol. 1998, 114, 427–433. [Google Scholar] [CrossRef]
- Zheng, M.; Tian, Z. Liver-Mediated Adaptive Immune Tolerance. Front. Immunol. 2019, 10, 2525. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, T.; Tagami, T.; Yamazaki, S.; Uede, T.; Shimizu, J.; Sakaguchi, N.; Mak, T.W.; Sakaguchi, S. Immunologic self-tolerance maintained by CD25(+)CD4(+) regulatory T cells constitutively expressing cytotoxic T lymphocyte-associated antigen 4. J. Exp. Med. 2000, 192, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Wang, D.; Zhang, G.; Guo, X. The Role Of PD-1/PD-L1 Axis in Treg Development and Function: Implications for Cancer Immunotherapy. OncoTargets Ther. 2019, 12, 8437–8445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vignali, D.A.A.; Collison, L.W.; Workman, C.J. How regulatory T cells work. Nat. Rev. Immunol. 2008, 8, 523–532. [Google Scholar] [CrossRef] [Green Version]
- Sakaguchi, S.; Sakaguchi, N.; Asano, M.; Itoh, M.; Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 1995, 155, 1151–1164. [Google Scholar]
- Kido, M.; Watanabe, N.; Okazaki, T.; Akamatsu, T.; Tanaka, J.; Saga, K.; Nishio, A.; Honjo, T.; Chiba, T. Fatal autoimmune hepatitis induced by concurrent loss of naturally arising regulatory T cells and PD-1-mediated signaling. Gastroenterology 2008, 135, 1333–1343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wherry, E.J. T cell exhaustion. Nat. Immunol. 2011, 12, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Wang, G.; Lv, Y.; Wan, Y.Y.; Zheng, J. Inhibition of Cdk8/Cdk19 Activity Promotes Treg Cell Differentiation and Suppresses Autoimmune Diseases. Front. Immunol. 2019, 10, 1988. [Google Scholar] [CrossRef] [Green Version]
- Diehl, L.; Schurich, A.; Grochtmann, R.; Hegenbarth, S.; Chen, L.; Knolle, P.A. Tolerogenic maturation of liver sinusoidal endothelial cells promotes B7-homolog 1-dependent CD8+ T cell tolerance. Hepatology 2008, 47, 296–305. [Google Scholar] [CrossRef]
- Dong, H.; Zhu, G.; Tamada, K.; Flies, D.B.; van Deursen, J.M.A.; Chen, L. B7-H1 Determines Accumulation and Deletion of Intrahepatic CD8+ T Lymphocytes. Immunity 2004, 20, 327–336. [Google Scholar] [CrossRef] [Green Version]
- Zeng, C.; Wen, W.; Morgans, A.K.; Pao, W.; Shu, X.-O.; Zheng, W. Disparities by Race, Age, and Sex in the Improvement of Survival for Major Cancers: Results from the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) Program in the United States, 1990 to 2010. JAMA Oncol. 2015, 1, 88–96. [Google Scholar] [CrossRef]
- Patel, J.D.; Krilov, L.; Adams, S.; Aghajanian, C.; Basch, E.; Brose, M.S.; Carroll, W.L.; De Lima, M.; Gilbert, M.R.; Kris, M.G.; et al. Clinical cancer advances 2013: Annual report on progress against cancer from the American Society of Clinical Oncology. J. Clin. Oncol. 2014, 32, 129–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chester, C.; Sanmamed, M.F.; Wang, J.; Melero, I. Immunotherapy targeting 4-1BB: Mechanistic rationale, clinical results, and future strategies. Blood 2018, 131, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Wolchok, J.D.; Kluger, H.; Callahan, M.K.; Postow, M.A.; Rizvi, N.A.; Lesokhin, A.M.; Segal, N.H.; Ariyan, C.E.; Gordon, R.A.; Reed, K.; et al. Nivolumab plus Ipilimumab in advanced melanoma. N. Engl. J. Med. 2013, 369, 122–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonnans, C.; Thomas, G.; He, W.; Jung, B.; Chen, W.; Liao, M.; Heyen, J.; Buetow, B.; Pillai, S.; Matsumoto, D.; et al. CD40 agonist-induced IL-12p40 potentiates hepatotoxicity. J. Immunother. Cancer 2020, 8, e000624. [Google Scholar] [CrossRef] [PubMed]
- Bhave, P.; Buckle, A.; Sandhu, S.; Sood, S. Mortality due to immunotherapy related hepatitis. J. Hepatol. 2018, 69, 976–978. [Google Scholar] [CrossRef] [Green Version]
- Doherty, G.J.; Duckworth, A.M.; Davies, S.E.; Mells, G.F.; Brais, R.; Harden, S.V.; Parkinson, C.A.; Corrie, P.G. Severe steroid-resistant anti-PD1 T-cell checkpoint inhibitor-induced hepatotoxicity driven by biliary injury. ESMO Open 2017, 2, e000268. [Google Scholar] [CrossRef] [Green Version]
- Zen, Y.; Yeh, M.M. Hepatotoxicity of immune checkpoint inhibitors: A histology study of seven cases in comparison with autoimmune hepatitis and idiosyncratic drug-induced liver injury. Mod. Pathol. 2018, 31, 965–973. [Google Scholar] [CrossRef] [Green Version]
- Johncilla, M.; Misdraji, J.; Pratt, D.S.; Agoston, A.T.; Lauwers, G.Y.; Srivastava, A.; Doyle, L.A. Ipilimumab-associated Hepatitis: Clinicopathologic Characterization in a Series of 11 Cases. Am. J. Surg. Pathol. 2015, 39, 1075–1084. [Google Scholar] [CrossRef]
- Clynes, R.A.; Towers, T.L.; Presta, L.G.; Ravetch, J. V Inhibitory Fc receptors modulate in vivo cytotoxicity against tumor targets. Nat. Med. 2000, 6, 443–446. [Google Scholar] [CrossRef]
- Pincetic, A.; Bournazos, S.; DiLillo, D.J.; Maamary, J.; Wang, T.T.; Dahan, R.; Fiebiger, B.-M.; Ravetch, J. V Type I and type II Fc receptors regulate innate and adaptive immunity. Nat. Immunol. 2014, 15, 707–716. [Google Scholar] [CrossRef]
- Uchida, J.; Hamaguchi, Y.; Oliver, J.A.; Ravetch, J.V.; Poe, J.C.; Haas, K.M.; Tedder, T.F. The innate mononuclear phagocyte network depletes B lymphocytes through Fc receptor-dependent mechanisms during anti-CD20 antibody immunotherapy. J. Exp. Med. 2004, 199, 1659–1669. [Google Scholar] [CrossRef]
- Gül, N.; Babes, L.; Siegmund, K.; Korthouwer, R.; Bögels, M.; Braster, R.; Vidarsson, G.; ten Hagen, T.L.M.; Kubes, P.; van Egmond, M. Macrophages eliminate circulating tumor cells after monoclonal antibody therapy. J. Clin. Investig. 2014, 124, 812–823. [Google Scholar] [CrossRef] [PubMed]
- Simpson, T.R.; Li, F.; Montalvo-Ortiz, W.; Sepulveda, M.A.; Bergerhoff, K.; Arce, F.; Roddie, C.; Henry, J.Y.; Yagita, H.; Wolchok, J.D.; et al. Fc-dependent depletion of tumor-infiltrating regulatory T cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J. Exp. Med. 2013, 210, 1695–1710. [Google Scholar] [CrossRef]
- Gordon, S.R.; Maute, R.L.; Dulken, B.W.; Hutter, G.; George, B.M.; McCracken, M.N.; Gupta, R.; Tsai, J.M.; Sinha, R.; Corey, D.; et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 2017, 545, 495–499. [Google Scholar] [CrossRef]
- Gubin, M.M.; Esaulova, E.; Ward, J.P.; Malkova, O.N.; Runci, D.; Wong, P.; Noguchi, T.; Arthur, C.D.; Meng, W.; Alspach, E.; et al. High-Dimensional Analysis Delineates Myeloid and Lymphoid Compartment Remodeling during Successful Immune-Checkpoint Cancer Therapy. Cell 2018, 175, 1014–1030.e19. [Google Scholar] [CrossRef] [Green Version]
- Sun, N.-Y.; Chen, Y.-L.; Wu, W.-Y.; Lin, H.-W.; Chiang, Y.-C.; Chang, C.-F.; Tai, Y.-J.; Hsu, H.-C.; Chen, C.-A.; Sun, W.-Z.; et al. Blockade of PD-L1 Enhances Cancer Immunotherapy by Regulating Dendritic Cell Maturation and Macrophage Polarization. Cancers 2019, 11, 1400. [Google Scholar] [CrossRef] [Green Version]
- Stecher, C.; Battin, C.; Leitner, J.; Zettl, M.; Grabmeier-Pfistershammer, K.; Höller, C.; Zlabinger, G.J.; Steinberger, P. PD-1 Blockade Promotes Emerging Checkpoint Inhibitors in Enhancing T Cell Responses to Allogeneic Dendritic Cells. Front. Immunol. 2017, 8, 572. [Google Scholar] [CrossRef]
- Wang, Y.; Xiang, Y.; Xin, V.W.; Wang, X.-W.; Peng, X.-C.; Liu, X.-Q.; Wang, D.; Li, N.; Cheng, J.-T.; Lyv, Y.-N.; et al. Dendritic cell biology and its role in tumor immunotherapy. J. Hematol. Oncol. 2020, 13, 107. [Google Scholar] [CrossRef] [PubMed]
- Peng, Q.; Qiu, X.; Zhang, Z.; Zhang, S.; Zhang, Y.; Liang, Y.; Guo, J.; Peng, H.; Chen, M.; Fu, Y.-X.; et al. PD-L1 on dendritic cells attenuates T cell activation and regulates response to immune checkpoint blockade. Nat. Commun. 2020, 11, 4835. [Google Scholar] [CrossRef] [PubMed]
- Garris, C.S.; Arlauckas, S.P.; Kohler, R.H.; Trefny, M.P.; Garren, S.; Piot, C.; Engblom, C.; Pfirschke, C.; Siwicki, M.; Gungabeesoon, J.; et al. Successful Anti-PD-1 Cancer Immunotherapy Requires T Cell-Dendritic Cell Crosstalk Involving the Cytokines IFN-γ and IL-12. Immunity 2018, 49, 1148–1161.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Sun, H.-W.; Yang, Y.-Y.; Chen, H.-T.; Yu, X.-J.; Wu, W.-C.; Xu, Y.-T.; Jin, L.-L.; Wu, X.-J.; Xu, J.; et al. Reprogramming immunosuppressive myeloid cells by activated T cells promotes the response to anti-PD-1 therapy in colorectal cancer. Signal Transduct. Target. Ther. 2021, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Liu, T.; Zhu, W.; Xie, S.; Zhao, Z.; Feng, B.; Guo, H.; Yang, R. Targeting MDSC for Immune-Checkpoint Blockade in Cancer Immunotherapy: Current Progress and New Prospects. Clin. Med. Insights Oncol. 2021, 15, 11795549211035540. [Google Scholar] [CrossRef] [PubMed]
- Schetters, S.T.T.; Rodriguez, E.; Kruijssen, L.J.W.; Crommentuijn, M.H.W.; Boon, L.; Van den Bossche, J.; Den Haan, J.M.M.; Van Kooyk, Y. Monocyte-derived APCs are central to the response of PD1 checkpoint blockade and provide a therapeutic target for combination therapy. J. Immunother. Cancer 2020, 8, e000588. [Google Scholar] [CrossRef] [PubMed]
- De Cicco, P.; Ercolano, G.; Ianaro, A. The New Era of Cancer Immunotherapy: Targeting Myeloid-Derived Suppressor Cells to Overcome Immune Evasion. Front. Immunol. 2020, 11, 1680. [Google Scholar] [CrossRef]
- Romano, E.; Kusio-Kobialka, M.; Foukas, P.G.; Baumgaertner, P.; Meyer, C.; Ballabeni, P.; Michielin, O.; Weide, B.; Romero, P.; Speiser, D.E. Ipilimumab-dependent cell-mediated cytotoxicity of regulatory T cells ex vivo by nonclassical monocytes in melanoma patients. Proc. Natl. Acad. Sci. USA 2015, 112, 6140–6145. [Google Scholar] [CrossRef] [Green Version]
- Yajuk, O.; Baron, M.; Toker, S.; Zelter, T.; Fainsod-Levi, T.; Granot, Z. The PD-L1/PD-1 Axis Blocks Neutrophil Cytotoxicity in Cancer. Cells 2021, 10, 1510. [Google Scholar] [CrossRef]
- Zhang, Y.; Guoqiang, L.; Sun, M.; Lu, X. Targeting and exploitation of tumor-associated neutrophils to enhance immunotherapy and drug delivery for cancer treatment. Cancer Biol. Med. 2020, 17, 32–43. [Google Scholar] [CrossRef]
- Sun, L.; Clavijo, P.E.; Robbins, Y.; Patel, P.; Friedman, J.; Greene, S.; Das, R.; Silvin, C.; Van Waes, C.; Horn, L.A.; et al. Inhibiting myeloid-derived suppressor cell trafficking enhances T cell immunotherapy. JCI Insight 2019, 4, e126853. [Google Scholar] [CrossRef] [Green Version]
- Yamauchi, Y.; Safi, S.; Blattner, C.; Rathinasamy, A.; Umansky, L.; Juenger, S.; Warth, A.; Eichhorn, M.; Muley, T.; Herth, F.J.F.; et al. Circulating and Tumor Myeloid-derived Suppressor Cells in Resectable Non-Small Cell Lung Cancer. Am. J. Respir. Crit. Care Med. 2018, 198, 777–787. [Google Scholar] [CrossRef]
- Yoshidome, H.; Kohno, H.; Shida, T.; Kimura, F.; Shimizu, H.; Ohtsuka, M.; Nakatani, Y.; Miyazaki, M. Significance of monocyte chemoattractant protein-1 in angiogenesis and survival in colorectal liver metastases. Int. J. Oncol. 2009, 34, 923–930. [Google Scholar] [CrossRef] [Green Version]
- Buchbinder, E.I.; Desai, A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am. J. Clin. Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis-Marcisak, E.F.; Fitzgerald, A.A.; Kessler, M.D.; Danilova, L.; Jaffee, E.M.; Zaidi, N.; Weiner, L.M.; Fertig, E.J. A novel mechanism of natural killer cell response to anti-CTLA-4 therapy identified by integrative analysis of mouse and human tumors. bioRxiv 2020. [Google Scholar] [CrossRef]
- Krummel, M.F.; Allison, J.P. CTLA-4 engagement inhibits IL-2 accumulation and cell cycle progression upon activation of resting T cells. J. Exp. Med. 1996, 183, 2533–2540. [Google Scholar] [CrossRef]
- Leach, D.R.; Krummel, M.F.; Allison, J.P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 1996, 271, 1734. [Google Scholar]
- Peeraphatdit, T.; Wang, J.; Odenwald, M.A.; Hu, S.; Hart, J.; Charlton, M.R. Hepatotoxicity from Immune Checkpoint Inhibitors: A Systematic Review and Management Recommendation. Hepatology 2020, 72, 315–329. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Giaccone, G.; de Marinis, F.; Reinmuth, N.; Vergnenegre, A.; Barrios, C.H.; Morise, M.; Felip, E.; Andric, Z.; Geater, S.; et al. Atezolizumab for First-Line Treatment of PD-L1–Selected Patients with NSCLC. N. Engl. J. Med. 2020, 383, 1328–1339. [Google Scholar] [CrossRef]
- Weber, J.; Thompson, J.A.; Hamid, O.; Minor, D.; Amin, A.; Ron, I.; Ridolfi, R.; Assi, H.; Maraveyas, A.; Berman, D.; et al. A randomized, double-blind, placebo-controlled, phase II study comparing the tolerability and efficacy of ipilimumab administered with or without prophylactic budesonide in patients with unresectable stage III or IV melanoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2009, 15, 5591–5598. [Google Scholar] [CrossRef] [Green Version]
- Postow, M.A.; Chesney, J.; Pavlick, A.C.; Robert, C.; Grossmann, K.; McDermott, D.; Linette, G.P.; Meyer, N.; Giguere, J.K.; Agarwala, S.S.; et al. Nivolumab and Ipilimumab versus Ipilimumab in Untreated Melanoma. N. Engl. J. Med. 2015, 372, 2006–2017. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Ran, Y.; Wang, K.; Zhu, Y.; Li, J. Incidence and risk of hepatic toxicities with PD-1 inhibitors in cancer patients: A meta-analysis. Drug Des. Dev. Ther. 2016, 10, 3153–3161. [Google Scholar] [CrossRef] [Green Version]
- Tiegs, G.; Lohse, A.W. Immune tolerance: What is unique about the liver. J. Autoimmun. 2010, 34, 1–6. [Google Scholar] [CrossRef]
- Bogdanos, D.P.; Gao, B.; Gershwin, M.E. Liver immunology. Compr. Physiol. 2013, 3, 567–598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horst, A.K.; Neumann, K.; Diehl, L.; Tiegs, G. Modulation of liver tolerance by conventional and nonconventional antigen-presenting cells and regulatory immune cells. Cell. Mol. Immunol. 2016, 13, 277–292. [Google Scholar] [CrossRef] [PubMed]
- Kubes, P.; Jenne, C. Immune Responses in the Liver. Annu. Rev. Immunol. 2018, 36, 247–277. [Google Scholar] [CrossRef]
- Metushi, I.G.; Hayes, M.A.; Uetrecht, J. Treatment of PD-1-/- mice with amodiaquine and anti-CTLA4 leads to liver injury similar to idiosyncratic liver injury in patients. Hepatology 2015, 61, 1332–1342. [Google Scholar] [CrossRef] [PubMed]
- Medina-Echeverz, J.; Ma, C.; Duffy, A.G.; Eggert, T.; Hawk, N.; Kleiner, D.E.; Korangy, F.; Greten, T.F. Systemic agonistic anti-CD40 treatment of tumor-bearing mice modulates hepatic myeloid-suppressive cells and causes immune-mediated liver damage. Cancer Immunol. Res. 2015, 3, 557–566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Affolter, T.; Llewellyn, H.P.; Bartlett, D.W.; Zong, Q.; Xia, S.; Torti, V.; Ji, C. Inhibition of immune checkpoints PD-1, CTLA-4, and IDO1 coordinately induces immune-mediated liver injury in mice. PLoS ONE 2019, 14, e0217276. [Google Scholar] [CrossRef] [PubMed]
- Said, E.A.; Dupuy, F.P.; Trautmann, L.; Zhang, Y.; Shi, Y.; El-Far, M.; Hill, B.J.; Noto, A.; Ancuta, P.; Peretz, Y.; et al. Programmed death-1-induced interleukin-10 production by monocytes impairs CD4+ T cell activation during HIV infection. Nat. Med. 2010, 16, 452–459. [Google Scholar] [CrossRef] [Green Version]
- Bally, A.P.R.; Lu, P.; Tang, Y.; Austin, J.W.; Scharer, C.D.; Ahmed, R.; Boss, J.M. NF-κB regulates PD-1 expression in macrophages. J. Immunol. 2015, 194, 4545–4554. [Google Scholar] [CrossRef] [Green Version]
- Suzman, D.L.; Pelosof, L.; Rosenberg, A.; Avigan, M.I. Hepatotoxicity of immune checkpoint inhibitors: An evolving picture of risk associated with a vital class of immunotherapy agents. Liver Int. 2018, 38, 976–987. [Google Scholar] [CrossRef] [Green Version]
- Fujimura, T.; Sato, Y.; Tanita, K.; Kambayashi, Y.; Otsuka, A.; Fujisawa, Y.; Yoshino, K.; Matsushita, S.; Funakoshi, T.; Hata, H.; et al. Serum levels of soluble CD163 and CXCL5 may be predictive markers for immune-related adverse events in patients with advanced melanoma treated with nivolumab: A pilot study. Oncotarget 2018, 9, 15542–15551. [Google Scholar] [CrossRef] [Green Version]
- McElroy, A.K.; Shrivastava-Ranjan, P.; Harmon, J.R.; Martines, R.B.; Silva-Flannery, L.; Flietstra, T.D.; Kraft, C.S.; Mehta, A.K.; Lyon, G.M.; Varkey, J.B.; et al. Macrophage Activation Marker Soluble CD163 Associated with Fatal and Severe Ebola Virus Disease in Humans. Emerg. Infect. Dis. 2019, 25, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Horiike, N.; Akbar, S.M.F.; Michitaka, K.; Matsuyama, T.; Onji, M. Soluble CD163 in patients with liver diseases: Very high levels of soluble CD163 in patients with fulminant hepatic failure. J. Gastroenterol. 2005, 40, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Grewal, I.S.; Flavell, R.A. CD40 and CD154 in cell-mediated immunity. Annu. Rev. Immunol. 1998, 16, 111–135. [Google Scholar] [CrossRef] [PubMed]
- Caux, C.; Massacrier, C.; Vanbervliet, B.; Dubois, B.; Van Kooten, C.; Durand, I.; Banchereau, J. Activation of human dendritic cells through CD40 cross-linking. J. Exp. Med. 1994, 180, 1263–1272. [Google Scholar] [CrossRef] [PubMed]
- Diehl, L.; den Boer, A.T.; Schoenberger, S.P.; van der Voort, E.I.H.; Schumacher, T.N.M.; Melief, C.J.M.; Offringa, R.; Toes, R.E.M. CD40 activation in vivo overcomes peptide-induced peripheral cytotoxic T-lymphocyte tolerance and augments anti-tumor vaccine efficacy. Nat. Med. 1999, 5, 774–779. [Google Scholar] [CrossRef] [PubMed]
- French, R.R.; Chan, H.T.C.; Tutt, A.L.; Glennie, M.J. CD40 antibody evokes a cytotoxic T-cell response that eradicates lymphoma and bypasses T-cell help. Nat. Med. 1999, 5, 548–553. [Google Scholar] [CrossRef]
- Vonderheide, R.H.; Burg, J.M.; Mick, R.; Trosko, J.A.; Li, D.; Shaik, M.N.; Tolcher, A.W.; Hamid, O. Phase I study of the CD40 agonist antibody CP-870,893 combined with carboplatin and paclitaxel in patients with advanced solid tumors. Oncoimmunology 2013, 2, e23033. [Google Scholar] [CrossRef] [Green Version]
- Hoechst, B.; Ormandy, L.A.; Ballmaier, M.; Lehner, F.; Krüger, C.; Manns, M.P.; Greten, T.F.; Korangy, F. A New Population of Myeloid-Derived Suppressor Cells in Hepatocellular Carcinoma Patients Induces CD4+CD25+Foxp3+ T Cells. Gastroenterology 2008, 135, 234–243. [Google Scholar] [CrossRef]
- Kapanadze, T.; Gamrekelashvili, J.; Ma, C.; Chan, C.; Zhao, F.; Hewitt, S.; Zender, L.; Kapoor, V.; Felsher, D.W.; Manns, M.P.; et al. Regulation of accumulation and function of myeloid derived suppressor cells in different murine models of hepatocellular carcinoma. J. Hepatol. 2013, 59, 1007–1013. [Google Scholar] [CrossRef] [Green Version]
- Connolly, M.K.; Mallen-St. Clair, J.; Bedrosian, A.S.; Malhotra, A.; Vera, V.; Ibrahim, J.; Henning, J.; Pachter, H.L.; Bar-Sagi, D.; Frey, A.B.; et al. Distinct populations of metastases-enabling myeloid cells expand in the liver of mice harboring invasive and preinvasive intra-abdominal tumor. J. Leukoc. Biol. 2010, 87, 713–725. [Google Scholar] [CrossRef]
- Jaillon, S.; Ponzetta, A.; Di Mitri, D.; Santoni, A.; Bonecchi, R.; Mantovani, A. Neutrophil diversity and plasticity in tumour progression and therapy. Nat. Rev. Cancer 2020, 20, 485–503. [Google Scholar] [CrossRef] [PubMed]
- Steele, C.W.; Karim, S.A.; Leach, J.D.G.; Bailey, P.; Upstill-Goddard, R.; Rishi, L.; Foth, M.; Bryson, S.; McDaid, K.; Wilson, Z.; et al. CXCR2 Inhibition Profoundly Suppresses Metastases and Augments Immunotherapy in Pancreatic Ductal Adenocarcinoma. Cancer Cell 2016, 29, 832–845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melero, I.; Shuford, W.W.; Newby, S.A.; Aruffo, A.; Ledbetter, J.A.; Hellström, K.E.; Mittler, R.S.; Chen, L. Monoclonal antibodies against the 4-1BB T-cell activation molecule eradicate established tumors. Nat. Med. 1997, 3, 682–685. [Google Scholar] [CrossRef] [PubMed]
- Hammers, H.J.; Plimack, E.R.; Infante, J.R.; Rini, B.I.; McDermott, D.F.; Lewis, L.D.; Voss, M.H.; Sharma, P.; Pal, S.K.; Razak, A.R.A.; et al. Safety and Efficacy of Nivolumab in Combination with Ipilimumab in Metastatic Renal Cell Carcinoma: The CheckMate 016 Study. J. Clin. Oncol. 2017, 35, 3851–3858. [Google Scholar] [CrossRef] [Green Version]
- Hellmann, M.D.; Ciuleanu, T.E.; Pluzanski, A.; Lee, J.S.; Otterson, G.A.; Audigier-Valette, C.; Minenza, E.; Linardou, H.; Burgers, S.; Salman, P.; et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N. Engl. J. Med. 2018, 378, 2093–2104. [Google Scholar] [CrossRef]
- Karamchandani, D.M.; Chetty, R. Immune checkpoint inhibitor-induced gastrointestinal and hepatic injury: Pathologists’ perspective. J. Clin. Pathol. 2018, 71, 665–671. [Google Scholar] [CrossRef]
- Everett, J.; Srivastava, A.; Misdraji, J. Fibrin Ring Granulomas in Checkpoint Inhibitor-induced Hepatitis. Am. J. Surg. Pathol. 2017, 41, 134–137. [Google Scholar] [CrossRef]
- Prendergast, G.C.; Smith, C.; Thomas, S.; Mandik-Nayak, L.; Laury-Kleintop, L.; Metz, R.; Muller, A.J. Indoleamine 2,3-dioxygenase pathways of pathogenic inflammation and immune escape in cancer. Cancer Immunol. Immunother. 2014, 63, 721–735. [Google Scholar] [CrossRef]
- Godin-Ethier, J.; Hanafi, L.-A.; Piccirillo, C.A.; Lapointe, R. Indoleamine 2,3-Dioxygenase Expression in Human Cancers: Clinical and Immunologic Perspectives. Clin. Cancer Res. 2011, 17, 6985–6991. [Google Scholar] [CrossRef] [Green Version]
- Meireson, A.; Devos, M.; Brochez, L. IDO Expression in Cancer: Different Compartment, Different Functionality? Front. Immunol. 2020, 11, 531491. [Google Scholar] [CrossRef]
- Sumpter, T.L.; Dangi, A.; Matta, B.M.; Huang, C.; Stolz, D.B.; Vodovotz, Y.; Thomson, A.W.; Gandhi, C.R. Hepatic Stellate Cells Undermine the Allostimulatory Function of Liver Myeloid Dendritic Cells via STAT3-Dependent Induction of IDO. J. Immunol. 2012, 189, 3848–3858. [Google Scholar] [CrossRef] [PubMed]
- Nagano, J.; Shimizu, M.; Hara, T.; Shirakami, Y.; Kochi, T.; Nakamura, N.; Ohtaki, H.; Ito, H.; Tanaka, T.; Tsurumi, H.; et al. Effects of Indoleamine 2,3-Dioxygenase Deficiency on High-Fat Diet-Induced Hepatic Inflammation. PLoS ONE 2013, 8, e73404. [Google Scholar] [CrossRef]
- Ito, H.; Hoshi, M.; Ohtaki, H.; Taguchi, A.; Ando, K.; Ishikawa, T.; Osawa, Y.; Hara, A.; Moriwaki, H.; Saito, K.; et al. Ability of IDO To Attenuate Liver Injury in α-Galactosylceramide–Induced Hepatitis Model. J. Immunol. 2010, 185, 4554–4560. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Thomas, L.; Bondarenko, I.; O’Day, S.; Weber, J.; Garbe, C.; Lebbe, C.; Baurain, J.-F.; Testori, A.; Grob, J.-J.; et al. Ipilimumab plus Dacarbazine for Previously Untreated Metastatic Melanoma. N. Engl. J. Med. 2011, 364, 2517–2526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langer, C.J.; Gadgeel, S.M.; Borghaei, H.; Papadimitrakopoulou, V.A.; Patnaik, A.; Powell, S.F.; Gentzler, R.D.; Martins, R.G.; Stevenson, J.P.; Jalal, S.I.; et al. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: A randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol. 2016, 17, 1497–1508. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Luft, A.; Vicente, D.; Tafreshi, A.; Gümüş, M.; Mazières, J.; Hermes, B.; Çay Şenler, F.; Csőszi, T.; Fülöp, A.; et al. Pembrolizumab plus Chemotherapy for Squamous Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2040–2051. [Google Scholar] [CrossRef]
- Horn, L.; Mansfield, A.S.; Szczęsna, A.; Havel, L.; Krzakowski, M.; Hochmair, M.J.; Huemer, F.; Losonczy, G.; Johnson, M.L.; Nishio, M.; et al. First-Line Atezolizumab plus Chemotherapy in Extensive-Stage Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2220–2229. [Google Scholar] [CrossRef]
- Schmid, P.; Adams, S.; Rugo, H.S.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Hegg, R.; Im, S.-A.; Shaw Wright, G.; et al. Atezolizumab and Nab-Paclitaxel in Advanced Triple-Negative Breast Cancer. N. Engl. J. Med. 2018, 379, 2108–2121. [Google Scholar] [CrossRef]
- Socinski, M.A.; Jotte, R.M.; Cappuzzo, F.; Orlandi, F.; Stroyakovskiy, D.; Nogami, N.; Rodríguez-Abreu, D.; Moro-Sibilot, D.; Thomas, C.A.; Barlesi, F.; et al. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N. Engl. J. Med. 2018, 378, 2288–2301. [Google Scholar] [CrossRef]
- West, H.; McCleod, M.; Hussein, M.; Morabito, A.; Rittmeyer, A.; Conter, H.J.; Kopp, H.-G.; Daniel, D.; McCune, S.; Mekhail, T.; et al. Atezolizumab in combination with carboplatin plus nab-paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): A multicentre, randomised, open-label, phase 3 tria. Lancet Oncol. 2019, 20, 924–937. [Google Scholar] [CrossRef]
- Gandhi, L.; Rodríguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab plus Chemotherapy in Metastatic Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef] [PubMed]
- Heinhuis, K.M.; Ros, W.; Kok, M.; Steeghs, N.; Beijnen, J.H.; Schellens, J.H.M. Enhancing antitumor response by combining immune checkpoint inhibitors with chemotherapy in solid tumors. Ann. Oncol. 2019, 30, 219–235. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Humeau, J.; Buqué, A.; Zitvogel, L.; Kroemer, G. Immunostimulation with chemotherapy in the era of immune checkpoint inhibitors. Nat. Rev. Clin. Oncol. 2020, 17, 725–741. [Google Scholar] [CrossRef]
- Dimeloe, S.; Frick, C.; Fischer, M.; Gubser, P.M.; Razik, L.; Bantug, G.R.; Ravon, M.; Langenkamp, A.; Hess, C. Human regulatory T cells lack the cyclophosphamide-extruding transporter ABCB1 and are more susceptible to cyclophosphamide-induced apoptosis. Eur. J. Immunol. 2014, 44, 3614–3620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otsubo, D.; Yamashita, K.; Fujita, M.; Nishi, M.; Kimura, Y.; Hasegawa, H.; Suzuki, S.; Kakeji, Y. Early-phase Treatment by Low-dose 5-Fluorouracil or Primary Tumor Resection Inhibits MDSC-mediated Lung Metastasis Formation. Anticancer Res. 2015, 35, 4425–4431. [Google Scholar] [PubMed]
- Schaer, D.A.; Geeganage, S.; Amaladas, N.; Lu, Z.H.; Rasmussen, E.R.; Sonyi, A.; Chin, D.; Capen, A.; Li, Y.; Meyer, C.M.; et al. The Folate Pathway Inhibitor Pemetrexed Pleiotropically Enhances Effects of Cancer Immunotherapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2019, 25, 7175–7188. [Google Scholar] [CrossRef]
- Wanderley, C.W.; Colón, D.F.; Luiz, J.P.M.; Oliveira, F.F.; Viacava, P.R.; Leite, C.A.; Pereira, J.A.; Silva, C.M.; Silva, C.R.; Silva, R.L.; et al. Paclitaxel Reduces Tumor Growth by Reprogramming Tumor-Associated Macrophages to an M1 Profile in a TLR4-Dependent Manner. Cancer Res. 2018, 78, 5891–5900. [Google Scholar] [CrossRef] [Green Version]
- Galluzzi, L.; Buqué, A.; Kepp, O.; Zitvogel, L.; Kroemer, G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 2017, 17, 97–111. [Google Scholar] [CrossRef]
- Vanmeerbeek, I.; Sprooten, J.; De Ruysscher, D.; Tejpar, S.; Vandenberghe, P.; Fucikova, J.; Spisek, R.; Zitvogel, L.; Kroemer, G.; Galluzzi, L.; et al. Trial watch: Chemotherapy-induced immunogenic cell death in immuno-oncology. Oncoimmunology 2020, 9, 1703449. [Google Scholar] [CrossRef] [Green Version]
- Dosset, M.; Vargas, T.R.; Lagrange, A.; Boidot, R.; Végran, F.; Roussey, A.; Chalmin, F.; Dondaine, L.; Paul, C.; Lauret Marie-Joseph, E.; et al. PD-1/PD-L1 pathway: An adaptive immune resistance mechanism to immunogenic chemotherapy in colorectal cancer. Oncoimmunology 2018, 7, e1433981. [Google Scholar] [CrossRef] [Green Version]
- Peng, J.; Hamanishi, J.; Matsumura, N.; Abiko, K.; Murat, K.; Baba, T.; Yamaguchi, K.; Horikawa, N.; Hosoe, Y.; Murphy, S.K.; et al. Chemotherapy Induces Programmed Cell Death-Ligand 1 Overexpression via the Nuclear Factor-κB to Foster an Immunosuppressive Tumor Microenvironment in Ovarian Cancer. Cancer Res. 2015, 75, 5034–5045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fournel, L.; Wu, Z.; Stadler, N.; Damotte, D.; Lococo, F.; Boulle, G.; Ségal-Bendirdjian, E.; Bobbio, A.; Icard, P.; Trédaniel, J.; et al. Cisplatin increases PD-L1 expression and optimizes immune check-point blockade in non-small cell lung cancer. Cancer Lett. 2019, 464, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Li, W.; Hu, J.; Zhu, E.C.; Su, Q. Hepatotoxicity in patients with solid tumors treated with PD-1/PD-L1 inhibitors alone, PD-1/PD-L1 inhibitors plus chemotherapy, or chemotherapy alone: Systematic review and meta-analysis. Eur. J. Clin. Pharmacol. 2020, 76, 1345–1354. [Google Scholar] [CrossRef] [PubMed]
- Mak, A.; Uetrecht, J. The Combination of Anti-CTLA-4 and PD1-/- Mice Unmasks the Potential of Isoniazid and Nevirapine to Cause Liver Injury. Chem. Res. Toxicol. 2015, 28, 2287–2291. [Google Scholar] [CrossRef]
- Adam, K.; Iuga, A.; Tocheva, A.S.; Mor, A. A novel mouse model for checkpoint inhibitor-induced adverse events. PLoS ONE 2021, 16, e0246168. [Google Scholar] [CrossRef]
- Park, R.; Lopes, L.; Saeed, A. Anti-PD-1/L1-associated immune-related adverse events as harbinger of favorable clinical outcome: Systematic review and meta-analysis. Clin. Transl. Oncol. 2021, 23, 100–109. [Google Scholar] [CrossRef]
- Cathcart-Rake, E.J.; Sangaralingham, L.R.; Henk, H.J.; Shah, N.D.; Riaz, I.B.; Mansfield, A.S. A Population-based Study of Immunotherapy-related Toxicities in Lung Cancer. Clin. Lung Cancer 2020, 21, 421–427.e2. [Google Scholar] [CrossRef]
- Chang, C.-Y.; Park, H.; Malone, D.C.; Wang, C.-Y.; Wilson, D.L.; Yeh, Y.-M.; Van Boemmel-Wegmann, S.; Lo-Ciganic, W.-H. Immune Checkpoint Inhibitors and Immune-Related Adverse Events in Patients with Advanced Melanoma: A Systematic Review and Network Meta-analysis. JAMA Netw. Open 2020, 3, e201611. [Google Scholar] [CrossRef]
- MacParland, S.A.; Liu, J.C.; Ma, X.-Z.; Innes, B.T.; Bartczak, A.M.; Gage, B.K.; Manuel, J.; Khuu, N.; Echeverri, J.; Linares, I.; et al. Single cell RNA sequencing of human liver reveals distinct intrahepatic macrophage populations. Nat. Commun. 2018, 9, 4383. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Qu, Y.; Xia, P.; Chen, Y.; Zhu, X.; Zhang, J.; Wang, G.; Tian, Y.; Ying, J.; Fan, Z. Transdifferentiation of tumor infiltrating innate lymphoid cells during progression of colorectal cancer. Cell Res. 2020, 30, 610–622. [Google Scholar] [CrossRef]
- Peng, S.; Hebert, L.L.; Eschbacher, J.M.; Kim, S. Single-Cell RNA Sequencing of a Postmenopausal Normal Breast Tissue Identifies Multiple Cell Types That Contribute to Breast Cancer. Cancers 2020, 12, 3639. [Google Scholar] [CrossRef] [PubMed]
- Aliya, S.; Lee, H.; Alhammadi, M.; Umapathi, R.; Huh, Y.S. An Overview on Single-Cell Technology for Hepatocellular Carcinoma Diagnosis. Int. J. Mol. Sci. 2022, 23, 1402. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, D.; Peng, M.; Tang, L.; Ouyang, J.; Xiong, F.; Guo, C.; Tang, Y.; Zhou, Y.; Liao, Q.; et al. Single-cell RNA sequencing in cancer research. J. Exp. Clin. Cancer Res. 2021, 40, 81. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Xu, Z.; Marignani, P.A. Single-cell RNA sequencing for the identification of early-stage lung cancer biomarkers from circulating blood. NPJ Genom. Med. 2021, 6, 87. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Chen, J.; Yang, L.; Cao, M.; Yu, Y.; Zhang, R.; Quan, H.; Jiang, Q.; Hua, Y.; Wei, W.; et al. Single-Cell Sequencing-Enabled Hexokinase 2 Assay for Noninvasive Bladder Cancer Diagnosis and Screening by Detecting Rare Malignant Cells in Urine. Anal. Chem. 2020, 92, 16284–16292. [Google Scholar] [CrossRef] [PubMed]
- Jing, Y.; Liu, J.; Ye, Y.; Pan, L.; Deng, H.; Wang, Y.; Yang, Y.; Diao, L.; Lin, S.H.; Mills, G.B.; et al. Multi-omics prediction of immune-related adverse events during checkpoint immunotherapy. Nat. Commun. 2020, 11, 4946. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, Z.; Wu, D.; Chen, L.; Xie, L. Single-Cell RNA-Seq Analysis Reveals Microenvironmental Infiltration of Plasma Cells and Hepatocytic Prognostic Markers in HCC With Cirrhosis. Front. Oncol. 2020, 10, 596318. [Google Scholar] [CrossRef]
- Fustero-Torre, C.; Jiménez-Santos, M.J.; García-Martín, S.; Carretero-Puche, C.; García-Jimeno, L.; Ivanchuk, V.; Di Domenico, T.; Gómez-López, G.; Al-Shahrour, F. Beyondcell: Targeting cancer therapeutic heterogeneity in single-cell RNA-seq data. Genome Med. 2021, 13, 187. [Google Scholar] [CrossRef]
- Iivanainen, S.; Ekstrom, J.; Virtanen, H.; Kataja, V.V.; Koivunen, J.P. Electronic patient-reported outcomes and machine learning in predicting immune-related adverse events of immune checkpoint inhibitor therapies. BMC Med. Inform. Decis. Mak. 2021, 21, 205. [Google Scholar] [CrossRef]
- Shankaran, V.; Ikeda, H.; Bruce, A.T.; White, J.M.; Swanson, P.E.; Old, L.J.; Schreiber, R.D. IFNγ and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 2001, 410, 1107–1111. [Google Scholar] [CrossRef]
- Dudley, M.E.; Wunderlich, J.R.; Robbins, P.F.; Yang, J.C.; Hwu, P.; Schwartzentruber, D.J.; Topalian, S.L.; Sherry, R.; Restifo, N.P.; Hubicki, A.M.; et al. Cancer Regression and Autoimmunity in Patients After Clonal Repopulation with Antitumor Lymphocytes. Science 2002, 298, 850–854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosenberg, S.A.; Spiess, P.; Lafreniere, R. A new approach to the adoptive immunotherapy of cancer with tumor-infiltrating lymphocytes. Science 1986, 233, 1318–1321. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A.; Packard, B.S.; Aebersold, P.M.; Solomon, D.; Topalian, S.L.; Toy, S.T.; Simon, P.; Lotze, M.T.; Yang, J.C.; Seipp, C.A.; et al. Use of Tumor-Infiltrating Lymphocytes and Interleukin-2 in the Immunotherapy of Patients with Metastatic Melanoma. N. Engl. J. Med. 1988, 319, 1676–1680. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).