Limitations of Nerve Fiber Density as a Prognostic Marker in Predicting Oncological Outcomes in Hepatocellular Carcinoma
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Staging and Surgical Technique
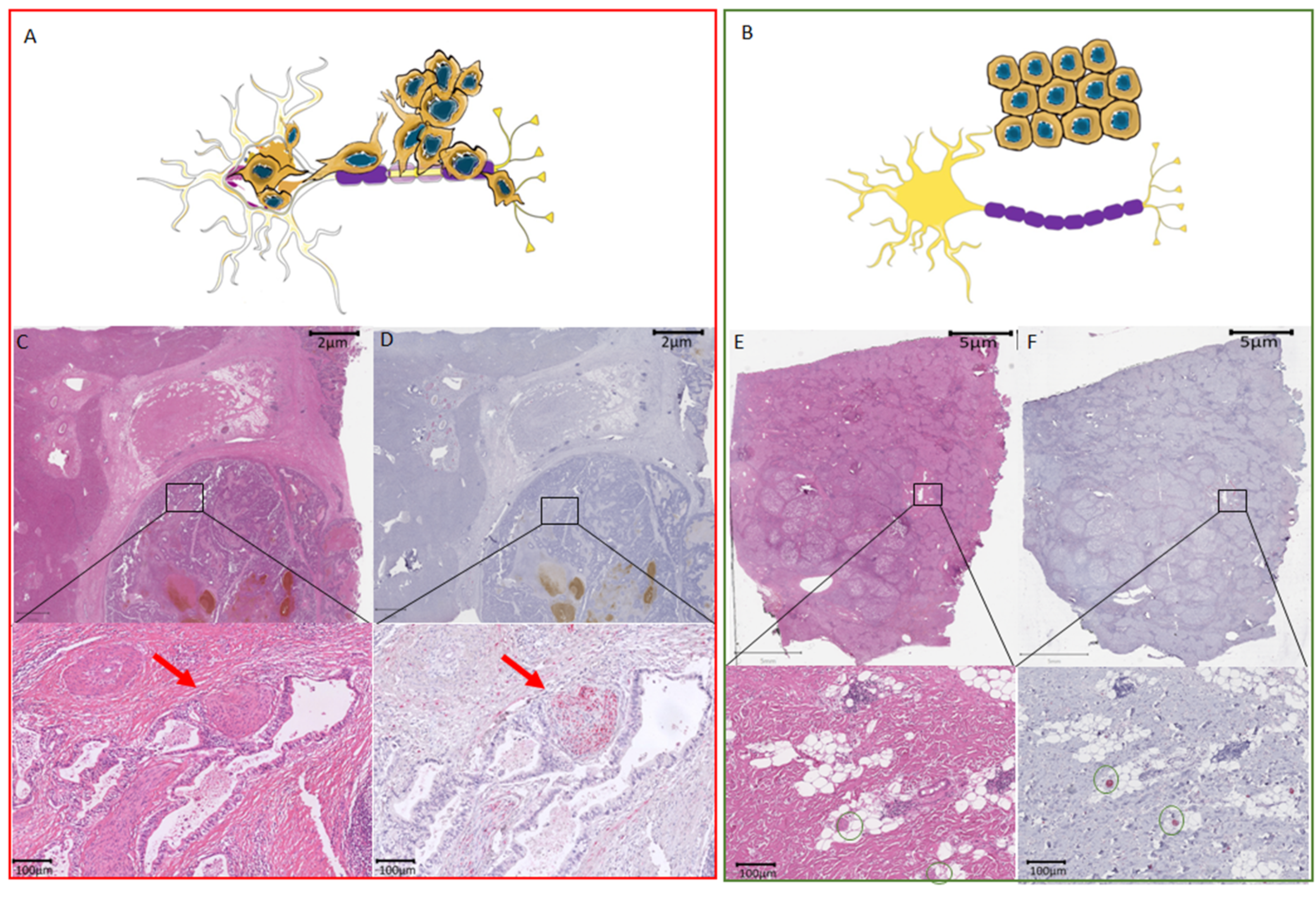
2.3. Assessment of Nerve Fibers
2.4. Statistical Analysis
3. Results
3.1. Patient Cohort
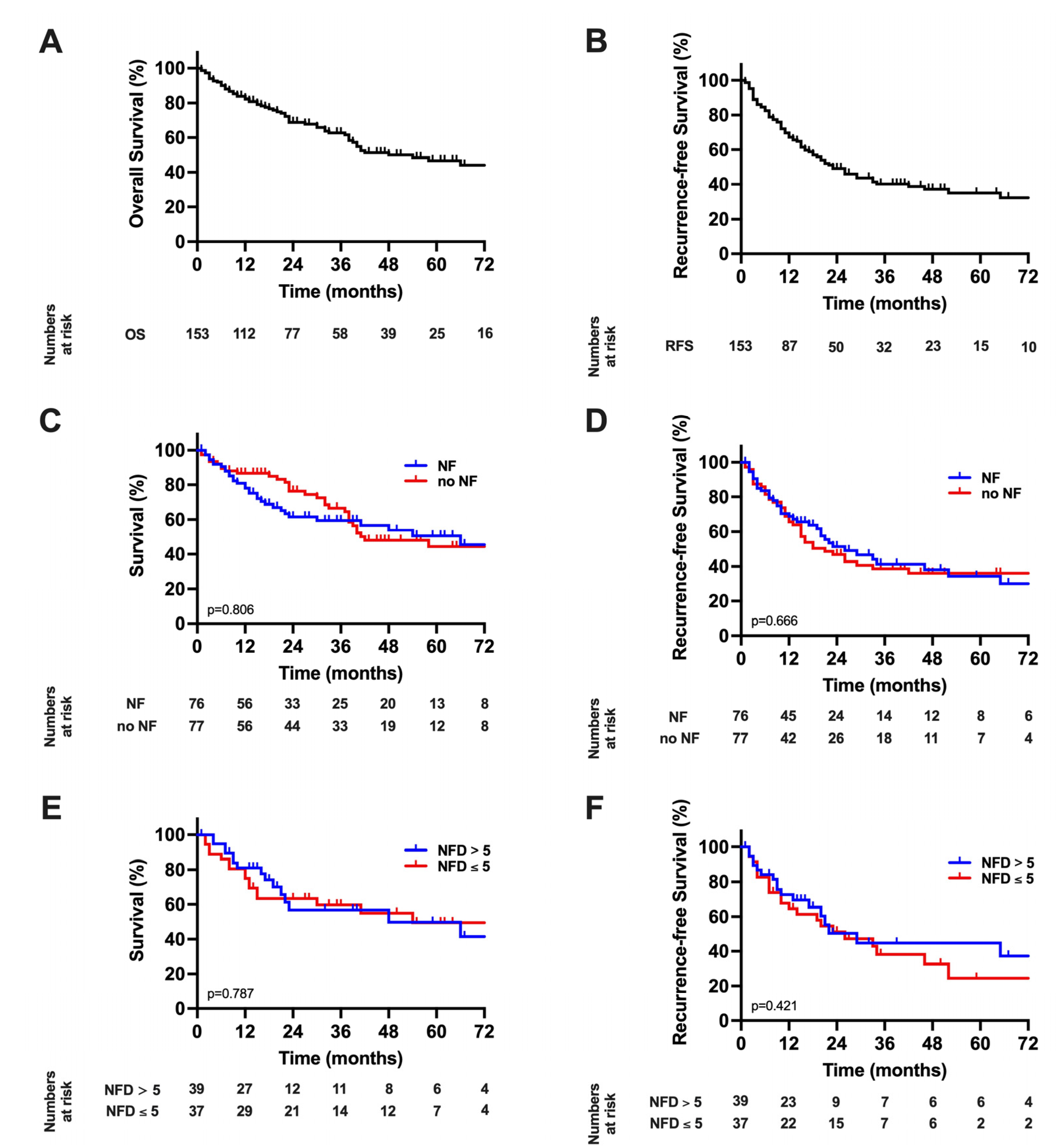
3.2. Survival Analysis with Respect to the Presence of Nerve Fibers in the Tumor Microenvironment and Nerve Fiber Density
3.3. Cox Regression Analysis of the Overall Cohort
3.4. Comparative Analysis of the Overall Patient Cohort with Respect to Nerve Fibers
3.5. Histological Characteristics
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics for Hispanics/Latinos. CA Cancer J. Clin. 2012, 62, 283–298. [Google Scholar] [CrossRef] [PubMed]
- Lurje, G.; Bednarsch, J.; Czigany, Z.; Amygdalos, I.; Meister, F.; Schoning, W.; Ulmer, T.F.; Foerster, M.; Dejong, C.; Neumann, U.P. Prognostic factors of disease-free and overall survival in patients with hepatocellular carcinoma undergoing partial hepatectomy in curative intent. Langenbeck’s Arch. Surg./Dtsch. Ges. Chir. 2018, 403, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Reig, M.; Sherman, M. Evidence-Based Diagnosis, Staging, and Treatment of Patients With Hepatocellular Carcinoma. Gastroenterology 2016, 150, 835–853. [Google Scholar] [CrossRef] [Green Version]
- Vitale, A.; Burra, P.; Frigo, A.C.; Trevisani, F.; Farinati, F.; Spolverato, G.; Volk, M.; Giannini, E.G.; Ciccarese, F.; Piscaglia, F.; et al. Survival benefit of liver resection for patients with hepatocellular carcinoma across different Barcelona Clinic Liver Cancer stages: A multicentre study. J. Hepatol. 2015, 62, 617–624. [Google Scholar] [CrossRef]
- Roayaie, S.; Jibara, G.; Tabrizian, P.; Park, J.W.; Yang, J.; Yan, L.; Schwartz, M.; Han, G.; Izzo, F.; Chen, M.; et al. The role of hepatic resection in the treatment of hepatocellular cancer. Hepatology 2015, 62, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Chen, P.H.; Yeh, J.H.; Hsiao, P.; Lo, G.H.; Tan, T.; Cheng, P.N.; Lin, H.Y.; Chen, Y.S.; Hsieh, K.C.; et al. Clinical outcomes of surgical resection versus radiofrequency ablation in very-early-stage hepatocellular carcinoma: A propensity score matching analysis. BMC Gastroenterol. 2021, 21, 418. [Google Scholar] [CrossRef] [PubMed]
- Kokudo, N.; Takemura, N.; Hasegawa, K.; Takayama, T.; Kubo, S.; Shimada, M.; Nagano, H.; Hatano, E.; Izumi, N.; Kaneko, S.; et al. Clinical practice guidelines for hepatocellular carcinoma: The Japan Society of Hepatology 2017 (4th JSH-HCC guidelines) 2019 update. Hepatol. Res. Off. J. Jpn. Soc. Hepatol. 2019, 49, 1109–1113. [Google Scholar] [CrossRef]
- Rahbari, N.N.; Mehrabi, A.; Mollberg, N.M.; Muller, S.A.; Koch, M.; Buchler, M.W.; Weitz, J. Hepatocellular carcinoma: Current management and perspectives for the future. Ann. Surg. 2011, 253, 453–469. [Google Scholar] [CrossRef]
- Nault, J.C.; Villanueva, A. Biomarkers for Hepatobiliary Cancers. Hepatology 2021, 73 (Suppl. 1), 115–127. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [Green Version]
- Bednarsch, J.; Czigany, Z.; Heise, D.; Joechle, K.; Luedde, T.; Heij, L.; Bruners, P.; Ulmer, T.F.; Neumann, U.P.; Lang, S.A. Prognostic evaluation of HCC patients undergoing surgical resection: An analysis of 8 different staging systems. Langenbeck’s Arch. Surg./Dtsch. Ges. Chir. 2021, 406, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Bednarsch, J.; Kather, J.; Tan, X.; Sivakumar, S.; Cacchi, C.; Wiltberger, G.; Czigany, Z.; Ulmer, F.; Neumann, U.P.; Heij, L.R. Nerve Fibers in the Tumor Microenvironment as a Novel Biomarker for Oncological Outcome in Patients Undergoing Surgery for Perihilar Cholangiocarcinoma. Liver Cancer 2021, 10, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Bednarsch, J.; Tan, X.; Czigany, Z.; Liu, D.; Lang, S.A.; Sivakumar, S.; Kather, J.N.; Appinger, S.; Rosin, M.; Boroojerdi, S.; et al. The Presence of Small Nerve Fibers in the Tumor Microenvironment as Predictive Biomarker of Oncological Outcome Following Partial Hepatectomy for Intrahepatic Cholangiocarcinoma. Cancers 2021, 13, 3661. [Google Scholar] [CrossRef] [PubMed]
- Albo, D.; Akay, C.L.; Marshall, C.L.; Wilks, J.A.; Verstovsek, G.; Liu, H.; Agarwal, N.; Berger, D.H.; Ayala, G.E. Neurogenesis in colorectal cancer is a marker of aggressive tumor behavior and poor outcomes. Cancer 2011, 117, 4834–4845. [Google Scholar] [CrossRef]
- Zhao, C.M.; Hayakawa, Y.; Kodama, Y.; Muthupalani, S.; Westphalen, C.B.; Andersen, G.T.; Flatberg, A.; Johannessen, H.; Friedman, R.A.; Renz, B.W.; et al. Denervation suppresses gastric tumorigenesis. Sci. Transl. Med. 2014, 6, 250ra115. [Google Scholar] [CrossRef] [Green Version]
- Iwasaki, T.; Hiraoka, N.; Ino, Y.; Nakajima, K.; Kishi, Y.; Nara, S.; Esaki, M.; Shimada, K.; Katai, H. Reduction of intrapancreatic neural density in cancer tissue predicts poorer outcome in pancreatic ductal carcinoma. Cancer Sci. 2019, 110, 1491–1502. [Google Scholar] [CrossRef]
- Heij, L.R.; Tan, X.; Kather, J.N.; Niehues, J.M.; Sivakumar, S.; Heussen, N.; van der Kroft, G.; Damink, S.W.M.O.; Lang, S.; Aberle, M.R.; et al. Nerve Fibers in the Tumor Microenvironment Are Co-Localized with Lymphoid Aggregates in Pancreatic Cancer. J. Clin. Med. 2021, 10, 490. [Google Scholar] [CrossRef]
- Bednarsch, J.; Czigany, Z.; Lurje, I.; Trautwein, C.; Ludde, T.; Strnad, P.; Gaisa, N.T.; Barabasch, A.; Bruners, P.; Ulmer, T.; et al. Intraoperative Transfusion of Fresh Frozen Plasma Predicts Morbidity Following Partial Liver Resection for Hepatocellular Carcinoma. J. Gastrointest. Surg. Off. J. Soc. Surg. Aliment. Tract. 2020, 25, 1212–1223. [Google Scholar] [CrossRef]
- Buechter, M.; Thimm, J.; Baba, H.A.; Bertram, S.; Willuweit, K.; Gerken, G.; Kahraman, A. Liver Maximum Capacity: A Novel Test to Accurately Diagnose Different Stages of Liver Fibrosis. Digestion 2019, 100, 45–54. [Google Scholar] [CrossRef]
- Balzan, S.; Belghiti, J.; Farges, O.; Ogata, S.; Sauvanet, A.; Delefosse, D.; Durand, F. The “50-50 criteria” on postoperative day 5: An accurate predictor of liver failure and death after hepatectomy. Ann. Surg. 2005, 242, 824–828, discussion 828–829. [Google Scholar] [CrossRef]
- Rahbari, N.N.; Garden, O.J.; Padbury, R.; Brooke-Smith, M.; Crawford, M.; Adam, R.; Koch, M.; Makuuchi, M.; Dematteo, R.P.; Christophi, C.; et al. Posthepatectomy liver failure: A definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery 2011, 149, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, A. Hepatocellular Carcinoma. Reply. N. Engl. J. Med. 2019, 381, e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Preston, M.; Sherman, L.S. Neural stem cell niches: Roles for the hyaluronan-based extracellular matrix. Front. Biosci. 2011, 3, 1165–1179. [Google Scholar] [CrossRef] [Green Version]
- Gritsenko, P.G.; Ilina, O.; Friedl, P. Interstitial guidance of cancer invasion. J. Pathol. 2012, 226, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Godinho-Silva, C.; Cardoso, F.; Veiga-Fernandes, H. Neuro-Immune Cell Units: A New Paradigm in Physiology. Annu. Rev. Immunol. 2019, 37, 19–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, X.; Sivakumar, S.; Bednarsch, J.; Wiltberger, G.; Kather, J.N.; Niehues, J.; de Vos-Geelen, J.; Valkenburg-van Iersel, L.; Kintsler, S.; Roeth, A.; et al. Nerve fibers in the tumor microenvironment in neurotropic cancer-pancreatic cancer and cholangiocarcinoma. Oncogene 2020, 40, 899–908. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.P.; Tay, S.S.; Leong, S.; Schemann, M. Colocalization of ChAT, DbetaH and NADPH-d in the pancreatic neurons of the newborn guinea pig. Cell Tissue Res. 1998, 294, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Dang, N.; Meng, X.; Song, H. Nicotinic acetylcholine receptors and cancer. Biomed. Rep. 2016, 4, 515–518. [Google Scholar] [CrossRef] [Green Version]
- Sha, M.; Cao, J.; Sun, H.Y.; Tong, Y.; Xia, Q. Neuroendocrine regulation of cholangiocarcinoma: A status quo review. Biochim. Biophys. Acta Rev. Cancer 2019, 1872, 66–73. [Google Scholar] [CrossRef]
- Franchitto, A.; Onori, P.; Renzi, A.; Carpino, G.; Mancinelli, R.; Alvaro, D.; Gaudio, E. Recent advances on the mechanisms regulating cholangiocyte proliferation and the significance of the neuroendocrine regulation of cholangiocyte pathophysiology. Ann. Transl. Med. 2013, 1, 27. [Google Scholar] [CrossRef]
- Shirai, K.; Ebata, T.; Oda, K.; Nishio, H.; Nagasaka, T.; Nimura, Y.; Nagino, M. Perineural invasion is a prognostic factor in intrahepatic cholangiocarcinoma. World J. Surg. 2008, 32, 2395–2402. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.B.; Patel, S.H.; Kooby, D.A.; Weber, S.; Bloomston, M.; Cho, C.; Hatzaras, I.; Schmidt, C.; Winslow, E.; Staley, C.A., 3rd; et al. Lymphovascular and perineural invasion as selection criteria for adjuvant therapy in intrahepatic cholangiocarcinoma: A multi-institution analysis. HPB Off. J. Int. Hepato Pancreato Biliary Assoc. 2012, 14, 514–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Partecke, L.I.; Kading, A.; Trung, D.N.; Diedrich, S.; Sendler, M.; Weiss, F.; Kuhn, J.P.; Mayerle, J.; Beyer, K.; von Bernstorff, W.; et al. Subdiaphragmatic vagotomy promotes tumor growth and reduces survival via TNFalpha in a murine pancreatic cancer model. Oncotarget 2017, 8, 22501–22512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamiya, A.; Hayama, Y.; Kato, S.; Shimomura, A.; Shimomura, T.; Irie, K.; Kaneko, R.; Yanagawa, Y.; Kobayashi, K.; Ochiya, T. Genetic manipulation of autonomic nerve fiber innervation and activity and its effect on breast cancer progression. Nat. Neurosci. 2019, 22, 1289–1305. [Google Scholar] [CrossRef]
- Zhao, Q.; Yang, Y.; Liang, X.; Du, G.; Liu, L.; Lu, L.; Dong, J.; Han, H.; Zhang, G. The clinicopathological significance of neurogenesis in breast cancer. BMC Cancer 2014, 14, 484. [Google Scholar] [CrossRef] [Green Version]
- Meister, F.A.; Lurje, G.; Verhoeven, S.; Wiltberger, G.; Heij, L.; Liu, W.J.; Jiang, D.; Bruners, P.; Lang, S.A.; Ulmer, T.F.; et al. The Role of Sarcopenia and Myosteatosis in Short- and Long-Term Outcomes Following Curative-Intent Surgery for Hepatocellular Carcinoma in a European Cohort. Cancers 2022, 14, 720. [Google Scholar] [CrossRef]
- Andreou, A.; Bahra, M.; Schmelzle, M.; Ollinger, R.; Sucher, R.; Sauer, I.M.; Guel-Klein, S.; Struecker, B.; Eurich, D.; Klein, F.; et al. Predictive factors for extrahepatic recurrence of hepatocellular carcinoma following liver transplantation. Clin. Transplant. 2016, 30, 819–827. [Google Scholar] [CrossRef]
- Shimozawa, N.; Hanazaki, K. Longterm prognosis after hepatic resection for small hepatocellular carcinoma. J. Am. Coll. Surg. 2004, 198, 356–365. [Google Scholar] [CrossRef]
- Yau, T.; Tang, V.Y.; Yao, T.J.; Fan, S.T.; Lo, C.M.; Poon, R.T. Development of Hong Kong Liver Cancer staging system with treatment stratification for patients with hepatocellular carcinoma. Gastroenterology 2014, 146, 1691–1700.e3. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Regalia, E.; Doci, R.; Andreola, S.; Pulvirenti, A.; Bozzetti, F.; Montalto, F.; Ammatuna, M.; Morabito, A.; Gennari, L. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N. Engl. J. Med. 1996, 334, 693–699. [Google Scholar] [CrossRef]
- Chen, S.H.; Zhang, B.Y.; Zhou, B.; Zhu, C.Z.; Sun, L.Q.; Feng, Y.J. Perineural invasion of cancer: A complex crosstalk between cells and molecules in the perineural niche. Am. J. Cancer Res. 2019, 9, 1–21. [Google Scholar] [PubMed]
- Karak, S.G.; Quatrano, N.; Buckley, J.; Ricci, A., Jr. Prevalence and significance of perineural invasion in invasive breast carcinoma. Connect. Med. 2010, 74, 17–21. [Google Scholar]
- Knijn, N.; Mogk, S.C.; Teerenstra, S.; Simmer, F.; Nagtegaal, I.D. Perineural Invasion is a Strong Prognostic Factor in Colorectal Cancer: A Systematic Review. Am. J. Surg. Pathol. 2016, 40, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zheng, Q.; Lu, Z.; Wang, L.; Ding, L.; Xia, L.; Zhang, H.; Wang, M.; Chen, Y.; Li, G. Role of the nervous system in cancers: A review. Cell Death Discov. 2021, 7, 76. [Google Scholar] [CrossRef] [PubMed]
- Sirica, A.E.; Gores, G.J. Desmoplastic stroma and cholangiocarcinoma: Clinical implications and therapeutic targeting. Hepatology 2014, 59, 2397–2402. [Google Scholar] [CrossRef] [Green Version]
- Kepper, M.; Keast, J. Immunohistochemical properties and spinal connections of pelvic autonomic neurons that innervate the rat prostate gland. Cell Tissue Res. 1995, 281, 533–542. [Google Scholar] [CrossRef]
| Variables | Overall Cohort (n = 153) | NF Positive (n = 76) | NF Negative (n = 77) | p Value |
|---|---|---|---|---|
| Demographics | ||||
| Gender, m/f (%) | 105 (68.6)/48 (31.4) | 47 (61.8)/29 (38.2) | 58 (75.3)/19 (24.7) | 0.072 |
| Age (years) | 69 (61–75) | 68 (59–75) | 70 (63–76) | 0.071 |
| BMI (kg/m2) | 26 (23–29) | 26 (23–29) | 27 (23–30) | 0.512 |
| Recurrence resection, n (%) | 13 (8.5) | 9 (11.8) | 4 (5.2) | 0.374 |
| Preoperative treatment | ||||
| Preoperative PVE, n (%) | 7 (4.6) | 3 (3.9) | 4 (5.2) | 0.712 |
| Preoperative TACE, n (%) | 8 (5.2) | 4 (5.3) | 4 (5.2) | 0.985 |
| Preoperative TARE, n (%) | 2 (1.3) | 2 (2.6) | 0 | 0.152 |
| ASA, n (%) | 0.508 | |||
| I | 2 (1.3) | 2 (2.6) | 0 | |
| II | 50 (32.7) | 23 (30.3) | 27 (35.1) | |
| III | 97 (63.4) | 49 (64.5) | 48 (62.3) | |
| IV | 4 (2.6) | 2 (2.6) | 2 (2.6) | |
| V | 0 | 0 | 0 | |
| Liver disease, n (%) | 0.575 | |||
| ALD | 34 (22.2) | 15 (19.7) | 19 (24.7) | |
| NAFLD | 60 (39.2) | 28 (36.8) | 32 (41.6) | |
| Viral | 39 (25.5) | 23 (30.3) | 16 (20.8) | |
| Cryptogenic/others | 20 (13.1) | 10 (13.2) | 10 (13.0) | |
| Preoperative liver function | ||||
| MELD Score | 6 (6–7) | 6 (6–7) | 6 (6–7) | 0.965 |
| AFP (ng/mL) | 8 (3–53) | 11 (3–95) | 6 (3–45) | 0.202 |
| Albumin (g/dL) | 4.1 (3.7–4.5) | 4.1 (3.6–4.4) | 4.1 (3.8–4.5) | 0.269 |
| AST (U/L) | 40 (27–58) | 40 (28–63) | 38 (26–58) | 0.374 |
| ALT (U/L) | 33 (23–54) | 37 (25–58) | 30 (21–51) | 0.123 |
| GGT (U/L) | 90 (51–213) | 92 (55–178) | 90 (50–267) | 0.822 |
| Total bilirubin (mg/dL) | 0.5 (0.4–0.8) | 0.5 (0.4–0.8) | 0.6 (0.4–0.8) | 0.515 |
| Platelet count (/nL) | 221 (163–279) | 225 (161–278) | 206 (168–282) | 0.818 |
| Alkaline Phosphatase (U/L) | 100 (77–140) | 95 (75–180) | 103 (79–134) | 0.860 |
| Prothrombin time (%) | 93 (85–101) | 92 (83–104) | 93 (85–100) | 0.868 |
| INR | 1.05 (0.98–1.11) | 1.05 (0.98–1.10) | 1.04 (0.98–1.11) | 0.898 |
| Creatinine (mg/dL) | 0.85 (0.70–1.04) | 0.84 (0.70–1.01) | 0.87 (0.72–1.09) | 0.526 |
| Hemoglobin (g/dL) | 13.2 (11.7–14.4) | 12.8 (11.7–14.1) | 13.5 (11.9–14.8) | 0.131 |
| Child-Pugh, n (%) | 0.088 | |||
| A | 139 (90.8) | 66 (86.8) | 73 (94.8) | |
| B | 14 (9.2) | 10 (13.2) | 4 (5.2) | |
| Preoperative Imaging features | ||||
| Number of nodules | 1 (1–2) | 1 (1–2) | 1 (1–2) | 0.324 |
| Largest nodule diameter (mm) | 50 (32–80) | 49 (32–78) | 53 (34–84) | 0.340 |
| Tumor burden >50%, n (%) | 7 (4.6) | 4 (5.3) | 3 (3.9) | 0.686 |
| Overall macrovascular invasion, n (%) | 39 (25.5) | 21 (27.6) | 18 (23.4) | 0.546 |
| Portal vein invasion, n (%) | 24 (15.7) | 14 (18.4) | 10 (13.0) | 0.355 |
| Extrahepatic vascular invasion, n (%) | 8 (5.2) | 3 (3.9) | 5 (6.5) | 0.479 |
| Portal vein thrombosis, n (%) | 6 (3.9) | 3 (3.9) | 3 (3.9) | 0.987 |
| Ascites, n (%) | 6 (3.9) | 3 (3.9) | 3 (3.9) | 0.987 |
| BCLC, n (%) | 0.709 | |||
| 0 | 7 (4.6) | 4 (5.3) | 3 (3.9) | |
| A | 89 (58.2) | 41 (53.9) | 48 (62.3) | |
| B | 33 (21.6) | 17 (22.4) | 16 (20.8) | |
| C | 24 (15.7) | 14 (18.4) | 10 (13.0) | |
| D | 0 | 0 | 0 | |
| Operative Data | ||||
| Laparoscopic resection, n (%) | 58 (37.9) | 28 (36.8) | 30 (39.0) | 0.787 |
| Conversation rate, n (%) | 5 (8.6) | 2 (7.1) | 3 (10.0) | 0.698 |
| Operative time (minutes) | 204 (146–274) | 206 (140–274) | 199 (150–273) | 0.469 |
| Operative procedure, n (%) | 0.575 | |||
| Atypical | 59 (38.6) | 27 (35.5) | 32 (41.6) | |
| Segmentectomy | 21 (13.7) | 8 (10.5) | 13 (16.9) | |
| Bisegmentectomy | 15 (9.8) | 8 (10.5) | 7 (9.1) | |
| Hemihepatectomy | 34 (22.2) | 17 (22.4) | 17 (22.1) | |
| Extended liver resection | 17 (11.1) | 12 (15.8) | 5 (6.5) | |
| ALPPS/TSH/other | 7 (4.6) | 4 (5.2) | 3 (3.9) | |
| Additional procedures (RFA, etc.), n (%) | 7 (4.6) | 3 (3.9) | 4 (5.2) | 0.712 |
| Pringle maneuver, n (%) | 10 (6.6) | 4 (5.3) | 6 (7.9) | 0.513 |
| Duration of pringle maneuver (min) * | 18 (10–24) | 11 (6–33) | 20 (14–24) | 0.352 |
| Intraoperative blood transfusion, n (%) | 42 (28.0) | 21 (27.6) | 21 (27.6) | 0.919 |
| Intraoperative FFP, n (%) | 58 (38.7) | 25 (33.8) | 33 (43.4) | 0.226 |
| Intraoperative platelet transfusion, n (%) | 4 (2.7) | 1 (1.4) | 3 (3.9) | 0.324 |
| Pathological examination | ||||
| R0 resection, n (%) | 147 (96.1) | 75 (98.7) | 72 (93.5) | 0.099 |
| T category, n (%) | 0.532 | |||
| T1 | 67 (34.8) | 36 (47.4) | 31 (40.3) | |
| T2 | 57 (37.3) | 25 (32.9) | 32 (41.6) | |
| T3/T4 | 29 (19.0) | 15 (19.7) | 14 (18.2) | |
| Microvascular invasion, n (%) | 62 (44.0) | 32 (45.1) | 30 (42.9) | 0.791 |
| Tumor grading, n (%) | 0.253 | |||
| G1/G2 | 122 (80.3) | 63 (84.0) | 59 (76.6) | |
| G3/G4 | 30 (19.7) | 12 (16.0) | 18 (23.4) | |
| NF, n (%) | 76 (49.7) | 76 (100) | 0 | <0.001 |
| NFD | 0 (0–5) | 6 (2–10) | 0 (0–0) | <0.001 |
| Postoperative Data | ||||
| Intensive care stay, days | 1 (1–1) | 1 (1–1) | 1 (1–1) | 0.946 |
| Hospitalization, days | 8 (6–8) | 8 (5–15) | 8 (6–13) | 0.772 |
| Postoperative complications, n (%) | 0.520 | |||
| No complications | 81 (52.9) | 38 (50.0) | 43 (55.8) | |
| Clavien-Dindo I | 15 (9.8) | 11 (14.5) | 4 (5.2) | |
| Clavien-Dindo II | 24 (15.7) | 12 (15.8) | 12 (15.6) | |
| Clavien-Dindo IIIa | 19 (12.4) | 8 (10.5) | 11 (14.3) | |
| Clavien-Dindo IIIb | 7 (4.6) | 4 (5.3) | 3 (3.9) | |
| Clavien-Dindo IVa | 6 (3.9) | 3 (3.9) | 3 (3.9) | |
| Clavien-Dindo IVb | 1 (0.7) | 0 | 1 (1.3) | |
| Clavien-Dindo V | 0 | 0 | 0 | |
| PHLF 50-50 criteria *, n (%) | 0 | 0 | 0 | n.a. |
| PHLF ISGLS *, n (%) | 25 (16.3) | 12 (15.8) | 13 (16.9) | 0.855 |
| ISGLS Grade, n (%) | 0.755 | |||
| A | 20 (80.0) | 9 (75.0) | 11 (84.6) | |
| B | 4 (16.0) | 2 (16.7) | 2 (15.4) | |
| C | 1 (4.0) | 1 (8.3) | 0 | |
| Postoperative blood transfusion | 19 (12.7) | 8 (10.8) | 11 (14.5) | 0.500 |
| Postoperative FFP | 6 (4.0) | 4 (5.4) | 2 (2.6) | 0.386 |
| Postoperative platelet transfusion | 1 (0.7) | 1 (1.4) | 1 (1.3) | 0.309 |
| Follow-up Data | ||||
| Recurrence-free survival (months) | 23 (16–30) | 26 (12–40) | 18 (9–27) | 0.666 |
| Overall survival (months) | 54 (34–74) | 66 (30–102) | 42 (21–63) | 0.804 |
| Variables | Univariate Analysis | Multivariable Analysis | ||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Demographics | ||||
| Gender (male = 1) | 1.82 (1.04–3.16) | 0.035 | 0.123 | |
| Age (≤65 years = 1) | 1.03 (0.62–1.70) | 0.915 | ||
| BMI (≤25 kg/m2 = 1) | 0.95 (0.58–1.57) | 0.851 | ||
| Recurrence resection (no = 1) | 0.62 (0.20–1.99) | 0.424 | ||
| ASA (I/II = 1) | 2.09 (1.18–3.69) | 0.011 | 0.188 | |
| Liver disease | 0.116 | |||
| ALD | 1 | |||
| NAFLD | 0.52 (0.28–0.96) | |||
| Viral | 0.65 (0.33–1.21) | |||
| Cryptogenic/others | 0.41 (0.16–1.04) | |||
| Preoperative liver function | ||||
| MELD Score (≤6 = 1) | 1.91 (1.12–3.24) | 0.017 | 2.08 (1.07–4.05) | 0.032 |
| Albumin (≤40 g/L = 1) | 0.57 (0.34–0.94) | 0.027 | 0.726 | |
| AFP (≤10 µg/L = 1) | 2.56 (1.40–4.67) | 0.002 | excl. | |
| AST (≤40 U/L = 1) | 1.89 (1.12–3.18) | 0.016 | 0.551 | |
| ALT (≤40 U/L = 1) | 1.64 (0.94–2.84) | 0.079 | ||
| GGT (≤100 U/L = 1) | 2.66 (1.54–4.60) | <0.001 | 0.354 | |
| Bilirubin (≤1 mg/dL = 1) | 1.82 (0.96–3.43) | 0.066 | ||
| AP (≤100 U/L = 1) | 1.95 (1.16–3.28) | 0.011 | 0.462 | |
| Platelet count (≤200/nL = 1) | 1.00 (0.60–1.65) | 0.988 | ||
| INR (≤1 = 1) | 1.82 (1.03–3.20) | 0.039 | 0.130 | |
| Creatinine (≤1 = 1) | 1.19 (0.70–2.02) | 0.531 | ||
| Hemoglobin (≤12 g/dL = 1) | 0.80 (0.48–1.34) | 0.399 | ||
| Child Pugh (A = 1) | 2.96 (1.44–6.07) | 0.003 | 0.556 | |
| Preoperative Imaging features | ||||
| Number of nodules (1 = 1) | 3.20 (1.95–5.24) | <0.001 | 2.01 (1.20–4.05) | 0.010 |
| Largest nodule diameter (≤50 mm = 1) | 1.89 (1.14–3.13) | 0.013 | 0.405 | |
| Tumor burden (≤50% = 1) | 2.90 (1.25–6.76) | 0.014 | 0.484 | |
| Macrovascular invasion (no = 1) | 2.23 (1.33–3.71) | 0.002 | 0.999 | |
| Portal vein invasion (no = 1) | 2.88 (1.63–5.09) | <0.001 | 0.084 | |
| Extrahepatic vascular invasion (no = 1) | 2.40 (1.03–5.59) | 0.042 | 0.700 | |
| Portal vein thrombosis (no = 1) | 3.09 (1.23–7.72) | 0.016 | 0.117 | |
| Ascites (no = 1) | 3.24 (1.15–9.09) | 0.025 | 6.24 (1.30–29.98) | 0.022 |
| BCLC | <0.001 | 0.190 | ||
| 0/A | 1 | |||
| B | 3.17 (1.80–5.58) | |||
| C | 4.30 (2.29–8.07) | |||
| Operative Data | ||||
| Laparoscopic resection (no = 1) | 1.94 (1.05–3.58) | 0.034 | 0.664 | |
| Operative time (≤180 min = 1) | 1.44 (0.86–2.41) | 0.163 | ||
| Operative procedure (minor = 1) | 1.15 (0.70–1.87) | 0.588 | ||
| Additional procedures (no = 1) | 1.25 (0.39–4.02) | 0.705 | ||
| Pringle maneuver (yes = 1) | 0.56 (0.24–1.32) | 0.185 | ||
| Intraop blood transfusion (no = 1) | 1.50 (0.89–2.53) | 0.128 | ||
| Intraop FFP (no = 1) | 1.36 (0.83–2.23) | 0.219 | ||
| Pathological data | ||||
| R1 resection (no = 1) | 3.58 (1.54–8.33) | 0.003 | 5.52 (1.86–16.38) | 0.002 |
| pT category | <0.001 | 0.192 | ||
| T1 | 1 | |||
| T2 | 2.86 (1.51–5.43) | |||
| T3/T4 | 6.19 (3.15–12.18) | |||
| Tumor grading (G1/G2 = 1) | 1.41 (0.79–2.51) | 0.248 | ||
| MVI (no = 1) | 4.27 (2.39–7.63) | <0.001 | 4.27 (2.18–8.37) | <0.001 |
| NF (no = 1) | 1.06 (0.65–1.73) | 0.806 | ||
| Postoperative Data | ||||
| Intensive care stay (≤1 day = 1) | 1.19 (0.57–2.51) | 0.641 | ||
| Hospitalization (≤7 days = 1) | 2.44 (1.35–4.42) | 0.003 | 0.094 | |
| Postop complications (I/II = 1) | 1.22 (0.70–2.13) | 0.482 | ||
| PHLF ISGLS (no = 1) | 1.14 (0.62–2.09) | 0.682 | ||
| Postop blood transfusion (no = 1) | 1.33 (0.67–2.61) | 0.414 | ||
| Postop FFP (no = 1) | 0.51 (0.12–2.09) | 0.348 | ||
| Univariate Analysis | Multivariable Analysis | |||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Demographics | ||||
| Gender (male = 1) | 1.00 (0.63–1.58) | 0.985 | ||
| Age (≤65 years = 1) | 0.77 (0.49–1.20) | 0.249 | ||
| BMI (≤25 kg/m2 = 1) | 0.83 (0.54–1.30) | 0.830 | ||
| Recurrence resection (no = 1) | 1.07 (0.49–2.33) | 0.863 | ||
| ASA (I/II = 1) | 1.05 (0.67–1.66) | 0.836 | ||
| Liver disease | 0.316 | |||
| ALD | 1 | |||
| NAFLD | 0.63 (0.35–1.14) | |||
| Viral | 1.00 (0.55–1.79) | |||
| Cryptogenic/others | 0.77 (0.35–1.71) | |||
| Preoperative liver function | ||||
| MELD Score (≤6 = 1) | ||||
| Albumin (≤40 g/L = 1) | 1.42 (0.88–2.33) | 0.155 | ||
| AFP (≤10 µg/L = 1) | 0.91 (0.58–1.41) | 0.662 | ||
| AST (≤40 U/L = 1) | 2.15 (1.29–3.57) | 0.003 | Excl. | |
| ALT (≤40 U/L = 1) | 2.45 (1.53–3.93) | <0.001 | 2.35 (1.30–4.25) | 0.005 |
| GGT (≤100 U/L = 1) | 2.05 (1.25–3.36) | 0.005 | 0.743 | |
| Bilirubin (≤1 mg/dL = 1) | 1.84 (1.15–2.93) | 0.011 | 0.303 | |
| AP (≤100 U/L = 1) | 1.77 (0.97–3.23) | 0.062 | ||
| Platelet count (≤200/nL = 1) | 1.85 (1.17–2.92) | 0.009 | 0.215 | |
| INR (≤1 = 1) | 0.90 (0.57–1.41) | 0.631 | ||
| Creatinine (≤1 = 1) | 1.50 (0.92–2.45) | 0.108 | ||
| Hemoglobin (≤12 g/dL = 1) | 0.77 (0.46–1.26) | 0.297 | ||
| Child Pugh (A = 1) | 0.79 (0.50–1.26) | 0.330 | ||
| Preoperative Imaging features | 2.20 (1.00–4.84) | 0.050 | ||
| Number of nodules (1 = 1) | ||||
| Largest nodule diameter (≤ 50 mm = 1) | ||||
| Tumor burden (≤50% = 1) | 3.78 (2.38–6.00) | <0.001 | 0.663 | |
| Macrovascular invasion (no = 1) | 1.76 (1.13–2.74) | 0.013 | 0.519 | |
| Portal vein invasion (no = 1) | 2.39 (0.96–5.96) | 0.061 | ||
| Extrahepatic vascular invasion (no = 1) | 1.93 (1.19–3.13) | 0.007 | 0.669 | |
| Portal vein thrombosis (no = 1) | 2.42 (1.37–4.26) | 0.002 | 2.44 (1.09–5.45) | 0.030 |
| Ascites (no = 1) | 2.48 (0.99–6.20) | 0.051 | ||
| BCLC | 5.90 (2.06–16.91) | 0.001 | 0.689 | |
| 0/A | 1.34 (0.33–5.51) | 0.685 | ||
| B | <0.001 | 0.725 | ||
| C | 1 | |||
| Operative Data | 3.13 (1.89–5.19) | |||
| Laparoscopic resection (no = 1) | 3.42 (1.86–6.26) | |||
| Operative time (≤180 min = 1) | ||||
| Operative procedure (minor = 1) | ||||
| Additional procedures (no = 1) | 1.32 (0.81–2.16) | 0.263 | ||
| Pringle maneuver (yes = 1) | 1.25 (0.80–1.97) | 0.327 | ||
| Intraop blood transfusion (no = 1) | 1.29 (0.83–2.02) | 0.259 | ||
| Intraop FFP (no = 1) | 1.30 (0.47–3.55) | 0.616 | ||
| Pathological data | 0.55 (0.24–1.26) | 0.156 | ||
| R1 resection (no = 1) | 1.23 (0.75–2.00) | 0.414 | ||
| pT category | 1.07 (0.68–1.68) | 0.784 | ||
| T1 | ||||
| T2 | 4.91 (2.10–11.49) | <0.001 | 0.243 | |
| T3/T4 | <0.001 | <0.001 | ||
| Tumor grading (G1/G2 = 1) | 1 | 1 | ||
| MVI (no = 1) | 3.28 (1.91–5.64) | 6.04 (2.89–12.60) | ||
| NF (no = 1) | 5.98 (3.15–11.38) | 6.02 (2.35–15.43) | ||
| Postoperative Data | 1.18 (0.68–2.04) | 0.565 | ||
| Intensive care stay (≤1 day = 1) | 2.38 (1.55–3.96) | <0.001 | 0.897 | |
| Hospitalization (≤7 days = 1) | 0.80 (0.52–1.24) | 0.322 | ||
| Postop complications (I/II = 1) | ||||
| PHLF ISGLS (no = 1) | 1.22 (0.64–2.31) | 0.547 | ||
| Postop blood transfusion (no = 1) | 1.14 (0.72–1.79) | 0.572 | ||
| Postop FFP (no = 1) | 0.92 (0.52–1.62) | 0.774 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bednarsch, J.; Tan, X.; Czigany, Z.; Wiltberger, G.; Buelow, R.D.; Boor, P.; Lang, S.A.; Ulmer, T.F.; Neumann, U.P.; Heij, L.R. Limitations of Nerve Fiber Density as a Prognostic Marker in Predicting Oncological Outcomes in Hepatocellular Carcinoma. Cancers 2022, 14, 2237. https://doi.org/10.3390/cancers14092237
Bednarsch J, Tan X, Czigany Z, Wiltberger G, Buelow RD, Boor P, Lang SA, Ulmer TF, Neumann UP, Heij LR. Limitations of Nerve Fiber Density as a Prognostic Marker in Predicting Oncological Outcomes in Hepatocellular Carcinoma. Cancers. 2022; 14(9):2237. https://doi.org/10.3390/cancers14092237
Chicago/Turabian StyleBednarsch, Jan, Xiuxiang Tan, Zoltan Czigany, Georg Wiltberger, Roman David Buelow, Peter Boor, Sven Arke Lang, Tom Florian Ulmer, Ulf Peter Neumann, and Lara Rosaline Heij. 2022. "Limitations of Nerve Fiber Density as a Prognostic Marker in Predicting Oncological Outcomes in Hepatocellular Carcinoma" Cancers 14, no. 9: 2237. https://doi.org/10.3390/cancers14092237







