Transplant Oncology: An Emerging Discipline of Cancer Treatment
Abstract
:Simple Summary
Abstract
1. Introduction
2. Concept and History of Transplant Oncology
2.1. Liver Transplantation for Hepatocellular Carcinoma
2.1.1. Milan Criteria
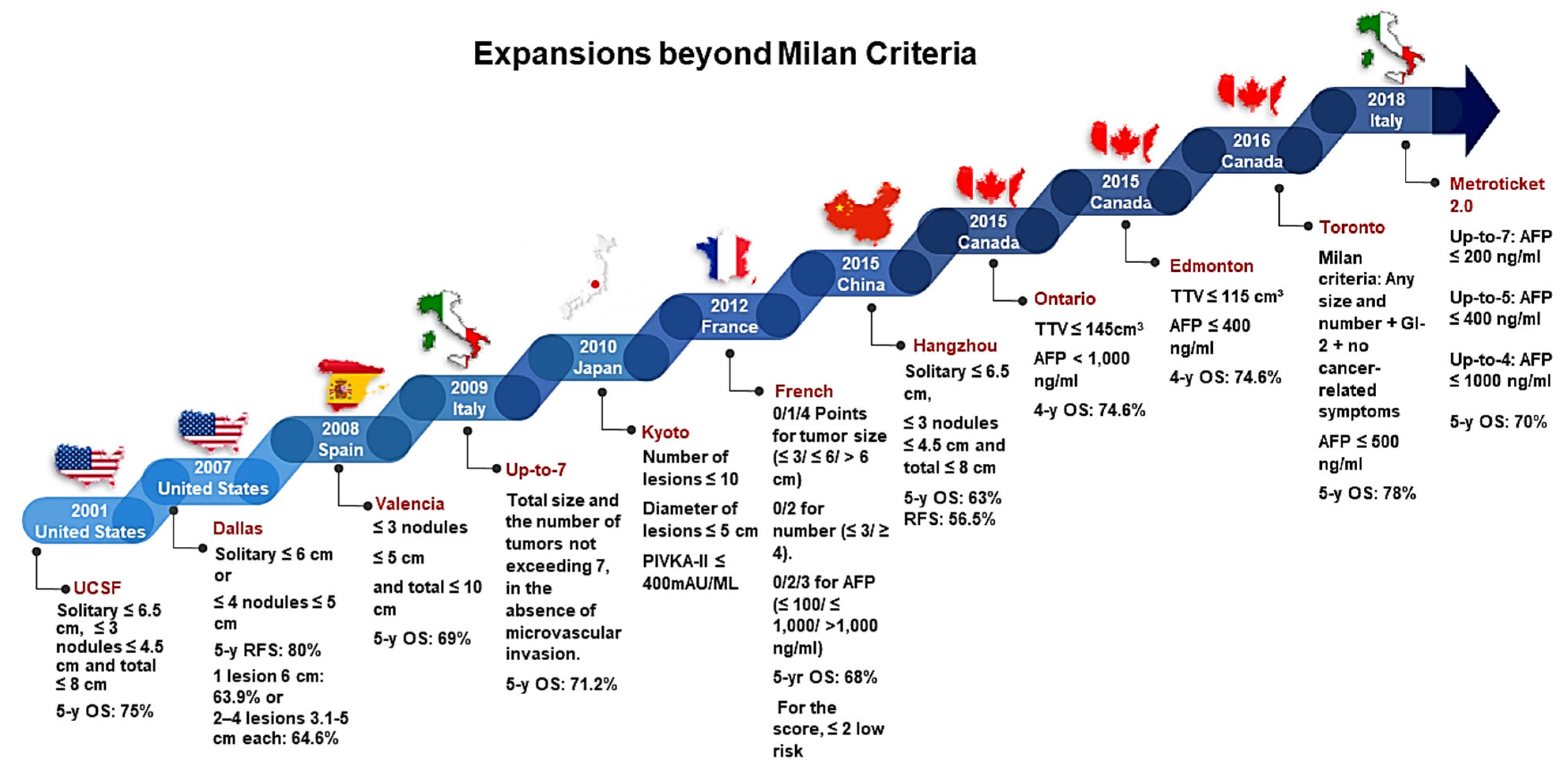
2.1.2. Beyond Milan Criteria
- A.
- University of California San Francisco Criteria
- B.
- Beyond USCF Criteria
2.1.3. Portal Vein Tumor Thrombus
2.1.4. Salvage Liver Transplantation
2.2. Liver Transplant for Non-Hepatocellular Carcinoma Tumors
2.2.1. Cholangiocarcinoma
- A.
- Hilar Cholangiocarcinoma
- B.
- Intrahepatic Cholangiocarcinoma
2.2.2. Hepatoblastoma
3. Liver Metastases
3.1. Neuroendocrine Tumor Liver Metastases (NETLM)
3.2. Colorectal Cancer
4. Emerging Concepts in Transplant Oncology
4.1. Immune Therapy in the Peri-Transplant Period
4.1.1. Pretransplant Bridging Therapy
| Age/Sex | ICPI Agent | ICPI Cycle | ICPI Class | Interval Time from Last Dose of ICPIs to Transplant | IST | Type of Response | Graft Outcome | References |
|---|---|---|---|---|---|---|---|---|
| 66 M | Atezolizumab Bevacizumab | (6) (5) | PD-L1 VEGF | 60 days | Tacrolimus/MMF | R | No rejection | Abdelrahim et al. [134] |
| 64 M | Nivolumab | (23) | PD-1 | 16 days | MMF/Prednisone/tacrolimus | R | Resolved rejection | Aby et al. [135] |
| 39 M | Toripalimab Lenvatinib | 10 UK | PD-1 TK | 93 days | Tacrolimus/Methylprednisolone | D | Graft rejection | Chen, G.H. et al. [136] |
| 64 M | Nivolumab | (1) | PD-1 | 7 days | Tacrolimus/MMF | RC | No rejection | Chen, Z. et al. [137] |
| 47 F | Nivolumab | (1) | PD-1 | 122 days | Tacrolimus/MMF | RC | No rejection | Chen, Z. et al. [137] |
| 50 M | Nivolumab | (1) | PD-1 | 62 days | Tacrolimus/MMF | R | No rejection | Chen, Z. et al. [137] |
| 38 M | Nivolumab | (6) | PD-1 | 59 days | Tacrolimus/MMF | R | No rejection | Chen, Z. et al. [137] |
| 67 M | Nivolumab | (6) | PD-1 | 67 days | Tacrolimus/MMF | R | No rejection | Chen, Z. et al. [137] |
| 60 M | Nivolumab | (17) | PD-1 | 5 weeks | Tacrolimus/MMF/steroid | R | Graft rejection | Dehghan et al. [138] |
| 14 M | Pembrolizumab | (3) | PD-1 | 138 days | Sirolimus/tacrolimus | R | No rejection | Kang et al. [139] |
| 63 M | Nivolumab Ipilimumab | UK | PD-1 CTLA-4 | 9 weeks | Methylprednisolone/Thymoglobulin | R | No rejection | Lizaola et al. [140] |
| 65 M | Nivolumab | UK | PD-1 | 8 days | Tacrolimus/MMF/Prednisone | D | Graft rejection | Nordness et al. [141] |
| 68 M | Nivolumab | UK | PD-1 | 10 months | UK | R | No rejection | Peterson et al. [142] |
4.1.2. Post-Transplant Palliative Therapy
| Age/Sex | ICPI Agent | ICPI Cycle | ICPI Class | Interval Time from Transplant to ICPIs | IST | Type of Response | Graft Outcome | References |
|---|---|---|---|---|---|---|---|---|
| 70 M | Nivolumab | (4) | PD-1 | 33 months | Tacrolimus/high-dose steroids. | PD | No rejection | Al Jarroudi et al. [146] |
| 62 F | Nivolumab | (5) | PD-1 | 12 months | Tacrolimus | PD | No rejection | Al Jarroudi et al. [146] |
| 66 M | Nivolumab | (6) | PD-1 | 24 months | Tacrolimus | PD | No rejection | Al Jarroudi et al. [146] |
| 56 M | Nivolumab | (6) | PD-1 | 32 months | Tacrolimus | PD | No rejection | DeLeon et al. [147] |
| 55 M | Nivolumab | (5) | PD-1 | 94 months | Sirolimus/MMF | PD | No rejection | DeLeon et al. [147] |
| 34 F | Nivolumab | UK | PD-1 | 44 months | Tacrolimus | PD | No rejection | DeLeon et al. [147] |
| 63 M | Nivolumab | UK | PD-1 | 14 months | Tacrolimus | UK | No rejection | DeLeon et al. [147] |
| 68 M | Nivolumab | UK | PD-1 | 13 months | Sirolimus | UK | Graft rejection | DeLeon et al. [147] |
| 41 M | Nivolumab | (15) | PD-1 | 16 months | Tacrolimus | PD | No rejection | De Toni and Gerbes et al. [148] |
| 70 M | Pembrolizumab | PD-1 | 96 months | Low-dose (50%) Tacrolimus | PD | No rejection | Varkaris et al. [149] | |
| 53 F | Nivolumab | (1) | PD-1 | 36 months | Everolimus/MMF | D due to OF (2 weeks after start ICPI) | Graft rejection | Gassmann et al. [150] |
| 14 M | Nivolumab | (1) | PD-1 | 36 months | Tacrolimus | D due to OF (5 weeks after start ICPI) | Graft rejection | Friend et al. [151] |
| 20 M | Nivolumab | (2) | PD-1 | 48 months | Sirolimus | D due to OF (4 weeks after start ICPI) | Graft rejection | Friend et al. [151] |
| 61 M | Nivolumab | (2) | PD-1 | 24 months | UK | R | Graft rejection | Gomez et al. [152] |
| 57 M | Pembrolizumab | (13) | PD-1 | 36 months | Tacrolimus/MMF/ steroid | R | No rejection | Rohmann et al. [144] |
| 64 M | Nivolumab | Less than (1) | PD-1 | 24 months | Thymoglobulin | R | Graft rejection | Kumar et al. [153] |
| 54 F | Ipilimumab | (17) | CTLA-4 | 84 months | Tacrolimus/Everolimus | PR | No rejection | Pandey et al. [154] |
| 54 M | Camrelizumab | (13) | PD-1 | 48 months | Tacrolimus | PD | No rejection | Qui et al. [155] |
| 54 M | Nivolumab | (12) | PD-1 | 24 months | Tacrolimus | PD | No rejection | Zhuang et al. [156] |
| 46 M | Toripalimab | (6) | PD-1 | 12 months | Sirolimus | PD | No rejection | Shi Gm et al. [157] |
| 35 M | Atezolizumab | (12) | PD-L1 | 48 months | UK | PD | No rejection | Ben Khaled et al. [158] |
| 35 M | Pembrolizumab | (2) | PD-1 | 240 months | MMF/Steroid | R | No rejection | Schvartsman et al. [159] |
| 54 M | Nivolumab | (3) | PD-1 | 156 months | Tacrolimus/Everolimus/Prednisone | PD | No rejection | Biondani P et al. [160] |
| 62 F | Ipilimumab Pembrolizumab | (4) (25) | CTLA-4 PD-1 | 14 months | Sirolimus/MMF | PR | No rejection | Kuo JC et al. [161] |
| 52 M | Nivolumab | (4) | PD-1 | 32 months | Cyclosporine/MMF | PD | No rejection | Kondo et al. [162] |
| 72 M | Nivolumab | (2) | PD-1 | 84 months | MMF/Budesonide | UK | No rejection | Deylon J et al. [163] |
| 59 M | Toripalimab | (8) | PD-1 | 16 months | Sirolimus | PD | No rejection | Shi GM et al. [157] |
4.2. Utility of Circulating Tumor DNA (ctDNA) for Cancer Minimal Residual Disease (MRD) Evaluation and Surveillance
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Kim, D.H.; Choi, S.H.; Park, S.H.; Kim, K.W.; Byun, J.H.; Kim, S.Y.; Lee, S.S.; Choi, J.I. The Liver Imaging Reporting and Data System tumor-in-vein category: A systematic review and meta-analysis. Eur. Radiol. 2021, 31, 2497–2506. [Google Scholar] [PubMed]
- Sapisochin, G.; Hibi, T.; Ghobrial, M.; Man, K. The ILTS Consensus Conference on Transplant Oncology: Setting the Stage. Transplantation 2020, 104, 1119–1120. [Google Scholar] [PubMed]
- Abdelrahim, M.; Esmail, A.; Abudayyeh, A.; Murakami, N.; Saharia, A.; McMillan, R.; Victor, D.; Kodali, S.; Shetty, A.; Nolte Fong, J.V.; et al. Transplant Oncology: An Evolving Field in Cancer Care. Cancers 2021, 13, 4911. [Google Scholar] [PubMed]
- Bruix, J.; Sherman, M. Management of hepatocellular carcinoma: An update. Hepatology 2011, 53, 1020–1022. [Google Scholar] [PubMed]
- Bruix, J.; Reig, M.; Sherman, M. Evidence-Based Diagnosis, Staging, and Treatment of Patients with Hepatocellular Carcinoma. Gastroenterology 2016, 150, 835–853. [Google Scholar] [PubMed]
- de Villa, V.; Lo, C.M. Liver transplantation for hepatocellular carcinoma in Asia. Oncologist 2007, 12, 1321–1331. [Google Scholar]
- Kim, W.R.; Lake, J.R.; Smith, J.M.; Skeans, M.A.; Schladt, D.P.; Edwards, E.B.; Harper, A.M.; Wainright, J.L.; Snyder, J.J.; Israni, A.K.; et al. OPTN/SRTR 2013 Annual Data Report: Liver. Am. J. Transplant. 2015, 15 (Suppl. 2), 1–28. [Google Scholar]
- Adam, R.; Karam, V.; Delvart, V.; O’Grady, J.; Mirza, D.; Klempnauer, J.; Castaing, D.; Neuhaus, P.; Jamieson, N.; Salizzoni, M.; et al. Evolution of indications and results of liver transplantation in Europe. A report from the European Liver Transplant Registry (ELTR). J. Hepatol. 2012, 57, 675–688. [Google Scholar]
- ILTS 2019 Consensus Conference: Transplant Oncology—The Future of Multidisciplinary Management. In Proceedings of the Consensus Conference, Rotterdam, The Netherlands, 7 February 2019.
- Hibi, T.; Sapisochin, G. What is transplant oncology? Surgery 2019, 165, 281–285. [Google Scholar]
- Balogh, J.; Victor, D., 3rd; Asham, E.H.; Burroughs, S.G.; Boktour, M.; Saharia, A.; Li, X.; Ghobrial, R.M.; Monsour, H.P., Jr. Hepatocellular carcinoma: A review. J. Hepatocell. Carcinoma 2016, 3, 41–53. [Google Scholar]
- Zhang, K.; Chen, R.; Gong, X.; Gao, Y. Survival outcomes of liver transplantation versus liver resection among patients with hepatocellular carcinoma: A SEER-based longitudinal study. J. Formos. Med. Assoc. 2019, 118, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Koh, J.H.; Tan, D.J.H.; Ong, Y.; Lim, W.H.; Ng, C.H.; Tay, P.W.L.; Yong, J.N.; Muthiah, M.D.; Tan, E.X.; Pang, N.Q.; et al. Liver resection versus liver transplantation for hepatocellular carcinoma within Milan criteria: A meta-analysis of 18,421 patients. Hepatobiliary Surg. Nutr. 2022, 11, 78–93. [Google Scholar] [CrossRef] [PubMed]
- Kow, A.W.C. Transplantation versus liver resection in patients with hepatocellular carcinoma. Transl. Gastroenterol. Hepatol. 2019, 4, 33. [Google Scholar] [CrossRef] [PubMed]
- Akoad, M.E.; Pomfret, E.A. Surgical resection and liver transplantation for hepatocellular carcinoma. Clin. Liver Dis. 2015, 19, 381–399. [Google Scholar]
- Fan, S.T.; Mau Lo, C.; Poon, R.T.P.; Yeung, C.; Leung Liu, C.; Yuen, W.K.; Ming Lam, C.; Ng, K.K.C.; Ching Chan, S. Continuous Improvement of Survival Outcomes of Resection of Hepatocellular Carcinoma: A 20-Year Experience. Ann. Surg. 2011, 253, 745–758. [Google Scholar] [CrossRef]
- Fan, S.T. Hepatocellular carcinoma—Resection or transplant? Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 732–737. [Google Scholar] [CrossRef]
- Cho, S.M.; Esmail, A.; Raza, A.; Dacha, S.; Abdelrahim, M. Timeline of FDA-Approved Targeted Therapy for Cholangiocarcinoma. Cancers 2022, 14, 2641. [Google Scholar] [CrossRef]
- Zhang, Y.; Esmail, A.; Mazzaferro, V.; Abdelrahim, M. Newest Therapies for Cholangiocarcinoma: An Updated Overview of Approved Treatments with Transplant Oncology Vision. Cancers 2022, 14, 5074. [Google Scholar] [CrossRef]
- Daoud, A.; Soliman, K.; Teeter, L.; Ali, H.; Graviss, E.A.; Mogawer, S.; Sholkamy, A.; El-Shazli, M.; Gaber, A.O. Alpha-Fetoprotein as a Modifier of Anatomic Criteria for Transplantation of HCC Patients. Transplant. Proc. 2021, 53, 833–838. [Google Scholar] [CrossRef]
- Daoud, A.; Teeter, L.; Ghobrial, R.M.; Graviss, E.A.; Mogawer, S.; Sholkamy, A.; El-Shazli, M.; Gaber, A.O. Transplantation for Hepatocellular Carcinoma: Is There a Tumor Size Limit? Transplant. Proc. 2018, 50, 3577–3581. [Google Scholar] [CrossRef]
- Frenette, C.T.; Boktour, M.; Burroughs, S.G.; Kaseb, A.; Aloia, T.A.; Galati, J.; Gaber, A.O.; Monsour, H., Jr.; Ghobrial, R.M. Pre-transplant utilization of sorafenib is not associated with increased complications after liver transplantation. Transpl. Int. 2013, 26, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Lobe, T.E.; Vera, S.R.; Bowman, L.C.; Fontanesi, J.; Britt, L.G.; Gaber, A.O. Hepaticopancreaticogastroduodenectomy with transplantation for metastatic islet cell carcinoma in childhood. J. Pediatr. Surg. 1992, 27, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Boeker, M.; França, F.; Bronsert, P.; Schulz, S. TNM-O: Ontology support for staging of malignant tumours. J. Biomed. Semant. 2016, 7, 64. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K.; Ohtsuki, T.; Obata, H.; Tomimatsu, M.; Okazaki, N.; Hasegawa, H.; Nakajima, Y.; Ohnishi, K. Natural history of hepatocellular carcinoma and prognosis in relation to treatment. Study of 850 patients. Cancer 1985, 56, 918–928. [Google Scholar] [CrossRef] [PubMed]
- Ringe, B.; Pichlmayr, R.; Wittekind, C.; Tusch, G. Surgical treatment of hepatocellular carcinoma: Experience with liver resection and transplantation in 198 patients. World J. Surg. 1991, 15, 270–285. [Google Scholar] [CrossRef]
- Iwatsuki, S.; Dvorchik, I.; Marsh, J.W.; Madariaga, J.R.; Carr, B.; Fung, J.J.; Starzl, T.E. Liver transplantation for hepatocellular carcinoma: A proposal of a prognostic scoring system11No competing interests declared. J. Am. Coll. Surg. 2000, 191, 389–394. [Google Scholar] [CrossRef]
- Iwatsuki, S.; Starzl, T.E.; Sheahan, D.G.; Yokoyama, I.; Demetris, A.J.; Todo, S.; Tzakis, A.G.; Van Thiel, D.H.; Carr, B.; Selby, R.; et al. Hepatic resection versus transplantation for hepatocellular carcinoma. Ann. Surg. 1991, 214, 221–228; discussion 228–229. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Regalia, E.; Doci, R.; Andreola, S.; Pulvirenti, A.; Bozzetti, F.; Montalto, F.; Ammatuna, M.; Morabito, A.; Gennari, L. Liver Transplantation for the Treatment of Small Hepatocellular Carcinomas in Patients with Cirrhosis. N. Engl. J. Med. 1996, 334, 693–699. [Google Scholar] [CrossRef]
- Hibi, T.; Shinoda, M.; Itano, O.; Kitagawa, Y. Current status of the organ replacement approach for malignancies and an overture for organ bioengineering and regenerative medicine. Organogenesis 2014, 10, 241–249. [Google Scholar] [CrossRef]
- Yao, F.Y.; Xiao, L.; Bass, N.M.; Kerlan, R.; Ascher, N.L.; Roberts, J.P. Liver transplantation for hepatocellular carcinoma: Validation of the UCSF-expanded criteria based on preoperative imaging. Am. J. Transplant. 2007, 7, 2587–2596. [Google Scholar] [CrossRef]
- Soejima, Y.; Taketomi, A.; Yoshizumi, T.; Uchiyama, H.; Aishima, S.; Terashi, T.; Shimada, M.; Maehara, Y. Extended indication for living donor liver transplantation in patients with hepatocellular carcinoma. Transplantation 2007, 83, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Kaido, T.; Ogawa, K.; Mori, A.; Fujimoto, Y.; Ito, T.; Tomiyama, K.; Takada, Y.; Uemoto, S. Usefulness of the Kyoto criteria as expanded selection criteria for liver transplantation for hepatocellular carcinoma. Surgery 2013, 154, 1053–1060. [Google Scholar] [CrossRef]
- Ito, T.; Takada, Y.; Ueda, M.; Haga, H.; Maetani, Y.; Oike, F.; Ogawa, K.; Sakamoto, S.; Ogura, Y.; Egawa, H.; et al. Expansion of selection criteria for patients with hepatocellular carcinoma in living donor liver transplantation. Liver Transplant. 2007, 13, 1637–1644. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, V.; Llovet, J.M.; Miceli, R.; Bhoori, S.; Schiavo, M.; Mariani, L.; Camerini, T.; Roayaie, S.; Schwartz, M.E.; Grazi, G.L.; et al. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: A retrospective, exploratory analysis. Lancet Oncol. 2009, 10, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, Y.; Tamura, S.; Makuuchi, M. Living donor liver transplantation for hepatocellular carcinoma: Tokyo University series. Dig. Dis. 2007, 25, 310–312. [Google Scholar] [CrossRef]
- Lee, S.G.; Hwang, S.; Moon, D.B.; Ahn, C.S.; Kim, K.H.; Sung, K.B.; Ko, G.Y.; Park, K.M.; Ha, T.Y.; Song, G.W. Expanded indication criteria of living donor liver transplantation for hepatocellular carcinoma at one large-volume center. Liver Transplant. 2008, 14, 935–945. [Google Scholar] [CrossRef]
- Yao, F.Y.; Ferrell, L.; Bass, N.M.; Bacchetti, P.; Ascher, N.L.; Roberts, J.P. Liver transplantation for hepatocellular carcinoma: Comparison of the proposed UCSF criteria with the Milan criteria and the Pittsburgh modified TNM criteria. Liver Transplant. 2002, 8, 765–774. [Google Scholar] [CrossRef]
- Onaca, N.; Davis, G.L.; Goldstein, R.M.; Jennings, L.W.; Klintmalm, G.B. Expanded criteria for liver transplantation in patients with hepatocellular carcinoma: A report from the International Registry of Hepatic Tumors in Liver Transplantation. Liver Transplant. 2007, 13, 391–399. [Google Scholar] [CrossRef]
- Duvoux, C.; Roudot–Thoraval, F.; Decaens, T.; Pessione, F.; Badran, H.; Piardi, T.; Francoz, C.; Compagnon, P.; Vanlemmens, C.; Dumortier, J.; et al. Liver Transplantation for Hepatocellular Carcinoma: A Model Including α-Fetoprotein Improves the Performance of Milan Criteria. Gastroenterology 2012, 143, 986–994.e3. [Google Scholar] [CrossRef]
- Xu, X.; Lu, D.; Ling, Q.; Wei, X.; Wu, J.; Zhou, L.; Yan, S.; Wu, L.; Geng, L.; Ke, Q.; et al. Liver transplantation for hepatocellular carcinoma beyond the Milan criteria. Gut 2016, 65, 1035–1041. [Google Scholar] [CrossRef]
- Zheng, S.S.; Xu, X.; Wu, J.; Chen, J.; Wang, W.L.; Zhang, M.; Liang, T.B.; Wu, L.M. Liver transplantation for hepatocellular carcinoma: Hangzhou experiences. Transplantation 2008, 85, 1726–1732. [Google Scholar] [CrossRef] [PubMed]
- Toso, C.; Meeberg, G.; Hernandez-Alejandro, R.; Dufour, J.-F.; Marotta, P.; Majno, P.; Kneteman, N.M. Total tumor volume and alpha-fetoprotein for selection of transplant candidates with hepatocellular carcinoma: A prospective validation. Hepatology 2015, 62, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Gorgen, A.; Muaddi, H.; Zhang, W.; McGilvray, I.; Gallinger, S.; Sapisochin, G. The New Era of Transplant Oncology: Liver Transplantation for Nonresectable Colorectal Cancer Liver Metastases. Can. J. Gastroenterol. Hepatol. 2018, 2018, 9531925. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, V.; Sposito, C.; Zhou, J.; Pinna, A.D.; De Carlis, L.; Fan, J.; Cescon, M.; Di Sandro, S.; Yi-Feng, H.; Lauterio, A.; et al. Metroticket 2.0 Model for Analysis of Competing Risks of Death After Liver Transplantation for Hepatocellular Carcinoma. Gastroenterology 2018, 154, 128–139. [Google Scholar] [CrossRef]
- Sapisochin, G.; Goldaracena, N.; Laurence, J.M.; Dib, M.; Barbas, A.; Ghanekar, A.; Cleary, S.P.; Lilly, L.; Cattral, M.S.; Marquez, M.; et al. The extended Toronto criteria for liver transplantation in patients with hepatocellular carcinoma: A prospective validation study. Hepatology 2016, 64, 2077–2088. [Google Scholar] [CrossRef]
- Mehta, N.; Bhangui, P.; Yao, F.Y.; Mazzaferro, V.; Toso, C.; Akamatsu, N.; Durand, F.; Ijzermans, J.; Polak, W.; Zheng, S.; et al. Liver Transplantation for Hepatocellular Carcinoma. Working Group Report from the ILTS Transplant Oncology Consensus Conference. Transplantation 2020, 104, 1136–1142. [Google Scholar] [CrossRef]
- Silva, M.; Moya, A.; Berenguer, M.; Sanjuan, F.; López-Andujar, R.; Pareja, E.; Torres-Quevedo, R.; Aguilera, V.; Montalva, E.; De Juan, M.; et al. Expanded criteria for liver transplantation in patients with cirrhosis and hepatocellular carcinoma. Liver Transplant. 2008, 14, 1449–1460. [Google Scholar] [CrossRef]
- Qu, Z.; Ling, Q.; Gwiasda, J.; Xu, X.; Schrem, H.; Beneke, J.; Kaltenborn, A.; Krauth, C.; Mix, H.; Klempnauer, J.; et al. Hangzhou criteria are more accurate than Milan criteria in predicting long-term survival after liver transplantation for HCC in Germany. Langenbeck’s Arch. Surg. 2018, 403, 643–654. [Google Scholar] [CrossRef]
- Notarpaolo, A.; Layese, R.; Magistri, P.; Gambato, M.; Colledan, M.; Magini, G.; Miglioresi, L.; Vitale, A.; Vennarecci, G.; Ambrosio, C.D.; et al. Validation of the AFP model as a predictor of HCC recurrence in patients with viral hepatitis-related cirrhosis who had received a liver transplant for HCC. J. Hepatol. 2017, 66, 552–559. [Google Scholar] [CrossRef]
- Cerrito, L.; Annicchiarico, B.E.; Iezzi, R.; Gasbarrini, A.; Pompili, M.; Ponziani, F.R. Treatment of hepatocellular carcinoma in patients with portal vein tumor thrombosis: Beyond the known frontiers. World J. Gastroenterol. 2019, 25, 4360–4382. [Google Scholar] [CrossRef]
- Manzano-Robleda Mdel, C.; Barranco-Fragoso, B.; Uribe, M.; Méndez-Sánchez, N. Portal vein thrombosis: What is new? Ann. Hepatol. 2015, 14, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Novi, M.; Lauritano, E.C.; Piscaglia, A.C.; Barbaro, B.; Zocco, M.A.; Pompili, M.; Gasbarrini, A. Portal Vein Tumor Thrombosis Revascularization During Sorafenib Treatment for Hepatocellular Carcinoma. Off. J. Am. Coll. Gastroenterol. ACG 2009, 104, 1852–1854. [Google Scholar] [CrossRef] [PubMed]
- Ponziani, F.R.; Zocco, M.A.; Senzolo, M.; Pompili, M.; Gasbarrini, A.; Avolio, A.W. Portal vein thrombosis and liver transplantation: Implications for waiting list period, surgical approach, early and late follow-up. Transplant. Rev. 2014, 28, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, G.; Wang, Y.; Liang, L.; Peng, B.; Fan, W.; Yang, J.; Huang, Y.; Yao, W.; Li, J. Is Salvage Liver Resection Necessary for Initially Unresectable Hepatocellular Carcinoma Patients Downstaged by Transarterial Chemoembolization? Ten Years of Experience. Oncologist 2016, 21, 1442–1449. [Google Scholar] [CrossRef] [PubMed]
- Lendoire, J.; Raffin, G.; Cejas, N.; Duek, F.; Schelotto, P.B.; Trigo, P.; Quarin, C.; Garay, V.; Imventarza, O. Liver transplantation in adult patients with portal vein thrombosis: Risk factors, management and outcome. HPB 2007, 9, 352–356. [Google Scholar] [CrossRef] [PubMed]
- Piscaglia, F.; Gianstefani, A.; Ravaioli, M.; Golfieri, R.; Cappelli, A.; Giampalma, E.; Sagrini, E.; Imbriaco, G.; Pinna, A.D.; Bolondi, L. Criteria for diagnosing benign portal vein thrombosis in the assessment of patients with cirrhosis and hepatocellular carcinoma for liver transplantation. Liver Transplant. 2010, 16, 658–667. [Google Scholar] [CrossRef]
- Lim, C.; Shinkawa, H.; Hasegawa, K.; Bhangui, P.; Salloum, C.; Gomez Gavara, C.; Lahat, E.; Omichi, K.; Arita, J.; Sakamoto, Y.; et al. Salvage liver transplantation or repeat hepatectomy for recurrent hepatocellular carcinoma: An intent-to-treat analysis. Liver Transplant. 2017, 23, 1553–1563. [Google Scholar] [CrossRef]
- de Haas, R.J.; Lim, C.; Bhangui, P.; Salloum, C.; Compagnon, P.; Feray, C.; Calderaro, J.; Luciani, A.; Azoulay, D. Curative salvage liver transplantation in patients with cirrhosis and hepatocellular carcinoma: An intention-to-treat analysis. Hepatology 2018, 67, 204–215. [Google Scholar] [CrossRef]
- Zheng, S.; Xie, Q.; Cheng, J. Salvage liver transplant for hepatocellular carcinoma: Rescues and benefits. Transl. Gastroenterol. Hepatol. 2018, 3, 65. [Google Scholar] [CrossRef]
- Pagano, D.; Mamone, G.; Petridis, I.; Gruttadauria, S. Hepatocellular Carcinoma Recurrence: How to Manage. In Hepatocellular Carcinoma; Ettorre, G.M., Ed.; Springer International Publishing: Cham, Switzerland, 2023; pp. 191–197. [Google Scholar]
- Pavel, M.; O’Toole, D.; Costa, F.; Capdevila, J.; Gross, D.; Kianmanesh, R.; Krenning, E.; Knigge, U.; Salazar, R.; Pape, U.F.; et al. ENETS Consensus Guidelines Update for the Management of Distant Metastatic Disease of Intestinal, Pancreatic, Bronchial Neuroendocrine Neoplasms (NEN) and NEN of Unknown Primary Site. Neuroendocrinology 2016, 103, 172–185. [Google Scholar] [CrossRef]
- Li, H.Y.; Wei, Y.G.; Yan, L.N.; Li, B. Salvage liver transplantation in the treatment of hepatocellular carcinoma: A meta-analysis. World J. Gastroenterol. 2012, 18, 2415–2422. [Google Scholar] [CrossRef] [PubMed]
- Cucchetti, A.; Vitale, A.; Del Gaudio, M.; Ravaioli, M.; Ercolani, G.; Cescon, M.; Zanello, M.; Morelli, M.C.; Cillo, U.; Grazi, G.L.; et al. Harm and benefits of primary liver resection and salvage transplantation for hepatocellular carcinoma. Am. J. Transplant. 2010, 10, 619–627. [Google Scholar] [CrossRef] [PubMed]
- De Vreede, I.; Steers, J.L.; Burch, P.A.; Rosen, C.B.; Gunderson, L.L.; Haddock, M.G.; Burgart, L.; Gores, G.J. Prolonged disease-free survival after orthotopic liver transplantation plus adjuvant chemoirradiation for cholangiocarcinoma. Liver Transplant. 2000, 6, 309–316. [Google Scholar] [CrossRef]
- Hong, J.C.; Jones, C.M.; Duffy, J.P.; Petrowsky, H.; Farmer, D.G.; French, S.; Finn, R.; Durazo, F.A.; Saab, S.; Tong, M.J.; et al. Comparative Analysis of Resection and Liver Transplantation for Intrahepatic and Hilar Cholangiocarcinoma: A 24-Year Experience in a Single Center. Arch. Surg. 2011, 146, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Robles, R.; Figueras, J.; Turrión, V.S.; Margarit, C.; Moya, A.; Varo, E.; Calleja, J.; Valdivieso, A.; Valdecasas, J.C.; López, P.; et al. Spanish experience in liver transplantation for hilar and peripheral cholangiocarcinoma. Ann. Surg. 2004, 239, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Casavilla, F.A.; Marsh, J.W.; Iwatsuki, S.; Todo, S.; Lee, R.G.; Madariaga, J.R.; Pinna, A.; Dvorchik, I.; Fung, J.J.; Starzl, T.E. Hepatic resection and transplantation for peripheral cholangiocarcinoma. J. Am. Coll. Surg. 1997, 185, 429–436. [Google Scholar] [CrossRef]
- Meyer, C.G.; Penn, I.; James, L. Liver transplantation for cholangiocarcinoma: Results in 207 patients. Transplantation 2000, 69, 1633–1637. [Google Scholar] [CrossRef]
- Panayotova, G.; Lunsford, K.E.; Latt, N.L.; Paterno, F.; Guarrera, J.V.; Pyrsopoulos, N. Expanding indications for liver transplantation in the era of liver transplant oncology. World J. Gastrointest. Surg. 2021, 13, 392–405. [Google Scholar] [CrossRef]
- Sapisochin, G.; Facciuto, M.; Rubbia-Brandt, L.; Marti, J.; Mehta, N.; Yao, F.Y.; Vibert, E.; Cherqui, D.; Grant, D.R.; Hernandez-Alejandro, R.; et al. Liver transplantation for “very early” intrahepatic cholangiocarcinoma: International retrospective study supporting a prospective assessment. Hepatology 2016, 64, 1178–1188. [Google Scholar] [CrossRef]
- Pham, T.A.; Gallo, A.M.; Concepcion, W.; Esquivel, C.O.; Bonham, C.A. Effect of Liver Transplant on Long-term Disease-Free Survival in Children With Hepatoblastoma and Hepatocellular Cancer. JAMA Surg. 2015, 150, 1150–1158. [Google Scholar] [CrossRef]
- Le Treut, Y.P.; Grégoire, E.; Klempnauer, J.; Belghiti, J.; Jouve, E.; Lerut, J.; Castaing, D.; Soubrane, O.; Boillot, O.; Mantion, G.; et al. Liver transplantation for neuroendocrine tumors in Europe-results and trends in patient selection: A 213-case European liver transplant registry study. Ann. Surg. 2013, 257, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Le Treut, Y.P.; Grégoire, E.; Belghiti, J.; Boillot, O.; Soubrane, O.; Mantion, G.; Cherqui, D.; Castaing, D.; Ruszniewski, P.; Wolf, P.; et al. Predictors of long-term survival after liver transplantation for metastatic endocrine tumors: An 85-case French multicentric report. Am. J. Transplant. 2008, 8, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Gedaly, R.; Daily, M.F.; Davenport, D.; McHugh, P.P.; Koch, A.; Angulo, P.; Hundley, J.C. Liver transplantation for the treatment of liver metastases from neuroendocrine tumors: An analysis of the UNOS database. Arch. Surg. 2011, 146, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Rosenau, J.; Bahr, M.J.; von Wasielewski, R.; Mengel, M.; Schmidt, H.H.J.; Nashan, B.; Lang, H.; Klempnauer, J.; Manns, M.P.; Boeker, K.H.W. Ki67, E-cadherin, and p53 as prognostic indicators of long-term outcome after liver transplantation for metastatic neuroendocrine tumors. Transplantation 2002, 73, 386–394. [Google Scholar] [CrossRef]
- Hagness, M.; Foss, A.; Line, P.D.; Scholz, T.; Jørgensen, P.F.; Fosby, B.; Boberg, K.M.; Mathisen, O.; Gladhaug, I.P.; Egge, T.S.; et al. Liver transplantation for nonresectable liver metastases from colorectal cancer. Ann. Surg. 2013, 257, 800–806. [Google Scholar] [CrossRef]
- DeOliveira, M.L.; Cunningham, S.C.; Cameron, J.L.; Kamangar, F.; Winter, J.M.; Lillemoe, K.D.; Choti, M.A.; Yeo, C.J.; Schulick, R.D. Cholangiocarcinoma: Thirty-one-year experience with 564 patients at a single institution. Ann. Surg. 2007, 245, 755–762. [Google Scholar] [CrossRef]
- Jarnagin, W.R.; Fong, Y.; DeMatteo, R.P.; Gonen, M.; Burke, E.C.; Bodniewicz, B.J.; Youssef, B.M.; Klimstra, D.; Blumgart, L.H. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann. Surg. 2001, 234, 507–517; discussion 517–519. [Google Scholar] [CrossRef]
- Kobayashi, A.; Miwa, S.; Nakata, T.; Miyagawa, S. Disease recurrence patterns after R0 resection of hilar cholangiocarcinoma. Br. J. Surg. 2010, 97, 56–64. [Google Scholar] [CrossRef]
- Darwish Murad, S.; Kim, W.R.; Harnois, D.M.; Douglas, D.D.; Burton, J.; Kulik, L.M.; Botha, J.F.; Mezrich, J.D.; Chapman, W.C.; Schwartz, J.J.; et al. Efficacy of neoadjuvant chemoradiation, followed by liver transplantation, for perihilar cholangiocarcinoma at 12 US centers. Gastroenterology 2012, 143, 88–98.e3, quiz e14. [Google Scholar] [CrossRef]
- Petrowsky, H.; Hong, J.C. Current Surgical Management of Hilar and Intrahepatic Cholangiocarcinoma: The Role of Resection and Orthotopic Liver Transplantation. Transplant. Proc. 2009, 41, 4023–4035. [Google Scholar] [CrossRef]
- Kennedy, T.J.; Yopp, A.; Qin, Y.; Zhao, B.; Guo, P.; Liu, F.; Schwartz, L.H.; Allen, P.; D’Angelica, M.; Fong, Y.; et al. Role of preoperative biliary drainage of liver remnant prior to extended liver resection for hilar cholangiocarcinoma. HPB 2009, 11, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Maguchi, H.; Takahashi, K.; Katanuma, A.; Osanai, M.; Nakahara, K.; Matuzaki, S.; Urata, T.; Iwano, H. Preoperative biliary drainage for hilar cholangiocarcinoma. J. Hepato-Biliary-Pancreat. Surg. 2007, 14, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Abdelrahim, M.; Al-Rawi, H.; Esmail, A.; Xu, J.; Umoru, G.; Ibnshamsah, F.; Abudayyeh, A.; Victor, D.; Saharia, A.; McMillan, R.; et al. Gemcitabine and Cisplatin as Neo-Adjuvant for Cholangiocarcinoma Patients Prior to Liver Transplantation: Case-Series. Curr. Oncol. 2022, 29, 3585–3594. [Google Scholar] [CrossRef] [PubMed]
- Abdelrahim, M.; Esmail, A.; Xu, J.; Umoru, G.; Al-Rawi, H.; Saharia, A.; Abudayyeh, A.; Victor, D.; McMillan, R.; Kodali, S.; et al. Gemcitabine Plus Cisplatin Versus Non-Gemcitabine and Cisplatin Regimens as Neoadjuvant Treatment for Cholangiocarcinoma Patients Prior to Liver Transplantation: An Institution Experience. Front. Oncol. 2022, 12, 908687. [Google Scholar] [CrossRef] [PubMed]
- Sapisochin, G.; de Lope, C.R.; Gastaca, M.; de Urbina, J.O.; Suarez, M.A.; Santoyo, J.; Castroagudín, J.F.; Varo, E.; López-Andujar, R.; Palacios, F.; et al. “Very Early” Intrahepatic Cholangiocarcinoma in Cirrhotic Patients: Should Liver Transplantation Be Reconsidered in These Patients? Am. J. Transplant. 2014, 14, 660–667. [Google Scholar] [CrossRef]
- Hong, J.C.; Petrowsky, H.; Kaldas, F.M.; Farmer, D.G.; Durazo, F.A.; Finn, R.S.; Saab, S.; Han, S.-H.; Lee, P.; Markovic, D.; et al. Predictive Index for Tumor Recurrence after Liver Transplantation for Locally Advanced Intrahepatic and Hilar Cholangiocarcinoma. J. Am. Coll. Surg. 2011, 212, 514–520. [Google Scholar] [CrossRef]
- Wong, M.; Kim, J.; George, B.; Eriksen, C.; Pearson, T.; Robbins, J.; Zimmerman, M.A.; Hong, J.C. Downstaging Locally Advanced Cholangiocarcinoma Pre-Liver Transplantation: A Prospective Pilot Study. J. Surg. Res. 2019, 242, 23–30. [Google Scholar] [CrossRef]
- Buettner, S.; van Vugt, J.L.; JN, I.J.; Groot Koerkamp, B. Intrahepatic cholangiocarcinoma: Current perspectives. Oncol. Targets Ther. 2017, 10, 1131–1142. [Google Scholar] [CrossRef]
- Spolverato, G.; Vitale, A.; Cucchetti, A.; Popescu, I.; Marques, H.P.; Aldrighetti, L.; Gamblin, T.C.; Maithel, S.K.; Sandroussi, C.; Bauer, T.W.; et al. Can hepatic resection provide a long-term cure for patients with intrahepatic cholangiocarcinoma? Cancer 2015, 121, 3998–4006. [Google Scholar] [CrossRef]
- Spolverato, G.; Kim, Y.; Ejaz, A.; Alexandrescu, S.; Marques, H.; Aldrighetti, L.; Gamblin, T.C.; Pulitano, C.; Bauer, T.W.; Shen, F.; et al. Conditional Probability of Long-term Survival After Liver Resection for Intrahepatic Cholangiocarcinoma: A Multi-institutional Analysis of 535 Patients. JAMA Surg. 2015, 150, 538–545. [Google Scholar] [CrossRef]
- Spolverato, G.; Kim, Y.; Alexandrescu, S.; Popescu, I.; Marques, H.P.; Aldrighetti, L.; Clark Gamblin, T.; Miura, J.; Maithel, S.K.; Squires, M.H.; et al. Is Hepatic Resection for Large or Multifocal Intrahepatic Cholangiocarcinoma Justified? Results from a Multi-Institutional Collaboration. Ann. Surg. Oncol. 2015, 22, 2218–2225. [Google Scholar] [CrossRef] [PubMed]
- Buettner, S.; Koerkamp, B.G.; Ejaz, A.; Buisman, F.E.; Kim, Y.; Margonis, G.A.; Alexandrescu, S.; Marques, H.P.; Lamelas, J.; Aldrighetti, L.; et al. The effect of preoperative chemotherapy treatment in surgically treated intrahepatic cholangiocarcinoma patients-A multi-institutional analysis. J. Surg. Oncol. 2017, 115, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Horgan, A.M.; Amir, E.; Walter, T.; Knox, J.J. Adjuvant therapy in the treatment of biliary tract cancer: A systematic review and meta-analysis. J. Clin. Oncol. 2012, 30, 1934–1940. [Google Scholar] [CrossRef] [PubMed]
- Sapisochin, G.; Ivanics, T.; Subramanian, V.; Doyle, M.; Heimbach, J.K.; Hong, J.C. Multidisciplinary treatment for hilar and intrahepatic cholangiocarcinoma: A review of the general principles. Int. J. Surg. 2020, 82, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Lunsford, K.E.; Javle, M.; Heyne, K.; Shroff, R.T.; Abdel-Wahab, R.; Gupta, N.; Mobley, C.M.; Saharia, A.; Victor, D.W.; Nguyen, D.T.; et al. Liver transplantation for locally advanced intrahepatic cholangiocarcinoma treated with neoadjuvant therapy: A prospective case-series. Lancet Gastroenterol. Hepatol. 2018, 3, 337–348. [Google Scholar] [CrossRef]
- Meza-Junco, J.; Montano-Loza, A.J.; Ma, M.; Wong, W.; Sawyer, M.B.; Bain, V.G. Cholangiocarcinoma: Has there been any progress? Can. J. Gastroenterol. 2010, 24, 52–57. [Google Scholar] [CrossRef]
- Moris, D.; Kostakis, I.D.; Machairas, N.; Prodromidou, A.; Tsilimigras, D.I.; Ravindra, K.V.; Sudan, D.L.; Knechtle, S.J.; Barbas, A.S. Comparison between liver transplantation and resection for hilar cholangiocarcinoma: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0220527. [Google Scholar]
- Ortega, J.A.; Douglass, E.C.; Feusner, J.H.; Reynolds, M.; Quinn, J.J.; Finegold, M.J.; Haas, J.E.; King, D.R.; Liu-Mares, W.; Sensel, M.G.; et al. Randomized comparison of cisplatin/vincristine/fluorouracil and cisplatin/continuous infusion doxorubicin for treatment of pediatric hepatoblastoma: A report from the Children’s Cancer Group and the Pediatric Oncology Group. J. Clin. Oncol. 2000, 18, 2665–2675. [Google Scholar] [CrossRef]
- Perilongo, G.; Maibach, R.; Shafford, E.; Brugieres, L.; Brock, P.; Morland, B.; de Camargo, B.; Zsiros, J.; Roebuck, D.; Zimmermann, A.; et al. Cisplatin versus cisplatin plus doxorubicin for standard-risk hepatoblastoma. N. Engl. J. Med. 2009, 361, 1662–1670. [Google Scholar] [CrossRef]
- Trobaugh-Lotrario, A.D.; Meyers, R.L.; Tiao, G.M.; Feusner, J.H. Pediatric liver transplantation for hepatoblastoma. Transl. Gastroenterol. Hepatol. 2016, 1, 44. [Google Scholar] [CrossRef] [PubMed]
- Gangi, A.; Howe, J.R. The Landmark Series: Neuroendocrine Tumor Liver Metastases. Ann. Surg. Oncol. 2020, 27, 3270–3280. [Google Scholar] [PubMed]
- Foster, J.H.; Lundy, J. Liver metastases. Curr. Probl. Surg. 1981, 18, 157–202. [Google Scholar] [PubMed]
- Foster, J.H.; Berman, M.M. Solid liver tumors. Major Probl. Clin. Surg. 1977, 22, 1–342. [Google Scholar] [PubMed]
- McEntee, G.P.; Nagorney, D.M.; Kvols, L.K.; Moertel, C.G.; Grant, C.S. Cytoreductive hepatic surgery for neuroendocrine tumors. Surgery 1990, 108, 1091–1096. [Google Scholar]
- Que, F.G.; Nagorney, D.M.; Batts, K.P.; Linz, L.J.; Kvols, L.K. Hepatic resection for metastatic neuroendocrine carcinomas. Am. J. Surg. 1995, 169, 36–43. [Google Scholar]
- Moris, D.; Tsilimigras, D.I.; Ntanasis-Stathopoulos, I.; Beal, E.W.; Felekouras, E.; Vernadakis, S.; Fung, J.J.; Pawlik, T.M. Liver transplantation in patients with liver metastases from neuroendocrine tumors: A systematic review. Surgery 2017, 162, 525–536. [Google Scholar]
- Mazzaferro, V.; Pulvirenti, A.; Coppa, J. Neuroendocrine tumors metastatic to the liver: How to select patients for liver transplantation? J. Hepatol. 2007, 47, 460–466. [Google Scholar] [CrossRef]
- Hibi, T.; Rela, M.; Eason, J.D.; Line, P.D.; Fung, J.; Sakamoto, S.; Selzner, N.; Man, K.; Ghobrial, R.M.; Sapisochin, G. Liver Transplantation for Colorectal and Neuroendocrine Liver Metastases and Hepatoblastoma. Working Group Report From the ILTS Transplant Oncology Consensus Conference. Transplantation 2020, 104, 1131–1135. [Google Scholar] [CrossRef]
- Nagai, S. Transplant oncology: Multivisceral transplantation for neuroendocrine tumor and liver metastasis. Curr. Opin. Organ Transplant. 2023, 28, 222–227. [Google Scholar] [CrossRef]
- Alagusundaramoorthy, S.S.; Gedaly, R. Role of surgery and transplantation in the treatment of hepatic metastases from neuroendocrine tumor. World J. Gastroenterol. 2014, 20, 14348–14358. [Google Scholar] [CrossRef] [PubMed]
- Sher, L.S.; Levi, D.M.; Wecsler, J.S.; Lo, M.; Petrovic, L.M.; Groshen, S.; Ji, L.; Uso, T.D.; Tector, A.J.; Hamilton, A.S.; et al. Liver transplantation for metastatic neuroendocrine tumors: Outcomes and prognostic variables. J. Surg. Oncol. 2015, 112, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Valderrama-Treviño, A.I.; Barrera-Mera, B.; Ceballos-Villalva, J.C.; Montalvo-Javé, E.E. Hepatic Metastasis from Colorectal Cancer. Euroasian J. Hepato-Gastroenterol. 2017, 7, 166–175. [Google Scholar]
- Wong, M.C.S.; Huang, J.; Lok, V.; Wang, J.; Fung, F.; Ding, H.; Zheng, Z.-J. Differences in Incidence and Mortality Trends of Colorectal Cancer Worldwide Based on Sex, Age, and Anatomic Location. Clin. Gastroenterol. Hepatol. 2021, 19, 955–966.e61. [Google Scholar] [CrossRef]
- National Cancer Institute. SEER Cancer Stat Facts: Colorectal Cancer; National Cancer Institute: Bethesda, MD, USA, 2020. [Google Scholar]
- Adam, R.; Delvart, V.; Pascal, G.; Valeanu, A.; Castaing, D.; Azoulay, D.; Giacchetti, S.; Paule, B.; Kunstlinger, F.; Ghémard, O.; et al. Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: A model to predict long-term survival. Ann. Surg. 2004, 240, 644–657; discussion 657–648. [Google Scholar] [CrossRef]
- Ito, K.; Govindarajan, A.; Ito, H.; Fong, Y. Surgical Treatment of Hepatic Colorectal Metastasis: Evolving Role in the Setting of Improving Systemic Therapies and Ablative Treatments in the 21st Century. Cancer J. 2010, 16, 103–110. [Google Scholar]
- Clavien, P.-A.; Selzner, N.; Morse, M.; Selzner, M.; Paulson, E. Downstaging of hepatocellular carcinoma and liver metastases from colorectal cancer by selective intra-arterial chemotherapy. Surgery 2002, 131, 433–442. [Google Scholar] [CrossRef]
- Kemeny, N.E.; Melendez, F.D.; Capanu, M.; Paty, P.B.; Fong, Y.; Schwartz, L.H.; Jarnagin, W.R.; Patel, D.; D’Angelica, M. Conversion to resectability using hepatic artery infusion plus systemic chemotherapy for the treatment of unresectable liver metastases from colorectal carcinoma. J. Clin. Oncol. 2009, 27, 3465–3471. [Google Scholar] [CrossRef]
- Sugihara, K.; Uetake, H. Therapeutic strategies for hepatic metastasis of colorectal cancer: Overview. J. Hepato-Biliary-Pancreat. Sci. 2012, 19, 523–527. [Google Scholar] [CrossRef]
- She, W.H.; Cheung, T.T. Bridging and downstaging therapy in patients suffering from hepatocellular carcinoma waiting on the list of liver transplantation. Transl. Gastroenterol. Hepatol. 2016, 1, 34. [Google Scholar] [CrossRef]
- Abdelrahim, M.; Esmail, A.; Saharia, A.; Abudayyeh, A.; Abdel-Wahab, N.; Diab, A.; Murakami, N.; Kaseb, A.O.; Chang, J.C.; Gaber, A.O. Utilization of Immunotherapy for the Treatment of Hepatocellular Carcinoma in the Peri-Transplant Setting: Transplant Oncology View. Cancers 2022, 14, 1760. [Google Scholar] [CrossRef] [PubMed]
- Abboud, K.; Umoru, G.; Esmail, A.; Abudayyeh, A.; Murakami, N.; Al-Shamsi, H.O.; Javle, M.; Saharia, A.; Connor, A.A.; Kodali, S. Immune checkpoint inhibitors for solid tumors in the adjuvant setting: Current progress, future directions, and role in transplant oncology. Cancers 2023, 15, 1433. [Google Scholar] [CrossRef] [PubMed]
- Mellman, I.; Coukos, G.; Dranoff, G. Cancer immunotherapy comes of age. Nature 2011, 480, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Buchbinder, E.I.; Desai, A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am. J. Clin. Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef]
- Munker, S.; De Toni, E.N. Use of checkpoint inhibitors in liver transplant recipients. United Eur. Gastroenterol. J. 2018, 6, 970–973. [Google Scholar] [CrossRef]
- Abdel-Wahab, N.; Shah, M.; Suarez-Almazor, M.E. Adverse Events Associated with Immune Checkpoint Blockade in Patients with Cancer: A Systematic Review of Case Reports. PLoS ONE 2016, 11, e0160221. [Google Scholar] [CrossRef]
- Tabrizian, P.; Florman, S.S.; Schwartz, M.E. PD-1 inhibitor as bridge therapy to liver transplantation? Am. J. Transplant. 2021, 21, 1979–1980. [Google Scholar] [CrossRef]
- Monden, M.; Okamura, J.; Sakon, M.; Gotoh, M.; Kobayashi, K.; Umeshita, K.; Yamada, T.; Kuroda, C.; Sakurai, M.; Mori, T. Significance of transcatheter chemoembolization combined with surgical resection for hepatocellular carcinomas. Cancer Chemother. Pharmacol. 1989, 23, S90–S95. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, Q.; He, J.; Yang, J.; Yang, G.; Wu, M. The effect of preoperative transcatheter hepatic arterial chemoembolization on disease-free survival after hepatectomy for hepatocellular carcinoma. Cancer 2000, 89, 2606–2612. [Google Scholar] [CrossRef]
- Lei, J.; Zhong, J.; Wang, W.; Yan, L.; Zhou, Q.; Li, B.; Wen, T.; Xu, M.; Yang, J.; Wei, Y. Preoperative transcatheter arterial chemoembolization for resectable hepatocellular carcinoma: A single center analysis. Ann. Hepatol. 2014, 13, 394–402. [Google Scholar]
- Abdelrahim, M.; Esmail, A.; Umoru, G.; Westhart, K.; Abudayyeh, A.; Saharia, A.; Ghobrial, R.M. Immunotherapy as a Neoadjuvant Therapy for a Patient with Hepatocellular Carcinoma in the Pretransplant Setting: A Case Report. Curr. Oncol. 2022, 29, 4267–4273. [Google Scholar] [CrossRef] [PubMed]
- Aby, E.S.; Lake, J.R. Immune Checkpoint Inhibitor Therapy Before Liver Transplantation-Case and Literature Review. Transplant. Direct 2022, 8, e1304. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.H.; Wang, G.B.; Huang, F.; Qin, R.; Yu, X.J.; Wu, R.L.; Hou, L.J.; Ye, Z.H.; Zhang, X.H.; Zhao, H.C. Pretransplant use of toripalimab for hepatocellular carcinoma resulting in fatal acute hepatic necrosis in the immediate postoperative period. Transpl. Immunol. 2021, 66, 101386. [Google Scholar] [CrossRef]
- Chen, Z.; Hong, X.; Wang, T.; Guo, Y.; Huang, C.; Li, M.; He, X.; Ju, W.; Chen, M. Prognosis after liver transplantation in patients treated with anti-PD-1 immunotherapy for advanced hepatocellular carcinoma: Case series. Ann. Palliat. Med. 2021, 10, 9354–9361. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, Y.; Schnickel, G.T.; Hosseini, M.; Burgoyne, A.M.; Ajmera, V.H.; Morris, G.P.; Mendler, M.H.; Parekh, J.R.; Abushamat, F.; Vodkin, I.; et al. Rescue liver re-transplantation after graft loss due to severe rejection in the setting of pre-transplant nivolumab therapy. Clin. J. Gastroenterol. 2021, 14, 1718–1724. [Google Scholar] [CrossRef]
- Kang, E.; Martinez, M.; Moisander-Joyce, H.; Saenger, Y.M.; Griesemer, A.D.; Kato, T.; Yamashiro, D.J.; Remotti, H.; Gartrell, R.D. Stable liver graft post anti-PD1 therapy as a bridge to transplantation in an adolescent with hepatocellular carcinoma. Pediatr. Transplant. 2022, 26, e14209. [Google Scholar] [CrossRef]
- Lizaola-Mayo, B.C.; Mathur, A.K.; Borad, M.J.; Jadlowiec, C.C.; Lam-Himlin, D.M.; Corey, R.L.; Iqbal, S.; Okubo, K.; Byrne, T.J.; Moss, A.A.; et al. Immunotherapy as a Downstaging Tool for Liver Transplantation in Hepatocellular Carcinoma. Am. J. Gastroenterol. 2021, 116, 2478–2480. [Google Scholar] [CrossRef]
- Nordness, M.F.; Hamel, S.; Godfrey, C.M.; Shi, C.; Johnson, D.B.; Goff, L.W.; O’Dell, H.; Perri, R.E.; Alexopoulos, S.P. Fatal hepatic necrosis after nivolumab as a bridge to liver transplant for HCC: Are checkpoint inhibitors safe for the pretransplant patient? Am. J. Transpl. 2020, 20, 879–883. [Google Scholar] [CrossRef]
- Woo, S.M.; Kimchy, A.V.; Sequeira, L.M.; Dorris, C.S.; He, A.R.; Rangnekar, A.S. Immunotherapy Use Prior to Liver Transplant in Patients with Hepatocellular Carcinoma. Curr. Oncol. 2022, 29, 9813–9825. [Google Scholar] [CrossRef]
- De Bruyn, P.; Van Gestel, D.; Ost, P.; Kruse, V.; Brochez, L.; Van Vlierberghe, H.; Devresse, A.; Del Marmol, V.; Le Moine, A.; Aspeslagh, S. Immune checkpoint blockade for organ transplant patients with advanced cancer: How far can we go? Curr. Opin. Oncol. 2019, 31, 54–64. [Google Scholar] [CrossRef]
- Rammohan, A.; Reddy, M.S.; Farouk, M.; Vargese, J.; Rela, M. Pembrolizumab for metastatic hepatocellular carcinoma following live donor liver transplantation: The silver bullet? Hepatology 2018, 67, 1166–1168. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Wahab, N.; Safa, H.; Abudayyeh, A.; Johnson, D.H.; Trinh, V.A.; Zobniw, C.M.; Lin, H.; Wong, M.K.; Abdelrahim, M.; Gaber, A.O.; et al. Checkpoint inhibitor therapy for cancer in solid organ transplantation recipients: An institutional experience and a systematic review of the literature. J. Immunother. Cancer 2019, 7, 106. [Google Scholar] [CrossRef] [PubMed]
- Al Jarroudi, O.; Ulusakarya, A.; Almohamad, W.; Afqir, S.; Morere, J.F. Anti-Programmed Cell Death Protein 1 (PD-1) Immunotherapy for Metastatic Hepatocellular Carcinoma After Liver Transplantation: A Report of Three Cases. Cureus 2020, 12, e11150. [Google Scholar] [CrossRef] [PubMed]
- DeLeon, T.T.; Salomao, M.A.; Aqel, B.A.; Sonbol, M.B.; Yokoda, R.T.; Ali, A.H.; Moss, A.A.; Mathur, A.K.; Chascsa, D.M.; Rakela, J.; et al. Pilot evaluation of PD-1 inhibition in metastatic cancer patients with a history of liver transplantation: The Mayo Clinic experience. J. Gastrointest. Oncol. 2018, 9, 1054–1062. [Google Scholar] [CrossRef]
- De Toni, E.N.; Gerbes, A.L. Tapering of Immunosuppression and Sustained Treatment with Nivolumab in a Liver Transplant Recipient. Gastroenterology 2017, 152, 1631–1633. [Google Scholar] [CrossRef]
- Varkaris, A.; Lewis, D.W.; Nugent, F.W. Preserved Liver Transplant After PD-1 Pathway Inhibitor for Hepatocellular Carcinoma. Am. J. Gastroenterol. 2017, 112, 1895–1896. [Google Scholar] [CrossRef]
- Gassmann, D.; Weiler, S.; Mertens, J.C.; Reiner, C.S.; Vrugt, B.; Nägeli, M.; Mangana, J.; Müllhaupt, B.; Jenni, F.; Misselwitz, B. Liver Allograft Failure After Nivolumab Treatment-A Case Report with Systematic Literature Research. Transplant. Direct 2018, 4, e376. [Google Scholar] [CrossRef]
- Friend, B.D.; Venick, R.S.; McDiarmid, S.V.; Zhou, X.; Naini, B.; Wang, H.; Farmer, D.G.; Busuttil, R.W.; Federman, N. Fatal orthotopic liver transplant organ rejection induced by a checkpoint inhibitor in two patients with refractory, metastatic hepatocellular carcinoma. Pediatr. Blood Cancer 2017, 64. [Google Scholar] [CrossRef]
- Gomez, P.; Naim, A.; Zucker, K.; Wong, M. A Case of Hepatocellular Carcinoma (HCC) Immunotherapy Inducing Liver Transplant Rejection: 2416. Off. J. Am. Coll. Gastroenterol. ACG 2018, 113, S1347. [Google Scholar] [CrossRef]
- Kumar, S. 2235°Nivolumab-Induced Severe Allograft Rejection in Recurrent Post-Transplant Hepatocellular Carcinoma. Off. J. Am. Coll. Gastroenterol. ACG 2019, 114, S1251. [Google Scholar] [CrossRef]
- Pandey, A.; Cohen, D.J. Ipilumumab for hepatocellular cancer in a liver transplant recipient, with durable response, tolerance and without allograft rejection. Immunotherapy 2020, 12, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Tang, W.; Du, C. Immune Checkpoint Inhibitors in Patients with Recurrent Hepatocellular Carcinoma after Liver Transplantation: A Case Report and Literature Review. Curr. Cancer Drug Targets 2020, 20, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, L.; Mou, H.B.; Yu, L.F.; Zhu, H.K.; Yang, Z.; Liao, Q.; Zheng, S.S. Immune checkpoint inhibitor for hepatocellular carcinoma recurrence after liver transplantation. Hepatobiliary Pancreat. Dis. Int. 2020, 19, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.M.; Wang, J.; Huang, X.W.; Huang, X.Y.; He, Y.F.; Ji, Y.; Chen, Y.; Wu, D.; Lu, J.C.; Sun, Q.M.; et al. Graft Programmed Death Ligand 1 Expression as a Marker for Transplant Rejection Following Anti-Programmed Death 1 Immunotherapy for Recurrent Liver Tumors. Liver Transplant. 2021, 27, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Ben Khaled, N.; Roessler, D.; Reiter, F.P.; Seidensticker, M.; Guba, M.; De Toni, E.N. Extending the Use of Atezolizumab and Bevacizumab to a Liver Transplant Recipient: Need for a Posttransplant Registry. Liver Transplant. 2021, 27, 928–929. [Google Scholar] [CrossRef]
- Schvartsman, G.; Perez, K.; Sood, G.; Katkhuda, R.; Tawbi, H. Immune Checkpoint Inhibitor Therapy in a Liver Transplant Recipient with Melanoma. Ann. Intern. Med. 2017, 167, 361–362. [Google Scholar] [CrossRef]
- Biondani, P.; De Martin, E.; Samuel, D. Safety of an anti-PD-1 immune checkpoint inhibitor in a liver transplant recipient. Ann. Oncol. 2018, 29, 286–287. [Google Scholar] [CrossRef]
- Kuo, J.C.; Lilly, L.B.; Hogg, D. Immune checkpoint inhibitor therapy in a liver transplant recipient with a rare subtype of melanoma: A case report and literature review. Melanoma Res. 2018, 28, 61–64. [Google Scholar] [CrossRef]
- Kondo, T.; Kawachi, S.; Nakatsugawa, M.; Takeda, A.; Kikawada, N.; Aihara, Y.; Okimura, A.; Hirano, H.; Ogawa, Y.; Tsukahara, K. Nivolumab for recurrent/metastatic hypopharyngeal squamous cell carcinoma in a liver transplant recipient. Auris Nasus Larynx 2022, 49, 721–726. [Google Scholar] [CrossRef]
- Delyon, J.; Zuber, J.; Dorent, R.; Poujol-Robert, A.; Peraldi, M.N.; Anglicheau, D.; Lebbe, C. Immune Checkpoint Inhibitors in Transplantation-A Case Series and Comprehensive Review of Current Knowledge. Transplantation 2021, 105, 67–78. [Google Scholar] [CrossRef]
- De Rubis, G.; Rajeev Krishnan, S.; Bebawy, M. Liquid Biopsies in Cancer Diagnosis, Monitoring, and Prognosis. Trends Pharmacol. Sci. 2019, 40, 172–186. [Google Scholar] [CrossRef] [PubMed]
- Reddy, T.; Esmail, A.; Chang, J.C.; Ghobrial, R.M.; Abdelrahim, M. Utility of Cell-Free DNA Detection in Transplant Oncology. Cancers 2022, 14, 743. [Google Scholar] [CrossRef] [PubMed]
- Abdelrahim, M.; Esmail, A.; Saharia, A.; McMillan, R.; He, A.R.; Starr, J.; Dhani, H.; Aushev, V.N.; Koyen, A.; Malashevich, N.H. The feasibility of tumor recurrence detection in liver post-transplantation for patients with hepatocellular carcinoma via personalized, tumor-informed ctDNA testing. Ann. Oncol. 2022, 33, S308–S309. [Google Scholar] [CrossRef]
- Ono, A.; Fujimoto, A.; Yamamoto, Y.; Akamatsu, S.; Hiraga, N.; Imamura, M.; Kawaoka, T.; Tsuge, M.; Abe, H.; Hayes, C.N.; et al. Circulating Tumor DNA Analysis for Liver Cancers and Its Usefulness as a Liquid Biopsy. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 516–534. [Google Scholar] [CrossRef] [PubMed]
- Kasi, P.M.; Budde, G.; Dayyani, F.; Botta, G.P.; Diehl, A.; King, G.T.; Malla, M.; Abdelrahim, M.; Hanna, D.L.; Schafer, L.N.; et al. Tumor-informed assessment of circulating tumor DNA and its incorporation into practice for patients with hepatobiliary cancers. J. Clin. Oncol. 2021, 39, 4103. [Google Scholar] [CrossRef]
- Jiang, P.; Chan, C.W.; Chan, K.C.; Cheng, S.H.; Wong, J.; Wong, V.W.; Wong, G.L.; Chan, S.L.; Mok, T.S.; Chan, H.L.; et al. Lengthening and shortening of plasma DNA in hepatocellular carcinoma patients. Proc. Natl. Acad. Sci. USA 2015, 112, E1317–E1325. [Google Scholar] [CrossRef]

| Malignancy | OS (5 y) | DFS (5 y) | Recurrence | References |
|---|---|---|---|---|
| HCC | 57.7% | 65.7% | NA | Li et al. [63] |
| 71% | 72% | NA | Lim et al. [58] | |
| 75% (4 y) | 83% (4 y) | NA | Milan Criteria [29] | |
| 75% | NA | NA | UCSF Criteria [38] | |
| 61.8% | 80% | NA | Dallas Criteria [39] | |
| 69% | NA | 14% | Valencia Criteria [48] | |
| 71.2% | NA | NA | Up-to-7 Criteria [35] | |
| 82% | NA | 7% | Kyoto Criteria [33] | |
| 68% | NA | NA | French Criteria [40] | |
| 62.4% | 56.5% | NA | Hangzhou Criteria [41] | |
| 74.6% (4 y) | NA | NA | Edmonton Criteria [43] | |
| 78% | NA | NA | Toronto Criteria [46] | |
| 70% | NA | NA | Metroticket 2.0 [45] | |
| HCCA | 17% | 92% | 9% | De Vreede et al. [65] |
| 29% | 33% | NA | Hong et al. [66] | |
| 30% | 30% | 53% | Robles et al. [67] | |
| ICCA | 18% | 31% | 60% | Casavilla et al. [68] |
| 23% | NA | 51% | Meyer et al. [69] | |
| 21.5% | 21.5% | >50% | Panayotova et al. [70] | |
| 65% | 18% | NA | Sapisochin et al. [71] | |
| HBL | 78% | 82% | 28% | Pham et al. [72] |
| NETLM | 52% | 30% | NA | Le Treut et al. (2013) [73] |
| 47% | 20% | NA | Le Treut et al. (2008) [74] | |
| 48% | 32% | NA | Gedaly et al. [75] | |
| 80% | 21% | NA | Rosenau et al. [76] | |
| CRLM | 60% | NA | 90% * | Hagness et al. [77] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelrahim, M.; Esmail, A.; Abudayyeh, A.; Murakami, N.; Victor, D.; Kodali, S.; Cheah, Y.L.; Simon, C.J.; Noureddin, M.; Connor, A.; et al. Transplant Oncology: An Emerging Discipline of Cancer Treatment. Cancers 2023, 15, 5337. https://doi.org/10.3390/cancers15225337
Abdelrahim M, Esmail A, Abudayyeh A, Murakami N, Victor D, Kodali S, Cheah YL, Simon CJ, Noureddin M, Connor A, et al. Transplant Oncology: An Emerging Discipline of Cancer Treatment. Cancers. 2023; 15(22):5337. https://doi.org/10.3390/cancers15225337
Chicago/Turabian StyleAbdelrahim, Maen, Abdullah Esmail, Ala Abudayyeh, Naoka Murakami, David Victor, Sudha Kodali, Yee Lee Cheah, Caroline J. Simon, Mazen Noureddin, Ashton Connor, and et al. 2023. "Transplant Oncology: An Emerging Discipline of Cancer Treatment" Cancers 15, no. 22: 5337. https://doi.org/10.3390/cancers15225337





