Simple Summary
It is unknown whether different types of exercise effect physical function, quality of life, mental wellbeing, and symptoms in older patients with blood cancers. No studies specifically focusing on patients over 65 years were identified. However, across adult age groups, we found that exercise has small to moderate positive effects on physical function, aerobic capacity, muscle strength, quality of life, fatigue, pain, anxiety, and depression. These benefits were generally consistent regardless of age, except for physical function and pain, which favored younger adults. Overall, exercise improves physical function and quality of life and reduces symptoms in adults with blood cancers undergoing treatment, but the role of age remains uncertain.
Abstract
Older patients receiving antineoplastic treatment face challenges such as frailty and reduced physical capacity and function. This systematic review and meta-analysis aimed to evaluate the effects of exercise interventions on physical function outcomes, health-related quality of life (QoL), and symptom burden in older patients above 65 years with hematological malignancies undergoing antineoplastic treatment. This review adheres to Cochrane guidelines, with the literature searches last updated on 27 March 2024, including studies with patients above 18 years. Screening of identified studies, data extraction, risk of bias, and GRADE assessments were performed independently by two authors. Meta-analyses evaluated the impact of exercise, considering advancing age. Forty-nine studies contributed data to the meta-analyses. Five studies included patients with a mean age above 60 years, and none included only patients above 60. Exercise interventions had moderate to small positive effects on QoL global (SMD 0.34, 95% CI [0.04–0.64]) and physical function (SMD 0.29, 95% CI [0.12–0.45]). Age did not explain the variability in exercise effects, except for physical function (slope 0.0401, 95% CI [0.0118–0.0683]) and pain (slope 0.0472, 95% CI [0.01–0.09]), which favored younger patients. Exercise interventions improve physical function and QoL and reduce symptoms in adults with hematological malignancies undergoing antineoplastic treatment; however, the influence of age remains inconclusive.
1. Introduction
The global increase in the aging population, in tandem with the delayed onset of most hematological malignancies, has led to a higher incidence of these cancers among older individuals [1,2]. Approximately half of all hematological malignancies occur in adults aged 65 years and older, and 70% of cancer-related deaths occur within this age group [3]. Overall survival in adults above 75 years old with hematological cancers, particularly acute leukemia, is low [4]. Older adults with hematological malignancies undergoing medical antineoplastic treatment experience challenges, including frailty and decline in physical and functional capacity, which can further burden their cancer treatment and overall recovery [5]. Among older patients with solid and hematological cancers, frailty is a prevalent concern associated with suboptimal therapeutic responses, increased treatment-related toxicity, and overall poorer survival outcomes [6]. Moreover, challenges like pain, fatigue, insomnia, and mood disturbances are prevalent among older cancer patients, significantly impacting their quality of life [7]. The estimated prevalence of cancer-related fatigue (CRF) is as high as 70% in individuals above 65 years [8]. This not only hinders everyday functioning but may also lead to significant disability and increased reliance on assistance for activities necessary for independent living [9].
Effective management of hematological malignancies in older patients requires a comprehensive, multidisciplinary approach that addresses both the disease and overall health and wellbeing [5,10]. In addition to the benefits provided by onco-geriatric interventions, which include screening for comorbidities and frailty to improve treatment outcomes, there is an increased interest in exploring nonpharmacological clinical interventions to improve health-related quality of life (QoL) and outcomes in older adults with solid and hematological cancers [11,12,13,14,15]. In recent years, growing recognition of the feasibility, safety, and potential benefits of exercise in improving clinical outcomes and QoL in patients with hematological malignancies [16,17] aligns with studies showing positive effects on physical capacity and function, psychological wellbeing, and the mitigation of fatigue and other treatment-related side effects in this patient population [18,19,20,21]. To guide exercise practices in older adults, several recommendations are available for clinical and research practices, such as the International exercise recommendations for older adults from 2021 [22], the American College of Sports Medicine (ACSM) exercise guidelines for older adults from 2009 [23], recommendations from 2015 for designing exercise clinical trials for older adults with cancer [24], and the 2019 roundtable on exercise guidelines for cancer survivors, which addresses issues of older cancer patients [25]. There remains, however, limited evidence supporting the benefits of exercise in older adults with hematological malignancies in these guidelines.
Consequently, there is a clear imperative to explore the impact of exercise in older adults with hematological cancers [26]. Given age-related physiological changes coupled with the complexities of hematological diseases and intensive treatments, there is a need for a comprehensive investigation into the potential benefits of exercise for this population. A systematic review and meta-analysis can provide insights into the effectiveness of exercise interventions on diverse outcomes and guide clinical strategies to optimize the health and wellbeing of older patients undergoing neoplastic treatment for hematological malignancies.
The aim of this systematic review and meta-analysis was to investigate the effect of exercise interventions on physical function, QoL, and symptom burden in older adults with hematological malignancies receiving medical antineoplastic treatments.
2. Materials and Methods
This systematic review and meta-analysis adheres to Cochrane guidelines [27] and is registered in The International Prospective Register of Systematic Reviews (PROSPERO) database (registration no. CRD42019130979 amended 24 May 2021 and 8 August 2023) and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [28,29].
2.1. Information Sources and Search Strategy
A systematic search was conducted in March 2019 across four databases: MEDLINE, EMBASE, Cochrane Central Register of Controlled Trials (CENTRAL), and CINAHL. No randomized controlled trials exclusively including participants above 65 years with hematological malignancies were identified (Figure S1). In October 2020, the search was expanded to include individuals above 18 years old, and an additional database, Science Citation Index Expanded/Social Sciences Citation Index (SCIE/SSCI), was added (Figure S2). On 27 March 2024, an updated search was carried out in MEDLINE, EMBASE, CENTRAL, CINAHL, and SCIE/SSCI (Figure S3). Using the PICO framework (Problems, Interventions, Comparisons and Outcomes) [30], the searches included three main areas: (1) exercise-based interventions, (2) hematological malignancies during medical antineoplastic treatment, and (3) randomized controlled trials. The searches involved a combination of MeSH/Thesaurus/indexed terms and relevant synonyms (Figures S1–S3).
2.2. Eligibility Criteria
Studies included were randomized controlled trials, randomized pilot trials, randomized crossover trials, and randomized feasibility studies with adult participants (≥18 years) diagnosed with hematologic malignancies undergoing medical antineoplastic treatment, such as chemotherapy, chemoradiation, stem cell transplantation including allogeneic hematopoietic stem cell transplantation (allo HSCT), autologous stem cell transplantation (autologous HSCT), and high-dose chemotherapy with stem cell support. Studies that included participants just prior to starting treatment and up to six months post-treatment were considered. All types of exercise-based interventions and complementary alternative medicine (CAM) such as yoga, compared to standard care were considered for inclusion, including multimodal interventions where the exercise component constituted at least 50% of the intervention sessions. Interventions could be initiated prior to, during hospitalization or outpatient clinic visits, or after discharge, including home-based training. All durations of interventions were included. For comparator(s) or control groups, all types, whether active or inactive, were considered. Studies included measured outcome effects. No restrictions were applied for language or publication date.
2.3. Data Collection Process
Records were imported to Covidence, a data management program (www.covidence.org; https://app.covidence.org/reviews/59783, accessed on 14 June 2019; https://app.covidence.org/reviews/122435, accessed on 11 November 2020; https://app.covidence.org/reviews/425529; accessed on 27 March 2024) for duplicate removal, screening, and data extraction. Two authors independently performed screening of identified studies and data extraction on author details (year, country), population characteristics (age, gender, diagnosis, treatment), study characteristics (design, sample size, recruitment, timing, retention, feasibility, adherence, safety), exclusion criteria, intervention characteristics (delivery mode, type, length, intensity), control group specifics, and outcome measurements. Outcome data were extracted at baseline and post-intervention, the time point nearest to the intervention completion date.
To identify ongoing exercise-based randomized trials, a comprehensive search was carried out in ClinicalTrials.gov, accessed on 13 November 2023, including only patients above 60 years with hematological malignancies undergoing medical antineoplastic treatment (Table S1). Information extracted includes trial identifier, author information, population characteristics (age, gender, diagnosis, treatment modalities), study design, type and duration of exercise intervention, primary outcomes, and current study status.
2.4. Outcomes
The primary outcomes were changes in physical function (e.g., 6-min walk test (6MWT)) and QoL global (e.g., The European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire (EORTC QLQ-C30)). Measurements of QoL global included both generic and disease-specific patient-reported outcome measures (PROMs).
Secondary outcomes included changes in aerobic capacity (e.g., Volume Oxygen Peak, (VO2 Peak)), muscle strength (e.g., Sit to Stand test (STS)), body composition (e.g., fat-free mass measured by Dual X-ray Absorptiometry (DEXA)), and PROMs evaluating physical activity (e.g., International Physical Activity Questionnaire (IPAQ)), physical, emotional, and functional well-being (e.g., EORTC QLQ-C30). Patient-reported symptoms comprised anxiety (e.g., Hospital Anxiety and Depression Scale (HADS)), depression (e.g., HADS), fatigue (e.g., The Functional Assessment of Chronic Illness Therapy Fatigue Scale (FACIT-F)), and pain (e.g., EORTC QLQ-C30), in addition to an assessment of feasibility and safety.
For the meta-analysis, and for each outcome, distinct measurement instruments and tests were chosen, prioritizing those most widely applied for assessing the outcomes of exercise interventions, considering validity and reliability (Table S2). For each study, a single measurement instrument was selected for each outcome.
2.5. Risk of Bias Assessment
Risk of bias for each study was independently assessed by two of six authors (STL, GK, RFL, CG, MSE, MJ) using the Revised Cochrane risk-of-bias tool for randomized trials (RoB 2) [31]. Any disagreements were resolved through discussions with MJ.
2.6. Certainty of Evidence
The certainty of evidence for each outcome was assessed using the Grading of Recommendation, Assessment, Development and Evaluation tool (GRADE) [32] by three authors (STL, GK, MJ) utilizing the GRADEpro Guideline Development Tool [Software] (McMaster University and Evidence Prime, 2024; Available from gradepro.org; accessed on 21 August 2024). The certainty of evidence was evaluated in five domains: study quality, inconsistency, indirectness, imprecision, and publication bias. Risk of publication bias (small study bias) was assessed using Egger’s test and illustrated with a funnel plot for all outcomes, which was inspected for asymmetry.
2.7. Data Synthesis and Analysis
The effect size of each intervention was estimated as the standardized mean difference (SMD) with a 95% CI using the restricted maximum likelihood model (REML). The SMD was calculated as the difference in mean change between the intervention and control group, divided by the pooled standard deviation (SD), and adjusted to Hedges’ g to account for a small overestimation of the effect in small studies. A random effects model was used for all meta-analyses, as it was expected that participants, interventions, and outcome measures might differ between studies [27]. To facilitate a unified analysis across multiple outcomes, we analyzed changes in measurement scores from baseline to post-test, and studies not reporting change scores were converted to change scores by using a correlation of 0.6. Interpretation of the SMD adhered to Cohen’s recommendations: small effect was defined as 0.2, moderate effect as 0.5, and large effect as 0.8 [27].
Heterogeneity was evaluated using the I2 index, and the between-study variance as tau-square (t2). Meta-regression analyses employing STATA version 18 (StataCorp 2023. Stata Statistical Software: Release 18. College Station, TX, USA) were conducted to assess potential influences of mean age of participants on effects in the meta-analysis. The number of participants for each outcome corresponds to those who completed the study with data, which may not necessarily be the same as the total number included in the studies.
In subgroup analyses, we examined covariates including mean age as a dichotomous variable (<60 years or >60 years), diagnosis (acute leukemia, lymphoma, multiple myeloma, mixed diagnoses), and antineoplastic treatment (chemotherapy, allogeneic HSCT, autologous HSCT or both, different treatments). Given the anticipated heterogeneity in exercise interventions, we categorized the exercise interventions based on type (aerobic, strength, combined aerobic and strength, either aerobic or strength exercise, CAM), timing of exercise (before, during, after treatment, or combination), delivery mode (supervised, partly supervised, unsupervised), individual or group-based, and extensiveness of intervention, and risk of bias (high risk, some concerns, and low risk). Extensiveness of exercise interventions was categorized based on number of sessions, session length, and intensity, inspired by guidelines from the American College of Sports Medicine [25,33] and two systematic reviews of exercise interventions [34,35]. Each exercise intervention was categorized as less, moderate, or extensive. The categorization process was collaboratively conducted by three authors (MJ, STL, GK), with STL providing expertise as a physiotherapist.
3. Results
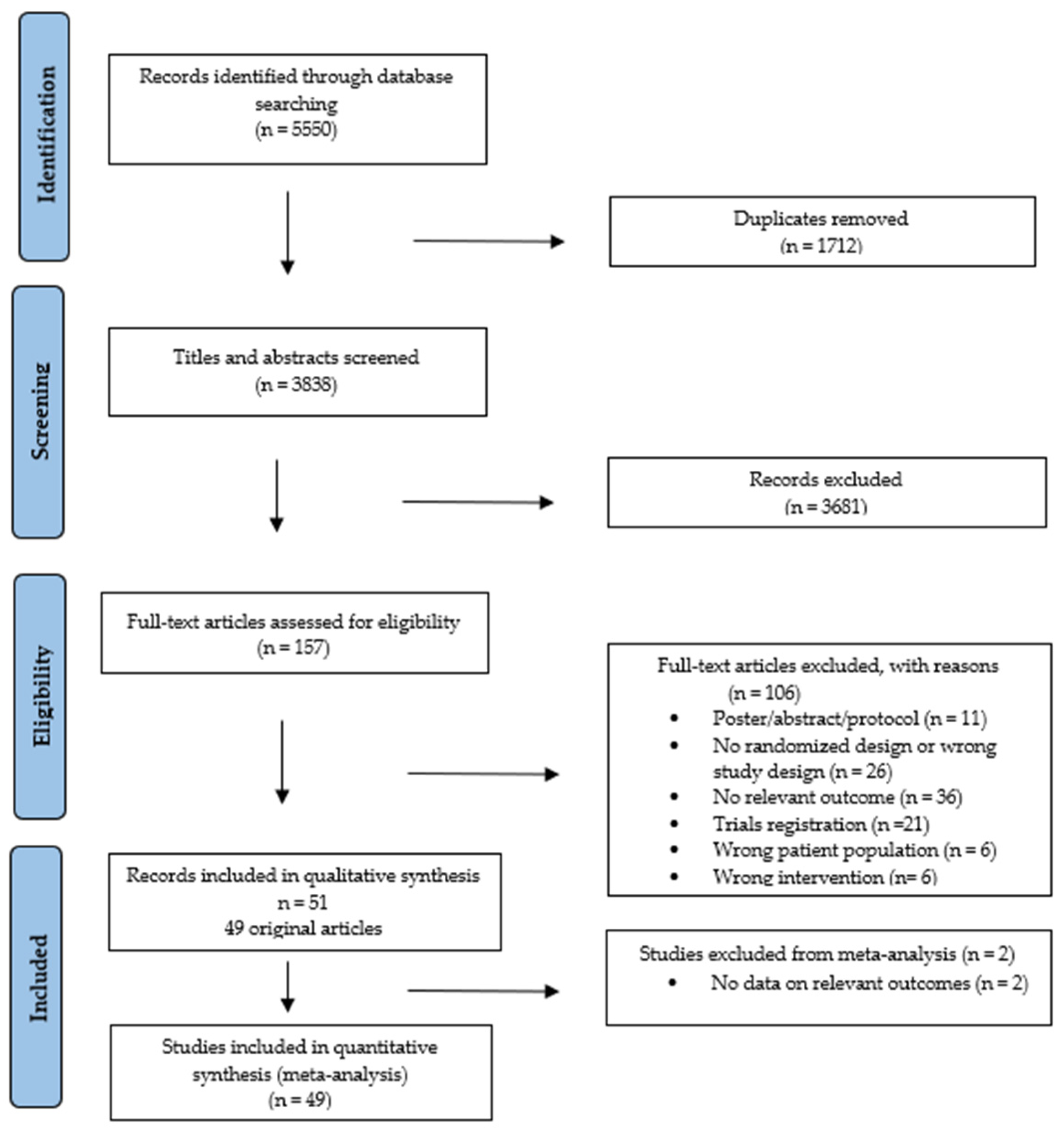
The searches identified 5550 records (Figures S2 and S3). After removing duplicates and screening, 49 studies were included [36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84], reported in 51 publications, with two of these being follow-up analyses [85,86]. All studies were included in the narrative review (Figure 1).
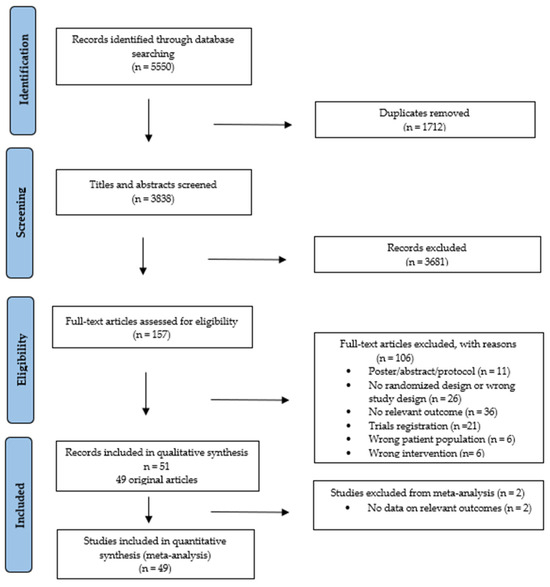
Figure 1.
PRISMA flowchart of study selection process.
3.1. Study Characteristics
In total, 3494 patients were included, with sample sizes ranging from 17–711 (Table 1). In 41 studies, the mean age was 50 years (range 18–90) [36,37,38,39,40,41,42,43,44,45,47,48,49,50,51,54,55,56,57,58,59,60,62,63,64,65,66,68,69,70,71,72,73,74,75,77,78,79,80,82,84], and in seven studies, the median age was 53 years [46,52,61,67,76,81,83]. Only five studies reported a mean age of participants ≥ 60 years (n = 359) [47,57,58,67,68]. Females constituted 43% of participants.

Table 1.
Characteristics of included studies.
Studies included mixed hematological cancer diagnoses (n = 30) [36,39,40,41,42,46,52,53,54,56,59,60,61,62,64,65,66,69,70,71,72,73,74,75,76,77,78,79,82,83], acute leukemia (n = 9) [37,38,43,44,45,55,63,80,81], multiple myeloma (n = 5) [49,50,57,67,68], and lymphoma (n = 5) [47,48,51,58,84]. Studies comprised patients undergoing chemotherapy (n = 20) [36,38,41,43,44,45,47,48,51,55,58,59,63,66,70,71,75,78,81,84], allo-HSCT (n = 10) [40,52,53,62,64,69,72,77,82,83], autologous HSCT (n = 4) [57,60,68,73], allogeneic and autologous HSCT (n = 6) [42,56,61,65,74,76], and different treatments (n = 8) [37,39,46,49,50,54,67,79]. One study did not specify the type of medical antineoplastic treatment [80].
Thirty-four studies were published within the last decade (2013–2024), most originating from the USA (n = 14) [43,46,48,49,50,52,53,56,57,59,61,74,77,83] and Germany (n = 9) [39,40,70,71,72,76,78,81,82]. The 49 trials were mainly designed as 2-arm (n = 46) [36,37,38,39,40,42,43,44,45,46,47,48,49,50,51,52,53,54,56,57,58,59,60,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,82,83,84], with 29% being pilot (n = 14) [39,43,45,46,49,55,57,59,60,68,70,71,81,83] or feasibility (n = 3) [37,53,76] randomized trials.
The control groups received usual care in 28 studies [38,39,40,43,45,47,48,49,50,51,52,57,59,60,62,63,64,65,67,68,70,73,74,77,78,79,80,84]. To counteract socio-psychological bias, 20 studies [36,38,41,42,44,46,53,54,55,56,58,61,66,71,72,75,76,81,82,83] implemented physical or attention activities (activity tracker, diary, phone calls) in the control groups.
3.2. Exercise Interventions
The type, intensity, length, and duration of the exercise interventions are presented in Table 1. The most common type of exercise intervention was a combination of aerobic (cycling, running, walking) and resistance (machines, resistance bands, body weight) exercise (n = 27) [36,37,38,39,40,41,42,43,49,50,54,57,58,60,62,63,65,66,67,68,69,70,73,76,77,78,82,86], followed by aerobic exercise (n = 11) [44,46,51,52,55,61,74,79,80,81,83], resistance exercise (n = 6) [55,56,71,72,75,81], and CAM practices such as yoga, Qi gong, relaxation, and breathing exercises (n = 7) [45,47,48,53,59,64,84]. The duration of exercise interventions in the included studies varied (range 1–36 weeks), with 12 weeks being the most common (n = 11) [37,51,53,54,58,59,60,63,65,79,80]. The most common frequency of intervention was three times a week (n = 11) [51,54,58,63,67,68,71,74,77,80,81], and some studies allowed variation and range in frequency, i.e., 2–3 times a week or 3–5 times a week (n = 5) [49,50,53,55,59]. The timing of the intervention was mostly during treatment (n = 21) [38,39,40,41,43,44,45,47,49,50,55,62,63,66,70,71,72,75,78,81,84], and after treatment (n = 13) [37,42,46,53,54,57,60,64,65,67,73,77,80].
The exercise interventions were mainly supervised (n = 27) [38,39,40,41,42,43,44,48,51,55,62,63,64,65,66,69,70,71,72,73,74,75,76,77,78,80,81]. Twenty-four took place at the hospital [38,39,40,42,43,44,45,48,51,62,63,64,66,69,70,71,72,74,75,76,77,78,80,81], and 12 were home-based [46,47,49,50,53,58,59,60,61,79,83,84]. Most studies offered individualized interventions (n = 44) [36,37,38,39,40,41,43,44,45,46,47,49,50,52,53,54,55,56,57,58,59,60,61,62,64,65,66,67,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,86]. The level of extensiveness of exercise interventions was predominantly moderate (n = 32) [37,38,39,40,43,49,50,51,52,54,55,56,57,58,60,61,62,65,67,69,70,71,72,74,75,76,77,79,80,81,82,83].
3.3. Effects of Exercise Interventions on Primary Outcomes
Data on physical function and QoL global were available for meta-analysis in 25 and 29 studies, including 1219 and 1447 participants, respectively. Exercise had significant moderate to small effects on physical function, SMD 0.29 (95% CI 0.12–0.45); I2; 48,17% and QoL global, SMD 0.34 (95% CI 0.04–0.64); I2: 87.39% (Table 2 and Figures S4 and S5).

Table 2.
Summary of findings. Population: Adults with hematological malignancies. Setting: Hospital-based, home-based, or a combination, other (e.g., clinics, nature). Exercise intervention: Aerobic, resistance, or combination, or CAM. Comparison: Usual care, no intervention, placebo control. Test timepoint: Post-intervention.
Meta-regression analyses on exercise benefits on age for QoL global showed a non-significant coefficient 0.0247 (95% CI −0.0140–0.0634, p = 0.210), favoring older patients, and physical function showed a significant negative coefficient 0.0401 (95% CI 0.0118–0.0683, p = 0.005), favoring younger patients (Figure S6).
Subgroup analyses on QoL global showed the largest effect in the age group over 60 years (SMD 1.29), and for physical function, it was below 60 years (SMD 0.32). Results for subgroup analyses for physical function and QoL global are presented in Figures S7 and S8, respectively.
3.4. Effects of Exercise Interventions on Secondary Outcomes
The largest effects of exercise were observed in aerobic capacity, SMD 0.53 (95% CI 0.27–0.79) and in muscle strength (SMD 0.47 (95% CI 0.17–0.78)). Moderate to small significant symptom-related benefits were found in fatigue (SMD 0.44), pain (SMD 0.43), and depression (SMD 0.37), and a small non-significant effect on anxiety (SMD 0.21). Significant moderate to small effects on QoL emotional (SMD 0.33), QoL functional (SMD 0.33), and QoL physical domains (SMD 0.32), as well as non-significant on the physical activity outcome (SMD 0.32), were observed. There was a small significant effect in body composition (SMD 0.20), all favoring exercise interventions (Table 2 and Figures S9–S19).
Except for pain (coefficient 0.0472 (95% CI 0.0078–0.0866, p = 0.019), favoring younger patients, age did not influence secondary outcomes (Figure S20).
Subgroup analyses showed that patients aged above 60 years yielded the largest effect on anxiety (SMD 0.44), fatigue (SMD 0.97), muscle strength (SMD 0.98), pain (SMD 1.31), QoL emotional (SMD 0.95), and QoL physical (SMD 1.50). Results for subgroup analyses are presented in Figures S21–S31.
3.5. Feasibility, Adverse Events, Adherence and Exclusion Criteria
Feasibility, adverse events, and adherence are presented in Table 3, and exclusion criteria in Table 1. One third of the studies (n = 16) did not provide information on adverse events (AE) [37,39,45,46,48,50,52,56,57,64,69,74,77,80,82,83]. Twenty-six studies reported no AE [36,40,41,42,43,44,47,49,53,55,58,59,60,61,62,65,66,67,70,71,75,76,78,79,81,84], and seven studies reported non-serious AE such as back, hip, and knee pain, and cardiorespiratory and gastrointestinal symptoms [38,51,54,63,68,72,73]. In the five studies with mean/median above 60 years, exercise interventions were tolerated, with three studies reporting no AE [47,58,67], one reporting non-serious AE [68], and one not reporting AE [57] (Table 3).

Table 3.
Feasibility and adverse events.
Thirty-three studies estimated sample sizes either through non-formal estimation or power calculations [36,37,38,39,40,41,42,43,45,46,47,48,50,51,55,58,61,62,63,64,65,66,67,70,73,75,77,78,79,80,81,83,84]. Twenty-two studies met sample size estimation [36,38,39,40,41,45,46,47,48,51,58,61,62,63,64,65,66,75,79,80,81,84], and eleven were underpowered [37,42,43,50,55,67,70,73,77,78,83]. Seventeen studies did not report sample size estimation [44,49,52,53,54,56,57,59,60,68,69,71,72,74,76,82]. Of those reporting (n = 41), a total of 7262 patients were screened for eligibility across the studies. Of these, 3552 were included in the studies, and 2924 participants completed the post-test, resulting in a retention rate of 82.3%.
Adherence to exercise was reported in 28 studies with a mean exercise adherence of 70% (range 15–100%), though 21 studies did not report adherence [39,40,42,44,49,50,53,55,57,58,60,61,62,64,68,69,70,75,76,80,83]. Two studies with patients mean age above 60 reported high adherences to the exercise intervention (range 75–96%) [47,67].
Inclusion and exclusion criteria varied across the 49 studies. The number of individual exclusion criteria per study was mean five (range 1–17), with the most frequent exclusion criteria being cardiovascular disease (n = 24), malignancy (n = 23), musculoskeletal disorder (n = 22), and mental health challenges (n = 18). Seven studies did not report exclusion criteria [42,44,49,57,69,73,82]. Despite detailed descriptions of inclusion and exclusion criteria in most studies, several did not report reasons for study exclusion (n = 14) [39,40,42,45,46,48,49,50,61,64,66,72,77,80]. In studies that reported (n = 34), the three most prevalent reasons for study exclusion were related to a) medical and health status: comorbidity, unstable conditions, adverse effects, malignancy, symptoms, low blood counts; b) physical performance: mobility challenges and frailty; and c) mental health issues: psychological instability and cognitive impairment. Some studies were not transparent, detailed, or systematic in reporting reasons for exclusion; and in some cases, it was just stated that participants did not meet inclusion criteria without providing explanation.
3.6. Risk of Bias in Individual Studies and across Studies
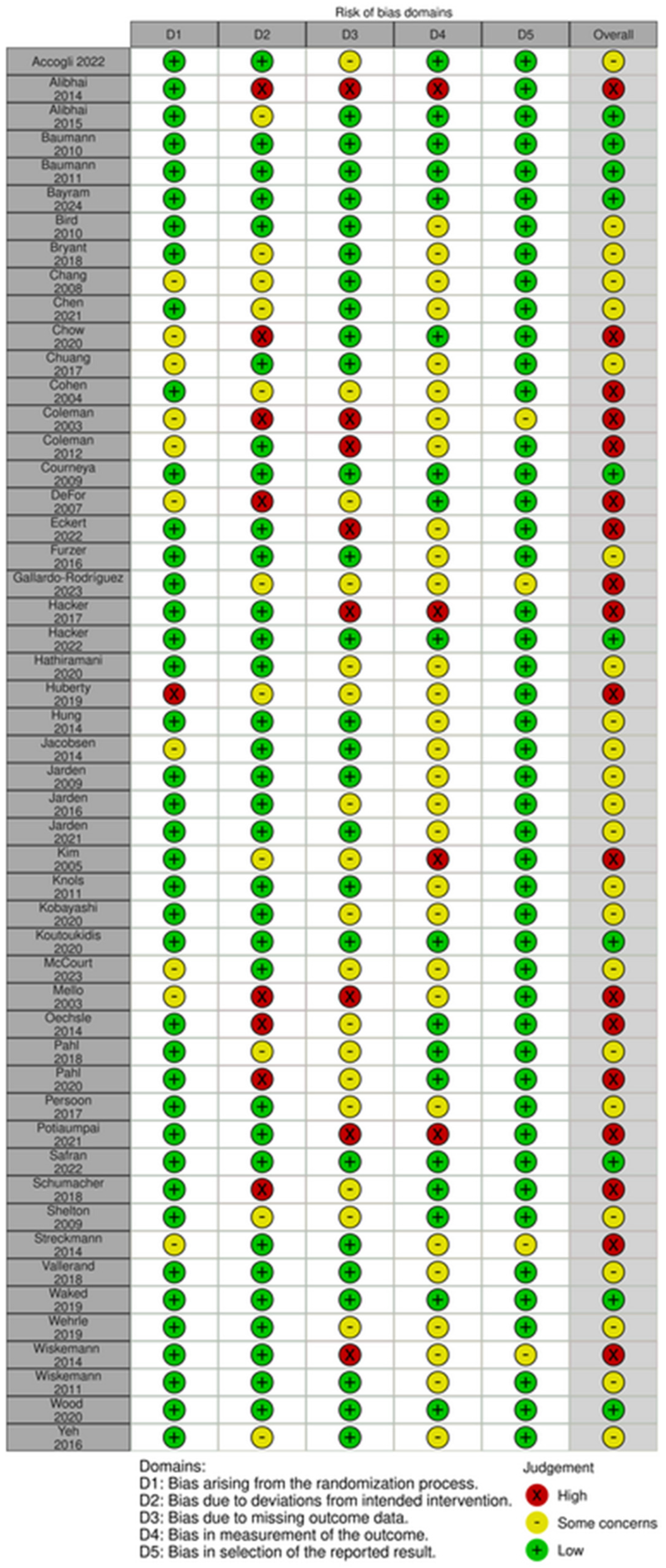
Risk of bias assessment for each study is reported in Figure 2. Most (n = 23) were assessed to have some concerns [36,42,43,44,45,47,54,58,60,61,62,63,65,66,68,71,73,77,79,81,82]. Eighteen were evaluated as high risk [37,46,48,49,50,52,53,55,56,59,64,69,70,72,74,76,78,86] and ten as low risk [38,39,40,41,51,57,67,75,80,83]. High risk and some concerns were mainly due to Domain 2, deviations from the intended intervention, or Domain 3, missing outcome data.
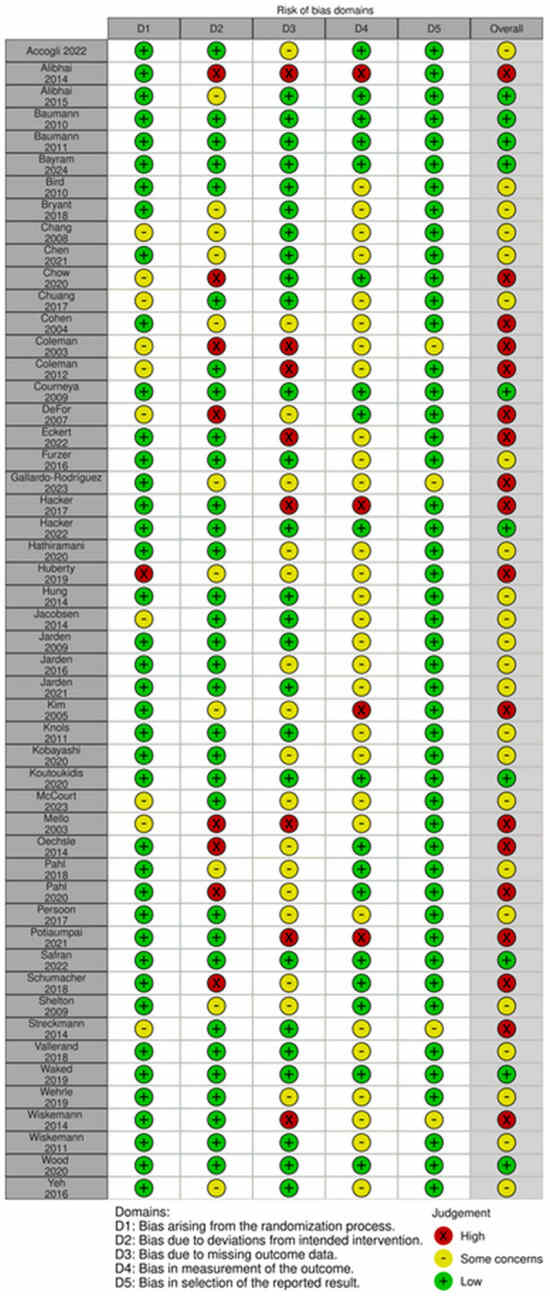
Figure 2.
Risk of Bias [36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86].
3.7. Quality of Evidence (GRADE)
Low to very low quality of evidence was found for a small effect on primary outcomes; physical function and QoL global, as evidence was downgraded due to high risk of bias, inconsistency, and risk of publication bias (Table 2). For secondary outcomes, only body composition showed high quality of evidence but for a small effect, while the remaining secondary outcomes ranged from very low to moderate levels of evidence.
3.8. Ongoing Studies Registered in Clinical Trials
There are currently four ongoing randomized exercise trials for older adults with hematological malignancies. Two studies recruit patients over 60 years, and the other two recruit patients over 65 and 70 (Table 4).

Table 4.
Ongoing exercise randomized trials in patients above 60 years with hematological malignancies (Clinicaltrials.gov—accessed on 13 November 2023).
4. Discussion
This systematic review and meta-analysis is a synthesis of data on the effect of exercise interventions in adults with hematological malignancies undergoing medical antineoplastic treatments. Notably, we did not find any studies that only included adults aged 65 years and older. Therefore, the scope was expanded to include all adult age groups (above 18 years), and we found exercise interventions to have significant positive benefits on outcomes, irrespective of age. Among the 49 studies analyzed, only five included a sample with mean or median age over 60 years, indicating a scarcity of evidence specifically addressing the older population with hematological malignancies. Likewise, Knowles et al. 2022 in a review of reviews of older people with cancer did not find any systematic reviews including a population over 65 years [87]. Similarly, Mikkelsen et al. 2020 in a systematic review of exercise interventions in older cancer patients identified only four studies that included a sample of patients above 65 years and found inconclusive evidence regarding benefits of exercise in this age group [88].
Given the rising prevalence of hematological malignancies among older adults and the increasing demand for more comprehensive approaches to treatment and care for this population [5,10], evidence regarding exercise remains notably lacking for older adults. We identified four ongoing exercise-based randomized trials in patients with hematological malignancies above 60 years registered in clinicaltrials.gov, accessed on 13 November 2023 (Table 4. These ongoing studies, though few, highlight the growing need to understand the role of exercise in improving physical function and QoL, and mitigating symptoms in older adults undergoing antineoplastic treatment.
In this review, we found exercise to have small to moderate certainty evidence across most outcomes across age groups. Likewise, several systematic reviews and meta-analyses, each with relatively few studies, also found exercise benefits in adults with hematological malignancies. Abo et al. (2021) reported moderate evidence in functional capacity, QoL global, and fatigue [20]. Moore et al. (2023) found evidence for improvements in physical function [21]. Yang et al. 2022 found significant improvements in QoL, emotional functioning, and pain [16]. Knips et al. (2019) found low-certainty evidence on depression and anxiety and moderate evidence for fatigue [18]. Research across hematological diagnoses and treatments consistently indicates that exercise positively impacts a broad range of outcomes in adults generally. However, our meta-regression analysis found that impact on QoL global favored advancing age, while physical function and pain favored younger adults. Our subanalyses, which should be interpreted with caution due to the limited number of studies included, showed that those aged above 60 had the largest effects on QoL domains (global, emotional, and physical), muscle strength, pain, fatigue, and anxiety, whereas those below 60 years showed the largest effects in physical function, aerobic capacity, QoL functional, and body composition.
Exercise interventions in our systematic review were reported as feasible and safe in 24 studies, with seven studies documenting nonserious AEs. However, one-third of the studies (n = 16) did not report on AEs. Similarly, GroBek et al.’s (2023) systematic review on the safety and feasibility of exercise interventions in patients with hematological cancer (12 studies), which includes the same studies as our systematic review, concluded that exercise is feasible and safe in the studies reporting on it, but also found a similar lack of AE reporting, and when provided, the information was often insufficient [17]. We recommend systematic monitoring and adequate documentation of feasibility and safety, including AEs, especially when providing exercise to hematological patients, given the potential comorbidities. This approach would facilitate targeted delivery of exercise interventions, not only to older adults but also to specific patient populations experiencing comorbidities.
Studies included in our review did not exclude patients based on older age; however, most studies used stringent exclusion criteria that targeted comorbidities and reduced function and performance inherent to this age group. This is evident in the detailed and high number of inclusion and exclusion criteria (up to 16), but importantly, reasons for exclusion were either not reported at all or reported with lack of detail, a pattern consistent with other exercise reviews of older cancer patients [87,88]. The rigorous exclusion criteria may contribute to the underrepresentation of older patients with comorbidities in exercise studies, as pointed out in several reviews [89,90,91]. However, there are exceptions, as in a feasibility pilot strength training program among an older population of patients (mean 68 years) with multiple myeloma, which included those with osteolytic lesions, establishing both feasibility and safety [92]. Older adults with the greatest need for exercise may not be considered suitable to participate due to strict safety criteria. This emphasizes the challenge of striking a balance between prioritizing safety and unintentionally excluding patients who could derive benefits from exercise.
Future exercise studies are likely to be designed to target the individual needs of older cancer patients, ultimately incorporating a more representative sample of patients. This will increase the generalizability of findings to the broader population of patients with hematological malignancies. Rosko et al. (2022) tested the feasibility of implementing an exercise intervention in older adults with hematologic malignancy and found a higher completion rate with in-person, physiotherapist supervision compared to at-home independent exercise. Though these older patients were motivated to follow a structured exercise program, health status changes were the main barrier to exercise [93].
In our review, adherence to exercise was predominantly high (72%). Two studies with a mean sample over 60 years reported high adherence (range 75–96%) [47,67]. Slightly lower, Mazzoni et al. (2020) found exercise adherence in a mixed cancer diagnosis group was ≥50% [94]. In our review, 20 studies did not provide adherence rates. Adherence is addressed in an umbrella scoping review by Collado-Mateo et al. (2021) of 53 studies of chronic disease and older individuals and identified multiple modifiable factors influencing adherence to exercise in this population [95]. Emphasizing factors that can improve adherence when planning and designing exercise interventions for the older population may improve feasibility and safety, and ultimately lead to better outcomes.
The extensiveness of exercise interventions varied across studies in this review, with moderate extensiveness (n = 32) being the most frequently designed intervention for the population with hematological malignancy. This pattern remained consistent in five studies where the mean or median age was above 60 years (n = 3). Conducting extensiveness rating across studies used in systematic reviews by Andersen et al. (2022) [34] and Ramírez-Vélez et al. (2021) [35] provides a more uniformed overview of the type of intervention that may be most beneficial for different populations. It is important to emphasize the significance of tailored exercise interventions for older adults with hematological malignancies, considering a more comprehensive supportive approach that takes individual needs and preferences into account, including not only physical and QoL outcomes but also clinical outcomes.
Although exercise recommendations for adults with cancer are available, evidence specifically for older cancer patients is limited and based on only a few studies [25]. There are no evidence-based recommendations specifically tailored to older patients with hematological cancers. However, it is suggested that greater focus on exercise in the older cancer population to optimize exercise delivery, participation, safety, and efficacy is justified [26]. Recognizing and understanding the benefits of exercise for older patients with hematological malignancies can be an important initiative to integrate into clinical practice.
Further research is needed on exercise interventions specifically designed for older patients with hematological cancers, including those with typical age-related comorbidities. Emphasis should be on addressing their individual needs, considering exercise types and contexts, and implementing strategies to improve recruitment accrual, minimize attrition, and enhance adherence rates. Involvement of patients in the design of these studies is important for relevance and overall quality of the study. Colton et al. (2022) captured the voices of older patients with hematological cancer, highlighting their specific needs and preferences on diet and exercise behavior [96]. As per our review, Kilari et al. (2016) also emphasized the notable lack of exercise studies specifically tailored to the older cancer population. To effectively inform future practices for this population, merely extracting results and insights from studies of a wide age range is not adequate for developing exercise prescriptions for older patients with hematological cancer. Future studies should incorporate outcomes beyond physical function and PROMs such as QoL and symptom burden and should also provide knowledge on the clinical benefits of exercise including admission days, complications, and survival [24].
Strengths and Limitations
This systematic review and meta-analysis provides the largest overview of exercise studies in a total of 3494 patients with hematological cancer undergoing neoplastic treatments. Notably, no study exclusively included participants over 65 years, or even 60 years. Consequently, drawing definitive conclusions regarding the benefit of exercise for older patients based solely on our meta-regression analysis and sub-analyses is not possible. Despite the inclusion of a large number of studies in this meta-analysis, we need to consider the substantial heterogeneity across studies, regarding variations in diagnosis groups, types of antineoplastic treatments, and the extensiveness of exercise interventions. The secondary outcomes and subgroup analyses are exploratory; therefore, interpreting results at a significance level of 0.05 can potentially pose a risk of false positive results (mass significance). We conducted three systematic searches but also acknowledge the possibility of having overlooked relevant studies in these repeated searches.
5. Conclusions
Exercise in adults with hematological malignancy undergoing neoplastic treatment provides significant benefits in physical function, aerobic capacity, muscle strength, physical activity, body composition, and QoL. Symptoms of depression, anxiety, fatigue, and pain were significantly reduced. The influence of age on the benefits of exercise remains inconclusive. The absence of studies specifically in older adults is staggering given that hematological malignancies predominantly affect the older population. Notably, there is a modest number of ongoing exercise-based randomized studies in older patients with hematological malignancy (clinicaltrials.gov, accessed on 13 November 2023), and this reflects the increasing interest and urgent need for evidence-based guidelines tailored to this population.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cancers16172962/s1, Table S1: Search strategy on 13 November 2023, implemented in ClinicalTrials.gov, limited to randomized trials only including patients above 60 years with hematological malignancies; Table S2: Outcome measurement tests and instruments chosen for meta-analysis, prioritizing those most widely applied for assessing outcomes of exercise interventions; Figure S1: Search strategy and PRISMA flow diagram for study selection (March 2019), implemented in four electronic databases, and results of total records, study population ≥ 65 years; Figure S2: Search strategy on 20 October 2020, implemented in five electronic databases, and results of total records; Figure S3: Updated search strategy on 27 March 2024, implemented in five electronic databases, and results of total records; Figure S4: Forest plot depicting the effects of exercise on physical function; Figure S5: Forest plot depicting the effects of exercise on QoL global; Figure S6: Meta-regression analyses on exercise benefits related to age for primary outcomes; physical function and QoL global; Figure S7: Subgroup analyses on primary outcome; physical function; Figure S8: Subgroup analyses on primary outcome; QoL global; Figure S9: Forest plot depicting the effects of exercise on aerobic capacity; Figure S10: Forest plot depicting the effects of exercise on muscle strength; Figure S11: Forest plot depicting the effects of exercise on body composition; Figure S12: Forest plot depicting the effects of exercise on physical activity; Figure S13: Forest plot depicting the effects of exercise on QoL emotional; Figure S14: Forest plot depicting the effects of exercise on QoL functional; Figure S15: Forest plot depicting the effects of exercise on QoL physical; Figure S16: Forest plot depicting the effects of exercise on anxiety; Figure S17: Forest plot depicting the effects of exercise on depression; Figure S18: Forest plot depicting the effects of exercise on fatigue; Figure S19: Forest plot depicting the effects of exercise on pain; Figure S20: Meta-regression analyses on exercise benefits related to age for secondary outcomes; Figure S21: Subgroup analyses on secondary outcome; aerobic capacity; Figure S22: Subgroup analyses on secondary outcome; muscle strength; Figure S23: Subgroup analyses on secondary outcome; body composition; Figure S24: Subgroup analyses on secondary outcome; physical activity; Figure S25: Subgroup analyses on secondary outcome; QoL emotional; Figure S26: Subgroup analyses on secondary outcome; QoL functional; Figure S27: Subgroup analyses on secondary outcome; QoL physical; Figure S28: Subgroup analyses on secondary outcome; anxiety; Figure S29: Subgroup analyses on secondary outcome; depression, Figure S30: Subgroup analyses on secondary outcome; fatigue; Figure S31: Subgroup analyses on secondary outcome; pain.
Author Contributions
Conceptualization, M.J., M.K.M., D.N., A.V. and C.B.J.; methodology, M.J., C.B.J., M.K.M., D.N., A.V., A.T., S.T.L. and G.K.; literature search, A.T.S.L., M.J., S.T.L., G.K., C.G. and R.F.L.; data curation, M.J., S.T.L., G.K., C.G., R.F.L. and M.S.E.; formal analysis, C.B.J., A.T., S.T.L., G.K. and M.J.; writing and review, M.J., S.T.L., G.K., A.T. and C.B.J.; project administration, M.J.; funding acquisition, M.J.; and editing, M.J., S.T.L., G.K., C.G., R.F.L., A.T.S.L., M.K.M., D.N., A.V., M.S.E., N.A., A.T. and C.B.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financially supported by the Novo Nordisk Foundation (NNF16OC0022338). The Novo Nordisk Foundation did not contribute to the drafting of the study protocol and was not involved in the execution or publication of the study.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
This study is part of the research program Models of Cancer Care, Copenhagen University Hospital, Rigshospitalet, Denmark.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Fitzmaurice, C.; Akinyemiju, T.F.; Al Lami, F.H.; Alam, T.; Alizadeh-Navaei, R.; Allen, C.; Alsharif, U.; Alvis-Guzman, N.; Amini, E.; Anderson, B.O.; et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-Years for 29 Cancer Groups, 1990 to 2016: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2019, 5, 1749–1768. [Google Scholar]
- Zhang, N.; Wu, J.; Wang, Q.; Liang, Y.; Li, X.; Chen, G.; Ma, L.; Liu, X.; Zhou, F. Global burden of hematologic malignancies and evolution patterns over the past 30 years. Blood Cancer J. 2023, 13, 82. [Google Scholar] [CrossRef] [PubMed]
- Howlader, N.A.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; Chen, H.S.; et al. (Eds.) SEER Cancer Statistics Review, 1975–2018, National Cancer Institute. Bethesda, MD. Based on November 2020 SEER Data Submission, Posted to the SEER Web Site. Available online: https://seer.cancer.gov/csr/1975_2018/ (accessed on 21 April 2021).
- Krok-Schoen, J.L.; Fisher, J.L.; Stephens, J.A.; Mims, A.; Ayyappan, S.; Woyach, J.A.; Rosko, A.E. Incidence and survival of hematological cancers among adults ages ≥75 years. Cancer Med. 2018, 7, 3425–3433. [Google Scholar] [CrossRef]
- Cordoba, R.; A Eyre, T.; Klepin, H.D.; Wildes, T.M.; Goede, V. Haematological Malignancies in Older People 1 A comprehensive approach to therapy of haematological malignancies in older patients. Lancet Haematol. 2021, 8, E840–E852. [Google Scholar] [CrossRef]
- Handforth, C.; Clegg, A.; Young, C.; Simpkins, S.; Seymour, M.T.; Selby, P.J.; Young, J. The prevalence and outcomes of frailty in older cancer patients: A systematic review. Ann. Oncol. 2015, 26, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.K.; Lee, D.T. Effects of pain, fatigue, insomnia, and mood disturbance on functional status and quality of life of elderly patients with cancer. Crit. Rev. Oncol. Hematol. 2011, 78, 127–137. [Google Scholar] [CrossRef]
- Su, W.Y.E.; Chen, H.; Wu, M.; Lai, Y. Fatigue among older advanced cancer patients. Int. J. Gerontol. 2011, 5, 84–88. [Google Scholar] [CrossRef]
- Soones, T.; Ombres, R.; Escalante, C. An update on cancer-related fatigue in older adults: A narrative review. J. Geriatr. Oncol. 2022, 13, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Wall, S.A.; Stevens, E.; Vaughn, J.; Bumma, N.; Rosko, A.E.; Borate, U. Multidisciplinary Approach to Older Adults with Hematologic Malignancies—A Paradigm Shift. Curr. Hematol. Malign- Rep. 2022, 17, 31–38. [Google Scholar] [CrossRef]
- Coelho, A.; Parola, V.; Cardoso, D.; Bravo, M.E.; Apóstolo, J. Use of non-pharmacological interventions for comforting patients in palliative care: A scoping review. JBI Database Syst. Rev. Implement Rep. 2017, 15, 1867–1904. [Google Scholar] [CrossRef]
- Abdelbasset, W.K.; Nambi, G.; Elsayed, S.H.; Osailan, A.M.; Eid, M.M. Falls and potential therapeutic interventions among elderly and older adult patients with cancer: A systematic review. Afr. Health Sci. 2021, 21, 1776–1783. [Google Scholar] [CrossRef] [PubMed]
- Brick, R.; Turner, R.; Bender, C.; Douglas, M.; Eilers, R.; Ferguson, R.; Leland, N.; Lyons, K.D.; Toto, P.; Skidmore, E. Impact of non-pharmacological interventions on activity limitations and participation restrictions in older breast cancer survivors: A scoping review. J. Geriatr. Oncol. 2022, 13, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, C.S.; Matos, J.R.; Prazeres, F.; Gomes, B. Non-pharmacological interventions in primary care to improve the quality of life of older patients with palliative care needs: A systematic review of randomised controlled trials. BMJ Open 2023, 13, e073950. [Google Scholar] [CrossRef]
- Pedersen, M.; Engedal, M.S.; Tolver, A.; Larsen, M.T.; Kornblit, B.T.; Lomborg, K.; Jarden, M. Effect of non-pharmacological interventions on symptoms and quality of life in patients with hematological malignancies—A systematic review. Crit. Rev. Oncol. 2024, 196, 104327. [Google Scholar] [CrossRef]
- Yang, Y.-P.; Pan, S.-J.; Qiu, S.-L.; Tung, T.-H. Effects of physical exercise on the quality-of-life of patients with haematological malignancies and thrombocytopenia: A systematic review and meta-analysis. World J. Clin. Cases 2022, 10, 3143–3155. [Google Scholar] [CrossRef]
- Großek, A.; Großek, K.; Bloch, W. Safety and feasibility of exercise interventions in patients with hematological cancer undergoing chemotherapy: A systematic review. Support. Care Cancer 2023, 31, 335. [Google Scholar] [CrossRef]
- Knips, L.; Bergenthal, N.; Streckmann, F.; Monsef, I.; Elter, T.; Skoetz, N. Aerobic physical exercise for adult patients with haematological malignancies. Cochrane Database Syst. Rev. 2019, 2019, CD009075. [Google Scholar] [CrossRef]
- Xu, W.; Yang, L.; Wang, Y.; Wu, X.; Wu, Y.; Hu, R. Effects of exercise interventions for physical fitness, fatigue, and quality of life in adult hematologic malignancy patients without receiving hematopoietic stem cell transplantation: A systematic review and meta-analysis. Support. Care Cancer 2022, 30, 7099–7118. [Google Scholar] [CrossRef] [PubMed]
- Abo, S.; Denehy, L.; Ritchie, D.; Lin, K.-Y.; Edbrooke, L.; McDonald, C.; Granger, C.L. People With Hematological Malignancies Treated With Bone Marrow Transplantation Have Improved Function, Quality of Life, and Fatigue Following Exercise Intervention: A Systematic Review and Meta-Analysis. Phys. Ther. 2021, 101, pzab130. [Google Scholar] [CrossRef]
- Moore, M.; Northey, J.M.; Crispin, P.; Semple, S.; Toohey, K. Effects of Exercise Rehabilitation on Physical Function in Adults With Hematological Cancer Receiving Active Treatment: A Systematic Review and Meta-Analysis. Semin. Oncol. Nurs. 2023, 39, 151504. [Google Scholar] [CrossRef]
- Izquierdo, M.; Merchant, R.A.; Morley, J.E.; Anker, S.D.; Aprahamian, I.; Arai, H.; Aubertin-Leheudre, M.; Bernabei, R.; Cadore, E.L.; Cesari, M.; et al. International Exercise Recommendations in Older Adults (ICFSR): Expert Consensus Guidelines. J. Nutr. Health Aging 2021, 25, 824–853. [Google Scholar] [CrossRef]
- Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. Exercise and Physical Activity for Older Adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef] [PubMed]
- Kilari, D.; Soto-Perez-De-Celis, E.; Mohile, S.G.; Alibhai, S.M.; Presley, C.J.; Wildes, T.M.; Klepin, H.D.; Demark-Wahnefried, W.; Jatoi, A.; Harrison, R.; et al. Designing exercise clinical trials for older adults with cancer: Recommendations from 2015 Cancer and Aging Research Group NCI U13 Meeting. J. Geriatr. Oncol. 2016, 7, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.L.; Winters-Stone, K.M.; Wiskemann, J.; May, A.M.; Schwartz, A.L.; Courneya, K.S.; Zucker, D.S.; Matthews, C.E.; Ligibel, J.A.; Gerber, L.H.; et al. Exercise Guidelines for Cancer Survivors: Consensus Statement from International Multidisciplinary Roundtable. Med. Sci. Sports Exerc. 2019, 51, 2375–2390. [Google Scholar] [CrossRef] [PubMed]
- Klepin, H.D.; Mohile, S.G.; Exterman, S.M.M.; Karger, S. (Eds.) Cancer and Aging: From Bench to Clinics. Exercise for Older Cancer Patients: Feasible and Helpful? 2012; Available online: https://karger.com/books/book/241/chapter-abstract/5166165/Exercise-for-Older-Cancer-Patients-Feasible-and?redirectedFrom=PDF (accessed on 11 July 2024).
- Higgins, J.P.T.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A.; Cochrane (Eds.) Cochrane Handbook for Systematic Reviews of Interventions Version 6.4; 2023; Available online: https://training.cochrane.org/handbook (accessed on 11 July 2024).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetz-laff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef]
- Rethlefsen, M.L.; Kirtley, S.; Waffenschmidt, S.; Ayala, A.P.; Moher, D.; Page, M.J.; Koffel, J.B.; PRISMA-S Group. PRISMA-S: An extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Syst. Rev. 2021, 10, 39. [Google Scholar] [CrossRef]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med. Inform. Decis. Mak. 2007, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Ryan, R.H.S. How to GRADE the Quality of the Evidence. Cochrane Consumers and Communication Group Version 3.0. Available online: http://cccrg.cochrane.org/author-resources (accessed on 1 January 2020).
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; LaMonte, M.J.; Lee, I.-M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sport. Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Andersen, H.H.; Vinther, A.; Lund, C.M.; Paludan, C.; Jørgensen, C.T.; Nielsen, D.; Juhl, C.B. Effectiveness of different types, delivery modes and extensiveness of exercise in patients with breast cancer receiving systemic treatment—A systematic review and meta-analysis. Crit. Rev. Oncol. 2022, 178, 103802. [Google Scholar] [CrossRef]
- Ramírez-Vélez, R.; Zambom-Ferraresi, F.; García-Hermoso, A.; Kievisiene, J.; Rauckiene-Michealsson, A.; Agostinis-Sobrinho, C. Evidence-Based Exercise Recommendations to Improve Mental Wellbeing in Women with Breast Cancer during Active Treatment: A Systematic Review and Meta-Analysis. Cancers 2021, 13, 264. [Google Scholar] [CrossRef] [PubMed]
- Accogli, M.A.; Denti, M.; Costi, S.; Fugazzaro, S. Therapeutic education and physical activity are feasible and safe in hematologic cancer patients referred to chemotherapy: Results of a randomized controlled trial. Support. Care Cancer 2022, 31, 61. [Google Scholar] [CrossRef]
- Alibhai, S.M.H.; O’neill, S.; Fisher-Schlombs, K.; Breunis, H.; Timilshina, N.; Brandwein, J.M.; Minden, M.D.; Tomlinson, G.A.; Culos-Reed, S.N. A pilot phase II RCT of a home-based exercise intervention for survivors of AML. Support. Care Cancer 2014, 22, 881–889. [Google Scholar] [CrossRef]
- Alibhai, S.; Durbano, S.; Breunis, H.; Brandwein, J.; Timilshina, N.; Tomlinson, G.; Oh, P.; Culos-Reed, S. A phase II exercise randomized controlled trial for patients with acute myeloid leukemia undergoing induction chemotherapy. Leuk. Res. 2015, 39, 1178–1186. [Google Scholar] [CrossRef] [PubMed]
- Baumann, F.T.; Kraut, L.; Schüle, K.; Bloch, W.; A Fauser, A. A controlled randomized study examining the effects of exercise therapy on patients undergoing haematopoietic stem cell transplantation. Bone Marrow Transplant. 2010, 45, 355–362. [Google Scholar] [CrossRef]
- Baumann, F.T.; Zopf, E.M.; Nykamp, E.; Kraut, L.; Schüle, K.; Elter, T.; Fauser, A.A.; Bloch, W. Physical activity for patients undergoing an allogeneic hematopoietic stem cell transplantation: Benefits of a moderate exercise intervention. Eur. J. Haematol. 2011, 87, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Bayram, S.; Barğı, G.; Çelik, Z.; Güçlü, M.B. Effects of pulmonary rehabilitation in hematopoietic stem cell transplantation recipients: A randomized controlled study. Support. Care Cancer 2024, 32, 72. [Google Scholar] [CrossRef]
- Bird, L.; Arthur, A.; Niblock, T.; Stone, R.; Watson, L.; Cox, K. Rehabilitation programme after stem cell transplantation: Randomized controlled trial. J. Adv. Nurs. 2010, 66, 607–615. [Google Scholar] [CrossRef]
- Bryant, A.L.; Deal, A.M.; Battaglini, C.L.; Phillips, B.; Pergolotti, M.; Coffman, E.; Foster, M.C.; Wood, W.A.; Bailey, C.; Hackney, A.C.; et al. The Effects of Exercise on Patient-Reported Outcomes and Performance-Based Physical Function in Adults With Acute Leukemia Undergoing Induction Therapy: Exercise and Quality of Life in Acute Leukemia (EQUAL). Integr. Cancer Ther. 2018, 17, 263–270. [Google Scholar] [CrossRef]
- Chang, P.-H.; Lai, Y.-H.; Shun, S.-C.; Lin, L.-Y.; Chen, M.-L.; Yang, Y.; Tsai, J.-C.; Huang, G.-S.; Cheng, S.-Y. Effects of a Walking Intervention on Fatigue-Related Experiences of Hospitalized Acute Myelogenous Leukemia Patients Undergoing Chemotherapy: A Randomized Controlled Trial. J. Pain Symptom Manag. 2008, 35, 524–534. [Google Scholar] [CrossRef]
- Chen, F.; Mao, L.; Wang, Y.; Xu, J.; Li, J.; Zheng, Y. The Feasibility and Efficacy of Self-help Relaxation Exercise in Symptom Distress in Patients With Adult Acute Leukemia: A Pilot Randomized Controlled Trial. Pain Manag. Nurs. 2021, 22, 791–797. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.J.; Doody, D.R.; Di, C.; Armenian, S.H.; Baker, K.S.; Bricker, J.B.; Gopal, A.K.; Hagen, A.M.; Ketterl, T.G.; Lee, S.J.; et al. Feasibility of a behavioral intervention using mobile health applications to reduce cardiovascular risk factors in cancer survivors: A pilot randomized controlled trial. J. Cancer Surviv. 2021, 15, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.-Y.; Yeh, M.-L.; Chung, Y.-C. A nurse facilitated mind-body interactive exercise (Chan-Chuang qigong) improves the health status of non-Hodgkin lymphoma patients receiving chemotherapy: Randomised controlled trial. Int. J. Nurs. Stud. 2017, 69, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Cohen, L.; Warneke, C.; Fouladi, R.T.; Rodriguez, M.A.; Chaoul-Reich, A. Psychological adjustment and sleep quality in a randomized trial of the effects of a Tibetan yoga intervention in patients with lymphoma. Cancer 2004, 100, 2253–2260. [Google Scholar] [CrossRef] [PubMed]
- Coleman, E.A.; Coon, S.; Hall-Barrow, J.; Richards, K.; Gaylor, D.; Stewart, B. Feasibility of Exercise During Treatment for Multiple Myeloma. Cancer Nurs. 2003, 26, 410–419. [Google Scholar] [CrossRef]
- Coleman, E.A.; Goodwin, J.A.; Kennedy, R.; Coon, S.K.; Richards, K.; Enderlin, C.; Stewart, C.B.; McNatt, P.; Lockhart, K.; Anaissie, E.J. Effects of Exercise on Fatigue, Sleep, and Performance: A Randomized Trial. Oncol. Nurs. Forum 2012, 39, 468–477. [Google Scholar] [CrossRef]
- Courneya, K.S.; Sellar, C.M.; Stevinson, C.; McNeely, M.L.; Peddle, C.J.; Friedenreich, C.M.; Tankel, K.; Basi, S.; Chua, N.; Mazurek, A.; et al. Randomized Controlled Trial of the Effects of Aerobic Exercise on Physical Functioning and Quality of Life in Lymphoma Patients. J. Clin. Oncol. 2009, 27, 4605–4612. [Google Scholar] [CrossRef]
- DeFor, T.E.; Burns, L.J.; Gold, E.-M.A.; Weisdorf, D.J. A Randomized Trial of the Effect of a Walking Regimen on the Functional Status of 100 Adult Allogeneic Donor Hematopoietic Cell Transplant Patients. Biol. Blood Marrow Transplant. 2007, 13, 948–955. [Google Scholar] [CrossRef]
- Eckert, R.; Huberty, J.; Kurka, J.; Laird, B.; Mesa, R.; Palmer, J. A Randomized Pilot Study of Online Hatha Yoga for Physical and Psychological Symptoms Among Survivors of Allogenic Bone Marrow Transplant. Int. J. Yoga Ther. 2022, 32, 12. [Google Scholar] [CrossRef]
- Furzer, B.J.; Ackland, T.R.; Wallman, K.E.; Petterson, A.S.; Gordon, S.M.; Wright, K.E.; Joske, D.J.L. A randomised controlled trial comparing the effects of a 12-week supervised exercise versus usual care on outcomes in haematological cancer patients. Support. Care Cancer 2016, 24, 1697–1707. [Google Scholar] [CrossRef]
- Gallardo-Rodríguez, A.G.; Fuchs-Tarlovsky, V.; Ocharán-Hernández, M.E.; Ramos-Peñafiel, C.O. Cross-Training and Resistance Training in Adults with Type B Acute Lymphoblastic Leukemia during the Induction Phase: A Randomized Blind Pilot Study. J. Clin. Med. 2023, 12, 5008. [Google Scholar] [CrossRef] [PubMed]
- Hacker, E.D.; Collins, E.; Park, C.; Peters, T.; Rondelli, D. Strength training to enhance early recovery after hematopoietic stem cell transplantation. J. Clin. Oncol. 2016, 34, 190. [Google Scholar] [CrossRef]
- Hacker, E.D.P.; Richards, R.L.; Abu Zaid, M.; Chung, S.-Y.; Perkins, S.; Farag, S.S. STEPS to Enhance Physical Activity After Hematopoietic Cell Transplantation for Multiple Myeloma. Cancer Nurs. 2022, 45, 211–223. [Google Scholar] [CrossRef]
- Hathiramani, S.; Pettengell, R.; Moir, H.; Younis, A. Relaxation versus exercise for improved quality of life in lymphoma survivors—A randomised controlled trial. J. Cancer Surviv. 2021, 15, 470–480. [Google Scholar] [CrossRef]
- Huberty, J.; Eckert, R.; Dueck, A.; Kosiorek, H.; Larkey, L.; Gowin, K.; Mesa, R. Online yoga in myeloproliferative neoplasm patients: Results of a randomized pilot trial to inform future research. BMC Complement. Altern. Med. 2019, 19, 121. [Google Scholar] [CrossRef]
- Hung, Y.-C.; Bauer, J.D.; Horsely, P.; Coll, J.; Bashford, J.; A Isenring, E. Telephone-delivered nutrition and exercise counselling after auto-SCT: A pilot, randomised controlled trial. Bone Marrow Transplant. 2014, 49, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, P.B.; Le-Rademacher, J.; Jim, H.; Syrjala, K.; Wingard, J.R.; Logan, B.; Wu, J.; Majhail, N.S.; Wood, W.; Rizzo, J.D.; et al. Exercise and Stress Management Training Prior to Hematopoietic Cell Transplantation: Blood and Marrow Transplant Clinical Trials Network (BMT CTN) 0902. Biol. Blood Marrow Transplant. 2014, 20, 1530–1536. [Google Scholar] [CrossRef]
- Jarden, M.; Baadsgaard, M.T.; Hovgaard, D.J.; Boesen, E.; Adamsen, L. A randomized trial on the effect of a multimodal intervention on physical capacity, functional performance and quality of life in adult patients undergoing allogeneic SCT. Bone Marrow Transplant. 2009, 43, 725–737. [Google Scholar] [CrossRef]
- Jarden, M.; Møller, T.; Christensen, K.B.; Kjeldsen, L.; Birgens, H.S.; Adamsen, L. Multimodal intervention integrated into the clinical management of acute leukemia improves physical function and quality of life during consolidation chemotherapy: A randomized trial ‘PACE-AL. Haematologica 2016, 101, e316–e319. [Google Scholar] [CrossRef]
- Kim, S.-D.; Kim, H.-S. Effects of a Relaxation Breathing Exercise on Anxiety, Depression, and Leukocyte in Hemopoietic Stem Cell Transplantation Patients. Cancer Nurs. 2005, 28, 79–83. [Google Scholar] [CrossRef]
- Knols, R.H.; de Bruin, E.D.; Uebelhart, D.; Aufdemkampe, G.; Schanz, U.; Stenner-Liewen, F.; Hitz, F.; Taverna, C.; Aaronson, N.K. Effects of an outpatient physical exercise program on hematopoietic stem-cell transplantation recipients: A randomized clinical trial. Bone Marrow Transplant. 2011, 46, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, D.; Watanabe, R.; Yamamoto, M.; Kizaki, M. Efficacy of physical exercise using the balance board game on physical and psychological function in patients with hematological malignancies confined to a bioclean room. Phys. Ther. Res. 2020, 23, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Koutoukidis, D.A.; Land, J.; Hackshaw, A.; Heinrich, M.; McCourt, O.; Beeken, R.J.; Philpott, S.; DeSilva, D.; Rismani, A.; Rabin, N.; et al. Fatigue, quality of life and physical fitness following an exercise intervention in multiple myeloma survivors (MASCOT): An exploratory randomised Phase 2 trial utilising a modified Zelen design. Br. J. Cancer 2020, 123, 187–195. [Google Scholar] [CrossRef]
- McCourt, O.; Fisher, A.; Ramdharry, G.; Land, J.; Roberts, A.L.; Rabin, N.; Yong, K. Exercise prehabilitation for people with myeloma undergoing autologous stem cell transplantation: Results from PERCEPT pilot randomised controlled trial. Acta Oncol. 2023, 62, 696–705. [Google Scholar] [CrossRef]
- Mello, M.; Tanaka, C.; Dulley, F.L. Effects of an exercise program on muscle performance in patients undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant. 2003, 32, 723–728. [Google Scholar] [CrossRef]
- Oechsle, K.; Aslan, Z.; Suesse, Y.; Jensen, W.; Bokemeyer, C.; de Wit, M. Multimodal exercise training during myeloablative chemotherapy: A prospective randomized pilot trial. Support. Care Cancer 2014, 22, 63–69. [Google Scholar] [CrossRef]
- Pahl, A.; Wehrle, A.; Kneis, S.; Gollhofer, A.; Bertz, H. Feasibility of whole body vibration during intensive chemotherapy in patients with hematological malignancies—A randomized controlled pilot study. BMC Cancer 2018, 18, 920. [Google Scholar] [CrossRef]
- Pahl, A.; Wehrle, A.; Kneis, S.; Gollhofer, A.; Bertz, H. Whole body vibration training during allogeneic hematopoietic cell transplantation—The effects on patients’ physical capacity. Ann. Hematol. 2020, 99, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Persoon, S.; ChinAPaw, M.J.M.; Buffart, L.M.; Liu, R.D.K.; Wijermans, P.; Koene, H.R.; Minnema, M.C.; Lugtenburg, P.J.; Marijt, E.W.A.; Brug, J.; et al. Randomized controlled trial on the effects of a supervised high intensity exercise program in patients with a hematologic malignancy treated with autologous stem cell transplantation: Results from the EXIST study. PLoS ONE 2017, 12, e0181313. [Google Scholar] [CrossRef]
- Potiaumpai, M.; Cutrono, S.; Medina, T.; Koeppel, M.; Pereira, D.L.; Pirl, W.F.; Jacobs, K.A.; Eltoukhy, M.; Signorile, J.F. Multidirectional Walking in Hematopoietic Stem Cell Transplant Patients. Med. Sci. Sports Exerc. 2021, 53, 258–266. [Google Scholar] [CrossRef]
- Safran, E.E.; Mutluay, F.; Uzay, A. Effects of neuromuscular electrical stimulation combined with resistance exercises on muscle strength in adult hematological cancer patients: A randomized controlled study. Leuk. Res. 2022, 121, 106932. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, H.; Stüwe, S.; Kropp, P.; Diedrich, D.; Freitag, S.; Greger, N.; Junghanss, C.; Freund, M.; Hilgendorf, I. A prospective, randomized evaluation of the feasibility of exergaming on patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant. 2018, 53, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Shelton, M.L.; Lee, J.Q.; Morris, G.S.; Massey, P.R.; Kendall, D.G.; Munsell, M.F.; Anderson, K.O.; Simmonds, M.J.; Giralt, S.A. A randomized control trial of a supervised versus a self-directed exercise program for allogeneic stem cell transplant patients. Psycho-Oncol. 2009, 18, 353–359. [Google Scholar] [CrossRef]
- Streckmann, F.; Kneis, S.; Leifert, J.A.; Baumann, F.T.; Kleber, M.; Ihorst, G.; Herich, L.; Grüssinger, V.; Gollhofer, A.; Bertz, H. Exercise program improves therapy-related side-effects and quality of life in lymphoma patients undergoing therapy. Ann. Oncol. 2014, 25, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Vallerand, J.R.; Rhodes, R.E.; Walker, G.J.; Courneya, K.S. Feasibility and preliminary efficacy of an exercise telephone counseling intervention for hematologic cancer survivors: A phase II randomized controlled trial. J. Cancer Surviv. 2018, 12, 357–370. [Google Scholar] [CrossRef]
- Waked, I.S. A Randomized Controlled Trial of the Effects of Supervised Aerobic Training Program on Anthropometry, Lipid Profile and Body Composition in Obese Adult Leukemic Patients. Iran. J. Blood Cancer 2019, 11, 26–32. [Google Scholar]
- Wehrle, A.; Kneis, S.; Dickhuth, H.-H.; Gollhofer, A.; Bertz, H. Endurance and resistance training in patients with acute leukemia undergoing induction chemotherapy—A randomized pilot study. Support. Care Cancer 2019, 27, 1071–1079. [Google Scholar] [CrossRef]
- Wiskemann, J.; Dreger, P.; Schwerdtfeger, R.; Bondong, A.; Huber, G.; Kleindienst, N.; Ulrich, C.M.; Bohus, M. Effects of a partly self-administered exercise program before, during, and after allogeneic stem cell transplantation. Blood 2011, 117, 2604–2613. [Google Scholar] [CrossRef]
- Wood, W.A.; Weaver, M.; Smith-Ryan, A.E.; Hanson, E.D.; Shea, T.C.; Battaglini, C.L. Lessons learned from a pilot randomized clinical trial of home-based exercise prescription before allogeneic hematopoietic cell transplantation. Support. Care Cancer 2020, 28, 5291–5298. [Google Scholar] [CrossRef]
- Yeh, M.-L.; Chung, Y.-C. A randomized controlled trial of qigong on fatigue and sleep quality for non-Hodgkin’s lymphoma patients undergoing chemotherapy. Eur. J. Oncol. Nurs. 2016, 23, 81–86. [Google Scholar] [CrossRef]
- Jarden, M.; Møller, T.; Christensen, K.B.; Buchardt, A.; Kjeldsen, L.; Adamsen, L. Longitudinal symptom burden in adult patients with acute leukaemia participating in the PACE-AL randomised controlled exercise trial—An explorative analysis. Eur. J. Cancer Care 2021, 30, e13462. [Google Scholar] [CrossRef]
- Wiskemann, J.; Kuehl, R.; Dreger, P.; Schwerdtfeger, R.; Huber, G.; Ulrich, C.M.; Jaeger, D.; Bohus, M. Efficacy of exercise training in SCT patients-who benefits most? Bone Marrow Transplant. 2014, 49, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Knowles, R.; Kemp, E.; Miller, M.; Davison, K.; Koczwara, B. Physical activity interventions in older people with cancer: A review of systematic reviews. Eur. J. Cancer Care 2022, 31, e13637. [Google Scholar] [CrossRef]
- Mikkelsen, M.K.; Juhl, C.B.; Lund, C.M.; Jarden, M.; Vinther, A.; Nielsen, D.L. The effect of exercise-based interventions on health-related quality of life and physical function in older patients with cancer receiving medical antineoplastic treatments: A systematic review. Eur. Rev. Aging Phys. Act. 2020, 17, 18. [Google Scholar] [CrossRef] [PubMed]
- Sheill, G.; Guinan, E.; Brady, L.; Hevey, D.; Hussey, J. Exercise interventions for patients with advanced cancer: A systematic review of recruitment, attrition, and exercise adherence rates. Palliat. Support. Care 2019, 17, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Allen, N.E.; Sherrington, C.; Suriyarachchi, G.D.; Paul, S.S.; Song, J.; Canning, C.G. Exercise and motor training in people with Parkinson’s disease: A systematic review of participant characteristics, intervention delivery, retention rates, adherence, and adverse events in clinical trials. Park. Dis. 2011, 2012, 854328. [Google Scholar] [CrossRef] [PubMed]
- Jansons, P.S.; Haines, T.P.; O’brien, L. Interventions to achieve ongoing exercise adherence for adults with chronic health conditions who have completed a supervised exercise program: Systematic review and meta-analysis. Clin. Rehabil. 2017, 31, 465–477. [Google Scholar] [CrossRef]
- Larsen, R.F.; Jarden, M.; Minet, L.R.; Frølund, U.C.; Abildgaard, N. Supervised and home-based physical exercise in patients newly diagnosed with multiple myeloma-a randomized controlled feasibility study. Pilot Feasibility Stud. 2019, 5, 130. [Google Scholar] [CrossRef]
- Rosko, A.; Huang, Y.; Jones, D.; Presley, C.J.; Jaggers, J.; Owens, R.; Naughton, M.; Krok-Schoen, J.L. Feasibility of implementing an exercise intervention in older adults with hematologic malignancy. J. Geriatr. Oncol. 2022, 13, 234–240. [Google Scholar] [CrossRef]
- Mazzoni, A.-S.; Brooke, H.L.; Berntsen, S.; Nordin, K.; Demmelmaier, I. Exercise Adherence and Effect of Self-Regulatory Behavior Change Techniques in Patients Undergoing Curative Cancer Treatment: Secondary Analysis from the Phys-Can Randomized Controlled Trial. Integr. Cancer Ther. 2020, 19, 1534735420946834. [Google Scholar] [CrossRef]
- Collado-Mateo, D.; Lavín-Pérez, A.; Peñacoba, C.; Del Coso, J.; Leyton-Román, M.; Luque-Casado, A.; Gasque, P.; Fernández-Del-Olmo, M.; Amado-Alonso, D. Key Factors Associated with Adherence to Physical Exercise in Patients with Chronic Diseases and Older Adults: An Umbrella Review. Int. J. Environ. Res. Public Health 2021, 18, 2023. [Google Scholar] [CrossRef] [PubMed]
- Colton, A.; Smith, M.A.; Broadbent, S.; Rune, K.T.; Wright, H.H. Perceptions of Older Adults with Hematological Cancer on Diet and Exercise Behavior and Its Role in Navigating Daily Tasks. Int. J. Environ. Res. Public Health 2022, 19, 15044. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).