Compassionate Use Program of Ipilimumab and Nivolumab in Intermediate or Poor Risk Metastatic Renal Cell Carcinoma: A Large Multicenter Italian Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Inclusion Criteria
- -
- Histological confirmation of RCC;
- -
- Eastern Cooperative Oncology Group (ECOG) performance status 0 to 2;
- -
- Advanced (not amenable to curative surgery or radiation therapy)/metastatic RCC Intermediate or poor IMDC score;
- -
- No prior treatment for metastatic disease;
- -
- Prior treatment with checkpoint inhibitors as adjuvant therapy;
- -
- active or suspected autoimmune disease requiring the use of systemic immunosuppressive agents or steroids (>10 mg of prednisone or equivalents);
- -
- active (untreated) brain metastases requiring steroid therapy (>10 mg/day prednisone equivalents);
- -
- any other conditions requiring systemic treatment with corticosteroids (>10 mg daily of prednisone or equivalents) or other immunosuppressive medications within 14 days prior to the first dose of IPI-NIVO
- -
- any positive test result for hepatitis B or hepatitis C virus indicating presence of virus, e.g., Hepatitis B surface antigen (HBsAg) positive, or Hepatitis C antibody (anti-HCV) positive (except if HCV-RNA negative);
- -
- a known history of testing positive for human immunodeficiency virus (HIV) or known acquired immunodeficiency syndrome (AIDS);
- -
- major surgery (e.g., nephrectomy) less than 28 days prior to the first dose.
2.2. Treatment Regimen and Evaluation of Response
2.3. Statistical Analysis
3. Results
3.1. Patients’ Charactheristics
3.2. Early Interruption of Treatment
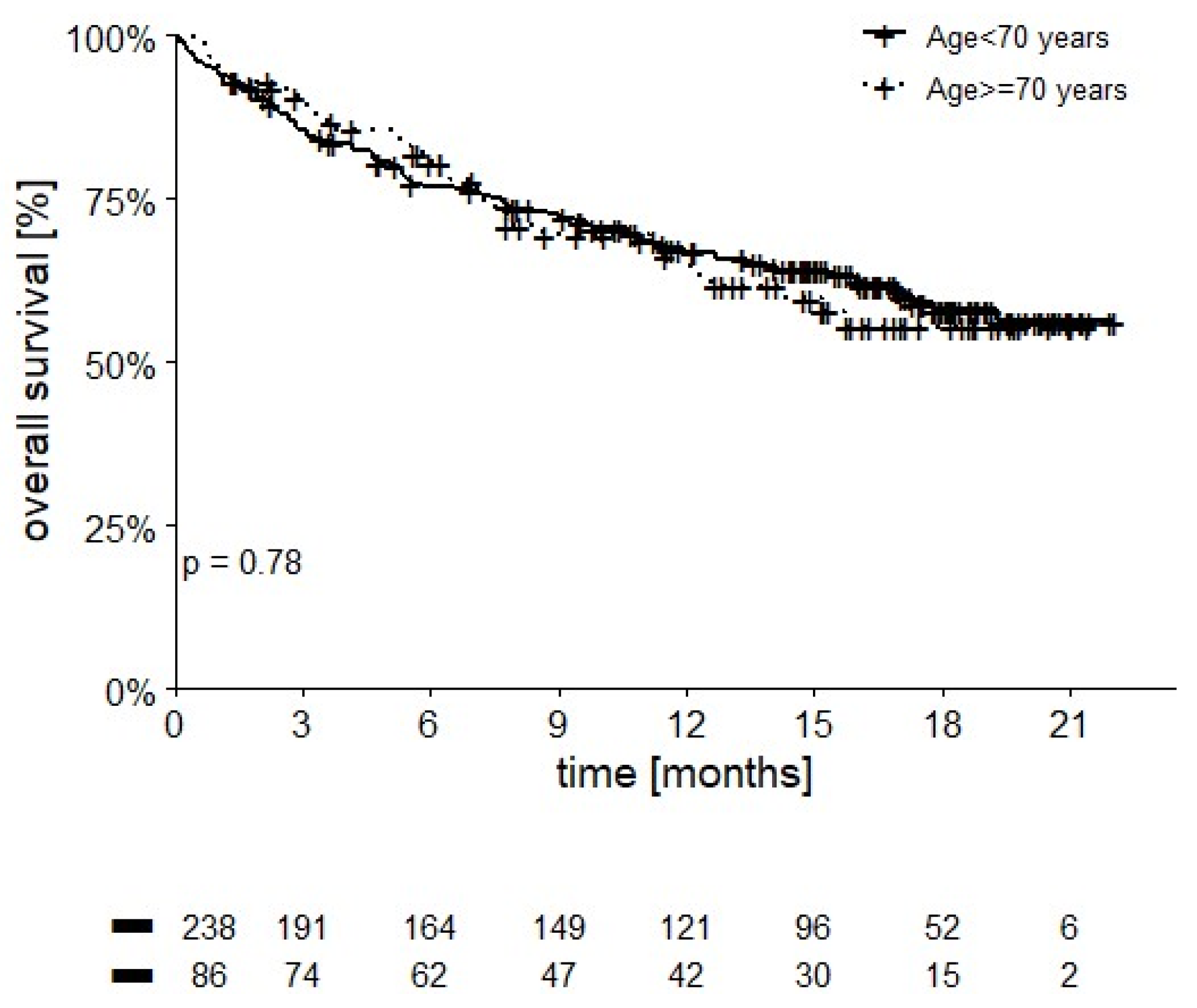
3.3. Overall Survival
3.4. Progression-Free Survival
3.5. Response Rate
3.6. Safety
3.7. Second-Line Therapy
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Capitanio, U.; Bensalah, K.; Bex, A.; Boorjian, S.A.; Bray, F.; Coleman, J.; Gore, J.L.; Sun, M.; Wood, C.; Russo, P. Epidemiology of Renal Cell Carcinoma. Eur. Urol. 2019, 75, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Escudier, B.; McDermott, D.F.; George, S.; Hammers, H.J.; Srinivas, S.; Tykodi, S.S.; Sosman, J.A.; Procopio, G.; Plimack, E.R.; et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N. Engl. J. Med. 2015, 373, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Cella, D.; Grünwald, V.; Nathan, P.; Doan, J.; Dastani, H.; Taylor, F.; Bennett, B.; DeRosa, M.; Berry, S.; Broglio, K.; et al. Quality of life in patients with advanced renal cell carcinoma given nivolumab versus everolimus in CheckMate 025: A randomised, open-label, phase 3 trial. Lancet Oncol. 2016, 17, 994–1003. [Google Scholar] [CrossRef] [Green Version]
- Hammers, H.J.; Plimack, E.R.; Infante, J.R.; Rini, B.I.; McDermott, D.F.; Lewis, L.D.; Voss, M.H.; Sharma, P.; Pal, S.K.; Razak, A.R.A.; et al. Safety and efficacy of nivolumab in combination with ipilimumab in metastatic renal cell carcinoma: The CheckMate 016 study. J. Clin. Oncol. 2017, 35, 3851–3858. [Google Scholar] [CrossRef] [Green Version]
- Motzer, R.J.; Tannir, N.M.; McDermott, D.F.; Arén Frontera, O.; Melichar, B.; Choueiri, T.K.; Plimack, E.R.; Barthélémy, P.; Porta, C.; George, S.; et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N. Engl. J. Med. 2018, 378, 1277–1290. [Google Scholar] [CrossRef]
- Heng, D.Y.; Xie, W.; Regan, M.M.; Warren, M.A.; Golshayan, A.R.; Sahi, C.; Eigl, B.J.; Ruether, J.D.; Cheng, T.; North, S.; et al. Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor-targeted agents: Results from a large, multicenter study. J. Clin. Oncol. 2009, 27, 5794–5799. [Google Scholar] [CrossRef]
- Cella, D.; Grünwald, V.; Escudier, B.; Hammers, H.J.; George, S.; Nathan, P.; Grimm, M.O.; Rini, B.I.; Doan, J.; Ivanescu, C.; et al. Patient-reported outcomes of patients with advanced renal cell carcinoma treated with nivolumab plus ipilimumab versus sunitinib (CheckMate 214): A randomised, phase 3 trial. Lancet Oncol. 2019, 20, 297–310. [Google Scholar] [CrossRef]
- Haanen, J.B.A.G.; Carbonnel, F.; Robert, C.; Kerr, K.M.; Peters, S.; Larkin, J.; Jordan, K. ESMO Guidelines Committee. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2018, 29 (Suppl. 4), iv264–iv266. [Google Scholar] [CrossRef]
- Schneider, B.J.; Naidoo, J.; Santomasso, B.D.; Lacchetti, C.; Adkins, S.; Anadkat, M.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J. Clin. Oncol. 2021, 39, 4073–4126. [Google Scholar] [CrossRef]
- Common Terminology Criteria for Adverse Events (CTCAE). Available online: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf (accessed on 12 March 2022).
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Wolchok, J.D.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.J.; Rutkowski, P.; Lao, C.D.; Cowey, C.L.; Schadendorf, D.; Wagstaff, J.; Dummer, R. Long-Term Outcomes With Nivolumab Plus Ipilimumab or Nivolumab Alone Versus Ipilimumab in Patients With Advanced Melanoma. J. Clin. Oncol. 2022, 40, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Ciuleanu, T.E.; Cobo, M.; Schenker, M.; Zurawski, B.; Menezes, J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O.; et al. First-line nivolumab plus ipilimumab with two cycles of chemotherapy versus chemotherapy alone (four cycles) in advanced non-small-cell lung cancer: CheckMate 9LA 2-year update. ESMO Open. 2021, 6, 100273. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.; Tannir, N.M.; David, F.; McDermott, M.B.; Toni, K.; Choueiri, H.J.; Hammers, E.R.; Plimack, C.P.; Powles, S.G.T.; Donskov, F.; et al. Conditional survival and 5-year follow-up in CheckMate 214: First-line nivolumab plus ipilimumab versus sunitinib in advanced renal cell carcinoma. Ann. Oncol. 2021, 32, S678–S724. [Google Scholar] [CrossRef]
- Tykodi, S.S.; Gordan, L.N.; Alter, R.S.; Arrowsmith, E.; Harrison, M.R.; Percent, I.; Singal, R.; Van Veldhuizen, P.; George, D.J.; Hutson, T.; et al. Safety and efficacy of nivolumab plus ipilimumab in patients with advanced non-clear cell renal cell carcinoma: Results from the phase 3b/4 CheckMate 920 trial. J. Immunother. Cancer 2022, 10, e003844. [Google Scholar] [CrossRef]
- Thana, M.; Basappa, N.S.; Ghosh, S.; Kollmannsberger, C.K.; Heng, D.Y.C.; Hansen, A.R.; Graham, J.; Soulières, D.; Reaume, M.N.; Lalani, A.A.; et al. Utilization and Safety of Ipilimumab Plus Nivolumab in a Real-World Cohort of Metastatic Renal Cell Carcinoma Patients. Clin. Genitourin. Cancer 2021, 10. [Google Scholar] [CrossRef]
- Alhalabi, O.; Hasanov, E.; Wilson, N.R.; Araujo, J.; Wang, J.; Campbell, M.T.; Goswami, S.; Shah, A.Y.; Gao, J.; Msaouel, P.; et al. Outcomes of patients with intermediate-risk or poor-risk metastatic renal cell carcinoma who received their first cycle of nivolumab and ipilimumab in the hospital because of symptomatic disease: The MD Anderson Cancer Center experience. Int. J. Cancer 2021, 149, 387–393. [Google Scholar] [CrossRef]
- Iacovelli, R.; De Giorgi, U.; Galli, L.; Zucali, P.; Nolè, F.; Sabbatini, R.; Fraccon, A.P.; Basso, U.; Mosca, A.; Atzori, F.; et al. Is It Possible to Improve Prognostic Classification in Patients Affected by Metastatic Renal Cell Carcinoma With an Intermediate or Poor Prognosis? Clin. Genitourin. Cancer 2018, 16, 355–359. [Google Scholar] [CrossRef]
- Takagi, T.; Fukuda, H.; Kondo, T.; Ishihara, H.; Yoshida, K.; Kobayashi, H.; Iizuka, J.; Okumi, M.; Ishida, H.; Tanabe, K. Prognostic Markers for Refined Stratification of IMDC Intermediate-Risk Metastatic Clear Cell Renal Cell Carcinoma Treated with First-Line Tyrosine Kinase Inhibitor Therapy. Target Oncol. 2019, 14, 179–186. [Google Scholar] [CrossRef]
- Lalani, A.A.; Bossé, D.; McGregor, B.A.; Choueiri, T.K. Immunotherapy in the Elderly. Eur. Urol. Focus. 2017, 3, 403–412. [Google Scholar] [CrossRef]
- Presley, C.J.; Gomes, F.; Burd, C.E.; Kanesvaran, R.; Wong, M.L. Immunotherapy in Older Adults with Cancer. J. Clin. Oncol. 2021, 39, 2115–2127. [Google Scholar] [CrossRef]
- Méjean, A.; Ravaud, A.; Thezenas, S.; Colas, S.; Beauval, J.B.; Bensalah, K.; Geoffrois, L.; Thiery-Vuillemin, A.; Cormier, L.; Lang, H.; et al. Sunitinib Alone or after Nephrectomy in Metastatic Renal-Cell Carcinoma. N. Engl. J. Med. 2018, 379, 417–427. [Google Scholar] [CrossRef]
- Hall, M.E.; Bhindi, B.; Luckenbaugh, A.N.; Laviana, A.A.; Moses, K.A.; Satkunasivam, R.; Rini, B.; Klaassen, Z.; Wallis, C.J.D. Association between cytoreductive nephrectomy and survival among patients with metastatic renal cell carcinoma receiving modern therapies: A systematic review and meta-analysis examining effect modification according to systemic therapy approach. Cancer Causes Control 2021, 32, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, T.; Mione, C.; Montoriol, P.F.; Molnar, I.; Ginzac, A.; Durando, X.; Mahammedi, H. Body Mass Index, Sarcopenia, and Their Variations in Predicting Outcomes for Patients Treated with Nivolumab for Metastatic Renal Cell Carcinoma. Oncology 2022, 100, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Woodall, M.J.; Neumann, S.; Campbell, K.; Pattison, S.T.; Young, S.L. The Effects of Obesity on Anti-Cancer Immunity and Cancer Immunotherapy. Cancers 2020, 12, 1230. [Google Scholar] [CrossRef] [PubMed]
- Fontes-Sousa, M.; Calvo, E. First-line immune checkpoint inhibitors in advanced or metastatic renal cell carcinoma with sarcomatoid features. Cancer Treat. Rev. 2022, 105, 102374. [Google Scholar] [CrossRef]
- Motzer, R.J.; Escudier, B.; McDermott, D.F.; Arén Frontera, O.; Melichar, B.; Powles, T.; Donskov, F.; Plimack, E.R.; Barthélémy, P.; Hammers, H.J.; et al. Survival outcomes and independent response assessment with nivolumab plus ipilimumab versus sunitinib in patients with advanced renal cell carcinoma: 42-month follow-up of a randomized phase 3 clinical trial. J. Immunother. Cancer 2020, 8, e000891. [Google Scholar] [CrossRef]
- Tarhini, A.A.; Kang, N.; Lee, S.J.; Hodi, F.S.; Cohen, G.I.; Hamid, O.; Hutchins, L.F.; Sosman, J.A.; Kluger, H.M.; Eroglu, Z.; et al. Immune adverse events (irAEs) with adjuvant ipilimumab in melanoma, use of immunosuppressants and association with outcome: ECOG-ACRIN E1609 study analysis. J. Immunother. Cancer 2021, 9, e002535. [Google Scholar] [CrossRef]
- Franzke, A.; Peest, D.; Probst-Kepper, M.; Buer, J.; Kirchner, G.I.; Brabant, G.; Kirchner, H.; Ganser, A.; Atzpodien, J. Autoimmunity resulting from cytokine treatment predicts long-term survival in patients with metastatic renal cell cancer. J. Clin. Oncol. 1999, 17, 529–533. [Google Scholar] [CrossRef]
- Schadendorf, D.; Wolchok, J.D.; Hodi, F.S.; Chiarion-Sileni, V.; Gonzalez, R.; Rutkowski, P.; Grob, J.J.; Cowey, C.L.; Lao, C.D.; Chesney, J.; et al. Efficacy and Safety Outcomes in Patients With Advanced Melanoma Who Discontinued Treatment With Nivolumab and Ipilimumab Because of Adverse Events: A Pooled Analysis of Randomized Phase II and III Trials. J. Clin. Oncol. 2017, 35, 3807–3814. [Google Scholar] [CrossRef] [Green Version]
- Verzoni, E.; Cartenì, G.; Cortesi, E.; Giannarelli, D.; De Giglio, A.; Sabbatini, R.; Buti, S.; Rossetti, S.; Cognetti, F.; Rastelli, F.; et al. Italian Nivolumab Renal Cell Cancer Early Access Program group. Real-world efficacy and safety of nivolumab in previously-treated metastatic renal cell carcinoma, and association between immune-related adverse events and survival: The Italian expanded access program. J. Immunother. Cancer 2019, 7, 99. [Google Scholar]
- Kato, T.; Nagahara, A.; Kawamura, N.; Nakata, W.; Soda, T.; Matsuzaki, K.; Hatano, K.; Kawashima, A.; Ujike, T.; Imamura, R.; et al. The prognostic impact of immune-related adverse events in metastatic renal cell carcinoma patients treated with nivolumab: A real-world multi-institutional retrospective study. Int. J. Clin. Oncol. 2021, 26, 954–961. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Plimack, E.R.; Soulières, D.; Waddell, T.; Stus, V.; Gafanov, R.; Nosov, D.; Pouliot, F.; Melichar, B.; Vynnychenko, I.; et al. Pembrolizumab plus axitinib versus sunitinib monotherapy as first-line treatment of advanced renal cell carcinoma (KEYNOTE-426): Extended follow-up from a randomised, open-label, phase 3 trial. Lancet Oncol. 2020, 21, 1563–1573. [Google Scholar] [CrossRef]
- Choueiri, T.K.; Motzer, R.J.; Rini, B.I.; Haanen, J.; Campbell, M.T.; Venugopal, B.; Kollmannsberger, C.; Gravis-Mescam, G.; Uemura, M.; Lee, J.L.; et al. Updated efficacy results from the JAVELIN Renal 101 trial: First-line avelumab plus axitinib versus sunitinib in patients with advanced renal cell carcinoma. Ann. Oncol. 2020, 31, 1030–1039. [Google Scholar] [CrossRef] [PubMed]
- Choueiri, T.K.; Powles, T.; Burotto, M.; Escudier, B.; Bourlon, M.T.; Zurawski, B.; Oyervides Juárez, V.M.; Hsieh, J.J.; Basso, U.; Shah, A.Y.; et al. CheckMate 9ER Investigators. Nivolumab plus Cabozantinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2021, 384, 829–841. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.; Alekseev, B.; Rha, S.Y.; Porta, C.; Eto, M.; Powles, T.; Grünwald, V.; Hutson, T.E.; Kopyltsov, E.; Méndez-Vidal, M.J.; et al. Lenvatinib plus Pembrolizumab or Everolimus for Advanced Renal Cell Carcinoma. N. Engl. J. Med. 2021, 384, 1289–1300. [Google Scholar] [CrossRef]
- Cattrini, C.; Messina, C.; Airoldi, C.; Buti, S.; Roviello, G.; Mennitto, A.; Caffo, O.; Gennari, A.; Bersanelli, M. Is there a preferred first-line therapy for metastatic renal cell carcinoma? A network meta-analysis. Ther. Adv. Urol. 2021, 13, 17562872211053189. [Google Scholar] [CrossRef]
- Navani, V.; Heng, D.Y.C. Treatment Selection in First-line Metastatic Renal Cell Carcinoma-The Contemporary Treatment Paradigm in the Age of Combination Therapy: A Review. JAMA Oncol. 2021, 8, 292–299. [Google Scholar] [CrossRef]
- Choueiri, T.K.; Tomczak, P.; Park, S.H.; Venugopal, B.; Ferguson, T.; Chang, Y.H.; Hajek, J.; Symeonides, S.N.; Lee, J.L.; Sarwar, N.; et al. Adjuvant Pembrolizumab after Nephrectomy in Renal-Cell Carcinoma. N. Engl. J. Med. 2021, 385, 683–694. [Google Scholar] [CrossRef]







| n = 324 | |||
|---|---|---|---|
| Age, median (range) 70–79 years; ≥80 years | 62 years (24–87) 72 pts (22.5%)/14 pts (4.3%) | ||
| Gender | 240 Males (74.1%)/84 Females (25.9%) | ||
| n | % | ||
| Symptoms | yes | 128 | 39.5 |
| no | 196 | 60.5 | |
| ECOG PS | 0 | 177 | 55 |
| 1 | 115 | 36 | |
| 2 | 32 | 10 | |
| BMI | ≥25 kg/m2 | 143 | 44.1 |
| <25 kg/m2 | 181 | 55.9 | |
| Autoimmune disease | yes | 10 | 3.1 |
| no | 314 | 96.9 | |
| Nephrectomy | before treatment | 211 | 65.1 |
| during treatment | 8 | 2.5 | |
| None | 105 | 32.4 | |
| Histology | clear cell | 269 | 83.0 |
| papillary | 21 | 6.5 | |
| chromophobe | 8 | 2.5 | |
| unclassified | 26 | 8.0 | |
| WHO grade | G1-2 | 48 | 14.8 |
| G3-4 | 130 | 40.1 | |
| NA | 146 | 45.1 | |
| Metastases at diagnosis | yes | 200 | 61.7 |
| no | 95 | 29.3 | |
| undefined | 29 | 9 | |
| Sites of metastases | lung | 231 | 71.3 |
| bone | 100 | 30.9 | |
| liver | 59 | 18.2 | |
| brain | 26 | 8.0 | |
| pancreas | 15 | 4.6 | |
| other | 80 | 24.7 | |
| IMDC risk score | Intermediate | 221 | 68.2 |
| 1 risk factor | 119 | 36.7 | |
| 2 risk factors | 102 | 31.5 | |
| poor | 103 | 31.8 | |
| n = 324 | ||
|---|---|---|
| Number of cycles of IPI-NIVO received | n | % |
| 1 | 37 | 11.4 |
| 2 | 33 | 10.2 |
| 3 | 34 | 10.5 |
| 4 (as planned) | 220 | 67.9 |
| Reason for early discontinuation | n | % |
| Progression | 49 | 15.1 |
| Adverse events | 55 | 17.0 |
| First Evaluation after IPI-NIVO | Additional Responses during Nivolumab Monotherapy | Overall Response Rate | |
|---|---|---|---|
| Response | Patients (%) | Patients (%) | Patients (%) |
| Complete response | 5 (1.9) | 3 (1.1) | 8 (2.8) |
| Partial response | 76 (28.1) | 22 (7.8) | 98 (34.8) |
| Stable | 96 (35.6) | - | 80 (28.4) |
| Progression | 93 (34.4) | - | 96 (34.0) |
| Total evaluable | 270 (100) | - | 282 (100) |
| AE | n = 55 | % |
|---|---|---|
| Gastrointestinal | 18 * | 32.7 |
| Hepatic | 8 † | 14.5 |
| Pulmonary | 5 | 9.1 |
| Pancreatic | 5 | 9.1 |
| Neurological | 4 | 7.3 |
| Asthenia | 2 | 3.6 |
| Cardiac | 1 | 1.8 |
| Endocrine | 1 | 1.8 |
| Allergic event | 1 | 1.8 |
| Skin toxicity | 1 | 1.8 |
| Renal | 1 # | 1.8 |
| Anemia | 1 | 1.8 |
| Others | 12 | 21.8 |
| irAE | Grade 1–2 (%) | Grade 3–4 (%) | Grade 5 (%) |
|---|---|---|---|
| Gastrointestinal | 39 (12.0) | 21 (6.5) | 2(0.6) |
| Hepatic | 12 (3.7) | 16 (4.9) | 1 (0.3) |
| Pulmonary | 4 (1.2) | 8 (2.5) | 0 |
| Pancreatic | 4 (1.2) | 9 (2.8%) | 0 |
| Neurological | 13 (4.0) | 6 (1.9) | 0 |
| Asthenia | 15 (4.6) | 2 (0.6) | 0 |
| Cardiac | - | 1 (0.3) | 0 |
| Thyroid | 51 (15.7) | 4 (1.2) | 0 |
| Hypophysitis | 5 (1.5) | 5 (1.5) | 0 |
| Skin | 54 (16.7) | 5 (1.5) | 0 |
| Anemia | 3 (0.9) | 2 (0.6) | 0 |
| Others | 3 (0.9%) | 8 (2.5%) | 1 (0.3%) |
| Total | 203 (62.7) | 67 (26.9%) | 4 (1.2%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basso, U.; Paolieri, F.; Rizzo, M.; De Giorgi, U.; Bracarda, S.; Antonuzzo, L.; Atzori, F.; Cartenì, G.; Procopio, G.; Fratino, L.; et al. Compassionate Use Program of Ipilimumab and Nivolumab in Intermediate or Poor Risk Metastatic Renal Cell Carcinoma: A Large Multicenter Italian Study. Cancers 2022, 14, 2293. https://doi.org/10.3390/cancers14092293
Basso U, Paolieri F, Rizzo M, De Giorgi U, Bracarda S, Antonuzzo L, Atzori F, Cartenì G, Procopio G, Fratino L, et al. Compassionate Use Program of Ipilimumab and Nivolumab in Intermediate or Poor Risk Metastatic Renal Cell Carcinoma: A Large Multicenter Italian Study. Cancers. 2022; 14(9):2293. https://doi.org/10.3390/cancers14092293
Chicago/Turabian StyleBasso, Umberto, Federico Paolieri, Mimma Rizzo, Ugo De Giorgi, Sergio Bracarda, Lorenzo Antonuzzo, Francesco Atzori, Giacomo Cartenì, Giuseppe Procopio, Lucia Fratino, and et al. 2022. "Compassionate Use Program of Ipilimumab and Nivolumab in Intermediate or Poor Risk Metastatic Renal Cell Carcinoma: A Large Multicenter Italian Study" Cancers 14, no. 9: 2293. https://doi.org/10.3390/cancers14092293






